Introduction
Chuanxiong (Ligusticum wallichi Franchat) has been
commonly used in Chinese traditional medicine for >2,000 years.
This medicine’s bioactive component, tetramethylpyrazine (TMP), was
identified and extracted from Chuanxiong in 1973 (1). There have been an overwhelming number
of applications of tetramethylpyrazine hydrochloride (TMPH) as a
highly efficient clinical treatment for ischemic neurovascular
diseases with only mild side effects (2–4).
Moreover, many studies have characterised the effects of TMP on
tumours as inhibiting the viability and attenuating the metastasis
of cancer cells (5,6). Clinical evidence has confirmed that
combining TMPH or Chuanxiong with other treatments can
significantly attenuate multidrug resistance (MDR) of chemotherapy
and increase the sensitivity of cancer cells to radiation in such
cancers as nasopharyngeal, lung, breast, renal and ovarian cancer
(7–9). Wang et al also confirmed by
glioma-neuronal co-culturing that TMP could inhibit the viability
of glioma cells while protecting hippocampal neurons and TMP could
promote the regression of malignant gliomas in vivo(10). Therefore, TMP may be a potential
therapeutic agent for the treatment of gliomas.
Many previous studies have demonstrated that TMP can
reduce blood viscosity, manage coagulation and improve the
microcirculation around ischaemic tissue (11,12).
Therefore, most research groups hypothesise that TMP may increase
the radiation and chemotherapy sensitivity of patients by improving
the microcirculation around tumour tissue, thereby reducing the
side effects on the organs at risk (7,13).
Thus, it seems logical to suggest that a correlation exists between
the tumour-inhibition and neural-protection bioactivities of TMP.
However, the exact molecular mechanisms underlying the actions of
TMP have not been fully elucidated.
CXCR4 is a G-protein-coupled receptor with 7
transmembrane-spanning domains that is expressed in various
endothelial and tumour cells. Previous studies have demonstrated
that CXCR4 plays an important role in all three fundamental aspects
of cancer: proliferation, migration, invasion and metastases
(13–17). Zhou et al confirmed that
CXCR4 was a major chemokine receptor in glioma cells and mediated
their survival (13). A recent
study reported that the clinically approved drug, AMD3100 (a small
molecule inhibitor of SDF-1/CXCR4 interactions), could
significantly prevent the recurrence of glioblastoma in mice after
irradiation (18). Currently, CXCR4
has been targeted in the treatment of brain tumours (19–21).
In addition, CXCR4 takes different roles when modulating
neurotransmission, neurotoxicity and neurological interactions in
the mature central nervous system (CNS) (22,23).
Abnormal activation of CXCR4 occurs in the pathogenesis of CNS
disorders such as stroke, ischemia and multiple sclerosis.
Therefore, to determine if TMP regulates CXCR4 expression in
glioma, this study was undertaken to test the clinical-grade
product, TMPH, both in vitro and in vivo.
Materials and methods
Cell culture
C6 rat glioma cells were obtained from the ATCC
Collection (Manassas, VA) and maintained in DMEM (Invitrogen, USA)
supplemented with 10% FBS plus 100 U/ml penicillin and 100 mg/ml
streptomycin in a humidified atmosphere of 5% CO2 at
37°C. TMPH was purchased from Harbin Medisan Pharmaceutical Co.
(China), AMD3100 was purchased from Sigma (USA), and both were
dissolved in normal saline to appropriate concentrations. Normal
saline was applied as the control in all experiments.
Real-time RT-PCR analysis
Total RNA was isolated with TRIzol Reagent
(Invitrogen). Total RNA (1 μg) was subjected to reverse
transcription using the SYBR PrimeScript™ RT-PCR kit (Takara,
China) following the manufacturer’s protocol. Real-time PCR was
employed to measure CXCR4, SDF-1 and VEGF expression using the SYBR
Green system (Takara). The following primer pairs were used: for
CXCR4, 5′-cttatcctgcctggtattgtc-3′ and 5′-caatgtagtaaggcagccaac-3′;
for SDF-1, 5′-gtcagcctgagctacagatgc-3′ and
5′-ttgtttaaggctttgtccaggt-3′; for VEGF,
5′-attgagaccctggtggacatct-3′ and 5′-tctctcctatgtgctggctttg-3′; and
for β-actin, 5′-caccacaccttctacaatgag-3′ and
5′-tagcacagcctggatagcaac-3′. The quantity of target gene mRNA
relative to the internal control gene, β-actin, was calculated
using the ΔCT method, as follows: the relative expression =
2−ΔCT, ΔCT = CT(target gene) -
CT(β-actin). The data were analysed in triplicate.
Western blot analyses
Cells were lysed with radio-immunoprecipitation
assay buffer. Whole-cell lysates were separated by sodium dodecyl
sulphate/polyacrylamide electrophoresis and transferred after 1 h
to a nitrocellulose polyvinylidene fluoride membrane. CXCR4 was
detected using primary antibodies against CXCR4 (1:100; Santa Cruz,
USA) and a horseradish peroxidase-conjugated goat anti-rabbit
secondary antibody (1:500; Santa Cruz). β-actin served as a loading
control. Protein bands were detected using an enhanced
chemiluminescence detection system (Santa Cruz, USA).
