Introduction
Upper tract urothelial carcinoma (UTUC) is a
relatively uncommon malignancy, accounting for 5–10% of all UCs
worldwide (1). Notably, its
incidence is disproportionately higher in certain geographic
regions, such as Southwestern Taiwan, possibly due to environmental
exposures or underlying genetic susceptibility (2). Radical nephroureterectomy (RNU) with
excision of the bladder cuff remains the gold-standard treatment
for high-risk localized UTUC, especially in cases presenting with
multifocality, large tumor burden (>2 cm), high-grade histology
or advanced clinical stage at diagnosis (3–5).
Postoperative management following RNU is guided by established
prognostic indicators, including pathological stage, tumor grade,
lymphovascular invasion and margin status, with adjuvant
chemotherapy or salvage radiotherapy considered for patients at
increased risk of recurrence or progression (6).
More recently, neoadjuvant chemotherapy has emerged
as a potential strategy for selected patients with high-risk UTUC,
aiming to optimize cisplatin eligibility prior to
nephrectomy-induced renal function decline, while immune checkpoint
inhibitors have shown promise in advanced and metastatic UC and are
being explored in perioperative settings (7,8).
However, despite these systemic treatment advances, their precise
role in localized UTUC is still evolving, and robust preoperative
biomarkers to guide patient selection are still lacking. As for
adjuvant chemotherapy, platinum-based treatment was demonstrated to
improve outcomes for patients with high-risk UTUC following RNU in
the POUT trial (9). Hence, an
increasing number of studies have been dedicated to discovering
more reliable and feasible prognosticators for selecting high-risk
patients who may truly benefit from adjuvant therapy.
Despite adherence to surgical guidelines, clinical
outcomes in UTUC remain heterogeneous. Risk stratification after
RNU typically incorporates established pathological factors such as
tumor stage, grade, lymphovascular invasion (LVI) and surgical
margin status (6). However, these
parameters alone often fail to capture the broader immunological
and systemic context influencing tumor progression. Consequently,
recent efforts have turned toward identifying systemic biomarkers,
particularly those linked to host immunity and inflammation, to
improve prognostication (10–12).
Studies have highlighted the prognostic relevance of
various systemic inflammatory markers in UTUC, including the
neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio,
monocyte-to-lymphocyte ratio and the systemic immune-inflammation
index (10–13). Yet, their predictive precision
remains limited. Given the immune-mediated nature of cancer
progression, a more integrated approach involving immune organ
metrics may enhance clinical risk models.
The spleen, as the largest secondary lymphoid organ
and a key component of the reticuloendothelial system, plays a
central role in both innate and adaptive immunity (14). Emerging evidence suggests that
immune dysregulation, manifesting as splenic enlargement due to the
accumulation of immune cells, may influence oncological outcomes in
various malignancies, including hepatocellular carcinoma (HCC),
colorectal cancer and lung cancer (15–18).
In particular, the spleen has been implicated in the production and
mobilization of tumor-associated macrophages, tumor-associated
neutrophils and myeloid-derived suppressor cells (MDSCs), all of
which contribute to tumor immune evasion and progression (19). Clinical studies in diverse cancer
types, including HCC, colorectal cancer and lung cancer, have
suggested that increased splenic volume (SV) may correlate with
immune suppression and a poor prognosis, possibly mediated through
depletion of cytotoxic lymphocyte subsets such as CD4+ T
cells and natural killer cells (16–18,20,21).
To date, no study has systematically examined the
prognostic relevance of SV or its interplay with circulating
lymphocyte parameters in UTUC. Therefore, the current study aimed
to explore the independent and synergistic prognostic value of
baseline standardized SV and lymphocyte percentage (lym%) in
patients with non-metastatic UTUC undergoing RNU.
Materials and methods
Study design and patient
selection
The present retrospective cohort study included
