Introduction
Prostate cancer frequently develops in older males.
It is the second leading cause of cancer death in American men.
Prostate cancer has serious impact on the survival and quality of
living for patients due to its high potential of metastasis to
other parts of the body. Therefore, deeper understanding of the
mechanisms responsible for the metastasis of prostate cancer is
important for the prevention and therapy of prostate cancer. To
identify important genes that contribute to metastasis of prostate
cancer, previously we performed cDNA microarrays to screen
differentially expressed genes between two PC-3M prostate cell
lines, i.e., PC-3M-1E8 (a high metastatic subline) and PC-3M-2B4 (a
low metastatic subline) (1).
Phosphoprotein associated with glycosphingolipid microdomains 1
(PAG) was one of the down-regulated genes in the high metastatic
PC-3M-1E8 subline.
PAG, also called Csk binding protein (Cbp), is a
ubiquitously expressed transmembrane adaptor protein. The structure
of PAG is characterized by a substantial intracytoplasmic region
that contains multiple tyrosine residues which can be
phosphorylated by Src-family kinases (2). In vitro, PAG could bind a
number of signaling molecules via their SH2 domains, such as Lyn,
Fyn and Csk, a negative regulator of Src-family kinases (3). Previous studies showed that PAG is an
important negative regulator of immunoreceptor signaling in T
lymphocyte (4,5). The binding of PAG and Csk induced the
recruitment of Csk to the membrane and suppressed the activity of
membrane associated Src-family kinases, therefore playing an
important role in the regulation of essential cellular activity to
keep the resting state of T lymphocytes (6,7). In
addition, PAG was able to inhibit the malignancy transformation and
proliferation of tumor cells by inhibiting the activation of Src
(8). In the stimulated T lymphocyte
PAG could interact with RasGAP (Ras GTP activation protein) as
revealed by immunoprecipitation, indicating that PAG may negatively
regulate Ras activity (9).
The recruitment of RasGAP to the membrane stimulates
the rapid hydrolysis of Ras-GTP formed in the lipid rafts, thus
effectively blocking Ras activation. RasGAP could perform this
regulatory function because it possesses two SH2 domains and could
bind PAG via its N-terminal SH2 domain (10,11).
Therefore, we speculate that the interaction of RasGAP with PAG is
not exclusive in T lymphocytes, but also in other kinds of cells
including tumor cells, which could lead to the inhibition of Ras
activity and the alteration of biological behaviors of tumor cells.
However, this hypothesis has never been studied and reported so
far.
The present study aimed to investigate the role of
PAG in the proliferation, invasion and metastasis of prostate
cancer cells and the underlying mechanisms. Our results
demonstrated that increased expression of PAG could inhibit the
proliferation and invasion of PC-3M-1E8 and DU145 cells in
vitro through interacting with RasGAP and regulating the
effectors of Ras signaling, such as Ras, p-ERK and cyclin D1.
Materials and methods
Cell culture
PC-3M-1E8, a variant subline of PC-3M with high
metastatic potential, and PC-3M-2B4, a variant subline of PC-3M
with low metastatic potential, and DU145, a high metastatic
prostate cancer cell line, were cultured in RPMI-1640 medium
(Gibco) supplemented with 10% heat-inactivated fetal bovine serum
(Gibco) at 37°C in a humidified incubator with 5%
CO2.
Real-time RT-PCR
Total RNA was extracted from the PC-3M-1E8,
PC-3M-2B4 and DU145 cells by TRIzol reagent (Invitrogen), and used
as the template for the synthesis of cDNA by M-MLV reverse
transcriptase (Promega). The mRNA level of PAG was detected by ABI
PRISM 7500 fluorescent quantitative polymerase chain reaction
instrument. The forward primer (GATGTTCAGCCGTTCAGTTAC) and reverse
primer (TCTGGACTTCCTCGTAATGC) were designed using primer premier
5.0 software. The reaction conditions were: 95°C for 5 min;
followed by 40 cycles at 95°C for 1 min, 60°C for 30 sec and 72°C
for 30 sec.
Western blot analysis
PC-3M-1E8, PC-3M-2B4 and DU145 cells were collected
and resuspended in 2X SDS cell lysis solution (100 mM Tris-HCl pH
6.8, 4% SDS, 20% glycerol, 200 mM DTT, 0.2% bromophenol blue). The
cell lysate was separated by sodium dodecyl sulfate-polyacrylamide
gel electrophoresis (SDS-PAGE) and then transferred to
nitrocellulose membranes. The membranes were subsequently probed by
PAG antibody (1:400, Abcam) and tubulin antibody (1:400,
Neomarker), and secondary antibody. The membranes were developed
using ECL chemiluminescence reagent (Pierce).
Plasmids and transfection
The cDNA of human PAG was amplified from total RNA
isolated from PC-3M-2B4 cells by RT-PCR (forward primer:
CGGAATTCGCCACCATGGGGC CCG CGGGGAGCCT, reverse primer: CCGCTCGAGCTA
GAGCCTGGTAATATCTCTG CCTTGCTGCAAGTCAC). The PCR products were cloned
into the eukaryotic expression vector pcDNA3 (Invitrogen) by
EcoRI and XhoI sites. The recombined plasmid
pcDNA3-PAG was verified by sequencing and prepared by Pureyield™
plasmid Midprep kit (Promega). PC-3M-1E8 and DU145 cells were
transfected with pcDNA3-PAG using Lipofectamine™ 2000 (Invitrogen)
and selected with G418 (600 μg/ml, Biochem). Four clones of each
cell with high PAG expression level identified by western blot were
mixed for the subsequent studies. PC-3M-1E8 and DU145 transfected
with pcDNA3 and parental PC-3M-1E8 and DU145 cells were used as the
controls.
Cell proliferation assay
MTT assay was performed to evaluate cell
proliferation as previously described (12). Briefly, cells were seeded in a
96-well-plate at 1.0×103 cells/well in triplicate. MTT
solution (20 μl) (5 mg/ml, Sigma) was added into each well and 150
μl DMSO was added 4 h later. Then the plate was measured for the
absorbance at 570 nm by ELISA.
Soft agar colony formation assay
Low melting-point agarose (1.2%) and 2X DMEM culture
solution with 20% fetal bovine serum were mixed to be used as the
bottom-layer agar in the 60-mm culture dishes. Single-cell
suspensions of 4×104 cells were subcultured into 3 ml
DMEM containing 20% fetal bovine serum and 0.7% low melting-point
agarose. Twenty days after plating, the number of colonies
containing >50 cells was counted under inverted microscope.
Flow cytometry for cell cycle
analysis
Cells were digested by 10% trypsin and washed twice
in cold PBS and subsequently fixed by 75% alcohol at 4°C for 24 h.
Three hundred-mesh nylon net was applied to filter the cells and
then 10 μl RNase was added and incubated at 37°C for 30 min.
Following staining with PI, samples were subjected to flow
cytometry analysis (BD FACSCalibur).
Flow cytometry for analysis of
apoptosis
The Apoptosis Assay kit (Gene Research Center of
Peking University) was used to evaluate prostate cancer cell
apoptosis. Cells were digested by 10% trypsin and washed twice in
cold PBS. Three hundred-mesh nylon net was used to filter the
cells. Cells were resuspended in 200 μl binding buffer. Then 10 μl
Annexin-V-FITC was added and incubated at 4°C for 30 min. Cells
were stained with 5 μl propidium iodide (PI) and subjected to flow
cytometry analysis.
