Introduction
Breast cancer is one of the most frequently
diagnosed types of cancer and is also the leading cause of
cancer-related mortality among women, representing 23% of total
cancer cases and 14% of cancer-related mortality (1). Breast cancer is classified into
different categories according to the expression of three
receptors, estrogen receptor (ER), progesterone receptor (PR), and
HER2/Neu (2). A triple-negative
breast cancer (TNBC), which is ER-negative, PR-negative, and
HER2-negative, is one of the most aggressive forms that accounts
for 15-25% of all breast cancer cases and is associated with a poor
prognosis and unresponsiveness to the usual endocrine therapies
(3-5).
The tumor necrosis factor-related apoptosis-inducing
ligand (TRAIL) is a member of the TNF superfamily that can initiate
apoptosis via the activation of death receptor 4 (DR4) and death
receptor 5 (DR5). As TRAIL can selectively induce apoptosis in
cancer cells, including breast cancer cells, without causing
toxicity to normal cells (6-8), it
could be a safe medication for cancer patients; however, primary or
acquired resistance to TRAIL is often observed which may limit its
efficacy in cancer patients (6).
Therefore, increasing TRAIL efficacy by combining with natural
medicines or other chemotherapeutic agents is an important strategy
in the treatment of breast cancer in the clinical setting. We
previously identified that vanillin, an active constituent of
vanilla, enhances TRAIL-induced apoptosis in cancer cells via
suppression of NF-κB (9). This
finding prompted us to search for other natural products that can
sensitize cancer cells to TRAIL or that can overcome the
TRAIL-resistant mechanism.
In the present study, we screened 138 medicinal
plant extracts in TNBC cells to identify candidates that are not
cytotoxic as a single agent but that can increase TRAIL-induced
cytotoxicity synergistically. Among the tested extracts, 5 extracts
(Uvaria dac, Artemisia vulgaris, Cortia depressa, Dichasia
bengalensis and Cinnamomum obtusifolium) were found to activate
TRAIL effects in TRAIL-sensitive TNBC MDA-MB-231 cells and to
overcome unresponsiveness in TRAIL-insensitive TNBC MDA-MB-468
cells. We herein report the TRAIL modulatory activity by these
identified extracts and their mechanisms of action.
Materials and methods
Plant extracts and reagents
Plant extracts were dissolved in dimethyl sulfoxide
(DMSO) at a concentration of 1,000 μg/ml and these stock solutions
were stored at -20°C. Recombinant human TRAIL was purchased from
PeproTech (Peprotech, London, UK). The plants used in this study
are listed in Table I.
 | Table IEvaluation of 138 plant extracts for
their cytotoxicity as a single regimen and/or additive or
synergistic effect with TRAIL. |
Table I
Evaluation of 138 plant extracts for
their cytotoxicity as a single regimen and/or additive or
synergistic effect with TRAIL.
| Extract | MDA-MB-231 | Predicted additive
effect | Synergy Index | MDA-MB-468 | Predicted additive
effect | Synergy Index |
|---|
|
|
|---|
| TRAIL (−) | TRAIL (+) | TRAIL (−) | TRAIL (+) |
|---|
|
|
|---|
| 1.00 | 0.76 | 1.00 | 0.97 |
|---|
| Zizyphi
Fructus | 0.86 | 0.69 | 0.66 | 1.05 | 1.06 | 1.00 | 1.03 | 0.98 |
| Cinnamomi
Cortex | 0.88 | 0.59 | 0.67 | 0.88 | 1.06 | 1.04 | 1.03 | 1.01 |
| Rhei
Rhizoma | 0.80 | 0.62 | 0.61 | 1.01 | 0.99 | 0.98 | 0.96 | 1.01 |
| Araliae Cordatae
Rhizoma | 0.95 | 0.68 | 0.72 | 0.94 | 1.01 | 0.95 | 0.98 | 0.97 |
| Condonopsitis
Radix | 0.97 | 0.72 | 0.74 | 0.98 | 0.96 | 0.94 | 0.93 | 1.01 |
| Ginseng Radix
Rubra | 1.03 | 0.80 | 0.79 | 1.02 | 0.95 | 0.93 | 0.93 | 1.00 |
| Salviae
miltiorrhizae Radix | 1.00 | 0.75 | 0.76 | 0.98 | 1.01 | 1.02 | 0.98 | 1.04 |
| Armeniacae
Semen | 0.96 | 0.72 | 0.74 | 0.98 | 1.04 | 1.03 | 1.01 | 1.02 |
| Eucommiae
Cortex | 0.89 | 0.61 | 0.68 | 0.89 | 0.99 | 0.94 | 0.96 | 0.98 |
| Zanthoxyli
Fructus | 0.99 | 0.65 | 0.76 | 0.86 | 1.00 | 0.95 | 0.97 | 0.98 |
| Acanthopanax
senticosus Harms | 1.03 | 0.70 | 0.78 | 0.89 | 1.03 | 1.01 | 1.00 | 1.01 |
| Morus bombycis
