Introduction
Upper tract urothelial carcinoma (UTUC) is a
relatively rare subtype of urothelial carcinoma, accounting for
5–10% of all urothelial malignancies. In Western countries, the
estimated annual incidence is ~2 cases per 100,000 individuals
(1). In comparison to urothelial
carcinoma of the lower urinary tract, UTUC is frequently diagnosed
at more advanced stages, largely due to anatomical factors that
limit early detection and endoscopic surveillance. UTUC therefore
represents a substantial clinical challenge, particularly in
advanced disease. Patients with pathological stage T2 or T3
(pT2/pT3) tumors exhibit a 5-year overall survival (OS) rate of
<50%, which declines to <10% in those with pT4 disease
(2,3). Radical nephroureterectomy (RNU), the
standard treatment for localized UTUC, remains essential for
oncological control; however, this is associated with significant
deterioration of renal function, further complicating subsequent
management.
Advances in cancer biology have underscored the
critical role of cancer stem cells (CSCs) in tumor initiation,
progression, metastasis and therapeutic resistance (4). CSCs possess self-renewal capacity and
multipotency, contributing to intratumoral heterogeneity and
treatment failure through specific mechanisms, such as enhanced DNA
repair, resistance to apoptosis and activation of drug efflux
pathways. Sex determining region-Y-related high mobility group box
2 (SOX2), a transcription factor central to stemness maintenance,
is widely recognized as a marker of CSCs in various malignancies
(5). SOX2 regulates
pluripotency-associated genes and self-renewal signaling pathways,
and plays a pivotal role in embryonic development and stem cell
biology. Together with other CSC markers, including aldehyde
dehydrogenase 1 (ALDH1) (6),
elevated SOX2 expression has been associated with aggressive tumor
phenotypes and unfavorable clinical outcomes across multiple cancer
types (7).
In UTUC, the results of preliminary studies
suggested an association between CSC markers, including SOX2 and
ALDH1, and adverse patient outcomes (8,9).
However, these investigations were limited by small sample sizes
and heterogeneous methodologies, resulting in inconsistent
conclusions regarding the prognostic significance of individual CSC
markers. The clinicopathological characteristics of CSC-positive
UTUC, their molecular subtype distribution and the therapeutic
implications of CSC-driven disease are yet to be fully elucidated.
In addition, potential differences in response to systemic
therapies, including chemotherapy and immunotherapy, between
CSC-positive and CSC-negative UTUC have not been systematically
examined. This knowledge gap limits risk stratification and the
development of optimized treatment strategies for patients with
biologically aggressive disease.
Given these limitations, the present study
systematically evaluated SOX2 expression in UTUC and examined its
association with clinicopathological features. The present study
further assessed whether SOX2 expression functions as an
independent prognostic biomarker beyond established
clinicopathological parameters (10,11).
In addition, the expression of programmed cell death ligand 1
(PD-L1) and antibody-drug conjugate (ADC) targets, including
Nectin-4 and trophoblast cell surface antigen 2 (TROP-2), was
analyzed to determine whether SOX2-positive UTUC represents a
distinct therapeutic subgroup that may benefit from tailored
systemic approaches. These findings may provide clinically relevant
evidence to support improved risk stratification and personalized
management in UTUC.
Materials and methods
Patients
The medical records of 87 consecutive patients with
histologically confirmed UTUC who underwent RNU at Toyama
University Hospital between January 2012 and December 2021 were
retrospectively reviewed. Of these patients, 69 were male and 18
were female, with a median age of 74 years [interquartile range
(IQR), 69–78; range, 51–87 years]. The present study was approved
by the Institutional Review Board of the University of Toyama
(approval no. R2019113). The requirement for informed consent was
waived due to the retrospective design of the study and the use of
anonymized data. Tumor staging was determined according to the 2017
Union for International Cancer Control (UICC) TNM classification
system, and tumor grading was performed in accordance with the
World Health Organization (WHO) 2016 grading criteria.
Immunohistochemistry and scoring
Formalin-fixed paraffin-embedded tumor sections (4
µm thick) underwent heat-induced epitope retrieval in Tris-EDTA
buffer (pH, 9.0) in an autoclave prior to immunostaining with
monoclonal antibodies against SOX2 (NBP2-29623, 1:200; Novus
Biologicals, Ltd.; Bio-Techne) and ALDH1 (611194, 1:500; BD
Transduction Laboratories; BD Biosciences). The chromogen was
applied using Dako Envision FLEX+ (Agilent Technologies, Inc.),
followed by counterstaining with hematoxylin, rinsing, dehydration
and placing on a cover slip. Negative controls were prepared via
substituting the primary antibody with dilution buffer. The
criteria for determining positive and negative staining were based
on previously published studies (12,13).
