Introduction
Primary lung cancer is the most commonly diagnosed
malignant tumor in the world. It is reported that the number of new
lung cancer cases in 2008 was 1.6 million, accounting for 13% of
the total cancer cases. Due to its insidious symptoms, late
clinical presentation and rapid progression, there are 1.4 million
deaths annually making it the leading cause of cancer-related
mortality worldwide (1). Non-small
cell lung cancer (NSCLC) accounts for ~75–80% of cases (2). It is widely accepted that genetic
heterogeneity and environmental factors result in the onset of lung
cancer simultaneously. Smoking is the most well-established cause
of lung cancer, yet non-smokers take up a very large proportion of
lung cancer patients, suggesting the genetic variants also play a
role (3–5). It is of utmost importance to identify
novel molecules for the detection and diagnosis of lung cancer.
Lysosomal-associated protein transmembrane-4β
(LAPTM4B), a novel oncogene candidate, was initially
identified in hepatocellular carcinoma. It is cloned using
fluorescence differential display, rapid amplification of cDNA ends
and reverse transcription-polymerase chain reaction (RT-PCR).
According to BLAST program analysis, it is located in chromosomes
8q22 and is composed of seven exons separated by six introns
(6,7). It was previously reported that
LAPTM4B protein was markedly overexpressed in various
malignant tumors, including pancreatic (8), gallbladder (9), ovarian (10) and cervical cancer (11). Two alleles of the LAPTM4B
gene have been recognized, designated as LAPTM4B*1 and
LAPTM4B*2 (GenBank nos. AY219176 and AY219177,
respectively). The difference between LAPTM4B*1 and
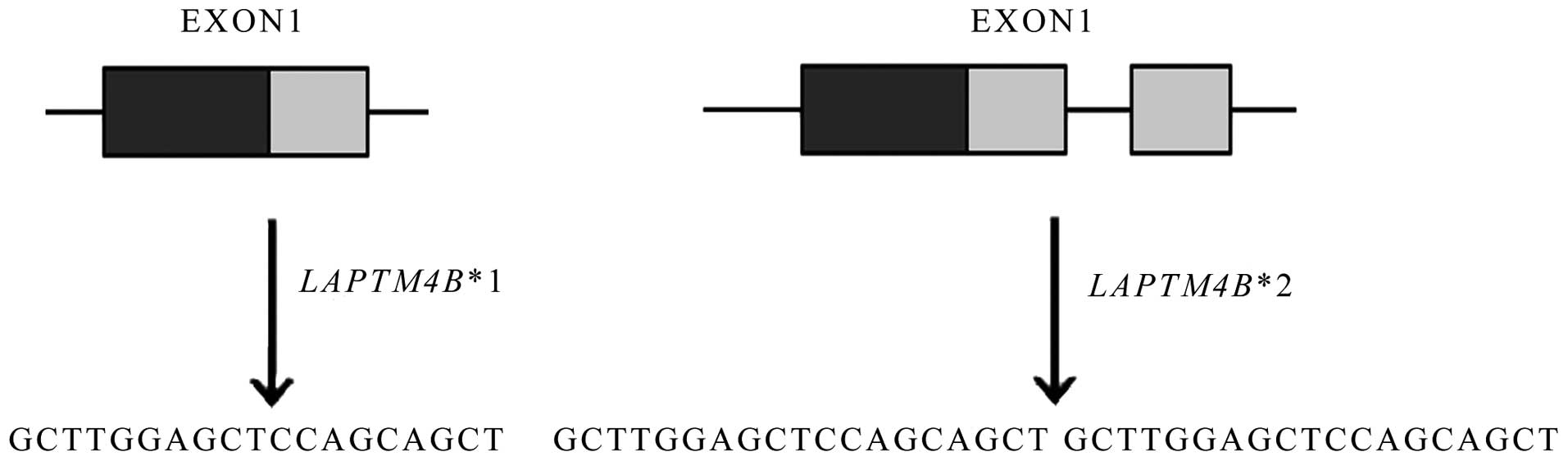
LAPTM4B *2 is the sequence at the 5′ untranslated region
(UTR) in the first exon. As shown in Fig. 1, LAPTM4B*1 contains only one
copy of a 19-bp sequence whereas LAPTM4B*2 contains two
tight tandem segments.
