Introduction
Colorectal cancer (CRC) is one of the most commonly
diagnosed cancers and is a common cause of cancer-related deaths in
developed countries (1). Prognosis
strongly depends on tumor stage at presentation. The 5-year overall
survival ranges from 93% for stage I patients to 8% for stage IV
patients (2). Currently, the only
curative treatment is surgical resection, with a modest survival
benefit from chemotherapy, particularly in stage III disease. Stage
III disease is defined by the presence of positive lymph nodes. The
impact of adjuvant chemotherapy (CT) for patients with stage III
CRC has been addressed in several clinical trials and
practice-based studies demonstrating a benefit of adjuvant therapy
in these patients (3,4). However, current clinical experience
with certain patients that may not be suitable for CT-based
therapies (due to age, comorbid disease and/or potential
treatment-related toxicity) faces a significant number of stage III
CRC patients who do not suffer from relapse in the absence of
adjuvant therapy. To date, substantial efforts have been given to
the identification of clinicopathological and molecular parameters
that improve the prognostic stratification of patients. To date,
there are no available biomarkers to predict CRC recurrence.
microRNAs (miRNAs) are a newly discovered class of
short 22 nucleotide non-coding RNAs that repress protein
translation through binding to target mRNAs. Bioinformatics and
cloning studies have estimated that miRNAs may regulate 30% of all
human genes and control hundreds of gene targets (5). There is growing evidence that
expression of miRNAs plays an important role in various cellular
processes commonly implicated in cancer, such as differentiation,
invasion, cell growth, angiogenesis and epithelial-mesenchymal
transition (6–8). Data from several studies show that
different miRNAs are aberrantly expressed in various hematological
and solid malignant diseases including CRC (9–11).
Functional studies indicate that miRNAs have both tumor-suppressive
and oncogenic potential in human cancer (12) and can control various aspects of
cancer biology such as chemoresistance (13) and metastasis (14). Accordingly, an increasing number of
pro-metastatic and anti-metastatic miRNAs have been identified from
expression profiling of tumor versus non-tumor tissue (15).
Accumulating data indicate that miRNAs are present
in body fluids including blood plasma and serum, urine, saliva and
semen (16,17). Circulating miRNAs are stable and
have been revealed as successful biomarkers for different
malignancies including solid tumors (16,18).
Mammalian cells in culture have also been reported to export miRNAs
into the extracellular environment (19,20).
To date, the discovery of miRNAs as novel biomarkers in serum or
plasma have opened up a new and interesting field in the screening
and monitoring of cancer patients.
In the present study, we aimed to establish
comprehensive miRNA expression patterns in stage III CRC patients.
Using microarray profiling we identified differentially expressed
upregulated and downregulated miRNAs between CRC tissues and paired
neighboring noncancerous colorectal tissues. To investigate
candidate circulating cancer-associated miRNAs we determined the
expression profile of upregulated tumoral miRNAs in serum samples
of stage III CRC patients and healthy individuals.
Patients and methods
Ethics statement
The study was approved by the institutional medical
ethics boards of our hospital (Corporació Sanitària Parc Taulí,
Institut Universitari Parc Taulí-UAB). Serum samples of the
patients and control were prospectively obtained, and both gave
written informed consent to participate in the study. The tissue
samples were obtained from formalin-fixed paraffin-embedded (FFPE)
blocks stored in the Pathology Department of the hospital. For
these patients, informed consent was not obtained in accordance
with the Spanish Law 14/2007 for Biomedical Research. According to
the law, archive samples may be used for biomedical research
without informed consent when obtaining informed consent is not
possible or extremely difficult, provided that i) the investigation
performed is of general interest, ii) there is no previous
declaration of the patient against the use of the samples for
research, iii) data confidentiality is granted and iv) the ethics
committee of the hospital evaluates and approves the study
protocol.
Patients and samples
For the microarray analysis, 12 FFPE stage III
colonic tumor tissue specimens and their surrounding non-cancerous
normal mucosa were collected from 12 CRC patients who underwent
surgical resection between 2000 and 2005 at the Department of
Oncology at the hospital of Sabadell (Corporació Sanitària Parc
Taulí, Institut Universitari Parc Taulí-UAB), Barcelona, Spain.
Tumors were staged according to the Tumor-Node-Metastasis (TNM)
staging system.
Peripheral blood samples (4 ml/individual) were
collected from 56 individuals: 30 stage III CRC patients and 26
healthy volunteers (13 female and 13 male with a median age of
64.1±7.4 years) with no prior history of solid or hematological
malignancy. No neoadjuvant therapy was used at the time when the
blood samples were obtained.