For SOX2, a 10% cut-off defined positive and negative specimens
(12). ALDH1 positivity was defined
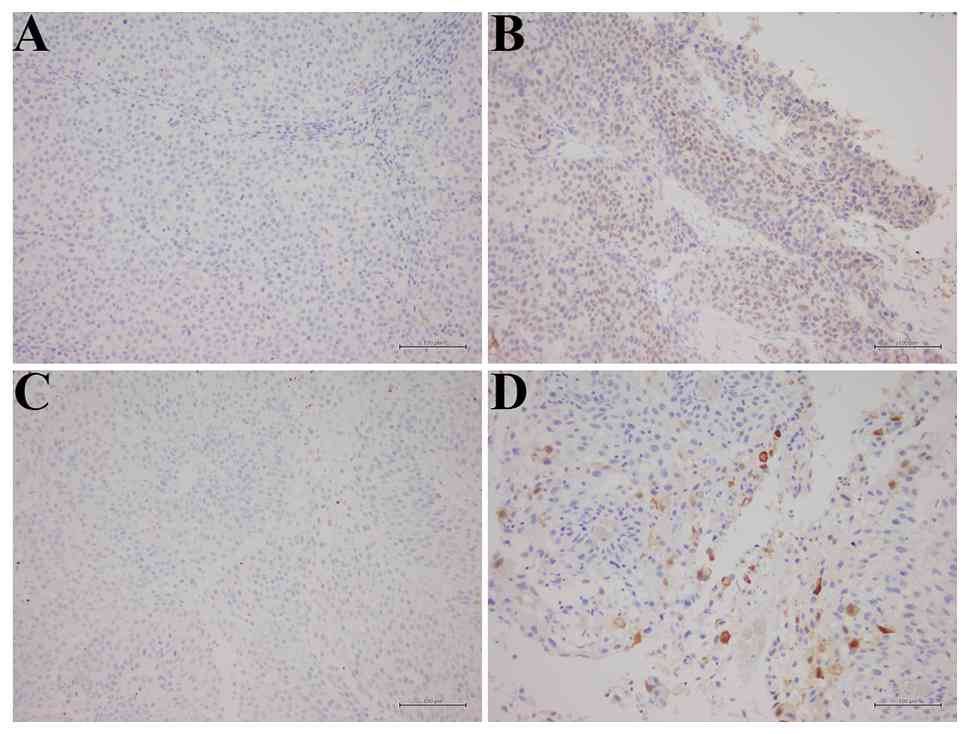
as staining in >1% of tumor cells (13). Representative immunohistochemical
staining results for SOX2 and ALDH1 are displayed in Fig. 1.
Molecular subtypes were evaluated using antibodies
against CK5/6 (418081, 1:1; Nichirei Biosciences, Inc.), CK20
(413491, 1:1; Nichirei Biosciences, Inc.), GATA3 (418201, 1:1;
Nichirei Biosciences, Inc.), UPK2 (418121, 1:1; Nichirei
Biosciences, Inc.) and CK14 (NCL-L-LL002, Leica, 1:50; Leica
Biosystems; Fig. 2A) (14,15).
Immunohistochemical scoring for all markers was based on the
percentage of positive tumor cells. Molecular subtypes were
hierarchically clustered using the average scores of CK20, GATA3
and UPK2 for the luminal type, and CK5/6 and CK14 for the basal
type. Immunohistochemical analysis was also performed to assess
PD-L1 (ab237726, 1:1,000; Abcam) and the ADC targets, Nectin-4
(ab192033, 1:4,000; Abcam) and TROP-2 (SC-376181, 1:1,000; Santa
Cruz Biotechnology). A cut-off value of 1% was applied for PD-L1
expression in tumor cell membranes (Fig. 2B) (16). Nectin-4 expression was quantified
using a H-score defined as the sum of staining intensity (0–3)
multiplied by the percentage (0–100) of tumor cells at each
intensity level. Specimens were classified as negative (H-score,
0–14) or positive (H-score, ≥15; Fig.
2B) (17). TROP-2
immunoreactivity localized to the cell membrane in ≥10% of tumor
cells was considered positive (Fig.
