Introduction
Hereditary breast and ovarian cancer syndrome (HBOC)
is caused by pathogenic germline variants in BRCA1 or
BRCA2 and significantly elevates the risk of breast and
ovarian cancers. Emerging evidence suggests that there is also a
modestly increased risk of uterine cancers, especially uterine
papillary serous carcinoma (UPSC) (1–3). A
2021 meta-analysis reported a 2.2-fold greater risk of uterine
cancer, particularly UPSC, in BRCA1/2 mutation carriers
[standardized incidence ratio (SIR) 17.97 for UPSC] (4). However, most endometrial carcinomas in
carriers are histologically heterogeneous, so the precise
relationship between the mutation and uterine carcinosarcoma (UCS)
is hard to quantify (3). In
addition, a recent genomic study has suggested that UCS shares
molecular characteristics with high-grade endometrial carcinomas,
highlighting the importance of molecular profiling in understanding
tumor biology and guiding therapeutic strategies (5).
UCS is a rare, aggressive neoplasm comprised
epithelial and mesenchymal components. It accounts for fewer than
5% of uterine cancers (6). It is
typically seen in postmenopausal women, and its incidence peaks in
patients over 60. It is exceptionally uncommon in young adults
(6). Furthermore, the molecular
features and clinical behavior of UCS arising in patients with
hereditary cancer syndromes remain poorly characterized.
This report presents a 21-year-old female patient
with a BRCA1 mutation who developed UCS. To the best of our
knowledge, this represents one of the youngest reported cases of
