Introduction
Epidemiological statistics confirm cervical
carcinoma as a leading global health burden, ranking fourth in
incidence among all malignancies worldwide (with an estimated
661,021 new cases and 348,189 deaths globally in 2022) preceded
only by breast, colorectal and lung cancer, and with significant
female morbidity and mortality across all regions, including China
(with ~150,700 new cases and 55,700 deaths in 2022) (1,2).
Although cervical cancer screening and human papillomavirus
vaccination programs have been successfully implemented and a
significant decline in incidence rates has been observed in
high-income countries (3,4), cervical cancer remains a persistent
global health challenge, disproportionately impacting low- and
middle-income countries, where disease burden reaches critical
levels (5,6).
Findings from five large-scale randomized controlled
trials (RCTs) conducted between 1999 and 2000 demonstrate that
concurrent chemoradiotherapy (CCRT) significantly enhances local
disease control in locally advanced cervical cancer (LACC), with
corresponding improvements in both progression-free survival (PFS)
and overall survival (OS) rates compared with conventional
radiotherapy alone (7–11). For instance, a systematic review and
meta-analysis revealed that CCRT led to a 10.2% increase in
complete response (CR) rates, an 8.4% increase in local regional
control and a 7.5% enhancement in OS rates (12). Notwithstanding these advances, the
5-year OS rate for LACC treated with CCRT remains suboptimal,
ranging from 65 to 70%, with distant metastasis representing a
primary mode of treatment failure (10,13–15).
This persistent efficacy ceiling underscores the imperative to
intensify and optimize systemic therapy within the standard CCRT
framework.
In recent years, in order to improve the treatment
outcome of LACC, research has shifted from merely adjusting
empirical treatment plans to a deeper understanding of tumor
biology and heterogeneity. Molecular studies are elucidating
factors underlying differential treatment responses and prognosis.
For example, the identification of specific gene mutations (such as
DCHS2, DNAH10, RYR1 and WDFY4) and the assessment of tumor
mutational burden (TMB) have enabled the construction of a
recurrence-related score (RRS) that stratifies patients by distinct
immune microenvironment features, such as increased infiltration of
activated CD8+ T cells and NK cells in high-risk groups
versus exhausted CD8+PD-1+ T cells in
low-risk groups, and correlates with clinical outcomes, thus
underscoring the potential of biomarker-driven strategies (16). Concurrently, the exploration of
novel agents, such as bioactive natural compounds capable of
inducing dual cell death pathways, points to alternative mechanisms
to overcome therapeutic resistance (17). These insights into the molecular
determinants of tumor invasiveness and treatment failure further
strengthen the theoretical basis for enhancing and optimizing
systemic treatment within the framework of standard concurrent
radiotherapy and chemotherapy.
In this context, the intensification of systemic
treatment has been investigated through both adjuvant and
neoadjuvant approaches. The adjuvant strategy, exemplified by the
OUTBACK trial, demonstrated that adding chemotherapy after CCRT
significantly increased treatment-related toxicity without
conferring OS benefits (18,19).
By contrast, the neoadjuvant approach, termed induction
chemotherapy (IC) prior to CCRT, aims to reduce primary tumor
burden and eradicate microscopic metastases earlier. However, the
historical role of IC has been fraught with contradiction and
controversy (20). Early
meta-analyses, most notably an individual patient data
meta-analysis of 21 trials, concluded that IC (often followed by
radiotherapy alone) provided no survival benefit over radiotherapy
alone and might even increase the risk of local recurrence
(21). This was largely attributed
to suboptimal, protracted IC regimens that delayed definitive CCRT,
potentially allowing for tumor repopulation (22).
This long-standing negative perception has been
fundamentally challenged and reversed by recent high-quality
evidence, which highlights that the schedule and dose-intensity of
IC are pivotal determinants of its efficacy (23). The landmark phase III INTERLACE
trial (NCT01566240) demonstrated that a short, dose-intense course
of weekly carboplatin and paclitaxel for 6 weeks, followed
immediately by standard CCRT, significantly improved 5-year PFS (72
vs. 64%) and OS (80 vs. 72%) rates compared with CCRT alone,
representing a 40% relative reduction in the hazard (risk) of dying
(24). Critically, despite an
increased incidence of hematological adverse events, this regimen
did not compromise radiotherapy compliance and, importantly, did
not lead to a clinically significant detriment in patient quality
of life compared with CCRT alone (25). Other modern, dose-dense IC regimens,
such as weekly cisplatin and paclitaxel, have also shown promising
efficacy and feasibility (26,27).
This evolving evidence signifies a paradigm shift, transforming IC
from a historically discouraged strategy into a promising new
standard of care, yet it simultaneously introduces new questions
regarding the optimal integration of different IC regimens and the
identification of patients who derive the greatest benefit.
Therefore, the central clinical question has evolved. The
contradiction is no longer about whether IC can be effective, but
rather how to analyze the totality of modern evidence to clearly
define the magnitude of benefit and safety profile of contemporary,
dose-intense IC+CCRT versus CCRT alone. Older meta-analyses are
outdated as they cannot incorporate these practice-changing trials
(28). To provide contemporary and
evidence-based clarity, the present systematic review and
meta-analysis was performed to compare the efficacy and safety of
modern IC+CCRT versus CCRT alone in patients with LACC.
Materials and methods
Protocol and registration
This systematic review with meta-analysis adhered to
the Preferred Reporting Items for Systematic Reviews and
Meta-Analyses (PRISMA) guidelines. The study protocol was
registered in PROSPERO (CRD42024613530).
Eligibility criteria [population,
intervention, comparison, outcomes and study (PICOS)]
The PICOS criteria were as follows. i) Population:
Patients with histologically or cytologically confirmed LACC
according to the International Federation of Gynecology and
Obstetrics 2018 staging system (stages IB2-IVA) (29). ii) Intervention: Patients receiving
sequential IC followed by standard CCRT, i.e., the IC + CCRT
regimen. iii) Comparison: Patients receiving standard CCRT alone.
iv) Outcomes: Primary outcomes of OS and PFS. OS was defined as the
time from randomization to death from any cause. PFS was defined as
the time from randomization to documented tumor progression
(radiological or clinical) or death. Treatment response was
evaluated per the Response Evaluation Criteria in Solid Tumors
version 1.1 (30), where CR
indicated disappearance of all target lesions, while partial
response (PR) required ≥30% reduction in the sum of target lesion
diameters. The objective response rate (ORR) comprised CRs and PRs.
Secondary outcomes were the incidence of adverse reactions, which
were classified into hematological toxicity (such as anemia and
myelosuppression) and non-hematological toxicity (including
gastrointestinal toxicity, such as diarrhea, nausea and vomiting).
v) Study design: Published or unpublished RCTs with no restrictions
on country or language were included.
Exclusion criteria
The exclusion criteria consisted of the following:
i) Reviews, systematic evaluations, animal-based experiments or
case reports; ii) non-RCTs, observational studies or retrospective
studies; iii) duplicate publications, studies with methodologically
flawed designs or inconsistent/incomplete outcome reporting; iv)
ongoing trials lacking published results; and v) studies not
fulfilling the inclusion criteria.
Literature search and study
screening
Two independent investigators systematically
searched the PubMed (https://pubmed.ncbi.nlm.nih.gov/), EMBASE (https://www.embase.com/), Cochrane Library (https://www.cochranelibrary.com/central), Web of
Science (https://www.webofscience.com/wos/woscc), CNKI
(https://www.cnki.net/), SinoMed (https://www.sinomed.ac.cn/), WanFang (https://www.wanfangdata.com.cn/) and VIP
(http://qikan.cqvip.com/) databases up to March
2025. Concurrent searches were performed in the World Health
Organization International Clinical Trials Registry Platform
(https://trialsearch.who.int/) and the
Chinese Clinical Trial Registry (http://www.chictr.org.cn/). The search strategy
incorporated medical subject headings for ((‘Uterine Cervical
Neoplasms’[Mesh]) AND (‘Chemoradiotherapy’[Mesh]) AND (‘Induction
Chemotherapy’ [Mesh] OR ‘Neoadjuvant Therapy‘[Mesh])). Retrieved
records were screened against predefined eligibility criteria by
both researchers. Discrepancies in study selection were resolved
through third-party adjudication.
Data extraction and quality
appraisal
Two independent reviewers performed dual extraction
of study characteristics from eligible trials, encompassing first
author, publication year, sample size, patient age, treatment
protocols (radiotherapy dosing and chemotherapy regimens) and
clinical outcomes. The Cochrane (RoB 2.0) tool (https://www.riskofbias.info/welcome/rob-2-0-tool)
was employed to assess risk of bias across the following key
domains: Selection bias (randomization and allocation concealment),
performance bias (blinding), attrition bias (incomplete data),
reporting bias and other sources. Studies were categorized as low,
high or unclear risk for each domain.
GRADE assessment
The certainty of evidence for each outcome was
evaluated using the Grading of Recommendations Assessment,
Development and Evaluation (GRADE) framework (31,32).
Two reviewers independently assessed the following domains: Risk of
bias, inconsistency, indirectness, imprecision and publication
bias. Evidence certainty was categorized as high, moderate, low or
very low. For each outcome, reasons for downgrading (such as
imprecision due to limited sample sizes or wide confidence
intervals and inconsistency due to high heterogeneity) were
documented. Summary tables were generated using GRADEpro GDT
software (https://www.gradepro.org/), and the
full evidence profile is presented in Table SI.
Statistical analysis
All meta-analyses were performed using Cochrane
RevMan (version 5.4) (The Cochrane
Collaboration;https://training.cochrane.org/online-learning/core-software-cochrane-reviews/revman)
and R (version 4.5.2) (R Core Team; http://www.r-project.org/). The results were reported
as pooled odds ratios (ORs) with 95% confidence intervals (95%
CIs). Cochran's Q test and I2 statistics were used to
evaluate the heterogeneity of all the studies. Taking into account
the diversity in both clinical and methodological aspects,
regardless of the degree of heterogeneity, the random-effects model
was adopted to provide more conservative and more general
estimates. Sensitivity analyses were conducted to explore the
influence of individual studies and to assess the robustness of the
findings. Funnel plots were created using Egger's/Begg's regression
tests for outcomes including ≥10 studies. For outcomes with fewer
studies, the potential for publication bias was acknowledged as a
limitation. Two-tailed P<0.05 was considered to indicate a
statistically significant difference.
