Introduction
Breast cancer is by far the most frequently
diagnosed cancer in woman; more than a million women are diagnosed
with breast cancer every year. More than half the cases are in
industrialized countries, with approximately 426,900 new cases of
breast cancer occurring each year in Europe (in 2006 there were
45,822 new cases diagnosed in the UK: 45,508 woman and 314 men) and
an estimated 182,460 in the USA (Office for National Statistics,
2006). These statistics underlie the intense activity in recent
years to identify markers for prognosis and treatment of this
disease. One area of scrutiny is the expression and distribution of
cancer stem cell markers.
Stem cells are required for the maintenance of high
cell turnover tissues where cells continually need to be replaced.
They constitute a small population of relatively undifferentiated
cells that express no differentiation markers of the tissue. They
are slowly self-renewing with each cell division producing an
average of one stem cell and one transit-amplifying cell. The
transit-amplifying cell has limited proliferative potential,
undergoing terminal differentiation to form the functioning cells
of the tissue.
The ‘tumor stem cell’ theory suggests that a small
percentage of cells in a tumor harbor intrinsic characteristics
making them resistant to treatment. This could explain how patients
with metastatic disease show clinical relapse several months after
starting treatment due to the survival of a small group of cells
with unique characteristics, including the ability to give rise to
a new population of cells with a resistant phenotype.
Putative cancer stem and progenitor cells have been
detected in a variety of epithelial cancers using markers that are
associated with normal tissue stem cells but highly contextual
(1). Previous studies have
identified CD44+/CD24− breast cancer cells as
candidate breast cancer stem cells (2), and CD133 (3,4). Park
et al(5) found that the
expression of CD44 seemed to decrease with tumor progression in
breast cancer and that CD44+/CD24− cells were
most common in basal-like tumors. In a wide-range study of cell
lines, expression of various stem cell marker (CD15, CD24, CD44,
CD133, CD166, CD326) patterns was correlated with tumor entities
such as basal breast cancer; other expression patterns occurred
across different tumor types and were largely related to expression
of a more mesenchymal phenotype in individual breast, lung, renal
and melanoma cell lines (6).
Due to the renewed interest in the ‘seed and soil’
hypothesis of cancer first put forth by Paget (7,8) in
1889, there has been intense interest in ‘cancer stem cell’
markers. But as yet, few conclusions have been drawn as to their
use as markers or prognostic indicators in solid human tumors. We
chose 8 cancer stem cell markers in order to determine the
distribution and expression pattern of these stem cell markers in
human breast cancer. We found a significant association between
loss of expression of CD24, CD34, CD44, CD49B, CD133 and PSCA and
metastatic disease in patients with breast cancer. Such
differential expression may play a part in breast cancer disease
progression, and suggests that the current cancer stem cell theory
may not hold true for all cancer types.
Materials and methods
Reagents and antibodies
Anti-CD44, -CD133, -CD49b and -PSCA antibodies were
purchased from Pharmingen International (San Diego, CA, USA).
Tissue collection and preparation
Breast tissue samples (124 tumor and 33 matched
adjacent tissues (Table I)
(9) were collected and immediately
frozen in liquid nitrogen before processing a portion of each
sample for quantitative-PCR analysis, a portion for
immunohistochemical analysis and a portion for routine histological
examination. RNA was isolated from tissue samples using standard
RNAzol procedures. For RT-PCR, cDNA was synthesized in a 20-μl
reaction mixture using 1 μg RNA, as described in the protocol
(ABgene Reverse Transcription System, Surrey, UK). The anonymized
breast tissue samples were obtained according to the guidelines of
the appropriate ethics committee (Bro Taf Health Authority 01/4303
and 01/4046). Informed patient consent was not applicable in this
instance (as stated in the Human Tissue Act 2004, UK).
 | Table IClinical characteristics of the
patient samples. |
Table I
Clinical characteristics of the
patient samples.
| Characteristics | No. of patient
samples |
|---|
| Tissue type |
| Background | 30 |
| Tumor | 124 |
| Grade |
| 1 | 24 |
| 2 | 42 |
| 3 | 58 |
| NPI |
| 1 | 68 |
| 2 | 38 |
| 3 | 16 |
| Unknown | 2 |
| TNM status |
| 1 | 70 |
| 2 | 40 |
| 3 | 7 |
| 4 | 4 |
| Unknown | 3 |
| Histology |
| Ductal | 94 |
| Lobular | 14 |
| Other | 16 |
| Patient
outcome |
| Alive and
well | 85 |
| Metastatic
disease | 7 |
| Death from breast
cancer | 15 |
| All poor
outcomes | 27 |
Quantitative-PCR
The q-PCR system used the Amplifluor™ UniPrimer™
system (Intergen Company, Oxford, UK) and Thermo-Start®
(ABgene, Epsom, Surrey, UK). Specific primer pairs for CD24, CD29,
CD34, CD44, CD49B, CD49F, CD133, PSCA and TSA-1 were designed by
the authors using a Beacon Designer software (Biosoft
International, Palo Alto, CA, USA) and manufactured by Invitrogen
(Invitrogen Life Technologies, Paisley, Scotland, UK), each
amplifying a region that spans at least 1 intron, generating an
~100 base pair product from both the control plasmid and cDNA. The
primers are as follows (5′-3′): CK-19QF1, caggtccgaggttactgac;
CK19QZR1, actgaac ctgaccgtacacagtttctgccagtgtgtcttc; GAPDHF2,
ctgagtacgtcgtg gagtc; GAPDHZR2,
actgaacctgaccgtacacacagagatgatgacccttt tg; CD24F1,
aactaatgccaaccaccaag; CD24ZR1, actgaacctgacc
gtacataagagtagagatgcagaagag; CD29F1, cctgccttggtgtctgtg; CD29ZR1,
actgaacctgaccgtacacctgtgtgcatgtgtctttc; CD34F1,
tcagcaaagtggaagttat; CD34ZR1, actgaacctgaccgtacagtagtttgg
gaatagctctg; CD44F1, accatggacaagttttggtggca; CD44ZR, act
gaacctgaccgtacactgtagcgaccatttttctc; CD49bF1, gcctgcagaaga
atatggta; CD49bZR1, actgaacctgaccgtacatccagactgatgtccacac; CD49F1,
gcgagccttcattgatgtg; CD49ZR1, actgaacctgaccgtacac
tacagtctttgagggaaacac; CD133F1, gcaaatgtggaaaaactgat; CD133ZR1,
actgaacctgaccgtacattaaatagcttcccagagaga; PSCAF1,
atgaaggctgtgctgctt; PSCAZR, actgaacctgaccgtacaag tcctcgttgctcacct;
TSA1F1, cttgaaccagaagagcaatc; TSA1ZR1,
actgaacctgaccgtacacactagcagacacagtcacg.
