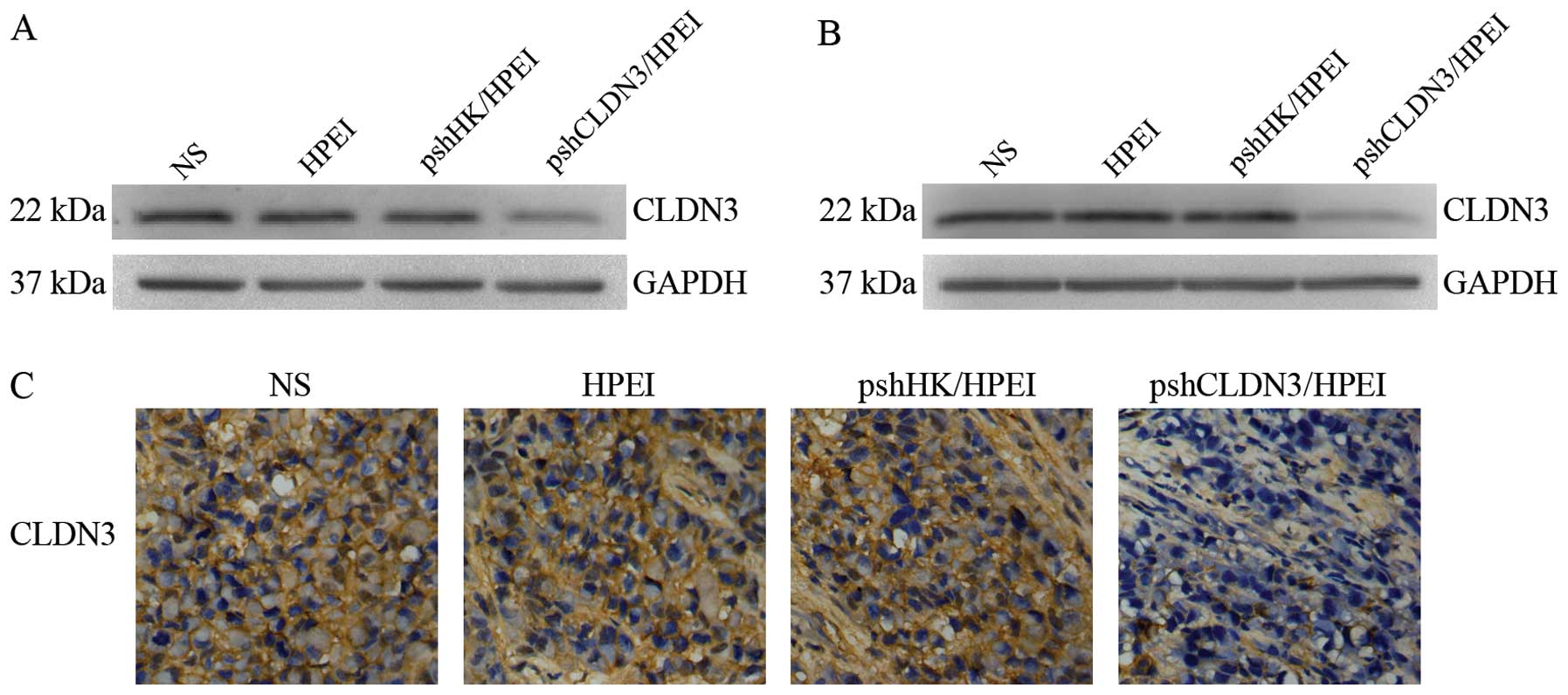
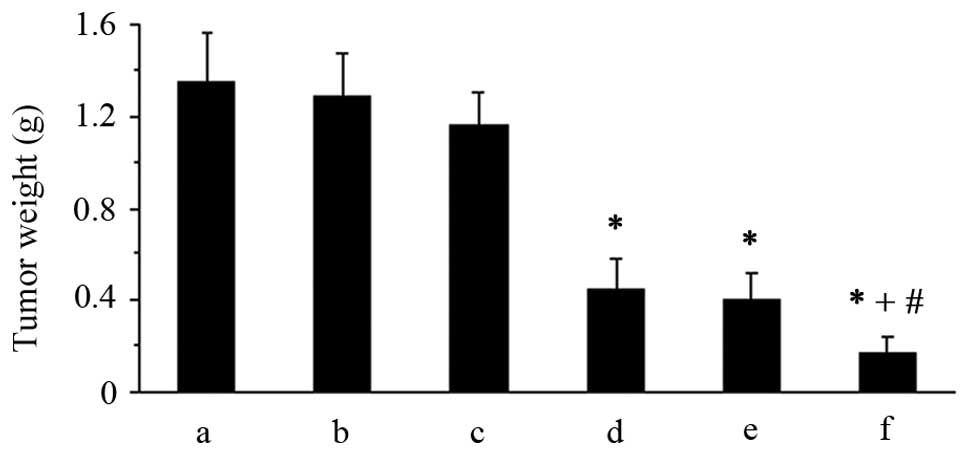
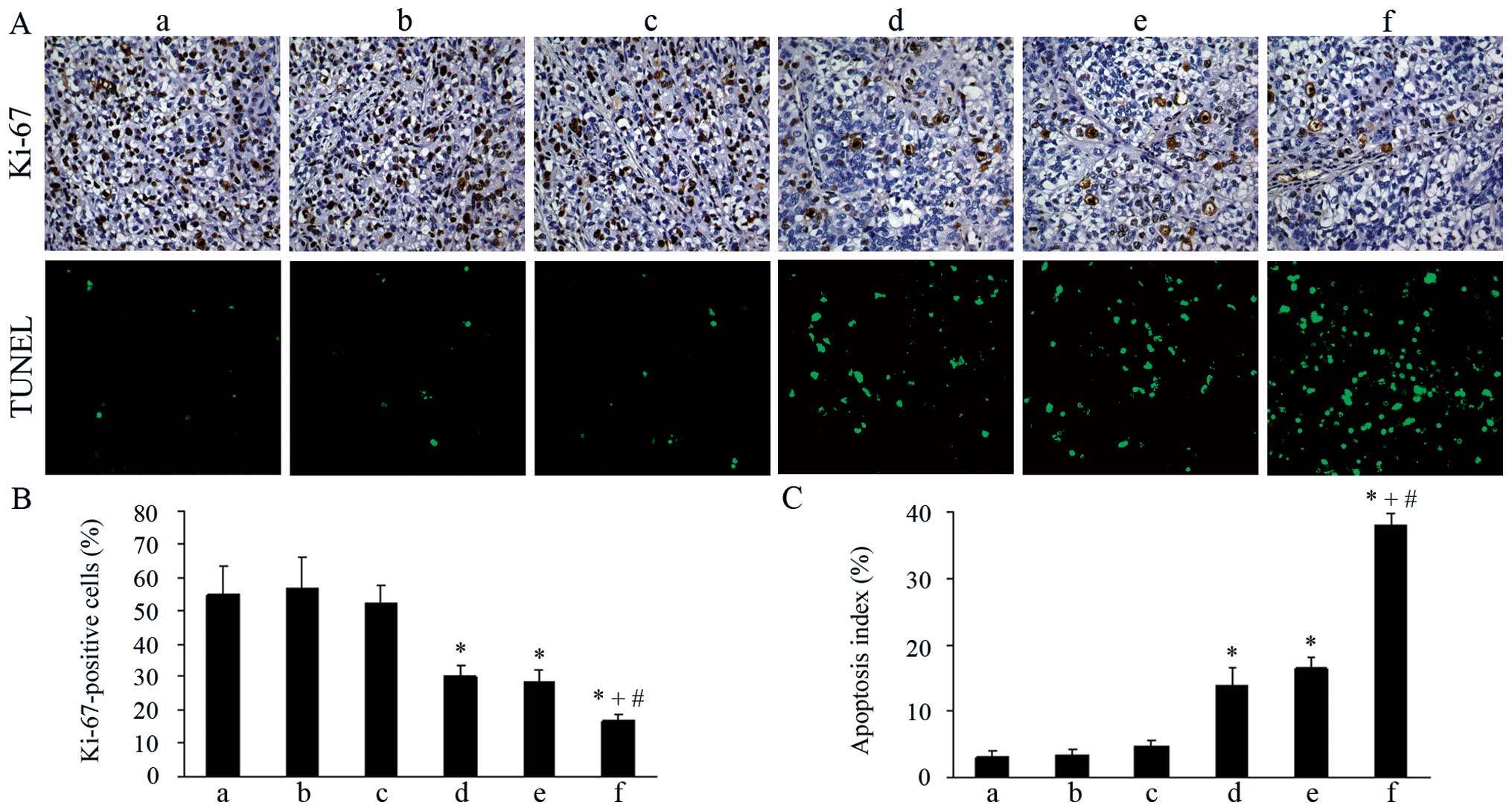
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2012. CA Cancer J Clin. 62:10–29. 2012. View Article : Google Scholar
|
|
2
|
McKeage MJ: Comparative adverse effect
profiles of platinum drugs. Drug Saf. 13:228–244. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Piccart MJ, Lamb H and Vermorken JB:
Current and future potential roles of the platinum drugs in the
treatment of ovarian cancer. Ann Oncol. 12:1195–1203. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Morita K, Furuse M, Fujimoto K and Tsukita
S: Claudin multigene family encoding four-transmembrane domain
protein components of tight junction strands. Proc Natl Acad Sci
USA. 96:511–516. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tsukita S and Furuse M: Pores in the wall:
claudins constitute tight junction strands containing aqueous
pores. J Cell Biol. 149:13–16. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tsukita S, Furuse M and Itoh M:
Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol.
2:285–293. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hough CD, Sherman-Baust CA, Pizer ES, et
al: Large-scale serial analysis of gene expression reveals genes