Clinical and pathologic data were extracted from the
medical records and reviewed for the purpose of this study.
Detailed patient information is documented in Table I.
 | Table IDemographic and clinicopathological
characteristics of the colorectal cancer patients. |
Table I
Demographic and clinicopathological
characteristics of the colorectal cancer patients.
| Variables | Tissue samples | Serum samples |
|---|
| No. of patients | 12 | 30 |
| Median age (years ±
SD) | 78.3±8 | 68.1±11 |
| Gender |
| Male | 9 | 18 |
| Female | 3 | 12 |
| Tumour size (cm) |
| <4 | 2 | 12 |
| ≥4 | 10 | 18 |
| Degree of
differentiation |
| Poor | 7 | 23 |
| Moderate-Well | 5 | 7 |
| Vascular
invasion |
| Yes | 4 | 10 |
| No | 8 | 20 |
| Perineural
invasion |
| Yes | 4 | 14 |
| No | 8 | 16 |
| T stage |
| T3 | 5 | 20 |
| T4 | 7 | 10 |
| N stage |
| N1 | 6 | 18 |
| N2 | 6 | 12 |
| M stage |
| M0 | 12 | 30 |
| Clinical stage |
| III | 12 | 30 |
| Localization |
| Left | 6 | 9 |
| Right | 5 | 17 |
| Rectum | 1 | 4 |
Sample processing and RNA isolation
miRNA-enriched total RNA from tissue specimens was
extracted using RecoverAll™ Total Nucleic Acid isolation kit for
FFPE samples (Ambion, Austin, TX, USA) following the manufacturer’s
protocol. Purity and concentration of the RNA were determined from
OD260/280 readings using a spectrophotometer. RNA integrity was
determined using the Bioanalyzer 2100 (Agilent Technologies Inc.,
Santa Clara, CA, USA).
A blood sample from each participant was collected
in a serum clot activator tube (Vacuette ref. 454067) before
surgical resection and centrifuged at 3,500 × g for 10 min at 4°C
to spin down the blood cells. Serum was then aliquoted and stored
at −80°C until use. RNA was isolated from 200 μl serum using TRIzol
LS (Invitrogen Life Technologies, Paisley, UK) as described by the
manufacturer and finally eluted into a 50-μl volume of RNA-free
water.
Microarray analysis
Microarray analysis was carried out by an external
core facility (www.crg.es) using Agilent Human miRNA microarray
including 887 human miRNAs, representing the miRBase set v14.0
(Agilent Technologies, Palo Alto, CA, USA).
Total RNA (100 ng) was labeled using Agilent miRNA
Complete Labeling and Hyb kit (Agilent, cat. no. 5190-0456)
following the manufacturer’s instructions. Briefly, total RNA from
each sample was dephosphorylated using calf intestinal alkaline
phospatase (CIP), and cyanine 3-pCp was then ligated using the T4
RNA ligase. Labeled samples were dried and resuspended in 18 μl of
nuclease-free water and co-hybridized with in situ
hybridization buffer for 20 h at 55°C on Agilent Human miRNA
microarray release 14.0, 8×15K (G4471A-029297). Arrays were washed
at room temperature for 5 min in Gene Expression Wash Buffer 1 and
5 min at 37°C in Gene Expression Wash Buffer 2 (both from Agilent
Technologies Inc.). Slides were scanned using Agilent’s
High-Resolution Scanner (G2539A) at 5 μm resolution and quantified
using Feature Extraction software (Agilent Technologies Inc.).
Quantification of miRNAs by qRT-PCR
Quantitative real-time polymerase chain reaction
(qRT-PCR) was used to validate miRNAs upregulated in the microarray
experiment data to determine the level of the same miRNAs in serum
samples of CRC patients and healthy controls.
cDNA synthesis was performed using miRCURY LNA™
Universal RT microRNA PCR kit (Exiqon A/S, Vedbaek, Denmark)
according to the manufacturer’s instructions. Briefly, 20 ng of
total RNA from tissue or 4 μl of total RNA from 200 μl of serum was
polyadenylated, and cDNA was synthesized using a poly(T) primer
with a 3′ degenerate anchor and a 5′ universal tag. Then, cDNA
served as a template for miRNA qPCR using FastStart Universal
SYBR-Green Master (Rox) (Roche Applied Science, Indianapolis, IN,
USA; cat no. 04913850001) and Exiqon-validated specific primer
sets. The amplification profile was denatured in 96-well optical
plates at 95°C for 10 min, followed by 40 cycles of 95°C for 10 sec
and 60°C for 60 sec. At the end of the PCR cycles, melting curve
analyses were performed. For each sample, both the cDNA synthesis
and real-time PCR assays were performed in duplicate in a final
volume of 20 μl using an Applied Biosystems 7500 Sequence Detection
system (Applied Biosystems, Foster City, CA, USA).