Previous studies showed that there was an
association between LAPTM4B polymorphism and increased risk
of hepatocellular carcinoma (12),
gastric cancer (13), colorectal
cancer (14), gallbladder (15) and ovarian carcinoma (16), and breast cancer (17). Two laboratories studied the
relationship between LAPTM4B polymorphism and susceptibility
of lung cancer, however, they drew completely different conclusions
(18,19); one reported that LAPTM4B*2
was associated with NSCLC susceptibility, but the other found no
relationship. Less than 200 patients were included in each group,
therefore, their conclusions are not very conclusive. Meanwhile,
the two groups included all types of histological lung cancer in
the samples. It is known that the development of small cell lung
cancer and NSCLC has a considerable difference, and LAPTM4B
polymorphism might influence only a certain histological type, thus
it is less rigorous not to divide them. Hence, it is necessary to
enrol more patients to rigorously and deeply explore whether there
is a relationship between LAPTM4B polymorphism and risk of
NSCLC. In addition, LAPTM4B*2 is also a marker of poor
prognosis in gallbladder (20) and
hepatocellular carcinoma (21),
breast (17) and endometrial cancer
(22), while its function in the
prognosis of lung cancer patients has not been clarified. The
present study was designed to study the effects of LAPTM4B
gene variants on the susceptibility of NSCLC via a large sample
size. Furthermore, the relationship between LAPTM4B genotype
and prognosis of NSCLC was also analyzed.
Materials and methods
Patients and controls
A total of 392 blood samples of patients were
collected from Qilu Hospital of Shandong University between July
2007 and October 2010. All patients underwent surgical resection
and were diagnosed as NSCLC by at least two pathologists. For all
patients, histological type of lung cancer was determined by the
World Health Organization classifications, and pathological staging
was based on the international staging system revised in 2009
(23). Also, 437 cancer-free
individuals attending the physical examination in Qilu Hospital of
Shandong University were recruited as controls. All blood samples
were stored at -80°C for further studies. To analyze the
association between NSCLC patients prognosis and LAPTM4B
polymorphism, we performed a retrospective study. September 30,
2013 was the end date of follow-up, therefore, a total of 101 cases
diagnosed as NSCLC before September 31, 2008 were investigated and
analyzed. Of these 101 patients, 4 were excluded (3 patients were
lost to follow-up, 1 patient died of perioperative complications),
thus, 97-long-term follow-up patients were enrolled in this
retrospective study. Each patient signed an informed consent
according to the Helsinki Declaration and the present study was
approved by the Ethics Committee of Qilu Hospital.
DNA extraction
Genomic DNA was extracted from a 1 ml peripheral
blood sample obtained from each participant using a RelaxGene Blood
DNA System (Tiangen, China) according to the protocol provided by
manufacturer. Then, the newly extracted genomic DNA was stored at
-20°C for the subsequent PCR analysis.
DNA genotyping
Polymorphism of LAPTM4B was determined by PCR
analysis using the specific primers. The primer sequences were:
forward, 5′-GCCGACTAGGGGACTGGC GGA-3′ and reverse,
5′-CGAGAGCTCCGAGCTTCTGCC-3′ (7).
The PCR was conducted under the standard procedure in a 20 μl
reaction mixture, which included 10 μl 2XPCR mix (TransGen, China),
1 μl sense primer, 1 μl reverse primer, 2 μl template DNA and 6 μl
ddH2O. The PCR conditions were: pre-denaturation at 95°C
for 5 min, 35 cycles of denaturation at 94°C for 30 sec, annealing
at 68°C for 30 sec, extension at 72°C for 30 sec, then final
extension at 72°C for 5 min. Human glyceraldehyde-3-phosphate
dehydrogenase (GAPDH) was regarded as the positive inner control,
which was a 258-bp fragment. The primer sequences were: sense,
5′-TGTCG CTGTTGAAGTCAGAGGAGA-3′ and reverse, 5′-AGAACA
TCATCCCTGCCTCTACTG-3′. PCR products were analyzed by
electrophoresis in a 2.5% agarose gel and visualized with ethidium
bromide.
Statistical analysis
Statistical analyses were performed using SPSS 18.0
software (SPSS, Inc., Chicago, IL, USA). The Chi-squared test or
the Fisher’s exact was used to test the genotypic frequencies of
the patients and controls for Hardy-Weinberg equilibrium and to
examine the association between LAPTM4B genotype and the
patient clinicopathological factors. The relationship between
LAPTM4B polymorphisms and susceptibility to NSCLC was
estimated using unconditional logistic regression method. The
Kaplan-Meier method and log-rank test were used to calculate
survival curve and to compare the statistical significance of
survival differences among patient subgroups. Multivariate Cox
regression analysis was carried out to identify the potential
prognostic factor of NSCLC patients. P-value <0.05 was
considered to indicate a statistically significant difference.
Results
LAPTM4B genotypes
Three different LAPTM4B genotypes designated
LAPTM4B*1/1, LAPTM4B*2/2 and LAPTM4B*1/2 were
identified by PCR-agarose gel electrophoresis analysis. As shown in
Fig. 2, 10 representative
individuals were chosen to display the polymorphism of
LAPTM4B genotypes. LAPTM4B*1/1 displayed a 204-bp
fragment, LAPTM4B*2/2 displayed a 223-bp fragment and
LAPTM4B*1/2 had both fragments. The upper 258-bp band in
each lane in Fig. 2 was
GAPDH as the positive inner control.