2B) (18).
 | Figure 2.Immunohistochemical staining and
molecular marker expression in UTUC. (A) Immunohistochemical
staining patterns for molecular subtype markers (CK5/6, CK14,
Uroplakin 2, CK20 and GATA3). Scale bar, 200 µm. (B)
Immunohistochemical expression of PD-L1 and ADC targets (Nectin-4
and TROP-2) in UTUC tissue. (a) Negative PD-L1 expression. (b)
Positive PD-L1 expression. Scale bar, 100 µm. (c) Negative Nectin-4
expression (intensity, 0). (d) Weak Nectin-4 expression (intensity,
1+). (e) Moderate Nectin-4 expression (intensity, 2+). (f) Negative
TROP-2 expression. (g) Positive TROP-2 expression. Scale bar, 200
µm. UTUC, upper tract urothelial carcinoma; PD-L1, programmed cell
death ligand 1; ADC, antibody-drug conjugate; TROP-2, trophoblast
cell surface antigen 2; CK, cytokeratin. |
Statistical analysis
OS, CSS and RFS were defined as the duration from
RNU to death from any cause, cancer-specific death, or recurrence,
respectively. Fisher's exact test was used to compare categorical
variables between subgroups. The Mann-Whitney U test was applied to
compare continuous variables. Survival curves were generated using
the Kaplan-Meier method and compared using the log-rank test. The
Cox proportional hazards model was used for univariate and
multivariate analyses to identify mortality risk factors. Variables
with P<0.05 in univariate analyses were entered into the
multivariate model. All statistical tests were two-sided, and
P<0.05 was considered to indicate a statistically significant
difference. Statistical analyses were performed using SPSS
(version, 27.0; IBM Corp.).
Results
Patient characteristics and positivity
of cancer stem cell markers
Table I summarizes
the characteristics of all 87 patients included in the present
study. The median follow-up period was 39.3 months (interquartile
range, 17.0–70.5 months). SOX2 and ALDH1 expression were positive
in 24% (21/87) and 37% (32/87) of patients, respectively.
 | Table I.Patient characteristics (N=87). |
Table I.
Patient characteristics (N=87).
| Characteristic | n (%) or median
(IQR) |
|---|
| Median age,
years | 74 (69–78) |
| Sex |
|
| Male | 69 (79) |
|
Female | 18 (21) |
| ECOG PS |
|
| 0/1 | 73 (84) |
| 2-4 | 14 (16) |
| Renal function, eGFR
(ml/min/1.73 m2) |
|
| ≥60 | 34 (39) |
|
45-59 | 22 (25) |
|
<45 | 31 (36) |
| Primary site |
|
| Renal
pelvis | 38 (44) |
|
Ureter | 44 (51) |
| Both | 5 (6) |
| Side |
|
|
Right | 49 (56) |
|
Left | 36 (41) |
|
Bilateral | 2 (2) |
| Hydronephrosis
grade |
|
|
0/1 | 33 (38) |
|
2-4 | 54 (62) |
| Pathological T
stage |
|
|
pTis/pTa/pT1 | 34 (39) |
|
pT2 | 10 (12) |
|
pT3 | 41 (47) |
|
pT4 | 2 (2) |
| Pathological N
stage |
|
|
pN- | 23 (26) |
|
pN+ | 8 (9) |
|
pNx | 56 (64) |
| Tumor grade |
|
| Low
grade | 16 (18) |
| High
grade | 66 (76) |
|
Unknown | 5 (6) |
| Lymphovascular
invasion |
|
|
Negative | 46 (53) |
|
Positive | 29 (33) |
|
Unknown | 12 (14) |
| Tumor
histology |
|
| Pure
UC | 82 (94) |
| UC with
variant histology | 5 (6) |
|
Squamous differentiation | 3 (3) |
|
Glandular differentiation | 1 (1) |
|
Sarcomatoid variant | 1 (1) |
| History of bladder
cancer |
|
| No | 74 (85) |
|
Yes | 13 (15) |
| Perioperative
chemotherapy |
|
| Neoadjuvant
chemotherapy |
|
| No | 83 (95) |
|
Yes | 4 (5) |
| GEM +
CDDP | 3 (3) |
| GEM +
PTX | 1 (1) |
| Adjuvant
chemotherapy |
|
| No | 69 (79) |
|
Yes | 18 (21) |
| GEM +
CDDP | 16 (18) |
| GEM +
CBDCA | 2 (2) |
Association between SOX2 expression
and clinicopatholo-gical characteristics
Clinicopathological characteristics were compared
between patients with SOX2-positive tumors and those with
SOX2-negative tumors. No significant associations were identified
between SOX2 expression and clinicopathological factors, with the
exception of hydronephrosis. Moreover, no significant correlations
were observed between SOX2 expression and molecular subtypes, PD-L1
expression or ADC targets, including Nectin-4 and TROP-2 (Table II).
 | Table II.Associations between SOX2 expression
and clinicopathological characteristics. |
Table II.