Koidz | 0.94 | 0.66 | 0.71 | 0.92 | 1.03 | 1.03 | 1.00 | 1.03 |
| Rehmanniae
Radix | 1.13 | 0.88 | 0.86 | 1.02 | 1.04 | 1.01 | 1.01 | 1.01 |
| Platycodi
Radix | 1.53 | 1.00 | 1.17 | 0.86 | 1.04 | 1.07 | 1.00 | 1.07 |
| Corni
Fructus | 1.45 | 0.81 | 1.10 | 0.73 | 0.97 | 1.01 | 0.94 | 1.07 |
| Sparganii
Rhizoma | 1.08 | 0.95 | 0.83 | 1.15 | 0.96 | 1.02 | 0.93 | 1.09 |
| Trichosanthis
Radix | 1.28 | 1.03 | 0.97 | 1.06 | 0.93 | 0.97 | 0.91 | 1.07 |
| Lycii
Cortex | 1.16 | 1.05 | 0.89 | 1.19 | 0.99 | 1.00 | 0.96 | 1.04 |
| Cannabis
Fructus | 1.12 | 0.75 | 0.86 | 0.88 | 1.05 | 1.10 | 1.02 | 1.08 |
| Tribuli
Fructus | 1.05 | 0.73 | 0.80 | 0.91 | 1.02 | 1.11 | 0.99 | 1.12 |
| Gentianae
macrophyllae Radix | 0.97 | 0.64 | 0.74 | 0.86 | 1.02 | 1.04 | 0.99 | 1.05 |
| Puerariae
Radix | 1.01 | 0.79 | 0.77 | 1.03 | 1.08 | 1.05 | 1.05 | 1.00 |
| Zingiberis
Rhizoma | 0.99 | 0.71 | 0.75 | 0.94 | 0.94 | 0.91 | 0.91 | 1.00 |
| Alismatis
Rhizoma | 0.87 | 0.63 | 0.66 | 0.94 | 0.93 | 0.92 | 0.90 | 1.03 |
| Zingiberis
Siccatum Rhizoma | 1.10 | 0.78 | 0.84 | 0.93 | 0.94 | 0.96 | 0.91 | 1.05 |
| Sophorae
Radix | 1.26 | 0.93 | 0.96 | 0.96 | 1.00 | 1.01 | 0.97 | 1.05 |
| Rehmannia
glutinosa | 1.05 | 0.70 | 0.80 | 0.87 | 1.38 | 1.43 | 1.34 | 1.07 |
| Zedoariae
Rhizoma | 0.76 | 0.66 | 0.58 | 1.14 | 0.94 | 0.97 | 0.91 | 1.07 |
| Ephedrae
Herba | 1.05 | 0.73 | 0.80 | 0.92 | 0.87 | 0.87 | 0.84 | 1.03 |
| Paeoniae Moutan
Cortex | 0.93 | 0.65 | 0.71 | 0.91 | 0.99 | 0.94 | 0.96 | 0.98 |
| Evodiae
Fructus | 0.93 | 0.66 | 0.71 | 0.93 | 1.05 | 1.11 | 1.02 | 1.09 |
| Paeoniae
Radix | 0.78 | 0.91 | 0.60 | 1.52 | 0.88 | 0.88 | 0.86 | 1.03 |
| Arctii
Fructus | 0.88 | 0.93 | 0.67 | 1.39 | 0.68 | 0.74 | 0.66 | 1.13 |
| Polygalae
Radix | 0.75 | 0.73 | 0.57 | 1.29 | 0.87 | 0.85 | 0.85 | 1.01 |
| Glehniae Radix
cum Rhizoma | 0.85 | 0.75 | 0.65 | 1.16 | 0.93 | 0.93 | 0.91 | 1.02 |
| Sinomeni Caulis
et Rhizoma | 0.77 | 0.78 | 0.58 | 1.33 | 0.91 | 0.89 | 0.88 | 1.01 |
| Dipsaci
Radix | 0.99 | 0.99 | 0.76 | 1.31 | 0.94 | 0.91 | 0.91 | 1.00 |
| Achyranthis
Radix | 0.84 | 0.60 | 0.64 | 0.93 | 0.96 | 0.95 | 0.93 | 1.02 |
| Leonuri
Herba | 0.89 | 0.77 | 0.68 | 1.14 | 0.94 | 1.18 | 0.91 | 1.29 |
| Woodforidia
fruticosaa | 0.68 | 0.62 | 0.52 | 1.19 | 0.50 | 0.49 | 0.48 | 1.02 |
| Quercus
infectoria | 0.48 | 0.47 | 0.37 | 1.26 | 0.41 | 0.34 | 0.40 | 0.86 |
| Gardenia
oronaria | 0.77 | 0.60 | 0.59 | 1.02 | 0.82 | 0.76 | 0.80 | 0.96 |
| Penthum
Operculina | 0.83 | 0.69 | 0.63 | 1.09 | 0.88 | 0.87 | 0.85 | 1.01 |
| Iris
florentia | 0.85 | 0.69 | 0.65 | 1.06 | 0.78 | 0.76 | 0.76 | 1.00 |
| Myrica
nagi | 0.82 | 0.76 | 0.63 | 1.21 | 0.82 | 0.83 | 0.79 | 1.05 |
| Chamaecyparis
obtusa (MeOH Ext., upper layer) | 0.18 | 0.17 | 0.14 | 1.24 | 0.24 | 0.27 | 0.24 | 1.13 |
| Chamaecyparis
obtusa (MeOH Ext., lower layer) | 0.72 | 0.21 | 0.55 | 0.39 | 0.75 | 0.37 | 0.73 | 0.51 |
| Cordyceps
sinensis | 0.91 | 0.77 | 0.70 | 1.11 | 1.05 | 0.88 | 1.02 | 0.86 |
| Derris
floribumda (CHCl3 Ext.) | 0.82 | 0.55 | 0.63 | 0.88 | 0.87 | 0.65 | 0.85 | 0.77 |
| Derris
floribumda (MeOH Ext.) | 0.57 | 0.53 | 0.44 | 1.22 | 0.79 | 0.65 | 0.77 | 0.84 |
| Cinnamomum
obtusifolium Nees. | 0.89 | 0.39 | 0.68 | 0.58 | 0.97 | 0.68 | 0.94 | 0.72 |
| Cryptomeria
japonica | 0.94 | 0.26 | 0.71 | 0.37 | 0.66 | 0.39 | 0.64 | 0.61 |
| Uvaria
dac | 1.13 | 0.47 | 0.86 | 0.54 | 1.09 | 0.74 | 1.06 | 0.70 |
| Artemisia
vulgaris | 0.87 | 0.34 | 0.66 | 0.52 | 0.93 | 0.35 | 0.90 | 0.39 |
| Scoparia
dulsis | 0.54 | 0.12 | 0.41 | 0.30 | 0.57 | 0.36 | 0.56 | 0.65 |
| Unidentified | 1.01 | 0.88 | 0.77 | 1.14 | 0.82 | 0.83 | 0.80 | 1.04 |
| Polygonum
barbatum (L.) | 0.92 | 0.88 | 0.70 | 1.26 | 0.81 | 0.78 | 0.79 | 0.99 |
| Euphorbia hirta
L. | 0.75 | 0.63 | 0.57 | 1.11 | 0.84 | 0.75 | 0.81 | 0.93 |
| Eclipta
prostrata | 0.62 | 0.26 | 0.47 | 0.55 | 0.66 | 0.59 | 0.64 | 0.93 |
| Chinopodium
ambrosioides | 1.05 | 0.82 | 0.80 | 1.03 | 0.81 | 0.87 | 0.79 | 1.10 |
| Senna
alata | 1.05 | 0.86 | 0.80 | 1.07 | 0.84 | 0.78 | 0.82 | 0.96 |
| Allium
wallichii | 0.62 | 0.55 | 0.47 | 1.18 | 0.60 | 0.62 | 0.59 | 1.05 |