patients with non-metastatic UTUC who underwent RNU at National
Cheng Kung University (Tainan, Taiwan) between November 2008 and
December 2020. The inclusion criteria were as follows: i)
Histologically confirmed UTUC; ii) no evidence of distant
metastasis at diagnosis; and iii) availability of preoperative
abdominal computed tomography (CT) imaging. Patients were excluded
due to any of the following criteria: i) Prior splenectomy; ii)
known splenic pathology (e.g., thrombosed splenic aneurysm); iii)
no accessible preoperative CT scan; or iv) a postoperative
follow-up duration of <24 months. Clinical and pathological
variables were reviewed from electronic medical records, including
age, sex, renal function, tumor characteristics and concomitant
bladder cancer. Peripheral lym% was extracted from preoperative
complete blood count results, collected within 1 month prior to
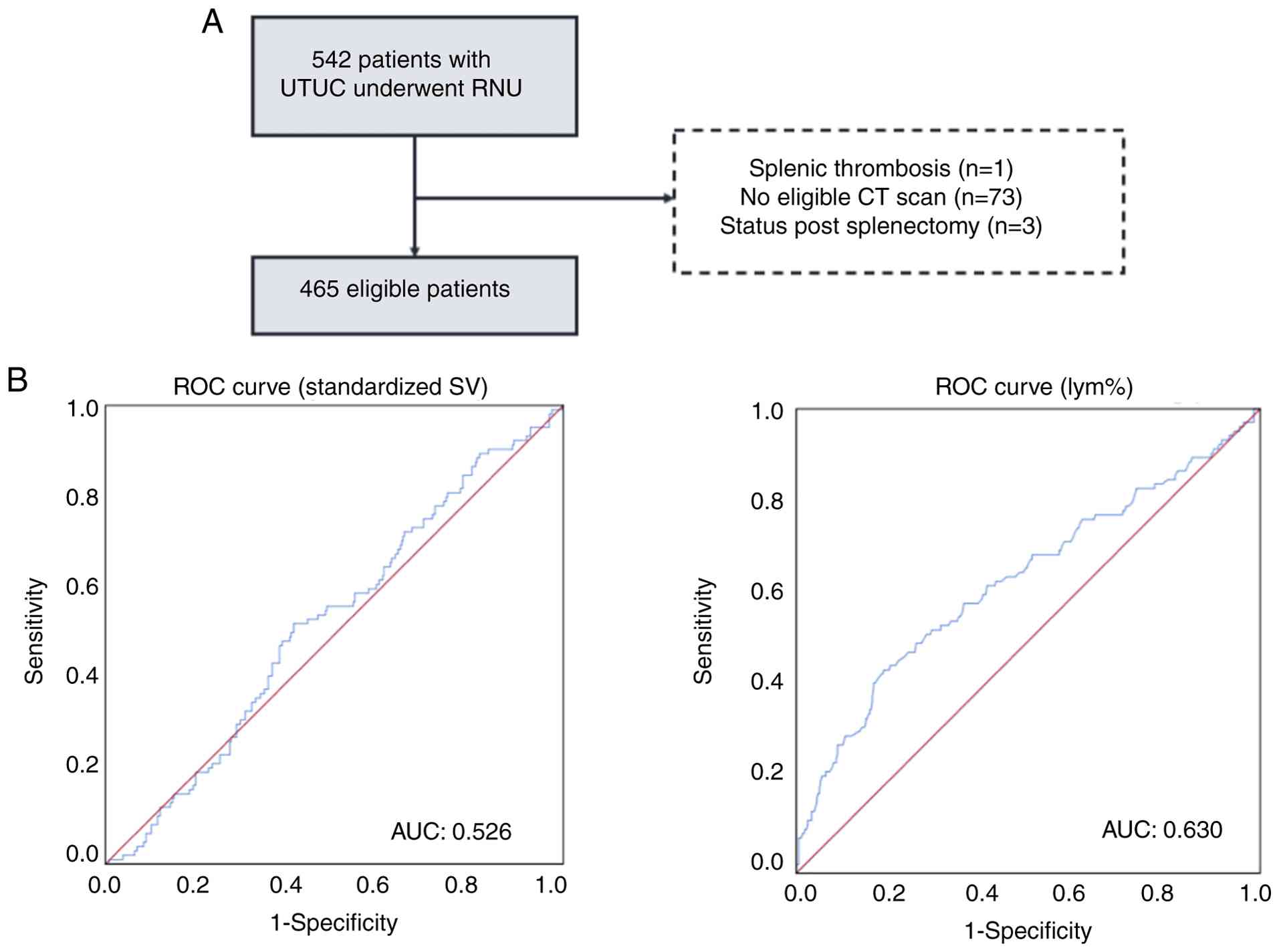
RNU. A diagram of the study design is shown in Fig. 1A.
Imaging analysis and SV
measurement
For each patient, the most recent abdominal CT scan
prior to surgery was retrieved from the Picture Archiving and
Communication System. Two experienced readers independently
delineated the spleen on axial slices from upper to lower poles
using a manual contouring method. The SV was calculated by summing
the area of each outlined slice multiplied by the corresponding
slice thickness. To account for individual body size differences,
SV was standardized to body surface area (m2), resulting
in a unit of cm3/m2. Details of the
segmentation procedure are shown in Fig. S1. Intra- and inter-observer
reliabilities of the SV measurement were assessed by intraclass
correlation coefficient (ICC). An ICC value >0.75 was considered
indicative of excellent agreement (22). The SV measurements demonstrated
excellent reliability, with an intra-observer ICC of 0.943 (95% CI,
0.791–0.984) and an inter-observer ICC of 0.951 (95% CI,
0.821–0.989).
Statistical analysis
All analyses were performed using IBM SPSS
Statistics for Windows, version 25.0 (IBM Corp.). Receiver
operating characteristic (ROC) curve analysis was applied to
determine optimal cut-off values for standardized SV and lym%,
using all-cause mortality as the endpoint. Continuous variables
were compared using independent t-tests or the Mann-Whitney U test,
as appropriate, and categorical variables were compared using
χ2 tests. Correlations between SV and hematological
parameters were evaluated using Pearson's correlation
coefficient.
Survival outcomes, including overall survival (OS),
cancer-specific survival (CSS) and metastasis-free survival (MFS),
were defined as the interval from RNU to death or last follow-up.
Patients lost to follow-up or alive at the final follow-up were
censored. Survival curves were generated using Kaplan-Meier
analysis, and differences were assessed by the log-rank test.
Prognostic factors were analyzed using univariate and multivariate
Cox proportional hazards models. Two-tailed P<0.05 was
considered to indicate a statistically significant difference.
Results
Clinical characteristics of patients
with UTUC
Between November 2008 and December 2020, a total of
542 patients who underwent RNU for UTUC at National Cheng Kung
University were identified. After excluding 1 patient with a
history of a splenic calcified aneurysm with thrombosis, 3 patients
who had undergone a splenectomy and 73 patients lacking
preoperative abdominal CT imaging, 465 patients were included in
the final analysis (Fig. 1A).
The mean age of the cohort was 69.5±10.4 years
(range, 29–95 years). Based on ROC analysis using all-cause
mortality as the endpoint, the optimal threshold for standardized
SV was determined to be 79 cm3/m2
[sensitivity, 52%; specificity, 58%; area under the curve (AUC),
0.526; 95% confidence interval (CI), 0.466–0.587], while the
optimal cut-off for lym% was 21% (sensitivity, 58%; specificity,
64%; AUC, 0.630; 95% CI, 0.565–0.696) (Fig. 1B). Of the 465 patients, 206 (44.3%)
were categorized into the high SV (SV >79
cm3/m2) group. These patients had a
significantly higher proportion of males (P=0.015) and worse
baseline renal function (P=0.017) than those in the low SV group
(n=259). Detailed clinicopathological features are presented in
Table I.
 | Table I.Baseline characteristics of the
cohort (n=465) according to low (n=259) or high (n=206) SV. |
Table I.
