Introduction
According to data from 2024, lung cancer remains the
most commonly diagnosed type of cancer worldwide (1). Despite recent advancements in surgical
techniques, and the application of targeted therapies and
immunotherapies, the overall survival rate for patients with lung
cancer remains low, with the age-standardized 5-year overall
survival rate estimated at 10–20% (2). Notably, immune escape, metastasis and
recurrence are factors that collectively represent major
therapeutic hurdles for patients with lung cancer (3). Therefore, identifying key driver genes
of immune evasion that can be targeted, elucidating their oncogenic
mechanisms and developing targeted combination strategies are
urgently needed to overcome therapeutic bottlenecks and prolong
patient survival.
The biological characteristics of lung cancer; for
example, proliferation, anti-apoptosis, invasion, metastasis, drug
resistance and immune evasion, are essentially the result of
dysregulated gene expression programs, with transcription factors
(TFs) serving as the primary switches for these programs (4). T-box TFs (TBXs), a family of proteins
containing T-box DNA-binding domains, are widely distributed across
the animal kingdom. TBXs serve crucial roles in embryonic
development, cellular differentiation, organ formation, and various
physiological and pathological processes. Numerous studies have
demonstrated their pivotal roles in regulating
epithelial-mesenchymal transition (EMT), maintaining tumor stem
cell properties, inducing apoptosis, promoting tumor cell
proliferation and enhancing tumor drug resistance (5–9).
Growing evidence has indicated that the T-box family serves as a
promising biomarker, with pivotal roles in cancer diagnosis,
identification of therapeutic targets and prognosis (10). As a key member of the T-box family,
TBX20 has garnered attention because of its roles in various
physiological and pathological processes. In cardiomyocytes, TBX20
has been shown to activate the bone morphogenetic protein 2
(BMP2)/phosphorylated (p)-Smad1/5/8 and PI3K/AKT/GSK3β/β-catenin
signaling pathways, promoting cell proliferation and survival
(11). In colorectal cancer, TBX20
directly binds to Ku70/Ku80 through its intermediate domain,
blocking the formation of heterodimers between the two proteins.
This inhibits non-homologous end joining-mediated double-strand
break repair, leading to cumulative DNA damage and genomic
instability, thereby suppressing cell proliferation and enhancing
sensitivity to radiotherapy/daunorubicin (12). TBX20 is clearly not merely a
developmental regulator but also a key TF with the potential to
modulate cancer progression. Notably, the role of TBX20 in heart
and lung development has been extensively studied (13,14),
but the specific mechanism of TBX20 in lung cancer remains unclear.
To elucidate the tumorigenic role of TBX20 in lung cancer, in
vitro and in vivo experiments were performed to
elucidate how TBX20 promotes cell proliferation and immune evasion.
The value of TBX20 as a prognostic marker and therapeutic target
was also evaluated.
Materials and methods
Cell culture and transfection
The human normal lung epithelial cell line BEAS-2B,
and the human non-small cell lung cancer cell lines A549 and
HCI-H1975 used in this study were all purchased from The Cell Bank
of Type Culture Collection of The Chinese Academy of Sciences. The
cells were cultured in DMEM (Wuhan Servicebio Technology Co., Ltd.)
supplemented with 10% fetal bovine serum (FBS; Nanjing BioChannel
Biotechnology Co., Ltd.) and 1% penicillin-streptomycin (Wuhan
Servicebio Technology Co., Ltd.), at 5% CO2 and 37°C.
Small interfering (si)RNA-TBX20 (si-TBX20) and siRNA-negative
control (NC) were transfected into A549 cells using
Lipofectamine® 3000 transfection reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). TBX20 overexpression plasmid
(oe-TBX20) was also transfected into A549 cells using
pRP[Exp]-EGFP/Puro-CAG>hTBX20 [Yunzhou Biosciences (Guangzhou)
Co., Ltd.); at the same time, the empty vector
pRP[Exp]-EGFP/Puro-CAG (without the TBX20 insertion fragment) was
used as the negative control. A549 cells were added to six-well
plates, with 2×106 cells per well, and 2 µl DMEM medium
was added. A total of 5 µg of DNA or siRNA, was added at 25°C for 6
h. Culture medium was removed, and 10% serum-containing DMEM medium
was added, and the cells were further cultured at 37°C for 24 h.
Finally, the transfection efficiency was observed under a
fluorescence microscope. The sequences of siRNA-TBX20, siRNA-NC and
oe-TBX20 were designed and synthesized by Sangon Biotech (Shanghai)
Co., Ltd. The siRNA sequences used are as follows: siRNA-TBX20,
forward 5′-CCGAGAUGAUCAUCACCAAGU-3′, reverse
5′-UUGGUGAUGAUCAUCUCGGUG-3′; siRNA-NC, forward
5′-UUCUCCGAACGUGUCACGUUU-3′ and reverse
5′-ACGUGACACGUUCGGAGAAUU-3′.
Western blotting
Total cellular protein was extracted from A549 and
HCI-H1975 using a protein extraction kit (cat. no. BC3710; Beijing
Solarbio Science & Technology Co., Ltd.) according to the
manufacturer's protocol. After determining the protein
concentration using BCA Protein Quantification kit, loading buffer
was added and the samples were incubated at 95°C for denaturation.
SDS-PAGE was performed on 12% gels with 30 µg protein/lane.
Proteins were transferred to a PVDF membrane, which was blocked
with 5% skim milk solution for 2 h at 25°C. The PVDF membrane was
incubated with diluted primary antibodies at 4°C for 12 h, washed
and incubated with diluted secondary antibodies on ice in the dark
for 2 h. An ECL reagent (cat. no. MA0186; Dalian Meilun Biology
Technology Co., Ltd.) was used for visualization and a gel imaging
system (SH-523; Hangzhou Shenhua Technology Co., Ltd.) was used for
development. ImageJ (National Institutes of Health, V1.8.0.112) was
used to calculate the gray values of proteins. The following
antibodies were used in the present study: TBX20 (cat. no.
83414-5-RR; 1:1,000; Wuhan Sanying Biotechnology), E-cadherin (cat.
no. 60335-1-Ig; 1:1,000; Wuhan Sanying Biotechnology), N-cadherin
(cat. no. 66219-1-Ig; 1:2,000; Wuhan Sanying Biotechnology),
vimentin (cat. no. A19607; 1:1,000; ABclonal Biotech Co., Ltd.),
CD44 (cat. no. 84854-5-RR; 1:500; Wuhan Sanying Biotechnology),
Sox2 (cat. no. 66411-1-Ig; 1:500; Wuhan Sanying Biotechnology),
Nanog (cat. no. 14295-1-AP; 1:500; Wuhan Sanying Biotechnology),
PD-L1 (cat. no. 66248-1-Ig; 1:100; Wuhan Sanying Biotechnology),
IL-6 (cat. no. A0286; 1:200; ABclonal Biotech Co., Ltd.), CCL2
(cat. no. MA5-17040; 1:500; Thermo Fisher Scientific, Inc.) and
β-actin (cat. no. 81115-1-RR; 1:2,000; Wuhan Sanying Biotechnology)
primary antibodies, and HRP-conjugated rabbit anti-mouse and goat
anti-rabbit secondary antibodies (cat. nos. ab6728 and ab6721;
1:5,000; Abcam).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from A549 and HCI-H1975
cells using a TRIzol kit (Invitrogen), and the RNA concentration
and purity were measured with a NanoDrop microplate
spectrophotometer (Thermo Fisher Scientific, Inc.). cDNA was
subsequently synthesized by RT using a PrimeScript RT-PCR kit
(Takara Bio, Inc.), performed according to manufacturer's protocol.
Finally, qPCR was performed on cDNA using the eQ9600 real-time
fluorescent qPCR detection system (Suzhou Dongsheng Xingye
Scientific Instruments Co., Ltd.), Thermocycling conditions were as
follows: Pre-denaturation at 95°C for 3 min; Denaturation at 95°C
for 5 sec, annealing at 60°C for 30 sec, extension at 72°C for 20
sec, a total of 40 cycles. The mRNA expression levels of TBX20,
vimentin, N-cadherin, E-cadherin, CD44, Sox2, Nanog, PD-L1, IL-6
and CCL2 were analyzed using the 2−ΔΔCq method (15), with β-actin as the internal
reference gene. The primer sequences are shown in Table I.
 | Table I.Primer sequences. |
Table I.
Primer sequences.
| Gene | Primer sequence,
5′-3′ |
|---|
| β-actin | F:
GATTCCTATGTGGGCGACGA |
|
| R:
AGGTCTCAAACATGATCTGGGT |
| TBX20 | F:
CAACCCCAAATCGAGGGTCA |
|
| R:
CCGATGGTGTCAGAGGCATT |
| N-cadherin | F:
CCTGCTTTCATTCTGACATACCT |
|
| R:
GCTTCTCACGGCATACACCA |
| E-cadherin | F:
TGGTACCTGGCAAGATGCAG |
|
| R:
GGGGGCTTCATTCACATCCA |
| Vimentin | F:
CAGATGCGTGAAATGGAAGAGA |
|
| R:
GTGATGCTGAGAAGTTTCGCTG |
| CD44 | F:
ACTGGAACCCAGAAGCACAC |
|
| R:
TGTCCCTGTTGTCGAATGGG |
| Sox2 | F:
ATGGACAGTTACGCGCACAT |
|
| R:
CGAGCTGGTCATGGAGTTGT |
| Nanog | F:
AGATGTCTTCTGCTGAGATGC |
|
| R:
TTGCGTCACACCATTGCTA |
| PD-L1 | F:
TGCCGACTACAAGCGAATTACTG |
|
| R:
CTGCTTGTCCAGATGACTTCGG |
| IL-6 | F:
AGTGAGGAACAAGCCAGAGC |
|
| R:
AGCTGCGCAGAATGAGATGA |
| CCL2 | F:
TCTCGCCTCCAGCATGAAAG |
|
| R:
GGCATTGATTGCATCTGGCT |
Cell Counting Kit (CCK)-8
After 48 h of transfection, A549 cells from each
group were seeded at 2,000 cells/well in a 96-well plate. After 24
h of incubation, 20 µl CCK-8 reagent (GlpBio, GK10001) was added
per well, followed by a 2-h, 37°C incubation. The absorbance was
measured at 450 nm using a microplate reader to assess cell
viability, as follows: Cell viability (%)=(ODexperimental
group/ODpositive drug group/ODcontrol
group) ×100.