Cell migration assay
The wound healing assay was performed to quantify
the rate of glioma cell migration. C6 glioma cells
(1×106) were seeded in a 60-mm dish to create a
confluent monolayer. After treatment with 200 μM TMPH, 10 μg/ml
AMD3100 or the control vehicle for 48 h, a wound was created by
manually scraping the cell monolayer with a p200-pipette tip. The
initial wound quantification was performed on images collected 1 h
after wounding when the wound size had stabilised. Additional
images were collected randomly from the wound areas at 6 and 12 h
after wounding.
Cell cycle assay
After treatment with 200 μM TMPH, 10 μg/ml AMD3100
or the control vehicle for 48 h, the cells were harvested, fixed
with 75% ice-cold ethanol in PBS and stored at 4°C. Before
analysis, the cells were washed twice with PBS and were incubated
for 30 min in a propidium iodide staining solution (0.05 mg/ml
propidium iodide, 1 mM EDTA, 0.1% Triton-X-100™ and 1 mg/ml
ribonuclease A) (Sigma-Aldrich, USA). The staining fluorescence
intensity was measured by BD FACSort™ (BD Biosciences, USA) and
used to determine the G2/M ratio.
Viability assays by MTT
C6 glioma cells (5×103) were seeded in a
96-well plate in 100 μl of medium 1 day prior to addition of 200 μM
TMPH, 10 μg/ml AMD3100 or the control vehicle. Cell viability was
determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenylte
tetrazolium bromide (MTT) assay using the optical density ratio of
a treated culture over an untreated control.
Colony formation assay
C6 glioma cells (1,000) were seeded in 6-well
culture plates in triplicate. After 10 days growth with 200 μM
TMPH, 10 μg/ml AMD3100 or the control vehicle, the cells were
stained with Giemsa for 20 min. The colony formation rate was
calculated using Image Pro Plus software (Media Cybernetics)
(24).
Tumour implantation
Thirty adult SD rats, each weighing 250–300 g, were
provided by the Laboratory Animal Centre of Southern Medical
University (Guangzhou, China). The rats were reared in the
laboratory animal centre of the Zhongshan Ophthalmic Centre. All
experimental procedures were approved by The Ethics Committee of
Zhongshan Ophthalmic Centre. Glioma cells (~106) were
injected into the frontal lobe of the right cerebral hemisphere of
the SD rats using a stereotaxic apparatus. Coordinates were set
according to the atlas of Paxinos and Watson (25). Seven days after tumour implantation,
the SD rats were randomised (15 rats for each group) to a treatment
group receiving daily i.p. injections of TMPH at a dose of 100
mg/kg in 500 μl for 20 days or to a control groups receiving i.p.
injections of the control vehicle.
Blood vessel staining
To assess tumour angiogenesis, the rats were
anesthetised, and 0.1 ml of FITC-dextran solution (50 mg/ml in PBS;
Sigma-Aldrich, USA) was administered retro-orbitally. Within 5 min
after the injection, animals were sacrificed by cervical
dislocation to prevent the FITC-dextran from diffusing distantly
from the perfused vessels. Next, the brains were rapidly removed
from the cranial cavity and fixed with 4% paraformaldehyde at 4°C
for 24 h. Coronal sections (30 μm) were cut and observed under a
fluorescence microscope. The tumour angiogenesis was analysed using
Image Pro Plus software (24).
Statistical analyses
All experiments in vitro were carried out in
triplicate. Data are expressed as means ± SE. The differences
between mean values were evaluated with a two-tailed Student’s
t-test (for 2 groups) and the analysis of variance (ANOVA, for
>2 groups). All calculations and statistical tests were
performed using Excel 2003 (Microsoft) or SPSS 11.5 (SPSS). A
p<0.05 was considered to be significant for all analyses.