Baseline characteristics of the
cohort (n=465) according to low (n=259) or high (n=206) SV.
|
|
| Standardized
SV |
|
|---|
|
|
|
|
|
|---|
|
Characteristics | All patients | Low SV | High SV | P-value |
|---|
| Mean age at
diagnosis of UTUC, years | 69.5±10.4 | 70.8±9.6 | 68.4±11.5 | 0.015 |
| Median follow-up
duration after RNU, months | 45.8±25.1 | 50.0±25.0 | 38.2±24.9 | 0.011 |
| Age, n (%) |
|
|
| 0.112 |
| ≤69
years | 219 (47) | 113 (44) | 106 (51) |
|
| >69
years | 246 (53) | 146 (56) | 100 (49) |
|
| Sex, n (%) |
|
|
| 0.015 |
|
Male | 203 (44) | 100 (39) | 103 (50) |
|
|
Female | 262 (56) | 159 (61) | 103 (50) |
|
| Renal function, n
(%) |
|
|
| 0.017 |
| eGFR
≤60 ml/min/1.73 m2 | 274 (59) | 141 (54) | 133 (65) |
|
| eGFR
>60 ml/min/1.73 m2 | 191 (41) | 118 (46) | 73 (35) |
|
| HTN or DM, n
(%) |
|
|
| 0.511 |
| No | 204 (44) | 110 (42) | 94 (46) |
|
|
Yes | 261 (56) | 149 (58) | 112 (54) |
|
| Previous BC, n
(%) |
|
|
| 0.195 |
| No | 395 (85) | 225 (87) | 170 (83) |
|
|
Yes | 70 (15) | 34 (13) | 36 (17) |
|
| Concomitant BC, n
(%) |
|
|
| 0.480 |
| No | 375 (81) | 212 (82) | 163 (79) |
|
|
Yes | 90 (19) | 47 (18) | 43 (21) |
|
| Tumor location, n
(%) |
|
|
| 0.683 |
|
Pelvis | 213 (46) | 119 (46) | 94 (46) |
|
|
Ureter | 156 (34) | 90 (35) | 66 (32) |
|
|
Both | 96 (21) | 50 (19) | 46 (22) |
|
| Pathological T
stage, n (%) |
|
|
| 0.309 |
|
Tis/a/1 | 158 (34) | 86 (33) | 72 (35) |
|
| T2 | 89 (19) | 56 (22) | 33 (16) |
|
|
T3/4 | 218 (47) | 117 (45) | 101 (49) |
|
| Lymph node status,
n (%) |
|
|
| >0.999 |
|
N0/x | 435 (94) | 242 (93) | 193 (94) |
|
| N+ | 30 (6) | 17 (7) | 13 (6) |
|
| Tumor grade, n
(%) |
|
|
| 0.291 |
|
Low | 15 (3) | 6 (2) | 9 (4) |
|
|
High | 450 (97) | 253 (98) | 197 (96) |
|
| Lymphovascular
invasion, n (%) |
|
|
| >0.999 |
|
Absent | 324 (70) | 180 (69) | 144 (70) |
|
|
Present | 141 (30) | 79 (31) | 62 (30) |
|
| Adjuvant
chemotherapy, n (%) |
|
|
| 0.467 |
| No | 411 (88) | 226 (87) | 185 (90) |
|
|
Yes | 54 (12) | 33 (13) | 21 (10) |
|
| Tumor necrosis, n
(%) |
|
|
| 0.185 |
| No | 380 (82) | 206 (80) | 174 (84) |
|
|
Yes | 85 (18) | 53 (20) | 32 (16) |
|
Association between SV and peripheral
blood parameters
Given that SV is associated with systemic immune
status (17,23), the differences in peripheral immune
cell counts and percentages between high and low SV were evaluated.
Results from a comparative analysis (Table II) showed significantly lower
lymphocyte count (P=0.001), lym% (P<0.001) and neutrophil
percentage (P=0.001) in patients with high SV compared to those
with low SV. No significant difference in platelet count (P=0.167),
monocyte count (P=0.959), monocyte percentage (P=0.795), white
blood cell count (0.079) or neutrophil count (0.251) was noted.
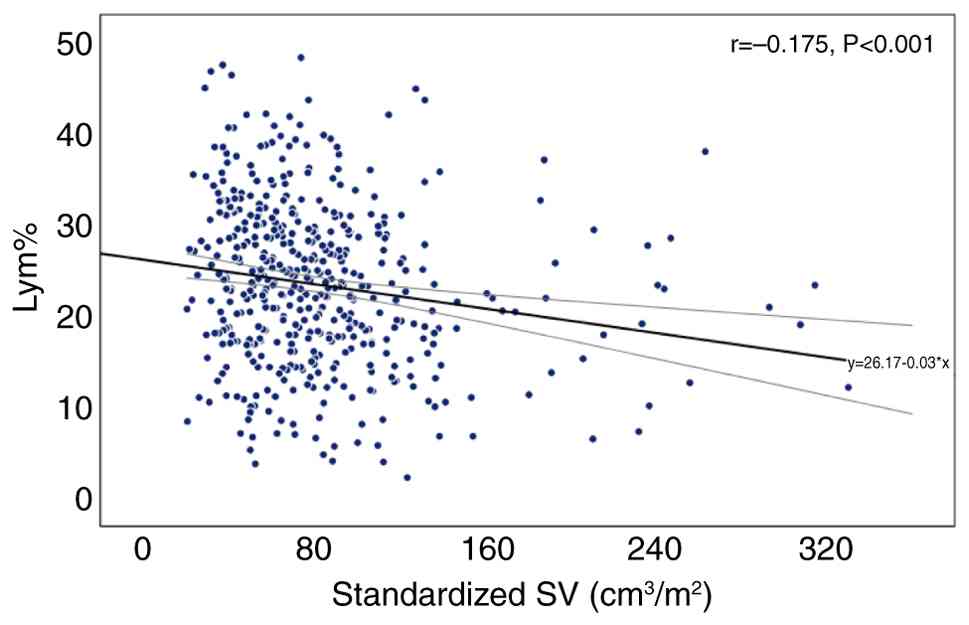
Furthermore, Pearson's correlation showed that the standardized SV
was negatively associated with lym% (r=−0.175, P<0.001)
(Fig. 2). Additionally, when
patients were stratified based on SV and lym%, higher neutrophil
percentage showed a trend toward lower lym%, independent of SV
(Fig. S2).
 | Table II.Comparison of peripheral blood
parameters in patients with low and high SV. |
Table II.
