Introduction
Lung cancer is a malignancy with high incidence and
mortality rates. Woldwide, 13% of patients with tumors are
diagnosed with lung cancer, and the mortality due to lung cancer
accounts for 18% of all cancer-related deaths (1,2).
Conventional strategies for treating advanced lung cancer include
surgery, chemotherapy and radiotherapy. However, most patients do
not have the opportunity to undergo surgical resection when they
are diagnosed with lung cancer, and the common therapeutic strategy
is radiation therapy. While radiotherapy has shown commendable
antitumor activity, serious side-effects always accompany
treatment. These include radiation-induced lung injury and
radioactive tracheal injury, resulting in the discontinuation of
antitumor treatment limiting its application (3–6).
Clearly, new therapeutic strategies are imminently needed to
optimize available regimens.
Angiogenesis plays an important role in a large
variety of physiological and pathological conditions, such as
embryonic development, wound healing and diabetic retinopathy
(7,8). Evidence indicates that most malignant
solid tumor growth and metastases are partly dependent on
angiogenesis (9–11). Accordingly, antiangiogenesis is an
effective strategy of antitumor treatment by preventing new vessel
formation in tumors and destroying the abnormal microvascular
system which provides oxygen and nutrients to the tumor tissue.
Endostatin, originally purified from the culture
supernatant of murine hemangioendothelioma (EOMA) cells, is a type
of endogenous angiogenesis inhibitor, which locates in the basement
membrane of blood vessels (12).
Amino acid sequence analysis shows that endostatin is a 20-kDa
C-terminal fragment of collagen XVIII which belongs to the
multiplexin family (13,14). Extensive basic and preclinical
research reveals that endostatin not only inhibits the
proliferation and migration of endothelial cells, but also induces
the apoptosis of vascular endothelial cells, thereby suppressing
angiogenesis (15–17). Further studies have demonstrated
that endostatin also inhibits the growth and metastasis of many
types of solid tumors. Therefore, endostatin targeting angiogenesis
has been increasingly used in preclinical and clinical research
(12,18,19).
Most therapeutic strategies of endostatin have utilized the
purified protein (20,21). However, as a protein drug,
endostatin has a short half-life in vivo, and thus needs
continuous administration or increased concentrations to maintain
effective serum levels. Moreover, the process of protein
purification may denature endostatin, which reduces the resultant
yield rates. These problems seriously hamper the clinical
application of endostatin (2,22). One
possible strategy to overcome these issues is the utilization of
gene therapy, and adenoviral vectors are widely utilized for gene
therapy and show no disadvantages of purified protein injection
(2,23–25).
Although tumor treatment with endostatin has shown
various effects, the inhibitory effect of endostatin is limited
since numerous factors are secreted by endothelial cells to promote
angiogenesis, while endostatin inhibits just one of these
angiogenic factors (3). In
addition, the endothelium can acquire resistance to angiogenesis
inhibitors through epigenetic changes as recently demonstrated
(26). Thus, the combination of
endostatin with other treatment strategies (such as radiotherapy)
may improve the antitumor effect of endostatin.
In the present study, we established an LL/2 cell
subcutaneous tumor model and a lung metastasis model, and evaluated
the efficacy of the combined treatment of recombinant endostatin
adenovirus (Ad-Endo) with low-dose irradiation. The present study
revealed that this strategy enhances the antitumor effect without
serious side-effects, and provides a rational alternative treatment
for lung cancer and other solid tumors.
Materials and methods
Cell culture and Ad-Endo preparation
Lewis lung carcinoma (LL/2) and human embryonic
kidney (HEK293) cell lines were obtained from the American Type
Culture Collection (ATCC; Manassas, VA, USA). They were cultured in
Dulbecco’s modified Eagle’s medium (DMEM) which included 10% fetal
bovine serum (FBS) plus amikacin and streptomycin routinely. The
viral particles which were amplified in HEK293 cells were purified
by CsCl gradient ultracentrifugation and measured by absorption (at
A260). The viral titer was quantified with the standard 50% tissue
culture infectious dose (TCID50) assay.
Determination of the multiplicity of
infection (MOI)
LL/2 cells were infected with Ad-GFP at multiple
MOIs [5, 10, 50, 100 and 200, the plaque-forming units (pfu)/cells
in 1.0 ml complete medium) or involved no infection. After
culturing at 37°C for 48 h, the cells containing GFP were observed
by fluorescence microscopy and then subjected to flow cytometry and
analyzed using CellQuest software.
Detection of endostatin expression by
western blot analysis
LL/2 cells were infected with Ad-Endo and Ad-null at
an MOI of 100 or not infected. Cells were conditioned at 37°C for
48 h, and the supernatant was collected and concentrated by
ultrafiltration (Millipore, Darmstadt, Germany). The retentate was
mixed with 5× SDS sample buffer and separated on a 12% SDS-PAGE gel
and then transferred onto a polyvinylidene difluoride (PVDF)
membrane. After the membrane was blocked by TBST containing 5%
defatted milk for 2 h, it was probed with goat anti-human
endostatin polyclonal antibody (1:1,000; R&D Systems) overnight
at 4°C, followed by horseradish peroxidase-conjugated anti-goat
immunoglobulin (1:5,000; ZSGB-BIO, Beijing, China) for 2 h at 37°C.
Three washes (15 min per wash) were carried out after each
incubation step. The protein bands were detected by an enhanced
chemiluminescence detection system (Pierce, Rockford, IL, USA).
Determination of transgene expression by
enzyme-linked immunosorbent assay
Human endostatin expression in the supernatant of
cultured cells or in mouse serum was detected with the human
endostatin ELISA kit (Sigma-Aldrich, St. Louis, MO, USA) according
to the manufacturer’s instructions. For the supernatant of cells
infected with viral particles, the supernatant was collected for
testing after 24, 48 and 72 h. For the mouse serum, on days 1, 7,
14 and 21 after tail vein administration of viral particles
(1×109 pfu) in the LL/2 cell subcutaneous tumor model,
the blood samples were obtained and centrifuged for 10 min at 3,000
rpm, and then the serum was collected for testing.
The LL/2 subcutaneous tumor model and the
in vivo antitumor effect of Ad-Endo combined with radiotherapy
To evaluate the antitumor effect of Ad-Endo combined
with radiotherapy in the established animal model, C57BL/6 female
mice (6–8 weeks of age) were injected s.c. with 3×106
LL/2 cells in 100 μl phosphate-buffered saline (PBS) in the right
flank. Seven days later, the tumors were palpable on each mouse,
and the mice were then randomly divided into 6 groups (10
mice/group). i) Ad-Endo group received an i.v. injection of
1×108 pfu/100 μl recombinant adenovirus. ii) RT group
received irradiation performed with the radiation treatment system
Elekta Compact™ (Elekta AB, Stockholm, Sweden) at a dose of 2 Gy
and locally operated on the tumors by shielding the rest of the
body. iii) Ad-Endo plus RT group received Ad-Endo administration
i.v. and 2 Gy dose of irradiation confined to the tumors. iv)
Ad-null group received an i.v. injection of 1×108
pfu/100 μl control adenovirus. v) Ad-null plus RT group received
Ad-null administration i.v. and 2 Gy dose of irradiation confined
to tumors. vi) NS group received an i.v. administration of 100 μl
of PBS at the same schedule as the other groups. When the groups
were completed, treatment was initiated at this time (day 0). The
viral particles were administered once every three days from day 0,
and the irradiation was delivered each day from day 2 to 6 and day
9 to 13.
Tumor growth was monitored by measuring tumor width
(W) and length (L) every three days. The tumor volume (V) was
calculated using the fomula: V = 0.52 × L × W2. The body
weight of mice was monitored every three days until day 22 after
treatment. On day 22, the mice were sacrificed, and the major
organs were obtained for histologic analysis. All experiments
involving mice were guided by the Animal Care and Use Committee of
our institute.
To assay the survival time, 60 mice were challenged
with LL/2 cells as above. When the tumors were palpable, the mice
were grouped and treated as above. The time of death of each mouse
was recorded.
Assessment of tumor vascularization and
anoxia factors
To analyze the mechanism of the enhanced antitumor
effect of Ad-Endo combined with irradiation, the tumor microvessel
density (MVD) and the level of anoxia factors was detected by
immunohistochemistry of CD31 and HIF-1α.
The quantification of MVD was carried out as
previously described (27).
Briefly, when the mice were sacrificed, the tumors were obtained
and fixed with formalin. Then the paraffin-embedded sections of the
tumors were probed with a monoclonal rat anti-mouse CD31 antibody
(1:400; Santa Cruz Biotechnology, Santa Cruz, CA, USA), followed by
a biotinylated polyclonal rabbit anti-rat IgG antibody (1:200;
Vector Laboratories, Burlingame, CA, USA). The positive reaction
was detected using the DAB substrate kit (Vector Laboratories), and
the sections were counterstained with hematoxylin. Finally, the
microvessels were calculated with an Olympus microscope at a ×200
magnification.