differentially expressed in ovarian cancer. Cancer Res.
60:6281–6287. 2000.PubMed/NCBI
|
|
8
|
Rangel LB, Agarwal R, D’Souza T, et al:
Tight junction proteins claudin-3 and claudin-4 are frequently
overexpressed in ovarian cancer but not in ovarian cystadenomas.
Clin Cancer Res. 9:2567–2575. 2003.PubMed/NCBI
|
|
9
|
Hibbs K, Skubitz KM, Pambuccian SE, et al:
Differential gene expression in ovarian carcinoma: identification
of potential biomarkers. Am J Pathol. 165:397–414. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu KH, Patterson AP, Wang L, et al:
Selection of potential markers for epithelial ovarian cancer with
gene expression arrays and recursive descent partition analysis.
Clin Cancer Res. 10:3291–3300. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Santin AD, Zhan F, Bellone S, et al: Gene
expression profiles in primary ovarian serous papillary tumors and
normal ovarian epithelium: identification of candidate molecular
markers for ovarian cancer diagnosis and therapy. Int J Cancer.
112:14–25. 2004. View Article : Google Scholar
|
|
12
|
Agarwal R, D’Souza T and Morin PJ:
Claudin-3 and claudin-4 expression in ovarian epithelial cells
enhances invasion and is associated with increased matrix