Using the iCycler iQ™ system (Bio-Rad), which
incorporates a gradient thermocycler and a 96-channel optical unit,
the plasmid standards and breast cancer cDNA were simultaneously
assayed in duplicated reactions using a standard HotStart q-PCR
Master Mix. q-PCR conditions were as follows: enzyme activation at
95°C for 12 min for 1 cycle; followed by 60 cycles of denaturing at
95°C for 15 sec; annealing at 55°C for 40 sec; and extension at
72°C for 25 sec. Using purified plasmids as internal standards, the
levels of cDNA (copies/50 ng RNA) in the breast cancer samples were
calculated. q-PCR for β-actin was also performed on the same
samples, to correct for any residual differences in the initial
level of RNA in the specimens (in addition to spectrophotometry).
Results were then normalized using cytokeratin-19 and GAPDH levels
in the same tissues. The products of q-PCR were verified on agarose
gels (data not shown). The data were then analyzed after a 10-year
follow-up.
Immunohistochemistry
Cryostat sections of frozen tissue were cut at 6 μm,
placed on SuperFrost Plus slides (LSL UK, Rochdale, UK), air dried
and fixed in a 50:50 solution of alcohol:acetone. The sections were
then air dried again and stored at −20°C until used. Immediately
before commencement of immunostaining, the sections were washed in
buffer for 5 min and treated with horse serum for 20 min as a
blocking agent to non-specific binding. Sections were stained using
PSCA, CD44, CD133 and CD49b antibodies (Pharmingen International).
Negative controls were used where necessary. Primary antibodies
were used at a 1:100 dilution for 60 min and then washed in buffer.
The secondary biotinylated antibody at a 1:100 dilution (Universal
Secondary, Vectastain Elite ABC; Vector Laboratories Inc.,
Burlingame, CA, USA) was added (in horse serum/buffer solution) for
30 min, followed by numerous washings. Avidin/biotin complex was
added for 30 min, again followed by washes. Diaminobenzidine was
used as a chromogen to visualize the antibody/antigen complex.
Sections were counterstained in Mayer’s haematoxylin for 1 min,
dehydrated, cleared and mounted in DPX. Following this, the
sections were analyzed for staining intensity as previously
described (9,10).
Statistical analysis
Statistical analysis was performed by Minitab
version 13.32 (Minitab Inc., State College, PA, USA) using a
two-sample Student’s t-test and the non-parametric Mann-Whitney
confidence interval and test or Kruskal-Wallis, where appropriate.
In addition, SPSS 12.01 was used to calculate the survival
curves.
Results
Immunohistochemical staining of human
breast tissues
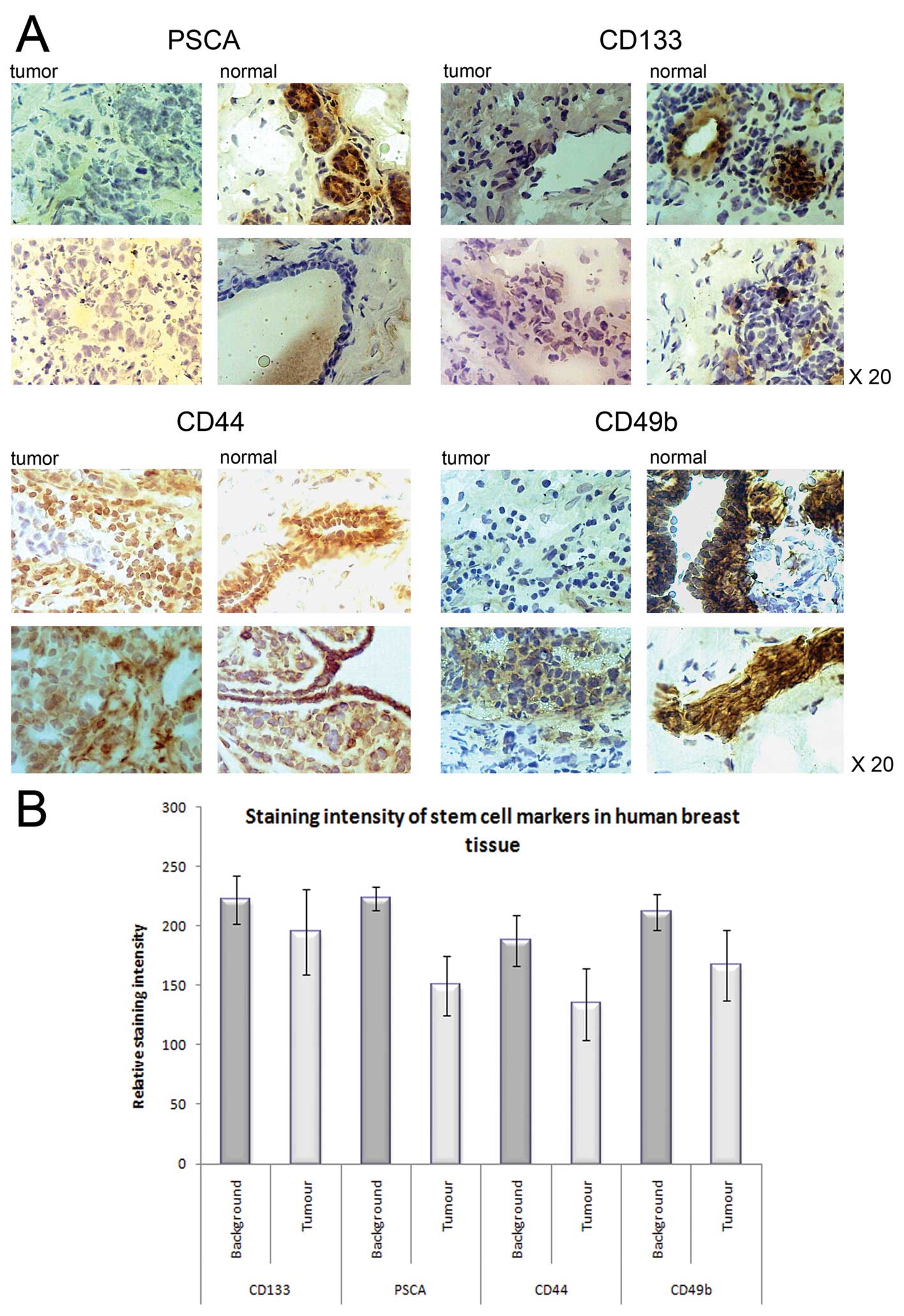
Representative sections of tumor and normal human
breast tissue sections are shown in Fig. 1A. IHC densitometry (Fig. 1B) revealed that there was a loss of
expression of PSCA in tumor sections (149.85±24.89) when compared
to that in the background tissue (222.85±9.88, n=20, p<0.0001).
