Introduction
Oligometastatic breast or recurrent gynecological
tumor (OBRGT) patients, if inoperable, have a poor prognosis
particularly when multiple therapeutic approaches have been already
attempted and further oncologic treatment may contribute to
unacceptable morbidity.
In the last few years, locoregional radiation
treatment of oligometastatic disease in gynecological and breast
cancer has become more prevalent (1–4).
In this context, radiotherapy should be ideally able
to deliver the entire dose necessary for the ablative intent in the
shortest delivery time. To date, this goal may be obtained by
combining two fundamental technological advances: stereotactic body
radiotherapy (SBRT), known as stereotactic body radiosurgery (SRS)
when administered in a single fraction (5,6) and
intensity modulated radiotherapy with volumetric arc technique
known with the acronym of VMAT.
Briefly, SBRT couples a high degree of anatomic
targeting accuracy and reproducibility with very high doses of
precisely delivered radiation, thereby maximizing the cell killing
effect on the target(s) while minimizing radiation-related injury
in adjacent normal tissues (7). Few
experiences have been reported concerning the use of stereotactic
techniques for administering an ablative dose in 3–5 fractions in
recurrent gynecological tumors (1–3,8–13).
Nevertheless, the available data allow consideration of this
technique as a promising palliative treatment strategy for OBRGT
due to the achievement of a >80% local control (LC) rate and a
low incidence of serious toxicity despite the high dose fractions
of administered radiation (1–3,10,12,13).
Moreover, VMAT, a novel technique characterized by
dynamic arc dose delivery (14),
administers radiation through a rotational movement of the linear
accelerator gantry, while a continuous variation in beam profile
and intensity is obtained. The expected advantages of this approach
are represented by increased delivery efficiency and reduced risk
of intra-fraction deviations both in terms of set-up errors and
organ motion.
Therefore, VMAT may represent a valuable technique
for SRS treatment; however, while little evidence exists concerning
the feasibility of SRS-VMAT in different clinical settings
(15), no data concerning SRS-VMAT
have been reported in the treatment of OBRGT.
In the context of our continuing efforts to improve
treatment efficacy in oligometastatic cancer patients (3,16), we
launched a prospective phase I clinical trial (DESTROY-2), aimed at
primarily defining the maximum tolerated dose (MTD) of SRS-VMAT in
patients with oligometastatic disease; secondary objectives are
represented by feasibility evaluation in terms of dose-volume
constraints, analysis of correlation between dosimetric and
toxicity data, clinical response and assessment of local control
rate (15).
Herein, we present the preliminary results of
feasibility, toxicity and efficacy of our SRS-VMAT experience in
OBRGT patients.
Materials and methods
This is a preliminary analysis of feasibility,
toxicity and clinical efficacy of SRS-VMAT administered to a cohort
of OBRGT patients enrolled in the DESTROY-2 study, as previously
reported (15).
Eligibility
The DESTROY-2 trial was approved by the local Ethics
Committee and the Institutional Review Board (P#988/CE/2010) and
all patients signed a written informed consent.
Patients who entered the present analysis were
selected among patients enrolled from August 2010 and March 2013
into the DESTROY-2 protocol, and inclusion criteria were: age
>18 years, diagnosis of breast and gynecologic cancer
recurrences not indicated for resection or other locally ablative
treatments, ECOG performance status ≤3, adequate bone marrow
function (neutrophil >1,500/mm3, platelets
>100,000/mm3), adequate renal function (blood urea
nitrogen <25 mg/dl, creatinine <1.5 mg/dl), normal liver
function (bilirubin <3 mg/dl).
Exclusion criteria were as follows: ECOG >3,
uncontrolled severe infection and/or medical problems unrelated to
malignancy, severe heart disease (if thorax site), diverticulitis
or ulcerative recto-colitis or pelvic inflammatory diseases (if
pelvic site). Patient should not have received SRS-VMAT prior to
clinical trial enrollment.
