Introduction
Perianal mucinous adenocarcinoma (PMA) is a rare
disease and accounts for <5% of all gastrointestinal
malignancies (1,2). Its occurrence is associated with
chronic perianal infections such as anal fistula and Crohn's
disease (3). The common symptoms
are recurrent perianal abscess, perianal pain and bleeding
(4–6), and the manifestation of a painless
perianal mass is rare (7) with no
clear data in the literature. As the early symptoms of PMA overlap
with benign conditions such as perianal abscesses and anal
fissures, the pathological biopsy is also affected by the presence
of mucus alone and may result in a missed diagnosis (1,7). The
pathological features of PMA are similar to those of colorectal
mucinous adenocarcinoma, including invasive glands lined with
columnar cells that secrete abundant extracellular mucus (7,8). A
KRAS gene mutation is common in colorectal mucinous adenocarcinoma
(CMA) (9), but to the best of our
knowledge, there are fewer reports of KRAS gene mutations in PMA
(5). There is no standardized
treatment protocol, and the main treatment techniques used in a few
cases have been radical surgery, adjuvant radiotherapy and
chemotherapy, and partial wide resection (7,10,11).
Studies on painless PMA are relatively scarce, and research on its
pathogenesis, diagnostic procedures and prognosis assessment is
limited. There is a lack of systematic analysis of painless cases
without chronic inflammation or a history of fistula, which leads
to a lack of understanding and diagnostic experience among doctors
for this special type of case.
The painless PMA in the present case report was
unique: The patient had no history of chronic perianal diseases
such as anal fistula or Crohn's disease, and there were no clear
chronic inflammatory or fistula-related pathogenic factors, which
are notably different from the disease backgrounds of the majority
of patients with PMA. Due to the presence of atypical clinical
manifestations and based on auxiliary examinations, painless PMA is
easily misdiagnosed as a benign tumor, resulting in a lack of
further examinations and the inability to perform the extended
radical resection. In the present case report, relevant studies
(1,2,7,9) have
been combined to analyze the clinical, pathological,
immunohistochemical, treatment and prognosis characteristics of
this disease, aiming to improve the understanding among doctors and
pathologists.
Case report
A 79-year old male patient with a 15-day old
painless perianal mass was admitted to the First People's Hospital
of Xiaoshan (Hangzhou, China) in January 2024. While conducting an
anal examination in the lithotomy position, a mass under the
epidermis was palpated. The mass was located between the 12 O'clock
and 2 O'clock directions, 3.0 cm away from the anal margin. It had
a volume of 4.0×3.0×2.0 cm, clear boundaries, soft texture and no
notable tenderness or fluctuation sensation. Bilateral superficial
inguinal lymph nodes were not enlarged. Digital rectal examination
showed no anal fistula or ulcer, and smooth rectal mucosa.
Additionally, no other mass was touched, and no blood or purulent
discharge was observed on the glove of the examining physician.
Colonoscopy of the patient revealed no abnormalities in the colon.
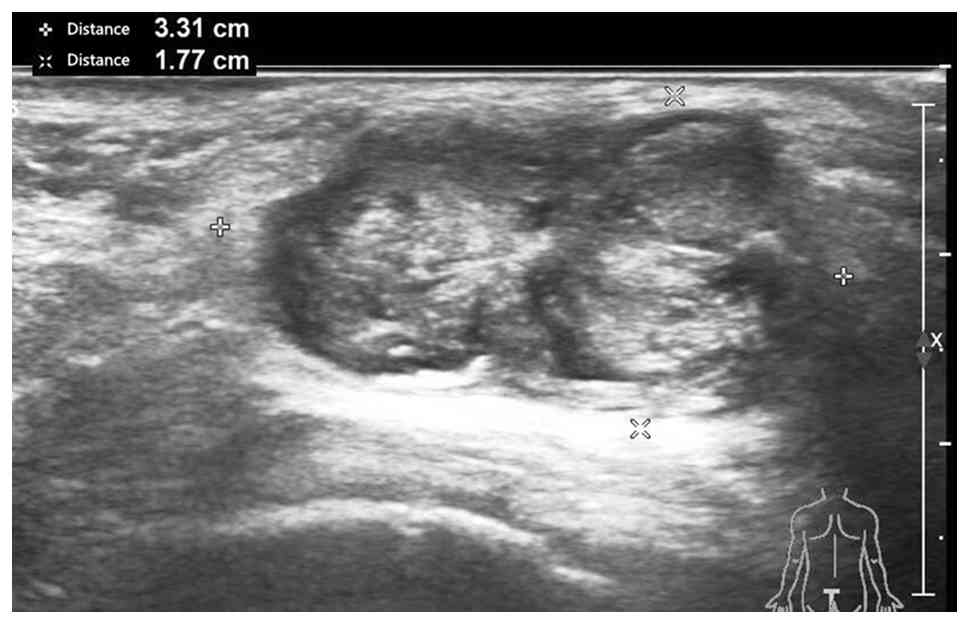
A B-ultrasound examination revealed a low heterogeneous echogenic
structure (3.31×1.90×1.77 cm) on the left side of the anal canal.
The area had distinct boundaries and a regular shape, with no
notable blood flow signal (Fig. 1);
thus, the mas was considered to be a benign tumor. Detection of
tumor serological markers was performed by following the
manufacturer's instructions (Abbott Trading (Shanghai) Co., Ltd.)
showed that squamous cell carcinoma-associated antigen was 0.92
ng/ml (normal range <1.50 ng/ml), AFP was 0.92 ng/ml (normal
range, 0.89–8.78 ng/ml), sugar chain antigen 125 was 5.80 U/ml
(normal range, 0–35 U/ml), carbohydrate antigen 199 was 6.70 U/ml
(normal range, 0–37 U/ml) and carcinoembryonic antigen was 3.94
ng/ml (normal range, 0–5 ng/ml). The normal ranges of all reagents
were provided by Abbott Trading (Shanghai) Co., Ltd. and verified
by the Clinical Laboratory Department of the First People's
Hospital of Xiaosha, Hangzhou, Zhejiang.
Medical history. The patient was previously healthy
and did not have a history of any disease such as cardiovascular or
cerebrovascular diseases, type 2 diabetes mellitus, chronic kidney
disease, chronic bronchitis, bronchial asthma, chronic gastritis,
chronic diarrhea or chronic pancreatitis. The patient denied
experiencing anal fistula, hemorrhoid disease or any other surgical
history. The patient also did not have a family history of
hereditary or similar diseases.
