Pancreatic cancer (PC) is a fatal disease, with
~51,980 deaths in the US each year and a 5-year survival rate of
only 13%, making it a malignancy with one of the worst prognosis
among all cancer types (1).
Currently, the primary clinical treatments for PC include surgical
resection and chemotherapy. However, due to the aggressive
progression of the disease and the fact that ~80% of patients are
diagnosed at an advanced stage, surgical resection is often not
feasible (2). Even the modified
version of folinic acid, irinotecan and oxaliplatin (mFOLFIRINOX)
chemotherapy regimen, which is considered one of the most effective
treatments for PC, achieves a 3-year overall survival rate of
merely 63.4% (3). mFOLFIRINOX is an
effective combined chemotherapy regimen. The name is derived from
the abbreviations of its four constituent drugs: FOL, FIRI and NOX.
The prefix ‘m’ indicates ‘improved’. This regimen was developed by
the classic FOLFIRINOX protocol. Currently, it is mainly used as a
first-line treatment option for patients with good physical
condition who have locally advanced or metastatic PC. FOLFIRINOX is
currently one of the key chemotherapy regimens in the field of PC.
However, chemotherapy for PC is often limited by two major
challenges: The development of chemoresistance, which prevents
patients from completing the full course of treatment and the
cumulative toxicity of prolonged regimens. Therefore, both
treatment efficacy and quality of life of patients are compromised
(4). The exact etiology of PC
remains to be elucidated; however, the tumor microenvironment
(TME), environmental factors and genetic predisposition are
recognized as major contributors to its initiation and progression
(5). For instance, smoking is the
primary non-genetic factor associated with an increased risk of
pancreatic ductal adenocarcinoma (PDAC), with an odds ratio of 3.7
(95% confidence interval: 1.8–7.6) (6). Additionally, smoking is significantly
associated with a notable decrease in the average age of diagnosis
among patients with familial PDAC. These findings underscore the
critical role of smoking-related environmental factors in the
pathogenesis and progression of PDAC (7,8). The
TME of PC consists of cancer, stromal and immune cells and
extracellular matrix components, all of which serve key roles in
promoting tumor growth and metastasis (9). Key features include driver gene
mutations in cancer cells, excessive collagen and extracellular
matrix deposition by stromal cells leading to tissue fibrosis and
markedly expressed inflammatory cytokines that create a
pro-inflammatory and immunosuppressive milieu. This unique TME not
only presents a notable barrier to treatment but also offers
potential targets for novel therapeutic strategies. Therefore,
developing early diagnostic methods and effective treatments for PC
remains an urgent and key challenge.
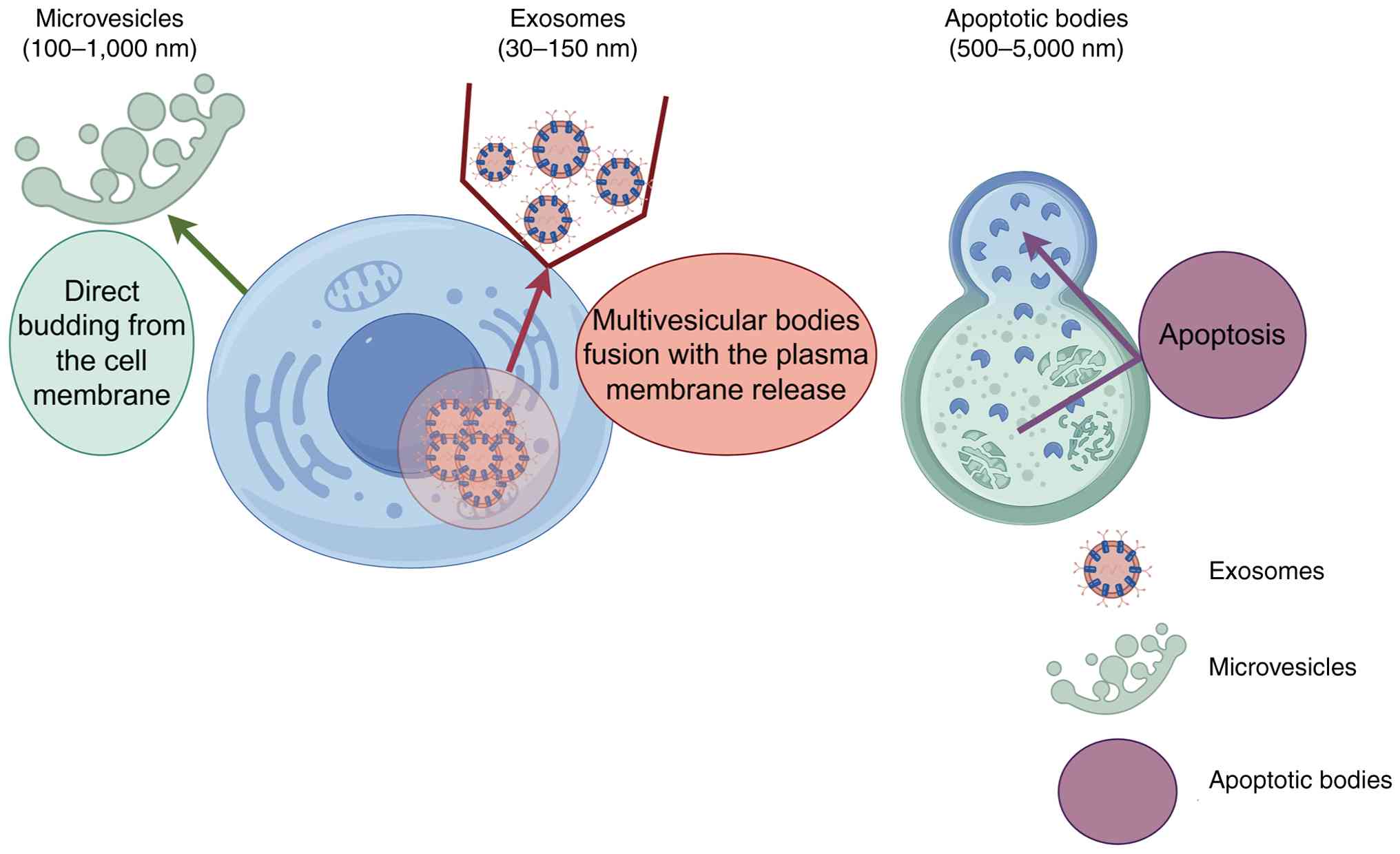
Exosomes are a type of extracellular vesicle (EV)
with a diameter of 30–150 nm. Exosomes carry a diverse cargo of
lipids, proteins and nucleic acids (10), enabling them to serve as exceptional
messengers for complex information transfer between cells (11), across distant tissues (12) and between tumor and stromal
compartments (13). Exosomes are
present in nearly all bodily fluids, including blood, sweat, tears,
urine, saliva, breast milk, ascites and cerebrospinal fluid.
Exosomes derived from different sources can exert distinct effects
on tumors (14). Tumor-derived
exosomes can transfer oncogenic molecules (15), promote anti-apoptotic effects
(16), stimulate angiogenesis
(17) and alter tumor cell
metabolism (18), thereby
facilitating tumor growth. However, a previous study has also
demonstrated that exosomes hold promise as biomarkers for the early
diagnosis of PC (19). Furthermore,
exosomes can serve as notable drug delivery vehicles, offering
potential therapeutic strategies for an extensive subset of
patients with PC with chemoresistance (20). Furthermore, elucidating the
interrelationship between the TME of PC and exosomes may help
researchers identify novel therapeutic targets, thereby opening
novel avenues for treatment. Thus, exosomes serve a dual role in
PC. The present review aims to summarize the mechanisms underlying
the dual functions of exosomes in the initiation and progression of
PC, provide novel insights and directions in optimally utilizing
exosomes in clinical therapy and accelerate the clinical
translation of exosome-based targeted therapies for PC.