Invasion assay
The conditional medium of NIH3T3 cells was used as
the chemotactic factor in the inferior part of the Boyden chambers.
A polycarbonate 8-μm thick millipore membrane was placed between
the superior and inferior chambers. Matrigel (50 μl) (1 g/l, BD)
was evenly distributed on the membrane. After complete
polymerization of the matrigel, ~2×105 cells were seeded
into the superior chamber of the well. Then the cells were
cultivated at 37°C in 5% CO2 for 8 h. The cells that
crossed the membrane were fixed by methanol and stained by
hematoxylin and eosin (H&E), and counted under a light
microscope.
Immunoprecipitation
For immunoprecipitation, 2×106 cells were
lysed in 500 μl cold NP-40 lysis buffer (1% NP-40, 50 mM
Tris-buffer saline pH 8.2, 100 mM NaCl, 5 mM EDTA) containing 5 μl
protease inhibitor cocktail and 5 μl phosphatase inhibitor for 30
min and then centrifuged for 15 min at 4°C, 12000 rpm to collect
the supernatant. PAG antibody (2 μg, Abcam) and protein A agarose
beads (Santa Cruz) were added into the supernatant and the mixture
was rotated at 4°C, 100 rpm for 12 h. In the control group, PAG
antibody was replaced by mouse IgG. Beads were washed three times
with PBS containing protease inhibitor cocktail and phosphatase
inhibitor, resuspended in 2X SDS loading buffer (100 mM Tris-HCl pH
6.8, 4% SDS, 20% glycerol, 200 mM DTT, 0.2% bromophenol blue) and
boiled for 10 min. After centrifuging, the supernatant was
collected for western blot analysis using RasGAP antibody (1:300,
Abcam).
GST pull-down assay
The expression of the fusion protein
pGEX-GST-Raf1-Ras-binding-domain (RBD) (amino acids 51–131 of human
Raf-1) in JM109 E. coli was induced by IPTG (final 1 mmol/l,
Gold Biotechnology). SDS-PAGE and coomassie brilliant blue staining
were performed using 20 μl lysate of the bacterium to estimate the
expression of the fusion protein. Total bacterial lysate was mixed
with 30 μl GST-sepharose 4 beads (Pharmacia) and incubated for 1 h
at room temperature. After being washed in PBS three times, the
beads were mixed with 300 μl cell lysate (~2×106 cells)
and incubated at 4°C for 12 h. Then the beads were washed three
times with NP-40 solution, resuspended in 2X SDS loading buffer and
boiled for 10 min. After centrifuging, the supernatant was
collected for western blot analysis using pan-Ras antibody (1:500,
Cell Signaling).
Western blot analysis
PC-3M-1E8 cells were lysed in cold NP-40 lysis
buffer and the cell lysate was collected for western blot analysis.
The antibodies used were as follows: anti-ERK1/2 (1:1000),
anti-p-ERK1/2 (1:200), anti-cyclin D1 (1:500),
anti-P21WAF1/CIP1 (1:300) and anti-p-AKT (1:500). All of
them were purchased from Santa Cruz Biotechnology.
TRITC-phalloidine staining
The cell suspension was dropped onto the sterile
glass slide and incubated at 37°C for 24 h in a humidified
incubator with 5% CO2. Then the slides were fixed in
cold acetone for 30 min. After being washed in PBS three times, the
slides were treated with 0.2% Triton X-100 for 2 min for adequate
permeabilization. Next the slides were washed by PBS three times
and stained by phalloidine labeled by
tetramethylrhodamine-5-(and-6)-isothiocyanate (TRITC) (1:50
dilution, Sigma) for 45 min at room temperature in the dark.
Finally, the slides were mounted by the mixture of glycerin and PBS
at equal volume. The cell morphology and F-actin arrangement were
observed under a laser passing confocal microscope. F-actin was
stained red.
Statistical analysis
Statistical analysis was performed using SPSS13.0
software. P<0.05 was considered statistically significant.
Results
Low expression of PAG in high metastatic
prostate cancer cells
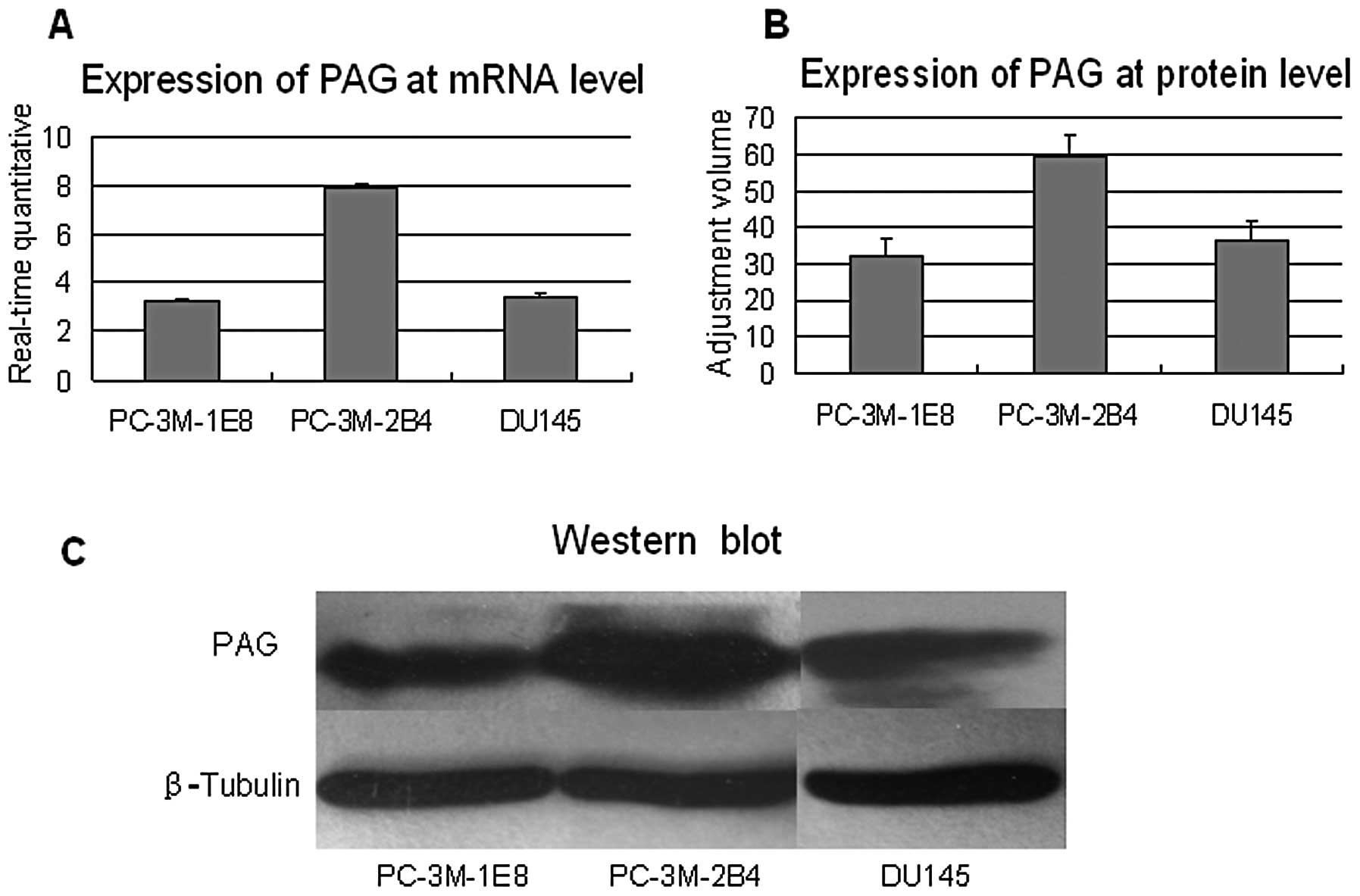
To confirm our previous cDNA microarray results
showing that PAG was down-regulated in the PC-3M-1E8 cells compared
to PC-3M-2B4 cells, we performed real-time quantitative RT-PCR and
western blot analysis to evaluate the expression level of PAG in
these prostate cancer cells. Real-time RT-PCR analysis showed that
the expression of PAG at mRNA level was twice that in PC-3M-2B4
cells than in PC-3M-1E8 and DU145 (P<0.5, Fig. 1A). Western blot analysis further
demonstrated that the expression of PAG at protein level was low in
PC-3M-1E8 and DU145 cells compared to that in PC-3M-2B4 (P<0.5,
Fig. 1B and C). Taken together,
these results demonstrate that PAG is down-regulated in high
metastatic prostate cancer cells, suggesting that PAG may inhibit
prostate cancer development and metastasis.
