Introduction
Liver cancer is the fifth most prevalent malignancy
in men and the seventh in women worldwide (1). The incidence of liver cancer is
increasing possibly due to the epidemic of obesity and the rise in
hepatitis C virus infection (1).
Current treatment for liver cancer includes surgical resection,
liver transplantation, local ablation, transarterial
chemoembolization and radioembolization, and targeted molecular
therapy (2,3). Despite improved therapeutic methods,
the curative outcome of advanced liver cancer patients remains
elusive. Liver cancer remains the second leading cause of
cancer-related death in men and the sixth in women (1). The overall 5-year survival rate of
liver cancer patients has remained below 12% in the United States
(3). Thus, there is a great need to
develop novel agents or alternative strategy to treat liver
cancer.
In China, Traditional Chinese Medicine (TCM) has
played a positive role in liver cancer treatment (4). Based on varied syndromes, different
therapeutic methods and Chinese herbs can be employed to ameliorate
clinical symptoms and local disease focus. Yujin (Curcuma
aromatica) is a commonly used Chinese herb for treating liver
cancer with blood-stasis and depressed liver-Qi with or without
jaundice. Curcuma aromatica possesses anticancer potential
against liver cancer and colon cancer (5,6).
Huzhang (Polygonum cuspidatum) is a well-tolerated Chinese
herb used for treating liver diseases with damp-heat and
blood-stasis syndrome, and has been regarded as an anticancer herb.
It is frequently used in liver cancer treatment. Polygonum
cuspidatum was found to display anticancer effects in liver
cancer, oral cancer and lung cancer cells (7–9).
Curcuma aromatica and Polygonum cuspidatum are
commonly used as a paired-herbal medication for liver cancer
treatment.
Curcumin is an important ingredient of Curcuma
aromatica, and has been used as a quality control standard for
Curcuma aromatica(10).
Curcumin inhibits proliferation and induces apoptosis in numerous
types of cancer cells including liver cancer (11). One of the major components of
Polygonum cuspidatum is resveratrol (12). In addition to chemo-preventive
activity against tumorigenesis, resveratrol exhibits anticancer
effects against various types of cancer cells such as lung
carcinoma, pancreatic cancer and hepatocellular carcinoma cells and
inhibits cancer cell invasion and metastasis (13–18).
Both curcumin and resveratrol have displayed synergistic effects
with current cancer therapeutics (19,20).
The combination of curcumin and resveratrol was found to
demonstrate a synergistic anticancer effect in colon cancer
(21). However, as constituents
from paired-herbs, the combined effect of curcumin and resveratrol
against hepatocarcinoma cells remains unknown. In the present
study, we evaluated the anticancer effect of curcumin combined with
resveratrol in hepatocarcinoma cells.
Materials and methods
Chemicals and reagents
Curcumin, resveratrol and dimethyl sulfoxide (DMSO)
were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). Fetal
bovine serum (FBS), Dulbecco’s modified Eagle’s medium (DMEM) and
0.25% (w/v) Trypsin-EDTA were obtained from Gibco-BRL
(Gaithersburg, MD, USA). The MTT cell proliferation and
cytotoxicity assay kit, 2′,7′-dichlorofluorescein diacetate
(DCFH-DA), N-acetylcysteine (NAC), caspase-3, -8 and -9
colorimetric assay kits, and Hoechst 33258 were obtained from
Beyotime Institute of Biotechnology (Jiangsu, China). Z-VAD-FMK was
obtained from R&D Systems (Minneapolis, MN, USA). Propidium
iodide (PI) and Annexin V-FITC were purchased from BD Pharmingen
(Minneapolis, MN, USA). Antibodies against XIAP and survivin were
purchased from Bioworld Technology Co., Ltd. (St. Louis Park, MN,
USA). Antibodies against β-actin were from Cell Signaling
Technology (Danvers, MA, USA).
Cell culture and treatment
Murine hepatocarcinoma cell line Hepa1-6 was
purchased from the Cell Bank of the Type Culture Collection of the
Chinese Academy of Sciences (Shanghai, China) and was grown in DMEM
with 10% FBS and 1% penicillin/streptomycin, and maintained at 37°C
in a humidified incubator with a 5% CO2 atmosphere.
Exponentially growing Hepa1-6 cells were seeded into desired plates
and allowed to attach for 24 h before treatment. Cells were treated
with various doses of curcumin and resveratrol dissolved in DMSO.
The identical volume of DMSO was used as a control.
Cell proliferation assay
The effects of curcumin and resveratrol on cell
proliferation were detected by MTT assay. Briefly, Hepa1-6 cells
were seeded into 96-well plates (3.5×103 cells/well) and
allowed to attach for 24 h before treatment. The cells were exposed
to various doses of curcumin and/or resveratrol for 48 h, and cell
viability was evaluated every 24 h by MTT assay according to the
manufacturer’s instructions. The cell survival rate was calculated
as follows: Cell survival rate (%) = experimental OD value/control
OD value × 100.
Observation of apoptotic morphology
Morphological changes characteristic of apoptosis
were detected by Hoechst 33258 staining. Briefly, 6×104
Hepa1-6 cells were seeded into 6-well plates and incubated for 24
h. Hepa1-6 cells were treated with curcumin and/or resveratrol for
48 h, and stained with Hoechst 33258 for 5 min at room temperature.
The cells were observed and photographed using an inverted
fluorescence microscope (AFM010-2, Nikon, Japan).
Quantification of apoptosis
Hepa1-6 cells were treated as indicated, collected,
and stained with Annexin V-FITC and PI as recommended by the
manufacturer. Apoptotic cells were detected by flow cytometry, and
the extent of apoptosis was calculated with FlowJo software
(version 7.6.1).
Detection of reactive oxygen species
(ROS)
Intracellular ROS production was detected by DCFH-DA
staining. DCFH-DA is cleaved intracellularly by non-specific
esterases to form DCFH, which is further oxidized by ROS to form
the fluorescent compound DCF (22).
Briefly, Hepa1-6 cells (1×105) were placed in 60-mm
culture dishes. On the second day, cells were exposed to the
indicated concentrations of agents for a 48 h treatment, and
stained with DCFH-DA (10 μM) at 37°C for 20 min. The stained cells
were collected and added to black 96-well plates at a density of
2×105 cells/well. The presence of DCF fluorescence was
quantified with a fluorescence microplate reader (Thermo Scientific
Varioskan Flash; Thermo Fischer Scientific, Waltham, MA, USA) at
wavelengths of 488 nm for excitation and 525 nm for emission. For
ROS inhibition, cells were pretreated with NAC (2.5 mmol/l for 2
h), followed by curcumin and/or resveratrol treatment.
