Introduction
Pancreatic ductal adenocarcinoma (PDAC) is an
aggressive malignancy with one of the poorest outcomes among all
cancers, reflected in a 5-year survival rate of only 9% (1,2).
Although early detection allows for curative surgical resection,
approximately 85% of cases are diagnosed at advanced or metastatic
stages due to the disease's subtle onset and rapid progression
(3). Consequently, the development
of novel, preferably noninvasive biomarkers with high sensitivity
and specificity is essential for facilitating early diagnosis,
increasing resectability, and improving survival outcomes.
Carbohydrate antigen 19-9 (CA19-9) remains the most
widely used tumor marker for PDAC. However, its diagnostic
performance is limited, with median sensitivity and specificity of
79 and 82%, respectively (4–9), and
sensitivity dropping below 50% for tumors ≤2 cm (10). Moreover, individuals who lack Lewis
antigens may exhibit false-negative CA19-9 results, further
reducing its clinical utility (10–13).
These limitations underscore the pressing need for complementary or
alternative biomarkers.
The p53 protein is a key tumor suppressor frequently
mutated across many cancer types. Mutant p53 accumulates in tumor
cells, increasing immunogenicity and leading to serum autoantibody
production in a subset of patients. Thus, serum p53 antibody
(s-p53-Ab) positivity may function as an indirect marker of p53
mutation and tumor burden (14).
Our research group have previously demonstrated the diagnostic
relevance of s-p53-Ab in several solid tumors, including esophageal
(15), gastric (16), colorectal (17), hepatocellular (18), and breast cancers (19), and has also explored the value of
multi-autoantibody panels (17,18,20).
Despite ongoing interest, reports on s-p53-Ab in PDAC are limited
to five studies with small cohorts (22–82 cases), showing
positivity rates of 5–28% (21–25).
Importantly, no prior studies have focused solely on resectable
PDAC cases or examined the prognostic implications of s-p53-Ab
status.
Considering these gaps, this study aimed to clarify
the clinicopathological features and prognostic significance of
s-p53-Ab positivity in patients with PDCA.
Materials and methods
Patients
This study was registered as UMIN000014530.
Preoperative serum samples were collected from 124 patients with
PDAC who underwent surgery at Omori Medical Center, Toho University
School of Medicine, between January 2012 and December 2023. The
cohort included 63 men (51%) and 61 women (49%), with a median age
of 72 years (range, 33–87 years). Patients with active coexisting
malignancies-defined as synchronous cancers or metachronous cancers
occurring within 5-year disease-free intervals were excluded. PDAC
staging was determined pathologically according to the eighth
edition of the International Union Against Cancer
tumor-node-metastasis classification system (26). Preoperative assessment, surgical
procedures, and postoperative follow-up adhered to established
clinical practice guidelines for pancreatic cancer. Tumors
presenting with distant metastasis, including peritoneal
dissemination, were classified as unresectable.
Data collection and serum tumor marker
analyses
Clinicopathological data, s-p53-Ab, CA19-9, and CEA
levels were analyzed. Preoperative variables, pathological
findings, postoperative outcomes, and survival data were compiled
in a spreadsheet and transferred to a dedicated database. The
prognostic significance and clinical utility of s-p53-Ab in PDAC
were assessed. Overall survival (OS) and relapse-free survival
(RFS) were defined from the date of surgery to death or October
2025.
The cutoff value for s-p53-Ab was set at 1.3 U/ml
based on the manufacturer's instructions (27). CEA and CA19-9 cutoff values were 5.0
ng/ml and 37 U/ml, respectively, according to the assay kit
guidelines.
Statistical analysis
Statistical analyses were conducted using JMP
version 13 (SAS Institute, Cary, NC, USA). Clinicopathological
variables and recurrence sites were analyzed using Fisher's exact
probability test. OS was estimated using the Kaplan-Meier method,
and survival differences were assessed using the log-rank test.
Significant predictors were identified through univariate and
multivariate analyses using Cox proportional hazard models, with
hazard ratios and 95% confidence intervals calculated. A P-value
<0.05 was considered statistically significant.
