Introduction
The management of locally advanced rectal cancer
(LARC) has long been anchored by total mesorectal excision (TME)
following neoadjuvant chemoradiotherapy (CRT) (1,2). This
approach successfully curtailed local recurrence rates but at the
cost of marked functional morbidity and permanent stomas in up to
30% of patients (3,4). The 21st century ushered in the era of
total neoadjuvant therapy (TNT) (5,6), which
not only addresses micrometastases earlier but also drives
pathological complete response (pCR) rates to 25–30% (7), as demonstrated in trials such as OPRA
(8). This breakthrough prompted a
critical question; for patients achieving a clinical complete
response (cCR) after TNT, is mandatory radical surgery always
necessary, or could it represent overtreatment in some cases?
Pioneered by Habr-Gama et al (9), the watch-and-wait (WaW) strategy
challenges the surgical dogma, offering a pathway to preserve
rectal function and potentially enhance quality of life (QoL)
(10). However, this path is
complicated by the central conundrum of clinical assessment
ambiguity and tumor biological heterogeneity. The traditional cCR
evaluation toolkit, digital rectal exam, magnetic resonance imaging
(MRI) and endoscopy, carries a non-negligible false-negative rate,
while the long-term fate of clinically ‘vanished’ tumors remains
only partially charted (11).
In the current treatment landscape, LARC management
typically integrates neoadjuvant CRT or TNT, followed by TME as the
standard surgical component. In parallel, emerging modalities such
as immunotherapy (particularly for mismatch repair-deficient
tumors) and targeted agents are reshaping therapeutic algorithms
(12–16). The present review aimed to
critically deconstruct the ‘pros’ and ‘risk’ dimensions of the WaW
strategy by synthesizing the latest evidence, evaluating emerging
biomarkers such as circulating tumor DNA (ctDNA) and proposing
data-driven considerations. The current review presents that
technology-facilitated precision in patient selection and dynamic
risk monitoring is pivotal to mitigating risks and maximizing
benefits (17,18). Therefore, the present article
provides a critical analysis of the WaW strategy by interrogating
key tensions: The balance between organ preservation and
oncological safety; the limitations and evolving technologies in
clinical response assessment; the management and implications of
local regrowth; and the practical challenges of implementing
rigorous surveillance protocols.
Methods for the literature search
Given the narrative nature of the present review, a
systematic literature search was performed to ensure transparency.
The databases PubMed (https://pubmed.ncbi.nlm.nih.gov/), Web of Science
(https://www.webofscience.com) and the
Cochrane Library (https://www.cochranelibrary.com/) were searched for
articles published between January 2000 and December 2025 using the
following terms: ‘Rectal cancer’, ‘watch-and-wait’, ‘total
neoadjuvant therapy’, ‘clinical complete response’, ‘organ
preservation’, ‘ctDNA’ and ‘local regrowth’. Included manuscripts
were original articles, systematic reviews, randomized controlled
trials (RCTs) and large registry studies reporting oncological or
QoL outcomes in English. The reference lists of included articles
were also screened for additional relevant studies. This approach,
while not systematic in the strictest sense, provides a
reproducible framework for the evidence synthesis presented.
Evolution of evidence for organ
preservation: From clinical trials to real-world practice
The journey toward mainstream acceptance of WaW is
underpinned by a sequential body of evidence, evolving from
hypothesis-generating cohort studies to RCTs and large-scale
real-world registries.
Pioneering observational studies and
the proof of concept
The foundational hypothesis was posited by Habr-Gama
et al (9), whose 2004
prospective observational study of 265 patients (71 achieving cCR
managed non-operatively vs. 22 pCR patients undergoing surgery)
demonstrated, with a mean follow-up of ~4 years, that patients with
a sustained cCR after CRT could achieve excellent long term
outcomes without immediate surgery (5-year OS, 100 vs. 88%; 5-year
DFS, 92 vs. 83%), with salvage therapy effectively managing the
minority who experienced local regrowth (9,19).
This work challenged the surgical imperative and established the
essential framework for patient selection and intensive
surveillance.
Role of RCTs: Establishing comparative
efficacy
While RCTs directly comparing WaW to standard TME in
patients with cCR are ethically unacceptable (randomizing cCR
patients to radical surgery without proven benefit) and practically
infeasible (requiring thousands of patients due to >85% survival
in both arms), several pivotal trials have provided indirect but
powerful evidence supporting organ preservation.
GRECCAR 2 trial
This phase III RCT directly tested a more
conservative surgical approach (local excision vs. TME) in good
responders after CRT. Although its primary composite endpoint
(death, recurrence, major morbidity) was not met (56 vs. 48%;
P=0.43), it highlighted the importance of accurate response
assessment and showed that organ-preserving strategies could be
feasible in a subset of patients, setting the stage for
non-operative management (4).
Trials of TNT
Modern RCTs of TNT regimens, such as RAPIDO
(20), PRODIGE 23 (21) and OPRA (8), were designed to improve systemic
disease control with mandated surgery for all participants
(6,7). However, a critical secondary finding
was the marked increase in pCR rates (28–30%) compared with
standard chemoradiotherapy (6–8). This
phenomenon is of notable importance for organ preservation, as it
substantially enlarges the population of patients who may achieve
cCR after TNT and thus become potential candidates for a
non-operative WaW strategy. The OPRA trial specifically used organ
preservation as a primary endpoint, demonstrating that a TNT-first
approach could successfully spare nearly half of the patients from
radical resection without compromising 3-year disease-free
survival, while RAPIDO and PRODIGE 23 reported significantly
improved 3-year disease-free survival and higher pCR rates compared
with standard chemoradiotherapy (8,20,21).
Synthesis of real-world evidence:
Validation and refinement
Large-scale prospective registries have been
instrumental in translating trial findings into clinical practice,
addressing questions of generalizability and long-term safety.
While most evidence supports the oncological safety of W&W in
strictly selected patients, a small number of real-world studies
have raised concerns, including a Norwegian population-based study
that reported higher than expected local regrowth (53%) and
metachronous metastases (14%) rates among patients with cCR
(22).
International WaW database (IWWD)
As the largest pooled registry, its analysis of
nearly 1,000 patients provided the first high-quality, multicenter
evidence on oncological outcomes. The reported 5-year overall
survival of 85% and cancer-specific survival of 94% for patients
with cCR offered powerful validation of the safety of the strategy
in a real-world, non-selected cohort (23).
Propensity-matched cohort studies
Studies such as that by Smith et al (24) performed rigorous statistical
matching between patients treated by WaW and patients treated with
surgery with a pCR (24). Their
finding of equivalent survival outcomes provides the strongest
non-randomized comparative evidence to date, effectively mitigating
concerns about survival trade-offs, a conclusion further supported
by the OnCoRe propensity-matched cohort (25).
National registry analyses
Studies from national cancer databases (such as the
Netherlands Cancer Registry, the Chinese Watch-and-Wait Database)
have corroborated these findings on a population level, confirming
that WaW outcomes are reproducible outside expert centers (26,27)
(Table I).
 | Table I.Key clinical studies shaping the
organ preservation paradigm. |
Table I.
Key clinical studies shaping the
organ preservation paradigm.
| Study (year) | Design | Key
intervention/group | Primary finding
relevant to WaW | Evidence level/key
details | (Refs.) |
|---|
| Habr-Gama et
al, 2004 | Prospective
cohort | CRT → cCR →
WaW | Demonstrated
feasibility and safety of non-operative management |
Hypothesis-generating. n=265; 71 cCR
patients (26.8%) managed with W&W; median follow-up 57.3
months; 5-year OS 100% in W&W vs. 88% in pCR resection group;
5-year DFS 92 vs. 83%; no stomas in W&W vs. 9 permanent
colostomies in resection group. | (9) |
| GRECCAR 2,
2017 | Phase III RCT | Local excision vs.