Results
Literature search and study
inclusion
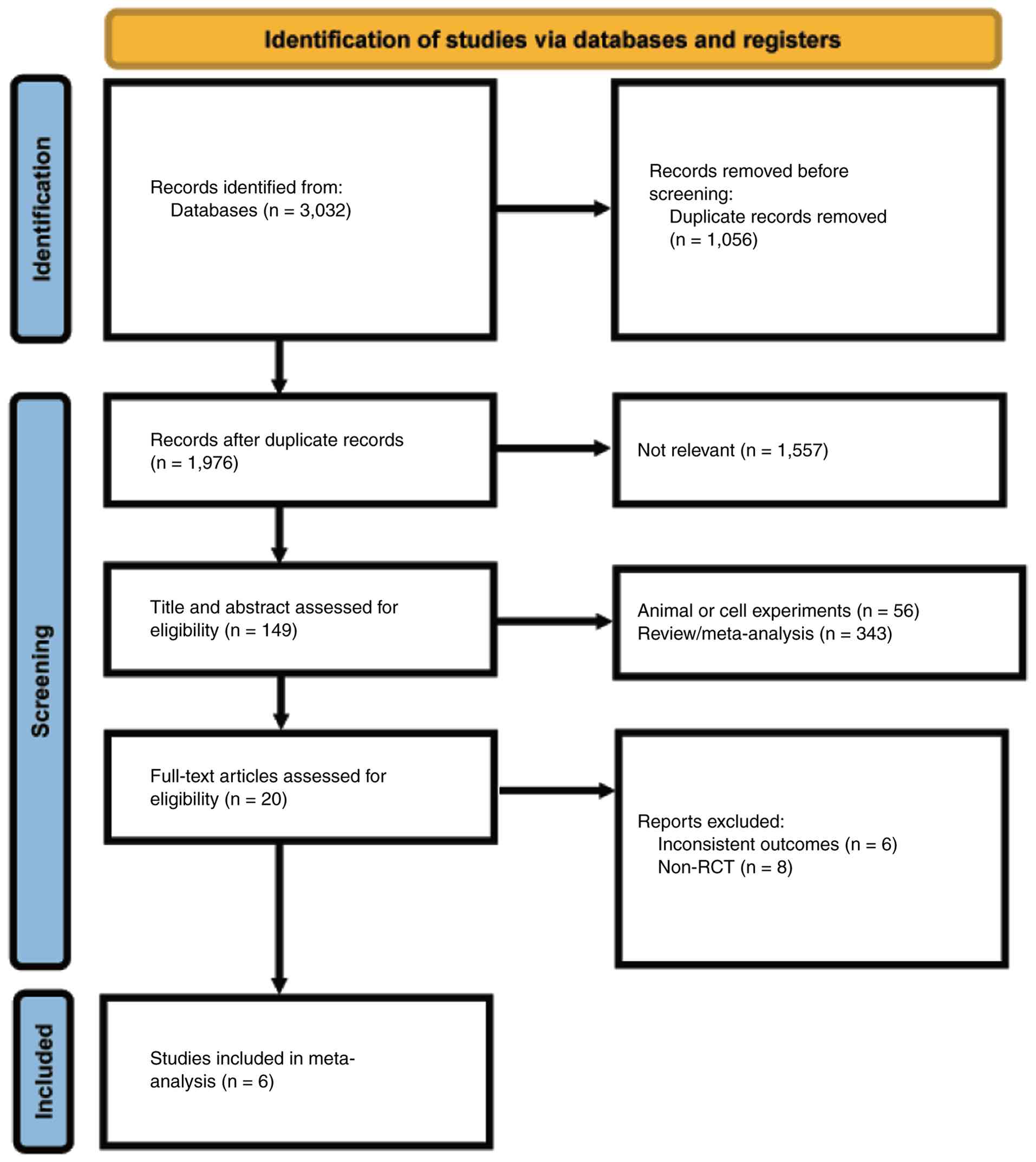
A total of 3,032 records were initially retrieved:
PubMed (n=515), EMBASE (n=408), Web of Science (n=1,085), Cochrane
Library (n=124), CNKI (n=330), SinoMed (n=287), WanFang (n=150) and
VIP (n=133). After excluding 1,056 duplicate articles, the titles
and abstracts of the remaining studies were reviewed. A total of
1,557 articles were excluded due to irrelevant content and 399
articles were not included as they were systematic reviews, animal
studies or case reports. Full-text articles were excluded for the
following reasons: Inconsistent outcomes (6 articles) and non-RCT
(8 articles). Following eligibility assessment, 6 trials involving
504 experimental and 502 control participants were included
(24,33–37).
Fig. 1 illustrates the literature
screening workflow, while Tables I
and II summarize the key
characteristics of the included studies.
 | Table I.Baseline characteristics of included
studies. |
Table I.
Baseline characteristics of included
studies.
|
|
|
| Number of
patients |
|
|
|
|
|
|---|
|
|
|
|
| Age, years |
|
|
|
|---|
|
|
|
|
|
|
|
|
|
|
|---|
| First author,
year | Country | Study design | IC + CCRT | CCRT | IC + CCRT | CCRT | FIGO | Pathology | (Refs.) |
|---|
| Tripathi and | India | RCT | 40 | 40 | Mean ± SD, | 47.13± | IIA-IIIB | SCC | (33) |
| Rawat, 2019 |
|
|
|
| 46.85± | 10.281 |
|
|
|
|
|
|
|
|
| 8.448 |
|
|
|
|
| Banerjee et
al, 2022 | India | RCT | 21 | 30 | NA | NA | IIB-IVA2 | SCC | (34) |
| Li et al,
2023 | China | RCT | 73 | 68 | Median, 51 | Median, 53 | IB2-IVA | SCC/AC/ASC | (35) |
| McCormack et
al, 2024 | Multicenter | RCT | 250 | 250 | Median, 46 | Median, 46 | IB2-IVA | SCC/AC/ASC | (24) |
| Singh et al,
2019 | India | RCT | 54 | 62 | Median, 50 | Median, 49 | IIB-IIIB | SCC | (36) |
| da Costa et
al, 2019 | Brazil | RCT | 55 | 52 | Median, 48 | Median, 45 | IIB-IVA | SCC/AC/ASC | (37) |
 | Table II.Treatment regimens across included
studies. |
Table II.
Treatment regimens across included
studies.
|
|
| Interventions |
|
|---|
|
|
|
|
|
|---|
| First author,
year | RT regimens | IC+CCRT | CCRT | (Refs.) |
|---|
| Tripathi and Rawat,
2019 | EBRT [50 Gy/25 f (5
f/week)]+ ICRT (HDR 700 cGy × 3 f) | Paclitaxel +
cisplatin + CCRT | Cisplatin + RT | (33) |
| Banerjee et
al, 2022 | EBRT [50 Gy/25 f (5
f/week)]+ ICRT (HDR 700 cGy × 3 f) | Paclitaxel +
cisplatin + CCRT | Cisplatin + RT | (34) |
| Li et al,
2023 | EBRT/IMRT
[56.35–60.2 Gy for LN, 50.4 Gy for CTV/28 f (5 f/week)] + ICRT (600
cGy × 5 f) | Paclitaxel +
cisplatin + CCRT | Cisplatin + RT | (35) |
| McCormack et
al, 2024 | EBRT [45.0–50.4
Gy/20-28 f (5 f/week)] + ICRT | Paclitaxel +
carboplatin + CCRT | Cisplatin + RT | (24) |
| Singh et al,
2019 | EBRT [46 Gy/23 f (5
f/week)]+ ICRT (HDR 700 cGy × 3 f) | 5-Fu + carboplatin
+ CCRT | Cisplatin + 5-Fu +
RT | (36) |
| da Costa et
al, 2019 | EBRT [45-50.4
Gy/25-28 f (5 f/week)] + ICRT (HDR 700–750 cGy × 4 f) | Gemcitabine +
cisplatin + CCRT | Cisplatin + RT | (37) |
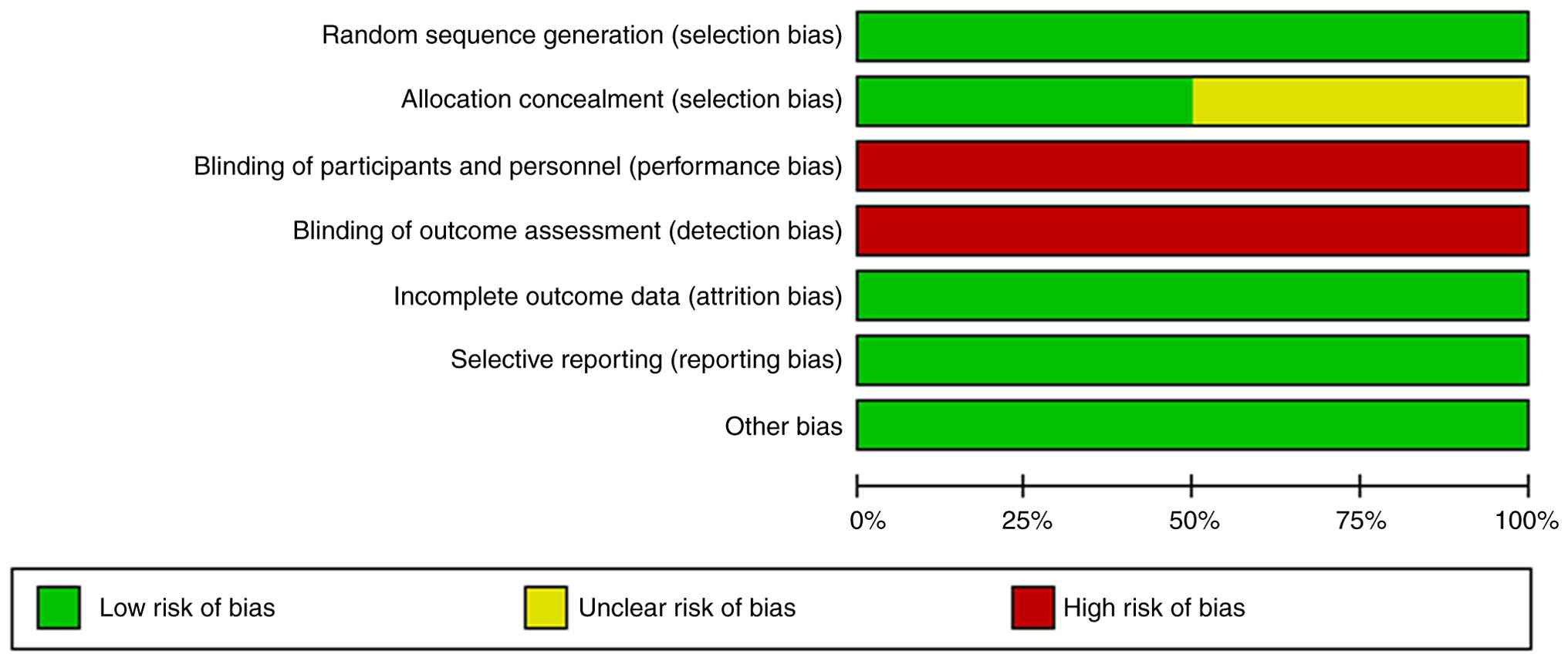
Quality assessment
The methodological quality was evaluated using the
Cochrane Risk of Bias Assessment Tool (RoB 2.0) (Figs. 2 and 3). Random sequence generation performed
well. However, in terms of allocation concealment, some studies
showed unclear risks. Due to the nature of the treatment regimens,
blinding of participants, researchers and outcome assessors
presented practical difficulties, resulting in a relatively high
risk of bias in this area. It is worth noting that all studies
demonstrated low risks in terms of the completeness of outcome
data, selective reporting and other biases.