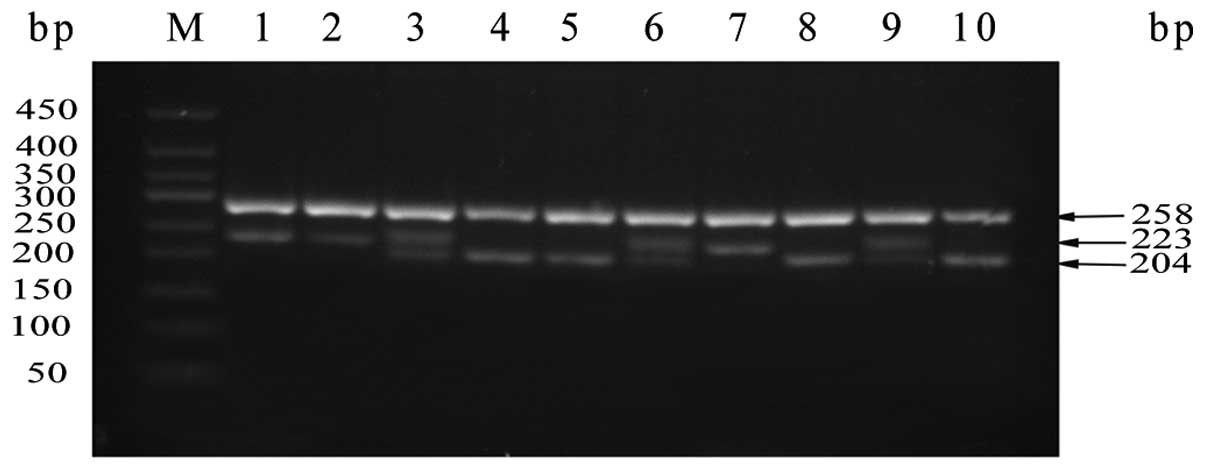 | Figure 2Genotyping of LAPTM4B. Samples were
analyzed by separation in 2.5% agarose gel electrophoresis. Lane M,
DNA marker (50, 100, 150, 200, 250, 300, 350, 400 and 450 bp). The
lower bands represent the target products. Lanes 4, 5, 8 and 10:
genotype *1/1; lanes 1, 2 and 7: genotype *2/2; lanes 3, 6 and 9:
genotype *1/2. The upper bands show the amplified product of human
GAPDH (258 bp) that served as the positive internal control.
LAPTM4B, lysosome-associated protein transmembrane-4β. |
LAPTM4B polymorphism and NSCLC
susceptibility
We collected 392 NSCLC subjects and 437 cancer-free
control subjects for the present study. The main characteristics of
the patients and controls are presented in Table I. The mean age of cases and controls
was 58.8 and 55.4 years, respectively. We divided the ages into two
groups on the basis of the median age. There was no statistical
significance in gender between cases and controls, while the age
distribution revealed a significant difference (P<0.001).
 | Table IDistribution of gender and age in
case and control groups. |
Table I
Distribution of gender and age in
case and control groups.
| Controls (n=437)
(%) | Cases (n=392)
(%) | Pa |
|---|
| Gender, n (%) | | | P=0.17 |
| Male | 257 (58.8) | 212 (54.1) | |
| Female | 180 (41.2) | 180 (45.9) | |
| Age, years, n
(%) | | |
P<0.001 |
| <60 | 253 (57.9) | 158 (40.3) | |
| ≥60 | 184 (42.1) | 234 (59.7) | |
The genotype and allele frequencies of
LAPTM4B in cases and controls in the present case-control
study are summarized in Table II.
We proved that the observed genotype frequencies for this
polymorphism were in agreement with the Hardy-Weinberg equilibrium
in the controls, suggesting that the control group could represent
the whole population. Table II
shows that there was a higher proportion of LAPTM4B*1/2 and
LAPTM4B*2/2 in cases (43.6 and 16.1%, respectively) than in
controls (40.3 and 8%, respectively). Odds ratio analysis indicated
that LAPTM4B*1/2, *2/2 were correlated with a significant
increased risk of NSCLC compared with LAPTM4B*1/1 (OR, 1.48;
95% CI, 1.076–2.037; OR, 2.855; 95% CI, 1.722–4.734, respectively).
In addition, the frequency of LAPTM4B*2 was notably higher
in cases than in controls (37.9 vs. 28.1%, respectively).
LAPTM4B*2 carriers had a 1.649-fold (95% CI, 1.316–2.068)
higher risk of developing NSCLC than non-LAPTM4B*2 carriers.