CD44 protein was strongly expressed in the background endothelia
and epithelia but was weaker and diffuse in tumor cells
(134.5±29.89 and 187.9±21.28 respectively, p<0.0001).
CD133 expression was lost in the tumor-associated
endothelial cells with diffuse and weak staining in tumors when
compared to the background (194.9±35.76 and 222.2±20.46
respectively, p<0.006). Conversely, CD49b was strongly stained
in the tumors and associated vessels and ducts but was weakly
stained in the background epithelia, albeit that it was strongly
stained in background ducts and vessels (166.7±29.78 and
211.75±15.32 respectively, p<0.0001).
Quantitative PCR analysis of human breast
tissues
q-PCR analysis of gene transcript levels (normalized
using CK-19/GAPDH) further revealed the expression of the following
stem cell markers: CD24, CD29, CD34, CD44, CD49b, CD49F, CD133,
PSCA and TSA.
Stem cell marker expression and
prognostic indicators
We compared the expression of all the stem cell
markers with the diagnostic indicators Nottingham Prognostic
Indicator (NPI), grade and TNM status (tumor nodal involvement).
CD24, CD29, CD44 and CD133 were reduced with increasing NPI status
(Table II), but did not reach
statistical significance. CD49B, CD49F and PSCA showed no overall
trend. Interestingly, CD34 was increased with increasing NPI (but
did not reach significance). When looking at node-positive vs.
node-negative tumors, CD133 was reduced in node-positive
(node-negative 433±275 vs. node-positive 147±102) as was CD44
(1067±810 vs. 437±338), CD29 (240±153 vs. 67.8±22.3) and CD24
(1234±1075 vs. 435±239). However, node-positive tumors showed
increased expression of CD49F (702329±480290 vs. 1175685±469885),
CD49B (15.7±11.9 vs. 31.5±17.9), CD34 (2754±1422 vs. 7676±4316) and
PSCA (11.78±5.91 vs. 125±104).
 | Table IIExpression of stem cell markers in
human breast cancer, prognostic indicators and the relationship
between survival and ER status. |
Table II
Expression of stem cell markers in
human breast cancer, prognostic indicators and the relationship
between survival and ER status.
| NPI status of ductal
carcinoma | Grade of ductal
carcinoma | TNM status of ductal
carcinoma |
|---|
|
|
|
|
|---|
| Stem cell marker | NPI 1 | NPI 2 | NPI 3 | Grade 1 | Grade 2/3 | TNM 1 | TNM 2/3/4 |
|---|
| CD24 | 1234±1075 | 400±289 | 515±442 | 274±150 | 995±618 | 1153±1007 | 454±249 |
| CD29 | 240±153 | 68.2±22 | 66.3±55.2 | 125±57 | 157±86 | 78±33 | 287±172 |
| CD34 | 2754±1422 | 6983±5898 | 9155±5218 | 1491±1491 | 5586±2477a | 3677±1668 | 6393±4542 |
| CD44 | 1067±810 | 593±466 | 24.4±24.2 | 5±4.3 | 881±512 | 1154±803 | 306±232 |
| CD49B | 15.7±11.9 | 45.1±25.7 | 1.05±1.03 | 16.9±17 | 24±12 | 29±18 | 15.7±8.1 |
| CD49F | 702329±480290 | 1438189±659135 | 569906±298853 | 392570±232230 | 980353±367634 | 1307666±596557 | 452655±190439 |
| CD133 | 433±275 | 177±148 | 82.9±51.1 | 80±50 | 315±164 | 182±94 | 694±401 |
| PSCA | 11.78±5.91 | 153±148 | 61.3±55.1 | 0.98±0.59 | 74±57 | 121±95 | 5.3±2.6 |
| Patients remaining
alive and well | Patients with poor
outcome (metastatic disease and/or died from breast cancer) |
|
|
|
| ER− | ER+ |
ERβ− |
ERβ+ | ER− | ER+ |
ERβ− |
ERβ+ |
|
| CD24 | 362±202 | 2825±2782 | 1267±997 | 234±214 | 15.3±10.6 | 1776±1173 | 1190±803 | 7.22±7 |
| CD29 | 219±150 | 141±119 | 257±145 | 16.2±14 | 23.1±13.7 | 99±89.7 | 73±59.7 | 27.7±26.3 |
| CD34 | 4188±1745 | 4653±3803 | 2674±1245 | 9589±5553 | 121.7±90.3 | 220±220 | 208±149 | NA |
| CD44 | 1309±849 | 180±158 | 1208±750 | 25.3±22.5 | 1.49±1.47 | 0.915±0.796 | 1.379±0.869 | 0.034±0.032 |
| CD49B | 17.4±15.5 | 67.3±34.4 | 36.3±18.8 | 13.4±13.3 | >0.000001 | 1.68±1.7 | 1.12±1.1 | <0.000001 |
| CD49F | 866042±428289 | 316842±157460 | 886945±403449 | 119026±42608 | 116487±38778 |
3196146±2609191 |
2172667±1753441 | 98040±9515 |
| CD133 | 494±281 | 757±645 | 727±341 | 10.57±7.93a | 93.8±93.5 | 67.8±55.8 | 45.3±37.6 | 281±281 |
| PSCA | 28.5±16.7 | 0.307±0.194 | 25.3±157 | 5.05±4.61 | 2.33±2.24 | 6.89±6.71 | 5.72±4.51 | 0.198±0.19 |
When comparing grade 1 tumors with grade 2 and 3, it
was found that all 8 stem cell markers were increased with
increasing grade (Table III).