Rationale for risk of bias
judgments
The assessments of high or unclear risk in specific
domains are detailed as follows. There was a high risk of bias in
blinding. All six trials were judged as high risk concerning the
blinding of participants, personnel and outcome assessment. This
stems from their open-label design, where the distinct treatment
pathways (IC + CCRT involving additional chemotherapy cycles versus
CCRT alone) were impossible to mask. Furthermore, the
characteristic toxicities of induction chemotherapy (such as hair
loss and more pronounced bone marrow suppression) acted as overt
unblinding factors. This knowledge of allocation introduces a
potential for expectancy bias in the assessment of subjective
outcomes such as tumor response and adverse event grading.
There was an unclear risk of bias in allocation
concealment. Three studies [Li et al (35) 2023; Banerjee et al (34) 2022; and Tripathi and Rawat (33) 2019] were rated as having an unclear
risk regarding allocation concealment. While describing
randomization, their reports omitted the specific methods used to
conceal the allocation sequence (for example, sealed envelopes and
central randomization) until after participants were enrolled. This
lack of detailed reporting prevents a definitive judgment on
whether selection bias was prevented, reflecting a common
limitation in methodological transparency rather than evidence of
actual bias.
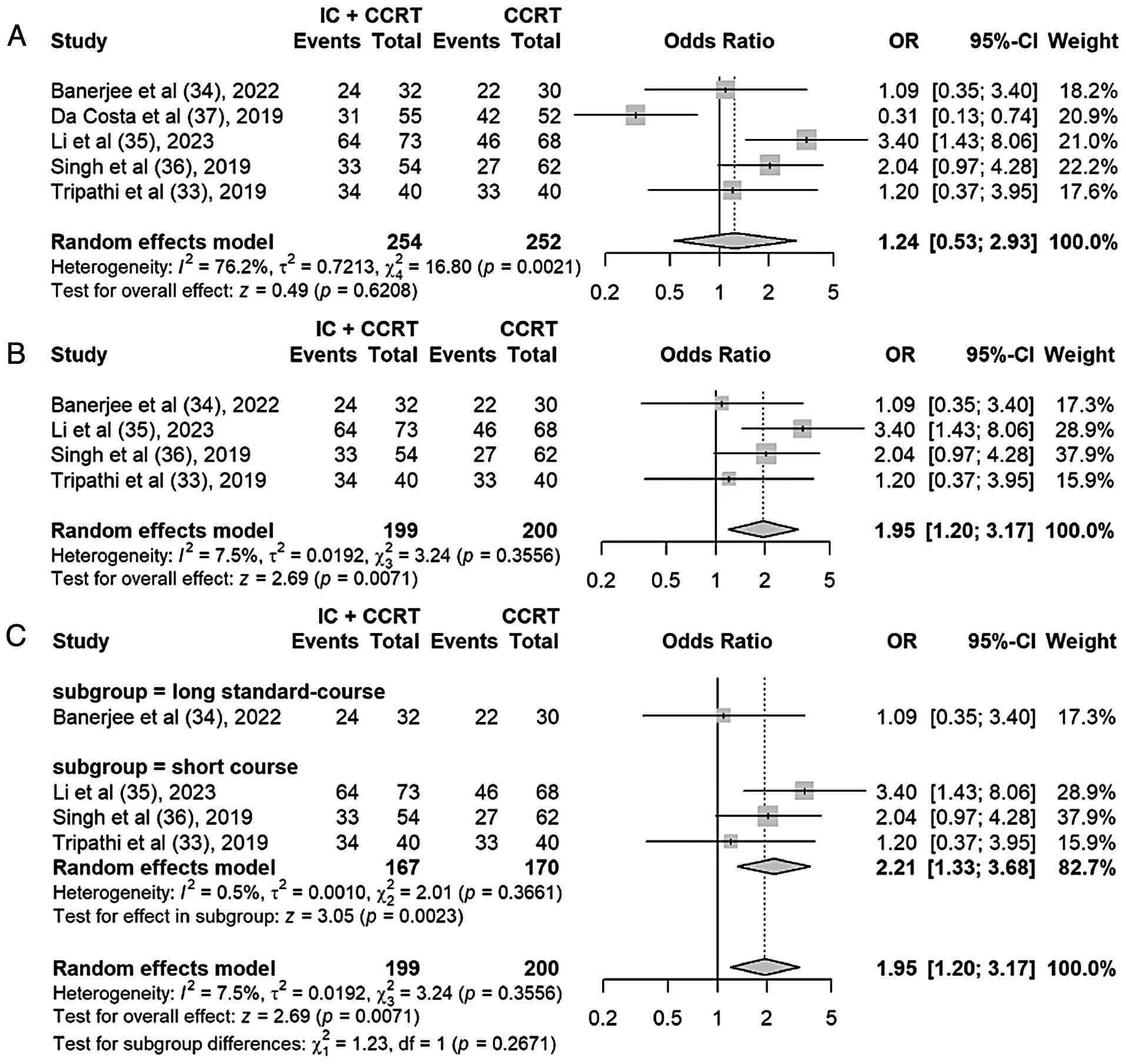
CR
In total, 5 of the included studies reported the CR
rate (33–37). The random-effects model was adopted
for the meta-analysis, which showed higher CR rates in the IC +
CCRT group compared with those in the CCRT alone group (OR, 1.24;
95% CI, 0.53–2.93; P=0.62), but there was significant heterogeneity
(P=0.002; I2=76%) (Fig.
4A). A sensitivity analysis conducted using the leave-one-out
method demonstrated that excluding the study by da Costa et
al (37) 2019 markedly reduced
this heterogeneity, with the I2 statistic decreasing
from 76 to 7%. The overall effect size shifted to 1.95 (95% CI,
1.20–3.17; P=0.007), indicating statistically significant results
(Fig. 4B). These findings suggest
that the study by da Costa 2019 was a substantial contributor to
the observed heterogeneity, and its exclusion improved the
consistency of the pooled estimates.
Based on the impact on overall treatment timeline,
induction chemotherapy regimens were categorized into long
standard-course (3 cycles of 3-weekly chemotherapy, extending over
~9 weeks) and short-course regimens. The latter included protocols
designed to minimize delay to definitive chemoradiation,
characterized by either a reduced number of cycles (1–2 cycles) or
an intensive weekly schedule over a shorter total duration. A
subgroup analysis was performed (Fig.
4C). The short-course subgroup demonstrated a significant
improvement in CR with IC + CCRT (OR, 2.21; 95% CI, 1.33–3.68;
P=0.002). This indicates that the total duration and intensity of
the induction regimen, which directly affect the timing of
definitive therapy, have a significant impact on treatment
outcome.
