Introduction
Breast cancer is a major life-threating disease in
females worldwide (1). Despite
current advances in chemotherapy, the outcome for patients with
metastatic breast cancer remains unsatisfactory. Certain patients
refuse to accept this type of treatment due to the severe
side-effects. Therefore, a more effective chemotherapeutic
combination treatment associated with manageable toxicity is highly
desired. As a potential strategy, suicide gene therapy is a
promising therapeutic approach for improving treatment efficacy and
the application of nucleoside kinases as suicide genes in gene
therapy has been extensively studied. The basic concept is to
transduce cells with the gene encoding herpes simplex virus type I
thymidine kinase (HSV-TK) and subsequently expose the cells to a
ganciclovir (GCV) prodrug. To date, this enzyme-directed prodrug
therapeutic combination with HSV-TK/GCV has been approved for phase
III clinical trials.
With the development of suicide gene therapy, a
deoxyribonucleoside kinase of Drosophila melanogaster
(Dm-dNK) in cultured Drosophila S-2 cells has been
introduced (2). Compared with
HSV-TK and other enzymes, Dm-dNK has a remarkably broad
substrate specificity and a higher catalytic rate with the capacity
to phosphorylate all 4 deoxyribonucleosides when transferred to
human cells. The isolated kinase has a preference for a pyrimidine
nucleoside, such as brivudine [BVDU,
(E)-5-(2-bromovinyl)-2′-deoxyuridine]. It has been confirmed that
the catalytic rate of Dm-dNK for pyrimidines is 100–600-fold
higher compared to purines (3).
Taken together, these data underline the importance of its
potential role as a suicide gene. Therefore, Dm-dNK has been
used successfully in suicide gene therapy against a wide variety of
cancers (4–8).
In terms of delivery vehicles, the efficacy of this
approach may be further enhanced. For this purpose, in this study,
we investigated recombinant vectors derived from
replication-defective adenovirus and lentivirus. Adenovirus is the
most commonly used viral delivery tool for gene therapy. However,
the major limitation in the application of this vector is the rapid
inactivation of adenovirus from systemic delivery, thus losing the
sustained gene expression (9–11). By
contrast, lentiviral vectors may be incorporated into the host
genome, thereby ensuring a prolonged gene expression with a limited
host immune response (12). To
date, the benefit of this therapy coupled with the
replication-defective adenoviral gene for breast cancer remains to
be elucidated.
The purpose of the present study was to compare the
efficacy of replication-defective adenoviral and lentiviral gene
therapy for breast cancer. The data presented in this study will
provide a theoretical reference for future studies on cancer
therapy.
Materials and methods
Cell line and cell culture
The Bcap37 human breast cancer cell line was
obtained from the Cancer Institute of the China Medical University
(Shenyang, China) and cultured in RPMI-1640 medium (Invitrogen
Corp., Carlsbad, CA, USA) supplemented with 10% fetal bovine serum,
100 U/ml penicillin and 100 μg/ml streptomycin at 37°C in a 5%
CO2 incubator.
Construction of viral vectors
The coding sequence of Dm-dNK was released
from plasmid PGEM-T-dNK with endonucleases EcoRI and
BamHI (New England Biolabs, Beverley, MA) and cloned
in-frame into plasmid Pentr13 [containing the sequence of a mouse
CMV promoter, His-tag(N) and multiple cloning sites] and plasmid
PGC-FU [Genechem, Shanghai, China; containing the sequence of a
mouse CMV promoter, multiple cloning sites and a green fluorescent
protein (GFP) sequence] to generate PENTR13-dNK and PGC-FU-dNK,
respectively. The fragment expression cassette of dNK containing a
mouse CMV promoter, His-tag(N) and the dNK gene from plasmid
PENTR13-dNK were excised and inserted into plasmid pShuttle-basic
(Sinogenomax, Inc., Beijing, China) to generate shuttle plasmid
pShuttle-basic-His-tag(N)-dNK (plasmid Ad-CMV-dNK). The fragment
expression of GFP from plasmid PGC-FU-dNK was removed with
endonucleases AgeI and EcoRI (NEB, UK) to generate
the recombinant plasmid PGC-FU-CMV-dNK (plasmid Lenti-CMV-dNK).
Plasmids PGC-FU, Ad-CMV-dNK and Lenti-CMV-dNK were individually
transfected into the human embryonic kidney HEK293 cells using
Lipofectamine™ 2000 reagent (Invitrogen Corp.) according to the
manufacturer's instructions. After homologous recombination, we
obtained a replication-deficient adenovirus known as Ad-CMV-dNK and
2 lentiviruses known as Lenti-GFP and Lenti-CMV-dNK. The lentivirus
and replication-deficient adenovirus harboring GFP alone (Lenti-GFP
and Ad-GFP) driven by the CMV promoter were used to determine the
infection rate of the Bcap37 cells.
Fluorescent-activated cell sorting (FACS)
analysis
The Bcap37 breast caner cells were seeded in 6-well
plates in 2 ml of growth medium at a population of 3×105
cells/well. After culturing for 24 h, Ad-GFP and Lenti-GFP were
added to the medium at a multiplicity of infection (MOI) of 1 and
10 to achieve the gene transfer efficiency. Three days later, all
cells were harvested and suspended in PBS with 0.5% bovine serum
albumin at the population of 1×106 cells/ml. The samples
were used to detect the expression profile of GFP by a FACScan flow
cytometer equipped with CellQuest and Modfit LT for Mac V1.01
software (Becton-Dickinson, San Jose, CA).