However, only CD34 reached statistical significance (p=0.034).
 | Table IIIExpression of stem cell markers in
ductal carcinoma and prognostic indicators. |
Table III
Expression of stem cell markers in
ductal carcinoma and prognostic indicators.
| NPI status of
ductal carcinoma | Grade of ductal
carcinoma | TNM status of
ductal carcinoma |
|---|
|
|
|
|
|---|
| Stem cell
marker | NPI 1 | NPI 2 | NPI 3 | Grade 1 | Grade 2/3 | TNM 1 | TNM 2/3/4 |
|---|
| CD24 | 1613±1524 | 421±309 | 668±573 | 282±199 | 1045±752 | 1450±1350 | 267±176a |
| CD29 | 512±217 | 54.3±16.4 | 79.4±71.9 | 103±35.8 | 74.3±24.9 | 67.4±39 | 280±201 |
| CD34 | 3787±1951 | 7449±6288 | 11444±6399 | 2113±2113 | 6711±2979 | 4635±2095 | 7804±5567 |
| CD44 | 1333±1084 | 685±538 | 13.1±30.8 | 0.795±0.465 | 1027±626 | 1507±1054 | 285±270 |
| CD49B | 5.6±3.76 | 48.4±27.5 | 1.35±1.35 | 26.6±26 | 21.2±12 | 24.8±20 | 19.98±9.75 |
| CD49F | 857356±679008 | 1528859±703798 |
718463±37624b | 555992±354824 | 1137533±450792 | 1649884±793017 |
526514±229112c |
| CD133 | 240±129 | 190±160 | 42.8±41.7 | 84.2±84.1 | 195.5±91 | 227±126 | 159±121 |
| PSCA | 16.42±8.28 | 5.04±2.3 | 79.6±71.4 | 0.697±0.634 | 22.5±11.9 | 35±3 | 6.39±3.09 |
Increasing TNM status was correlated with increased
expression of CD29, CD34, CD44 and CD133 (Table II). CD24, CD49B, CD49B and PSCA
were decreased with increasing TNM status. Again, this did not
reach statistical significance.
Stem cell marker expression and estrogen
receptor (ER) status
In ER-positive tumors, CD133 expression was
increased (400±223 vs. 452±370, ER-negative vs. ER-positive,
respectively), as was CD49F (710328±339907 vs. 1261327±730947),
CD49B (15.5±12.5 vs. 38.6±20.2), CD34 (3521±1394 vs. 8448±6030),
PSCA (24.1±13.3 vs. 150±148) and CD24 (311±161 vs. 2082±1595).
Conversely, expression of CD44 was decreased in ER-positive tumors
(1122±683 vs. 111±93.4) as was CD29 (189±119 vs. 143.5±72).
Positive significance was not reached.
In the ERβ-positive tumors, expression of CD133 was
decreased (518±242 vs. 36.7±28.2, ERβ-negative vs. ERβ-positive
tumors, respectively), as was CD49F (1016335±412127 vs.
442909±317945), CD49B (27±13.3 vs. 10.04±9.96), CD44 (942±550 vs.
18.1±16.1, p=0.097), CD29 (203±102 vs. 53.6±29.1) and CD24
(1108±706 vs. 187±160). In comparison, CD34 was increased in
ERβ-positive tumors (4568±2605 vs. 6974±4110, ERβ-negative vs.
ERβ-positive tumors, respectively), as was the level of PSCA
(19.7±11 vs. 226±222).
Stem cell marker expression and patient
clinical outcome
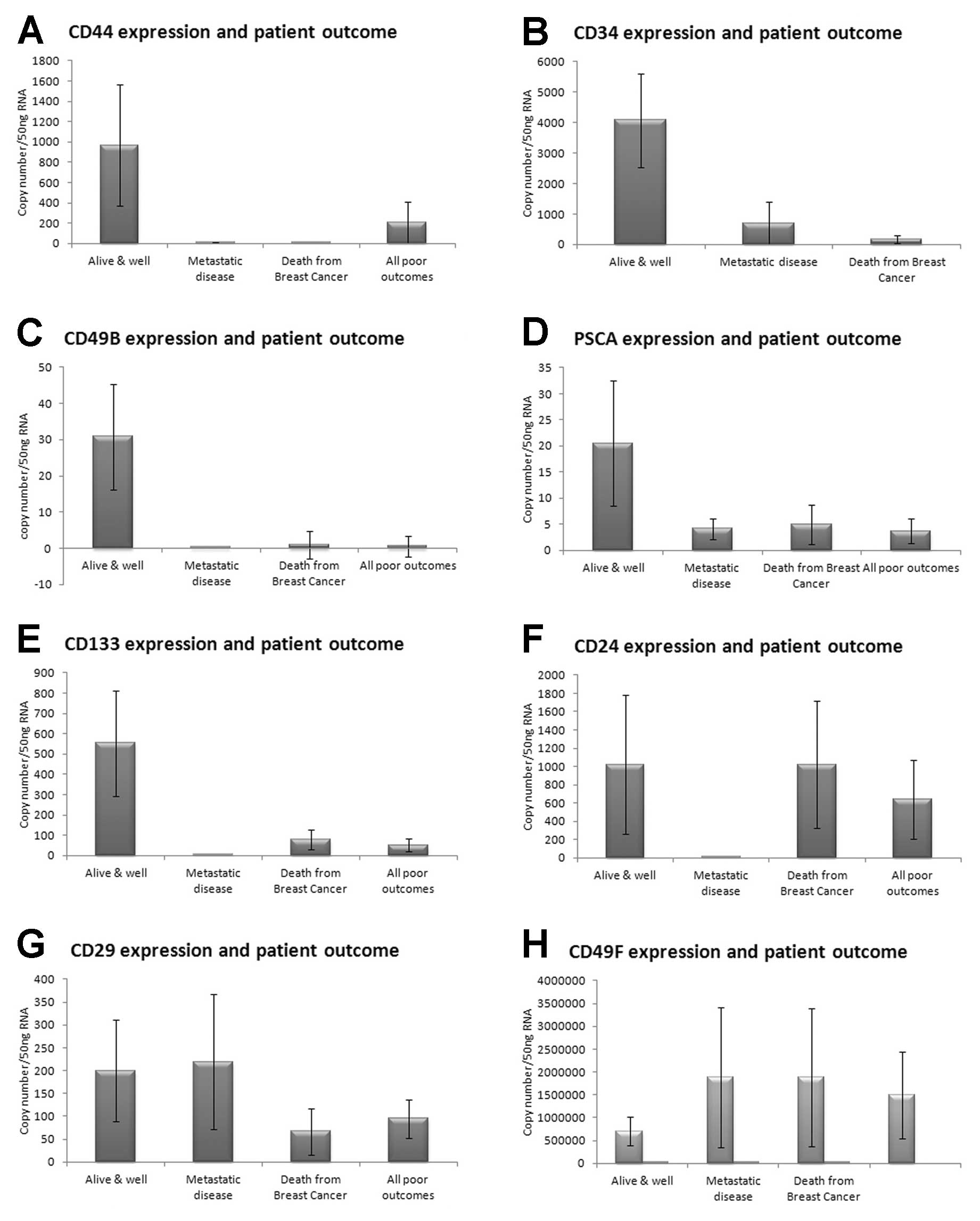
CD44, CD34, CD49B and PSCA were reduced in tumors of
patients with poor outcome (metastatic disease and death from
breast cancer) (Fig. 2A–D). CD133
was significantly reduced in tumors from patients with metastatic
disease (1.66±1.02) when compared with those remaining alive and
well after the 10-year follow-up (553±261, p=0.038) (Fig. 2E). CD24 was dramatically reduced in
metastatic disease, but not in the patients who had died from
breast cancer (Fig. 2F).
