Introduction
Colorectal cancer (CRC) is the second leading cause
of cancer-related mortality and a major global health burden
(1). Epidemiological data show that
the incidence of CRC is steadily rising, especially among young
individuals (2). Although the
latest advances in screening modes and therapeutic interventions,
including immune checkpoint inhibitors and molecular targeted
drugs, have improved clinical outcomes, metastatic CRC continues to
pose significant treatment challenges. Treatment resistance
development and early metastasis remain the key obstacles, leading
to a 5-year survival of <15% for stage IV patients (3). This clinical reality highlights the
pressing need to explore innovative, safer, and more effective
treatment strategies for CRC.
Exosomes are nanovesicles with a lipid bilayer
membrane structure. They are released by almost all cell types.
Exosomes are rich in bioactive substances, such as nucleic acids,
lipids and proteins (4), which play
an important role in intercellular transmission, information
exchange and immune regulation (5).
Plant-derived exosome-like nanoparticles (ELNs) have attracted more
attention in the fields of anti-inflammatory, antitumor and
radioprotective effects as a result of their low immunogenicity,
favorable biocompatibility, stability and biosafety (6). In addition, Lycium ruthenicum
Murray-derived ELNs hinder Aβ-induced apoptosis of HT22 cells
and reduce oxidative stress levels by activating the Nrf2/HO-1/NQO1
signaling pathway (7). Polygonum
multiflorum-derived ELNs significantly inhibit proliferation
and migration of liver cancer cells (8) and exert anti-photoaging effects by
reducing ultraviolet-induced oxidative stress and promoting
collagen formation (9).
Atractylodes macrocephala-derived ELNs significantly improve
ulcerative colitis by remodeling gut microbiota balance, modulating
tryptophan metabolism, and suppressing the Th17 cell
differentiation pathway (10).
Gardenia-derived ELNs alleviate Parkinson's disease
progression by attenuating dopaminergic neuronal apoptosis and
inhibiting the p38 MAPK/p53 signaling pathway (11). Pomegranate vesicle extracts have
been shown to prevent intestinal barrier dysfunction and liver
injury/fibrosis in a mouse model of metabolic
dysfunction-associated steatotic liver disease (12).
Pomegranate [Punica granatum Linn (Punica
granatum L.)] is a highly nutritious fruit that has attracted
considerable attention due to its notable pharmacological
properties. The edible pomegranate flesh contains abundant
bioactive compounds, including anthocyanins and ellagitannins,
which have powerful antioxidant and vasculoprotective effects
(13). The non-edible pomegranate
peel accounts for ~40% of the total fruit weight and is typically
discarded as agricultural waste. However, it has been found to
contain beneficial active substances. It has been indicated that
the levels of antioxidant components and activities in pomegranate
peel are markedly higher than in the seeds and pulp (14). Pomegranate-derived ELNs have shown
significant anticancer effects against lung, breast and skin
cancers (15). However, the effect
of pomegranate peel-derived nanovesicles on CRC cells remains
unexplored.
The present study explored the intervention
mechanism of CRC with a focus on pomegranate peel-derived ELNs
(PELNs). Differential ultra-centrifugation was successfully
utilized for the first time to isolate and purify PELNs from the
pericarp of Punica granatum L. A series of functional
experiments was subsequently carried out in vitro, including
cell internalization, proliferation, apoptosis and migration.
Transcriptome sequencing experiment was then conducted to analyze
the anti-CRC mechanism of PELNs.
Materials and methods
PELN isolation
ELNs were isolated from the juice found in the
pomegranate peel. A total of ~50 g of pomegranate peel was washed
with ultra-pure water, ground with phosphate-buffered saline (PBS)
buffer, and filtered through a strainer to collect the leaching
filtrate solution. The filtrate was then centrifuged at 500 × g for
10 min, 2,000 × g for 20 min, and 12,000 × g for 60 min to remove
dead cells and cell debris (Fig.
1A). The supernatant was subsequently ultra-centrifuged at
100,000 × g for 70 min at 4°C (Thermo Fisher Scientific, Inc.). The
pellet was resuspended in PBS and then ultra-centrifuged again for
70 min. Finally, the PELNs were resuspended in PBS and stored at
−80°C until further analysis.
PELN characterization
PELN size and concentration were measured using a
Nanoparticle Tracking Analysis (NTA) instrument (ZetaView; Particle
Metrix GmbH). PELNs were then stained with 2% uranyl acetate for 1
min at room temperature, and images of their morphology structure
were captured using transmission electron microscopy (TEM; Hitachi
HT-7700; JEOL, Ltd.).
Cell culture
The CRC cell line SW480 utilized in the present
study was obtained from Procell Life Science & Technology Co.,
Ltd. SW480 cells were cultured in Dulbecco's Modified Eagle medium
(DMEM; Invitrogen; Thermo Fisher Scientific, Inc.) with 10% fetal
bovine serum (FBS; HyClone™; Cytiva) and 1% penicillin/streptomycin
(PS) solution in a 37°C cell culture incubator with 5%
CO2.
Cell internalization
Next, 50 µl of PELNs was mixed with 4 µl of PKH26
red fluorescent cell membrane dye (MilliporeSigma) in 2 ml of PBS.
The mixture was incubated at room temperature for 5 min and 2 ml of
FBS was added to stop the staining reaction. The mixtures were
subsequently ultra-centrifuged at 100,000 × g for 70 min at 4°C.
The pellet was re-suspended in PBS buffer and then
ultra-centrifuged again for 70 min. Finally, the PKH26-labeled
PELNs were re-suspended in PBS for future analysis.