Comparison of peripheral blood
parameters in patients with low and high SV.
| Serum immune
cells | Low SV | High SV | P-value |
|---|
| White blood cell
counts (×109/l) | 6,600 (2,600) | 6,300 (2,900) | 0.079 |
| Lymphocyte count
(×109/l) | 1,602 (801) | 1,389 (727) | 0.001 |
| Lymphocyte
percentage | 24.8 (13.0) | 21.7 (12.2) | <0.001 |
| Neutrophil count
(×109/l) | 4,320 (2,406) | 4,163 (2,156) | 0.241 |
| Neutrophil
percentage | 64.9 (15.2) | 67.6 (11.5) | 0.001 |
| Monocyte count
(×109/l) | 507 (234) | 504 (297) | 0.959 |
| Monocyte
percentage | 7.9 (3.2) | 7.7 (3.5) | 0.795 |
| Platelet
(×109/l) | 225 (88) | 212 (100) | 0.167 |
Impact of SV and lym% on oncological
outcomes
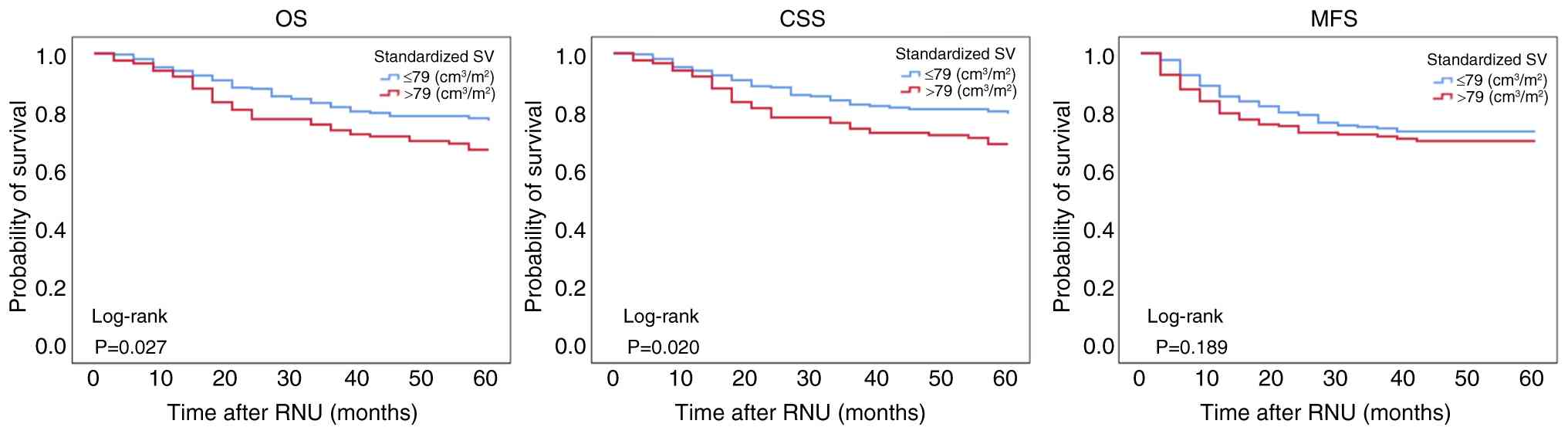
Kaplan-Meier analysis revealed that patients with
high SV had significantly shorter OS (P=0.027) and CSS (P=0.020)
times, and also showed a non-significant trend toward poorer MFS
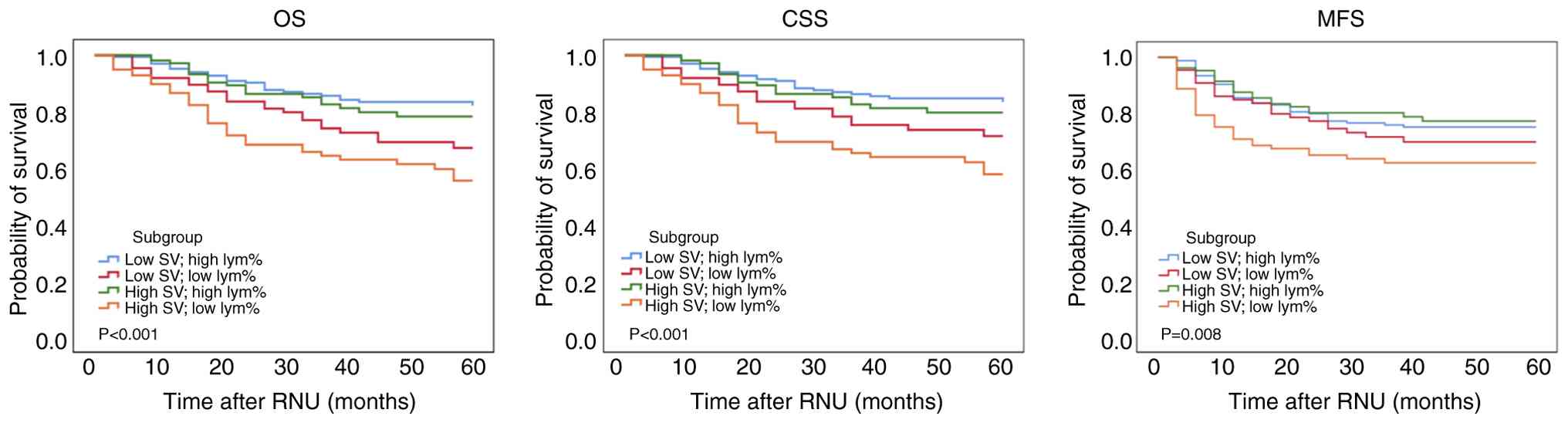
time (P=0.189) (Fig. 3). Given the
observed negative correlation between SV and peripheral lym%, both
parameters were further integrated to assess their combined impact
on patient outcomes. Patients with high SV and low lym% exhibited
significantly worse OS (P<0.001), CSS (P<0.001) and MFS
(P=0.008) times compared with those with high SV and high lym%, or
low SV regardless of lym% status (Fig.
4).