uterine carcinosarcoma associated with hereditary breast and
ovarian cancer syndrome. This case suggests a potential need to
expand the HBOC tumor spectrum to include early UCS, as well as
possible reconsideration of surveillance and risk-reduction
strategies in young BRCA mutation carriers.
Case report
A 21-year-old woman presented with continuous
uterine bleeding. When ultrasound revealed a tumor in the uterine
cavity, she was referred to our institution for further evaluation.
Relevant family history included a paternal grandmother who had
suffered breast and peritoneal cancer, and prostate cancer in a
maternal grandfather and a maternal uncle. The patient's laboratory
results identified severely low hemoglobin levels (5.5 g/dl). Her
hormone levels, including estradiol, luteinizing hormone,
follicle-stimulating hormone, and prolactin, were within normal
limits. Her levels of lactate dehydrogenase (330 U/l) and cancer
antigen (CA) 19-9 (81.6 U/ml) were mildly elevated. Other tumor
markers, including CA125 and carcinoembryonic antigen, were within
normal limits.
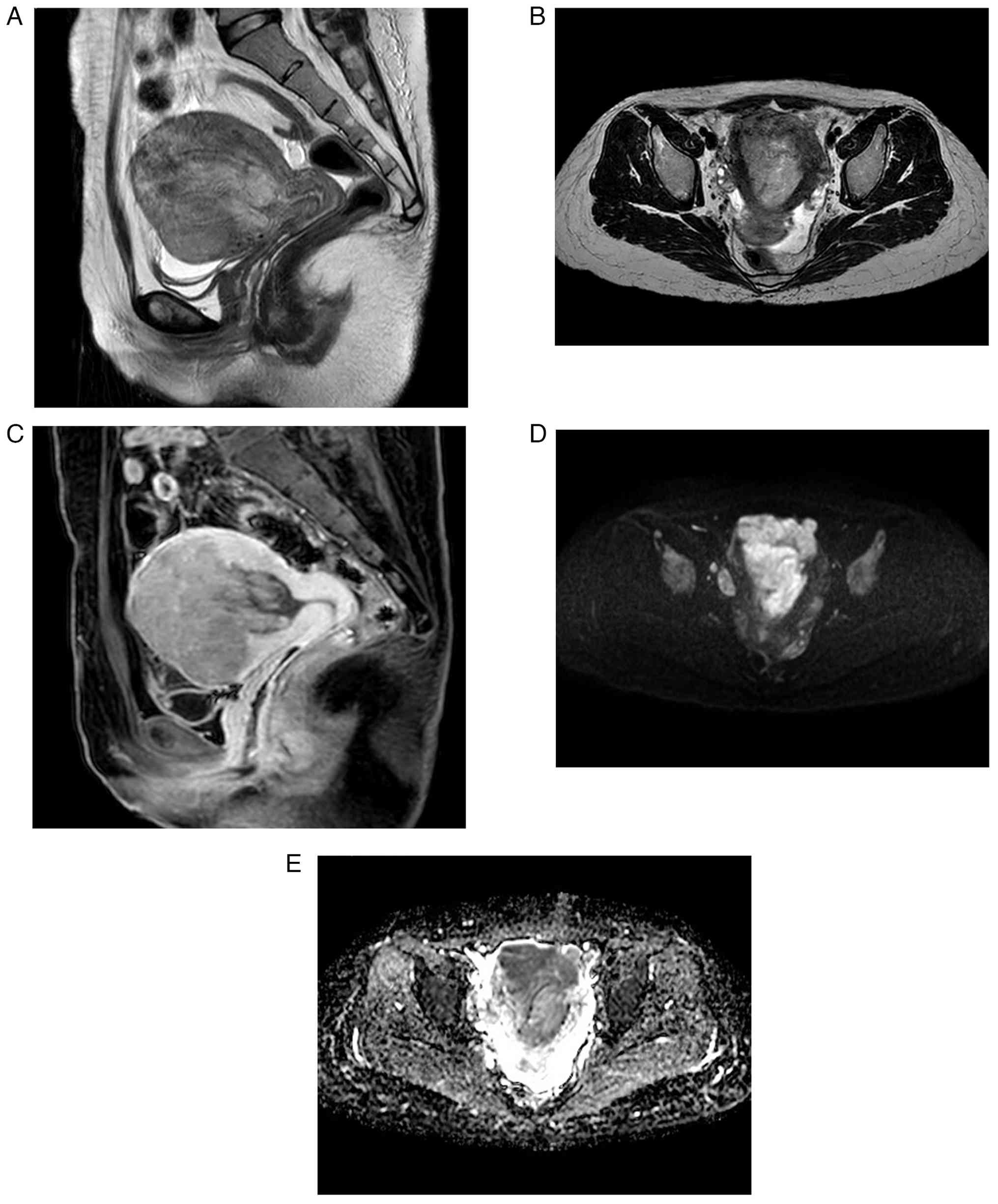
Pelvic magnetic resonance imaging revealed an
irregular mass with mild contrast enhancement and restricted
diffusion. This protruded from the myometrium into the uterine
cavity (Fig. 1A-E). As both
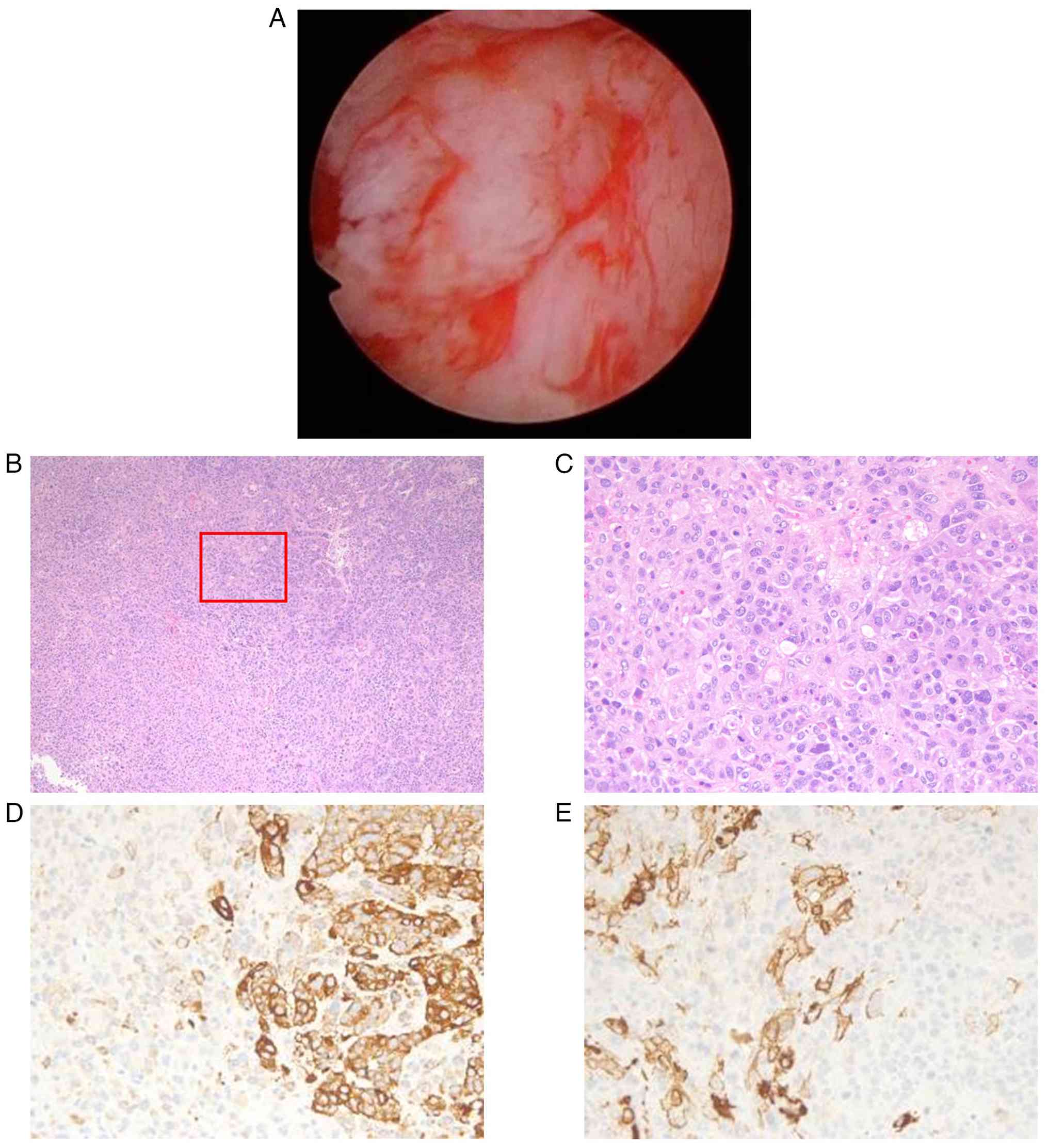
cervical and endometrial cytology were negative, a hysteroscopic
biopsy of the tumor was performed. The tumor was white, irregular,
and showed neovascularization (Fig.
