Introduction
Acute lymphoblastic leukemia (ALL) is a severe
hematological malignancy characterized by impaired lymphocyte
differentiation, which causes the proliferation of leukemic blasts
in the bone marrow, peripheral blood and extramedullary tissues
(1,2). ALL primarily affects children, with an
incidence peaking between the ages of 1 and 4. The disease accounts
for ~20% of adult leukemias, and ~60% of patients with ALL are
diagnosed before the age of 20 (3–5).
According to immunophenotypic classification, B-cell acute
lymphoblastic leukemia (B-ALL) is the most common type, accounting
for ~75% of cases (6).
High-risk B-ALL primarily comprises
relapsed/refractory (R/R) cases and Philadelphia
chromosome-positive (Ph+) cases. In R/R B-ALL,
conventional chemotherapy has limited efficacy, low complete
remission (CR) rates and high toxicity. For example, in pediatric
patients receiving conventional four-drug regimens, the incidence
of life-threatening infections reaches 39.8%, and treatment-related
mortality is 5%, while long-term survival in adult patients is only
20–40% (7,8). Ph+ B-ALL accounts for
22–31% of adult ALL cases. Standard treatment regimens include
intensive chemotherapy combined with tyrosine kinase inhibitors
(TKIs). However, mutations in the ABL1 kinase domain (e.g.,
Thr315Ile) often lead to TKI resistance, significantly shortening
post-relapse survival (9,10). Furthermore, elderly patients have a
worse prognosis due to a poor tolerance to high-intensity
chemotherapy. Analysis of the Surveillance, Epidemiology and End
Results database (https://seer.cancer.gov/) shows that the median
overall survival (mOS) time in this population is only 7–8 months.
The disease in this group often exhibits aggressive biological
characteristics, including complex karyotypes and TP53 mutations,
which complicate treatment (11,12).
Over the past decade, targeted treatments utilizing
B-cell targeted drugs and TKIs have demonstrated significant
clinical efficacy in B-ALL (13,14).
The anti-CD22 antibody-drug conjugate inotuzumab ozogamicin (INO)
exhibits potent anti-leukemic activity and lower myelosuppression
compared with conventional chemotherapy (15–18).
Additionally, the bispecific T-cell engager blinatumomab has shown
promising results in relapsed or refractory B-ALL cases (19).
Blinatumomab is a bispecific T-cell engager antibody
that simultaneously binds CD19 on B cells and CD3 on T cells. This
dual binding activates T cells and redirects their cytotoxic
activity against CD19-positive tumor cells, leading to targeted
cell lysis (20–22). CD19 is uniformly expressed on
malignant B cells but absent on hematopoietic stem cells (HSCs),
making it an optimal therapeutic target for B-ALL. Due to this
mechanism, blinatumomab has demonstrated clinical efficacy in R/R
B-ALL and received U.S. Food and Drug Administration approval for
this indication in 2014 (16,20,23,24).
In a phase 2 trial of adults with newly diagnosed Ph+
B-ALL, dasatinib and blinatumomab yielded a 98% CR rate. At the
18-month median follow-up, the OS rate was 95% and the disease-free
survival (DFS) rate was 88% (25).
Blinatumomab has been studied in combination with
other therapies. When added to low-intensity chemotherapy
[mini-hyperfractionated cyclophosphamide, vincristine, and
dexamethasone (mini-Hyper-CVD)] plus INO, blinatumomab prolonged
the interval between inotuzumab administration and HSC
transplantation (HSCT). This approach may decrease the risk of
veno-occlusive disease/sinusoidal obstruction syndrome.
Importantly, no cytokine release syndrome (CRS) or neurotoxicity
was reported with this regimen (26). In patients with Ph+
B-ALL, the chemotherapy-free combination of blinatumomab and
ponatinib has shown promising results. This regimen can induce
sustained deep remissions, potentially eliminating the need for
allogeneic HSCT in some patients (27).
Despite advances with targeted therapies (such as
the third-generation TKI ponatinib) and immunotherapies (including
bispecific antibody blinatumomab and antibody-drug conjugate INO),
single-agent treatment has yielded limited response rates and
duration (19). For example,
blinatumomab monotherapy achieves only a 36% overall response rate
and 7.1-month mOS time in R/R Ph+ B-ALL (28). Therefore, investigating combination
regimens to enhance the depth of response, reduce treatment
toxicity and address drug resistance has become a key direction for
improving the prognosis of patients with high-risk B-ALL.
Given these factors and the severity of the
condition, a meta-analysis of clinical trials on high-risk B-ALL
was conducted. The present study aimed to evaluate the efficacy and
safety of blinatumomab-based combination therapy in patients with
high-risk B-ALL.
Methods
Study registration
The present systematic review and meta-analysis was
conducted according to the Preferred Reporting Items for Systematic
Reviews and Meta-Analyses statement (29). The study protocol was prospectively
registered in the International Prospective Register of Systematic
Reviews database (https://www.crd.york.ac.uk/prospero/), with
registration number CRD420251108271.
Search strategy
A search of the PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase (https://www.embase.com/), Web of Science (https://www.webofscience.com/), and the Cochrane
Library (https://www.cochranelibrary.com/) databases was
performed from inception to July 20, 2025. To ensure comprehensive
coverage, no language restrictions were applied to the initial
database search. The search strategy combined Medical Subject
Headings terms and free-text words, including: ‘blinatumomab’ AND
(‘precursor cell lymphoblastic leukemia-lymphoma’ OR ‘acute
lymphoblastic leukemia’ OR ‘acute lymphoid leukemia’ OR ‘leukemia,
lymphoblastic, acute’). Additionally, reference lists of included
studies were reviewed to identify further relevant studies.
Inclusion and exclusion criteria
Inclusion criteria
Eligible studies could be either prospective
(covering randomized controlled trials and single-arm trials) or
retrospective in design. The intervention under investigation
needed to be blinatumomab in combination with other therapies. The
target study population had to consist of patients with high-risk
B-ALL. Lastly, only English-language studies were considered
eligible for inclusion.
Exclusion criteria
Publication types such as reviews, conference
abstracts, letters, guidelines, case reports, as well as cellular
or animal experiments were excluded. Additionally, studies with
unavailable full texts were excluded. Duplicate studies, including
those involving secondary analyses, were also excluded.
Furthermore, studies of very low quality or with severe design
flaws, as evaluated by the Newcastle-Ottawa Scale (NOS) (30) or Methodological Index for
Non-Randomized Studies (MINORS) (31), were excluded from the study.
Two researchers independently screened articles to
evaluate their eligibility based on predefined inclusion and
exclusion criteria. Discrepancies in study selection were resolved
through discussions between the two researchers. If no consensus
was reached, a third researcher was consulted for further
resolution.
Data extraction and quality
assessment
Two researchers independently extracted data from
included studies and evaluated their quality. In the present study,
CR was defined as bone marrow leukemic blasts ≤5% with or without
complete blood cell recovery. Minimal residual disease (MRD)
response was defined as a negative MRD status, with bone marrow
leukemic blasts <0.01%. Complete molecular remission (CMR) was
defined as sustained negative MRD, specifically bone marrow
leukemic blasts <0.01% confirmed by sensitive testing at time
points defined by individual study protocols. The Common
Terminology Criteria for Adverse Events (CTCAE) version 5 (32) was used to categorize adverse events
(AEs). Other outcomes assessed were 1-, 2- and 3-year OS rates.
Statistical analysis
The present meta-analysis used Stata 14.0 software
to conduct the data analysis (StataCorp LP). Heterogeneity among
studies was assessed using Cochran's Q test and I2
statistic. Given the inherent clinical and methodological diversity
among the included studies, a random-effects model was employed for
all meta-analyses to account for potential between-study
heterogeneity, in accordance with the Cochrane Handbook
recommendations (33). Sensitivity
analysis was performed to assess the robustness of the results. The
test of potential publication bias was performed using Begg's test
and Egger's test. A two-sided P<0.05 was considered
statistically significant. Data were presented as pooled rates (ES)
with 95% confidence intervals (CIs).
