Introduction
Colorectal cancer (CRC) is the third most common
cancer type diagnosed worldwide and the second leading cause of
cancer-related death; it also presents the highest total lifetime
risk of developing and dying from gastrointestinal cancer (1,2).
The introduction of immune checkpoint inhibitors
(ICIs) has reshaped treatment and outcomes in patients with a
variety of solid tumors including metastatic CRC (mCRC). Notably,
patients with cancer harboring the mismatch repair-deficient
(dMMR)/microsatellite instability-high (MSI-H) phenotype have shown
a higher response and better efficacy to ICI treatment, with a
significantly prolonged 5-year survival rate (76±4 vs. 54±2%;
P<0.001) (3). However, only 15%
of early-stage CRC and 5% of mCRC cases are dMMR/MSI-H (4). The vast majority of patients with mCRC
are in a MMR proficient (pMMR)/microsatellite stable (MSS) genomic
state characterized by an immunosuppressive tumor microenvironment
(TME) with a low tumor mutational burden and a lack of
tumor-infiltrating lymphocytes (5).
Therefore, unsurprisingly, ICI monotherapy has failed to
demonstrate meaningful clinical activity in this patient
population.
To overcome immunoresistance and enhance effective
antitumor immune response, an increasing number of basic research
and clinical trials are exploring ICI-based combination strategies
to synergistically stimulate the transformation of an immune-desert
TME into an immunosupportive TME. A series of exploratory studies
have reported mixed results from ICI combined with chemotherapy
(CT), radiotherapy (RT), antiangiogenic targeted agents (such as
regorafenib and fruquintinib) or other cytotoxic drugs in treating
mCRC (6–11).
However, to the best of our knowledge, routine
clinical practice data on immune-based combination modalities by
line of treatment for patients with pMMR/MSS mCRC are still
lacking. Therefore, the present retrospective study was conducted
to evaluate the real-world ICI-based treatment patterns and
outcomes in patients with pMMR/MSS mCRC across all treatment lines,
especially those in third-line or above (3L+) settings who have
limited treatment options under current guidelines and are in
urgent need of effective treatment strategies.
Patients and methods
Study design and population
This retrospective study was conducted at the Renmin
Hospital of Wuhan University (Wuhan, China). Patients who were
treated at the Department of Oncology of Renmin Hospital of Wuhan
University from June 10, 2019 to December 20, 2023 were screened
and enrolled. Patients were selected according to the following
inclusion criteria: i) Patients who were ≥18 years of age; ii) had
histologically or cytologically confirmed metastatic pMMR/MSS
colorectal adenocarcinoma; iii) had received at least one dose of
ICI at the first-line (1L), second-line (2L) or 3L+ setting; iv)
and had available follow-up data. The exclusion criteria included:
i) Patients with other malignancies (except skin basal cell
carcinoma and cervical carcinoma in situ); ii) a history of
or active autoimmune disease or being treated with
immunosuppressive therapy; and iii) with incomplete medical
data.
Follow-up for survival analysis continued until July
20, 2024, which served as the final cut-off date for the present
study. The patients typically returned to Renmin Hospital of Wuhan
University for regular re-examination, including in outpatient or
inpatient clinics. For patients who did not attend clinic visits,
follow-up was conducted via telephone. The clinical data from June
10, 2019 to July 20, 2024 were collected and accessed
retrospectively through the medical electronic system at the
hospital, which included age, sex, histological type, primary tumor
location, type of metastasis, metastatic site, RAS/BRAF mutation
status, MSI/MMR status, treatment information, adverse events (AEs)
and survival status. MSI or MMR status was tested using
immunohistochemistry, polymerase chain reaction or next-generation
sequencing.
In addition, there are currently diverse definitions
and similar terms for ‘ICI reuse’, represented by ‘ICI
rechallenge’, ‘ICI retreatment’, ‘ICI re-administration’, ‘ICI
restart’, ‘ICI re-exposure’, ‘ICI reinitiation’, ‘ICI
reintroduction’ and ‘ICI resumption’, which mainly vary based on
the cause of interruption, with or without in-between therapy and
the subsequent treatments. In the present study, to achieve
terminological uniformity, ‘ICI cross-line therapy’ was used as a
surrogate for a series of similar concepts. ICI cross-line therapy
was defined as the reuse of ICI in the same patient across
different lines of treatment after prior interruption, regardless
of the reason for discontinuation (such as disease progression,
toxicity, completion of planned treatment or other clinical
decisions).
Outcome variables
The primary outcomes of interest were median
progression-free survival (PFS), overall survival (OS), objective
response rate (ORR), disease control rate (DCR) and safety. PFS was
defined as the time from 1L, 2L or 3L+ treatment initiation to
disease progression or death from any cause (whichever occurred
first) or last visit (for censored patients). OS was measured from
the treatment initiation to death or date of last follow-up. The
last follow-up date was recorded as the censored data for survival
analysis when death or progression time could not be confirmed or
if the patient was still alive.
Tumor response was assessed according to the
Response Evaluation Criteria in Solid Tumors v1.1 (12) and classified as complete response
(CR), partial response (PR), stable disease (SD) or progressive
disease (PD). ORR was determined as the rate of a best response of
CR or PR. DCR included CR, PR and SD. Data on treatment-related AEs
(TRAEs) and immune-related AEs (irAEs) were collected and graded
for severity according to the National Cancer Institute Common
Terminology Criteria for Adverse Events version 5.0 (13).
Statistical analysis
All statistical analyses in the present study were
performed using SPSS statistical software (version 26.0; IBM Corp.)
and R (version 4.2.3; R Project for Statistical Computing;
http://www.r-project.org/). Categorical
variables are summarized by percentages and are compared by the
χ2 test or Fisher's exact test. Continuous variables are
described as the median and interquartile range (IQR), and are
compared using the Kruskal-Wallis rank-sum test. The median
follow-up time was determined using the reverse Kaplan-Meier
estimator. PFS and OS were plotted and compared using Kaplan-Meier
curves and log-rank tests under the assumption of proportional
hazard rates. The two-stage method was used for the correct
comparison of two crossing survival curves. A Cox proportional
hazards regression model analysis was performed to calculate the
hazard ratio (HR) and bilateral 95% confidence interval (CI).
Propensity score matching (PSM) was used to control confounding and
balance baseline characteristics between groups with a 1:1 matching
ratio and a caliper of 0.02. Demographic characteristics, AEs and
other clinical data are summarized descriptively. P<0.05 was
considered to indicate a statistically significant difference.
Results
Patient characteristics
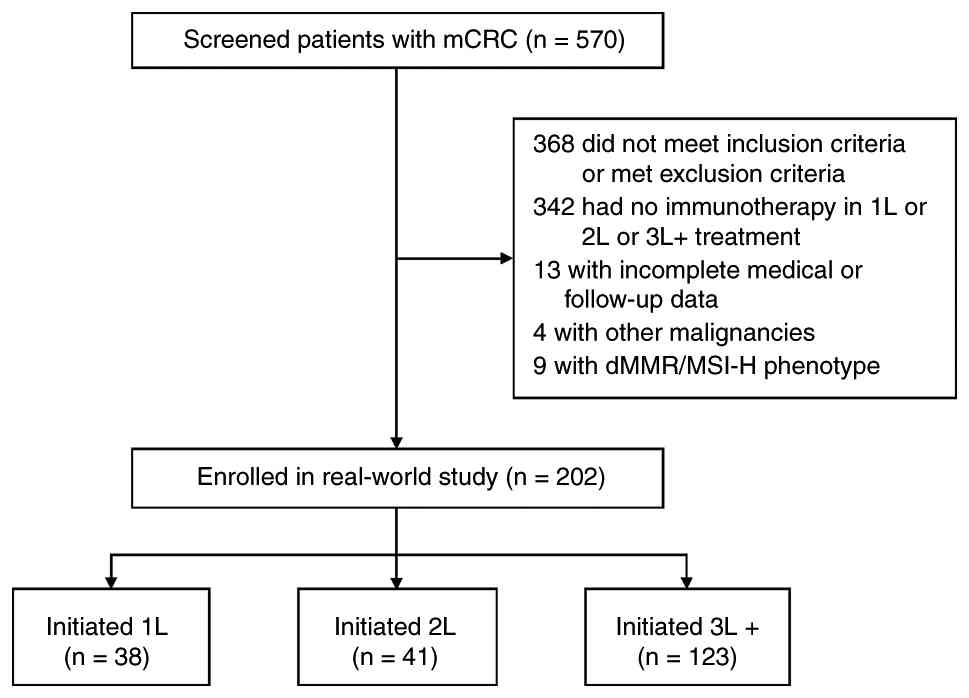
In total, 202 patients with mCRC were included in
the present study (Fig. 1), of whom
38 (18.8%) had a 1L ICI-based treatment initiation (1L cohort), 41
(20.3%) had an initiated 2L ICI-based treatment (2L cohort) and 123
(60.9%) received initiated ICI-based treatment at 3L+ settings (3L+
cohort). Of the entire cohort, the median age of the patients was
60 years (IQR, 51–68 years), 137 (67.8%) patients were male, 156
(77.2%) had a primary tumor located in left-side colon and rectum,
150 (74.3%) had ≥2 metastatic organs and 134 (66.3%) had liver
metastasis. The incidence of lung metastasis increased with the
progression of treatment lines (28.9 vs. 43.9 vs. 57.7%). Gene
mutation data were available for 132 patients (65.3%) and 67
(33.2%) harbored RAS/BRAF mutations. All patients had pMMR and/or
MSS phenotypes. Notably, 80.7% of patients underwent surgical
resection of the primary lesion. None of the patients in the 1L
cohort had received previous targeted therapy, while in the 2L and
3L+ cohorts, 73.2 and 91.9% of the patients, respectively, had a
history of targeted therapy. Patient baseline characteristics,
stratified according to the line of therapy, are presented in
Table I.
 | Table I.Patient demographics and baseline
characteristics. |
Table I.