TME (good responders) | Highlighted
challenge of response assessment; supported selective organ
preservation | Level I (for local
excision). n=148; primary endpoint (composite of death, recurrence,
major morbidity at 2 years) not met (56 vs. 48%; OR 1.33, 95% CI
0.62–2.86; P=0.43); 5-year follow-up showed local excision
non-inferior to TME for local recurrence and OS. | (4) |
| RAPIDO, 2020 | Phase III RCT | Short-course RT +
TNT vs. CRT | Notably increased
pCR (28% vs. 14%), expanding pool for WaW | Level I. n=920; pCR
rates: 28% (120/423) in TNT group vs. 14% (57/398) in CRT group (OR
2.37, 95% CI 1.67–3.36, P<0.001); 3-year DFS improved in TNT
group (76 vs. 68%). | (20) |
| OPRA, 2022 | Phase II RCT (organ
preservation endpoint) | TNT → cCR →
WaW | 3-year DFS
identical whether cCR achieved via chemo-first or CRT-first
TNT | Prospective
validation. n=292; 3-year DFS 76% in both arms; organ preservation
rate 41% (INCT-CRT) vs. 53% (CRT-CNCT); salvage TME successful in
>85% of regrowths. | (8) |
| IWWD, 2018 | International
registry | Pooled patients
with cCR from >30 centers | Provided robust
real-world 5-year survival benchmarks (OS 85%, CSS 94%) | Large-scale RWE.
n=867; median follow-up 3.3 years; 5-year OS 85% (95% CI 80–90%),
CSS 94% (95% CI 91–97%); 2-year cumulative local regrowth rate 25%;
80% of regrowths occurred within first 2 years. | (23) |
| Smith et al,
2019 | Propensitymatched
analysis | WaW (cCR) vs. TME
(pCR) | No difference in
3-year OS or DMFS between matched groups | Comparative RWE.
n=113 W&W matched to 136 pCR patients; 3-year OS: 92.4%
(W&W) vs. 95.0% (pCR); 3-year DMFS: 88.9% vs. 92.9%; P=NS for
both; median follow-up 38 months. | (24) |
Evolving consensus: From ‘if’ to
‘how’
The convergence of evidence from RCTs and real-world
studies has shifted the clinical discourse. The question is no
longer if WaW is a valid strategy for selected patients, but how to
implement it optimally. This involves refining patient selection
through improved biomarkers (such as ctDNA), standardizing response
assessment with artificial intelligence and personalizing
surveillance intensity.
Favorable outcomes: The case for organ
preservation
Oncological outcomes: Evidence from
large datasets
Early skepticism toward WaW focused primarily on its
oncological safety. Today, long-term follow-up data from large
international registries provide a robust response. The IWWD, in a
pooled analysis of 867 patients with cCR, demonstrated a 5-year
overall survival rate of 85% and a cancer-specific survival rate of
94% (23). These outcomes are
comparable to those observed in patients who achieve a pCR and
undergo standard TME surgery [e.g., Smith et al (24) reported 5-year OS of 94% in the pCR
group]. This indicates that for the cohort of patients who truly
achieve cCR, delaying or even avoiding surgery does not confer a
survival disadvantage in the mid-term.
A large 2019 multicenter propensity score-matched
analysis further corroborates this view. Comparing patients treated
by W&W from several leading cancer centers with patients with
pCR who underwent standard TME surgery (113 W&W patients
matched to 136 patients with pCR) (24). Comparing patients treated by WaW
from several leading cancer centers with patients with pCR who
underwent standard TME surgery, the study found that after
propensity score matching, the 3-year overall survival and
metastasis-free survival rates were markedly similar between the
two groups (3-year OS: 92.4% in W&W vs. 95.0% in pCR; 3-year
MFS: 88.9 vs. 92.9%; P-value not significant for both) (24). This accumulating real-world evidence
is gradually dismantling the traditional dogma that ‘only surgical
resection can cure’ (25) (Table II).
 | Table II.Comparative oncological outcomes of
WaW vs. standard TME in patients with cCR/pCR. |
Table II.
Comparative oncological outcomes of
WaW vs. standard TME in patients with cCR/pCR.
| Study (year) | Patient group | Sample size, n | Follow-up,
years | OS, % | Disease-free
survival, % | Local
regrowth/recurrence rate, % | Key conclusion | (Refs.) |
|---|
| IWWD, 2018 | WaW (cCR) | 867 | 3 | 91 | 87 | 25 (2-year
cumulative) | Excellent OS/CSS;
most regrowths early and salvageable | (23) |
| Smith et al,
2019 | WaW (cCR) vs. TME
(pCR) | 113 vs. 136
(matched) | 3 | ~95 vs. ~95 | ~88 vs. ~89 | Not directly
compared | Notably comparable
survival outcomes between strategies | (24) |
| OPRA Trial,
2022 | WaW post-TNT | 292 | 3 | 91 | 76 | ~25 | TNT followed by WaW
provides satisfactory organ preservation without compromising
survival | (8) |
Hierarchy of oncological endpoints in
WaW
It is important to distinguish between endpoints
when interpreting these results. Overall survival and
cancer-specific survival (CSS) are the most robust measures of
oncological safety (28).
Disease-free survival (DFS) and local regrowth rates are secondary
endpoints; favorable DFS or regrowth-free survival does not
automatically equate to long-term overall survival equivalence
(29). Currently, most WaW data
support overall survival and CSS up to 5 years; definitive evidence
beyond 10 years remains limited (30).
QoL: A multidimensional benefit
While oncological safety is the non-negotiable
foundation, the potential advantage of WaW lies in its ability to
deliver improvement in patient QoL. This extends beyond the mere
avoidance of a stoma; it represents a comprehensive preservation of
pelvic anatomical and functional integrity. Evidence from
prospective QoL studies using validated instruments (EORTC QLQ-C30,
CR38, LARS score) consistently demonstrates a multifaceted benefit.
For example, a prospective cohort study of 278 patients (221
W&W, 18 local excision, 39 TME) reported that W&W patients
had good QoL and significantly less major bowel dysfunction than
those requiring surgery (25 vs. 56% after local excision and higher
rates after TME) (31). The most
immediate and tangible benefit is the circumvention of major
colorectal resection and its associated sequelae.
Avoidance of permanent stoma
For patients who would otherwise require an
abdominoperineal resection, WaW eliminates the lifelong physical
and psychological burden of a permanent colostomy. Studies report
notably higher scores in body image and social functioning domains
for patients treated by WaW compared with patients with stoma (mean
difference of +15 to +20 points on relevant EORTC subscales)
(32,33).
Prevention of low anterior resection
syndrome (LARS)
Even with sphincter-preserving low anterior
resection, up to 80% of patients experience LARS, a debilitating
cluster of symptoms including fecal incontinence, urgency and
clustering. By forgoing any rectal resection, patients treated by
WaW maintain near-normal bowel continence and function. Severe LARS
is virtually absent in the WaW cohort, whereas it affects 25–40% of
patients after LAR (31).
Reduction in surgical
complications
WaW avoids the risks of anastomotic leakage (5–15%),
pelvic sepsis and re-operation inherent to TME surgery, along with
prolonged hospitalizations and recovery periods (34,35).
Protection of genitourinary and sexual
function
Surgical dissection within the narrow confines of
the pelvis carries a high risk of injury to the autonomic nerve
plexi responsible for bladder control and sexual function. WaW
obviates this iatrogenic injury at its source. Post-operative
sexual dysfunction, particularly erectile dysfunction in men and
dyspareunia in women, is common. A prospective study found that in
men, impotence occurred in 32%, partial impotence in 52%; in women,
dyspareunia occurred in 46% and lubrication difficulties in 56%
(36). A long-term study by Custers
et al (31) found that
>85% of men in a WaW cohort preserved erectile function,
compared with rates often below 30–40% in historical surgical
series. In addition, bladder voiding dysfunction and urinary
incontinence are notably less prevalent in patients treated by WaW,
who are spared injury to the pelvic splanchnic nerves and
hypogastric plexus. A 2024 meta-analysis of 55 studies including
15,072 patients reported that 25% of patients experienced urination
dysfunction ≥12 months after rectal cancer surgery, with long-term
incontinence (23%) and difficulty in bladder emptying (30.6%) as
common complications (37,38).