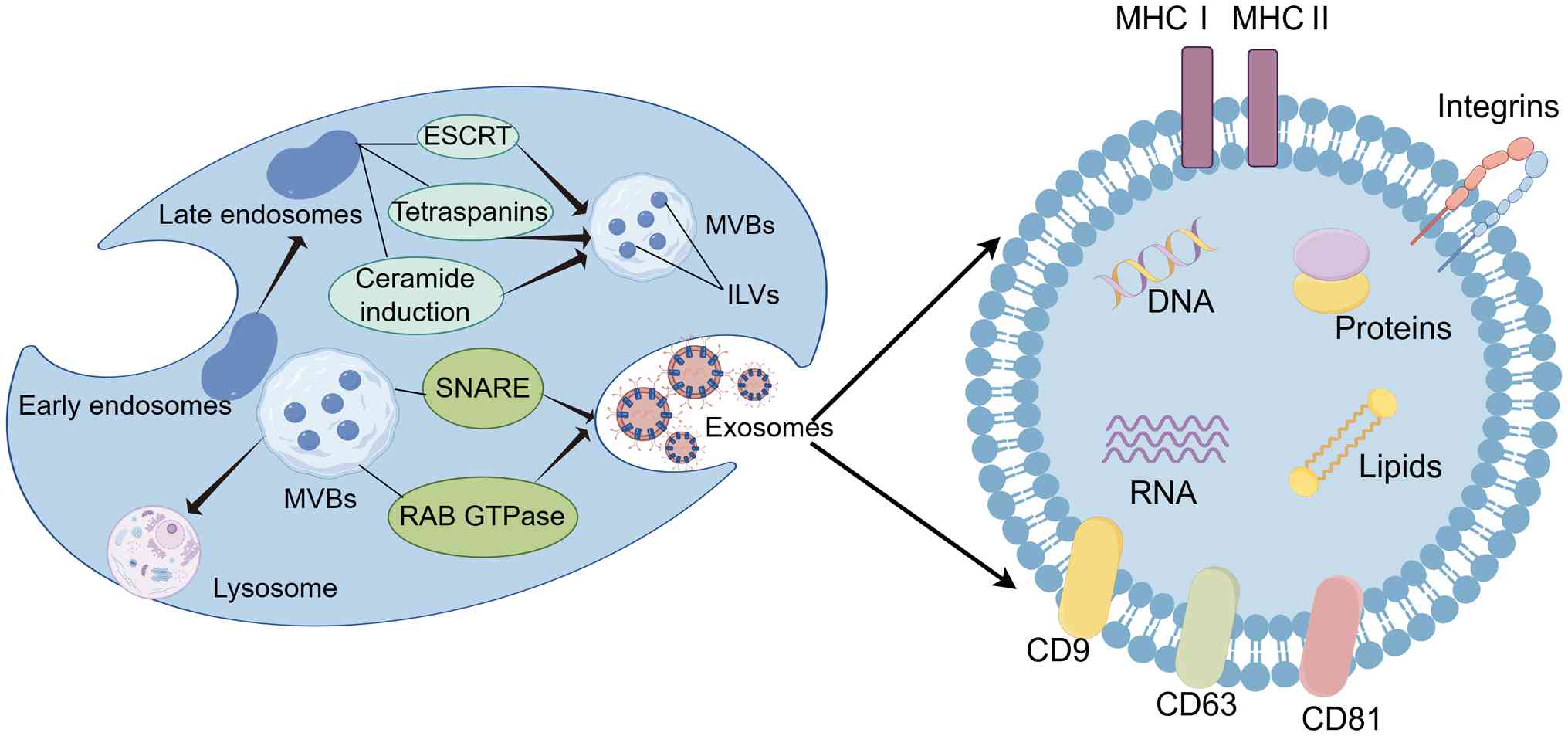
The biogenesis and synthesis of exosomes is a
complex process, which begins with endocytosis at the cell membrane
surface, where early endosomes are formed through inward budding.
Over time, early endosomes mature into late endosomes. Late
endosomes primarily form intraluminal vesicles (ILVs) through three
pathways: Endosomal sorting complex required for transport,
tetraspanin proteins and ceramide induction, and these ILVs
gradually develop into multivesicular bodies (MVBs) (32–34).
Subsequently, most MVBs fuse with lysosomes, leading to the
degradation of ILVs, while a small number of MVBs fuse with the
plasma membrane and release exosomes into the extracellular
environment (32,35,36).
Exosomes carry a rich cargo of lipids, proteins and nucleic acids.
The lipid components in exosomes include cholesterol,
sphingomyelin, glycosphingolipids, phosphatidylserine,
phosphatidylinositol, phosphatidic acid and ceramide (37). Common proteins in exosomes include
those associated with membrane transport, such as Ras-related in
brain GTPases, annexins, flotillins and MVB biogenesis proteins
including apoptosis-linked gene 2-interacting protein X (38). Exosomes also contain tetraspanin
proteins, including CD9, CD63 and CD81, as well as heat shock
proteins (HSP) such as HSP60 and HSP90. The nucleic acid components
in exosomes are also highly diverse, such as microRNA (miRNA/miR)
and messenger RNA, which were among the first two types identified
(39,40), followed by long non-coding RNA
(lncRNA), transfer RNA, small nuclear RNA and circular RNA
(circRNA/circ) (39,41) (Fig.
2). These RNA not only act as regulators of gene expression but
also serve as potential biomarkers (42). For instance, miR-141 can be detected
in the blood of patients with prostate cancer, and its expression
level is correlated with the size and malignancy of the tumor
(43). Saliva miR-3679-5p and
miR-940 have good discriminatory ability and can be used for the
detection of resectable PC, with reasonable specificity and
sensitivity (44). Furthermore,
lncRNA can be regarded as an independent predictor of pathological
cardiac remodeling and diastolic dysfunction in patients with type
2 diabetes (45).
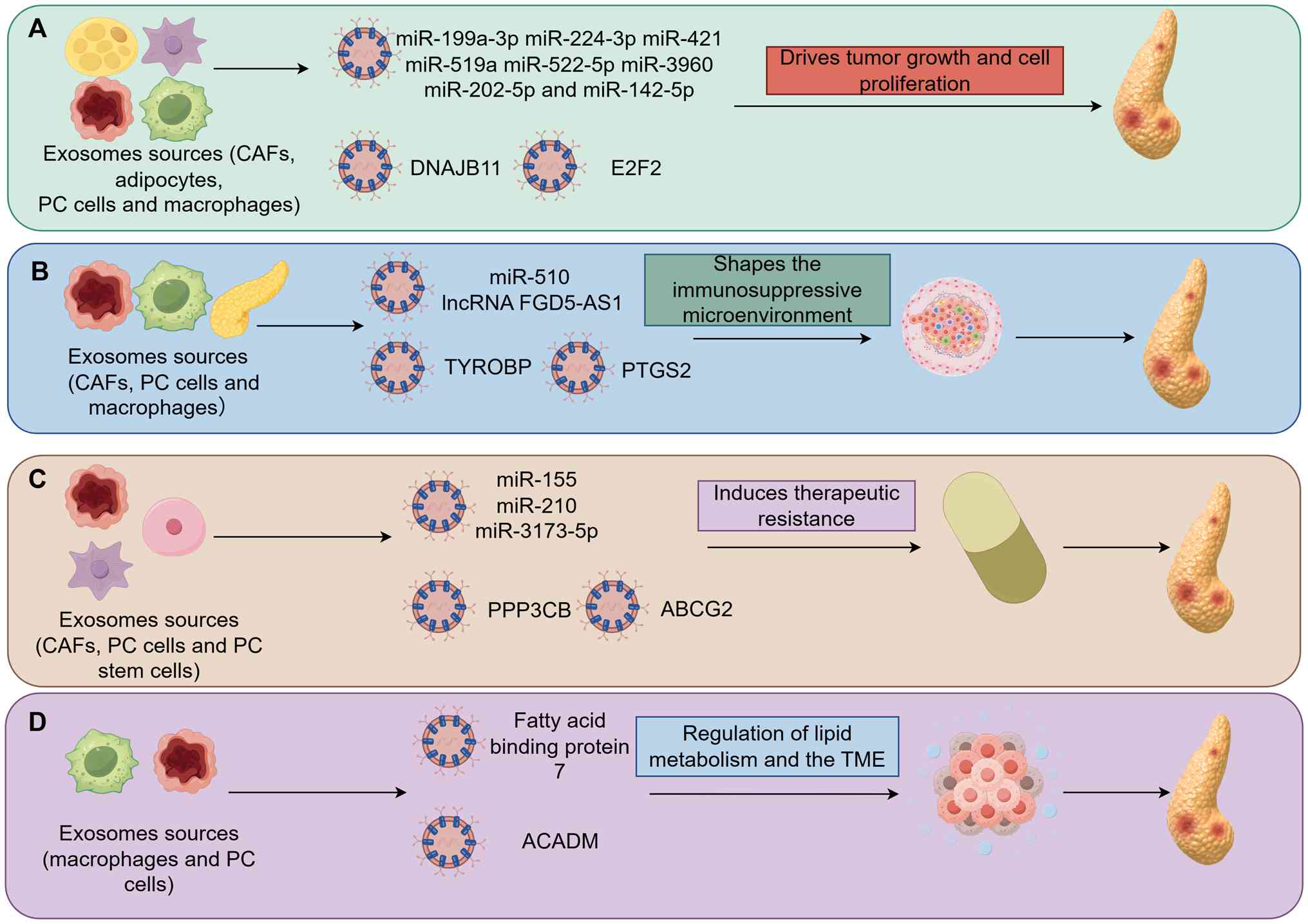
Exosomes serve a non-negligible ‘dark’ role in the
initiation and progression of PC, driving tumor development and
inducing therapy resistance through multiple mechanisms.