Transwell migration and invasion
assays
After the A549 and HCI-H1975 cells were resuspended
in serum-free DMEM, 1×10^4 cells were inoculated into each well of
a 24-well low-adhesion plate and total volume is 500 µl. A 200-µl
cell suspension was added to the upper chamber of the Transwell
system. The lower chamber was supplemented with 500 µl DMEM
containing 20% fetal bovine serum. After incubation at 37°C for 24
h, the supernatant and cell debris were removed. At 25°C, the cells
were fixed with 100% methanol for 30 min, stained with 0.1% crystal
violet for 20 min and washed with PBS. Finally, three randomly
selected fields were observed under a ×200 inverted light
microscope to assess cell migration. For the invasion assay, the
same protocol was performed; however, the upper chamber of the
Transwell system was coated with 8% Matrigel at 37°C for 3 h before
cell seeding.
Spheroid formation assay
A549 cells were seeded at 1×104 cells/500
µl volume per well into low-adhesion 24-well plates. DMEM/F12
(Wuhan Servicebio Technology Co., Ltd, containing 20 ng/ml
epidermal growth factor, 10 ng/ml basic fibroblast growth factor
and 10 µg/ml B27) was added and the cells were incubated at 37°C
for 24, 48 and 72 h. Images were captured using an light microscope
at 72 h post-seeding, after which the spheroids were counted.
Nude mouse transplantation tumor
model
A total of 12 male BALB/c nude mice (age, 5–6 weeks;
body weight, ~16 g) were provided by Hunan Sileke Jingda
Experimental Animal Co., Ltd. [license no. SCXK (Xiang) 2025-0004]
and the experiment was approved by the Animal Ethics Committee of
Fuzhou University Affiliated Provincial Hospital [approval no.
IACUC-FPH-SL-20250527[1092]). Human peripheral blood mononuclear
cells (PBMCs) were purchased from the American Type Culture
Collection (cat. no. PCS-800-011). The use of commercially
available anonymous primary cells was exempted for ethics review by
the Ethics Committee of Fuzhou University Affiliated Provincial
Hospital. The mice were randomly divided into four groups: A549,
A549 + sh-NC, A549 + short hairpin (sh)RNA-TBX20 (sh-TBX20; A549
cell line stably transduced with sh-TBX20 lentiviral plasmid) and
A549 + oe-TBX20 (A549 cell line stably transfected with
pcDNA3.1-oe-TBX20). Each group contained 3 mice.
Using pcDNA3.1 as the vector, restriction
endonucleases NheI and XbaI were used as the cutting sites.
Mutant primers were designed and synthesized by Shanghai Sangon
Biotech Co., Ltd., and the target gene was amplified by PCR to
obtain the mutant TBX20 cDNA sequence. The forward primer sequence
was 5′-GCTAGCATGGCGGCGGCGGCGGCGGCGGC-3′ (containing NheI
site) and the reverse primer sequence was
5′-TCTAGATCAGTCATCATCATCGTCATCAT-3′ (containing XbaI sit). For the
T262M point mutation, the mutagenic primer sequences were as
follows: Forward 5′-GCTAGCATGGCGGCGGCGGCGGCGGCGGC-3′ and reverse
5′-TCTAGATCAGTCATCATCATCGTCATCAT-3′, with the mutation introduced
by site-directed mutagenesis using PrimeSTAR high-fidelity DNA
polymerase (Takara Bio, Inc.; cat. no. R045A). PCR amplification
was performed with the following conditions: Initial denaturation
at 98°C for 2 min; 35 cycles of 98°C for 10 sec, 60°C for 15 sec,
and 72°C for 2 min; final extension at 72°C for 5 min. The target
gene was recombined and transformed with pcDNA3.1, and the obtained
sequence was confirmed by Sanger sequencing (Shanghai Sangon
Biotech Co., Ltd.) to ensure its accuracy. Wild-type TBX20-pcDNA3.1
and mutant TBX20-T262M-pcDNA3.1 overexpression plasmids were
constructed by Shanghai Sangon Biotech Co., Ltd.
The second-generation lentiviral packaging system
was used for lentivirus production. 293T cells (human embryonic
kidney cells; American Type Culture Collection, Cat. No. CRL-3216)
were seeded at 4×106 cells per 10-cm dish 24 h prior to
transfection. At 70–80% confluence, cells were co-transfected with
7.5 µg shRNA-TBX20 plasmid (forward,
5′-CCGGGCAGAAGATCAAGATCAATTCTCGAGAATTGATCTTGATCTTCTGCTTTTTG-3′ and
reverse,
5′-AATTCAAAAAGCAGAAGATCAAGATCAATTCTCGAGAATTGATCTTGATCTTCTGC-3′),
plasmid backbone (pL KO.1-puro Lentiviral shRNA expression vector
(purinomycin resistance); Sigma-Aldrich, Cat. No. SHC001), 5.6 µg
psPAX2 packaging plasmid (Addgene, 12260), and 1.9 µg of pMD2.G
envelope plasmid (Addgene, Inc.; cat. no. 12259) using
Lipofectamine 3000 (Thermo Fisher Scientific, Inc., L3000015)
according to the manufacturer's instructions. The plasmid ratio was
4:3:1 (shRNA:psPAX2:pMD2.G). Transfection was performed in Opti-MEM
reduced serum medium at 37°C, 5% CO2 for 12 h, after
which the medium was replaced with fresh DMEM containing 10% FBS.
At 48 and 72 h after transfection, supernatants and centrifuge at
500 × g for 5 min at 4°C. Then filter through a 0.45 µm PVDF
membrane. After aliquoting, store at −80°C. Viral titer was
determined by RT-qPCR.
For lentiviral transduction, A549 cells were seeded
at 2×105 cells per well in 6-well plates. When cell
density reached 50% confluence, lentivirus at MOI of 8 were added.
The cells were gently mixed and cultured at 37°C for 24 h. Cells
were selected using puromycin (2 µg/ml). Continuous screening was
conducted for 5 days. Cells were cultured for 24 h before
subsequent experiments. Infection efficiency of the lentivirus was
verified by RT-qPCR and Western blot. Culture medium containing
puromycin (1 µg/ml) was used to maintain the cells.
Stable overexpression and interference cell lines
(oe-TBX20 and shTBX20) of the TBX20 gene were constructed. A549
cells that stably expressed oe-TBX20 and shTBX20 were resuspended
in PBS (2×107 cells/ml) and gently mixed with an equal
volume of Matrigel to obtain a cell-scaffold mixture with a final
concentration of 1.0×107 cells/ml. Subsequently, 100 µl
mixture was injected subcutaneously into the right axilla of each
nude mouse. PBMCs were expanded by culturing in a complete medium
containing 100 ng/ml IL-2. The cells were resuspended in sterile
PBS to a concentration of 1.0×108 cells/ml and stored on
ice for later use. The PBMCs were resuspended in PBS at a
concentration of 1.0×108 cells/ml, and 100 µl PBMC
suspension was injected subcutaneously at the edge of the tumor
nodule (not directly injected into the tumor center). At 30 days
after the injection of tumor cells, the mice were euthanized by
intraperitoneal injection with 1% pentobarbital sodium (200 mg/kg)
and decapitation, and the tumors were removed and weighed. Humane
endpoints were as follows: Maximum tumor diameter exceeded 20 mm;
maximum tumor volume exceeded 2,000 mm3; obvious signs
of cachexia. No mice were sacrificed prematurely.
Immunohistochemistry (IHC)
Tumor tissues were fixed with 4% paraformaldehyde
solution for 24 h at 4°C. The tissue blocks were sectioned into 4
µm slices, baked at 60°C for 30 min. The sections underwent routine
xylene dewaxing, followed by alcohol hydration with a gradient, and
three washes with distilled water for 3 min each. Subsequently,
they were rinsed three times with PBS for 3 min each. The antigen
retrieval solution containing EDTA, pH 9.0, Wuhan Servicebio
Technology Co., Ltd.) was boiled in a high-pressure cooker for 2
min. The sections were then placed in an incubation chamber, and 3%
hydrogen peroxide solution was added. After incubation at 25°C for
12 min, the sections were rinsed three times with PBS solution for
3 min each. Then, 5% goat serum (Nanjing SenBeiJia Biological
Technology Co., Ltd.) was added, and the sections were incubated at
25°C for 20 min. TBX20 primary antibody (Thermo Fisher Scientific,
Inc., cat. no. PA5-110464; 1:500) was added overnight at 4°C.
Sections were rinsed three times with PBS buffer for 2 min each to
remove excess primary antibody. The secondary antibody (HRP-labeled
rabbit anti-IgG, 1:500; cat. no. TBAG0030, Wuhan Servicebio
Technology Co., Ltd.) was added, and the sections were incubated at
25°C for 30 min. After rinsing three times with PBS buffer for 2
min each, the prepared DAB chromogenic solution (Wuhan Servicebio
Technology Co., Ltd.) was added. The staining was observed under an
light microscope. Sections were stained with hematoxylin (Beijing
Biosharp Technology Co., Ltd) for 5 min, followed by rinsing with
distilled water. Staining was evaluated independently by two
pathologists who were blinded to the clinical data. Positive
staining was defined as brown nuclear staining. Images of five
representative high-power fields (magnification, ×400) per tumor
were photographed. The percentage of positive tumor cells was
recorded.