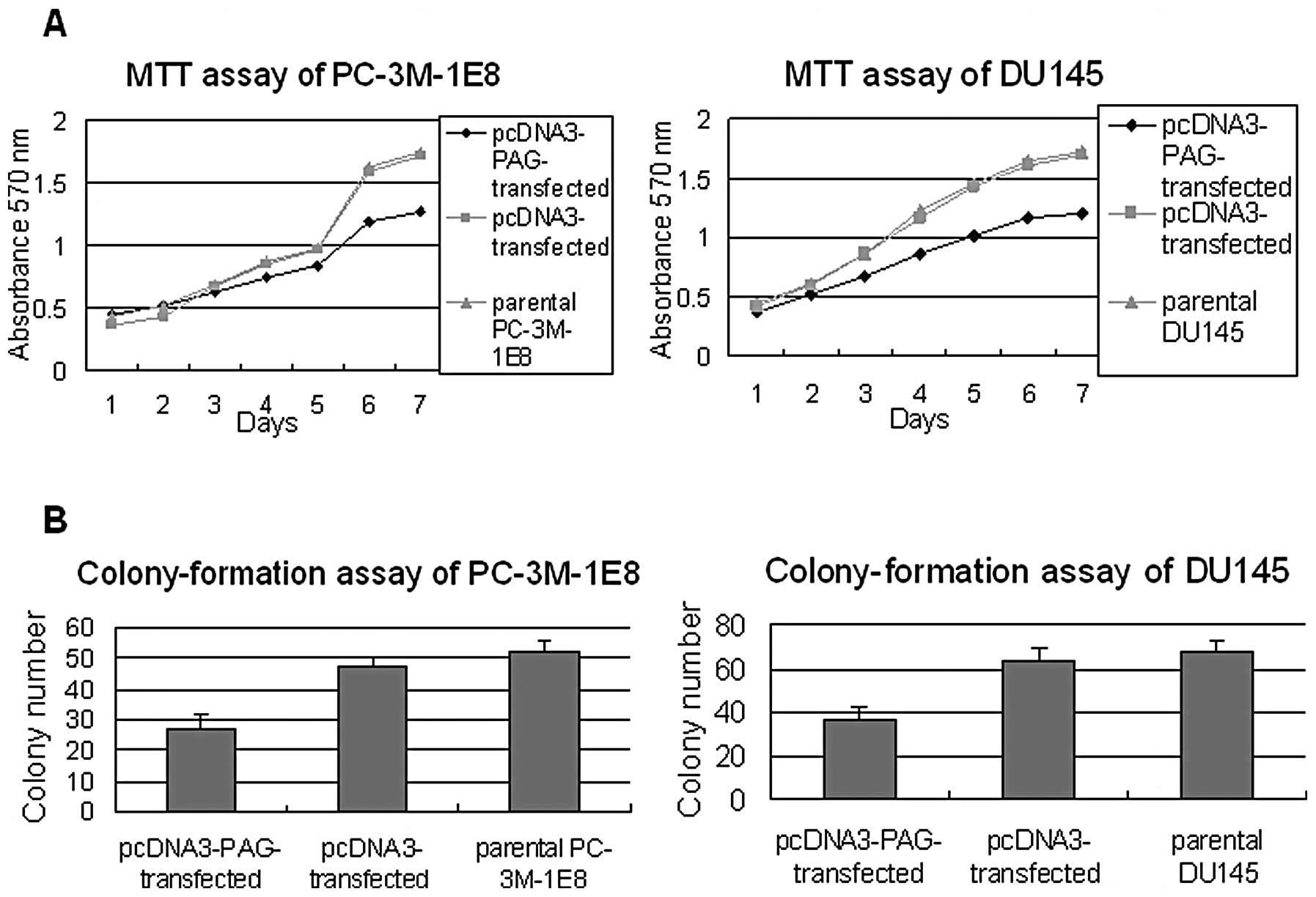
PAG inhibits the proliferation and
invasion of high metastatic prostate cancer cells
To provide experimental evidence that PAG inhibits
prostate cancer development and metastasis, first we performed MTT
assay and soft-agar colony-formation assay to examine the
proliferation ability of PC-3M-1E8 and DU145 cells that exogenously
expressed PAG. The results showed that PC-3M-1E8 and DU145 cells
expressing exogenous PAG grew more slowly and formed fewer colonies
than the control cells under the same circumstance (P<0.5,
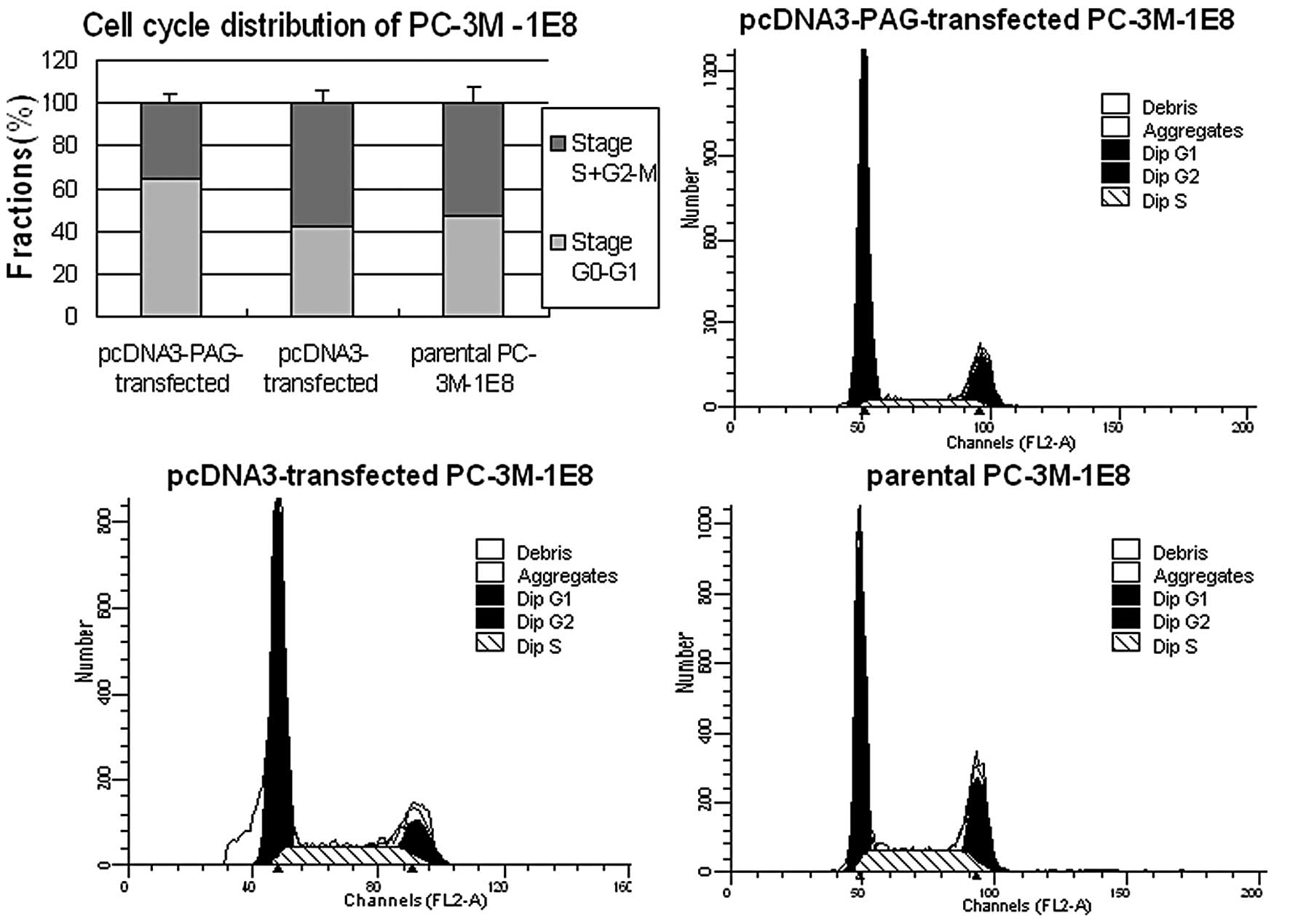
