Introduction
Diffuse large B-cell lymphoma (DLBCL), the most
common aggressive subtype of non-Hodgkin lymphoma (NHL), accounts
for 30–40% of newly diagnosed cases globally. The 5-year overall
survival rates range from 20 to 30% (1). R-CHOP chemotherapy, the standard
first-line immunochemotherapy regimen for diffuse large B-cell
lymphoma (DLBCL) comprising rituximab (R), cyclophosphamide (C),
doxorubicin (H), vincristine (O) and prednisone (P), achieves
durable remission in ~60% of patients. However, 30–40% of patients
develop relapsed or refractory (R/R) disease, with a subsequent
median overall survival of only 6–12 months (2). Conventional chemotherapy is often
associated with poor patient tolerance, prolonged treatment
duration and notable toxicity (2,3).
Research efforts have increasingly focused on addressing these
clinical challenges and improving patient outcomes in DLBCL
(3). Resistance mechanisms to
R-CHOP are multifaceted, including CD20 downregulation, MYC and
BCL-2 co-expression, upregulation of anti-apoptotic proteins,
constitutive activation of NF-κB and ERK pathways and TP53
mutations (4,5). Overcoming this resistance requires
exploring alternative cell death pathways and combining novel
agents, such as natural compounds from traditional medicine, with
conventional therapies such as radiotherapy.
In the era of gene editing and artificial
intelligence, traditional Chinese medicine (TCM) has gained renewed
attention because of its holistic approach and unique therapeutic
wisdom. Previous studies on TCM-derived compounds have revealed
novel avenues of modern medicine. For instance, tanshinone inhibits
vascular endothelial growth factor and angiogenesis (6), safranin modulates gut microbiota and
ameliorates metabolic syndrome (7)
and curcumin has demonstrated anticancer potential via epigenetic
modulation (8,9). Curcumin, a natural polyphenol derived
from the rhizome of Curcuma longa (Zingiberaceae), is the
primary active component of turmeric, which is a TCM (10). Turmeric was first documented in the
‘Xinxiu Ben Cao’ (Newly Revised Materia Medica) during the Tang
Dynasty (11). Curcumin, a natural
polyphenol, curcumin has garnered considerable interest because of
its broad-spectrum antitumor activity (12). It modulates multiple signaling
pathways by inhibiting NF-κB and STAT3, downregulating BCL-2 and
activating caspase-dependent apoptosis (13,14).
Curcumin exhibits a wide range of pharmacological activities,
including anti-inflammatory (15),
antibacterial (16), antioxidant
(17), antitumor (inhibiting
proliferation and metastasis) (8),
radiosensitizing and chemosensitizing (18) and photobiological effects (19). It can also induce multiple modes of
cell death, including apoptosis (20), autophagy (21), necrosis (22), necroptosis (23) and ferroptosis (24). Its anticancer efficacy has been
demonstrated in both solid tumors (such as lung, colorectal and
liver cancers) and hematological malignancies (such as leukemia and
lymphoma) (25,26). Epigenetically, curcumin influences
tumor progression by inhibiting histone acetyltransferases and
modulating microRNA expression (27). Advanced delivery systems (such as
liposomes and nanoparticles) improve its tumor-targeting efficiency
and therapeutic efficacy (28). In
lymphoma, curcumin has been shown to overcome bortezomib resistance
(29) and enhance radiosensitivity
(30), indicating its potential as
a treatment for DLBCL treatment. However, the mechanism by which
curcumin induces non-apoptotic cell death, in particularly
parthanatos, remains poorly understood, and research in this area
is limited. Thus, critical knowledge gaps regarding its application
in DLBCL therapy remain to be addressed.
According to the Cell Death Nomenclature Committee,
cell death can be classified based on morphological, biochemical
and functional criteria (31).
Physiologically regulated programmed cell death includes apoptosis,
necroptosis, parthanatos, autophagy, ferroptosis and cyclophilin
D-dependent necrosis (31).
Parthanatos was first described by Wang et al (32) in 2009. Initially characterized in
the context of neurodegenerative diseases (32–35),
its relevance has been increasingly recognized in oncology, with
roles identified in neuroblastoma (35), lung cancer (36), breast cancer (37), hepatocellular carcinoma (38), colon cancer (5), prostate cancer (39), leukemia (40) and myeloma (41). It is characterized by rapid
poly(ADP-ribose) (PAR) polymerase 1 (PARP-1) activation, PAR
polymer accumulation, apoptosis-inducing factor (AIF)/macrophage
migration inhibitory factor (MIF) nuclear translocation and
extensive DNA fragmentation (42).
Parthanatos is a distinct form of programmed cell death that
depends on PARP-1 and has been previously characterized (31). Unlike apoptosis and necrosis,
parthanatos does not involve cellular swelling, apoptotic body
formation or autophagosome accumulation. It is characterized by
excessive cytoplasmic accumulation of PAR polymers. This
accumulation leads to loss of mitochondrial membrane potential,
translocation of AIF to the nucleus and extensive DNA
fragmentation, resulting in cell death (38). This pathway has been extensively
studied in neurodegenerative diseases (43). In oncology, parthanatos can be
triggered by DNA-damaging agents (such as temozolomide) or PARP
inhibitors, offering a therapeutic advantage in p53-deficient
tumors (5,42). Recent studies have indicated that
natural compounds (such as tanshinone) can induce parthanatos in
leukemia cells (4,6). However, systematic studies of
parthanatos in DLBCL are lacking.
Materials and methods
Cells and reagents
The human DLBCL cell lines SU-DHL-4, SU-DHL-6,
SU-DHL-8, SU-DHL-10 and DoHH2 were provided by Professor Fengting
Liu. Curcumin and Q-VD-OPh were obtained from MedChemExpress and
olaparib was acquired from Selleck Chemicals.
Preparation and storage of
experimental drugs
To ensure consistent drug concentrations and
maintained bioactivity under experimental conditions, the storage
and preparation protocols for all drugs used in the present study
were standardized. Curcumin, Q-VD-OPh and olaparib were dissolved
in dimethyl sulfoxide (DMSO) to prepare stock solutions of 50, 1
and 50 mM, respectively. Aliquots were stored at −80°C in a
cryogenic freezer. Working concentrations were prepared by diluting
in complete RPMI-1640 medium (Gibco; Thermo Fisher Scientific,
Inc.) immediately before use.
Cell thawing and recovery
To ensure that post-thaw cells retained their
original biological functions, morphology and capacity for normal
proliferation and metabolism-thereby providing a stable and healthy
cellular source for subsequent culture and pharmacological
assays-the cryopreservation and thawing procedures were carefully
optimized. The cells were recovered using standard cryopreservation
reversal procedures. Briefly, the water bath was pre-warmed to
37°C. Cryovials containing frozen cells were promptly removed from
the liquid nitrogen and immersed in a water bath with gentle
agitation to accelerate thawing. After thawing, the external
surfaces of the cryovials were wiped with 75% ethanol and
transferred to a biological safety cabinet. The cell suspension was
transferred into a centrifuge tube containing 5 ml pre-warmed
complete medium [RPMI-1640 basal medium + 10% fetal bovine serum
(FBS; ZETA Life) + 1% penicillin-streptomycin (dual antibiotics);
pre-warmed to 37°C), followed by centrifugation at 200 × g for 5
min at room temperature. The supernatant was carefully aspirated
and the cell pellet was gently resuspended in fresh complete medium
(RPMI-1640 basal medium + 10% FBS + 1% penicillin-streptomycin).
The resuspended cells were then transferred into a labeled culture
flask and incubated in an upright position in a humidified
incubator maintained at 37°C with 5% CO2.
Cell culture and passaging
To maintain cells in the logarithmic growth phase
and ensure consistent cell state and absence of contamination
during experiments, cell culture and subculturing were performed
according to the following standard procedures.
Cell lines (SU-DHL-8, SU-DHL-10 and DoHH2) were
maintained in RPMI-1640 (Gibco; Thermo Fisher Scientific, Inc.)
medium containing 10% FBS (ZETA Life) and 1%
penicillin-streptomycin at 37°C in a humidified 5% CO2
incubator. The cells were routinely monitored for growth, medium
clarity and signs of contamination. Depending on the cell
condition, the culture medium was replaced every 2–3 days to
prevent bacterial contamination. All cell lines used in the present
study were suspension cultures. At a high density, the cells formed
aggregates and the medium became turbid upon gentle agitation. The
cells were subcultured or diluted when reaching to appropriate
densities to maintain exponential growth.
Cell proliferation activity assay:
Cell Counting Kit-8 (CCK-8) method
To evaluate the inhibitory effect of curcumin on
DLBCL cell proliferation and calculate its IC50 values,
the following procedures were performed: SU-DHL-8 and SU-DHL-10
cells were seeded in 24-well plates at 1×105 cells/well
(n=5). The cells were then treated with increasing concentrations
of curcumin (0, 10, 20, 40 and 60 µM). After 24 h, cell viability
was measured using a CCK-8 assay according to the manufacturer's
protocol. Specifically, 10 µl CCK-8 solution was added to each well
and incubated for 1–1.5 h. Absorbance was measured at 450 nm using
a microplate reader. Cell viability was calculated as:
(ODsample-ODblank)/(ODcontrol-ODblank) ×100%, with control groups
set as 100% viability.
Cellular protein extraction
Following treatment, cells were collected by
centrifugation at 200 × g for 5 min at 4°C. The supernatant was
discarded, and the cell pellet was washed with PBS, transferred to
a new microcentrifuge tube and centrifuged again under the same
conditions. The supernatant was then carefully removed. The cell
pellet was resuspended in 70–110 µl ice-cold lysis buffer (Beyotime
Biotechnology) supplemented with 1% protease and phosphatase
inhibitors. The suspension was vortexed for 15 sec at 5 min
intervals and incubated on ice for 30 min. The lysate was
centrifuged at 15,000 × g for 20 min at 4°C. The supernatant was
collected, and the protein concentration was determined using a BCA
protein assay kit (cat. no. 23227; Thermo Fisher Scientific, Inc.).
Protein samples were mixed with 5X loading buffer (1:4 ratio) and
denatured by heating at 95°C for 10 min. Samples were either used
immediately or stored at −80°C for subsequent western blot analysis
of relevant protein expression.
Cytoplasmic and nuclear protein
isolation
Treated cells were collected by centrifugation at
200 × g for 5 min at 4°C. After discarding the medium, the cell
pellet was washed with PBS, transferred to a new tube and
centrifuged as previously described. The supernatants were
carefully aspirated. The pellet was resuspended in 200 µl ice-cold
Cytoplasmic Extraction Reagent A (cat. no. P0028-1; Beyotime
Biotechnology) containing 1% protease/phosphatase inhibitors,
vortexed vigorously for 5 sec and incubated on ice for 15 min.