Results
Clinicopathological significance of
s-p53-Ab status
No significant differences were observed between the
s-p53-Ab (+) and (−) groups in demographic factors (age and sex),
tumor location, tumor size, lymph-node involvement, histological
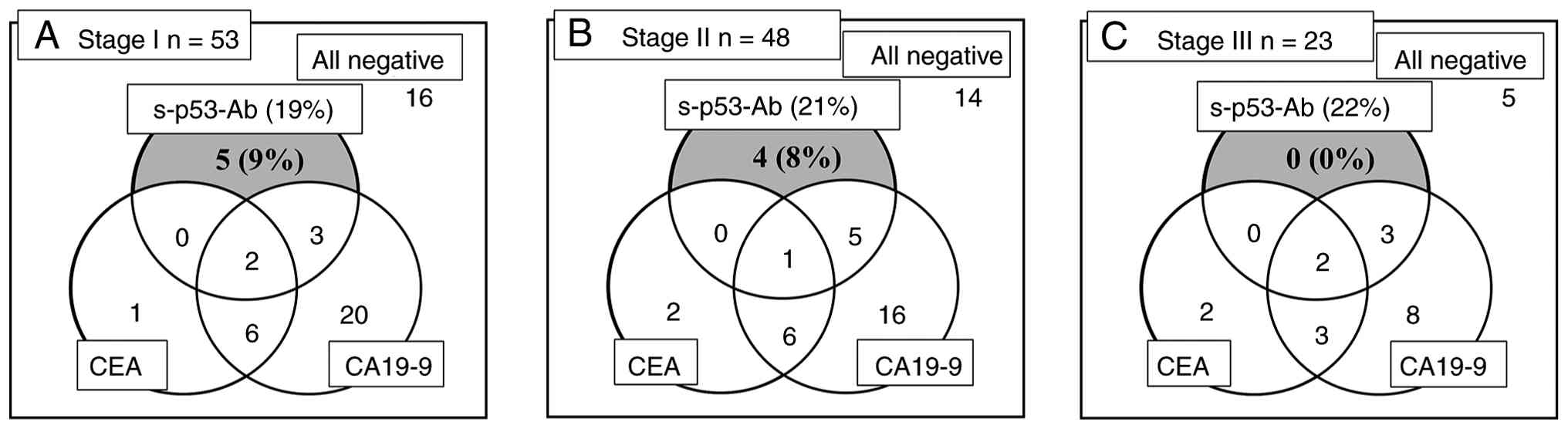
grade, or other pathological features (Table I). Among the 124 patients, 25 (20%)
were s-p53-Ab (+). Seropositivity appeared even in early disease
stages, including 19% (10/53) of stage I and 21% (10/48) of stage
II cases, indicating that elevated s-p53-Ab levels occur even in
the initial phase of PDAC progression. Furthermore, several
patients were solely positive for s-p53-Ab in stages I (n=5) and II
(n=4) (Fig. 1). These findings
suggest that combining s-p53-Ab with other tumor markers improve
diagnostic sensitivity.
 | Table I.Comparison of clinicopathological
characteristics between the s-p53-Ab(+) group and the s-p53-Ab(−)
group in patients with pancreatic cancer. |
Table I.
Comparison of clinicopathological
characteristics between the s-p53-Ab(+) group and the s-p53-Ab(−)
group in patients with pancreatic cancer.
| Variables | No. of patients
(n=124) | s-p53-Ab(+), n (%)
(n=25) | s-p53-Ab(−), n (%)
(n=99) | P-valuea |