Conversely, CD29 was slightly elevated in metastatic disease, but
reduced in those patients who had died from breast cancer (Fig. 2G). The only marker to show increased
expression in all poor outcomes, compared to those patients who
remained alive and well was CD49F (Fig.
2H).
Stem cell marker expression and
metastatic disease to bone
Since tissues from patients with metastatic disease
had a correlation with the stem cell markers in relation to patient
outcome, we decided to dissect the metastatic data further to
consider the expression of these markers. CD133 was again markedly
reduced in metastasis to bone (alive and well, 553±261; bone
metastasis, 110±91; p=0.067). The expression of CD49B was also
significantly reduced in bone metastasis and in bone metastasis
leading to death (alive and well, 31.15±15; bone metastasis,
0.00054±0.0005; p=0.039; bone metastasis and death, 0.75±0.7;
p=0.044). CD44 was also reduce in bone metastasis, but did not
reach significance (alive and well, 968±599; bone metastasis,
2.58±1.6). There was also a reduction in expression of PSCA, CD29
and CD24 in bone metastasis (PSCA, 20.5±12 vs. 0.06±0.05; p=0.09;
CD29, 200±111 vs. 63±50; CD24, 1021±761 vs. 50±44, alive and well
vs. bone metastasis, respectively). The results for CD29 were in
contrast to the levels found in metastatic disease, which were
increased. Moreover, CD34 was increased in bone metastasis, which
was in contrast to metastatic disease overall (alive and well,
4080±1541 vs. bone metastasis, 25166±23318 vs. all metastatic
disease, 700±701) but this did not reach significance. Again, CF49F
was increased in tissues from patients with bone metastasis (alive
and well, 704108±309573 vs. bone metastasis, 3109444±3000851) but
did not reach significance.
Stem cell marker expression and ER status
in patient survival
We further dissected the results obtained for
survival in regard to ER status. Overall, in patients who had
remained alive and well, as shown in Table II, CD24, CD34, CD49B, CD49F and
CD133 were increased in ER-positive tumors, with CD29, CD44 and
PSCA being reduced. This increase was significant for CD133
(p=0.041). CD24, CD29, Cd44, CD49B, CD133 and PSCA were reduced in
ERβ-positive tumors, with CD34 and CD49F being decreased. In
patients who had poor outcome (i.e. had metastatic disease or died
from breast cancer), CD24, CD29, CD34, CD49B, CD49F and PSCA were
increased in ER-negative tumors, whereas CD44 and CD133 were
decreased. CD34, CD29, CD44, CD49F, PSCA and CD49B were reduced in
ERβ-positive tumors (data not available for CD34), with CD133 being
the only marker to be increased.
Stem cell marker expression and ductal
carcinoma
When tumor type was considered, the data showed that
CD44, CD29 and CD34 were elevated in ductal carcinoma in comparison
to other tumor types (CD44 ductal, 8719±536 vs. other types,
13.1±12.9; CD29 ductal, 173±95.6 vs. other types, 148±121; CD34
ductal, 6054±2573 vs. other types, 101±79; p=0.023). CD133, CD49B,
PSCA and CD49F were reduced in ductal carcinoma (CD133 ductal,
186±83 vs. other types, 1162±757; CD49B ductal, 21.7±11.1 vs. other
types, 24.6±24.5; PSCA ductal, 20.4±10.7 vs. other types, 212±211;
CD49F ductal, 1080198±407865 vs. other types, 221874±113113;
p=0.046). We then further investigated how these stem cell markers
were expressed in ductal carcinoma.
Stem cell marker expression in ductal
carcinoma and prognostic indicators
When considering ductal carcinomas alone, it was
noted that with increasing NPI status, there was a corresponding
increase in expression of CD34 and PSCA (Table III). Overall, the other stem cell
markers showed reduced expression with increasing NPI, which was
significant for CD49F (p=0.012). CD34 was also increased with
increasing grade as was CD44, CD49F, CD133 and PSCA. There was a
decrease in expression of CD24 (p=0.02), CD29 and CD49B with
increasing grade of ductal carcinomas. Only CD34 and CD29 showed an
elevated expression in high (2–4) TNM
status (Table III). CD49F was
significantly decreased (p=0.047).