Results
TMPH treatment reduces CXCR4 expression
in C6 glioma cells
Considering that CXCR4, stromal cell-derived
factor-1 (SDF-1) and VEGF play important roles in cell migration
and proliferation, we first measured the expression of CXCR4, SDF-1
and VEGF in C6 glioma cells treated with 100 μM TMPH. Forty-eight
hours after treatment, total RNA was extracted for analysis by
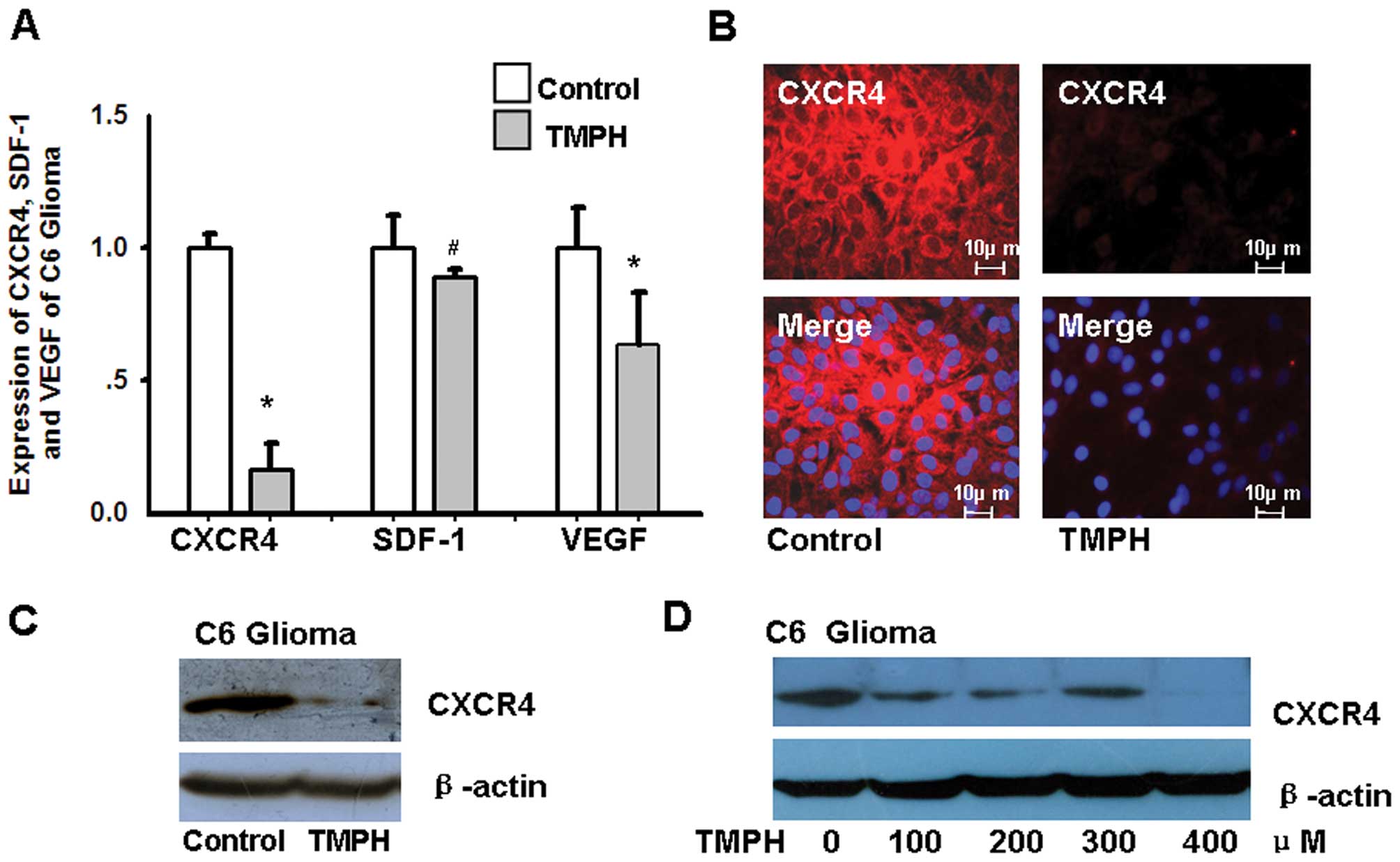
real-time RT-PCR. As shown in Fig.
1A, the expression of CXCR4 and VEGF in C6 glioma cells is
down-regulated after TMPH treatment; the decrease in CXCR4
expression is 3.94-fold greater than the decrease in VEGF
expression. CXCR4 is the only known receptor for SDF-1, and SDF-1
expression is only slightly altered upon TMPH treatment. The
analysis of immunofluorescence staining and western blot data
further confirms that TMPH significantly inhibits CXCR4 expression
in C6 glioma cells (Fig. 1B and C).
Moreover, TMPH treatment downregulates the expression of CXCR4 in
glioma cells in a dose-dependent manner (Fig. 1D).
TMPH inhibits cell migration and
viability
The SDF-1/CXCR4 interaction plays a pivotal role in
cell migration. AMD3100 is a specific inhibitor of CXCR4.
