Introduction
Primary liver cancer (PLC) is the sixth most common
cancer and fourth leading cause of cancer-related mortality
worldwide, with >840,000 novel cases and 780,000 deaths annually
(1). There are three subtypes of
PLC: Hepatocellular carcinoma (HCC), intrahepatic
cholangiocarcinoma (ICC) and combined
hepatocellular-cholangiocarcinoma (CHC).
CHC is an uncommon and unique type of PLC with the
phenotypic characteristics of both HCC and ICC. The incidence of
CHC varies from 2.4 to 14.2% among studies (2–4). This
type of carcinoma contains explicit mixed HCC and ICC components.
Currently, there is no unified pathological classification of CHC.
The first scientific description of CHC was published in a study by
Allen and Lisa in 1949 (5), in
which this tumor was defined into three types: Type A (double
cancer; HCC and ICC located in different parts of the same liver),
type B (combined type; HCC and ICC existing in adjacent parts with
continuous growth) and type C (mixed type; HCC and ICC components
completely combined within the same tumor). In 1985, Goodman et
al (6) divided CHC into type I
(collision), characterized by the coincidence of HCC and ICC in the
same patient; type II (transition), characterized by the presence
of an identifiable transition between HCC and ICC; and type III
(fibrolamellar), similar to the fibrolamellar variants of HCC but
with the existence of pseudodoglands secreting mucin. However, in
2010, the World Health Organization classified CHC into two main
types: A typical type, which is similar to type C, as explained by
Allen and Lisa, and subtypes with stem cell characteristics, which
are divided into classic, intermediate and bile duct cell subtypes
(7).
However, due to the low prevalence of CHC, its
clinicopathological characteristics and prognosis vary among
studies. Certain studies have reported that the biological
characteristics of CHC are analogous to those of HCC, rather than
those of ICC (8–10). However, at the same time, other
studies have indicated that CHC is genetically more similar to ICC
compared with HCC (11,12). In order to gain a more comprehensive
understanding of the clinical pathologies and outcomes of CHC, the
present study, which aimed to compare the clinicopathological
characteristics and prognosis of CHC with HCC and ICC, and to
investigate the clinical prognostic factors in patients with CHC,
investigated the clinical characteristics, prognosis and
histogenesis of CHC after surgery.
Patients and methods
Patients
The clinical data of patients with PLC who underwent
a hepatectomy at Guangxi Medical University Cancer Hospital
(Nanning, China) between January 2014 and December 2018 were
retrospectively analyzed. R0 margins were observed in all cases.
CHC was diagnosed according to the definitions of types B and C
according to the classification by Allen and Lisa (5). Meanwhile, the exclusion criteria were
as follows: i) Patients who received preoperative chemotherapy,
radiotherapy or other non-surgical treatment; and ii) patients with
incomplete clinical data.
Patient history was reviewed for clinicopathological
information, including sex, age, viral hepatitis B and C status,
background liver disease, serum bilirubin, alanine transaminase
(ALT), aspartate transaminase (AST), carcinoembryonic antigen
(CEA), α-fetoprotein (AFP), carbohydrate antigen 19-9 (CA19-9) and
Child-Pugh status (13).
Pathological data included tumor number and size, presence of
capsules, cirrhosis, Edmondson-Steiner grading (14), satellite lesions, macrovascular and
microvascular invasion, and lymph node metastasis. Clinical and
pathological features, overall survival (OS) and disease-free
survival (DFS) were compared between patients with CHC and those
with HCC or ICC. The prognostic factors for OS and DFS, recurrence
patterns and management of patients with CHC were also
investigated.
Patients were regularly monitored for tumor
recurrence or distant metastasis every 3 months in the first 2
years and then every 3–6 months, with measurements of serum tumor
markers, liver function tests, chest radiography, color ultrasound,
dynamic contrast-enhanced computed tomography or magnetic resonance
imaging.
Immunohistochemistry
The present study performed immunohistochemical
analysis of CD90 and EpCAM to examine whether CHC arose from the
HPCs in the present study. Surgical tissues were fixed in 10%
formalin at room temperature for 24 h, embedded in paraffin, cut
into 5-µm tissue slices, deparaffinized in xylene and rehydrated
using graded alcohol solutions. Antigen retrieval was performed for
5 min at 100°C in citrate buffer (10 mmol/l; pH 6.0) in a pressure
cooker. Endogenous peroxidases were blocked at room temperature by
immersing sections in 3% hydrogen peroxide for 20 min. Tissue
samples were then incubated at 37°C for 2 h with a mouse monoclonal
antibody against human CD90 (1:200; cat. no. ab92574; Abcam) and a
mouse monoclonal antibody against human epithelial cell adhesion
molecule (EpCAM; 1:200; cat. no. ab223582; Abcam). The tissue
slices were washed with phosphate-buffered saline (PBS), incubated
with biotinylated anti-mouse immunoglobulin diluted in PBS for 30
min at room temperature and washed again with PBS. The sections
were incubated with anti-HRP conjugate for 10 min, rinsed with PBS
and incubated with diaminobenzidine for 10 min. Lastly, the
sections were counterstained at room temperature with hematoxylin.
A light microscope was used for analysis. For statistical analysis
(ImageJ; National Institutes of Health), patients were categorized
as positive for these markers if the percentage of cells positive
for CD90 or EpCAM was ≥5%.
Statistical analysis
Continuous variables are presented as the mean ± SD
or median ± IQR, with normally distributed data as the mean ± SD
and non-normally distributed data as the median ± IQR, and
categorical variables are presented as the frequency (percentage).