The preoperative diagnosis was a perianal mass and
the lesion was resected under general anesthesia. During the
operation, a mass ~4.0×3.0 cm in size was found; it had distinct
boundaries and appeared as a capsule. It was movable and found
between the 12 O'clock and the 2 O'clock position in the lithotomy
position, 3 cm away from the anal margin. It did not involve the
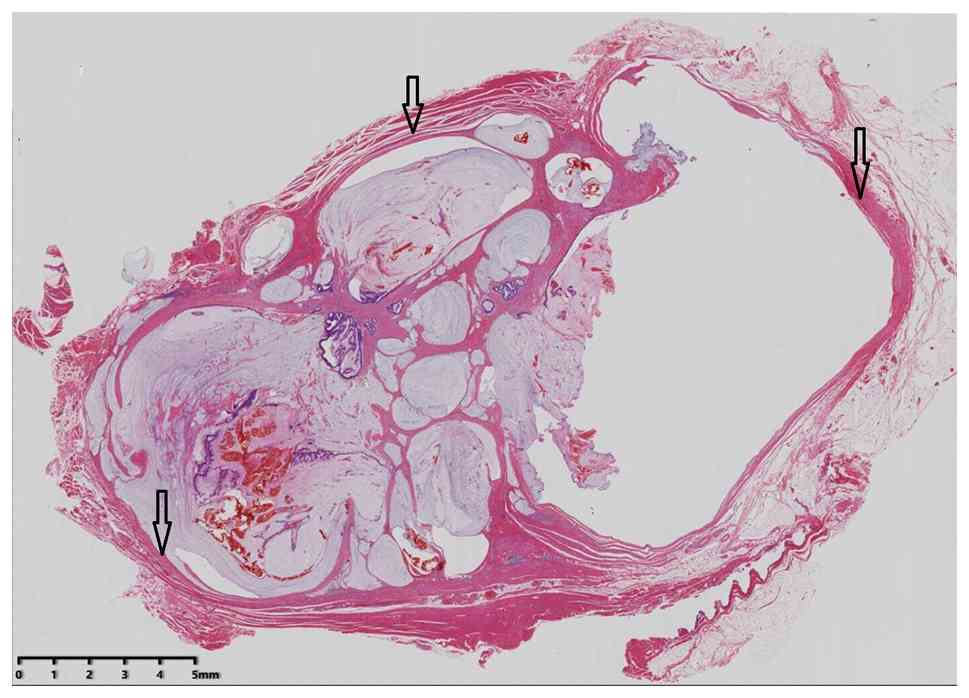
anal canal or the rectal wall. A gross pathological examination
revealed soft, gray-yellow tissue with a volume of 3.5×3.0×3.0 cm,
with a cystic, solid cut surface and jelly-like substance
inside
The tissue was fixed with 10% neutral formalin for
24 h at 25°C and embedded in paraffin. Next, serial sections (3-µm
thick) were prepared and subjected to hematoxylin and eosin
(H&E) staining (Shanghai Rongbai Biological Technology
Development Co., Ltd., and Sinopharm Chemical Reagent Co., Ltd. for
3 h at 25°C. The samples were observed using a light microscope.
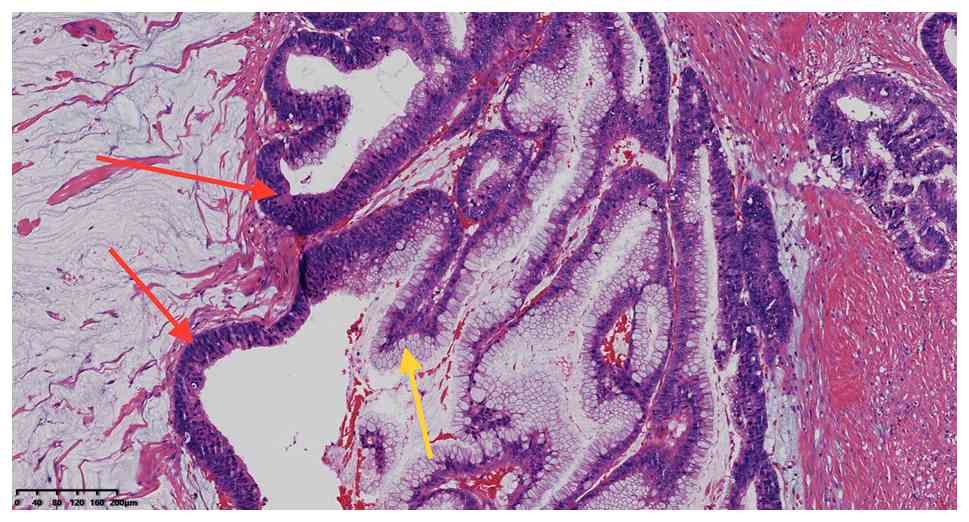
Microscopic examination revealed a nodular, expansile growth of the
tumor within fibrous and striated muscle tissue, irregular glands
secreting large amounts of mucus (Fig.
2) and the columnar and goblet cells presenting with low-grade
intraepithelial neoplasia (Fig. 3).
The tumor margin was negative under the microscope.
Immunohistochemistry (IHC) analysis was performed
using an EnVision IHC kit (polymer method; cat. no. KIT-0014;
Fuzhou Maixin Biotechnology Development Co., Ltd.) using primary
antibodies (incubated for 50 min at 25°C) purchased from Beijing
Zhongshan Jinqiao Biological Co., Ltd. and Fuzhou Maixin
Biotechnology Development Co., Ltd., to target the following