Psychosocial well-being and the
‘burden of surveillance’
The holistic impact extends into psychological and
social domains. Without the physical trauma of major surgery and
the constant reminder of a stoma or bowel dysfunction, patients
often experience a qualitatively different recovery. Avoidance of a
permanent alteration in body function may mitigate risks of
anxiety, depression and social isolation (39). However, the WaW pathway is not
without psychological demands. The intensive surveillance schedule
and the ever-present, albeit low, risk of regrowth can generate
‘scan anxiety’ and a state of chronic uncertainty for some
patients. Prospective QoL studies generally indicate that this
burden of surveillance is, for most, preferable to the definite and
permanent functional losses imposed by radical surgery (32). This underscores the importance of
thorough patient counseling and shared decision-making, ensuring
that candidates are not only clinically suitable but also
psychologically prepared for the WaW journey.
In summary, the QoL argument for WaW is
multi-dimensional. It is not merely an ancillary benefit but a core
outcome that redefines the therapeutic goal in rectal cancer
management, from survival at any cost to survival with optimal
function and dignity.
Evolution of the treatment
paradigm
The increasing prevalence of WaW signifies a
fundamental shift in the philosophy of rectal cancer treatment,
moving from ‘maximal tolerable therapy’ toward ‘minimal effective
therapy’. Using TNT as an in vivo biopsy, biological
subtypes of tumors that are notably sensitive to CRT can be
screened for. Resecting an organ that has effectively been ‘cured’
in situ can, in a sense, be viewed as overtreatment
(40). WaW thus represents an
application of precision medicine in the surgical management of
solid tumors (41). Despite these
encouraging data, it is critical to recognize that comparative
survival evidence largely stems from non-randomized studies with
inherent selection bias. The favorable QoL outcomes, while
compelling, must be weighed against the psychological burden of
intensive surveillance, a trade-off that requires further
patient-centered research.
Calculated risk: Challenges and perils
Weaknesses of cCR assessment
Currently, the determination of cCR is a clinical
composite judgment, typically based on digital rectal examination,
MRI and high-resolution endoscopy (42,43)
using criteria such as those from Memorial Sloan Kettering Cancer
Center (44) However, this system
has inherent limitations.
Limitations of MRI
While diffusion-weighted imaging provides functional
information, the resolution of MRI for detecting residual
microscopic tumor foci, particularly within the bowel wall, is
limited. Meta-analyses indicate that the false-negative rate of cCR
assessment (cCR without pCR) lies within a range of 10–25%
(10,23,45).
Blind spots of endoscopy and
biopsy
The submucosa underlying endoscopically
normal-appearing mucosa may harbor tumor cells. Random biopsies
suffer from notable sampling error and cannot represent the entire
tumor bed (43,46).
Advances
To break through this bottleneck, research is
focusing on artificial intelligence (AI)-assisted image analysis
(47) and quantitative radiomics.
Deep learning models that analyze texture features imperceptible to
the human eye in MRI images promise more accurate prediction of
tumor regression (43).
Furthermore, new techniques such as endoscopic contrast-enhanced
ultrasound are being explored to improve assessment accuracy
(48–50).
Local regrowth
A major clinical endpoint in the W&W
strategy
Local regrowth represents the most common and
clinically significant adverse event following a watch-and-wait
approach (51). According to IWWD
data (23), the cumulative 2-year
local regrowth rate is ~25%, with the majority (~80%) of these
events occurring within the first 24 months of surveillance
(Fig. 1).
Analysis of regrowth patterns brings a positive
signal, and ~90% of regrowths are local, non-invasive and usually
occur at the primary tumor site (intraluminal regeneration).
Notably, the majority (>85%) of these regrowths can be
curatively treated with timely salvage TME surgery (52,53),
and long-term survival rates after salvage surgery are comparable
with those of patients who had initial surgery. This underscores
the importance of early detection, which relies entirely on a
strict follow-up protocol (54,55).
Strategies to mitigate regrowth
risk
Risk-stratified surveillance intensity based on
initial response depth is a promising approach (54). Patients with a ‘deep’ cCR (complete
normalization of MRI and endoscopy with negative ctDNA) might be
eligible for less intensive follow-up, whereas those with
‘near-cCR’ could be considered for consolidative local therapies
(such as local excision) within clinical trials (26). Emerging evidence suggests that ctDNA
may help identify patients at highest risk of early regrowth,
enabling closer monitoring or prophylactic intervention (56), although this remains
investigational.
Long-term doubts and occult
metastasis: Exploring the unknown
Despite encouraging mid-term data, long-term
oncological outcome data beyond 10 years for the WaW strategy
remain scarce (57). There is
concern about the possibility of occult residual disease in
unresected mesorectal lymph nodes acting as ‘seeds’ for future
distant metastasis (58).
ctDNA
Liquid biopsy, particularly ctDNA detection, is
considered a potential tool for this dilemma (41). ctDNA can detect trace amounts of DNA
fragments from tumor cells in the blood, with assays capable of
detecting mutations at a variant allele frequency as low as 0.01%,
far below the spatial resolution of traditional imaging (59). Studies show that patients with
persistently positive ctDNA after TNT have a markedly high risk of
recurrence; such patients are likely unsuitable for the WaW
strategy. For patients with cCR who become ctDNA-negative after TNT
and enter the WaW pathway, periodic ctDNA monitoring may serve as a
sensitive ‘liquid sentinel’. A reconversion of ctDNA to positivity
often occurs months before radiological or endoscopic detection of
recurrence (60,61), providing a valuable window for
intervention.
Nevertheless, ctDNA remains investigational in this
setting. Standardized assays, validated thresholds and prospective
trials demonstrating its utility as a standalone decision-making
tool are still needed. The future WaW model will likely be a
tripartite precision monitoring system integrating clinical
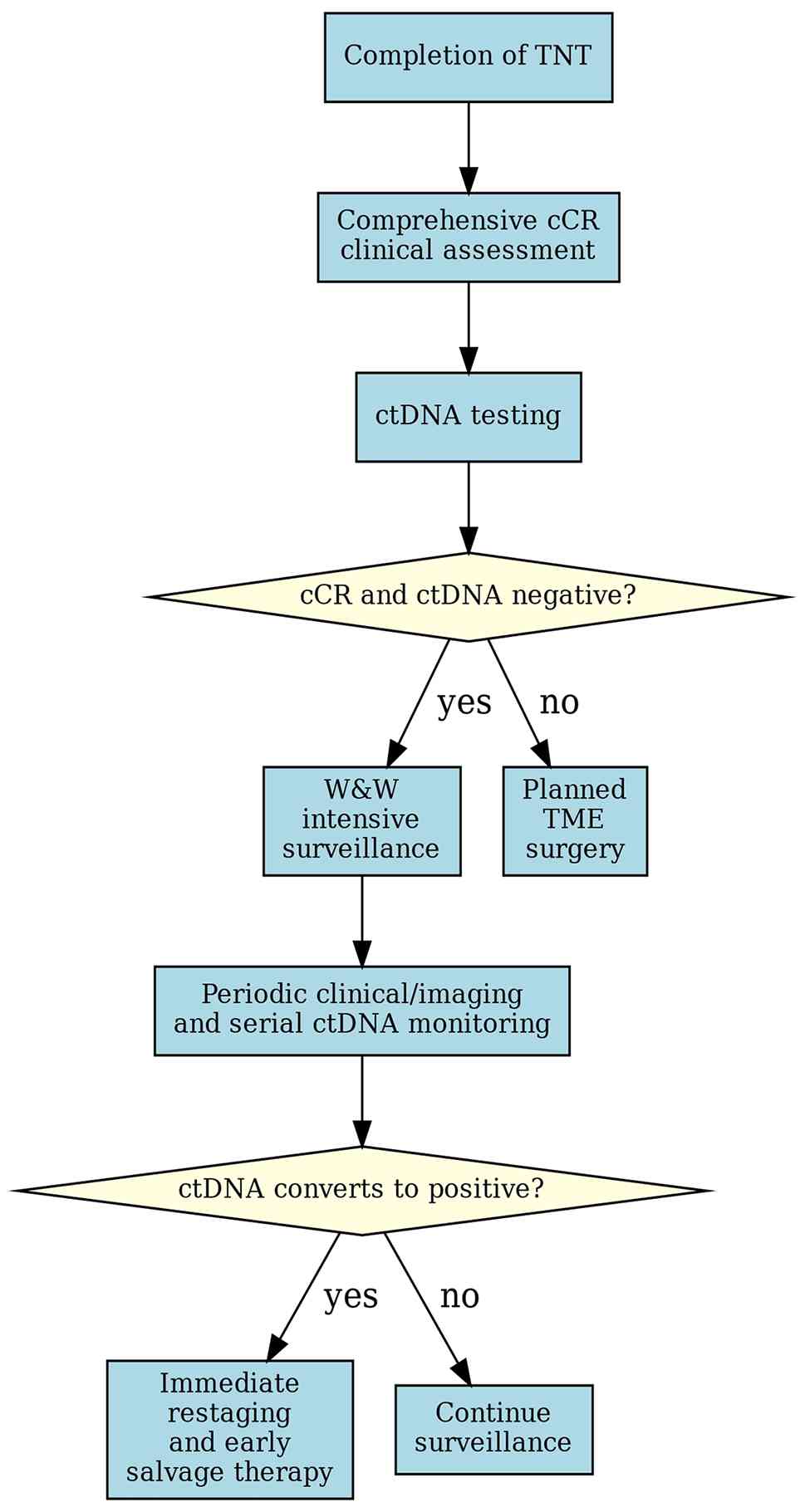
assessment, imaging and ctDNA (Fig.