The significance of the baseline differences among the three groups
was evaluated using analysis of one-way variance with Bonferroni's
post hoc test for continuous variables or the Mann-Whitney U test
with Bonferroni's post hoc test for non-parametric variables. When
multiple comparisons are made among different groups, the risk of
Type I errors (false-positives) increases. Therefore, P-value
correction was applied to control for this, reducing the P-value
threshold by dividing the α level (0.05) by the number of
comparisons (n=3), resulting in a new significance threshold of
0.0167. The χ2 test or Fisher's exact test was used to
compare categorical data. Survival analysis was performed using the
Kaplan-Meier method and intergroup differences were assessed using
the log-rank test. The prognostic risk factors for poor DFS and OS
were assessed using univariate and multivariate Cox regression
analyses. The SPSS statistical software (version 24.0; IBM Corp.)
was used for all analyses and P<0.05 was considered to indicate
a statistically significant difference.
Results
Clinical data
The baseline characteristics of the patients with
CHC, HCC and ICC are provided in Table
I. There were 22 men (84.6%) in the CHC group, the male:female
ratio was 5.5:1 and the mean age was 53.2±9.2 years (range, 34–68
years). There were no significant differences in sex distribution
or mean age in the three groups. In total, 13 patients (50.0%) had
hepatitis B virus (HBV), while no patient had hepatitis C virus
(HCV) infection with CHC, and significantly fewer of these patients
had HBV infection compared with those in the HCC group
(P<0.001). There were no significant differences in serum total
bilirubin, ALT, AST and CA19-9 among the CHC, HCC and ICC groups
(P=0.057). The serum CEA level in the CHC group was significantly
higher compared with that in the HCC group (P=0.001) but similar to
that in the ICC group (P=0.704). No significant differences were
observed in terms of Child-Pugh status, tumor capsule, tumor
number, tumor size, Edmondson-Steiner grading and satellite lesions
among the three groups (all P>0.05).
 | Table I.Baseline characteristics of patients
with CHC, HCC and ICC. |
Table I.
Baseline characteristics of patients
with CHC, HCC and ICC.
|
|
|
|
| P-value |
|---|
|
|
|
|
|
|
|---|
|
Characteristics | CHC (n=26) | HCC (n=901) | ICC (n=40) | CHC vs. HCC vs.
ICC | CHC vs. HCC | CHC vs. ICC | HCC vs. ICC |
|---|
| Sex, n (%) |
|
|
| 0.001a | 0.839 | 0.080 |
<0.001a |
|
Male | 22 (84.6) | 775 (86.0) | 26 (65.0) |
|
|
|
|
|
Female | 4 (15.4) | 126 (14.0) | 14 (35.0) |
|
|
|
|
| Mean age ± SD,
years | 53.2±9.2 | 49.8±10.8 | 55.9±11.1 | 0.001a | 0.085 | 0.264 | 0.001a |
| Diabetes, n
(%) |
|
|
| 0.625 | 0.340 | 0.356 | 0.886 |
| No | 25 (96.2) | 817 (90.7) | 36 (90.0) |
|
|
|
|
|
Yes | 1 (3.8) | 84 (9.3) | 4 (10.0) |
|
|
|
|
| HBV viral
infection, n (%) |
|
|
|
<0.001a |
<0.001a | 0.843 |
<0.001a |
| No | 13 (50.0) | 112 (12.4) | 21 (52.5) |
|
|
|
|
|
Yes | 13 (50.0) | 789 (87.6) | 19 (47.5) |
|
|
|
|
| HCV viral
infection, n (%) |
|
|
| 0.691 | 0.589 |
| 0.503 |
| No | 26 (100.0) | 891 (98.9) | 40 (100.0) |
|
|
|
|
|
Yes | 0 (0.0) | 10 (1.1) | 0 (0.0) |
|
|
|
|
| Total bilirubin,
U/mlb | 12.3±4.8 | 15.0±10.0 | 14.2±18.4 | 0.001a | 0.100 | 0.256 | 0.001a |
| ALT,
U/mlb | 46.5±54.2 | 44.6±42.8 | 32.1±31.2 |
<0.001a | 0.098 | 0.748 |
<0.001a |
| AST,
U/mlb | 44.3±31.0 | 52.6±47.0 | 33.1±16.7 |
<0.001a | 0.101 | 0.284 | <0.001 |
| CEA,
ng/mlb | 73.1±234.3 | 3.0±5.4 | 33.9±98.6 |
<0.001a | 0.001a | 0.704 | 0.002a |
| CA19-9,
U/mlb | 111.4±265.8 | 28.8±53.4 | 343.1±437.9 |