To assess the prognostic significance of the
combined SV and lym%, univariate Cox regression analyses were
performed for OS, CSS and MFS (Table
III, Table IV, Table V). In the univariate analysis,
factors significantly associated with poorer outcomes across all
three survival endpoints included the combination of high SV with
low lym%, LVI, lymph node involvement, multifocal tumor location
(involving both renal pelvis and ureter), advanced pathological T
stage (pT stage), male sex and concomitant bladder cancer. Notably,
concomitant bladder cancer and the low SV with low lym% subgroup
were associated with worse OS and CSS, but not MFS. Multivariate
analyses identified LVI, lymph node involvement, advanced pT stage,
multifocal tumor location (involving both renal pelvis and ureter),
male sex, and high SV with low lym% as consistent independent
predictors of inferior OS, CSS and MFS.
 | Table III.Univariate and multivariate Cox
regression analyses for predicting cancer-specific survival in
patients with upper tract urothelial carcinoma after RNU. |
Table III.
Univariate and multivariate Cox
regression analyses for predicting cancer-specific survival in
patients with upper tract urothelial carcinoma after RNU.
|
| Univariate | Multivariate |
|---|
|
|
|
|
|---|
| Characteristic | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age at RNU |
|
|
|
|
| >69
vs. ≤69 years | 1.472
(0.997–2.172) | 0.052 | 1.722
(1.154–2.568) | 0.008 |
| Sex |
|
|
|
|
| Female
vs. male | 0.475
(0.321–0.702) | <0.001 | 0.561
(0.373–0.845) | 0.006 |
| Renal function
status |
|
|
|
|
| eGFR
<60 vs. ≥60 ml/min/1.73 m2 | 1.317
(0.882–1.965) | 0.178 |
|
|
| Hematuria |
|
|
|
|
| Yes vs.
no | 0.890
(0.523–1.516) | 0.668 |
|
|
| Hydronephrosis |
|
|
|
|
| Yes vs.
no | 1.187
(0.7361.915) | 0.483 |
|
|
| Previous BC |
|
|
|
|
| Yes vs.
no | 1.120
(0.666–1.882) | 0.670 |
|
|
| Concomitant BC |
|
|
|
|
| Yes vs.
no | 1.614
(1.052–2.476) | 0.028 | 0.937
(0.583–1.505) | 0.788 |
| Tumor location |
|
|
|
|
| Ureter
vs. renal pelvis | 0.870
(0.539–1.404) | 0.569 | 1.241
(0.751–2.050) | 0.400 |
| Both
vs. renal pelvis | 1.979
(1.263–3.100) | 0.003 | 1.739
(1.078–2.805) | 0.023 |
| Pathological T
stage |
|
|
|
|
| pT2 vs.
pTa/1 | 4.230
(1.641–10.906) | 0.003 | 3.468
(1.320–9.110) | 0.012 |
| pT3/4
vs. pTa/1 | 12.886
(5.627–29.511) | <0.001 | 9.216
(3.887–21.852) | <0.001 |
| Lymph node
involvement |
|
|
|
|
| N+ vs.
Nx/0 | 6.097
(3.728–9.969) | <0.001 | 3.862
(2.187–6.819) | <0.001 |
| Tumor grade |
|
|
|
|
| High
vs. low | 3.236
(0.451–23.190) | 0.243 |
|
|
| Lymphovascular
invasion |
|
|
|
|
|
Presence vs. absence | 3.854
(2.613–5.686) | <0.001 | 1.857
(1.197–2.883) | 0.006 |
| Adjuvant
chemotherapy |
|
|
|
|
| Yes vs.
no | 1.321
(0.764–2.283) | 0.319 | 0.382
(0.203–0.717) | 0.003 |
| Standardized SV and
lym% subgroup |
|
|
|
|
| Low SV
with high lym% | Reference |
| Reference |
|
| Low SV
with low lym% | 1.795
(1.029–3.131) | 0.039 | 1.611
(0.911–2.850) | 0.101 |
| High SV
with high lym% | 1.176
(0.654–2.116) | 0.588 | 1.140
(0.626–2.077) | 0.668 |
| High SV
with low lym% | 2.847
(1.727–4.692) | <0.001 | 2.142
(1.253–3.662) | 0.005 |
 | Table IV.Univariate and multivariate Cox
regression analyses for predicting overall survival in patients
with upper tract urothelial carcinoma after RNU. |
Table IV.
Univariate and multivariate Cox
regression analyses for predicting overall survival in patients
with upper tract urothelial carcinoma after RNU.
|
| Univariate | Multivariate |
|---|
|
|
|
|
|---|
| Characteristic | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age at RNU |
|
|
|
|
| >69
vs. ≤69 years | 1.493
(1.025–2.173) | 0.037 | 1.729
(1.176–2.542) | 0.005 |
| Sex |
|
|
|
|
| Female
vs. male | 0.525
(0.362–0.763) | 0.001 | 0.603
(0.408–0.892) | 0.011 |
| Renal function
status |
|
|
|
|
| eGFR
<60 vs. ≥60 ml/min/1.73 m2 | 1.474
(0.995–2.184) | 0.053 |
|
|
| Hematuria |
|
|
|
|
| Yes vs.
no | 0.907
(0.541–1.519) | 0.710 |
|
|
| Hydronephrosis |
|
|
|
|
| Yes vs.
no | 1.161
(0.734–1.836) | 0.523 |
|
|
| Previous BC |
|
|
|
|
| Yes vs.
no | 1.254
(0.773–20.32) | 0.359 |
|
|
| Concomitant BC |
|
|
|
|
| Yes vs.
no | 1.670
(1.109–2.515) | 0.014 | 1.013
(0.645–1.592) | 0.954 |
| Tumor location |
|
|
|
|
| Ureter
vs. renal pelvis | 0.948
(0.601–1.495) | 0.818 | 1.333
(0.827–2.150) | 0.238 |
| Both
vs. renal pelvis | 2.003
(1.293–3.102) | 0.002 | 1.759
(1.106–2.799) | 0.017 |
| Pathological T
stage |
|
|
|
|
| pT2 vs.
pTa/1 | 2.391
(1.142–5.008) | 0.021 | 1.932
(0.903–4.137) | 0.090 |
| pT3/4
vs. pTa/1 | 6.489
(3.543–11.887) | <0.001 | 4.601
(2.397–8.831) | <0.001 |
| Lymph node
involvement |
|
|
|
|
| N+ vs.