2A). Endometrial biopsy revealed densely proliferating
epithelioid cells with nuclear atypia and prominent mitoses,
consistent with poorly differentiated carcinoma. However, atypical
polygonal cells were also present. Immunohistochemistry showed
reduced epithelial marker expression and weak positivity for smooth
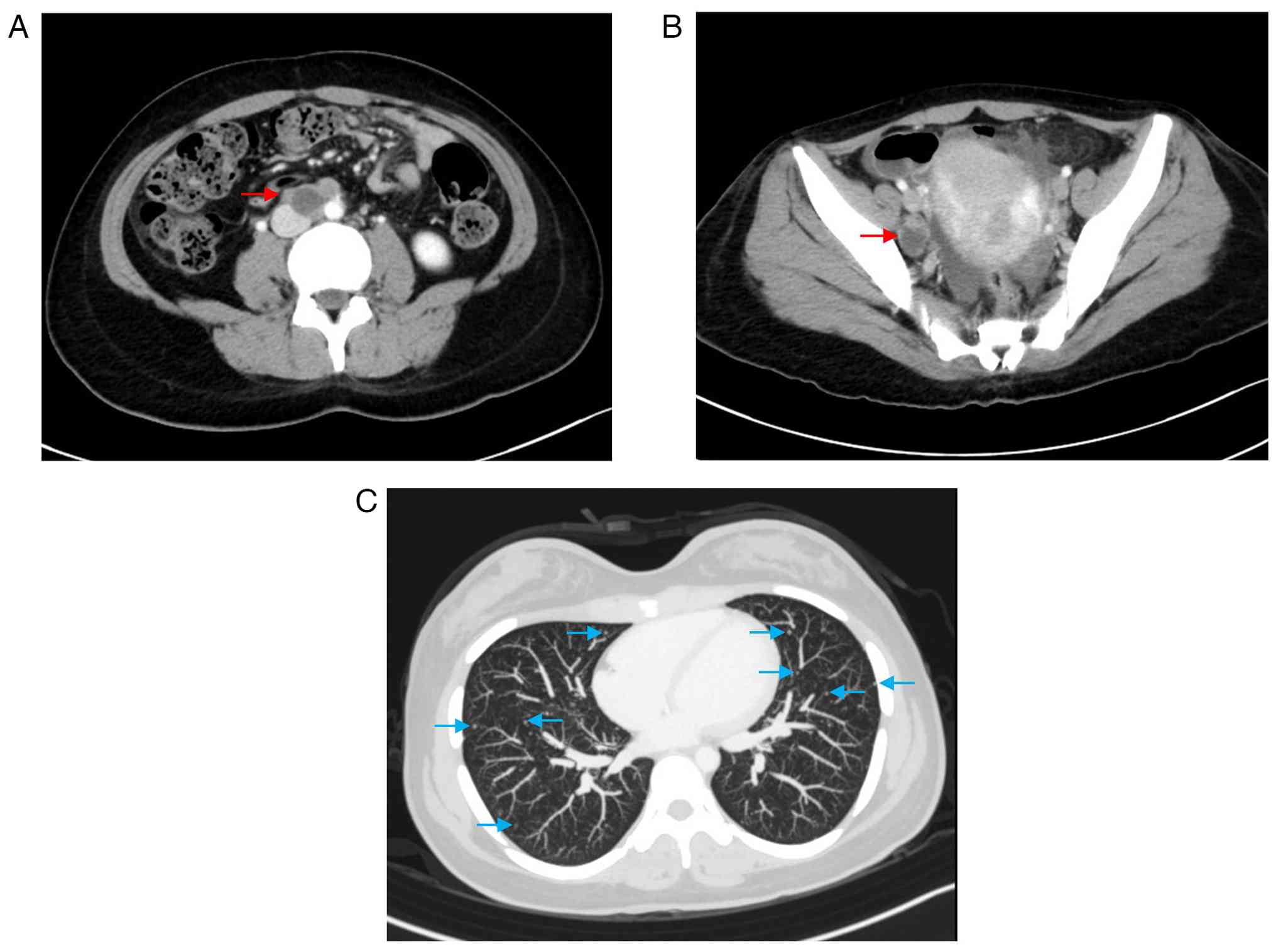
muscle markers, raising the possibility of carcinosarcoma (Fig. 2B-E). Computed tomography (CT)
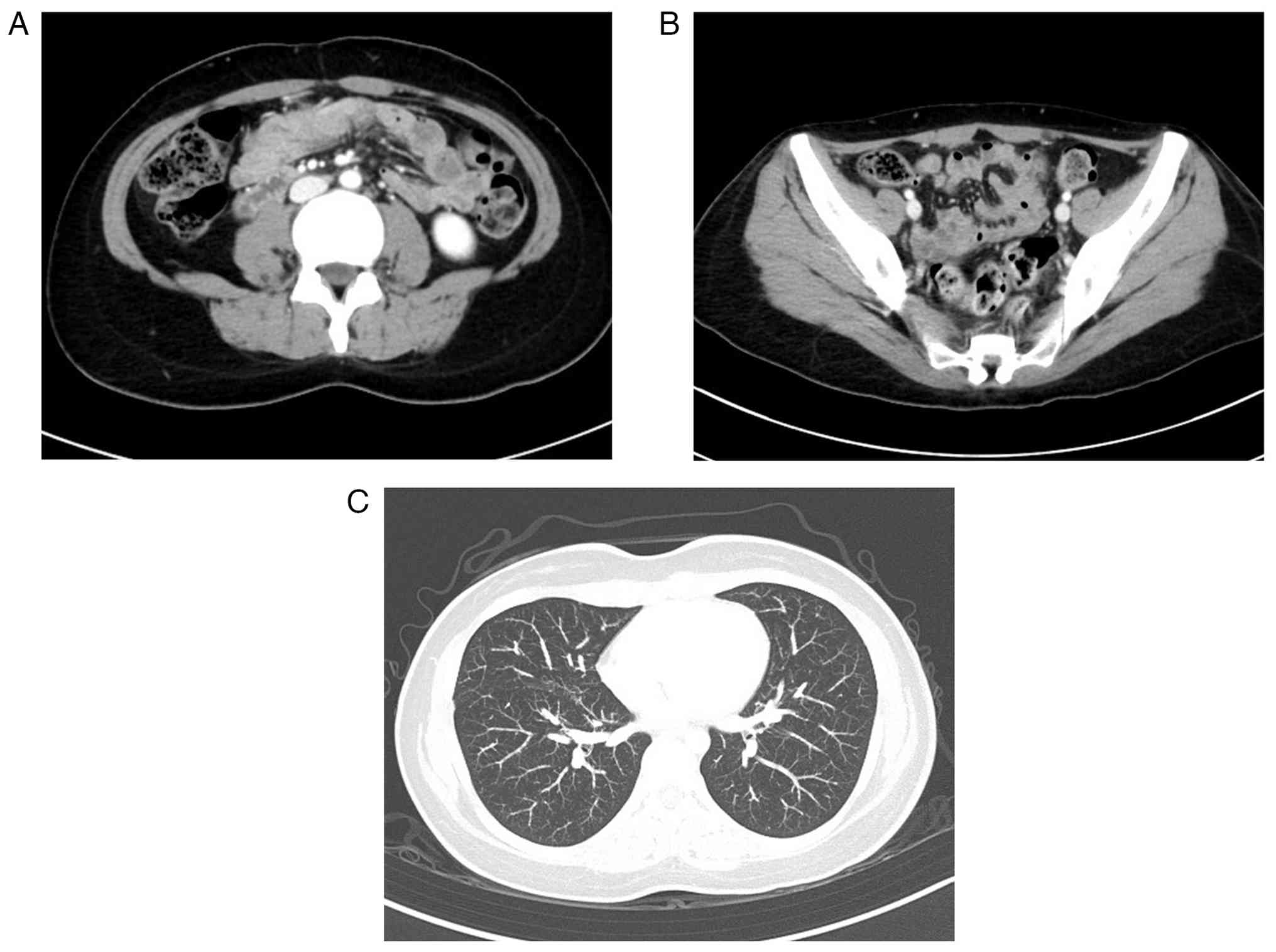
revealed para-aortic and pelvic lymph node metastasis and multiple
pulmonary metastases (Fig. 3A-C).
The patient underwent total hysterectomy with bilateral
salpingo-oophorectomy to control genital bleeding and obtain a
definitive diagnosis.
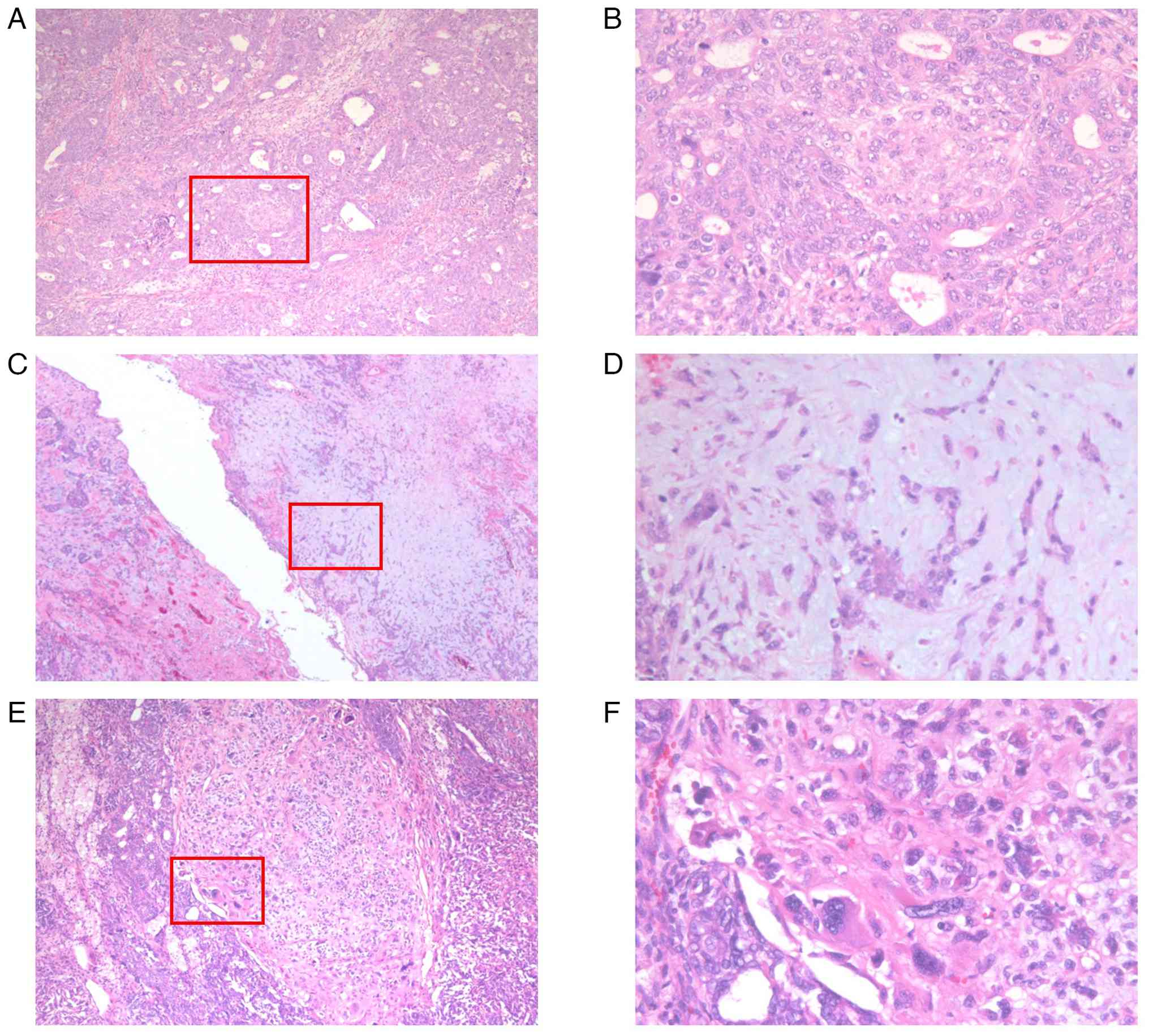
Histopathological examination revealed a mixture of
poorly differentiated endometrioid carcinoma and heterologous
sarcomatous components with chondrosarcomatous and
rhabdomyosarcomatous differentiation (Fig. 4A-F). This led to a diagnosis of UCS
at FIGO stage IVB. The patient underwent comprehensive genomic
profiling with FoundationOne® CDx (Foundation Medicine,
Cambridge, MA, USA), a hybrid-capture next-generation sequencing
(NGS) assay that interrogate 324 cancer-related genes, including
POLE, and selected introns to detect base substitutions,
insertions/deletions, copy-number alterations, and gene
rearrangements. The report also included tumor mutational burden
(TMB), microsatellite (MS) status, and homologous recombination