|---|
| Sex |
|
|
| 0.302 |
| Male | 63 | 15 (60) | 48 (48) |
|
|
Female | 61 | 10 (40) | 51 (52) |
|
| Age, years |
|
|
| 0.970 |
|
<70 | 50 | 10 (40) | 40 (40) |
|
| ≥70 | 74 | 15 (60) | 59 (60) |
|
| Tumor location |
|
|
| 0.374 |
| Ph | 79 | 14 (56) | 65 (66) |
|
| Pb-t | 45 | 11 (44) | 34 (34) |
|
| CRP, mg/dl |
|
|
| 0.342 |
| ≤0.2 | 89 | 16 (64) | 73 (74) |
|
|
>0.2 | 35 | 9 (36) | 26 (26) |
|
| Albumin, g/dl |
|
|
| 0.421 |
|
>3.5 | 97 | 21 (84) | 76 (77) |
|
| ≤3.5 | 27 | 4 (16) | 23 (23) |
|
| Tumor size, mm |
|
|
| 0.599 |
|
<25 | 44 | 10 (40) | 34 (34) |
|
| ≥25 | 80 | 15 (60) | 65 (66) |
|
| Tumor
classification |
|
|
| 0.552 |
| Well,
moderate | 113 | 22 (88) | 91 (92) |
|
| Poor | 11 | 3 (12) | 8 (8) |
|
| Lymphovascular
invasion |
|
|
| 0.982 |
|
Negative | 25 | 5 (20) | 20 (20) |
|
|
Positive | 99 | 20 (80) | 79 (80) |
|
| Perineural
invasion |
|
|
| 0.623 |
|
Negative | 60 | 11 (44) | 49 (49) |
|
|
Positive | 64 | 14 (56) | 50 (51) |
|
| Tumor depth |
|
|
| 0.598 |
| T1 | 25 | 6 (24) | 19 (19) |
|
| T2, 3,
4 | 99 | 19 (76) | 80 (81) |
|
| Nodal status |
|
|
| 0.755 |
|
Negative | 58 | 11 (44) | 47 (47) |
|
|
Positive | 66 | 14 (56) | 52 (53) |
|
| CEA, ng/ml |
|
|
| 0.982 |
|