Data analysis and statistics
For statistical analysis of microarray data,
extracted intensities were background corrected using the normexp
method with an offset of 50 (21).
To assure comparability across samples, we used quantile
normalization on background corrected log2-transformed intensities.
Microarray probes were collapsed to miRNAs by taking the median
intensity of the respective probes per miRNA. For determining
differentially regulated miRNAs, moderated t-tests were applied
using limma (22). miRNAs with FDR
adjusted p-value <5% and additionally a fold-change (FC) >1.5
in absolute value were selected as the relevant ones. All
statistical analyses were performed with the Bioconductor project
in the R statistical environment (23).
According to Exiqon’s guidelines for microRNA qPCR
in serum/plasma, we chose miR-16, miR-103 and miR-let-7a as
reference miRNAs for normalization of expression levels. Using
DataAssist™ Software v3.0 (Applied Biosystems) the fold-change in
the miRNA expression was calculated by the equation
2−ΔΔCT(24). Paired
t-test was performed and p-values were adjusted using the Benjamini
and Yekutieli false discovery rate method (25).
Results
Differentially expressed miRNAs in stage
III CRC tumors and adjacent non-tumor tissues
Comparison of the expression profiles of 887 human
miRNAs between normal and tumor tissue revealed a total of 43
differentially expressed miRNAs between tumors and corresponding
non-tumor samples with a fold change (FC) >1.5 and adjusted
p-value below the threshold level of 0.05. This miRNA signature was
able to separate the tumors from the normal group into two main
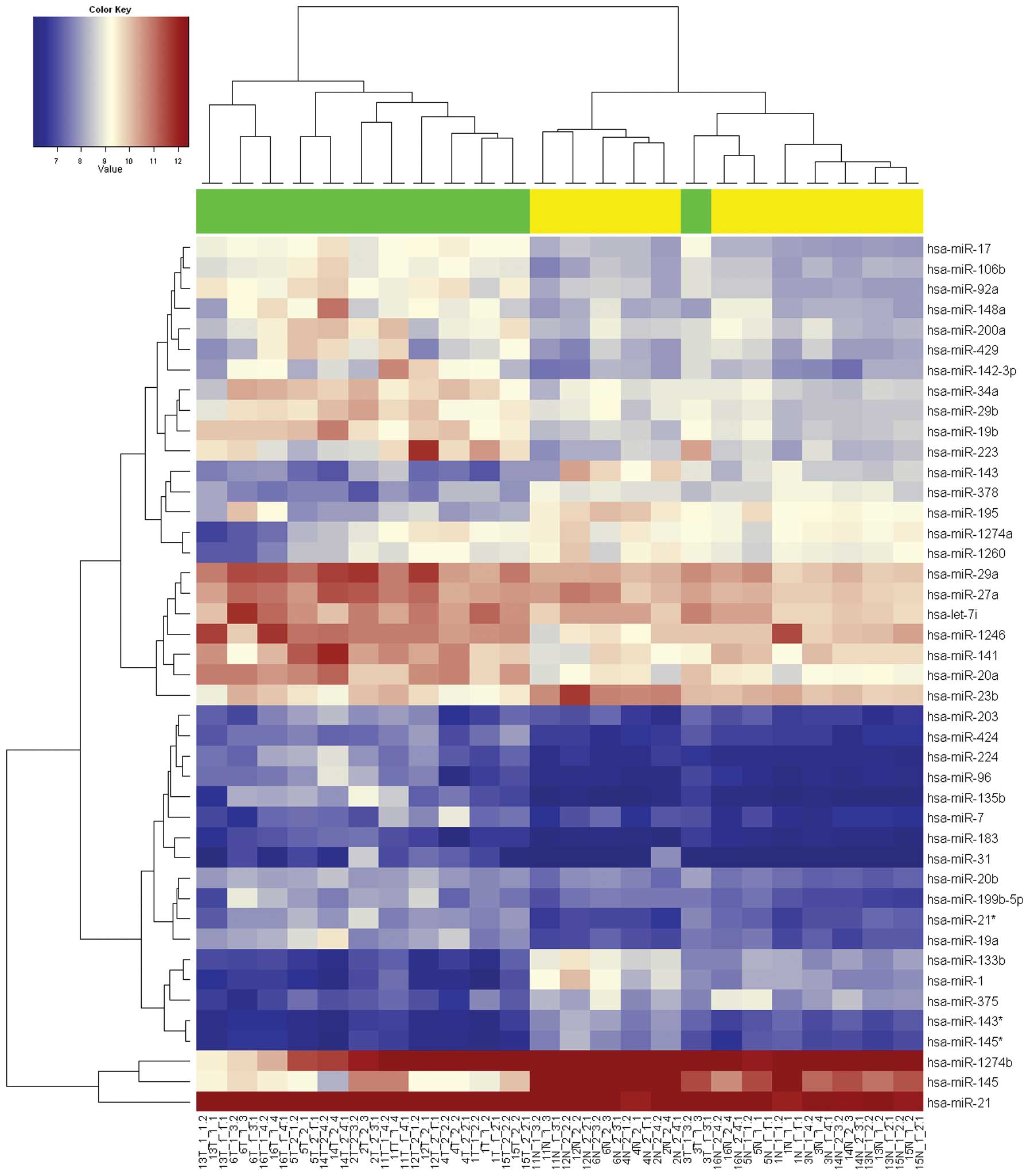
unsupervised hierarchial clusters shown in Fig. 1. The first cluster comprised all of
the non-tumor samples and one tumor sample (case 3). The second