<0.001a | 0.176 | 0.149 |
<0.001a |
| AFP, n (%) |
|
|
|
<0.001a | 0.049a | 0.032a |
<0.001a |
| ≤400
ng/ml | 19 (73.1) | 483 (53.6) | 37 (92.5) |
|
|
|
|
| >400
ng/ml | 7 (26.9) | 418 (46.4) | 3 (7.5) |
|
|
|
|
| Child-Pugh, n
(%) |
|
|
| 0.223 | 0.083 | 0.322 | 0.945 |
| A | 24 (92.3) | 880 (97.7) | 39 (97.5) |
|
|
|
|
| B | 2 (7.7) | 21 (2.3) | 1 (2.5) |
|
|
|
|
| Liver cirrhosis, n
(%) |
|
|
|
<0.001a | 0.021a |
<0.01a |
<0.001a |
| No | 7 (26.9) | 449 (49.8) | 33 (82.5) |
|
|
|
|
|
Yes | 19 (73.1) | 452 (50.2) | 7 (17.5) |
|
|
|
|
| Tumor capsular, n
(%) |
|
|
| 0.057 | 0.241 | 0.033a | 0.042a |
| No | 4 (15.4) | 230 (25.5) | 16 (40.0) |
|
|
|
|
|
Yes | 22 (84.6) | 671 (74.5) | 24 (60.0) |
|
|
|
|
| Tumor number, n
(%) |
|
|
| 0.517 | 0.610 | 0.318 | 0.592 |
|
Solitary | 17 (65.4) | 631 (70.0) | 31 (77.5) |
|
|
|
|
|
Multiple | 9 (34.6) | 270 (30.0) | 9 (22.5) |
|
|
|
|
| Tumor size, n
(%) |
|
|
| 0.539 | 0.636 | 0.305 | 0.322 |
| ≤5
cm | 11 (42.3) | 340 (37.7) | 12 (30.0) |
|
|
|
|
| >5
cm | 15 (57.7) | 561 (62.3) | 28 (70.0) |
|
|
|
|
| Macrovascular
invasion, n (%) |
|
|
| 0.440 | 0.442 | 0.219 | 0.318 |
| No | 20 (76.9) | 630 (69.9) | 25 (62.5) |
|
|
|
|
|
Yes | 6 (23.1) | 271 (30.1) | 15 (37.5) |
|
|
|
|
| Microvascular
invasion, n (%) |
|
|
| 0.091 | 0.067 | 0.491 | 0.210 |
| No | 19 (73.1) | 495 (54.9) | 26 (65.0) |
|
|
|
|
|
Yes | 7 (26.9) | 406 (45.1) | 14 (35.0) |
|
|
|
|
| Edmondson-Steiner
grading, n (%) |
|
|
| 0.164 | 0.517 | 0.105 | 0.078 |
| I and
II | 9 (34.6) | 369 (41.0) | 22 (55.0) |
|
|
|
|
| III and
IV | 17 (65.4) | 532 (59.0) | 18 (45.0) |
|
|
|
|
| Satellite lesions,
n (%) |
|
|
| 0.971 | 0.852 | 0.966 | 0.872 |
| No | 22 (84.6) | 774 (85.9) | 34 (85.0) |
|
|
|
|
|
Yes | 4 (15.4) | 127 (14.1) | 6 (15.0) |
|
|
|
|
| Lymph node
metastasis, n (%) |
|
|
|
<0.001a |
<0.001a | 0.286 |
<0.001a |
| No | 21 (80.8) | 889 (98.7) | 36 (90.0) |
|
|
|
|
|
Yes | 5 (19.2) | 12 (1.3) | 4 (10.0) |
|
|
|
|
| CD34, n (%) |
|
|
|
<0.001a | 0.257 | 0.020a |
<0.001a |
|
Negative | 5 (19.2) | 107 (11.9) | 19 (47.5) |
|
|
|
|
|
Positive | 21 (80.8) | 794 (88.1) | 21 (52.5) |
|
|
|
|
| CK7, n (%) |
|
|
|
<0.001a |
<0.001a | 0.959 |
<0.001a |
|
Negative | 7 (26.9) | 697 (77.4) | 11 (27.5) |
|
|
|
|
|
Positive | 19 (73.1) | 204 (22.6) | 29 (72.5) |
|
|
|
|
| CK19, n (%) |
|
|
|
<0.001a |
<0.001a | 0.257 |
<0.001a |
|
Negative | 2 (7.7) | 684 (75.9) | 7 (17.5) |
|
|
|
|
|
Positive | 24 (92.3) | 217 (24.1) | 33 (82.5) |
|
|
|
|
| Glypican-3, n
(%) |
|
|
|
<0.001a | 0.119 |
<0.001a |
<0.001a |
|
Negative | 7 (26.9) | 118 (13.1) | 32 (80.0) |
|
|
|
|
|
Positive | 19 (73.1) | 783 (86.9) | 8 (20.0) |
|
|
|
|
In the CHC group, liver cirrhosis was significantly
more regular compared with that in the ICC group (P<0.01) but
was similar to that in the HCC group (P=0.021). However, there were
no significant differences in macrovascular and microvascular
invasion between the CHC group and the HCC and ICC groups (all
P>0.05). Lymph node metastasis was significantly more frequent
in the CHC group compared with that in the HCC group
(P<0.001).
No significant difference was observed in the
percentage of CD34 between the CHC group and the HCC and ICC
groups. The percentage of patients with glypcian-3 positivity in
the CHC group was similar to that in the HCC group (P>0.05) but
significantly higher compared with that in the ICC group
(P<0.05). By contrast, the distributions of both CK7- and
CK19+ patients in the CHC group were similar to those in
the ICC group (both P>0.05) but higher compared with those in
the HCC group (both P<0.05).