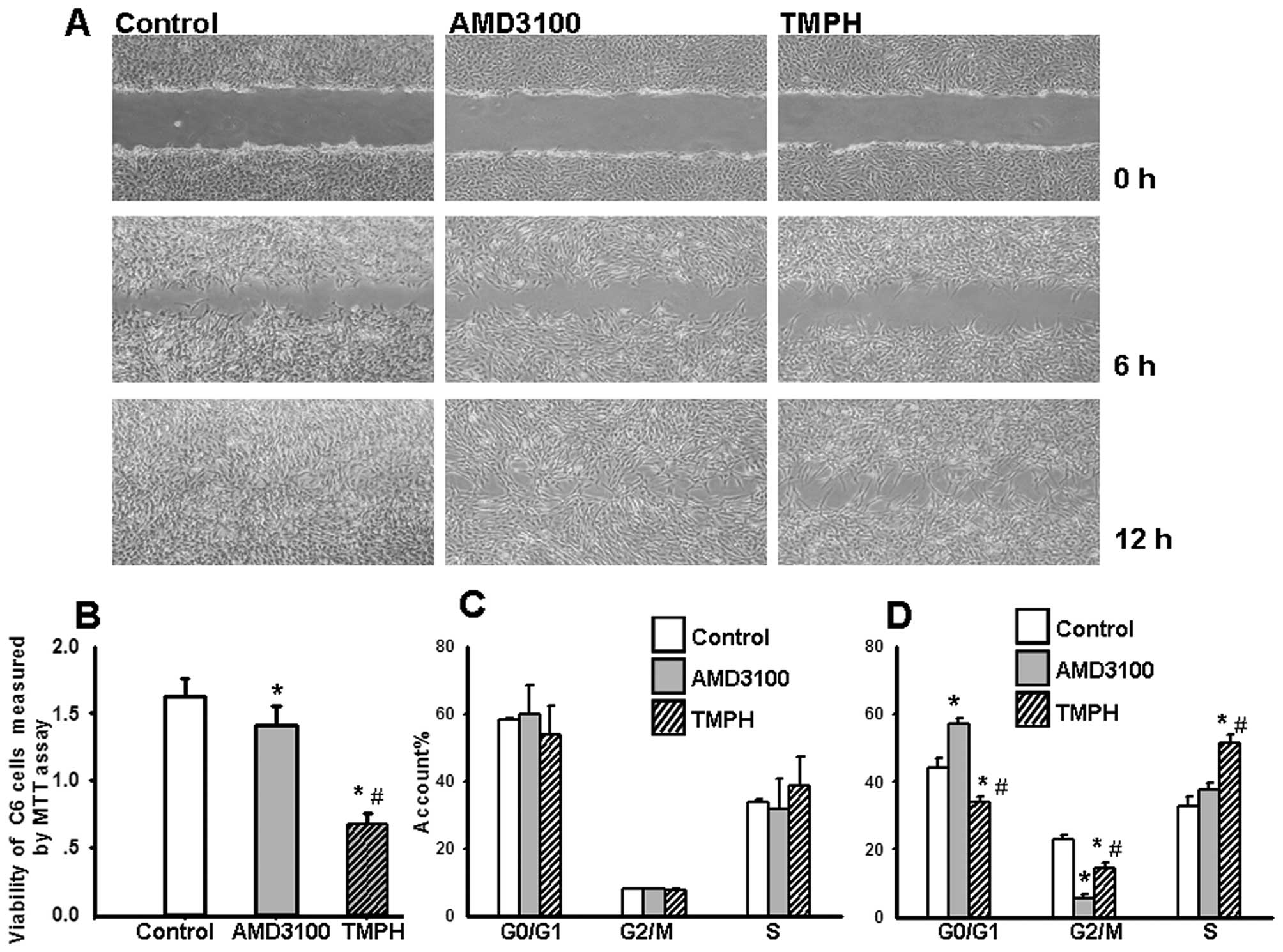
Therefore, the effects of TMPH (200 μM) on cell migration were
evaluated using a scratch-wound assay, and the results were
compared to those from cells treated with 10 μg/ml of AMD3100. As
shown in Fig. 2A, TMPH
significantly decreases the migration of C6 glioma cells compared
to the control. More open spaces are observed at 6 and 12 h after
treatment with TMPH. Moreover, the inhibition of cell migration by
TMPH is more effective than that of AMD3100. Cell viability was
measured by staining with MTT. Fig.
2B shows that TMPH inhibits the viability of cultured glioma
cells and that this inhibition is much stronger than that caused by
AMD3100.
Previous data regarding the effects of AMD3100 on
the cell cycle were controversial (13,26,27).
To determine the bioactivity of TMPH on cell growth and to compare
the bioactivity of TMPH and AMD3100, we analysed the cell cycle
profile of C6 glioma cells at different confluencies. As shown in
Fig. 2C, at 48 h after treatment,
when the cells are 50–80% confluent, TMPH does not alter the cell
cycle profile of glioma cells. However, when the cells are 100%
confluent, TMPH treatment induces an arrest in S phase
(51.52±2.29%) compared to treatment with AMD3100 (37.5±2.63%) and
the control (32.95±1.28%). The rate of cell growth in the G1
(34.10±1.53%) and G2 phases (14.03±1.54%) is significantly reduced
in glioma cells treated with TMPH compared to the control (G1 phase
44.22±1.70%, G2 phase 22.83±2.16%). AMD3100 does not affect S phase
(37.51±2.62%), but there are more cells in G1 phase (57.10±2.88%)
(p<0.001) and fewer in G2 phase (5.39±1.51%) (p<0.001)
(Fig. 2D). These data indicate that
TMPH and AMD3100 affect different phases of the cell cycle.