Specifically, exosomes serve as key mediators of intercellular
communication and can be derived from various cell types, including
PC cells, adipocytes, cancer-associated fibroblasts (CAFs) and
immune cells (46,47). By carrying specific molecules such
as miRNAs, proteins and circRNAs, exosomes activate multiple
signaling pathways [such as signal transducer and activator of
transcription 3 (STAT3), hypoxia inducible factor 1α (HIF-1α),
epidermal growth factor receptor/mitogen-activated protein kinase
(EGFR/MAPK)], directly promoting tumor cell proliferation, invasion
and metastasis. Simultaneously, exosomes act as key modulators of
the immunosuppressive TME. Exosomes can induce M2 macrophage
polarization and regulate pathways such as protein kinase
B/extracellular signal-regulated kinase (AKT/ERK), establishing a
local environment conducive to immune evasion and tumor metastasis.
Furthermore, exosomes notably contribute to chemoresistance. They
can function as drug efflux pumps or deliver miRNAs (such as
miR-155 and miR-210) to inhibit apoptosis and ferroptosis, thereby
mediating resistance to gemcitabine. Furthermore, exosomes can also
promote the progression of PC by regulating lipid metabolism and
the TME (Table I) (48–66).
These findings not only highlight exosomes as key drivers of
malignant progression in PC but also provide a theoretical basis in
targeting them to reverse drug resistance and improve therapeutic
efficacy (Fig. 3).
Exosomes can drive the proliferation of tumor cells
through multiple signaling pathways. The mechanism of their action
mainly involves exosomes derived from tumor cells, CAFs and immune
cells. A previous study using in vitro cell models reported
that exosomes derived from cancer-associated adipocytes generated
by co-culturing human adipocytes with human PC cells could promote
the proliferation, invasion, migration and drug resistance of PC
cells through the suppressor of cytokine signaling 7/STAT3/serum
amyloid A1 pathway (48).
Similarly, Zhang et al (49)
reported that exosomes derived from CAFs could promote the
proliferation of PC cells by regulating Abelson murine leukemia
viral oncogene homolog 2 through miR-224-3p. Based on an in
vivo animal model study, Zhou et al (50) reported that exosomes derived from
CAFs could also exert effects through the silent information
regulator 3/histone 3 lysine 9 acetylation/HIF-1α axis.
By contrast, exosomes secreted by PC cells
themselves also serve a key role in tumor progression. For example,
miR-519a/522-5p in exosomes can promote PC progression by enhancing
the Warburg effect (51), miR-3960
promotes the proliferation, invasion and metastasis of PC cells
through the transcription factor activator protein-2α axis
(52), and the DnaJ heat shock
protein family (HSP40) member B11 protein participates in PC
progression by regulating the EGFR/MAPK pathway (53). Furthermore, exosomes derived from
immune cells also regulate the progression of PC. For instance,
miR-202-5p and miR-142-5p in exosomes derived from macrophages can
enhance the invasiveness of PC cells and promote the metastasis of
pancreatic ductal adenocarcinoma (54). In addition, exosomes derived from
M2-type macrophages can further support tumor growth by targeting
E2F transcription factor 2 and inducing tumor angiogenesis
(55). These results collectively
indicated that exosomes can jointly serve a role in promoting the
occurrence and development of PC through various mechanisms.
The mechanism by which exosomes promote the
progression of PC by shaping an immunosuppressive microenvironment
has been a notable research direction in this field. Although
existing studies have revealed the relevant pathways, the strength
of the evidence varies, mainly based on in vitro cell models
or in vivo animal models. In in vitro cell models, it
has been reported that exosomes derived from PC cells can induce
M2-type macrophage polarization by delivering miR-510 and lncRNA
FGD5-AS1, thereby enhancing the vitality, migration and invasion of
cancer cells (56,57). In in vivo animal models, the
relevant mechanisms of action have been further verified and
expanded. For example, Yang et al (58) reported that exosomes derived from
CAFs can carry prostaglandin-endoperoxide synthase 2 and induce
M2-type macrophage polarization by activating the
nucleotide-binding oligomerization domain-containing protein 1,
thereby promoting the metastasis of PC. The exosomes of
tumor-associated macrophages, through the tyrosine kinase-binding
protein, promote the metastasis process of PC via the CD44/AKT/ERK
pathway (59). These results
revealed the multi-cellular origin and multi-pathway synergy of
exosomes in shaping the immunosuppressive microenvironment,
providing key clues for further understanding of the progression
mechanism of PC.
PC is prone to develop treatment resistance at an
early stage, markedly threatening the life and quality of life of
patients. Currently, the mechanism of drug resistance in PC has not
been fully elucidated, but exosomes may serve a key role in this
process. Exosomes may mediate PC resistance through multiple
pathways (67). They can either
remove drugs from cancer cells directly or deliver miRNA mutations
or upregulated proteins to drug-sensitive cells to exert an
indirect effect (20,68,69).
The drug resistance mechanism of exosomes has been further
clarified in different research models. In vitro cell models
have demonstrated that exosomes derived from PC cells can
upregulate STAT3 expression by inhibiting miR-298 through protein
phosphatase 3 catalytic subunit β (60). Furthermore, exosomes can induce the
high expression level of ATP-binding cassette sub-family G member 2
as a drug efflux pump in cells in vitro (61). The miR-210 in exosomes derived from
PC stem cells can trigger the mTOR signaling pathway, thereby
inducing gemcitabine resistance (62).
Exosomes participate in metabolic reprogramming and
immune microenvironment remodeling in PC by delivering lipid
metabolism-related molecules. For instance, previous research
indicated that the fatty acid binding protein 7 in macrophages can
transfer the induced lipids to CD8+ T cells and tumor
cells through exosomes. This process leads to dysfunction of
CD8+ T cells and proliferation of tumor cells through
metabolic reprogramming (65).
Furthermore, a previous study demonstrated that the medium-chain
acyl-CoA dehydrogenase present in exosomes enhances gemcitabine
resistance by regulating fatty acid metabolism and ferroptosis in
PC (66). These findings indicated
that exosomes serve not only as carriers but also as key mediators
of lipid metabolic reprogramming, indirectly regulating the
immunosuppressive state of the TME through metabolic
intervention.
Although exosomes serve an undeniable ‘dark side’
role in the occurrence and development of PC, they can promote
disease progression by directly driving tumor growth and cell
proliferation, shaping an immunosuppressive microenvironment and
inducing treatment resistance. However, its unique biological
characteristics also provide a novel ‘bright side’ role for the
treatment of PC. This duality of opposites lays the foundation for
its transformation from a disease promoter to a diagnostic and
therapeutic tool. Based on further understanding of the mechanism
of action of exosomes in PC, researchers are actively developing
their application potential in early diagnosis, targeted therapy
and drug resistance reversal, thereby maximizing the positive role
of exosomes in PC. The following section systematically elaborates
on the latest research progress of exosomes as diagnostic markers,
direct therapeutic tools and drug delivery carriers.
