Introduction
The incidence of bladder cancer has shown a steady
increase over the past decade with an estimated 72,000 newly
diagnosed cases in 2009 compared to approximately 54,000 in 1999 in
the US (1,2). At primary contact with medical health
care, the majority of patients present with non-muscle invasive
stages of this disease (pTa and pT1) and are commonly treated with
bladder-sparing procedures such as transurethral resection of the
bladder. Clinicopathologic parameters, such as tumor stage and
grade, concomitant carcinoma in situ (CIS) and multifocality
are known to be predictors of recurrence, progression and survival.
Investigations over the past years have increased the understanding
of the biological behavior of this disease and have led to novel
prognostic and therapeutic recommendations and approaches (3). However, more than 50 to 70% of
non-muscle invasive bladder cancer cases recur within 3 years after
primary treatment with a 10 to 30% progression rate to muscle
invasive tumor stages (pT2–pT4) (4,5).
Maspin, a 42-kDa non-serine protease inhibitory
protein of the serpin family, has versatile biological functions.
It is an epithelial-specific gene and is the only serpin whose
absence is lethal in early embryogenesis (6,7). Its
less specific but broad functions indicate that Maspin may be the
oldest component of the serpin family. Maspin plays a role in the
inhibition of angiogenesis, thereby regulating tumor growth,
invasion and metastasis by blocking VEGF-/bFGF-induced vascular
endothelial cell migration (8).
Recombinant Maspin (rMaspin) is produced by fusion with glutathione
S-transferase. Investigations involving rMaspin as well as secreted
Maspin may show various anti-tumor effects besides angiogenesis
inhibition, such as restraint of tumor adhesion and motility,
thereby also modulating invasion and metastasis. Urokinase-type
plasminogen activator, urokinase-type plasminogen activator
receptor and collagen type I and III were previously identified as
extracellular targets that strengthen focal adhesion contacts
(9,10). Among its biological functions,
Maspin was found to induce apoptosis by regulating Bcl-2 family
proteins and/or interacting with Hsp90 (11,12).
The diverse functions of Maspin and its involvement
in tumor development have resulted in an extensive number of
investigational studies. Only four studies involving the role of
Maspin in bladder cancer have been published thus far, with
controversial results (13–16). The present study
immunohistochemically evaluated the expression of Maspin to
determine the correlations between tumor expression in non-muscle
invasive transitional cell carcinoma of the bladder (TCC) and
patient recurrence- and progression-free survival.
Materials and methods
Patients and specimens
Archival specimens from 162 non-muscle invasive
bladder cancer patients treated by transurethral resection between
1993 and 2003 were included in the present investigation. The same
cohort was validated in a previous biomarker study investigating
galectin-3 (17). The study was
approved by the Ethics Committee of Tübingen University. Table I shows details of patient and tumor
characteristics. Follow-up information was available for all 162
patients with a minimum follow-up time of 3 years. In case of tumor
recurrence or progression, tumor specimens were included regardless
of the time when either recurrence or progression occurred.
Recurrence was defined as the reappearance of tumors of the same
stage. Progression was defined as tumor development from non-muscle
to muscle invasive disease. For classification, the tumor, node and
metastasis (TNM) staging system was used. The tumors were initially
graded by pathologists according to the 1973 World Helath
Organization grading system (18).
 | Table IFrequency distribution of
clinicopathological parameters and average Maspin expression
levels. |
Table I
Frequency distribution of
clinicopathological parameters and average Maspin expression
levels.