Cytoplasmic Extraction Reagent B (10 µl) (cat. no. P0028-2;
Beyotime Biotechnology) was added, followed by vortexing for 5 sec,
incubation on ice for 1 min and vigorous shaking for 1 min. The
tube was vortexed again for 5 sec and centrifuged at 14,000 × g for
10 min at 4°C. The cytoplasmic supernatant was transferred to a
pre-chilled tube for immediate use or storage at −80°C. The nuclear
pellet was washed three times with 200 µl ice-cold PBS by
centrifugation at 3,000 × g (4°C). The pellet was resuspended in 50
µl Nuclear Extraction Reagent (cat. no. P0028-3; Beyotime
Biotechnology) with inhibitors and vortexed vigorously for 15–30
sec. The suspension was incubated on ice with intermittent
vortexing (15–30 sec every 1–2 min) for 30 min. After
centrifugation at 14,000 × g for 10 min (4°C), the nuclear extract
(supernatant) was collected. The protein concentrations of the
cytoplasmic and nuclear fractions were determined separately using
a BCA assay. The fractions were mixed with 5X loading buffer (1:4)
and denatured at 95°C for 10 min. Samples were used immediately or
stored at −80°C for analysis of nuclear translocation of proteins
such as AIF and MIF.
Trypan blue exclusion assay
Cell suspensions were mixed with an equal volume of
0.4% trypan blue solution. A 10 µl aliquot was loaded onto a
hemocytometer, incubated for 3 min at room temperature and viable
cells were counted using an automated cell counter. Cell viability
was expressed as a percentage of the control group (set as 100%).
This was conducted for direct evaluation of the effects of curcumin
and its combined treatments on cell viability.
Protein concentration determination by
BCA assay
Protein concentrations were determined using the BCA
assay to ensure consistent sample loading in western blot and
related experiments, as detailed in the following protocol.
Briefly, a working solution was prepared by mixing
the BCA reagents A and B in a 50:1 ratio. A standard curve was
generated by adding 0–20 µg standard protein to a 96-well plate,
followed by volume adjustment to 25 µl with deionized water.
Subsequently, 2 µl each protein sample was loaded into the plate
and also adjusted to 25 µl with deionized water. Then, 200 µl BCA
working solution was added to each well, followed by incubation at
37°C for 30 min protected from light. Finally, the absorbance was
measured at 562 nm using a microplate reader, and sample protein
concentrations were calculated based on a standard curve.
Western blot
Western blot was performed to detect the expression
and localization changes of PAR polymers, PARP-1, AIF, MIF and
apoptosis- and necroptosis-related proteins, following the detailed
procedures below.
Whole-cell lysates were prepared using lysis buffer
(Sigma-Aldrich; Merck KGaA) with 1% protease/phosphatase inhibitors
on ice for 30 min, followed by centrifugation at 14,000 × g for 30
min at 4°C. Protein concentration was determined using the BCA
method. Nuclear and cytoplasmic fractions were isolated using a
commercial extraction kit (cat. no. P0028; Beyotime Biotechnology)
according to the manufacturer's instructions. Proteins (20 µg) were
separated using 7.5, 10 and 12.5% SDS-PAGE gels according to their
molecular sizes and transferred to PVDF membranes. Membranes were
blocked with 5% skim milk and incubated with primary antibodies at
4°C overnight. After washing with TBST (0.1% Tween, 3×10 min), the
membranes were incubated with horseradish peroxidase-conjugated
secondary antibodies (1:5,000; cat. nos. AS014 and AS0003; ABclonal
Biotech. Co., Ltd.) for 1 h at room temperature. Signals were
detected using an ECL substrate and a chemiluminescence imaging
system (Tanon). The following primary antibodies were used: GAPDH
(1:10,000; cat. no. 600-GAPDHt; Trevigen Inc.), AIF (1:1,000; cat.
no. #4642; Cell Signaling Technology, Inc.), MIF (1:1,000; cat. no.
ab7207; Abcam), PAR (1:1,000; cat. no. #84510; Cell Signaling
Technology, Inc.), PARP (1:1,000; cat. no. #9542; Cell Signaling
Technology, Inc.), BCL-2 (1:1,000; cat. no. #3498; Cell Signaling
Technology, Inc.), BCL-6 (1:1,000; cat. no. WL03134; Wanleibio Co.,
Ltd.), c-Myc (1:1,000; cat. no. A5011; Selleck Chemicals), Bax
(1:1,000; cat. no. A19684; ABclonal Biotech Co., Ltd.), receptor
interacting protein 1 (RIP1; 1:1,000; cat. no. #3493; Cell
Signaling Technology, Inc.), receptor interacting protein 3 (RIP3;
1:1,000; cat. no. #13526; Cell Signaling Technology, Inc.), mixed
lineage kinase domain like pseudokinase (MLKL; 1:1,000; cat. no.
#14993; Cell Signaling Technology, Inc.), cleaved caspase-3
(1:1,000; cat. no. #9661; Cell Signaling Technology, Inc.) and
caspase-3 (1:1,000; cat. no. #9662; Cell Signaling Technology,
Inc.). Band intensities from three independent experiments were
quantified using ImageJ 1.53t software (National Institute of
Health).
Ultraviolet B (UVB) irradiation
UVB irradiation was employed to mimic the
DNA-damaging stimuli of radiotherapy, thereby investigating the
synergistic effects of curcumin combined with ultraviolet
treatment. A Bio-Rad UVB irradiation system (Bio-Rad Laboratories,
Inc.) was used, which emits ultraviolet light at wavelengths of
280–320 nm to generate UVB radiation. Before UVB exposure, the
irradiation device was thoroughly sterilized with 75% ethanol.
Subsequently, the cells were covered with 6-well plate lids and
placed on a UV-transparent tray and irradiated at an intensity of
50 mJ/cm2. Following UVB exposure, the cells were
returned to the incubator for further incubation, allowing
subsequent experiments to proceed. To ensure consistency and
comparability in the present study and based on preliminary
research findings (data not shown), the UVB irradiation time for
SU-DHL-8 cells across all groups was set at 60 sec, whereas for
SU-DHL-10 cells it was set at 120 sec across all groups.
Immunofluorescence staining
Immunofluorescence staining was conducted to
directly visualize the subcellular localization of target
proteins.
SU-DHL-10 cells (1×106 cells/ml) were
seeded in 6-well plates and treated as indicated for 24 h. Cells
were fixed with 4% paraformaldehyde (20 min, room temperature),
permeabilized with 0.5% Triton X-100 (20 min) and washed with PBST.
After blocking with 5% BSA (15 min, room temperature), cells were
incubated with primary antibodies (AIF, cat. no. #4642; 1:50;
PARP-1, cat. no. #9542; 1:200; Cell Signaling Technology, Inc.)
overnight at 4°C. The cells were then incubated with an
FITC-conjugated secondary antibody (1:200; cat. no. AS014; ABclonal
Biotechnology Co., Ltd.) for 1 h at room temperature counterstained
with DAPI and mounted with an anti-fade reagent. Images were
acquired using a fluorescence microscope (Nikon Corporation).
DAPI/PI staining
To distinguish between apoptotic and non-apoptotic
cell death, cell death modalities were assessed via nuclear
staining and membrane integrity markers. SU-DHL-10 cells
(1×106 cells/ml) were seeded in 6-well plates, treated
as indicated and incubated for 24 h. The cells were harvested and
incubated in the dark at room temperature for 20 min with a
solution containing DAPI (1:100 in PBS; Beijing Solarbio Science
& Technology Co., Ltd.) and propidium iodide (PI; 1:20 in PBS;
Beyotime Biotechnology). The cells were then washed three times
with PBS. The stained cells were visualized using a fluorescence
microscope (Nikon Corporation).
Statistics
Statistical analyses were performed using SPSS 22.0
(IBM Corp.) and GraphPad Prism 8.0.1 (Dotmatics). For comparisons
among multiple groups, one-way analysis of variance (ANOVA) was
used, followed by Tukey's HSD test for post hoc pairwise
comparisons. Data are presented as the mean ± SD from at least
three independent experiments. A P-value of <0.05 was considered
statistically significant, and a P-value of <0.01 was considered
highly statistically significant. All experiments were performed at
least in triplicate.
Results
Curcumin induces non-apoptotic cell
death
The effect of curcumin on DLBCL cell viability was
assessed using the CCK-8 assay. Cells were treated with curcumin
(0, 10, 20, 40 or 60 µmol/l) for 24 h. Curcumin significantly
inhibited the viability of SU-DHL-8 and SU-DHL-10 cells in a
concentration-dependent manner, with IC50 values of
22.12 and 39.22 µmol/l, respectively (Fig. 1A). To distinguish apoptotic from
non-apoptotic cell death, the pan-caspase inhibitor Q-VD-OPh was
used (44,45). Based on preliminary data and
literature (45), cells were
treated with 20 nmol/l Q-VD-OPh and 10 or 20 µmol/l curcumin for 24
h. In SU-DHL-8 cells, co-treatment with Q-VD-OPh (Cur20 + Q)
significantly rescued cell viability compared with treatment with
curcumin alone (Cur20). By contrast, Q-VD-OPh had no protective
effect on SU-DHL-10 cells (Fig.
1B). Consistently, DAPI/PI staining showed a significant
increase in stained cells after curcumin (20 µmol/l) treatment in
both cell lines. This increase was attenuated by Q-VD-OPh in
SU-DHL-8 cells, but not in SU-DHL-10 cells (Fig. 1C and D), indicating that
curcumin-induced death in SU-DHL-10 cells was largely
caspase-independent.