Our data indicated that LAPTM4B*2 was likely to be
associated with an increased susceptibility to NSCLC in a Chinese
population.
 | Table IIDistribution of genotypes and alleles
of LAPTM4B in case and control groups. |
Table II
Distribution of genotypes and alleles
of LAPTM4B in case and control groups.
| Controls
n (%) | Cases
n (%) | OR (95%CI)a |
|---|
| Genotypes |
| *1/1 | 226 (51.7) | 158 (40.3) | |
| *1/2 | 176 (40.3) | 171 (43.6) | 1.48
(1.076–2.037) |
| *2/2 | 35 (8) | 63 (16.1) | 2.855
(1.722–4.734) |
| Total | 437 (100) | 392 (100) | |
| Alleles |
| *1 | 628 (71.9) | 487 (62.1) | |
| *2 | 246 (28.1) | 297 (37.9) | 1.649
(1.316–2.068) |
| Total | 874 (100) | 874 (100) | |
LAPTM4B polymorphism and
clinicopathological variables
We investigated the distribution of clinical
parameters such as gender, age, pathological type, differentiation
degree, lymph node metastasis, smoking and tumor-node-metastasis
(TNM) stage in different genotypes of LAPTM4B in these 392
patients. We found that LAPTM4B*2 was significantly
associated with poor histopathologic differentiation (P=0.012), but
not with gender, age, pathological type, lymph node metastasis,
smoking and TNM stage (P>0.05), shown in Table III.
 | Table IIIDistribution of three genotypes of
LAPTM4B in relation to clinicopathological variables in 392
patients with non-small cell lung cancer. |
Table III
Distribution of three genotypes of
LAPTM4B in relation to clinicopathological variables in 392
patients with non-small cell lung cancer.
| Variables | No. of cases | LAPTM4B
genotypes | χ2 | Pa |
|---|
|
|---|
| *1/1 | *1/2 | *2/2 |
|---|
| Gender | | | | | 1.907 | 0.358 |
| Male | 212 | 82 | 91 | 39 | | |
| Female | 180 | 76 | 80 | 24 | | |
| Age (years) | | | | | 0.048 | 0.976 |
| <60 | 158 | 64 | 68 | 26 | | |
| ≥60 | 234 | 94 | 103 | 37 | | |
| Pathological
type | | | | | 0.741 | 0.69 |
| AC | 220 | 85 | 100 | 35 | | |
| SCC | 172 | 73 | 71 | 28 | | |
|
Differentiation | | | | | 12.924 | 0.012 |
| Well | 109 | 56 | 40 | 13 | | |
| Moderately | 129 | 54 | 58 | 17 | | |
| Poorly | 154 | 48 | 73 | 33 | | |
| Tumor size
(cm) | | | | | 0.622 | 0.733 |
| <4 | 200 | 77 | 89 | 34 | | |
| ≥4 | 192 | 81 | 82 | 29 | | |
| Lymph node
metastasis | | | | | 3.003 | 0.223 |
| No | 186 | 83 | 77 | 26 | | |
| Yes | 206 | 75 | 94 | 37 | | |
| Classification of
TNM | | | | | 3.155 | 0.789 |
| I | 101 | 46 | 42 | 13 | | |
| II | 130 | 53 | 54 | 23 | | |
| III | 124 | 47 | 56 | 21 | | |
| IV | 37 | 12 | 19 | 6 | | |
| Smokingb | | | | | 1.396 | 0.498 |
| Yes | 168 | 64 | 79 | 25 | | |
| No | 224 | 94 | 92 | 38 | | |
LAPTM4B genotype and NSCLC prognosis
We conducted survival analysis in these 97
followed-up patients to examine the impact of LAPTM4B
polymorphism on NSCLC prognosis. As of September 30, 2013 which was
the end date for follow-up, 23 (23.7%) patients were alive and 74