deficiency (HRD) signature. Results showed a BRCA1 V923fs*76
mutation [variant allele frequency (VAF): 81.5%], a TP53
splice-site mutation (920-2A>G; VAF: 62.8%), and a PIK3CA
Q546K mutation (VAF: 10.2%). The tumor was HRD signature-positive,
MS-stable, and had a TMB of 2 mutations per megabase (2 Muts/Mb).
Germline genetic testing for BRCA1 and TP53 was
performed after genetic counseling, and a germline mutation was
identified in BRCA1.
The patient was treated with four cycles of
paclitaxel and carboplatin chemotherapy. Subsequently, durvalumab
and olaparib were approved for reimbursement under the Japanese
national health insurance system for the treatments of endometrial
cancer. Therefore, the patient received an additional four cycles
of combination chemotherapy with paclitaxel, carboplatin, and
durvalumab. Post-chemotherapy CT showed a complete response
according to RECIST criteria (Fig.
5A-C). Immunohistochemistry confirmed mismatch repair, and
maintenance therapy with durvalumab and olaparib was initiated. The
patient has remained progression-free for 10 months.
Discussion
Carcinosarcoma is unusual in young women, with only
sporadic cases reported (7–9), and its occurrence within the HBOC
spectrum is exceptionally rare and poorly defined. Prior
young-onset reports have lacked broad molecular interrogation: Al
Dallal et al (7) assessed
p53 and mismatch-repair proteins by immunohistochemistry without
comprehensive genomic testing, whereas Soror et al (8) applied a 34-gene NGS panel that