Patient demographics and baseline
characteristics.
| Variables | Total (n=202) | 1L cohort
(n=38) | 2L cohort
(n=41) | 3L+ cohort
(n=123) |
P-valuea |
|---|
| Median age (IQR),
years | 60 (51–68) | 60 (50–70) | 62 (52–69) | 59 (51–66) | 0.260 |
|
>60 | 93 (46.0) | 18 (47.4) | 24 (58.5) | 51 (41.5) | 0.162 |
|
≤60 | 109 (54.0) | 20 (52.6) | 17 (41.5) | 72 (58.5) |
|
| Sex |
|
|
|
| 0.349 |
|
Male | 137 (67.8) | 26 (68.4) | 24 (58.5) | 87 (70.7) |
|
|
Female | 65 (32.2) | 12 (31.6) | 17 (41.5) | 36 (29.3) |
|
| ECOG PS score |
|
|
|
| 0.865 |
|
0-1 | 187 (92.1) | 35 (92.1) | 39 (95.1) | 113 (91.9) |
|
| 2 | 15 (7.4) | 3 (7.9) | 2 (4.9) | 10 (8.1) |
|
| Primary site |
|
|
|
| 0.771 |
|
Left | 156 (77.2) | 28 (73.7) | 33 (80.5) | 95 (77.2) |
|
|
Right | 46 (22.8) | 10 (26.3) | 8 (19.5) | 28 (22.8) |
|
| Metastatic
organs |
|
|
|
| 0.217 |
| 1 | 52 (25.7) | 14 (36.8) | 9 (22.0) | 29 (23.6) |
|
| ≥2 | 150 (74.3) | 24 (63.2) | 32 (78.0) | 94 (76.4) |
|
| Type of
metastasis |
|
|
|
|
|
| With
liver metastasis | 134 (66.3) | 26 (68.4) | 29 (70.7) | 79 (64.2) | 0.714 |
| With
lung metastasis | 100 (49.5) | 11 (28.9) | 18 (43.9) | 71 (57.7) | 0.006 |
| RAS/BRAF
status |
|
|
|
| 0.050 |
|
RAS/BRAF MT | 67 (33.2) | 8 (21.1) | 11 (26.8) | 48 (39.0) |
|
|
RAS/BRAF WT | 65 (32.2) | 10 (26.3) | 14 (34.1) | 41 (33.3) |
|
|
Unknown | 70 (34.7) | 20 (52.6) | 16 (39.0) | 34 (27.6) |
|
| MMR/MSI status |
|
|
|
| >0.999 |
|
pMMR/MSS | 202 (100.0) | 38 (100.0) | 41 (100.0) | 123 (100.0) |
|
| Prior
treatment |
|
|
|
|
|
| Surgery
of primary lesion | 163 (80.7) | 27 (71.1) | 36 (87.8) | 100 (81.3) | 0.163 |
|
Chemotherapy | 174 (86.1) | 10 (26.3) | 41 (100.0) | 123 (100.0) | <0.001 |
|
Radiotherapy | 73 (36.1) | 3 (7.9) | 16 (39.0) | 54 (43.9) | <0.001 |
|
Targeted drugsb | 143 (70.8) | 0 | 30 (73.2) | 113 (91.9) | <0.001 |
|
Anti-EGFR mAb | 30 (14.9) | 0 | 4 (9.8) | 26 (21.1) | 0.003 |
|
Anti-VEGF(R) mAb | 119 (58.9) | 0 | 22 (53.7) | 97 (78.9) | <0.001 |
|
TKIs | 45 (22.3) | 0 | 4 (9.8) | 41 (33.3) | <0.001 |
Treatment patterns
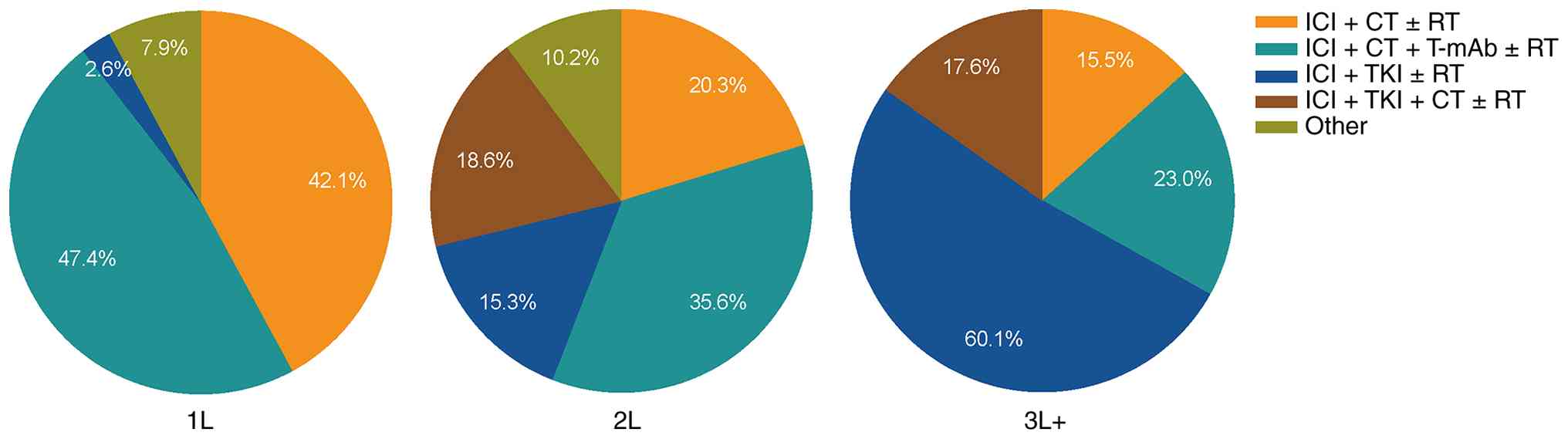
ICI-based treatment patterns are described in
Table II and Fig. 2. The most commonly used regimens in
1L therapy were ICI combined with CT alone (31.6%) and ICI combined
with CT and anti-VEGF(R)/EGFR/HER2 targeted agent (28.9%) (Table II). The 2L therapy was still
dominated by CT, among which ICI plus CT and anti-VEGF(R)/EGFR/HER2
monoclonal antibody accounted for 30.5%. However, in the 3L+
treatment of pMMR/MSS mCRC patients, the predominant regimen has
shifted to the combination of ICI and TKI, accounting for 77.7%
(115/148). Among them, the ICI+TKI±RT regimen accounted for 60.1%
(89/148), and the ICI+TKI+CT±RT regimen accounted for 17.6%
(26/148) (Fig. 2). The proportion
of patients receiving ICI combined with TKI alone (1L: 2.6%; 2L:
11.9%; 3L: 56.1%; P<0.0001) increased gradually with each
advancing line of therapy (Table
II). However, CT lost its dominance in the 3L+ setting, and the
proportion of patients receiving ICI combined with CT alone (1L:
31.6%; 2L: 13.6%; 3L: 12.8%; P=0.016) decreased gradually with each
advancing line of therapy (Table
II).
 | Table II.Treatment patterns in the patient
cohort. |
Table II.
Treatment patterns in the patient
cohort.
| Variables | 1L | 2L | 3L+ |
P-valuea |
|---|
| Total patients | 38 (18.8) | 59 (29.2) | 148
(73.3)b | <0.0001 |
| Initiated line of
ICIc | 38 (18.8) | 41 (20.3) | 123 (60.9) | <0.0001 |
| ICI cross-line
therapy | 0 (0.0) | 18 (8.9) | 54
(26.7)d | <0.0001 |
| Treatment
regimens |
|
|
|
|
| ICI ± CT ± RT | 17 (44.7) | 14 (23.7) | 32 (21.6) | 0.013 |
| ICI
alone | 1 (2.6) | 1 (1.7) | 7 (4.7) | 0.539 |
| ICI +
CT | 12 (31.6) | 8 (13.6) | 19 (12.8) | 0.016 |
| ICI +
RT | 0 (0.0) | 1 (1.7) | 2 (1.4) | 0.741 |
| ICI +
CT + RT | 4 (10.5) | 4 (6.8) | 4 (2.7) | 0.102 |
| ICI +
anti-VEGF(R)/EGFR/HER2 ± CT ± RT | 20 (52.6) | 24 (40.7) | 44 (29.7) | 0.022 |
| ICI +
anti-VEGF(R)/EGFR/HER2e | 1 (2.6) | 2 (3.4) | 9 (6.1) | 0.562 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + CT | 11 (28.9) | 18 (30.5) | 26 (17.6) | 0.076 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + RT | 1 (2.6) | 1 (1.7) | 1 (0.7) | 0.577 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + CT + RT | 7 (18.4) | 3 (5.1) | 8 (5.4) | 0.017 |
| ICI + TKI ± CT ±
RT | 1 (2.6) | 20 (33.9) | 115 (77.7) | <0.0001 |
| ICI +
TKIf | 1 (2.6) | 7 (11.9) | 83 (56.1) | <0.0001 |
| ICI +
TKI + CT | 0 (0.0) | 10 (16.9) | 21 (14.2) | 0.033 |
| ICI +
TKI + RT | 0 (0.0) | 2 (3.4) | 6 (4.1) | 0.454 |
| ICI +
TKI + CT + RT | 0 (0.0) | 1 (1.7) | 5 (3.4) | 0.443 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + TKI ± CT ± RT | 0 (0.0) | 1 (1.7) | 5 (3.4) | 0.443 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + TKI | 0 (0.0) | 0 (0.0) | 2 (1.4) | 0.516 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + TKI + CT | 0 (0.0) | 0 (0.0) | 2 (1.4) | 0.516 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + TKI + RT | 0 (0.0) | 0 (0.0) | 1 (0.7) | 0.720 |
| ICI +
anti-VEGF(R)/EGFR/HER2 + TKI + CT + RT | 0 (0.0) | 1 (1.7) | 0 (0.0) | 0.205 |
Given the long-lasting immunological memory
properties of immunotherapy, increasing evidence supports the
survival benefit of ICI cross-line therapy in clinical practice
(14–18). In the present study, among the 148
patients receiving 3L+ ICI-based treatments, 25 patients had
immunotherapy initiation at 1L and/or 2L, known as ICI cross-line
therapy. Of the 123 patients who received initial ICI at the 3L+
setting, 29 received subsequent ICI cross-line therapy.