Nx/0 | 5.749
(3.529–9.365) | <0.001 | 3.968
(2.253–6.990) | <0.001 |
| Tumor grade |
|
|
|
|
| High
vs. low | 1.738
(0.429–7.036) | 0.439 |
|
|
| Lymphovascular
invasion |
|
|
|
|
|
Presence vs. absence | 3.534
(2.436–5.129) | <0.001 | 1.898
(1.237–2.912) | 0.003 |
| Adjuvant
chemotherapy |
|
|
|
|
| Yes vs.
no | 1.227
(0.712–2.114) | 0.461 | 0.385
(0.206–0.723) | 0.003 |
| Standardized SV and
lym% subgroup |
|
|
|
|
| Low SV
with high lym% |
|
|
|
|
| Low SV
with low lym% | 1.902
(1.120–3.231) | 0.017 | 1.743
(1.014–2.996) | 0.044 |
| High SV
with high lym% | 1.160
(0.656–2.051) | 0.609 | 1.158
(0.647–2.073) | 0.621 |
| High SV
with low lym% | 2.821
(1.738–4.577) | <0.001 | 2.201
(1.314–3.688) | 0.003 |
 | Table V.Univariate and multivariate Cox
regression analyses for predicting metastasis-free survival in
patients with upper tract urothelial carcinoma after radical
nephroureterectomy. |
Table V.
Univariate and multivariate Cox
regression analyses for predicting metastasis-free survival in
patients with upper tract urothelial carcinoma after radical
nephroureterectomy.
|
| Univariate | Multivariate |
|---|
|
|
|
|
|---|
| Characteristic | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age at RNU,
years |
|
|
|
|
| >69
vs. ≤69 | 1.005
(0.704–1.435) | 0.978 | 1.203
(0.833–1.737) | 0.325 |
| Sex |
|
|
|
|
| Female
vs. male | 0.572
(0.399–0.817) | 0.002 | 0.658
(0.453–0.954) | 0.037 |
| Renal function
status, ml/min/1.73 m2 |
|
|
|
|
| eGFR
<60 vs. ≥60 | 0.942
(0.657–1.350) | 0.942 |
|
|
| Hematuria |
|
|
|
|
| Yes vs.
no | 0.832
(0.505–1.373) | 0.473 |
|
|
| Hydronephrosis |
|
|
|
|
| Yes vs.
no | 0.991
(0.646–1.521) | 0.969 |
|
|
| Previous BC |
|
|
|
|
| Yes vs.
no | 1.239
(0.774–1.983) | 0.372 |
|
|
| Concomitant BC |
|
|
|
|
| Yes vs.
no | 1.461
(0.971–2.197) | 0.069 |
|
|
| Tumor location |
|
|
|
|
| Ureter
vs. renal pelvis | 1.048
(0.680–1.616) | 0.832 | 1.674
(1.063–2.637) | 0.026 |
| Both
vs. renal pelvis | 2.057
(1.339–3.162) | 0.001 | 2.212
(1.428–3.425) | <0.001 |
| Pathological T
stage |
|
|
|
|
| pT2 vs.
pTa/1 | 2.952
(1.224–7.122) | 0.016 | 2.320
(0.949–5.672) | 0.065 |
| pT3/4
vs. pTa/1 | 12.885
(6.266–26.495) | <0.001 | 8.707
(4.100–18.490) | <0.001 |
| Lymph node
involvement |
|
|
|
|
| N+ vs.
Nx/0 | 5.842
(3.506–9.733) | <0.001 | 3.292
(1.843–5.880) | <0.001 |
| Tumor grade |
|
|
|
|
| High
vs. low | 4.029
(0.563–28.838) | 0.165 |
|
|
| Lymphovascular
invasion |
|
|
|
|
|
Presence vs. absence | 4.305
(2.996–6.186) | <0.001 | 2.060
(1.388–3.057) | <0.001 |
| Adjuvant
chemotherapy |
|
|
|
|
| Yes vs.
no | 2.584
(1.673–3.993) | <0.001 | 0.786
(0.475–1.302) | 0.350 |
| Standardized SV and
lym% subgroup |
|
|
|
|
| Low SV
with high lym% |
|
|
|
|
| Low SV
with low lym% | 1.234
(0.744–2.046) | 0.416 | 1.076
(0.641–1.807) | 0.782 |
| High SV
with high lym% | 0.889
(0.528–1.495) | 0.656 | 0.857
(0.502–1.463) | 0.571 |
| High SV
with low lym% | 1.834
(1.165–2.888) | 0.009 | 1.649
(1.031–2.636) | 0.037 |
Discussion
In the present retrospective cohort study of 465
patients with non-metastatic UTUC who underwent RNU, a higher
preoperative SV and lower peripheral lym% were found to be
independently associated with inferior OS, CSS and MFS. Notably,
the combined presence of high SV and low lym% was the strongest
prognostic indicator across all endpoints. These findings suggest
that baseline immune and hematological parameters, quantified
through imaging and routine blood tests, may offer valuable
insights into host-tumor dynamics and support individualized risk
stratification.
The present analysis expands on existing literature
linking splenomegaly to adverse outcomes in solid malignancies.
Prior studies have demonstrated that elevated SV is associated with
a poor prognosis in hepatocellular carcinoma, pancreatic cancer,
gastric cancer, metastatic colorectal cancer and non-small cell
lung cancer (17,20,21,24).
Consistent with these observations, the present study found that
larger baseline SV predicted worse oncological outcomes in UTUC.
Importantly, to the best of our knowledge, the present study is the
first to evaluate SV in the context of urothelial carcinoma of the
upper tract and to integrate peripheral immune status (lym%) as a
complementary biomarker.
While the mechanistic link between splenic
remodeling and cancer outcomes remains under investigation,
accumulating evidence from preclinical models supports the role of
the spleen in tumor-induced immune suppression (23,25).
Han et al (26) found that
tumor-bearing mice produced erythroblast-like Ter cells in the
spleen that promoted hepatocellular carcinoma progression.