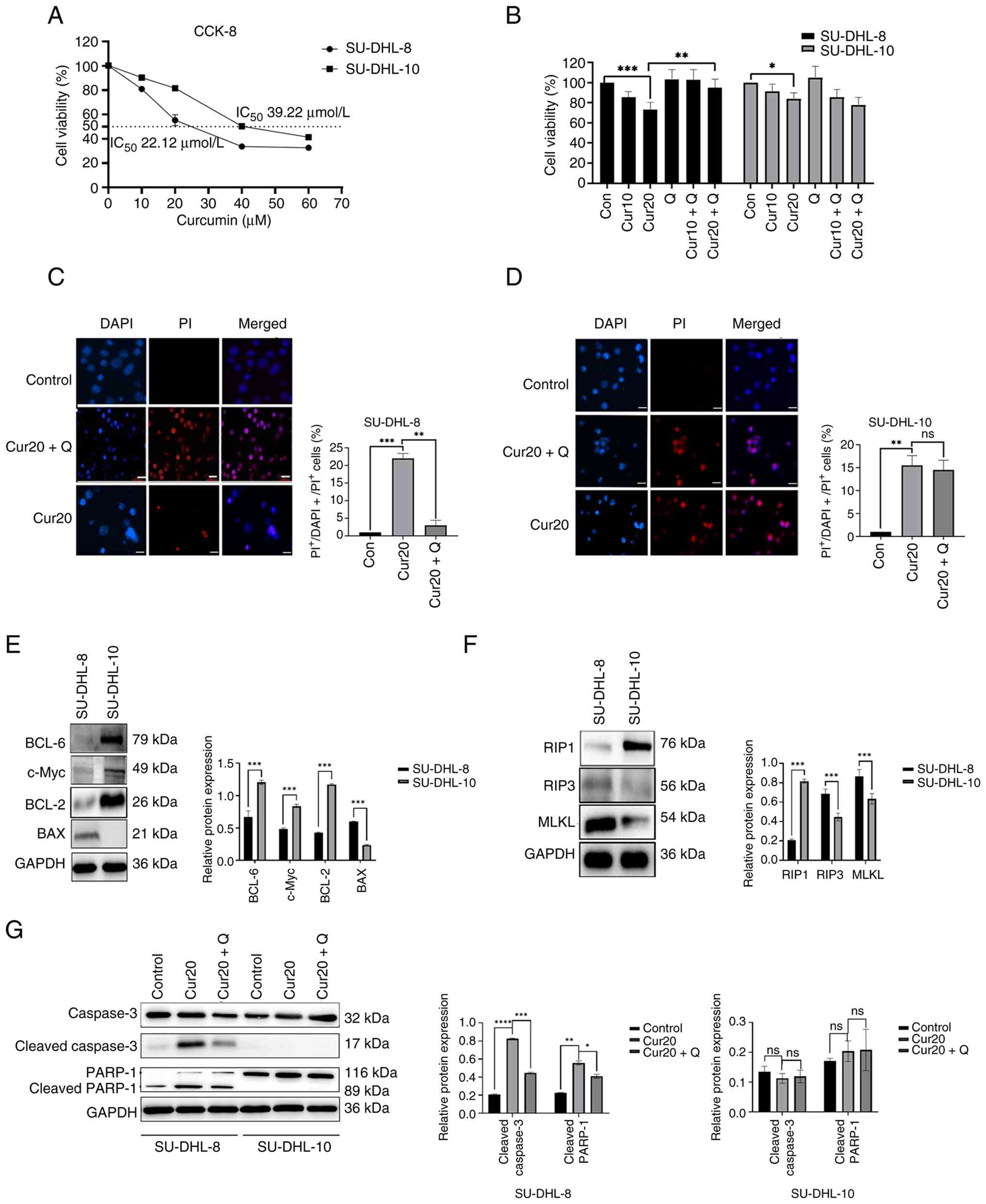 | Figure 1.Curcumin induces non-apoptotic cell
death. (A) Percentage cell viability after 0, 10, 20, 40 and 60 µM
curcumin treatment for 24 h. (B) Cell viability percentage with or
without curcumin or Q-VD-OPH for 24 h. Representative images of
DAPI+/PI+ staining in (C) SU-DHL-8 and (D)
SU-DHL-10 cells treated with curcumin ± Q-VD-OPH for 24 h. The
percentage of DAPI+/PI+ staining cell in
SU-DHL-8 and SU-DHL-10 cells treated with curcumin ± Q-VD-OPH for
24 h. (E) The protein levels of BAX, BCL-2, BCL-6 and c-Myc in
SU-DHL-8 and SU-DHL-10 cells. (F) The protein levels of RIP1, RIP3
and MLKL in SU-DHL-8 and SU-DHL-10 cells. (G) The protein levels of
cleaved caspase-3 and cleaved PARP-1 of SU-DHL-8 and SU-DHL-10
cells treated with 20 µM curcumin for 24 h. Scale bar, 25 µm.
*P<0.05, **P<0.01,***P<0.001 and ****P<0.0001; ns,
P≥0.05. Con, blank control group; Cur10, curcumin 10 µmol/l; Cur20,
curcumin 20 µmol/l; Q, Q-VD-OPH; RIP, receptor interacting protein;
PI, propidium iodide; MLKL, mixed lineage kinase domain like
pseudokinase; PARP-1, poly(ADP-ribose) polymerase-1. |
To investigate the mechanism of curcumin-induced
cell death, the basal expression of c-Myc, BAX, BCL-2 and BCL-6 was
examined. The effects of curcumin on caspase 3 and PARP-1 cleavage
were assessed using western blotting. SU-DHL-10 cells lacked BAX
expression, but showed higher levels of BCL-2, BCL-6 and c-Myc than
SU-DHL-8 cells (Fig. 1E). Upon
curcumin treatment, cleaved caspase-3 and cleaved PARP-1 levels
significantly increased in SU-DHL-8 cells. This effect was
suppressed by Q-VD-OPh. By contrast, cleaved caspase-3 and cleaved
PARP-1 were undetectable in SU-DHL-10 cells under all conditions
(Fig. 1G). These results confirmed
that curcumin induces caspase-dependent apoptosis in SU-DHL-8 cells
but triggers a non-apoptotic cell death pathway in SU-DHL-10 cells.
Although RIP1 expression was higher in SU-DHL-10 cells, the key
necroptosis mediators RIP3 and MLKL were expressed at very low
levels (Fig. 1F), which is
consistent with a previous report (46). Thus, necroptosis is unlikely to
contribute to cell death in SU-DHL-10 cells.
Curcumin causes the increase of PAR
and upregulation of nuclear PARP1, AIF and MIF and nuclear
translocation of AIF in DLBCL cells
To investigate the role of parthanatos, SU-DHL-10
cells were treated with 20 µM curcumin for 24 h, and the levels of
PAR polymers were measured. The PARP-1 inhibitor olaparib (10 µM)
and the pan-caspase inhibitor Q-VD-OPh (20 nM) were used for
mechanistic validation. Western blot analysis showed a significant
increase in PAR polymer levels in curcumin-treated SU-DHL-10 cells
compared with those in the untreated control group (Fig. 2A and B). This increase was
significantly suppressed by olaparib (Cur20 + Ola), but not by
Q-VD-OPh (Cur20 + Q) (Fig. 2A and
B). Furthermore, curcumin treatment significantly increased the
nuclear protein levels of PARP-1, AIF and MIF, which were reversed
by olaparib (Fig. 2C and D).
Immunofluorescence staining consistently revealed the enhanced
nuclear localization of both AIF and PARP-1 after curcumin
treatment, which was attenuated by olaparib treatment (Fig. 2E and F).
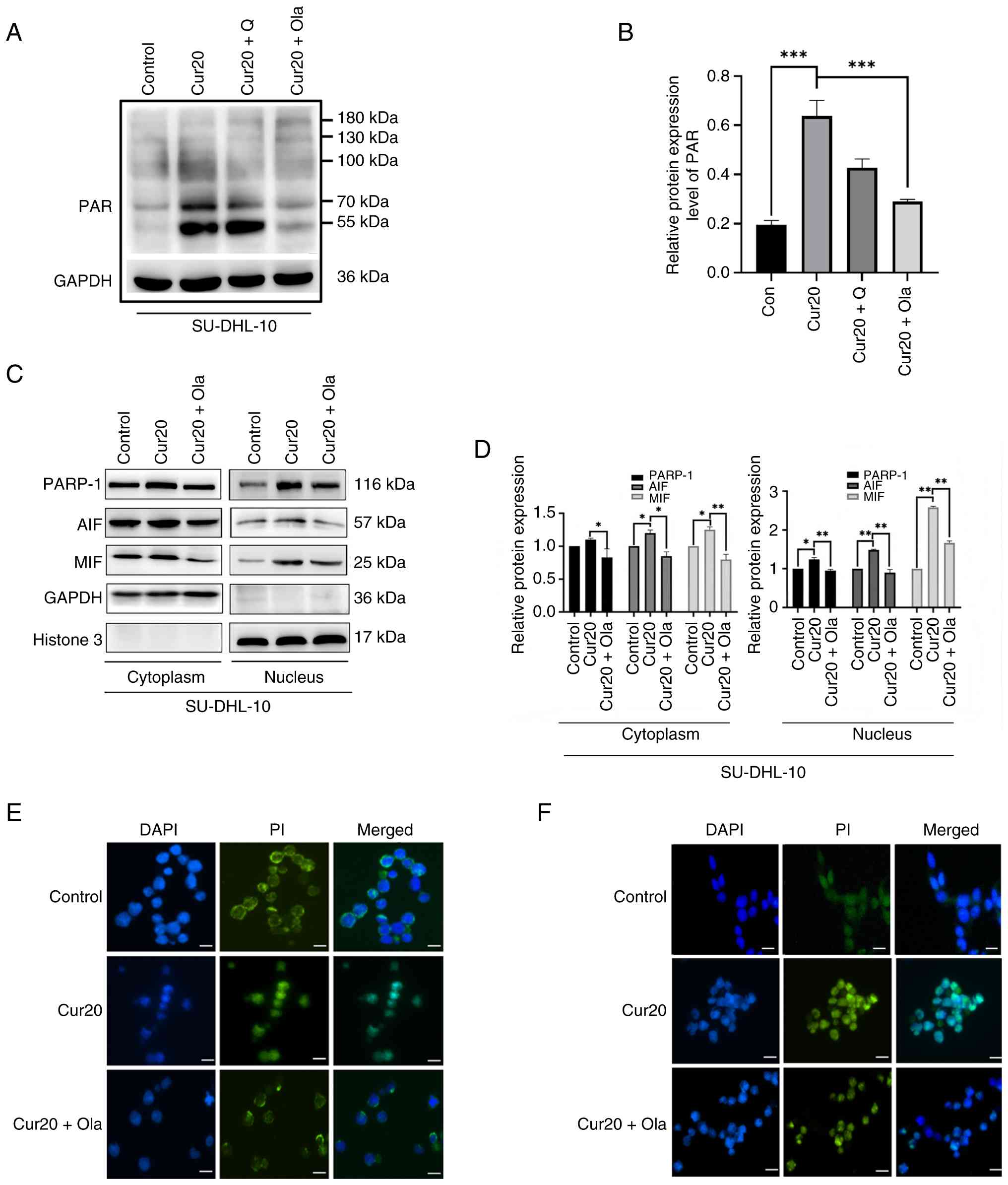 | Figure 2.Curcumin causes the increase of PAR
and upregulation of nuclear PARP-1, AIF and MIF, and nuclear
translocation of AIF in DLBCL cells. (A) The protein level of PAR
polymers in the SU-DHL-10 cells treated with 20 µM curcumin ± 20 nM
Q-VD-OPh or 10 µM olaparib for 24 h (B) Relative protein expression
levels of PAR in the SU-DHL-10 cells treated with 20 µM curcumin ±
20 nM Q-VD-OPh or 10 µM olaparib for 24 h. (C) The protein levels
of PARP-1, AIFand MIF in the nucleus and cytoplasm of SU-DHL-10
cells treated with 20 µM curcumin ± 10 µM olaparib for 24 h. (D)
Relative protein expression levels of PARP-1, AIF and MIF in the
nucleus and cytoplasm of SU-DHL-10 cells treated with 20 µM
curcumin ± 10 µM olaparib for 24 h. (E) Representative images of
AIF immunofluorescence staining in SU-DHL-10 cells treated with 20
µM curcumin for 24 h. (F) Representative images of PARP-1
immunofluorescence staining in SU-DHL-10 cells treated with 20 µM
curcumin for 24 h. *P<0.05, **P<0.01 and ***P<0.001; ns,
P≥0.05. Scale bar, 25 µm. Cur20, curcumin 20 µmol/l; Q, Q-VD-OPH;
Ola, olaparib; DLBCL, diffuse large B-cell lymphoma; PAR,
poly(ADP-ribose); PARP-1, poly(ADP-ribose) polymerase-1; AIF,
apoptosis-inducing factor; MIF, macrophage migration inhibitory
factor; PI, propidium iodide. |
Curcumin combined with UVB induces
non-apoptotic death
SU-DHL-10 cells are relatively insensitive to
DNA-damaging stimuli such as UVB (47) (Fig.