proteins (pre-diluted working solutions unless otherwise
indicated): CK7 (1:200; cat. no. ZM-0071), CK20 (1:200; cat. no.
ZA-0574), CDX2 (working fluid; cat. no. RMA-0631), Mucin 2 (MUC2;
working fluid; cat. no. ZM-0392), MutL homolog 1 (MLH1; 1:100; cat.
no. MAB-0838), MutS Homolog 2 (MSH2; 1:200; cat. no. MAB0836), MutS
Homolog 6 (MSH6; 1:200; cat. no. MAB-0831), Mismatch repair
endonuclease PMS2 (PMS2; 1:200; cat. no. RMA-0775), p53 (1:100;
cat. no. ZM-0408) and Ki-67 proliferation index (1:200; cat. no.
ZM-0167). IHC sections were observed under a light microscope
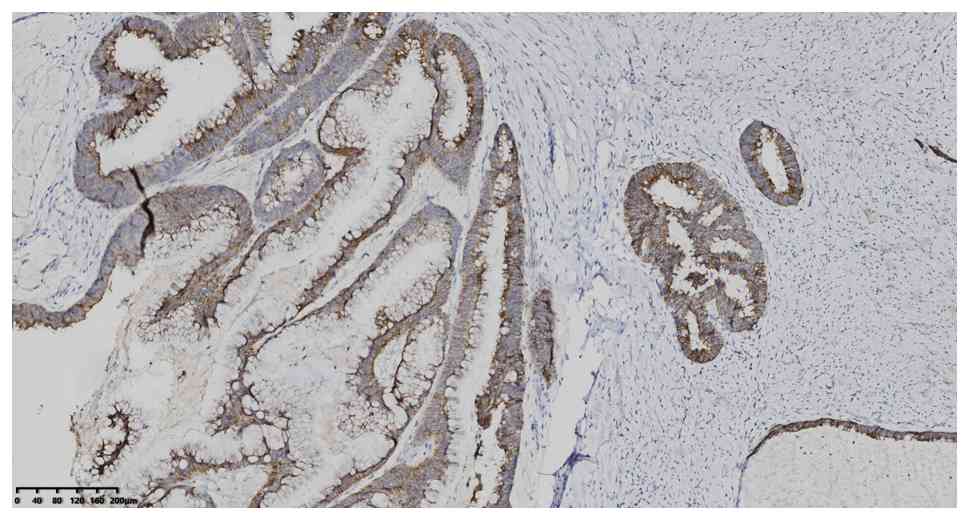
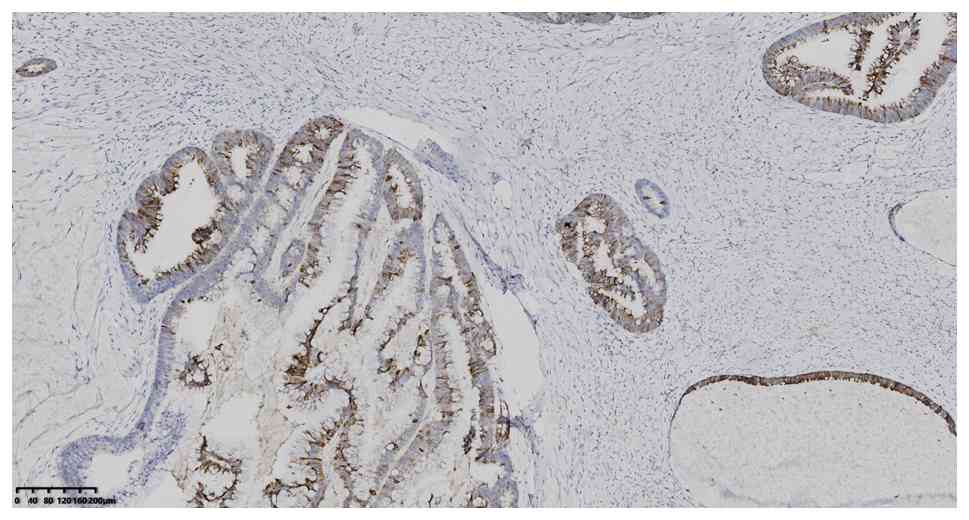
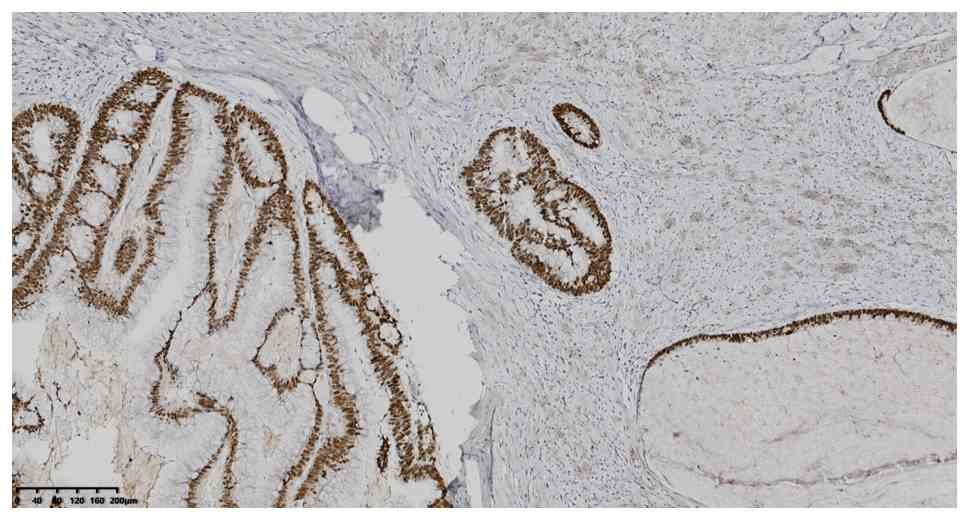
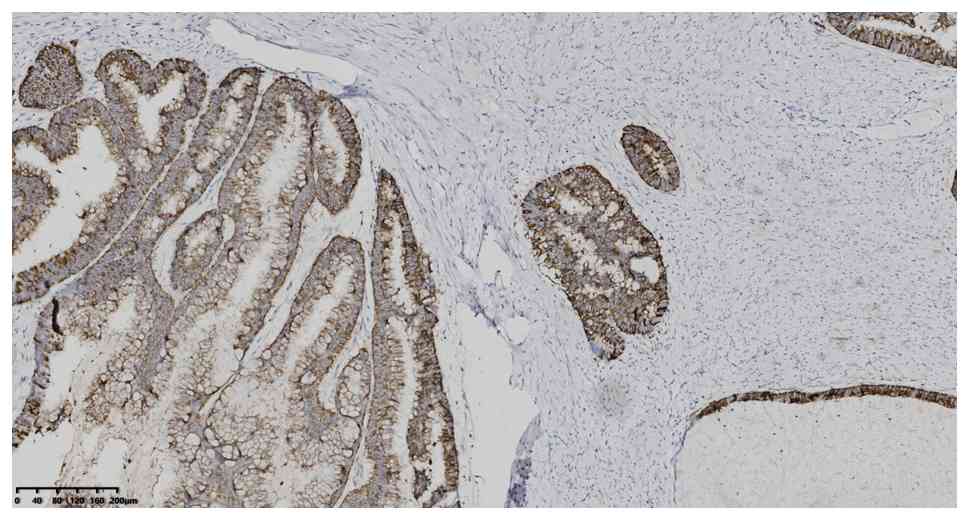
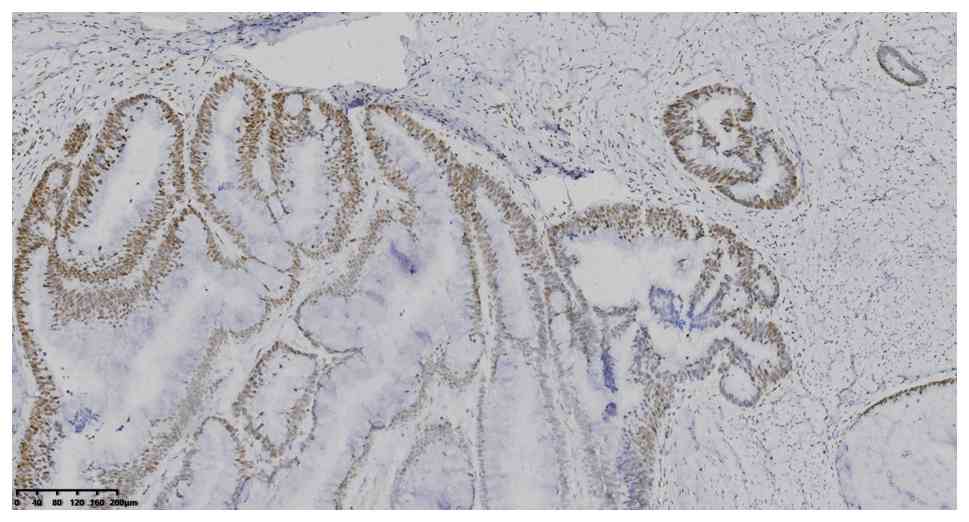
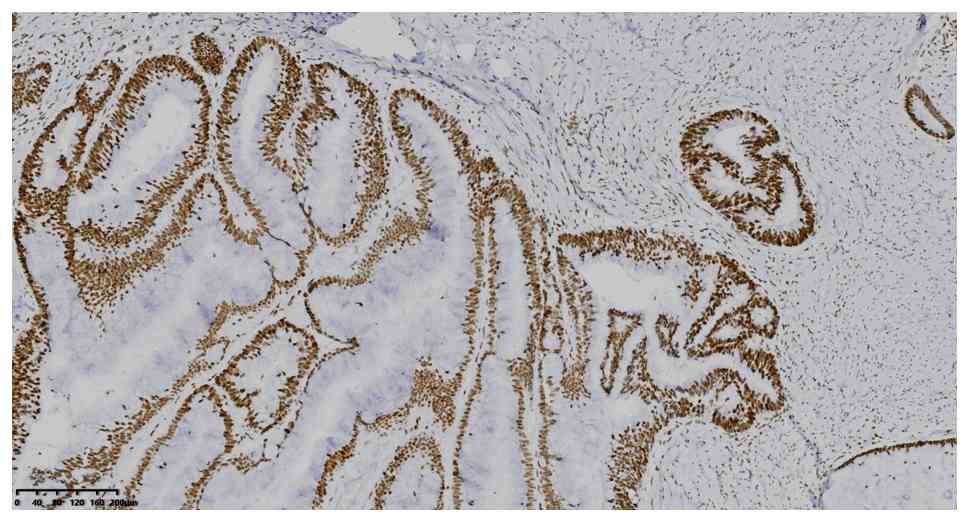
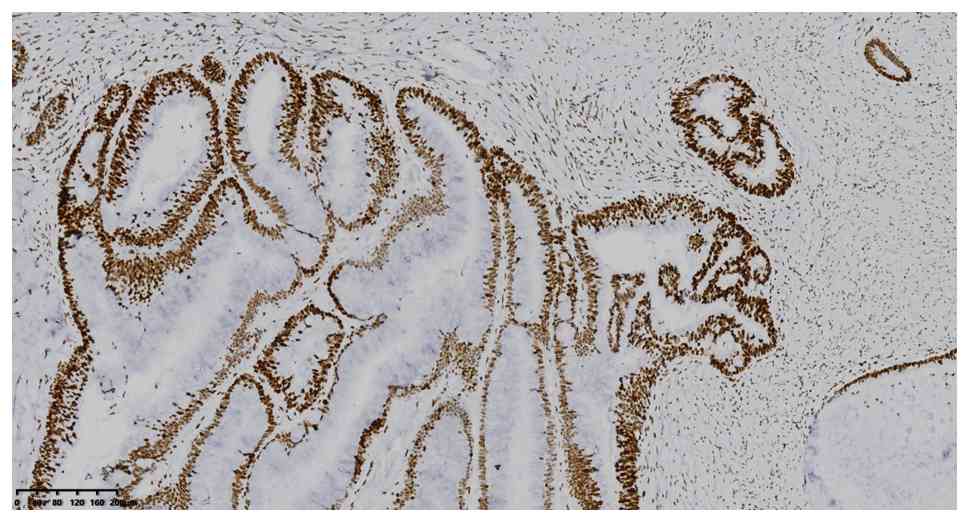
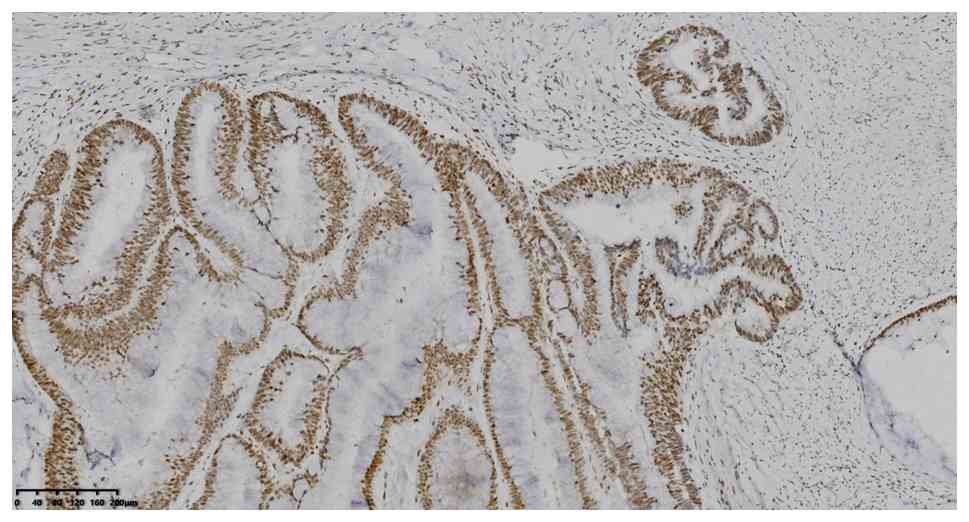
without software analysis. IHC analysis showed positive expression
of CK7, CK20, CDX2 and MUC2 (Fig.
4, Fig. 5, Fig. 6, Fig.
7), MLH1, MSH2, MSH6 and PMS2 (Fig.
8, Fig. 9, Fig. 10, Fig.
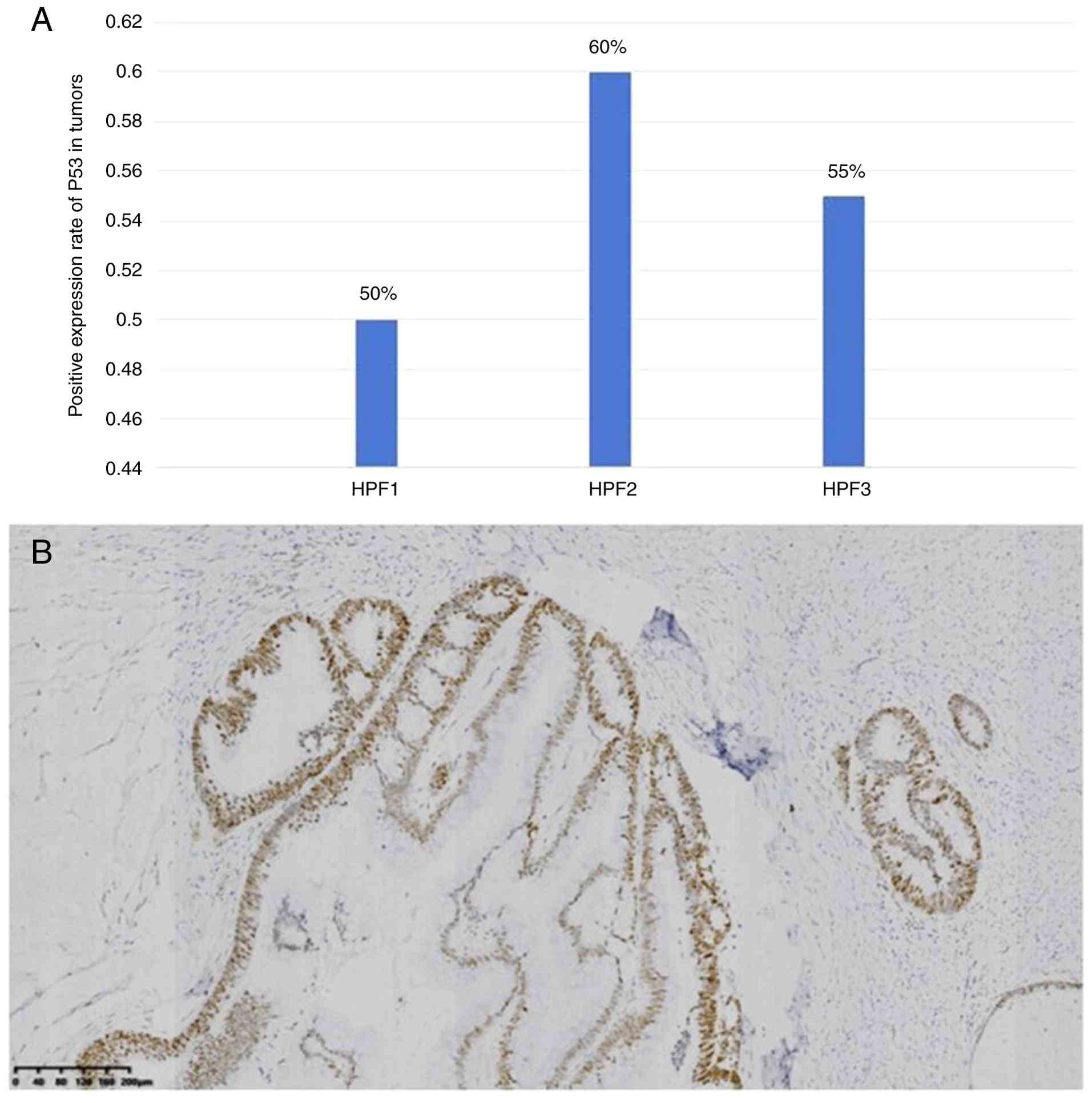
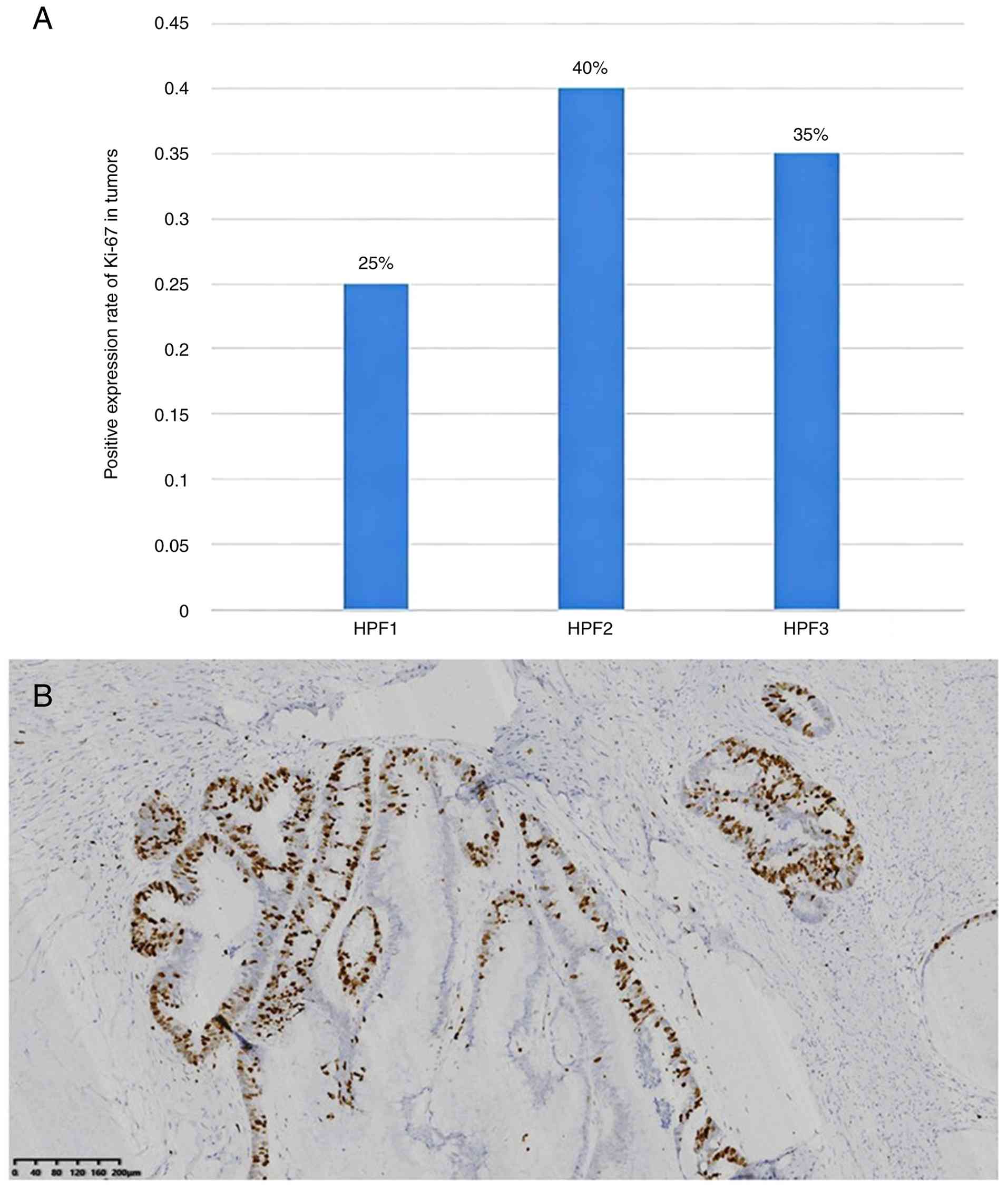
11). The positive expression rate of p53 was 55% (Fig. 12A-B), and the Ki-67 proliferation
index was 33% (Fig. 13A and B).
The methods described in the case report are the same for the
immunohistochemical staining shown in Fig. 8, Fig.
9, Fig. 10, Fig. 11, Fig.
12 (MLH1, MSH2, MSH6, PMS2 and p53), and this standardized IHC
protocol was also used for the other IHC stains (CK7, CK20, CDX2,
MUC2 and Ki-67) reported in the present study. Immunohistochemical