Stem cell marker expression in ductal
carcinoma and patient outcome
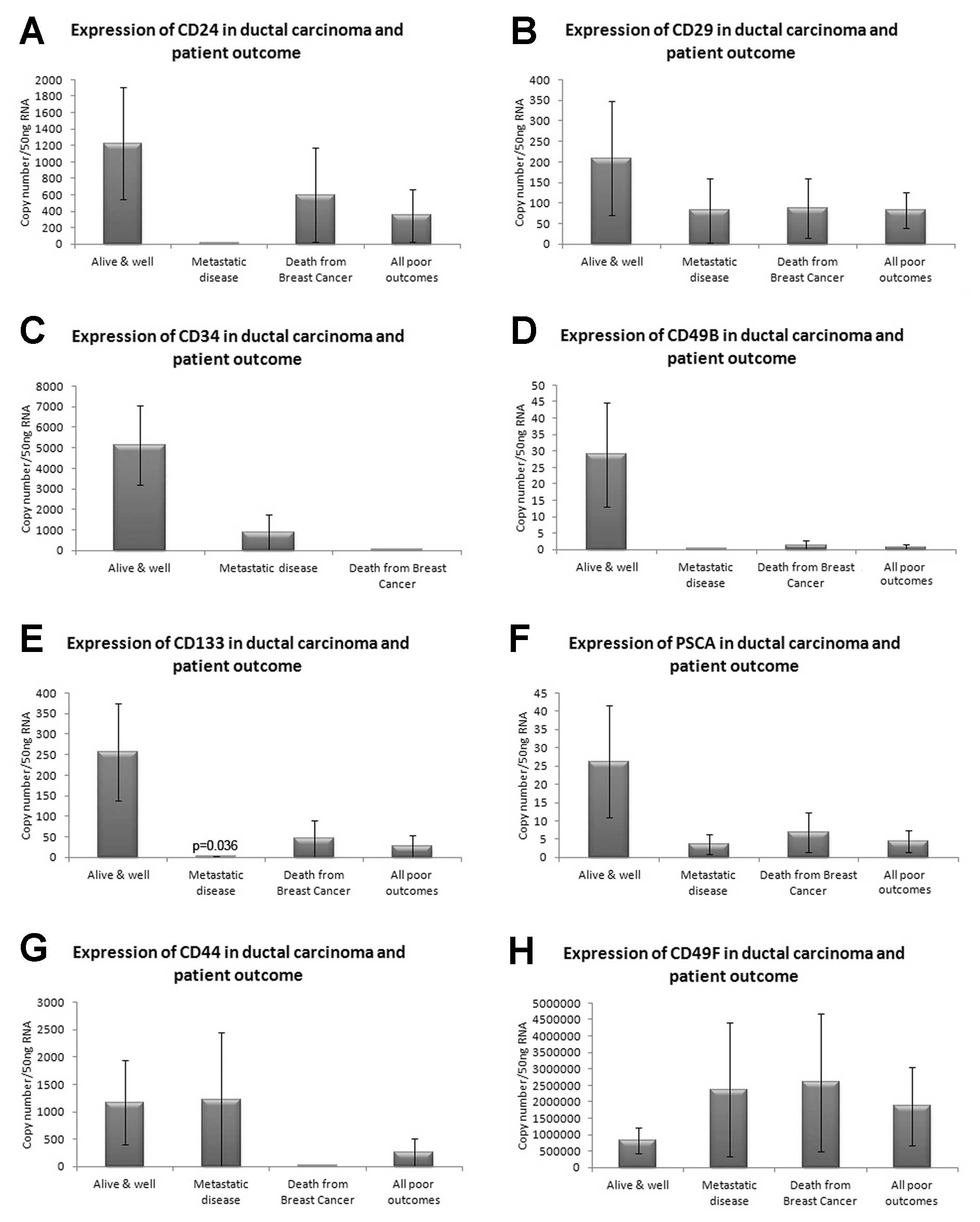
In the ductal carcinomas, CD24 expression was
reduced in metastatic disease and in those patients who had died
from breast cancer (Fig. 3A).
Similar results were observed for CD29, CD34, CD49B, CD133
(p=0.036) and PSCA (Fig. 3B–F). In
contrast, expression of CD44 showed some increase in metastatic
disease, although levels were reduced in patients who had died from
ductal carcinoma (Fig. 3G).
Moreover, CD49F was markedly increased in metastatic disease and
all poor outcomes (Fig. 3H). This
distribution was similar to that found in breast cancer overall
(discussed above), apart from CD44. When looking more closely at
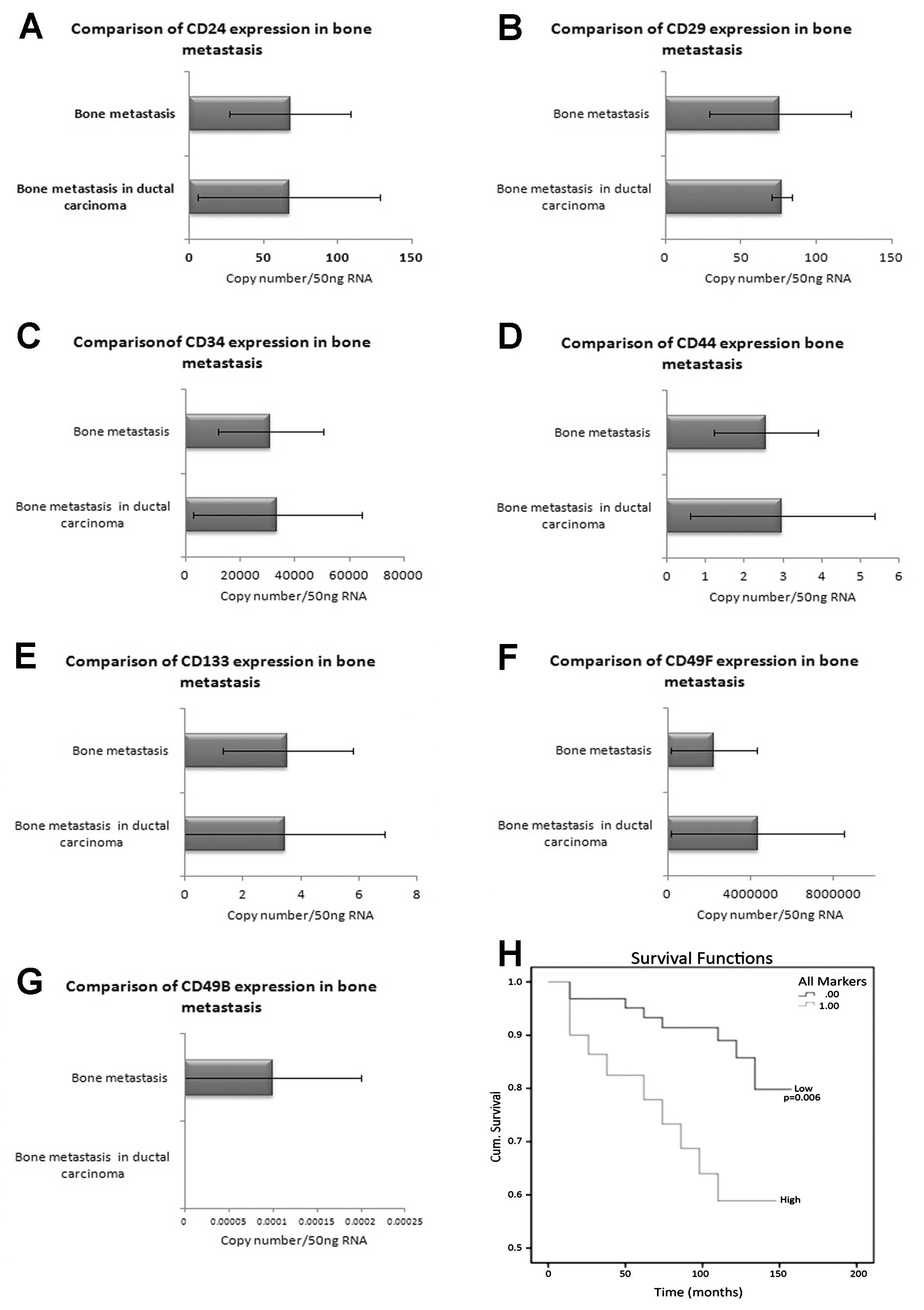
bone metastasis, it was found that the expression of CD24, CD29,
CD34, CD44 and CD133 in bone metastasis in ductal carcinoma was not
different to that in bone metastasis overall (Fig. 4A–E). However, levels of CD49F were
increased in bone metastasis in ductal carcinoma and reduced CD49B
expression was noted (Fig. 4F and
G). Levels for PSCA were unavailable.