RT-PCR
Bcap37 cells were seeded in 6-well plates at a
density of 105 cells/well for 24 h. Subsequently,
Dm-dNK transduced by different viruses at a MOI of 10 was
added to the cell cultures. Polybrene (6 μg/ml) was added to all
cultures. Total RNA was isolated from the cultured cells using
TRIzol reagent (Sigma, USA) after 3 days of infection. The cDNA was
synthesized from 1 ng of RNA using an RT-PCR kit (Takara, Japan)
following the manufacturer's instructions. The primer sequences
were as follows: sense, 5′-CCG GAA TTC ACC ATG GAG GCA-3′ and
antisense, 5′-CGC GGA TCC TCA TTA TCT GGC GAC-3′ for Dm-dNK
(779 bp); sense, 5′-ACC ACA GTC CAT GCC ATC AC-3′ and antisense,
5′-TCC ACC ACC CTG TTG CTG TTG CTG TA-3′ for GAPDH (452 bp). The
PCR conditions were as follows: a pre-heating step at 94°C for 4
min, 35 cycles with denaturing at 94°C for 1 min, annealing at 60°C
(55°C for GAPDH) for 1 min and an extension at 72°C for 1.5 min.
PCR products were electrophoresed on a 2% agarose gel and the
optical density of the bands was determined. The PCR product of the
GAPDH gene was used as the internal control.
Activity assays of Dm-dNK
In order to assess the function of Dm-dNK
transduced by different viruses, we prepared cell extracts as
previously described (13) from
Bcap37 cells at 72 h after viral infection at a MOI of 10. The
activity of Dm-dNK was determined by DE-81 filter paper
assay using tritium-labeled substrates. Briefly, the assay was
conducted in a 35-ml reaction mixture containing 50 mM Tris-HCl at
pH 7.6, 5 mM MgCl2, 2 mM dithiothreitol, 15 mM NaF, 100
mM KCl, 5 mM ATP, 0.5 mg/ml BSA and 0.6 mg protein extract.
Equivalent amounts of unlabeled substrates were mixed with 2.5 mM
[methyl-3H]dThd (Moravek Biochem, CA, USA) and incubated
at 37°C for 10, 20 and 30 min. Subsequently, aliquots of the
reaction mixtures were applied to Whatman DE-81 filter paper disks
and dried for 1 h. After washing 3 times with 5 mM ammonium
formate, the filter bound with nucleoside monophosphates was eluted
with 0.5 M KCl, and the radioactivity was quantified using a
scintillation counter.
Cell viability and proliferation
analysis
Exponentially growing Bcap37 cells were plated into
96-well plates (104 cells/well) and incubated for 24 h.
The cells were then infected with Lenti-GFP, Ad-CMV-dNK and
Lenti-CMV-dNK at a MOI of 10. Post-transfection, the medium was
treated with BVDU at various concentrations ranging from 0.001 to
10 μM for 72 h. Prior to measurement, a volume of 20 μl of 5 mg/ml
tetrazolium salt 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl
tetrazolium bromide (MTT) (Promega, Madison, WI) in
phosphate-buffered saline (PBS) was added to each well. After 4 h
of incubation, the culture medium was removed and 200 μl of
dimethyl sulphoxide (DMSO) were added to dissolve the purple
crystal followed by vibration for 10 min. The absorbance was
determined at 570 nm to evaluate the viable cells. Each experiment
was performed in triplicate. In order to determine whether the
cells transfected with different viruses were functional, we
conducted proliferation assays in vitro. Briefly, Bcap37
cells transduced by Lenti-GFP, Ad-CMV-dNK and Lenti-CMV-dNK at a
MOI of 10 were seeded to 24-well plates with a pre-coating of BVDU
(1 μM). During the incubation, cells were trypsinized and suspended
in a serum-free medium. Cell counting was conducted under a
microscope using a hemocytometer.
Cell apoptosis assay
Quantitative assessment of apoptosis was conducted
by flow cytometry using the Annexin V-FITC/propidium iodide (PI)
double staining kit (Genmed Biosciences, China) according to the
manufacturer's instructions. Following transfection and treatment
with BVDU, Bcap37 cells were harvested, washed with PBS and
resuspended in 200 μl of binding buffer. A volume of 5 μl of
Annexin V-FITC and 10 μl of PI was added and the mixture was
incubated for 15 min in the dark. The apoptotic cells were analyzed
by a FACSCalibar equipped with CellQuest and Modfit LT for Mac
V1.01 software (Becton-Dickinson).
In vivo experiment
Female BALB/C nude mice 6–7 weeks old were purchased
from the Experimental Animal Center, Chinese Academy of Sciences
(Shanghai, China). All animals were handled strictly in compliance
with the Guidelines for the Care and Use of Laboratory Animals
(National Research Council, 1996). The mice were inoculated
subcutaneously in the flank with 1.0×107 Bcap37 cells.
When the average volume of tumors reached ~100 mm3, the
mice were randomly divided into 4 groups (6 mice/group) as follows:
i) BVDU with PBS; ii) BVDU with Lenti-GFP; iii) BVDU with
Ad-CMV-dNK and iv) BVDU with Lenti-CMV-dNK. The mice were injected
intratumorally with the viruses at a dose of 109 pfu 3
times with a 2-day interval. Subsequently, 5 mg/kg of BVDU was
administered daily into the peritoneal cavity for 7 consecutive
days. Tumor growth was monitored every 5 days and the dimension of
the tumors was recorded with a caliper for up to 30 days.
Statistical analysis
The data are expressed as the means ± SD and
analyzed using the statistical software SPSS (version 10.1,
Chicago, IL). P<0.05 was considered to indicate a statistically
significant difference.