analysis was performed using the EnVision IHC kit (polymer method;
cat. no. KIT-0014; Fuzhou Maixin Biotechnology Development Co.,
Ltd.) with primary antibodies targeting MLH1, MSH2, MSH6, PMS2, and
p53; these primary antibodies were obtained from Beijing Zhongshan
Jinqiao Biological Co., Ltd. and Fuzhou Maixin Biotechnology
Development Co., Ltd., with specified dilutions for each antibody
(MLH1, 1:100; MSH2, 1:200; MSH6, 1:200; PMS2, 1:200; p53, 1:100).
The primary antibody incubation step was carried out at 25°C for 50
min for all targeted proteins in Fig.
8, Fig. 9, Fig. 10, Fig.
11, Fig. 12. Following the
antibody incubation and subsequent kit-based staining procedures,
the prepared IHC sections were examined directly under a light
microscope and no software-based image or quantitative analysis was
conducted for the initial qualitative staining assessment. For p53
(Fig. 12), additional quantitative
analysis was performed by calculating the mean percentage of
positively stained cells across three high-power fields (HPFs) to
determine a 55% positive nuclear staining rate reported for the
tumor cells. All staining for the proteins in Fig. 8, Fig.
9, Fig. 10, Fig. 11, Fig.
12 utilized hematoxylin (for 3 min at 25°C) as a counterstain,
which produced blue nuclear staining to contrast with the brown
cytoplasmic/nuclear staining indicative of positive antibody
binding for the target proteins. The stained sections were imaged
at a magnification of 100×, with a 200 µm scale bar included for
all micrographs presented in Fig.
8, Fig. 9, Fig. 10, Fig.
11, Fig. 12B.
Molecular testing was performed by KingMed
Diagnostics (next-generation sequencing; Illumina sequencing