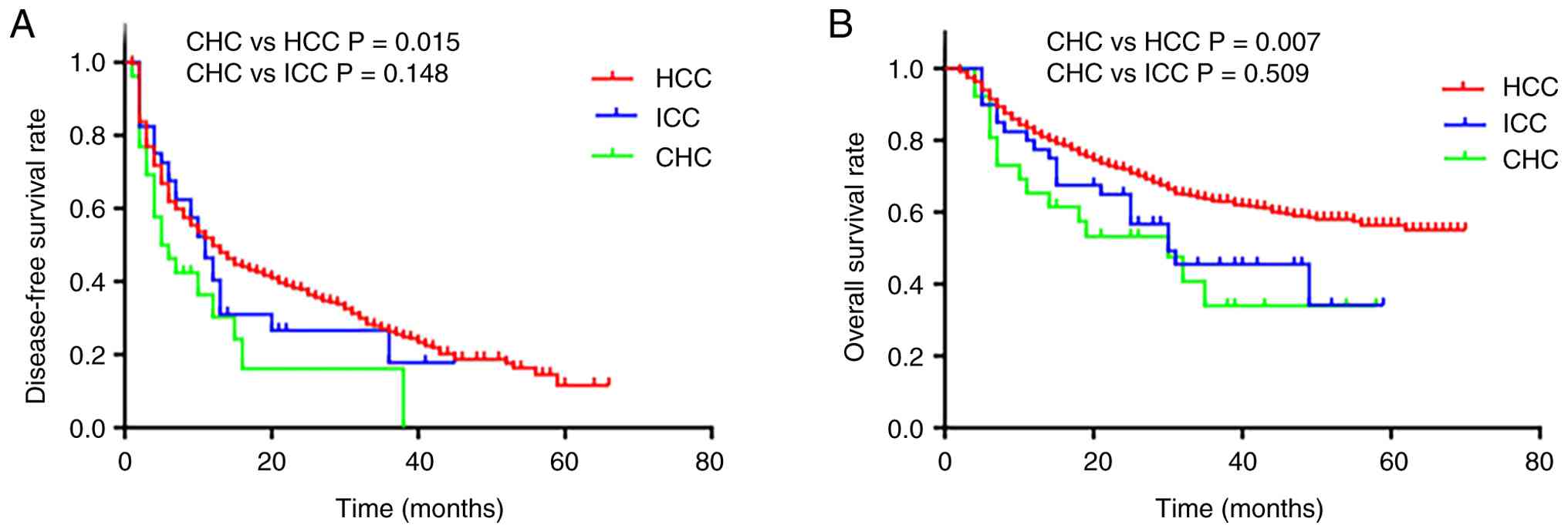
OS and DFS
The median OS time in the CHC group was 30 months
and the 1- and 3-year OS rates were 65.4 and 33.9%, respectively.
The median OS time in the HCC group was not reached and the 1- and
3-year OS rates were 82.1 and 63.2%, respectively. The median OS
time in the ICC group was 30 months and the 1- and 3-year OS rates
were 77.5 and 45.4%, respectively. The OS and DFS in the CHC group
were significantly lower compared with those in the HCC group (both
P<0.05) but not significantly different from those in the ICC
group (both P>0.05) (Fig.
1).
Univariate and multivariate
analysis
Univariate analysis demonstrated that AFP, CA19-9
and CEA levels, tumor size, macrovascular invasion and lymph node
metastasis were significant predictive factors for lower OS and DFS
in patients with CHC (both P<0.05) (Table II).
 | Table II.Univariate and multivariate analyses
of prognostic factors in patients with combined hepatocellular
cholangiocarcinoma. |
Table II.
Univariate and multivariate analyses
of prognostic factors in patients with combined hepatocellular
cholangiocarcinoma.
|
| Overall
survival | Disease-free
survival |
|---|
|
|
|
|
|---|
| Variables | Univariate analysis
P-value | Multivariate
analysis | Univariate
analysis | Multivariate
analysis |
|---|
|
|
|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Sex | 0.455 |
|
| 0.308 |
|
|
| Age (>50
years) | 0.374 |
|
| 0.144 |
|
|
| Diabetes | 0.284 |
|
| 0.578 |
|
|
| HBV viral
infection | 0.202 |
|
| 0.599 |
|
|
| Total bilirubin
(>21 U/ml) | 0.683 |
|
| 0.968 |
|
|
| AFP (>400
ng/ml) | 0.029 | 1.481
(0.363–6.041) | 0.584 | 0.015a | 2.509
(0.763–8.251) | 0.130 |
| CEA (>5
ng/ml) | 0.005 | 3.157
(0.775–12.866) | 0.109 | 0.009a | 1.645
(0.484–5.592) | 0.426 |
| CA19-9 (>5
U/ml) | 0.001 | 4.235
(0.938–19.131) | 0.061 | 0.013a | 2.350
(0.729–7.576) | 0.152 |
| ALT (>40
U/ml) | 0.551 |
|
| 0.469 |
|
|
| AST (>40
U/ml) | 0.245 |
|
| 0.318 |
|
|
| Child-Pugh | 0.817 |
|
| 0.986 |
|
|
| Liver
cirrhosis | 0.329 |
|
| 0.548 |
|
|
| Tumor capsular | 0.927 |
|
| 0.400 |
|
|
| Tumor number | 0.738 |
|
| 0.299 |
|
|
| Tumor size (>5
cm) | 0.003 | 3.394
(0.618–18.625) | 0.160 | 0.009a | 1.762
(0.505–6.148) | 0.374 |
| Macrovascular
invasion | 0.002 | 4.528
(1.001–20.470) | 0.050 | 0.043a | 2.765
(0.785–9.741) | 0.113 |
| Microvascular
invasion | 0.193 |
|
| 0.139 |
|
|
| Edmondson-Steiner
grading | 0.485 |
|
| 0.560 |
|
|
| Satellite
lesions | 0.103 |
|
| 0.337 |
|
|
| Lymph node
metastasis | <0.001 | 3.818
(0.918–15.879) | 0.065 | 0.006a | 2.176
(0.587–8.067) | 0.245 |
Recurrence patterns of CHC and
management after recurrence
The recurrence patterns and management of CHC are
summarized in Table III. Tumor
recurrence occurred in 20 of the 26 patients with CHC. There were
13 cases of intrahepatic recurrence, 4 cases of extrahepatic
recurrence, and 3 cases of both intrahepatic and extrahepatic
recurrence. Among the patients with extrahepatic metastases, 3
patients exhibited lymph node metastasis, 2 patients exhibited
peritoneal implantation and 2 patients exhibited bone
metastases.
 | Table III.Recurrence patterns and management of
recurrence after hepatectomy of combined
hepatocellular-cholangiocarcinoma. |
Table III.