Caspase activity assay
After treatment with curcumin and/or resveratrol,
caspase-3, -8 and -9 activities were measured by the cleavage of
the specific chromogenic substrate according to the manufacturer’s
instructions. For inhibiton of caspase activity, Hepa1-6 cells were
pretreated with Z-VAD-FMK (50 μmol/l for 2 h) and further treated
with curcumin and/or resveratrol.
Western blot analysis
Western blot analysis was performed as previously
described (23,24). Briefly, after drug treatment,
Hepa1-6 cells were collected, lysed and subjected to 6–12% sodium
dodecyl sulfate-polyacrylamide (SDS-PAGE) gel, and transferred to
nitrocellulose membranes (Amersham Biosciences, Buckinghamshire,
UK). The transferred membranes were blocked with 5% non-fat milk,
washed and probed with the indicated antibodies. The specific
antigen-antibody complex was visualized using an enhanced
chemiluminescence detection method (Amersham, Buckinghamshire,
UK).
Statistical analysis
Data are expressed as the means ± standard deviation
of at least two independent experiments, each conducted in
triplicate. Statistical significances between the control and drug
treatment were determined by one-way ANOVA. A value of P<0.05
was considered to indicate a statistically significant result.
Results
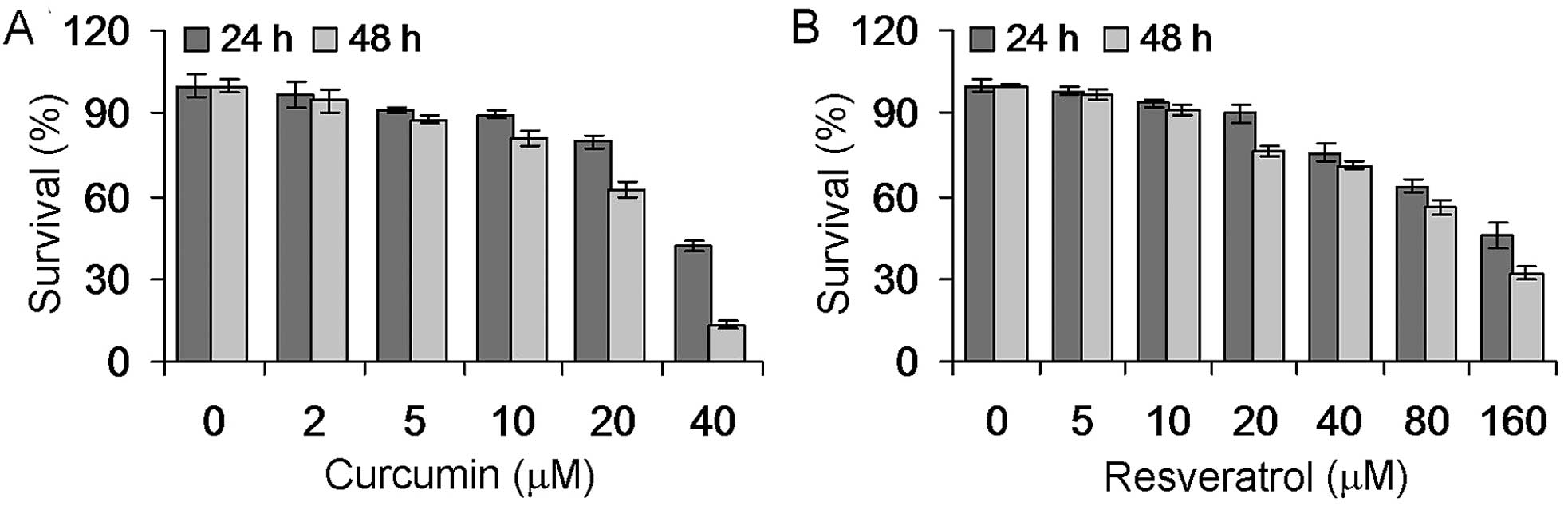
Effects of curcumin and resveratrol on
Hepa1-6 cell proliferation
The antiproliferative effects of curcumin and
resveratrol on hepatocarcinoma Hepa1-6 cells were assessed with the
MTT assay. As shown in Fig. 1,
curcumin (5–40 μM) and resveratrol (10–160 μM) significantly
inhibited the proliferation of Hepa1-6 cells in a dose- and
time-dependent manner (P<0.05).
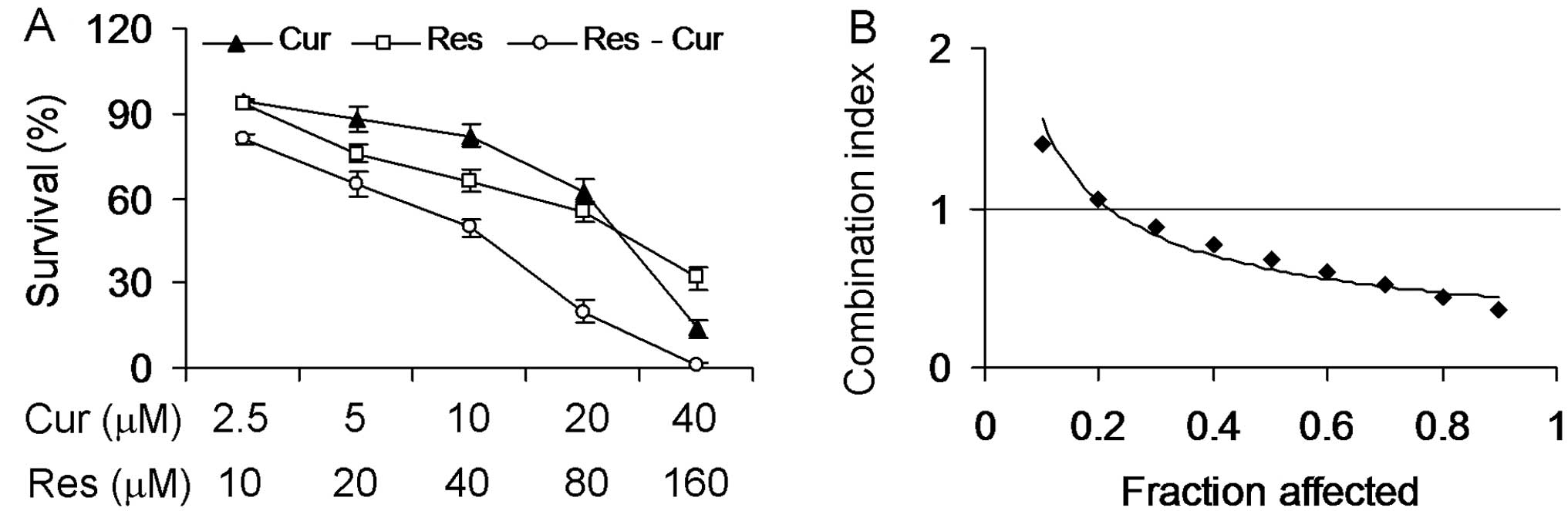
Synergistic antiproliferative effect of
curcumin and resveratrol on Hepa1-6 cells
To assess the combined effects of curcumin and
resveratrol on Hep1-6 cells, we treated Hepa1-6 cells with
curcumin, resveratrol or both agents in a constant ratio to one
another. As shown in Fig. 2A, the
combination treatment of curcumin and resveratrol was more
effective in inhibiting the proliferation of Hepa1-6 cells when
compared with the treatment of either agents alone (P<0.01),
which indicated an interaction between the two drugs. The precise
nature of this interaction was further analyzed by the
median-effect method (25), where
the combination indices (CI) of less than, equal to, and more than
1 indicate synergistic, additive and antagonistic effects,
respectively. The CI value was <1 for the combination treatment
when the proliferation inhibition was >20%, indicating a
synergistic effect between curcumin and resveratrol on Hepa1-6 cell
proliferation (Fig. 2B). Based on
these observations, we selected 10 μM of curcumin and 40 μM of
resveratrol (~50% inhibitory effect for the combination treatment)
to carry out the subsequent studies.