Treatment response
The ORR of the 1L, 2L and 3L+ cohorts was 63.2, 22.0
and 11.4%, respectively, and the DCR was 94.7, 75.6 and 70.7%,
respectively (Fig. 3A). In total,
there was 1 (2.6%) case of confirmed CR and 23 (60.5%) cases of PR
in the 1L cohort and 9 (22.0%) cases of PR and 22 (53.7%) cases of
SD in the 2L cohort. As expected, the ORR and DCR in the 3L+ cohort
was lower than that in 2L and 1L cohorts. However, the antitumor
response in the 14 (11.4%) PR cases and 73 (59.3%) SD cases in the
3L+ cohort was acceptable compared with previous reports (19,20).
In the efficacy-evaluable population, a reduction in the size of
target lesions was achieved in 29/38 (76.3%), 27/40 (67.5%) and
52/121 (43.0%) patients in the IL, 2L and 3L+ cohorts, respectively
(Fig. 3B).
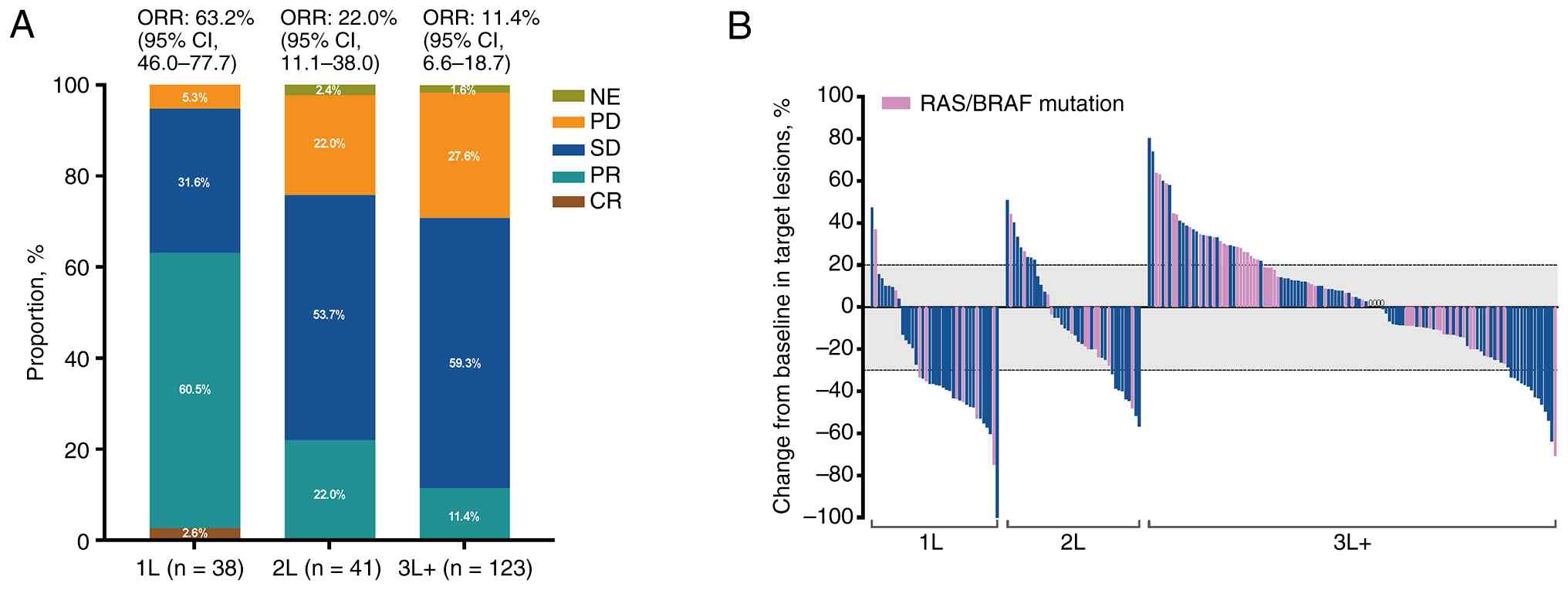 | Figure 3.Tumor response. (A) Treatment
efficacy in each cohort. (B) Waterfall plot showing the best
percentage change in the size of the target lesions from the
baseline in the efficacy-evaluable population in each cohort. The
dashed lines at +20 and −30% indicate thresholds for progressive
disease and partial response, respectively, according to RECIST
v1.1. 1L, first-line treatment; 2L, second-line treatment; 3L+,
third-line or above treatment; CI, confidence interval; CR,
complete response; CT, chemotherapy; NE, not evaluable; ORR,
objective response rate; PD, progressive disease; PR, partial
response; SD, stable disease. |
Survival and prognosis
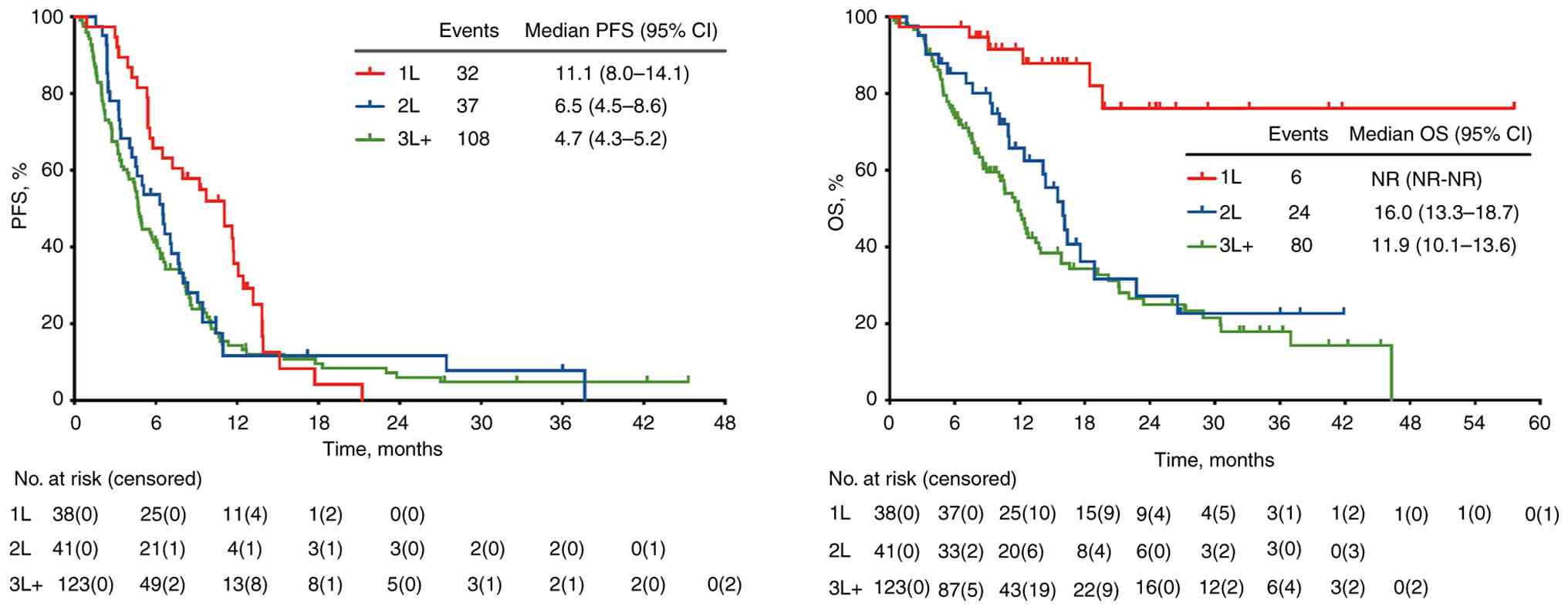
As of the data cut-off in July 20, 2024, the median
follow-up was 19.8 (range, 0.4–57.6) months, the median PFS of the
1L, 2L and 3L+ cohorts was 11.1 (95% CI, 8.0–14.1), 6.5 (95% CI,
4.5–8.6) and 4.7 (95% CI, 4.3–5.2) months, respectively, and the
median OS time was not reached, 16.0 (95% CI, 13.3–18.7) and 11.9
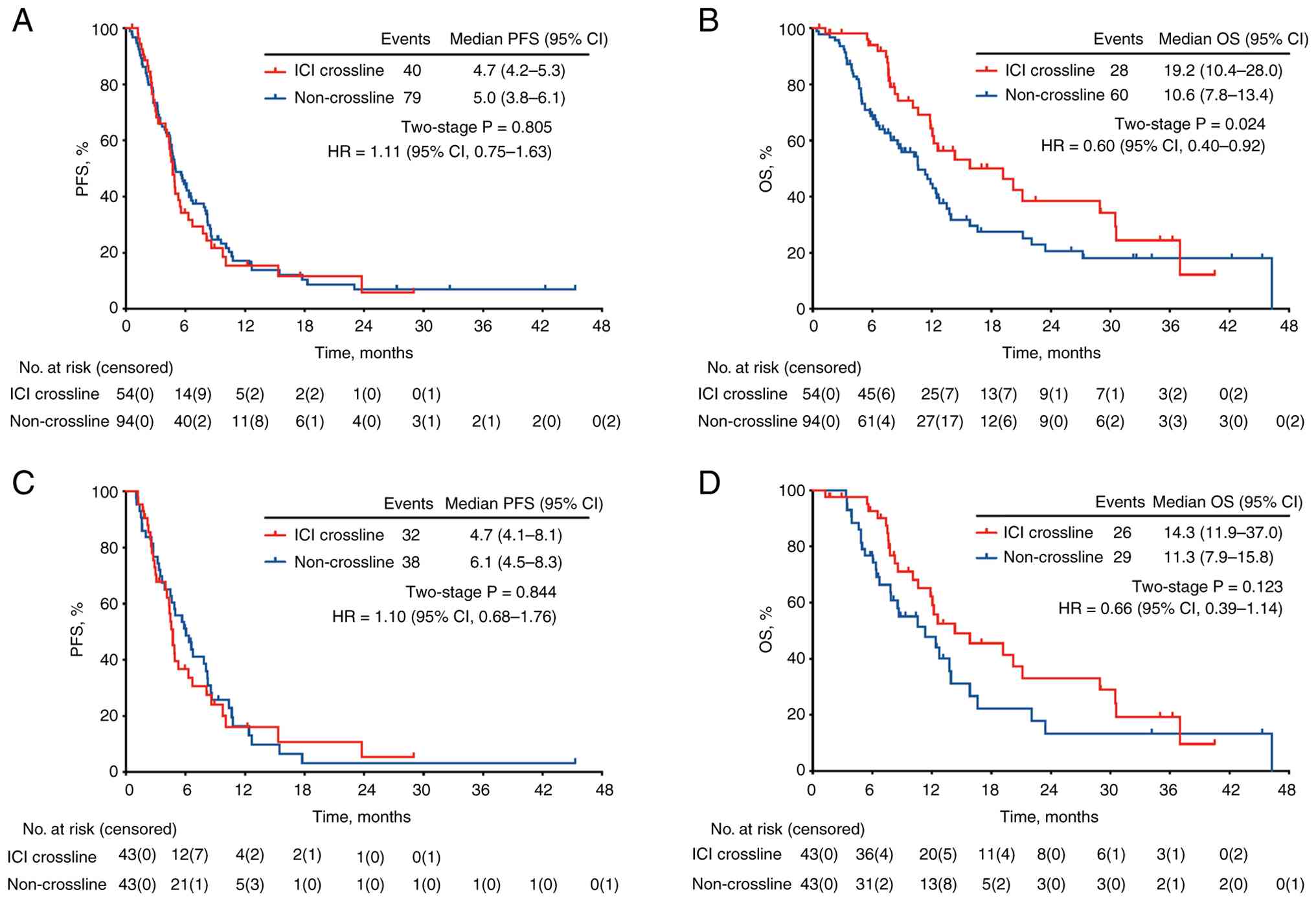
(95% CI, 10.1–13.6) months, respectively (Fig. 4A and B). In the 3L+ cohort, 54
(26.7%) patients received ICI cross-line therapy at least once,
while the remaining 94 patients did not. After PSM, 43 pairs of
patients with balanced baseline features were matched (Table SI, Table SII, Table SIII). Compared with patients in the
non-ICI cross-line group, no significant difference was found in
the PFS rate before and after PSM (Fig.