3A). To explore the combined effects of radiotherapy, SU-DHL-10
cells were treated with a low-dose of curcumin and UVB irradiation
for 24 h. The CCK-8 assay showed that combined treatment with
curcumin (10 or 20 µM) and UVB significantly reduced cell viability
compared with single-agent treatments (Fig. 3B). Notably, the addition of Q-VD-OPh
to the curcumin (10 µM) and UVB combination further decreased cell
survival in the trypan blue exclusion assay (Fig. 3C). DAPI/PI staining confirmed a
synergistic increase in cell death with the Cur10 + UV combination,
which was not inhibited by Q-VD-OPh (Fig. 3D and E). Western blotting analysis
confirmed the absence of cleaved caspase-3 and cleaved PARP-1 in
cells treated with curcumin alone or in combination with UVB
(Fig. 3F).
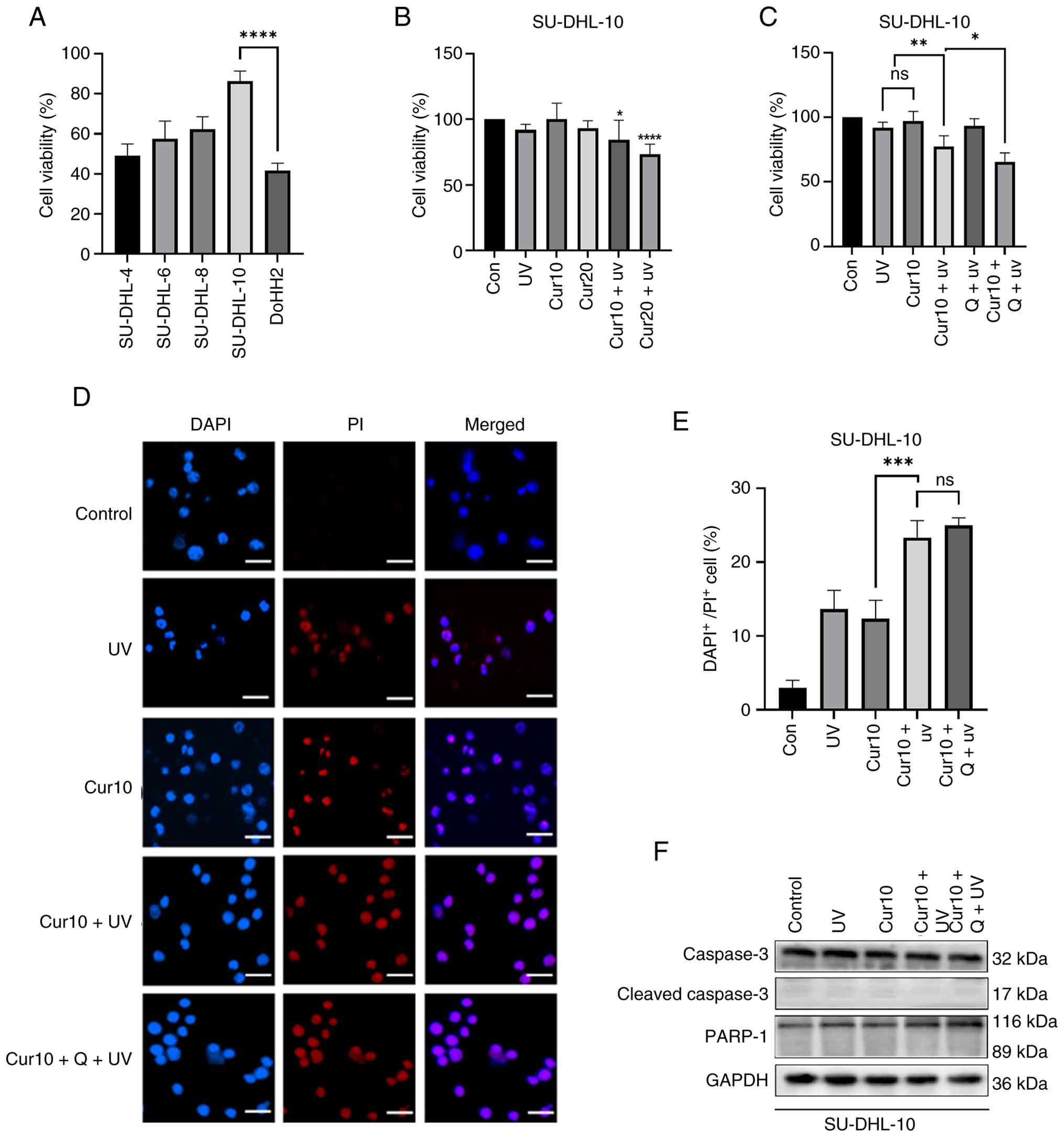 | Figure 3.Curcumin combined with UVB induces
non-apoptotic death. (A) Cell survival rates of different cell
lines after UVB treatment. (B) Percentage of cell viability after
10 and 20 µM curcumin ± UVB treatment for 24 h. (C) Percentage of
cell viability after 10 µM curcumin ± UVB or 20 nM Q-VD-OPh
treatment for 24 h. (D) Representative images of
DAPI+/PI+ staining in cells treated with
curcumin ± UVB or 20 nM Q-VD-OPh for 24 h. (E) The percentage of
DAPI+/PI+ staining cell in cells treated with
10 µM curcumin ± UVB or 20 nM Q-VD-OPh for 24 h. (F) The protein
levels of cleaved caspase 3 and cleaved PARP-1 of cells treated
with 10 µM curcumin ± UVB or 20 nM Q-VD-OPh for 24 h. *P<0.05,
**P<0.01, ***P<0.001, ****P<0.0001 and ns, P≥0.05. Scale
bar, 25 µm. Cur10, curcumin 10 µmol/l; Q, Q-VD-OPH; UVB,
ultraviolet B radiation; PI, propidium iodide, PARP-1,
poly(ADP-ribose) polymerase-1. |
Curcumin sensitizes the antitumor
effects of UVB through parthanatos
To determine whether parthanatos mediated the
combined effect, PAR polymer expression was evaluated in SU-DHL-10
cells treated with curcumin and UVB. The PAR polymer levels were
significantly higher in the Cur10 + UV group than in either
treatment alone. Olaparib, but not Q-VD-OPh, effectively suppressed
this increase (Fig. 4A and B).
Furthermore, the nuclear levels of PARP-1, AIF and MIF were
significantly elevated by the Cur10 + UV combination compared with
the single treatments, and this elevation was reversed by olaparib
(Fig. 4C and D). Immunofluorescence
staining confirmed the enhanced nuclear translocation of AIF and
PARP-1 induced by the Cur10 + UV combination, which was inhibited
by olaparib (Fig. 4E and F).
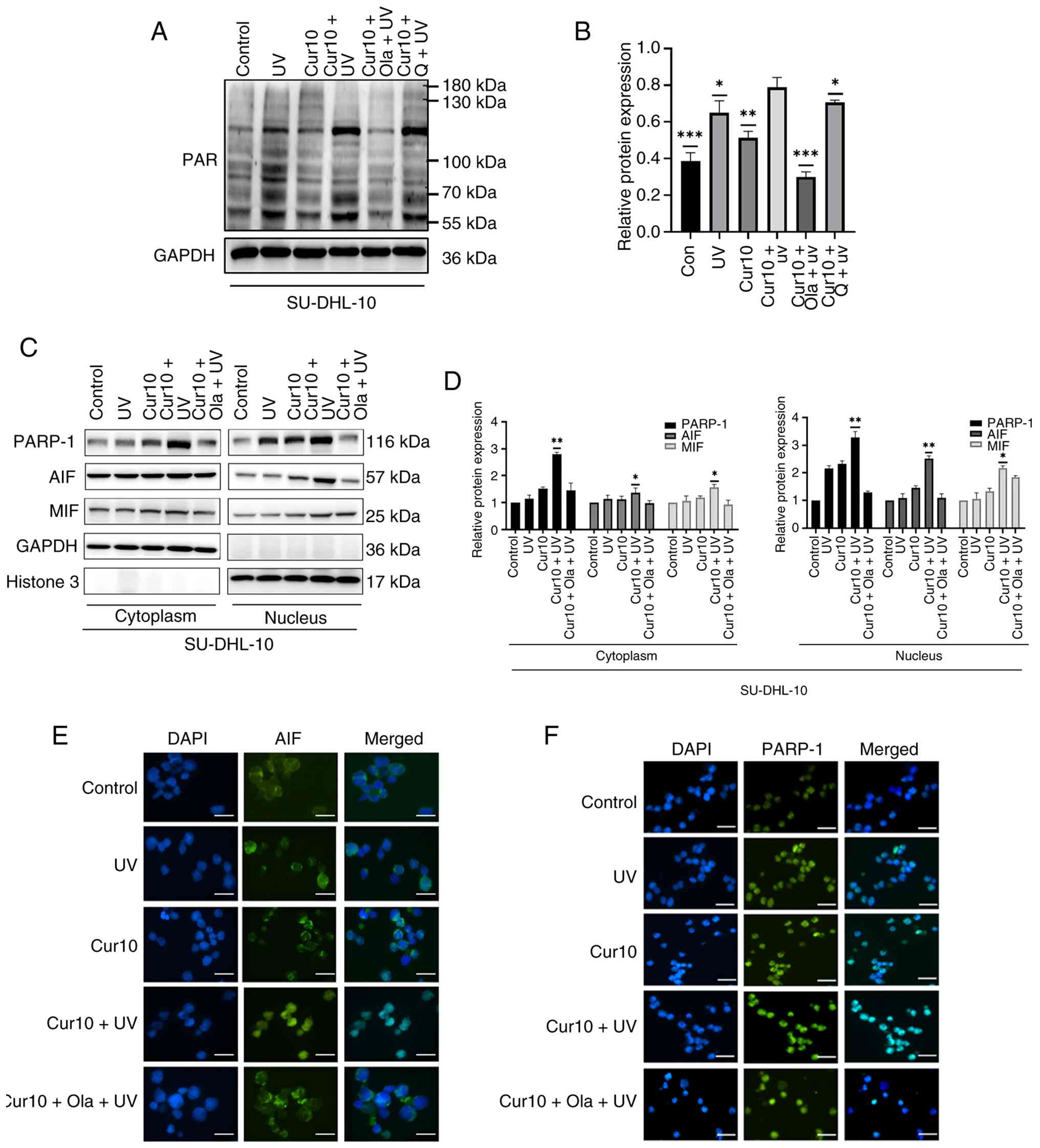 | Figure 4.Curcumin sensitizes the antitumor
effects of UVB through parthanatos. (A) The protein level of PAR
polymers in the SU-DHL-10 cells treated with 10 µM curcumin
combined UVB ± 20 nM Q-VD-OPh or 10 µM olaparib treatment for 24 h.