Results
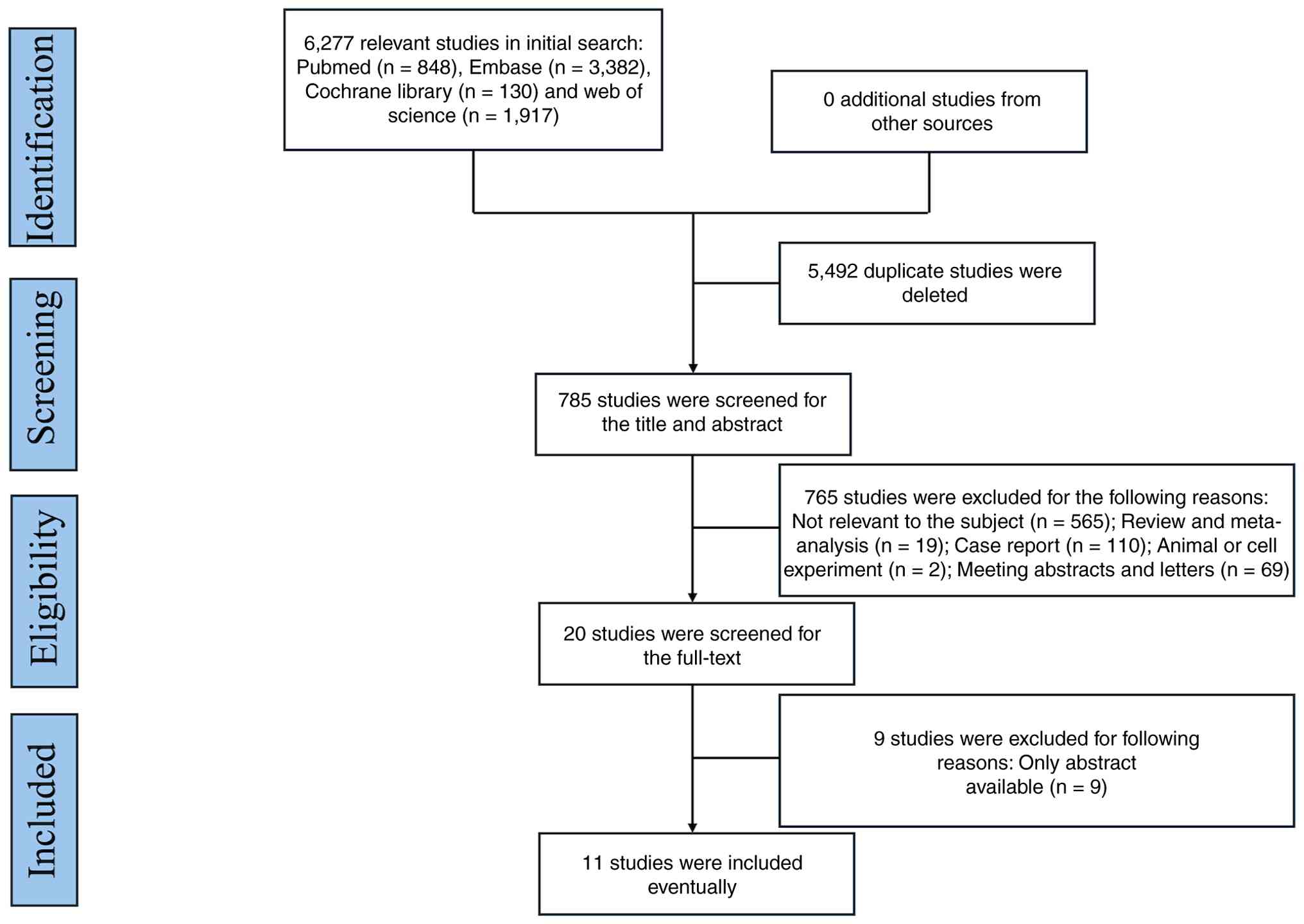
Study selection
An initial search of four databases yielded 6,277
relevant studies: 848 studies in PubMed, 3,382 in Embase, 130 in
the Cochrane Library and 1,917 in Web of Science. After potential
duplicates were eliminated, titles and abstracts were screened and
a total of 20 studies were selected for full-text review. A
subsequent full-text evaluation led to the exclusion of 9 studies
due to the unavailability of full texts. Ultimately, 11 studies
(19,25,34–42)
involving 402 patients met the inclusion criteria and were included
in this meta-analysis. The study selection process is shown in
Fig. 1, and detailed information on
each included study is provided in Table I.
 | Table I.Characteristics of the studies
included in the meta-analysis. |
Table I.
Characteristics of the studies
included in the meta-analysis.
| First author,
year | Country | Study design | Phase | Median age (range),
years | Treatment 1 | Treatment 2 | Blinatumomab group,
n | Safety analysis,
n | Ph+, %
(n/total n) | Prior HSCT, %
(n/total n) | Dose of
Blinatumomab | Median follow-up
time, months | Treatment
Setting | (Refs.) |
|---|
| Jabbour et
al, 2023 | USA | Prospective
study | II | 68 (60–87) | Blinatumomab +
mini-Hyper-CVD + fractionated low-dose INO | mini-Hyper-CVD +
standard-dose INO | 31 | 31 | - | - | Blinatumomab: 9
µg/day for first 4 days, then 28 µg/day for 4 weeks per cycle
(total 4 cycles) | 29.7(8.8–41.0) | First-line | (19) |
| Foà et al,
2020 | Italy | Prospective
study | II | 54 (24–82) | Blinatumomab +
Dasatinib | - | 63 | 60 | 100 (63/63) | 0 (0/63) | Blinatumomab: 28
µg/day for 4 weeks per cycle (minimum 2 cycles) | 18.0
(1.0–25.0) | First-line | (25) |
| Sokolov et
al, 2017 | Russia | Retrospective
study | - | 32 (24–49) | Blinatumomab +
TKI | - | 11 | 11 | 73 (8/11) | - | Blinatumomab: 9
mcg/day in first week of first cycle, then 28 mcg/day for
subsequent three weeks; subsequent cycles of 28 mcg/day for 4
weeks | 12.0
(3.0–19.0) | R/R | (34) |
| Assi et al,
2017 | USA | Retrospective
study | - | 65 (30–77) | Blinatumomab +
TKI | - | 9 | 9 | 100 (9/9) | - | Blinatumomab: 9
mcg/day in first week of first cycle, then 28 mcg/day by continuous
infusion for 3 weeks; second cycle and onwards at 28 mcg/day for 4
weeks, repeated every 6 weeks | 8.0 (2.0–14.0) | R/R | (35) |
| Couturier et
al, 2021 | France | Retrospective
study | - | 58 (18–81) | Blinatumomab +
ponatinib | - | 26 | 26 | 100 (26/26) | 34.6 (9/26) | Blinatumomab: 9
mg/day for first week of cycle 1, then escalated to 28 mg/day, and
28 mg/day for subsequent cycles | 34.4 58.7)
−58.7) | R/R | (36) |
| Gibson et
al, 2024 | USA | Retrospective
study | - | 8.5 (2–17) | Blinatumomab +
Mini-Hyper-CVD | - | 9 | 5 | - | 0 (0/5) | Blinatumomab and
rituximab: Median, 1 cycle (1–2 cycles) | 17.1
(4.8–39.4) | R/R | (37) |
| Hogan et al,
2023 | USA | Prospective
study | III | 11 (1–30) | Blinatumomab +
Chemotherapy | Chemotherapy | 127 | 121 | - | 0 (0/255) | Blinatumomab: 15
µg/m2/day for 28 days per cycle (3 cycles) | 3.5 (0.1–6.6) | R/R | (38) |
| Jabbour et
al, 2023 | USA | Prospective
study | II | 51 (36–68) | Blinatumomab +
ponatinib | - | 54 | 54 | 100 (54/54) | - | Blinatumomab: 9 µg
per day on days 1–4 of cycle one, then 28 µg per day on days 5–28,
28 µg per day for cycles two to five (28 days each cycle), in up to
five 42-day cycles | 16.0
(11.0–24.0) | First-line | (39) |
| Kantarjian et
al, 2023 | USA | Prospective
study | II | 37 (17–87) | Blinatumomab +
Mini-Hyper-CVD + fractionated low-dose inotuzumab | Mini-Hyper-CVD +
standard-dose inotuzumab | 43 | 43 | - | 5 (2/43) | Blinatumomab: 9
µg/day for first 4 days of course 1, then 28 µg/day thereafter, and
28 µg/day for subsequent courses | 48.0
(9.0–115.0) | R/R | (40) |
| King et al,
2019 | USA | Retrospective
study | - | 61.2 (27–72.1) | Blinatumomab +
TKI | - | 11 | 11 | 100 (11/11) | 27 (3/11) | Blinatumomab: 9
mcg/day in first week of cycle 1, then 28 mcg/day for 3 weeks;
subsequent cycles of 28 mcg/day for 4 weeks. MRD dosing: 28 mcg/day
for 4 weeks, repeated every 6 weeks (1 patient transitioned to
conventional dosing) | 10.8
(3.5–20.0) | R/R | (41) |
| Stolz et al,
2025 | Switzerland | Retrospective
study | - | 65(18+) | Blinatumomab +
TKI | TKI +
chemotherapy | 18 | 18 | 100 (18/18) | - | Blinatumomab: 28
µg/day for 28 days per cycle (2–6 cycles) | 24.0
(2.0–100.0) | R/R | (42) |
Quality assessment
Quality assessment of 4 cohort studies (comprising
prospective and retrospective designs) was performed using the NOS
(30). The scale evaluates three
domains (selection of study groups, comparability of groups and
outcome/exposure determination for cohort/case-control studies)
based on eight specific criteria, awarding a maximum of 9 stars.
Studies with a score of ≥7 were considered high quality. For 7
single-arm studies, the MINORS tool (31) was applied. This index scores 8 items
from 0 (not reported) to 2 (reported and adequate), giving a
maximum total score of 16 for non-comparative studies, with higher
scores indicating better methodological quality. Detailed results
are also shown in Table II.
 | Table II.Quality assessment of the studies
included in the meta-analysis. |
Table II.
Quality assessment of the studies
included in the meta-analysis.
| A, NOS for
prospective and retrospective cohort studies |
|---|
|
|---|
| First author,
year | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Total | (Refs.) |
|---|
| Kantarjian et
al, 2023 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 9 | (40) |
| Jabbour et
al, 2023 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 9 | (19) |
| Stolz et al,
2025 | 0.5 | 1 | 1 | 1 | 0.5 | 0.5 | 2 | 1 | 7.5 | (42) |
| Hogan et al,
2023 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 9 | (38) |
|
| B, MINORS for
single-arm studies |
|
| First author,
year | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Total | (Refs.) |
|
| Gibson et
al, 2024 | 2 | 2 | 1 | 2 | 2 | 2 | 1 | 1 | 13 | (37) |
| Couturier et
al, 2021 | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 1 | 14 | (36) |
| Jabbour et
al, 2023 | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 1 | 16 | (39) |
| Foà et al,
2020 | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 1 | 16 | (25) |
| King et al,
2019 | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 1 | 14 | (41) |
| Assi et al,
2017 | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 1 | 14 | (35) |
| Sokolov et
al, 2017 | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 1 | 14 | (34) |
NOS for prospective and retrospective
cohort studies
The numerical labels Q1-Q8 in the Table II headers for the scoring system
correspond to the following criteria: i) Representativeness of the
exposed cohort; ii) representativeness of the non-exposed cohort;
iii) selection of the exposed cohort; iv) absence of the outcome of
interest at baseline; v) comparability of cohorts in design or
analysis; vi) assessment of outcomes; vii) sufficiency of follow-up
duration for outcome occurrence; and 8) adequacy of cohort
follow-up completion.
MINORS for single-arm studies
The numerical labels Q1-Q8 in the Table II header for the scoring system
correspond to the following criteria: i) Clear statement of
research objective; ii) consistency of patient inclusion; iii)
prospective data collection; iv) appropriateness of outcome
indicators to reflect research objective; v) objectivity of outcome
indicator evaluation; vi) sufficiency of follow-up duration; vii)
follow-up loss rate <5%; and viii) sample size estimation.