Flow cytometry
Flow cytometry was used to detect immune cells.
Fresh tumor tissues were placed in cold PBS and minced with
ophthalmic scissors. The tissue was then transferred to a 15-ml
centrifuge tube and digested with 5 ml digestion buffer (RPMI-1640
+ 2 mg/ml collagenase IV + 20 µg/ml DNase I) in a 37°C water bath
with oscillation. After the digestion was complete, 5 ml RPMI-1640
containing 10% FBS was added to terminate the reaction. The
suspension was filtered through a 70-µm cell sieve to collect the
single-cell suspension and was then centrifuged at 400 × g for 5
min at 4°C, after which the supernatant was discarded, and the
cells were resuspended in PBS. Next, 1 ml ACK lysis buffer was
added, the mixture was incubated for 2 min and the reaction was
terminated with PBS. The cell suspension was adjusted to
1×106 cells/100 µl, and was transferred to a flow
cytometry tube. Fc blocking agent (Rat Anti-Mouse CD16/CD32, BD
Biosciences, cat. No. 553142) was added, and the sample was
incubated at 4°C for 10 min. A surface antibody mixture (CD3, CD4,
CD8, Foxp3, CD86 and CD206) was then added and incubated at 4°C in
the dark for 30 min. The labelled antibodies for CD4+ T cells are:
CD3 (FITC anti-human CD3, BioLegend, Inc., cat. No. 317306), CD4
(PE-anti-human CD4, BioLegend, Inc., Cat. No. 300508). The labelled
antibodies for CD8+ T cells are: CD3, CD8 (PE anti-human CD8
Antibody, BioLegend, Inc., Cat. No. 344705). The labelled
antibodies for Treg cells are: CD3, CD4, Foxp3 (APC
Anti-FOXP3Antibody[3G3], Abcam, Cat. No. ab200568). The labelled
antibody for M1 cells is: CD86 (FITC anti-human CD86 Antibody,
BioLegend, Inc., Cat. No. 374203). The labelled antibody for M1
cells is: CD206 (FITC anti-human CD206 (MMR) Antibody, BioLegend,
Inc., Cat. No. 321103). The suspension was subsequently washed
twice with PBS (4°C, 300 × g, 5 min each), the supernatant was
discarded, and a FoxP3 fixation/permeabilization kit (eBioscience™
Foxp3/Transcription Factor Staining Buffer Set; Cat. no.
00-5523-00, Thermo Fisher Scientific, Inc.) was used. Briefly, 100
µl fixation/membrane disruption solution was added and the sample
was incubated at 25°C in the dark for 30–60 min. The sample was
then washed once with 1X membrane disruption buffer and then
centrifuged (4°C and 400 × g for 5 min) to remove the supernatant.
The sample was then resuspended in 50 µl membrane disruption
buffer, PE-labelled anti-FoxP3 antibody was added and the sample
was incubated at 4°C in the dark for 30 min. The cells were then
washed once with membrane disruption buffer, resuspended in 200–300
µl PBS and prepared for flow cytometry. A BD LSRFortessa flow
cytometer (BD Biosciences) with appropriate laser and filter
settings was used. At least 50,000 CD45+ leukocyte
events per sample were collected, isotype control (Mouse IgG1, κ
isotype Ctrl antibody, BioLegend, Inc., Cat. no. 400105) and
single-stain compensation tubes were used for normalization, and
the data were analyzed using FlowJo v10 software (BD
Biosciences).
Statistical analysis
Statistical analysis was performed using SPSS26.0
(IBM Corp.) and GraphPad Prism 8.0.2 (Dotmatics), with all
experiments repeated three times. Quantitative data (conforming to
normality and variance homogeneity) are presented as the mean ±
standard deviation. One-way ANOVA was used for inter-group
comparisons, after which, the SNK post hoc test was used to analyze
datasets containing ≤3 groups, whereas multiple comparisons between
>3 groups were analyzed using the Tukey post hoc test.
Independent samples t-test was employed for comparisons between two
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
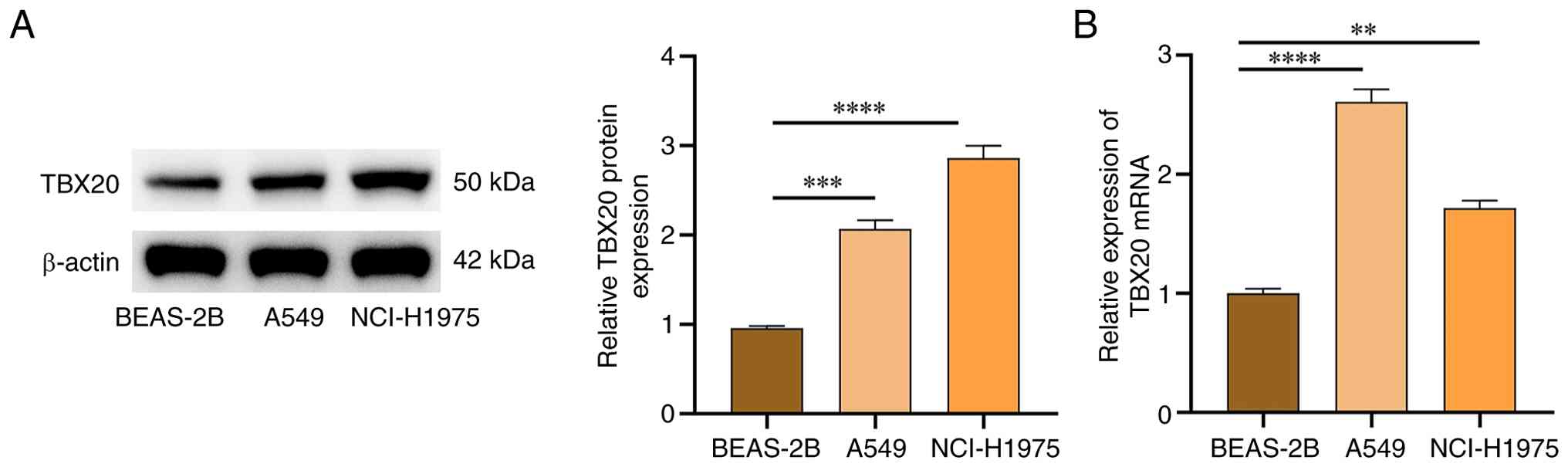
TBX20 expression in cancer cells
The expression levels of TBX20 in lung cancer cells
were assessed by western blotting, which revealed that TBX20
protein expression was significantly higher in in A549 and
NCI-H1975 cancer cells compared with that in BEAS-2B cells
(P<0.05; Fig. 1A). qPCR further
confirmed the statistically significant increases in TBX20 mRNA
expression levels in these cell lines (P<0.05; Fig. 1B). As the expression of TBX20 mRNA
was highest in A549, A549 cells were used for subsequent
experiments.
Effects of TBX20 on lung cancer cell
viability, migration, invasion and spheroid formation
The effects of TBX20 overexpression/silencing were
subsequently investigated in lung cancer cells. The successful
transfection of si-TBX20 and oe-TBX20 is shown in Fig. 2A and B. Compared with the BEAS-2B
group, the A549 group exhibited significantly greater cell
viability (Fig. 2C), migration
(Fig. 2D and G), invasion (Fig. 2E and G) and spheroid formation
capacity (Fig. 2F and G)
(P<0.05), whereas no significant differences were observed
between the A549 and A549-siNC groups (P>0.05). Following TBX20
overexpression, compared with in the A549-NC group, cell viability,
migration and invasion, and spheroid formation capacity were
significantly increased (P<0.05). By contrast, after TBX20
silencing, cell viability, migration and invasion and spheroid
formation capacity were significantly reduced compared with in the
A549-siNC group (P<0.05).