The levels of anoxia factors in the tumors were
detected as followed. First, the tumors were obtained and fixed
with formalin. Then the paraffin-embedded sections were probed with
a monoclonal rabbit anti-mouse HIF-1α antibody (1:200, Abcam,
Cambridge, MA, USA), followed by a biotinylated goat anti-rabbit
IgG antibody (1:200; Vector Laboratories). A positive reaction was
detected using the DAB substrate kit, and sections were
counterstained with hematoxylin. Finally, the sections were mounted
with glass coverslips, and images were captured with an Olympus
microscope at a ×200 magnification.
Detection of apoptosis
The apoptotic cells in the tumor tissues were
detected using the terminal deoxynucleotidyl transferase-mediated
nick end labeling assay (TUNEL) (In Situ Cell Death Detection kit;
Roche, Basel, Switzerland), according to the manufacturer’s
guidelines. Images were obtained by an Olympus fluorescence
microscope at a ×200 magnification. The apoptotic cells were
counted in 5 high power fields in each slide. The percentage of
apoptotic cells among the total number of tumor cells was
calculated as the apoptotic index.
Alginate encapsulation assay
The alginate encapsulation assay was performed as
previously described (28).
Briefly, LL/C cells were resuspended with 1.5% sodium alginate
(m/v; Sigma-Aldrich), and then the suspension was added into a
oscillation pool containing 250 mM CaCl2 to form
alginate beads. Each bead contained 1×105 LL/2 cells,
and each mouse was implanted s.c. with four beads on the back. The
mice were treated with Ad-Endo or/and irradiation as above, and
with Ad-null or saline as control. On day 15, the mice were
administered i.v. 100 μl of FITC-dextran solution (100 mg/kg; Sigma
Chemicals) and sacrificed 20 min later. Images of the alginate
beads were captured using a SPOT Flex camera. The beads were then
removed and were transferred into tubes containing 2 ml of
physiological saline. After being pounded, the beads were rested
for 1 h and centrifuged for 5 min at 1,000 × g. The supernatant was
collected and the fluorescence was determined to assess blood
vessel formation.
The LL/2 cell pulmonary metastasis tumor
model and the in vivo antitumor effect of Ad-Endo combined with
radiotherapy
The 6- to 8-week-old female C57BL/6 mice were
injected with 3×105 LL/2 cells in 100 μl PBS by tail
vein. Three days later, the mice were randomly divided into 6
groups identical to those described above. On the same day, the
mice were treated with viral particles or/and irradiation as
above.
On day 25, the mice were sacrificed, and the lungs
were obtained. The weight and metastatic nodules of the lungs were
determined. The lungs were then fixed with formalin for
histological analysis.
Observation of toxicity
Drug toxicity indices, such as weight loss, ruffled
fur, diarrhea, anorexia, skin ulcerations and behavior change, were
closely observed during the entire couse of treatment. In addition,
the major organs (heart, liver, spleen, lung and kindey) were fixed
in 4% paraformaldehyde, and embedded in paraffin to obtain
sections. The sections were stained with hematoxylin and eosin
(H&E) and observed using the double-blind method.
Statistical analysis
SPSS 17.0 was used for statistical analysis. For
comparison of individual time-points, differences between groups
were tested by performing analysis of variance (ANOVA) and the
unpaired Student’s t-test. A diference was regarded as significant
at P<0.05.
Results
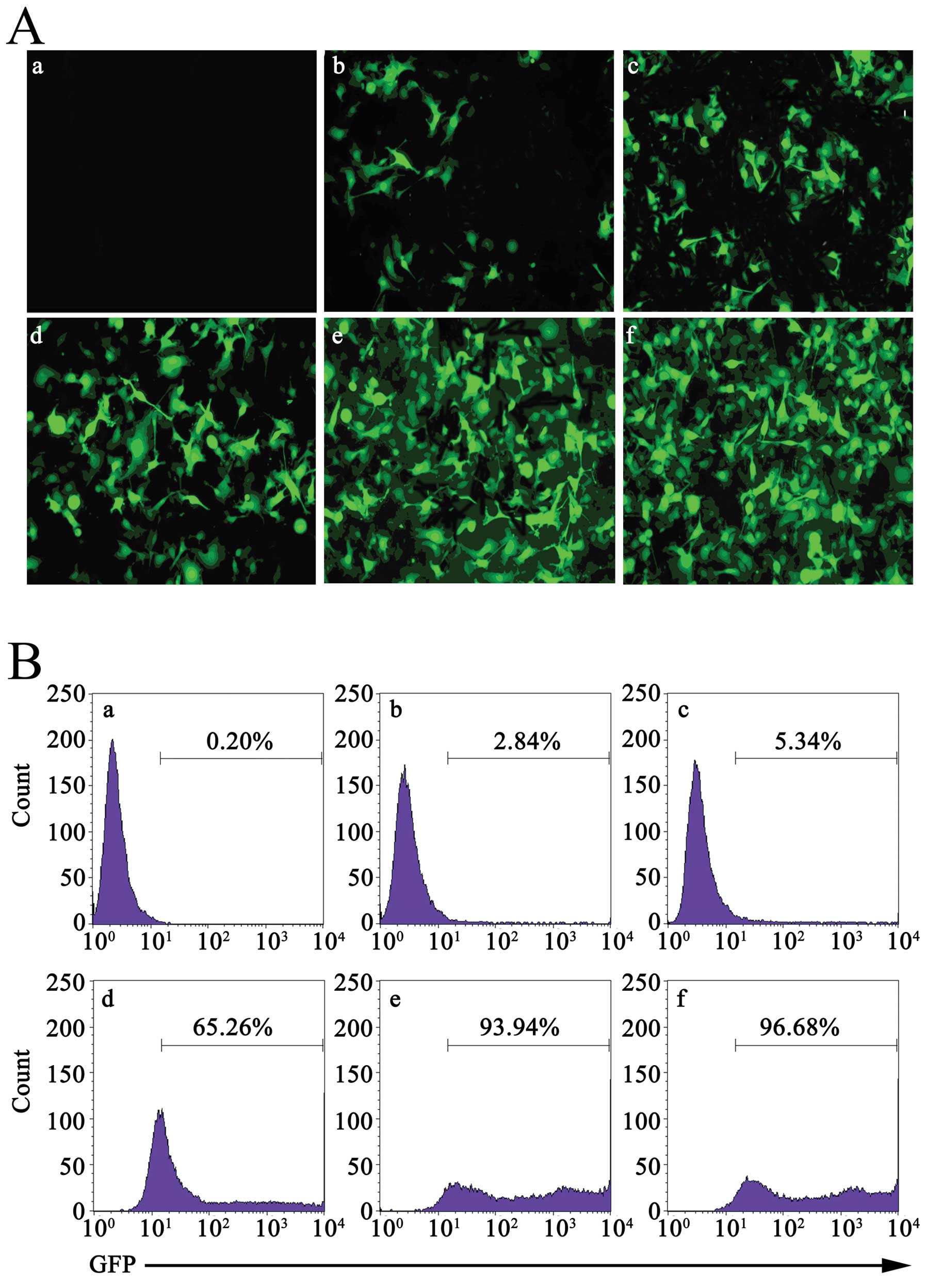
Determination of the MOI
LL/2 cells were infected with multiple MOIs (5, 10,
50, 100 and 200) of Ad-GFP. Forty-eight hours later, the level of
GFP in cells was detected with fluorescence microscopy. As shown in
Fig. 1A, with the increase in MOI,
the proportion of LL/2 cells expressing GFP was increased from 3.1
to 98%. Although there were no differences in proportion between
MOI 100 and MOI 200, the cells which were infected with MOI 200
were seriously damaged. Similar results were obtained through flow
cytometry (Fig. 1B). These results
indicated that Ad-GFP could effectively infect LL/2 cells, in a
concentration-dependent manner. Based on this, we selected MOI 100
to continue the following studies.