platform, reference genome: GRCh37/hg19). Sample Preparation
Reagents and Verification Methods. Reagents for sample preparation
used in the present study are summarized in Table I. Specifically, for nucleic acid
extraction from FFPE samples, the One-step extraction
(High-sensitivity FFPE Genomic DNA Kit; cat. no. RC1102) was
purchased from Kaishuo Biotechnology (Xiamen) Co., Ltd. Library
construction and hybridization were performed using the products
developed by NanJing Vazyme Biotech Co., Ltd. (VAHTS Universal Plus
DNA Library Prep Kit V4). Its composition related to library
construction and hybridization is specified in detail as follows:
For hybridization, the core reagents include a set of biotinylated
DNA/RNA capture probes (120–150 bp), Human Cot-1 DNA and Universal
blocking oligos (serving as blocking reagents), high-salt
hybridization buffer containing formamide, low-stringency wash
buffer (2× SSC; 0.1% SDS), high-stringency wash buffer (0.1× SSC;
0.1% SDS), streptavidin-coated magnetic beads and elution buffer
(0.1 M NaOH or Tris-HCl); for library construction, the required
reagents consist of fragmentation enzyme, T4 DNA polymerase, Klenow
fragment, T4 polynucleotide kinase (for DNA fragmentation and end
repair), Klenow fragment (3′→5′ exo-) and T4 DNA ligase (for
A-tailing and adapter ligation), as well as dual-indexed sequencing
adapters (with UMIs), high-fidelity DNA polymerase, dNTP mix (dATP,
dTTP, dCTP, dGTP) and primer mix (for library amplification). In
addition, quality control reagents including qPCR master mix with
reference standards and fragment analyzer reagents were also used
in the whole process to ensure the reliability of the experiment.