5A and C). However, for OS, although the difference was
statistically significant before PSM (Fig. 5B), only directional OS improvement
(HR, 0.66; 95% CI, 0.39–1.14; P=0.123) was observed after PSM
(Fig. 5D), with a median OS time of
14.3 months in the ICI cross-line group and 11.3 months in the
non-ICI cross-line group.
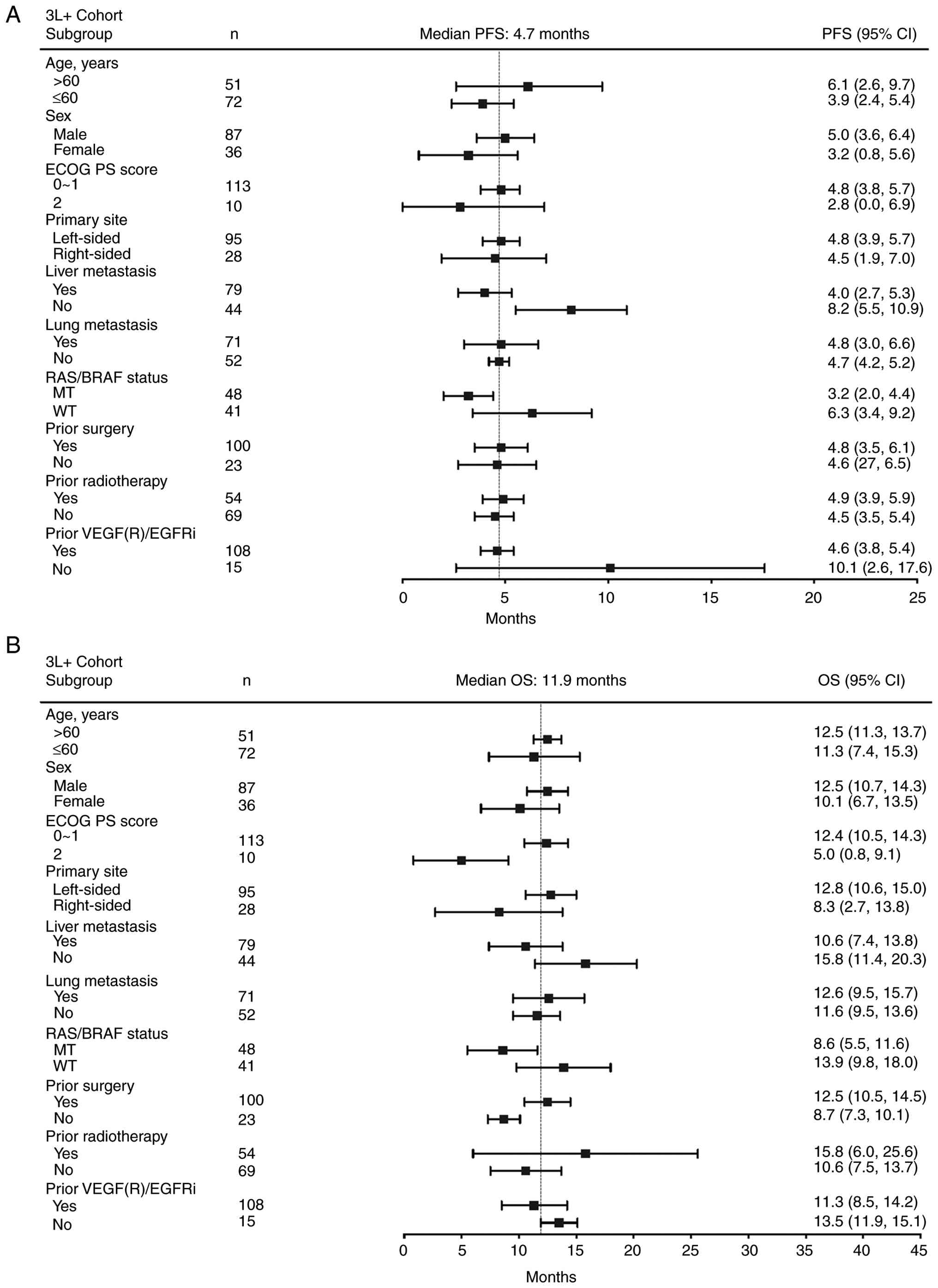
Univariate analysis showed that patients with liver
metastasis (4.0 vs. 8.2 months; HR, 1.97; 95% CI: 1.35–2.87;
P=0.001), RAS/BRAF mutations (3.2 vs. 6.3 months; HR, 1.62; 95% CI,
1.03–2.54; P=0.031) and previous anti-VEGF/EGFR therapy (4.6 vs.
10.1 months; HR, 1.97; 95% CI, 1.21–3.21; P=0.028) had poorer
median PFS times in the 3L+ cohort (Fig. 6A and Table III). Multivariate analysis
indicated that liver metastasis (HR, 2.08; 95% CI, 1.24–3.50;
P=0.006), RAS/BRAF mutation (HR, 1.83; 95% CI, 1.11–3.02; P=0.018)
and age >60 years (HR, 0.58; 95% CI, 0.35–0.98; P=0.041) were
independent prognostic factors for PFS in patients with pMMR/MSS
mCRC who received ICI at the 3L+ setting (Table III). As for OS, Eastern
Cooperative Oncology Group performance status (ECOG PS) and
RAS/BRAF mutation were two independent prognostic factors in the
3L+ cohort (Fig. 6B and Table III). Univariate and multivariate
analyses were also conducted for the 1L and 2L cohorts (Figs. S1 and S2 and Table
SIV, Table SV, Table SVI, Table SVII). Specifically, ECOG PS of 0–1
(HR, 0.15; 95% CI, 0.04–0.56; P=0.005) was an independent
protective factor for PFS in the 1L cohort, while liver metastasis
(HR, 3.99; 95% CI, 1.59–10.00; P=0.003) was an independent risk
factor for PFS in this cohort (Table
SIV and SV). In the 2L cohort,
male sex (HR, 0.41; 95% CI, 0.19–0.87; P=0.020) was an independent
protective factor for PFS (Tables
SVI and SVII).
 | Table III.Univariate and multivariate analyses
of the risk factors for PFS and OS in the 3L+ cohort. |
Table III.