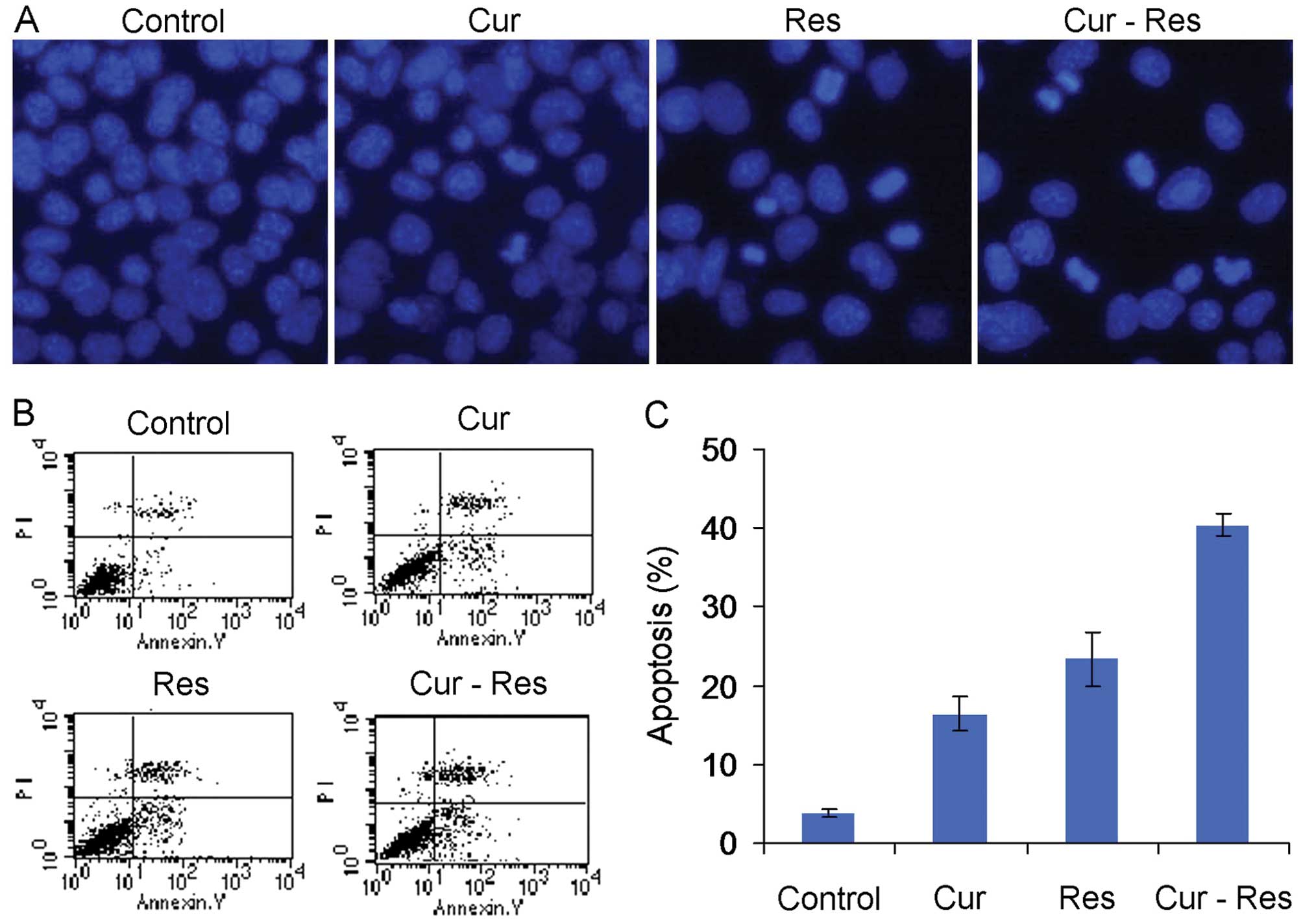
Combination of curcumin and resveratrol
induces apoptosis in Hepa1-6 cells
To assess the effects of curcumin and resveratrol on
apoptosis, Hepa1-6 cells were treated with curcumin and/or
resveratrol and subjected to Hoechst 33258 staining to visualize
the apoptotic morphological alterations. As shown in Fig. 3A, following treatment with curcumin
and/or resveratrol for 48 h, a portion of Hepa1-6 cells exhibited
nuclear shrinkage or fragmentation, indicating the occurrence of
apoptotic processes. Flow cytometric analysis was used to quantify
the extent of apoptosis. As shown in Fig. 3B and C, curcumin and resveratrol
significantly induced apoptosis of the Hepa1-6 cells (P<0.01).
Combination treatment of curcumin and resveratrol was found more
effective in inducing apoptosis than either agent alone
(P<0.01).