Additional studies by Zhao et al (27) and Sano et al (28) identified
CD71+TER119+ erythroid progenitor cells in
the spleen that exert immunosuppressive effects on effector T
cells. Moreover, the spleen has been shown to facilitate the
generation and expansion of MDSCs in the tumor-bearing state,
further contributing to an immunosuppressive milieu and enhanced
tumor aggressiveness (23,29,30).
Jiang et al (31)
demonstrated a positive correlation between splenic weight and
serum mMDSC levels in a mouse model of hepatocellular carcinoma.
The clinical relevance of these findings is underscored by the
study by Levy et al (32),
which demonstrated that splenectomy inhibited tumor growth and
metastasis in non-small cell lung cancer by depleting MDSCs and
enhancing cytotoxic T-cell activation. The present findings may
therefore reflect a broader impairment of systemic immunity in
UTUC. An enlarged spleen may indicate an activated yet functionally
exhausted immune state, accompanied by peripheral lymphocyte
depletion, diminished antitumor immune surveillance and a
pro-inflammatory milieu. Additionally, tumor-stroma interactions
likely amplify this process by promoting cytokine-driven immune
evasion, neutrophil recruitment and microenvironmental
immunosuppression (17,19,29).
Together, these mechanisms offer a biological explanation for why
patients with high standardized SV and low circulating lym% are
prone to early metastasis and inferior oncological outcomes.
The combination of imaging-derived SV and routine
lym% may serve as a non-invasive, cost-effective method for
identifying high-risk patients with UTUC who might benefit from
intensified surveillance or consideration of adjuvant systemic
therapies. Given the lack of robust predictive biomarkers in this
population, the present results provide a potentially actionable
stratification tool that complements established
clinicopathological factors such as stage, LVI and nodal
status.
Nevertheless, several limitations warrant
consideration. First, the retrospective nature and single-center
design of the present study may introduce selection bias and limit
generalizability. Second, the study was lacking post-operative
immune profiling or longitudinal SV measurements, which restricts
the ability to evaluate dynamic immunological changes over time.
Finally, although ROC-based cutoffs were used to dichotomize SV and
lym%, external validation of these thresholds is necessary before
clinical application.
In conclusion, higher standardized SV combined with
lower circulating lym% was identified as a novel, independent
predictor of worse survival outcomes in patients with UTUC
following RNU. These findings suggest that incorporating baseline
standardized SV and lym%, alongside established clinicopathological
risk factors, can enhance risk stratification and support more
individualized postoperative management strategies.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Professor
Sheng-Hsiang Lin (Biostatistics Consulting Center, National Cheng
Kung University Hospital, Tainan, Taiwan), for providing
statistical consulting services.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CHL and HCJ were responsible for the study concept
and design. Data acquisition was performed by CHL, CYH, CHO and
HCJ. Quality control of data and algorithms was performed by KCL,
CYH and CHO. Data analysis and interpretation was performed by CHL,
KCL and CAW. The statistical analysis was performed by CHL and KCL.
CHL and KCL prepared the manuscript. The manuscript was edited by
CHO and HCJ, and reviewed by CAW, CYH, CHO and HCJ. CHL and HCJ
confirm the authenticity of all the raw data. All authors have read
and agreed to the final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Review Board of National Cheng Kung University Hospital (Tainan,
Taiwan; approval no. B-ER-112-214). The requirement for informed
consent was waived by the IRB due to the retrospective nature of
the study, and access to follow-up clinical records was granted.
All procedures were conducted in accordance with the ethical
standards outlined in the Declaration of Helsinki.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Raman JD, Messer J, Sielatycki JA and
Hollenbeak CS: Incidence and survival of patients with carcinoma of
the ureter and renal pelvis in the USA, 1973–2005. BJU Int.
107:1059–1064. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Colin P, Koenig P, Ouzzane A, Berthon N,
Villers A, Biserte J and Rouprêt M: Environmental factors involved
in carcinogenesis of urothelial cell carcinomas of the upper
urinary tract. BJU Int. 104:1436–1440. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rouprêt M, Babjuk M, Burger M, Capoun O,
Cohen D, Comperat EM, Cowan NC, Dominguez-Escrig JL, Gontero P,
Hugh Mostafid A, et al: European Association of Urology guidelines
on upper urinary tract urothelial carcinoma: 2020 update. Eur Urol.
79:62–79. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Clark PE, Agarwal N, Biagioli MC,
Eisenberger MA, Greenberg RE, Herr HW, Inman BA, Kuban DA, Kuzel
TM, Lele SM, et al: Bladder cancer. J Natl Compr Canc Netw.
11:446–475. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rouprêt M, Zigeuner R, Palou J, Boehle A,
Kaasinen E, Sylvester R, Babjuk M and Oosterlinck W: European
guidelines for the diagnosis and management of upper urinary tract
urothelial cell carcinomas: 2011 update. Eur Urol. 59:584–594.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rouprêt M, Seisen T, Birtle AJ, Capoun O,
Compérat EM, Dominguez-Escrig JL, Gürses Andersson I, Liedberg F,
Mariappan P, Hugh Mostafid A, et al: European Association of
Urology guidelines on upper urinary tract urothelial carcinoma:
2023 update. Eur Urol. 84:49–64. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Oswald D, Pallauf M, Deininger S, Törzsök
P, Sieberer M and Eiben C: Neoadjuvant chemotherapy before
nephroureterectomy in high-risk upper tract urothelial cancer: A
systematic review and meta-analysis. Cancers (Basel). 14:48412022.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Piombino C, Tonni E, Oltrecolli M, Pirola
M, Pipitone S, Baldessari C, Dominici M, Sabbatini R and Vitale MG:
Immunotherapy in urothelial cancer: Current status and future
directions. Expert Rev Anticancer Ther. 23:1141–1155. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Birtle A, Johnson M, Chester J, Jones R,
Dolling D, Bryan RT, Harris C, Winterbottom A, Blacker A, Catto
JWF, et al: Adjuvant chemotherapy in upper tract urothelial
carcinoma (the POUT trial): A phase 3, open-label, randomised
controlled trial. Lancet. 395:1268–1277. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jan HC, Hu CY, Yang WH and Ou CH:
Combination of Platelet-lymphocyte ratio and Monocyte-lymphocyte
ratio as a new promising prognostic factor in upper tract
urothelial carcinoma with large Tumor Sizes >3 cm. Clin
Genitourin Cancer. 18:e484–e500. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li T, Xu H, Yang L, Tan P and Wei Q:
Predictive value of preoperative lymphocyte-to-monocyte ratio for
patients with upper tract urothelial carcinoma. Clin Chim Acta.