| Potentilla
peduncularis | 1.10 | 0.84 | 0.84 | 1.00 | 0.67 | 0.77 | 0.65 | 1.18 |
| Cortia
depressa | 0.97 | 0.44 | 0.74 | 0.60 | 0.81 | 0.57 | 0.79 | 0.72 |
| Rhododendron
campanulatum | 0.85 | 0.97 | 0.65 | 1.49 | 0.78 | 0.77 | 0.76 | 1.02 |
| Rhododendron
ciliatum Hook. | 0.90 | 0.75 | 0.69 | 1.09 | 0.81 | 0.79 | 0.79 | 1.00 |
| Primula
rotundifolia | 0.93 | 0.77 | 0.71 | 1.09 | 1.17 | 1.06 | 1.13 | 0.93 |
| Boschniakia
himalaica | 1.05 | 0.94 | 0.80 | 1.18 | 1.09 | 1.05 | 1.06 | 0.99 |
| Clematis
montana | 1.04 | 1.04 | 0.79 | 1.31 | 1.18 | 1.11 | 1.15 | 0.97 |
| Bistorta
macrophylla | 1.07 | 1.05 | 0.82 | 1.29 | 1.10 | 1.09 | 1.07 | 1.02 |
| Primula
denticulata | 0.90 | 0.90 | 0.69 | 1.30 | 1.08 | 1.02 | 1.05 | 0.98 |
| Ohiglossum
valgatum | 0.77 | 0.28 | 0.59 | 0.48 | 0.57 | 0.26 | 0.55 | 0.47 |
| Unidentified | 0.99 | 0.97 | 0.76 | 1.28 | 1.13 | 1.13 | 1.10 | 1.03 |
| Semecarpus
anacardium | 0.15 | 0.15 | 0.11 | 1.30 | 0.14 | 0.14 | 0.14 | 1.02 |
| Dichasia
bengalensis | 0.82 | 0.42 | 0.63 | 0.67 | 0.81 | 0.37 | 0.79 | 0.47 |
| Senna
fistula | 0.97 | 0.93 | 0.74 | 1.25 | 1.25 | 1.10 | 1.21 | 0.91 |
| Euphorbia
hirta | 0.97 | 0.86 | 0.74 | 1.16 | 1.17 | 1.04 | 1.14 | 0.91 |
| Curcuma
caesia | 0.08 | 0.08 | 0.06 | 1.31 | 0.08 | 0.07 | 0.07 | 1.02 |
| Cedrus
deodara | 0.74 | 0.63 | 0.56 | 1.12 | 0.96 | 0.83 | 0.93 | 0.89 |
|
Argemonum | 1.10 | 0.72 | 0.84 | 0.86 | 0.94 | 0.79 | 0.91 | 0.87 |
|
Chrysenthamum | 0.73 | 0.57 | 0.56 | 1.02 | 0.84 | 0.78 | 0.81 | 0.96 |
| Adiantum
venustum | 0.92 | 0.53 | 0.70 | 0.75 | 0.77 | 0.51 | 0.75 | 0.69 |
| Nerium
oleander | 0.97 | 0.92 | 0.74 | 1.25 | 1.00 | 0.92 | 0.97 | 0.94 |
| Euphorbia
royleana | 0.08 | 0.08 | 0.06 | 1.27 | 0.09 | 0.09 | 0.09 | 1.03 |
| Pogostemon
benghalensis | 0.88 | 0.36 | 0.67 | 0.53 | 0.65 | 0.29 | 0.63 | 0.45 |
| Bauhinia
variegata | 0.88 | 0.89 | 0.67 | 1.32 | 0.91 | 0.77 | 0.88 | 0.87 |
| Tamarindus
indica | 0.59 | 0.58 | 0.45 | 1.28 | 0.64 | 0.57 | 0.62 | 0.92 |
| Woodforidia
fruticosaa | 0.59 | 0.58 | 0.45 | 1.28 | 0.70 | 0.63 | 0.68 | 0.93 |
| Argemone
mexicana | 0.72 | 0.56 | 0.55 | 1.03 | 0.51 | 0.39 | 0.50 | 0.80 |
| Ficus
lacor | 0.72 | 0.79 | 0.55 | 1.43 | 0.67 | 0.67 | 0.65 | 1.02 |
| Oroxylum
indicum | 0.78 | 0.36 | 0.60 | 0.61 | 0.58 | 0.26 | 0.56 | 0.46 |
| Lathyrus
sativus | 0.61 | 0.15 | 0.47 | 0.33 | 0.44 | 0.16 | 0.43 | 0.37 |
| Dichroa
febrifuga | 0.85 | 0.77 | 0.65 | 1.19 | 0.93 | 0.89 | 0.90 | 0.99 |
| Origanum
majorana | 0.92 | 0.93 | 0.70 | 1.33 | 1.04 | 0.98 | 1.01 | 0.98 |
| Calotropis
gigantea | 0.62 | 0.36 | 0.48 | 0.75 | 0.48 | 0.16 | 0.46 | 0.35 |
| Rhus trichocarpa
bark | 1.03 | 0.96 | 0.78 | 1.23 | 0.97 | 0.92 | 0.94 | 0.98 |
| Rhus trichocarpa
wood | 1.00 | 0.97 | 0.76 | 1.27 | 0.88 | 0.83 | 0.85 | 0.98 |
| Syzygium
cumini | 1.04 | 1.15 | 0.79 | 1.46 | 0.73 | 0.69 | 0.70 | 0.98 |
| Artium
lappa | 0.91 | 0.83 | 0.70 | 1.20 | 1.14 | 1.16 | 1.10 | 1.05 |
| Rheum
austale | 0.65 | 0.65 | 0.50 | 1.31 | 0.68 | 0.69 | 0.66 | 1.05 |
| Nelumbium
speciosum | 0.82 | 0.83 | 0.62 | 1.34 | 0.83 | 0.85 | 0.80 | 1.06 |
| Carum
copticum | 0.55 | 0.16 | 0.42 | 0.39 | 0.66 | 0.35 | 0.64 | 0.54 |
| Solanum
xanthocarpum schrad | 0.25 | 0.30 | 0.19 | 1.56 | 0.10 | 0.11 | 0.10 | 1.09 |
| Vernonia
anthelmintica willd | 0.11 | 0.11 | 0.08 | 1.29 | 0.11 | 0.11 | 0.11 | 1.05 |
| Aconitum
spicatum | 0.94 | 0.71 | 0.72 | 0.99 | 0.98 | 0.87 | 0.95 | 0.92 |
| Nardostachys
grandiflora | 0.07 | 0.07 | 0.05 | 1.31 | 0.08 | 0.08 | 0.08 | 1.01 |
| Delphinium
denudatum | 0.96 | 0.81 | 0.73 | 1.10 | 1.11 | 0.96 | 1.07 | 0.89 |
| Kaunia
longipetiolata | 0.85 | 0.90 | 0.65 | 1.39 | 0.85 | 0.82 | 0.83 | 0.99 |
| Mucuna
nigricans | 0.96 | 0.88 | 0.73 | 1.20 | 1.14 | 1.07 | 1.11 | 0.96 |
| Strychnos
nuxvomica | 0.92 | 0.82 | 0.70 | 1.16 | 1.00 | 0.97 | 0.97 | 0.99 |
| Adhatoda vasica
Nees | 0.96 | 0.86 | 0.74 | 1.16 | 1.03 | 0.92 | 1.00 | 0.92 |
| Wrightia
tinctoria | 0.76 | 0.70 | 0.58 | 1.21 | 0.76 | 0.54 | 0.74 | 0.73 |
| Symplocos
racemosa | 0.88 | 0.96 | 0.67 | 1.44 | 0.91 | 0.87 | 0.88 | 0.99 |