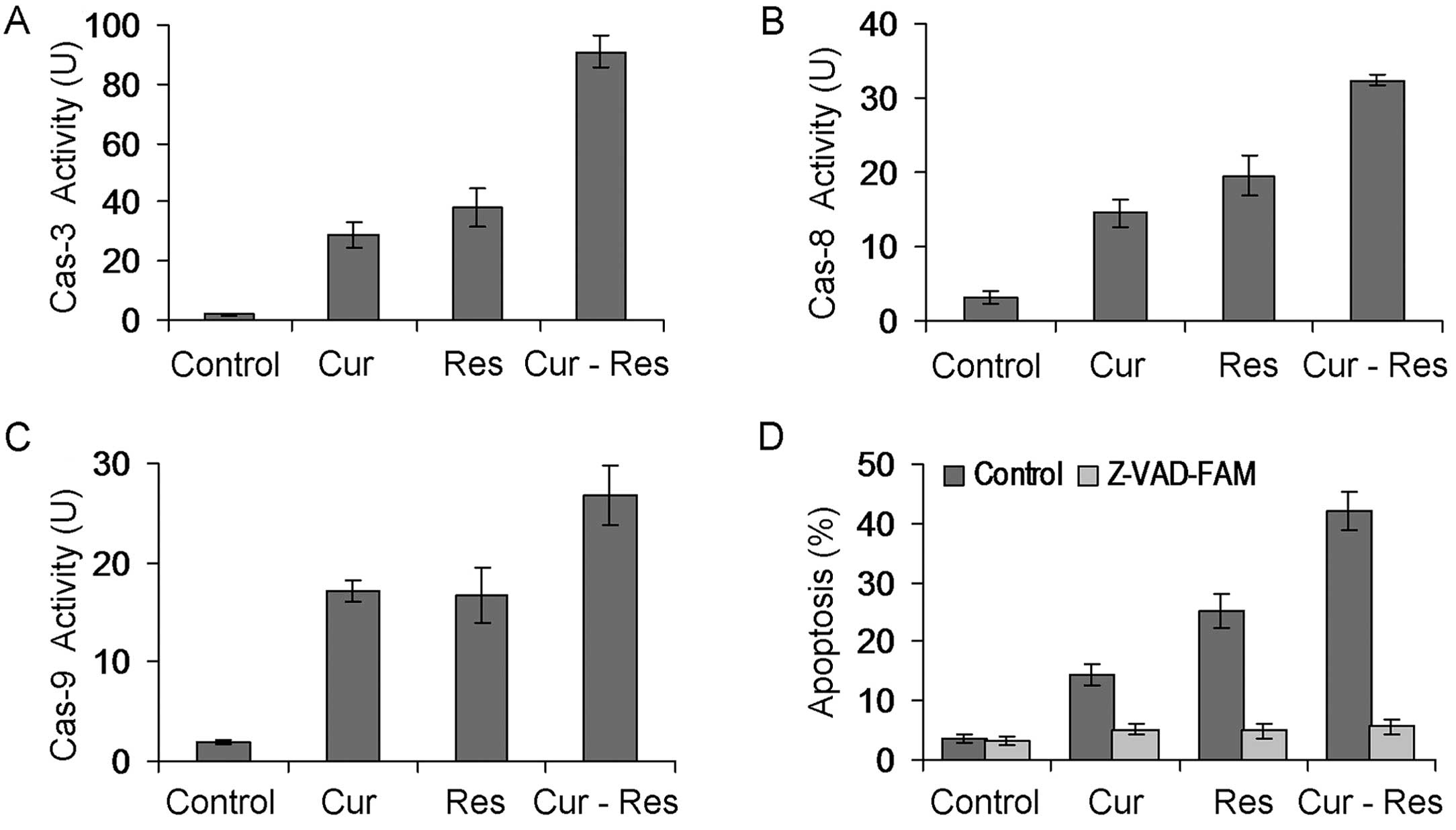
Combination of curcumin and resveratrol
activates caspases in Hepa1-6 cells
Cell apoptosis is executed by a caspase (cysteine
aspartate-specific proteinase) cascade, and activation of caspases
has been recognized as a hallmark of apoptosis (26). We further observed the effects of
curcumin and/or resveratrol on caspase activation in Hepa1-6 cells.
As shown in Fig. 4A-C, curcumin
and/or resveratrol treatment significantly activated caspase-3, -8
and -9 in Hepa1-6 cells (P<0.01). Combination treatment of
curcumin and resveratrol significantly enhanced caspase-3, -8 and
-9 activation when compared with that following treatment with
either agent alone (P<0.01). In addition, Z-VAD-FMK, a pan
caspase inhibitor, completely inhibited curcumin and
resveratrol-induced apoptosis in Hepa1-6 cells (P<0.01)
(Fig. 4D). These results suggest
that caspase-3, -8 and -9 activation may be an important mechanism
involved in the apoptosis of Hepa1-6 cells induced by the
combination treatment of curcumin and resveratrol.
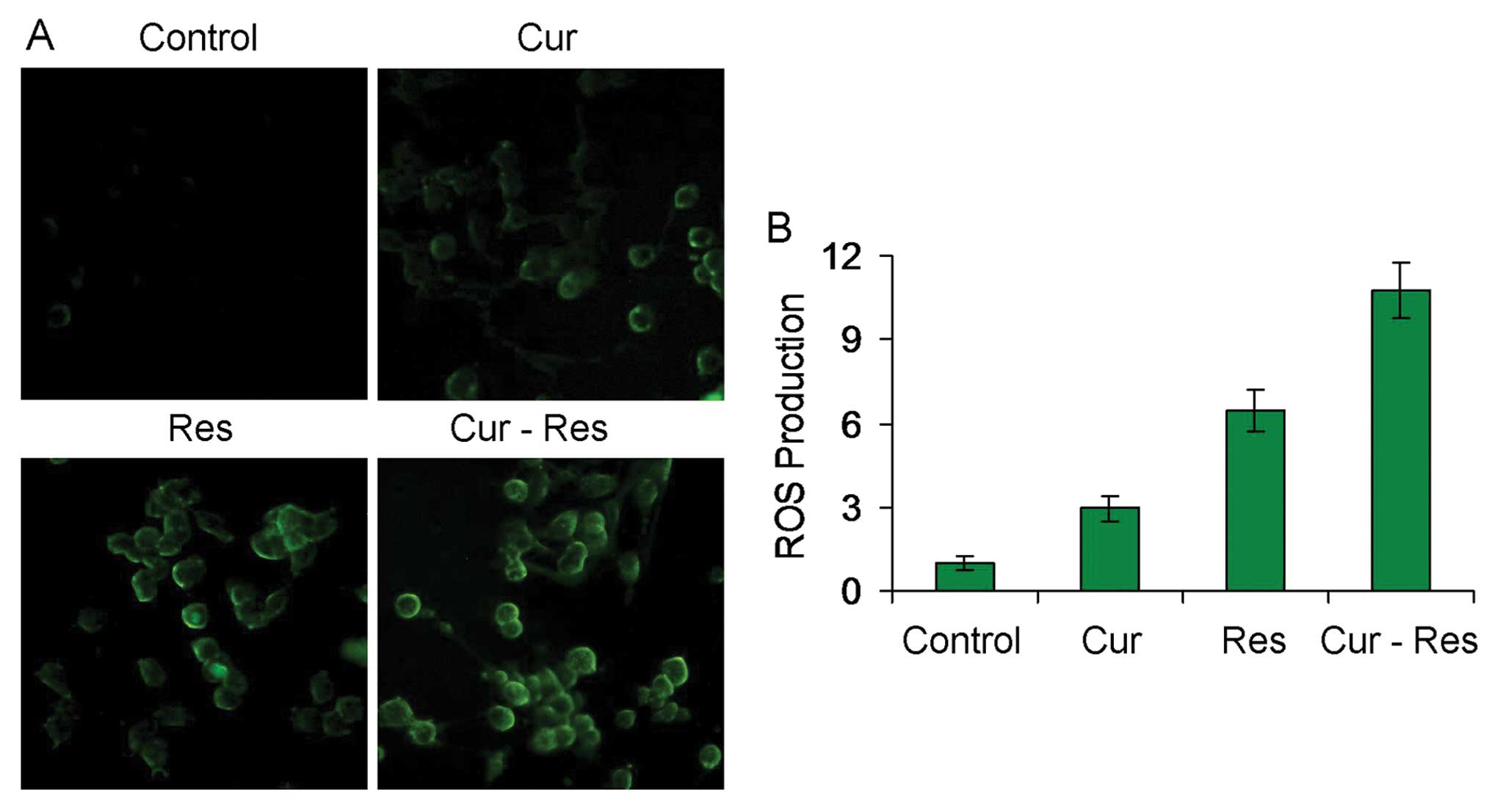
Combination of curcumin and resveratrol
induces ROS generation in Hepa1-6 cells
It has been confirmed that excess ROS promotes cell
death, and both curcumin and resveratrol upregulate ROS to induce
apoptosis in cancer cells (27–29).