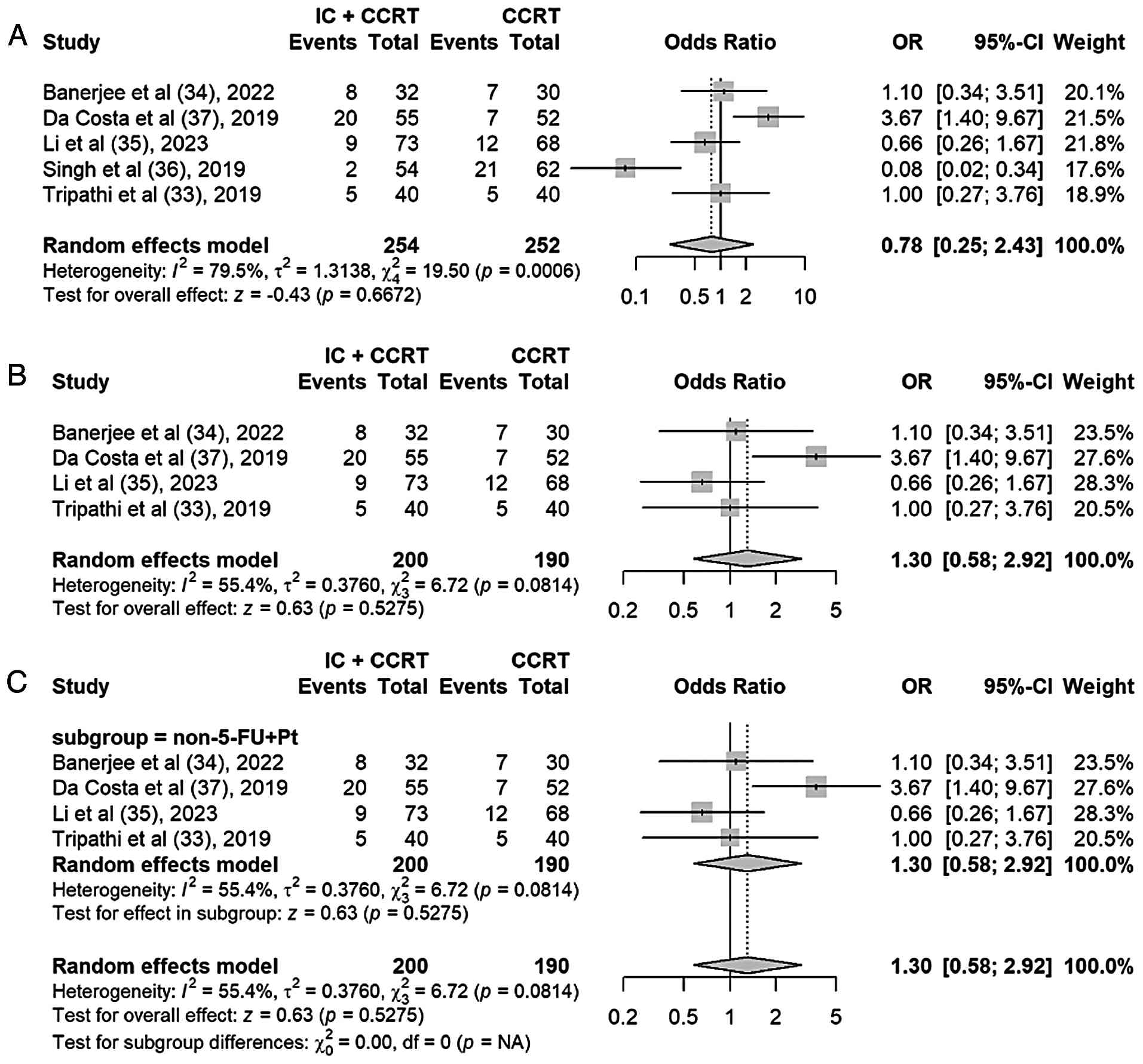
Partial response
A total of 5 of the included studies reported the PR
rate. In the meta-analysis of PR rates, the initial random-effects
model showed high heterogeneity (I2=79%) (Fig. 5A). Sensitivity analysis revealed
that the study by Singh et al (36) in 2019 significantly influenced the
results. The control group in this study used a two-drug regimen of
cisplatin + 5-fluorouracil, while the other studies all used
single-agent cisplatin. This difference may be the main reason for
it becoming an outlier and causing high heterogeneity. After
excluding this study, the heterogeneity decreased to a moderate
level (I2=55%) (Fig.
5B), and the control group regimens of the remaining studies
tended to be consistent.
Further subgroup analysis, stratified by the
inclusion of fluorouracil in the control regimen, revealed that in
the non-fluorouracil subgroup (four studies), heterogeneity
decreased to a moderate level (I2=55%), whereas the
fluorouracil subgroup consisted of a single study (Singh et
al 2019), precluding the calculation of within-subgroup
heterogeneity. The test for subgroup differences was significant
(P=0.0011), confirming that the difference in control regimens
(cisplatin + 5-fluorouracil vs. cisplatin alone) was a major source
of the initial high heterogeneity. Both subgroup analyses did not
show that IC + CCRT had a significant advantage in improving the PR
rate. Given the remaining heterogeneity, a random-effects model was
used for the pooled analysis. The results showed that IC + CCRT did
not significantly improve the PR rate compared with simple CCRT
(combined OR, 1.30; 95% CI, 0.58–2.92; P=0.53) (Fig. 5C).
ORR
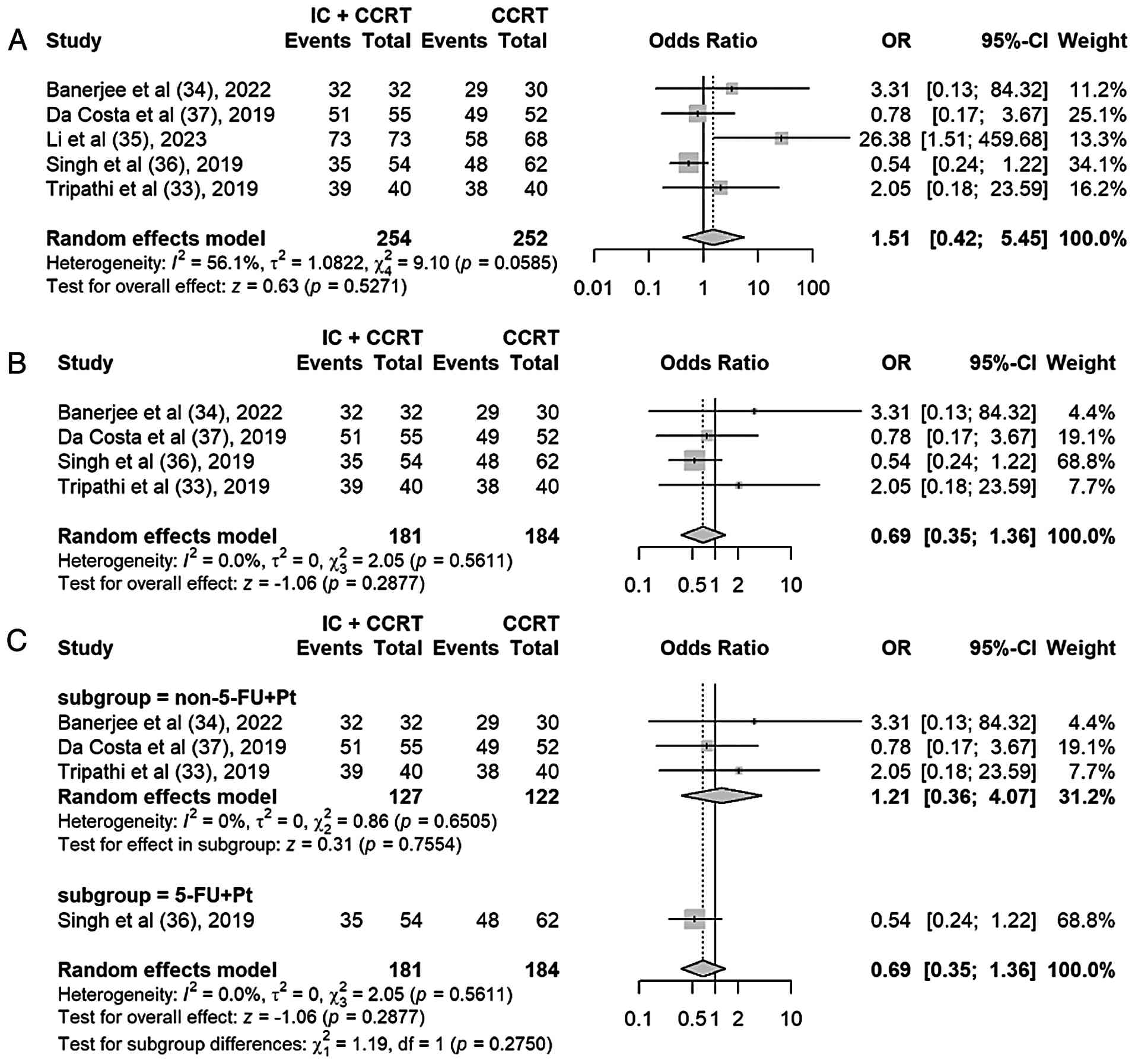
A total of 5 studies reported the ORR. The pooled
analysis using the random-effects model showed that there was no
statistically significant difference in ORR between the IC + CCRT
group and the CCRT group (OR, 1.51; 95% CI, 0.42–5.45; P=0.53), but
there was moderate heterogeneity among the studies
(I2=56%; P=0.06) (Fig.
6A). Sensitivity analysis using the leave-one-out method
identified the study by Li et al (35) as the primary source of
heterogeneity. After excluding this study, the I2
statistic decreased from 56 to 0%, while the pooled OR remained
non-significant (OR, 0.69; 95% CI, 0.35–1.36; P=0.29) (Fig. 6B). The extreme ORR (100%) in the
experimental arm of the study by Li et al (35) may be attributable to the later
assessment time point (1 year post-treatment versus 4–12 weeks in
other studies) and a more intensive radiotherapy regimen (higher
nodal dose and more brachytherapy fractions). Notably, the
remaining four studies still included one trial with an intensified
CCRT regimen (cisplatin + 5-fluorouracil) in the control arm
(36). A further subgroup analysis
restricted to studies using single-agent cisplatin CCRT [Tripathi
and Rawat (33), Banerjee et
al (34) and da Costa et
al (37)] yielded a pooled OR
of 1.21 (95% CI, 0.36–4.07; P=0.76), with low heterogeneity
(I2=0%) (Fig. 6C). These
findings indicate that while IC may show a non-significant trend
toward improved ORR in the context of single-agent cisplatin CCRT,
the overall evidence does not support a statistically significant
benefit.
1-, 3- and 5-year PFS and OS
rates
A total of 3 studies reported the 1-year PFS and OS
rates, while 2 studies reported the 3- and 5-year PFS and OS
rates.