Associations between SOX2 expression
and clinicopathological characteristics.
| Characteristic | SOX2 positive
(n=21) | SOX2 negative
(n=66) | P-value |
|---|
| Median age,
years | 74 (65–77) | 75 (70–78) | 0.188 |
| Sex |
|
|
|
|
Male | 18 (86) | 51 (77) | 0.543 |
|
Female | 3 (14) | 15 (23) |
|
| ECOG PS |
|
|
|
|
0/1 | 15 (71) | 58 (88) | 0.078 |
|
2-4 | 6 (29) | 8 (12) |
|
| eGFR (ml/min/1.73
m2) |
|
|
|
|
≥45 | 11 (52) | 45 (68) | 0.202 |
|
<45 | 10 (48) | 21 (32) |
|
| Primary site |
|
|
|
| Renal
pelvis | 7 (33) | 31 (47) | 0.490 |
|
Ureter | 13 (62) | 31 (47) |
|
|
Both | 1 (5) | 4 (6) |
|
| Bilateral
disease |
|
|
|
|
Unilateral | 21 (100) | 64 (97) | 1.000 |
|
Bilateral | 0 (0) | 2 (3) |
|
| Hydronephrosis
grade |
|
|
|
|
0/1 | 3 (14) | 30 (45) | 0.011 |
|
2-4 | 18 (86) | 36 (55) |
|
| pT stage |
|
|
|
|
pTis/pTa/pT1 | 8 (38) | 26 (39) | 0.574 |
|
pT2 | 1 (5) | 9 (14) |
|
|
pT3 | 11 (52) | 30 (45) |
|
|
pT4 | 1 (5) | 1 (2) |
|
| pN stage |
|
|
|
|
pN- | 7 (33) | 16 (24) | 0.388 |
|
pN+ | 3 (14) | 5 (8) |
|
|
pNx | 11 (52) | 45 (68) |
|
| Tumor grade |
|
|
|
| Low
grade | 4 (19) | 12 (18) | 0.434 |
| High
grade | 13 (62) | 53 (80) |
|
|
Unknown | 4 (19) | 1 (2) |
|
| Lymphovascular
invasion |
|
|
|
|
Negative | 11 (52) | 35 (53) | 0.982 |
|
Positive | 7 (33) | 22 (33) |
|
|
Unknown | 3 (14) | 9 (14) |
|
| Tumor
histology |
|
|
|
| Pure
UC | 20 (95) | 62 (94) | 0.824 |
| UC with
variant histology | 1 (5) | 4 (6) |
|
|
Squamous
differentiation | 1 (5) | 2 (3) |
|
|
Glandular
differentiation | 0 | 1 (2) |
|
|
Sarcomatoid
variant | 0 | 1 (2) |
|
| Molecular
subtype |
|
|
|
|
Luminal | 14 (67) | 52 (79) | 0.258 |
|
Basal | 7 (33) | 14 (21) |
|
| PD-L1 |
|
|
|
|
Positive | 6 (29) | 22 (33) | 0.792 |
|
Negative | 15 (71) | 44 (67) |
|
| ADC targets |
|
|
|
|
Nectin-4 |
|
|
|
|
Positive | 17 (81) | 56 (85) | 0.736 |
|
Negative | 4 (19) | 10 (15) |
|
| TROP-2 |
|
|
|
|
Positive | 17 (81) | 52 (79) | 1.000 |
|
Negative | 4 (19) | 14 (21) |
|
Survival outcomes and prognostic
significance
In the overall cohort analysis, patients with
SOX2-positive tumors exhibited significantly worse clinical
outcomes than those with SOX2-negative tumors. SOX2 expression was
associated with significantly shorter OS, CSS and RFS (P=0.004,
P=0.005 and P=0.011, respectively; Fig.
3A, C and E). The 5-year OS, CSS and RFS rates in the
SOX2-negative and SOX2-positive groups were 65 vs. 38%, 69 vs. 43%
and 42 vs. 14%, respectively. By contrast, no significant
differences were observed between the ALDH1-positive and
ALDH1-negative groups with respect to OS, CSS or RFS (Fig. 3B, D and F).