Results
Dm-dNK expression
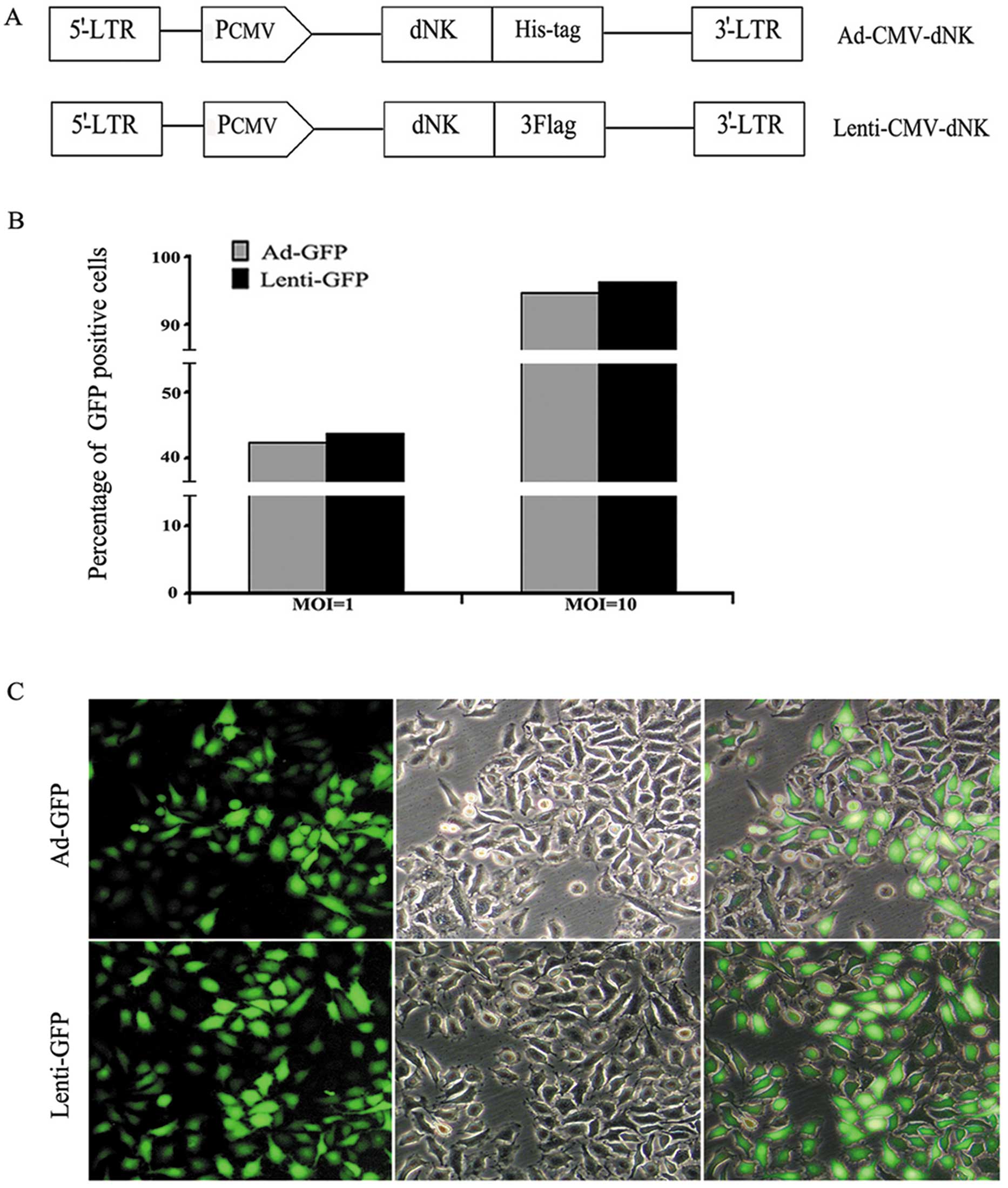
Dm-dNK-expressed replication-defective
adenoviral and lentiviral vectors were successfully constructed in
the 293T cells. Both expression cassettes contained a CMV promoter
required to drive the expression of the Dm-dNK gene
(Fig. 1A). The titers of both types
of viruses were 2×1011 and 2×109 TU/ml,
respectively. The infection efficiency was assessed by transducing
the Bcap37 human breast cancer cell line with adenoviral and
lentiviral vectors carrying the GFP construct (Fig. 1B). Compared with the vector
infection at a MOI of 1, a significantly higher population
(~90–95%) of GFP-positive cells was observed under the condition of
a vector infection at a MOI of 10. Moreover, both vectors were
efficiently transduced in the Bcap37 cell line at a MOI of 10.
Therefore, a MOI of 10 was the optimal dose for the transfection of
Bcap37 cells. Furthermore, no significant difference in the
infection efficiency at a MOI of 10 between both types of vectors
was observed; therefore, Lenti-GFP was selected as the
representative for the following studies. Similarly, as
demonstrated in Fig. 1C, GFP
expression was stronger at a MOI of 10 in the Bcap37 cells.
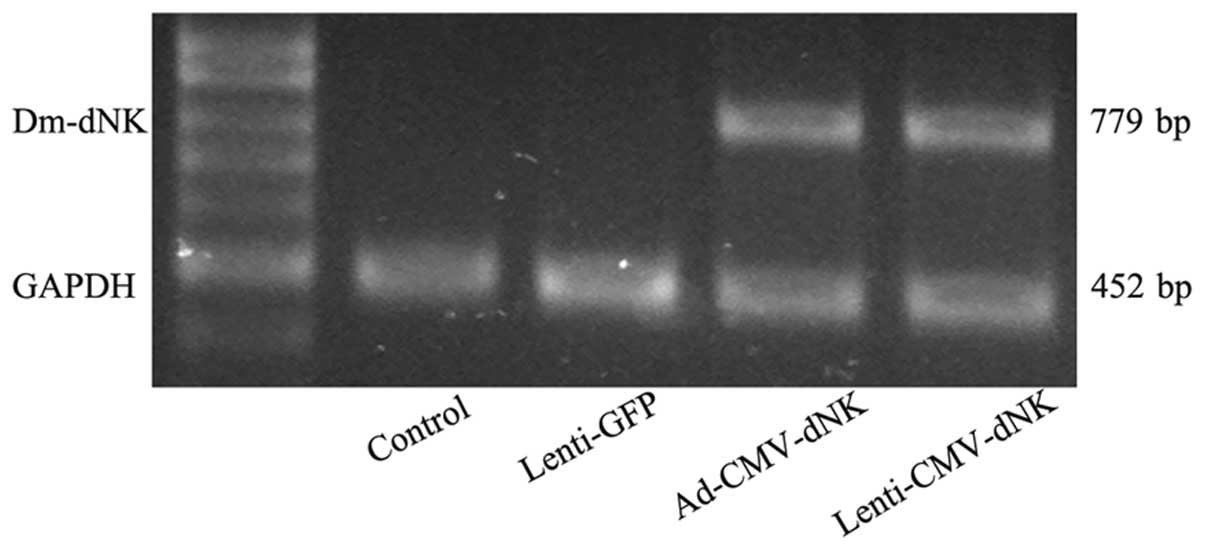
The expression of Dm-dNK was also evaluated
by RT-PCR. Bcap37 cells transduced with Ad-CMV-dNK and
Lenti-CMV-dNK revealed a significantly higher expression of
Dm-dNK mRNA (Fig. 2).