SW480 cells were seeded in 24-well plates and
treated with PKH26-labeled PELNs for 5 h. The cells were then fixed
with 4% paraformaldehyde for 15 min at room temperature, stained
with DAPI blue-fluorescent dye for 15 min at room temperature, and
images were captured using confocal microscopy (TCS SP8; Leica
Microsystems GmbH).
Cell proliferation assay
The effect of PELNs on SW480 cell proliferation was
assessed using a Cell Counting Kit-8 (CCK-8; Beijing Solarbio
Science & Technology Co., Ltd.). SW480 cells were seeded in
96-well plates and treated with PBS (Control group) or PELNs (PELN
group) for 0, 24, 48 and 72 h. Next, 10 µl of CCK-8 reagent was
added to each well and further cultured for 1 h at 37°C. The
absorbance was measured at 450 nm using an enzymatic reader
(Bio-Rad Laboratories, Inc.).
Colony formation assay
SW480 cells were seeded in 6-well plates and treated
with PBS or PELNs. After 14 days, cells were fixed with 4%
paraformaldehyde for 15 min at room temperature and stained with
crystal violet staining solution (cat. no. C0121; Beyotime
Biotechnology) for 15 min at room temperature. The areas of cell
colony formation were quantified using ImageJ software (v1.46r;
National Institutes of Health).
Flow cytometric analysis of
apoptosis
SW480 cells were seeded in 6-well plates and treated
with PBS or PELNs. The cells were harvested using trypsin upon
reaching 80% confluency, resuspended in PBS, and stained with 5 µl
of Annexin V-FITC and 5 µl of propidium iodide (PI; Beijing
Solarbio Science & Technology Co., Ltd.) for 15 min in the
dark. Apoptotic cells were then quantified using a flow cytometer
(MoFLO XDP; Beckman Coulter, Inc.) and CytExpert software
(v2.3.1.22; Beckman Coulter, Inc.).
Wound healing assay
SW480 cells were seeded in 6-well plates until
reaching 90% confluency. Then, straight scratches were made in each
well using a 1-ml sterile pipette tip. After removing cell debris
with a PBS wash, cells were cultured for 0, 24, or 48 h in 2 ml of
DMEM with 1% FBS and 1% PS solution, containing PBS or PELNs
(1.0×109 particles/ml), at 37°C in a 5% CO2 incubator. Wound
healing was observed using a light microscope (bright-field). The
migration distance was quantified by measuring the wound width
using ImageJ software.
Transwell migration assay
SW480 cells were seeded in the upper chamber of a
24-well Transwell plate in 200 µl of DMEM containing PBS or PELNs.
The lower chamber was filled with 500 µl of DMEM supplemented with
10% FBS and 1% penicillin/streptomycin. Cells were fixed with 4%
paraformaldehyde for 15 min at room temperature after treatment for
24 h and stained with crystal violet staining solution (cat. no.
C0121; Beyotime Biotechnology) for 15 min. The areas of cell
migration were quantified using ImageJ software.
RNA extraction
The total RNA samples from SW480 cells treated with
PBS or PELNs were extracted using TRIzol reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). TRIzol reagent was added to the
sample, and the mixture was incubated for 5 min and mixed with
chloroform, phenol, and isopropanol for 3 min according to the
manufacturer instructions. The RNA samples were collected with DEPC
water by centrifugation at 12,000 × g for 15 min at 4°C. RNA
quality was assessed prior to library construction. Purity and
concentration were measured using a NanoDrop spectrophotometer
(Thermo Fisher Scientific, Inc.) and a Qubit 4.0 fluorometer
(Thermo Fisher Scientific, Inc.), with a 260/280 nm ratio >1.8
required for sample acceptance. The integrity and quantity of the
extracted RNA were measured using the Agilent 2100 system (Agilent
Technologies, Inc.).
Transcriptome sequencing and
analysis
The SW480 cells were seeded in 6-well plates and
treated with PELNs or PBS for 24 h for RNA-sequencing (RNA-Seq)
analysis. Total RNA was extracted from the CRC cell samples, and
mRNA was purified using the HieffNGS® mRNA isolation
master kit (cat. no. 12603; Shanghai Yeasen Biotechnology Co.,
Ltd.). First-strand cDNA was synthesized using random hexamers,
followed by second-strand synthesis with DNA polymerase I and RNase
H. Double-stranded cDNA was end-repaired, adenylated, and ligated
to sequencing adaptors. For purification and fragment selection,
Hieff NGS® DNA Selection Beads (cat. no. 12601ES56;
Yeasen Biotechnology Co., Ltd.) were used. After adapter ligation,
bead-based size selection was performed using 0.6× and 0.2× ratios
to enrich for fragments of the desired size. The final libraries
were purified using a 0.8 × bead ratio and subsequently amplified
by PCR. The purified libraries were quantified using a Qubit
fluorometer (Thermo Fisher Scientific, Inc.) and initially measured
in ng/µl. The molar concentration was then calculated based on the
average fragment size determined by an Agilent 2100 system (Agilent
Technologies, Inc.). Library circularization was performed using
the MGIEasy Dual Barcode Circularization Module (cat. no. A0508;
MGI Tech Co., Ltd.) prior to sequencing. Finally, the libraries
were sequenced on the DNBSEQ-T7 platform (MGI Tech Co., Ltd.) using
the DNBSEQ-T7RS High-throughput Sequencing Set (App-D FCL PE150,
V3.0; cat. no. Group 100-000016-00; MGI Tech Co., Ltd.) in
paired-end 150 bp (PE150) mode, following the manufacturer's
instructions.
StringTie (v2.1.5; http://ccb.jhu.edu/software/stringtie/index.shtml) was
used to compare the read count values for each gene as the original
expression level. Fragments per kilobase of transcript per million
mapped reads were then utilized to standardize the expression.