| Clinicopathological
parameters | No. of
patientsa | % | Maspin expression
levels |
|---|
| Clinical data |
| Male | 119 | 73 | 176 |
| Female | 43 | 27 | 157 |
| Age, years | 67.3 (mean) | 69 (median) | 171 |
| Carcinoma in
situ + | 8 | 5 | 122 |
| Multifocality
(+) | 60 | 37 | 164 |
| Lymph node
(+)c | - | - | - |
| Pathological
distribution |
| pTa | 91 | 56 | 179 |
| pT1 | 71 | 44 | 168 |
| G1 | 59 | 36 | 161 |
| G2 | 90 | 56 | 195 |
| G3 | 13 | 8 | 102 |
| Follow-up data |
| Time to
recurrenceb | 14.6 (mean) | 10 (median) | - |
| Time to
progressionb | 23.1 (mean) | 12 (median) | - |
| pTa+pT1 recurrence
(+) | 84 | 52 | 154 |
| pTa+pT1 recurrence
(−) | 78 | 48 | 191 |
| pTa recurrence
(+) | 47 | 52 | 156 |
| pTa recurrence
(−) | 44 | 48 | 196 |
| pT1 recurrence
(+) | 37 | 52 | 153 |
| pT1 recurrence
(−) | 34 | 48 | 184 |
| pTa+pT1 progression
(+) | 28 | 17 | 82 |
| pTa+pT1 progression
(−) | 134 | 83 | 195 |
| pTa progression
(+) | 9 | 10 | 80 |
| pTa progression
(−) | 82 | 90 | 195 |
| pT1 progression
(+) | 19 | 27 | 83 |
| pT1 progression
(−) | 52 | 73 | 193 |
Tissue microarray and immunohistochemical
analysis
Tumor specimens were fixed in formalin, dehydrated
and embedded in paraffin. The paraffin blocks were cut into 4-μm
sections. Hematoxylin and eosin staining was performed for each
tumor specimen to validate tumor stage and grade. Tissue
microarrays (TMAs) were constructed and stained as previously
described (17,19–21). A
commercially available Maspin antibody (monoclonal mouse antibody;
NCL-Maspin, Novocastra Laboratories Ltd.) in a dilution of 1:500
was used. For the negative control, the primary anti-human Maspin
antibody was replaced by non-immune mouse serum.
The TMA slides were reviewed and classified by two
independent investigators (M.W.K. and A.S.M) in a blinded manner.
For statistical analysis, the immunohistochemical staining reaction
was classified according to a semiquantitative reference scale
ranging from ‘0’ to ‘3+’, depending on the intensity of the Maspin
protein expression. The relative amount of tumor cells stained
positively for Maspin (0–100%) in conjunction with the rating of
the staining intensity, resulted in a staining score ranging from 0
to 300 as previously described (19). The same evaluation method was
validated in previous studies (17,21).
The concordance rate of the investigators was 90%.
Statistical analysis
The JMP program was used for statistical
evaluations. A D’Augostino and Pearson omnibus normality test was
performed to determine whether all data sets were parametric or
non-parametric. One-way ANOVA and the Student’s t-test were applied
to correlate the Maspin expression with various tumor
characteristics. Time-to-event probabilities were estimated by the
univariate Kaplan-Meier method. The Cox proportional hazard model
was applied for the multivariate analysis. P<0.05 was considered
to indicate statistically significant differences.
Results
Clinicopathological data
Specimens of 162 patients [119 (73%) male and 43
(27%) female] with non-muscle invasive TCC as detected using
cystoscopy were included in the present study. Tumor stages
according to the TNM system were determined as follows: pTa, n=91
(56%) and pT1, n=71 (44%). Since exclusively non-muscle invasive
tumors were investigated, no information on lymph node invasion and
distant metastasis was recorded. Of the 162 specimens, 59 (36%)
were graded as G1, 90 (56%) as G2 and 13 (8%) as G3. Follow-up data
were available for patients with a median follow-up of 58.5 months.
Tumor recurrence was observed in 84 (52%) patients, and tumor
progression in 28 (17%) patients. The average time interval between
initial treatment and time of recurrence was 14 (3–72) and 23
(3–79) months for tumor progression (Table I).