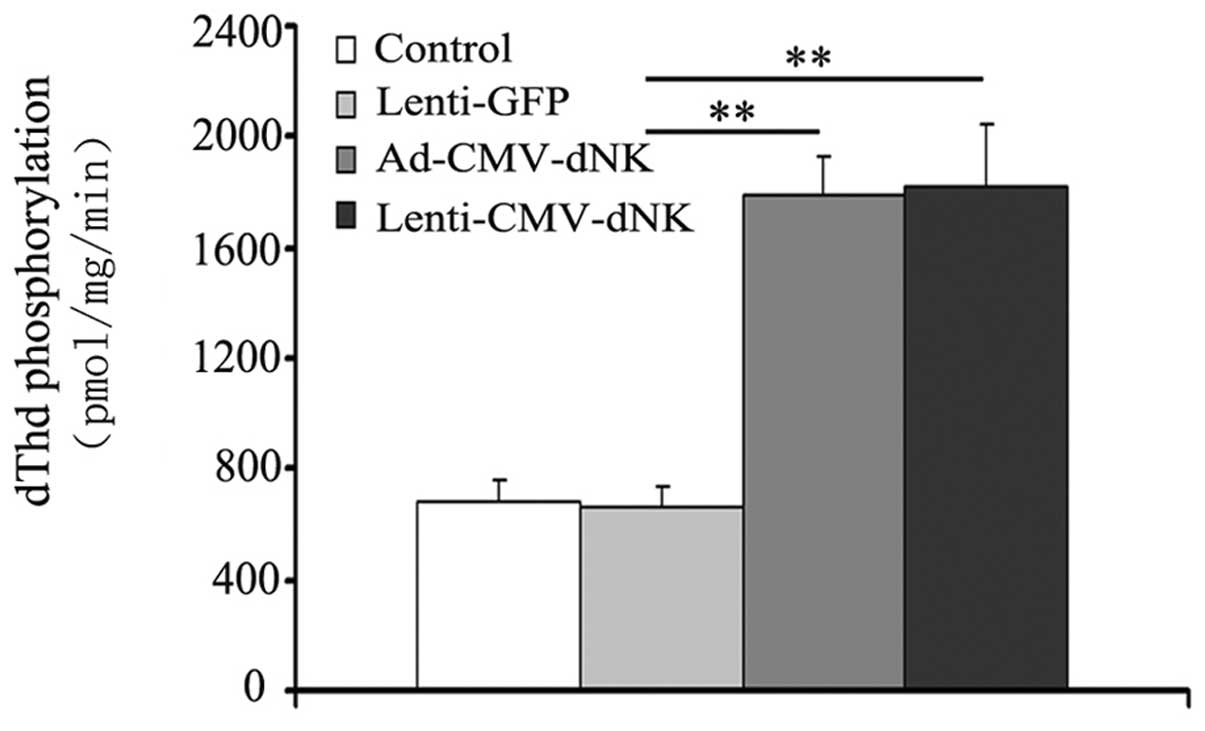
Moreover, the phosphorylation of dThd in cell extracts was examined
in order to evaluate the activity of Dm-dNK when imported
into cancer cells. The activity of Dm-dNK in the Bcap37
cells transduced with Ad-CMV-dNK and Lenti-CMV-dNK revealed a
3-fold increase compared to that in the cells transduced with
Lenti-GFP (Fig. 3), with a
significant difference (P<0.05), although no obvious difference
(P>0.05) between the cells transduced with Ad-CMV-dNK and
Lenti-CMV-dNK was observed.