≤5.0 | 99 | 20 (80) | 79 (80) |
|
|
>5.0 | 25 | 5 (20) | 20 (20) |
|
| CA19-9, U/ml |
|
|
| 0.686 |
|
≤37 | 49 | 9 (36) | 40 (40) |
|
|
>37 | 75 | 16 (64) | 59 (60) |
|
| Neoadjuvant
chemotherapy |
|
|
| 0.556 |
|
Yes | 8 | 1 (4) | 7 (7) |
|
| No | 116 | 24 (96) | 92 (93) |
|
Prognostic significance of s-p53-Ab
status
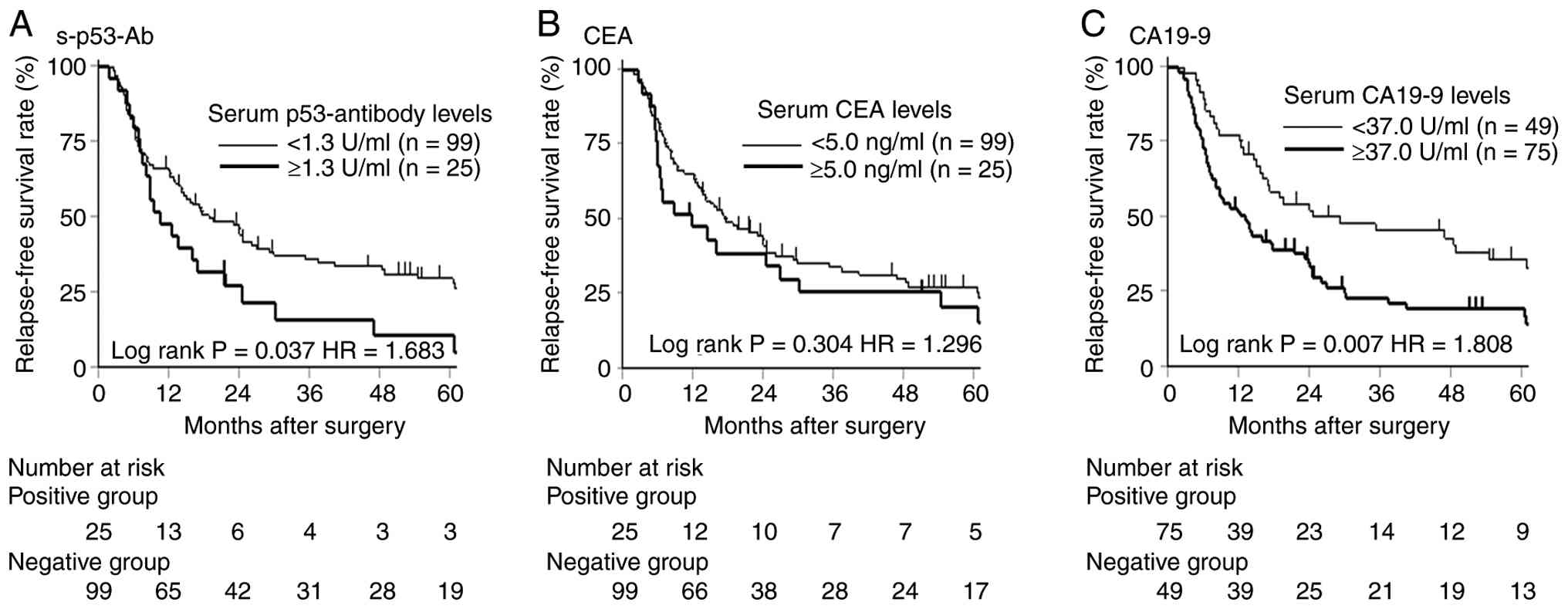
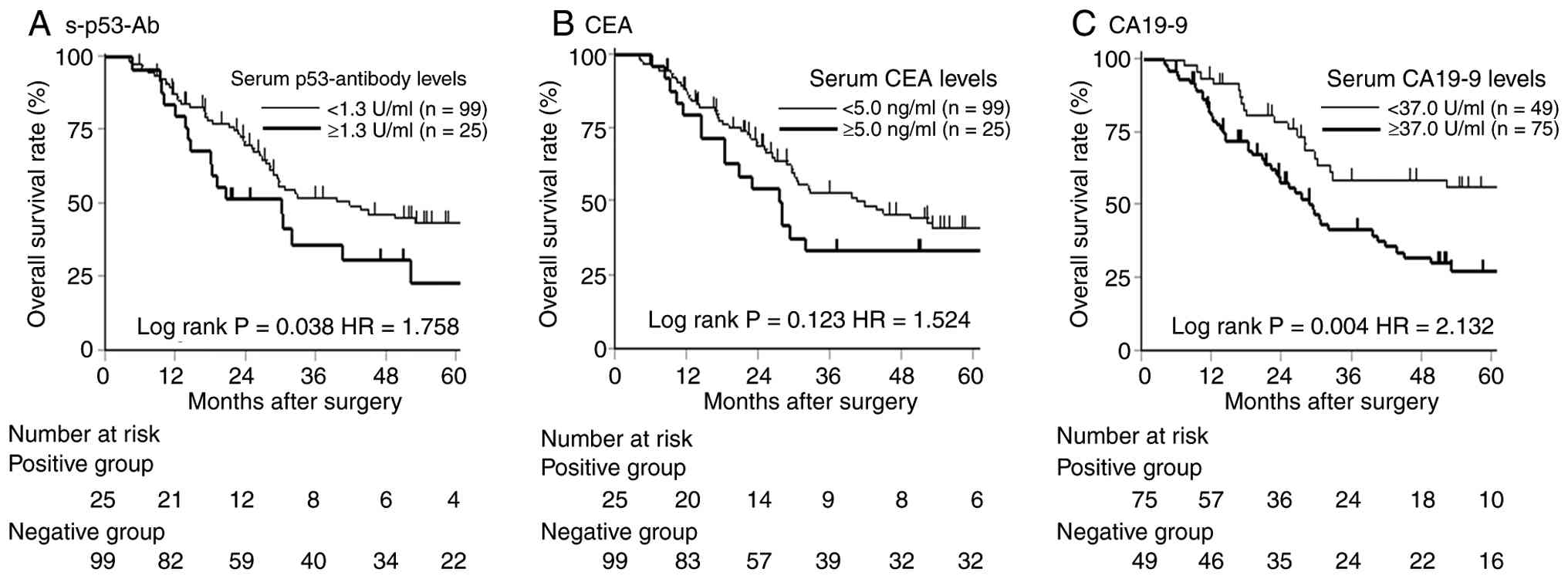
Kaplan-Meier survival analysis showed that patients
with s-p53-Ab (+) had significantly worse RFS (P=0.037) and OS
(P=0.038) than those who were s-p53-Ab (−) (Figs. 2 and 3). Among CA19-9 (+) patients, those who
were also s-p53-Ab (+) showed significantly poorer RFS than CA19-9
(+)/s-p53-Ab (−) patients (P=0.032). Multivariate Cox analysis
further identified s-p53-Ab (+) as an independent prognostic factor
for reduced RFS [hazard ratio (HR)=1.683, 95% confidence interval
(CI): 1.102–2.693, P=0.047] and OS (HR=1.758, 95% CI: 1.036–2.915,
P=0.048) (Tables II and III).
 | Table II.Univariate and multivariate analysis
of risk factors for relapse-free survival in patients with
pancreatic cancer. |
Table II.
Univariate and multivariate analysis
of risk factors for relapse-free survival in patients with
pancreatic cancer.