cluster included 11 of the 12 tumor samples grouped together. Among
these differentially expressed miRNAs, 30 miRNAs showed increased
expression in colon tumors in relation to normal colon tissue, with
miR-21 being the most significantly upregulated miRNA in the tumors
at 3.6-fold. Expression of miR-20a, miR-19b, miR-17, miR-92a,
miR-19a, miR-92a, mapping to the same region of chromosome 13, were
highly correlated with a FC >2. Additionally, 13 miRNAs showed
decreased expression in colon tumors, with miR-145 and miR-1 being
the most downregulated at −5.2-fold and −3.1-fold,
respectively.
miRNA selection and qRT-PCR
validation
Thirteen overexpressed miRNAs in CRC tumors were
selected for validation using qRT-PCR in the same cohort of 12
paired samples investigated initially by the microarray assay. Ten
highly upregulated miRNAs (FC >1.9, p<0.05): miR-135b,
miR-142-3p, miR-20a, miR-21, miR-223, miR-224, miR-29a, miR-34a,
miR-92a, miR-96, along with 3 miRNAs which were selected on the
basis of previous studies supporting their involvement in CRC
prognosis: miR-141 (FC 1.76, p=0.04), miR-18a (FC 1.36, p=0.0035)
and miR-31 (FC 1.57, p=0.0035). Table
II summarizes the 13 selected miRNAs and their experimentally
observed associated target genes. Notably, the determined heat map
using only unsupervised clustering of these 13 miRNAs showed that
the entire normal tissue profile clustered together in one group
(including sample 3) and separately from the group of colon tumor
samples (data not shown).
 | Table IIList of 13 deregulated miRNAs
identified by array profiling in stage III CRC and selected for
experimental validation by qRT-PCR. |
Table II
List of 13 deregulated miRNAs
identified by array profiling in stage III CRC and selected for
experimental validation by qRT-PCR.