Efficacy
CR
A total of 10 studies reported the CR rate of
blinatumomab-based combination therapy for treating high-risk
B-ALL. High heterogeneity was observed among these studies
(I2=77.0%; P<0.001). Using a random-effects model,
the meta-analysis showed a pooled CR rate of 87% [95% confidence
interval (CI), 78–95%] (Fig.
2A).
 | Figure 2.Forest plot of the pooled CR rate.
(A) Forest plot of all the studies. (B-F) Forest plots for CR rates
based on (B) study types (prospective studies and retrospective
studies), (C) different age groups (<18 and >18 years old),
(D) different combination therapy regimens (plus TKI and plus
chemotherapy), (E) different Ph-positive expression rates (all
Ph+ and non-all Ph+) and (F) different
treatment settings (first-line and R/R). CR, complete remission;
Ph+, Philadelphia chromosome-positive; TKI, tyrosine
kinase inhibitor; R/R, relapsed/refractory; CI, confidence
interval; ES, effect size; DL, DerSimonian-Laird. |
Subgroup analyses were conducted based on study type
(prospective vs. retrospective), age group (<18 vs. >18
years), combination therapy regimen (plus TKI vs. plus
chemotherapy), stratification by Philadelphia chromosome status
(all-Ph+ cohort vs. non-all Ph+ cohort) and
treatment setting (first-line vs. R/R). The latter was
pre-specified given the distinct prognosis of newly diagnosed vs.
R/R patients.
For study type, the pooled CR rates were 92% (95%
CI, 83–100%; I2=34.8%; P=0.175) in 6 retrospective
studies and 83% (95% CI, 67–99%; I2=90.4%; P<0.001)
in 4 prospective studies (Fig. 2B).
For age groups, the pooled CR rates were 80% (95% CI, 28–99%) in
patients <18 years and 87% (95% CI, 78–95%; I2=79.2%;
P<0.001) in patients >18 years (Fig. 2C). For combination therapy, the
pooled CR rates were 89% (95% CI, 79–98%; I2=76.3%;
P<0.001) in the plus TKI subgroup and 81% (95% CI, 61–100%;
I2=75.5%; P=0.017) in the plus chemotherapy subgroup
(Fig. 2D). In the analysis
stratified by Ph status, the pooled CR rates were 88% (95% CI,
78–99%; I2=80.1%; P<0.001) in the all Ph+
subgroup and 84% (95% CI, 69–98%; I2=64.6%; P=0.037) in
the non-all Ph+ subgroup (Fig. 2E). For treatment setting, the pooled
CR rates were 88% (95% CI, 72–100%; I2=89.2%;
P<0.001) in the first-line subgroup and 85% (95% CI, 73–97%;
I2=68.2%; P=0.004) in the R/R subgroup (Fig. 2F).
MRD response
A total of 8 studies were included to analyze the
effect of blinatumomab-based combination therapy on MRD response
rate in high-risk B-ALL, with a pooled MRD response rate of 81%
(95% CI, 75–87%; I2=0.0%; P=0.871) (Fig. 3A). For subgroup analyses by study
type, the pooled MRD response rates were 84% (95% CI, 74–93%;
I2=0.0%; P=0.795) in 5 retrospective studies and 80%
(95% CI, 72–87%; I2=0.0%; P=0.567) in 3 prospective
studies (Fig. 3B). By age group,
the pooled MRD response rates were 80% (95% CI, 28–99%) in patients
<18 years and 81% (95% CI, 75–87%; I2=0.0%; P=0.791)
in patients >18 years (Fig. 3C).
By combination therapy regimen, the pooled MRD response rates were
82% (95% CI, 75–89%; I2=0.0%; P=0.700) in the subgroup
with TKI addition and 79% (95% CI, 67–91%; I2=0.0%;
P=0.962) in the subgroup with chemotherapy addition (Fig. 3D). When stratified by Ph status
(all-Ph+ vs. non-all Ph+), the pooled MRD
response rates were 82% (95% CI, 75–89%; I2=0.0%;
P=0.557) in the all Ph+ subgroup and 80% (95% CI,
69–91%; I2=0.0%; P=0.981) in the non-all Ph+
subgroup (Fig. 3E). By treatment
setting, the pooled MRD response rates were 80% (95% CI, 70–90%;
I2=10.1%; P=0.292) in the first-line subgroup and 82%
(95% CI, 74–90%; I2=0.0%; P=0.854) in the R/R subgroup
(Fig. 3F).
 | Figure 3.Forest plot of the pooled MRD
response rates. (A) Forest plot of all the studies. (B-F) Forest
plots for MRD response rates based on (B) study types (prospective
studies and retrospective studies), (C) different age groups
(<18 and >18 years old), (D) different combination therapy
regimens (plus tyrosine kinase inhibitor and plus chemotherapy),
(E) different Ph-positive expression rates (all Ph+ and
non-all Ph+) and (F) different treatment settings
(first-line and relapsed/refractory). MRD, minimal residual
disease; Ph+, Philadelphia chromosome-positive; TKI,
tyrosine kinase inhibitor; R/R, relapsed/refractory; CI, confidence
interval; ES, effect size; DL, DerSimonian-Laird. |
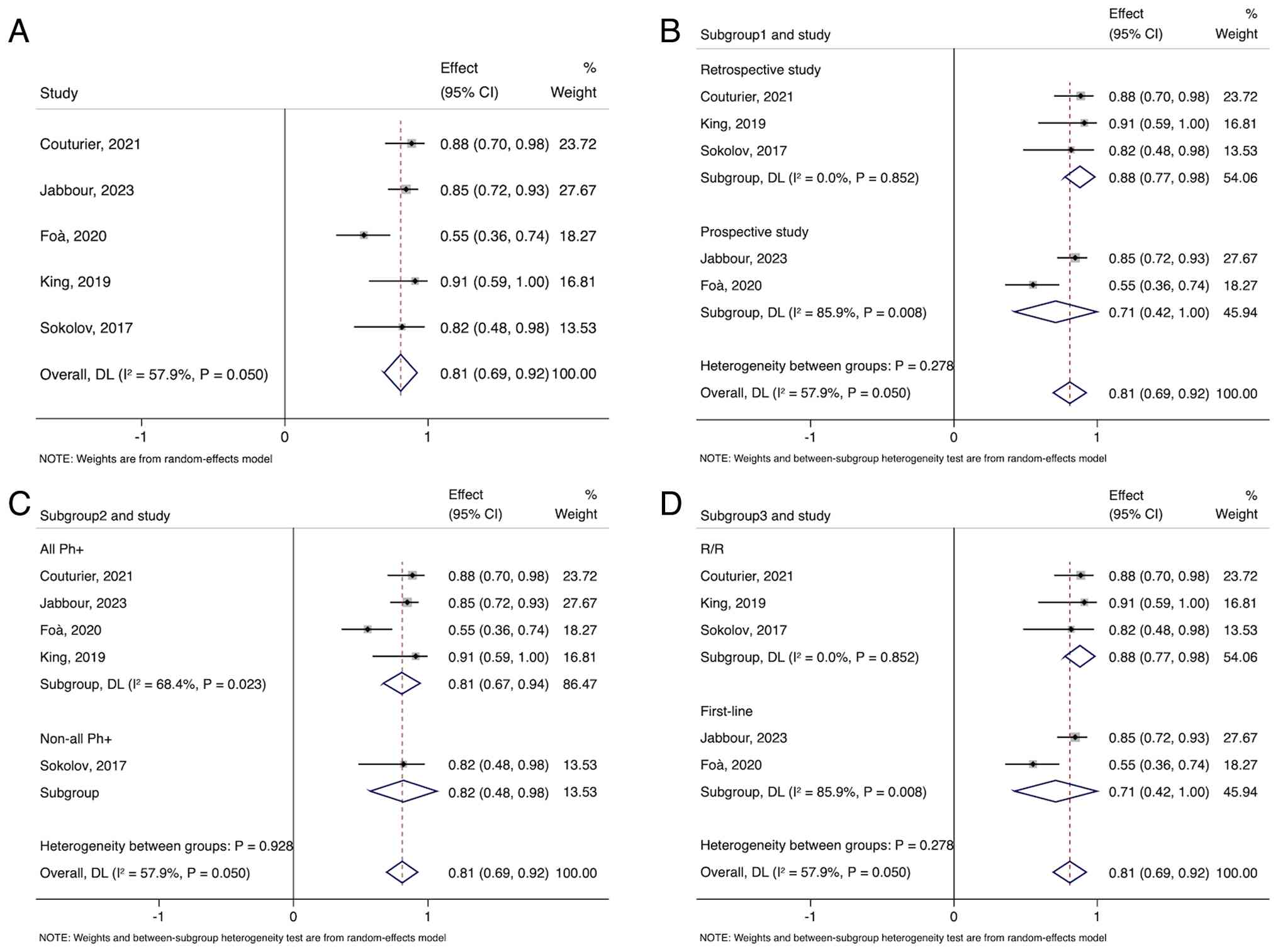
CMR
A total of 5 studies were included that analyzed the
effect of blinatumomab-based combination therapy on CMR in
high-risk B-ALL, with a pooled CMR rate of 81% (95% CI, 69–92%;
I2=57.9%; P=0.050) (Fig.
4A). The pooled CMR rates of 3 retrospective studies and 2
prospective studies were 88% (95% CI, 77–98%; I2=0.0%;
P=0.852) and 71% (95% CI, 42–100%; I2=85.9%; P=0.008),
respectively (Fig. 4B). For the all
Ph+ subgroup and non-all Ph+ subgroup, the
pooled CMR rates were 81% (95% CI, 67–94%; I2=68.4%;
P=0.023) and 82% (95% CI, 48–98%), respectively (Fig. 4C). For the R/R subgroup and the
first-line subgroup, the pooled CMR rates were 88% (95% CI, 77–98%;
I2=0.0%; P=0.852) and 71% (95% CI, 42–100%;
I2=85.9%; P=0.008), respectively (Fig. 4D).