Univariate and multivariate
analyses
The univariate analysis identified several factors
associated with poor prognosis, including female sex,
hydronephrosis grade, advanced pathological T stage, positive
pathological N stage, lymphovascular invasion and SOX2 expression
(Table III). Results of the
multivariate analysis demonstrated that SOX2 expression acted as an
independent prognostic indicator for CSS (P=0.001), demonstrating
its prognostic value beyond conventional clinicopathological
parameters (Table III).
 | Table III.Prognostic factors for
cancer-specific survival in univariate and multivariate
analyses. |
Table III.
Prognostic factors for
cancer-specific survival in univariate and multivariate
analyses.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Factor | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, ≥75 vs.
<75 | 0.855
(0.399–1.829) | 0.855 | N/A | N/A |
| Sex, male vs.
female | 0.305
(0.141–0.661) | 0.003a | 0.286
(0.093–0.881) | 0.029a |
| ECOG PS, 2–4 vs.
0/1 | 0.709
(0.459–1.095) | 0.143 | N/A | N/A |
| Reduced renal
function, eGFR <45 vs. ≥45 | 0.733
(0.501–1.071) | 0.121 | N/A | N/A |
| Hydronephrosis
grade, 2–4 vs. 0/1 | 2.868
(1.156–7.119) | 0.014a | 1.117
(0.648–1.924) | 0.691 |
| pT stage, ≥ pT2 vs.
≤ pT1 | 3.686
(1.390–9.772) | 0.009a | 1.777
(0.459–7.019) | 0.412 |
| pN stage, pN+ vs.
pN- | 5.632
(2.215–14.316) |
<0.001a | 4.591
(1.510–13.957) | 0.007a |
| Tumor grade, high
vs. low | 1.590
(0.546–4.624) | 0.370 | N/A | N/A |
| LVI, positive vs.
negative | 4.509
(1.921–10.580) |
<0.001a | 2.173
(0.754–6.265) | 0.151 |
| History of bladder
cancer, yes vs. no | 0.724
(0.218–2.407) | 0.599 | N/A | N/A |
| SOX2, positive vs.
negative | 2.850
(1.321–6.148) | 0.008a | 4.820
(1.842–12.614) | 0.001a |
| ALDH1, positive vs.
negative | 1.812
(0.851–3.859) | 0.123 | N/A | N/A |
| Molecular subtype,
basal vs. luminal | 1.663
(0.727–3.801) | 0.228 | N/A | N/A |
| PD-L1, positive vs.
negative | 1.841
(0.853–3.974) | 0.120 | N/A | N/A |
Treatment response analysis
Impact of SOX2 expression on treatment response was
analyzed via comparing the outcomes of patients who received or did
not receive platinum-based chemotherapy (neoadjuvant, adjuvant or
both; Fig. 4). Among patients who
did not receive neoadjuvant or adjuvant chemotherapy, those with
SOX2-positive tumors consistently showed poorer outcomes than those
with SOX2-negative tumors. The 5-year CSS rate was 73% in patients
with SOX2-negative tumors, vs. 49% in those with SOX2-positive
tumors (Fig. 4D). The 5-year RFS
rates were 50 and 19%, respectively (Fig. 4F).
Discussion
The present study provides a comprehensive analysis
demonstrating that SOX2 expression is a strong and independent
prognostic biomarker in patients with UTUC undergoing radical
nephroureterectomy. A clinically significant difference in 5-year
CSS was observed (43% for SOX2-positive vs. 69% for SOX2-negative),
underscoring the importance of identifying this highly aggressive
patient subset. SOX2 expression remained an independent prognostic
factor in multivariate analyses, irrespective of conventional
clinicopathological variables, such as tumor stage and grade. This
independence suggests that SOX2 may define a biologically distinct
disease entity characterized by high stemness and intrinsic
malignancy, independent of morphological tumor characteristics.
In the present cohort, the prevalence of SOX2
expression (24%) was consistent with previous reports in urothelial
carcinomas, where SOX2 positivity ranged from 15 to 30% (8,9). The
26% absolute difference in CSS may represent a clinically
meaningful prognostic distinction with implications for treatment
planning and patient counseling. Multivariate analyses (P=0.001)
confirmed that SOX2 may provide prognostic data beyond established
parameters, such as tumor stage, grade and lymphovascular invasion
(19). Notably, SOX2 expression did
not correlate with traditional prognostic variables, further
supporting its role in identifying a biologically distinct,
intrinsically aggressive subset of UTUC. The function of SOX2 as a
master regulator of stemness and pluripotency provides biological
plausibility for its association with poor prognosis (5).