(B) Relative protein expression levels of PAR in the SU-DHL-10
cells treated with 10 µM curcumin combined UVB ± 20 nM Q-VD-OPh or
10 µM olaparib treatment for 24 h. (C) The protein level of PARP-1,
AIF and MIF in the nucleus and cytoplasm of SU-DHL-10 cells treated
with 10 µM curcumin combined UVB ± 20 nM Q-VD-OPh or 10 µM olaparib
treatment for 24 h. (D) Relative protein expression levels of
PARP-1, AIF and MIF in the nucleus and cytoplasm of SU-DHL-10 cells
treated with 10 µM curcumin combined UVB ± 20 nM Q-VD-OPh/10 µM
olaparib treatment for 24 h. (E) Representative images of AIF
immunofluorescence staining in SU-DHL-10 cells treated with 10 µM
curcumin combined UVB ± 20 nM Q-VD-OPh/10 µM olaparib treatment for
24 h. (F) Representative images of PARP-1 immunofluorescence
staining in SU-DHL-10 cells treated with 10 µM curcumin combined
UVB ± 20 nM Q-VD-OPh/10 µM Olaparib treatment for 24 h. Scale bar,
25 µm. *P<0.05, **P<0.01 and ***P<0.001; ns, P≥0.05. Scale
bar, 25 µm. Cur10, curcumin 10 µmol/l; Q, Q-VD-OPH; Ola, olaparib;
UVB, ultraviolet radiation B; PAR, poly (ADP-ribose); PARP-1, poly
(ADP-ribose) polymerase-1; AIF, apoptosis-inducing factor; MIF,
macrophage migration inhibitory factor. |
Discussion
In the present study, curcumin reduced the survival
rate of SU-DHL-8 and SU-DHL-10 cells in a dose-dependent manner,
and low-dose curcumin enhanced the cytotoxic effects of
DNA-damaging agents (UVB irradiation) on the cells, consistent with
the findings of Wang et al (48). In the CCK-8 assay, the cell survival
rates in the SU-DHL-8 Cur10 and SU-DHL-10 Cur20 groups were
significantly lower than those in the control group. Although a
decrease was observed in the trypan blue exclusion assay, the
difference was not statistically significant. The difference in the
experimental principles between the two methods lies in the fact
that cells that have lost viability or membrane integrity exhibit
increased membrane permeability and are stained blue by trypan
blue, whereas normal live cells with intact membrane structures
exclude trypan blue, preventing it from entering the cell. In the
CCK-8 assay, WST-8 was reduced by mitochondrial dehydrogenases in
the presence of the electron carrier 1-methoxy-5-methylphenazinium
methyl sulfate to form an orange-yellow soluble called formazan.
The amount of formazan generated was proportional to the number and
activity of viable cells. When the cell membrane is mildly damaged,
but not to an extent that affects cell survival, the cells may not
be stained or only lightly stained with trypan blue. Automated
counting machines may consider these cells normal rather than
damaged, resulting in the trypan blue exclusion assay being less
sensitive than the CCK-8 assay (49,50).
Furthermore, previous studies have shown that the CCK-8 assay is
more sensitive than the trypan blue exclusion assay, which is
consistent with the present experimental results.
Apoptosis resistance is a major clinical obstacle
that contributes to the failure of traditional cancer therapies
such as chemotherapy and radiotherapy, as well as the development
of multidrug resistance. Developing novel anticancer agents or drug
combination strategies that bypass the apoptotic pathway to induce
cancer cell death is considered an effective approach to overcome
this challenge (51,52). The present experimental results
indicated that SU-DHL-10 cells did not express the crucial
apoptosis-promoting protein BAX, yet exhibited high expression
levels of c-Myc and the anti-apoptotic proteins BCL-2 and BCL-6.
Curcumin was capable of inducing SU-DHL-10 cells death, and this
process was not inhibited by the broad-spectrum caspase inhibitor
Q-VD-OPh. The key molecular markers of apoptosis, cleaved caspase-3
and cleaved PARP-1, were not detected during this process.
Furthermore, although SU-DHL-10 cells showed high of RIP1
expression, they also showed low RIP3 and MLKL expression. This
finding is consistent with previous literature reporting that
SU-DHL-10 does not express RIP3 or MLKL (38). RIP1, phosphorylated RIP3 and
phosphorylated MLKL form the key complex of necroptosis (53). RIP3 and MLKL are indispensable
molecules in this cell death pathway, and knockout or inhibition of
both can impede the occurrence of necroptosis (54,55).
Based on these observations, it was concluded that curcumin induced
non-apoptotic cell death in SU-DHL-10 cells but did not induce
necroptosis.
To the best of our knowledge, the present study is
the first report demonstrating curcumin-induced activation of the
parthanatos pathway in DLBCL cells. Curcumin significantly
increased PAR polymer accumulation, an effect that was insensitive
to Q-VD-OPh but was potently inhibited by the PARP-1 inhibitor
olaparib. Furthermore, curcumin promoted the nuclear accumulation
of PARP-1, AIF and MIF. Immunofluorescence analysis confirmed the
enhanced nuclear translocation of PARP-1 and AIF, which was
attenuated by olaparib treatment These findings align with the
established hallmarks of parthanatos (56,57):
Namely, cell death associated with excessive PAR polymer formation,
which is dependent on PARP-1 activity (58).
Regarding the execution mechanism, AIF itself lacks
nuclease activity, whereas MIF possesses both exonuclease and
endonuclease activities capable of cleaving DNA. AIF binds to MIF
in the cytoplasm to form a complex that, upon nuclear
translocation, mediates large-scale DNA fragmentation and chromatin
condensation upon its nuclear translocation leading to cell death
(59). Notably, AIF is required for
nuclear translocation of MIF, but not vice versa, highlighting the
pivotal role of AIF in this process (60).
The clinical PARP inhibitor olaparib exerts its
effects through mechanisms including inhibition of PARP enzymatic
activity and ‘trapping’ of PARP-DNA complexes, leading to impaired
DNA repair and genomic instability (61). In the context of parthanatos,
olaparib blocks the downstream consequences of PARP-1
hyperactivation (61), thereby
suppressing curcumin-induced PARP-1-dependent cell death induced by
curcumin in SU-DHL-10 cells. Curcumin has been reported to
synergize with radiotherapy and chemotherapy, often by promoting
apoptotic cell death (62,63). Resistance to apoptosis is a major
cause of treatment failure in conventional cancer therapies.
Therefore, strategies that bypass apoptotic resistance by inducing
alternative cell death pathways are of great therapeutic interest
(64–66). In the present study, UVB radiation
was used as a DNA-damaging stimulus, analogous to radiotherapy
(67). Notably, curcumin can
protect healthy tissues from UVB damage (68) while acting as a photosensitizer in
tumor cells (69), suggesting a
selective advantage. As anticipated, it was found that low-dose
curcumin synergized with UVB to induce parthanatos in SU-DHL-10
cells. These results suggest that curcumin may serve as a
radiosensitizer via the induction of parthanatos. However,
translational studies are warranted to evaluate its clinical
potential in combination with radiotherapy.
It is important to note that the present findings
are primarily based on the SU-DHL-10 cell line, which serves as a
model for high-risk, treatment-resistant DLBCL. This cell line
models key features of resistance, including lack of BAX expression
(68), low levels of key apoptotic
(such as caspase-8) and necroptotic (such as RIP3 and MLKL)
mediators (70,71), BCL-2 and MYC rearrangements
characteristic of double-hit lymphoma (72,73)
and inherent insensitivity to DNA damage (74). It should be noted that the SU-DHL-10
cell line used in the present study is a double-hit lymphoma model
characterized by concurrent rearrangements in the MYC and BCL2
genes (75,76). Although the results obtained with
this cell line are promising, their molecular profiles differ from
those of major DLBCL subtypes such as germinal center B-cell or
activated B-cell lymphoma, which may limit the generalizability of
the findings (77,78). Further validation using a broader
panel of DLBCL cell lines representing distinct molecular subtypes,
as well as primary patient-derived cells, is necessary to
comprehensively assess the therapeutic potential of curcumin across
heterogeneous DLBCL populations.
Curcumin, a multi-target natural compound, has
demonstrated notable antitumor potential in preclinical studies
DLBCL, particularly exhibiting a radiosensitizing effect when
combined with radiotherapy (79–81).
However, translating this potential into clinical practice faces
multidimensional and complex challenges; for instance, curcumin
exhibits extremely low oral bioavailability (typically <1%) due
to its poor water solubility, low intestinal absorption rate and
rapid hepatic first-pass metabolism (through reduction and
conjugation) (82–85). Moreover, the complexity of the in
vivo microenvironment may substantially influence these
effects. DLBCL is highly heterogeneous, and different molecular
subtypes may vary in their sensitivity to curcumin. Currently, most
studies are based on single cell lines or animal models and lack
stratified analyses across different patient subtypes.
Consequently, it remains difficult to identify the optimal patient
population that would benefit the most from curcumin combined with
radiotherapy. Future research must advance synergistically across
multiple fronts, including the optimization of drug delivery
systems, in-depth elucidation of mechanisms of action, development
of precise animal models and rigorous clinical trial designs, to
gradually promote the clinical translation of curcumin in
combination with radiotherapy for DLBCL (86–88).
Future development of targeted delivery systems, such as the
creation of curcumin nanocarriers that specifically accumulate in
lymphoma lesions or lymph node regions (such as CD19/CD20-targeted
nanoparticles), is key to achieving effective local
radiosensitization while minimizing systemic toxicity (89–91).