(76.3%) patients had succumbed to the disease. We first performed
univariate analysis of clinicopathological variables for prognosis
using the Kaplan-Meier method and log-rank test. According to the
survival analysis, the 5-year overall and disease-specific survival
were 62.7 and 68.1%, respectively. As shown in Table IV, it was of no statistically
significant difference in some clinical parameters, such as age,
gender and pathological type (P>0.05). However, the overall
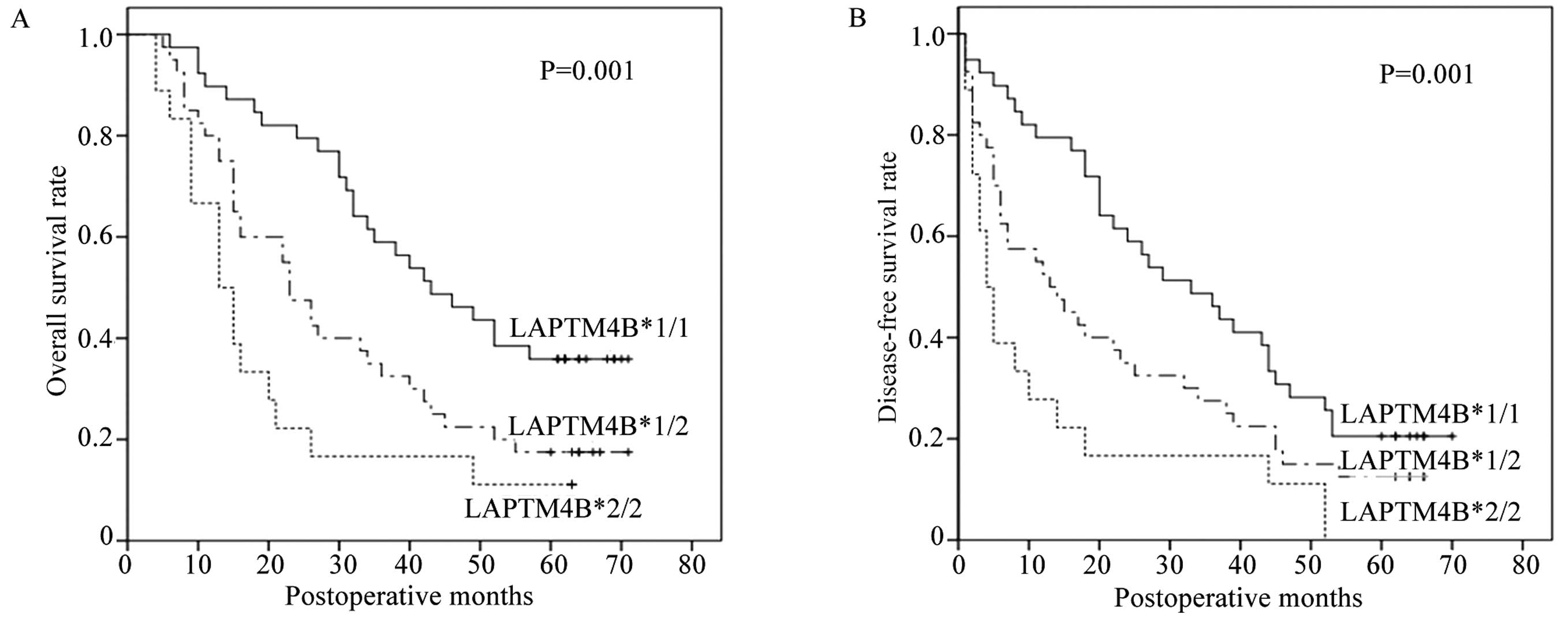
5-year survival rate of patients with LAPTM4B*2/2 and
LAPTM4B*1/2 (11.1 and 17.5%, respectively, vs. 35.9%,
P=0.001) was significantly lower than that of LAPTM4B*1/1
carriers. In addition, Kaplan-Meier analysis of disease-free
survival also demonstrated poor 5-year survival rate in patients
with LAPTM4B*2/2 and LAPTM4B*1/2 (0 and 12.5%, respectively,
vs. 20.5%, P=0.001) shown in Fig.
3. Furthermore, poor prognosis was strikingly associated with
large tumor size (P<0.01 and P<0.01, respectively), high
grade of histopathological differentiation (P=0.016 and P=0.056,
respectively), positive lymph node metastasis (P<0.01 and
P<0.01, respectively) and high TNM stage (P<0.01 and
P<0.01, respectively).
 | Table IVUnivariate Kaplan-Meier survival
analysis of overall survival and disease-free survival in 97
patients with non-small cell lung cancer. |
Table IV
Univariate Kaplan-Meier survival
analysis of overall survival and disease-free survival in 97
patients with non-small cell lung cancer.
| Prognostic
variables | No. of cases | OS (months) | DFS (months) |
|---|
|
|
|---|
| Mean ± SE | 95% CI | Pa | Mean ± SE | 95% CI | Pa |
|---|
| Gender | | | | 0.727 | | | 0.954 |
| Male | 59 | 35.2±3.1 | 29.1–41.4 | | 25.7±3.1 | 19.6–31.8 | |
| Female | 38 | 35.1±3.7 | 27.7–42.4 | | 26.0±3.6 | 19.0–33.0 | |
| Age (years) | | | | 0.963 | | | 0.933 |
| <60 | 45 | 35.0±3.6 | 27.8–42.1 | | 25.1±3.4 | 18.5–31.8 | |
| ≥60 | 52 | 35.3±3.2 | 29.1–41.6 | | 26.3±3.2 | 19.9–32.6 | |
| Pathological
type | | | | 0.172 | | | 0.198 |
| AC | 63 | 39.5±4.2 | 31.2–47.8 | | 23.8±2.8 | 18.2–29.3 | |
| SCC | 34 | 32.8±2.9 | 27.2–38.4 | | 29.4±4.0 | 21.5–37.2 | |
|
Differentiation | | | | 0.016 | | | 0.056 |
| Poorly | 42 | 27.3±3.2 | 21.0–33.6 | | 19.1±3.4 | 12.4–25.8 | |
| Moderately | 24 | 35.7±5.2 | 25.6–45.8 | | 26.0±4.7 | 16.9–35.2 | |
| Well | 31 | 44.5±3.8 | 37.2–51.9 | | 33.8±3.7 | 26.7–41.0 | |
| Tumor size
(cm) | | | |
<0.001 | | |
<0.001 |
| <4 | 53 | 43.4±3.4 | 36.7–50.0 | | 34.9±3.4 | 28.2–41.7 | |
| ≥4 | 44 | 24.8±2.5 | 19.8–29.7 | | 15.1±2.3 | 10.6–19.6 | |
| Lymph node
metastasis | | | |
<0.001 | | |
<0.001 |
| No | 51 | 47.4±3.1 | 41.3–53.5 | | 37.6±3.3 | 31.3–44.0 | |
| Yes | 46 | 21.4±2.4 | 16.6–26.2 | | 12.9±2.3 | 8.5.4–17.4 | |
| Classification of
TNM | | | |
<0.001 | | |
<0.001 |
| I | 30 | 49.9±3.7 | 42.7–57.1 | | 40.3±3.9 | 32.8–47.9 | |