| Piper
chabo | 0.68 | 0.17 | 0.52 | 0.32 | 0.38 | 0.15 | 0.37 | 0.42 |
| Paris
poyphilla | 0.25 | 0.24 | 0.19 | 1.21 | 0.10 | 0.09 | 0.09 | 1.00 |
| Withania
somnifera | 1.01 | 1.04 | 0.77 | 1.35 | 1.01 | 0.89 | 0.98 | 0.91 |
| Linum
usitatissimum | 0.96 | 0.87 | 0.73 | 1.20 | 0.98 | 1.00 | 0.95 | 1.05 |
| Rhododendron
anthopogon | 0.82 | 0.68 | 0.63 | 1.08 | 0.70 | 0.62 | 0.68 | 0.91 |
| Inula
cappade | 0.96 | 0.94 | 0.73 | 1.29 | 0.89 | 0.92 | 0.86 | 1.06 |
| Citrullus
colocynthis | 0.99 | 0.83 | 0.75 | 1.11 | 0.85 | 0.88 | 0.82 | 1.08 |
| Calotropis
gigantean | 0.88 | 0.94 | 0.67 | 1.39 | 0.95 | 0.87 | 0.92 | 0.95 |
| Lyonia
ovalifolia | 0.99 | 1.10 | 0.75 | 1.47 | 1.13 | 1.22 | 1.10 | 1.11 |
| Periploca
calophylla | 0.93 | 1.04 | 0.71 | 1.46 | 1.31 | 1.41 | 1.27 | 1.11 |
| Zanthoxylum
armatum | 0.92 | 1.02 | 0.70 | 1.44 | 1.56 | 1.40 | 1.52 | 0.92 |
| Unidentified | 1.02 | 1.10 | 0.78 | 1.42 | 1.59 | 1.51 | 1.55 | 0.97 |
| Achyranthus
aspera | 0.96 | 1.03 | 0.73 | 1.40 | 1.54 | 1.47 | 1.49 | 0.99 |
| Woodforidia
fruticosaa | 0.68 | 0.67 | 0.52 | 1.31 | 0.86 | 0.89 | 0.84 | 1.06 |
| Ficus
lacor | 0.91 | 0.97 | 0.70 | 1.40 | 1.35 | 1.36 | 1.31 | 1.04 |
| Paris
polyphylla | 0.63 | 0.70 | 0.48 | 1.45 | 0.42 | 0.47 | 0.41 | 1.15 |
| Solanum
xanthocarpum | 0.28 | 0.32 | 0.22 | 1.50 | 0.37 | 0.32 | 0.36 | 0.91 |
| Equisetum
debile | 1.06 | 1.07 | 0.81 | 1.32 | 1.00 | 0.86 | 0.98 | 0.89 |
| Pogostemon
benghalensis | 0.91 | 1.03 | 0.69 | 1.48 | 1.13 | 1.15 | 1.10 | 1.05 |
| Girardiana
heterophylla | 0.94 | 0.85 | 0.72 | 1.19 | 1.04 | 0.99 | 1.01 | 0.97 |
| Gaultheria
fragrantisia | 0.98 | 0.92 | 0.75 | 1.23 | 1.07 | 1.07 | 1.04 | 1.03 |
| Crateva
unilocularis | 1.00 | 1.04 | 0.76 | 1.37 | 1.23 | 1.24 | 1.20 | 1.04 |
| Rabdosia
rugosa | 0.99 | 1.00 | 0.75 | 1.33 | 1.13 | 1.21 | 1.09 | 1.11 |
| Curcuma
amada | 0.97 | 0.87 | 0.74 | 1.18 | 0.78 | 0.62 | 0.76 | 0.82 |
| Rhododendron
anthopogon | 0.82 | 0.87 | 0.62 | 1.40 | 0.90 | 0.87 | 0.87 | 1.00 |
Cell culture
MDA-MB-231, MDA-MB-468, MDA-MB-453 and MCF-7 cells
(ATCC, Rockville, MD, USA) were cultured in Dulbecco’s modified
Eagle’s medium (DMEM) supplemented with 10% heat-inactivated fetal
calf serum, 2 mM L-glutamine, 100 U/ml penicillin, and 10 μg/ml
streptomycin. Cells were maintained at 37°C in a humidified
atmosphere of 5% CO2/95% air.
Cell viability assay
Viability of cells following treatment was
determined using the WST-1 Cell Counting kit (Wako Pure Chemical
Industries, Osaka, Japan) as previously described (10). Briefly, cells were seeded into
96-well plates (7×103/80 μl/well). After 24 h, 10 μl
medium containing each plant extract was added and cells were
incubated for an additional 30 min. After the addition of TRAIL (50
ng/ml), cells were incubated for 24 h, and 10 μl WST-1 solution was
added to each well and incubated at 37°C for 1 h. The absorbance at
450 nm was measured. Relative cell viability was calculated by the
formula: Relative cell viability = [average absorbance of
experimental wells/average absorbance of control wells].
Synergistic effects were determined when cell viability treated
with both TRAIL and plant extract (Vcomb) was less than
predicted additive effects [= cell viability of TRAIL alone
(VTRAIL) × cell viability of plant extract alone
(Vext)]. The synergy index was calculated by
Vcom/(VTRAIL × Vext).
Western blot analysis
Western blot analysis was performed as previously
described (11). Cells were seeded
and incubated overnight in a 60-mm culture dish
(0.25×106 cells/ml). Following treatment, whole cell
lysates were collected in lysis buffer [25 mM HEPES pH 7.7, 0.3 M
NaCl, 1.5 mM MgCl2, 0.2 mM EDTA, 0.1% Triton X-100, 20
mM β-glycerophosphate, 1 mM sodium orthovanadate, 1 mM
phenylmethylsulfonyl fluoride (PMSF), 1 mM dithiothreitol (DTT), 10
mg/ml aprotinin and 10 mg/ml leupeptin]. Equal amounts of protein
were resolved by electrophoresis on acrylamide gels. Antibodies
against caspase-3, PARP, XIAP, MCL-1 and phosphorylated p65 were
purchased from Cell Signaling Technology (Danvers, MA, USA).