metalloproteinase-2 activity. Cancer Res. 65:7378–7385. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sun C, Yi T, Song X, et al: Efficient
inhibition of ovarian cancer by short hairpin RNA targeting
claudin-3. Oncol Rep. 26:193–200. 2011.PubMed/NCBI
|
|
14
|
Santin AD, Cané S, Bellone S, et al:
Treatment of chemotherapy-resistant human ovarian cancer xenografts
in C.B-17/SCID mice by intraperitoneal administration of
Clostridium perfringens enterotoxin. Cancer Res.
65:4334–4342. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yoshida H, Sumi T, Zhi X, Yasui T, Honda K
and Ishiko O: Claudin-4: a potential therapeutic target in
chemotherapy-resistant ovarian cancer. Anticancer Res.
31:1271–1277. 2011.PubMed/NCBI
|
|
16
|
Boussif O, Lezoualc’h F, Zanta MA, et al:
A versatile vector for gene and oligonucleotide transfer into cells
in culture and in vivo: polyethylenimine. Proc Natl Acad Sci USA.
92:7297–7301. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lungwitz U, Breunig M, Blunk T and
Göpferich A: Polyethylenimine-based non-viral gene delivery
systems. Eur J Pharm Biopharm. 60:247–266. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Neu M, Fischer D and Kissel T: Recent
advances in rational gene transfer vector design based on
poly(ethylene imine) and its derivatives. J Gene Med. 7:992–1009.