included BRCA1/2 and found no pathogenic BRCA1/2
variants. Given the very limited number of young-onset cases
reported in the literature, it remains difficult to discuss whether
or not differences between races and ethnicities may play a role in
disease presentation in this age group. In this regard, our
21-year-old patient underwent comprehensive gene testing that
identified a germline BRCA1 frameshift mutation and HRD
positivity, establishing HBOC and enabling prediction of
sensitivity to platinum chemotherapy and PARP inhibition. This case
therefore supports heightened clinical vigilance and a low
threshold for early germline evaluation and tumor
profiling-particularly in young patients with suggestive personal
or family histories-and provides the rationale for the genomically
guided treatment strategy described below.
Advances in molecular profiling over the last decade
have transformed our understanding of UCS. Integrated genomic
analyses, most notably through The Cancer Genome Atlas (TCGA), have
characterized UCS as a metaplastic carcinoma arising from a single
epithelial clone, with sarcomatous components emerging through
epithelial-mesenchymal transition (5). TP53 mutations have been found
in around 90% of UCS cases. As in endometrioid and serous uterine
carcinomas, alterations are also often seen in PTEN, PIK3CA,
PPP2R1A, FBXW7, and KRAS (5). TCGA and the Proactive Molecular Risk
Classifier for Endometrial Cancer (ProMisE) have identified four
novel molecular endometrial cancer subgroups (10,11).