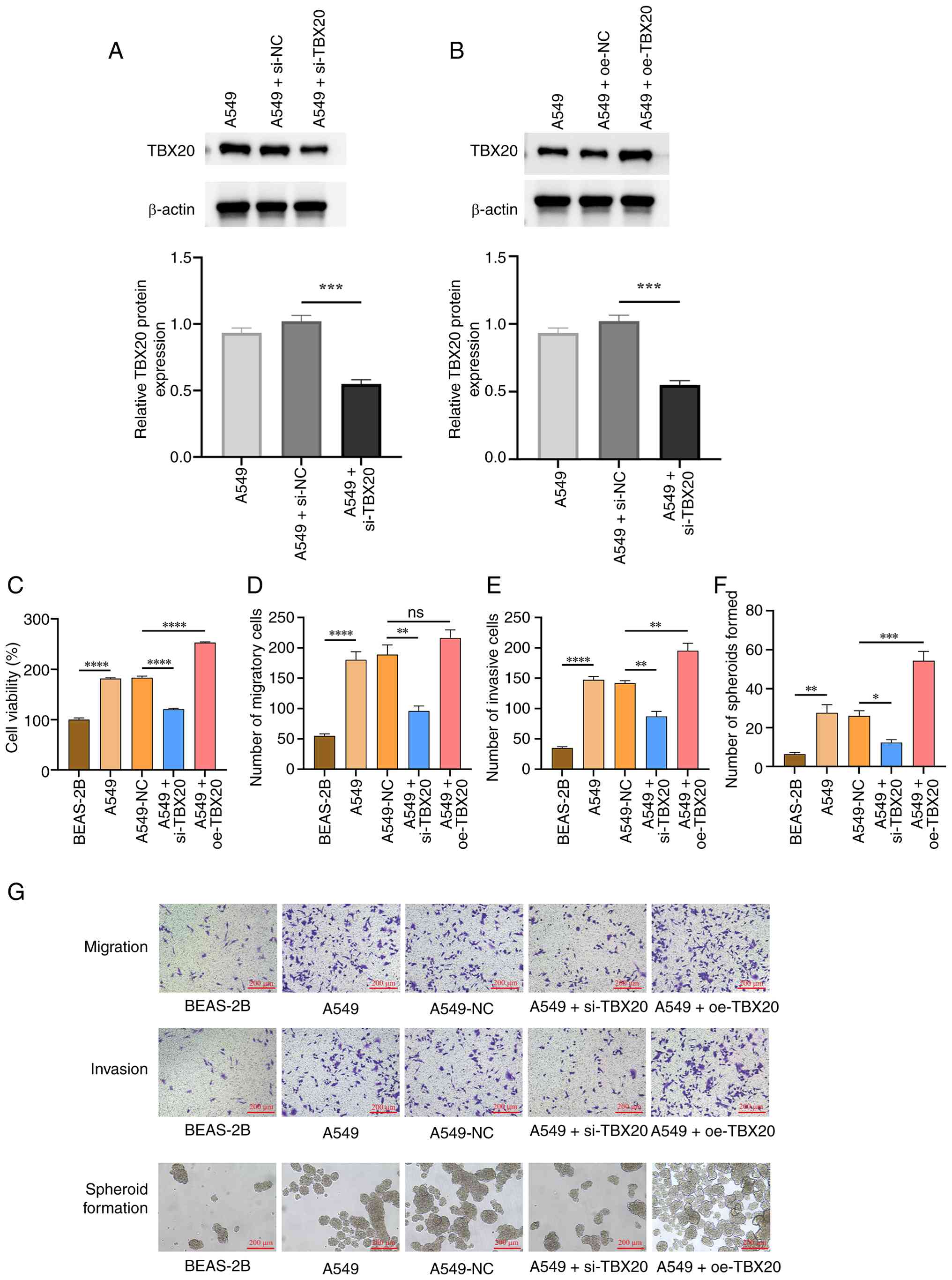 | Figure 2.Effects of oe-TBX20 and si-TBX20 on
lung cancer cell viability, migration, invasion and spheroid
formation. (A and B) Western blotting confirmed that TBX20
transfection was successful. Protein expression levels of TBX20 in
A549 cells after (A) si-TBX20 and (B) oe-TBX20 transfection. (C)
Cell viability, (D) cell migration, (E) cell invasion and (F) cell
spheroid formation were assessed. (G) Transwell and sphere
formation assay images (magnification, ×100). Data are presented as
the mean ± standard deviation (n=3). *P<0.05, **P<0.01,
***P<0.001 and ****P<0.0001. NC, negative control; oe,
overexpression; si, small interfering; TBX20, T-box transcription
factor 20; ns, not significant (P>0.05).. |
Effects of TBX20 on the expression
levels of EMT-, stemness- and immune-associated protein and genes
in lung cancer cells
To evaluate the effects of TBX20
overexpression/silencing on EMT, stemness and immune activity in
lung cancer cells, western blotting and RT-qPCR were used to
measure the expression levels of EMT markers (E-cadherin,
N-cadherin and vimentin) (16),
stemness-related markers (CD44 and Nanog) (17) and immune-related cytokines (PD-L1,
IL-6 and CCL2) (18). The results
of western blotting (Fig. 3A)
revealed that, compared with in the BEAS-2B group, the A549 group
exhibited significantly reduced E-cadherin protein expression;
higher N-cadherin and vimentin levels; higher CD44 and Nanog
expression; and higher expression levels of PD-L1, IL-6 and CCL2
(P<0.05; Fig. 3B-D and F-J).
Compared with the A549 + NC group, the expression of Sox2 in the
A549 + oe-TBX20 group showed no significant change (Fig. 3E). However, no significant
differences were observed between the A549 and A549-NC groups
(P>0.05). After TBX20 overexpression, the expression levels of
E-cadherin were significantly lower, whereas those of N-cadherin,
vimentin, CD44, Nanog, PD-L1 and IL-6 levels were higher than those
in the A549-NC group (P<0.05). Conversely, compared with in the
A549-NC group, TBX20 silencing led to increased E-cadherin
expression, but significantly decreased N-cadherin, vimentin, CD44,
PD-L1 and IL-6 levels (P<0.05).
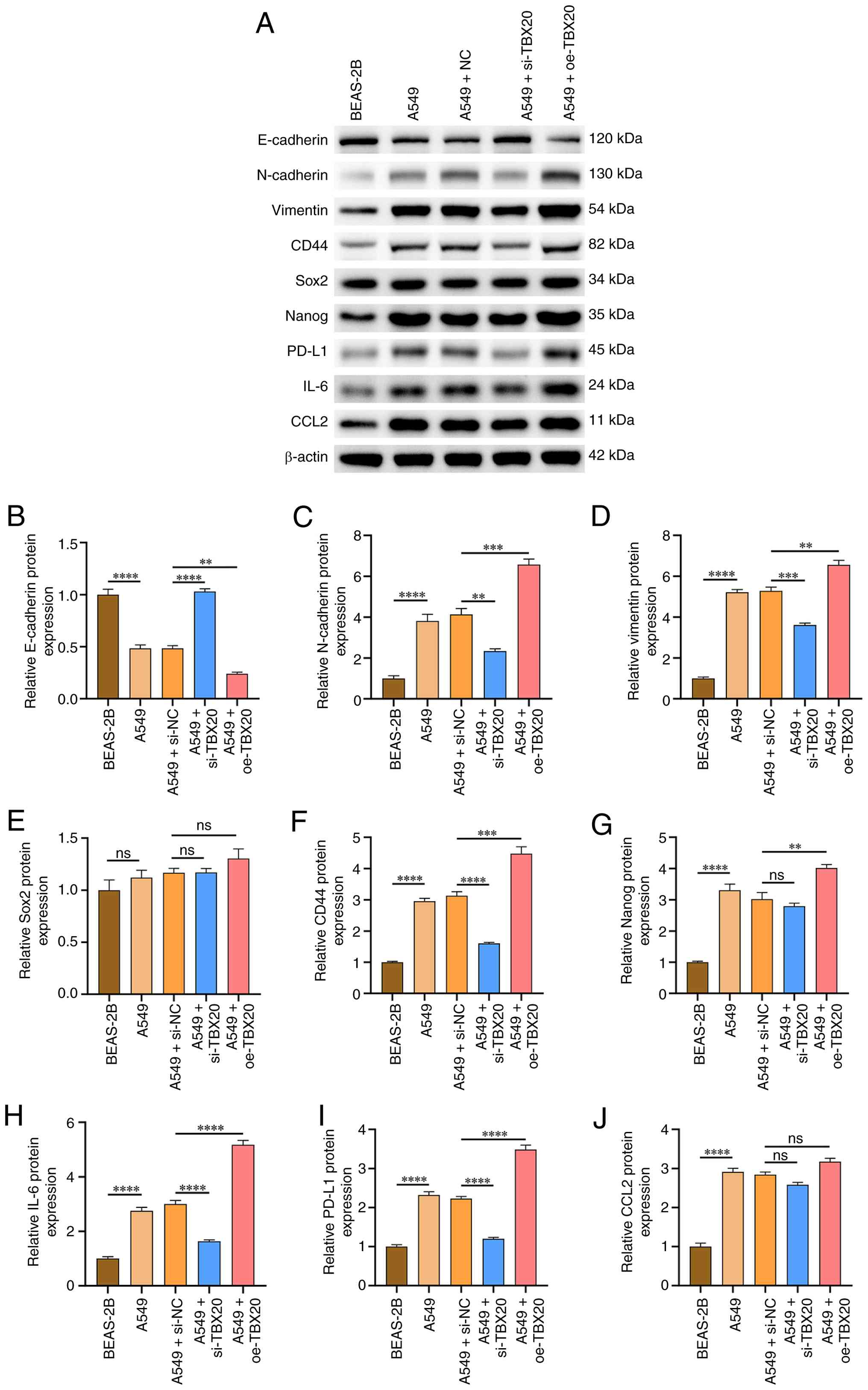 | Figure 3.Effects of oe-TBX20 and si-TBX20 on
epithelial-mesenchymal transition- and stemness phenotype-related
proteins in lung cancer cells. (A) Western blot analysis. Protein
expression levels of (B) E-cadherin, (C) N-cadherin, (D) vimentin,
(E) Sox2, (F) CD44, (G) Nanog, (H) IL-6, (I) PD-L1 and (J) CCL2.
Data are presented as the mean ± standard deviation (n=3).
**P<0.01, ***P<0.001, ****P<0.0001 and
nsP>0.05. NC, negative control; oe, overexpression;
si, small interfering; TBX20, T-box transcription factor 20. |
The RT-qPCR results revealed that compared with in
the BEAS-2B group, the A549 group exhibited significantly lower
E-cadherin mRNA expression, and significantly higher mRNA
expression levels of N-cadherin, vimentin, Sox2, CD44, Nanog,
PD-L1, IL-6 and CCL2 (P<0.05; Fig.
4). However, no significant differences were observed between
the A549 and A549-NC groups (P>0.05). After TBX20 was
overexpressed, compared with the A549-NC group, E-cadherin
expression was significantly decreased, whereas the levels of
vimentin, Sox2, CD44, Nanog, PD-L1, IL-6 and CCL2 were
significantly increased (P<0.05). Conversely, after TBX20
silencing, compared with in the A549-NC group, E-cadherin
expression was significantly increased, and the levels of
N-cadherin, vimentin, Sox2, CD44, Nanog, PD-L1, IL-6 and CCL2 were
decreased (P<0.05).