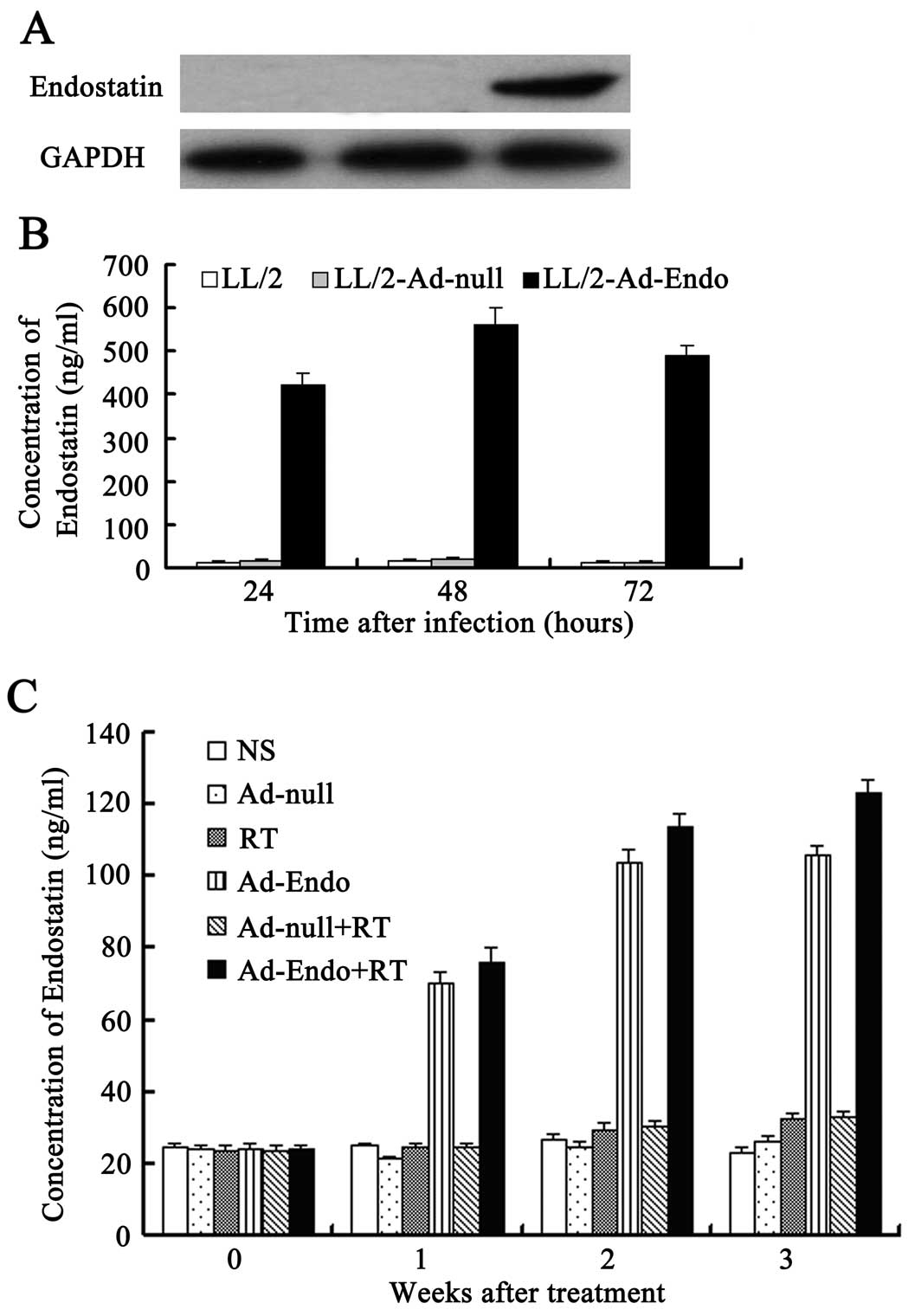
Expression of recombinant human
endostatin in vitro and in vivo
LL/2 cells were infected with Ad-Endo or Ad-null at
MOI 100. Forty-eight hours later, the culture supernatants were
collected and concentrated to detect the expression of endostatin
using western blot analysis. As shown in Fig. 2A, a distinct band ~20 kDa, similar
to the volume of endostatin, was visualized in the supernatants of
the Ad-Endo-infected cells, but not in that of the control
cells.
To further analyze the level and duration of
endostatin, we detected the level of endostatin in the culture
supernatants of LL/2 cells, which were separately infected with
Ad-Endo or Ad-null for 24, 48 and 72 h using ELISA. The results
showed that after 24 h, in the LL/2 cells infected with Ad-Endo,
endostatin was detected in the supernatants and the level reached
420±30 ng/ml (Fig. 2B). The level
peaked (560±40 ng/ml) after 48 h and decreased slightly after 72 h
(490±25 ng/ml). However, we could not detect the expression of
endostatin in the cells which were infected with Ad-null or in
cells not infected. Similar results also were detected in
vivo (Fig. 2C). These
observations indicated that LL/2 cells infected with Ad-Endo stably
and persistently expressed secretory endostatin.
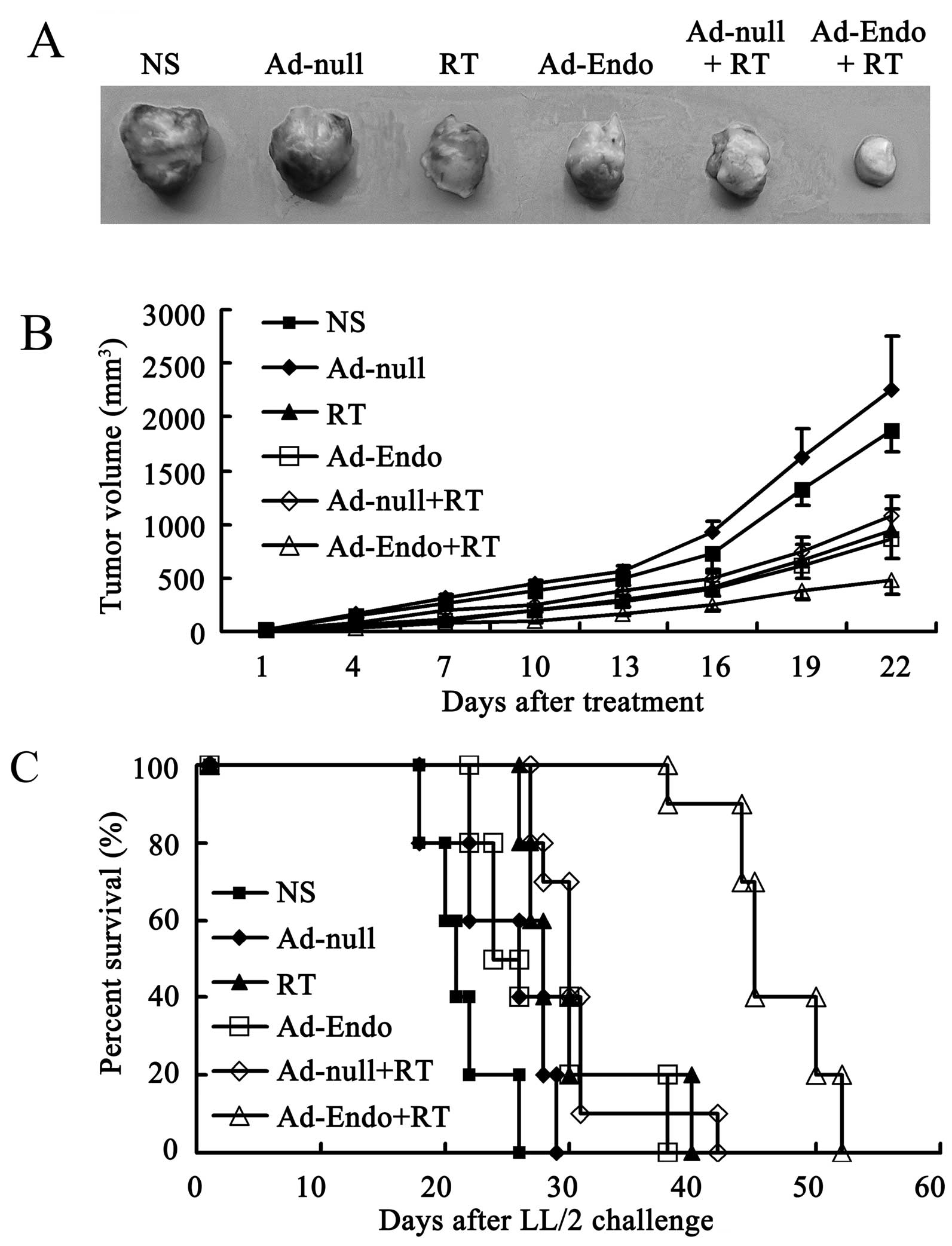
Combination treatment significantly
reduces tumor growth and prolonges the life-span in the LL/2 cell
subcutaneous tumor model
To assay the antitumor effect of Ad-Endo combined
with low-dose irradiation, the LL/2 cell subcutaneous tumor model
was established, and the mice were randomly divided into 6 groups
after 7 days. The mice were then treated as described in Materials
and methods, and the tumor growth was monitored every 3 days. As
shown in Fig. 3A, the tumors in the
Ad-Endo + RT group were much smaller than tumors in the other
groups. Similar to the results in Fig.