2), but this remains a hypothesis-generating framework
requiring prospective validation. The challenges outlined here
underscore that WaW is not a passive strategy but an active,
high-stakes management plan.
Precision frontier: Microsatellite
instability-high (MSI-H) rectal cancer and immunotherapy-driven
organ preservation
A critical dimension absent from the earlier
discussion of TNT and WaW is the distinct biological subset of
MSI-H or mismatch repair-deficient (dMMR) rectal cancers, which
account for 5–10% of non-metastatic cases (62). The management of this subgroup is
undergoing a radical transformation, potentially redefining the
concept of neoadjuvant therapy and organ preservation (63).
A unique biology with marked
therapeutic sensitivity
MSI-H tumors harbor a high mutational burden and a
robust tumor-infiltrating lymphocyte presence, rendering them
notably sensitive to immune checkpoint inhibitors (ICIs), such as
anti-PD-1 antibodies (pembrolizumab and dostarlimab). Unlike
conventional chemotherapy and radiotherapy, ICIs can induce deep
and durable antitumor immune responses (64).
Pioneering clinical evidence: Dawn of
‘chemo-radiation-free’ curative intent therapy
Emerging clinical trial data suggest that
neoadjuvant immunotherapy alone may be sufficient for achieving
curative outcomes in locally advanced MSI-H rectal cancer,
challenging the necessity of cytotoxic therapy or radiation. While
focusing on colon cancer, the NICHE study demonstrated a 100%
pathological response rate (660% pCR) (65) to neoadjuvant nivolumab + ipilimumab
in patients with dMMR. This principle is being actively translated
to rectal cancer (66). Numerous
phase II studies investigating neoadjuvant pembrolizumab
monotherapy in MSI-H LARC have reported cCR rates of 60–70% and pCR
rates of 50–60% after several cycles of treatment, with minimal
toxicity (62,67).
A notable single-arm, phase II trial (NCT04165772)
evaluated neoadjuvant dostarlimab in patients with dMMR, locally
advanced rectal adenocarcinoma (68). In an interim analysis, all evaluable
patients (12/12) who completed the planned 6-month course achieved
a cCR, with no evidence of tumor on MRI, fluorodeoxyglucose-PET,
endoscopic evaluation or digital rectal examination. None of the
patients required subsequent CRT or surgery, and no cases of
progression or recurrence were reported during follow-up. At the
time of interim analysis, the median follow-up duration was 6.8
months (range, 0.7–23.8 months) (68).
These response rates, particularly the 100% cCR rate
with dostarlimab, challenge the necessity of cytotoxic therapy or
radiation for this molecular subset. This raises the possibility of
a ‘chemo-radiation-free and surgery-free’ curative pathway for
selected patients with MSI-H. The WaW strategy in this context
evolves from a post-chemoradiation option to the primary intended
management following neoadjuvant immunotherapy alone.
Integration into the WaW framework:
New opportunities and unanswered questions
The integration of MSI-H management into organ
preservation strategies necessitates novel considerations. Patient
selection-including confirmation of dMMR/MSI-H status by
immunohistochemistry or PCR, and exclusion of metastatic
disease-requires universal MSI/MMR testing at diagnosis to identify
candidates for this paradigm. Patterns of response to immunotherapy
(such as pseudoprogression, delayed response) differ from those
seen with CRT, as immunotherapy can manifest unique response
patterns, including pseudoprogression and delayed tumor regression.
The definition of cCR and optimal imaging modalities require
re-evaluation. In addition, the role of ctDNA for monitoring
post-immunotherapy is of interest but not yet standardized. Ongoing
randomized trials (such as comparing immunotherapy-based TNT to
standard CRT-based TNT in patients with MSI-H) will provide Level I
evidence to solidify this approach.
In summary, the MSI-H subtype represents the
vanguard of truly biomarker-driven, response-adapted organ
preservation in rectal cancer. It illustrates the future direction
of the field: Moving beyond a ‘one-size-fits-all’ TNT approach
toward personalized neoadjuvant strategies where the choice of
therapy, and the possibility of WaW, is dictated by the fundamental
molecular characteristics of the tumor (Table III).
 | Table III.Evolving neoadjuvant strategies for
MSI-H locally advanced rectal cancer and their organ preservation
potential. |
Table III.
Evolving neoadjuvant strategies for
MSI-H locally advanced rectal cancer and their organ preservation
potential.
| Strategy | Therapeutic
agents | Reported pCR/cCR
rate, % | Key advantages | Challenges and
unknowns |
|---|
| Standard CRT-based
TNT | Fluoropyrimidine +
Radiation → Chemo | 25-30 | Established safety
profile, long-term data | Likely
‘over-treatment’ for this sensitive subtype; excess toxicity |
| Immunotherapy-based
TNT | Anti-PD-1 ± other
agents → (chemo/RT) | 50-70 (early
data) | Exceptional
efficacy, less acute toxicity, systemic immune activation | Long-term
durability of response? Optimal treatment duration? |
| Immunotherapy
alone | Anti-PD-1
monotherapy | ~60 cCR (early
data) | Maximally
de-escalated; avoids all cytotoxic therapy toxicity | Definitive efficacy
vs. combination approaches? Optimal patient selection? |
Towards consensus: Balancing hope and
risk
Confronting both potential benefits and risks, the
international academic community is converging on a consensus: WaW
is not a binary option but a nuanced strategy to be offered to
carefully selected patients within a markedly structured pathway
(57).
Patient selection and structured
follow-up
Ideal candidate selection
Baseline tumor characteristics include: Initial
stage T2-3 (some selected T4a), N0-1, no distant metastasis and no
extramural vascular invasion. The patient should display excellent
response to TNT, strictly meeting cCR criteria. In addition, the
patient needs to understand the procedure and provide full informed
consent, high compliance and commitment to completing the intensive
follow-up plan (Table IV).
 | Table IV.Ideal candidate selection criteria
for a watch-and-wait strategy. |
Table IV.