|
|
|
| Multivariate
analysis |
|---|
|
|
|
|
|
|---|
| Variables | No. of patients
(n=124) | Univariate analysis
P-valuea | HRb | 95% CIc |
P-valued |
|---|
| Sex |
|
|
|
|
|
|
Male | 63 | 0.908 |
|
|
|
|
Female | 61 |
|
|
|
|
| Age, years |
|
|
|
|
|
|
<70 | 50 | 0.410 |
|
|
|
|
≥70 | 74 |
|
|
|
|
| Tumor location |
|
|
|
|
|
| Ph | 79 | 0.627 |
|
|
|
|
Pb-t | 45 |
|
|
|
|
| CRP (mg/dl) |
|
|
|
|
|
|
≤0.2 | 89 | 0.003 | 1 |
| 0.107 |
|
>0.2 | 35 |
| 1.468 | 0.918-2.307 |
|
| Albumin (g/dl) |
|
|
|
|
|
|
>3.5 | 97 | 0.809 |
|
|
|
|
≤3.5 | 27 |
|
|
|
|
| Tumor
classification |
|
|
|
|
|
| Well,
moderate | 113 | 0.982 |
|
|
|
|
Poor | 11 |
|
|
|
|
| Lymphovascular
invasion |
|
|
|
|
|
|
Negative | 25 | 0.079 |
|
|
|
|
Positive | 99 |
|
|
|
|
| Perineural
invasion |
|
|
|
|
|
|
Negative | 60 | 0.124 |
|
|
|
|
Positive | 64 |
|
|
|
|
| Tumor depth |
|
|
|
|
|
| T1 | 25 | 0.459 |
|
|
|
| T2, 3,
4 | 99 |
|
|
|
|
| Nodal status |
|
|
|
|
|
|
Negative | 58 | 0.002 | 1 |
| 0.087 |
|
Positive | 66 |
| 1.481 | 0.944–2.348 |
|
| s-p53-Ab, U/ml |
|
|
|
|
|
|
≤1.3 | 99 | 0.037 | 1 |
| 0.047 |
|
>1.3 | 25 |
| 1.683 | 1.102–2.693 |
|
| CEA, ng/ml |
|
|
|
|
|
|
≤5.0 | 99 | 0.303 |
|
|
|
|
>5.0 | 25 |
|
|
|
|
| CA19-9, U/ml |
|
|
|
|
|
|
≤37 | 49 | 0.006 | 1 |
| 0.042 |
|
>37 | 75 |
| 1.598 | 1.106–2.447 |
|
| Neoadjuvant
chemotherapy |
|
|
|
|
|
|
Yes | 8 | 0.172 |
|
|
|
| No | 116 |
|
|
|
|
| Resection
margin |
|
|
|
|
|
| R0 | 96 | <0.001 | 1 |
| 0.011 |
| R1 | 28 |
| 1.972 | 1.175–3.234 |
|
 | Table III.Univariate and multivariate analysis
of risk factors for overall survival in patients with pancreatic
cancer. |
Table III.
Univariate and multivariate analysis
of risk factors for overall survival in patients with pancreatic
cancer.
|
|
|
| Multivariate
analysis |
|---|
|
|
|
|
|
|---|
| Variables | No. of patients
(n=124) | Univariate analysis
P-valuea | HRb | 95% CIc |
P-valued |
|---|
| Sex |
|
|
|
|
|
|
Male | 63 | 0.915 |
|
|
|
|
Female | 61 |
|
|
|
|
| Age, years |
|
|
|
|
|
|
<70 | 50 | 0.885 |
|
|
|
|
≥70 | 74 |
|
|
|
|
| Tumor location |
|
|
|
|
|
| Ph | 79 | 0.763 |
|
|
|
|
Pb-t | 45 |
|
|
|
|
| CRP, mg/dl |
|
|
|
|
|
|
≤0.2 | 89 | 0.003 | 1 |
| 0.050 |
|
>0.2 | 35 |
| 1.700 | 0.999–2.836 |
|
| Albumin, g/dl |
|
|
|
|
|
|
>3.5 | 97 | 0.911 |
|
|
|
|
≤3.5 | 27 |
|
|
|
|
| Tumor
classification |
|
|
|
|
|
| Well,
moderate | 113 | 0.322 |
|
|
|
|
Poor | 11 |
|
|
|
|
| Lymphovascular
invasion |
|
|
|
|
|
|
Negative | 25 | 0.241 |
|
|
|
|
Positive | 99 |
|
|
|
|
| Perineural
invasion |
|
|
|
|
|
|
Negative | 60 | 0.452 |
|
|
|
|
Positive | 64 |
|
|
|
|
| Tumor depth |
|
|
|
|
|
| T1 | 25 | 0.676 |
|
|
|
| T2, 3,
4 | 99 |
|
|
|
|
| Nodal status |
|
|
|
|
|
|
Negative | 58 | 0.003 | 1 |
| 0.097 |
|
Positive | 66 |
| 1.566 | 0.921–2.711 |
|
| s-p53-Ab, U/ml |
|
|
|
|
|
|
≤1.3 | 99 | 0.038 | 1 |
| 0.048 |
|
>1.3 | 25 |
| 1.758 | 1.036–2.915 |
|
| CEA, ng/ml |
|
|
|
|
|
|
≤5.0 | 99 | 0.123 |
|
|
|
|
>5.0 | 25 |
|
|
|
|
| CA19-9, U/ml |
|
|
|
|
|
|
≤37 | 49 | 0.004 | 1 |
| 0.028 |
|
>37 | 75 |
| 1.808 | 1.066–3.157 |
|
| Neoadjuvant
chemotherapy |
|
|
|
|
|
|
Yes | 8 | 0.086 |
|
|
|
| No | 116 |
|
|
|
|
| Resection
margin |
|
|
|
|
|
| R0 | 96 | 0.002 | 1 |
| 0.126 |
| R1 | 28 |
| 1.579 | 0.874–2.764 |
|
Comparison of recurrence sites between
s-p53-Ab positive and negative group
The s-p53-Ab (+) group demonstrated a significantly
higher rate of lymph-node recurrence (P=0.013) and peritoneal
dissemination (P=0.045) than the s-p53-Ab (−) group (Table IV).
 | Table IV.Comparison of recurrence sites
between the s-p53-Ab(+) group and the s-p53-Ab(−) group (n=91). |
Table IV.