TMPH inhibits colony formation of C6
glioma cells
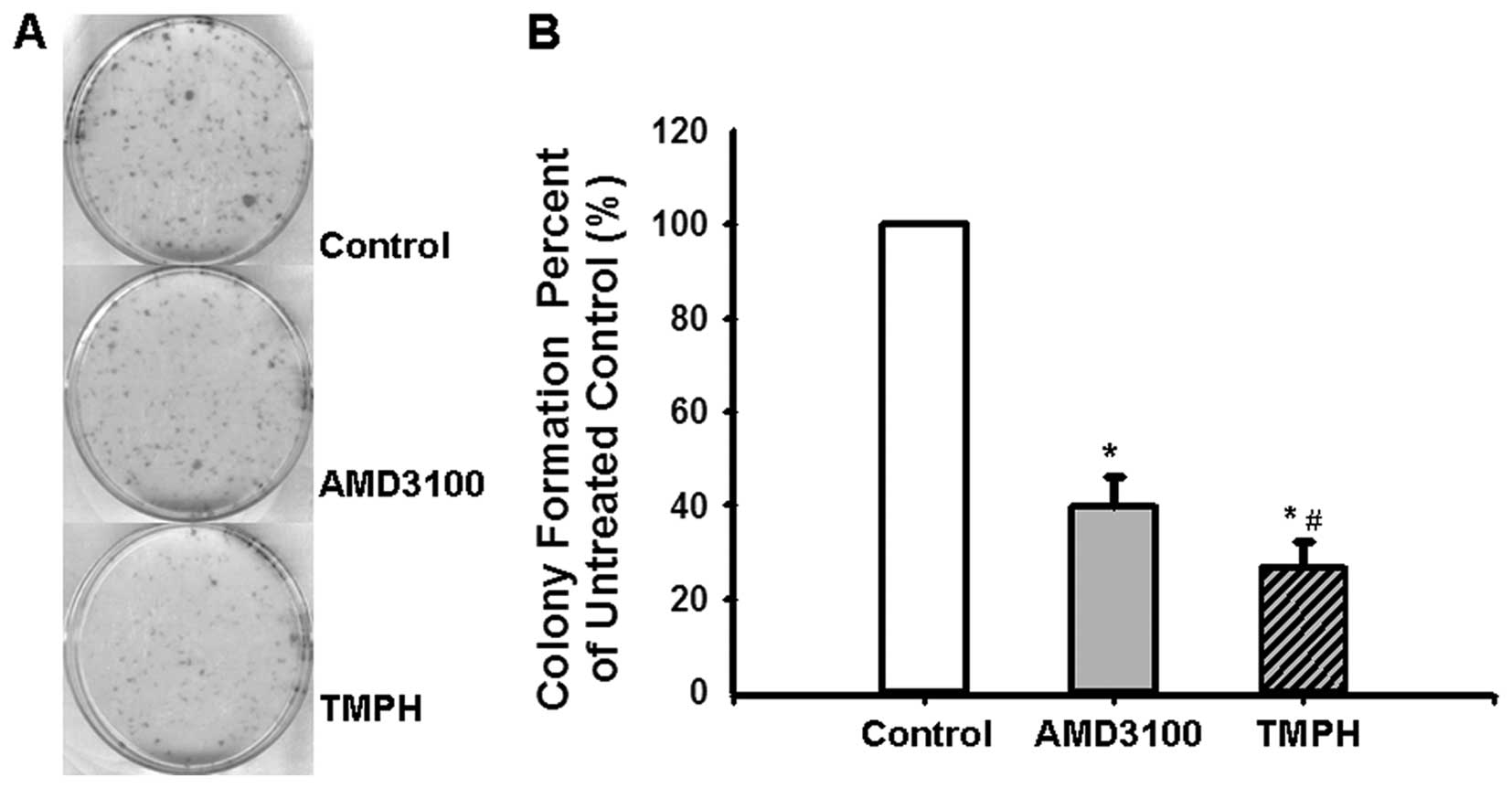
There is increasing evidence that the capacity of
cancer cells to form colonies is critical for the process of tumour
development (28,29). To determine the impact of TMP on
colony formation, 1,000 C6 glioma cells were seeded into a 6-well
plate, and colony formation was recorded after 10 days, at which
time colonies are visible. As shown in Fig. 3, TMPH effectively inhibits colony
formation of the seeded cells compared with the control. Moreover,
the colony formation rate was significantly decreased in glioma
cells treated with TMPH (26.49±5.65%) compared with AMD3100
(39.68±6.50%).