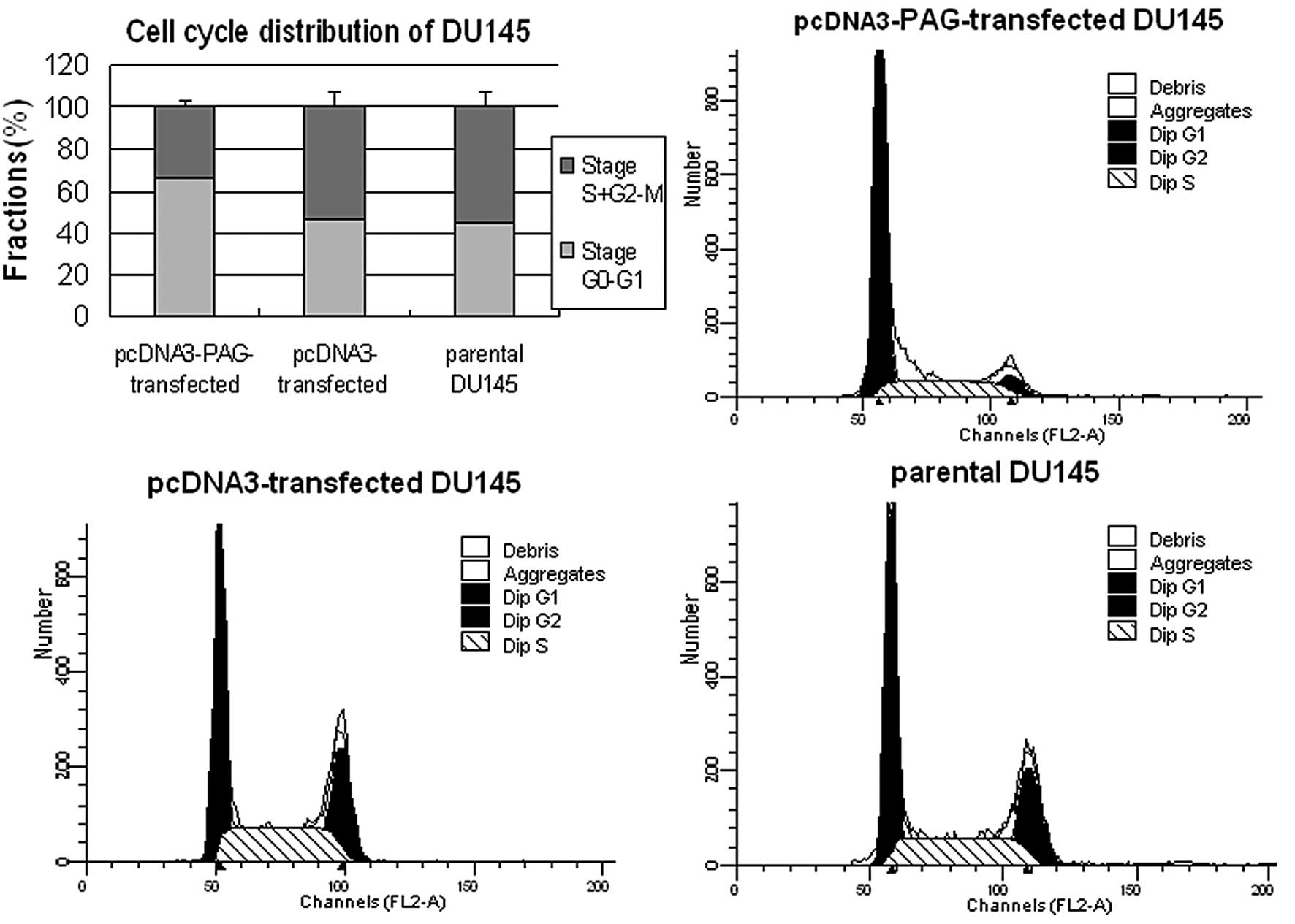
Fig. 2). Next we performed flow
cytometry analysis to investigate the cell cycle progression and
apoptosis of these cells. The results demonstrated that higher
percentage of cells was distributed in stage
G0–G1 in PC-3M-1E8 (Fig. 3) and DU145 cells (Fig. 4) expressed exogenous PAG more than
in the controls (P<0.5). However, no significant difference in
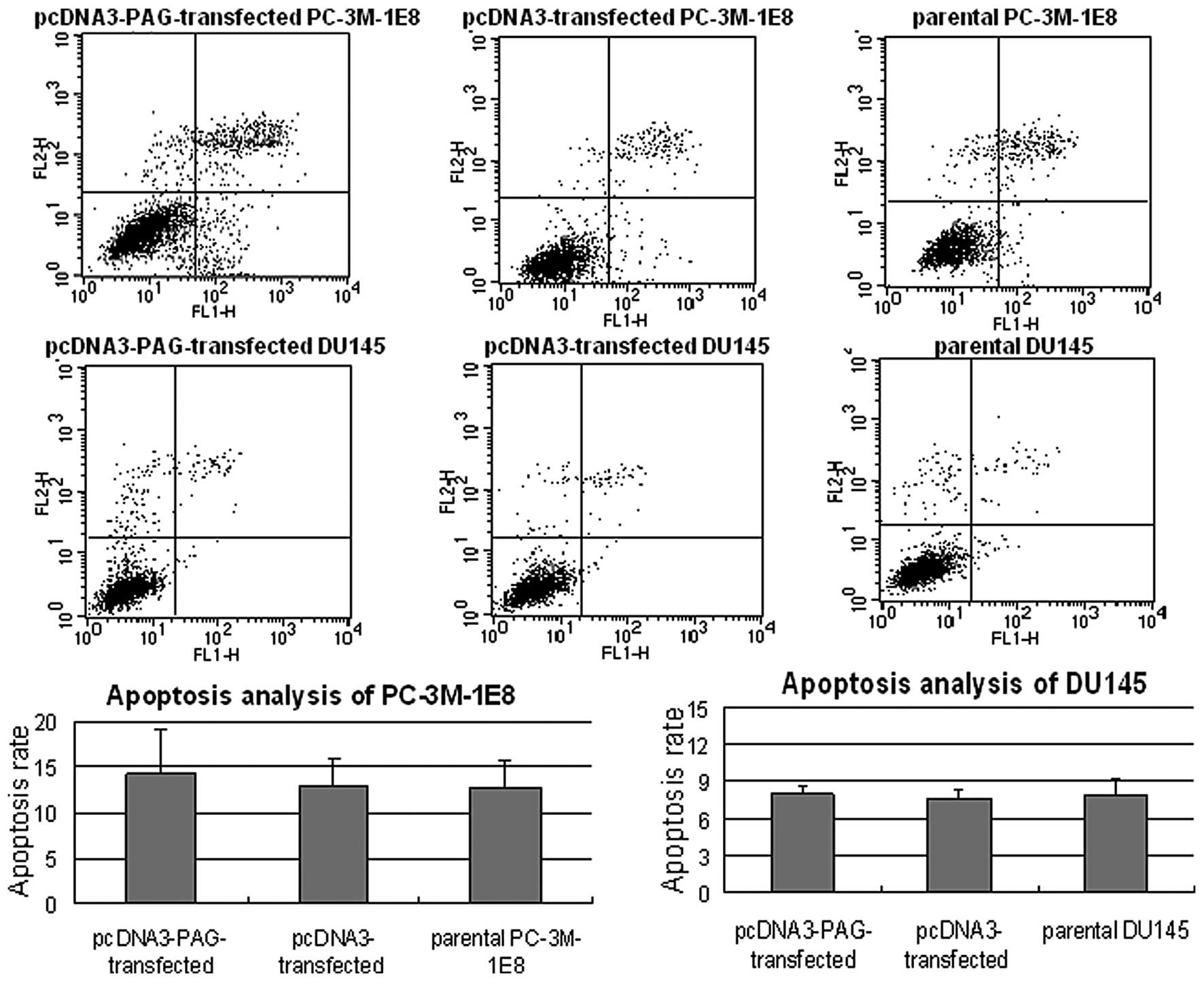
the apoptosis rate was found between PC-3M-1E8 and DU145 cells
expressing exogenous PAG and the controls (P>0.5, Fig. 5). Taken together, these results