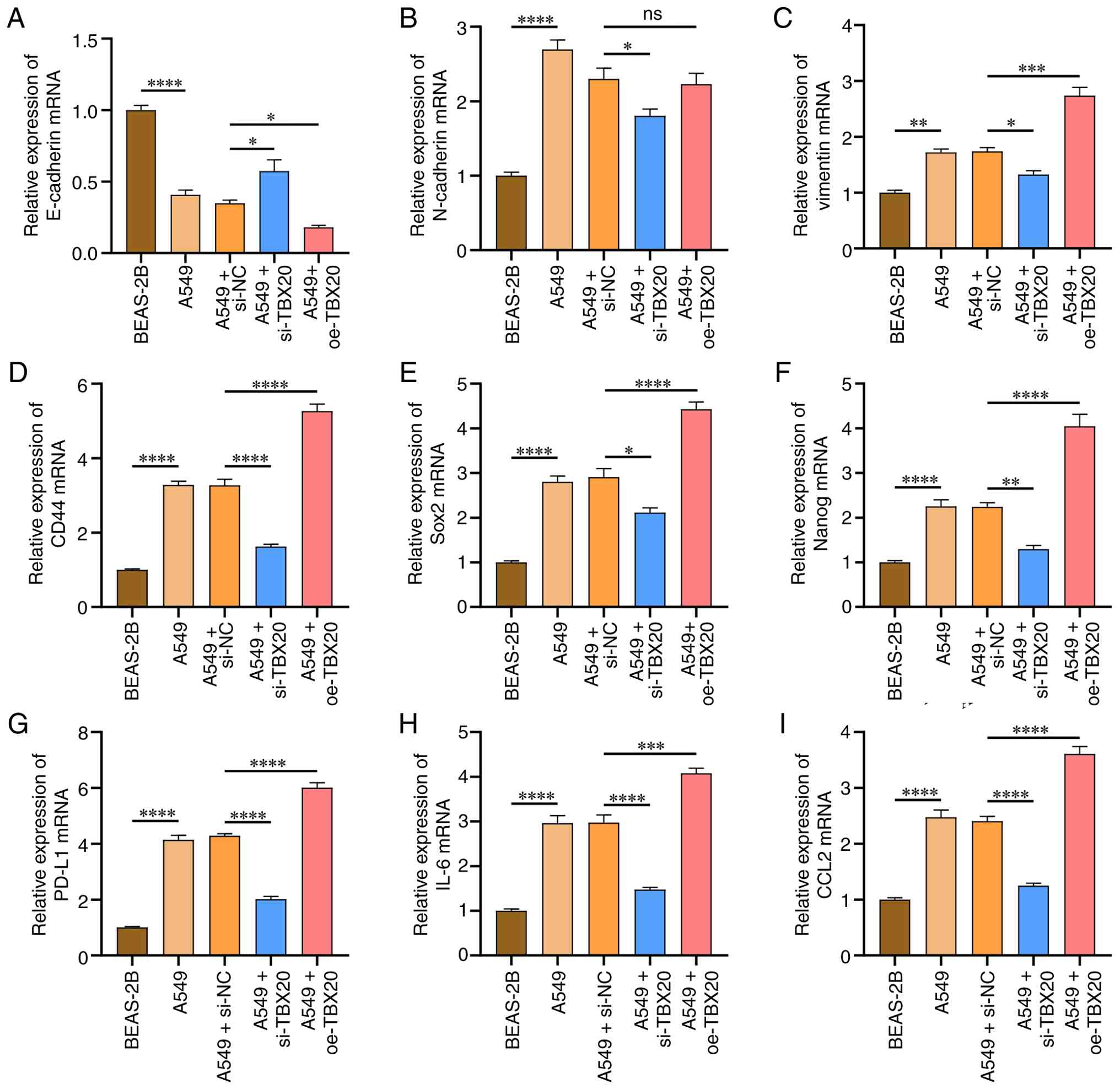 | Figure 4.Effects of oe-TBX20 and si-TBX20 on
epithelial-mesenchymal transition- and stemness
phenotype-associated mRNA expression in lung cancer cells. mRNA
expression levels of (A) E-cadherin, (B) N-cadherin, (C) vimentin,
(D) CD44, (E) Sox2, (F) Nanog, (G) PD-L1, (H) IL-6 and (I) CCL2.
Data are presented as the mean ± standard deviation (n=3).
*P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 and
nsP>0.05. NC, negative control; oe, overexpression;
si, small interfering; TBX20, T-box transcription factor 20. |
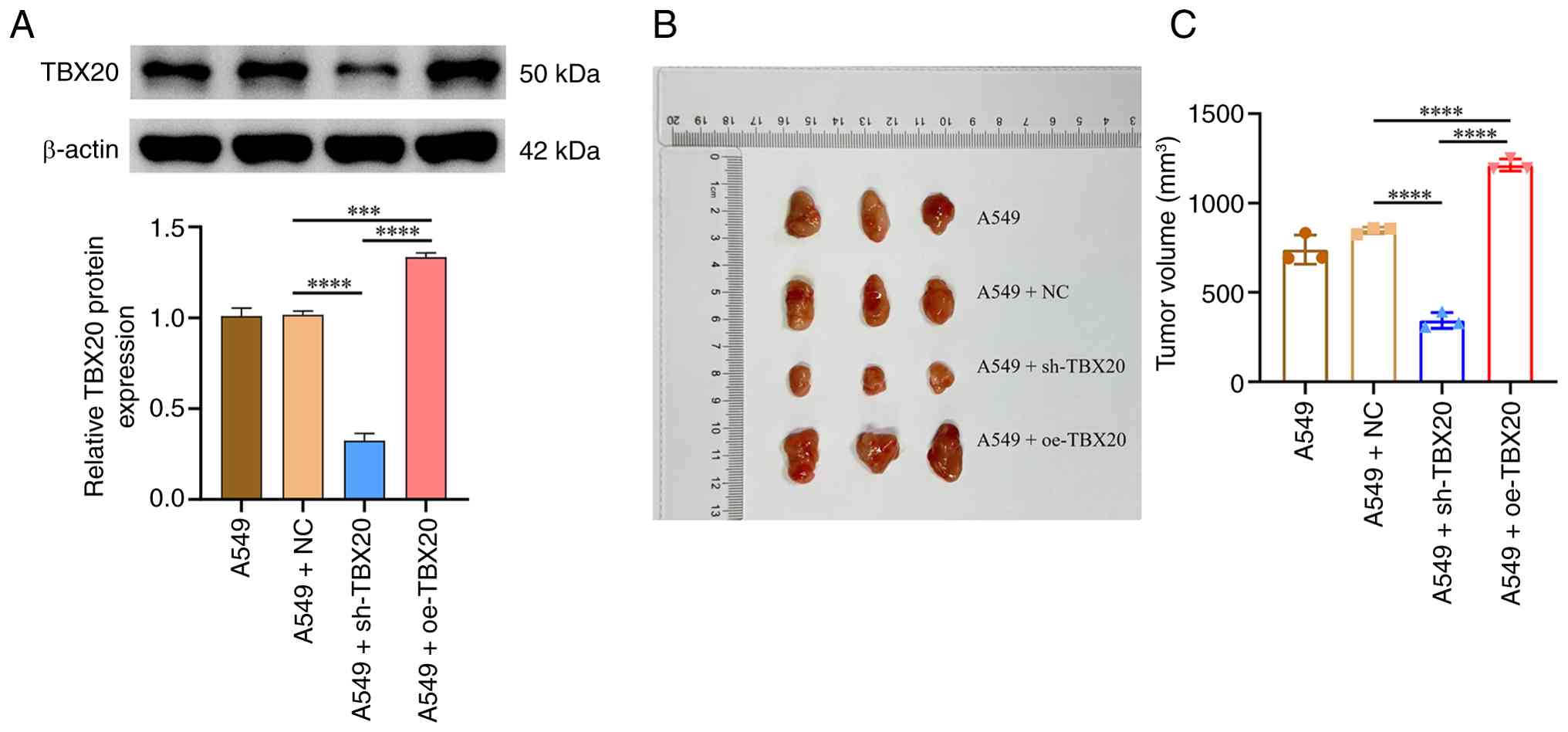
Effects of TBX20 overexpression and
silencing on subcutaneous tumor growth in nude mice
In vivo experiments were conducted to
evaluate the effects of TBX20 overexpression and silencing on tumor
size. The expression levels of TBX20 in the A549 + oe-TBX20 group
were significantly greater than those in the A549 + NC group
(P<0.05), whereas the A549 + sh-TBX20 group exhibited
significantly lower expression (P<0.05), confirming successful
transfection (Fig. 5A). At 4 weeks
after subcutaneous tumor transplantation, tumor volume measurements
revealed that the tumors in the A549 + oe-TBX20 group were
significantly larger than those in the A549-NC group (P<0.05);
the maximum diameter and tumor volume in the A549 + oe-TBX20 group
were 20 mm and 1,273 mm3, respectively, whereas those in
the A549 + sh-TBX20 group were significantly smaller (P<0.05;
Fig. 5B and C).