Ideal candidate selection criteria
for a watch-and-wait strategy.
| Dimension | Ideal criteria |
Cautionary/exclusionary criteria |
|---|
| Baseline tumor | cT2-3b, cN0-1, no
EMVI, no lateral pelvic node involvement | cT4a/b, cN2,
presence of EMVI, low-lying tumors with sphincter invasion |
| Treatment
response | Excellent response
to TNT, meeting stringent cCR criteria (such as MSKCC
criteria) | Near-cCR, residual
small ulceration or nodule |
| Patient
factors | Comprehensive
informed consent, high compliance, commitment to intensive
follow-up | Poor psychological
resilience, anticipated low follow-up adherence |
| Biomarkers
(emerging)a | ctDNA clearance
post-TNT | Persistently
positive ctDNA post-TNT |
Central role of the multidisciplinary
team (MDT)
The determination of cCR and the subsequent decision
to embark on a WaW pathway is a high-stakes clinical judgment that
should not be rendered by a single specialist. International
consensus guidelines uniformly mandate that this decision should
arise from a formal consensus deliberation by a specialized MDT
(54). An effective rectal cancer
MDT for this purpose should, at a minimum, include colorectal
surgeons, medical oncologists, radiation oncologists, dedicated
abdominal radiologists and gastrointestinal pathologists. This
collaborative synthesis is a notable safeguard against the inherent
limitations and subjectivity of any single assessment modality
(42,43). Studies have shown that MDT review
changes management plans in a notable proportion of complex rectal
cancer cases, improving staging accuracy and adherence to treatment
guidelines (69–72). Therefore, MDT consensus is a
non-negotiable prerequisite for safe entry into a WaW program.
Structured follow-up protocol
The safety of the WaW strategy is contingent upon an
unwavering, protocol-driven surveillance schedule designed for
early detection of local regrowth. There is broad international
consensus on an intensive schedule that de-escalates over time
(54,57). Years 1–2 (high-risk period):
Assessments every 2–3 months, including clinical history, digital
rectal examination, serum carcinoembryonic antigen (CEA),
high-resolution pelvic MRI (with diffusion-weighted imaging) and
rigid or flexible proctoscopy (23,54,55).
This high frequency is justified by the observation that ~80% of
local regrowths occur within the first 24 months after treatment
completion (23). Years 3–5:
Interval can be safely extended to every 6 months, maintaining the
same combination of clinical, biomarker and imaging assessments
(54). After 5 years: Annual
surveillance is generally considered sufficient, mirroring the
long-term follow-up for patients who underwent curative resection
(40,54).
Integrating ctDNA into surveillance:
Technical considerations and investigational status
The incorporation of ctDNA analysis holds
transformative potential for personalizing and enhancing the safety
of WaW surveillance. However, its implementation requires a nuanced
strategy and an understanding of available technologies. It is
important to emphasize that ctDNA monitoring remains an
investigational tool and is not yet incorporated into formal
clinical guidelines; its use should be within clinical trials or
institutional protocols.
Proposed ctDNA monitoring
strategy
A rational, evidence-informed approach involves
three key timepoints (41,60,73):
Baseline (post-TNT): Obtain a plasma sample 4–8 weeks after
completing TNT. A negative ctDNA result is a favorable prognostic
marker, supporting the decision for WaW. A persistently positive
result notably predicts residual disease and imminent recurrence;
such patients should be strongly reconsidered for immediate salvage
TME surgery. During active surveillance (years 1–2): Perform serial
testing every 3–4 months, synchronized with clinical and imaging
visits. The goal is to monitor for molecular recurrence (ctDNA
reconversion from negative to positive), which typically precedes
radiographic detection of regrowth by 3–6 months. At suspicion of
recurrence: In cases of ambiguous imaging or clinical findings, a
contemporaneous ctDNA test can serve as a markedly specific ‘liquid
biopsy’ to clarify the likelihood of true disease recurrence vs.
post-treatment change.
Technical approaches to ctDNA
detection: Pros and cons
Table V compares the
main ctDNA assay types. For the WaW setting, where the clinical
question is the detection of minimal residual disease and early
molecular recurrence, the tumor-informed, patient-specific approach
currently offers the most robust performance characteristics,
despite its logistical demands. As evidence matures and costs
decrease, ctDNA monitoring may evolve from an investigational tool
to a cornerstone of precision surveillance, but current use should
be considered hypothesis-generating (Fig. 2).
 | Table V.Comparison of primary ctDNA testing
strategies in the WaW context. |
Table V.
Comparison of primary ctDNA testing
strategies in the WaW context.
| Assay type | Methodology | Pros | Cons | Best suited
for |
|---|
| Tumor-informed,
patient-specific (such as Signatera™) | Custom-designed PCR
or NGS assay targeting 16–50 somatic variants unique to the
patient's tumor (from archival tissue) | Highest sensitivity
and specificity; can detect minute quantities of ctDNA (limit of
detection ~0.01%); quantitative tracking of variant allele
frequency | Requires tumor
tissue; longer turnaround time; higher cost | Optimal for MRD
monitoring where ultra-sensitivity is critical |
| Tumor-agnostic,
fixed panel (such as Guardant Reveal™) | NGS panel targeting
a fixed set of genes and epigenetic markers common across
cancers | No tumor tissue
needed; faster, standardized workflow; can detect unknown/novel
mutations | Lower sensitivity
for MRD; potential false positives from clonal hematopoiesis
(CHIP) | Screening tool if
tissue is unavailable; may be useful for detecting later,
higher-volume recurrence |
| Methylation-based
assays | Detects
cancer-specific hypermethylation patterns in plasma cell-free
DNA | High
cancer-specificity; methylation changes are abundant and early in
carcinogenesis; tissue-of-origin prediction possible | Clinical validity
in rectal cancer WaW setting still under investigation;
standardization evolving | Emerging modality;
likely to be integrated into future multi-analyte panels |
Comprehensive informed consent
An honest discussion with the patient and family is
mandatory, covering: The rationale for the WaW strategy, the
precise risk of local regrowth, the success rates and risks of
salvage surgery and an explanation of long-term uncertainties.
Conclusions and future directions: Towards a
mature, risk-adapted organ preservation paradigm
The WaW strategy for rectal cancer stands at a
pivotal juncture. It has demonstrated that for a biologically
select group of patients achieving a cCR, organ preservation with
oncological safety is an achievable goal in the mid-term. The
accumulated evidence from registries and comparative studies
confirms that WaW can deliver survival outcomes comparable to
radical surgery up to 5 years, while offering a transformative
improvement in QoL by preserving anorectal, urinary and sexual
function. This firmly establishes it as a valid component of the
modern therapeutic arsenal for locally advanced rectal cancer, but
only for carefully selected patients within a structured program.
However, as the present review has elucidated, current
implementation of WaW is fundamentally constrained by its reliance
on an imperfect, binary decision point, the cCR. This artifact of
assessment leads to inevitable rates of local regrowth and
perpetuates a ‘one-size-fits-all’ surveillance model. Therefore,
the true future of organ preservation lies not in refining the cCR
construct alone, but in orchestrating a shift toward dynamic,
risk-adapted management. To navigate this shift and solidify WaW as
a robust, equitable standard of care, several future directions
must be prioritized.
Precision in selection and
assessment
Prospective validation of ctDNA as a tool for
confirming molecular complete response and monitoring for molecular
recurrence is paramount (73).
Simultaneously, AI and radiomics must be translated from research
tools into clinical aids that standardize and objectify MRI-based
response evaluation, reducing inter-observer variability.
Integration of tissue-based biomarkers (such as Immunoscore,
MSI/MMR status) will further refine upfront patient
stratification.
Development of risk-stratified
pathways
The next generation of clinical trials should move
beyond simply offering WaW to all patients with cCR. They must test
risk-adapted surveillance and intervention protocols. For instance,
patients with a ‘deep’ clinical, radiological and molecular
response could be enrolled in trials of de-intensified, less
frequent follow-up. Conversely, those with ‘near-cCR’ or specific
high-risk biologic features could be offered novel consolidative
therapies (such as short-course immunotherapy, targeted agents)
within clinical trials to increase their chances of successful
organ preservation.
Implementation science and equity
Research must expand beyond academic centers to
address pragmatic questions. Cost-effectiveness analyses comparing
lifelong surveillance to upfront surgery are needed for healthcare
system planning. Studies should also focus on implementation
barriers, such as standardizing MDT protocols across community and
tertiary settings and developing technological solutions (such as
centralized AI imaging review, accessible ctDNA testing) to
mitigate geographic and socioeconomic disparities in access to
precision WaW programs.