| miRNA | Chromosome
localization | FC | Experimentally
observed target genes |
|---|
| hsa-miR-21 | 17q23 | 3.56 | ACTA2, APAF1,
BMPR2, BTG2, CDC25A, CDK6, CDKN1A, E2F1, FAS, FASLG, IL6R, JAG1,
MARCKS, NFIB, PIK3R1, PTEN, SERPINB5, SOCS5, SPRY2, TGFBR2, TIMP3,
TPM1 |
| hsa-miR-135b | 1q32.1 | 2.76 | APC, JAK2, RUNX2,
SMAD5 |
| hsa-miR-20a | 13q31.3 | 2.48 | BAMBI, BCL2,
BCL2L11, BMPR2, CCND1, CDKN1A, E2F1, ESR1, IL8, JAK1, MICA, NCOA3,
PPARG, PTEN, RB1, RBL2, RUNX1, S1PR1, STAT3, TGFBR2, TP63, VEGFA,
VIM |
| hsa-miR-223 | Xq22 | 2.40 | IRS1, MEF2C, RHOB,
STMN1, VIM |
| hsa-miR-224 | Xq28 | 2.18 | AP2M1, KLK1 |
| hsa-miR-92a | 13q31.3
Xq26.2 | 2.08 | BCL2L11, BMPR2,
CDKN1A, CDKN1C, ITGA5, MAP2K4 |
| hsa-miR-96 | 1q32.2
7q32.2 | 2.05 | FOXO1, IRS1, MITF,
RYK |
| hsa-miR-142-3p | 17q22 | 2.01 | BCL2L1, LIFR |
| hsa-miR-34a | 1p36.22 | 1.92 | AXIN2, BCL2, CCND1,
CD47, CDK6, CREB1, DLL1, E2F3, HDAC1, JAG1, MAP2K1, MET, MYC, MYCN,
NOTCH1, NOTCH2, VEGFA, WNT1 |
| hsa-miR-29a | 7q32.3 | 1.90 | ACVR2A, CAV2,
CDC42, CDK6, COL1A1, COL1A2, COL3A1, DNMT3A, DNMT3B, DUSP2, HDAC4,
MCL1, PIK3R1, PPM1D, TCL1A, TGFB3, TPM1, YY1 |
| hsa-miR-141 | 12p13.31 | 1.76 | BAP1, CTNNB1,
CYP1B1, EPHA2, EXOC5, MAP2K4, PRKACB, TGFB2 |
| hsa-miR-31 | 9p21.3 | 1.57 | HIF1A, ITGA5,
PDGFB, PPP2R2A, RDX, RHOA |
| hsa-miR-18a | 13q31.3 | 1.36 | BCL2L11, CDKN1A,
CTGF, E2F1, ESR1, THBS1, TPG3 |
Statistically significant increases in 11 miRNAs
(miR-135b, miR-141, miR-18a, miR-20a, miR-21, miR-224, miR-29a,
miR-31, miR-34a, miR-92a, miR-96) were validated by qRT-PCR in the
12 CRC tissue samples compared with non-cancerous adjacent
colorectal mucosa. No significant differences were observed for
miR-142-3p and miR-223 (data not shown).
Evaluation of miRNAs in serum
samples
Employing qRT-PCR assay we then compared the
expression levels of the 11 validated miRNAs in serum samples from
56 individuals: 30 stage III CRC patients and 26 healthy donors.
Our data reflected that the relative levels of miR-135b, miR-141
and miR-224 were not consistently measurable in serum. miR-31
showed two peaks in the derivative melting curve and was therefore
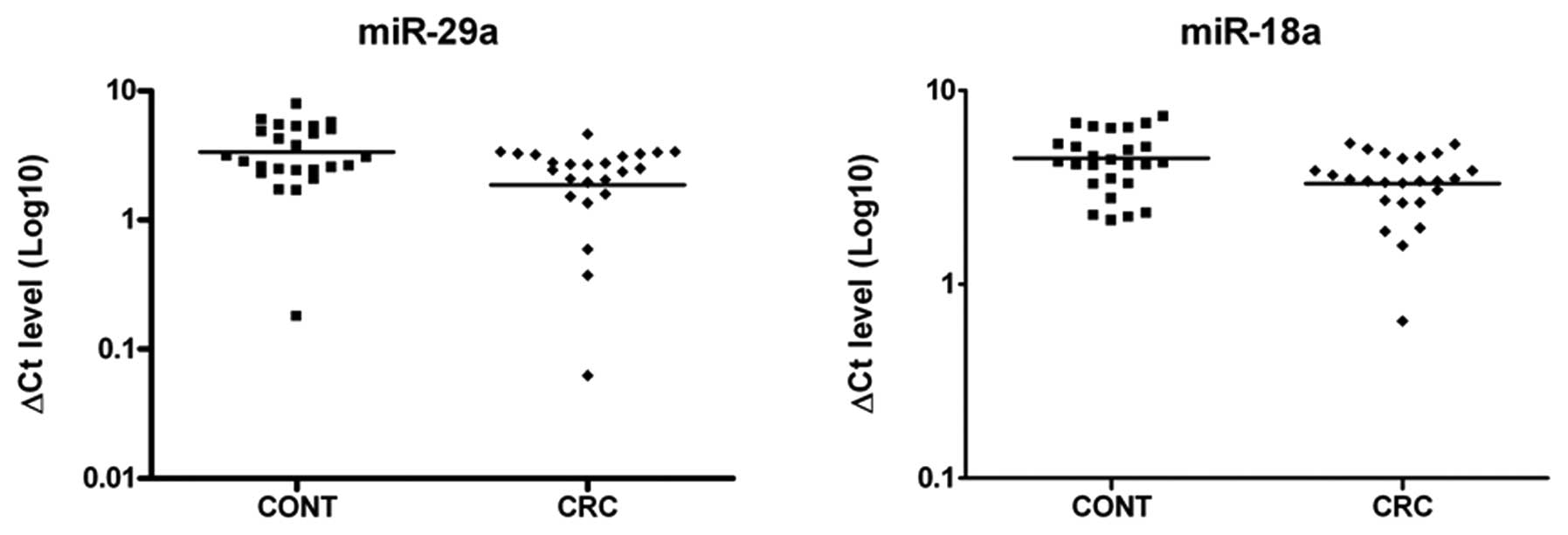
not considered for the analysis. Eventually, 7 miRNAs (miR-18a,
miR-20a, miR-21, miR-29a, miR-34a, miR-92a and miR-96) were
efficiently extracted and amplified from the serum of stage III CRC
patients and healthy individuals. The levels of expression of these
miRNAs are shown in Table III. We
found that the expression levels of miR-18a and miR-29a, normalized
to the level of miR-103, miR-16 and miR-let-7a endogenous controls,
were significantly elevated (Benjamini-Hochberg adjusted p<0.05)
in the serum of CRC patients compared with the healthy individuals
(Fig. 2).