Thus, we further examined the effects of the combination treatment
of curcumin and resveratrol on ROS production in Hepa1-6 cells by
DCFH-DA staining. As shown in Fig.
5, curcumin and/or resveratrol treatment resulted in
significant ROS generation as indicated by brightly green
fluorescence in the Hepa1-6 cells (P<0.01). Combination of
curcumin and resveratrol significantly enhanced the ROS generation
when compared to that following treatment with either agent alone
(P<0.01).
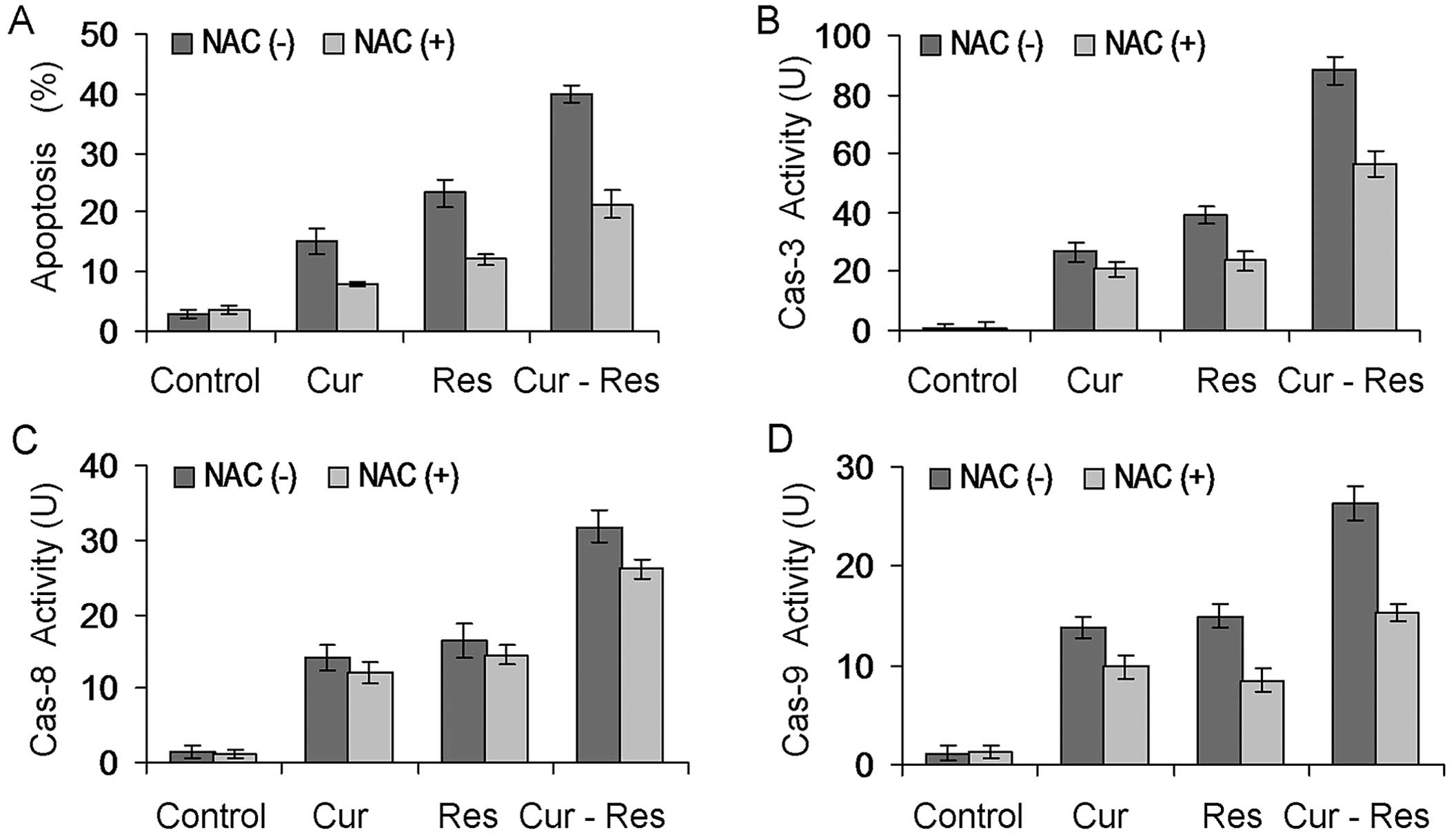
Role of ROS in curcumin and
resveratrol-induced apoptosis
To elucidate the effects of ROS on curcumin and
resveratrol-induced apoptosis, we further evaluated cell apoptosis
after NAC (ROS scavenger) pretreatment by flow cytometric analysis.
When compared without the blocking of NAC, apoptosis induced by
curcumin and/or resveratrol was partially but significantly
abrogated by NAC pretreatment (P<0.05) (Fig. 6A). Particularly in the combination
treatment group, the percentage of apoptosis was reduced to almost
half of the apoptosis noted in the absence of NAC. In addition,
curcumin and/or resveratrol-induced activation of caspase-3, -8 and
-9 was also reduced by NAC pretreatment, particularly in the
combination treatment group (P<0.05) (Fig. 6B-D). These observations suggest that
the combination treatment of curcumin and resveratrol induced
apoptosis, and caspase activation was associated with ROS
production.