Cell cytotoxicity, proliferation and
induction of apoptosis
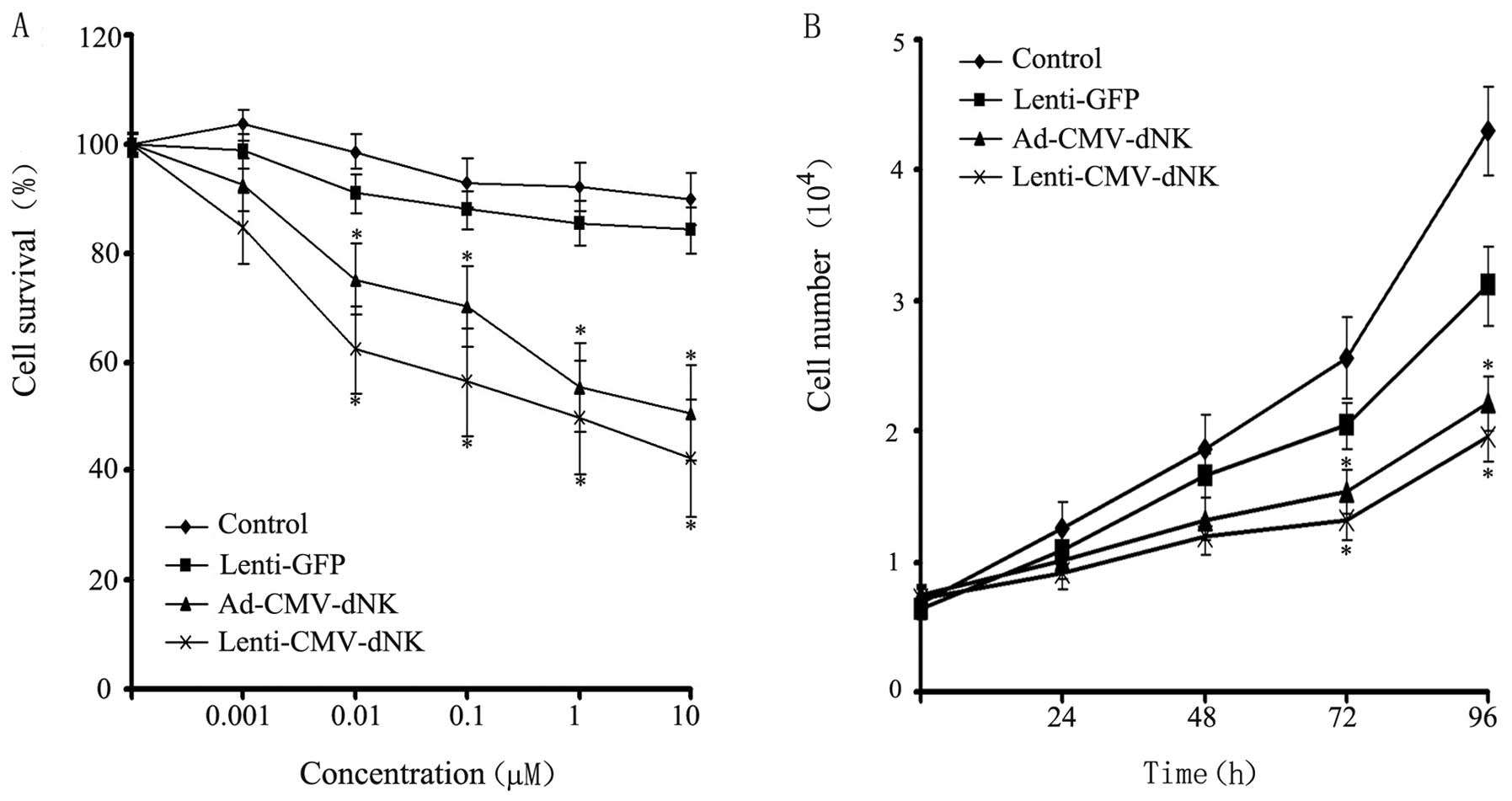
Bcap37 cells were transduced with Lenti-GFP,
Ad-CMV-dNK and Lenti-CMV-dNK at a MOI of 10 and incubated with BVDU
at increasing concentrations for 72 h, followed by viability
analysis using MTT assay. Cytotoxicity was observed in the Bcap37
cells infected with Ad-CMV-dNK and Lenti-CMV-dNK in the presence of
BVDU (Fig. 4A). The survival rates
of the Bcap37 cells transduced with Ad-CMV-dNK and Lenti-CMV-dNK in
the presence of 1 μM BVDU were ~55.3 and 49.7%, respectively. A
more prominent cytotoxic effect was observed in the presence of
high concentrations of BVDU, suggesting that the viability of the
cells revealed a decreasing trend in a dose-dependent manner.
Moreover, the Bcap37 cells treated with both Ad-CMV-dNK and
Lenti-CMV-dNK exhibited a decreasing trend in viability when
compared with the control or with the cells treated with Lenti-GFP.
The proliferation of the transduced Bcap37 cells in the presence of
1 μM BVDU was further analyzed. Both Ad-CMV-dNK and Lenti-CMV-dNK
revealed an apparent inhibitory effect on cell growth when compared
with the Lenti-GFP group after 72 h (P<0.05) (Fig. 4B). However, no significant
difference between the Ad-CMV-dNK and Lenti-CMV-dNK groups was
observed (P>0.05).
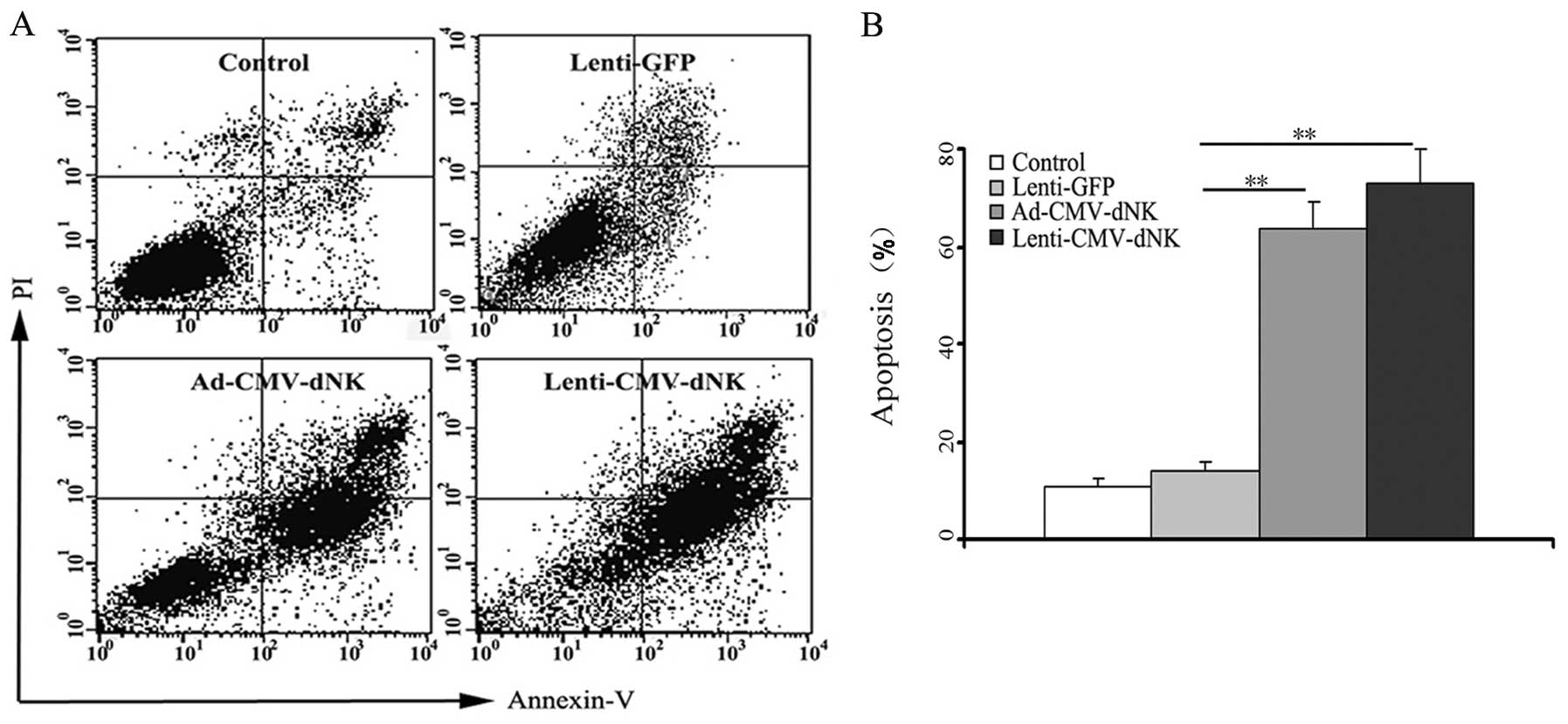
In an attempt to gain further insight into the
possible mechanisms of this suicide gene therapy, FACS analysis was
used to evaluate the apoptosis of Bcap37 cells. Ad-CMV-dNK and
Lenti-CMV-dNK resulted in higher apoptotic rates of 63.5±5.8 and