 | Table IIIMean fold change of the 7 miRNAs
identified in the serum samples of CRC patients and healthy
individuals. |
Table III
Mean fold change of the 7 miRNAs
identified in the serum samples of CRC patients and healthy
individuals.
| miRNA | Type | Mean FC | P-value |
|---|
| miR-18a | Target | 2.229 | 0.038 |
| miR-20a | Target | 1.378 | 0.593 |
| miR-21 | Target | 2.081 | 0.087 |
| miR-29a | Target | 2.811 | 0.038 |
| miR-34a | Target | 3.205 | 0.055 |
| miR-92a | Target | 0.444 | 0.087 |
| miR-96 | Target | 2.267 | 0.107 |
| miR-103 | Selected
control | 1.324 | 0.496 |
| miR-16 | Selected
control | 0.829 | 0.593 |
| miR-let-7a | Selected
control | 0.910 | 0.702 |
Discussion
The identification of molecular markers that can
predict prognosis is an important aim in cancer research.
Accumulating evidence suggess that miRNAs play an active role in
controlling the development, differentiation and cancer
progression. miRNAs are well preserved in tissue samples even after
formalin-fixation and paraffin-embedding and can be efficiently
extracted and evaluated in tumors and also in body fluids such as
serum or plasma. Therefore, currently, there is a growing interest
in the study of miRNAs as molecular biomarkers with potential
clinical applicability to tumor classification, cancer diagnosis
and disease follow-up.
In the present study, we used a commercial platform
including 887 validated human miRNAs to investigate miRNA
expression profiling in stage III colon adenocarcinomas and
adjacent non-tumor tissues. In agreement with previous reports, our
data confirmed that numerous miRNAs are aberrantly expressed in CRC
in relation to normal tissue. Heat map and cluster analyses showed
that miRNA signatures were able to separate cancer tissue and
non-tumor tissue in most cases. We found concordant expression of
previously reported miRNAs altered in CRC (i.e. downregulation of
miR-143, miR.145 or miR-195 and upregulation of miR-21, miR-17-92
cluster or miR-135b). Moreover, we identified several other miRNAs
which have not previously been reported in CRC, probably since they
have been more recently included in array platforms (i.e.
downregulation of miR-1274a, miR-1274b, miR-1260 and upregulation
of miR-1246, miR-199b-5p, miR-29b). The current results support
other published studies and contribute to the better
characterization of differentially expressed miRNAs in stage III
CRC tumors.
The discovery of the presence of miRNAs in blood
(serum or plasma) has generated great scientific and clinical
interest. The expression pattern of miRNAs in the circulation could
be relevant to identify the high risk of disease progression in
CRC. While many studies have shown the overexpression or
downregulation of specific miRNAs in CRC tissue or cell lines,
there is less data concerning circulating tumor-associated
miRNAs.
Therefore, we investigated the circulating levels of
11 miRNAs overexpressed in tumors and identified in serum samples
of stage III CRC patients and healthy individuals. Our results
identified at least 2 highly overexpressed miRNAs (miR-135b and
miR-224) in tumor tissue that have not been previously detected in
the serum of patients nor in healthy individuals. Future studies
determining the cellular localization of these 2 miRNAs as well as
an increasing understanding of miRNA transportation outside cells
are warranted to address the absence of circulating miR-135b and
miR-224.