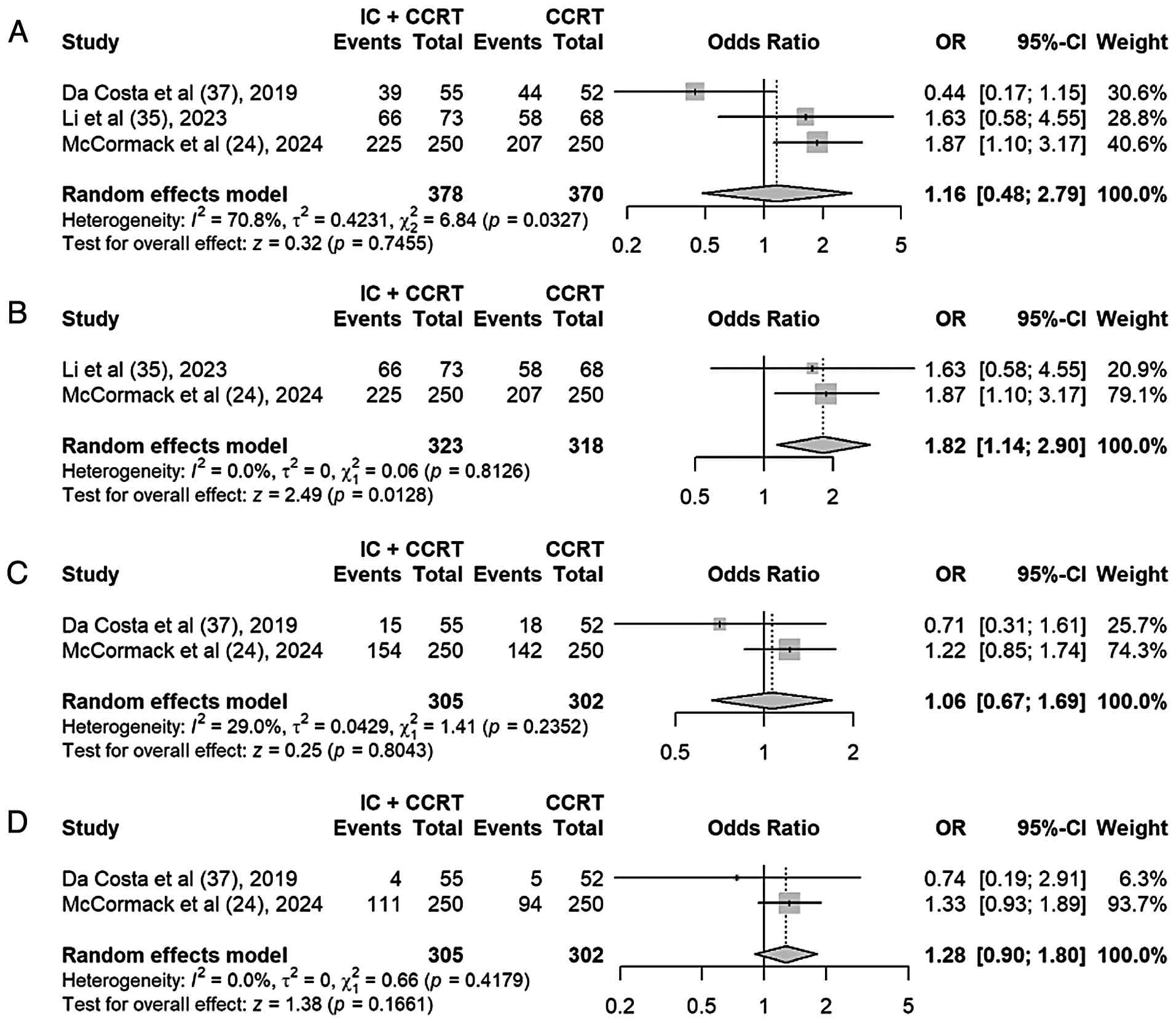
Based on a preliminary meta-analysis, the addition
of IC to standard CCRT showed a non-significant trend toward
improved 1-year PFS (OR, 1.16; 95% CI, 0.48–2.79; P=0.75), with
substantial heterogeneity (I2=71%) (Fig. 7A). Given that the outlier study [da
Costa et al (37) 2019]
employed a long standard-course regimen, a sensitivity analysis was
performed excluding it to reduce protocol-related heterogeneity.
After exclusion, heterogeneity was eliminated (I2=0%),
and IC + CCRT demonstrated a significant improvement in 1-year PFS
rate compared with CCRT alone (OR, 1.82; 95% CI, 1.14–2.90; P=0.01)
(Fig. 7B).
For longer-term outcomes, random-effect models
showed non-significant point estimates favoring IC + CCRT at 3
years (P=0.80) (Fig. 7C) and 5
years (P=0.17) (Fig. 7D), with the
magnitude of benefit appearing to increase over time, although
neither reached statistical significance.
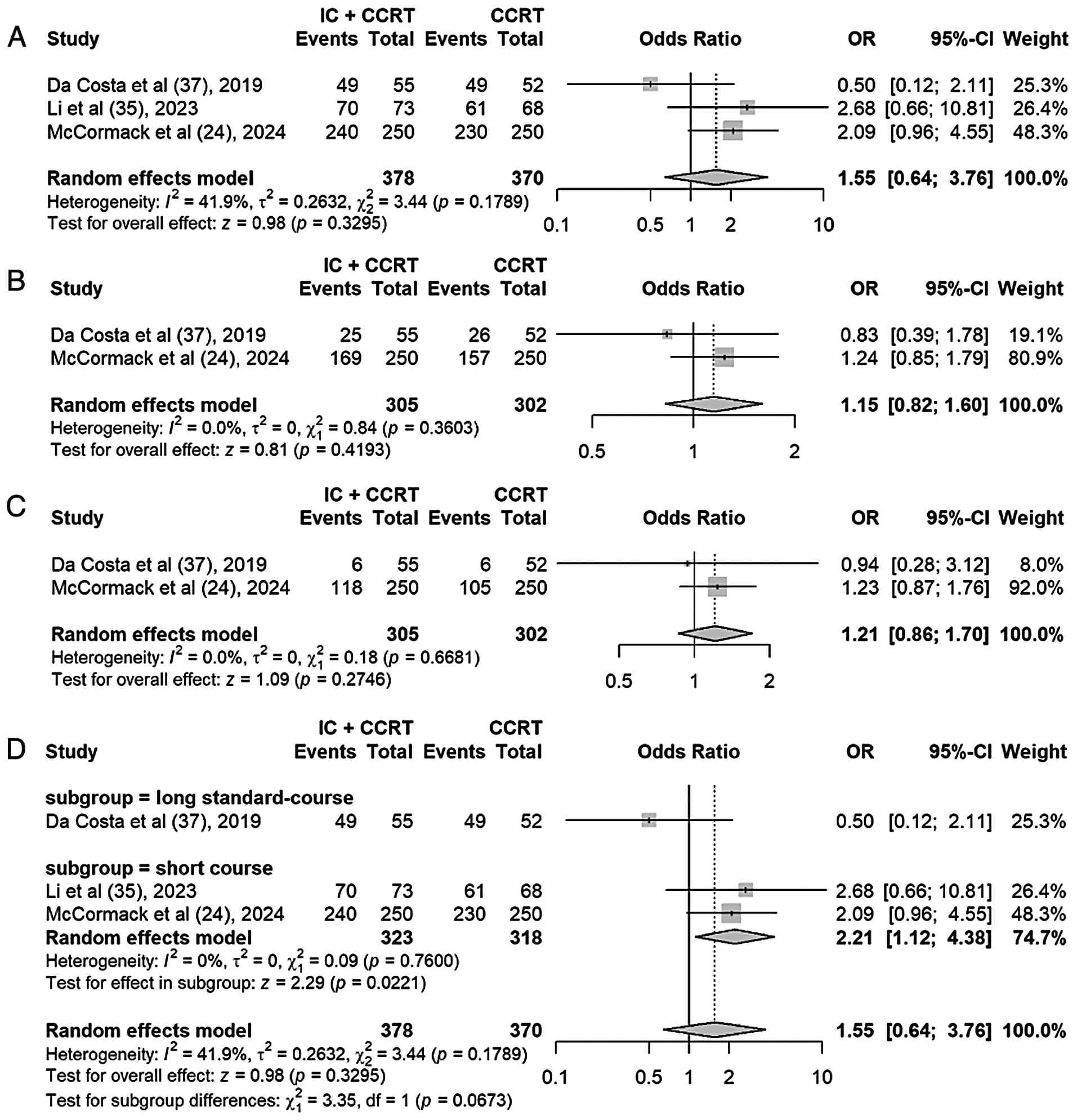
OS
A random-effects model was applied for the
meta-analysis of OS rate. The pooled analysis demonstrated
consistent but non-significant improvements in OS rate favoring IC
+ CCRT: 1-year OS rate (OR, 1.55; 95% CI, 0.64–3.76; P=0.33),
3-year OS rate (OR, 1.15; 95% CI, 0.82–1.60; P=0.42) and 5-year OS
rate (OR, 1.21; 95% CI, 0.86–1.70; P=0.27). These results were not
statistically significant (Fig.
8A-C).
Although the initial 1-year OS rate combined
analysis did not show statistical significance, it was observed
that the results of the study by da Costa et al (37) were in contrast to those of the other
two studies. In the study by da Costa et al (37), which utilized a ‘long-course
standard type’ induction regimen comprising three cycles of
cisplatin and gemcitabine administered every 3 weeks (extending
over ~9 weeks and delaying the initiation of definitive
chemoradiation by 3–4 weeks after the last chemotherapy cycle), the
odds ratio was <1, favoring CCRT alone. By contrast, both the
study by Li et al (35) 2023
and that by McCormack et al (23) 2024 employed ‘short-course type’
regimens designed to minimize delays to definitive chemoradiation,
and both reported odds ratios >1, favoring IC+CCRT. Considering
that this study used the ‘long-course standard type’ induction
regimen, while the other studies used the ‘short-course type’
regimen, a subgroup analysis was conducted based on treatment
course design. In the ‘short-course type’ subgroup (n=2), IC + CCRT
significantly improved 1-year OS rate (OR, 2.21; 95% CI, 1.12–4.38;
P=0.02) (Fig. 8D), and there was no
heterogeneity (I2=0%). This result suggests that the
benefit of induction chemotherapy on survival may be limited to the
‘short-course type’ regimen.