Recurrence patterns and management of
recurrence after hepatectomy of combined
hepatocellular-cholangiocarcinoma.
| Variables | Patients, n
(%) |
|---|
| Recurrence | 20 (76.9) |
| Location |
|
|
Intrahepatic | 13 (65.0) |
|
Extrahepatic | 4 (20.0) |
|
Both | 3 (15.0) |
| Extrahepatic
metastases | 7 (27.0) |
| Lymph
node metastasis | 3 (42.8) |
|
Peritoneal implantation | 2 (28.6) |
| Bone
metastases | 2 (28.6) |
| Management |
|
|
Resection | 2 (10.0) |
|
TACE | 8 (40.0) |
|
RFA | 1 (5.0) |
|
Chemotherapy | 3 (15.0) |
|
Radiotherapy | 1 (5.0) |
|
Comprehensive treatment | 5 (25.0) |
| TACE +
chemotherapy + radiotherapy | 1 (5.0) |
| TACE+
radiotherapy + percutaneous | 1 (5.0) |
| ethanol
injection |
|
| TACE +
RFA | 2 (10.0) |
|
Resection + TACE | 1 (5.0) |
However, the management of tumor recurrence
differed. In the present study, intrahepatic recurrence was treated
with resection (n=2), transcatheter arterial chemoembolization
(TACE; n=8), radiofrequency ablation (RFA; n=1) and comprehensive
treatment with TACE + radiotherapy + percutaneous ethanol injection
(n=1) and TACE + RFA (n=1). Extrahepatic relapses were treated with
chemotherapy (n=3) or radiotherapy (n=1). Both intrahepatic and
extrahepatic recurrences were comprehensively treated using TACE +
chemotherapy + radiotherapy (n=1), TACE + RFA (n=1) or resection +
TACE (n=1).
CD90 and EpCAM expression in CHC
tissues
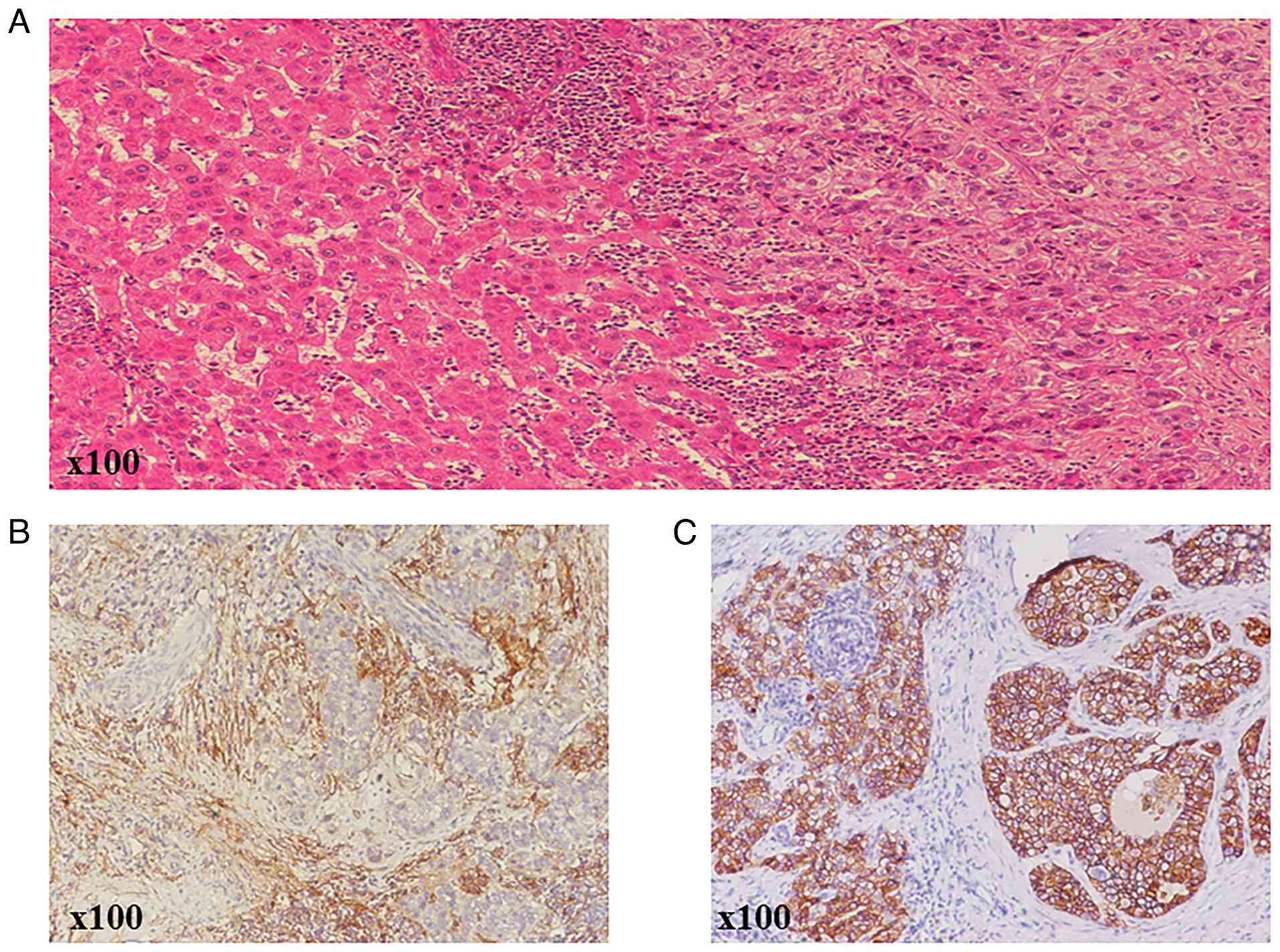
All 26 CHC samples were immunohistochemically
positive for the classic hepatic progenitor cell (HPC) markers CD90
and EpCAM (Fig. 2). In total, 22/26
patients (84.6%) expressed CD90, 21/26 patients (80.8%) expressed
EpCAM and 19/26 patients (73.1%) patients demonstrated simultaneous
co-expression of CD90 and EpCAM (Table
IV).
 | Table IV.Expression levels of CD90 and EpCAM
in 26 patients with combined hepatocellular-cholangiocarcinoma. |
Table IV.