The NovaSeq 6000 S1 v1.5 Kit (cat. no. 20028317) from Illumina,
Inc. was employed for sequencing.
 | Table I.Reagents for sample preparation |
Table I.
Reagents for sample preparation
| Reagent
application | Kit name | Cat.
no./specification | Supplier |
|---|
| Nucleic acid
extraction | One-step extraction
(High-sensitivity | RC1102 | Xiamen Biosan |
| (FFPE samples) | FFPE Genomic DNA
Kit) |
| Biotech Co.,
Ltd. |
| Library
construction | Jinyu In-house LDT
Solid | No cat. no.
(in-house | KingMed |
| and
hybridization | Tumor Mini panel | reagent) | Diagnostics |
| Sequencing | NovaSeq 6000 S1
v1.5 Kit | 20028317 | Illumina, Inc. |
Methods for verification of processed sample
quality/integrity. The nucleic acid concentration and library
concentration of the processed samples were verified using the
Qubit assay to ensure that the samples met the requirements for
subsequent sequencing and analysis.
Sequencing type. Next-generation sequencing was
adopted as the sequencing type, with a read length and sequencing
mode of 2×150 bp paired-end sequencing.
Sequencing platform. The Illumina NovaSeq 6000
platform was used for sequencing experiments.
Final library loading concentration and
quantification method. For the Illumina NovaSeq 6000 platform, the
final library loading concentration was set to 1 nM, and the
concentration was quantified by the Qubit assay.
Analysis revealed a KRAS mutation, 35G>A p. G12d,
with a mutation frequency of 15.1% (Table II). MSI testing was stable. The
final diagnosis was confirmed as PMA, pathological tumor 3, node 0,
metastasis 0 (pT3N0M0), Stage IIA
 | Table II.Results of genetic testing. |
Table II.
Results of genetic testing.
| Genes | Nucleotide
changes | Amino acid
changes | Frequency of
variation | Classification of
variation |
Targeting/immunization treatment
options |
|---|
| KRAS | c.35G>A | p.G12D | 15.10% | I | NA |
| BRAF | ND | NA | NA | NA | NA |
| MLH1 | ND | NA | NA | NA | NA |
| MSH2 | ND | NA | NA | NA | NA |
| MSH6 | ND | NA | NA | NA | NA |
| NRAS | ND | NA | NA | NA | NA |
| NTRK1 | ND | NA | NA | NA | NA |
| PIK3CA | ND | NA | NA | NA | NA |
| PMS2 | ND | NA | NA | NA | NA |
| POLE | ND | NA | NA | NA | NA |
| RET | ND | NA | NA | NA | NA |
| POLD1 | ND | NA | NA | NA | NA |
| AKT1 | ND | NA | NA | NA | NA |
| ARAF | ND | NA | NA | NA | NA |
| RAF1 | ND | NA | NA | NA | NA |
| FGFR1 | ND | NA | NA | NA | NA |
| FGFR2 | ND | NA | NA | NA | NA |
| FGFR3 | ND | NA | NA | NA | NA |
Treatment and follow-up. After the first operation,
the doctor found that the tumor boundary was distinct and covered.
The pathological examination showed no residual cancer cells at the
resection edge. The doctor was not knowledgeable about the precise
postoperative examination selection for malignant tumors.
Therefore, only the enhanced computed tomography (CT) scanning of
the upper abdomen of the patient and the middle part of the lower
abdomen was performed and magnetic resonance imaging (MRI) was
omitted. The results revealed soft tissue swelling in the perianal
area after the surgery. The size, shape and density of the liver,
spleen, gallbladder, pancreas, bilateral ureters and bladder were
normal, and no significant thickening of the rectal wall was found.
Small cysts and stones were found in the bilateral kidneys.
At that time, the clinician discharged the patient
following the tumor resection, and the patient recuperated at home
while awaiting the results of the routine pathological examination.
After a pathological diagnosis confirmed the perianal tumor to be
malignant, the clinician determined that the initial surgical
procedure had merely involved the resection of the lesion under a
benign tumor presumption, resulting in an inadequate oncologic
safety margin. Concurrently, the age of the patient (79 years) was
not suitable for immediate surgery, therefore sphincter-preserving
local extended radical resection was performed 1 month after the
diagnosis of PMA. The surgeon made a circular incision on the
surface of the skin 4.0 cm from the perimeter of the previous
surgical scar, cut the subcutaneous and adipose tissue of the skin
and extended the resection of the previous surgical scar and
surrounding tissue, inward to the external anal sphincter. Frozen
section examination is used during radical resection, and the
procedure is as follows: i) The pathology department verified the
specimen and requisition form, then macroscopically selected the
lesion tissue. ii) The tissue was embedded in optimal cutting
temperature compound and frozen to a hard block at −25°C. iii)
Sections that were 5 µm thick were prepared and rapidly fixed with
95% ethanol (25°C; 1–2 min). iv) Rapid H&E staining and
mounting was preformed (25°C; 8 min). v) A pathologist performed
rapid microscopic diagnosis, issues an intraoperative report and
informed the surgeon. Upon examining frozen sections of the
surgical margin, no cancer tissue was found.
Pathological examination of the resected specimen
(performed byhF&E staining and light microscopy observation)
showed focal chronic inflammatory cell infiltration (predominantly
lymphocytes and plasma cells) in the fibroadipose tissue, and
histological evaluation of the surrounding surgical margins
revealed no residual tumor cells, and no vascular or lymphatic
invasion, indicating a negative margin. The comprehensive
evaluation included postoperative clinical assessment (physical
examination, routine laboratory tests and ECOG score),
contrast-enhanced CT/MRI of the chest, abdomen and pelvis for
excluding local and distant metastasis, pathological TNM staging
and risk factor analysis. The patient did not require any
additional treatment, including radiotherapy or chemotherapy.
During the postoperative period (≤30 days of surgery), the patient
had normal stool passage, no abnormal anal discharge and no
soiling. Recurrence or metastasis was not detected at the 12-month
follow-up.
Discussion
PMA is a rare disease and accounts for <5% of all
gastrointestinal malignancies (1,2). A
literature summary from 1990–2024 by Gkegkes et al (7) revealed that among the 150 cases
analysed in Gkegkes' study, the average age of patients with PMA
was 60.5 years, and 82.7% were men (men to women ratio, 4.77:1.00).
The majority of cases were associated with chronic anal fistula or
Crohn's disease (disease duration, 5–20 years). Commonly reported
symptoms were perianal pain, bleeding or recurrent abscess
(7). Other study has reported that
anal fistulas may become malignant 2 years after its formation
(12), however the patient in the
present case report had no definite history of anal fistula, and
only a painless mass was found that was 15 days old. This suggests
that there may be the presence of a subclinical glandular malignant
process, and further emphasizing the need for clinicians to be
alert to atypical perianal masses.
Regarding the pathogenesis, PMA may be associated
with chronic inflammation, the upregulation of MUC2 and mutations
in the Ras/MAPK pathway (8,9,13,14).
MUC2 is a glycoprotein secreted by intestinal goblet cells, and its
upregulation is closely associated with CMA (13). KRAS gene mutation is a common
carcinogenic mechanism, and the mutation leads to the continuous
proliferation of cells due to the loss of GTPase activity (15,16).