Maspin expression
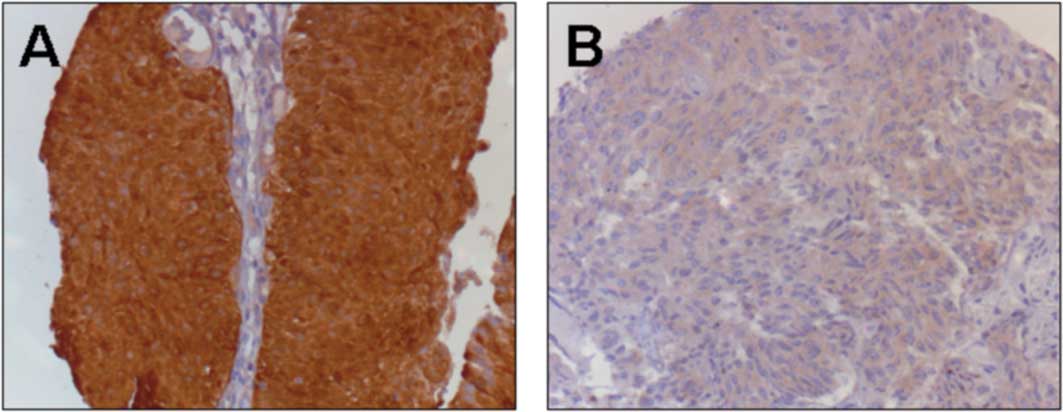
Maspin protein was detected in the nucleus and
cytoplasm of the TCC specimens. A clear shift from high to faint
staining within the nucleus was observed with more aggressive forms
of non-muscle invasive TCC specimens in terms of risk of recurrence
and progression. In 12 muscle-invasive tumor tissues used as
comparative samples, no nuclear staining was observed (unpublished
data).
Statistical analysis of Maspin protein expression in
the TMAs is shown in Table II.
Urothelium-defined (pTa) and minimally invasive TCC (pT1) were not
distinguished by immunohistochemically detected Maspin protein
expression (p=0.42). TCCs with low and high tumor grades (G1 and
G3) showed a lower Maspin expression compared to grade 2 tumors.
CIS was noted in 8 patients. While the presence of CIS correlated
with Maspin staining scores (p<0.05), multifocal tumors at the
time of diagnosis showed no significant correlation (p=0.41).
 | Table IIUnivariate and multivariate
analyses.a |
Table II
Univariate and multivariate
analyses.a
| Parameters | P-value | Chi square | Odds ratio | Standard error |
|---|
| Univariate analyses:
Student’s t-test |
| Fit Y by X
(Y=Maspin) |
| Gender | 0.180 | 1.58 | 1.00 | 0.00 |
| Age | 0.580 | - | - | - |
| Carcinoma in
situ |
<0.050 | 2.28 | 7.68 | 0.00 |
| Multifocality | 0.410 | 1.68 | 1.31 | 0.00 |
| Ta vs. T1 | 0.420 | 0.43 | 1.52 | 0.00 |
| G1 vs. G2 |
<0.050 | 4.42 | 4.28 | 0.00 |
| G1 vs. G3 | >0.050 | 1.67 | 1.30 | 0.00 |
| G2 vs. G3 |
<0.010 | 7.12 | 9.77 | 0.00 |
| Recurrence vs.
non-recurrence (Ta+T1) |
<0.050 | 5.16 | 4.57 | 0.00 |
| Recurrence vs.
non-recurrence (Ta) |
<0.050 | 3.73 | 6.36 | 0.00 |
| Recurrence vs.
non-recurrence (T1) | >0.050 | 1.46 | 1.77 | 0.00 |
| Progression vs.
non-progression (Ta+T1) |
<0.001 | 12.16 | 15.50 | 0.00 |
| Progression vs.
non-progression (Ta) |
<0.001 | 10.96 | 12.24 | 0.00 |
| Progression vs.