Antibodies against β-actin and p65 were purchased from Santa Cruz
Biotechnology Inc., (Santa Cruz, CA, USA).
Results
TRAIL sensitivity in breast cancer cell
lines
Since each cell line shows various sensitivities to
TRAIL due to different expression levels of BAX, MCL-1, IAPs and
others (12-17), we initially tried four breast cancer
cell lines, MDA-MB-231, -468, -453 and MCF-7, to distinguish
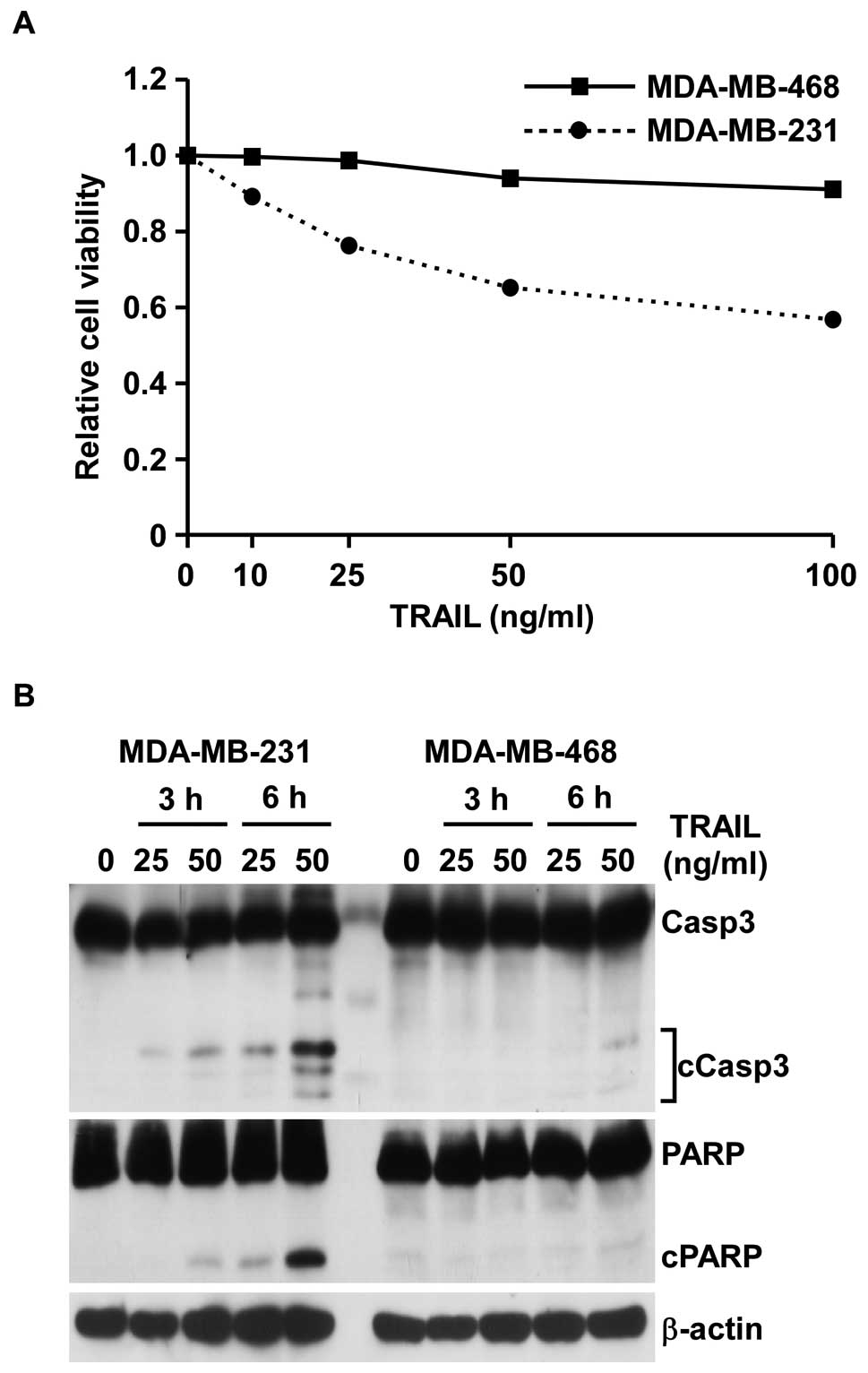
TRAIL-sensitive from -insensitive cells. Of note, the cell
viability assay showed that only MDA-MB-231 cells were sensitive to
TRAIL and the others were insensitive (Fig. 1A and data not shown). Both
MDA-MB-231 and MDA-MB-468 are triple-negative (ER-,
PR- and HER2-) breast cancer cells, and are
reported to be the most severe forms of breast cancer (3-5);
therefore, we focused on these two cell lines in the present study.
Furthermore, cleaved PARP and cleaved caspase-3, which are known as
apoptotic markers, were also observed in MDA-MB-231 cells with
TRAIL in a time- and concentration-dependent manner, but not in
MDA-MB-468 cells (Fig. 1B). This
showed that MDA-MB-231 cells are sensitive to TRAIL and MDA-MB-468
cells are insensitive, and that the cell death induced by TRAIL
might be due to apoptosis.