The unfavorable outcomes observed in patients with
SOX2-positive UTUC emphasized the requirement for novel therapeutic
strategies targeting this high-risk population. CSCs, characterized
by self-renewal and multilineage differentiation capacities,
frequently exhibit resistance to conventional anticancer agents and
radiotherapy (20). SOX2 positivity
may reflect enhanced tumor-initiating capacity, increased
resistance to systemic therapies and a greater metastatic potential
(21).
Recent advances in molecular classification have
stratified muscle-invasive urothelial carcinoma (MIUC) into
distinct molecular subtypes with differing prognoses (22). Emerging evidence indicated that SOX2
regulates key genes within the basal/squamous (Ba/Sq) subtype of
MIUC and modulates chemotherapeutic response (23). Results of a previous study
demonstrated that SOX2 depletion reduced Ba/Sq markers, increased
luminal markers and enhanced cisplatin sensitivity in MIUC
(23). Although UTUC and bladder
urothelial carcinoma differ in epidemiological and
clinicopathological characteristics (24), therapeutic strategies targeting SOX2
may also be applicable to UTUC through modulating molecular subtype
features and improving chemotherapy responsiveness. These findings
raise the possibility that neoadjuvant chemotherapy (NAC),
historically lacking clearly established efficacy in UTUC (25), may confer benefit when combined with
SOX2-targeted strategies. If SOX2 inhibition enhances cisplatin
efficacy, as demonstrated in bladder cancer models, combination
approaches may improve outcomes in patients with SOX2-positive
UTUC.
To the best of our knowledge, the present study is
the first to evaluate the association between SOX2 expression and
therapeutic biomarkers, including PD-L1, and ADC targets; namely,
Nectin-4 and TROP-2, in a UTUC cohort undergoing RNU. Comparable
expression levels of PD-L1, Nectin-4 and TROP-2 between
SOX2-positive and SOX2-negative tumors suggest that immune
checkpoint inhibitors (ICIs) and ADC therapies, including
enfortumab vedotin and sacituzumab govitecan, may remain effective
treatment options in this high-risk subgroup.
Notably, the present study exhibits limitations. The
retrospective, single-institution design and relatively small
sample size may limit generalizability, and validation in larger,
multi-center cohorts is required. Although the prognostic
significance of SOX2 was demonstrated, the molecular mechanisms
underlying the adverse outcomes in SOX2-positive patients remain to
be fully elucidated. Treatment response analysis was limited by
chemotherapy heterogeneity and retrospective treatment selection.
Furthermore, we were unable to assess longitudinal changes in SOX2
expression following NAC or at the time of recurrence. Although
four patients received NAC, pre-treatment biopsy specimens were
available for only one case [the remaining three were diagnosed via
urinary cytology (n=2) or imaging (n=1)]. Consequently, we were
unable to evaluate the impact of NAC on SOX2 expression in the
current cohort. This remains an important subject for future
investigations. In addition, the association between SOX2
expression and specific molecular subtypes of UTUC, as well as the
feasibility of SOX2-targeted therapies, should be further explored
in prospective studies. While the present study was limited to an
immunohistochemistry-based classification for molecular subtype
assessment, previously proposed molecular classifications based on
whole-exome sequencing require further investigation to clarify
their association with SOX2 expression (26,27).
Although the present study included a
single-institution retrospective analysis, a limitation common in
studies of rare malignancies such as UTUC, the statistical
robustness of SOX2 as an independent prognostic factor supports its
clinical relevance. Moreover, the present study provided the first
evidence evaluating the association between SOX2 expression and
PD-L1 and ADC targets. The comparable expression of these markers
in SOX2-positive and SOX2-negative tumors suggested that ICIs and
ADC therapies, such as enfortumab vedotin or sacituzumab govitecan,
may represent viable therapeutic options for this high-risk
subgroup, offering an immediate and clinically meaningful treatment
opportunity.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HY and HK contributed to the study design and
formulation of the framework. HY prepared the initial draft of the
manuscript. YI and NN conducted the data analysis of molecular
subtype. HY, YI and NN confirm the authenticity of all the raw
data. HK supervised the study. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Review Board of The University of Toyama (approval no. R2019113).