non-progression (T1) |
<0.001 | 10.36 | 11.59 | 0.00 |
|
| Multivariate
analysis: Cox proportional hazard |
| Censor: recurrence
of Ta/T1 tumors |
| Maspin
expression |
<0.05 | 6.69 | - | 0.00 |
| T-stage | 0.70 | 0.15 | - | 0.16 |
| Grade | 0.17 | 3.50 | - | 0.29 |
| Multifocality | 0.51 | 0.41 | - | 0.14 |
| Carcinoma in
situ | 0.25 | 1.35 | - | 0.36 |
| Gender | 0.75 | 0.10 | - | 0.16 |
| Age | 0.18 | 1.76 | - | 0.01 |
|
| Multivariate
analysis: Cox proportional hazard |
| Censor: progression
of Ta/T1 tumors |
| Maspin
expression |
<0.0001 | 26.53 | - | 0.00 |
| T-stage | 0.2500 | 1.32 | - | 0.32 |
| Grade | 0.7900 | 0.47 | - | 0.60 |
| Multifocality | 0.0900 | 2.97 | - | 0.25 |
| Carcinoma in
situ | 0.8100 | 0.05 | - | 0.63 |
| Gender | 0.8500 | 0.04 | - | 0.26 |
| Age | 0.2100 | 2.12 | - | 0.13 |
Maspin expression in correlation with
follow-up information
We evaluated whether the Maspin protein expression
in non-muscle invasive bladder cancer specimens predicts tumor
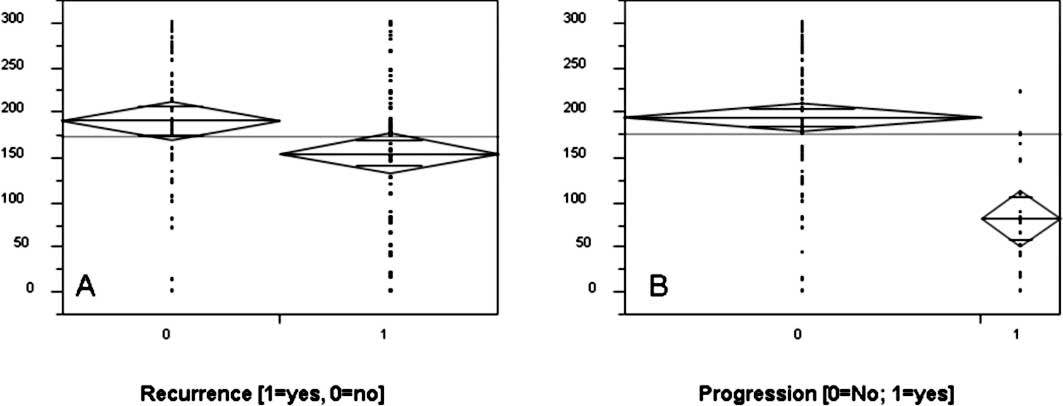
recurrence and/or muscle invasion. Results are shown in Table II. Decreased staining levels in
pTa, as well as in combination with pT1 tumors showed a higher
incidence of tumor relapse (p<0.05, Student’s t-test) as
confirmed by Chi-square analyses (Table II, Fig.
1). However, the Kaplan-Meier analysis did not indicate any
significant prognostic value in respect to tumor recurrence. As
shown in Table III, sensitivity
and specificity were low, at 52 and 67%, respectively. The
univariate analysis of the prognostic significance of the staining
score in relation to the likelihood of tumor progression showed
that a strong Maspin expression and nuclear staining were
associated with better survival in terms of tumor progression
(Table II, Fig. 3). Significant lower staining scores
were noted in pTa and pT1 tumors and a reduction in nuclear
staining in tissue samples of patients who showed tumor progression
within the follow-up time period (p<0.001; Student’s t-test).