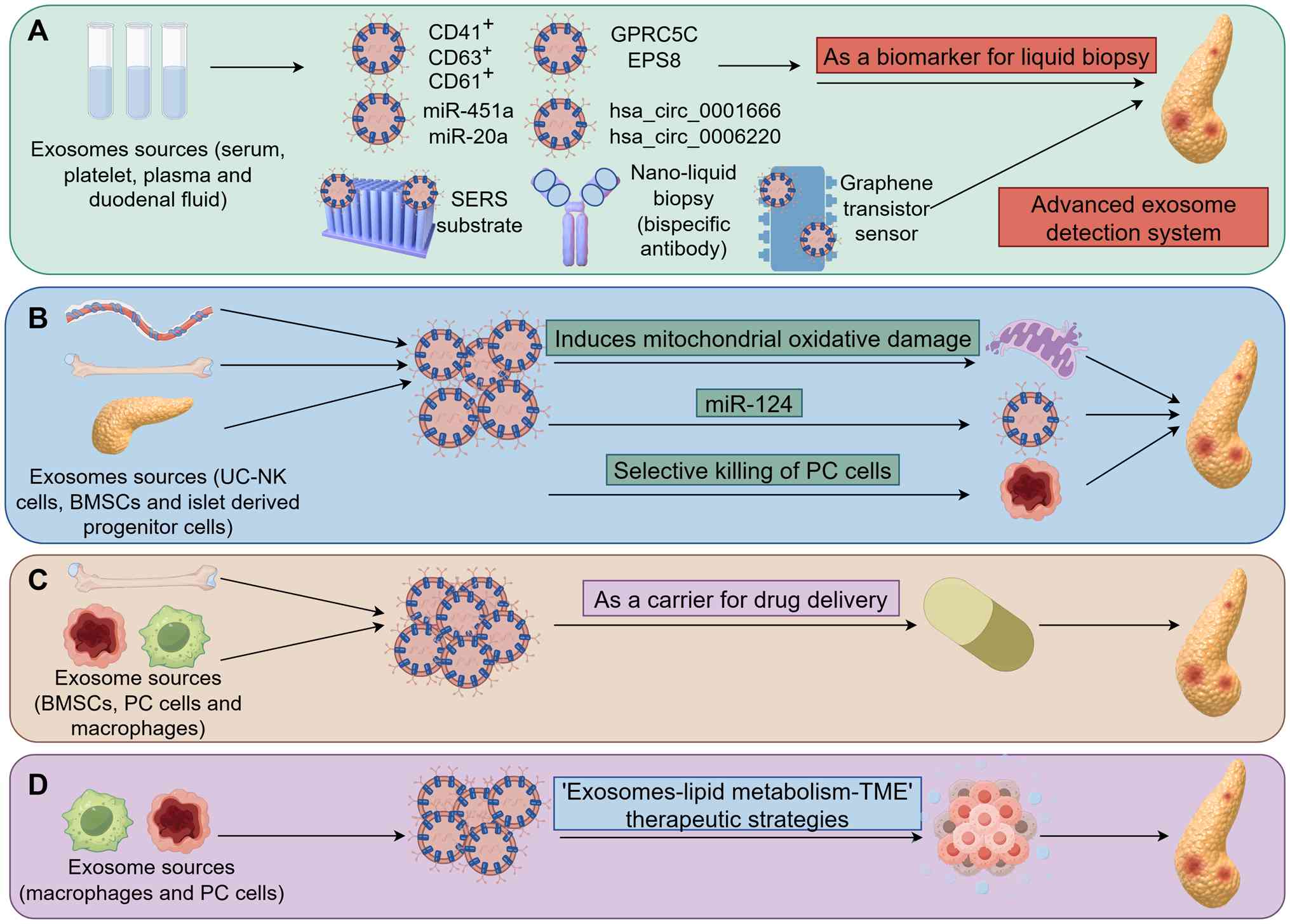
In recent years, exosomes have demonstrated notable
‘bright side’ potential in the diagnosis and treatment of PC. In
terms of diagnosis, due to the protective effects of their lipid
bilayer membrane and the CD47 transmembrane protein, exosomes
carrying PC-specific information remain relatively stable in the
circulatory system and are less susceptible to clearance by
monocytes. This stability provides a possibility for early
diagnosis through liquid biopsy (70–72).
Regarding therapeutic applications, exosomes exhibit a key role: i)
Exosomes can function as natural therapeutic agents originating
directly from immune cells or stem cells, capable of effectively
inhibiting tumor cell proliferation and selectively killing cancer
cells; ii) exosomes represent a highly promising class of drug
delivery vehicles. By loading chemotherapeutic drugs or
immunomodulators, exosomes enable precise targeting to overcome
drug resistance and reprogram the TME, thereby offering notable
potential for effective PC treatment; and iii) exosomes can exert
an active effect through the ‘exosome-lipid metabolism-TME’ axis
(Table II) (73–86)
(Fig. 4).
In recent years, exosomes have demonstrated
potential to serve as a biomarker for the early diagnosis of PC.
Several studies based on clinical samples have systematically
verified their value. In 2022, Odaka et al (73) identified that CD63+ cells
in serum-derived exosomes and CD41/CD61+ cells in
platelet-derived exosomes serve as potential diagnostic biomarkers
for pancreatic ductal adenocarcinoma. Furthermore, miR-451a in
serum-derived exosomes (74), as
well as G protein-coupled receptor class C group 5 member C and
EGFR pathway substrate 8 (75) have
been proposed as candidate markers for the early detection of PC.
In the same year, further research revealed that high expression
levels of circRNAs (hsa_circ_0001666 and hsa_circ_0006220) in
plasma-derived exosomes also possess diagnostic value (76). miR-20a, which is present in exosomes
derived from duodenal fluid, has also been proposed as a biomarker
for pancreatic ductal adenocarcinoma (77). These findings were all based on
systematic validation using clinical samples and had a high level
of evidence strength.
Furthermore, in order to enhance the sensitivity and
specificity of exosome detection, novel detection technologies are
constantly emerging. For example, Li et al (87) constructed an immunoassay system
based on a hierarchical surface-enhanced Raman scattering
substrate, enabling highly sensitive detection of exosomes. The
study identified that a novel combination of leucine-rich
α-2-glycoprotein 1-derived exosomes and glypican-1-derived exosomes
could enhance the diagnostic efficiency for PC. Yu et al
(88) developed a simple nanoliquid
biopsy method that achieves specific, ultrasensitive and
cost-effective exosome detection through a dual-specific biomarker
antigen co-recognition and capture strategy. Yin et al
(89) designed a graphene
field-effect transistor-based biosensor for accurate and rapid
detection of PC-associated exosomes. These technological
advancements have collectively facilitated the transformation of
exosomes from biomarker identification to clinical application,
providing a multi-level solution for the early diagnosis of PC.
Exosomes can serve as a direct therapeutic tool in
inhibiting the development of PC and their effects have been
verified through various research models. In in vitro cell
models, exosomes derived from multiple cell sources exhibited clear
antitumor activity. For instance, exosomes derived from natural
killer cells can upregulate the expression level of let-7b-5p in PC
cells and target the cell cycle regulatory factor CDK6, thereby
inhibiting tumor cell proliferation (78). Furthermore, Hasoglu and Karatug
Kacar (79) identified that
exosomes derived from pancreatic islet progenitor cells can
selectively kill PC cells without notable damage to normal cells,
demonstrating good biological safety. In in vivo animal
models, these therapeutic potentialities can be further verified. A
previous study has demonstrated that exosomes derived from
umbilical cord blood natural killer cells can enter PANC-1 cells
through endocytosis, induce mitochondrial oxidative damage and
inhibit the progression of this cell line, revealing a marked
anti-PC effect (80). Furthermore,
miR-124 contained in exosomes derived from bone marrow mesenchymal
stem cells (BMSCs) has also exhibited anti-PC activity in both
in vitro and in vivo experiments (81). These studies collectively
demonstrated that exosomes have the potential for multi-mechanism
synergy, extensive sources and good safety in the treatment of PC,
providing notable basis for the development of novel biological
therapies.
Exosomes, as an efficient drug delivery carrier, are
expected to provide a safer and more effective delivery strategy
for traditional anti-PC drugs due to their notable biocompatibility
and low toxicity (90,91). An in vitro cell model study
demonstrated that exosomes derived from human BMSCs carrying
gemcitabine can enhance apoptosis to inhibit the proliferation of
PC cells, thereby inhibiting the progression of PC (82). Furthermore, exosomes derived from
M1-type macrophages loaded with gemcitabine and deferasirox can
effectively overcome drug resistance and provide a potential
effective treatment strategy for patients with PC with drug
resistance (83). The in
vivo animal models further demonstrated the therapeutic
advantages of the exosome delivery system, autologous exosomes
carrying gemcitabine can markedly promote the uptake of the drug in
cancer cells and enhance its antitumor effect (84). Furthermore, the emerging engineered
exosome platform offers a novel direction for the regulation of the
TME; for instance, Sun et al (92) developed a novel exosome-based drug
delivery platform, cmExoaCD11b, aiming to precisely
target and reprogram the TME for immunotherapy of PC. This
technology can cause M2 macrophages to polarize to the M1
phenotype, thereby reprogramming the TME and markedly inhibiting
the development of PC. These studies collectively demonstrated that
exosomes not only serve as efficient drug carriers but can also
achieve multi-mechanism synergistic antitumor effects through
dynamic interactions with the TME.