3A, the tumor growth rate in the RT group and Ad-Endo group was
relatively slower than that in the NS and Ad-null groups
(P<0.05). Compared with the RT, Ad-Endo and Ad-null + RT groups,
the combination Ad-Endo + RT group exhibited a more marked
inhibition of tumor growth, and the difference was noted from day
16 after treatment. Dramatically, from day 4 after treatment, the
tumor growth rate in the combination group showed significant
differences when compared with the NS and Ad-null groups. The
inhibitory effect was determined by the weight reduction. At the
end of the present study, the tumors were removed and measured
(data not shown). Compared with the NS group, the tumor weight in
the Ad-Endo + RT group was reduced 79%, which was higher than that
in the other groups (58.4% in RT group, 52% in Ad-Endo group, and
61.7% in Ad-null + RT group). Furthermore, the combination group
significantly showed a longer life-span than that of the other
groups (Fig. 3C). These results
revealed that Ad-Endo + RT improved the antitumor effect of
radiotherapy and that of Ad-Endo alone, which reflects the
synergism between radiotherapy and Ad-Endo.
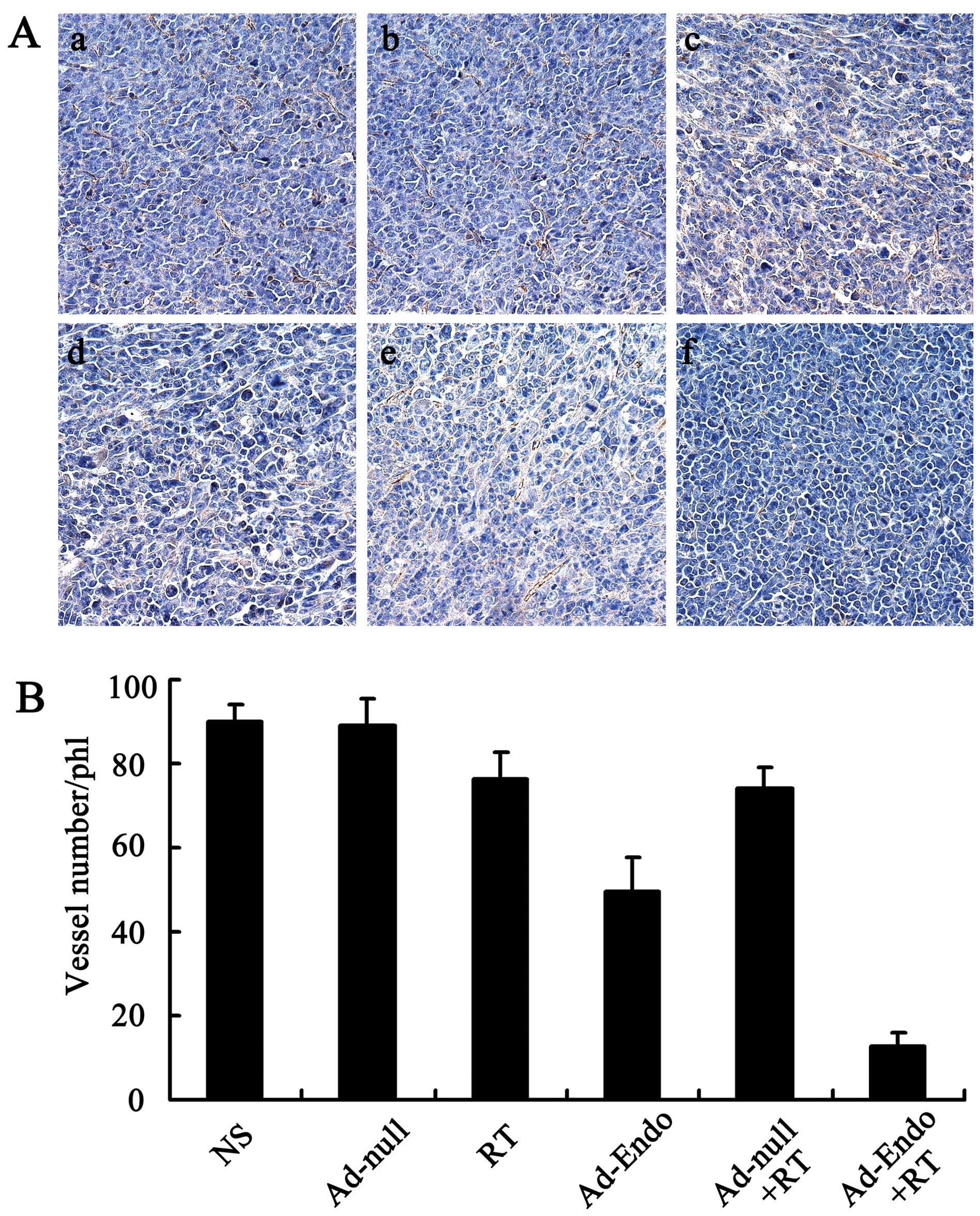
Inhibition of tumor-induced angiogenesis
and increase in apoptosis in vivo
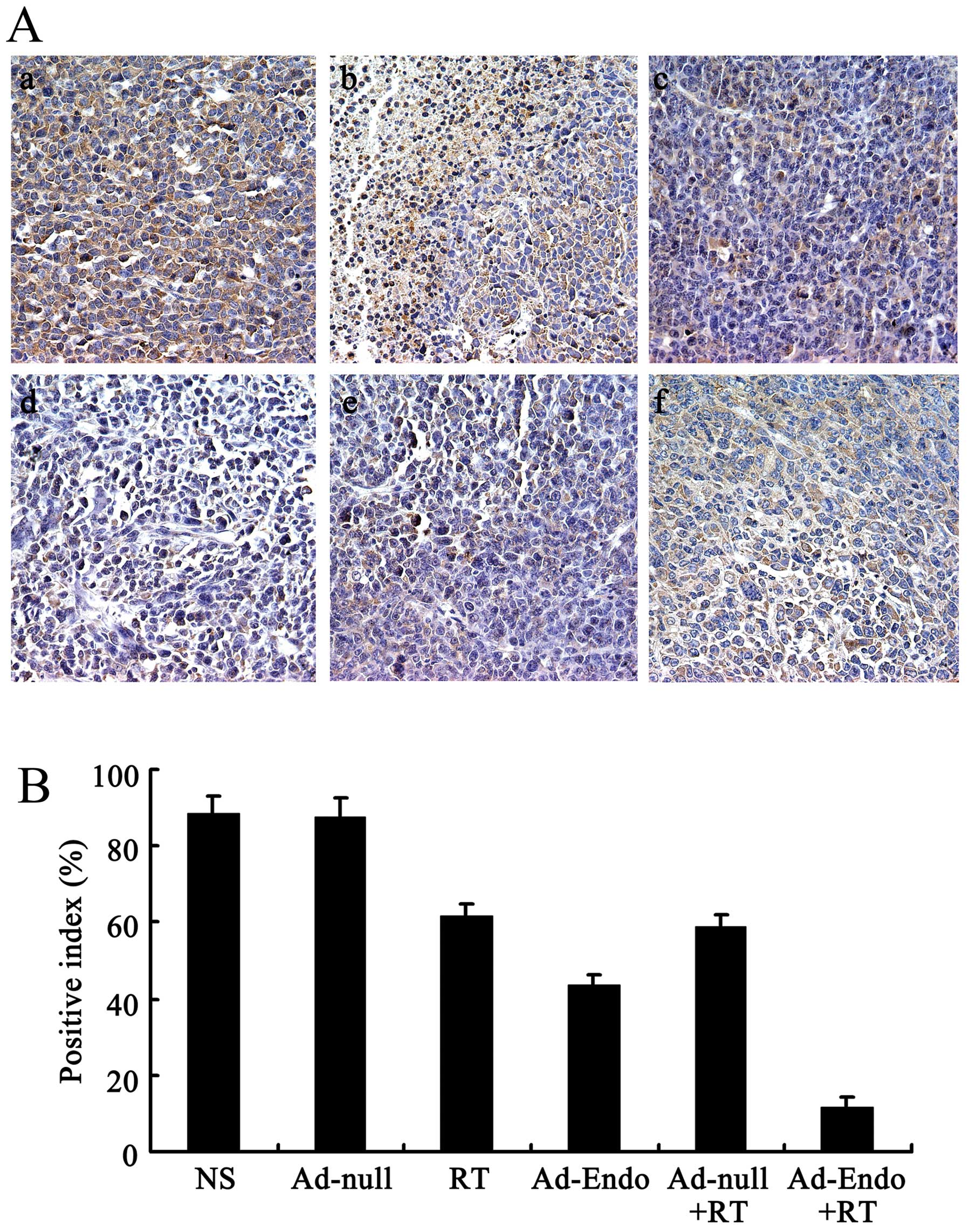
To evaluate the mechanism of the enhanced antitumor
effect of Ad-Endo combined with irradiation, the tumor sections
were stained with the anti-CD31 antibody. As shown in Fig. 4A, tumors of the control groups
(including NS group, Ad-null group, RT group, Ad-Endo group and
Ad-null + RT group) exhibited larger microvessel counts than those
of the Ad-Endo + RT group. The MVD in the Ad-Endo + RT group was
12.6±3.2%, while the MVD was 49.4±8.2% in the Ad-Endo group,
74.3±4.6% in the Ad-null + RT group, 76.5±6.1% in the RT group,
88.9±6.4% in the Ad-null group and 90.2±3.7% in the NS group,
respectively (Fig. 4B).