TMPH causes malignant glioma regression
in vivo
To validate our findings that TMPH-mediated
inhibition of glioma cells involves inhibition of CXCR4-dependent
migration in vivo, ~106 glioma cells were
implanted into the frontal lobe of the left cerebral hemisphere of
SD rats using a stereotaxic apparatus. Seven days after tumour
implantation, the SD rats were randomised to treatment groups
receiving i.p. injections of TMPH or normal saline daily for 20
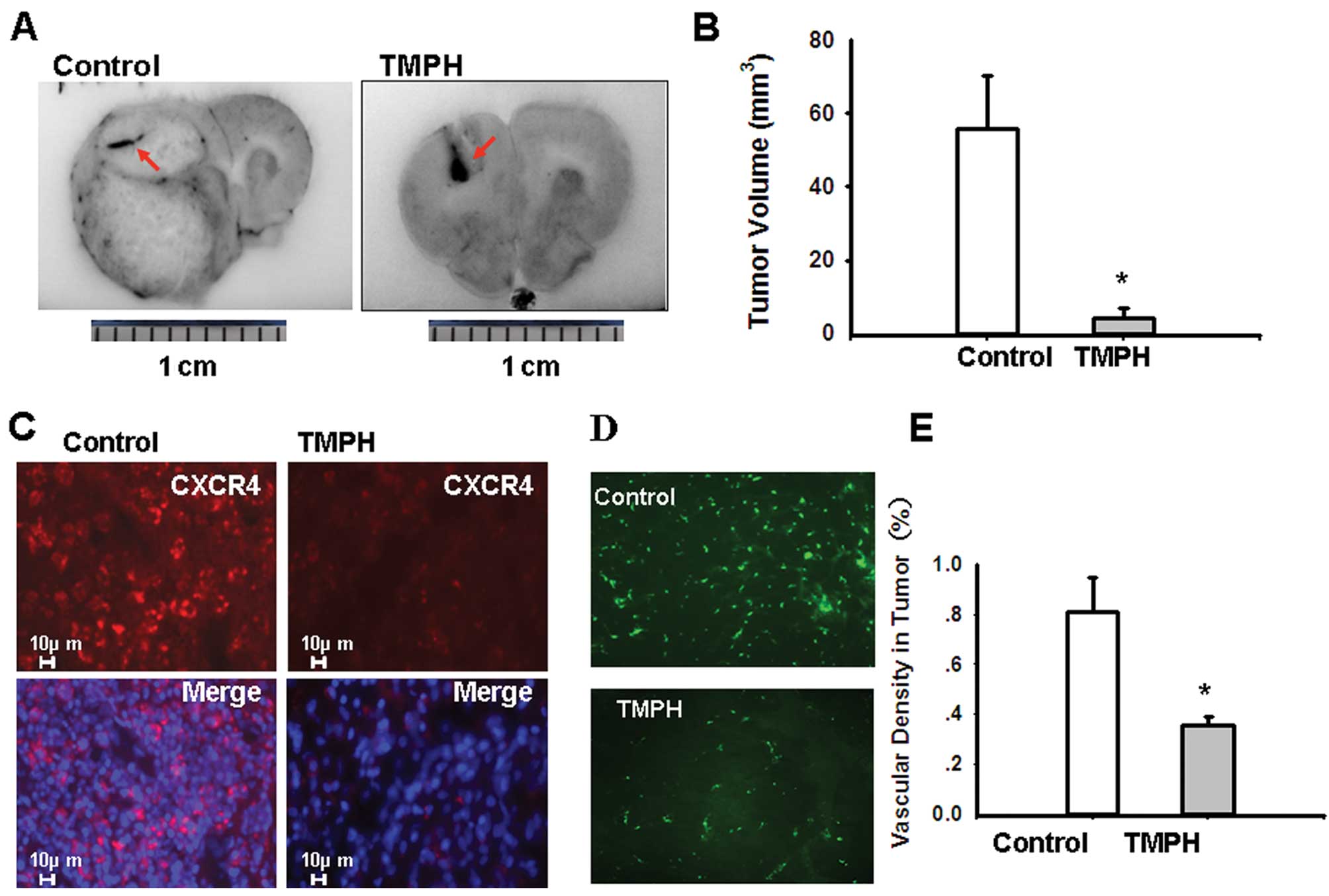
days. Fig. 4A shows that the size
of a C6 glioma implantation treated with TMPH is significantly
smaller than that of the control. There is obvious haemorrhaging in
the C6 glioma implantation after TMPH treatment (red arrowhead).
Tumour growth is significantly inhibited in rats treated with 0.8
mg TMPH day-1 (4.14±2.81 mm3) compared with the growth
in control rats (55.9±14.12 mm3) (p<0.001) (Fig. 4B). These data are consistent with a
previous report on the effects of TMP treatment in a rat model of
C6 glioma (10). Consistent with
our results from C6 glioma cells in vitro, TMPH
downregulates CXCR4 in glioma implantation compared with the
control (Fig. 4C).
To verify the role of CXCR4 signalling during
vascular response in vivo, FITC-dextran was injected as a
perfusion marker before the tumours were harvested. As shown in
Fig. 4D, the microcirculation in
the tumour is denser in the control rats than in the TMPH-treated
rats. Fig. 4E shows the
quantification of microvasculature formation as measured by
microscopy (p<0.001).
Discussion
In the present study, we demonstrated that the
CXCR4/SDF-1 pathway is a novel mechanism underlying TMP-mediated
glioma inhibition. Our results demonstrated that TMPH significantly
decreased CXCR4 expression from rat C6 glioma cells (Fig. 1A–C). We also found that TMPH
treatment downregulates the VEGF expression in C6 glioma cells
(Fig. 1A), which has previously
been considered to be the main factor in TMP treatment of tumours.