Securing long-term data and
patient-reported outcomes
Continued long-term follow-up (>10 years) of
existing WaW cohorts remains essential to definitively confirm the
non-inferiority of CSS. Equally important is the systematic
collection of long-term patient-reported outcomes, capturing not
only functional results but also the psychological experience of
long-term surveillance, to fully understand the benefit-burden
balance from the perspective of the patient.
In conclusion, the WaW strategy offers a promising
alternative to radical surgery for selected patients (74), but it remains a calculated risk,
with the calculation currently based on imperfect inputs. By
embracing the aforementioned future directions focusing on
precision biomarkers, risk-adapted pathways, equitable
implementation and long-term data, the oncology community can
transform this promising strategy into a mature, reliable and
widely accessible pillar of rectal cancer care. The goal is no
longer just to ‘WaW’, but to stratify, guide, and preserve with
confidence.
Acknowledgements
Not applicable.
Funding
The present review was funded by ‘Suqian Talents’ Project
Achievement Contribution Award (grant no. SQCG202506), Project of
Natural Science Foundation of Suqian City in 2025 (grant no.
K202522) and Suqian Sci&Tech Program (grant no. K202221).
Availability of data and materials
Not applicable.
Authors' contributions
The present review was conceptualized by XQZ, BBG
and LCW. The formal analysis was performed by XQZ, BBG and SWS. The
literature search, data extraction, and synthesis was conducted by
XQZ and LCW. Project administration was conducted by XQZ, BBG and
SWS. XQZ and BBG wrote the original draft and reviewed and edited
the manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Fokas E, Williams H, Diefenhardt M, Lin S,
Qin LX, Piso P, Dapper H, Germer CT, Grützmann R, Tim Friede J, et
al: Chemoradiotherapy plus induction or consolidation chemotherapy
as total neoadjuvant therapy for locally advanced rectal cancer:
Pooled analysis of the CAO/ARO/AIO-12 and the OPRA randomized phase
2 trials. Eur J Cancer. 210:1142912024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Benson AB, Venook AP, Al-Hawary MM, Arain
MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D,
Garrido-Laguna I, et al: NCCN guidelines insights: Rectal cancer,
version 6.2020. J Natl Compr Canc Netw. 18:806–815. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rullier E, Rouanet P, Tuech JJ, Valverde
A, Lelong B, Rivoire M, Faucheron JL, Jafari M, Portier G, Meunier
B, et al: Organ preservation for rectal cancer (GRECCAR 2): A
prospective, randomised, open-label, multicentre, phase 3 trial.
Lancet. 390:469–479. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Marr R, Birbeck K, Garvican J, Macklin CP,
Tiffin NJ, Parsons WJ, Dixon MF, Mapstone NP, Sebag-Montefiore D,
Scott N, et al: The modern abdominoperineal excision: The next
challenge after total mesorectal excision. Ann Surg. 242:74–82.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kasi A, Abbasi S, Handa S, Al-Rajabi R,
Saeed A, Baranda J and Sun W: Total neoadjuvant therapy vs standard
therapy in locally advanced rectal cancer: A systematic review and
meta-analysis. JAMA Netw Open. 3:e20300972020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Turri G, Ostuzzi G, Vita G, Barresi V,
Scarpa A, Milella M, Mazzarotto R, Ruzzenente A, Barbui C and
Pedrazzani C: Treatment of locally advanced rectal cancer in the
era of total neoadjuvant therapy: A systematic review and network
meta-analysis. JAMA Netw Open. 7:e24147022024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kagawa Y, Watanabe J, Uemura M, Ando K,
Inoue A, Oba K, Takemasa I and Oki E: Short-term outcomes of a
prospective multicenter phase II trial of total neoadjuvant therapy
for locally advanced rectal cancer in Japan (ENSEMBLE-1). Ann
Gastroenterol Surg. 7:968–976. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Garcia-Aguilar J, Patil S, Gollub MJ, Kim
JK, Yuval JB, Thompson HM, Verheij FS, Omer DM, Lee M, Dunne RF, et
al: Organ preservation in patients with rectal adenocarcinoma
treated with total neoadjuvant therapy. J Clin Oncol. 40:2546–2556.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Habr-Gama A, Perez RO, Nadalin W, Sabbaga
J, Ribeiro U Jr, Silva e Sousa AH Jr, Campos FG, Kiss DR and
Gama-Rodrigues J: Operative versus nonoperative treatment for stage
0 distal rectal cancer following chemoradiation therapy: Long-term
results. Ann Surg. 240:711–718. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dattani M, Heald RJ, Goussous G,
Broadhurst J, São Julião GP, Habr-Gama A, Perez RO and Moran BJ:
Oncological and survival outcomes in watch and wait patients with a
clinical complete response after neoadjuvant chemoradiotherapy for
rectal cancer: A systematic review and pooled analysis. Ann Surg.
268:955–967. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Flom E, Schultz KS, Pantel HJ and Leeds
IL: The predictors of complete pathologic response in rectal cancer
during the total neoadjuvant therapy era: A systematic review.
Cancers (Basel). 15:58532023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Haggstrom L, Chan WY, Nagrial A, Chantrill
LA, Sim HW, Yip D and Chin V: Chemotherapy and radiotherapy for
advanced pancreatic cancer. Cochrane Database Syst Rev.
12:CD0110442024.PubMed/NCBI
|
|
13
|
Gao S, Ye K, Zhang Z, Wang W, Li J, Xu T
and Tan H: Polysaccharides of pseudostellaria heterophylla (Miq.)
Pax ex Pax et Hoffm: Extraction, purification, structural
characteristics, pharmacological activities, and structure-activity
relationships: A review. Int J Biol Macromol. 330:1480822025.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gao S, Zhang Z, Ye K, Wang W, Li J, Xu T
and Tan H: Comprehensive characterization of Rubus idaeus L.
Polysaccharides: Extraction, purification, structural diversity,
biological efficacy, and structure-activity relationships. J
Ethnopharmacol. 355:1206772026. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhao T and Dhillon SK: CD8+ T-cell
signatures as prognostic and immunotherapy response predictors in
non-small cell lung cancer. Folia Biol (Praha). 70:196–208. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu L, Hua F, Li C and Xu T: Application
and evaluation of minimally invasive surgical treatment options for
early endometrial cancer. Technol Health Care. 32:4403–4415. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dossa F, Chesney TR, Acuna SA and Baxter
NN: A watch-and-wait approach for locally advanced rectal cancer
after a clinical complete response following neoadjuvant
chemoradiation: A systematic review and meta-analysis. Lancet
Gastroenterol Hepatol. 2:501–513. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fernandez LM, São Julião GP, Renehan AG,
Beets GL, Papoila AL, Vailati BB, Bahadoer RR, Kranenbarg EM,
Roodvoets AGH, Figueiredo NL, et al: The risk of distant metastases
in patients with clinical complete response managed by watch and
wait after neoadjuvant therapy for rectal cancer: The influence of
local regrowth in the international watch and wait database. Dis
Colon Rectum. 66:41–49. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wen RB and Zhang W: Thoughts on the watch
and wait strategy of patients with rectal cancer after neoadjuvant
therapy. Zhonghua Wei Chang Wai Ke Za Zhi. 23:230–236. 2020.(In
Chinese). PubMed/NCBI
|
|
20
|
Bahadoer RR, Dijkstra EA, van Etten B,
Marijnen CAM, Putter H, Kranenbarg EM, Roodvoets AGH, Nagtegaal ID,
Beets-Tan RGH, Blomqvist LK, et al: Short-course radiotherapy
followed by chemotherapy before total mesorectal excision (TME)
versus preoperative chemoradiotherapy, TME, and optional adjuvant
chemotherapy in locally advanced rectal cancer (RAPIDO): A
randomised, open-label, phase 3 trial. Lancet Oncol. 22:29–42.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Conroy T, Bosset JF, Etienne PL, Rio E,
François É, Mesgouez-Nebout N, Vendrely V, Artignan X, Bouché O,
Gargot D, et al: Neoadjuvant chemotherapy with FOLFIRINOX and
preoperative chemoradiotherapy for patients with locally advanced
rectal cancer (UNICANCER-PRODIGE 23): A multicentre, randomised,
open-label, phase 3 trial. Lancet Oncol. 22:702–715. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wasmuth HH and Færden AE: The norwegian
watch and wait study: Norwait for rectal cancer. A report from a
failed study-a word of caution. Updates Surg. 76:1635–1639. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
van der Valk MJM, Hilling DE, Bastiaannet
E, Meershoek-Klein Kranenbarg E, Beets GL, Figueiredo NL, Habr-Gama
A, Perez RO, Renehan AG and van de Velde CJH; IWWD Consortium, :
Long-term outcomes of clinical complete responders after
neoadjuvant treatment for rectal cancer in the international watch
& wait database (IWWD): An international multicentre registry
study. Lancet. 391:2537–2545. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Smith JJ, Strombom P, Chow OS, Roxburgh
CS, Lynn P, Eaton A, Widmar M, Ganesh K, Yaeger R, Cercek A, et al:
Assessment of a watch-and-wait strategy for rectal cancer in
patients with a complete response after neoadjuvant therapy. JAMA
Oncol. 5:e1858962019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Renehan AG, Malcomson L, Emsley R, Gollins
S, Maw A, Myint AS, Rooney PS, Susnerwala S, Blower A, Saunders MP,
et al: Watch-and-wait approach versus surgical resection after
chemoradiotherapy for patients with rectal cancer (the OnCoRe
project): A propensity-score matched cohort analysis. Lancet Oncol.