Univariate and multivariate analyses
of the risk factors for PFS and OS in the 3L+ cohort.
|
| PFS | OS |
|---|
|
|
|
|
|---|
|
| Univariate
analysis | Multivariate
analysis | Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|
|
|---|
| Variables | HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, >/≤60
years | 0.61 | 0.010 | 0.58 | 0.041 | 0.97 | 0.890 | - | - |
|
| (0.42–0.88) |
| (0.35–0.98) |
| (0.62–1.52) |
|
|
|
| Sex,
male/female | 0.54 | 0.002 | 0.67 | 0.141 | 0.69 | 0.111 | - | - |
|
| (0.34–0.86) |
| (0.39–1.14) |
| (0.42–1.14) |
|
|
|
| ECOG PS, 0–1/2 | 0.55 | 0.061 | - | - | 0.35 | 0.001 | 0.24 | <0.001 |
|
| (0.23–1.27) |
|
|
| (0.13–0.98) |
| (0.11–0.53) |
|
| Primary site,
left/right | 0.92 | 0.729 | - | - | 0.65 | 0.093 | - | - |
|
| (0.60–1.46) |
|
|
| (0.37–1.16) |
|
|
|
| Liver metastasis,
yes/no | 1.97 | 0.001 | 2.08 | 0.006 | 1.50 | 0.076 | - | - |
|
| (1.35–2.87) |
| (1.24–3.50) |
| (0.96–2.33) |
|
|
|
| Lung metastasis,
yes/no | 1.02 | 0.934 | - | - | 0.87 | 0.547 | - | - |
|
| (0.69–1.49) |
|
|
| (0.56–1.37) |
|
|
|
| RAS/BRAF
status, | 1.62 | 0.031 | 1.83 | 0.018 | 1.68 | 0.048 | 1.75 | 0.046 |
| MT/WT | (1.03–2.54) |
| (1.11–3.02) |
| (0.99–2.87) |
| (1.01–3.03) |
|
| Prior surgery,
yes/no | 0.70 | 0.130 | - | - | 0.61 | 0.069 | - | - |
|
| (0.41–1.19) |
|
|
| (0.32–1.16) |
|
|
|
| Prior
radiotherapy, | 0.77 | 0.171 | - | - | 0.76 | 0.222 | - | - |
| yes/no | (0.53–1.12) |
|
|
| (0.49–1.18) |
|
|
|
| Prior
VEGF(R)/EGFRi, | 1.97 | 0.028 | 2.23 | 0.131 | 1.33 | 0.391 | - | - |
| yes/no | (1.21–3.21) |
| (0.79–6.33) |
| (0.73–2.42) |
|
|
|
Safety
All patients received at least one dose of ICI and
were included in the safety analysis. Most patients (94.6%)
experienced at least one TRAE (Table
IV). However, no unexpected safety concerns were reported. The
most common TRAEs leading to dose reduction and/or treatment
interruption were thrombocytopenia (67/202, 33.2%), leukopenia
(65/202, 32.2%), abnormal liver function (63/202, 31.2%),
electrolyte disturbance (60/202, 29.7%), anemia (59/202, 29.2%) and
thyroid dysfunction (54/202, 26.7%). Grade ≥3 AEs were reported in
88 (43.6%) patients, including thrombocytopenia (19/202, 9.4%),
leukopenia (14/202, 6.9%), neutropenia (11/202, 5.4%), abnormal
hepatic function (10/202, 5.0%) and thyroid dysfunction (8/202,
4.0%). For the incidence of TRAEs in specific ICI-based
combinations, no significant differences were observed among the
1L, 2L and 3L+ cohorts (Table
SVIII). The AEs of the combined ICI and CT regimen were still
mainly bone marrow hematopoietic function inhibition and
gastrointestinal reactions, while the AEs of the ICI-TKI
combination were mainly hand-foot skin reactions, edema,
proteinuria and abnormal thyroid function. The addition of TKI
and/or CT to ICI does not seem to significantly increase the
occurrence and severity of irAEs in each cohort (Table SVIII). But we acknowledge that this
finding is specific to our observation and does not constitute a
direct comparison with ICI monotherapy. The safety profile of each
line of therapy was generally manageable and well-tolerated and no
treatment-related deaths occurred.
 | Table IV.Safety of the treatments. |
Table IV.
Safety of the treatments.
|
| Any grade AE | Grade ≥3 AE |
|---|
|
|
|
|
|---|
| Variables | 1L, n=38 | 2L, n=41 | 3L+, n=123 |
P-valuea | 1L, n=38 | 2L, n=41 | 3L+, n=123 |
P-valuea |
|---|
| All AEs | 37 (97.4) | 38 (92.7) | 116 (94.3) | 0.645 | 17 (44.7) | 23 (56.1) | 48 (39.0) | 0.160 |
| irAEs | 13 (34.2) | 13 (31.7) | 52 (42.3) | 0.400 | 6 (15.8) | 7 (17.1) | 18 (14.6) | 0.929 |
| Leukopenia | 19 (50.0) | 10 (24.4) | 36 (29.3) | 0.028 | 3 (7.9) | 3 (7.3) | 8 (6.5) | 0.952 |
| Neutropenia | 12 (31.6) | 13 (31.7) | 22 (17.9) | 0.078 | 3 (7.9) | 2 (4.9) | 6 (4.9) | 0.761 |
|
Thrombocytopenia | 20 (52.6) | 18 (43.9) | 29 (23.6) | 0.001 | 6 (15.8) | 3 (7.3) | 10 (8.1) | 0.323 |
| Anemia | 17 (44.7) | 16 (39.0) | 26 (21.1) | 0.006 | 1 (2.6) | 2 (4.9) | 3 (2.4) | 0.721 |
| Nausea and/or
vomiting | 10 (26.3) | 8 (19.5) | 21 (17.1) | 0.451 | 0 (0.0) | 1 (2.4) | 0 (0.0) | 0.139 |
| Diarrhea | 4 (10.5) | 7 (17.1) | 18 (14.6) | 0.702 | 0 (0.0) | 1 (2.4) | 5 (4.1) | 0.424 |
| Electrolyte
disturbance | 15 (39.5) | 8 (19.5) | 37 (30.1) | 0.151 | 1 (2.6) | 1 (2.4) | 6 (4.9) | 0.705 |
| Abnormal liver
function | 17 (44.7) | 14 (34.1) | 32 (26.0) | 0.084 | 1 (2.6) | 3 (7.3) | 6 (4.9) | 0.630 |
| Ileus | 0 (0.0) | 2 (4.9) | 12 (9.8) | 0.099 | 0 (0.0) | 0 (0.0) | 3 (2.4) | 0.376 |
| Thromboembolic
event | 1 (2.6) | 1 (2.4) | 3 (2.4) | 0.998 | 0 (0.0) | 1 (2.4) | 1 (0.8) | 0.523 |
| Infusion-related
reaction | 1 (2.6) | 2 (4.9) | 4 (3.3) | 0.844 | 0 (0.0) | 0 (0.0) | 2 (1.6) | 0.523 |
| Neurotoxicity | 8 (21.1) | 6 (14.6) | 10 (8.1) | 0.082 | 0 (0.0) | 0 (0.0) | 0 (0.0) | >0.999 |
| Proteinuria | 6 (15.8) | 6 (14.6) | 19 (15.4) | 0.989 | 0 (0.0) | 2 (4.9) | 4 (3.3) | 0.424 |
| Urine occult
blood | 6 (15.8) | 7 (17.1) | 22 (17.9) | 0.955 | 1 (2.6) | 2 (4.9) | 3 (2.4) | 0.721 |
|
Hypoalbuminemia | 5 (13.2) | 6 (14.6) | 19 (15.4) | 0.941 | 0 (0.0) | 0 (0.0) | 1 (0.8) | 0.724 |
| Hypertension | 2 (5.3) | 5 (12.2) | 23 (18.7) | 0.109 | 0 (0.0) | 0 (0.0) | 1 (0.8) | 0.724 |
| Hyperglycemia | 2 (5.3) | 4 (9.8) | 11 (8.9) | 0.730 | 1 (2.6) | 1 (2.4) | 3 (2.4) | 0.998 |
|
Mucositisb | 3 (7.9) | 4 (9.8) | 16 (13.0) | 0.642 | 0 (0.0) | 1 (2.4) | 1 (0.8) | 0.523 |
| Hoarseness | 6 (15.8) | 5 (12.2) | 27 (22.0) | 0.334 | 0 (0.0) | 1 (2.4) | 0 (0.0) | 0.139 |
| Hand-foot skin
reaction | 3 (7.9) | 8 (19.5) | 39 (31.7) | 0.008 | 1 (2.6) | 2 (4.9) | 4 (3.3) | 0.844 |
| RCCEP | 2 (5.3) | 2 (4.9) | 8 (6.5) | 0.912 | 0 (0.0) | 1 (2.4) | 1 (0.8) | 0.523 |
| Rash | 2 (5.3) | 5 (12.2) | 14 (11.4) | 0.510 | 0 (0.0) | 0 (0.0) | 1 (0.8) | 0.724 |
| Myasthenia
gravis | 0 (0.0) | 0 (0.0) | 2 (1.6) | 0.523 | 0 (0.0) | 0 (0.0) | 1 (0.8) | 0.724 |
| Immune
myositis | 1 (2.6) | 0 (0.0) | 5 (4.1) | 0.410 | 0 (0.0) | 0 (0.0) | 0 (0.0) | >0.999 |
| Cardiac
dysfunction | 3 (7.9) | 5 (12.2) | 11 (8.9) | 0.776 | 1 (2.6) | 0 (0.0) | 2 (1.6) | 0.614 |
| Thyroid
dysfunction | 10 (26.3) | 12 (29.3) | 32 (26.0) | 0.918 | 2 (5.3) | 2 (4.9) | 4 (3.3) | 0.810 |
|
Pneumonitisc | 7 (18.4) | 6 (14.6) | 15 (12.2) | 0.616 | 1 (2.6) | 1 (2.4) | 2 (1.6) | 0.902 |
The incidence of irAEs of any grade in the entire
cohort was 38.6% (78/202). Most of the irAEs were graded 1–2. The
incidence of grade ≥3 irAEs was 15.3% (31/202), including rash,
abnormal liver function and hypothyroidism. All irAEs were
controllable after appropriate symptomatic therapy.
Discussion
To the best of our knowledge, the present study is
the first to explore the real-world ICI-based treatment patterns
and clinical outcomes at different lines of therapy for patients
with pMMR/MSS mCRC. The findings indicated that, unlike
immuno-monotherapy, ICI-based combinations resulted in mixed
results in patients with pMMR/MSS mCRC. An improved survival
outcome can be realized by rational combination of ICI and
chemotherapy and/or targeted agents. Notably, ICI in combination
with TKI and ICI cross-line therapy may be clinically valuable
options for heavily pretreated patients with mCRC at the 3L+
setting.