However, the decrease in CXCR4 expression induced by TMPH is
3.94-fold greater than the decrease in VEGF expression. Moreover, a
recent study demonstrated that the CXCR4/SDF-1 axis was the major
cause of ectopic overexpression of VEGF in tumours. The CXCR4/SDF-1
axis regulates VEGF expression through Yin Yang 1 (YY1), which is a
positive regulator of VEGF (30).
Based on this evidence, we deduce that the downregulation of CXCR4
induced by TMP treatment occurs prior to reducing VEGF
expression.
SDF-1/CXCR4 plays a key role in promoting the
migration and growth of tumour cells (17). Our study demonstrated that TMPH
treatment significantly decreased the migration and viability of C6
glioma cells (Fig. 2A). Moreover,
this inhibition by TMPH is more effective than the CXCR4
antagonist, AMD3100.
We analysed the cell cycle profiles of C6 glioma
cells after TMPH treatment. We found that TMPH and AMD3100 do not
affect the cell cycle when the cells are 50–80% confluent. However,
at 100% confluency, TMPH induces arrest in S phase, significantly
reducing the G1 and G2 populations of C6 glioma cells compared with
that of the control. Similarly, AMD3100 also affects the cell cycle
when the cells are 100% confluent. These results are consistent
with the previous controversial reports on the effects of AMD3100
(13,26,27) on
the cell cycle. TMP and AMD3100 affect cell cycle in a
dose-dependent manner in some studies but not in others. Moreover,
there is a significant difference in cell cycle profiles between
the treatment with TMP and AMD3100 at 100% confluency. The cells
treated with AMD3100 were arrested in the G1 phase compared with
the control. Therefore, the basis of the fundamental physiological
roles of TMPH and AMD3100 in the cell cycle or cell migration may
be that TMP directly downregulates CXCR4, while AMD3100 directly
blocks the active domain of CXCR4.
We further evaluated the bioactivity of TMPH on C6
glioma cell implantation in vivo. Our data show that tumour
growth is significantly inhibited in rats treated with TMPH
compared with the control, which is in agreement with previous
reports (10,18). Consistent with our in vitro
results with C6 glioma cells, the expression of CXCR4 is abolished
in C6 glioma cell implantation after TMPH treatment.
Moreover, we also confirmed that TMPH could protect
cerebral neurocytes by downregulating the expression of CXCR4 and
inhibiting glutamate release from cerebral neurocytes in
vitro (unpblished data), which build up a correlation existing
between the tumour-inhibition and neural-protection bioactivities
of TMP.
In conclusion, we believe we have shown for the
first time that TMP treatment results in inhibition of cell
migration, thereby blocking the cell cycle and eventually
suppressing tumour growth by downregulating CXCR4 expression in
glioma cells both in vitro and in vivo. This finding
is in contrast to the widely held notion that TMP exerts
bioactivity only through improving microcirculation (7,13).
Additionally, TMPH could be a potential agent for treatment of
malignant brain tumours.
Acknowledgements
This study was supported was supported by the grant
from the National Natural Science Foundation (Project:
30872811).
References
|
1
|
|
|
2
|
Liu SF, Cai YN, Evans TW, et al:
Ligustrazine is a vasodilator of human pulmonary and bronchial
arteries. Eur J Pharmacol. 191:345–350. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shen SY, Fu XD and Fei ZY: Assessment and
explorations on the mechanism of neuroprotection of patients in
ischemic stroke by traditional Chinese medicine. Chin J Integr Med.
11:237–240. 2005.(In Chinese).
|
|
4
|
Han Z, Wang Q, Gao F, Ren Y and Yang KJ:
The clinic evaluation of treatment of Ligustrazine and Radix
Astragalus to 36 patients. J Fourth Military Medical University.
27:1207–1209. 2006.(In Chinese).
|
|
5
|
Wang XB, Wang SS, Zhang QF, et al:
Inhibition of tetramethylpyrazine on P-gp, MRP2, MRP3 and MRP5 in
multidrug resistant human hepatocellular carcinoma cells. Oncol
Rep. 23:211–215. 2010.PubMed/NCBI
|
|
6
|
Mei Y, Shi Y, Zuo G, Gong J and Liu C:
Study on ligustrazine in reversing multidrug resistance of
HepG2/ADM cell in vitro. Zhongguo Zhong Yao Za Zhi. 29:970–973.