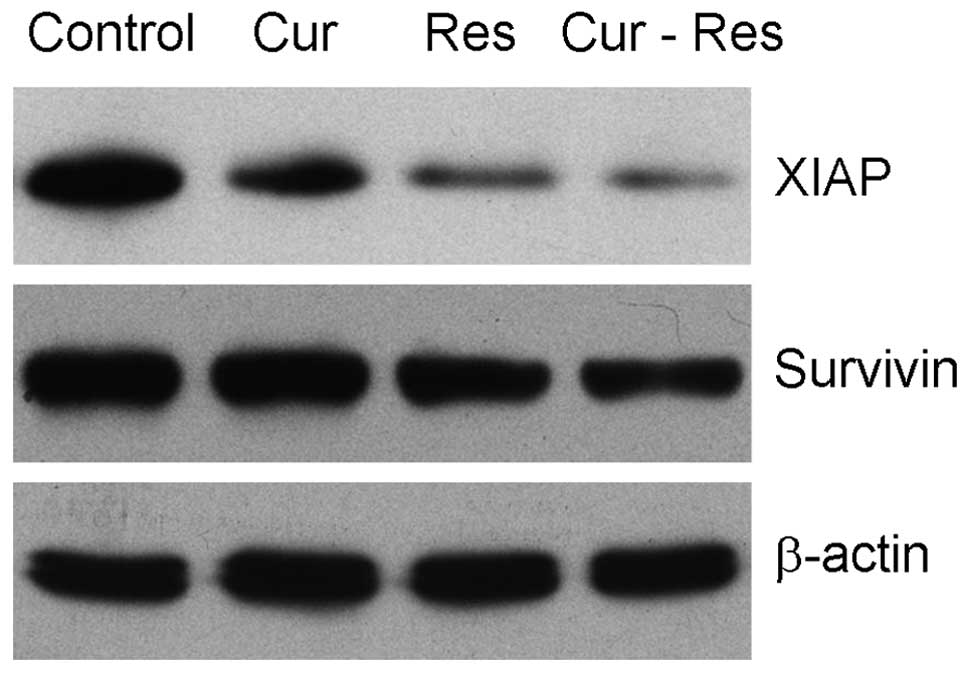
Combined effects of resveratrol and
curcumin on expression of the inhibitors of apoptosis proteins
It has been reported that the inhibitors of
apoptosis proteins, such as XIAP and survivin, are abnormally
expressed in hepatocarcinoma cells and they have been confirmed as
therapeutic targets for natural products (30–32).
We further evaluated the effects of the combination treatment of
curcumin and resveratrol on XIAP and survivin expression in Hepa1-6
cells. As shown in Fig. 7,
combination treatment with curcumin and resveratrol significantly
inhibited XIAP and survivin expression in the Hepa1-6 cells, when
compared to the expression in the control or following treatment
with either agent alone.
Discussion
In Chinese herbalism, every herb has its own
characteristics, and TCM physicians believe that illness can be
effectively treated by combining herbs based on their various
features. Combinations of multiple herbs guided by the theories of
TCM, called Chinese herbal formula, are the major methods for the
application of Chinese herbs. Due to the lack of appropriate
ancient Chinese herbal formulas for cancer, most TCM physicians
combine multiple herbs for a formula or prescription based on the
illness and body condition of patients, TCM principles, progress in
pharmacological research and personal experience. However, the
combination of different anticancer herbs may elicit additive,
synergisic or antagonistic effects (33). Thus, there is a great need to
explore the combination effects of anticancer herbs, and establish
effective herbal formulas for various situations in cancer
treatment (33,34).
Paired-herbs, consisting of two fixed Chinese herbs
that play a complementary role, are another simplified form of
herbal combinations to strengthen treatment efficacy. Paired-herbs
usually originate from clinical experience or an ancient Chinese
herbal formula. It is rational to explore the combination rule of
anticancer herbs based on paired-herbs. Curcuma aromatica
and Polygonum cuspidatum have been frequently used as a
paired-herbal medication for liver cancer treatment. Curcumin and
resveratrol are the major anticancer constituents from Curcuma
aromatica and Polygonum cuspidatum, respectively.
Combination treatment of curcumin and resveratrol has been found to
demonstrate a synergistic anticancer effect in colon cancer
(21). Recent research found that
resveratrol enhanced curcumin-induced apoptosis in osteosarcoma
cells (35). In the present study,
we observed that curcumin and resveratrol significantly inhibited
Hepa1-6 cell proliferation. Combination treatment with curcumin and
resveratrol significantly enhanced the antiproliferative effects
when compared with either agent alone. The CI value was <1 for
most of the concentrations tested, indicating a synergistic effect
between curcumin and resveratrol in Hepa1-6 cells.
Apoptosis, first proposed by Kerr et al in
1972, is a normal physiological process that maintains cell
homeostasis (36). Apoptosis plays
a key role in the pathogenesis of diseases, such as degenerative
diseases and cancer (37).
Apoptosis has been recognized as a major anticancer therapeutic
response (37,38). Manipulation of apoptosis has become
one of the popular strategies of cancer treatment. For example,
doxorubicin and cisplatin induce apoptosis in HepG2 hepatoma cells
via different signaling pathways (39,40).
Our data demonstrated that the combination of curcumin and
resveratrol induced apoptotic cell death as indicated by nuclear
morphological alterations and positive Annexin/PI staining. This
suggests a similar mechanism as other anticancer therapy.
Therefore, induction of apoptosis is an important mechanism in the
elicited anticancer effects of the combination treatment of
curcumin and resveratrol.
The death receptor pathway and mitochondrial pathway
are the primary apoptotic pathways (37,38).
Caspase-8 is the initiator in the death receptor pathway, activated
by conformational change. It is reported that curcumin induces
apoptosis via activation of the Fas/caspase-8 pathway (41). Caspase-9, an important initial
factor in mitochondrial pathways, could be activated by the complex
consisting of cytochrome c, pro-caspase-9 and apoptotic protease
activating factor-1 (Apaf-1). Resveratrol markedly promotes
caspase-9 activation, which is associated with provoking the
release of CytC (42). Once
activated, the two pathways converge to a common execution phase
and stimulation of caspase-3 activation (43). In the present study, the activities
of caspase-8, -9 and -3 were dramatically enhanced by the
combination treatment of curcumin and resveratrol. In addition, the
apoptosis of Hepa1-6 cells induced by the combination treatment of
curcumin and resveratrol was completely abrogated by the pan
caspase inhibitor, Z-VAD-FMK. These results suggest that both
extrinsic and intrinsic apoptotic signaling pathways are involved
in the apoptosis induced by the combination treatment of curcumin
and resveratrol.
ROS are natural products and are generated by
metabolism and xenobiotic exposure. High levels of ROS promote cell
death via the mitochondrial pathway and/or death receptor pathway
(27,44). Natural products, such as wogonin,
emodin and cordycepin may induce ROS generation to mediate their
proapoptosis effects (45–47). In the present study, the combination
treatment of curcumin and resveratrol resulted in significant ROS
generation in Hepa1-6 cells. Moreover, NAC, a scavenger of free
radicals, significantly abrogated curcumin and resveratrol-induced
apoptosis. Furthermore, the caspase activation induced by the
combination of curcumin and resveratrol was also reduced by NAC
pretreatment, suggesting that ROS contributed to the increased
apoptosis caused by the combination treatment of curcumin and
resveratrol.