The present study identified 7 miRNAs that were
stable and detectable in all serum samples analyzed. To our
knowledge, this is the first study to investigate circulating
levels of miR-18a, miR-142-3p, miR-20a and miR-223 in serum samples
of CRC patients. Our results identified miR-18a and miR-29a, whose
expression was found to differ significantly between CRC patients
and normal donor sera. miR-18a, located at region 13q31.3, is one
of the 6 miRNAs encoded by the miR-17-92 cluster. This cluster,
also named oncomir-1, is known as a body of oncogenic miRNAs and
typically exhibits increased expression in tumors, including CRC.
Previous studies have reported significantly elevated levels of
both miR-17-3p and miR-92 in the blood of patients with CRC
(26,27). In our study, no significant
difference was observed in the levels of miR-92a between CRC
patients and healthy individuals (RQ=1.7362, p=0.453). In contrast,
we identified miR-18a as another miRNA included in the same cluster
which was significantly elevated in the serum of patients with CRC
(RQ=2.670, p<0.031). Of interest, in CRC, miR-18a*
targets specifically on K-Ras (28), and has recently been found to be
significantly higher in the plasma of pancreatic cancer patients
than that in the controls (29). To
our knowledge, our study is the first report to demonstrate miR-18a
as a potential novel non-invasive biomarker for CRC patients.
Consistent with another published report, we found
that serum miR-29a was significantly higher in CRC cancer patients
than in healthy individuals (27).
Results from recent studies revealed miR-29a as a significant
potential candidate prognostic biomarker in CRC patients. Higher
expression of miR-29a has been reported in stage II CRC tumoral
samples, and was associated with a longer disease-free survival
(30). Furthermore, upregulation of
miR-29a was observed in colorectal tumor tissue from liver
metastatic (CRLM) patients when compared with non-metastatic CRC
patients. Other authors, demonstrated that serum miR-29a can
discriminate CRLM patients from non-metastatic CRC patients with a
specificity and sensitivity higher than serum CEA (60% sensitivity
and 34% specificity) (31). In
addition, one study that combined in silico analysis and an
empirical experiment also indicated miR-29a as a potential
biomarker to predict the early recurrence of CRC. All of these
findings together indicate that the circulating level of miR-29a
warrants further testing as a promising serum biomarker of
prognosis in CRC patients.
In summary, the data presented in this study confirm
that miRNAs are differentially expressed between colonic tumor
tissues and adjacent tissues. Our results provide a list of 43
miRNAs that are differentially expressed between stage III CRC
tissue and corresponding normal tissue, which could contribute to
further explain the role of miRNAs in colonic tumors with lymph
node metastasis. Our data showed that not all overexpressed tumor
miRNAs are detectable in serum fluids. Further investigation of
miR-18a and miR-29a, preferably in large prospective studies, to
validate their potential use as circulating biomarkers in CRC
patients is warranted.
Acknowledgements
We are grateful to all the patients and healthy
individuals who kindly allowed samples to be collected and used in
this study. The authors would also like to acknowledge the nursing
staff Francisca Cuadras-Lacasa and Marta Fernández-Ros of
Corporació Sanitària Parc Taulí, Institut Universitari Parc
Taulí-UAB for their continued technical assistance and
collaboration. Finally, we thank Sarah Bonnin of the CRG Genomics
Unit for the excellent technical assistance. This study was
supported by ‘Fundació Benèfica Privada Barnola Vallribera Sant
Josep’ from Sabadell and by ‘Fundació Parc Taulí - Institut
Universitari UAB’ of the Sabadell Hospital (CIR2010029).
References
|
1
|
Weitz J, Koch M, Debus J, Höhler T, Galle
PR and Büchler MW: Colorectal cancer. Lancet. 365:153–165. 2005.
View Article : Google Scholar
|
|
2
|
O’Connell JB, Maggard MA and Ko CY: Colon
cancer survival rates with the new American Joint Committee on
Cancer 6th edition staging. J Natl Cancer Inst. 96:1420–1425.
2004.
|
|
3
|
No authors listed. Efficacy of adjuvant
fluorouracil and folinic acid in B2 colon cancer. International
Multicentre Pooled Analysis of B2 Colon Cancer Trials (IMPACT B2)
Investigators. J Clin Oncol. 17:1356–1363. 1999.
|
|
4
|
No authors listed. Efficacy of adjuvant
fluorouracil and folinic acid in colon cancer. International
Multicentre Pooled Analysis of Colon Cancer Trials (IMPACT)
investigators. Lancet. 345:939–944. 1995. View Article : Google Scholar
|
|
5
|
Wu H, Neilson JR, Kumar P, et al: miRNA
profiling of naïve, effector and memory CD8 T cells. PLoS One.