73.1±6.8%, respectively, at a BVDU dose of 1 μM (Fig. 5). Conversely, the apoptotic rate was
only 13.8±2.1% in the cells transduced with Lenti-GFP, indicating
that Dm-dNK had a slight contribution to the cytotoxicity.
Furthermore, there was no difference in apoptosis between the
Ad-CMV-dNK and Lenti-CMV-dNK groups (P>0.05), which was
consistent with its expression level in the previous experiment,
thus establishing a connection between the apoptotic level and the
activity of Dm-dNK in the experimental cell line.
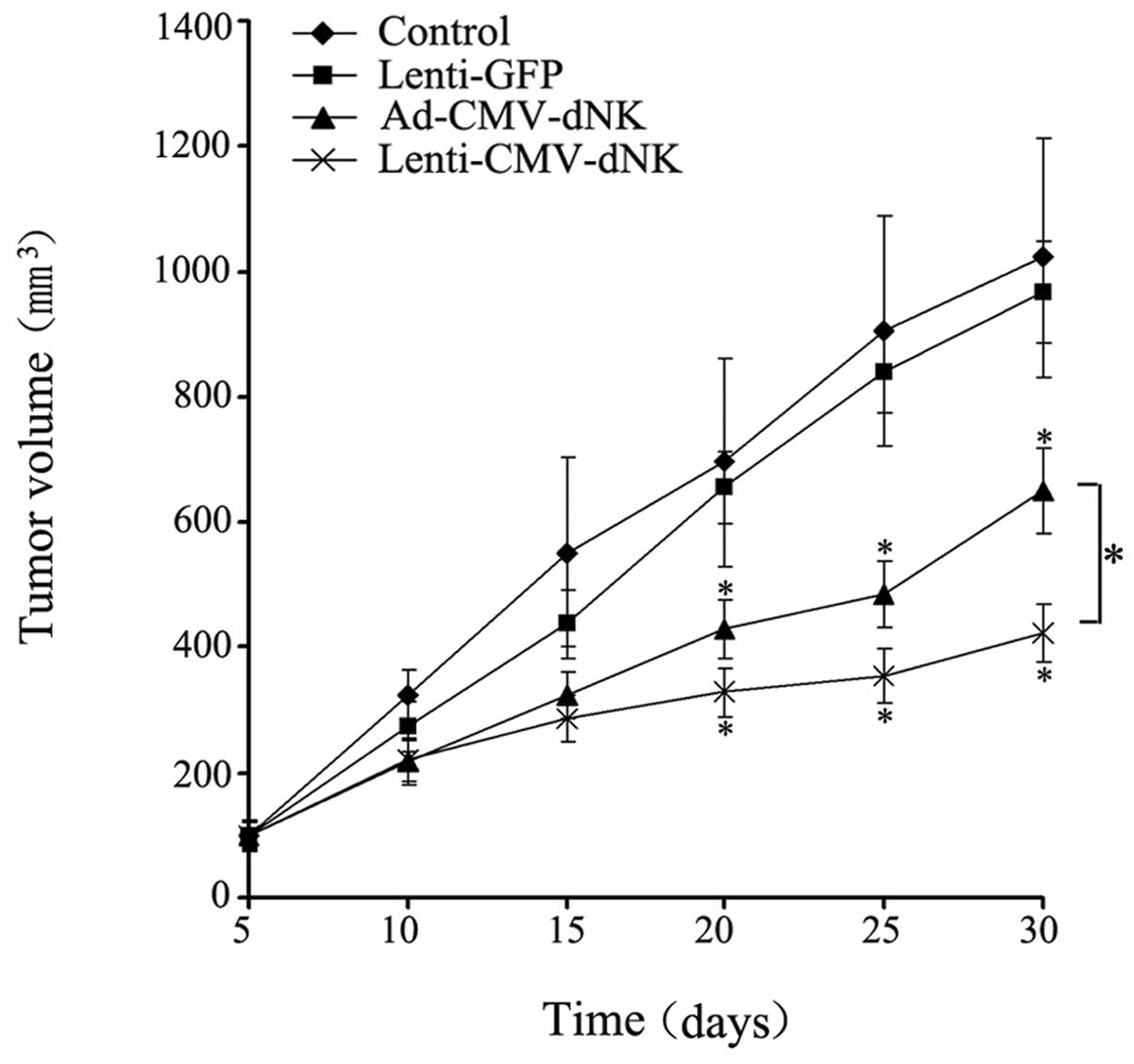
Antitumor efficacy of Dm-dNK in vivo
The therapeutic efficacy of lentiviral and
adenoviral vectors encoding a targeted gene in nude mice was
compared, revealing a dramatic difference. A significant inhibitory
effect on tumor growth in either the Ad-CMV-dNK or Lenti-CMV-dNK
groups combined with BVDU treatment when compared with the control
or Lenti-GFP group combined with BVDU treatment was demonstrated
(Fig. 6). The average volumes of
the tumors from the control or Lenti-GFP injection group combined
with BVDU treatment after 30 days were 1,023.48±191.39 and
967.79±81.21 mm3, respectively, which were much larger
compared to those from either the Ad-CMV-dNK or Lenti-CMV-dNK group
combined with BVDU treatment (650.34±68.30 and 424.22±46.74
mm3, respectively) (P<0.05). Moreover, the average
volume of tumors at day 30 from the Ad-CMV-dNK group combined with
BVDU was larger compared to that from the Lenti-CMV-dNK group
combined with BVDU (P<0.05). These findings indicate that
lentiviral vectors may the improve the therapeutic benefit in
long-term therapy in vivo.