Comparison of recurrence sites
between the s-p53-Ab(+) group and the s-p53-Ab(−) group (n=91).
| Variables | Recurrent patients,
n (%) (n=91) | s-p53-Ab(+) group,
n (%) (n=22) | s-p53-Ab(−) group,
n (%) (n=69) |
P-valuea |
|---|
| Multiple organ
recurrence | 20 (22) | 4 (18) | 16 (23) | 0.984 |
| Initial recurrence
site |
|
|
|
|
|
Liver | 28 (31) | 4 (18) | 24 (35) | 0.364 |
| Lymph
nodes | 26 (29) | 10 (45) | 16 (23) | 0.013 |
|
Local | 20 (22) | 1 (5) | 19 (28) | 0.037 |
|
Peritoneum | 18 (20) | 7 (32) | 11 (16) | 0.045 |
|
Lung | 12 (13) | 3 (14) | 9 (13) | 0.667 |
| Recurrence within 1
year | 49 (54) | 13 (59) | 36 (52) | 0.156 |
Discussion
This study examined the clinicopathological and
prognostic significance of s-p53-Ab (+) in 124 patients with PDAC
and demonstrated that 20% were s-p53-Ab (+), a status significantly
associated with poorer RFS and OS.
No clear relationship was observed between s-p53-Ab
positivity and clinicopathological variables, consistent with
findings in other malignancies (14–18).
Notably, the positivity rate in stage I (19%) and stage II (21%)
patients were comparable to the overall cohort, aligning with
previous reports in gastric (28),
esophageal (29), colorectal
(30), and hepatocellular cancers
(18). Although s-p53-Ab alone was
positive in 10% of stage I and 8% of stage II cases, CEA positivity
was far lower (2 and 4%, respectively). These results suggest that
s-p53-Ab is a supplementary diagnostic marker, particularly for
early-stage PDAC, and we propose as a high-specificity
‘confirmatory’ adjunct to CA19-9, given the low sensitivity.
Regarding the prognostic significance of s-p53-Ab
(+), it was identified as an independent poor prognostic factor for
both RFS and OS in PDAC, suggesting that s-p53-Ab reflects the
tumor's biological aggressiveness. The HRs for RFS and OS were
comparable, with the HR for OS approximately 1.13 times that for
RFS-similar to ratios observed with CA19-9 (×1.18) and CEA (×1.17).
This suggests that patients positive for s-p53-Ab have a resistance
to post-recurrence treatments comparable to those positive for
CA19-9 or CEA.
Although preoperative s-p53-Ab (+) was not
associated with lymph-node metastasis at surgery, it was
significantly associated with postoperative lymph-node recurrence.
Among patients with pathologically confirmed lymph-node metastasis,
those with preoperative s-p53-Ab (+) had a markedly higher rate of
lymph-node recurrence than s-p53-Ab (−) patients (8/14 vs. 8/52,
P=0.028). This implies that elevated s-p53-Ab levels indicate early
dissemination of cancer cells to extra-regional lymph nodes,
possibly explaining the poorer RFS observed in s-p53-Ab (+)
patients. s-p53-Ab might be a more sensitive indicator of occult
micro-metastasis than current pathological staging.
Despite these promising results, several limitations
must be acknowledged. First, the retrospective nature and
single-institution design may introduce selection and information
biases. Second, the unavailability of immunohistochemical analyses
of tumor tissues by costs. Serum autoantibodies and expression in
tumor tissue may be correlated (14). Since micro heterogeneity of p53
expression may affect induction of autoantibodies, further precise
immunohistochemical analysis should be performed. Third, the
biological mechanisms linking s-p53-Ab positivity to aggressive
tumor behavior remain unclear; it is unknown whether s-p53-Ab
simply reflects tumor immunogenicity or actively influences tumor
progression or the host immune response. Further molecular and
translational studies are needed to clarify this relationship.