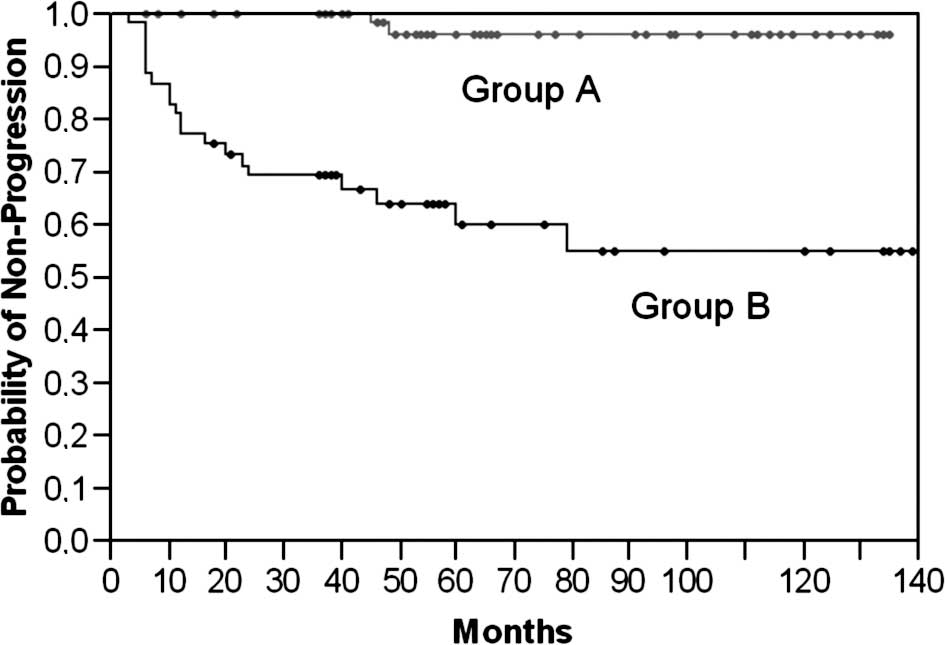
Subsequently, a Kaplan-Meier analysis was performed to further
stratify the prognostic value of Maspin. Therefore, we classified
the analyzed cohort into groups (A vs. B) according to a receiver
operating curve. The cut-off limit (Group A ≥ 175 vs. B < 175)
was based on the highest area under the curve and selected using
the JMP statistical program (JMP 6 software, SAS Institute, Cary,
NC, USA). Sensitivity and specificity were then calculated as 95
and 70%, respectively (Table
III). As illustrated in Fig. 2,
low Maspin protein expression levels were highly associated with a
shorter progression-free survival (46 vs. 18 months; p<0.0001,
Log-rank test). To determine which of the studied parameters and/or
Maspin staining are independent prognostic indicators of tumor
progression to muscle invasion, we used the Cox proportional hazard
model. The statistical analysis affirmed only Maspin as an
independent prognostic predictor for the likelihood of progression
(p<0.0001; Table II). Further
parameters, such as tumor stage and grade, presence of concomitant
CIS, multifocality, age and gender, were included. None of these
parameters were statistically significant.
 | Table IIISensitivities and
specificities.a |
Table III
Sensitivities and
specificities.a
| Sensitivity | Specificity | Log-rank value |
|---|
| Recurrence | 52% | 67% | p=0.1820 |
| Progression | 95% | 70% | p<0.0001 |
Discussion
Maspin is a 42-kDa protein associated with various
tumor-related processes such as the inhibition of cell migration,
cell invasion, angiogenesis, as well as improvement in cell
adhesion and the induction of programmed cell death, thus
classifying it as a tumor suppressor (8–12).
Maspin expression has been observed in multiple tissues, e.g.,
epithelium of the breast, prostate, epidermis and lung (10,22–24).
Localization within the cell appears to define its function. Loss
of nuclear expression in ovarian, breast and lung cancer coincides
with more aggressive phenotypes and decreased survival (25–27).
We found a shift from nuclear to a predominantly cytoplasmic
staining in 12 muscle-invasive tissue samples and, notably, in
highly aggressive forms of non-muscle invasive bladder cancer.
Tissues from patients that showed progression to muscle-invasive
disease detected by subsequent cytoscopies following initial
diagnosis showed less nuclear staining in the primary tissue. This
observation stresses the assumption that cytoplasmic Maspin in the
early steps of tumor progression may signal an imperfect
suppressive effect. Currently, only four studies evaluating Maspin
expression in bladder cancer have been published. Of these, only
Blandamura et al described the possible role of nuclear vs.
cytoplasmic staining (16). These
authors found a correlation between nuclear Maspin expression and
lower histological grades (PUNLMP and low grade). This stresses the
prognostic value of Maspin since low-grade and PUNLMP tumors are
less likely to progress to muscle-invasive tumor stages (pT≥2).