492:50–56. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vartolomei MD, Kimura S, Ferro M,
Vartolomei L, Foerster B, Abufaraj M and Shariat SF: Is
neutrophil-to-lymphocytes ratio a clinical relevant preoperative
biomarker in upper tract urothelial carcinoma? A meta-analysis of
4385 patients. World J Urol. 36:1019–1029. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jan HC, Yang WH and Ou CH: Combination of
the preoperative systemic Immune-inflammation index and
Monocyte-lymphocyte ratio as a novel prognostic factor in patients
with Upper-tract urothelial carcinoma. Ann Surg Oncol. 26:669–684.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Steenbrugge J, De Jaeghere EA, Meyer E,
Denys H and De Wever O: Splenic hematopoietic and stromal cells in
cancer progressionthe spleen in cancer progression. Cancer Res.
81:27–34. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mebius RE and Kraal G: Structure and
function of the spleen. Nat Rev Immunol. 5:606–616. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bae JS, Lee DH, Yoo J, Yi NJ, Lee KW, Suh
KS, Kim H and Lee KB: Association between spleen volume and the
post-hepatectomy liver failure and overall survival of patients
with hepatocellular carcinoma after resection. Eur Radiol.
31:2461–2471. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zeng Z, Liu Z, Li J, Sun J, Ma M, Ye X, Yu
J and Kang W: Baseline splenic volume as a biomarker for clinical
outcome and circulating lymphocyte count in gastric cancer. Front
Oncol. 12:10657162023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Galland L, Lecuelle J, Favier L, Fraisse
C, Lagrange A, Kaderbhai C, Truntzer C and Ghiringhelli F: Splenic
volume as a surrogate marker of immune checkpoint inhibitor
efficacy in metastatic non small cell lung cancer. Cancers.
13:30202021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cortez-Retamozo V, Etzrodt M, Newton A,
Rauch PJ, Chudnovskiy A, Berger C, Ryan RJ, Iwamoto Y, Marinelli B,
Gorbatov R, et al: Origins of tumor-associated macrophages and
neutrophils. Proc Natl Acad Sci USA. 109:2491–2496. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Niogret J, Limagne E, Thibaudin M, Blanc
J, Bertaut A, Le Malicot K, Rinaldi Y, Caroli-Bosc FX, Audemar F,
Nguyen S, et al: Baseline splenic volume as a prognostic biomarker
of FOLFIRI efficacy and a surrogate marker of MDSC accumulation in
metastatic colorectal carcinoma. Cancers (Basel). 12:14292020.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Müller L, Gairing SJ, Kloeckner R,
Foerster F, Weinmann A, Mittler J, Stoehr F, Emrich T, Düber C,
Galle PR and Hahn F: Baseline splenic volume outweighs
Immuno-modulated size changes with regard to survival outcome in
patients with hepatocellular carcinoma under immunotherapy. Cancers
(Basel). 14:35742022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Koo TK and Li MY: A guideline of selecting
and reporting intraclass correlation coefficients for reliability
research. J Chiropr Med. 15:155–163. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jordan KR, Kapoor P, Spongberg E, Tobin
RP, Gao D, Borges VF and McCarter MD: Immunosuppressive
myeloid-derived suppressor cells are increased in splenocytes from
cancer patients. Cancer Immunol Immunother. 66:503–513. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Aarnink A, Richard C, Truntzer C, Vincent
J, Bengrine L, Vienot A, Borg C and Ghiringhelli F: Baseline
splenic volume as a surrogate marker of FOLFIRINOX efficacy in
advanced pancreatic carcinoma. Oncotarget. 9:256172018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ugel S, Peranzoni E, Desantis G, Chioda M,
Walter S, Weinschenk T, Ochando JC, Cabrelle A, Mandruzzato S and
Bronte V: Immune tolerance to tumor antigens occurs in a
specialized environment of the spleen. Cell Rep. 2:628–639. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Han Y, Liu Q, Hou J, Gu Y, Zhang Y, Chen
Z, Fan J, Zhou W, Qiu S, Zhang Y, et al: Tumor-induced generation
of splenic Erythroblast-like Ter-cells promotes tumor progression.
Cell. 184:13922021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao L, He R, Long H, Guo B, Jia Q, Qin D,
Liu SQ, Wang Z, Xiang T, Zhang J, et al: Late-stage tumors induce
anemia and immunosuppressive extramedullary erythroid progenitor
cells. Nat Med. 24:1536–1544. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sano Y, Yoshida T, Choo MK,
Jiménez-Andrade Y, Hill KR, Georgopoulos K and Park JM: Multiorgan
signaling mobilizes Tumor-associated erythroid cells expressing
immune checkpoint molecules. Mol Cancer Res. 19:507–515. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu C, Ning H, Liu M, Lin J, Luo S, Zhu W,
Xu J, Wu WC, Liang J, Shao CK, et al: Spleen mediates a distinct
hematopoietic progenitor response supporting tumor-promoting
myelopoiesis. J Clin Invest. 128:3425–3438. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tavukcuoglu E, Horzum U, Yanik H, Uner A,
Yoyen-Ermis D, Nural SK, Aydin B, Sokmensuer C, Karakoc D, Yilmaz
KB, et al: Human splenic polymorphonuclear myeloid-derived
suppressor cells (PMN-MDSC) are strategically located immune
regulatory cells in cancer. Eur J Immunol. 50:2067–2074. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jiang W, Li Y, Zhang S, Kong G and Li Z:
Association between cellular immune response and spleen weight in
mice with hepatocellular carcinoma. Oncol Lett. 22:6252021.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Levy L, Mishalian I, Bayuch R, Zolotarov
L, Michaeli J and Fridlender ZG: Splenectomy inhibits non-small
cell lung cancer growth by modulating anti-tumor adaptive and
innate immune response. Oncoimmunology. 4:e9984692015. View Article : Google Scholar : PubMed/NCBI
|