Expression levels of CD90 and EpCAM
in 26 patients with combined hepatocellular-cholangiocarcinoma.
| Variables | n/total n (%) |
|---|
| Positive for
CD90 | 22⁄26 (84.6) |
| Positive for
EpCAM | 21⁄26 (80.8) |
| Positive for both
CD90 and EpCAM | 19⁄26 (73.1) |
Discussion
CHC is an extremely rare type of hepatic malignancy
with clinical characteristics and prognoses different from those of
HCC and ICC. The incidence of CHC varies widely from 2.4 to 14.2%
(2,4). The different rates of CHC may be due
to ambiguous definitions and the exclusion of unresectable cases in
different reports. The incidence of CHC in the present study was
2.7%, which falls within the range found in the aforementioned
studies.
The clinical and pathological features of CHC
compared with those of HCC or ICC vary in different studies. Among
the groups, the age at diagnosis, sex ratio and co-occurring
cirrhosis were similar in certain reports, as were the rates of
viral hepatitis infection (15,16).
Furthermore, patients with CHC had less capsule formation compared
with those with HCC (8,9). However, studies from Asia have
identified that patients with HCC and ICC are younger compared with
patients with CHC and that mainly men are affected. In Chu's and
Lee's series, the majority of patients with CHC were men, with a
high prevalence of HBV infection, suggesting that HBV infection is
a major cause of both HCC and CHC (17,18).
In the present study, the sex ratios were similar across groups;
however, men were the majority in all three groups, and the age and
tumor volume were similar in the CHC group compared with those in
the HCC and ICC groups. Furthermore, the proportion of HBV
infection was lower in CHC compared with that in HCC but similar
between CHC and ICC; HCV infection was similar across groups. The
percentage of patients with CHC with cirrhosis was higher compared
with that of patients with ICC but similar to that of patients with
HCC.
An increase in AFP was identified among the groups.
Several studies have revealed that a high increase in AFP level
(≥400 IU/l) is an independent prognostic factor in CHC (9,16).
However, certain reports demonstrated that the level of AFP in CHC
was lower compared with that in HCC but had no significant
difference (12,19). In the present study, there was no
significant difference in the AFP levels between the CHC group and
the HCC and ICC groups.
Previous research revealed that serum CEA and CA19-9
levels were higher in patients with CHC compared with those in
patients with ICC (19,20). However, the present study pointed
out that the serum CEA level in the CHC group was significantly
higher compared with that in the HCC group but similar to that in
the ICC group, and there was no statistically significant
difference in CA19-9 serum level between CHC and HCC, as well as
between CHC and ICC.
In addition, the immunohistochemical characteristics
of patients with CHC demonstrated intermediate features of HCC and
ICC. Both the ICC-specific markers CK7 and CK19, and the
HCC-specific marker glypcian-3, were elevated in the patients with
CHC. These findings concurred with those reported by Lee et
al (21).
Several previous studies have consistently reported
that the prognosis of CHC is worse compared with that of HCC,
whereas that of ICC varies (8,9,20,22).
The present study results demonstrated that the OS and DFS of
patients with CHC were significantly lower compared with those of
patients with HCC, but not significantly different from those of
patients with ICC. Factors associated with poor clinical prognosis
and tumor recurrence and survival have been reported in several
studies, including high levels of AFP, CEA and CA19-9, multiple
tumors, large tumor size (>5 cm), satellite lesions, vascular
invasion, advanced tumor stage and lymph node metastasis, which may
represent more advanced tumor states (17,20,23,24).
The present study identified that levels of CEA and lymph node
metastases in the CHC group were significantly higher compared with
those in the HCC group but similar to those in the ICC group, which
also suggested that patients with CHC had intermediate clinical
features compared with patients with HCC and ICC.
Few studies have reported on the recurrence of CHC
after surgery. A previous study identified that CHC recurred
continuously in the liver, whereas other studies demonstrated that
extrahepatic recurrence was generally observed in the lymph nodes
(9,25,26).
The present study determined that the most frequent form of
recurrence was intrahepatic metastasis. Currently, there are no
explicit guidelines for the treatment of recurrent CHC. Most of the
20 patients with recurrence in the present study underwent TACE.
However, the outcome after relapse is poor and therefore the
prognosis for CHC after surgery is poor due to the high rate of
recurrence and ineffective treatment after recurrence.
Comprehensive postoperative therapy may be a promising strategy for
patients with CHC in the future.
The origin of CHC remains complex and has been a
subject of debate. The predominant hypothesis is that it is derived
from HPCs, which are intermediate stem cells capable of undergoing
bidirectional differentiation into hepatocytes and bile duct
epithelial cells. HPCs may serve a key role in human CHC
development (27,28).