Differentially expressed genes (DEGs) were then analyzed using
DESeq2 (v1.30.1; http://bioconductor.org/packages//release/bioc/html/DESeq2.html),
with screening conditions set as follows:
|log2FoldChange| >1 and adjusted P-value (padj)
≤0.05. Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene
Ontology (GO) enrichment analyses of DEGs were performed using
clusterProfiler (v3.8.1; http://github.com/GuangchuangYu/clusterProfiler).
Significant enrichment results (P-adj ≤0.05) were selected to
identify connections between genes and pathways.
Statistical analysis
Statistical significance was analyzed using GraphPad
Prism 5.0 (Dotmatics) and Microsoft Excel (2010) software. All data
are presented as the mean ± standard deviation (mean ± SD). Paired
Student's t-test was performed to compare the means of two data
sets. P<0.05 was considered to indicate a statistically
significant difference.
Results
Isolation, characterization and
cellular internalization of PELNs
PELNs were isolated and purified from Punica
granatum L. using filtration and differential ultra-centrifugation
(Fig. 1A). TEM analysis revealed
that the isolated particles exhibited a cup-shaped nanostructure
with exosome-like characteristics (Fig.
1B). NTA analysis revealed that PELNs had an average diameter
of 152.0 nm, a concentration of 4.2×1010 particles/ml,
and a zeta potential of −26.14 mV (Fig.
1C and D).
An internalization experiment was carried out to
investigate the biological function of PELNs in the anti-CRC
mechanism. After treating SW480 cells with PKH26-labeled PELNs or
PBS, red fluorescence was only observed in the PELN-treated group
and located around the blue fluorescence, but not in the control
group (Fig. 1E). These results
indicated that PELNs were effectively received by the CRC
cells.
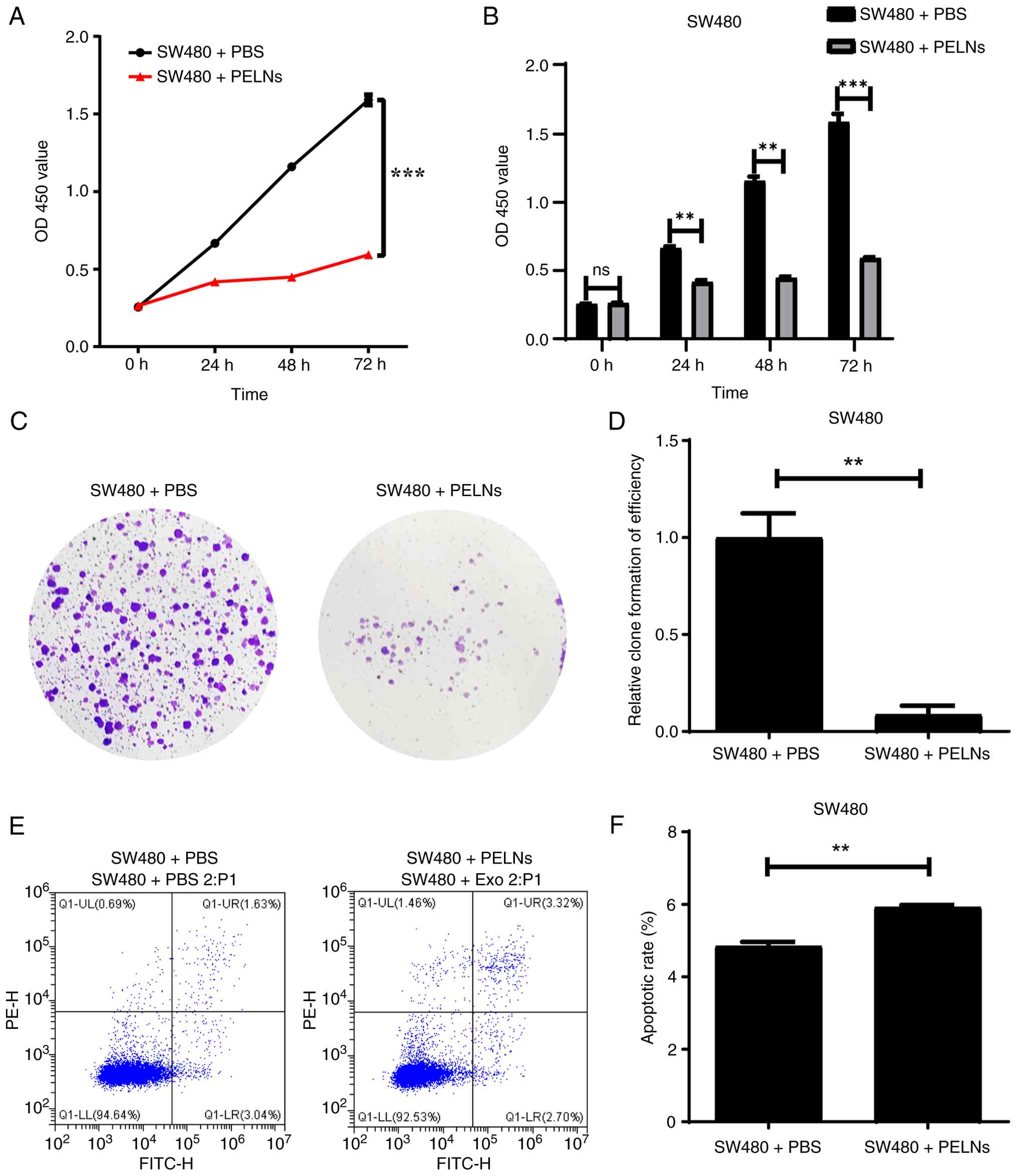
PELNs inhibit CRC cell proliferation
and induce apoptosis
CCK-8 and colony formation assays were performed to
further evaluate the effect of PELNs on CRC cell proliferation. The
proliferation ability of CRC cells treated with PELNs for 24, 48
and 72 h was significantly reduced compared with that of the
control group (P<0.001; Fig. 2A and
B). In the colony formation assay, the areas of SW480 cells
stained with crystal violet in the PELN group were significantly
decreased (P<0.01; Fig. 2C and
D).