Concurrently, there is a need to leverage advanced preclinical
models, such as patient-derived xenograft models or humanized mouse
models, to more accurately simulate the radiation response and
tumor microenvironment of DLBCL, thereby optimizing dosing
regimens. Therefore, exploratory clinical trials should be
conducted to systematically evaluate the safety, tolerability and
preliminary efficacy of curcumin combined with radiotherapy, while
dynamically refining the timing and dosage ratios (86,92,93).
In summary, translating the radiosensitizing effects of curcumin
from the laboratory to clinical practice for DLBCL requires deep
collaboration across multiple disciplines, including
pharmaceuticals, radiation biology and hematologic oncology. It is
hoped that curcumin-based combination regimens with novel targeted
therapies or immunotherapies will demonstrate convincing
synergistic effects in specific DLBCL subtypes, thereby offering
patients with more effective and gentler treatment options.
In summary, the present study demonstrated that
curcumin induced parthanatos in DLBCL cells through PARP-1
overactivation and AIF/MIF nuclear translocation. Furthermore, it
exhibited synergistic cytotoxicity with UVB irradiation (Fig. 5). The present study provides a
rationale for exploring parthanatos induction as a novel
therapeutic strategy for R/R DLBCL and highlights the potential of
curcumin as a radiosensitizer, which warrants further
investigation.
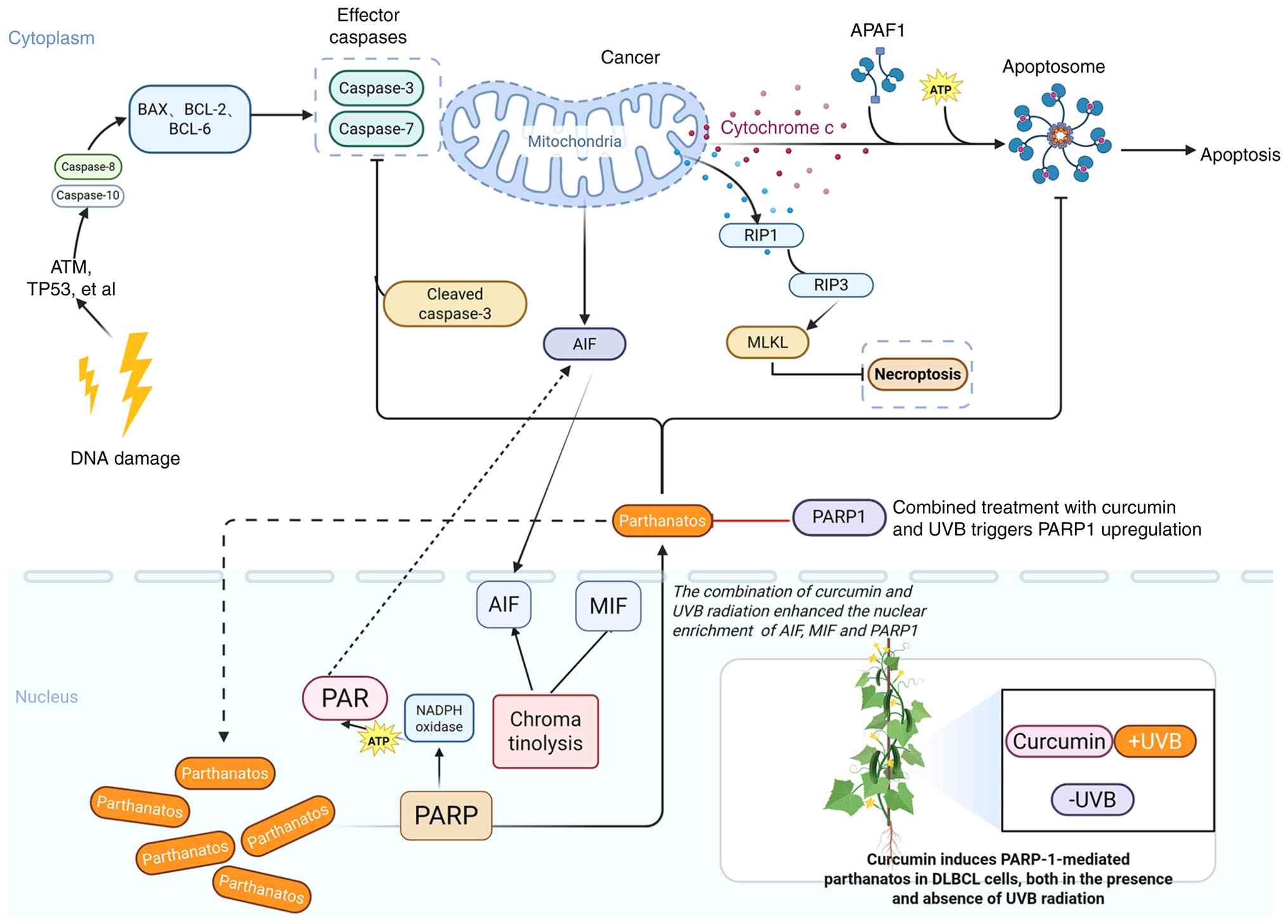 | Figure 5.Schematic illustration of how
curcumin ± UVB induces the PARP-1 mediated parthanatos in DLBCL.
When cells undergo severe DNA damage, necroptosis can be mediated
by RIP1, RIP3 and MLKL, and apoptosis can be induced by cleaved
caspase-3 and cleaved PARP. However, SU-DHL-10 cells did not
express RIP3 and MLKL, and no cleaved PARP was detected in the
presence of curcumin ± UVB, so it was speculated that curcumin ±
UVB did not cause necroptosis and apoptosis. On the contrary,
curcumin ± UVB caused an increase in PAR polymer in SU-DHL-10
cells, and an increase in AIF and MIF in the nucleus, leading to
chromatin shrinkage and DNA breakage, and eventually leading to
parthanatos in DLBCL cells. DLBCL, diffuse large B-cell lymphoma;
PAR, poly (ADP-ribose); PARP-1, poly (ADP-ribose) polymerase-1;
AIF, apoptosis-inducing factor; MIF, macrophage migration
inhibitory factor; UVB, ultraviolet radiation B; RIP, receptor
interacting protein, MLKL, mixed lineage kinase domain like
pseudokinase; BAX, Bcl-2-associated X protein; BCL, B-cell lymphoma
2; RIP, receptor-interacting protein kinase; ATP, adenosine
triphosphate; APAF1, apoptotic protease activating factor 1; TP53,
tumor protein p53. |
Acknowledgements
The authors would like to thank Dr Zhen Yang
(Tianjin Cancer Institute of Integrative Traditional Chinese and
Western Medicine, Tianjin Union Medical Center of Nankai
University, Tianjin, China) for their technical assistance and Dr
Ruxue Liu (School of Integrative Medicine, Tianjin University of
Traditional Chinese Medicine, Tianjin, China) for their helpful
discussions and critical reading of the manuscript.
Funding
The present work was supported by the National Natural Science
Foundation of China (grant no. 82070206) and Tianjin Key Medical
Discipline Construction Project (grant no. TJYXZDXK-3-003A-5).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
FL and HW contributed to the conceptualization of
the study. YW, RQ and MQ were responsible for data curation,
including organizing and verifying the research data. YW, RQ and XZ
performed the formal analysis, applying statistical and analytical
techniques to analyze the study data. MQ and HW acquired the
funding that supported this research. YW, RQ and HM conducted the
investigation, which involved performing the experiments and
collecting the data. FL and HW developed the methodology, designing
the procedures and frameworks used in the present study. MQ and HW
handled project administration, coordinating and managing the
research activities. FL and HW provided supervision, overseeing the
research progress and providing guidance. YW was responsible for
visualization, creating the figures and tables that present the
data. YW and RQ wrote the original draft of the manuscript. YW, RQ
and HW reviewed and edited the manuscript for important
intellectual content. YW, MQ and HW confirm the authenticity of all
the raw data. All authors read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools were used to improve the readability and
language of the manuscript or to generate images, and subsequently,
the authors revised and edited the content produced by the
artificial intelligence tools as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Gong IY, Crump M, Prica A, Calzavara A,
Liu N, Kordbacheh T, Rodin D, Hodgson D, Mozessohn L, Cheung MC and
Kuruvilla J: Outcomes and factors influencing survival in patients
with diffuse large B-cell lymphoma: A population-based analysis.
Blood Neoplasia. 2:1001172025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nastoupil LJ and Bartlett NL: Navigating
the evolving treatment landscape of diffuse large B-cell lymphoma.
J Clin Oncol. 41:903–913. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
He MY and Kridel R: Treatment resistance
in diffuse large B-cell lymphoma. Leukemia. 35:2151–2165. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu Y, Stockwell BR, Jiang X and Gu W:
p53-regulated non-apoptotic cell death pathways and their relevance
in cancer and other diseases. Nat Rev Mol Cell Biol. 26:600–614.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang Y, Zhang C, Li J, Jiang M, Guo S,
Yang G, Zhang L, Wang F, Yi S, Wang J, et al: Inhibition of AKT
induces p53/SIRT6/PARP1-dependent parthanatos to suppress tumor
growth. Cell Commun Signal. 20:932022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang WQ, Liu L, Sun HC, Fu YL, Xu HX, Chai
ZT, Zhang QB, Kong LQ, Zhu XD, Lu L, et al: Tanshinone IIA inhibits
metastasis after palliative resection of hepatocellular carcinoma
and prolongs survival in part via vascular normalization. J Hematol
Oncol. 5:692012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang N, Chen S, Xie Y, Liu X, Xi Z, Li J,
Xue C, Deng R, Min W, Kang R and Xie L: The Sanbi Decoction
alleviates intervertebral disc degeneration in rats through
intestinal flora and serum metabolic homeostasis modulation.
Phytomedicine. 127:1554802024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ming T, Tao Q, Tang S, Zhao H, Yang H, Liu
M, Ren S and Xu H: Curcumin: An epigenetic regulator and its
application in cancer. Biomed Pharmacother. 156:1139562022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang W, Li M, Wang L, Chen L and Goh BC:
Curcumin in cancer therapy: Exploring molecular mechanisms and
overcoming clinical challenges. Cancer Lett. 570:2163322023.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abd El-Hack ME, El-Saadony MT, Swelum AA,
Arif M, Abo Ghanima MM, Shukry M, Noreldin A, Taha AE and
El-Tarabily KA: Curcumin, the active substance of turmeric: Its
effects on health and ways to improve its bioavailability. J Sci
Food Agric. 101:5747–5762. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhao PY, Ren H, Xu LY, Lou MF, Qing Y, Lin
LT, Zhu ZP and Liao W: Research progress on pharmacological
activity and mechanism of traditional Chinese medicine
polysaccharides of Curcuma. Chin Tradit Herb Drugs. 56:5276–5287.