Simulation
Treatment set-up was performed with a CT simulator.
Women were treated in a head-first supine treatment position with
arms at their sides while lying on a customized body-frame
immobilization shell (Elekta Stereotactic Body-Frame or SBF; Elekta
Oncology Systems, Crawley, UK). To evaluate the reproducibility of
the set-up, three CT scan evaluations were performed on three
different days, aimed to verify set-up deviation <3 mm.
Moreover, to evaluate the organ motion, target displacement was
measured performing 30 free breathing axial CT scans on the same
slice. For displacement >5 mm, the SBF abdominal compressor was
applied and the CT scan for organ motion assessment was repeated.
The final CT simulation, for the acquisition of axial images
necessary for stereotactic localization and plan calculations, was
produced with a spiral technique. Three-millimeter scans were
acquired with a 3-mm interval between scans in the target region.
For treating abdominal or pelvic targets, patients received 2 cc of
oral Gastrografin, diluted in 500 cc water 30 min before CT scan.
In the case of mediastinal, abdominal or pelvic target volumes,
intravenous infusion of an iodinated contrast medium was also
used.
Target and normal tissue contouring
The contoured radiosurgical gross tumour volume
(GTV) consisted of identified cancer target(s), highlighted by CT
and/or 18F-FDG PET and/or MRI and agreed upon by both
the treating radiation oncologist and a gynecologic oncologist.
Each planning target volume (PTV) was individually defined for each
patient as follow: the internal margin was based on respiratory
excursions and the set-up margin was set at 3 mm according to the
ROSEL study (17). Nearby normal
tissue structures (OARs) according to the irradiated site were
contoured by the radiation oncologist or a certified medical
dosimetrist.
Treatment planning and dose delivery
SRS-VMAT treatment plans were optimized using the
ERGO++ treatment planning system with VMAT technique (Elekta). All
plans were generated with a single arc clockwise rotation,
described in the optimization process by a sequence of 86 control
points, i.e. one every 4°. The dose calculation was performed using
the pencil beam algorithm with inhomogeneity correction and a dose
grid resolution of 2 mm. A uniform method for the selection of the
prescription isodose surface (IDS) was used. According to the ROSEL
study (17), for each plan, the IDS
was selected as the greatest IDS fulfilling the two following
criteria: 95% of the PTV volume reached 100% of the prescription
dose and 99% of the PTV reached ≥90% of the prescription dose. The
maximum dose within the PTV should not exceed 140% of the
prescribed dose. Careful attention was paid to ensure that the
maximum dose always remained within the GTV according to OAR
constraints, previously reported in detail (15). SRS-VMAT plans were exported to the
record and verify (R&V) system, Mosaiq v. 1.6 (Impac Software;
Elekta) by DICOM-RT for later irradiation. Radiation prescription
doses ranged from 12 to 28 Gy according to different arms of the
dose escalation protocol as detailed elsewhere (15). All plans underwent dosimetric
verification by means of ion chamber array, using gamma-analyses
(18). For all patients, portal
images before irradiation were acquired on virtual orthogonal
beams. Deviations >3 mm in the isocenter position were
immediately corrected. For quality assurance through treatment
planning and delivery, two independent checks (IC1 and 2) were
performed by the medical and physics staff, as previously described
(19).
Supportive therapy
Patients receiving thorax irradiation were given
prescriptions for 0.5 mg oral betamethasone, 3 times/day for 1
month, followed by a gradual reduction, which are associated with
gastric protection (H2-inhibitors). Patients receiving abdominal
irradiation were prescribed 10 mg oral metoclopramide, 3 times/day,
for ≤1 week following radiation therapy and 40 mg oral rabeprazole,
daily for 12 months (in case of stomach and/or duodenum
irradiation). In addition, patients with upper abdomen lesions
received 3 mg intravenous granisetron plus 12 mg intravenous
dexamethasone immediately before radiosurgery and 6 h later.