Stem cell markers and survival
A long-term survival curve incorporating all of the
markers was calculated using Kaplan-Meier survival curves (Fig. 4H). Patients with high levels of
combined transcripts had a significantly shorter survival than
patients with low levels (p=0.006); high mean survival of 109.55
months (90.666–128.435 months, 95% CI) vs. low mean survival of
143.106 months (134.415–151.797 months, 95% CI) with cut-offs as
previously determined (9).
Discussion
The most widely accepted model for metastasis is the
‘seed and soil’ hypothesis postulated by Paget (7). He suggested that malignant tumor cells
are shed from the primary tumor and disseminated in the entire body
although they will metastasize when the shed (disseminated tumor
cells) and soil (secondary organ) are compatible. Subsequently,
knowledge in this area has expanded significantly. However, the
mechanisms underlying the entire process are still unclear, and
currently available therapies are mainly palliative. The tumor stem
cell hypothesis suggests that there exists within cancers, a subset
of cancer cells that are responsible for tumor recurrence following
chemotherapy and are causative of metastasis. Studies using human
cell lines and human tissues suggest that a pattern of cell surface
and functional markers define these cancer stem cells (6). Our study is one of the first to
determine the distribution and expression pattern of a number of
stem cell markers in human breast cancer.
As attractive as the cancer stem cell hypothesis
sounds, especially when applied to tumors that respond poorly to
current treatments, it has been argued that the proposal of a
stem-like cell that initiates and drives solid tissue cancer growth
and is responsible for therapeutic failure is far from proven
(11).
In the present study, we demonstrated that overall,
there was a reduction in expression of CD24, CD29, CD44 and CD133
with increasing NPI, an increase in all markers with increasing
grade and an increase in expression of CD29, CD34, CD44 and CD133
with increasing TNM status. Moreover, the majority of cancer stem
markers were reduced with metastatic disease and poor prognosis
overall. Only CD29 was increased with metastatic disease, and
patients who had died from breast cancer had increased levels of
CD24.
Although a broad range of pathological subtypes of
invasive breast cancer have been identified, the most commonly used
classification of invasive breast cancers divides them into ductal
and lobular types. Invasive ductal carcinoma represents the largest
group, accounting for up to 80% of invasive cancers (12). When comparing ductal carcinoma and
other cancer types, we found that CD34, CD29 and CD44 were
elevated. In the patients with ductal carcinoma, we found that
while for the most part the distribution of cancer stem cell
markers mirrored that of breast cancer overall, CD44 was increased
in metastatic disease and CD49F was increased in all poor
outcomes.
Breast carcinomas have been reported to contain a
subpopulation of CD44+/CD24− tumor cells with
stem cell-like properties. In fact, the majority of studies to date
have tended to concentrate on
CD44+/CD24−(13–17)
and there have been few that have examined a larger range of
markers in the same cohort. Giatromanolaki et al(18) investigated the significance of these
2 molecules in connection with tumor aggression and prognosis. The
phenotypic profile of 139 breast carcinomas was investigated in
paraffin sections using markers previously associated with stem
cell-like properties (CD44, CD24), the ‘triple-state’ (ER, PR,
c-erb-B2) and angiogenesis (CD31). The authors concluded that
assessment of the CD44/CD24 status may reveal distinct subgroups of
breast cancer patients with different clinical behavior, and that
CD44 targeting was an attractive therapeutic alternative for breast
cancer patients and that the strong association between the
CD44−/CD24− phenotype and prognosis required
further investigation.
Such studies are plainly in contrast to the study
presented here. However, Ahmed et al(19) suggested that the
CD44−/CD24+ phenotype is a poor prognostic
marker in early invasive breast cancer. Breast cancer cells with
high CD44 and low or absent CD24 are reported to have stem cell
features. The authors stated that CD24 and CD44 expression can
individually yield prognostic data in breast cancer, but
importantly, when both markers are considered,
CD44+/CD24− was associated with the best
prognosis, while CD44−/CD24+ was associated
with the worst prognosis. This shows that the relationship between
basic cell biology and clinical behavior is not always
straightforward and warrants further investigation of the true
clinical impact of breast cancer stem cells (19). It has been demonstrated that
individual CD44 isoforms can be associated with different breast
cancer subtypes and clinical markers such as HER2, ER and PgR,
which suggests involvement of CD44 splice variants in specific
oncogenic signaling pathways (20).
Efforts therefore, to link CD44 to cancer stem cells and tumor
progression should consider the expression of various CD44
isoforms. Moreover, in a recent study by Guler et
al(21), the occurrence of
CD24−/44+ and CD24+/44−
cells in primary tumors did not differ in primary vs. matched lymph
node or distant and locoregional metastatic lesions. Thus, the
frequency of CD24−/44+ cells does not differ
in metastases relative to the primary breast cancer but differs in
regards to tumor stage and subtype.
Approximately 70% of patients with breast cancer
have bone metastases. They are associated with poor prognosis and
the available treatment options are very limited (22). Bone metastasis usually presents with
severe pain, and these symptoms are usually noted in the femur and
pelvic region. Of the 2 types of breast cancer bone metastasis,
osteolytic lesions are the most common form and cause destruction
of the bone whereas osteoblastic lesions, which are less common,
cause new bone formation. Most patients have components of both
bone resorption and bone formation. When we examined our data more
intensely in order to determine any differences in bone metastasis,
we discovered that there was, overall, very little difference in
the expression of the cancer stem cell markers in all patients with
bone metastasis, compared to patients with ductal carcinoma. CD49F,
however, was increased in bone metastasis in ductal carcinoma,
whereas CD48B was completely lost.
The metadata analysis conducted by Zhou et
al(16) lent support to the
cancer stem cell hypothesis by showing a significant correlation
between cancer stem cells and common clinical parameters, such as
ER, PR, HER2 and tumor grade. Putative stem cell markers,
particularly ALDH1, were significantly associated with worse
survival based on currently obtained data. It was suggested by the
authors that these markers should be further evaluated for their
potential use in the identification of breast cancer stem cells in
clinical practice.
In conclusion, we demonstrated that there is a
significant association between loss of expression of certain stem
cell markers and metastatic disease in patients with breast cancer.
Such differential expression may play a part in breast cancer
disease progression, and suggests that the current stem cell theory
may not entirely hold true for all cancer types and that
investigations into wider types of solid tumors and covering a
broader range of ‘stem cell markers’ are required.