HPC markers are used to authenticate HPCs. There are
a large number of HPC markers, including the classic markers CD90
and EpCAM (29,30). The present study performed
immunohistochemical analysis of CD90 and EpCAM to examine whether
CHC arose from the HPCs in the present study. All 26 CHC samples
revealed positive results for the classical HPC markers CD90 and
EpCAM. The present study results indicated that CHC highly
expresses HPCs, which is consistent with previous studies
suggesting that CHC may be derived from HPCs (7,31);
therefore, the present study further suggested that the CHC
reported here may originate from the HPCs.
To the best of our knowledge, the present study is
the first systematic and comprehensive analysis of the clinical
pathologies, origins, risk factors for relapse and survival
prognosis, and relapse treatment models of CHC. However, the
present study had certain limitations. First, this was a
single-center, retrospective study and the number of patients with
CHC was relatively small; therefore, further larger multicenter
studies are necessary to validate the present study findings.
Second, it is difficult to diagnose primary biliary cholangitis,
which is considered a risk factor for CHC, and so this was not
included in the present study. Lastly, the present study did not
routinely test for tumor burden score; therefore, the present study
did not evaluate it in the CHC group.
In conclusion, CHC is an extremely rare type of PLC
with a poor outcome and intermediate clinical and pathological
features between HCC and ICC. CHC also has a high expression level
of HPCs, supporting the hypothesis that CHC may originate from HPCs
that have the potential to differentiate into hepatocytes and bile
duct cells. Most studies have revealed that surgical intervention
is a valid treatment for CHC; however, patients with CHC have
markedly worse survival outcomes after hepatic resection compared
with patients with HCC. Further studies on effective treatment
modalities and clinical predictors of CHC are warranted to extend
survival in these patients. Further research is necessary to
confirm the molecular biology of the histogenesis of inclusive
CHC.
Patients with CHC appear to have intermediate
clinical features compared with those with HCC and ICC.
Comprehensive postoperative therapy may be a promising strategy for
patients with CHC. The high expression level of HPC markers in
tumor tissues suggested that CHC may originate from HPCs.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the National
Natural Science Foundation of China (grant no. 81960450) and the
National Major Special Science and Technology Project (grant no.
2017ZX10203207).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
BX and JZho contributed to the conception of the
present study and provided feedback on the report. CY, YQ, JZha and
RS analyzed data. JH and JC performed the data analyses and wrote
the manuscript. All authors have read and approved the final
manuscript. BX and JZho confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
The present study protocol was reviewed and approved
by the Medical Ethics Committee of Guangxi Medical University
Cancer Hospital (Nanning, China; approval no. 6-20-2019).
Preoperatively, all patients provided written informed consent for
data collection and for research purposes.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CHC
|
combined hepatocellular-cholangiocar
cinoma
|
|
HCC
|
hepatocellular carcinoma
|
|
ICC
|
intrahepatic cholangiocarcinoma
|
|
HPC
|
hepatic progenitor cell
|
|
PLC
|
primary liver cancer
|
|
CEA
|
carcinoembryonic antigen
|
|
AFP
|
α-fetoprotein
|
|
CA19-9
|
carbohydrate antigen 19-9
|
|
OS
|
overall survival
|
|
DFS
|
disease-free survival
|
|
PBS
|
phosphate-buffered saline
|
|
HBV
|
hepatitis B virus
|
|
HCV
|
hepatitis C virus
|
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI
|
|
2
|
Stavraka C, Rush H and Ross P: Combined
hepatocellular cholangiocarcinoma (cHCC-CC): An update of genetics,
molecular biology, and therapeutic interventions. J Hepatocell
Carcinoma. 6:11–21. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang J, Li E, Yang H, Wu J, Lu HC, Yi C,
Lei J, Liao W and Wu L: Combined hepatocellular-cholangiocarcinoma:
A population level analysis of incidence and mortality trends.
World J Surg Oncol. 17:432019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brunt E, Aishima S, Clavien PA, Fowler K,
Goodman Z, Gores G, Gouw A, Kagen A, Klimstra D, Komuta M, et al:
cHCC-CCA: Consensus terminology for primary liver carcinomas with
both hepatocytic and cholangiocytic differentation. Hepatology.
68:113–126. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Allen RA and Lisa JR: Combined liver cell
and bile duct carcinoma. Am J Pathol. 25:647–655. 1949.PubMed/NCBI
|
|
6
|
Goodman ZD, Ishak KG, Langloss JM,
Sesterhenn IA and Rabin L: Combined
hepatocellular-cholangiocarcinoma. A histologic and
immunohistochemical study. Cancer. 55:124–135. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yeh MM: Pathology of combined
hepatocellular-cholangiocarcinoma. J Gastroenterol Hepatol.
25:1485–1492. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Koh KC, Lee H, Choi MS, Lee JH, Paik SW,
Yoo BC, Rhee JC, Cho JW, Park CK and Kim HJ: Clinicopathologic
features and prognosis of combined hepatocellular
cholangiocarcinoma. Am J Surg. 189:120–125. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yano Y, Yamamoto J, Kosuge T, Sakamoto Y,
Yamasaki S, Shimada K, Ojima H, Sakamoto M, Takayama T and Makuuchi
M: Combined hepatocellular and cholangiocarcinoma: A
clinicopathologic study of 26 resected cases. Jpn J Clin Oncol.