As is well-known, chemotherapy remains the
cornerstone of mCRC treatment. Fluorouracil based doublet or
triplet chemotherapy combined with anti-VEGF(R) or anti-EGFR
targeted therapy is the standard 1L and 2L therapy for patients
with mCRC, as recommended by the National Comprehensive Cancer
Network (NCCN) Clinical Practice Guidelines in Oncology for
Colorectal Cancer (21,22). The median PFS of 1L and 2L therapy
is reported to be ~12 months and 6 months, respectively (23). Current clinical practice has shown
that the breakthrough of further survival improvement is difficult.
A number of efforts are being explored by researchers, including
screening of benefit population and combining drugs with different
mechanisms of action.
The KEYNOTE-016 study explored the response of
patients with metastatic MSI-H and MSS tumors to pembrolizumab and
the results showed that the ORR of MSI-H patients was as high as
71%, while the ORR of MSS patients was 0%, with a median PFS time
of 2.2 months and OS of 5.0 months (24). Patients with cancer harboring the
dMMR/MSI-H phenotype are considered to be the right population for
ICI therapy. However, the vast majority of patients with mCRC are
pMMR/MSS type with low tumor mutation load and an immune-suppressed
TME (known as ‘cold tumor’) and show a low response to ICIs
(5). However, a series of clinical
studies based on immunotherapy for pMMR/MSS mCRC have also been
widely explored (25–30). The single-arm phase II NIVACOR trial
evaluated the efficacy of nivolumab combined with FOLFOXIRI and
bevacizumab as the 1L treatment for patients with RAS/BRAF-mutated
mCRC (26). The subgroup analysis
of 52 MSS patients showed an ORR of 78.9% and a median PFS of 9.82
months. The BBCAPX study evaluated sintilimab plus CPAEOX and
bevacizumab in the 1L treatment of patients with RAS mutant and MSS
mCRC, noting an ORR of 84% and a median PFS of 18.2 months. In the
study, some CR/PR patients converted into the ‘immune-hot’ subtype
after therapy (27). The AtezoTRIBE
study assessed the efficacy of atezolizumab plus FOLFOXIRI and
bevacizumab vs. FOLFOXIRI plus bevacizumab as the 1L treatment for
mCRC (28). The median PFS time
increased by 1.6 months in the ICI group (13.1 vs. 11.5 months;
P=0.012) in the whole population, but there was no statistical
difference in the PFS time of the pMMR/MSS sub-group (12.9 vs. 11.4
months; P=0.071). The updated data published in the 2023 ASCO
meeting suggested that patients with a high immune score may
benefit from this regimen (29).
The Checkmate 9X8 trial did not show a significant difference in
either the median PFS (11.9 vs. 11.9 months) or OS (29.2 months vs.
not reached) times regarding nivolumab plus standard-of-care (SOC;
FOLFOX and bevacizumab) vs. SOC in the 1L treatment of mCRC;
however, higher PFS rates after 12 months (28 vs. 9%), ORR (60 vs.
46%) and durable responses (12.9 vs. 9.3 months) were observed in
the ICI group (30).
In the present study, the results showed that the
overall ORR of the 1L ICI cohort was 63.2% (95% CI, 46.0–77.7), the
median PFS time was 11.1 months (95% CI, 8.0–14.1) and the OS was
not reached. Compared with the aforementioned prospective studies
that evaluated ICI combined with chemotherapy and anti-angiogenic
targeted therapy as 1L treatment, the PFS time of patients
receiving this triple regimen in the present study was 9.2 months
and the ORR was 63.6%. The data were generally consistent, but also
exhibited certain differences, which were related to multiple
factors such as the nature of each study, sample size, genotype and
ethnicity of the enrolled patients. Additionally, in comparison to
historical data on 1L SOC, the incorporation of ICIs did not result
in a statistically significant extension of PFS time in the
pMMR/MSS subpopulation. This observation is most clearly
exemplified by the subgroup analysis of the AtezoTRIBE study, which
reported a median PFS time of 12.9 months vs. 11.4 months
(P>0.05) (28). Further
investigation and biomarker exploration to identify subgroups of
patients with mCRC that may benefit from ICI-based combinations is
warranted. However, it has recently been shown that a small subset
of patients with MSS CRC harboring POLE and POLD1 proofreading
deficiency respond well to ICI therapy (31). CRC is no longer considered a single
unique disease and has complex molecular subtypes, each of which
have distinct genetic and epigenetic signatures (such as
chromosomal instability and CpG island methylator phenotype)
(32). Incorporating genomic
profiling (such as POLE/POLD1 mutations) into the routine clinical
practice of CRC is technically feasible with existing sequencing
platforms. However, its clinical utility remains limited. These
mutations are rare in MSS mCRC (<2%), making routine screening
inefficient (33). Although they
may predict optimal immunotherapy responses in a small subset,
robust prospective validation is lacking. At present, this approach
is best reserved for clinical trials or comprehensive molecular
profiling in refractory cases. Prioritizing standard biomarkers
(such as programmed death-ligand 1 (PD-L1), tumor mutational burden
and MSI/dMMR) remains the evidence-based standard.
For the 2L treatment of pMMR/MSS mCRC, chemotherapy
consisting of fluorouracil, irinotecan and/or oxaliplatin remains
an important component. In addition, continuing anti-angiogenesis
therapy (such as bevacizumab) beyond PD has been strongly supported
by the data from a series of phase III trials (34–37).
However, the median PFS is still only 5–6 months with this method.
At present, studies on ICI therapy for pMMR/MSS mCRC at the 2L
setting are limited. In the REGOMUNE trial, 5/47 (11%) patients
with MSS mCRC received the regorafenib-avelumab combination as ≥2L
treatment. The PFS of the 43 efficacy-evaluable patients was 3.6
months (95% CI, 1.8–5.4) and the 6-month ORR was 0% (7). In addition, regorafenib monotherapy as
the 2L treatment for RAS-mutant mCRC was explored in the STREAM
study, with an ORR of 10.9% and a PFS of 3.6 months (95% CI,
1.9–6.7) observed (38). In the
present study, the 2L ICI combined with TKI showed a PFS of 6.3
months and an ORR of 14.3%. Therefore, it seems that the 2L
treatment of ICI plus TKI exerts certain advantages in terms of
both median PFS and ORR. However, the survival data for the 2L
setting was not available separately in the REGOMUNE trial.
Additionally, the MSI status of the patients in the STREAM study
was unclear and the treatment was a chemotherapy-free, single-agent
TKI. Therefore, these data cannot be directly compared.
We previously evaluated the efficacy of TKI
(fruquintinib) combined with chemotherapy as a 2L treatment for
patients with pMMR/MSS mCRC. The updated results presented at the
ASCO meeting in 2024 showed a median PFS of 6.9 months (95% CI,
5.2–8.6) and an ORR of 26.0% (95% CI, 17.9–36.2) (39). In the present study, 41 patients
received ICI-based combinations (mainly ICI plus chemotherapy and
targeted therapy) as the 2L treatment and exhibited a PFS time of
6.5 months (95% CI, 4.5–8.6), an OS time of 16.0 months (95% CI,
13.3–18.7) and an ORR of 22.0% (95% CI, 11.1–38.0). Although the
vast majority of the 2L cohort patients also received chemotherapy
and/or targeted agents, the results were no better than the data we
previously reported for 2L therapy without ICIs. It is worth noting
that there may be certain differences between the present
real-world study and the previously published clinical trials.
Baseline characteristics such as the patient's ethnicity, gene
mutation status, comorbidities, different ICIs (programmed cell
death protein 1 or PD-L1 inhibitors), chemotherapies (triplet,
doublet or a single-agent), targeted agents (bevacizumab,
regorafenib or fruquintinib) and various combination strategies are
all factors that need to be taken into account when interpreting
these results.
Clinical trials of ICI-based combinations in the 3L+
treatment for MSS mCRC have been widely developed and have reported
mixed outcomes (6–11). Nivolumab plus regorafenib provided
an ORR of 33% and median PFS time of 7.9 month in 25 patients with
MSS mCRC (6). Pembrolizumab in
combination with lenvatinib showed an ORR of 22% and a median PFS
time of 2.3 months (40).
Durvalumab plus cabozantinib revealed an ORR of 27.6% and a median
PFS time of 4.4 months in 29 heavily treated patients with pMMR/MSS
mCRC (41). Tislelizumab plus
fruquintinib and stereotatic body radiotherapy exhibited an ORR of
26% and a PFS time of 8.5 months (9). Treatment with atezolizumab plus
capecitabine and bevacizumab has shown a median PFS time of 4.4
months in patients with refractory MSS mCRC (42). Tislelizumab combined with
fruquintinib and fecal microbiota transplantation have demonstrated
an ORR of 20% and a median PFS time of 9.6 months (10). In the present study, the results
showed that the ORR of ICI-based strategies for pMMR/MSS mCRC at
the 3L+ setting was 11.4%, the median PFS time was 4.7 months (95%
CI, 4.3–5.2) and the OS was 11.9 months (95% CI, 10.1–13.6). The
ICI-TKI combination accounted for 77.7%, including 60.1% of
ICI+TKI±RT and 17.6% of ICI+TKI+CT±RT. Although there is still a
gap compared with the survival outcomes reported in some of the
above-mentioned clinical trials, as real-world study data, it seems
to be better than the results of 3L treatment drugs such as
regorafenib, fruquintinib and TAS102 recommended by the current
NCCN guidelines for patients with mCRC. The scientific rationality
of TKI combined with ICI is mainly reflected in the following
aspects. On the one hand, small molecule TKIs, particularly
anti-angiogenic TKIs (such as those targeting VEGFR), could
normalize tumor vasculature, improve blood perfusion, decrease
hypoxia, reduce the formation of new blood vessels, induce
immunogenic cell death, activate dendritic cells, enhance T-cell
response and infiltration, directly or indirectly affect checkpoint
expression, reverse immunosuppressive signals that favor ICI
resistance and alter glucose and amino acid metabolism in the TME
(43). Ultimately, TKIs
synergistically enhance the antitumor effect of ICIs by reshaping
the TME into an immune-supportive type. On the other hand, the
positive feedback loop between ICI-induced immune reprogramming and
TKI-mediated normalization of tumor vascular further promotes the
killing and clearance of tumor cells mediated by antitumor
immunity.