Forth, although a standardized cutoff value for s-p53-Ab (1.3 U/ml)
was applied, the optimal threshold may vary depending on population
characteristics, assay sensitivity, and clinical application
(diagnosis vs. prognosis). Therefore, external validation in
independent, multi-institutional cohorts is essential before
routine clinical use. Future prospective research should confirm
these findings and assess the value of incorporating s-p53-Ab into
multi-marker panels or risk models to enhance pancreatic cancer
management.
In conclusion, this study shows that serum s-p53-Ab
is present in a subset of patients with resectable PDAC and that
its positivity independently predicts poorer prognosis. s-p53-Ab
may serve as a valuable adjunct biomarker for early detection and
outcome prediction in PDAC.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
RO conceived the study. RO designed the study. YO,
YK, YM and YI acquired data. RO and HS confirm the authenticity of
all the raw data. RO and YO were involved in quality control of
data and algorithms. RO, YO and HS analyzed and interpreted data.
RO performed statistical analysis. RO prepared the manuscript. RO
and HS edited the manuscript. All authors reviewed the manuscript.
All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
All study participants provided consent for future
analyses of their blood samples for research. The protocol for the
present study was approved by the Ethics Committee of the Toho
University (approval nos. T2024-2081, M22211, M21038_20197_19213
and A18103_A17052_A16035_A16001_26095_25024_24038_22047_22112;
Ota-ku, Tokyo, Japan). Patients provided written informed consent
before enrolment. The study was registered in the UMIN Clinical
Trials Registry (UMIN000014530) and was conducted in accordance
with The Declaration of Helsinki and the Japanese Ethical
Guidelines for Clinical Research.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33.
2021.PubMed/NCBI
|
|
2
|
McGuigan A, Kelly P, Turkington RC, Jones
C, Coleman HG and McCain RS: Pancreatic cancer: A review of
clinical diagnosis, epidemiology, treatment and outcomes. World J
Gastroenterol. 24:4846–4861. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chari ST, Kelly K, Hollingsworth MA,
Thayer SP, Ahlquist DA, Andersen DK, Batra SK, Brentnall TA, Canto
M, Cleeter DF, et al: Early detection of sporadic pancreatic
cancer: summative review. Pancreas. 44:693–712. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ballehaninna UK and Chamberlain RS: The
clinical utility of serum CA19-9 in the diagnosis, prognosis and
management of pancreatic adenocarcinoma. An evidence based
appraisal. J Gastrointest Oncol. 3:105–119. 2012.PubMed/NCBI
|
|
5
|
Brentnall TA, Bronner MP, Byrd DR, Haggitt
RC and Kimmey MB: Early diagnosis and treatment of pancreatic
dysplasia in patients with a family history of pancreatic cancer.
Ann Intern Med. 131:247–255. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bünger S, Laubert T, Roblick UJ and
Habermann JK: Serum biomarkers for improved diagnostic of
pancreatic cancer: A current overview. J Cancer Res Clin Oncol.
137:375–389. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ritts RE and Pitt HA: CA 19-9 in
pancreatic cancer. Surg Oncol Clin N Am. 7:93–101. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Goonetilleke KS and Siriwardena AK:
Systematic review of carbohydrate antigen (CA 19-9) as a
Biochemical marker in the diagnosis of pancreatic cancer. Eur J
Surg Oncol. 33:266–270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Luo G, Jin K, Deng S, Cheng H, Fan Z, Gong
Y, Qian Y, Huang Q, Ni Q, Liu C, et al: Roles of CA19-9 in
Pancreatic cancer: Biomarker, predictor and promoter. Biochim
Biophys Acta Rev Cancer. 1875:1884092021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Egawa S, Takeda K, Fukuyama S, Motoi F,
Sunamura M and Matsuno S: Clinicopathological aspects of small
pancreatic cancer. Pancreas. 28:235–240. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Safi F, Schlosser W, Falkenreck S and
Beger HG: CA 19-9 serum course and prognosis of pancreatic cancer.
Int J Pancreatol. 20:155–161. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Goggins M: Identifying molecular markers
for the early detection of pancreatic neoplasia. Semin Oncol.
34:303–310. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim JE, Lee KT, Lee JK, Paik SW, Rhee JC
and Choi KW: Clinical usefulness of carbohydrate antigen 19-9 as a
screening test for pancreatic cancer in an asymptomatic population.