Moreover, a study by Li et al (9) showed 57.1% (28/49) of patients with
CMA had KRAS mutations. In the present case report, the patient had
no chronic inflammation, no genetic history, but did have
microsatellite stability and MUC2-positive IHC. A KRAS p.G12d
activating mutation was detected in the present case report, which
aligns with the aforementioned literature findings on the CMA
pathogenic mechanism involving MUC2 upregulation and KRAS mutation,
and it is speculated that these two factors are the core pathogenic
factors in this case.
KRAS mutation is an important target in the
molecular mechanism of CRC, but the clinical value of different
subtypes of mutations differs. Drug treatment regimens
(sotorasib/adagrasib combined with cetuximab/panitumumab) have been
recommended by the National Comprehensive Cancer Network (NCCN)
guidelines for patients with CRC harboring KRAS p. G12c mutations
(15). Although the KRAS p. G12d
mutation has been detected in CRC (17), there is no approved drug for
targeted therapy.
Diagnostic and typing value. The KRAS p. G12d
mutation can be used as a molecular marker of PMA. Combined with
MUC2 expression and other pathological features, it can help
distinguish PMA from other perianal malignant tumors, such as sweat
gland mucinous adenocarcinoma of skin and lung metastatic
adenocarcinoma, especially for PMA cases without chronic
inflammation. Treatment direction. Although no targeted drug
is known, the presence of the KRAS p. G12d mutation provides a
direction for subsequent treatment (preclinical trials are ongoing)
(17).
Microscopically, the dysplastic glands within the
tumor showed infiltrative growth, mucus lake formation and mild to
severe glandular epithelial dysplasia (5). The typical immunohistochemical
findings of non-anal gland, non-fistula associated mucinous
adenocarcinomas suggest variable features: CK7(+), CK20(+) and
CDX2(+) (8,11) or CK7(−), CK20 (+) and CDX2(+)
(18). In the present case report,
microscopically, the patient presented with expansive nodular
growth with typical morphological changes of mucinous
adenocarcinoma. Immunohistochemically, the patient was positive for
CK20, CDX2 and CK7, which was consistent with the results reported
in the literature (7,10). Therefore, based on the medical
history of the patient, H&E staining results and
immunohistochemical findings, it is speculated that the tumor may
have arisen from non-fistula-related glandular structures (acquired
or congenital malformations or associated with embryonic residues)
(8,16)
The differential diagnosis of PMA include the
following: i) Infiltration or metastasis of CMA to the perianal
tissue, such as the case reported by Spiridakiset et al
(19), synchronous adenocarcinoma
of the rectum and sigmoid colon implanted in the anal fistula,
manifested as recurrent perianal abscess. However, colorectal
adenocarcinoma immunohistochemical phenotype (CK7) is usually
negative and can be distinguished; ii) mucinous adenocarcinoma
derived from sweat glands of the skin is also rare, with prominent
eosinophilic cytoplasm and positive expression of GCDFP-15 and no
expression of CK20 and CDX2; and iii) gastrointestinal
adenocarcinoma metastasis to the perianal region: A primary
gastrointestinal tumor was excluded in the patient through
colonoscopy and abdominal CT examination. Moreover, fistula or
Crohn's disease was absent in the anorectal area. Therefore, the
diagnosis of primary PMA was confirmed
There were prominent limitations in imaging
evaluation in the present case report: The first B-ultrasound
examination misjudged the hypoechoic mass as benign, and the
anatomical relationship between the tumor and the anorectal part
was not clear due to the lack of anal dual-plane ultrasound
equipment (EAUS) and pelvic CT/MRI before the operation, which
reflected that the doctor lacked clinical knowledge of PMA imaging
examinations. Combined with previous studies and clinical practice,
several preoperative imaging recommendations are proposed for
suspected perianal masses.
Basic examination. EAUS is preferred for the
diagnosis of perianal mass (7).
EAUS can identify the ‘multiple hypoechoic lesions’ characteristic
of mucinous tumors and can directly obtain pathological specimens
through ultrasound-guided biopsy, notably reducing the rate of
misdiagnosis (7,20). It is recommended as the first choice
of imaging examination for suspected perianal masses (7), especially for cases of painless masses
with clear boundaries. EAUS should be used to exclude the
possibility of mucinous adenocarcinoma.
Advanced evaluation. Pelvic enhanced MRI and
selective abdominal CT: Pelvic enhanced MRI has high resolution of
soft tissue, which can accurately reveal the anatomical
relationship between the tumor and anal canal, rectal wall and
sphincter, determine the depth of tumor invasion and provide an
important basis for establishing the scope of surgical resection
(6). Abdominal and chest CT can
assist in the detection of distant metastasis, such as liver and
lung metastasis (21).
If imaging (EAUS/MRI) suggests mucinous
adenocarcinoma features (such as mucinous lakes and invasive
growth), ultrasound-guided biopsy should be performed immediately
to avoid the possibility of accidentally ignoring malignancy due to
‘benign imaging findings’. Even if the initial biopsy is negative,
if PMA is still highly suspected (Such as perianal mass painless
short-term enlargement), biopsy or immunohistochemical staining
(such as CK7, CK20 and CDX2) should be performed again for further
verification.
Future prospects of imaging examinations: i)
Establish a standardized imaging examination process for perianal
tumors. Compare the differences in diagnostic efficacy among
B-ultrasound, EAUS, CT and MRI in the diagnosis of perianal tumors;
ii) explore the combined diagnostic model of MRI and pathological
examination. The combined diagnosis of ‘MRI imaging features and
puncture pathological results’ improves the accuracy of
preoperative examinations; and iii) conduct research on the
optimization of individualized surgical plans under the guidance of
MRI, further explore the application value of MRI in postoperative
follow-up, provide a basis for precise postoperative follow-up and
early intervention, and ultimately improve the long-term prognosis
of patients.
In terms of treatment, a radical surgical approach,
such as abdominoperineal resection and its variations, is often
recommended in cases of late diagnosis or locally advanced tumors
(11). Neoadjuvant
chemoradiotherapy is recommended for locally advanced and
metastatic diseases (7,22). NCCN guidelines recommend
sotorasib/adagrasib combined with cetuximab/panitumumab for
patients with CRC harboring KRAS p. G12c mutation (15). However, no drug targeting the KRAS
p. G12d mutation has yet been approved for treating CRC (8). In terms of prognosis, according to the
report by Gkegkes et al (7)
among the 150 patients with PMA, 41 (29.3%) had recurrence, 32
(21.3%) had complications and 44 (29.3%) were deceased, while the
rest were not clearly specified (7). In the present case, complete resection
(R0) was achieved by radical local extended resection, which agreed
with the treatment principles recommended by studies on early
localized tumors (7,10). The patient had a clinical stage II
tumor and did not need further adjuvant therapy after comprehensive
analysis. No recurrence or metastasis was found after 12 months of
follow-up, but regular follow-ups for local recurrence or distant
metastasis are needed. Therefore conducting thoracic and abdominal
contrast-enhanced CT scans plus MRI assessments every 3 months for
the first 2 years postoperatively is recommended, followed by
semiannual (every 6 months) examinations for the subsequent 3 years
and annual imaging evaluations thereafter for lifelong follow-up.
The key point in the present case report is that the vigilance of
presenting with a painless mass and no history of inflammation
should be increased in clinical practice. EAUS is the preferred
imaging modality for this assessment, and sole evaluation with
conventional B-ultrasound should be avoided due to its limited
spatial resolution and inability to accurately delineate the
tumor's local extent, depth of invasion, and perirectal soft tissue
involvement. Multiple sampling can avoid the missed diagnosis of a
single biopsy. The nature of the tumor can be determined by
examining frozen sections during the operation.