33:283–287. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu CL, Fan ST, Lo CM, Ng IO, Lam CM, Poon
RT and Wong J: Hepatic resection for combined hepatocellular and
cholangiocarcinoma. Arch Surg. 138:86–90. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cazals-Hatem D, Rebouissou S, Bioulac-Sage
P, Bluteau O, Blanché H, Franco D, Monges G, Belghiti J, Sa Cunha
A, Laurent-Puig P, et al: Clinical and molecular analysis of
combined hepatocellular-cholangiocarcinomas. J Hepatol. 41:292–298.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jarnagin WR, Weber S, Tickoo SK, Koea JB,
Obiekwe S, Fong Y, DeMatteo RP, Blumgart LH and Klimstra D:
Combined hepatocellular and cholangiocarcinoma: Demographic,
clinical, and prognostic factors. Cancer. 94:2040–2046. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kok B and Abraldes JG: Child-Pugh
classification: Time to abandon? Semin Liver Dis. 39:96–103. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhou L, Rui JA, Zhou WX, Wang SB, Chen SG
and Qu Q: Edmondson-steiner grade: A crucial predictor of
recurrence and survival in hepatocellular carcinoma without
microvascular invasio. Pathol Res Pract. 213:824–830. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang AQ, Zheng YC, Du J, Zhu CP, Huang HC,
Wang SS, Wu LC, Wan XS, Zhang HH, Miao RY, et al: Combined
hepatocellular cholangiocarcinoma: Controversies to be addressed.
World J Gastroenterol. 22:4459–4465. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Park H, Choi KH, Choi SB, Choi JW, Kim DY,
Ahn SH, Kim KS, Choi JS, Han KH, Chon CY and Park JY:
Clinicopathological characteristics in combined
hepatocellular-cholangiocarcinoma: A single center study in Korea.
Yonsei Med J. 52:753–760. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chu KJ, Lu CD, Dong H, Fu XH, Zhang HW and
Yao XP: Hepatitis B virus-related combined
hepatocellular-cholangiocarcinoma: Clinicopathological and
prognostic analysis of 390 cases. Eur J Gastroenterol Hepatol.
26:192–199. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lee CH, Hsieh SY, Chang CJ and Lin YJ:
Comparison of clinical characteristics of combined
hepatocellular-cholangiocarcinoma and other primary liver cancers.
J Gastroenterol Hepatol. 28:122–127. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tang D, Nagano H, Nakamura M, Wada H,
Marubashi S, Miyamoto A, Takeda Y, Umeshita K, Dono K and Monden M:
Clinical and pathological features of Allen's type C classification
of resected combined hepatocellular and cholangiocarcinoma: A
comparative study with hepatocellular carcinoma and
cholangiocellular carcinoma. J Gastrointest Surg. 10:987–998. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim KH, Lee SG, Park EH, Hwang S, Ahn CS,
Moon DB, Ha TY, Song GW, Jung DH, Kim KM, et al: Surgical
treatments and prognoses of patients with combined hepatocellular
carcinoma and cholangiocarcinoma. Ann Surg Oncol. 16:623–629. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lee JH, Chung GE, Yu SJ, Hwang SY, Kim JS,
Kim HY, Yoon JH, Lee HS, Yi NJ, Suh KS, et al: Long-term prognosis
of combined hepatocellular and cholangiocarcinoma after curative
resection comparison with hepatocellular carcinoma and
cholangiocarcinoma. J Clin Gastroenterol. 45:69–75. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zuo HQ, Yan LN, Zeng Y, Yang JY, Luo HZ,
Liu JW and Zhou LX: Clinicopathological characteristics of 15
patients with combined hepatocellular carcinoma and
cholangiocarcinoma. Hepatobiliary Pancreat Dis Int. 6:161–165.
2007.PubMed/NCBI
|
|
23
|
Gera S, Ettel M, Acosta-Gonzalez G and Xu
R: Clinical features, histology, and histogenesis of combined
hepatocellular-cholangiocarcinoma. World J Hepatol. 9:300–309.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Song S, Moon HH, Lee S, Kim TS, Shin M,
Kim JM, Park JB, Kwon CH, Kim SJ, Lee SK and Joh JW: Comparison
between resection and transplantation in combined hepatocellular
and cholangiocarcinoma. Transplant Proc. 45:3041–3046. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wakizaka K, Yokoo H, Kamiyama T, Ohira M,
Kato K, Fujii Y, Sugiyama K, Okada N, Ohata T, Nagatsu A, et al:
Clinical and pathological features of combined
hepatocellular-cholangiocarcinoma compared with other liver
cancers. J Gastroenterol Hepatol. 34:1074–1080. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee SD, Park SJ, Han SS, Kim SH, Kim YK,
Lee SA, Ko YH and Hong EK: Clinicopathological features and
prognosis of combined hepatocellular carcinoma and
cholangiocarcinoma after surgery. Hepatobiliary Pancreat Dis Int.
13:594–601. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guo Z: Cancer stem cell markers correlate
with early recurrence and survival in hepatocellular carcinoma.
World Journal of Gastroenterology. 20:2098–2106. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang F, Chen XP, Zhang W, Dong HH, Xiang
S, Zhang WG and Zhang BX: Combined hepatocellular
cholangiocarcinoma originating from hepatic progenitor cells:
Immunohistochemical and double-fluorescence immunostaining
evidence. Histopathology. 52:224–232. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Van Haele M and Roskams T: Hepatic
progenitor cells: An update. Gastroenterol Clin North Am.
46:409–420. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Herrera MB, Bruno S, Buttiglieri S, Tetta
C, Gatti S, Deregibus MC, Bussolati B and Camussi G: Isolation and
characterization of a stem cell population from adult human liver.
Stem Cells. 24:2840–2850. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Theise ND, Yao JL, Harada K, Hytiroglou P,
Portmann B, Thung SN, Tsui W, Ohta H and Nakanuma Y: Hepatic ‘stem
cell’ malignancies in adults: Four cases. Histopathology.
43:263–271. 2003. View Article : Google Scholar : PubMed/NCBI
|