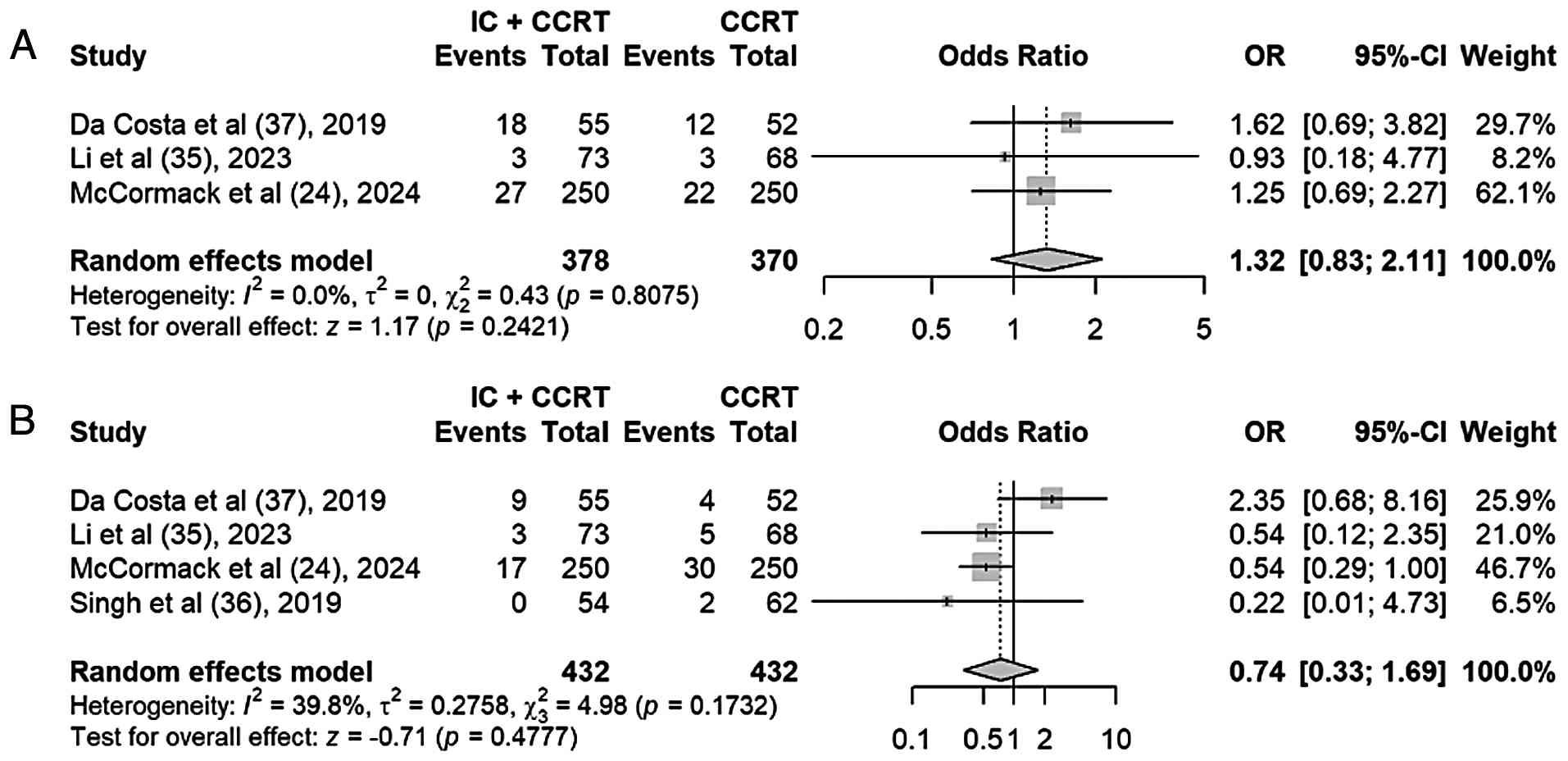
Local recurrence rate and distant
metastasis rate
Local recurrence rates were reported in 3 out of the
6 studies, and distant metastasis rates were reported in 4 out of
the 6 articles. A random-effects model was used for the
meta-analysis, and it was found that the local recurrence rate was
higher in the IC + CCRT group than that in the CCRT group (OR,
1.32; 95% CI, 0.83–2.11; P=0.24), but that the distant metastasis
rate was lower in the IC + CCRT group than that in the CCRT group
(OR, 0.74; 95% CI, 0.33–1.69; P=0.48). There were no statistically
significant differences in the local recurrence rate and distant
metastasis rate groups (Fig. 9A and
B).
Safety and adverse events
The adverse events that occur during treatment
mainly come under two categories: Hematological toxicity and
non-hematological toxicity. Regarding hematological toxicity,
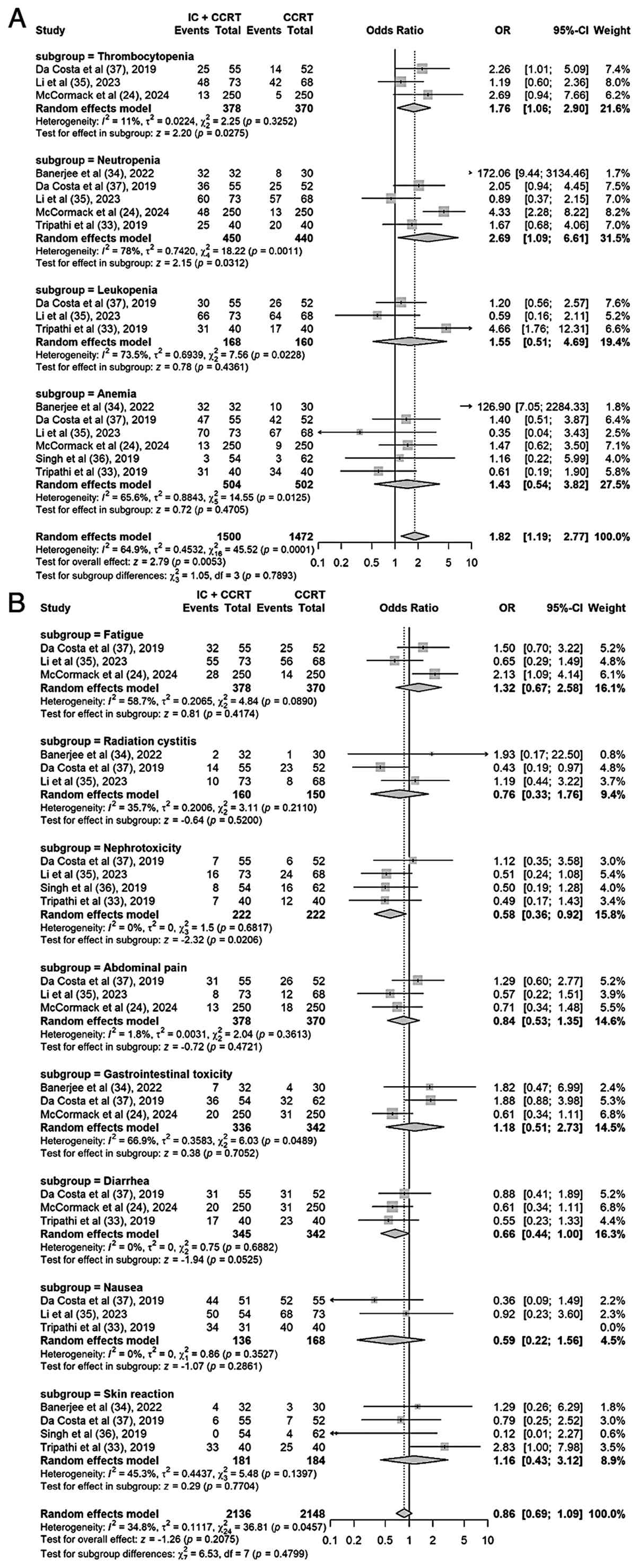
thrombocytopenia was documented in 3 studies, neutropenia in 5,
leukopenia in 3 and anemia in 6. Regarding hematological toxicity,
the IC + CCRT group demonstrated a significantly higher incidence
of thrombocytopenia (OR, 1.76; 95% CI, 1.06–2.90; P=0.03) and
neutropenia (OR, 2.69; 95% CI, 1.09–6.61; P=0.03) (Fig. 10A). However, for leukopenia (OR,
1.55; 95% CI, 0.51–4.69; P=0.44) and anemia (OR, 1.43; 95% CI,
0.54–3.82; P=0.47), although the IC + CCRT group showed a
numerically higher incidence, this difference did not reach
statistical significance. With regard to non-hematological
toxicity, fatigue was mentioned in 3 studies, radiation cystitis in
3, nephrotoxicity in 4, abdominal pain in 3, gastrointestinal
toxicity in 3, diarrhea in 3, nausea in 3 and skin reaction in 4.
The pooled analysis demonstrated no statistically significant
differences between the IC + CCRT and CCRT groups in the incidence
of fatigue (OR, 1.32; 95% CI, 0.67–2.58; P=0.42), radiation
cystitis (OR, 0.76; 95% CI, 0.33–1.76; P=0.52), abdominal pain (OR,
0.84; 95% CI, 0.53–1.35; P=0.47), gastrointestinal toxicity (OR,
1.18; 95% CI, 0.51–2.73; P=0.71), diarrhea (OR, 0.66; 95% CI,
0.44–1.00; P=0.05), nausea (OR, 0.59; 95% CI, 0.22–1.56; P=0.29) or
skin reaction (OR, 1.16; 95% CI, 0.43–3.12; P=0.77). The only
exception was nephrotoxicity, which occurred significantly less
frequently in the IC+CCRT group (OR, 0.58; 95% CI, 0.36–0.92;
P=0.02) (Fig. 10B). The pooled
odds ratios for hematological and non-hematological adverse events
are summarized in Table III. The
certainty of evidence for each outcome was assessed using the GRADE
framework. The evidence profile summarizing the certainty ratings
is provided in Table SI. Overall,
the certainty of evidence was high for hematological and
non-hematological toxicity outcomes, and moderate for most survival
outcomes, with downgrading primarily due to imprecision (from
limited sample sizes) or inconsistency (from high heterogeneity) in
several endpoints.
 | Table III.Summary of adverse events in IC+CCRT
versus CCRT for locally advanced cervical cancer. |
Table III.
Summary of adverse events in IC+CCRT
versus CCRT for locally advanced cervical cancer.
| Adverse event
category | Number of
studies | Odds ratio (95%
CI) | P-value | Interpretation |
|---|
| Hematological
toxicities |
|
|
|
|
|
Thrombocytopenia | 3 | 1.75
(1.10–2.79) | 0.02 | Significantly
higher in IC+CCRT |
|
Neutropenia | 5 | 2.89
(2.03–4.12) | <0.01 | Significantly
higher in IC+CCRT |
|
Leukopenia | 3 | 1.62
(0.96–2.72) | 0.07 | Not
significant |
|
Anemia | 6 | 1.81
(1.15–2.84) | 0.01 | Significantly
higher in IC+CCRT |
| Non-hematological
toxicities |
|
|
|
|
|
Fatigue | 3 | 1.39
(0.91–2.11) | 0.13 | Not
significant |
|
Radiation cystitis | 3 | 0.70
(0.38–1.27) | 0.24 | Not
significant |
|
Nephrotoxicity | 4 | 0.58
(0.36–0.92) | 0.02 | Significantly lower
in IC+CCRT |
|
Abdominal pain | 3 | 0.84
(0.53–1.33) | 0.46 | Not
significant |
|
Gastrointestinal toxicity | 3 | 1.01
(0.66–1.55) | 0.97 | Not
significant |
|
Diarrhea | 3 | 0.66
(0.44–1.00) | 0.05 | Borderline
significance |
|
Nausea | 3 | 0.58
(0.22–1.50) | 0.26 | Not
significant |
| Skin
reaction | 4 | 1.21
(0.65–2.27) | 0.55 | Not
significant |
Sensitivity analysis and publication
bias assessment
Sensitivity analyses were performed using a
leave-one-out approach, systematically excluding each study in
sequence to determine its individual impact on the pooled outcomes.