Furthermore, SW480 cells were treated with PELNs for
24 h to investigate whether PELNs affect CRC cell apoptosis. Early
and late apoptotic cells were labeled with Annexin V-FITC/PI. Flow
cytometric analysis indicated that the PELN treatment significantly
increased the rate of CRC cell apoptosis (P<0.01; Fig. 2E and F).
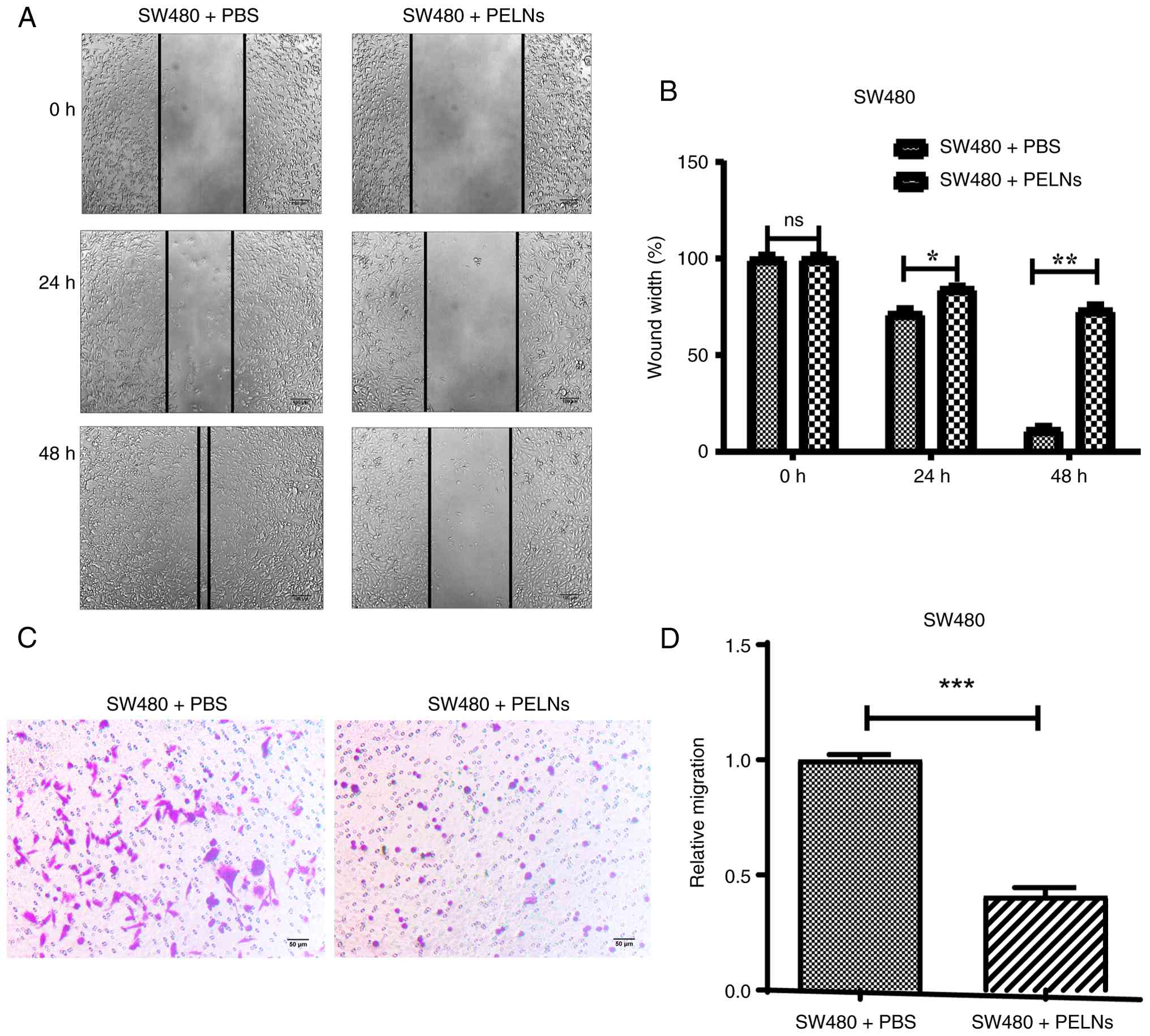
PELNs inhibit CRC cell migration
Wound healing and Transwell migration assays were
carried out to evaluate the effect of PELNs on the migration
ability of CRC cells. SW480 cells were treated with PELNs after
generating scratch wounds in the plate wells. The changes in wound
width were observed at 0, 24 and 48 h. The wound width in the
PELN-treated group was significantly increased compared with that
in the control group (P<0.001; Fig.
3A and B).
SW480 cells were incubated with PELNs in the upper
chamber of Transwell plates for 24 h. The migratory cells were then
quantified. Crystal violet-stained areas in the PELN group
demonstrated a significant reduction (P<0.05; Fig. 3C and D). These results indicated
that PELNs significantly inhibit CRC cell migration in
vitro.