Effects of TBX20 overexpression and
silencing on the expression of EMT and stemness markers in
subcutaneous tumors from nude mice
To assess the effects of TBX20 on tumor EMT and
stemness markers in nude mice, IHC was performed (Fig. 6A) to detect the expression levels of
E-cadherin, N-cadherin and vimentin (EMT markers), CD44 (cellular
stemness marker) and PD-L1 (tumor immune escape indicator) in the
subcutaneous tumor tissues of nude mice. Compared with in the A549
group, the A549 + oe-TBX20 group exhibited significantly lower
E-cadherin levels (Fig. 6B),
whereas N-cadherin (Fig. 6C),
vimentin (Fig. 6D), CD44 (Fig. 6E) and PD-L1 levels (Fig. 6F) were significantly greater
(P<0.05). Conversely, compared with in the control group, the
A549 + sh-TBX20 group demonstrated significantly higher E-cadherin
levels, with significant reductions in N-cadherin, vimentin, CD44
and PD-L1 levels (P<0.05).
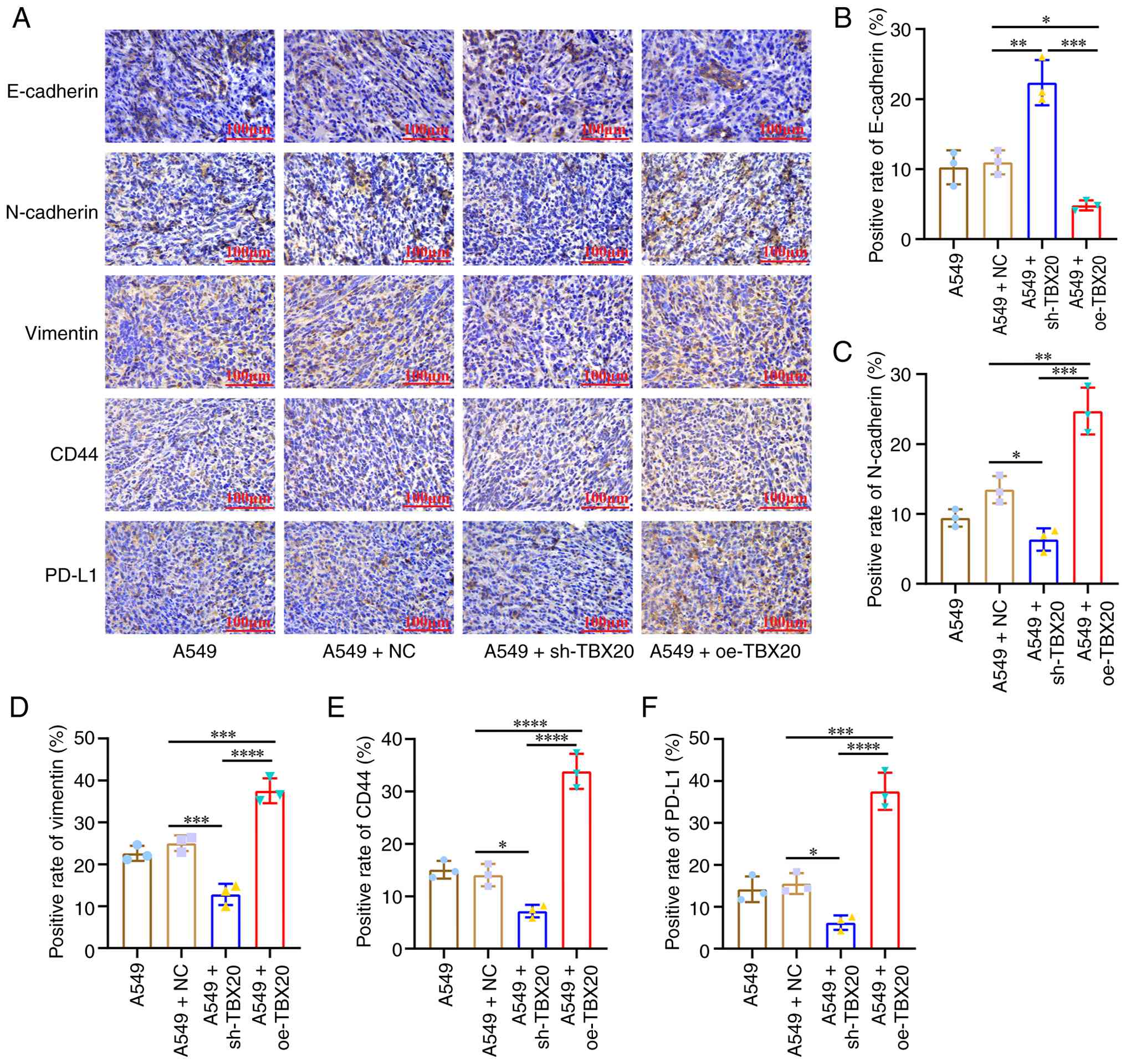 | Figure 6.IHC staining of EMT and stemness
indicators in subcutaneous tumor models of nude mice. (A) IHC
staining of E-cadherin, N-cadherin, vimentin, CD44 and PD-L1 in
A549 subcutaneous tumor models (magnification, ×400). (B)
E-cadherin, (C) N-cadherin, (D) vimentin, (E) CD44 and (F) PD-L1
positivity rates across different groups. Data are presented as the
mean ± standard deviation (n=3). *P<0.05, **P<0.01,
***P<0.001 and ****P<0.0001. IHC, immunohistochemistry; NC,
negative control; oe, overexpression; si, small interfering; TBX20,
T-box transcription factor 20. |
Effects of TBX20 overexpression and
silencing on immune cell infiltration in subcutaneous tumors from
nude mice
To assess the effects of TBX20 on immune cell
infiltration in nude mice, flow cytometry was performed to detect
the infiltration of CD8+ T cells and CD4+ T
cells in subcutaneous tumor tissues, along with the proportions of
M1 and M2 macrophages, and regulatory T cells (Tregs). The
inoculation of human PBMCs into nude mice (lacking functional T
cells) provides a reconstructed host environment capable of
eliciting an immune response for human-derived tumors.
CD8+ T cells and CD4+ T cells are the core
components of PBMCs, whereas Tregs are also a functional subgroup
present in PBMCs. Compared with in the A549 + sh-TBX20 group, the
A549 + oe-TBX20 group exhibited significantly lower infiltration
levels of CD4+ T cells, CD8+ T cells and M1
cells, but significantly greater infiltration levels of M2 cells
and Tregs (P<0.05; Fig. 7).
Conversely, compared with in the A549 + NC group, the A549 +
sh-TBX20 group exhibited significantly higher infiltration levels
of CD4+ T cells, CD8+ T cells, and M1
macrophages, but significantly lower infiltration levels of M2
macrophages and Tregs (P<0.05).
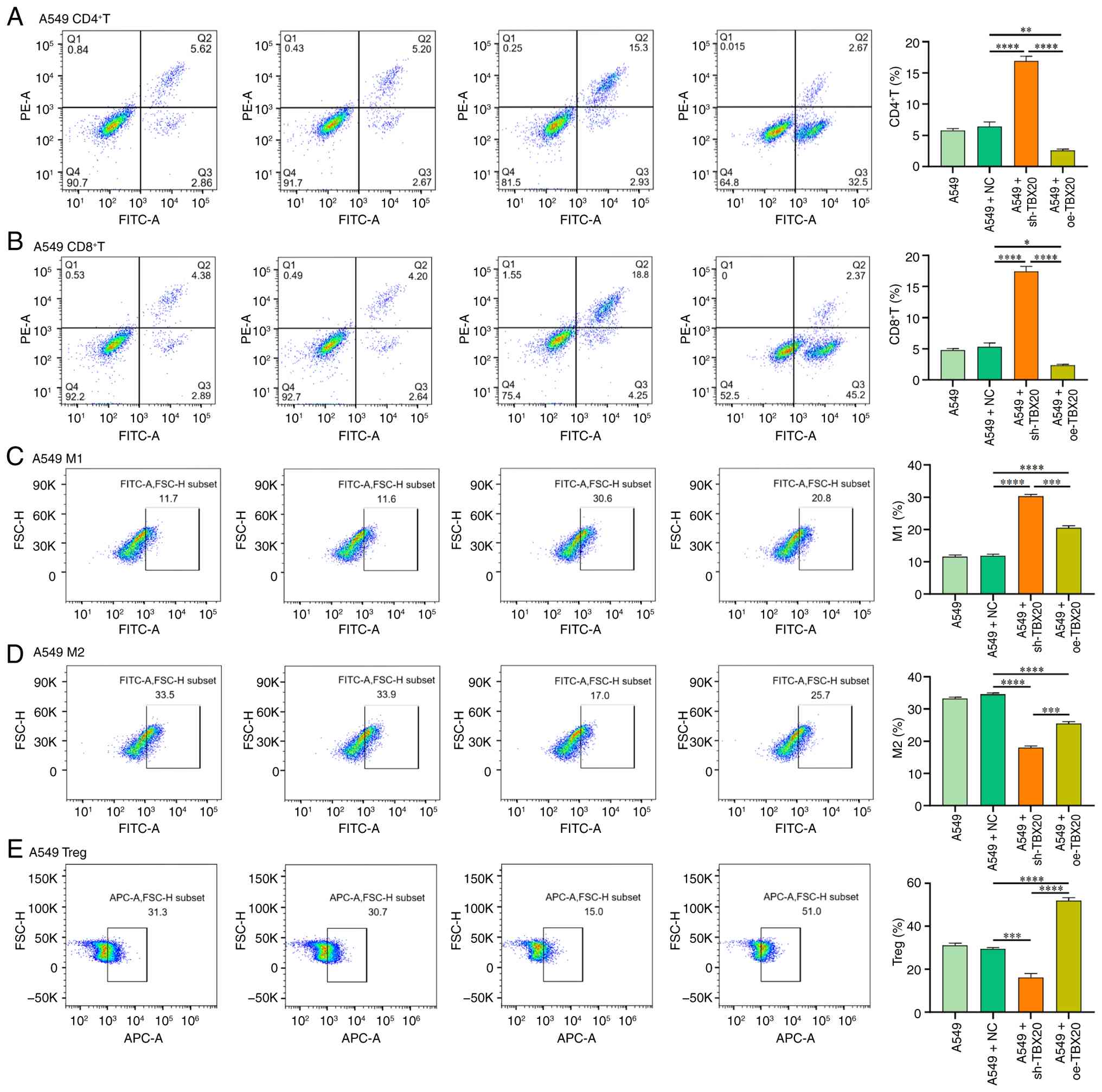 | Figure 7.Flow cytometric analysis of
CD4+ T cells, CD8+ T cells, M1 cells, M2
cells and Tregs. Infiltration levels of (A) CD4+ T
cells, (B) CD8+ T cells, (C) M1 macrophages, (D) M2
macrophages and (E) Tregs in A549 cell xenograft tumors across
different groups. Data are presented as the mean ± standard
deviation (n=3). *P<0.05, **P<0.01, ***P<0.001 and
****P<0.0001. NC, negative control; oe, overexpression; si,
small interfering; TBX20, T-box transcription factor 20; Treg,
regulatory T cell. |
Discussion
TBX20 is a TF that serves a key regulatory role
during development, although its expression patterns in tumor
tissues remain poorly understood. As a member of the TBX1
subfamily, TBX1 acts as a tumor activator in prostate cancer by
promoting ribosomal RNA gene transcription (7). In breast cancer, TBX1 expression
levels are significantly higher in tumor tissues than in adjacent
normal breast tissue, with gradual increases observed as tumor
stages progress. Knocking down TBX1 expression can markedly inhibit
the proliferation, colony formation, migration and invasion of
breast cancer cell (19). TBX20 is
classified as a member of the TBX1 subfamily, and shares homology
with TBX1 in terms of protein structure (conserved T-box
DNA-binding domain) and chromosomal distribution (20).