| II | 35 | 35.3±3.7 | 28.1–42.5 | | 25.7±3.8 | 18.3–33.0 | |
| III | 24 | 22.7±3.9 | 15.1–30.2 | | 14.7±3.6 | 7.7–21.7 | |
| IV | 8 | 12.3±2.5 | 7.4–17.1 | | 4.0±1.6 | 0.8–7.2 | |
| LAPTM4B
genotypes | | | | 0.001 | | | 0.001 |
| *1/1 | 39 | 45.5±3.6 | 38.6–52.5 | | 35.2±3.7 | 28.0–42.3 | |
| *1/2 | 40 | 31.3±3.5 | 24.4–38.1 | | 22.5±3.5 | 15.6–29.3 | |
| *2/2 | 18 | 20.4±4.3 | 12.1–28.8 | | 12.8±4.1 | 4.7–20.9 | |
LAPTM4B genotype is an independent
prognostic marker for NSCLC patients
To analyze the independent factor of LAPTM4B
polymorphism on prognosis, a multivariate Cox regression model
adjusted for statistically significant prognostic factors was
performed. Table V shows that
subjects with the LAPTM4B*2 allele had, respectively,
2.025-fold (95% CI, 1.21–3.388; P=0.007) increased mortality and
1.678-fold (95% CI, 1.044–2.696; P=0.033) increased recurrence of
NSCLC than those carrying LAPTM4B*1/1 genotype,
demonstrating that the LAPTM4B genotype was an independent
prognostic factor for NSCLC patients. In addition, lymph node
metastasis and TNM classification also retained their prognostic
significance.
 | Table VMultivariate Cox regression model of
overall survival and disease-free survival in 97 patients with
non-small cell lung cancer. |
Table V
Multivariate Cox regression model of
overall survival and disease-free survival in 97 patients with
non-small cell lung cancer.
| OS | DFS |
|---|
|
|
|
|---|
| Variables | RR | 95% CI | Pa | RR | 95% CI | Pa |
|---|
| Tumor size
(cm) |
| <4 | | | | | | |
| ≥4 | 1.184 | 0.62–2.261 | 0.61 | 1.393 | 0.75–2.587 | 0.294 |
| Lymph node
metastasis |
| No | | | | | | |
| Yes | 2.261 | 1.241–4.116 | 0.008 | 2.202 | 1.242–3.905 | 0.007 |
|
Differentiation |
| Well | | | | | | |
| Moderately | 0.967 | 0.473–1.974 | 0.926 | 0.952 | 0.498–1.821 | 0.883 |
| Poorly | 1.386 | 0.724–2.652 | 0.325 | 1.273 | 0.702–2.31 | 0.427 |
| Classification of
TNM |
| I | | | | | | |
| II | 1.527 | 0.783–2.981 | 0.214 | 1.288 | 0.705–2.353 | 0.411 |
| III | 2.478 | 1.128–5.445 | 0.024 | 2.102 | 1.031–4.285 | 0.041 |
| IV | 3.969 | 1.46–10.788 | 0.007 | 4.502 | 1.705–11.89 | 0.002 |
| LAPTM4B
genotypes |
| *1/1 | | | | | | |
| *1/2 + *2/2 | 2.025 | 1.21–3.388 | 0.007 | 1.678 | 1.044–2.696 | 0.033 |
Discussion
In the present study, we detected the polymorphism
of LAPTM4B genotypes in NSCLC patients, then analyzed the
relationship between distribution of LAPTM4B genotypes and
susceptibility, prognosis of lung cancer. Our finding was that
patients with LAPTM4B*2 showed a higher risk of
susceptibility and mortality of NSCLC compared with non-carriers.
To our knowledge, this is the first study to prove polymorphism of
LAPTM4B may act as an indicator for the prognosis of NSCLC.
Our results are consistent with the putative role LAPTM4B
plays in carcinogenesis and tumor progression.