Evaluation of response and follow-up. Tumor response
assessment was performed 8–12 weeks after treatment. Morphologic
imaging modalities were employed (CT with contrast medium and/or
MRI with or without contrast) in all patients. Tumour response was
based on the Response Evaluation Criteria in Solid Tumours (RECIST)
criteria (20). If feasible, the
response was also assessed by functional imaging, which included
(18F)-fluorodeoxyglucose (FDG)-PET. Herein, the European
Organisation for Research and Treatment of Cancer (EORTC) criteria
(21) were used as previously
detailed (15).
Follow-up was performed 2 weeks after SRS-VMAT to
evaluate acute toxicity, 3 months later to evaluate response by
abdominal CT and PET-CT and every 6 months thereafter.
Toxicity and quality of life (QoL)
evaluation
Adverse events were prospectively assessed. Grading
of toxicity was based on the Common Terminology Criteria for
Adverse Events (CTCAE V4.03), with the highest grade of any
observed toxicity reported for each patient (22). Several parameters were recorded to
evaluate the SRS-VMAT impact on pain, ECOG performance status,
weight and QoL by nursing staff. QoL indices were evaluated using
cancer linear analog scales, for well-being (CLAS1), fatigue
(CLAS2) and ability to perform daily activities (CLAS3),
respectively (23). The visual
analog scale (VAS) for pain (24),
the pain score (pain evaluation obtained by multiplying severity x
frequency) and the drugs score (analgesic assumption evaluation
obtained by multiplying severity x frequency) were used to record
and monitor pain (25).
Statistical analysis
Objective response rate (ORR) included complete and
partial response. Clinical benefit included ORR and stabilization
of disease. The 95% confidence intervals (95% CI) have been
provided.
Local control (LC) of irradiated lesions was
calculated using the Kaplan-Meier method (26) from the date of SRS-VMAT to the date
of the inside SRS-VMAT field relapse/progression of disease or the
date last seen. Metastasis-free survival (MFS) was calculated on a
per patient basis from the date of SRS-VMAT to the date of
relapse/progression of disease outside SRS-VMAT field or the date
last seen. Overall survival (OS) was calculated on a per patient
basis from the date of SRS-VMAT to the date of death or the date of
the last visit. Statistical analysis was carried out using SOLO
(BMDP Statistical Software, Los Angeles, CA, USA).
Results
Patient characteristics
Twenty-four patients meeting the inclusion criteria
and irradiated between August 2010 and March 2013 were analyzed.
Clinical and pathological characteristics of the patients are
summarized in Table I. The median
age was 63 years (range, 40–81) and the vast majority of patients
(N=18, 75.0%) had ECOG performance status <2. Patients were
considered as overweight/obese in 29.2% of cases. The primary tumor
was most frequently represented by breast carcinoma (75.0%),
followed by endometrial (8.3%) and cervical cancer (8.3%). Up to
two-thirds of patients suffer from comorbidities such as
hypertension, diabetes or vasculopathies. All patients were studied
for quality of life using CLAS score system, VAS and clinical
parameters as pain score, drug score. Seven patients (29.1%) had
been previously irradiated at the SRS-VMAT site and 3 (12.5%) had
already received >50 Gy to the SRS-VMAT site. Table II documents the details concerning
the SRS-VMAT sites, doses and treatment volume data. Doses were
according to the DESTROY-2 dose escalation protocol, as previously
detailed (15).
 | Table ICharacteristics of the entire study
population. |
Table I
Characteristics of the entire study
population.