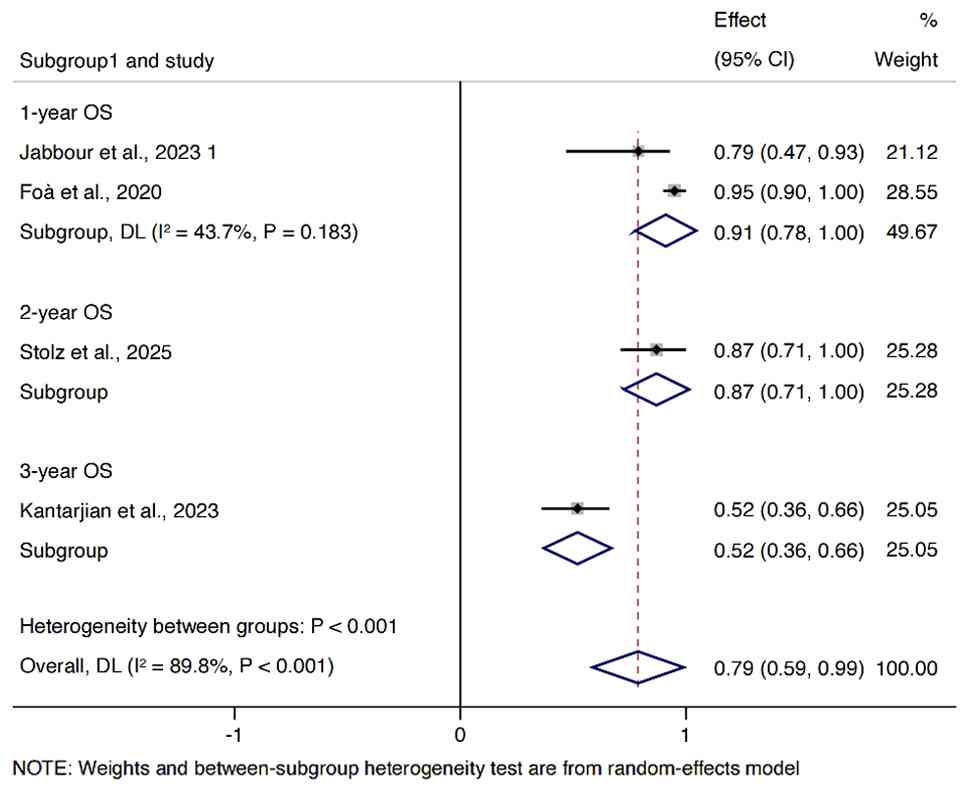
Survival
A meta-analysis was conducted using a random-effects
model, with results showing a pooled 1-year OS rate of 91% (95% CI,
78–100%; I2=43.7%; P=0.183), a 2-year OS rate of 87%
(95% CI, 71–100%) and a 3-year OS rate of 52% (95% CI, 36–66%)
(Fig. 5). Heterogeneity statistics
(I2 and P-value) were not applicable for the 2-year and
3-year estimates as data were derived from single studies.
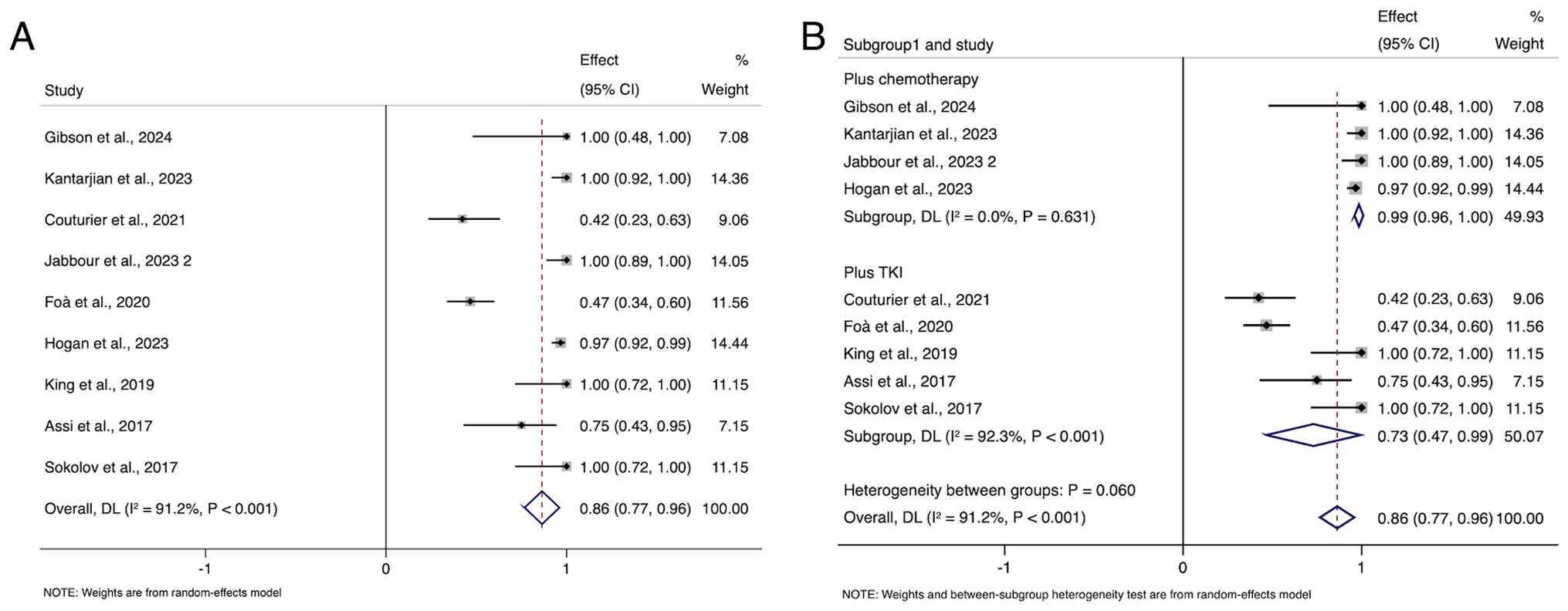
Toxicities
An analysis of the incidence of the most common
all-grade AEs associated with blinatumomab combination therapy for
high-risk B-ALL showed an overall incidence of 86% (95% CI, 77–96%;
I2=91.2%; P<0.001) (Fig.
6A), with results summarized in Table III. Most patients experienced AEs,
which were generally well-tolerated. The most common AEs were
thrombocytopenia, neutropenia and infections, with incidence rates
of 64% (95% CI, 36–92%), 39% (95% CI, 0–86%) and 37% (95% CI,
16–59%), respectively. Notably, grade ≥3 AE incidence rates were
much lower, with few exceeding an incidence rate of 20%. In
particular, the occurrence rates of the most frequent grade ≥3 AEs
(neutropenia, febrile neutropenia and hyperglycemia) were 38% (95%
CI, 0–84%), 26% (95% CI, 10–43%) and 21% (95% CI, 5–36%),
respectively.
 | Table III.AEs of the studies included in the
meta-analysis. |
Table III.
AEs of the studies included in the
meta-analysis.
| Adverse Event | n/N (All
grade) | All grades ES %
(95% CI) | I2
(%) | n/N (Grade ≥3) | Grade ≥3 ES % (95%
CI) | I2
(%) |
|---|
| CRS | 27/206 | 11.2
(4.9–17.5) | 28.5 | 2/127 | 1.6 (0.0–4.3) | - |
| Neutropenia | 67/160 | 39.0
(0.0–86.3) | 98.0 | 65/160 | 38.1
(0.0–83.6) | 97.8 |
| Febrile
Neutropenia | 63/270 | 26.4
(10.1–42.7) | 87.1 | 63/270 | 26.4
(10.1–42.7) | 87.1 |
| Infections | 125/285 | 37.4
(16.2–58.5) | 91.5 | 30/175 | 18.7
(7.3–30.1) | 55.9 |
| Hyperglycemia | 77/260 | 28.5
(6.1–51.0) | 94.3 | 59/260 | 20.7
(3.5–36.2) | 88.9 |
| Sepsis | 11/210 | 5.9 (0.0–14.6) | 72.7 | 11/210 | 5.9 (0.0–14.6) | 72.7 |
|
Thrombocytopenia | 165/207 | 64.2
(36.1–92.4) | 95.6 | 3/17 | 17.6
(3.8–43.4) | - |
|
Neurotoxicity/Neurological Events | 42/143 | 27.5
(9.8–45.2) | 64.0 | 8/132 | 5.4 (1.1–9.7) | 0.0 |
| Hepatic
SOS/VOD | 18/200 | 8.7 (4.4–12.9) | 0.0 | 17/200 | 8.0 (3.9–12.1) | 0.0 |
| Liver Toxicity | 73/223 | 34.3
(9.8–58.9) | 91.9 | 24/200 | 11.8
(7.0–16.6) | 0.0 |
| Hypokalemia | 50/190 | 28.3
(0.0–57.8) | 95.0 | 28/190 | 14.6
(9.3–20.0) | 0.0 |
|
Hyperbilirubinemia | 56/190 | 32.7
(0.0–75.9) | 97.8 | 21/190 | 11.0
(6.2–15.9) | 0.0 |
| Headache | 48/140 | 32.2
(4.8–59.6) | 92.0 | 7/140 | 4.7 (0.6–8.8) | 0.0 |
| Neuropathy | 38/91 | 33.9
(8.1–59.8) | 73.2 | 1/80 | 1.3 (0.0–6.8) | - |
| Constipation | 47/90 | 34.7
(11.8–81.3) | 92.9 | 4/80 | 5.0 (0.0–10.5) | - |
| CMV Reactivation or
Infection | 9/74 | 13.3
(0.0–27.6) | 32.3 | 6/63 | 9.5 (1.5–17.5) | - |
| All | 266/320 | 86.3
(77.0–96.0) | 91.2 | 104/121 | 85.0
(78.0–92.0) | - |
Furthermore, a focused analysis of the hallmark
toxicities of the blinatumomab, CRS and neurological events (Nes),
was conducted. As detailed in Table
III, the pooled incidence rate of all-grade CRS was 11.2%.