indicate that the decreased proliferation of PC-3M-1E8 and DU145
cells upon exogenous expression of PAG is attributed to the arrest
of cell cycle rather than the promotion of apoptosis.
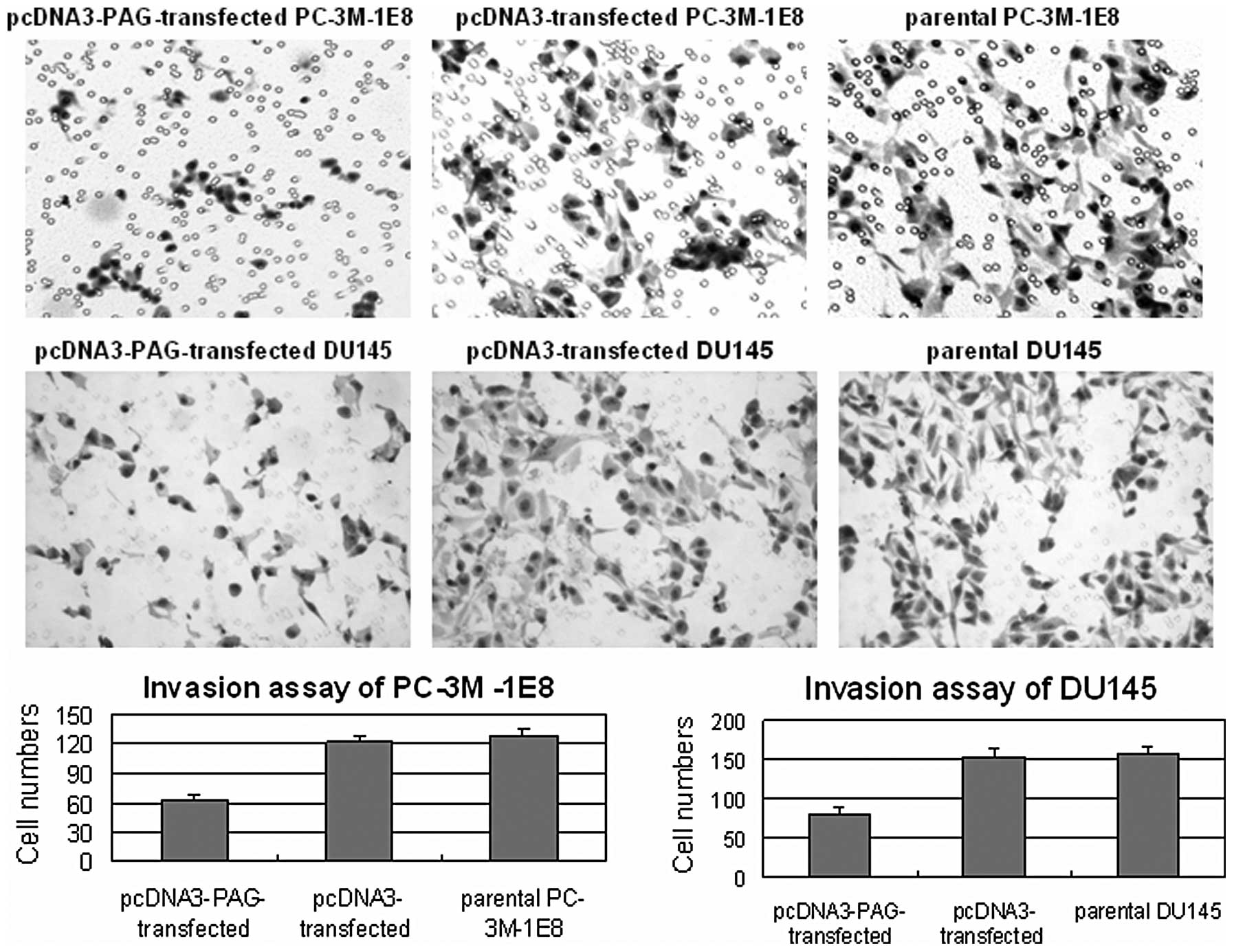
Furthermore, we employed matrigel invasion assay to
examine the invasion ability of PC-3M-1E8 and DU145 cells that
exogenously expressed PAG. The results showed that the number of
PC-3M-1E8 and DU145 cells passed the matrigel and millipore
membrane was reduced after exogenous expression of PAG (P<0.5,
Fig. 6), proving that PAG inhibits
the invasion of prostate cancer cells.