The results indicated that, with the exception of the CR, PR, ORR,
and 1-year PFS analyses, the removal of any single study did not
substantially influence the overall results (Fig. S1A, Fig. S2A, Fig. S3A, Fig. S4A, Fig. S5A, Fig. S6A, Fig. S7A, Fig. S8A, Fig. S9A, Fig. S10A, Fig. 11A). This indicates that the
meta-analysis results are relatively robust. Furthermore, after
performing Begg's and Egger's tests on the funnel plots of all
outcome indicators, the publication bias analysis of the 6 included
studies demonstrated no significant publication bias with regard to
the CR, PR and ORR, the 1-, 3- and 5-year PFS and OS rates, or the
LRR and DMR (all P>0.05) (Fig.
S1B, Fig. S2B, Fig. S3B, Fig. S4B, Fig. S5B, Fig. S6B, Fig. S7B, Fig. S8B, Fig. S9B, Fig. S10B, Fig. 11B). However, for outcomes with only
a small number of studies (for example, 3–5 studies), the
statistical power to detect publication bias is inherently low,
which is an important methodological limitation. Therefore, while
the available quantitative tests did not detect significant bias,
the possibility of unpublished data or small-study effects cannot
be definitively ruled out, particularly for the outcomes with very
few studies. The interpretation of funnel plots for these outcomes
is also highly challenging. Consequently, the conclusions,
especially those reliant on a small number of trials, should be
interpreted with caution regarding potential publication bias.
Discussion
A systematic review and meta-analysis was conducted
to evaluate the efficacy and safety of adding IC before standard
CCRT for LACC. The core results indicated that the IC + CCRT
strategy, especially when using modern short-course, dose-dense
regimens, significantly improved the CR rate and 1-year PFS rate of
patients compared with CCRT alone, but with a higher risk of
hematological toxicity; although the OS rate showed an improving
trend, it did not reach statistical significance. These findings
mark the transformation of IC from a historically controversial
strategy due to long-term and delayed radical treatment to a
promising treatment enhancement option based on precise protocol
design. Subgroup analysis confirmed that short-course intensive
regimens (1–2 cycles therapy or weekly therapy) are key to
achieving survival benefits, which is highly consistent with the
conclusion of the INTERLACE III phase trial that changed clinical
practice (24). The trial
demonstrated that immediate continuation of CCRT after 6 weeks of
carboplatin/paclitaxel induction therapy could significantly
increase the 5-year OS rate from 72 to 80%, and its successful
model lies in rapidly reducing tumors through dose-dense weekly
therapy while minimizing the risk of tumor recurrence due to long
treatment intervals, thereby converting the potential theoretical
advantages of IC into definite survival benefits (24). This principle, solidified by modern
practice-changing trials, finds precedent in earlier exploratory
studies (38,39). For instance, a phase II trial
demonstrated that a dense schedule of paclitaxel-cisplatin
administered every 10 days over ~1 month achieved a high clinical
response rate (90.7%) in patients with locally advanced disease
without delaying subsequent definitive treatment (39). This early evidence underscores the
feasibility and activity of compressing the induction period to
rapidly reduce tumor volume, a foundational concept for
contemporary protocols.
Based on the confirmation of the effectiveness of
the short-term treatment plan, an in-depth analysis was conducted
to investigate the ‘incremental value’ of adjuvant chemotherapy
prompts. It was found that this value is not absolute and is
largely influenced by the intensity of the subsequent comprehensive
CCRT. The present sensitivity analysis and subgroup analysis
revealed that when the control group in the study adopted
intensified dual-drug CCRT (such as cisplatin + 5-fluorouracil),
the trend of additional benefits brought by increasing IC was not
obvious. This finding aligns with the existing evidence-based
medical evidence. In the context of CCRT, multiple studies and
network meta-analyses have shown that the dual-drug combination
based on cisplatin does not demonstrate a clear superiority over
cisplatin monotherapy in terms of survival outcomes (12,40).
This suggests that there may be a ‘yield reduction’ effect in the
treatment intensity sequence for LACC. In other words, if the
radical CCRT stage itself is a potent treatment plan, then the
expected additional survival benefit from adding IC before it may
be relatively limited; conversely, in the clinical context where
the standard CCRT is based on single-agent cisplatin, adding
short-term IC can provide a clear improvement in treatment
intensity, which is consistent with the observed trend of ORR
improvement in the present study. Therefore, the clinical
decision-making for IC should be a meticulous balance process, with
the core purpose not being simply to stack chemotherapy drugs, but
rather to strategically utilize the systemic effect of IC to early
clear micrometastases and create more favorable local conditions
for subsequent radiotherapy by reducing the primary tumor size
based on the individual patient's condition and the treatment
background.
From the perspective of the mechanism, the benefits
of IC may go beyond the simple cytotoxic effect. Basic research
suggests that chemotherapy drugs such as platinum may have certain
immunomodulatory effects, such as reducing immunosuppressive cells
in the tumor microenvironment (41). This may partially explain the
positive trend observed in the present study of IC+CCRT in reducing
the rate of distant metastasis. Of course, the present study also
has several limitations. The total number and sample size of the
included RCTs are still limited, and in particular, there are few
studies reporting long-term survival outcomes. This may affect the
statistical confidence in the OS results. In addition, the
heterogeneity in radiotherapy techniques, specific chemotherapy
doses and adverse reaction recording standards among the studies,
although handled through random-effects models and sensitivity
analyses, may still affect the accuracy of some combined
estimates.
In conclusion, the present study provides important
evidence-based support for the application of modern short-course
intensive IC combined with CCRT in LACC, especially in patients who
subsequently receive standard-intensity CCRT. The future research
direction should focus on integrating multi-dimensional
information, such as tumor molecular characteristics, radiomics and
immune microenvironment markers, to construct a precise predictive
model, in order to more accurately identify the patient groups most
likely to benefit from this intensified treatment strategy. At the
same time, it should carefully explore the timing and sequence of
combining immunotherapy and other new modalities, ultimately
achieving individualized treatment and breaking through the
treatment bottleneck of LACC.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The study was supported by the Special Funds for Promoting
Scientific and Technological Innovation in Xuzhou in 2022 (grant
no. KC22255).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Author's contributions
YS and XL contributed to the conception, design and
writing of the manuscript. WX and TT were responsible for data
extraction and analysis. The first draft of the manuscript was
written by YS. FZ and DY contributed to the statistical analysis
and interpretation of results, ensuring the robustness of the
meta-analysis methodology. Manuscript revision and proofreading
were conducted by WX, XL, FZ, and DY. YS and XL confirm the
authenticity of all the raw data. YG, WX and YX contributed to the
study design and provided critical feedback on the methodology. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CCRT
|
concurrent chemoradiotherapy
|
|
CR
|
complete response
|
|
IC
|
induction chemotherapy
|
|
LACC
|
locally advanced cervical cancer
|
|
ORR
|
objective response rate
|
|
OS
|
overall survival
|
|
PFS
|
progression-free survival
|
|
PR
|
partial response
|
|
RCT
|
randomized controlled trial
|
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Zheng RS, Chen R, Han BF, Wang SM, Li L,
Sun KX, Zeng HM, Wei WW and He J: Cancer incidence and mortality in
China, 2022. Zhonghua Zhong Liu Za Zhi (Chinese). 46:221–231.
2024.PubMed/NCBI
|
|
3
|
Patel C, Brotherton JM, Pillsbury A,
Jayasinghe S, Donovan B, Macartney K and Marshall H: The impact of
10 years of human papillomavirus (HPV) vaccination in Australia:
What additional disease burden will a nonavalent vaccine prevent?
Euro Surveill. 23:17007372018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Giannone G, Giuliano AR, Bandini M,
Marandino L, Raggi D, Earle W, Ayres B, Pettaway CA, McNeish IA,
Spiess PE and Necchi A: HPV vaccination and HPV-related
malignancies: Impact, strategies and optimizations toward global
immunization coverage. Cancer Treat Rev. 111:1024672022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Simms KT, Steinberg J, Caruana M, Smith
MA, Lew JB, Soerjomataram I, Castle PE, Bray F and Canfell K:
Impact of scaled up human papillomavirus vaccination and cervical
screening and the potential for global elimination of cervical
cancer in 181 countries, 2020–99: A modelling study. Lancet Oncol.