2005. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Godbey WT, Wu KK and Mikos AG: Size
matters: molecular weight affects the efficiency of
poly(ethylenimine) as a gene delivery vehicle. J Biomed Mater Res.
45:268–275. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kunath K, von Harpe A, Fischer D, et al:
Low-molecular-weight polyethylenimine as a non-viral vector for DNA
delivery: comparison of physicochemical properties, transfection
efficiency and in vivo distribution with high-molecular-weight
polyethylenimine. J Control Release. 89:113–125. 2003. View Article : Google Scholar
|
|
21
|
Gou M, Men K, Zhang J, et al: Efficient
inhibition of C-26 colon carcinoma by VSVMP gene delivered by
biodegradable cationic nanogel derived from polyethyleneimine. ACS
Nano. 4:5573–5584. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xie C, Gou ML, Yi T, et al: Efficient
inhibition of ovarian cancer by truncation mutant of FILIP1L gene
delivered by novel biodegradable cationic heparin-polyethyleneimine
nanogels. Hum Gene Ther. 22:1413–1422. 2011. View Article : Google Scholar
|
|
23
|
Liu P, Gou M, Yi T, et al: The enhanced
antitumor effects of biodegradable cationic
heparin-polyethyleneimine nanogels delivering HSulf-1 gene combined
with cisplatin on ovarian cancer. Int J Oncol. 41:1504–1512.
2012.
|
|
24
|
Lin XJ, Chen XC, Wang L, et al: Dynamic
progression of an intraperitoneal xenograft model of human ovarian
cancer and its potential for preclinical trials. J Exp Clin Cancer
Res. 26:467–474. 2007.PubMed/NCBI
|
|
25
|
Mabuchi S, Altomare DA, Cheung M, et al:
RAD001 inhibits human ovarian cancer cell proliferation, enhances
cisplatin-induced apoptosis, and prolongs survival in an ovarian
cancer model. Clin Cancer Res. 13:4261–4270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mabuchi S, Terai Y, Morishige K, et al:
Maintenance treatment with bevacizumab prolongs survival in an in
vivo ovarian cancer model. Clin Cancer Res. 14:7781–7789. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang YH, Bao Y, Peng W, et al: Claudin-3
gene silencing with siRNA suppresses ovarian tumor growth and
metastasis. Proc Natl Acad Sci USA. 106:3426–3430. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kiuchi-Saishin Y, Gotoh S, Furuse M,
Takasuga A, Tano Y and Tsukita S: Differential expression patterns
of claudins, tight junction membrane proteins, in mouse nephron
segments. J Am Soc Nephrol. 13:875–886. 2002.
|
|
29
|
Hewitt KJ, Agarwal R and Morin PJ: The
claudin gene family: expression in normal and neoplastic tissues.
BMC Cancer. 6:1862006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vaisman A, Varchenko M, Said I and Chaney
SG: Cell cycle changes associated with formation of Pt-DNA adducts
in human ovarian carcinoma cells with different cisplatin
sensitivity. Cytometry. 27:54–64. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cohen SM and Lippard SJ: Cisplatin: from
DNA damage to cancer chemotherapy. Prog Nucleic Acid Res Mol Biol.
67:93–130. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang D and Lippard SJ: Cellular processing
of platinum anticancer drugs. Nat Rev Drug Discov. 4:307–320. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kominsky SL: Claudins: emerging targets
for cancer therapy. Expert Rev Mol Med. 8:1–11. 2006. View Article : Google Scholar
|
|
34
|
Relph K, Harrington K and Pandha H: Recent
developments and current status of gene therapy using viral vectors
in the United Kingdom. BMJ. 329:839–842. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Glover DJ, Lipps HJ and Jans DA: Towards
safe, non-viral therapeutic gene expression in humans. Nat Rev
Genet. 6:299–310. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ferber D: Gene therapy. Safer and
virus-free? Science. 294:1638–1642. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lv H, Zhang S, Wang B, Cui S and Yan J:
Toxicity of cationic lipids and cationic polymers in gene delivery.
J Control Release. 114:100–109. 2006. View Article : Google Scholar : PubMed/NCBI
|