17:174–183. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Geubels BM, Grotenhuis BA, van den
Esschert AJ, Lambregts DMJ, Maas M, Marijnen CAM, Beets-Tan RGH and
Beets GL; Dutch Watch-and-Wait consortium, : National
implementation of watch-and-wait in patients with rectal cancer. Br
J Surg. 112:znaf0402025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao YM, Wang WH, Zhang W, Wang L, Li S,
Wang JW, Liao LE, Yu GY, Sun Z, Qu YL, et al: Long-term outcome of
patients with rectal cancer who achieve complete or near complete
clinical responses after neoadjuvant therapy: A multicenter
registry study of data from the Chinese watch and wait database.
Zhonghua Wei Chang Wai Ke Za Zhi. 27:372–382. 2024.(In Chinese).
PubMed/NCBI
|
|
28
|
Fokas E, Glynne-Jones R, Appelt A,
Beets-Tan R, Beets G, Haustermans K, Marijnen C, Minsky BD, Ludmir
E, Quirke P, et al: Outcome measures in multimodal rectal cancer
trials. Lancet Oncol. 21:e252–e264. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yin J, Salem ME, Dixon JG, Jin Z, Cohen R,
DeGramont A, Van Cutsem E, Taieb J, Alberts SR, Wolmark N, et al:
Reevaluating disease-free survival as an endpoint vs overall
survival in stage III adjuvant colon cancer trials. J Natl Cancer
Inst. 114:60–67. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Felder SI and Hoffe SE: Redefining rectal
cancer priorities through a watch-and-wait paradigm: Balancing
cancer cure and quality of life. J Oncol Pract. 15:135–136. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Custers PA, van der Sande ME, Grotenhuis
BA, Peters FP, van Kuijk SMJ, Beets GL and Breukink SO; Dutch
Watch-and-Wait Consortium, : Long-term quality of life and
functional outcome of patients with rectal cancer following a
watch-and-wait approach. JAMA Surg. 158:e2301462023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Peacock O and Chang GJ: ‘Watch and wait’
for complete clinical response after neoadjuvant chemoradiotherapy
for rectal cancer. Minerva Chir. 74:481–495. 2019.PubMed/NCBI
|
|
33
|
Jung WB: Beyond survival: A comprehensive
review of quality of life in rectal cancer patients. Ann
Coloproctol. 40:527–537. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu Y, Hu X, Huang Y, Yin X, Zhang P, Hao
Y, Li H and Wang G: Does transanal drainage tubes placement have an
impact on the incidence of anastomotic leakage after rectal cancer
surgery? A systematic review and meta-analysis. BMC Cancer.
24:2632024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ochiai K, Hida K, Yamaguchi T, Fukuda M,
Akagi T, Akiyoshi T, Okamura R, Yamamoto S, Naitoh T and Konishi T;
Japan Society of Laparoscopic Colorectal Surgery, : Risk factors
for and oncologic impact of anastomotic leakage after
sphincter-preserving proctectomy for mid/low rectal cancer: A
multi-institutional cohort study in Japan. Ann Surg Oncol.
32:7315–7325. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hendren SK, O'Connor BI, Liu M, Asano T,
Cohen Z, Swallow CJ, Macrae HM, Gryfe R and McLeod RS: Prevalence
of male and female sexual dysfunction is high following surgery for
rectal cancer. Ann Surg. 242:212–223. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ágústsdóttir DH, Öberg S, Christophersen
C, Oggesen BT and Rosenberg J: The frequency of urination
dysfunction in patients operated on for rectal cancer: A systematic
review with meta-analyses. Curr Oncol. 31:5929–5942. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lange MM, Maas CP, Marijnen CAM, Wiggers
T, Rutten HJ, Kranenbarg EK and van de Velde CJ; Cooperative
Clinical Investigators of the Dutch Total Mesorectal Excision
Trial, : Urinary dysfunction after rectal cancer treatment is
mainly caused by surgery. Br J Surg. 95:1020–1028. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hupkens BJP, Martens MH, Stoot JH, Berbee
M, Melenhorst J, Beets-Tan RG, Beets GL and Breukink SO: Quality of
life in rectal cancer patients after chemoradiation: Watch-and-wait
policy versus standard resection-a matched-controlled study. Dis
Colon Rectum. 60:1032–1040. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yuval JB, Thompson HM and Garcia-Aguilar
J: Organ preservation in rectal cancer. J Gastrointest Surg.
24:1880–1888. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Airoldi M, Roselló S, Tarazona N, Huerta
M, Pérez-Santiago L, Fleitas T, Pla-Martí V, Puccini A, Roda D and
Cervantes A: Advances in the management of locally advanced rectal
cancer: A shift toward a patient-centred approach to balance
outcomes and quality of life. Cancer Treat Rev. 140:1030152025.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang L, Zhao YM, Sun TT, Xu YL, Li SJ,
Zhang XY, Cai Y, Li YH, Li ZW, Chen PJ, et al: Total neoadjuvant
therapy followed by watch and wait approach or organ preservation
for MRI stratified low-risk rectal cancer: Early result from a
prospective, single arm trial. Zhonghua Wei Chang Wai Ke Za Zhi.
23:258–265. 2020.(In Chinese). PubMed/NCBI
|
|
43
|
van der Sande ME, Maas M, Melenhorst J,
Breukink SO, van Leerdam ME and Beets GL: Predictive value of
endoscopic features for a complete response after chemoradiotherapy
for rectal cancer. Ann Surg. 274:e541–e547. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Polk JB, Campbell J, Drilon AE, Keating P
and Cambrosio A: Organizing precision medicine: A case study of
Memorial Sloan Kettering Cancer Center's engagement in/with
genomics. Soc Sci Med. 324:1157892023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chin RI, Roy A, Pedersen KS, Huang Y, Hunt
SR, Glasgow SC, Tan BR, Wise PE, Silviera ML, Smith RK, et al:
Clinical complete response in patients with rectal adenocarcinoma
treated with short-course radiation therapy and nonoperative
management. Int J Radiat Oncol Biol Phys. 112:715–725. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Liu X, Duan B, Liu R, Zhu M, Zhao G, Guan
N and Wang Y: Enhancing clinical complete response assessment in
rectal cancer: Integrating transanal multipoint full-layer puncture
biopsy criteria: A systematic review. Front Oncol. 14:14285832024.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ichimasa K, Kudo SE, Kouyama Y, Takashina
Y, Chung H, Maeda Y, Lwin WP, Toya Y, Hatta W, So JBY, et al:
Artificial intelligence-assisted whole slide image analysis for
lymph node status prediction in early colorectal and gastric
cancer. Dig Endosc. 38:e700422026. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Badea R, Gersak MM, Dudea SM, Graur F,
Hajjar NA and Furcea L: Characterization and staging of rectal
tumors: Endoscopic ultrasound versus MRI/CT. Pictorial essay. Med
Ultrason. 17:241–247. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Meyenberger C, Huch Böni RA, Bertschinger
P, Zala GF, Klotz HP and Krestin GP: Endoscopic ultrasound and
endorectal magnetic resonance imaging: A prospective, comparative
study for preoperative staging and follow-up of rectal cancer.