Of note, exosomes derived from M1-type macrophages
can inhibit the lipid metabolism pathway, thereby reducing the
proliferation, migratory and invasive abilities of PC cells, while
simultaneously promoting their apoptosis (85). Furthermore, a previous study has
demonstrated that exosomes may be involved in lipid metabolism and
regulate the TME, thereby participating in multiple processes of PC
(86). These findings provided
novel insights in developing metabolism-targeted exosome-based
therapies.
In summary, the present review systematically
elaborated on the dual roles of exosomes in PC and their specific
mechanisms of action. Exosomes serve a positive role in the
treatment of PC, however, their involvement in regulating PC cells,
shaping an immunosuppressive microenvironment and promoting
chemotherapy resistance remains to be explored in future research.
Further exploration of the key molecules, proteins and signaling
pathways underlying these adverse effects may provide novel
research directions and intervention strategies for targeted
therapy of PC.
However, numerous challenges remain in realizing the
optimal application of exosomes for PC therapy. First, current
techniques in isolating and extracting exosomes face notable
limitations (93). Although
multiple methods are available, such as ultracentrifugation and
size-exclusion chromatography, a standardized protocol that
simultaneously ensures high purity, simplicity and low cost is
still lacking (94,95). Establishing a unified separation
standard for exosomes is of notable importance for the
reproducibility of research and cross-study comparisons. Second,
the diameter of exosomes ranges from 30–150 nm, which is below the
diffraction limit of conventional optical microscopy. This poses
notable challenges in visualizing the release, transport and
real-time dynamics of exosomes. In recent years, super-resolution
microscopy holds promise in resolving exosomal ultrastructure; its
high spatial resolution often comes at the expense of temporal
efficiency, making dynamic studies in living cells particularly
challenging (96,97). Third, the extraction of exosomes
remains costly. While obtaining exosomes from patient urine or
saliva is a non-invasive approach with certain practical
advantages, the process is often time-consuming and requires
notable financial investment (98).
Of note, in recent years, EVs extracted from natural plants or food
sources have successively demonstrated notable biocompatibility and
therapeutic potential, providing a key direction and research
breakthrough in reducing extraction costs and developing stable and
controllable EV resources (99,100).
Fourth, although exosomes exhibit notable promise as drug delivery
vehicles, key issues such as high loss rates during drug loading
and elevated production costs should be addressed (101). Currently, the use of serum-free
culture systems to optimize the production process of exosomes, as
well as the development of synthetic biomimetic vesicles with
similar functions, provide a feasible technical direction in
addressing these issues (102).
Fifth, there is a notable lack of clinical data. Current research
on exosome-based therapies for PC is largely limited to in
vitro studies and in vivo mouse models. However, human
clinical trials are still lacking (78,82,84).
To promote its clinical application, the following key steps need
to be clarified: i) Pharmacological and toxicological research are
warranted to elucidate the pharmacokinetic and pharmacodynamic
characteristics, biodistribution and potential immunogenicity of
exosomes in the body; ii) in-depth clinical trials; and iii)
progressive research from safety assessment to efficacy validation.
Sixth, future studies should further elucidate how exosomes
regulate the TME through lipid metabolism reprogramming,
particularly their role in metabolic-immune crosstalk. The
development of therapeutic strategies targeting exosomal lipid
metabolism, such as exosome-based delivery systems for metabolic
enzyme inhibitors, may potentially offer novel avenues in reversing
immunosuppression and overcoming chemoresistance (103,104).
Addressing the aforementioned challenges will
notably advance the application of exosomes in PC research and
therapy. The development of more efficient, simpler and
cost-effective techniques in isolating and extracting exosomes
represents a key and urgent breakthrough warranted in the field.
Simultaneously, establishing standardized protocols for exosome
extraction is key to improving operational efficiency and
minimizing sample loss. Furthermore, systematic comparative studies
should be performed to clarify the advantages and suitable
scenarios of exosomes over traditional nanocarriers such as
liposomes in drug delivery. Lastly, active efforts are warranted to
promote the translation of exosome-based therapies for PC from
basic research to clinical trials.
In summary, exosomes hold notable potential as a
therapeutic strategy for PC. Future research should focus on
further understanding of their functional mechanisms, optimizing
their isolation and extraction techniques and facilitating their
clinical translation, with the potential goal of offering novel
treatment options for patients with PC in the clinic at the
earliest opportunity.
Exosomes, as key mediators of intercellular
communication, are involved in the occurrence and progression of
PC. The present review systematically elaborated on the dual roles
of exosomes in PC and analyzed their mechanisms of action at the
molecular and signaling pathway levels. Previous research has
identified that exosomes primarily promote the initiation and
progression of PC through the following pathways: i) Directly
regulating tumor cell behavior, driving the growth and
proliferation of PC cells; ii) remodeling the immunosuppressive
microenvironment, providing a more favorable environment for the
growth and proliferation of PC cells; iii) inducing therapeutic
resistance, thereby reducing the efficacy of drug treatments; and
iv) regulating lipid metabolism, by reprogramming metabolism,
affects the TME, thereby further supporting the progression of
PC.
However, exosomes also exhibit potential in
inhibiting the development of PC: i) Specific proteins, nucleic
acids and other components carried by exosomes potentially offer
novel avenues for the early diagnosis and prognosis assessment of
PC; ii) certain exosomes can inhibit the progression of PC through
different signaling pathways or by modulating the TME; iii)
exosomes can serve as notable carriers for drugs effectively
treating PC, enabling targeted delivery of therapeutic agents to
achieve enhanced efficacy and reduced toxicity; and iv) the
regulatory strategies based on the ‘exosome-lipid metabolism-TME’
axis may become a novel target for the treatment of PC.
The functional duality exhibited by exosomes in PC
reflects their complex role in tumor biology: Exosomes can both
promote malignant tumor progression and hold potential as
diagnostic and therapeutic tools. This paradoxical characteristic
suggests that their functions may be regulated by multiple factors,
including the source cells, cargo composition and recipient
microenvironment. Future research is warranted to further elucidate
the specific mechanisms by which exosomes transmit information
between different tumor stages and cell types and explore the
molecular basis of their transformation from the ‘dark side’ to the
‘bright side’.
In conclusion, a comprehensive understanding of the
dual roles of exosomes in PC will not only help further the
understanding of the communication network within the TME but also
provide a notable basis in developing novel diagnostic strategies
and therapeutic methods based on exosomes. Advancement in the
clinical translation of exosomes may potentially open novel paths
for the early detection and precision treatment of PC in the
future.
Not applicable.
Funding: No funding was received.
Not applicable.
ZG contributed to the conception and overall design
of the study, drafted the manuscript and prepared the figures and
tables. QL reviewed and revised the manuscript. All authors read
and approved the final manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Siegel RL, Giaquinto AN and Jemal A:
Cancer statistics, 2024. CA Cancer J Clin. 74:12–49.
2024.PubMed/NCBI
|
|
2
|
Rawla P, Sunkara T and Gaduputi V:
Epidemiology of pancreatic cancer: Global trends, etiology and risk
factors. World J Oncol. 10:10–27. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Conroy T, Hammel P, Hebbar M, Ben
Abdelghani M, Wei AC, Raoul JL, Choné L, Francois E, Artru P, Biagi
JJ, et al: FOLFIRINOX or gemcitabine as adjuvant therapy for
pancreatic cancer. N Engl J Med. 379:2395–2406. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stoop TF, Javed AA, Oba A, Koerkamp BG,
Seufferlein T, Wilmink JW and Besselink MG: Pancreatic cancer.
Lancet. 405:1182–1202. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Saba H and Goggins M: Familial pancreatic
cancer. Gastroenterol Clin North Am. 51:561–575. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wood LD, Yurgelun MB and Goggins MG:
Genetics of familial and sporadic pancreatic cancer.