LAPTM4B, a novel oncogene, was first detected
in hepatocellular carcinoma and was then found upregulated in
various solid malignant tumors (24). LAPTM4B is closely related
with the biological behaviors of malignant tumors. An increase in
LAPTM4B expression, as measured by mRNA and protein, was
associated with tumor progression and poorer survival in patients
with breast (25), pancreatic
(8), colon (26), ovary (27) and cervical cancer (11). Furthermore, extensive studies have
been performed to account for such outcomes. Studies have shown
that upregulation of LAPTM4B could promote cell
proliferation (28), invasion,
migration (29) and may inhibit
cell apoptosis (30,31) in vitro, while in nude mice
the time of tumorigenesis was markedly shortened (29). It was assumed that various signal
molecules were associated with cellular malignant transformation
after the alteration of LAPTM4B protein expression level. It
has been confirmed that LAPTM4B protein could upregulate
some proliferation-promoting transcription factors such as c-Myc,
c-Jun and c-Fos, and cell cycle-promoting proteins such as cyclin
D1 and E (28). Meanwhile, it could
also activate PI3K/AKT signaling pathway to motivate cellular
multidrug resistance (31). A
recent study clarified that cAMP responsive element binding
protein-1 (CREB1) played an important role in LAPTM4B
transcriptional regulation (32).
The unique region of 19-bp sequence at 5′UTR in the
first exon was identified as the difference between
LAPTM4B*1 and LAPTM4B *2. The 19-bp difference in the
first exon of the LAPTM4B gene altered the open reading
frame (ORF), so it may influence the structure and function of the
protein encoded by it. Previous studies have shown that
LAPTM4B*2 allele played important roles in the
susceptibility and prognosis of many tumors. To date, the exact
mechanism of the phenomenon has not yet been revealed. It has been
reported that a 40 kD protein was encoded by LAPTM4B*2
allele with an extra 53 amino acids compared with the
LAPTM4B*1 allele encoding a 35 kD protein, which may explain
such a difference. Different structures and activities of proteins
could alter the cellular metabolism and signal pathway, then induce
the malignant transformation. This area requires further study.
The present study was carried out under the strict
rules of clinical trial, hence our conclusions are firm. Our data
demonstrated that LAPTM4B*2 is associated with NSCLC
susceptibility. This is also the first study to clarify that the
polymorphism of LAPTM4B genotype is related to NSCLC
progression and prognosis. Although the exact molecular mechanisms
which underlie the function of LAPTM4B in lung
carcinogenesis have yet to be fully clarified, LAPTM4B*2
could be a novel potential marker to estimate susceptibility and
prognosis of NSCLC.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (no. 30571844), the Science and
Technology Development Foundation of Shandong Province (no.
2009GG10002007), and the National Natural Science Foundation of
Shandong Province (no. ZR2009CM090). The authors thank all the
people and patients who participated in the present study.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Brognard J, Clark AS, Ni Y and Dennis PA:
Akt/protein kinase B is constitutively active in non-small cell
lung cancer cells and promotes cellular survival and resistance to
chemotherapy and radiation. Cancer Res. 61:3986–3997.
2001.PubMed/NCBI
|
|
3
|
Couraud S, Zalcman G, Milleron B, Morin F
and Souquet PJ: Lung cancer in never smokers - a review. Eur J
Cancer. 48:1299–1311. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sun S, Schiller JH and Gazdar AF: Lung
cancer in never smokers - a different disease. Nat Rev Cancer.
7:778–790. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Toh CK, Gao F, Lim WT, et al:
Never-smokers with lung cancer: epidemiologic evidence of a
distinct disease entity. J Clin Oncol. 24:2245–2251. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu X, Zhou R, Zhang Q, et al:
Identification and characterization of LAPTM4B encoded by a human
hepatocellular carcinoma-associated novel gene. Beijing Da Xue Xue
Bao. 35:340–347. 2003.(In Chinese).
|
|
7
|
Shao GZ, Zhou RL, Zhang QY, et al:
Molecular cloning and characterization of LAPTM4B, a novel
gene upregulated in hepatocellular carcinoma. Oncogene.
22:5060–5069. 2003.PubMed/NCBI
|
|
8
|
Zhang G, Liang Y, Huang Y, Chen Y and Zhou
R: Elevated lysosome-associated protein transmembrane-4β-35 is an
independent prognostic marker in pancreatic carcinoma. J Int Med
Res. 40:1275–1283. 2012.PubMed/NCBI
|
|
9
|
Zhou L, He XD, Chen J, et al:
Overexpression of LAPTM4B-35 closely correlated with
clinicopathological features and post-resectional survival of
gallbladder carcinoma. Eur J Cancer. 43:809–815. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang Y, Yang H, McNutt MA, et al: LAPTM4B
overexpression is an independent prognostic marker in ovarian
carcinoma. Oncol Rep. 20:1077–1083. 2008.PubMed/NCBI
|
|
11
|
Meng F, Luo C, Hu Y, et al: Overexpression
of LAPTM4B-35 in cervical carcinoma: a clinicopathologic study. Int
J Gynecol Pathol. 29:587–593. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang S, Zhang QY and Zhou RL: Relationship
between LAPTM4B gene polymorphism and susceptibility of
primary liver cancer. Ann Oncol. 23:1864–1869. 2012.
|
|
13
|
Liu Y, Zhang QY, Qian N and Zhou RL:
Relationship between LAPTM4B gene polymorphism and
susceptibility of gastric cancer. Ann Oncol. 18:311–316. 2007.
|
|
14
|
Cheng XJ, Xu W, Zhang QY and Zhou RL:
Relationship between LAPTM4B gene polymorphism and
susceptibility of colorectal and esophageal cancers. Ann Oncol.