Classification of the plant extracts
according to their cytotoxic and/or synergistic effects
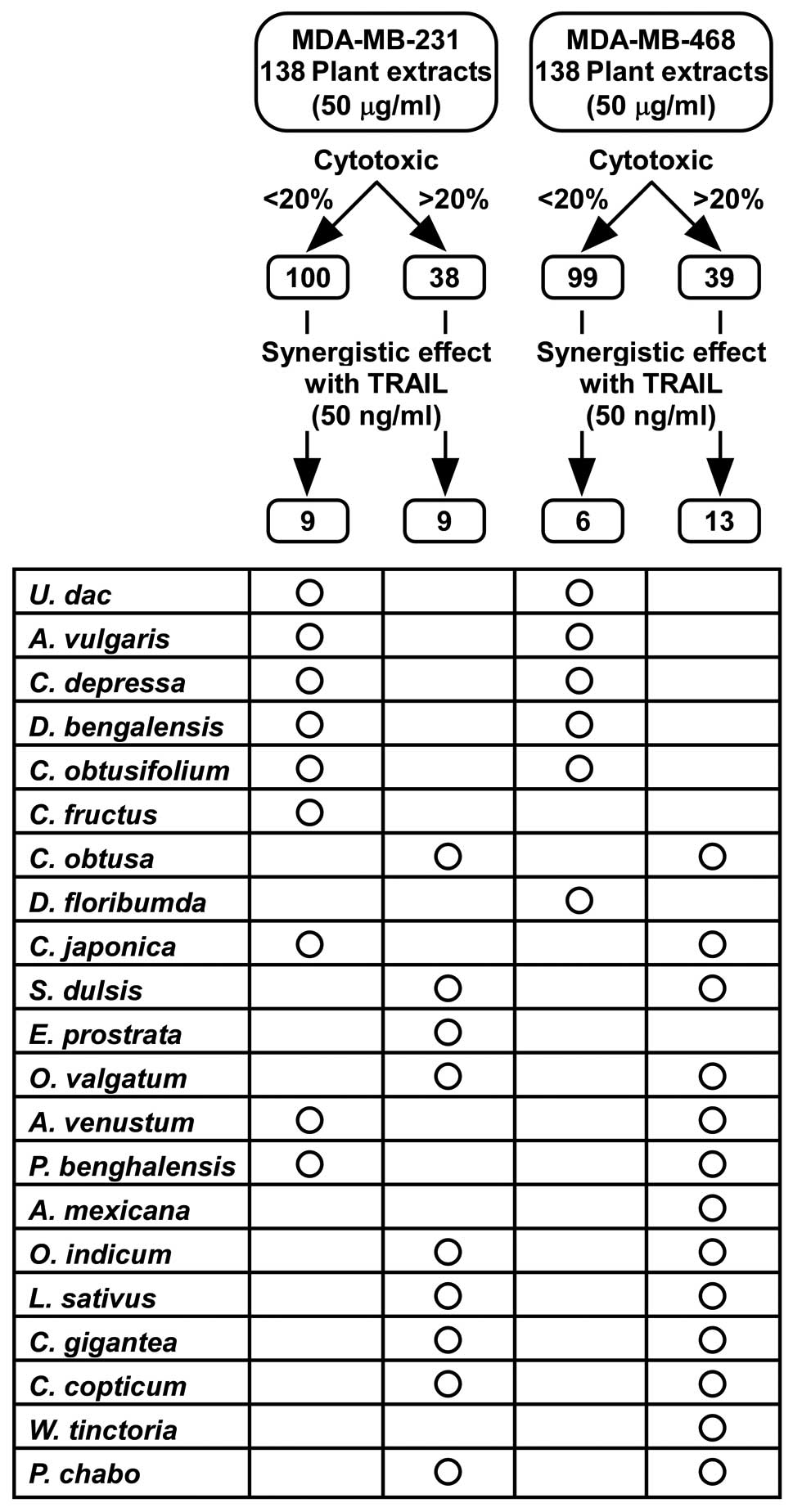
Subsequently, we identified the plant extracts that
showed less cytotoxicity as a single agent, but that enhanced TRAIL
effects in TRAIL-sensitive cells or that overcame unresponsiveness
in TRAIL-insensitive cells. One hundred and thirty-eight plant
extracts with or without TRAIL were screened by cell viability
assay (Fig. 2 and Table I). Two criteria were used to
classify the plant extracts; first, we determined whether cell
viability treated with the plant extracts alone (Vext)
was >0.8 (>80% cell survival) at the maximum tested
concentration of 50 μg/ml. Second, we checked whether the
combination of plant extract with TRAIL showed a synergistic
effect, indicating that the detected cell viability with both TRAIL
and plant extract (Vcomb), is less than its predicted
additive effect [= cell viability with TRAIL alone
(VTRAIL) × Vext] and the synergy index [=
Vcom/(VTRAIL × Vext)] is also
<0.8. In TRAIL-sensitive MDA-MB-231 cells, 100 extracts showed
low cytotoxicity (Vext >0.8). Among them, 9 extracts
(C. fructus, C. obtusifolium, C. japonica, U. dac, A. vulgaris,
C. depressa, D. bengalensis, A. venustum and P.
benghalensis) showed synergistic effects with TRAIL. In the
case of MDA-MB-468 cells, 99 extracts showed low cytotoxicity and 6
extracts (D. floribumda, C. obtusifolium, U. dac, A. vulgaris,
C. depressa and D. bengalensis) showed synergistic
effects with TRAIL. As shown in Fig.
2, only 5 extracts met these two criteria in both cell lines,
U. dac, A. vulgaris, C. depressa, D.
bengalensis and C. obtusifolium.
Five identified plant extracts induce
cell death in a concentration-dependent manner
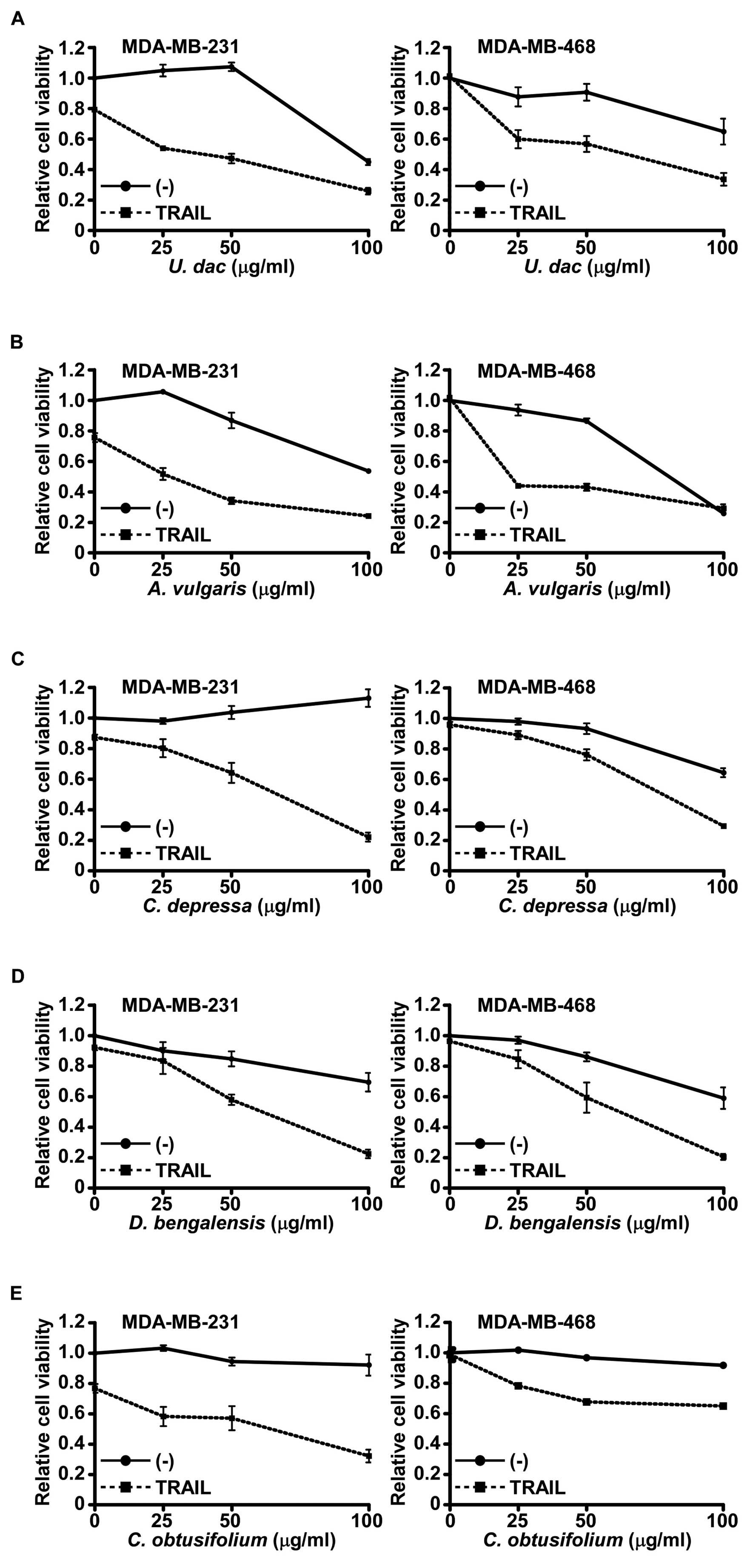
To check the concentration-dependency of the 5
selected plant extracts, we performed the cell viability assay at
various extract concentrations with TRAIL (Fig. 3). These extracts showed synergistic