Gastroenterology. 156:2041–2055. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ngamruengphong S and Canto MI: Screening
for pancreatic cancer. Surg Clin North Am. 96:1223–1233. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Klein AP, Brune KA, Petersen GM, Goggins
M, Tersmette AC, Offerhaus GJ, Griffin C, Cameron JL, Yeo CJ, Kern
S and Hruban RH: Prospective risk of pancreatic cancer in familial
pancreatic cancer kindreds. Cancer Res. 64:2634–2638. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Torphy RJ, Zhu Y and Schulick RD:
Immunotherapy for pancreatic cancer: Barriers and breakthroughs.
Ann Gastroenterol Surg. 2:274–281. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Raposo G and Stoorvogel W: Extracellular
vesicles: Exosomes, microvesicles, and friends. J Cell Biol.
200:373–383. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ham S, Lima LG, Chai EPZ, Muller A, Lobb
RJ, Krumeich S, Wen SW, Wiegmans AP and Möller A: Breast
cancer-derived exosomes alter macrophage polarization via
gp130/STAT3 signaling. Front Immunol. 9:8712018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Costa-Silva B, Aiello NM, Ocean AJ, Singh
S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, et
al: Pancreatic cancer exosomes initiate pre-metastatic niche
formation in the liver. Nat Cell Biol. 17:816–826. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Han S, Gonzalo DH, Feely M, Rinaldi C,
Belsare S, Zhai H, Kalra K, Gerber MH, Forsmark CE and Hughes SJ:
Stroma-derived extracellular vesicles deliver tumor-suppressive
miRNAs to pancreatic cancer cells. Oncotarget. 9:5764–5777. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang H, Freitas D, Kim HS, Fabijanic K,
Li Z, Chen H, Mark MT, Molina H, Martin AB, Bojmar L, et al:
Identification of distinct nanoparticles and subsets of
extracellular vesicles by asymmetric flow field-flow fractionation.
Nat Cell Biol. 20:332–343. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Al-Nedawi K, Meehan B, Micallef J, Lhotak
V, May L, Guha A and Rak J: Intercellular transfer of the oncogenic
receptor EGFRvIII by microvesicles derived from tumour cells. Nat
Cell Biol. 10:619–624. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Raimondo S, Saieva L, Corrado C, Fontana
S, Flugy A, Rizzo A, De Leo G and Alessandro R: Chronic myeloid
leukemia-derived exosomes promote tumor growth through an autocrine
mechanism. Cell Commun Signal. 13:82015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yukawa H, Suzuki K, Aoki K, Arimoto T,
Yasui T, Kaji N, Ishikawa T, Ochiya T and Baba Y: Imaging of
angiogenesis of human umbilical vein endothelial cells by uptake of
exosomes secreted from hepatocellular carcinoma cells. Sci Rep.
8:67652018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhao H, Yang L, Baddour J, Achreja A,
Bernard V, Moss T, Marini JC, Tudawe T, Seviour EG, San Lucas FA,
et al: Tumor microenvironment derived exosomes pleiotropically
modulate cancer cell metabolism. Elife. 5:e102502016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qin C, Li T, Lin C, Zhao B, Li Z, Zhao Y
and Wang W: The systematic role of pancreatic cancer exosomes:
distant communication, liquid biopsy and future therapy. Cancer
Cell Int. 24:2642024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oliveira C, Calmeiro J, Carrascal MA,
Falcão A, Gomes C, Miguel Neves B and Teresa Cruz M: Exosomes as
new therapeutic vectors for pancreatic cancer treatment. Eur J
Pharm Biopharm. 161:4–14. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chargaff E and West R: The biological
significance of the thromboplastic protein of blood. J Biol Chem.
166:189–197. 1946. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Trams EG, Lauter CJ, Salem N Jr and Heine
U: Exfoliation of membrane ecto-enzymes in the form of
micro-vesicles. Biochim Biophys Acta. 645:63–70. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Johnstone RM, Adam M, Hammond JR, Orr L
and Turbide C: Vesicle formation during reticulocyte maturation.
Association of plasma membrane activities with released vesicles
(exosomes). J Biol Chem. 262:9412–9420. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Johnstone RM, Bianchini A and Teng K:
Reticulocyte maturation and exosome release: Transferrin receptor
containing exosomes shows multiple plasma membrane functions.
Blood. 74:1844–1851. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yu LL, Zhu J, Liu JX, Jiang F, Ni WK, Qu
LS, Ni RZ, Lu CH and Xiao MB: A comparison of traditional and novel
methods for the separation of exosomes from human samples. Biomed
Res Int. 2018:36345632018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lathe GH and Ruthven CR: The separation of
substances on the basis of their molecular weights, using columns
of starch and water. Biochem J. 60:xxxiv1955.PubMed/NCBI
|
|
27
|
Cheng H, Fang H, Xu RD, Fu MQ, Chen L,
Song XY, Qian JY, Zou YZ, Ma JY and Ge JB: Development of a rinsing
separation method for exosome isolation and comparison to
conventional methods. Eur Rev Med Pharmacol Sci. 23:5074–5083.
2019.PubMed/NCBI
|
|
28
|
Yoo CE, Kim G, Kim M, Park D, Kang HJ, Lee
M and Huh N: A direct extraction method for microRNAs from exosomes
captured by immunoaffinity beads. Anal Biochem. 431:96–98. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wunsch BH, Smith JT, Gifford SM, Wang C,
Brink M, Bruce RL, Austin RH, Stolovitzky G and Astier Y: Nanoscale
lateral displacement arrays for the separation of exosomes and
colloids down to 20 nm. Nat Nanotechnol. 11:936–940. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li P, Kaslan M, Lee SH, Yao J and Gao Z:
Progress in exosome isolation techniques. Theranostics. 7:789–804.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Alderton GK: Diagnosis: Fishing for
exosomes. Nat Rev Cancer. 15:4532015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Colombo M, Raposo G and Théry C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Simons M and Raposo G: Exosomes-vesicular
carriers for intercellular communication. Curr Opin Cell Biol.
21:575–581. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Stoorvogel W, Strous GJ, Geuze HJ,
Oorschot V and Schwartz AL: Late endosomes derive from early
endosomes by maturation. Cell. 65:417–427. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hanson PI and Cashikar A: Multivesicular
body morphogenesis. Annu Rev Cell Dev Biol. 28:337–362. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sahu R, Kaushik S, Clement CC, Cannizzo
ES, Scharf B, Follenzi A, Potolicchio I, Nieves E, Cuervo AM and
Santambrogio L: Microautophagy of cytosolic proteins by late
endosomes. Dev Cell. 20:131–139. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Skotland T, Hessvik NP, Sandvig K and
Llorente A: Exosomal lipid composition and the role of ether lipids
and phosphoinositides in exosome biology. J Lipid Res. 60:9–18.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Théry C, Zitvogel L and Amigorena S:
Exosomes: Composition, biogenesis and function. Nat Rev Immunol.
2:569–579. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gusachenko ON, Zenkova MA and Vlassov VV:
Nucleic acids in exosomes: Disease markers and intercellular
communication molecules. Biochemistry (Mosc). 78:1–7. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Valadi H, Ekström K, Bossios A, Sjöstrand
M, Lee JJ and Lötvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sadik N, Cruz L, Gurtner A, Rodosthenous
RS, Dusoswa SA, Ziegler O, Van Solinge TS, Wei Z, Salvador-Garicano
AM, Gyorgy B, et al: Extracellular RNAs: A new awareness of old
perspectives. Methods Mol Biol. 1740:1–15. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Oliveira GP Jr, Zigon E, Rogers G,
Davodian D, Lu S, Jovanovic-Talisman T, Jones J, Tigges J, Tyagi S
and Ghiran IC: Detection of extracellular vesicle RNA using
molecular beacons. iScience. 23:1007822020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mitchell PS, Parkin RK, Kroh EM, Fritz BR,
Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O'Briant
KC, Allen A, et al: Circulating microRNAs as stable blood-based
markers for cancer detection. Proc Natl Acad Sci USA.