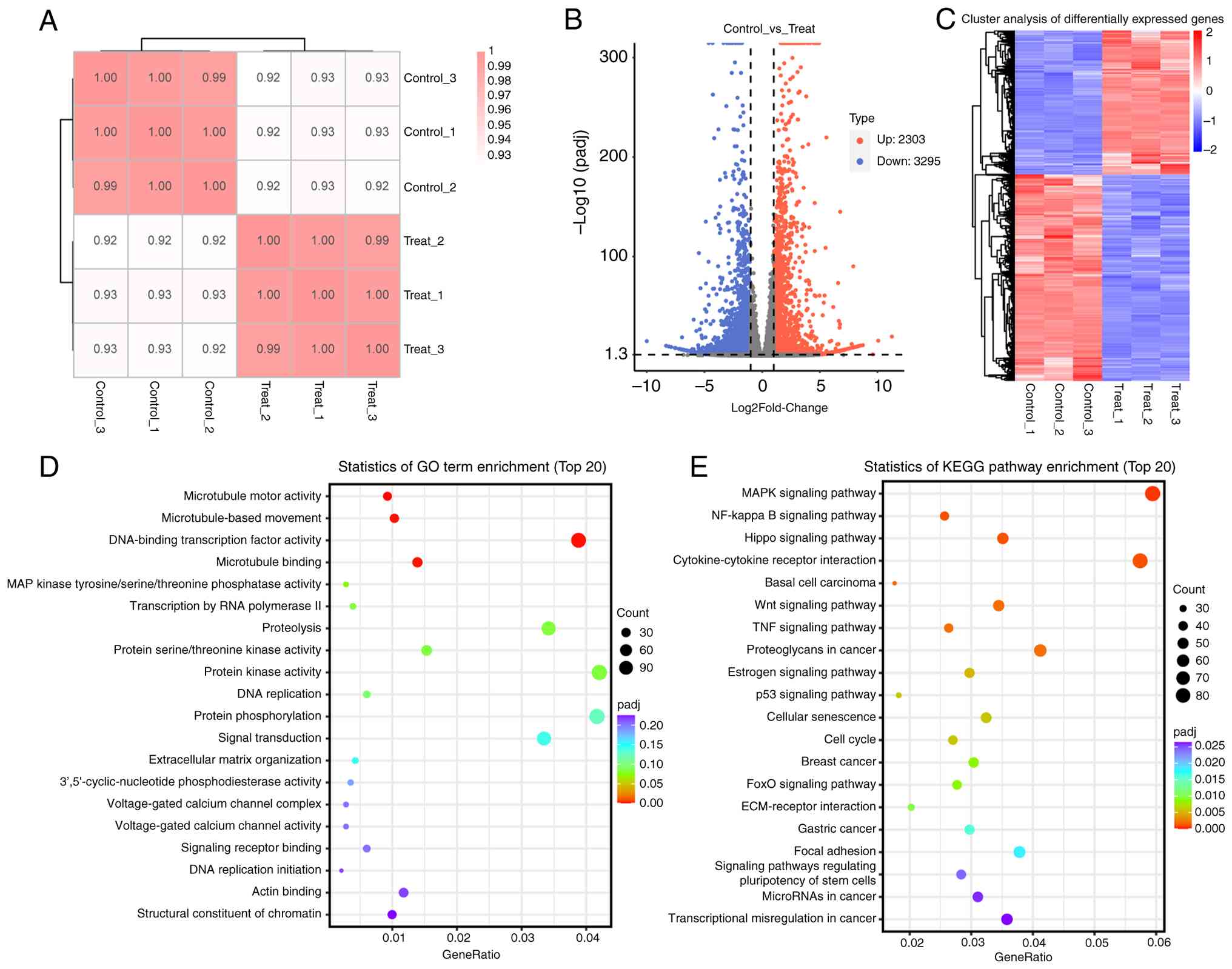
Transcriptomic analysis of
PELN-treated CRC cells
The sample correlation analysis heat map indicated
that the correlation between repeated samples was strong,
confirming experimental reproducibility (Fig. 4A). DEGs of SW480 cells after
treatment with PBS (Control-1, Control-2 and Control-3) or PELNs
(PELNs-1, PELNs-2 and PELNs-3) were statistically analyzed. The
results of a volcano plot and cluster analysis showed that there
were 2,303 upregulated and 3,295 downregulated genes (Fig. 4B and C). KEGG pathways and GO terms
related to DEGs were subsequently analyzed based on the functional
annotation results in the database.
Statistical analysis of KEGG pathway enrichment (top
20) indicated that multiple enriched pathways were associated with
signal transduction and cancer, including ‘MAPK signaling pathway’
(ID: map04010), ‘Hippo signaling pathway’ (ID: map04390), ‘NF-kappa
B signaling pathway’ (ID: 04064), ‘TNF signaling pathway’ (ID:
map04668), ‘Wnt signaling pathway’ (ID: map04310), ‘Estrogen
signaling pathway’ (ID: map04915), ‘Cellular senescence’ (ID:
map04218), ‘p53 signaling pathway’ (ID: map04115), ‘Cell cycle’
(ID: map04110), ‘FoxO signaling pathway’ (ID: map04068),
‘ECM-receptor interaction’ (ID: map04512), ‘MicroRNAs in cancer’
(ID: map05206) and ‘Transcriptional misregulation in cancer’ (ID:
map05202) (Fig. 4D, Table SI).
GO enrichment analysis of the top 20 molecular
function terms demonstrated significant enrichment for processes
directly related to cell cycle regulation and signal transduction,
including ‘MAP kinase tyrosine/serine/threonine phosphatase
activity’ (GO: 0017017), ‘protein kinase activity’ (GO: 0004672),
‘protein serine/threonine kinase activity’ (GO: 0004674), ‘DNA
replication’ (GO: 0006260), ‘protein phosphorylation’ (GO:
0006468), ‘signal transduction’ (GO: 0007165), ‘signaling receptor
binding’ (GO: 0005102), and ‘DNA replication initiation’ (GO:
0006270) (Fig. 4E, Table SII). Transcriptome sequencing
analysis results indicated that the DEGs associated with PELNs
specifically affected the proliferation and migration abilities of
CRC cells.