Notably, severe CRS (grade ≥3) was exceptionally rare, with a
pooled incidence of only 1.6%. Regarding neurological toxicity,
while the pooled incidence of all-grade Nes was 27.5%, the
incidence of grade ≥3 Nes remained low at 5.4%. Importantly, the
heterogeneity for grade ≥3 Nes was 0.0%, indicating a consistently
low risk of severe neurotoxicity across the included studies.
To further investigate the sources of toxicity, the
analysis was stratified on the basis of combination therapy
regimen. As shown in Fig. 6, the
pooled incidence of all-grade AEs in the blinatumomab +
chemotherapy subgroup was 99% (95% CI, 96–100%), with an
I2 value of 0.0% (P=0.631). By contrast, the pooled
incidence of AEs in the blinatumomab + TKI subgroup was 73% (95%
CI, 47–99%), with an I2 value of 92.3% (P<0.001). The
difference between subgroups approached but did not reach
statistical significance (P=0.060) (Fig. 6B).
Sensitivity analysis
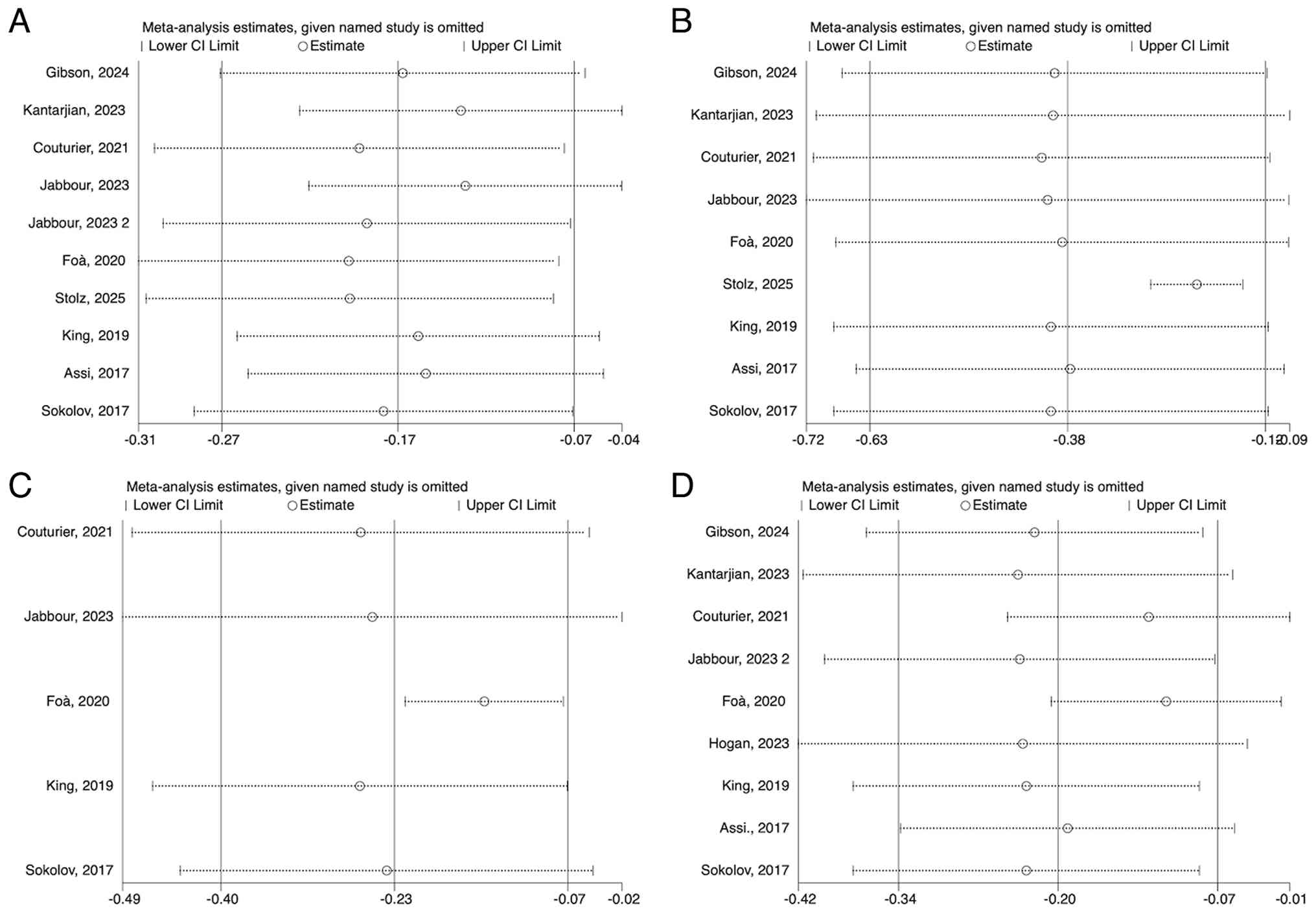
Sensitivity analysis was performed by removing
studies one at a time to determine the effect of this on the pooled
results. It was determined that the removal of a single study made
no significant difference to the pooled outcome and its 95%
confidence, which indicates the general accuracy of the
meta-analysis (Fig. 7).
Publication bias
Publication bias was assessed using Egger's and
Begg's tests. Both tests were applied to evaluate publication bias
for different outcomes with the following results. For MRD
response: Egger's test, P=0.05; and Begg's test, P=0.10 (Fig. S1A). For CMR: Egger's test, P=0.36;
and Begg's test, P=0.22 (Fig.
S1B). For AEs: Egger's test, P=0.09; and Begg's test, P=0.06
(Fig. S1C). No significant
publication bias was observed for these outcomes as aforementioned.
However, upon examining CR, publication bias was identified, with
the following results: Egger's test, P=0.02; and Begg's test,
P=0.05 (Fig. S1D). The observed
publication bias in CR outcomes may be attributed to heterogeneity
in study design, patient characteristics or treatment regimens.
Discussion
ALL, especially R/R, MRD-positive or Ph+
B-ALL, remains a high-risk hematological malignancy. The pivotal
TOWER study demonstrated that blinatumomab was significantly
superior to chemotherapy in R/R B-ALL patients, with mOS times of
7.7 and 4.0 months, respectively [hazard ratio (HR), 0.71]
(20). The BLAST study showed that
blinatumomab induced a negative status for MRD in 78% of
MRD-positive patients, effectively bridging them to HSCT (43). The ALCANTARA study further validated
the efficacy of blinatumomab in adult patients with R/R
Ph+ B-ALL, with a CR rate of 35.6% and a negative MRD
rate as high as 87.5% (44). In
children with R/R B-ALL, the AALL1331 trial showed that
blinatumomab significantly improved 2-year OS rate compared with
chemotherapy (79.4 vs. 59.2%) (45). Even though there is strong
effectiveness of blinatumomab monotherapy, it does not ensure the
long-term survival among the high-risk groups, necessitating
combination therapy strategies.
Combination therapy based on blinatumomab has shown
a synergistic effect in B-ALL. This is primarily attributed to the
immunomodulatory properties of the bispecific antibody, which can
enhance the antitumor activity of other drugs (36). Blinatumomab can activate T cells to
kill leukemia cells by simultaneously binding to CD3 on the surface
of T cells and CD19 on the surface of B cells, thereby triggering
immunogenic cell death and enhancing antigen presentation and T
cell activity in tumors (46). In
addition, combination therapy can upregulate the expression of
tumor cell surface-associated antigens, thereby improving their
sensitivity to targeted drugs. This synergistic effect may overcome
some drug resistance mechanisms associated with blinatumomab
monotherapy in B-ALL. For example, chemotherapy-induced changes in
the tumor microenvironment, including increased T cell infiltration
and enhanced antigen presentation, may help overcome barriers and
increase the proportion of patients benefiting from combination
therapy (20,47,48).
Ongoing and future clinical trials are expected to provide
high-level evidence supporting the efficacy and safety of
blinatumomab combination therapy in high-risk B-ALL (Table IV). These studies are crucial for
future clinical practice and reshaping the prospects of high-risk
B-ALL treatment.
 | Table IV.Ongoing trials of blinatumomab
combination therapy in high-risk acute lymphoblastic leukemia. |
Table IV.
Ongoing trials of blinatumomab
combination therapy in high-risk acute lymphoblastic leukemia.