105:10513–10518. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Xie Z, Yin X, Gong B, Nie W, Wu B, Zhang
X, Huang J, Zhang P, Zhou Z and Li Z: Salivary microRNAs show
potential as a noninvasive biomarker for detecting resectable
pancreatic cancer. Cancer Prev Res (Phila). 8:165–173. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
de Gonzalo-Calvo D, Kenneweg F, Bang C,
Toro R, van der Meer RW, Rijzewijk LJ, Smit JW, Lamb HJ,
Llorente-Cortes V and Thum T: Circulating long-non coding RNAs as
biomarkers of left ventricular diastolic function and remodelling
in patients with well-controlled type 2 diabetes. Sci Rep.
6:373542016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sun W, Ren Y, Lu Z and Zhao X: The
potential roles of exosomes in pancreatic cancer initiation and
metastasis. Mol Cancer. 19:1352020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Honselmann KC, Finetti P, Birnbaum DJ,
Monsalve CS, Wellner UF, Begg SKS, Nakagawa A, Hank T, Li A,
Goldsworthy MA, et al: Neoplastic-stromal cell cross-talk regulates
matrisome expression in pancreatic cancer. Mol Cancer Res.
18:1889–1902. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Noda K, Sato Y, Okada Y, Nishida K, Kawano
Y, Tanahashi T, Bando M, Okamoto K, Takehara M, Sogabe M, et al:
Exosomal miR-199a-3p secreted from cancer-associated adipocytes
promotes pancreatic cancer progression. Cancer Med. 13:e702652024.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang L, Chen Y, Dai Y, Mou W, Deng P and
Jin Y, Xu J and Jin Y: Cancer-associated fibroblast-derived exosome
Leptin promotes malignant biological lineage in pancreatic ductal
adenocarcinoma by regulating ABL2 via miR-224-3p. Mol Biol Rep.
51:9952024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhou B, Lei JH, Wang Q, Qu TF, Cha LC,
Zhan HX, Liu SL, Hu X, Sun CD, Guo WD, et al: Cancer-associated
fibroblast-secreted miR-421 promotes pancreatic cancer by
regulating the SIRT3/H3K9Ac/HIF-1α axis. Kaohsiung J Med Sci.
38:1080–1092. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Feng S, Chen R, Huang H, Ong M, Jiang J,
Cui J and Ling Q: Cancer cell-derived exosomal miR-519a/522-5p
promotes pancreatic cancer progression by enhancing warburg effect.
Ann Surg Oncol. 32:7068–7081. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wu J: Pancreatic cancer-derived exosomes
promote the proliferation, invasion, and metastasis of pancreatic
cancer by the miR-3960/TFAP2A axis. J Oncol. 2022:35903262022.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Liu P, Zu F, Chen H, Yin X and Tan X:
Exosomal DNAJB11 promotes the development of pancreatic cancer by
modulating the EGFR/MAPK pathway. Cell Mol Biol Lett. 27:872022.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Chen Y, Lei Y, Li J, Wang X and Li G:
Macrophage-derived exosomal microRNAs promote metastasis in
pancreatic ductal adenocarcinoma. Int Immunopharmacol.
129:1115902024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yang Y, Guo Z, Chen W, Wang X, Cao M, Han
X, Zhang K, Teng B, Cao J, Wu W, et al: M2 macrophage-derived
exosomes promote angiogenesis and growth of pancreatic ductal
adenocarcinoma by targeting E2F2. Mol Ther. 29:1226–1238. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang T, Ye L, Zhou Y, Zhang X, Li R, Zhou
Y, Weng J, Mo Q and Yu Y: Pancreatic cancer-derived exosomal
miR-510 promotes macrophage M2 polarization and facilitates cancer
cell aggressive phenotypes. Hum Cell. 38:172024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
He Z, Wang J, Zhu C, Xu J, Chen P, Jiang
X, Chen Y, Jiang J and Sun C: Exosome-derived FGD5-AS1 promotes
tumor-associated macrophage M2 polarization-mediated pancreatic
cancer cell proliferation and metastasis. Cancer Lett.
548:2157512022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yang W, Zheng Y, Zhou H, Liang R and Hu C:
Cancer-associated fibroblast-secreted exosomes regulate macrophage
polarization in pancreatic cancer via the NOD1 pathway. J Biochem
Mol Toxicol. 39:e701262025. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhong D, Liao Y, Chen W, Huang X, Liu J
and Wang Z: TYROBP promotes the spread of pancreatic cancer by
causing M2 TAM polarization. J Gastroenterol Hepatol. 39:2926–2939.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wang C, Xu S and Qin Y: Tumor-derived
exosome PPP3CB induce gemcitabine resistance by regulating
miR-298/STAT3 in pancreatic cancer. Heliyon. 10:e364342024.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Bhattacharya S, Pal K, Sharma AK, Dutta
SK, Lau JS, Yan IK, Wang E, Elkhanany A, Alkharfy KM, Sanyal A, et
al: GAIP interacting protein C-terminus regulates autophagy and
exosome biogenesis of pancreatic cancer through metabolic pathways.
PLoS One. 9:e1144092014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Yang Z, Zhao N, Cui J, Wu H, Xiong J and
Peng T: Exosomes derived from cancer stem cells of
gemcitabine-resistant pancreatic cancer cells enhance drug
resistance by delivering miR-210. Cell Oncol (Dordr). 43:123–136.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Qi R, Bai Y, Li K, Liu N, Xu Y, Dal E,
Wang Y, Lin R, Wang H, Liu Z, et al: Cancer-associated fibroblasts
suppress ferroptosis and induce gemcitabine resistance in
pancreatic cancer cells by secreting exosome-derived
ACSL4-targeting miRNAs. Drug Resist Updat. 68:1009602023.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Mikamori M, Yamada D, Eguchi H, Hasegawa
S, Kishimoto T, Tomimaru Y, Asaoka T, Noda T, Wada H, Kawamoto K,
et al: MicroRNA-155 controls exosome synthesis and promotes
gemcitabine resistance in pancreatic ductal adenocarcinoma. Sci
Rep. 7:423392017. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Xu S, Peng X, Wang Z, Le C, Wu X, Zeng Z,
Zeng S, Zhang C, Qiu M, Zou X, et al: FABP7-mediated lipid-laden
macrophages drive the formation of pre-metastatic niche and liver
metastasis. Int J Biol Sci. 21:4388–4409. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Yang Y, Gu H, Zhang K, Guo Z, Wang X, Wei
Q, Weng L, Han X, Lv Y, Cao M, et al: Exosomal ACADM sensitizes
gemcitabine-resistance through modulating fatty acid metabolism and
ferroptosis in pancreatic cancer. BMC Cancer. 23:7892023.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Mashouri L, Yousefi H, Aref AR, Ahadi AM,
Molaei F and Alahari SK: Exosomes: Composition, biogenesis, and
mechanisms in cancer metastasis and drug resistance. Mol Cancer.
18:752019. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Adamska A, Elaskalani O, Emmanouilidi A,
Kim M, Abdol Razak NB, Metharom P and Falasca M: Molecular and
cellular mechanisms of chemoresistance in pancreatic cancer. Adv
Biol Regul. 68:77–87. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Fan J, Wei Q, Koay EJ, Liu Y, Ning B,
Bernard PW, Zhang N, Han H, Katz MH, Zhao Z and Hu Y:
Chemoresistance transmission via exosome-mediated EphA2 transfer in
pancreatic cancer. Theranostics. 8:5986–5994. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Guo W, Ying P, Ma R, Jing Z, Ma G, Long J,
Li G and Liu Z: Liquid biopsy analysis of lipometabolic exosomes in
pancreatic cancer. Cytokine Growth Factor Rev. 73:69–77. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Søreide K, Ismail W, Roalsø M, Ghotbi J
and Zaharia C: Early diagnosis of pancreatic cancer: Clinical
premonitions, timely precursor detection and increased
curative-intent surgery. Cancer Control. 30:107327482311547112023.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Fang X, Lan H, Jin K and Qian J:
Pancreatic cancer and exosomes: role in progression, diagnosis,
monitoring, and treatment. Front Oncol. 13:11495512023. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Odaka H, Hiemori K, Shimoda A, Akiyoshi K
and Tateno H: CD63-positive extracellular vesicles are potential
diagnostic biomarkers of pancreatic ductal adenocarcinoma. BMC
Gastroenterol. 22:1532022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Chen J, Yao D, Chen W, Li Z, Guo Y, Zhu F
and Hu X: Serum exosomal miR-451a acts as a candidate marker for
pancreatic cancer. Int J Biol Markers. 37:74–80. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Yoshioka Y, Shimomura M, Saito K, Ishii H,
Doki Y, Eguchi H, Nakatsura T, Itoi T, Kuroda M, Mori M and Ochiya
T: Circulating cancer-associated extracellular vesicles as early
detection and recurrence biomarkers for pancreatic cancer. Cancer
Sci. 113:3498–3509. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Hong L, Xu L, Jin L, Xu K, Tang W, Zhu Y,
Qiu X and Wang J: Exosomal circular RNA hsa_circ_0006220, and
hsa_circ_0001666 as biomarkers in the diagnosis of pancreatic
cancer. J Clin Lab Anal. 36:e244472022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Taniguchi T, Ideno N, Araki T, Miura S,
Yamamoto M, Nakafusa T, Higashijima N, Yamamoto T, Tamura K,
Nakamura S, et al: MicroRNA-20a in extracellular vesicles derived
from duodenal fluid is a possible biomarker for pancreatic ductal
adenocarcinoma. DEN Open. 4:e3332024. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Di Pace AL, Pelosi A, Fiore PF, Tumino N,
Besi F, Quatrini L, Santopolo S, Vacca P and Moretta L: MicroRNA
analysis of Natural Killer cell-derived exosomes: the microRNA
let-7b-5p is enriched in exosomes and participates in their
anti-tumor effects against pancreatic cancer cells. Oncoimmunology.