19:527–532. 2008.
|
|
15
|
Yang H, Zhai G, Ji X, Xiong F, Su J and
McNutt MA: Correlation of LAPTM4B polymorphisms with gallbladder
carcinoma susceptibility in Chinese patients. Med Oncol.
29:2809–2813. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu Y, Liu Y, Zhou R, et al: LAPTM4B
polymorphisms is associated with ovarian cancer susceptibility and
its prognosis. Jpn J Clin Oncol. 42:413–419. 2012. View Article : Google Scholar
|
|
17
|
Li X, Kong X, Chen X, et al: LAPTM4B
allele *2 is associated with breast cancer susceptibility and
prognosis. PLoS One. 7:e449162012.
|
|
18
|
Deng LJ, Zhang QY, Liu B and Zhou RL:
Relationship between LAPTM4B gene polymorphism and
susceptibility of lung cancer. Beijing Da Xue Xue Bao. 37:302–305.
2005.(In Chinese).
|
|
19
|
Li C, Zhou Q, Wang Y, Chen X, Yang X and
Zhu D: Relationship between LAPTM4B gene polymorphism and
susceptibility of lung cancer. Zhongguo Fei Ai Za Zhi. 9:109–112.
2006.(In Chinese).
|
|
20
|
Zhai G, Yan K, Ji X, et al: LAPTM4B allele
*2 is a marker of poor prognosis for gallbladder carcinoma. PLoS
One. 7:e452902012.
|
|
21
|
Yang H, Zhai G, Ji X, Xiong F, Su J and
McNutt MA: LAPTM4B allele *2 is a marker of poor prognosis
following hepatic tumor resection for hepatocellular carcinoma.
PLoS One. 7:e349842012.
|
|
22
|
Meng F, Li H, Zhou R, Luo C, Hu Y and Lou
G: LAPTM4B gene polymorphism and endometrial carcinoma risk
and prognosis. Biomarkers. 18:136–143. 2013. View Article : Google Scholar
|
|
23
|
Sobin LGM and Wittekind C: TNM
Classification of Malignant Tumours. 7th edition. Wiley-Blackwell;
New York: 2010
|
|
24
|
Kasper G, Vogel A, Klaman I, et al: The
human LAPTM4b transcript is upregulated in various types of solid
tumours and seems to play a dual functional role during tumour
progression. Cancer Lett. 224:93–103. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xiao M, Jia S, Wang H, Wang J, Huang Y and
Li Z: Overexpression of LAPTM4B: an independent prognostic
marker in breast cancer. J Cancer Res Clin Oncol. 139:661–667.
2013.
|
|
26
|
Kang Y, Yin M, Jiang W, et al:
Overexpression of LAPTM4B-35 is associated with poor prognosis in
colorectal carcinoma. Am J Surg. 204:677–683. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yin M, Lou C, Zhang W, et al: LAPTM4B
overexpression is a novel independent prognostic marker for
metastatic ovarian tumors. Int J Gynecol Cancer. 22:54–62. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu X, Xiong F, Wei X, Yang H and Zhou R:
LAPTM4B-35, a novel tetratransmembrane protein and its PPRP motif
play critical roles in proliferation and metastatic potential of
hepatocellular carcinoma cells. Cancer Sci. 100:2335–2340. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang H, Xiong F, Wei X, Yang Y, McNutt MA
and Zhou R: Overexpression of LAPTM4B-35 promotes growth and
metastasis of hepatocellular carcinoma in vitro and in vivo. Cancer
Lett. 294:236–244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou L, He XD, Yu JC, Zhou RL, Shan Y and
Rui JA: Overexpression of LAPTM4B-35 attenuates epirubucin-induced
apoptosis of gallbladder carcinoma GBC-SD cells. Surgery.
150:25–31. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li L, Wei XH, Pan YP, et al: LAPTM4B: a
novel cancer-associated gene motivates multidrug resistance through
efflux and activating PI3K/AKT signaling. Oncogene. 29:5785–5795.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang M, Xu JJ, Zhou RL and Zhang QY: cAMP
responsive element binding protein-1 is a transcription factor of
lysosomal-associated protein transmembrane-4 beta in human breast
cancer cells. PLoS One. 8:e575202013. View Article : Google Scholar
|