Discussion
Exosomes were considered cellular metabolism waste
in 1983 (16,17). Thorough research studies gradually
discovered that exosomes have specific cup-shaped nanovesicle
structures and that they participate in cellular information
exchange, tumor occurrence and development, and wound healing
physiological and pathological processes. PELN advantages include
source abundance, low immunogenicity and high biocompatibility.
PELNs have demonstrated significant potential in medical
applications due to their anti-inflammatory properties (balloon
flower root ELNs) (18), antitumor
effects (garlic ELNs) (19) and
osteoporosis' prevention capacity (yam ELNs) (20). Pomegranate peel is discarded as
waste in daily life, but it is commonly used as a traditional
Chinese medicine for antimicrobial (21), antioxidant (22) and wound healing needs (23). However, the inhibitory effect of
pomegranate PELNs on cancer has not been explored. The present
study for the first time, to the best of our knowledge,
successfully isolated high concentrations of ELNs from the peel of
Punica granatum L. Functional experiments identified that
PELNs inhibit the proliferation and migration of CRC cells.
KEGG and GO analyses of RNA-Seq revealed that PELNs
inhibited cancer-associated pathways, including the MAPK, Hippo,
NF-kappa B and Wnt signaling pathways. Pellino 2 is an E3 ubiquitin
ligase that regulates the CRC cell cycle, apoptotic signals, and
downstream epithelial-mesenchymal transition program via the MAPK
signaling pathway (24). In
addition, Sakuma et al (25)
demonstrated that overexpression of heterogeneous nuclear
ribonucleoprotein L-like upregulated the expression levels of PCNA,
RFC3 and FEN1, promoted cell cycle progression, and thereby
enhanced CRC cell proliferation. PELNs may play an anti-CRC role by
downregulating the expression of DNA replication initiation
promoter genes PCNA and FEN1 (Table
SIII) and disrupting normal DNA repair and cell cycle in SW480
cells. The canonical Wnt signaling pathway depends on the β-catenin
protein to regulate gene expression, cell migration, proliferation
and differentiation during the pathological development of CRC
(26). Zheng et al (27) reported that as a therapeutic target
for CRC, LDL receptor related protein 2 (LRP2) enhances
GSK3β/β-catenin signaling to promote CRC metastasis. In the present
study, DEG LRP2 was significantly downregulated (Table SIII) after treatment with PELNs,
which may be the main reason for PELN SW480 cell migration
inhibition. In summary, the molecular mechanism by which PELNs
restrained CRC progression may primarily have acted through the
MAPK/Wnt signaling pathway and cell cycle regulation.
The primary limitation of the present study is that
the specific composition of PELNs remains unclarified. Accumulating
evidence indicates that ELNs carry abundant microRNAs (miRNAs or
miRs) (28), proteins (29), lipids (30) and metabolites (31). It has been reported that
pomegranate-ELNs are enriched with proteins including chitinase,
lipid transfer protein and chalcone synthase (12). Pomegranate-ELNs are effective in
improving pancreatitis and preventing metabolic
dysfunction-associated fatty liver disease (32). Centella Asiatica-ELNs are
enriched with miRNAs (for example, aof-miR159, fve-miR396c-3p and
aof-miR396b), lipid components (for example, ceramide, triglyceride
and, hexosyl-ceramide), and numerous proteins involved in key
biological processes (33). The
miR-7972 abundant in Rehmanniae Radix-ELNs is an active
component responsible for the anti-inflammatory activity of the
fresh plant (34). Momordica
charantia-ELNs, which are rich in metabolism-related proteins
and anti-inflammatory and antioxidant lipid components, have
demonstrated efficacy in treating diabetes and alleviating
ulcerative colitis, with studies revealing that their therapeutic
effects are mediated through the repair of insulin-secretory
function and comprehensive renovation of the intestinal
microenvironment (35,36). These precedents underscore that
elucidating molecular cargo is the critical step linking observed
phenotype to mechanistic understanding, thereby exemplifying the
established ‘phenotype, composition, mechanism’ research paradigm
applicable to ELNs studies. In vitro functional validation
serves not only as the foundational core of such research but also
provides critical direction and justification for subsequent
in-depth compositional analysis. Consequently, the conclusive in
vitro data presented in the present study confirm the potent
anti-CRC bioactivity of PELNs. This provides a solid basis for
further investigation and clearly delineates the future research
trajectory of the current study. First, the particle-free
supernatant will be used as a negative control to definitively
attribute the observed anticancer effects to PELNs. Subsequently,
integrated multi-omics analyses (for example, miRNA sequencing,
proteomics and lipidomics) will be performed. These analyses will
systematically identify the constituents of PELNs and pinpoint key
bioactive molecules, such as specific miRNAs or functional
proteins, to elucidate their anti-CRC mechanism of action. Finally,
comprehensive in vivo studies on the biodistribution, safety
and antitumor efficacy of PELNs will be conducted. Collectively,
these studies are crucial for laying the practical groundwork for
clinical translation and for advancing the role of plant-derived
nanotherapeutics in CRC treatment.
Pomegranate PELNs are a natural biological
nanomaterial that exhibit significant inhibitory effects on the
proliferation and migration of CRC cells. This finding greatly
enhanced the potential value of future research into the biological
functions and clinical applications of plant-derived ELNs, fruit
peel, and other ‘neglected waste’ materials. In addition,
engineering modifications of PELNs, such as surface modification of
targeted peptides or internal delivery of small molecule drugs, are
expected to further enhance their anti-cancer effects. In the
future, PELNs have great potential for precise targeted therapy of
malignant tumors. Nevertheless, advancing PELNs to the clinic will
require further validation of their composition, safety and in
vivo efficacy.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Dr Zhan Leilei
(Wuhan GeneCreate Biological Engineering Co., Ltd.) for critically
discussing the manuscript.