Acknowledgements
We would like to thank Cancer Research Wales (CRW)
for sponsoring the present study and Dr Anthony Douglas-Jones for
his invaluable help and assistance with patient samples.
References
|
1
|
Donnenberg VS, Donnenberg AD, Zimmerlin L,
Landreneau RJ, Bhargava R, Wetzel RA, Basse P and Brufsky AM:
Localization of CD44 and CD90-positive cells to the invasive front
of breast tumors. Cytometry B Clin Cytom. 78:287–301. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ginestier C, Korkaya H, Dontu G, Birnbaum
D, Wicha MS and Charafe-Jauffret EI: The cancer stem cell: the
breast cancer driver. Med Sci. 23:1133–1139. 2007.(In French).
|
|
3
|
Wright MH, Calcagno AM, Salcido CD,
Carlson MD, Ambudkar SV and Varticovski L: Brca1 breast
tumors contain distinct CD44+/CD24− and
CD133+ cells with cancer stem cell characteristics.
Breast Cancer Res. 10:R102008. View
Article : Google Scholar
|
|
4
|
Xiao Y, Ye Y, Yearsley K, Jones S and
Barsky SH: The lymphovascular embolus of inflammatory breast cancer
expresses a stem cell-like phenotype. Am J Pathol. 173:561–574.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Park SY, Lee HE, Li H, Shipitsin M, Gelman
R and Polyak K: Heterogeneity for stem cell-related markers
according to tumor subtype and histologic stage in breast cancer.
Clin Cancer Res. 16:876–887. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stuelten CH, Mertins SD, Busch JI, Gowens
M, Scudiero DA, Burkett MW, Hite KM, Alley M, Hollingshead M,
Shoemaker RH and Niederhuber JE: Complex display of putative tumor
stem cell markers in the NCI60 tumor cell line panel. Stem Cells.
28:649–660. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Paget S: The distribution of secondary
growths of the breast. Lancet. 1:571–573. 1889. View Article : Google Scholar
|
|
8
|
Lin EH, Jiang Y, Deng Y, Lapsiwala R, Lin
T and Blau CA: Cancer stem cells, endothelial progenitors, and
mesenchymal stem cells: ‘seed and soil’ theory revisited.
Gastrointest Cancer Res. 2:169–174. 2008.
|
|
9
|
Martin TA, Watkins G, Mansel RE and Jiang
WG: Loss of tight junction plaque molecules in breast cancer
tissues is associated with a poor prognosis in patients with breast
cancer. Eur J Cancer. 40:2717–2725. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Martin TA, Pereira G, Watkins G, Mansel RE
and Jiang WG: N-WASP is a putative tumour suppressor in breast
cancer cells, in vitro and in vivo, and is associated with clinical
outcome in patients with breast cancer. Clin Exp Metastasis.
25:97–108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rahman M, Deleyrolle L, Vedam-Mai V, Azari
H, Abd-El-Barr M and Reynolds BA: The cancer stem cell hypothesis:
failures and pitfalls. Neurosurgery. 68:531–545. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Weigelt B, Horlings HM, Kreike B, Hayes
MM, Hauptmann M, Wessels LF, de Jong D, Van de Vijver MJ, Van’t
Veer LJ and Peterse JL: Refinement of breast cancer classification
by molecular characterization of histological special types. J
Pathol. 216:141–150. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bhat-Nakshatri P, Appaiah H, Ballas C,
Pick-Franke P, Goulet R Jr, Badve S, Srour EF and Nakshatri H:
SLUG/SNAI2 and tumor necrosis factor generate breast cells with
CD44+/CD24− phenotype. BMC Cancer.
10:4112010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cipak A, Mrakovcic L, Ciz M, Lojek A,
Mihaylova B, Goshev I, Jaganjac M, Cindric M, Sitic S, Margaritoni
M, Waeg G, Balic M and Zarkovic N: Growth suppression of human
breast carcinoma stem cells by lipid peroxidation product
4-hydroxy-2-nonenal and hydroxyl radical-modified collagen. Acta
Biochim Pol. 57:165–171. 2010.
|
|
15
|
Oliveras-Ferraros C, Vazquez-Martin A,
Martin-Castillo B, Martin-Castillo B, Cufí S, Del Barco S,
Lopez-Bonet E, Brunet J and Menendez JA: Dynamic emergence of the
mesenchymal CD44posCD24neg/low phenotype in
HER2-gene amplified breast cancer cells with de novo
resistance to trastuzumab (Herceptin). Biochem Biophys Res Commun.
397:27–33. 2010.PubMed/NCBI
|
|
16
|
Zhou L, Jiang Y, Yan T, Di G, Shen Z, Shao
Z and Lu J: The prognostic role of cancer stem cells in breast
cancer: a meta-analysis of published literatures. Breast Cancer Res
Treat. 122:795–801. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fillmore C and Kuperwasser C: Human breast
cancer stem cell markers CD44 and CD24: enriching for cells with
functional properties in mice or in man? Breast Cancer Res.
9:3032007. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Giatromanolaki A, Sivridis E, Fiska A and
Koukourakis MI: The CD44+/CD24− phenotype
relates to ‘triple-negative’ state and unfavorable prognosis in
breast cancer patients. Med Oncol. 28:745–752. 2011.
|
|
19
|
Ahmed MA, Aleskandarany MA, Rakha EA,
Moustafa RZ, Benhasouna A, Nolan C, Green AR, Ilyas M and Ellis IO:
A CD44−/CD24+ phenotype is a poor prognostic
marker in early invasive breast cancer. Breast Cancer Res Treat.
133:979–995. 2012.
|
|
20
|
Olsson E, Honeth G, Bendahl PO, Saal LH,
Gruvberger-Saal S, Ringnér M, Vallon-Christersson J, Jönsson G,
Holm K, Lövgren K, Fernö M, Grabau D, Borg A and Hegardt C: CD44
isoforms are heterogeneously expressed in breast cancer and
correlate with tumor subtypes and cancer stem cell markers. BMC
Cancer. 11:4182011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guler G, Balci S, Costinean S, Ussakli CH,
Irkkan C, Suren D, Sari E, Altundag K, Ozisik Y, Jones S, Bacher J,
Shapiro CL and Huebner K: Stem cell-related markers in primary
breast cancers and associated metastatic lesions. Mod Pathol.
25:949–955. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Akhtari M, Mansuri J, Newman KA, Guise TM
and Seth P: Biology of breast cancer bone metastasis. Cancer Biol
Ther. 7:3–9. 2008. View Article : Google Scholar
|