XIAP and survivin proteins are members of the
inhibitors of the apoptotic protein (IAP) family. Both inhibit
caspase activation to protect cells from apoptosis. XIAP can
inhibit caspase-3 by a linker region between the first two BIR
domains, and suppress caspase-9 via its third BIR domains to
protect cells from apoptosis (48).
It has been reported that the activity of caspase-9 was
significantly elevated in lung cancer A549 cells transfected with
siRNA against survivin (49). Our
data showed that the combination of curcumin and resveratrol
downregulated the expression of XIAP and survivin, suggested that
the downregulation of XIAP and survivin may contribute to the
anticancer effects induced by the combination treatment of curcumin
and resveratrol.
In conclusion, our results demonstrated that
combination treatment of curcumin and resveratrol elicits a
synergistic anticancer effect in hepatocarcinoma cells via
extrinsic and intrinsic apoptosis, and is associated with ROS
generation and downregulation of XIAP and survivin. The present
study suggests that a combination of curcumin and resveratrol is a
promising novel anticancer treatment strategy for liver cancer. The
present study also provides new insights into the effective
mechanism of paired-herbs in Traditional Chinese Medicine.
Acknowledgements
This study was supported, in part, by the National
Natural Science Foundation of China (81273726), the Program of the
Science and Technology Commission of Shanghai Municipality
(11495801300), the Three-Year Action Program of Shanghai
Municipality for Traditional Chinese Medicine (ZYSNXD-CC-ZDYJ017),
the Long-Yi Scholars and the Research Team Program from the State
Clinical Research Center of TCM in Longhua Hospital (LYTD-04), the
Xing-Lin Scholars Program and the Key Discipline of the State
Administration of Traditional Chinese Medicine (Traditional Chinese
Medicine in Oncology, LHZLK-1107), and the Shanghai Shen Kang
Platform Grant (SHDC12007206). We also thank the Shanghai Key
Laboratory of Tissue Engineering for the technical assistance.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Rampone B, Schiavone B and Confuorto G:
Current management of hepatocellular cancer. Curr Oncol Rep.
12:186–192. 2010. View Article : Google Scholar
|
|
3
|
El-Serag HB: Hepatocellular carcinoma. N
Engl J Med. 365:1118–1127. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Du Q, Hu B, Shen KP and An HM:
Pathogenesis and treatment law of Traditional Chinese Medicine in
hepatocarcinoma. World J Integr Tradit West Med. 5:814–817.
2010.(In Chinese).
|
|
5
|
Tang CC, Qin KL and Huang KX: The chemical
constituents of the stems and leaves of Curcuma aromaticaand
its anticancer activities. J Wenzhou Med Coll. 37:110–113. 2007.(In
Chinese).
|
|
6
|
Hu B, Shen KP, An HM, Wu Y and Du Q:
Aqueous extract of Curcuma aromatica induces apoptosis and
G2/M arrest in human colon carcinoma LS-174-T cells independent of
p53. Cancer Biother Radiopharm. 26:97–104. 2011.
|
|
7
|
Dai GH, Yang F and Tong YL: Study on
effect of Polygoniextract on human hepatocarcinoma cell line
HepG-2 in vitro. Chinese J Tradit Med Sci Technol. 16:376–377.
2009.(In Chinese).
|
|
8
|
Shin JA, Shim JH, Jeon JG, Jeon JG, Choi
KH, Choi ES, Cho NP and Cho SD: Apoptotic effect of Polygonum
cuspidatum in oral cancer cells through the regulation of
specificity protein 1. Oral Dis. 17:162–170. 2011.
|
|
9
|
Lin YW, Yang FJ, Chen CL, Lee WT and Chen
RS: Free radical scavenging activity and antiproliferative
potential of Polygonum cuspidatum root extracts. J Nat Med.
64:146–152. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li M and Zhang N: To study the quality
control of Curcuma aromatica. J Chinese Med Mater.
31:540–543. 2008.(In Chinese).
|
|
11
|
Ravindran J, Prasad S and Aggarwal BB:
Curcumin and cancer cells: how many ways can curry kill tumor cells
selectively? AAPS J. 11:495–510. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Feng L, Zhang LF, Yan T, Jin J and Tao WY:
Studies on active substance of anticancer effect in Polygonum
cuspidatum. J Chinese Med Mater. 29:689–691. 2006.(In
Chinese).
|
|
13
|
Jang M, Cai L, Udeani GO, Slowing KV,
Thomas CF, Beecher CW, Fong HH, Farnsworth NR, Kinghorn AD, Mehta
RG, Moon RC and Pezzuto JM: Cancer chemopreventive activity of
resveratrol, a natural product derived from grapes. Science.
275:218–220. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Weng CJ, Yang YT, Ho CT and Yen GC:
Mechanisms of apoptotic effects induced by resveratrol,
dibenzoylmethane, and their analogues on human lung carcinoma
cells. J Agric Food Chem. 57:5235–5243. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mo W, Xu X, Xu L, Wang F, Ke A, Wang X and
Guo X: Resveratrol inhibits proliferation and induces apoptosis
through the hedgehog signaling pathway in pancreatic cancer cells.
Pancreatology. 11:601–609. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liao PC, Ng LT, Lin LT, Richardson CD,
Wang GH and Lin CC: Resveratrol arrests cell cycle and induces
apoptosis in human hepatocellular carcinoma Huh-7 cells. J Med
Food. 13:1415–1423. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tang FY, Su YC, Chen NC, Hsieh HS and Chen
KS: Resveratrol inhibits migration and invasion of human
breast-cancer cells. Mol Nutr Food Res. 52:683–691. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Weng YL, Liao HF, Li AF, Chang JC and
Chiou RY: Oral administration of resveratrol in suppression of
pulmonary metastasis of BALB/c mice challenged with CT26 colorectal
adenocarcinoma cells. Mol Nutr Food Res. 54:259–267. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Patel BB and Majumdar AP: Synergistic role
of curcumin with current therapeutics in colorectal cancer:
minireview. Nutr Cancer. 61:842–846. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rashid A, Liu C, Sanli T, Tsianli E, Singh
G, Bristow RG, Dayes I, Lukka H, Wright J and Tsakiridis T:
Resveratrol enhances prostate cancer cell response to ionizing
radiation. Modulation of the AMPK, Akt and mTOR pathways. Radiat
Oncol. 6:1442011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Majumdar AP, Banerjee S, Nautiyal J, Patel
BB, Patel V, Du J, Yu Y, Elliott AA, Levi E and Sarkar FH: Curcumin
synergizes with resveratrol to inhibit colon cancer. Nutr Cancer.