By exclusively including pTa and pT1 tissues in this
investigation, we were unable to show significance in respect to
tumor stage and grade of the 162 examined samples. Similar results
were published by Friedrich et al who found a predominantly
weak Maspin protein expression in pTa and pT1 tumors without any
correlation in respect to stage and grade (15). The two studies must be analyzed in
contrast to that of Blandamura and colleagues who reported a
statistical association between the Maspin staining pattern and
stage and grade. It appears that a strong Maspin expression was
associated with high-grade tumors whereas low-grade tumors and
PUNLMP expressed less Maspin (16).
When comparing Maspin protein expression in non-muscle invasive to
muscle-invasive bladder cancer, Sugimoto et al were the
first to describe an increased expression in higher tumor stages
(pT≥2 > pTa/pT1) (14). However,
in concordance to our results, no statistical difference was found
when comparing pTa and pT1 tumors as 23 of the 27 specimens showed
no Maspin expression at any rate.
Following loss of the Maspin protein expression in
muscle-invasive tumor samples which suggested prognostic
significance, Beecken and colleagues further examined Maspin mRNA
expression in different bladder cell lines (13). Highly aggressive tumor cell lines
(MGHU1, UMUC3) showed no Maspin mRNA expression. A subsequent
investigation of the in vivo tumor growth rate found a close
inverse correlation to the Maspin expression, highly emphasizing
its prognostic value. However, apart from the previous promising
results, no statistically significant correlation between Maspin
staining and tumor relapse of pTa tumors was observed. Notably, the
cohort of 24 patients for follow-up information in that study was
small. In the present study, follow-up information was collected
from 162 patients with a minimum follow-up time of 36 months
(median 58.5). A Chi-square analysis indicated Maspin to be a
prognostic indicator for tumor recurrence in pTa and pTa/pT1 tumors
(p<0.05), as well as an indicator for tumor progression in pTa,
pT1 and pTa/pT1 TCC (p<0.001). However, the sensitivity and
specificity of 52 and 67%, respectively, for tumor recurrence were
rather low compared to 95 and 70%, respectively, for tumor
progression. As described above, loss of Maspin appears to
aggravate tumors via the deregulation of tumor cell growth,
motility and angiogenesis. Accordingly, we found Maspin to be an
independent prognostic indicator for tumor progression to
muscle-invasive disease in TCC in a multivariate analysis
(p<0.0001). Findings of a Kaplan-Meier analysis showed a shorter
progression-free survival (18 vs. 46 months) for non-muscle
invasive TCC with low Maspin protein expression scores.
In conclusion, the findings of the studies
investigating the protein expression of Maspin in TCC indicated
that no definitive answers can be formulated at this point.
However, our study showed that Maspin may play a significant role
in predicting tumor progression since Maspin was found to be an
independent prognostic indicator. In order to promote diagnostic
and therapeutic possibilities, additional studies are warranted to
further clarify the role of Maspin in bladder cancer. Therefore,
further investigational approaches must be engineered.
References
|
1
|
Landis SH, Murray T, Bolden S and Wingo
PA: Cancer statistics. CA Cancer J Clin. 49:8–31. 1999.
|
|
2
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics. CA Cancer J Clin. 59:225–249. 2009.
|
|
3
|
Guney S, Guney N, Canogullari Z and
Ergenekon E: TaT1 low and intermediate transitional cell carcinoma
of the bladder: recurrence rates and the timing of check
cystoscopies within the first year. Urol Int. 80:124–128.
2008.PubMed/NCBI
|
|
4
|
Holmang S, Hedelin H, Anderstrom C,
Holmberg E, Busch C and Johansson SL: Recurrence and progression in
low grade papillary urothelial tumors. J Urol. 162:702–707. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Herr HW: Tumor progression and survival of
patients with high grade, noninvasive papillary (TaG3) bladder
tumors: 15-year outcome. J Urol. 163:60–62. 2000.PubMed/NCBI
|
|
6
|
Futscher BW, Oshiro MM, Wozniak RJ, et al:
Role for DNA methylation in the control of cell type specific
maspin expression. Nat Genet. 31:175–179. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gao F, Shi HY, Daughty C, Cella N and
Zhang M: Maspin plays an essential embryonic development.
Development. 131:1479–1489. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang M, Volpert O, Shi YH and Bouck N:
Maspin is an angiogenesis inhibitor. Nat Med. 6:196–199. 2000.