2004.PubMed/NCBI
|
|
7
|
Li Z, Shao M, Zhou Y, Wei R and Gong Y:
Resistance-related protein in rectal cancer tissue expression and
significance of combination chemotherapy with Ligustrazine. Int Med
Health Guidance News. 2:142–144. 2010.(In Chinese).
|
|
8
|
Li L, Xu K, Liu Y, Zheng J, Qiu S and Liu
C: The clinic evaluation of treatment of percutaneous implantation
of radioactive particles with the TMP to 20 patients with malignant
cancer by CT-guided. Shaanxi J Trad Chin Med. 29:542–544. 2008.
|
|
9
|
Wu Y, Dai X, Hu P and Chen Z: The
progresses of Chinese traditional medicine on brain tumors. J New
Chin Med. 38:76–79. 2004.
|
|
10
|
Fu YS, Lin YY, Chou SC, et al:
Tetramethylpyrazine inhibits activities of glioma cells and
glutamate neuro-excitotoxicity: potential therapeutic application
for treatment of gliomas. Neurooncology. 10:139–152. 2008.
|
|
11
|
Li M, Handa S, Ikeda Y and Goto S:
Specific inhibiting characteristics of tetramethylpyrazine, one of
the active ingredients of the Chinese herbal medicine ‘Chuanxiong’,
on platelet thrombus formation under high shear rates. Thromb Res.
104:15–28. 2001.PubMed/NCBI
|
|
12
|
Liu Y, Ren J, Jiang H, Du S and Zhang Y:
The influenced foundation of hemodynamics during recovery of blood
stasis rats by gustrazine and angelica parenteral solution. Chin J
Med Physics. 22:680–681. 2005.
|
|
13
|
Zhou Y, Larsen PH, Hao C, et al: CXCR4 is
a major chemokine receptor on glioma cells and mediates their
survival. J Biol Chem. 277:49481–49487. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang X and Chen X: Study on the effects of
tetramethylpyrazine on tumor cells: survey and prospects. Zhongguo
Zhong Yao Za Zhi. 28:295–298. 2003.PubMed/NCBI
|
|
15
|
Muller A, Homey B, Soto H, et al:
Involvement of chemokine receptors in breast cancer metastasis.
Nature. 410:50–56. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Righi E, Kashiwagi S, Yuan J, et al:
CXCL12/CXCR4 blockade induces multimodal antitumor effects that
prolong survival in an immunocompetent mouse model of ovarian
cancer. Cancer Res. 71:5522–5534. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ping YF, Yao XH, Jiang JY, et al: The
chemokine CXCL12 and its receptor CXCR4 promote glioma stem
cell-mediated VEGF production and tumour angiogenesis via PI3K/AKT
signaling. J Pathol. 224:344–354. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kioi M, Vogel H, Schultz G, et al:
Inhibition of vasculogenesis, but not angiogenesis, prevents the
recurrence of glioblastoma after irradiation in mice. J Clin
Invest. 120:694–705. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Terasaki M, Sugita Y, Arakawa F, Okada Y,
Ohshima K and Shigemori M: CXCL12/CXCR4 signaling in malignant
brain tumors: a potential pharmacological therapeutic target. Brain
Tumor Pathol. 28:89–97. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schulte A, Gunther HS, Phillips HS,
Kharbanda S, Soriano RH, Modrusan Z, Zapf S, Westphal M and Lamszus
K: A distinct subset of glioma cell lines with stem cell-like
properties reflects the transcriptional phenotype of glioblastomas
and overexpresses CXCR4 as therapeutic target. Glia. 59:590–602.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schulte A, Gunther HS, Phillips HS, et al:
CXCL12 (SDF1alpha)-CXCR4/CXCR7 pathway inhibition: an emerging
sensitizer for anticancer therapies? Clin Cancer Res. 17:2074–2080.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Calì C and Bezzi P: CXCR4-mediated
glutamate exocytosis from astrocytes. J Neuroimmunol. 224:13–21.
2010.PubMed/NCBI
|
|
23
|
Cartier L, Hartley O, Dubois-Dauphin M and
Krause KH: Chemokine receptors in the central nervous system: role
in brain inflammation and neurodegenerative diseases. Brain Res
Brain Res Rev. 48:16–42. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhou Q, Kiosses WB, Liu J and Schimmel P:
Tumor endothelial cell tube formation model for determining
anti-angiogenic activity of a tRNA synthetase cytokine. Methods.
44:190–195. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Paxinos G and Watson C: The rat brain in
stereotaxic coordinates. 2nd edition. Academic Press; San Diego,
CA: pp. 567–572. 1986
|
|
26
|
Redjal N, Chan JA, Segal RA and Kung AL:
CXCR4 inhibition synergizes with cytotoxic chemotherapy in gliomas.
Clin Cancer Res. 12:6765–6771. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rubin JB, Kung A, Klein RS, et al: A
small-molecule antagonist of CXCR4 inhibits intracranial growth of
primary brain tumors. Proc Natl Acad Sci USA. 100:13513–13518.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Egeblad M and Werb Z: New functions for
the matrix metalloproteinases in cancer progression. Nat Rev
Cancer. 2:161–174. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shoemaker RH, Wolpert-DeFilippes MK, Kern
DH, et al: Application of a human tumor colony-forming assay to new
drug screening. Cancer Res. 45:2145–2153. 1985.PubMed/NCBI
|
|
30
|
Nigris Fd, Crudele V, Giovane A, et al:
CXCR4/YY1 inhibition impairs VEGF network and angiogenesis during
malignancy. Proc Natl Acad Sci USA. 107:14484–14489. 2010.
View Article : Google Scholar : PubMed/NCBI
|