J Gastroenterol Hepatol. 19:182–186. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Okada R, Shimada H, Otsuka Y, Tsuchiya M,
Ishii J, Katagiri T, Maeda T, Kubota Y, Nemoto T and Kaneko H:
Serum p53 antibody as a potential tumor marker in extrahepatic
cholangiocarcinoma. Surg Today. 47:1492–1499. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shimada H: p53 Molecular approach to
diagnosis and treatment of esophageal squamous cell carcinoma. Ann
Gastroenterol Surg. 2:266–273. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nanami T, Hoshino I, Ito M, Yajima S,
Oshima Y, Suzuki T, Shiratori F, Nabeya Y, Funahashi K and Shimada
H: Prevalence of autoantibodies against Ras-Like GTPases, RalA, in
patients with gastric cancer. Mol Clin Oncol. 13:282020.PubMed/NCBI
|
|
17
|
Ushigome M, Nabeya Y, Soda H, Takiguchi N,
Kuwajima A, Tagawa M, Matsushita K, Koike J, Funahashi K and
Shimada H: Multi-panel assay of serum autoantibodies in colorectal
cancer. Int J Clin Oncol. 23:917–923. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Okada R, Otsuka Y, Wakabayashi T, Shinoda
M, Aoki T, Murakami M, Arizumi S, Yamamoto M, Aramaki O, Takayama
T, et al: Six autoantibodies as potential serum biomarkers of
hepatocellular carcinoma: A prospective multicenter study. Int J
Cancer. 147:2578–2586. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kubota Y, Ogata H, Otsuka S, Kuwajima A,
Saito F and Shimada H: Presence of Autoantibodies against Ras-Like
GTPases in Serum in Stage I/II Breast Cancer. Toho J Med.
3:125–130. 2017.
|
|
20
|
Hoshino I, Nagata M, Takiguchi N, Nabeya
Y, Ikeda A, Yokoi S, Kuwajima A, Tagawa M, Matsushita K, Satoshi Y
and Hideaki S: Panel of autoantibodies against multiple
Tumor-associated antigens for detecting gastric cancer. Cancer Sci.
108:308–315. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Laurent-Puig P, Lubin R, Semhoun-Ducloux
S, Pelletier G, Fourre C, Ducreux M, Briantais MJ, Buffet C and
Soussi T: Antibodies against p53 protein in serum of patients with
benign or malignant pancreatic and biliary diseases. Gut.
36:455–458. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Müller M, Meyer M, Schilling T, Ulsperger
E, Lehnert T, Zentgraf H, Stremmel W, Volkmann M and Galle PR:
Testing for Anti-p53 antibodies increases the diagnostic
sensitivity of conventional tumor markers. Int J Oncol. 29:973–980.
2006.PubMed/NCBI
|
|
23
|
Li J, Wang LJ, Ying X, Han SX, Bai E,
Zhang Y and Zhu Q: Immunodiagnostic value of combined detection of
autoantibodies to Tumor-associated antigens as biomarkers in
pancreatic cancer. Scand J Immunol. 75:342–349. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Okada T, Noji S, Goto Y, Iwata T, Fujita
T, Okada T, Matsuzaki Y, Kuwana M, Hirakata M, Horii A, et al:
Immune responses to DNA mismatch repair enzymes hMSH2 and hPMS1 in
patients with pancreatic cancer, dermatomyositis and polymyositis.
Int J Cancer. 116:925–933. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ohshio G, Suwa H and Imamura M: Clinical
implication of Anti-p53 antibodies and p53-protein in pancreatic
disease. Int J Gastrointest Cancer. 31:129–135. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Brierley JD, Gospodarowicz MK and
Wittekind C: International Union Against Cancer (UICC): TNM
Classification of Malignant Tumors (Wiley-Blackwell, 2017).
|
|
27
|
Shimada H, Ochiai T and Nomura F; Japan
p53 antibody research group, : Titration of serum p53 antibodies in
1085 patients with various types of malignant tumors. Cancer.
97:682–689. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Oshima Y, Suzuki T, Yajima S, Nanami T,
Shiratori F, Funahashi K and Shimada H: Serum p53 antibody: Useful
for detecting gastric cancer but not for predicting prognosis after
surgery. Surg Today. 50:1402–1408. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Suzuki T, Yajima S, Ishioka N, Nanami T,
Oshima Y, Washizawa N, Funahashi K, Otsuka S, Nemoto T and Shimada
H: Prognostic significance of high serum p53 antibody titers in
patients with esophageal squamous cell carcinoma. Esophagus.
15:294–300. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yajima S, Suzuki T, Oshima Y, Shiratori F,
Funahashi K, Kawai S, Nanki T, Muraoka S, Urita Y, Saida Y, et al:
New assay system elecsys Anti-p53 to detect serum Anti-p53
antibodies in esophageal cancer patients and colorectal cancer
patients: Multi-institutional study. Ann Surg Oncol. 28:4007–4015.
2021. View Article : Google Scholar : PubMed/NCBI
|