The requirement for written informed consent was waived due to the
retrospective design of the study. Consent was obtained via an
opt-out method approved by the ethics committee, with study
information provided publicly to allow patients to decline
participation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
UTUC
|
upper tract urothelial carcinoma
|
|
RNU
|
radical nephroureterectomy
|
|
CSC
|
cancer stem cell
|
|
SOX2
|
sex determining region-Y-related high
mobility group box 2
|
|
ALDH1
|
aldehyde dehydrogenase 1
|
|
PD-L1
|
programmed cell death ligand 1
|
|
ADC
|
antibody-drug conjugate
|
|
TROP-2
|
trophoblast cell surface antigen 2
|
|
CK
|
cytokeratin
|
|
UPK2
|
uroplakin 2
|
|
H-score
|
histochemical score
|
|
OS
|
overall survival
|
|
CSS
|
cancer-specific survival
|
|
RFS
|
recurrence-free survival
|
|
MIUC
|
muscle-invasive urothelial
carcinoma
|
|
Ba/Sq
|
basal/squamous
|
|
NAC
|
neoadjuvant chemotherapy
|
|
ICIs
|
immune checkpoint inhibitors
|
References
|
1
|
Gontero P, Birtle A, Compérat E, Escrig
JL, Liedberg F, Mariappan P, Masson-Lecomte A, Mostafid AH, Rhijn
BWG, Seisen T, et al: European association of urology guidelines on
non-muscle-invasive bladder cancer (TaT1 and carcinoma in situ)-a
summary of the 2024 guidelines update. Eur Urol. 86:531–549. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Abouassaly R, Alibhai SMH, Fleshner NE,
Timilshina N and Finelli A: Troubling outcomes from
population-level analysis of surgery for upper tract urothelial
carcinoma. Urology. 76:895–901. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jeldres C, Sun M, Isbarn H, Lughezzani G,
Budäus L, Alasker A, Shariat SF, Lattouf JB, Widmer H, Pharand D,
et al: A population-based assessment of perioperative mortality
after nephroureterectomy for upper-tract urothelial carcinoma.
Urology. 75:315–320. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang L, Shi P, Zhao G, Xu J, Peng W, Zhang
J, Zhang G, Wang X, Dong Z, Chenet F, et al: Targeting cancer stem
cell pathways for cancer therapy. Signal Transduct Target Ther.
5:82020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mirzaei S, Paskeh MDA, Entezari M,
Mirmazloomib S, Hassanpoorb A, Aboutalebib M, Rezaeib S, Hejazib
ES, Kakavandb A, Heidari H, et al: SOX2 function in cancers:
Association with growth, invasion, stemness and therapy response.
Biomed Pharmacother. 156:1138602022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ginestier C, Hur MH, Jauffret EC, Monville
F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG, Liu S, et
al: ALDH1 is a marker of normal and malignant human mammary stem
cells and a predictor of poor clinical outcome. Cell Stem Cell.
1:555–567. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wuebben EL and Rizzino A: The dark side of
SOX2: Cancer-a comprehensive overview. Oncotarget. 8:44917–44943.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kitamura H, Torigoe T, Hirohashi Y,
Asanuma H, Inoue R, Nishida S, Tanaka T, Fukuta F, Masumori N, Sato
N and Tsukamoto T: Prognostic impact of the expression of ALDH1 and
SOX2 in urothelial cancer of the upper urinary tract. Mod Pathol.
26:117–124. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bao Z, Zhan Y, He S, Li Y, Guan B, He Q,
Yang X, Li X, Fang D and Zhou L: Increased expression of SOX2
predicts a poor prognosis and promotes malignant phenotypes in
upper tract urothelial carcinoma. Cancer Manag Res. 11:9095–9106.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Leow JJ, Orsola A, Chang SL and Bellmunt
J: A contemporary review of management and prognostic factors of
upper tract urothelial carcinoma. Cancer Treat Rev. 41:310–319.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ito Y, Kikuchi E, Tanaka N, Miyajima A,
Mikami S, Jinzaki M and Oya M: Preoperative hydronephrosis grade
independently predicts worse pathological outcomes in patients
undergoing nephroureterectomy for upper tract urothelial carcinoma.