| NCT ID | Phase | Design | Target enrollment,
n | Primary
endpoint |
|---|
| NCT02744768 | II | Blinatumomab +
dasatinib | 60 | MRD |
| NCT04329325 | II | Blinatumomab +
TKI | 17 | CMR |
| NCT05645718 | II | Blinatumomab + INO
+ rituximab | 27 | AEs |
| NCT04524455 | I | Blinatumomab + AMG
404 | 17 | DLT |
| NCT03160079 | II | Blinatumomab +
pembrolizumab | 16 | ORR |
| NCT03512405 | II | Blinatumomab +
pembrolizumab | 36 | AE/CR |
| NCT05182385 | II | Blinatumomab +
pembrolizumab | 39 | MTD/MRD |
| NCT06308588 | II | Blinatumomab +
asciminib | 40 | AEs |
| NCT03739814 | II | Blinatumomab +
INO | 64 | EFS |
| NCT05931757 | II | Blinatumomab +
olverembatinib | 22 | CR |
| NCT03751709 | I | Blinatumomab +
HMCT | 22 | DLT |
| NCT03595917 | I | Blinatumomab +
ABL001 + dasatinib | 40 | MTD of ABL001 |
| NCT03263572 | II | Blinatumomab +
ponatinib | 90 |
CMR/ORR/OS/EFS/RFS |
| NCT02879695 | I | Blinatumomab +
ipilimumab + nivolumab | 28 | AEs |
| NCT06124157 | III | Blinatumomab +
dasatinib/imatinib | 222 | EFS/AEs |
| NCT04722848 | III | Blinatumomab +
ponatinib | 236 | EFS |
| NCT03518112 | II | Blinatumomab +
chemotherapy | 6 | EFS/CR/CRi/PR |
| NCT03147612 | II | Blinatumomab +
chemotherapy + ponatinib | 22 | CMR/OR |
| NCT04546399 | II | Blinatumomab +
nivolumab | 461 | EFS/AEs/PR |
| NCT04872790 | I | Blinatumomab +
dasatinib | 20 | AEs |
Research on blinatumomab-based combination therapies
for B-ALL has yielded promising results, with different combination
regimens demonstrating certain efficacy. In pediatric R/R B-ALL,
the Pedi-cRIB regimen (mini-Hyper-CVD chemotherapy combined with
INO, blinatumomab and/or rituximab) achieved an objective response
rate (ORR) of 75%, with no CRS or neurotoxicity observed (37). At a median follow-up time of 17.1
months, 67% of patients remained alive and in remission (37). For adult R/R B-ALL, the ORR of
mini-Hyper-CVD combined with INO with or without blinatumomab was
83%, with a 3-year OS rate of 40%. Specifically, for patients
receiving blinatumomab, the 3-year OS was 52%, and the incidence of
sinusoidal obstruction syndrome decreased from 13 to 2% (40). In Ph+ B-ALL, blinatumomab
combined with ponatinib in R/R adult patients resulted in a 96.2%
CR rate and an 88.5% CMR rate, with a mOS of 20 months and
favorable tolerability (36). In
elderly patients with newly diagnosed B-ALL, mini-Hyper-CVD
combined with INO with or without blinatumomab achieved a 2-year
progression-free survival (PFS) rate of 58.2% and a 5-year PFS rate
of 44%, with an ORR of 99%. However, vigilance is required
regarding AEs, such as infections and secondary myeloid neoplasms,
which require close monitoring (39). Furthermore, in children, adolescents
and young adults with B-ALL, blinatumomab combined with
chemotherapy achieved a 4-year DFS rate of 72.7% and an OS rate of
97.1% in patients with bone marrow relapse (with or without
extramedullary relapse), which was significantly better than
chemotherapy alone. Nevertheless, this therapy was not as effective
in individuals who experienced an isolated extramedullary relapse
(38). Such studies propose that
combination therapy can enhance the remission and survival rates,
yet the treatment options would have to be tailored according to
the age of the patient, the type of relapse and other clinical
factors.
The present meta-analysis reveals distinct efficacy
characteristics of combination treatment plans as compared with the
historical standards. There was a marked increase in the pooled CR
rate (87%) compared with the historical 34–44% CR rate reported
with the blinatumomab monotherapy in the TOWER trial (20), validating the synergistic effect of
adding a TKI or chemotherapy. Regarding MRD clearance, the pooled
rate of 81% in the present meta-analysis is close to the 78%
reported in the BLAST study (43).
However, long-term survival differed significantly: The 3-year OS
rate in the present meta-analysis was 52%, which was significantly
lower than the 85% in MRD-negative patients receiving blinatumomab
consolidation therapy in the E1910 trial (49). This difference may stem from patient
baseline characteristics, number of prior lines of therapy (the
present analysis included many R/R patients, while the E1910 study
focused on consolidation therapy) and heterogeneity in subsequent
maintenance therapy strategies. Specifically, in the TKI
combination therapy subgroup, while the high CR rate (89%) was
consistent with the ALCANTARA study results (44), the observed decline in 3-year
survival highlighted a key message: Durable targeted maintenance
therapy may be necessary to maintain remission, despite the
effectiveness of chemotherapy-free induction therapy in
Ph+ B-ALL. The cross-study disparity highlights the
importance of individualized treatment approaches based on disease
stage and an individual risk profile.
Furthermore, the present subgroup analysis according
to treatment stage clarified the optimal timing for blinatumomab
administration. Deep molecular response rates regarding MRD
negativity were comparable between patients receiving first-line
combination therapy and R/R patients (80 vs. 82%). Of note, the
pooled CMR rate was numerically higher in the R/R subgroup compared
with the first-line subgroup (88 vs. 71%), although morphological
CR rates were similar (88 vs. 85%). While the biological principle
that lower tumor burden and a more intact immune system contribute
to optimal blinatumomab activity supports the potential benefits of
early intervention (46), the lower
pooled CMR rate observed in the first-line subgroup may be
attributable to significant heterogeneity among the included
studies (I2=85.9%). Conversely, the high deep remission
rates achieved in the R/R setting underscore the potent
anti-leukemic efficacy of blinatumomab-based combinations,
demonstrating their ability to induce deep molecular remission even
in patients with advanced disease.
Crucially, indirect comparisons of survival outcomes
provide a clearer perspective on the long-term benefits of early
intervention. First-line studies [e.g., Jabbour et al
(39) and Foà et al
(25)] typically report
significantly higher 2-year OS rates compared with the 41% reported
in the R/R study by Couturier et al (36). Therefore, despite the variations in
molecular response rates observed in this meta-analysis, the
optimal window to achieve durable survival remains at the earliest
instance where blinatumomab combination therapy is administered.
This approach could potentially eliminate the need to continue with
intensive chemotherapy or serve as an improved bridge to HSCT.
While safety is generally manageable in both settings, clinicians
must remain vigilant regarding the accumulating toxicity in R/R
patients who possess extensive prior treatment histories.
In terms of safety, it is important to delineate the
source of toxicity. A notable difference was identified in the
present stratified analysis: The pooled incidence of all-grade AEs
was 99% (I2=0.0%) in chemotherapy-containing regimens,
compared with 73% in TKI-based regimens. This consistent and
near-universal toxicity in the chemotherapy subgroup suggests that
the adverse event burden is primarily driven by the cytotoxic drug
backbone rather than blinatumomab itself. By contrast, the
TKI-based strategy showed a significantly improved toxicity
profile. Furthermore, regarding the hallmark toxicities specific to
blinatumomab, the incidence of grade ≥3 CRS and Nes was low in all
combination therapy trials (2.1 and 5.4%, respectively). These
rates are within the range reported in prior blinatumomab
monotherapy studies, including the TOWER trial (20). Compared with the TOWER trial, the
incidence of CRS in the current meta-analysis was similar (11.2 vs.
14.2%), with no apparent increase in CRS signal in the combination
setting. Several non-CRS adverse events were higher, including
hyperglycemia (28.5 vs. 7.5%) and neurological events (27.5 vs.
9.4%), whereas febrile neutropenia (26.4 vs. 24.0%) and infections
(37.4 vs. 34.1%) were broadly comparable. These cross-trial
differences should be interpreted cautiously given heterogeneity in
patient populations, AE ascertainment and concomitant agents;
importantly, these toxicities are generally monitorable and
treatable with standard supportive measures, supporting an overall
manageable safety profile (Table
V). This favorable safety profile is possibly due to the
pre-emptive cytoreductive effect of the adjuvant (TKI or
chemotherapy), which reduces tumor burden prior to T-cell
involvement, thereby mitigating the intensity of immune-related
inflammatory responses.
 | Table V.AEs for subgroups and TOWER
trial. |
Table V.
AEs for subgroups and TOWER
trial.
| AEs | Blinatumomab
combination n/N | ES % (95% CI) | TOWER trial (N=267)
n | % | P-value |
|---|
| CRS | 27/206 | 11.2
(4.9–17.5) | 38 | 14.2 | 0.84 |
| Neutropenia | 67/160 | 39.0
(0.0–86.3) | 53 | 19.9 | 0.87 |
| Febrile
Neutropenia | 63/270 | 26.4
(10.1–42.7) | 64 | 24.0 | 0.98 |
| Infections | 125/285 | 37.4
(16.2–58.5) | 91 | 34.1 | 0.95 |
| Hyperglycemia | 77/260 | 28.5
(6.1–51.0) | 20 | 7.5 | 0.35 |
| Sepsis | 11/210 | 5.9 (0.0–14.6) | 13 | 4.9 | 0.96 |
|
Neurotoxicity/Neurological Events | 42/143 | 27.5
(9.8–45.2) | 25 | 9.4 | 0.45 |
| Liver Toxicity | 73/223 | 34.3
(9.8–58.9) | 34 | 12.7 | 0.70 |
| Hypokalemia | 50/190 | 28.3
(0.0–57.8) | 45 | 16.9 | 0.84 |
| Headache | 48/140 | 32.2
(4.8–59.6) | 77 | 28.8 | 0.99 |
| Constipation | 47/90 | 34.7
(11.8–81.3) | 34 | 12.7 | 0.75 |
| All AEs | 266/320 | 86.3
(77.0–96.0) | 263 | 98.5 | 0.45 |
Contextualizing these findings within the current
therapeutic landscape is essential. While CAR-T cell therapy
demonstrates efficacy in R/R cases (23), its clinical application is often
restricted by manufacturing delays, high costs and the requirement
for specialized centers to manage severe toxicities (such as
high-grade CRS and neurotoxicity) (50,51).
By contrast, blinatumomab-based combinations offer immediate
availability and a generally manageable safety profile (20,52),
making them particularly valuable in resource-limited settings
lacking CAR-T infrastructure. Furthermore, for patients ineligible
for transplantation, such as the elderly or those with marked
comorbidities, combining blinatumomab with TKIs or low-intensity
chemotherapy offers a viable, potentially curative and less toxic
alternative to high-dose chemotherapy and HSCT (25,53).
Future research continues to evolve beyond current immunotherapies.
Emerging CD19/CD22 bispecific antibodies address antigen escape,
while novel nanocarrier-based drug delivery systems are being
explored to enhance the pharmacokinetics and safety of therapeutic
agents (54,55). These technological innovations may
further optimize the delivery of combination therapies. Thus,
current blinatumomab-based strategies represent a robust and
accessible clinical standard that precedes these next-generation
advancements.
The present study has several limitations. First,
the absence of randomized controlled trials (RCTs) limits causal
inferences and the ability to definitively compare efficacy with
that of standard regimens. Second, data availability was limited
for long-term survival outcomes and specific subgroup analyses.