effects with TRAIL in both cell lines in a concentration-dependent
manner; therefore, we further examined whether the induced cell
death was due to increased apoptosis or due to other types of cell
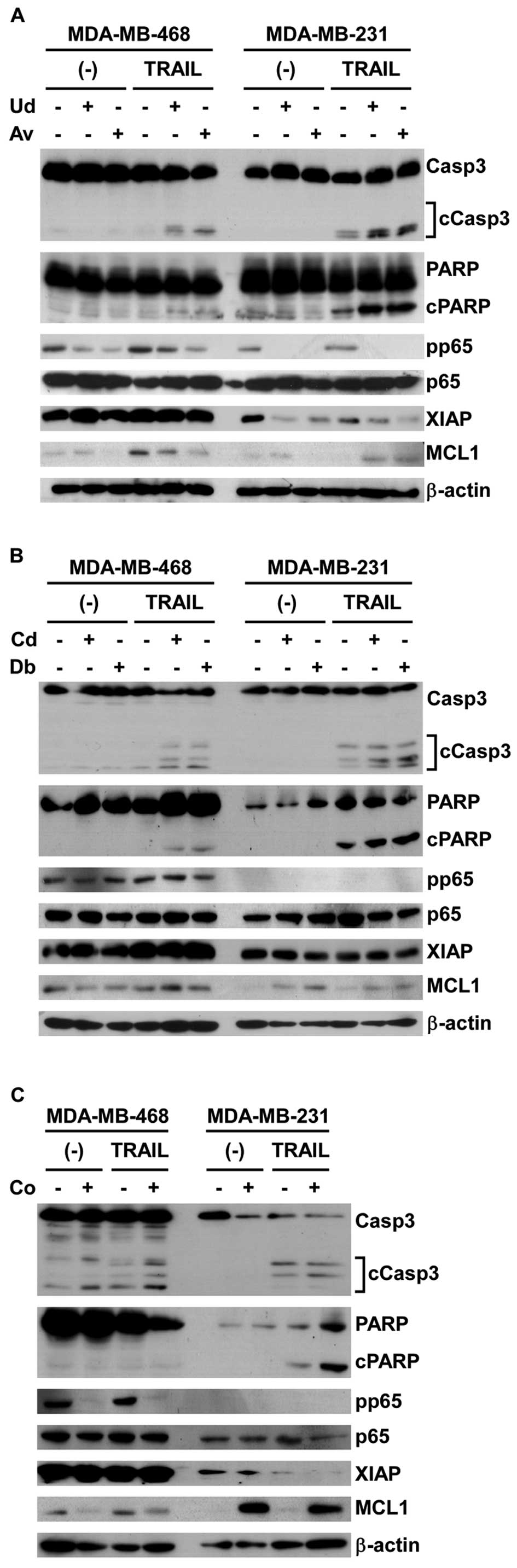
death (Fig. 4). In both cell lines,
the cleavage of PARP and caspase-3 was strongly detected when
treated with a combination of plant extract and TRAIL compared with
TRAIL alone. We then checked the expression and phosphorylation of
p65, in addition to the expression of XIAP and MCL-1, which are
involved in TRAIL-resistant mechanisms (9,12,14-16).
Markedly, the three extracts (U. dac, A. vulgaris and
C. obtusifolium) showed clear inhibition of the
phosphorylation of p65, suggesting that the reduced phosphorylation
of p65 could explain the synergistic effect of U. dac, A.
vulgaris and C. obtusifolium with TRAIL. On the other
hand, the two extracts, C. depressa and D.
bengalensis, that also showed synergistic effects with TRAIL
did not show the downregulation of MCL-1, XIAP or phosphorylation
of p65, suggesting that these two extracts may cause the
synergistic effects with TRAIL by affecting different proteins or
pathways.
Discussion
In the present study, we screened 138 medicinal
plant extracts for their ability to potentiate TRAIL effects in
TRAIL-sensitive cells and to overcome resistant mechanisms in
TRAIL-insensitive cells. From the screening, 5 potential medicinal
plant extracts were identified to sensitize TNBC cancer cells to
TRAIL. For identification, we used only two distinct criteria:
whether the plant extract displays only minimal toxicity (<20%
at 50 μg/ml concentration) and whether the extract potentiates
TRAIL-induced cell death in a synergistic manner. Our results
demonstrated that these two criteria suffice to identify extracts
as TRAIL-sensitizing reagents. This screening criteria can also be
applied for drugs other than TRAIL to identify drug-sensitizing
reagents.