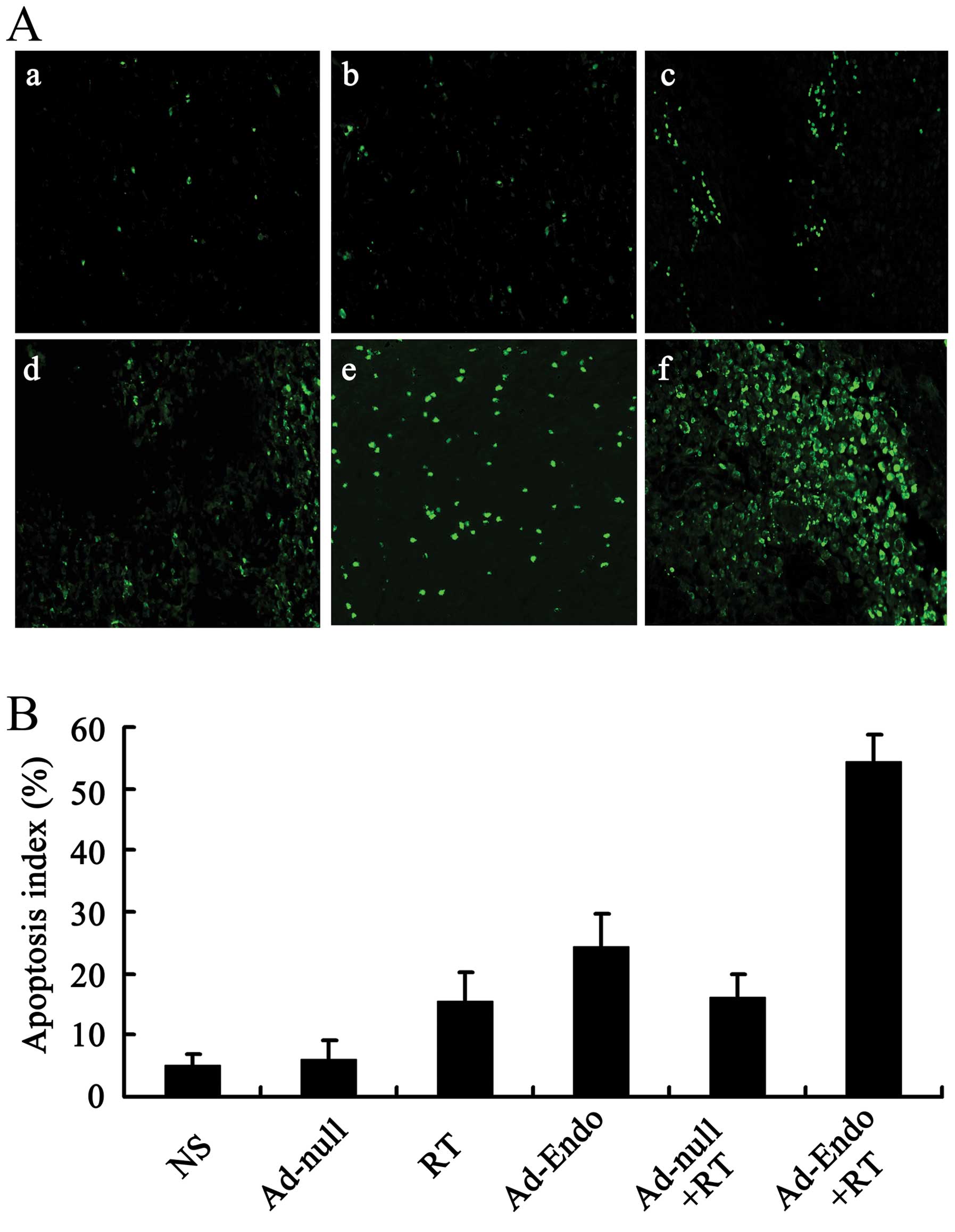
In order to estimate the apoptosis in tumor tissues,
the tumor sections were stained with TUNEL reagent, and the
positive results were confirmed as described in Materials and
methods. Compared with the control groups, the number of apoptotic
cells was increased in the Ad-Endo group, RT group and Ad-null + RT
group. Furthermore, more obvious apoptosis was noted in the tumor
sections of the Ad-Endo + RT group (Fig. 5A). In regards to the apoptotic
index, the Ad-Endo + RT group showed the highest index among all of
the groups (Fig. 5B). This suggests
that Ad-Endo combined with irradiation obviously inhibited the
angiogenesis in tumors, which promoted increased apoptosis.
Detection of anoxia factors with
immunohistochemistry of HIF-1α
Tumor growth can induce anoxia in tissues, which
increases the expression of HIF-1α. To assay the influence of
Ad-Endo combined with irradiation on the level of anoxia factors,
immunohistochemistry of HIF-1α was applied. As shown in Fig. 6A and B, compared with the control
groups, a decrease in expression of HIF-1α was noted in the
Ad-Endo, RT and Ad-null + RT groups. However, the combined Ad-Endo
+ RT group displayed the lowest level among all the groups, which
was far less than those in the radiotherapy groups and the Ad-Endo
group.
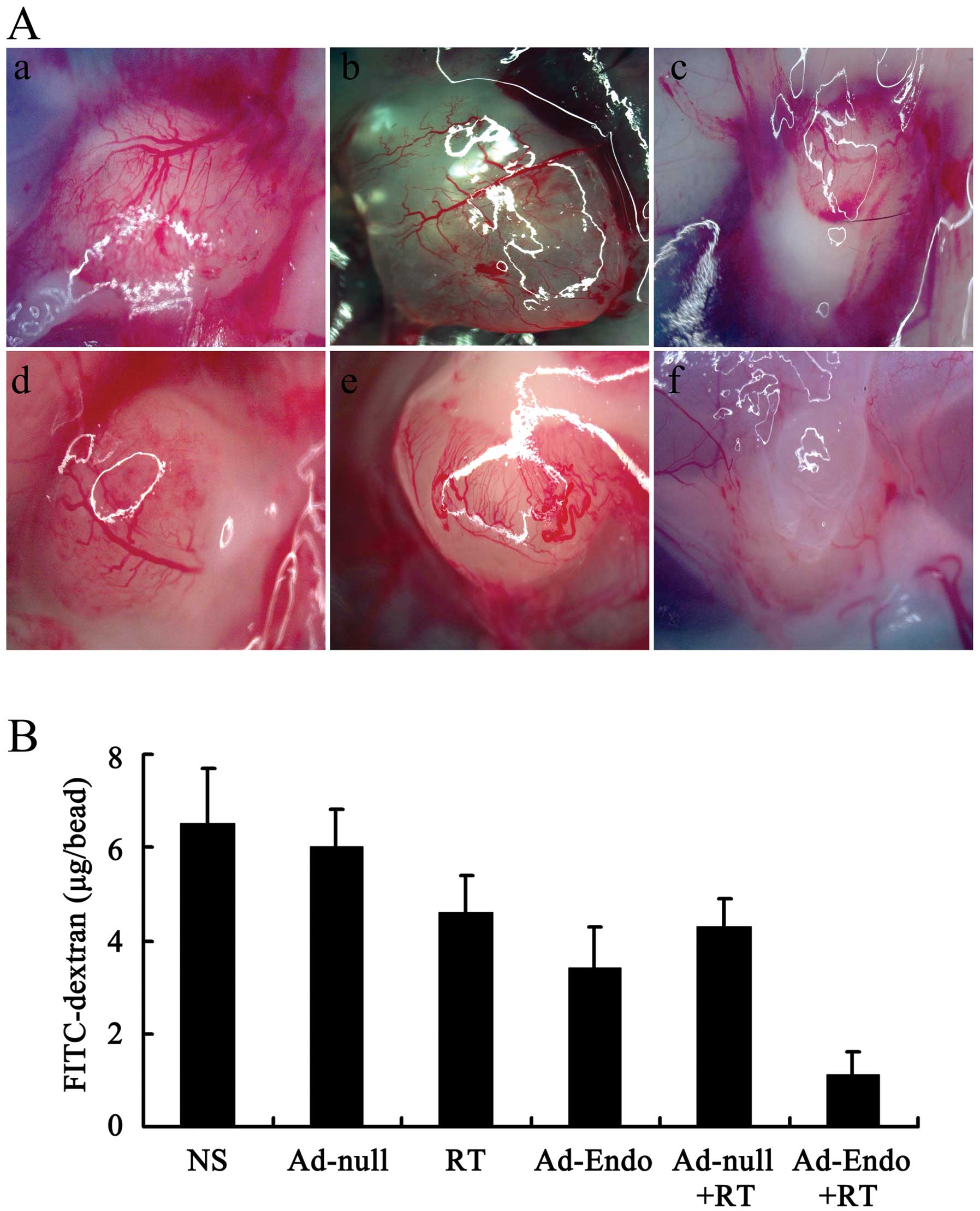
Inhibition of angiogenesis in the
alginate encapsulation assay
Alginates can provide the desired marix for tumor
angiogenesis and growth without affecting the release and delivery
of cytokines and drugs. Thus, we evaluated the effect of Ad-Endo
combined with irradiation on angiogenesis in vivo using the
alginate encapsulation assay. As shown in Fig. 7A, there was less micrangium on the
surface of beads in the Ad-Endo + RT group than that in the other
groups. The FITC-dextran uptake was nearly 83% less in the Ad-Endo
+ RT group than that in the NS group or Ad-null group and 67–76%
less than that in the other groups (Fig. 7B).