Notably, the estimates for 2- and 3-year OS were derived from
single studies, precluding the execution of a pooled meta-analysis
for these instances. Consequently, these results reflect findings
from individual studies rather than synthesized evidence, and it
remains to be confirmed whether high response rates definitively
translate into long-term survival benefits. Third, high
heterogeneity was observed among the included studies. The subgroup
analysis revealed that clinical heterogeneity, particularly
differences in efficacy between first-line and R/R treatments, and
differences in safety between chemotherapy and TKI regimens, is a
significant contributing factor. Furthermore, inconsistencies in
MRD response assessment methods (flow cytometry vs. next-generation
sequencing) and safety reporting methods (such as different CTCAE
versions) may also contribute to residual heterogeneity. Fourth,
some of the studies used small sample sizes, reducing the
statistical power. As such, large-scale, multicenter, RCTs with the
use of standardized assessment techniques and long-term follow up
would be necessary to confirm these findings.
In conclusion, the present meta-analysis shows that
blinatumomab-based combination therapy is an effective and
well-tolerated treatment option for high-risk B-ALL, exhibiting
high remission rates, durable remission and manageable toxicity.
The combination of blinatumomab and TKIs or low-intensity
chemotherapy has the potential to improve the prognosis of B-ALL.
Even though additional RCTs are required to confirm these results
and optimize treatment options, current evidence has established
blinatumomab combination therapy as a valuable alternative. This
offers a potential shift in the therapeutic paradigm of high-risk
B-ALL, especially for patients ineligible for intensive
chemotherapy or transplantation. Further investigations are needed
on how to optimize combination therapy regimens and to define the
optimal regimen and timing of combination therapy to ensure better
patient selection, as well as increase long-term survival
rates.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The study was funded by the Specialty of Orthopedics (Shang
Antong) of Sichuan Provincial Medical Association (grant no.
2025SAT05) and the Innovation Team Project of the Affiliated
Hospital of Chengdu University Clinical Medical College (grant no.
CDFYCX202202).
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article. The raw pooled data
generated in the present study may be requested from the
corresponding author.
Authors' contributions
Conceptualization was performed by XLY. Data
curation, encompassing the independent screening of articles for
eligibility, data extraction, and quality assessment of the
included studies, was performed by MW, XLY, ZQZ, YH, LLC and YL.
Formal analysis and investigation were performed by MW, XLY, LLC
and YL. Methodology design was the responsibility of MW, XLY, ZQZ,
YH, LLC and YL. Software was employed by XLY for statistical
analysis. Supervision was provided by YL and LLC. Data validation
was performed by YL and LLC. Visualization (preparation of figures
and tables) was the responsibility of MW, XLY, ZQZ, LLC and YL. The
original draft was written by MW and XLY. The manuscript was
reviewed and edited by MW, XLY, LLC and YL. All authors have read
and approved the final manuscript. MW and XLY confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Malard F and Mohty M: Acute lymphoblastic
leukaemia. Lancet. 395:1146–1162. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pui CH, Yang JJ, Hunger SP, Pieters R,
Schrappe M, Biondi A, Vora A, Baruchel A, Silverman LB, Schmiegelow
K, et al: Childhood acute lymphoblastic leukemia: Progress through
collaboration. J Clin Oncol. 33:2938–2948. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Inaba H, Greaves M and Mullighan CG: Acute
lymphoblastic leukaemia. Lancet. 381:1943–1955. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jabbour EJ, Faderl S and Kantarjian HM:
Adult acute lymphoblastic leukemia. Mayo Clin Proc. 80:1517–1527.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Howlader N, Noone A, Krapcho M, Miller D,
Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis D, et al:
SEER cancer statistics review, 1975–2018. National Cancer
Institute; Bethesda, MD: 2021, https://seer.cancer.gov/csr/1975_2018/July 20–2025
|
|
6
|
Arber DA, Orazi A, Hasserjian R, Thiele J,
Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M and Vardiman JW:
The 2016 revision to the World Health Organization classification
of myeloid neoplasms and acute leukemia. Blood. 127:2391–2405.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Parker C, Waters R, Leighton C, Hancock J,
Sutton R, Moorman AV, Ancliff P, Morgan M, Masurekar A, Goulden N,
et al: Effect of mitoxantrone on outcome of children with first
relapse of acute lymphoblastic leukaemia (ALL R3): an open-label
randomised trial. Lancet. 376:2009–2017. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun W, Orgel E, Malvar J, Sposto R, Wilkes
JJ, Gardner R, Tolbert VP, Smith A, Hur M, Hoffman J, et al:
Treatment-related adverse events associated with a modified UK
ALLR3 induction chemotherapy backbone for childhood
relapsed/refractory acute lymphoblastic leukemia. Pediatr Blood
Cancer. 63:1943–1948. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Thomas X, Thiebaut A, Olteanu N, Danaïla
C, Charrin C, Archimbaud E and Fiere D: Philadelphia chromosome
positive adult acute lymphoblastic leukemia: characteristics,
prognostic factors and treatment outcome. Hematol Cell Ther.
40:119–128. 1998.PubMed/NCBI
|
|
10
|
Burmeister T, Schwartz S, Bartram CR,
Gökbuget N, Hoelzer D and Thiel E: Patients' age and BCR-ABL
frequency in adult B-precursor ALL: A retrospective analysis from
the GMALL study group. Blood. 112:918–919. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Geyer MB, Hsu M, Devlin SM, Tallman MS,
Douer D and Park JH: Overall survival among older US adults with
ALL remains low despite modest improvement since 1980: SEER
analysis. Blood. 129:1878–1881. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim C, Molony JT, Chia VM, Kota VK, Katz
AJ and Li S: Patient characteristics, treatment patterns, and
mortality in elderly patients newly diagnosed with ALL. Leuk
Lymphoma. 60:1462–1468. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jabbour E, Pui CH and Kantarjian H:
Progress and innovations in the management of adult acute
lymphoblastic leukemia. JAMA Oncol. 4:1413–1420. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Short NJ, Kantarjian H and Jabbour E:
Optimizing the treatment of acute lymphoblastic leukemia in younger
and older adults: New drugs and evolving paradigms. Leukemia.
35:3044–3058. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chevallier P, Leguay T, Delord M, Salek C,
Kim R, Huguet F, Hicheri Y, Wartiovaara-Kautto U, Raffoux E,
Cluzeau T, et al: Inotuzumab ozogamicin and low-intensity
chemotherapy in older patients with newly diagnosed CD22(+)
philadelphia chromosome-negative B-cell precursor acute
lymphoblastic leukemia. J Clin Oncol. 42:4327–4341. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kantarjian HM, DeAngelo DJ, Stelljes M,
Martinelli G, Liedtke M, Stock W, Gökbuget N, O'Brien S, Wang K,
Wang T, et al: Inotuzumab ozogamicin versus standard therapy for
acute lymphoblastic leukemia. N Engl J Med. 375:740–753. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rizzari C: Inotuzumab ozogamicin in older
patients with acute lymphoblastic leukaemia: Premises and promises.
Lancet Oncol. 19:159–160. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Stelljes M, Alakel N, Wäsch R, Scholl S,
Nachtkamp K, Rank A, Haenel M, Spriewald B, Hanoun M, Martin S, et
al: Final induction therapy results of an open label phase II study
using inotuzumab ozogamicin for induction therapy, followed by a
conventional chemotherapy based consolidation and maintenance
therapy in patients aged 56 years and older with acute
B-lymphoblastic leukemia (INITIAL-1 trial). Blood. 138 (Suppl
1):S23002021. View Article : Google Scholar
|
|
19
|
Jabbour E, Short NJ, Senapati J, Jain N,
Huang X, Daver N, DiNardo CD, Pemmaraju N, Wierda W, Garcia-Manero
G, et al: Mini-hyper-CVD plus inotuzumab ozogamicin, with or
without blinatumomab, in the subgroup of older patients with newly
diagnosed Philadelphia chromosome-negative B-cell acute lymphocytic
leukaemia: Long-term results of an open-label phase 2 trial. Lancet
Haematol. 10:e433–e444. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kantarjian H, Stein A, Gökbuget N,
Fielding AK, Schuh AC, Ribera JM, Wei A, Dombret H, Foà R, Bassan
R, et al: Blinatumomab versus chemotherapy for advanced acute
lymphoblastic leukemia. N Engl J Med. 376:836–847. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
O'Brien MM, Ji L, Shah NN, Rheingold SR,
Bhojwani D, Yuan CM, Xu X, Yi JS, Harris AC, Brown PA, et al: Phase
II trial of inotuzumab ozogamicin in children and adolescents with
relapsed or refractory B-cell acute lymphoblastic leukemia:
Children's oncology group protocol AALL1621. J Clin Oncol.
40:956–967. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Thomas DA, O'Brien S, Jorgensen JL, Cortes
J, Faderl S, Garcia-Manero G, Verstovsek S, Koller C, Pierce S, Huh
Y, et al: Prognostic significance of CD20 expression in adults with
de novo precursor B-lineage acute lymphoblastic leukemia. Blood.