2025.(In Chinese).
|
|
12
|
Simamora A, Timotius KH, Yerer MB,
Setiawan H and Mun'im A: Xanthorrhizol, a potential anticancer
agent, from Curcuma xanthorrhiza Roxb. Phytomedicine.
105:1543592022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Koh YC, Tsai YW, Lee PS, Nagabhushanam K,
Ho CT and Pan MH: Amination potentially augments the ameliorative
effect of curcumin on inhibition of the IL-6/Stat3/c-Myc pathway
and gut microbial modulation in colitis-associated tumorigenesis. J
Agric Food Chem. 70:14744–14754. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chung SS, Dutta P, Chard N, Wu Y, Chen QH,
Chen G and Vadgama J: A novel curcumin analog inhibits canonical
and non-canonical functions of telomerase through STAT3 and NF-κB
inactivation in colorectal cancer cells. Oncotarget. 10:4516–4531.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Siriviriyakul P, Chingchit T, Klaikeaw N,
Chayanupatkul M and Werawatganon D: Effects of curcumin on
oxidative stress, inflammation and apoptosis in L-arginine induced
acute pancreatitis in mice. Heliyon. 5:e022222019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hu J, Lin S, Tan BK, Hamzah SS, Lin Y,
Kong Z, Zhang Y, Zheng B and Zeng S: Photodynamic inactivation of
Burkholderia cepacia by curcumin in combination with EDTA. Food Res
Int. 111:265–271. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Aadinath W, Bhushani A and
Anandharamakrishnan C: Synergistic radical scavenging potency of
curcumin-in-β-cyclodextrin-in-nanomagnetoliposomes. Mater Sci Eng C
Mater Biol Appl. 64:293–302. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zoi V, Galani V, Tsekeris P, Kyritsis AP
and Alexiou GA: Radiosensitization and radioprotection by curcumin
in glioblastoma and other cancers. Biomedicines. 10:3122022.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ailioaie LM and Litscher G: Curcumin and
photobiomodulation in chronic viral hepatitis and hepatocellular
carcinoma. Int J Mol Sci. 21:71502020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shetty NP, Prabhakaran M and Srivastava
AK: Pleiotropic nature of curcumin in targeting multiple
apoptotic-mediated factors and related strategies to treat gastric
cancer: A review. Phytother Res. 35:5397–5416. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Deng L, Wu X, Zhu X, Yu Z, Liu Z, Wang J
and Zheng Y: Combination effect of curcumin with docetaxel on the
PI3K/AKT/mTOR pathway to induce autophagy and apoptosis in
esophageal squamous cell carcinoma. Am J Transl Res. 13:57–72.
2021.PubMed/NCBI
|
|
22
|
Calaf GM, Ponce-Cusi R and Carrión F:
Curcumin and paclitaxel induce cell death in breast cancer cell
lines. Oncol Rep. 40:2381–2388. 2018.PubMed/NCBI
|
|
23
|
Lee YJ, Park KS and Lee SH: Curcumin
targets both apoptosis and necroptosis in acidity-tolerant prostate
carcinoma cells. Biomed Res Int. 2021:88591812021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tang X, Ding H, Liang M, Chen X, Yan Y,
Wan N, Chen Q, Zhang J and Cao J: Curcumin induces ferroptosis in
non-small-cell lung cancer via activating autophagy. Thorac Cancer.
12:1219–1230. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Giordano A and Tommonaro G: Curcumin and
cancer. Nutrients. 11:23762019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rahman AT, Rafia R, Jethro A, Santoso P,
Kharisma VD, Affan A, Murtadlo A, Purnamasari D, Soekamto NH,
Ansori ANM, et al: In silico study of the potential of endemic
sumatra wild turmeric rhizomes (curcuma sumatrana: Zingiberaceae)
as anti-cancer. Pharmacogn J. 14:806–812. 2022. View Article : Google Scholar
|
|
27
|
Li K, Wang J, Xie Y, Lu Z, Sun W, Wang K,
Liang J and Chen X: Reactive oxygen species/glutathione dual
sensitive nanoparticles with encapsulation of miR155 and curcumin
for synergized cancer immunotherapy. J Nanobiotechnology.
22:4002024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Belcaro G, Hosoi M, Pellegrini L,
Appendino G, Ippolito E, Ricci A, Ledda A, Dugall M, Cesarone MR,
Maione C, et al: A controlled study of a lecithinized delivery
system of curcumin (Meriva®) to alleviate the adverse
effects of cancer treatment. Phytother Res. 28:444–450. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pastorelli D, Fabricio ASC, Giovanis P,
D'Ippolito S, Fiduccia P, Soldà C, Buda A, Sperti C, Bardini R, Da
Dalt G, et al: Phytosome complex of curcumin as complementary
therapy of advanced pancreatic cancer improves safety and efficacy
of gemcitabine: Results of a prospective phase II trial. Pharmacol
Res. 132:72–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shah S, Rath H, Sharma G, Senapati SN and
Mishra E: Effectiveness of curcumin mouthwash on radiation-induced
oral mucositis among head and neck cancer patients: A triple-blind,
pilot randomised controlled trial. Indian J Dent Res. 31:718–727.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Galluzzi L, Vitale I, Aaronson SA, Abrams
JM, Adam D, Agostinis P, Alnemri ES, Altucci L, Amelio I, Andrews
DW, et al: Molecular mechanisms of cell death: Recommendations of
the nomenclature committee on cell death 2018. Cell Death Differ.
25:486–541. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang X, Zhang W, Ge P, Yu M and Meng H:
Parthanatos participates in glutamate-mediated HT22 cell injury and
hippocampal neuronal death in kainic acid-induced status
epilepticus rats. CNS Neurosci Ther. 28:2032–2043. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Buckley AM, Lynam-Lennon N, O'Neill H and
O'Sullivan J: Targeting hallmarks of cancer to enhance
radiosensitivity in gastrointestinal cancers. Nat Rev Gastroenterol
Hepatol. 17:298–313. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tuo QZ, Zhang ST and Lei P: Mechanisms of
neuronal cell death in ischemic stroke and their therapeutic
implications. Med Res Rev. 42:259–305. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Liu S, Luo W and Wang Y: Emerging role of
PARP-1 and PARthanatos in ischemic stroke. J Neurochem. 160:74–87.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
So KY and Oh SH: Arsenite-induced
cytotoxicity is regulated by poly-ADP ribose polymerase 1
activation and parthanatos in p53-deficient H1299 cells: The roles
of autophagy and p53. Biochem Biophys Res Commun. 656:78–85. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wen W, Jin K, Che Y, Du LY and Wang LN:
Arnicolide D inhibits oxidative stress-induced breast cancer cell
growth and invasion through apoptosis, ferroptosis, and
parthanatos. Anticancer Agents Med Chem. 24:836–844. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jiang X, Deng W, Tao S, Tang Z, Chen Y,
Tian M, Wang T, Tao C, Li Y, Fang Y, et al: A RIPK3-independent
role of MLKL in suppressing parthanatos promotes immune evasion in
hepatocellular carcinoma. Cell Discov. 9:72023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li C, Zhang J, Wu Q, Kumar A, Pan G and
Kelvin DJ: Nifuroxazide activates the parthanatos to overcome
TMPRSS2:ERG fusion-positive prostate cancer. Mol Cancer Ther.
22:306–316. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu L, Liu B, Guan G, Kang R, Dai Y and
Tang D: Cyclophosphamide-induced GPX4 degradation triggers
parthanatos by activating AIFM1. Biochem Biophys Res Commun.
606:68–74. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Boulos JC, Omer EA, Rigano D, Formisano C,
Chatterjee M, Leich E, Klauck SM, Shan LT and Efferth T:
Cynaropicrin disrupts tubulin and c-Myc-related signaling and
induces parthanatos-type cell death in multiple myeloma. Acta
Pharmacol Sin. 44:2265–2281. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Robinson N, Ganesan R, Hegedűs C, Kovács
K, Kufer TA and Virág L: Programmed necrotic cell death of
macrophages: Focus on pyroptosis, necroptosis, and parthanatos.
Redox Biol. 26:1012392019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yang L, Guttman L, Dawson VL and Dawson
TM: Parthanatos: Mechanisms, modulation, and therapeutic prospects
in neurodegenerative disease and stroke. Biochem Pharmacol.
228:1161742024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kuželová K, Grebeňová D and Brodská B:
Dose-dependent effects of the caspase inhibitor Q-VD-OPh on
different apoptosis-related processes. J Cell Biochem.
112:3334–3342. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Caserta TM, Smith AN, Gultice AD, Reedy MA
and Brown TL: Q-VD-OPh, a broad spectrum caspase inhibitor with
potent antiapoptotic properties. Apoptosis. 8:345–352. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Bhatti IA, Abhari BA and Fulda S:
Identification of a synergistic combination of Smac mimetic and
Bortezomib to trigger cell death in B-cell non-Hodgkin lymphoma
cells. Cancer Lett. 405:63–72. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang P, Wang P, Liu B, Zhao J, Pang Q,
Agrawal SG, Jia L and Liu FT: Dynamin-related protein Drp1 is
required for Bax translocation to mitochondria in response to
irradiation-induced apoptosis. Oncotarget. 6:22598–22612. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang Z, Liu F, Liao W, Yu L, Hu Z, Li M
and Xia H: Curcumin suppresses glioblastoma cell proliferation by
p-AKT/mTOR pathway and increases the PTEN expression. Arch Biochem
Biophys. 689:1084122020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Miao Y, Zheng Y, Geng Y, Yang L, Cao N,
Dai Y and Wei Z: The role of GLS1-mediated
glutaminolysis/2-HG/H3K4me3 and GSH/ROS signals in Th17 responses
counteracted by PPARγ agonists. Theranostics. 11:4531–4548. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Stepanenko AA and Dmitrenko VV: Pitfalls
of the MTT assay: Direct and off-target effects of inhibitors can
result in over/underestimation of cell viability. Gene.