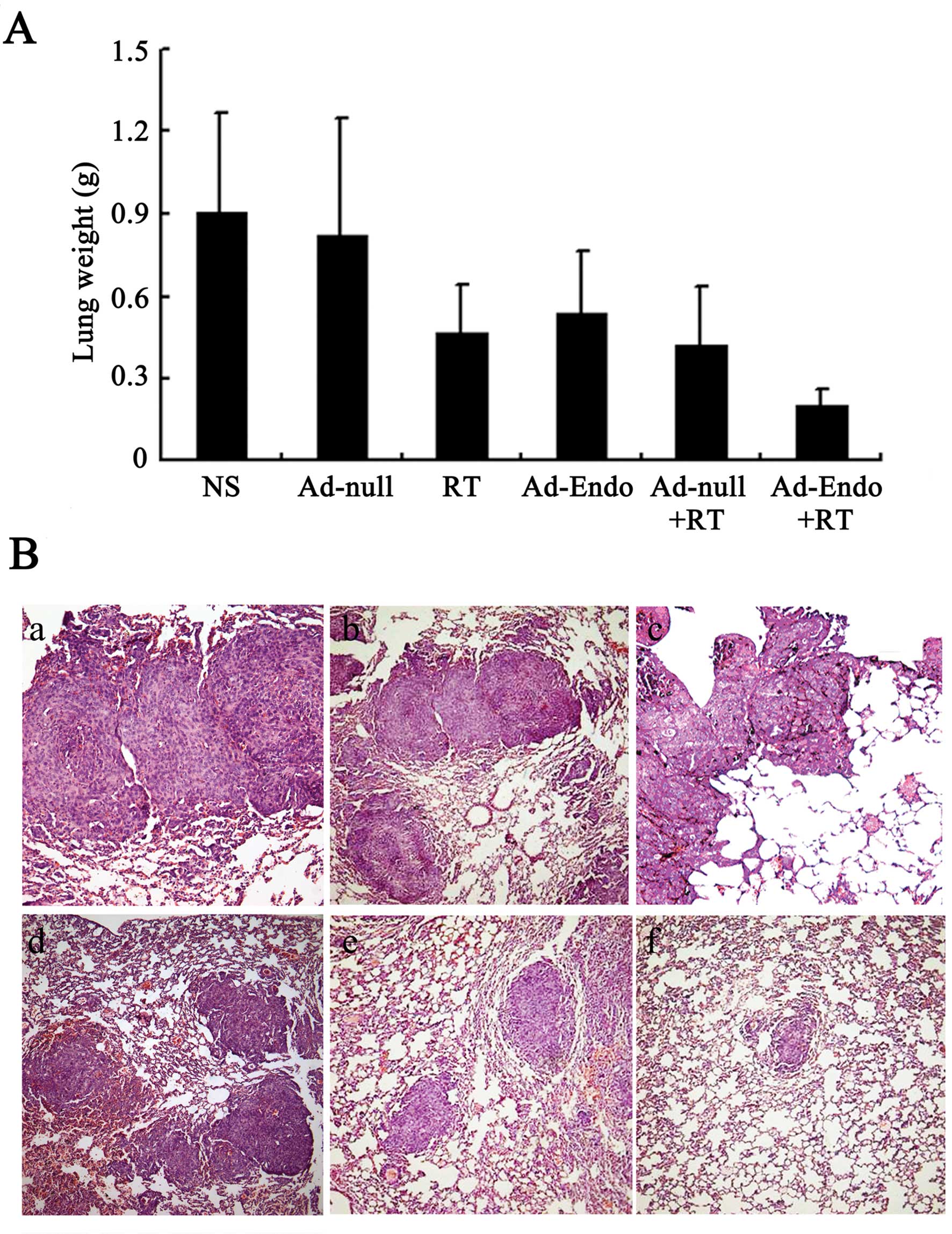
Combination treatment significantly
inhibits tumor metastasis in the LL/2 cell pulmonary metastasis
tumor model
Angiogenesis is not only relevant to tumor growth,
but also promotes tumor metastasis. Thus, the LL/2 cell pulmonary
metastasis tumor model was established and treatment with Ad-Endo
combined with low-dose of irradiation was carried out. The lung
weight of all mice is presented in Fig.
8A. The lung weight was 0.91±0.36 g in the NS group and
0.82±0.43 g in the Ad-null group; these weights were heavier than
those in the RT group (0.47±0.17 g), Ad-Endo group (0.54±0.22 g)
and Ad-null + RT group (0.42±0.21 g). Compared with the other
groups, the Ad-Endo + RT group had the lowest lung weight (0.2±0.06
g). Moreover, the number of pulmonary metastatic nodules was
counted, and the results are shown in Fig. 8B and Table I. The number of nodules in the NS
and Ad-null groups was 180 and 165, respectively, for which
metastatic nodules >3 mm occupied 30 and 28%. A smaller number
of nodules was noted in the RT group (92), Ad-Endo group (110) and
Ad-null + RT group (86), in which nodules >3 mm accounted for
15, 20 and 18%, respectively. Notably, the number of nodules in the
Ad-Endo + RT group was 10 and only 1.3% of nodules were >3 mm,
which indicates that Ad-Endo combined with low-dose irradiation
effectively inhibited the pulmonary metastasis of LL/2 cell
tumors.
 | Table IThe number of pulmonary metastatic
nodules in the LL/2 cell pulmonary metastasis tumor model. |
Table I
The number of pulmonary metastatic
nodules in the LL/2 cell pulmonary metastasis tumor model.
| Group | Median no. of
metastatic nodules | % large (>3 mm)
metastatic nodules |
|---|
| NS | 180 | 30 |
| Ad-null | 165 | 28 |
| RT | 92 | 15 |
| Ad-Endo | 110 | 20 |
| Ad-null+RT | 86 | 18 |
| Ad-Endo+RT | 10 | 1.3 |
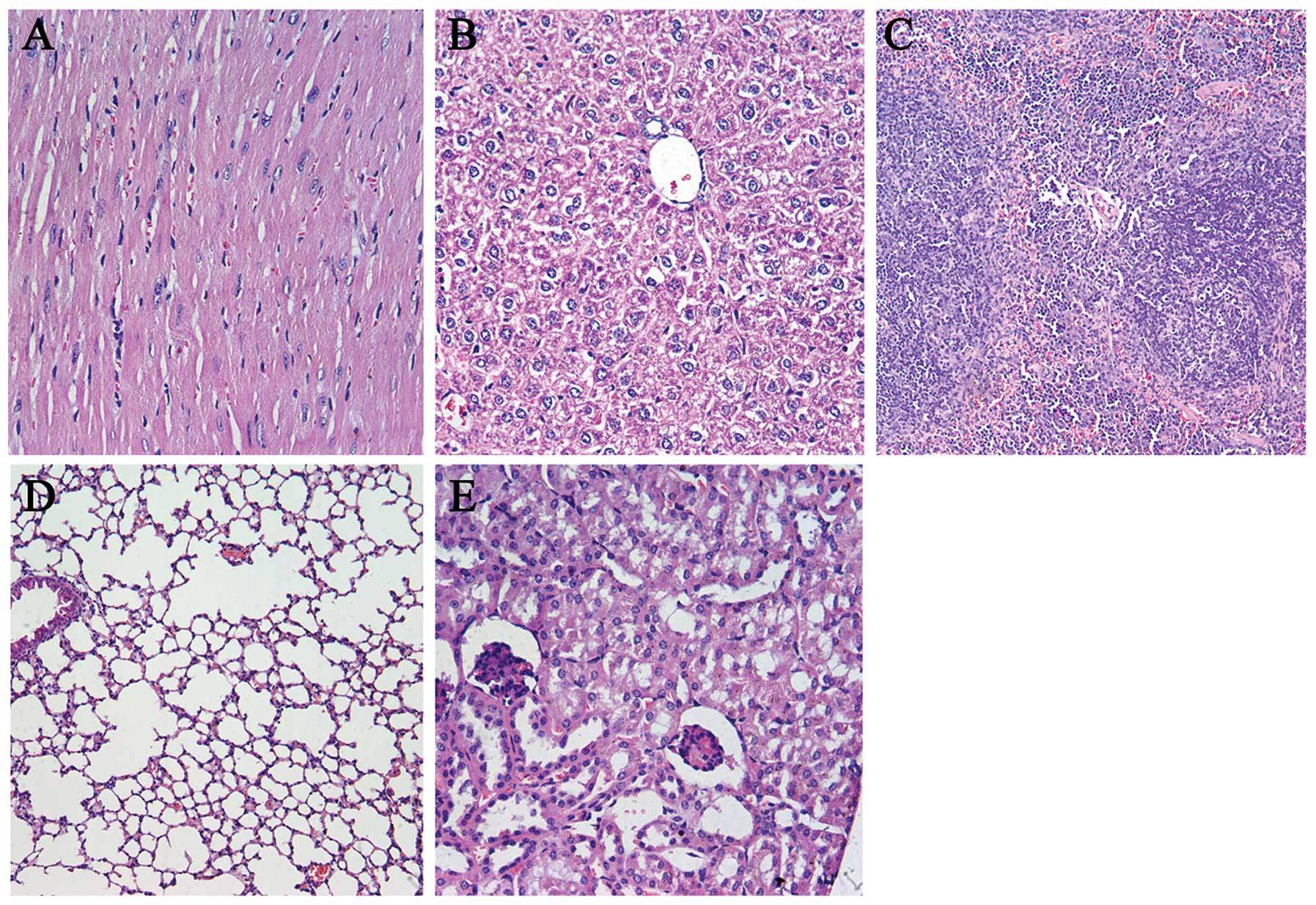
Observation of toxicity
In order to evaluate the potential side-effects of
the treatment, the toxicity assessment was executed in both models.