J Urol. 185:1621–1626. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nakatsugawa M, Takahashi A, Hirohashi Y,
Torigoe T, Inoda S, Murase M, Asanuma H, Tamura Y, Morita R,
Michifuri Y, et al: SOX2 is overexpressed in stem-like cells of
human lung adenocarcinoma and augments the tumorigenicity. Lab
Invest. 91:1796–1804. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nogami T, Shien T, Tanaka T, Nishiyama K,
Mizoo T, Iwamoto T, Ikeda H, Taira N, Doihara H and Miyoshi S:
Expression of ALDH1 in axillary lymph node metastases is a
prognostic factor of poor clinical outcome in breast cancer
patients with 1–3 lymph node metastases. Breast Cancer. 21:58–65.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Robertson AG, Kim J, Ahmadie HAI, Bellmunt
J, Guo G, Cherniack AD, Hinoue T, Laird PW, Hoadley KA, Akbani R,
et al: Comprehensive molecular characterization of muscle-invasive
bladder cancer. Cell. 171:540–556. e252017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Guo CC, Bondaruk J, Yao H, Wang Z, Zhang
L, Lee S, Lee JG, Cogdell D, Zhang M, Yang G, et al: Assessment of
luminal and basal phenotypes in bladder cancer. Sci Rep.
10:97432020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pichler R, Fritz J, Lachner F, Sprung S,
Brunner A, Horninger W, Loidl W, Pircher A and Heidegger I:
Prognostic value of testing PD-L1 expression after radical
cystectomy in high-risk patients. Clin Genitourin Cancer.
16:e1015–e1024. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Challita-Eid PM, Satpayev D, Yang P, An Z,
Morrison K, Shostak Y, Raitano A, Nadell R, Liu W, Lortie DR, et
al: Enfortumab vedotin antibody-drug conjugate targeting nectin-4
is a highly potent therapeutic agent in multiple preclinical cancer
models. Cancer Res. 76:3003–3013. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abbas M, Heitplatz B, Bernemann C,
Boegemann M, Trautmann M, Schrader A, Wardelmann E and Schlack K:
Immunohistochemical expression of TROP-2 (TACSTD2) on the
urothelial carcinoma of the urinary bladder and other types of
cancer. Oncol Lett. 26:5272023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kim DS, Lee YH, Cho KS, Cho NH, Chung BH
and Hong SJ: Lymphovascular invasion and pT stage are prognostic
factors in patients treated with radical nephroureterectomy for
localized upper urinary tract transitional cell carcinoma. Urology.
75:328–332. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wicha MS, Liu S and Dontu G: Cancer stem
cells: An old idea-a paradigm shift. Cancer Res. 66:1883–1890.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Javaeed A and Ghauri SK: Metastatic
potential and prognostic significance of SOX2: A meta-analysis.
World J Clin Oncol. 10:234–246. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kamoun A, Reyniès A, Allory Y, Sjödahl G,
Robertson AG, Seiler R, Hoadley KA, Groeneveld CS, Ahmadie HA, Choi
W, et al: A consensus molecular classification of muscle-invasive
bladder cancer. Eur Urol. 77:420–433. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nargis N, Lind A, Sczepanski A, Herndon R,
Smiley O, Somji S, Sens DA and Mehus AA: SOX2 regulates growth,
expression of basal/luminal markers, and chemotherapy response in
urothelial carcinoma. Cells. 14:9492025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sfakianos JP, Cha EK, Iyer G, Scott SN,
Zabor EC, Shah RH, Ren Q, Bagrodia A, Kim PH, Hakimi AA, et al:
Genomic characterization of upper tract urothelial carcinoma. Eur
Urol. 68:970–977. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rouprêt M, Seisen T, Birtle AJ, Capoun O,
Compe´rat EM, Escrig JLD, Andersson IG, Liedberg F, Mariappan P,
Mostafid AH, et al: European association of urology guidelines on
upper urinary tract urothelial carcinoma: 2023 update. Eur Urol.
84:49–64. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Moss TJ, Qi Y, Xi L, Peng B, Kim TB,
Ezzedine NE, Mosqueda ME, Guo CC, Czerniak BA, Ittmann M, et al:
Comprehensive genomic characterization of upper tract urothelial
carcinoma. Eur Urol. 72:641–649. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fujii Y, Sato Y, Suzuki H, Kakiuchi N,
Yoshizato T, Lenis AT, Maekawa S, Yokoyama A, Takeuchi Y, Inoue Y,
et al: Molecular classification and diagnostics of upper urinary
tract urothelial carcinoma. Cancer Cell. 39:793–809. 2021.
View Article : Google Scholar : PubMed/NCBI
|