12:22210812023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Hasoglu I and Karatug Kacar A: The
therapeutic effects of exosomes the first time isolated from
pancreatic islet-derived progenitor cells in the treatment of
pancreatic cancer. Protoplasma. 261:281–291. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Zheng Y, Zou X, Li Q, Jiang D, Zhu F and
Wu Y: Exosomes derived from umbilical cord blood NK cells inhibit
the progression of pancreatic cancer by targeting ROS-mediated
mitochondrial dysfunction. Saudi Pharm J. 33:82025. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xu Y, Liu N, Wei Y, Zhou D, Lin R, Wang X
and Shi B: Anticancer effects of miR-124 delivered by BM-MSC
derived exosomes on cell proliferation, epithelial mesenchymal
transition, and chemotherapy sensitivity of pancreatic cancer
cells. Aging (Albany NY). 12:19660–19676. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Tang ZG, Chen TM, Lu Y, Wang Z, Wang XC
and Kong Y: Human bone marrow mesenchymal stem cell-derived
exosomes loaded with gemcitabine inhibit pancreatic cancer cell
proliferation by enhancing apoptosis. World J Gastrointest Oncol.
16:4006–4013. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhao Y, Zheng Y, Zhu Y, Zhang Y, Zhu H and
Liu T: M1 macrophage-derived exosomes loaded with gemcitabine and
deferasirox against chemoresistant pancreatic cancer.
Pharmaceutics. 13:14932021. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Li YJ, Wu JY, Wang JM, Hu XB, Cai JX and
Xiang DX: Gemcitabine loaded autologous exosomes for effective and
safe chemotherapy of pancreatic cancer. Acta Biomater. 101:519–530.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zhan T, Zou Y, Han Z, Tian X, Chen M, Liu
J, Yang X, Zhu Q, Liu M, Chen W, et al: Single-cell sequencing
combined with spatial transcriptomics reveals that the IRF7 gene in
M1 macrophages inhibits the occurrence of pancreatic cancer by
regulating lipid metabolism-related mechanisms. Clin Transl Med.
14:e17992024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Li S, Dong R, Kang Z, Li H, Wu X and Li T:
Exosomes: Another intercellular lipometabolic communication
mediators in digestive system neoplasms? Cytokine Growth Factor
Rev. 73:93–100. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Li J, Li Y, Chen S, Duan W, Kong X, Wang
Y, Zhou L, Li P, Zhang C, Du L and Wang C: Highly sensitive exosome
detection for early diagnosis of pancreatic cancer using
immunoassay based on hierarchical surface-enhanced raman scattering
substrate. Small Methods. 6:e22001542022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Yu Z, Yang Y, Fang W, Hu P, Liu Y and Shi
J: Dual tumor exosome biomarker co-recognitions based nanoliquid
biopsy for the accurate early diagnosis of pancreatic cancer. ACS
Nano. 17:11384–11395. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Yin T, Xu L, Gil B, Merali N, Sokolikova
MS, Gaboriau DCA, Liu DSK, Muhammad Mustafa AN, Alodan S, Chen M,
et al: Graphene sensor arrays for rapid and accurate detection of
pancreatic cancer exosomes in patients' blood plasma samples. ACS
Nano. 17:14619–14631. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Jabbari N, Karimipour M, Khaksar M,
Akbariazar E, Heidarzadeh M, Mojarad B, Aftab H, Rahbarghazi R and
Rezaie J: Tumor-derived extracellular vesicles: Insights into
bystander effects of exosomes after irradiation. Lasers Med Sci.
35:531–545. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Jabbari N, Akbariazar E, Feqhhi M,
Rahbarghazi R and Rezaie J: Breast cancer-derived exosomes: Tumor
progression and therapeutic agents. J Cell Physiol. 235:6345–6356.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Sun M, Zhang H, Ma Y, Wang S, Chen J, Cui
Y, Zhang Y, Hu S, Zhou D, Zhang P, et al: In situ programming of
the tumor microenvironment to alleviate immunosuppression for
pancreatic cancer immunotherapy. Adv Sci (Weinh). 12:e040082025.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
van Niel G, Carter DRF, Clayton A, Lambert
DW, Raposo G and Vader P: Challenges and directions in studying
cell-cell communication by extracellular vesicles. Nat Rev Mol Cell
Biol. 23:369–382. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Royo F, Théry C, Falcón-Pérez JM,
Nieuwland R and Witwer KW: Methods for separation and
characterization of extracellular vesicles: Results of a worldwide
survey performed by the ISEV rigor and standardization
subcommittee. Cells. 9:19552020. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Ramirez MI, Amorim MG, Gadelha C, Milic I,
Welsh JA, Freitas VM, Nawaz M, Akbar N, Couch Y, Makin L, et al:
Technical challenges of working with extracellular vesicles.
Nanoscale. 10:881–906. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Ghanam J, Chetty VK, Zhu X, Liu X, Gelléri
M, Barthel L, Reinhardt D, Cremer C and Thakur BK: Single molecule
localization microscopy for studying small extracellular vesicles.
Small. 19:e22050302023. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhang YP, Lobanova E, Dworkin A, Furlepa
M, Yang WS, Burke M, Meng JX, Potter N, Sala RL, Kahanawita L, et
al: Improved imaging surface for quantitative single-molecule
microscopy. ACS Appl Mater Interfaces. 16:37255–37264. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Ju Y, Hu Y, Yang P, Xie X and Fang B:
Extracellular vesicle-loaded hydrogels for tissue repair and
regeneration. Mater Today Bio. 18:1005222022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Lu Y, Zhou H, Han C, Gong Y, Li Y, Xia Y,
Liang B, Yang H and Wang Z: Enhanced therapeutic impact of
Shikonin-encapsulated exosomes in the inhibition of colorectal
cancer progression. Nanotechnology. 35:2024. View Article : Google Scholar
|
|
100
|
Zhao B, Lin H, Jiang X, Li W, Gao Y, Li M,
Yu Y, Chen N and Gao J: Exosome-like nanoparticles derived from
fruits, vegetables, and herbs: Innovative strategies of therapeutic
and drug delivery. Theranostics. 14:4598–4621. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Liang Y, Duan L, Lu J and Xia J:
Engineering exosomes for targeted drug delivery. Theranostics.
11:3183–3195. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Herrmann IK, Wood MJA and Fuhrmann G:
Extracellular vesicles as a next-generation drug delivery platform.
Nat Nanotechnol. 16:748–759. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Phutela K, Bal A, Singh N and Sharma S:
Hydroxycitrate-loaded exosomes demonstrate enhanced therapeutic
efficacy against lung adenocarcinoma by inhibiting the metabolic
enzyme ATP citrate lyase. Nanoscale Adv. 7:3846–3858. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Mohammed O, Ahmed Assaye M, Alemayehu E,
Tufa A and Genet S: Exosomes in cancer metabolism and drug
resistance: A review. Biomol Biomed. 26:730–745. 2025. View Article : Google Scholar : PubMed/NCBI
|