In the present study, a directional OS improvement
(14.3 vs. 11.3 months; HR, 0.66; 95% CI, 0.39–1.14; P=0.123) was
observed among patients who received ICI cross-line therapy at the
3+ setting, especially during the initial 3-year follow-up period.
Due to the limited variety of 3L+ drugs available, ICI reinduction
or sequential use is relatively common in 3L+ treated patients with
mCRC without rare mutations. These preliminary results may provide
valuable support for further studies on ICI cross-line therapy in
patients with pMMR/MSS CRC. However, specific biomarkers and
molecular events should be investigated to optimize patient
identification for this ICI-based cross-line strategy.
Patients with colorectal liver metastasis (CRLM) are
historically characterized by low immune responses and poor
survival outcomes (44). Similar
results were observed in the present study. Patients with CRLM in
the 3L+ cohort had a significantly poorer median PFS time (4.0 vs.
8.2 months; P=0.001) than those without liver metastasis. CRLM was
also the independent risk factor most significantly associated with
PFS (HR, 2.08; 95% CI, 1.24–3.50; P=0.006). This may be closely
related to the immunosuppressive TME in patients with CRLM
(45). Animal studies have found
that CRLM can recruit and siphon peripheral circulating
CD8+ T cells to hepatocytes and then interact with
FasL+ monocyte-derived macrophages to drive
antigen-specific Fas+ CD8+ T cell apoptosis,
thereby shaping an immune-excluded TME and mediating ICI resistance
(46). Rational liver
metastases-oriented approaches have been designed to attenuate
immune resistance and stimulate immune response, such as ICI-based
combinations with radiotherapy, radiofrequency ablation,
antiangiogenic agents, cytotoxic drugs, oncolytic viruses, vaccines
and bispecific antibodies.
The safety data of ICI-based combinations in the
present study were consistent with those reported in other clinical
studies (7–10). Most of the TRAEs were graded 1–2 and
all the AEs were manageable. Later-line ICI therapy was not
reported to have a higher incidence of irAEs than front-line ICI
therapy in the present study. Moreover, the occurrence of grade ≥3
irAEs at front-line settings is not the only determining factor for
the subsequent ICI cross-line therapy. This also requires a
comprehensive consideration of the survival benefits and physical
condition of the patient. In addition, improved clinical outcomes
of ICI cross-line therapy were seen in patients with
discontinuation of front-line ICI treatment caused by irAEs other
than by disease progression. Exploration of potential mechanisms
underlying the differences in AEs among various ICI-based
combinations is needed for individualized treatment of clinical
patients.
Several limitations exist in the present study.
First, it was an observational study, which may have introduced
some selection bias. Second, the lack of a control group makes it
challenging to compare the results with other treatment options
directly. Third, due to the retrospective nature of the present
study, recall bias may exist and some results, including AEs, may
have been missed during data collection. Fourth, the PD-L1 and
genomic profiling of most patients were unavailable. Fifth, the
limited sample size reduced the statistical power. Moreover, the
follow-up time was short and the OS of the 1L cohort was not
reached. Last, real-world study outcomes may be affected by a
variety of confounding factors in routine clinical practice, such
as the baseline characteristics and comorbidities of the patients.
Therefore, real-world findings need to be interpreted with caution.
Despite these limitations, the present results provide valuable
information for ICI-based treatment in patients with pMMR/MSS mCRC.
A large-sample prospective study with dynamic biopsy collection as
well as genomic and immunologic profiling should be performed to
validate the present results and identify valuable biomarkers.
In conclusion, the results of the present study
suggest that chemotherapy still dominates 1L and 2L treatment for
pMMR/MSS mCRC and that the addition of ICI does not seem to
significantly extend the median PFS. The 3L+ treatment for pMMR/MSS
mCRC patients mainly consists of ICI plus TKI, demonstrating
manageable safety and improved survival.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
This work was funded by the National Natural Science Foundation
of China (grant no. 82102954).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
DDC and TZ conceived and designed the present study.
WSZ and SJM acquired, analyzed and interpreted the data. WSZ and
SJM performed the statistical analyses and drafted the manuscript.
WSZ obtained funding for the present study. DDC and TZ revised the
manuscript. DDC and TZ confirm the authenticity of all the raw
data. All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the Declaration of Helsinki. The study was approved by the Ethics
Committee of Renmin Hospital of Wuhan University (approval no.
2023K-K067). The need for informed consent was waived by the ethics
committee due to the retrospective nature of the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
Wen-Si Zhao ORCID: https://orcid.org/0000-0002-0610-7701; De-Dong Cao
ORCID: https://orcid.org/0000-0002-5777-4176.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
2
|
Wang S, Zheng R, Li J, Zeng H, Li L, Chen
R, Sun K, Han B, Bray F, Wei W and He J: Global, regional, and
national lifetime risks of developing and dying from
gastrointestinal cancers in 185 countries: A population-based
systematic analysis of GLOBOCAN. Lancet Gastroenterol Hepatol.
9:229–237. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gryfe R, Kim H, Hsieh ET, Aronson MD,
Holowaty EJ, Bull SB, Redston M and Gallinger S: Tumor
microsatellite instability and clinical outcome in young patients
with colorectal cancer. N Engl J Med. 342:69–77. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Boland CR and Goel A: Microsatellite
instability in colorectal cancer. Gastroenterology.
138:2073–2087.e3. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Al Zein M, Boukhdoud M, Shammaa H, Mouslem
H, El Ayoubi LM, Iratni R, Issa K, Khachab M, Assi HI, Sahebkar A
and Eid AH: Immunotherapy and immunoevasion of colorectal cancer.
Drug Discov Today. 28:1036692023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fukuoka S, Hara H, Takahashi N, Kojima T,
Kawazoe A, Asayama M, Yoshii T, Kotani D, Tamura H, Mikamoto Y, et
al: Regorafenib plus nivolumab in patients with advanced gastric or
colorectal cancer: An open-label, dose-escalation, and
dose-expansion phase Ib trial (REGONIVO, EPOC1603). J Clin Oncol.
38:2053–2061. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cousin S, Cantarel C, Guegan JP,
Gomez-Roca C, Metges JP, Adenis A, Pernot S, Bellera C, Kind M,
Auzanneau C, et al: Regorafenib-avelumab combination in patients
with microsatellite stable colorectal cancer (REGOMUNE): A
Single-arm, Open-label, phase II trial. Clin Cancer Res.
27:2139–2147. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gou M, Yan H, E LT, Wang Z, Si H, Chen H,
Pan Y, Fan R, Qian N and Dai G: Fruquintinib combination with
sintilimab in refractory metastatic colorectal cancer patients in
China. J Clin Oncol. 38 (Suppl 15):S40282020. View Article : Google Scholar
|
|
9
|
Yuan X, Zhang M and Hou H: Fruquintinib
combined with tislelizumab and SBRT as a later-line therapy in
microsatellite stability (MSS) metastatic colorectal cancer (mCRC):
Results from the FRUIT trial. J Clin Oncol. 41:1502023. View Article : Google Scholar
|
|
10
|
Zhao W, Lei J, Ke S, Chen Y, Xiao J, Tang
Z, Wang L, Ren Y, Alnaggar M and Qiu H: Fecal microbiota
transplantation plus tislelizumab and fruquintinib in refractory
microsatellite stable metastatic colorectal cancer: An open-label,
single-arm, phase II trial (RENMIN-215). EClinicalMedicine.
66:1023152023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang F, Jin Y, Wang M, Luo HY, Fang WJ,
Wang YN, Chen YX, Huang RJ, Guan WL, Li JB, et al: Combined
anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal
cancer: A randomized phase 2 trial. Nat Med. 30:1035–1043. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Freites-Martinez A, Santana N,
Arias-Santiago S and Viera A: Using the common terminology criteria
for adverse events (CTCAE-Version 5.0) to evaluate the severity of
adverse events of anticancer therapies. Actas Dermosifiliogr (Engl
Ed). 112:90–92. 2021.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fujita K, Yamamoto Y, Kanai O, Okamura M,
Hashimoto M, Nakatani K, Sawai S and Mio T: Retreatment with
anti-PD-1 antibody in non-small cell lung cancer patients
previously treated with anti-PD-L1 antibody. Thorac Cancer.
11:15–18. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nardin C, Hennemann A, Diallo K,
Funck-Brentano E, Puzenat E, Heidelberger V, Jeudy G, Samimi M,
Lesage C, Boussemart L, et al: Efficacy of immune checkpoint
inhibitor (ICI) rechallenge in advanced melanoma patients'
responders to a first course of ICI: A multicenter national
retrospective study of the french group of skin cancers (Groupe de
Cancerologie Cutanee, GCC). Cancers (Basel). 15:35642023.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scheiner B, Roessler D, Phen S, Lim M,
Pomej K, Pressiani T, Cammarota A, Fründt TW, von Felden J, Schulze
K, et al: Efficacy and safety of immune checkpoint inhibitor
rechallenge in individuals with hepatocellular carcinoma. JHEP Rep.