Discussion
Despite several set-backs, gene-directed enzyme
prodrug therapy (GDEPT) remains a promising approach for cancer
gene therapy. This strategy has a significant advantage over
conventional chemotherapy since it reduces the toxicity induced by
the prodrug to target cells (14,15).
With the efforts to improve the therapeutic efficacy, viral vectors
are useful for gene delivery. In particular, adenoviral and
lentiviral vectors as highly effective gene carriers have been
widely applied.
Adenoviral vectors are extensively utilized in a
wide range of cancer cells. Adenoviruses retain their
extrachromosomal form, transduce susceptible cells efficiently and
produce high titers. However, the systemic delivery of adenoviruses
causes a variety of host immune responses, which strongly limits
its safe application in vivo. Lentiviral vectors have
advantages over adenoviral vectors since they are incorporated into
the host genome to establish a stable transgene expression, which
provides a promising role in improving therapeutic efficacy of gene
therapy (16). The unique
properties of both vectors provide the impetus for us to conduct
experimental and clinical studies of cancer gene therapy. To date,
to the best of our knowledge, there are no reports related to the
therapeutic outcomes of adenoviral and lentiviral vectors
expressing Dm-dNK in the gene therapy of cancer. In the
present study, the adenovirus- and lentivirus-mediated expression
of Dm-dNK was observed to offer efficient treatment for
human breast cancer. Moreover, lentivirus exhibited great potential
for long-lasting expression of the transgene. Therefore, the
vectors may be used in the majority of clinical cases.
Over the past decade, Dm-dNK has been
reported to phosphorylate all precursor nucleosides. The
deoxyribonucleoside kinase may be expressed in human cells and no
difference in its cellular sensitivity to nucleoside analogs was
observed when this enzyme was expressed in the cytosol or nucleus
(17,18). The broad substrate specificity of
Dm-dNK, as well as its high catalytic rate, makes it a
promising candidate of the nucleoside kinase family. On the basis
of previous findings (5–7), Dm-dNK has been shown to exert
potential antitumor activity. However, the underlying mechanism
remains unclear. Current interest is focused on substrate
specificity in the activation of nucleoside analogue to improve
therapeutic efficiency. Knecht et al addressed the issue
that a few amino acid substitutes may change the substrate
specificity of Dm-dNK(19).
Moreover, the deoxyribonucleoside kinase mutant exhibits a
relatively increased sensitivity to nucleoside analogs and a
simultaneous positive therapeutic effect in cancer cells (20,21).
Therefore, further studies are required to quantify
interconversation rates of Dm-dNK and mutants and provide a
more effective gene therapeutic strategy.
Knecht et al convincingly demonstrated that
gemcitabine, 2′,2′-difluoro-deoxycytidine (dFdC), an anticancer
drug, is an efficient substrate for Dm-dNK, which may
explain the structural basis for the interaction between
Dm-dNK and gemcitabine (22). Nevertheless, the underlying
mechanisms of other efficient prodrug candidates such as
1-β-D-arabinofuranosylcytosine (AraC, cytarabine) remain an area of
active investigation. Further studies are required to decipher the
precise role(s) of the correlation between Dm-dNK and
nucleoside analogs in cancer treatments combined with gene
therapy.
For attempts to optimize the therapeutic efficacy,
adeno-associated virus (AAV) as a promising mammalian virus vector
is commonly used for gene therapy and is considered one of the
safest viral vectors due to its stability as a vector genome and
its persistent high expression level (23,24).
Compared with other viral vectors, AAV may elicit minimal immune
responses. Recently, studies using animal models of stromal
transduction with AAV vectors have shown the advantages of this
vector (25,26). However, it has been reported that
AAV integration is associated with hepatocellular carcinoma
(27). Clearly, additional research
is required to more precisely elucidate these findings.
In conclusion, in this study, we demonstrate that
replication-defective adenoviral and lentiviral vectors highly
expressing a suicide gene can be effectivley used in suicide gene
therapy for the treatment of human breast cancer. However, the
lentiviral vector demonstrates broad implications for the long-term
expression of a therapeutic gene in the strategy of suicide gene
therapy. By employing lentiviral vectors, the Dm-dNK/BVDU
system may provide a safe and effective modality for the treatment
of breast cancer.
Acknowledgements
The present study was supported by grants from the
Hi-Tech Research Development Program of China (863 Program,
2006AA02Z493) and National Natural Science Foundation of China
(Nos. 81071900, 81272920 and 81172199).