The present study investigated the biological
functions of TBX20 in lung cancer cells, particularly its effects
on cell viability, migration, invasion and spheroid formation. By
establishing an A549 cell line overexpressing TBX20, it was
revealed that TBX20 promoted malignant behaviors in lung cancer
cells. Conversely, TBX20 knockdown significantly inhibited these
malignant behaviors, indicating its crucial regulatory role in lung
cancer progression. Previous studies have suggested that TBX20 is
highly expressed in various tumors and is associated with poor
prognosis (21,22). Consistent with these findings, in
the present study, TBX20 was revealed to be highly expressed in
lung cancer cell lines, and was able to regulate four key malignant
phenotypes: Viability, migration, invasion and spheroid formation,
with particularly enhanced spheroid formation capacity. This
ability is a hallmark of cancer stem cells (CSCs), which possess
superior immune evasion capabilities (23). Furthermore, lung cancer cells with
spheroid formation capacity typically exhibit enhanced resistance
to chemotherapy and radiotherapy (24). Clinically, the spheroid formation
capacity of lung cancer cells is a potential prognostic biomarker
(25). Therefore, the development
of immunotherapy strategies targeting TBX20-regulated tumor stem
cells with spheroid formation and immune evasion capabilities could
lead to novel breakthroughs for improving outcomes for patients
with lung cancer.
To investigate the specific mechanisms of TBX20 in
lung cancer cells the current study further examined markers
associated with EMT, tumor stem cell characteristics and immune
evasion. By comparing TBX20-overexpressing lung cancer cells with
control cells, it was demonstrated that TBX20 overexpression
significantly reduced E-cadherin expression, but increased
N-cadherin, vimentin, CD44, Nanog, PD-L1, IL-6 and CCL2 expression.
Similar phenomena were observed in the in vivo experiments.
These findings indicated that TBX20 overexpression promotes
malignant progression through EMT induction, enhanced stemness and
immune evasion, thereby facilitating metastasis, therapy resistance
and immune escape in lung cancer.
E-cadherin, an adhesion molecule in epithelial
cells. Loss of E-cadherin weakens the adhesion force between cells,
making cancer cells more likely to detach and spread to other
parts. By contrast, N-cadherin, a mesenchymal cell adhesion
molecule, is typically upregulated in lung cancer tissues; this
elevated N-cadherin expression enhances cell adhesion and
migration, thereby promoting tumor invasion and metastasis
(26). Vimentin is an intermediate
filament protein. During the EMT process, lung cancer cells
increase the expression of Vimentin, acquiring the characteristics
of mesenchymal cells, thereby enhancing their motility and
invasiveness (27). In the current
study, TBX20 overexpression led to decreased E-cadherin expression,
and increased N-cadherin and vimentin expression, whereas TBX20
silencing resulted in elevated E-cadherin expression, and reduced
N-cadherin and vimentin levels. These findings suggest that TBX20
silencing may suppress lung cancer cell invasion and metastasis by
regulating the expression levels of EMT-associated proteins.
Both Sox2 and Nanog are stem cell TFs that have
pivotal roles in maintaining the characteristics of tumor stem
cells. In lung cancer, high expression levels of Sox2 and Nanog are
closely associated with tumor invasiveness, chemotherapy resistance
and EMT (28). CD44, a glycoprotein
on the cell surface, is also highly expressed in lung cancer and is
associated with the properties of tumor stem cells, promoting their
self-renewal and chemotherapy resistance. Furthermore, CD44
contributes to the formation and remodeling of the tumor
microenvironment (TME) by facilitating interactions between tumor
cells and stromal cells, thereby creating favorable conditions for
tumor growth and metastasis (29).
The present study demonstrated that TBX20 overexpression increased
the expression levels of Sox2, Nanog and CD44, whereas TBX20
silencing reduced these factors. These findings suggest that TBX20
suppression may inhibit lung CSC characteristics by regulating the
expression of the stem cell-associated factors Sox2 and Nanog, as
well as the cell surface glycoprotein CD44, thereby affecting the
invasiveness, chemotherapy resistance and metastatic capacity of
lung cancer cells.
PD-L1 is typically upregulated on tumor cell
surfaces (30). IL-6, a
multifunctional cytokine, serves key roles in inflammatory
responses, immune regulation and cell proliferation. IL-6
upregulates immune checkpoint molecules such as PD-L1, thereby
enhancing tumor cell immune evasion capabilities (31). CCL2 (also known as monocyte
chemotactic protein-1) is a key chemokine primarily secreted by
tumor cells and tumor-associated stromal cells, which recruits
immune cells such as monocytes, macrophages, myeloid-derived
suppressor cells and Tregs into the TME, thereby promoting immune
suppression and tumor progression (32). The present study demonstrated that
TBX20 overexpression increased the expression levels of PD-L1, IL-6
and CCL2, whereas TBX20 knockdown reduced these factors. These
findings suggest that inhibiting TBX20 may significantly modulate
immune evasion mechanisms in lung cancer cells by regulating the
expression of these immune factors.
In nude mouse tumor formation experiments, TBX20
overexpression significantly reduced the infiltration levels of
CD4+ T cells, CD8+ T cells, and M1-type
macrophages in subcutaneous tumor tissues, but increased the
proportions of M2-type macrophages and Tregs. Conversely, when
TBX20 was inhibited, these effects were reversed. These results
indicate that TBX20 not only serves as a key factor driving the
malignant behavior of tumor cells but also acts as a crucial
regulator of the tumor immune microenvironment.
M1-type macrophages typically exert antitumor
effects, whereas M2-type macrophages promote tumor growth,
angiogenesis and tissue remodeling through multiple mechanisms,
while suppressing adaptive immune responses (33). The present study demonstrated that
knocking down TBX20 led to an increase in M1 and a decrease in M2.
Compared to the TBX20 knockdown group, TBX20 overexpression
significantly shifted the M1/M2 balance towards M2, which may be a
critical factor in tumor progression. M2-type macrophages directly
inhibit T-cell activation and proliferation by secreting
immunosuppressive factors, such as IL-10 and TGF-β, and expressing
immune checkpoint molecules such as PD-L1 (33). While Tregs serve a crucial role in
maintaining immune tolerance, in the TME, they suppress antitumor
immunity through multiple mechanisms. The present study
demonstrated that the proportion of Tregs was significantly
elevated in the TBX20-overexpressing group, which is consistent
with the observed functional suppression of CD4+ and
CD8+ T cells. Tregs competitively deplete IL-2 in the
microenvironment, thereby inhibiting the effector functions of
coexisting CD8+ T cells, preventing these cells from
effectively eliminating cancer cells (34). The enrichment of TBX20-induced Tregs
may serve as a mechanism for tumor immune evasion. CD8+
T cells, the core executors of antitumor immunity, exhibit reduced
infiltration and functional suppression as hallmarks of immune
evasion (35). In the present
study, significantly decreased CD8+ T-cell infiltration
in the TBX20-overexpression group suggested that TBX20 created a
microenvironment unfavorable for the survival and function of
effector T cells.
The present study preliminarily demonstrated that
TBX20 can induce EMT, enhance stemness and upregulate
immune-related factors in A549 cells to promote immune evasion;
however, its downstream signaling pathways remain unexplored and
should be defined in future promoter deletion, DNA-binding
mutagenesis and rescue experiments. Future research should focus on
candidate pathways, such as BMP2/pSmad1/5/8 and
PI3K/AKT/GSK3β/β-catenin to identify functionally dependent nodes.
Furthermore, the animal sample size may have led to an
underestimation of significant differences; therefore, the cohort
will be expanded in subsequent experiments.