In conclusion, PMA is relatively rare in clinical
practice and occurs in various forms. The present case was unique
as the tumor had a rapid growth rate and an uncommon KRAS p. G12d
mutant subtype was identified through genetic testing. An accurate
diagnosis before the operation is difficult. For painless PMA, a
comprehensive diagnosis should be performed by combining clinical,
pathological, immunohistochemical, genetic testing and imaging
methods. Early and accurate diagnosis enables patients to receive
timely and appropriate treatment, which can help avoid missed
diagnoses and delayed treatment.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author. The data generated in the
present study may be found in the Genome Sequence Archive for Human
under accession number HRA014514 or at the following URL:
https://ngdc.cncb.ac.cn/gsa-human/browse/HRA014514.
Author's contributions
JS and JY drafted the manuscript and conceived the
study. JS and GL were responsible for the collection and analysis
of case data and literature. JY, XW, YZ and JS revised the
manuscript and interpreted the data. JS, XW and GL confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethical approval and consent to
participate
Ethical consent was provided by the Ethics Committee
of The First People's Hospital of Xiaoshan (approval no.
2024-12).
Patient consent for publication
Before the research was carried out, the patients
and their families were fully informed of the research purpose of
this case report, the scope of data use and the form of
publication. The patients and their families voluntarily signed the
written informed consent form, agreeing to use their clinical data,
pathological and molecular testing data for the writing and public
release of this case report. All data were anonymized to protect
the privacy of the patients.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hongo K, Kazama S, Sunami E and Kitayama J
and Kitayama J: Perianal adenocarcinoma associated with anal
fistula: A report of 11 cases in a single institution focusing on
treatment and literature review. Hepatogastroenterology.
60:720–726. 2013.PubMed/NCBI
|
|
2
|
Gaertner WB, Hagerman GF, Finne CO, Alavi
K Jessurun J, Rothenberger DA and Madoff RD: Fistula-associated
anal adenocarcinoma: Good results with aggressive therapy. Dis
Colon Rectum. 51:1061–1067. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
de Souza ABP, Lima AP, Lívia Genaro LM,
Geiger CPF, Ayrizono MLS and Leal RF: Mucinousadenocarcinoma in
perianal fistula in Crohn's disease: Case report and literature
review. Int J Surg Case Rep. 95:1072112022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Santos MD, Nogueira C and Lopes C:
Mucinous adenocarcinoma arising from chronicperianal fistula. Case
Rep Surg. 2014:3861502014.PubMed/NCBI
|
|
5
|
Koizumi M, Matsuda A, Yamada T, Morimoto
K, Kubota I, Kubota Y, Tamura S, Tominaga K, Sakatani T and Yoshida
H: A case report of anal fistula-associated mucinous adenocarcinoma
developing 3 years after treatment of perianal abscess. Surg Case
Rep. 9:1592023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chrysikos D, Mariolis-Sapsakos T,
Triantafyllou T, Karampelias V, Mitrousias A and Theodoropoulos G:
Laparoscopic abdominoperineal resection for the treatment of a
mucinous adenocarcinoma associated with an anal fistula. J Surg
Case Rep. 2018:rjy0362018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gkegkes ID, Milionis V, Goutas N,
Mantzoros I, Bourtzinakou AA and Stamatiadis AP: Perianal mucinous
adenocarcinoma: A case report and a systematic review of the
literature. J Gastrointest Cancer. 56:62024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
WHO Classifcation of Tumours Editorial
Board, . Tumours of the anal canal. Digestive system tumours. 5th
ed. Lyon, France: 2019, pp. 193–214
|
|
9
|
Li X, Sun K, Liao X, Gao H, Zhu H and Xu
R: Colorectal carcinomas with mucinous differentiation are
associated with high frequent mutation of KRAS or BRAF mutations,
irrespective of quantity of mucinous component. BMC Cancer.
20:4002020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cenac LA, Xiao P and Asarian A: Incidental
discovery of mucinous adenocarcinoma from a uspected infammatory
perianal mass. J Surg Case Rep. 2:rjz4132020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Feo CF, Veneroni S, Santoru A, Cossu ML,
Scanu AM, Ginesu GC and Porcu A: Perianal mucinous adenocarcinoma
with dysplastic polyps of the colon: A case report. Int J Surg Case
Rep. 78:99–102. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Purkayastha A, Sharma N, Dutta V, Bisht N
and Pandya T: Mucinous adenocarcinoma of perianal region: An
uncommon disease treated with neoadjuvant chemo-radiation. Transl
Gastroenterol Hepatol. 1:522016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Imai Y, Yamagishi H, Fukuda K, Ono Y,
Inoue T and Ueda Y: Differential mucin phenotypes and their
significance in a variation of colorectal carcinoma. World J
Gastroenterol. 19:3957–3968. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hugen N, Simons M, Halilović A, van der
Post RS, Bogers AJ, Marijnissen-van Zanten MA, de Wilt JH and
Nagtegaal ID: The molecular background of mucinous carcinoma beyond
MUC2. J Pathol Clin Res. 1:3–17. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
NCCN Clinical Practice Guidelines in
Oncology, . Colon Cancer. Version 1.2024.
|
|
16
|
Uprety D and Adjei AA: KRAS: From
undruggable to a druggable Cancer Target. Cancer Treat Rev.
89:1020702020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang R, Cong D, Bai Y and Zhang W: Case
report: Long-term sustained remission in a case of metastatic colon
cancer with high microsatellite instability and KRAS exon 2 p.G12D
mutation treated with fruquintinib after local radiotherapy: A case
report and literature review. Front Pharmacol. 14:12073692023.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Laohawetwanit T, Hareerak T, Thamwongskul
C and Boon-Ing N: Extramucosal anal canal adenocarcinoma, non-anal
gland type, and non-fistula-associated with mucinous appearance: A
recently described diagnostic entity. Pathol Int. 71:715–718. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Spiridakis KG, Sfakianakis EE, Flamourakis
ME, Intzepogazoglou DS, Tsagataki ES, Ximeris NE, Rachmanis EK,
Gionis IG, Kostakis GE and Christodoulakis MS: Synchronous mucinous
adenocarcinoma of the recto sigmoid revealed by and seeding an anal
fistula. (A case report and review of the literature). Int J Surg
Case Rep. 37:48–51. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Toyonaga T, Mibu R, Matsuda H, Tominaga Y,
Hirata K, Takeyoshi M, Tsuneyoshi M and Matsushima M: Endoanal
ultrasonography of mucinous adenocarcinoma arising from chronic
fistula-in-Ano: Three Case Reports. J Anus Rectum Colon. 1:100–105.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ghuman SS, Kochhar R, Mahajan H, Buxi TBS,
Gupta A, Arora A, Saxena KK, Sud S, Sud A, Rawat K, et al: CT and
MR imaging in colorectal carcinoma: A tool for diagnosis, staging,
response evaluation, and follow-up. South Asian J Cancer.
13:236–240. 2025.PubMed/NCBI
|
|
22
|
Ohta R, Sekikawa K, Goto M, Narita K,
Takahashi Y, Ikeda H, Oneyama M, Hirata Y, Nakayama M, Shimoda Y
and Sato S: A case of perianal mucinous adenocarcinoma arising from
an anorectal fistula successfully resected after preoperative
radiotherapy. Case Rep Gastroenterol. 7:219–223. 2013. View Article : Google Scholar : PubMed/NCBI
|