2:e10202007.
|
|
6
|
Wiemer EA: The role of microRNAs in
cancer: no small matter. Eur J Cancer. 43:1529–1544. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Esquela-Kerscher A and Slack FJ: Oncomirs
- microRNAs with a role in cancer. Nat Rev Cancer. 6:259–269. 2006.
View Article : Google Scholar
|
|
9
|
Michael MZ, O’ Connor SM, van Holst
Pellekaan NG, Young GP and James RJ: Reduced accumulation of
specific microRNAs in colorectal neoplasia. Mol Cancer Res.
1:882–891. 2003.PubMed/NCBI
|
|
10
|
Cummins JM, He Y, Leary RJ, et al: The
colorectal microRNAome. Proc Natl Acad Sci USA. 103:3687–3692.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bandrés E, Cubedo E, Agirre X, et al:
Identification by real-time PCR of 13 mature microRNAs
differentially expressed in colorectal cancer and non-tumoral
tissues. Mol Cancer. 5:292006.PubMed/NCBI
|
|
12
|
Garzon R, Calin GA and Croce CM: MicroRNAs
in cancer. Annu Rev Med. 60:167–179. 2009. View Article : Google Scholar
|
|
13
|
Ma J, Dong C and Ji C: MicroRNA and drug
resistance. Cancer Gene Ther. 17:523–531. 2010. View Article : Google Scholar
|
|
14
|
Hurst DR, Edmonds MD and Welch DR:
Metastamir: the field of metastasis-regulatory microRNA is
spreading. Cancer Res. 69:7495–7498. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang H, Li Y and Lai M: The microRNA
network and tumor metastasis. Oncogene. 29:937–948. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mitchell PS, Parkin RK, Kroh EM, et al:
Circulating microRNAs as stable blood-based markers for cancer
detection. Proc Natl Acad Sci USA. 105:10513–10518. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weber JA, Baxter DH, Zhang S, et al: The
microRNA spectrum in 12 body fluids. Clin Chem. 56:1733–1741. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen X, Ba Y, Ma L, et al:
Characterization of microRNAs in serum: a novel class of biomarkers
for diagnosis of cancer and other diseases. Cell Res. 18:997–1006.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Valadi H, Ekström K, Bossios A, Sjöstrand
M, Lee JJ and Lötvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kosaka N, Iguchi H, Yoshioka Y, Takeshita
F, Matsuki Y and Ochiya T: Secretory mechanisms and intercellular
transfer of microRNAs in living cells. J Biol Chem.
285:17442–17452. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ritchie ME, Silver J, Oshlack A, et al: A
comparison of background correction methods for two-colour
microarrays. Bioinformatics. 23:2700–2707. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Smyth GK: Linear models and empirical
bayes methods for assessing differential expression in microarray
experiments. Stat Appl Genet Mol Biol. 3:Article 3. 2004.PubMed/NCBI
|
|
23
|
Gentleman RC, Carey VJ, Bates DM, et al:
Bioconductor: open software development for computational biology
and bioinformatics. Genome Biol. 5:R802004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Benjamini Y, Drai D, Elmer G, Kafkafi N
and Golani I: Controlling the false discovery rate in behavior
genetics research. Behav Brain Res. 125:279–284. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ng EK, Chong WW, Jin H, et al:
Differential expression of microRNAs in plasma of patients with
colorectal cancer: a potential marker for colorectal cancer
screening. Gut. 58:1375–1381. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang Z, Huang D, Ni S, Peng Z, Sheng W
and Du X: Plasma microRNAs are promising novel biomarkers for early
detection of colorectal cancer. Int J Cancer. 127:118–126. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tsang WP and Kwok TT: The
miR-18a* microRNA functions as a potential tumor
suppressor by targeting on K-Ras. Carcinogenesis. 30:953–959.
2009.PubMed/NCBI
|
|
29
|
Morimura R, Komatsu S, Ichikawa D, et al:
Novel diagnostic value of circulating miR-18a in plasma of patients
with pancreatic cancer. Br J Cancer. 105:1733–1740. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Weissmann-Brenner A, Kushnir M, Lithwick
Yanai G, et al: Tumor microRNA-29a expression and the risk of
recurrence in stage II colon cancer. Int J Oncol. 40:2097–2103.
2012.PubMed/NCBI
|
|
31
|
Wang LG and Gu J: Serum microRNA-29a is a
promising novel marker for early detection of colorectal liver
metastasis. Cancer Epidemiol. 36:e61–e67. 2012. View Article : Google Scholar : PubMed/NCBI
|