These are POLE/ultramutated (POLE),
microsatellite-instable/hypermutated (MSI),
copy-number-high/TP53-mutant (CNH), and
copy-number-low/TP53-wild-type (CNL). A recent meta-analysis of
four studies reported the pooled prevalence of the TCGA groups
among patients with UCS. These were 5.3% POLE, 7.3% MSI, 73.9% CNH,
and 13.5% CNL (12). Travaglino
et al (13) performed a
systematic review and meta-analysis including 263 uterine
carcinosarcoma (UCS) classified according TCGA groups. They found
that POLE-mutated tumors had an excellent prognosis with no
recurrences or deaths, MSI tumors showed intermediate outcomes,
while TP53-mutated tumors (CNH group) and no specific molecular
profile (NSMP) tumors (CNL group) were associated with poor
survival, paralleling the prognostic pattern observed in
endometrioid and serous endometrial carcinoma (13). Clinically, poorer outcomes in
TP53-abnormal endometrial cancer likely reflect enrichment for
advanced stage at presentation (14,15)
and relative insensitivity to limited adjuvant radiotherapy
(15); in addition, p53
overexpression has been linked to radioresistance and inferior
survival (15,16). Complementing these observations, a
large real-world series (n=2,235) showed tiered survival by
molecular class: TP53-mutated UCS had a shorter median overall
survival than TP53-mutated endometrioid cancer [27.9 vs. 35.3
months; hazard ratio (HR), 1.3; 95% confidence interval (CI),
1.1–1.5], and even TP53-wild-type UCS underperformed TP53-wild-type
endometrioid disease (29.4 vs. 70.7 months; HR, 2.0; 95% CI,
1.5–2.7) (17). Together, these
data indicate that while TP53 alterations define the
poorest-prognosis subgroup, ‘being UCS’ adds risk beyond TP53 class
alone.
Our patient's tumor harbored a TP53
splice-site mutation and a PIK3CA Q546K mutation, consistent
with the molecular hallmarks of UCS that have been identified in
large cohorts. To the best of our knowledge, no studies have yet
investigated the functional implications of TP53 splice-site
mutations and PIK3CA Q546K mutations in UCS. Furthermore, the
absence of POLE mutations and a microsatellite-stable status
led to its classification as the CNH subtype. Importantly, our
patient also carried a germline BRCA1 mutation, and the high
variant allele frequency of 81% strongly suggests biallelic
inactivation within the tumor, highlighting a potential driver
event in tumorigenesis. This finding is particularly notable
because, in a cohort of 167 UCS, Sia et al (18) reported germline mutations in 19
patients (11%); however, all affected individuals were aged 52–81
years (median, 61), and individual-level data were not available.
In contrast, our patient was diagnosed at the exceptionally young
age of 21, making this case one of the earliest-onset UCS
associated with a germline BRCA1 mutation reported to date.
In the Sia cohort, six out of 167 cases (3.6%) harbored germline
BRCA1/2 mutations, and the corresponding tumors exhibited
biallelic inactivation, indicating these alterations are likely
oncogenic drivers in UCS (18).
Consistently, in a series of endometrial cancers with germline
BRCA mutations, UCS comprised 28.5% of cases (6/21), further
supporting a link between BRCA-associated tumorigenesis and
this aggressive histologic subtype (19). Nevertheless, a lack of IHC is a
limitation of the present study.
Based on the results of the GOG-232B and GOG-261
trials, the recommended first-line treatment for advanced UCS is a
carboplatin/ paclitaxel doublet due to its non-inferiority and good
toxicity profile (20,21). However, the outcomes previously
reported in stage IV cases have been poor, with a median overall
survival time of just 8 months (22). Notably, tumors exhibiting HRD,
particularly those with BRCA mutations, have increased
sensitivity to platinum chemotherapy due to their impaired DNA
repair capabilities. Thus, our patient's complete radiologic
response following four cycles of paclitaxel/carboplatin may be
attributable to an underlying HRD-driven susceptibility (23). This suggests the potential utility
of HRD and BRCA testing of patients with UCS to predict
chemosensitivity.
Recent preclinical studies have elucidated the
mechanisms behind the synergistic effects of combined PARP
inhibitors (PARPi) and immune checkpoint inhibitors (ICIs), and
support the superiority of this approach over PARPi monotherapy.
PARP inhibition induces DNA double-strand breaks. The accumulation
of these breaks causes the activation of cyclic GMP-AMP synthase
(cGAS). This is a stimulator of interferon genes (STING) pathway
that enhances type I interferon signaling and dendritic cell
recruitment, promoting antitumor immunity (24). PARP inhibition also upregulates
PD-L1 expression via the inactivation of GSK3β, contributing to
immune evasion and creating a therapeutic window for ICIs (25). These immunomodulatory effects are
further amplified in HRD tumor models, where the combination of
PARPi and anti-PD-1/PD-L1 antibodies provides superior cytotoxic
T-cell infiltration and tumor regression compared to monotherapy
(26). Collectively, these findings
provide a strong biological rationale for combined PARPi-ICI
therapy in HRD tumors. Combination ICIs and PARPi are garnering
increased attention in gynecological oncology. The phase III DUOE
trial included patients with advanced or recurrent endometrial
cancer, including UCS. The trial found that adding durvalumab to
carboplatin and paclitaxel treatment, and following with
maintenance durvalumab and olaparib, significantly improved
progression-free survival (PFS). Specifically, the PFS hazard
ratios (HRs) were 0.71 (durvalumab arm) and 0.55 (durvalumab and
olaparib arm) versus chemotherapy alone (27). In a subgroup analysis of tumors
harboring homologous recombination repair mutations, the PFS HR was
0.30 in the durvalumab and olaparib arm compared to chemotherapy
alone (27). This provides
compelling grounds for applying this combination strategy to
HRD-positive UCS.