Funding
The present study was supported by the Wuhan Traditional Chinese
Medicine Scientific Research Project (grant no. WZ22C24;
2022-2024).
Availability of data and materials
The data generated in the present study may be found
in the BioProject database under accession number PRJNA1307758 or
at the following URL: https://www.ncbi.nlm.nih.gov/bioproject/?term=PRJNA1307758.
Authors' contributions
YD and QY conceived and designed the study,
performed the experiments, and analyzed the data. ZZ and CC
participated in the design of experiments, data analysis and data
interpretation. All authors read and approved the final version of
the manuscript. YD and QY confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Siegel RL, Wagle NS, Cercek A, Smith RA
and Jemal A: Colorectal cancer statistics, 2023. CA Cancer J Clin.
73:233–254. 2023.PubMed/NCBI
|
|
3
|
Biller LH and Schrag D: Diagnosis and
treatment of metastatic colorectal cancer: A review. JAMA.
325:669–685. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Isaac R, Reis FCG, Ying W and Olefsky JM:
Exosomes as mediators of intercellular crosstalk in metabolism.
Cell Metab. 33:1744–1762. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen Q, Li Q, Liang Y, Zu M, Chen N, Canup
BSB, Luo L, Wang C, Zeng L and Xiao B: Natural exosome-like
nanovesicles from edible tea flowers suppress metastatic breast
cancer via ROS generation and microbiota modulation. Acta Pharm Sin
B. 12:907–923. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ling Y, Li X, Gao H, Liu Y, Liu Y, Zheng
J, Zhu J, Zhao C, Shi Y, Lu J and Yi J: Biyang floral
mushroom-derived exosome-like nanovesicles: Characterization,
absorption stability and ionizing radiation protection. Food Funct.
15:6900–6913. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang Y, Lu L, Li Y, Liu H, Zhou W and
Zhang L: Response surface methodology optimization of Exosome-like
nanovesicles extraction from Lyceum ruthenicum murray and their
inhibitory effects on Aβ-Induced apoptosis and oxidative stress in
HT22 cells. Foods. 13:33282024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang M, Xu L and Wang W: Molecular
anti-tumorigenic mechanism of Radix Polygoni Multiflori-derived
exosome-like nanoparticles. Heliyon. 11:e419182025. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
He J, Fu L, Shen Y, Teng Y, Huang Y, Ding
X, Xu D, Cui H, Zhu M, Xie J, et al: Polygonum multiflorum
extracellular Vesicle-Like nanovesicle for skin photoaging therapy.
Biomater Res. 28:00982024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tan X, Gao B, Xu Y, Zhao Q, Jiang J, Sun
D, Zhang Y, Zhou S, Fan JB, Zhang M and Zhao K: Atractylodes
macrocephala-derived extracellular vesicles-like particles enhance
the recovery of ulcerative colitis by remodeling intestinal
microecological balance. J Nanobiotechnology. 23:4332025.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen W, Wang H, Ye X, Hao X, Yan F, Wu J,
Li D, Wang Y and Xu L: Gardenia-derived extracellular vesicles
exert therapeutic effects on dopaminergic neuron apoptosis-mediated
Parkinson's disease. NPJ Parkinsons Dis. 11:2002025. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim JS, Song BJ and Cho YE:
Pomegranate-derived Exosome-like nanovesicles containing ellagic
acid alleviate gut leakage and liver injury in MASLD. Food Sci
Nutr. 13:e700882025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang D, Özen C, Abu-Reidah IM, Chigurupati
S, Patra JK, Horbanczuk JO, Jóźwik A, Tzvetkov NT, Uhrin P and
Atanasov AG: Vasculoprotective effects of pomegranate (Punica
granatum L.). Front Pharmacol. 9:5442018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mastrodi Salgado J, Ferreira TRB, De
Oliveira Biazotto F and Dos Santos Dias CT: Increased antioxidant
content in juice enriched with dried extract of pomegranate (Punica
granatum) Peel. Plant Foods Hum Nutr. 67:39–43. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sharma P, McClees SF and Afaq F:
Pomegranate for prevention and treatment of cancer: An update.
Molecules. 22:1772017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Harding C, Heuser J and Stahl P:
Receptor-mediated endocytosis of transferrin and recycling of the
transferrin receptor in rat reticulocytes. J Cell Biol. 97:329–339.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pan BT and Johnstone RM: Fate of the
transferrin receptor during maturation of sheep reticulocytes in
vitro: Selective externalization of the receptor. Cell. 33:967–978.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kim M, Jang H and Park JH: Balloon flower
Root-derived extracellular vesicles: In vitro assessment of
Anti-Inflammatory, proliferative, and antioxidant effects for
chronic wound healing. Antioxidants (Basel). 12:11462023.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu J, Yu Y, Zhang Y, Dai H, Yang Q, Wang
B, Ma Q, Chen Y, Xu F, Shi X, et al: Oral administration of
garlic-derived nanoparticles improves cancer immunotherapy by
inducing intestinal IFNγ-producing γδ T cells. Nat Nanotechnol.