61:544–553. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Smith JA and Weidemann MJ: Further
characterization of the neutrophil oxidative burst by flow
cytometry. J Immunol Methods. 162:261–268. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hu B, An HM, Shen KP, Xu L, Du Q, Deng S
and Wu Y: Liver Yin deficiency tonifying herbal extract induces
apoptosis and cell senescence in Bel-7402 human hepatocarcinoma
cells. Exp Ther Med. 3:80–86. 2012.PubMed/NCBI
|
|
24
|
Hu B, An HM, Shen KP, Xu L, Du Q, Deng S
and Wu Y: Modified Yi Guan Jian, a Chinese herbal formula, induces
anoikis in Bel-7402 human hepatocarcinoma cells in vitro.
Oncol Rep. 26:1465–1470. 2011.PubMed/NCBI
|
|
25
|
Chou TC: Drug combination studies and
their synergy quantification using the Chou-Talalay method. Cancer
Res. 70:440–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Meier P and Vousden KH: Lucifer’s
labyrinth - ten years of path finding in cell death. Mol Cell.
28:746–754. 2007.
|
|
27
|
Circu ML and Aw TY: Reactive oxygen
species, cellular redox systems, and apoptosis. Free Radic Biol
Med. 48:749–762. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang CL, Ma YG, Xue YX, Liu YY, Xie H and
Qiu GR: Curcumin induces small-cell lung cancer NCI-H446 cell
apoptosis via the reactive oxygen species-mediated mitochondrial
pathway and not the cell death receptor pathway. DNA Cell Biol.
31:139–150. 2012. View Article : Google Scholar
|
|
29
|
Juan ME, Wenzel U, Daniel H and Planas JM:
Resveratrol induces apoptosis through ROS-dependent mitochondria
pathway in HT-29 human colorectal carcinoma cells. J Agric Food
Chem. 56:4813–4818. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Augello C, Caruso L, Maggioni M, Donadon
M, Montorsi M, Santambrogio R, Torzilli G, Vaira V, Pellegrini C,
Roncalli M, Coggi G and Bosari S: Inhibitors of apoptosis proteins
(IAPs) expression and their prognostic significance in
hepatocellular carcinoma. BMC Cancer. 9:1252009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shyu MH, Kao TC and Yen GC: Oleanolic acid
and ursolic acid induce apoptosis in HuH7 human hepatocellular
carcinoma cells through a mitochondrial-dependent pathway and
downregulation of XIAP. J Agric Food Chem. 58:6110–6118. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhong X, Wu B, Pan YJ and Zheng S:
Brazilein inhibits survivin protein and mRNA expression and induces
apoptosis in hepatocellular carcinoma HepG2 cells. Neoplasma.
56:387–392. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Du Q, Hu B and Shen KP: Compatibility of
Chinese anti-cancer herb. Chinese J Exp Tradit Med Formu.
16:232–235. 2011.(In Chinese).
|
|
34
|
Hu B, Du Q, Shen KP and Xu L: Principles
and scientific basis of traditional Chinese medicine in cancer
treatment. J Bioanal Biomed. S6:0052012. View Article : Google Scholar
|
|
35
|
Masuelli L, Marzocchella L, Focaccetti C,
Tresoldi I, Palumbo C, Izzi V, Benvenuto M, Fantini M, Lista F,
Tarantino U, Modesti A, Galvano F and Bei B: Resveratrol and
diallyl disulfide enhance curcumin-induced sarcoma cell apoptosis.
Front Biosci. 17:498–508. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kerr JF, Wyllie AH and Currie AR:
Apoptosis: a basic biological phenomenon with wide-ranging
implications in tissue kinetics. Br J Cancer. 26:239–257. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wong RS: Apoptosis in cancer: from
pathogenesis to treatment. J Exp Clin Cancer Res. 30:872011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Brown JM and Attardi LD: The role of
apoptosis in cancer development and treatment response. Nat Rev
Cancer. 5:231–237. 2005.PubMed/NCBI
|
|
39
|
Lee TK, Lau TC and Ng IO:
Doxorubicin-induced apoptosis and chemosensitivity in hepatoma cell
lines. Cancer Chemother Pharmacol. 49:78–86. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Qin LF and Ng IO: Induction of apoptosis
by cisplatin and its effect on cell cycle-related proteins and cell
cycle changes in hepatoma cells. Cancer Lett. 175:27–38. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu SH, Hang LW, Yang JS, Chen HY, Lin HY,
Chiang JH, Lu CC, Yang JL, Lai TY, Ko YC and Chung JG: Curcumin
induces apoptosis in human non-small cell lung cancer NCI-H460
cells through ER stress and caspase cascade- and
mitochondria-dependent pathways. Anticancer Res. 30:2125–2133.
2010.PubMed/NCBI
|
|
42
|
Zuo Y, Xiang B, Yang J, Sun X, Wang Y,
Chang H and Yi J: Oxidative modification of caspase-9 facilitates
its activation via disulfide-mediated interaction with Apaf-1. Cell
Res. 19:449–457. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mazumder S, Plesca D and Almasan A:
Caspase-3 activation is a critical determinant of genotoxic
stress-induced apoptosis. Methods Mol Biol. 414:13–21.
2008.PubMed/NCBI
|
|
44
|
Lau AT, Wang Y and Chiu JF: Reactive
oxygen species: current knowledge and applications in cancer
research and therapeutic. J Cell Biochem. 104:657–667. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lin CC, Kuo CL, Lee MH, Lai KC, Lin JP,
Yang JS, Yu CS, Lu CC, Chiang JH, Chueh FS and Chung JG: Wogonin
triggers apoptosis in human osteosarcoma U-2 OS cells through the
endoplasmic reticulum stress, mitochondrial dysfunction and
caspase-3-dependent signaling pathways. Int J Oncol. 39:217–224.
2011.
|
|
46
|
Ma YS, Weng SW, Lin MW, Lu CC, Chiang JH,
Yang JS, Lai KC, Lin JP, Tang NY, Lin JG and Chung JG: Antitumor
effects of emodin on LS1034 human colon cancer cells in vitro and
in vivo: roles of apoptotic cell death and LS1034 tumor xenografts
model. Food Chem Toxicol. 50:1271–1278. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jeong JW, Jin CY, Park C, Hong SH, Kim GY,
Jeong YK, Lee JD, Yoo YH and Choi YH: Induction of apoptosis by
cordycepin via reactive oxygen species generation in human leukemia
cells. Toxicol In Vitro. 25:817–824. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sun C, Cai M, Gunasekera AH, Meadows RP,
Wang H, Chen J, Zhang H, Wu W, Xu N, Nq SC and Fesik SW: NMR
structure and mutagenesis of the inhibitor-of-apoptosis protein
XIAP. Nature. 401:818–822. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen XQ, Yang S, Li ZY, Lu HS, Kang MQ and
Lin TY: Effects and mechanism of downregulation of survivin
expression by RNA interference on proliferation and apoptosis of
lung cancer cells. Mol Med Rep. 5:917–922. 2012.PubMed/NCBI
|