Compared with our case, Wan et al (28) reported a 58-year-old postmenopausal
UCS with bilateral benign ovarian Brenner tumors showing diffuse
mutant-type p53 on IHC; the report focuses on pathology and does
not include TCGA/ProMisE molecular stratification, HRD status,
germline testing, or systemic-therapy outcomes. By contrast, our
case demonstrates the successful application of targeted and
immunological therapy guided by precise molecular characterization
in a young patient with UCS. Comprehensive genomic profiling
(TP53 mutation, PIK3CA alteration, BRCA1
frameshift, and HRD positivity) informed a personalized treatment
plan combining platinum-based chemotherapy, immune checkpoint
blockade, and PARP inhibition. This resulted in complete and
durable remission. Our findings support the use of routine genomic
profiling, including HRD testing, in UCS. Clinical trials targeting
HRD-positive, MMR-proficient UCS are needed to further investigate
treatment strategies such as that described herein. Such strategies
may transform the prognosis for this aggressive and historically
intractable cancer subtype.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The raw next-generation sequencing data generated in
the present study may be found in the Japanese Genotype-Phenotype
Archive, hosted by the DNA Data Bank of Japan, under accession
number JGAS000883 (dataset: JGAD001027) or at the following URL:
https://ddbj.nig.ac.jp/search/entry/jga-dataset/JGAD001027.
All other data generated in the present study may be requested from
the corresponding author.
Authors' contributions
YI, AK, YS, TI, JM, MI and ST contributed to study
conception and design. YI and AK prepared the materials and
collected and analyzed the data. YI drafted the manuscript. All
authors reviewed and edited the manuscript. YI and ST confirm the
authenticity of all the raw data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
This case report was conducted in accordance with
the tenets of the 1964 Declaration of Helsinki and its later
revisions. This case report was approved by the Ethics Committee of
Dokkyo Medical University Saitama Medical Center (approval no.
25094; Koshigaya, Japan).
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of their images and data.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
FIGO
|
International Federation of Gynecology
and Obstetrics
|
|
HBOC
|
hereditary breast and ovarian cancer
syndrome
|
|
HRD
|
homologous recombination
deficiency
|
|
MMR
|
mismatch repair
|
|
PARP
|
poly (ADP-ribose) polymerase
|
|
UCS
|
uterine carcinosarcoma
|
References
|
1
|
de Jonge MM, de Kroon CD, Jenner DJ,
Oosting J, de Hullu JA, Mourits MJE, Gómez Garcia EB, Ausems M,
Margriet Collée J, van Engelen K, et al: Endometrial cancer risk in
women with germline BRCA1 or BRCA2 mutations: Multicenter cohort
study. J Natl Cancer Inst. 113:1203–1211. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shu CA, Pike MC, Jotwani AR, Friebel TM,
Soslow RA, Levine DA, Nathanson KL, Konner JA, Arnold AG,
Bogomolniy F, et al: Uterine cancer after risk-reducing
salpingo-oophorectomy without hysterectomy in women with BRCA
mutations. JAMA Oncol. 2:1434–1440. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Laitman Y, Michaelson-Cohen R, Levi E,
Chen-Shtoyerman R, Reish O, Josefsberg Ben-Yehoshua S,
Bernstein-Molho R, Keinan-Boker L, Rosengarten O, Silverman BG, et
al: Uterine cancer in Jewish Israeli BRCA1/2 mutation carriers.
Cancer. 125:698–703. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nahshon C, Segev Y, Gemer O, Bar Noy T,
Schmidt M, Ostrovsky L and Lavie O: Should the risk for uterine
cancer influence decision making for prophylactic hysterectomy in
BRCA1/2 mutated patients- A systematic review and meta-analysis.
Gynecol Oncol. 160:755–762. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cherniack AD, Shen H, Walter V, Stewart C,
Murray BA, Bowlby R, Hu X, Ling S, Soslow RA, Broaddus RR, et al:
Integrated molecular characterization of uterine carcinosarcoma.
Cancer Cell. 31:411–423. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bogani G, Ray-Coquard I, Concin N, Ngoi
NYL, Morice P, Caruso G, Enomoto T, Takehara K, Denys H, Lorusso D,
et al: Endometrial carcinosarcoma. Int J Gynecol Cancer.
33:147–174. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Al Dallal HA, Jacobs TH, Bergman CL,
Narayanan S, Kaur A, Al-Quran SZ and Chopra HK: Uterine
carcinosarcoma with heterologous osseous elements: A case report of
an extremely rare clinical occurrence with literature review. Front
Oncol. 15:15055042025. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Soror NN, Woredekal D, Hemrock L, Gibson G
and Bennett R: Uterine carcinosarcoma in a young female: Case
report and literature review. Cureus. 13:e126422021.PubMed/NCBI
|
|
9
|
Al-Badawi IA, AlOmar O and Kornfeld I:
Carcinosarcoma of the uterus in a woman with down syndrome. Hematol
Oncol Stem Cell Ther. 3:161–162. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kandoth C, Schultz N, Cherniack AD, Akbani
R, Liu Y, Shen H, Robertson AG, Pashtan I, Shen R, Benz CC, et al:
Integrated genomic characterization of endometrial carcinoma.