PAG interacts with RasGAP and inhibits
the activation of Ras signaling in prostate cancer cells
To elucidate the potential mechanism by which PAG
modulates the proliferation and invasion of prostate cancer cells,
we decided to characterize the interacting partners of PAG in
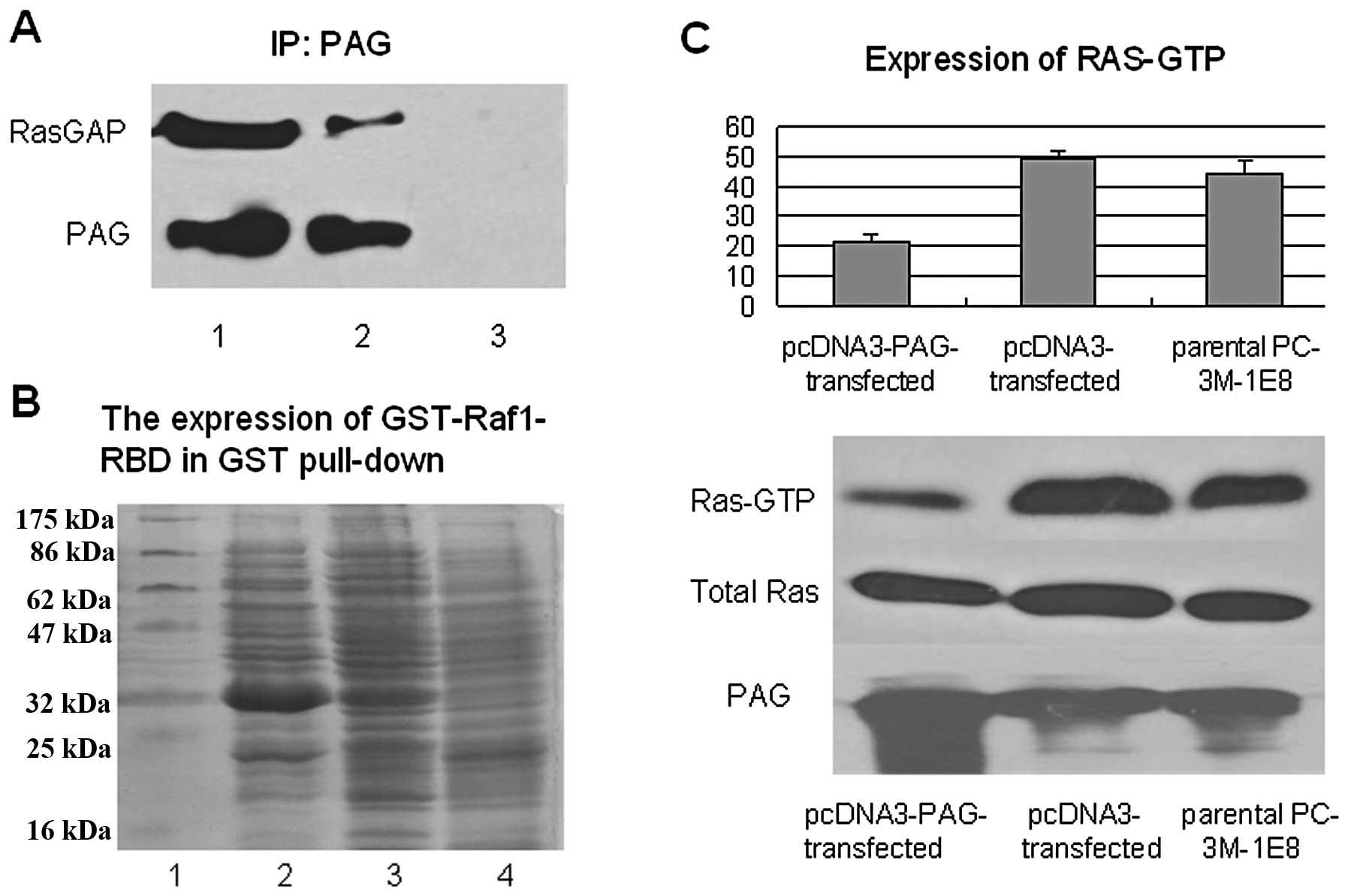
prostate cancer cells. Since RasGAP was suggested to bind directly
to PAG in T lymphocyte, immunoprecipitation experiment was
performed to examine the interaction between PAG and RasGAP in
PC-3M-1E8 cells. The results showed that endogenous RasGAP could be
co-precipitated with PAG in PC-3M-1E8 cells that stably transfected
with PAG expression plasmid by PAG antibody but not mouse IgG
control (Fig. 7A). Having
established that PAG interacted with RasGAP, next we examined
whether PAG could modulate the activation of Ras. To this end, we
performed GST pull-down assay in which the pGEX-Raf1-Ras-binding
domain (RBD) encoding amino acids 1–140 of c-Raf-1 fused to GST
could specially bind the activated Ras (Ras-GTP). After the
induction of IPTG, the GST-Raf1-RBD fusion protein with the
molecular of 33 kDa was highly expressed in JM109 (Fig. 7B). GST pull-down assay demonstrated
that the activated Ras (Ras-GTP) was reduced in PC-3M-1E8 cells
that stably transfected with PAG expression plasmid (P<0.5), but
the amount of total Ras did not change, compared to the controls
(Fig. 7C).