References
|
1
|
Akcay MN: Metastatic disease in the
breast. Breast. 11:526–528. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Munch-Petersen B, Piskur J and Sondergaard
L: Four deoxynucleoside kinase activities from Drosophila
melanogaster are contained within a single monomeric enzyme, a
new multifunctional deoxynucleoside kinase. J Biol Chem.
273:3926–3931. 1998.PubMed/NCBI
|
|
3
|
Munch-Petersen B, Knecht W, Lenz C,
Sondergaard L and Piskur J: Functional expression of a
multisubstrate deoxyribonucleoside kinase from Drosophila
melanogaster and its C-terminal deletion mutants. J Biol Chem.
275:6673–6679. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ito M, Suda Y, Harashima H and Kamiya H:
Cytotoxic effect of Drosophila deoxynucleoside kinase gene
on replicating plasmid in HeLa cells. Biol Pharm Bull.
33:1223–1227. 2010.
|
|
5
|
Zhu Z, Mao L, Zhao L, et al: Synergistic
therapeutic effect in gastric cancer cells produced by oncolytic
adenovirus encoding Drosophila melanogaster
deoxyribonucleoside kinase. Cancer Biol Ther. 11:874–882. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ma S, Zhao L, Zhu Z, et al: The
multisubstrate deoxyribonucleoside kinase of Drosophila
melanogaster as a therapeutic suicide gene of breast cancer
cells. J Gene Med. 13:305–311. 2011.
|
|
7
|
Ma S, Qu W, Mao L, et al: Antitumor
effects of oncolytic adenovirus armed with Drosophila
melanogaster deoxyribonucleoside kinase in colorectal cancer.
Oncol Rep. 27:1443–1450. 2012.PubMed/NCBI
|
|
8
|
Zhang N, Zhao L, Ma S, Gu M and Zheng X:
Lentivirus-mediated expression of Drosophila melanogaster
deoxyribonucleoside kinase driven by the hTERT promoter combined
with gemcitabine: a potential strategy for cancer therapy. Int J
Mol Med. 30:659–665. 2012.
|
|
9
|
Kojaoghlanian T, Flomenberg P and Horwitz
MS: The impact of adenovirus infection on the immunocompromised
host. Rev Med Virol. 13:155–171. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Poller W, Fechner H, Noutsias M, Tschoepe
C and Schultheiss HP: Highly variable expression of virus receptors
in the human cardiovascular system. Implications for cardiotropic
viral infections and gene therapy. Z Kardiol. 91:978–991. 2002.
View Article : Google Scholar
|
|
11
|
VandenDriessche T, Collen D and Chuah MK:
Viral vector-mediated gene therapy for hemophilia. Curr Gene Ther.
1:301–315. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sugiyama O, An DS, Kung SP, et al:
Lentivirus-mediated gene transfer induces long-term transgene
expression of BMP-2 in vitro and new bone formation in vivo. Mol
Ther. 11:390–398. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sandrini MP, Clausen AR, On SL, Aarestrup
FM, Munch-Petersen B and Piskur J: Nucleoside analogues are
activated by bacterial deoxyribonucleoside kinases in a
species-specific manner. J Antimicrob Chemother. 60:510–520. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bonini C, Bondanza A, Perna SK, et al: The
suicide gene therapy challenge: how to improve a successful gene
therapy approach. Mol Ther. 15:1248–1252. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yazawa K, Fisher WE and Brunicardi FC:
Current progress in suicide gene therapy for cancer. World J Surg.
26:783–789. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bastide C, Maroc N, Bladou F, et al:
Expression of a model gene in prostate cancer cells lentivirally
transduced in vitro and in vivo. Prostate Cancer Prostatic Dis.
6:228–234. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Johansson M, Brismar S and Karlsson A:
Human deoxycytidine kinase is located in the cell nucleus. Proc
Natl Acad Sci USA. 94:11941–11945. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhu C, Johansson M and Karlsson A:
Incorporation of nucleoside analogs into nuclear or mitochondrial
DNA is determined by the intracellular phosphorylation site. J Biol
Chem. 275:26727–26731. 2000.PubMed/NCBI
|
|
19
|
Knecht W, Sandrini MP, Johansson K, Eklund
H, Munch-Petersen B and Piskur J: A few amino acid substitutions
can convert deoxyribonucleoside kinase specificity from pyrimidines
to purines. EMBO J. 21:1873–1880. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Solaroli N, Bjerke M, Amiri MH, Johansson
M and Karlsson A: Active site mutants of Drosophila
melanogaster multisubstrate deoxyribonucleoside kinase. Eur J
Biochem. 270:2879–2884. 2003.
|
|
21
|
Zhu Z, Ma S, Zhao L, et al:
Adenovirus-mediated Drosophila melanogaster
deoxyribonucleoside kinase mutants combined with gemcitabine harbor
a safe cancer treatment profile. Int J Oncol. 38:745–753. 2011.
|
|
22
|
Knecht W, Mikkelsen NE, Clausen AR, et al:
Drosophila melanogaster deoxyribonucleoside kinase activates
gemcitabine. Biochem Biophys Res Commun. 382:430–433. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mueller C and Flotte TR: Clinical gene
therapy using recombinant adeno-associated virus vectors. Gene
Ther. 15:858–863. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Calcedo R, Vandenberghe LH, Gao G, Lin J
and Wilson JM: Worldwide epidemiology of neutralizing antibodies to
adeno-associated viruses. J Infect Dis. 199:381–390. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu J, Saghizadeh M, Tuli SS, et al:
Different tropism of adenoviruses and adeno-associated viruses to
corneal cells: implications for corneal gene therapy. Mol Vis.
14:2087–2096. 2008.PubMed/NCBI
|
|
26
|
Sharma A, Tovey JC, Ghosh A and Mohan RR:
AAV serotype influences gene transfer in corneal stroma in vivo.
Exp Eye Res. 91:440–448. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Donsante A, Miller DG, Li Y, et al: AAV
vector integration sites in mouse hepatocellular carcinoma.
Science. 317:4772007. View Article : Google Scholar : PubMed/NCBI
|