Nature. 497:67–73. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Talhouk A, McConechy MK, Leung S, Li-Chang
HH, Kwon JS, Melnyk N, Yang W, Senz J, Boyd N, Karnezis AN, et al:
A clinically applicable molecular-based classification for
endometrial cancers. Br J Cancer. 113:299–310. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Travaglino A, Raffone A, Gencarelli A,
Mollo A, Guida M, Insabato L, Santoro A, Zannoni GF and Zullo F:
TCGA classification of endometrial cancer: The place of
carcinosarcoma. Pathol Oncol Res. 26:2067–2073. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Travaglino A, Raffone A, Raimondo D,
Arciuolo D, Angelico G, Valente M, Scaglione G, D'Alessandris N,
Casadio P, Inzani F, et al: Prognostic value of the TCGA molecular
classification in uterine carcinosarcoma. Int J Gynaecol Obstet.
158:13–20. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hong JH, Cho HW, Ouh YT, Lee JK, Chun Y
and Gim JA: Genomic landscape of advanced endometrial cancer
analyzed by targeted next-generation sequencing and the cancer
genome atlas (TCGA) dataset. J Gynecol Oncol. 33:e292022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Horeweg N, Nout RA, Jürgenliemk-Schulz IM,
Lutgens L, Jobsen JJ, Haverkort MAD, Mens JWM, Slot A, Wortman BG,
de Boer SM, et al: Molecular classification predicts response to
radiotherapy in the randomized PORTEC-1 and PORTEC-2 trials for
early-stage endometrioid endometrial cancer. J Clin Oncol.
41:4369–4380. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Akiyama A, Minaguchi T, Fujieda K,
Hosokawa Y, Nishida K, Shikama A, Tasaka N, Sakurai M, Ochi H and
Satoh T: Abnormal accumulation of p53 predicts radioresistance
leading to poor survival in patients with endometrial carcinoma.
Oncol Lett. 18:5952–5958. 2019.PubMed/NCBI
|
|
17
|
Toboni MD, Dinkins K, Wu S, Mattox T,
Oberley MJ, Thaker PH, Herzog TJ, Powell MA and Jones N: Not all
uterine carcinosarcomas are created equal: Survival outcomes
according to molecular characterization of uterine carcinosarcoma.
Gynecol Oncol. 193:89–97. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sia TY, Gordhandas SB, Birsoy O, Kemel Y,
Maio A, Salo-Mullen E, Sheehan M, Hensley ML, Rubinstein M, Makker
V, et al: Germline drivers of gynecologic carcinosarcomas. Gynecol
Oncol. 174:34–41. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gordhandas S, Rios-Doria E, Cadoo KA,
Catchings A, Maio A, Kemel Y, Sheehan M, Ranganathan M, Green D,
Aryamvally A, et al: Comprehensive analysis of germline drivers in
endometrial cancer. J Natl Cancer Inst. 115:560–569. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Powell MA, Filiaci VL, Hensley ML, Huang
HQ, Moore KN, Tewari KS, Copeland LJ, Secord AA, Mutch DG, Santin
A, et al: Randomized phase III trial of paclitaxel and carboplatin
versus paclitaxel and ifosfamide in patients with carcinosarcoma of
the uterus or ovary: An NRG oncology trial. J Clin Oncol.
40:968–977. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Powell MA, Filiaci VL, Rose PG, Mannel RS,
Hanjani P, Degeest K, Miller BE, Susumu N and Ueland FR: Phase II
evaluation of paclitaxel and carboplatin in the treatment of
carcinosarcoma of the uterus: A gynecologic oncology group study. J
Clin Oncol. 28:2727–2731. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Matsuzaki S, Klar M, Matsuzaki S, Roman
LD, Sood AK and Matsuo K: Uterine carcinosarcoma: Contemporary
clinical summary, molecular updates, and future research
opportunity. Gynecol Oncol. 160:586–601. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maxwell KN, Wubbenhorst B, Wenz BM, De
Sloover D, Pluta J, Emery L, Barrett A, Kraya AA, Anastopoulos IN,
Yu S, et al: BRCA locus-specific loss of heterozygosity in germline
BRCA1 and BRCA2 carriers. Nat Commun. 8:3192017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pantelidou C, Sonzogni O, De Oliveria
Taveira M, Mehta AK, Kothari A, Wang D, Visal T, Li MK, Pinto J,
Castrillon JA, et al: PARP inhibitor efficacy depends on CD8(+)
T-cell recruitment via intratumoral STING pathway activation in
BRCA-deficient models of triple-negative breast cancer. Cancer
Discov. 9:722–737. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiao S, Xia W, Yamaguchi H, Wei Y, Chen
MK, Hsu JM, Hsu JL, Yu WH, Du Y, Lee HH, et al: PARP inhibitor
upregulates PD-L1 expression and enhances cancer-associated
immunosuppression. Clin Cancer Res. 23:3711–3720. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ding L, Kim HJ, Wang Q, Kearns M, Jiang T,
Ohlson CE, Li BB, Xie S, Liu JF, Stover EH, et al: PARP inhibition
elicits STING-dependent antitumor immunity in Brca1-deficient
ovarian cancer. Cell Rep. 25:2972–2980.e2975. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Westin SN, Moore K, Chon HS, Lee JY,
Thomes Pepin J, Sundborg M, Shai A, de la Garza J, Nishio S, Gold
MA, et al: Durvalumab plus carboplatin/paclitaxel followed by
maintenance durvalumab with or without olaparib as first-line
treatment for advanced endometrial cancer: The phase III DUO-E
trial. J Clin Oncol. 42:283–299. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wan X, Bi F, Xin B and Qiao C: A rare case
report of uterine carcinosarcoma with bilateral ovarian brenner
tumors. Front Oncol. 15:16127162025. View Article : Google Scholar : PubMed/NCBI
|