There were no obvious differences in the gross measures among all
the groups (data not shown). Moreover, no serious pathological
changes in main organs were noted in the subcutaneous tumor model
(Fig. 9) and pulmonary metastasis
tumor model (data not shown).
Discussion
Angiogenesis plays an important role in tumor growth
and metastasis (10,29,30).
In 1945, Algire et al (31)
proposed that ‘the growth of tumor transplants is dependent on the
development of a vascular system’. Subsequently, Folkman et
al (32) found that tumor
tissue could not exceed a size of 2–3 mm without the recruitment of
new blood vessels, which supply nutrients and gases to tumors.
Therefore, the authors hypothesized that the development of a
vascular system was critical to the growth of tumors and suggested
that antiangiogenesis may be an effective strategy for cancer
therapy
Angiogenesis is a complex process which includes the
proliferation and migration of vascular endothelial cells,
formation of new capillaries and extracelluar matrix turnover
(33–35). Several studies have shown that a
host of cytokines regulates this process (33,36,37).
Although the mechanism is not clearly elucidated, endostatin is a
significant factor which can inhibit endothelial cell proliferation
and suppress the growth and metastasis of tumors (18,38).
Wickstrom et al (39) found
that endostatin binds to α5β1 integrin and caveolin-1, which blocks
the adhesion of endothelium and inhibits the activity of
metalloproteases, further suppressing angiogenesis. Furthermore,
endostatin can reduce the expression of Bcl-2 and Bel-XI, suppress
the growth of endothelium and promote apoptosis, consequently
inhibiting the growth and metastasis of tumors (15). Thus, endostatin is an effective
target for tumor therapy. In the present study, we also
demonstrated that endostatin can effectively inhibit the growth and
metastasis of LL/2 tumors, and prolong the survival of
tumor-bearing mice.
Although research has shown that the recombinant
protein of endostatin can effectively inhibit tumor growth and
metastasis, its widespread application has been hampered by
difficulties in the large scale production of the protein and the
high concentration of endostatin needed to maintain an antitumor
effect (36,40,41).
Gene therapy with adenovirus vectors, which are gaining interest,
have been reported to have beneficial effects in tumor treatment
(24,42–44).
In the present study, an adenovirus expressing the secreted form of
endostatin was identified as an effective strategy for endostatin
gene therapy. After infection with Ad-GFP, LL/2 cells secreted GFP
in a concentration-dependent manner. In addition, the expression of
endostatin in vitro was examined by western blotting.
Endostatin protein could be probed in the supernatant of LL/2 cells
infected with Ad-Endo, which indicated that the adenovirus
effectively transferred the endostatin gene to the LL/2 cells and
that the endostatin gene was successfully expressed. Moreover, LL/2
cells infected with Ad-Endo stably and persistently expressed
secretory endostatin which was confirmed by ELISA assay in our
study.
However, as several angiogenesis factors are
produced by endothelial cells and endostatin inhibits one of these
factors, the inhibitory effect of endostatin is limited (3). Therefore, some researchers have
combined conventional treatment strategies with endostatin to
improve the effficacy of endostatin (17,36,45,46).
In the present study, we combined low-dose irradiation with Ad-Endo
to treat the LL/2 cell subcutaneous model and pulmonary metastasis
tumor model. Our study showed that either Ad-Endo or low-dose
irradiation alone partially suppressed tumor growth and metastasis
and prolonged the life-span of tumor-bearing mice along with no
serious toxicity. Notably, the combination therapy showed a more
effective antitumor activity when compared to the other groups.
Although the mechanism of the enhanced antitumor efficacy remains
to be elucidated, it may be related to a decrease in microvascular
density and an increase in apoptosis in tumors. Our present
findings revealed that there were less veins and more apoptotic
cells in the tumors treated with Ad-Endo combined with irradiation
when compared with the other groups. Although radiotherapy is an
effective strategy to treat tumors through direct killing of cells,
the activity is limited when hypoxia occurs in tumors (47–49).
It is known that the rapid growth of tumors can cause regions of
hypoxia in tumors, which contributes to the production of
angiogenesis factors and blood vessel formation (48). Inhibition of angiogenesis with
anti-angiogenic therapy may induce an increase in hypoxia in
tumors, leading to radiotherapy resistance (49). However, other studies have shown
that angiogenesis inhibitors can improve tumor oxygenation and
increase the response to radiotherapy by delaying tumor growth
(17,36,50–52).
In the present study, when the tumors were treated with Ad-Endo,
the blood vessel formation was inhibited and the vascular density
was decreased. Consequently, hypoxia was improved, which boosted
the activity of radiotherapy. Subsequently, more tumor cells were
killed by irradiation, and tumor growth and metastasis were
inhibited. These findings imply that radiotherapy and
antiangiogenic therapy synergistically play a role in tumor
treatment. Moreover, when patients are treated with effective doses
of radiation, side-effects are commonly noted. In the present
study, low-dose irradiation was adopted combined with Ad-Endo,
which not only ensured an antitumor effect, but also inhibited the
incidence of adverse side-effects.
In summary, our findings demonstrated that Ad-Endo
combined with low-dose irradiation effectively inhibited the growth
and metastasis of LL/2 cell tumors in a synergistic manner. The
enhanced inhibitory effects were related to the decrease in
microvascular density, inhibition of blood vessel formation,
improvement in hypoxia and an increase in apoptosis of LL/2 cells.
Further investigation of this strategy for the treatment of lung
cancer and other tumors is warranted.
Acknowledgements
The study was supported by the National Natural
Science Foundation of China (grant no. 81101604).
References
|
1
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics, 2006. CA Cancer J Clin. 56:106–130. 2006. View Article : Google Scholar
|
|
2
|
Ning T, Yan X, Lu ZJ, et al: Gene therapy
with the angiogenesis inhibitor endostatin in an orthotopic lung
cancer murine model. Hum Gene Ther. 20:103–111. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhuang HQ and Yuan ZY: Process in the
mechanisms of endostatin combined with radiotherapy. Cancer Lett.
282:9–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Govindan R, Bogart J and Vokes EE: Locally
advanced non-small cell lung cancer: the past, present, and future.
J Thorac Oncol. 3:917–928. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bunn PA Jr and Thatcher N: Systemic
treatment for advanced (stage IIIb/IV) non-small cell lung cancer:
more treatment options; more things to consider. Conclusion
Oncologist. 13(Suppl 1): 37–46. 2008.
|
|
6
|
Bayman NA, Blackhall F, Jain P, Lee L,
Thatcher N and Faivre-Finn C: Management of unresectable stage III
non-small-cell lung cancer with combined-modality therapy: a review
of the current literature and recommendations for treatment. Clin
Lung Cancer. 9:92–101. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang HL, Yuan C, Zhang DM, et al: A novel
combined conjugate vaccine: enhanced immunogenicity of bFGF with
CRM197 as a carrier protein. Mol Med Rep. 4:857–863.