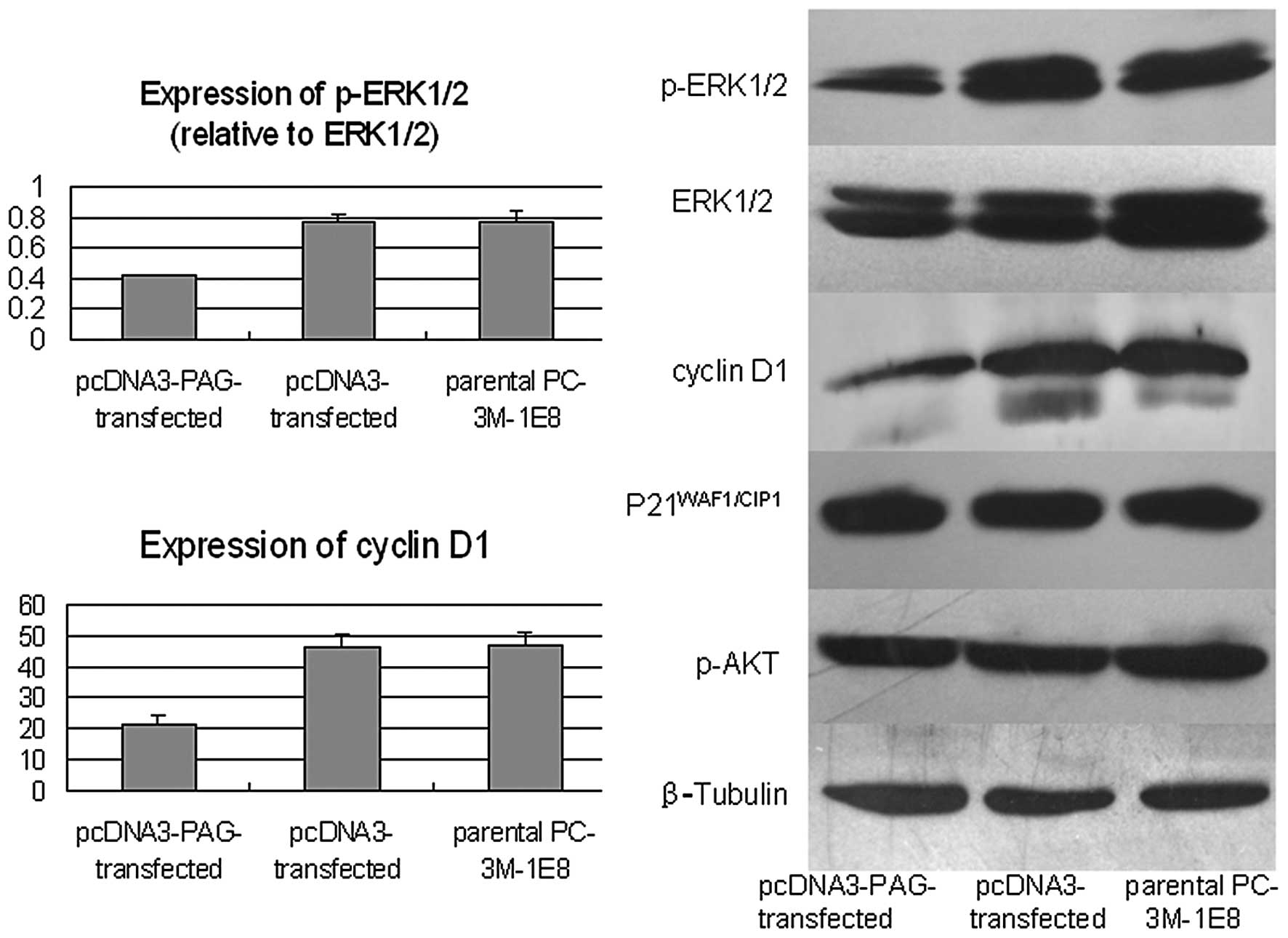
To provide further evidence that PAG modulates Ras
signal pathway, we examined the levels of components of Ras
signaling by western blot and found that the levels of p-ERK1/2 and
cyclin D1, two crucial factors involved in cell proliferation, were
decreased notably accompanied with the increased expression of PAG
in PC-3M-1E8 cells (P<0.5). However, the levels of
P21WAF1/CIP1 and p-AKT did not show significant changes
(Fig. 8). Collectively, these data
strongly suggest that PAG inhibits the activation of Ras signaling
in prostate cancer cells by interacting with RasGAP.
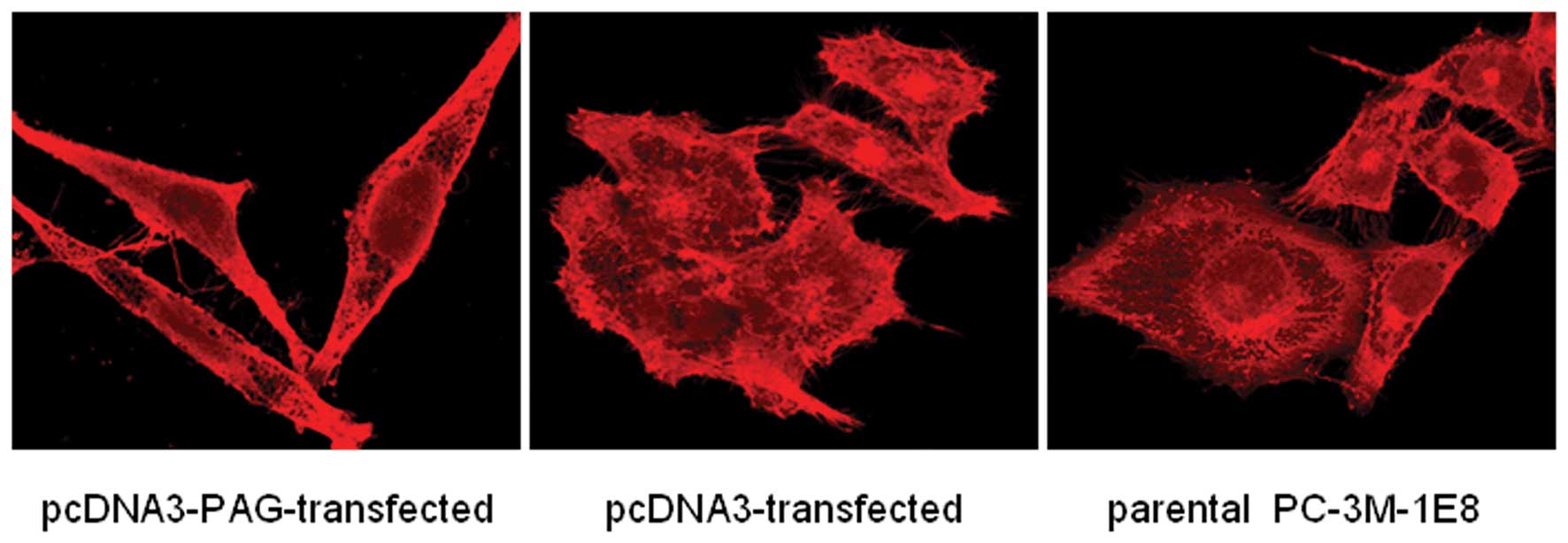
PAG modulates intracellular F-actin in
prostate cancer cells
Cell skeleton is crucially involved in cell motility
and invasion. Therefore, we examined the effects of PAG on the
skeleton remodeling and morphology of PC-3M-1E8 cells. Our results
showed that exogenous expression of PAG in PC-3M-1E8 cells led to
reduced formation of pseudopodia and disrupted structure of
intracellular F-actin as detected by TRITC staining under a
confocal microscope (Fig. 9).
Discussion
In this study we focused on PAG, a candidate gene
that was selected from cDNA microarray screening of genes
up-regulated or down-regulated in high metastatic subline PC-3M-1E8
cells. PAG is a transmembrane adaptor protein without any enzymatic
activity but acts as a scaffold in the signal transduction.
Interestingly, the 397-amino acid cytoplasmic domain of PAG
contains ten tyrosine residues which are the substrates of Src
family kinases (13). When the
tyrosine residues of PAG are phosphorylated, it could recruit
proteins with special domains such as SH2 domain to the membrane
and subsequently induce their phosphorylation or dephosphorylation
to regulate the signal transduction in T lymphocyte (14). Although PAG has been shown as an
important negative regulator of immunoreceptor signaling in T
lymphocyte, the role of PAG in tumor cells remains largely
unexplored.
First, we confirmed that the expression of PAG at
mRNA and protein levels in the high metastatic PC-3M-1E8 and DU145
cells was significantly lower than that in the low metastatic
PC-3M-2B4 cells, in agreement with the results of cDNA microarrays
we previously reported. Next we restored the expression of PAG in
PC-3M-1E8 and DU145 cells by establishing stable transfection of
pcDNA3-PAG expression vector and examined the resulting biological
effects. Our results demonstrated that reconstitution of PAG in
high metastatic PC-3M-1E8 and DU145 cells led to reduced
proliferation through cell cycle arrest, decreased
anchorage-independent growth and reduced invasion ability of
PC-3M-1E8 and DU145 cells in vitro. Collectively, these data
provide important evidence that PAG may play an important role in
inhibiting the proliferation and invasion of prostate cancer
cells.