View Article : Google Scholar
|
|
9
|
Odero-Marah VA, Khalkhali-Ellis Z,
Chunthapong J, et al: Maspin regulates different signaling pathways
for motility and adhesion in aggressive breast cancer cells. Cancer
Biol Ther. 2:398–403. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
McGowen R, Biliran H Jr, Sager R and Sheng
S: The surface of prostate carcinoma DU145 cells mediates the
inhibition of urokinase-type plasminogen activator by maspin.
Cancer Res. 60:4771–4778. 2000.PubMed/NCBI
|
|
11
|
Jiang N, Meng Y, Zhang S, Mensah-Osman E
and Sheng S: Maspin sensitizes breast carcinoma cells to induced
apoptosis. Oncogene. 21:4089–4098. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lockett J, Yin S, Li X, Meng Y and Sheng
S: Tumor suppressive maspin and epithelial homeostasis. J Cell
Biochem. 97:651–660. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Beecken WD, Engl T, Engels K, et al:
Clinical relevance of maspin expression in bladder cancer. World J
Urol. 24:338–344. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sugimoto S, Maass N, Takimoto Y, et al:
Expression and regulation of tumor suppressor gene maspin in human
bladder cancer. Cancer Lett. 203:209–215. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Friedrich MG, Toma MI, Petri S, et al:
Expression of maspin in non-muscle invasive bladder carcinoma:
Correlation with tumor angiogenesis and prognosis. Eur Urol.
45:737–743. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Blandamura S, D’Alessandro E, Giacomelli
L, et al: Expression of maspin in papillary Ta/T1 bladder
neoplasms. Anticancer Res. 28:471–478. 2008.PubMed/NCBI
|
|
17
|
Kramer MW, Kuczyk MA, Hennenlotter J, et
al: Decreased expression of galectin-3 predicts tumour recurrence
in pTa bladder cancer. Oncol Rep. 20:1403–1408. 2008.PubMed/NCBI
|
|
18
|
Mostofi FK, Sorbin L and Torloni H:
Histological typing of urinary bladder tumours International
classification of tumours. World Health Organisation. Geneva:
1973.
|
|
19
|
Kramer MW, Merseburger AS, Hennenlotter J
and Kuczyk M: Tissue microarrays in clinical urology – technical
considerations. Scand J Urol Nephrol. 41:478–484. 2007.
|
|
20
|
Merseburger AS, Anastasiadis AG,
Hennenlotter J, et al: Tissue microarrays: applications in
urological cancer research. World J Urol. 24:579–584. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Merseburger AS, Kramer MW, Hennenlotter J,
et al: Loss of galectin-3 expression correlates with clear cell
renal carcinoma progression and reduced survival. World J Urol.
26:637–642. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ngamkitidechakul C, Burke JM, O’Brien WJ
and Twining SS: Maspin: synthesis by human cornea and regulation of
in vitro stromal cell adhesion to extracellular matrix. Invest
Ophthalmol Vis Sci. 42:3135–3141. 2001.PubMed/NCBI
|
|
23
|
Reis-Filho JS, Milanezi F, Silva P and
Schmitt FC: Maspin expression in myoepithelial tumors of the
breast. Pathol Res Pract. 197:817–821. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Reis-Filho JS, Torio B, Albergaria A and
Schmitt FC: Maspin expression in normal skin and usual cutaneous
carcinomas. Virchows Arch. 441:551–558. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mohsin SK, Zhang M, Clark GM and Craig
Allred D: Maspin expression in invasive breast cancer: Association
with other prognostic factors. J Pathol. 199:432–435. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sood AK, Fletcher MS, Gruman LM, et al:
The paradoxical expression of maspin in ovarian carcinoma. Clin
Cancer Res. 8:2924–2932. 2002.PubMed/NCBI
|
|
27
|
Heighway J, Knapp T, Boyce L, et al:
Expression profiling of primary non-small cell lung cancer for
target identification. Oncogene. 21:7749–7763. 2002. View Article : Google Scholar : PubMed/NCBI
|