574:193–203. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Moyer A, Tanaka K and Cheng EH: Apoptosis
in cancer biology and therapy. Annu Rev Pathol. 20:303–328. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tian X, Srinivasan PR, Tajiknia V, Sanchez
Sevilla Uruchurtu AF, Seyhan AA, Carneiro BA, De La Cruz A,
Pinho-Schwermann M, George A, Zhao S, et al: Targeting apoptotic
pathways for cancer therapy. J Clin Invest. 134:e1795702024.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Grootjans S, Vanden Berghe T and
Vandenabeele P: Initiation and execution mechanisms of necroptosis:
An overview. Cell Death Differ. 24:1184–1195. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Remijsen Q, Goossens V, Grootjans S, Van
den Haute C, Vanlangenakker N, Dondelinger Y, Roelandt R, Bruggeman
I, Goncalves A, Bertrand MJ, et al: Depletion of RIPK3 or MLKL
blocks TNF-driven necroptosis and switches towards a delayed RIPK1
kinase-dependent apoptosis. Cell Death Dis. 5:e10042014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Dondelinger Y, Aguileta MA, Goossens V,
Dubuisson C, Grootjans S, Dejardin E, Vandenabeele P and Bertrand
MJ: RIPK3 contributes to TNFR1-mediated RIPK1 kinase-dependent
apoptosis in conditions of cIAP1/2 depletion or TAK1 kinase
inhibition. Cell Death Differ. 20:1381–1392. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li D, Kou Y, Gao Y, Liu S, Yang P,
Hasegawa T, Su R, Guo J and Li M: Oxaliplatin induces the
PARP1-mediated parthanatos in oral squamous cell carcinoma by
increasing production of ROS. Aging (Albany NY). 13:4242–4257.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Künzi L and Holt GE: Cigarette smoke
activates the parthanatos pathway of cell death in human bronchial
epithelial cells. Cell Death Discov. 5:1272019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang HF, Wang ZQ, Ding Y, Piao MH, Feng
CS, Chi GF, Luo YN and Ge PF: Endoplasmic reticulum stress
regulates oxygen-glucose deprivation-induced parthanatos in human
SH-SY5Y cells via improvement of intracellular ROS. CNS Neurosci
Ther. 24:29–38. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Fatokun AA, Dawson VL and Dawson TM:
Parthanatos: Mitochondrial-linked mechanisms and therapeutic
opportunities. Br J Pharmacol. 171:2000–2016. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wang Y, An R, Umanah GK, Park H, Nambiar
K, Eacker SM, Kim B, Bao L, Harraz MM, Chang C, et al: A nuclease
that mediates cell death induced by DNA damage and poly(ADP-ribose)
polymerase-1. Science. 354:aad68722016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Murai J, Huang SY, Das BB, Renaud A, Zhang
Y, Doroshow JH, Ji J, Takeda S and Pommier Y: Trapping of PARP1 and
PARP2 by clinical PARP inhibitors. Cancer Res. 72:5588–5599. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lai X, Geng X, Li M, Tang M, Liu Q, Yang
M, Shen L, Zhu Y and Wang S: Glutathione-responsive PLGA
nanocomplex for dual delivery of doxorubicin and curcumin to
overcome tumor multidrug resistance. Nanomedicine (Lond).
16:1411–1427. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zheng X, Yang X, Lin J, Song F and Shao Y:
Low curcumin concentration enhances the anticancer effect of
5-fluorouracil against colorectal cancer. Phytomedicine.
85:1535472021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Carneiro BA and El-Deiry WS: Targeting
apoptosis in cancer therapy. Nat Rev Clin Oncol. 17:395–417. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Herrera M, Pretelli G, Desai J, Garralda
E, Siu LL, Steiner TM and Au L: Bispecific antibodies: Advancing
precision oncology. Trends Cancer. 10:893–919. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sarmento-Ribeiro AB, Scorilas A, Gonçalves
AC, Efferth T and Trougakos IP: The emergence of drug resistance to
targeted cancer therapies: Clinical evidence. Drug Resist Updat.
47:1006462019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Pollet M, Shaik S, Mescher M, Frauenstein
K, Tigges J, Braun SA, Sondenheimer K, Kaveh M, Bruhs A, Meller S,
et al: The AHR represses nucleotide excision repair and apoptosis
and contributes to UV-induced skin carcinogenesis. Cell Death
Differ. 25:1823–1836. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ben Yehuda Greenwald M, Frušić-Zlotkin M,
Soroka Y, Ben Sasson S, Bitton R, Bianco-Peled H and Kohen R:
Curcumin protects skin against UVB-induced cytotoxicity via the
keap1-Nrf2 pathway: The use of a microemulsion delivery system.
Oxid Med Cell Longev. 2017:52054712017. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Park K and Lee JH: Photosensitizer effect
of curcumin on UVB-irradiated HaCaT cells through activation of
caspase pathways. Oncol Rep. 17:537–540. 2007.PubMed/NCBI
|
|
70
|
Galluzzi L, Kepp O, Chan FK and Kroemer G:
Necroptosis: Mechanisms and relevance to disease. Annu Rev Pathol.
12:103–130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Yuan J and Ofengeim D: A guide to cell
death pathways. Nat Rev Mol Cell Biol. 25:379–395. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Zhang J, Wang T, Shetty K, Alkan S, Xu S,
Gong Q, Liu X, Li Y, Hu Z, Huang W, et al: Genetic characterization
and drug sensitivity study of newly derived HGBL double/triple-hit
lymphoma cell lines. Blood Adv. 6:5067–5071. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Esteve-Arenys A, Valero JG,
Chamorro-Jorganes A, Gonzalez D, Rodriguez V, Dlouhy I, Salaverria
I, Campo E, Colomer D, Martinez A, et al: The BET bromodomain
inhibitor CPI203 overcomes resistance to ABT-199 (venetoclax) by
downregulation of BFL-1/A1 in in vitro and in vivo models of
MYC+/BCL2+ double hit lymphoma. Oncogene. 37:1830–1844. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Ennishi D, Jiang A, Boyle M, Collinge B,
Grande BM, Ben-Neriah S, Rushton C, Tang J, Thomas N, Slack GW, et
al: Double-hit gene expression signature defines a distinct
subgroup of germinal center B-cell-like diffuse large B-cell
lymphoma. J Clin Oncol. 37:190–201. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Attieh M and Asakrah S: B-ALL with
synchronous MYC and BCL-2 rearrangement. Blood. 141:18942023.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Collinge B, Ben-Neriah S, Hilton LK,
Alduaij W, Tucker T, Slack GW, Farinha P, Craig JW, Boyle M,
Meissner B, et al: Unbalanced MYC break-apart FISH patterns
indicate the presence of a MYC rearrangement in HGBCL-DH-BCL2.
Blood. 144:1611–1616. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Chapuy B, Stewart C, Dunford AJ, Kim J,
Kamburov A, Redd RA, Lawrence MS, Roemer MGM, Li AJ, Ziepert M, et
al: Molecular subtypes of diffuse large B cell lymphoma are
associated with distinct pathogenic mechanisms and outcomes. Nat
Med. 24:679–690. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Schmitz R, Wright GW, Huang DW, Johnson
CA, Phelan JD, Wang JQ, Roulland S, Kasbekar M, Young RM, Shaffer
AL, et al: Genetics and pathogenesis of diffuse large B-cell
lymphoma. N Engl J Med. 378:1396–1407. 2028. View Article : Google Scholar
|
|
79
|
Bozzuto G, Calcabrini A, Colone M,
Condello M, Dupuis ML, Pellegrini E and Stringaro A: Phytocompounds
and nanoformulations for anticancer therapy: A review. Molecules.
29:37842024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Qiao Q, Jiang Y and Li G: Curcumin
improves the antitumor effect of X-ray irradiation by blocking the
NF-κB pathway: An in-vitro study of lymphoma. Anticancer Drugs.
23:597–605. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Qiao Q, Jiang Y and Li G: Inhibition of
the PI3K/AKT-NF-κB pathway with curcumin enhanced radiation-induced
apoptosis in human Burkitt's lymphoma. J Pharmacol Sci.
121:247–256. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Pratama BP, Khairullah AR, Ma'ruf IF,
Akintunde AO, Mustofa I, Mulyati S, Ahmad AZ and Ansori ANM:
Revolutionizing curcumin bioavailability: From health benefits to
placement in food packaging products. Food Syst. 8:343–354. 2025.
View Article : Google Scholar
|
|
83
|
El-Saadony MT, Saad AM, Mohammed DM,
Alkafaas SS, Ghosh S, Negm SH, Salem HM, Fahmy MA, Mosa WFA,
Ibrahim EH, et al: Curcumin, an active component of turmeric:
Biological activities, nutritional aspects, immunological,
bioavailability, and human health benefits-a comprehensive review.
Front Immunol. 16:16030182025. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Heidari H, Bagherniya M, Majeed M,
Sathyapalan T, Jamialahmadi T and Sahebkar A: Curcumin-piperine
co-supplementation and human health: A comprehensive review of
preclinical and clinical studies. Phytother Res. 37:1462–1487.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Matthewman C, Krishnakumar IM and Swick
AG: Review: Bioavailability and efficacy of ‘free’ curcuminoids
from curcumagalactomannoside (CGM) curcumin formulation. Nutr Res
Rev. 37:14–31. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Zhang X, Cui Q, Yin L, Zhu J, Mao Y, Yin
R, Shao H, Wang W, Sun X, Zhang Z, et al: Ginger-derived
vesicle-like nanoparticles loaded with curcumin to alleviate
ionizing radiation-induced intestinal damage via gut microbiota
regulation. Gut Microbes. 17:25312102025. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Sak K: Radiosensitizing potential of
curcumin in different cancer models. Nutr Cancer. 72:1276–1289.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Sudarshan V, Shyamjith P, Kumar S,
Ravindran F, Teraiya N, Choudhary B and Karki SS: Assessing the
anti-leukemic efficacy of a curcuminoid in cellular and animal
models. Eur J Med Chem. 300:1181772025. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ipar VS, Dsouza A and Devarajan PV:
Enhancing curcumin oral bioavailability through nanoformulations.
Eur J Drug Metab Pharmacokinet. 44:459–480. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Zhao J, Jia W, Zhang R, Wang X and Zhang
L: Improving curcumin bioavailability: Targeted delivery of
curcumin and loading systems in intestinal inflammation. Food Res
Int. 196:1150792024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Chang R, Chen L, Qamar M, Wen Y, Li L,
Zhang J, Li X, Assadpour E, Esatbeyoglu T, Kharazmi MS, et al: The
bioavailability, metabolism and microbial modulation of
curcumin-loaded nanodelivery systems. Adv Colloid Interface Sci.
318:1029332023. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Karimi A, Naeini F, Niazkar HR, Tutunchi
H, Musazadeh V, Mahmoodpoor A, Asghariazar V, Mobasseri M and
Tarighat-Esfanjani A: Nano-curcumin supplementation in critically
ill patients with sepsis: A randomized clinical trial investigating
the inflammatory biomarkers, oxidative stress indices, endothelial
function, clinical outcomes and nutritional status. Food Funct.
13:6596–6612. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Karimi A, Pourreza S, Vajdi M, Mahmoodpoor
A, Sanaie S, Karimi M and Tarighat-Esfanjani A: Evaluating the
effects of curcumin nanomicelles on clinical outcome and cellular
immune responses in critically ill sepsis patients: A randomized,
double-blind, and placebo-controlled trial. Front Nutr.
9:10378612022. View Article : Google Scholar : PubMed/NCBI
|



