To further explore the potential mechanism
underlying PAG-induced inhibition of the proliferation and invasion
of prostate cancer cells, we characterized the interaction of PAG
with RasGAP, a known binding partner of PAG in T lymphocytes.
Immuno-coprecipitation assay proved that PAG interacted with RasGAP
in prostate cancer cells, indicating the potential link of PAG with
Ras signaling. To address this possibility we performed GST
pull-down utilizing Raf1-RBD domain to detect the level of
activated Ras and found that Ras activation was down-regulated
after the reconstitution of PAG in PC-3M-1E8 cells although the
total amount of Ras showed no significant change. In addition,
western blot analysis demonstrated that the levels of downstream
proteins of Ras signaling such as p-ERK1/2 and cyclin D1 were
decreased upon the reconstitution of PAG in PC-3M-1E8 cells. Taken
together, we speculate that PAG interacts with RasGAP in prostate
cancer cells to recruit RasGAP to the cell membrane where it
hydrolyzes GTP to GDP and reduces the level of activated Ras,
ultimately suppressing the activation of ERK1/2, cyclin D1 and
other effectors of Ras signal pathway.
In addition, we found that the formation of
pseudopodia in the cellular surface and the structure of F-actin
were disturbed after the reconstitution of PAG in PC-3M-1E8 cells.
Previous studies showed that PAG could inhibit the movement of the
membrane lipid raft and reduce the formation of pseudopodia in T
lymphocyte (15,16). Brdickova et al reported that
PAG could interact with EBP50 to modulate F-actin (3). The formation of pseudopodia has been
proposed as the early and critical step for the migration of tumor
cells into and out of the vascular space and metastasis to the
distant organs (17). Therefore, we
assume that the redistribution of F-actin and impairment of
pseudopodia of cellular surface could directly lead to the
decreased invasion and metastasis potential of PC-3M-1E8 cells.
In conclusion, our data demonstrated the increased
expression of PAG could inhibit the proliferation and invasion
potential of prostate cancer cells in vitro by suppressing
the activations of Ras and downstream effectors such as ERK and
cyclin D1 through the interaction with RasGAP. Morphologically, we
observed that the increased expression of PAG could diminish the
formation of pseudopodia on cellular surface and lead to the
redistribution of the intracellular F-actin in PC-3M-1E8 cells.
Taken together, these results suggested that PAG acts to inhibit
the development and metastasis of prostate cancer cells and
represents a novel therapeutic target for prostate cancer.
Acknowledgements
This study was supported by Nation 973 basal
research development assistance programs (2009CB521805) of
China.
References
|
1
|
Liu YX, Zheng J and Fang WG: Isolation and
characterization of human prostate cancer cell subclones with
different metastatic potential. Chin J Pathol. 28:361–364.
1999.PubMed/NCBI
|
|
2
|
Svec A: Phosphoprotein associated with
glycosphingolipid-enriched microdomains/Csk-binding protein: a
protein that matters. Pathol Res Pract. 204:785–792. 2008.
View Article : Google Scholar
|
|
3
|
Brdickova N, Brdicka T, Andera L, Spicka
J, Angelisova P, Milgram SL and Horejsi V: Interaction between two
adapter proteins, PAG and EBP50: a possible link between membrane
rafts and actin cytoskeleton. FEBS Lett. 507:133–136. 2001.
View Article : Google Scholar
|
|
4
|
Kawabuchi M, Satomi Y, Takao T, Shimonishi
Y, Nada S, Nagai K, Tarakhovsky A and Okada M: Transmembrane
phosphoprotein Cbp regulates the activities of Src-family tyrosine
kinases. Nature. 404:999–1003. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takeuchi S, Takayama Y, Ogawa A, Tamura K
and Okada M: Transmembrane phosphoprotein cbp positively regulates
the activity of the carboxyl-terminal src kinase Csk. J Biol Chem.
275:29183–29186. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brdicka T, Pavlistova D, Leo A, Bruyns E,
Korinek V, Angelisova P, Scherer J, Shevchenko A, Hilgert I, Cerny
J, Drbal K, Kuramitsu Y, Kornacker B, Horejsi V and Schraven B:
Phosphoprotein associated with glycosphingolipid-enriched
microdomains (PAG), a novel ubiquitously expressed transmembrane
adaptor protein, binds the protein tyrosine kinase csk and is
involved in regulation of T cell activation. J Exp Med.
191:1591–1604. 2000. View Article : Google Scholar
|
|
7
|
Torgersen KM, Vang T, Abrahamsen H, Yaqub
S, Horejsi V, Schraven B, Rolstad B, Mustelin T and Tasken K:
Release from tonic inhibition of T cell activation through
transient displacement of C-terminal Src kinase (Csk) from lipid
rafts. J Biol Chem. 276:29313–29318. 2001. View Article : Google Scholar
|
|
8
|
Jiang LQ, Feng X, Zhou W, Knyazev PG,
Ullrich A and Chen Z: Csk-binding protein (Cbp) negatively
regulates epidermal growth factor-induced cell transformation by
controlling Src activation. Oncogene. 25:5495–5506. 2006.
View Article : Google Scholar
|
|
9
|
Smida M, Posevitz-Fejfar A, Horejsi V,
Schraven B and Lindquist JA: A novel negative regulatory function
of the phosphoprotein associated with glycosphingolipid-enriched
microdomains: blocking Ras activation. Blood. 110:596–615. 2007.
View Article : Google Scholar
|
|
10
|
Durrheim GA, Garnett D, Dennehy KM and
Beyers AD: Thy-1 associated pp85–90 is a potential docking site for
SH2 domain-containing signal transduction molecules. Cell Biol Int.
25:33–42. 2001.PubMed/NCBI
|
|
11
|
Sanchez-Margalet V and Najib S: Sam68 is a
docking protein linking GAP and PI3K in insulin receptor signaling.
Mol Cell Endocrinol. 183:113–121. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang KT, Chen YH and Walker AM:
Inaccuracies in MTS assays: major distorting effects of medium,
serum albumin, and fatty acids. Biotechniques. 37:406–412.
2004.PubMed/NCBI
|
|
13
|
Simeoni L, Smida M, Posevitz V, Schraven B
and Lindquist JA: Right time, right place: the organization of
membrane proximal signaling. Semin Immunol. 17:35–49. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cloutier JF, Chow LM and Veillette A:
Requirement of the SH3 and SH2 domains for the inhibitory function
of tyrosine protein kinase p50csk in T lymphocytes. Mol Cell Biol.
15:5937–5944. 1995.
|
|
15
|
Itoh K, Sakakibara M, Yamasaki S, Takeuchi
A, Arase H, Miyazaki M, Nakajima N, Okada M and Saito T: Cutting
edge: negative regulation of immune synapse formation by anchoring
lipid raft to cytoskeleton through Cbp-EBP50-ERM assembly. J
Immunol. 168:541–544. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Harder T and Simons K: Clusters of
glycolipid and glycosylphosph atidylinositol-anchored proteins in
lymphoid cells: accumulation of actin regulated by local tyrosine
phosphorylation. Eur J Immunol. 29:556–562. 1999. View Article : Google Scholar
|
|
17
|
Condeelis JS, Wyckoff JB, Bailly M,
Pestell R, Lawrence D, Backer J and Segall JE: Lamellipodia in
invasion. Semin Cancer Biol. 11:119–128. 2001. View Article : Google Scholar : PubMed/NCBI
|