Although we only focused on plant extracts showing
low cytotoxicity and synergistic effects with TRAIL in both cells,
the screening results in Fig. 2 and
Table I provide significant insight
into the cytotoxic activity of each plant extract as well as their
combination effects with TRAIL. Eight plant extracts (C. obtusa,
S. dulsis, O. valgatum, O. indicum, L. sativus, C. gigantean, C.
copticum and P. chabo) showed cytotoxicity and
synergistic effects in both cell lines (Fig. 2). In addition, 38 plant extracts
(including S. anacardium, C. caesia, E. royleana)
revealed strong cytotoxicity with or without TRAIL in both cell
lines (Table I); therefore, the
active constituents present in these extracts need further
investigation as potential chemotherapeutic agents against breast
cancer. The extracts of C. fructus, C. japonica, A. venustum
and P. benghalensis showed low cytotoxicity with synergistic
effects with TRAIL in MDA-MB-231 cells but showed cytotoxicity and
synergistic effects in MDA-MB-468 cells. Therefore, further study
of these extracts may provide critical information and clues to
identify TRAIL-resistant mechanisms that are specifically active in
MDA-MB-468 cells.
Among the 5 plant extracts sensitizing breast cancer
cells to TRAIL, U. dac and its active constituent (+)
grandifloracin showed anti-austerity activity against PANC-1 human
pancreatic cancer cells in a nutrition-deficient tumor-mimicking
environment (18). The extract of
A. vulgaris has been reported to have cytotoxicity against
MCF-7 cells (19). Consistent with
these findings, U. dac and A. vulgaris also showed
cytotoxicity at higher concentrations (Fig. 3A and B) in the present study,
although we used lower non-cytotoxic concentrations to identify the
synergistic effects with TRAIL. The possible mechanisms of
sensitization by the newly identified plant extracts to
TRAIL-induced cell death of TNBC were investigated by western blot
analysis (Fig. 4). In particular,
U. dac, A. vulgaris and C. obtusifolium may
sensitize breast cancer cells via suppression of the
phosphorylation of p65, which is involved in the resistant
mechanism to TRAIL (9); however,
further investigation of the other two extracts, C. depressa
and D. bengalensis, is necessary to understand the mechanism
of action of these extracts.
In conclusion, the results of this study provide
valuable information about potential medicinal plants that could
enhance TRAIL activity and develop combination therapies against
breast cancer. The 5 medicinal plant extracts identified in the
present study were unique, as they enhanced the sensitivity to
TRAIL in both TRAIL-sensitive and -insensitive TNBC cells.
Furthermore, these extracts could have potential medical
applications to improve TRAIL efficacy and to overcome resistance
mechanisms not only in breast cancer, but in other types of cancer
as well.
Acknowledgements
We thank Professor Hiroaki Sakurai (Faculty of
Pharmaceutical Sciences, University of Toyama, Japan) for helpful
discussions.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global Cancer Statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Brenton JD, Carey LA, Ahmed AA and Caldas
C: Molecular classification and molecular forecasting of breast
cancer: ready for clinical application? J Clin Oncol. 23:7350–7360.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hudis CA and Gianni L: Triple-Negative
Breast Cancer: An Unmet Medical Need. Oncologist. 16:1–11. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Haffty BG, Yang Q, Reiss M, et al:
Locoregional relapse and distant metastasis in conservatively
managed triple negative early-stage breast cancer. J Clin Oncol.
24:5652–5657. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dent R, Trudeau M, Pritchard KI, et al:
Triple-negative breast cancer: clinical features and patterns of
recurrence. Clin Cancer Res. 13:4429–4434. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Keane MM, Ettenberg SA, Nau MM, Russell EK
and Lipkowitz S: Chemotherapy augments TRAIL induced apoptosis in
breast cell lines. Cancer Res. 59:734–741. 1999.PubMed/NCBI
|
|
7
|
Chinnaiyan AM, Prasad U, Shankar S, et al:
Combined effect of tumor necrosis factor-related apoptosis-inducing
ligand and ionizing radiation in breast cancer therapy. Proc Natl
Acad Sci USA. 97:1754–1759. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Walczak H, Miller RE, Ariail K, et al:
Tumoricidal activity of tumor necrosis factor-related
apoptosis-inducing ligand in vivo. Nat Med. 5:157–163. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lirdprapamongkol K, Sakurai H, Suzuki S,
et al: Vanillin enhances TRAIL-induced apoptosis in cancer cells
through inhibition of NF-κB activation. In Vivo. 24:501–506.
2010.PubMed/NCBI
|
|
10
|
Lirdprapamongkol K, Sakurai H, Kawasaki N,
et al: Vanillin suppresses in vitro invasion and in vivo metastasis
of mouse breast cancer cells. Eur J Pharm Sci. 25:57–65. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Choo MK, Kawasaki N, Singhirunnusorn P, et
al: Blockade of transforming growth factor-beta-activated kinase 1
activity enhances TRAIL-induced apoptosis through activation of a
caspase cascade. Mol Cancer Ther. 5:2970–2976. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Falschlehner C, Emmerich CH, Gerlach B and
Walczak H: TRAIL signalling: decisions between life and death. Int
J Biochem Cell Biol. 39:1462–1475. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Burns TF and El-Deiry WS: Identification
of inhibitors of TRAIL-induced death (ITIDs) in the TRAIL-sensitive
colon carcinoma cell line SW480 using a genetic approach. J Biol
Chem. 276:37879–37886. 2001.PubMed/NCBI
|
|
14
|
Clohessy JG, Zhuang J, de Boer J,
Gil-Gómez G and Brady HJ: Mcl-1 interacts with truncated Bid and
inhibits its induction of cytochrome c release and its role in
receptor-mediated apoptosis. J Biol Chem. 281:5750–5759. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chawla-Sarkar M, Bae SI, Reu FJ, Jacobs
BS, Lindner DJ and Borden EC: Downregulation of Bcl-2, FLIP or IAPs
(XIAP and survivin) by siRNAs sensitizes resistant melanoma cells
to Apo2L/TRAIL-induced apoptosis. Cell Death Differ. 11:915–923.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ravi R and Bedi A: Potential methods to
circumvent blocks in apoptosis in lymphomas. Curr Opin Oncol.
14:490–503. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shrader M, Pino MS, Lashinger L, Bar-Eli
M, Adam L, Dinney CP and McConkey DJ: Gefitinib reverses TRAIL
resistance in human bladder cancer cell lines via inhibition of
AKT-mediated X-linked inhibitor of apoptosis protein expression.
Cancer Res. 67:1430–1435. 2007. View Article : Google Scholar
|
|
18
|
Awale S, Ueda J, Athikomkulchai S,
Abdelhamed S, Yokoyama S, Saiki I and Miyatake R: Antiausterity
agents from Uvaria dac and their preferential cytotoxic
activity against human pancreatic cancer cell lines in a
nutrient-deprived condition. J Nat Prod. 75:1177–1183. 2012.
|
|
19
|
Taghizadeh Rabe SZ, Mahmoudi M, Ahi A and
Emami SA: Antiproliferative effects of extracts from Iranian
Artemisia species on cancer cell lines. Pharm Biol.
49:962–969. 2011.PubMed/NCBI
|