113:6330–6337. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maude SL, Laetsch TW, Buechner J, Rives S,
Boyer M, Bittencourt H, Bader P, Verneris MR, Stefanski HE, Myers
GD, et al: Tisagenlecleucel in children and young adults with
B-cell lymphoblastic leukemia. N Engl J Med. 378:439–448. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shah BD, Ghobadi A, Oluwole OO, Logan AC,
Boissel N, Cassaday RD, Leguay T, Bishop MR, Topp MS, Tzachanis D,
et al: KTE-X19 for relapsed or refractory adult B-cell acute
lymphoblastic leukaemia: Phase 2 results of the single-arm,
open-label, multicentre ZUMA-3 study. Lancet. 398:491–502. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Foà R, Bassan R, Vitale A, Elia L,
Piciocchi A, Puzzolo MC, Canichella M, Viero P, Ferrara F, Lunghi
M, et al: Dasatinib-blinatumomab for ph-positive acute
lymphoblastic leukemia in adults. N Engl J Med. 383:1613–1623.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Marrapodi MM, Mascolo A, di Mauro G,
Mondillo G, Pota E and Rossi F: The safety of blinatumomab in
pediatric patients with acute lymphoblastic leukemia: A systematic
review and meta-analysis. Front Pediatr. 10:9291222022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu H, Xi R, Mao D, Zhao X and Wu T:
Efficacy and safety of blinatumomab for the treatment of
relapsed/refractory acute lymphoblastic leukemia: A systemic review
and meta-analysis. Clin Lymphoma Myeloma Leuk. 23:e139–e149. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Martinelli G, Boissel N, Chevallier P,
Ottmann O, Gökbuget N, Topp MS, Fielding AK, Rambaldi A, Ritchie
EK, Papayannidis C, et al: Complete hematologic and molecular
response in adult patients with relapsed/refractory philadelphia
chromosome-positive B-precursor acute lymphoblastic leukemia
following treatment with blinatumomab: results from a phase II,
single-arm, multicenter study. J Clin Oncol. 35:1795–1802. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372:n712021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wells GA, Shea B, O'Connell D, Peterson J,
Welch V, Losos M and Tugwell P: The Newcastle-Ottawa Scale (NOS)
for assessing the quality of nonrandomised studies in
meta-analyses. Ottawa Hospital Research Institute; Ottawa, ON:
2000
|
|
31
|
Slim K, Nini E, Forestier D, Kwiatkowski
F, Panis Y and Chipponi J: Methodological index for non-randomized
studies (minors): Development and validation of a new instrument.
ANZ J Surg. 73:712–716. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Freites-Martinez A, Santana N,
Arias-Santiago S and Viera A: Using the common terminology criteria
for adverse events (CTCAE - version 5.0) to evaluate the severity
of adverse events of anticancer therapies. Actas Dermosifiliogr
(Engl Ed). 112:90–92. 2021.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Higgins JP and Green S: Cochrane handbook
for systematic reviews of interventions: Cochrane Book Series. The
Cochrane Collaboration; 2008, View Article : Google Scholar
|
|
34
|
Sokolov AN, Parovichnikova EN, Troitskaya
VV, Kuzmina LA, Galtseva IV, Kulikov SM, Bondarenko SN, Davidova
JO, Kapranov NM, Lukyanova IA, et al: Blinatumomab+ tyrosine kinase
inhibitors with no chemotherapy in BCR-ABL-positive or
IKZF1-deleted or FLT3-ITD-positive relapsed/refractory acute
lymphoblastic leukemia patients: high molecular remission rate and
toxicity profile. Blood. 130 (Suppl 1):S38842017.
|
|
35
|
Assi R, Kantarjian H, Short NJ, Daver N,
Takahashi K, Garcia-Manero G, DiNardo C, Burger J, Cortes J, Jain
N, et al: Safety and efficacy of blinatumomab in combination with a
tyrosine kinase inhibitor for the treatment of relapsed
philadelphia chromosome-positive leukemia. Clin Lymphoma Myeloma
Leuk. 17:897–901. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Couturier MA, Thomas X, Raffoux E, Huguet
F, Berthon C, Simand C, Gallego-Hernanz MP, Hicheri Y, Hunault
Berger M, Saillard C, et al: Blinatumomab + ponatinib for
relapsed/refractory Philadelphia chromosome-positive acute
lymphoblastic leukemia in adults. Leuk Lymphoma. 62:620–629. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gibson A, Nunez C, Robusto L, Kammerer B,
Garcia M, Roth M, Sheth R, Tewari P, Hittle A, Toepfer L, et al:
Combination low-intensity chemotherapy plus inotuzumab ozogamicin,
blinatumomab and rituximab for pediatric patients with
relapsed/refractory B-cell acute lymphoblastic leukemia.
Haematologica. 109:3042–3047. 2024.PubMed/NCBI
|
|
38
|
Hogan LE, Brown PA, Ji L, Xu X, Devidas M,
Bhatla T, Borowitz MJ, Raetz EA, Carroll A, Heerema NA, et al:
Children's oncology group AALL1331: Phase III trial of blinatumomab
in children, adolescents, and young adults with low-risk B-Cell ALL
in first relapse. J Clin Oncol. 41:4118–4129. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jabbour E, Short NJ, Jain N, Huang X,
Montalban-Bravo G, Banerjee P, Rezvani K, Jiang X, Kim KH,
Kanagal-Shamanna R, et al: Ponatinib and blinatumomab for
Philadelphia chromosome-positive acute lymphoblastic leukaemia: A
US, single-centre, single-arm, phase 2 trial. Lancet Haematol.
10:e24–e34. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kantarjian H, Haddad FG, Jain N, Sasaki K,
Short NJ, Loghavi S, Kanagal-Shamanna R, Jorgensen J, Khouri I,
Kebriaei P, et al: Results of salvage therapy with mini-hyper-CVD
and inotuzumab ozogamicin with or without blinatumomab in pre-B
acute lymphoblastic leukemia. J Hematol Oncol. 16:442023.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
King AC, Pappacena JJ, Tallman MS, Park JH
and Geyer MB: Blinatumomab administered concurrently with oral
tyrosine kinase inhibitor therapy is a well-tolerated consolidation
strategy and eradicates measurable residual disease in adults with
Philadelphia chromosome positive acute lymphoblastic leukemia. Leuk
Res. 79:27–33. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Stolz SM, Hofer KD, Rösler W, Deuel J,
Schwotzer R, Schneidawind C, Schneidawind D, Manz MG and Rieger MJ:
Tyrosine kinase inhibitors with blinatumomab versus chemotherapy in
Philadelphia-positive acute B-lymphoblastic leukemia. Int J Cancer.
157:1197–1204. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gökbuget N, Dombret H, Bonifacio M,
Reichle A, Graux C, Faul C, Diedrich H, Topp MS, Brüggemann M,
Horst HA, et al: Blinatumomab for minimal residual disease in
adults with B-cell precursor acute lymphoblastic leukemia. Blood.
131:1522–1531. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Martinelli G, Boissel N, Chevallier P,
Ottmann O, Gökbuget N, Rambaldi A, Ritchie EK, Papayannidis C,
Tuglus CA, Morris JD, et al: Long-term follow-up of blinatumomab in
patients with relapsed/refractory Philadelphia chromosome-positive
B-cell precursor acute lymphoblastic leukaemia: Final analysis of
ALCANTARA study. Eur J Cancer. 146:107–114. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Brown PA, Ji L, Xu X, Devidas M, Hogan LE,
Borowitz MJ, Raetz EA, Zugmaier G, Sharon E, Bernhardt MB, et al:
Effect of postreinduction therapy consolidation with blinatumomab
vs chemotherapy on disease-free survival in children, adolescents,
and young adults with first relapse of B-Cell acute lymphoblastic
leukemia: A randomized clinical Trial. JAMA. 325:833–842. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Franquiz MJ and Short NJ: Blinatumomab for
the treatment of adult B-Cell Acute lymphoblastic leukemia: Toward
a new era of targeted immunotherapy. Biologics. 14:23–34.
2020.PubMed/NCBI
|
|
47
|
Horibe K, Morris JD, Tuglus CA, Dos Santos
C, Kalabus J, Anderson A, Goto H and Ogawa C: A phase 1b study of
blinatumomab in Japanese children with relapsed/refractory B-cell
precursor acute lymphoblastic leukemia. Int J Hematol. 112:223–233.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hunger SP and Raetz EA: How I treat
relapsed acute lymphoblastic leukemia in the pediatric population.
Blood. 136:1803–1812. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Podoltsev NA, Sun Z, Litzow MR, Paietta E,
Roberts KG, Zhang Y, Racevskis J, Lazarus HM, Rowe JM, Arber DA, et
al: Addition of blinatumomab to consolidation therapy among older
newly diagnosed patients (pts) with BCR:: ABL1 Negative B-lineage
acute lymphoblastic leukemia (ALL) in the ECOG-ACRIN E1910
randomized phase III trial. Blood. 144 (Suppl 1):S42112024.
View Article : Google Scholar
|
|
50
|
Neelapu SS, Locke FL, Bartlett NL, Lekakis
LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T,
Lin Y, et al: Axicabtagene Ciloleucel CAR T-Cell therapy in
refractory large B-Cell lymphoma. N Engl J Med. 377:2531–2544.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lee DW, Santomasso BD, Locke FL, Ghobadi
A, Turtle CJ, Brudno JN, Maus MV, Park JH, Mead E, Pavletic S, et
al: ASTCT consensus grading for cytokine release syndrome and
neurologic toxicity associated with immune effector cells. Biol
Blood Marrow Transplant. 25:625–638. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Topp MS, Gökbuget N, Stein AS, Zugmaier G,
O'Brien S, Bargou RC, Dombret H, Fielding AK, Heffner L, Larson RA,
et al: Safety and activity of blinatumomab for adult patients with
relapsed or refractory B-precursor acute lymphoblastic leukaemia: A
multicentre, single-arm, phase 2 study. Lancet Oncol. 16:57–66.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Advani AS, Moseley A, O'Dwyer KM, Wood BL,
Fang M, Wieduwilt MJ, Aldoss I, Park JH, Klisovic RB, Baer MR, et
al: SWOG 1318: A phase II trial of blinatumomab followed by POMP
maintenance in older patients with newly diagnosed philadelphia
chromosome-negative B-Cell acute lymphoblastic leukemia. J Clin
Oncol. 40:1574–1582. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Nabih NW, Hassan HAFM, Preis E, Schaefer
J, Babker A, Abbas AM, Amin MU, Bakowsky U and Fahmy SA:
Antibody-functionalized lipid nanocarriers for RNA-based cancer
gene therapy: Advances and challenges in targeted delivery.
Nanoscale Adv. 7:5905–5931. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wafik Nabih N, Nafie MS, Babker A, Hassan
HAFM and Fahmy SA: Recent advances in nano vehicles encapsulating
cinnamic acid and its derivatives as promising anticancer agents.
RSC Adv. 15:20815–20847. 2025. View Article : Google Scholar : PubMed/NCBI
|