19:1569–1578. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hwang JH, Park YS, Kim HS, Kim DH, Lee SH,
Lee CH, Lee SH, Kim JE, Lee S, Kim HM, et al: Yam-derived
exosome-like nanovesicles stimulate osteoblast formation and
prevent osteoporosis in mice. J Control Release. 355:184–198. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Abu-Niaaj LF, Al-Daghistani HI, Katampe I,
Abu-Irmaileh B and Bustanji YK: Pomegranate Peel: Bioactivities as
antimicrobial and cytotoxic agents. Food Sci Nutr. 12:2818–2832.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhai X, Zhu C, Zhang Y, Sun J, Alim A and
Yang X: Chemical characteristics, antioxidant capacities and
hepatoprotection of polysaccharides from pomegranate peel.
Carbohydr Polym. 202:461–469. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yan H, Peng KJ, Wang QL, Gu ZY, Lu YQ,
Zhao J, Xu F, Liu YL, Tang Y, Deng FM, et al: Effect of pomegranate
peel polyphenol gel on cutaneous wound healing in alloxan-induced
diabetic rats. Chin Med J (Engl). 126:1700–1706. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu J, Hu S, Zhao L, Yang Y, Wu G, Duan Y,
Ma X, Wang P, Zhang Z and Zong H: PELI2 inhibits colorectal cancer
development through MAPK signaling pathway. Mol Med. 31:2352025.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sakuma K, Sasaki E, Kimura K, Komori K,
Shimizu Y, Yatabe Y and Aoki M: HNRNPLL stabilizes mRNA for DNA
replication proteins and promotes cell cycle progression in
colorectal cancer cells. Cancer Sci. 109:2458–2468. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cheng X, Xu X, Chen D, Zhao F and Wang W:
Therapeutic potential of targeting the Wnt/β-catenin signaling
pathway in colorectal cancer. Biomed Pharmacother. 110:473–481.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zheng X, Liu H, Zhong X, Liu X, Cai Z,
Chen Y, He X, Lan P and Wu X: LDL receptor related protein 2 to
promote colorectal cancer metastasis via enhancing GSK3β/β-catenin
signaling. J Clin Oncol. 39:e155072021. View Article : Google Scholar
|
|
28
|
Leng Y, Yang L, Pan S, Zhan L and Yuan F:
Characterization of blueberry exosome-like nanoparticles and miRNAs
with potential cross-kingdom human gene targets. Food Sci Human
Wellness. 13:869–878. 2024. View Article : Google Scholar
|
|
29
|
Sun J, Du L, Qu Z, Wang H, Dong S, Li X
and Zhao H: Integrated metabolomics and proteomics analysis to
study the changes in Scutellaria baicalensis at different growth
stages. Food Chemistry. 419:1360432023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sui Y, Sun X, Liu Q, Qi G, Yang Y, Zhang
X, Francis OB, Wang Y, Liu R, Li X, et al: Mori fructus-derived
extracellular vesicle-like nanoparticles regulate dyslipidemia and
prevent atherosclerosis progression via miRNAs. Phytomedicine.
146:1571282025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fu J, Liu Z, Feng Z, Huang J, Shi J, Wang
K, Jiang X, Yang J, Ning Y, Lu F, et al: Platycodon grandiflorum
exosome-like nanoparticles: The material basis of fresh Platycodon
grandiflorum optimality and its mechanism in regulating acute lung
injury. J Nanobiotechnol. 23:2702025. View Article : Google Scholar
|
|
32
|
Kang H, Hu Q, Yang Y, Huang G, Li J, Zhao
X, Zhu L, Su H, Tang W and Wan M: Urolithin A's role in alleviating
severe acute pancreatitis via endoplasmic Reticulum-mitochondrial
calcium channel modulation. ACS Nano. 18:13885–13898. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shi R, Tan W, Jin H, Chan SI, Li W, Lei
SS, Cui G, Wang Y, Yang DH and Zhong Z: MicroRNA-enriched
Plant-derived exosomes alleviate colitis by modulating systemic
immunity, metabolic homeostasis, and Gut microbiota. Adv Sci
(Weinh). 12:e059212025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Qiu FS, Wang JF, Guo MY, Li XJ, Shi CY, Wu
F, Zhang HH, Ying HZ and Yu CH: Rgl-exomiR-7972, a novel plant
Exosomal MicroRNA derived from fresh Rehmanniae radix, ameliorated
lipopolysaccharide-induced acute lung injury and Gut dysbiosis.
Biomed Pharmacother. 165:1150072023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Han M, Wang J, Zhang Z, Yan Z, Wang Z,
Guan X, Wang S, Mao Y and Zhang J: Momordica Charantia L.-derived
extracellular vesicles achieve pancreatic-targeted delivery and
repair insulin-secretory function through dual mechanisms via
lymphatic transport. Chemical Engineering J. 520:1657472025.
View Article : Google Scholar
|
|
36
|
Gao B, Huang X, Fu J, Chen L, Deng Z, Wang
S, Zhu Y, Xu C, Zhang Y, Zhang M, et al: Oral administration of
Momordica charantia-derived extracellular vesicles alleviates
ulcerative colitis through comprehensive renovation of the
intestinal microenvironment. J Nanobiotechnology. 23:2612025.
View Article : Google Scholar : PubMed/NCBI
|















![Isolation, characterization and
cellular internalization of PELNs. (A) Schematic diagram of the
preparation process for isolating PELNs from the peel of Punica
granatum. (B) Representative transmission electron microscopy image
of PELNs. Scale bar, 200 nm (left); 100 nm (right). (C) PELN
concentration and size distribution. (D) Zeta potential
distribution of PELNs. (E) PKH26-labeled PELNs (red) were received
by SW480 cells [nuclei stained by DAPI (blue)]. Scale bar, 25 µm.
PELNs, pomegranate peel-derived exosome-like nanoparticles; UC,
ultracentrifugation; PBS, phosphate-buffered saline.](/article_images/ol/31/5/ol-31-05-15546-g00.jpg)