5:1006202023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sun L, Cohen RB, D'Avella CA, Singh AP,
Schoenfeld JD and Hanna GJ: Overall survival, treatment duration,
and rechallenge outcomes with ICI therapy for recurrent or
metastatic HNSCC. JAMA Netw Open. 7:e24285262024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ravi P, Mantia C, Su C, Sorenson K, Elhag
D, Rathi N, Bakouny Z, Agarwal N, Zakharia Y and Costello BA:
Evaluation of the safety and efficacy of immunotherapy rechallenge
in patients with renal cell carcinoma. JAMA Oncol. 6:1606–1610.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gou M, Qian N, Zhang Y, Yan H, Si H, Wang
Z and Dai G: Fruquintinib in combination With PD-1 Inhibitors in
patients with refractory Non-MSI-H/pMMR metastatic colorectal
cancer: A Real-World study in China. Front Oncol. 12:8517562022.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu R, Ji Z, Wang X, Zhu L, Xin J, Ma L,
Zhang J, Ge S, Zhang L, Yang Y, et al: Regorafenib plus sintilimab
as a salvage treatment for microsatellite stable metastatic
colorectal cancer: A single-arm, open-label, phase II clinical
trial. Nat Commun. 16:14812025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Benson AB, Venook AP, Al-Hawary MM, Arain
MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, et
al: Colon cancer, version 2.2021, NCCN clinical practice guidelines
in oncology. J Natl Compr Canc Netw. 19:329–359. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Benson AB, Venook AP, Al-Hawary MM, Azad
N, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D,
Garrido-Laguna I, et al: Rectal cancer, version 2.2022, NCCN
clinical practice guidelines in oncology. J Natl Compr Canc Netw.
20:1139–1167. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ciardiello F, Ciardiello D, Martini G,
Napolitano S, Tabernero J and Cervantes A: Clinical management of
metastatic colorectal cancer in the era of precision medicine. CA
Cancer J Clin. 72:372–401. 2022.PubMed/NCBI
|
|
24
|
Le DT, Durham JN, Smith KN, Wang H,
Bartlett BR, Aulakh LK, Lu S, Kemberling H, Wilt C, Luber BS, et
al: Mismatch repair deficiency predicts response of solid tumors to
PD-1 blockade. Science. 357:409–413. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Baraibar I, Mirallas O, Saoudi N, Ros J,
Salvà F, Tabernero J and Élez E: Combined treatment with
Immunotherapy-based strategies for MSS metastatic colorectal
cancer. Cancers (Basel). 13:63112021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Damato A, Bergamo F, Antonuzzo L, Nasti G,
Pietrantonio F, Tonini G, Maiello E, Bordonaro R, Rosati G,
Romagnani A, et al: Phase II study of nivolumab in combination with
FOLFOXIRI/bevacizumab as first-line treatment in patients with
advanced colorectal cancer RAS/BRAF mutated (mut): NIVACOR trial
(GOIRC-03-2018). J Clin Oncol. 40 (16_suppl):S35092022. View Article : Google Scholar
|
|
27
|
Fang X, Zhu N, Zhong C, Wang L, Li J, Weng
S, Hu H, Dong C, Li D, Song Y, et al: Sintilimab plus bevacizumab,
oxaliplatin and capecitabine as first-line therapy in RAS-mutant,
microsatellite stable, unresectable metastatic colorectal cancer:
An open-label, single-arm, phase II trial. EClinicalMedicine.
62:1021232023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Antoniotti C, Rossini D, Pietrantonio F,
Catteau A, Salvatore L, Lonardi S, Boquet I, Tamberi S, Marmorino
F, Moretto R, et al: Upfront FOLFOXIRI plus bevacizumab with or
without atezolizumab in the treatment of patients with metastatic
colorectal cancer (AtezoTRIBE): A multicentre, open-label,
randomised, controlled, phase 2 trial. Lancet Oncol. 23:876–887.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Antoniotti C, Rossini D, Pietrantonio F,
Salvatore L, Lonardi S, Tamberi S, Marmorino F, Moretto R,
Prisciandaro M, Tamburini E, et al: Upfront fluorouracil,
leucovorin, oxaliplatin, and irinotecan plus bevacizumab with or
without atezolizumab for patients with metastatic colorectal
cancer: Updated and overall survival results of the ATEZOTRIBE
study. J Clin Oncol. 42:2637–2644. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lenz HJ, Parikh A, Spigel DR, Cohn AL,
Yoshino T, Kochenderfer M, Elez E, Shao SH, Deming D, Holdridge R,
et al: Modified FOLFOX6 plus bevacizumab with and without nivolumab
for first-line treatment of metastatic colorectal cancer: Phase 2
results from the CheckMate 9X8 randomized clinical trial. J
Immunother Cancer. 12:e0084092024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ambrosini M, Rousseau B, Manca P, Artz O,
Marabelle A, André T, Maddalena G, Mazzoli G, Intini R, Cohen R, et
al: Immune checkpoint inhibitors for POLE or POLD1
proofreading-deficient metastatic colorectal cancer. Ann Oncol.
35:643–655. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Armaghany T, Wilson JD, Chu Q and Mills G:
Genetic alterations in colorectal cancer. Gastrointest Cancer Res.
5:19–27. 2012.PubMed/NCBI
|
|
33
|
Keshinro A, Vanderbilt C, Kim JK, Firat C,
Chen CT, Yaeger R, Ganesh K, Segal NH, Gonen M, Shia J, et al:
Tumor-Infiltrating lymphocytes, tumor mutational burden, and
genetic alterations in microsatellite unstable, microsatellite
stable, or mutant POLE/POLD1 Colon Cancer. JCO Precis Oncol.
52021.doi: 10.1200/PO.20.00456. PubMed/NCBI
|
|
34
|
Bennouna J, Sastre J, Arnold D, Österlund
P, Greil R, Van Cutsem E, von Moos R, Viéitez JM, Bouché O, Borg C,
et al: Continuation of bevacizumab after first progression in
metastatic colorectal cancer (ML18147): A randomised phase 3 trial.
Lancet Oncol. 14:29–37. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Masi G, Salvatore L, Boni L, Loupakis F,
Cremolini C, Fornaro L, Schirripa M, Cupini S, Barbara C, Safina V,
et al: Continuation or reintroduction of bevacizumab beyond
progression to first-line therapy in metastatic colorectal cancer:
Final results of the randomized BEBYP trial. Ann Oncol. 26:724–730.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tabernero J, Yoshino T, Cohn AL,
Obermannova R, Bodoky G, Garcia-Carbonero R, Ciuleanu TE, Portnoy
DC, Van Cutsem E, Grothey A, et al: Ramucirumab versus placebo in
combination with second-line FOLFIRI in patients with metastatic
colorectal carcinoma that progressed during or after first-line
therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine
(RAISE): A randomised, double-blind, multicentre, phase 3 study.
Lancet Oncol. 16:499–508. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Van Cutsem E, Tabernero J, Lakomy R,
Prenen H, Prausová J, Macarulla T, Ruff P, van Hazel GA, Moiseyenko
V, Ferry D, et al: Addition of aflibercept to fluorouracil,
leucovorin, and irinotecan improves survival in a phase III
randomized trial in patients with metastatic colorectal cancer
previously treated with an oxaliplatin-based regimen. J Clin Oncol.
30:3499–3506. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cardone C, De Stefano A, Rosati G, Cassata
A, Silvestro L, Borrelli M, Di Gennaro E, Romano C, Nappi A,
Zanaletti N, et al: Regorafenib monotherapy as second-line
treatment of patients with RAS-mutant advanced colorectal cancer
(STREAM): An academic, multicenter, single-arm, two-stage, phase II
study. ESMO Open. 8:1007482023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhao W, Li J, Li L, Huang J, Gong W, Yu X,
Le K, He L, Han Q, Wang S, et al: Efficacy and safety of
fruquintinib plus investigator's choice of chemotherapy as
second-line therapy in metastatic colorectal cancer: Updated
results of a multicenter, single-arm, phase 2 trial. J Clin Oncol.
42:35712024. View Article : Google Scholar
|
|
40
|
Gomez-Roca C, Yanez E, Im SA, Alvarez EC,
Senellart H, Doherty M, García-Corbacho J, Lopez JS, Basu B,
Maurice-Dror C, et al: LEAP-005: A phase II multicohort study of
lenvatinib plus pembrolizumab in patients with previously treated
selected solid tumors-Results from the colorectal cancer cohort. J
Clin Oncol. 39:942021. View Article : Google Scholar
|
|
41
|
Saeed A, Park R, Dai J, Taiseer Al-Rajabi
RM, Kasi A, Saeed A, Collins Z, Thompson K, Barbosa L, Mulvaney K,
et al: Phase II trial of cabozantinib (Cabo) plus durvalumab
(Durva) in chemotherapy refractory patients with advanced mismatch
repair proficient/microsatellite stable (pMMR/MSS) colorectal
cancer (CRC): CAMILLA CRC cohort results. J Clin Oncol. 40
(4_Suppl):S1352022. View Article : Google Scholar
|
|
42
|
Mettu NB, Ou FS, Zemla TJ, Halfdanarson
TR, Lenz HJ, Breakstone RA, Boland PM, Crysler OV, Wu C, Nixon AB,
et al: Assessment of capecitabine and bevacizumab with or without
atezolizumab for the treatment of refractory metastatic colorectal
cancer: A randomized clinical trial. JAMA Netw Open.
5:e21490402022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Khan KA and Kerbel RS: Improving
immunotherapy outcomes with anti-angiogenic treatments and vice
versa. Nat Rev Clin Oncol. 15:310–324. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang C, Sandhu J, Ouyang C, Ye J, Lee PP
and Fakih M: Clinical response to immunotherapy targeting
programmed cell death Receptor 1/Programmed cell death ligand 1 in
patients with treatment-resistant microsatellite stable colorectal
cancer with and without liver metastases. JAMA Netw Open.
4:e21184162021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhou H, Liu Z, Wang Y, Wen X, Amador EH,
Yuan L, Ran X, Xiong L, Ran Y, Chen W and Wen Y: Colorectal liver
metastasis: Molecular mechanism and interventional therapy. Signal
Transduct Target Ther. 7:702022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yu J, Green MD, Li S, Sun Y, Journey SN,
Choi JE, Rizvi SM, Qin A, Waninger JJ, Lang X, et al: Liver
metastasis restrains immunotherapy efficacy via macrophage-mediated
T cell elimination. Nat Med. 27:152–164. 2021. View Article : Google Scholar : PubMed/NCBI
|