Endoscopy. 27:469–479. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gibiino G, Sbrancia M, Binda C, Coluccio
C, Fabbri S, Giuffrida P, Gallo G, Saragoni L, Maselli R, Repici A
and Fabbri C: Usefulness of contrast-enhanced endoscopic ultrasound
(CH-EUS) to guide the treatment choice in superficial rectal
lesions: A case series. Diagnostics (Basel). 13:22672023.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Socha J and Bujko K: The ultimate local
failure rate after the watch-and-wait strategy for rectal cancer: A
systematic review of literature and meta-analysis. Acta Oncol.
62:1052–1065. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
São Julião GP, Fernández LM, Vailati BB,
Habr-Gama A, Azevedo JM, Santiago IA, Parés O, Parvaiz A, Vendrely
V, Rullier A, et al: Local regrowth and the risk of distant
metastases among patients undergoing watch-and-wait for rectal
cancer: What is the best control group? Multicenter retrospective
study. Dis Colon Rectum. 67:73–81. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Peltrini R, Caruso E and Bucci L: Local
regrowth after ‘watch and wait’ strategy: Is salvage surgery enough
for disease control? Int J Colorectal Dis. 34:1505–1506. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cerdán-Santacruz C, Vailati BB, São Julião
GP, Habr-Gama A and Perez RO: Watch and wait: Why, to whom and how.
Surg Oncol. 43:1017742022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Habr-Gama A, São Julião GP, Vailati BB,
Castro I and Raffaele D: Management of the complete clinical
response. Clin Colon Rectal Surg. 30:387–394. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Manca P, Chen CT, Shah F, Lee C, Domenico
D, Omer D, Gonzalez S, De Bruijn I, Ahuno S, Chatila WK, et al:
Ultrasensitive ctDNA monitoring for organ preservation in patients
with locally advanced rectal cancer. NPJ Precis Oncol. 10:82025.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bernier L, Balyasnikova S, Tait D and
Brown G: Watch-and-wait as a therapeutic strategy in rectal cancer.
Curr Colorectal Cancer Rep. 14:37–55. 2018.PubMed/NCBI
|
|
58
|
Swellengrebel HA, Bosch SL, Cats A,
Vincent AD, Dewit LG, Verwaal VJ, Nagtegaal ID and Marijnen CA:
Tumour regression grading after chemoradiotherapy for locally
advanced rectal cancer: A near pathologic complete response does
not translate into good clinical outcome. Radiother Oncol.
112:44–51. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Kidess E, Heirich K, Wiggin M, Vysotskaia
V, Visser BC, Marziali A, Wiedenmann B, Norton JA, Lee M, Jeffrey
SS and Poultsides GA: Mutation profiling of tumor DNA from plasma
and tumor tissue of colorectal cancer patients with a novel,
high-sensitivity multiplexed mutation detection platform.
Oncotarget. 6:2549–2561. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Tie J, Cohen JD, Lahouel K, Lo SN, Wang Y,
Kosmider S, Wong R, Shapiro J, Lee M, Harris S, et al: Circulating
tumor DNA analysis guiding adjuvant therapy in stage II colon
cancer. N Engl J Med. 386:2261–2272. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yu D and Tai J: A new paradigm in
postoperative colorectal cancer surveillance: Integrating advanced
imaging and multi-omics. Front Physiol. 16:17583852026. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Petrelli F, Dottorini L, Zaniboni A,
Celotti A and Iaculli A: Immune checkpoint inhibitors for dMMR/MSI
localized rectal cancer: A systematic review of treatment
strategies. Crit Rev Oncol Hematol. 215:1049212025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Storandt MH and Sinicrope FA: Deficient
mismatch repair/microsatellite instability-high colorectal cancer:
Current treatment paradigms, limitations and future perspectives.
BMJ Oncol. 5:e0009802026. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Sahin IH, Akce M, Alese O, Shaib W,
Lesinski GB, El-Rayes B and Wu C: Immune checkpoint inhibitors for
the treatment of MSI-H/MMR-D colorectal cancer and a perspective on
resistance mechanisms. Br J Cancer. 121:809–818. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chalabi M, Fanchi LF, Dijkstra KK, Van den
Berg JG, Aalbers AG, Sikorska K, Lopez-Yurda M, Grootscholten C,
Beets GL, Snaebjornsson P, et al: Neoadjuvant immunotherapy leads
to pathological responses in MMR-proficient and MMR-deficient
early-stage colon cancers. Nat Med. 26:566–576. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
de Gooyer PGM, Verschoor YL, van den
Dungen LDW, Balduzzi S, Marsman HA, Geukes Foppen MH, Grootscholten
C, Dokter S, den Hartog AG, Verbeek WHM, et al: Neoadjuvant
nivolumab and relatlimab in locally advanced MMR-deficient colon
cancer: A phase 2 trial. Nat Med. 30:3284–3290. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ludford K, Ho WJ, Thomas JV, Raghav KPS,
Murphy MB, Fleming ND, Lee MS, Smaglo BG, You YN, Tillman MM, et
al: Neoadjuvant pembrolizumab in localized microsatellite
instability high/deficient mismatch repair solid tumors. J Clin
Oncol. 41:2181–2190. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Cercek A, Lumish M, Sinopoli J, Weiss J,
Shia J, Lamendola-Essel M, El Dika IH, Segal N, Shcherba M,
Sugarman R, et al: PD-1 blockade in mismatch repair-deficient,
locally advanced rectal cancer. N Engl J Med. 386:2363–2376. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Valentini V, Marijnen C, Beets G, Bujko K,
De Bari B, Cervantes A, Chiloiro G, Coco C, Gambacorta MA,
Glynne-Jones R, et al: The 2017 assisi think tank meeting on rectal
cancer: A positioning paper. Radiother Oncol. 142:6–16. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Bednarski BK, Taggart M and Chang GJ:
MDT-how it is important in rectal cancer. Abdom Radiol (NY).
48:2807–2813. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ma H, Li H, Xu T, Gao Y, Liu S, Wang W,
Wei L, Wang X, Jiang L, Chi Y, et al: Multidisciplinary team
quality improves the survival outcomes of locally advanced rectal
cancer patients: A post hoc analysis of the STELLAR trial.
Radiother Oncol. 200:1105242024. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Ioannidis A, Konstantinidis M, Apostolakis
S, Koutserimpas C, Machairas N and Konstantinidis KM: Impact of
multidisciplinary tumor boards on patients with rectal cancer. Mol
Clin Oncol. 9:135–137. 2018.PubMed/NCBI
|
|
73
|
Mögele T, Höck M, Sommer F, Friedrich L,
Sommer S, Schmutz M, Altenburger A, Messmann H, Anthuber M, Kröncke
T, et al: Circulating tumor DNA for prediction of complete
pathological response to neoadjuvant radiochemotherapy in locally
advanced rectal cancer (NEORECT trial). Cancers (Basel).
16:41732024. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Garcia-Aguilar J, Williams H, Fokas E and
Rodel C: Reply to the letter to the editor by Perez et al.
regarding ‘Survival among patients treated with total mesorectal
excision or selective watch-and-wait after total neoadjuvant
therapy: A pooled analysis of the CAO/ARO/AIO-12 and OPRA
randomized phase II trials’. Ann Oncol. 36:705–706. 2025.
View Article : Google Scholar : PubMed/NCBI
|