20:394–407. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Toliman PJ, Kaldor JM, Tabrizi SN and
Vallely AJ: Innovative approaches to cervical cancer screening in
low- and middle-income countries. Climacteric. 21:235–238. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peters WA III, Liu PY, Barrett RJ II,
Stock RJ, Monk BJ, Berek JS, Souhami L, Grigsby P, Gordon W Jr and
Alberts DS: Concurrent chemotherapy and pelvic radiation therapy
compared with pelvic radiation therapy alone as adjuvant therapy
after radical surgery in high-risk early-stage cancer of the
cervix. J Clin Oncol. 18:1606–1613. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Whitney CW, Sause W, Bundy BN, Malfetano
JH, Hannigan EV, Fowler WC Jr, Clarke-Pearson DL and Liao SY:
Randomized comparison of fluorouracil plus cisplatin versus
hydroxyurea as an adjunct to radiation therapy in stage IIB-IVA
carcinoma of the cervix with negative para-aortic lymph nodes: A
Gynecologic Oncology Group and Southwest Oncology Group study. J
Clin Oncol. 17:1339–1348. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Morris M, Eifel PJ, Lu J, Grigsby PW,
Levenback C, Stevens RE, Rotman M, Gershenson DM and Mutch DG:
Pelvic radiation with concurrent chemotherapy compared with pelvic
and para-aortic radiation for high-risk cervical cancer. N Engl J
Med. 340:1137–1143. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rose PG, Bundy BN, Watkins EB, Thigpen JT,
Deppe G, Maiman MA, Clarke-Pearson DL and Insalaco S: Concurrent
cisplatin-based radiotherapy and chemotherapy for locally advanced
cervical cancer. N Engl J Med. 340:1144–1153. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Keys HM, Bundy BN, Stehman FB, Muderspach
LI, Chafe WE, Suggs CL III, Walker JL and Gersell D: Cisplatin,
radiation, and adjuvant hysterectomy compared with radiation and
adjuvant hysterectomy for bulky stage IB cervical carcinoma. N Engl
J Med. 340:1154–1161. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Datta NR, Stutz E, Liu M, Rogers S,
Klingbiel D, Siebenhüner A, Singh S and Bodis S: Concurrent
chemoradiotherapy vs. radiotherapy alone in locally advanced cervix
cancer: A systematic review and meta-analysis. Gynecol Oncol.
145:374–385. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Thomas GM: Improved treatment for cervical
cancer-concurrent chemotherapy and radiotherapy. N Engl J Med.
340:1198–1200. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Akyildiz A, Gultekin M, Yigit E, Demir E,
Ismayilov R, Ahmed M, Buyukkor M, Yildirim HC, Yildirim N, Ucar G,
et al: Efficacy of cumulative cisplatin dose on survival in
patients with locally advanced cervical cancer treated with
definitive chemoradiotherapy: multicenter study by Turkish Oncology
Group. Int J Gynecol Cancer. 34:1359–1365. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chemoradiotherapy for Cervical Cancer
Meta-Analysis Collaboration, . Reducing uncertainties about the
effects of chemoradiotherapy for cervical cancer: A systematic
review and meta-analysis of individual patient data from 18
randomized trials. J Clin Oncol. 26:5802–5812. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao C, Zhou Q, Liu L, Liu H, Yang Y, Qu S,
He Q, Huang Y, He X and Wang H: Stratification by mutational
landscape reveals differential immune infiltration and predicts the
recurrence and clinical outcome of cervical cancer. Phenomics.
5:384–403. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pal A, Das S, Basu S and Kundu R:
Apoptotic and autophagic death union by Thuja occidentalis
homeopathic drug in cervical cancer cells with thujone as the
bioactive principle. J Integr Med. 20:463–472. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mileshkin LR, Moore KN, Barnes EH, Gebski
V, Narayan K, King MT, Bradshaw N, Lee YC, Diamante K, Fyles AW, et
al: Adjuvant chemotherapy following chemoradiotherapy as primary
treatment for locally advanced cervical cancer versus
chemoradiotherapy alone (OUTBACK): An international, open-label,
randomised, phase 3 trial. Lancet Oncol. 24:468–482. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gupta S, Maheshwari A, Parab P,
Mahantshetty U, Hawaldar R, Sastri Chopra S, Kerkar R, Engineer R,
Tongaonkar H, Ghosh J, et al: Neoadjuvant chemotherapy followed by
radical surgery versus concomitant chemotherapy and radiotherapy in
patients with stage IB2, IIA, or IIB squamous cervical cancer: A
randomized controlled trial. J Clin Oncol. 36:1548–1555. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
de Azevedo CR, Thuler LC, de Mello MJ and
Ferreira CG: Neoadjuvant chemotherapy followed by chemoradiation in
cervical carcinoma: A review. Int J Gynecol Cancer. 26:729–736.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Neoadjuvant Chemotherapy for Locally
Advanced Cervical Cancer Meta-analysis Collaboration, . Neoadjuvant
chemotherapy for locally advanced cervical cancer: A systematic
review and meta-analysis of individual patient data from 21
randomised trials. Eur J Cancer. 39:2470–2486. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Glynne-Jones R and Hoskin P: Neoadjuvant
cisplatin chemotherapy before chemoradiation: A flawed paradigm? J
Clin Oncol. 25:5281–5286. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
McCormack M, Kadalayil L, Hackshaw A,
Hall-Craggs MA, Symonds RP, Warwick V, Simonds H, Fernando I,
Hammond M, James L, et al: A phase II study of weekly neoadjuvant
chemotherapy followed by radical chemoradiation for locally
advanced cervical cancer. Br J Cancer. 108:2464–2469. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
McCormack M, Eminowicz G, Gallardo D, Diez
P, Farrelly L, Kent C, Hudson E, Panades M, Mathew T, Anand A, et
al: Induction chemotherapy followed by standard chemoradiotherapy
versus standard chemoradiotherapy alone in patients with locally
advanced cervical cancer (GCIG INTERLACE): An international,
multicentre, randomised phase 3 trial. Lancet. 404:1525–1535. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Eminowicz G, Vaja S, Gallardo D, Kent C,
Panades M, Mathew T, Anand A, Forrest J, Adusumalli M, Chan A, et
al: Induction chemotherapy followed by chemoradiation in locally
advanced cervical cancer: Quality of life outcomes of the GCIG
INTERLACE trial. Eur J Cancer. 220:1153752025. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li J, Li Y, Wang H, Shen L, Wang Q, Shao
S, Shen Y, Xu H, Liu H, Cai R and Feng W: Neoadjuvant chemotherapy
with weekly cisplatin and paclitaxel followed by chemoradiation for
locally advanced cervical cancer. BMC Cancer. 23:512023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gadducci A, Barsotti C, Laliscia C, Cosio
S, Fanucchi A, Tana R and Fabrini MG: Dose-dense paclitaxel- and
carboplatin-based neoadjuvant chemotherapy followed by surgery or
concurrent chemo-radiotherapy in cervical cancer: A preliminary
analysis. Anticancer Res. 37:1249–1255. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Borghi C, Biagioli E, Mauro J, Roberto A,
Borghese M and Buda A: Neoadjuvant chemotherapy prior to radical
hysterectomy in locally advanced cervical cancer: A systematic
review and meta-analysis. Int J Gynecol Cancer. 34:35–46. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bhatla N, Aoki D, Sharma DN and
Sankaranarayanan R: Cancer of the cervix uteri. Int J Gynaecol
Obstet. 143 (Suppl 2):22–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Guyatt GH, Oxman AD, Vist GE, Kunz R,
Falck-Ytter Y, Alonso-Coello P and Schünemann HJ; GRADE Working
Group, : GRADE: An emerging consensus on rating quality of evidence
and strength of recommendations. BMJ. 336:924–926. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schünemann HJ, Higgins JP, Vist GE,
Glasziou P, Akl EA, Skoetz N and Guyatt GH; on behalf of the
Cochrane GRADEing Methods Group (formerly Applicability and
Recommendations Methods Group) the Cochrane Statistical Methods
Group, : Completing ‘Summary of findings’ tables and grading the
certainty of the evidence. Cochrane Handbook for Systematic Reviews
of Interventions. Higgins JPT, Thomas J, Chandler J, Cumpston M, Li
T, Page MJ and Welch VA: Wiley; pp. 375–402. 2019, View Article : Google Scholar
|
|
33
|
Tripathi A and Rawat S: Comparative study
of neoadjuvant chemotherapy followed by definitive
chemoradiotherapy versus definitive chemoradiotherapy alone in
locally advanced carcinoma of cervix. J Obstet Gynecol India.
69:546–552. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Banerjee C, Banerjee S, Dutta S and Bera
A: Treatment outcome and toxicity of neoadjuvant chemotherapy
followed by concomitant chemoradiotherapy versus concomitant
chemoradiation alone in locally advanced cervical carcinoma: A
comparative study. Asian J Pharm Clin Res. 15:114–118. 2022.
View Article : Google Scholar
|
|
35
|
Li F, Mei F, Yin S, Du Y, Hu L, Hong W and
Li J: Improving the efficacy and safety of concurrent
chemoradiotherapy by neoadjuvant chemotherapy: A randomized
controlled study of locally advanced cervical cancer with a large
tumor. J Gynecol Oncol. 35:e102023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Singh R, Bhatt MLB, Kumar R, Srivastava K,
Grover RK, Shukla P, Raturi VP, Roopali Gaur J, Saha M and Kishan
D: A prospective randomized comparison of concurrent chemoradiation
with neoadjuvant and adjuvant chemotherapy with concurrent
chemoradiation alone for locally advanced carcinoma cervix. Indian
J Med Paediatr Oncol. 40:353–357. 2019. View Article : Google Scholar
|
|
37
|
da Costa SCS, Bonadio RC, Gabrielli FCG,
Aranha AS, Dias Genta MLN, Miranda VC, de Freitas D, Abdo Filho E,
Ferreira PAO, Machado KK, et al: Neoadjuvant chemotherapy with
cisplatin and gemcitabine followed by chemoradiation versus
chemoradiation for locally advanced cervical cancer: A randomized
phase II trial. J Clin Oncol. 37:3124–3131. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nguyen VT, Winterman S, Playe M, Benbara
A, Zelek L, Pamoukdjian F and Bousquet G: Dose-intense
cisplatin-based neoadjuvant chemotherapy increases survival in
advanced cervical cancer: An up-to-date meta-analysis. Cancers
(Basel). 14:8422022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Park DC, Kim JH, Lew YO, Kim DH and
Namkoong SE: Phase II trial of neoadjuvant paclitaxel and cisplatin
in uterine cervical cancer. Gynecol Oncol. 92:59–63. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kim YS, Shin SS, Nam JH, Kim YT, Kim YM,
Kim JH and Choi EK: Prospective randomized comparison of monthly
fluorouracil and cisplatin versus weekly cisplatin concurrent with
pelvic radiotherapy and high-dose rate brachytherapy for locally
advanced cervical cancer. Gynecol Oncol. 108:195–200. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li JY, Chen YP, Li YQ, Liu N and Ma J:
Chemotherapeutic and targeted agents can modulate the tumor
microenvironment and increase the efficacy of immune checkpoint
blockades. Mol Cancer. 20:272021. View Article : Google Scholar : PubMed/NCBI
|