2011.PubMed/NCBI
|
|
8
|
Carmeliet P and Jain RK: Angiogenesis in
cancer and other diseases. Nature. 407:249–257. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Folkman J: Angiogenesis in cancer,
vascular, rheumatoid and other disease. Nat Med. 1:27–31. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hahnfeldt P, Panigrahy D, Folkman J and
Hlatky L: Tumor development under angiogenic signaling: a dynamical
theory of tumor growth, treatment response, and postvascular
dormancy. Cancer Res. 59:4770–4775. 1999.PubMed/NCBI
|
|
11
|
Shi HS, Yang LP, Wei W, et al:
Systemically administered liposome-encapsulated Ad-PEDF potentiates
the anti-cancer effects in mouse lung metastasis melanoma. J Transl
Med. 11:862013. View Article : Google Scholar
|
|
12
|
O’Reilly MS, Boehm T, Shing Y, et al:
Endostatin: an endogenous inhibitor of angiogenesis and tumor
growth. Cell. 88:277–285. 1997.
|
|
13
|
Oh SP, Warman ML, Seldin MF, et al:
Cloning of cDNA and genomic DNA encoding human type XVIII collagen
and localization of the α1(XVIII) collagen gene to mouse chromosome
10 and human chromosome 21. Genomics. 19:494–499. 1994.PubMed/NCBI
|
|
14
|
Wenzel D, Schmidt A, Reimann K, et al:
Endostatin, the proteolytic fragment of collagen XVIII, induces
vasorelaxation. Circ Res. 98:1203–1211. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dhanabal M, Ramchandran R, Waterman MJ, et
al: Endostatin induces endothelial cell apoptosis. J Biol Chem.
274:11721–11726. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Skovseth DK, Veuger MJ, Sorensen DR, De
Angelis PM and Haraldsen G: Endostatin dramatically inhibits
endothelial cell migration, vascular morphogenesis, and
perivascular cell recruitment in vivo. Blood. 105:1044–1051. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu DS, Wu CM, Huang TH and Xie QD:
Combined effects of radiotherapy and endostatin gene therapy in
melanoma tumor model. Radiat Environ Biophys. 47:285–291. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Blezinger P, Wang J, Gondo M, et al:
Systemic inhibition of tumor growth and tumor metastases by
intramuscular administration of the endostatin gene. Nat
Biotechnol. 17:343–348. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
You ZY, Zhao Y, Liu F, Zhang YD and Wang
JJ: The radiosensitization effects of Endostar on human lung
squamous cancer cells H-520. Cancer Cell Int. 10:172010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cui R, Ohashi R, Takahashi F, et al:
Signal transduction mediated by endostatin directly modulates
cellular function of lung cancer cells in vitro. Cancer Sci.
98:830–837. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thomas JP, Arzoomanian RZ, Alberti D, et
al: Phase I pharmacokinetic and pharmacodynamic study of
recombinant human endostatin in patients with advanced solid
tumors. J Clin Oncol. 21:223–231. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li XP, Li CY, Li X, et al: Inhibition of
human nasopharyngeal carcinoma growth and metastasis in mice by
adenovirus-associated virus-mediated expression of human
endostatin. Mol Cancer Ther. 5:1290–1298. 2006. View Article : Google Scholar
|
|
23
|
Malecki M, Kolsut P and Proczka R:
Angiogenic and antiangiogenic gene therapy. Gene Ther. 12(Suppl 1):
S159–S169. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sauter BV, Martinet O, Zhang WJ, Mandeli J
and Woo SL: Adenovirus-mediated gene transfer of endostatin in
vivo results in high level of transgene expression and
inhibition of tumor growth and metastases. Proc Natl Acad Sci USA.
97:4802–4807. 2000.PubMed/NCBI
|
|
25
|
Mellon MJ, Ahn M, Jimenez JA, Kao C and
Gardner TA: Anti-angiogenic gene therapy for metastatic renal cell
carcinoma produces tumor growth suppression in an athymic nude
mouse model. J Urol. 179:737–742. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kerbel RS, Yu J, Tran J, et al: Possible
mechanisms of acquired resistance to anti-angiogenic drugs:
implications for the use of combination therapy approaches. Cancer
Metastasis Rev. 20:79–86. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Weidner N, Semple JP, Welch WR and Folkman
J: Tumor angiogenesis and metastasis - correlation in invasive
breast carcinoma. N Engl J Med. 324:1–8. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wu Y, Yang L, Hu B, et al: Synergistic
anti-tumor effect of recombinant human endostatin adenovirus
combined with gemcitabine. Anticancer Drugs. 16:551–557. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Folkman J: Tumor angiogenesis and tissue
factor. Nat Med. 2:167–168. 1996. View Article : Google Scholar
|
|
30
|
Bergers G, Javaherian K, Lo KM, Folkman J
and Hanahan D: Effects of angiogenesis inhibitors on multistage
carcinogenesis in mice. Science. 284:808–812. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Algire GH, Chalkley HW, Earle WE, et al:
Vascular reactions of normal and malignant tissues in vivo. III
Vascular reactions’ of mice to fibroblasts treated in vitro with
methylcholanthrene. J Natl Cancer Inst. 11:555–580. 1950.PubMed/NCBI
|
|
32
|
Folkman J, Merler E, Abernathy C and
Williams G: Isolation of a tumor factor responsible for
angiogenesis. J Exp Med. 133:275–288. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Risau W: Mechanisms of angiogenesis.
Nature. 386:671–674. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Carmeliet P: Mechanisms of angiogenesis
and arteriogenesis. Nat Med. 6:389–395. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Folkman J and Kalluri R: Cancer without
disease. Nature. 427:7872004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shi W, Teschendorf C, Muzyczka N and
Siemann DW: Gene therapy delivery of endostatin enhances the
treatment efficacy of radiation. Radiother Oncol. 66:1–9. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ehrmann RL and Knoth M: Choriocarcinoma.
Transfilter stimulation of vasoproliferation in the hamster cheek
pouch Studied by light and electron microscopy. J Natl Cancer Inst.
41:1329–1341. 1968.
|
|
38
|
Huang G and Chen L: Recombinant human
endostatin improves anti-tumor efficacy of paclitaxel by
normalizing tumor vasculature in Lewis lung carcinoma. J Cancer Res
Clin Oncol. 136:1201–1211. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wickstrom SA, Alitalo K and Keski-Oja J:
Endostatin associates with integrin α5β1 and
caveolin-1, and activates Src via a tyrosyl phosphatase-dependent
pathway in human endothelial cells. Cancer Res. 62:5580–5589.
2002.
|
|
40
|
Lan KL, Ou-Yang F, Yen SH, Shih HL and Lan
KH: Cationic liposome coupled endostatin gene for treatment of
peritoneal colon cancer. Clin Exp Metastasis. 27:307–318. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Boehm T, Pirie-Shepherd S, Trinh LB,
Shiloach J and Folkman J: Disruption of the KEX1 gene in Pichia
pastoris allows expression of full-length murine and human
endostatin. Yeast. 15:563–572. 1999.PubMed/NCBI
|
|
42
|
Griscelli F, Li H, Bennaceur-Griscelli A,
et al: Angiostatin gene transfer: inhibition of tumor growth in
vivo by blockage of endothelial cell proliferation associated
with a mitosis arrest. Proc Natl Acad Sci USA. 95:6367–6372.
1998.PubMed/NCBI
|
|
43
|
Lin P, Buxton JA, Acheson A, et al:
Antiangiogenic gene therapy targeting the endothelium-specific
receptor tyrosine kinase Tie2. Proc Natl Acad Sci USA.
95:8829–8834. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Regulier E, Paul S, Marigliano M, et al:
Adenovirus-mediated delivery of antiangiogenic genes as an
antitumor approach. Cancer Gene Ther. 8:45–54. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bai RZ, Wu Y, Liu Q, et al: Suppression of
lung cancer in murine model: treated by combination of recombinant
human endostsatin adenovirus with low-dose cisplatin. J Exp Clin
Cancer Res. 28:312009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Liao Z, Huang C, Zhou F, et al: Radiation
enhances suicide gene therapy in radioresistant laryngeal squamous
cell carcinoma via activation of a tumor-specific promoter. Cancer
Lett. 283:20–28. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Durand RE and LePard NE: Contribution of
transient blood flow to tumour hypoxia in mice. Acta Oncol.
34:317–323. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bussink J, Kaanders JH and van der Kogel
AJ: Tumor hypoxia at the micro-regional level: clinical relevance
and predictive value of exogenous and endogenous hypoxic cell
markers. Radiother Oncol. 67:3–15. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Cousins C and Sharp C: Medical
interventional procedures-reducing the radiation risks. Clin
Radiol. 59:468–473. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lee CG, Heijn M, di Tomaso E, et al:
Anti-vascular endothelial growth factor treatment augments tumor
radiation response under normoxic or hypoxic conditions. Cancer
Res. 60:5565–5570. 2000.PubMed/NCBI
|
|
51
|
Teicher BA, Holden SA, Ara G, et al:
Potentiation of cytotoxic cancer therapies by TNP-470 alone and
with other anti-angiogenic agents. Int J Cancer. 57:920–925. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jain RK: Normalizing tumor vasculature
with anti-angiogenic therapy: a new paradigm for combination
therapy. Nat Med. 7:987–989. 2001. View Article : Google Scholar : PubMed/NCBI
|