In conclusion, TBX20 was revealed to be highly
expressed in lung cancer cells. The overexpression of TBX20
significantly increased lung cancer cell viability, migration,
invasion and spheroid formation, whereas its knockdown reversed
these effects. Mechanistic studies revealed that TBX20 may induce
EMT, which is characterized by the downregulation of E-cadherin
expression, and the upregulation of N-cadherin and vimentin
expression. Additionally, TBX20 could enhance tumor cell stemness
by increasing the expression of the stem cell marker CD44, and may
promote immune evasion by upregulating immune-associated factors
such as PD-L1. These findings highlight the pivotal regulatory role
of TBX20 in the malignant phenotype of lung cancer cells, providing
a potential therapeutic target for targeted therapy. Inhibiting
TBX20 and its downstream factors could improve patient outcomes,
reduce tumor recurrence and metastasis risks, and ultimately
increase treatment efficacy and survival rates.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CX conceived and designed the study, analyzed data,
conducted experiments and wrote and revised the manuscript. PZ, YZ
and LL conducted experiments, performed analysis, and constructed
figures. GW conducted experiments, established the methodology,
analyzed data and revised the manuscript. All authors have read and
approved the final manuscript.. CX and GW confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate
The present study was approved by the Animal Ethics
Committee of Fuzhou University Affiliated Provincial Hospital
(approval no. IACUC-FPH-SL-20250527[1092]).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Bi JH, Tuo JY, Xiao YX, Tang DD, Zhou XH,
Jiang YF, Ji XW, Tan YT, Yuan HY and Xiang YB: Observed and
relative survival trends of lung cancer: A systematic review of
population-based cancer registration data. Thorac Cancer.
15:142–151. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li D, Tian J and Zou Y: Advancing the
understanding of the immune escape in lung cancer immunotherapy:
Global trends, collaborations, and future directions. Hum Vaccin
Immunother. 22:26283952026. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yi L, Zhou L, Shao B, Xiang T and Tang J:
Multifaceted role of T-box transcription factor 4: From embryonic
development to disease pathogenesis. Genes Dis. 13:1018112025.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shimoda M, Sugiura T, Imajyo I, Ishii K,
Chigita S, Seki K, Kobayashi Y and Shirasuna K: The T-box
transcription factor Brachyury regulates epithelial-mesenchymal
transition in association with cancer stem-like cells in adenoid
cystic carcinoma cells. BMC Cancer. 12:3772012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dong L, Lyu X, Faleti OD and He ML: The
special stemness functions of Tbx3 in stem cells and cancer
development. Semin Cancer Biol. 57:105–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang N, Li Y, Wei J, Pu J, Liu R, Yang Q,
Guan H, Shi B, Hou P and Ji M: TBX1 functions as a tumor suppressor
in thyroid cancer through inhibiting the activities of the PI3K/AKT
and MAPK/ERK pathways. Thyroid. 29:378–394. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cui J, Zhang Y, Ren X, Jin L and Zhang H:
TBX1 functions as a tumor activator in prostate cancer by promoting
ribosome RNA gene transcription. Front Oncol. 10:6161732021.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu H, Song M, Sun X, Zhang X, Miao H and
Wang Y: T-box transcription factor TBX1, targeted by
microRNA-6727-5p, inhibits cell growth and enhances cisplatin
chemosensitivity of cervical cancer cells through AKT and MAPK
pathways. Bioengineered. 12:565–577. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li S, Luo X, Sun M, Wang Y, Zhang Z, Jiang
J, Hu D, Zhang J, Wu Z, Wang Y, et al: Context-dependent T-BOX
transcription factor family: from biology to targeted therapy. Cell
Commun Signal. 22:3502024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chakraborty S, Sengupta A and Yutzey KE:
Tbx20 promotes cardiomyocyte proliferation and persistence of fetal
characteristics in adult mouse hearts. J Mol Cell Cardiol.
62:203–213. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Luo J, Chen JW, Zhou J, Han K, Li S, Duan
JL, Cao CH, Lin JL, Xie D and Wang FW: TBX20 inhibits colorectal
cancer tumorigenesis by impairing NHEJ-mediated DNA repair. Cancer
Sci. 113:2008–2021. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang Y, Aryal S, Geng X, Zhou X, Fast VG,
Zhang J, Lu R and Zhou Y: TBX20 improves contractility and
mitochondrial function during direct human cardiac reprogramming.
Circulation. 146:1518–1536. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang D, Shang X, Ji Q and Niu L:
Exploring genetic mapping and co-expression patterns to illuminate
significance of Tbx20 in cardiac biology. Transgenic Res. 34:52025.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Emile MH, Emile SH, El-Karef AA, Ebrahim
MA, Mohammed IE and Ibrahim DA: Association between the expression
of epithelial-mesenchymal transition (EMT)-related markers and
oncologic outcomes of colorectal cancer. Updates Surg.
76:2181–2191. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou J, Wang H, Che J, Xu L, Yang W, Li Y
and Zhou W: Silencing of microRNA-135b inhibits invasion,
migration, and stemness of CD24+CD44+ pancreatic cancer stem cells
through JADE-1-dependent AKT/mTOR pathway. Cancer Cell Int.
20:1342020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jin SX, Liu BN, Ji HJ, Wu JR, Li BL, Gao
XL, Li N, Zheng ZD and Du C: Serum cytokines and
creatinine/cystatin C ratio as prognostic biomarkers in advanced
cancer patients treated with anti-PD-1/PD-L1 therapy. Support Care
Cancer. 32:3702024.PubMed/NCBI
|
|
19
|
Huang S, Shu X, Ping J, Wu J, Wang J,
Shidal C, Guo X, Bauer JA, Long J, Shu XO, et al: TBX1 functions as
a putative oncogene of breast cancer through promoting cell cycle
progression. Carcinogenesis. 43:12–20. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Takatori N, Hotta K, Mochizuki Y, Satoh G,
Mitani Y, Satoh N, Satou Y and Takahashi H: T-box genes in the
ascidian Ciona intestinalis: Characterization of cDNAs and spatial
expression. Dev Dyn. 230:743–753. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li J, Huan J, Yang F, Chen H, Wang M and
Heng X: Identification and validation of a seizure-free-related
gene signature for predicting poor prognosis in lower-grade
gliomas. Int J Gen Med. 14:7399–7410. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang L, Li D, Du F, Huang H, Yuan C, Fu
J, Sun S, Tian T, Liu X, Sun H, et al: A panel of differentially
methylated regions enable prognosis prediction for colorectal
cancer. Genomics. 113:3285–3293. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Terry S, Savagner P, Ortiz-Cuaran S,
Mahjoubi L, Saintigny P, Thiery JP and Chouaib S: New insights into
the role of EMT in tumor immune escape. Mol Oncol. 11:824–846.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhao W, Luo Y, Li B and Zhang T:
Tumorigenic lung tumorospheres exhibit stem-like features with
significantly increased expression of CD133 and ABCG2. Mol Med Rep.
14:2598–2606. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Herreros-Pomares A, de-Maya-Girones JD,
Calabuig-Fariñas S, Lucas R, Martínez A, Pardo-Sánchez JM, Alonso
S, Blasco A, Guijarro R, Martorell M, et al: Lung tumorspheres
reveal cancer stem cell-like properties and a score with prognostic
impact in resected non-small-cell lung cancer. Cell Death Dis.
10:6602019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Loh CY, Chai JY, Tang TF, Wong WF, Sethi
G, Shanmugam MK, Chong PP and Looi CY: The E-Cadherin and
N-Cadherin switch in epithelial-to-mesenchymal transition:
Signaling, therapeutic implications, and challenges. Cells.
8:11182019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Berr AL, Wiese K, Dos Santos G, Koch CM,
Anekalla KR, Kidd M, Davis JM, Cheng Y, Hu YS and Ridge KM:
Vimentin is required for tumor progression and metastasis in a
mouse model of non-small cell lung cancer. Oncogene. 42:2074–2087.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chu X, Tian W, Ning J, Xiao G, Zhou Y,
Wang Z, Zhai Z, Tanzhu G, Yang J and Zhou R: Cancer stem cells:
advances in knowledge and implications for cancer therapy. Signal
Transduct Target Ther. 9:1702024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Alaei E, Farahani N, Orouei S,
Alimohammadi M, Daneshi S, Mousavi T, Mahmoodieh B, Taheriazam A,
Rahimzadeh P and Hashemi M: The clinicopathologic and prognostic
value of CD44 expression in patients with non-small cell lung
cancer: A systematic review and meta-analysis. Mol Cell Probes.
81:1020282025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li Z, Wang T, Liu J, Qi W, Lv Q, Xu Y and
Tian L: The mechanism and research progress of PD-1/PD-L1 on immune
escape of lung cancer. Transl Cancer Res. 14:6041–6051. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jing B, Wang T, Sun B, Xu J, Xu D, Liao Y,
Song H, Guo W, Li K, Hu M, et al: IL6/STAT3 signaling orchestrates
premetastatic niche formation and immunosuppressive traits in lung.
Cancer Res. 80:784–797. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kohli K, Pillarisetty VG and Kim TS: Key
chemokines direct migration of immune cells in solid tumors. Cancer
Gene Ther. 29:10–21. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hao D and Chen S: Targeting
tumor-associated macrophages in non-small cell lung cancer:
Mechanisms, prognosis, and therapeutic opportunities. Front
Immunol. 16:16795372025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li X, Pan L, Li W, Liu B, Xiao C, Chew V,
Zhang X, Long W, Ginhoux F, Loscalzo J, et al: Deciphering immune
predictors of immunotherapy response: A multiomics approach at the
pan-cancer level. Cell Rep Med. 6:1019922025. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yan D, Wang Y, Zhang L, Li Y, Gong Y, Liu
H, Ma X and Pang J: ARTN drives CD8+ T cell exhaustion via the
GFRα3-RET-PI3K/AKT Axis to promote TNBC progression. Cell Signal.
141:1123972026. View Article : Google Scholar : PubMed/NCBI
|