| Characteristics | Patients n (%) | Target lesions n
(%) |
|---|
| Total | 24 (100) | 36 (100) |
| Age, years |
| Median
(range) | 63 (40–81) | |
| ECOG PS
statusa |
| 0 | 18 (75.0) | |
| 1 | 3 (12.5) | |
| 2 | 3 (12.5) | |
| Body mass index,
kg/m2 |
| 18.5–24.9 | 9 | |
| 25.0–28.9 | 8 | |
| 30–34.9 | 2 | |
| 25.0–39.9 | 1 | |
| ≥40.0 | 4 | |
| Primary tumor |
| Breast cancer | 18 (75.0) | 27 (75.0) |
| Endometrial
cancer | 2 (8.3) | 4 (11.1) |
| Cervical
cancer | 2 (8.3) | 3 (8.3) |
| Ovarian
cancer | 1 (4.2) | 1 (2.7) |
| Vulvar cancer | 1 (4.2) | 1 (2.7) |
| Histotype |
| Breast lobular
carcinoma | 3 (12.5) | 6 (16.7) |
| Breast ductal
carcinoma | 13 (54.1) | 18 (50.0) |
| Breast
ductal-lobular carcinoma | 2 (8.3) | 3 (8.4) |
| Endometrial clear
cell carcinoma | 2 (8.3) | 4 (11.2) |
| Squamous cervical
carcinoma | 1 (4.2) | 2 (5.6) |
| Cervical
adenocarcinoma | 1 (4.2) | 1 (2.7) |
| Serous ovarian
carcinoma | 1 (4.2) | 1 (2.7) |
| Vulvar
melanoma | 1 (4.2) | 1 (2.7) |
| Comorbidities |
| No | 8 (33.3) | |
| Yes | 16 (66.6) | |
| Pain score |
| 0 | 17 (70.8) | |
| ≥1 | 7 (29.1) | |
| Drug score |
| 0 | 21 (87.5) | |
| ≥1 | 3 (12.5) | |
| VAS |
| 0 | 17 (70.8) | |
| 1–5 | 5 (20.8) | |
| ≥5 | 2 (8.3) | |
| CLAS 1 |
| ≤5 | 8 (33.3) | |
| >5 | 16 (66.6) | |
| CLAS 2 |
| ≤5 | 8 (33.3) | |
| >5 | 16 (66.6) | |
| CLAS 3 |
| ≤5 | 3 (12.5) | |
| >5 | 21 (87.5) | |
| Previous
radiotherapy on SRS site | 7 (29.2) | 8 (22.2) |
| RT ≥50 Gy to SRS
site | 3 (12.5) | 3 (8.3) |
 | Table IIDetails concerning the SRS-treated
lesions (N=36). |
Table II
Details concerning the SRS-treated
lesions (N=36).
| SRS-treated
lesions | n (%) |
|---|
| Site |
| Lung | 5 (13.9) |
| Liver | 11 (30.5) |
| Vulva | 1 (2.8) |
| Lymph nodes | 6 (16.7) |
| Bone | 13 (36.1) |
| Region of
SRS-treated lesion |
| Thorax | 17 (47.2) |
| Abdomen | 11 (30.5) |
| Pelvis | 6 (16.7) |
| Other | 2 (5.6) |
| Dose (Gy) |
| 12 | 4 (11.1) |
| 16 | 8 (22.2) |
| 18 | 13 (36.1) |
| 20 | 4 (11.1) |
| 26 | 3 (8.3) |
| 28 | 4 (11.1) |
| GTV volume
(cc) |
| Median
(range) | 4.4 (0.1–42.3) |
| PTV volume
(cc) |
| Median
(range) | 19.4
(3.7–133.4) |
Fourteen patients received radiosurgery on a single
lesion, while 8 and 2 patients were irradiated on 2 and 3 different
metastatic sites, respectively, thus leading to a total number of
36 lesions including lung (13.9%), liver (30.5%), vulva (2.8%),
lymph node (16.7%) and bone (36.1%) (Table II).
Median dose was 18 Gy (BED2 Gy, α/β:
10=50.4 Gy), minimal dose was 12 Gy (BED2 Gy, α/β:
10=26.4 Gy) and maximal dose was 28 Gy (BED2 Gy, α/β:
10=106.4 Gy). Median SRS-VMAT GTV was 4.4 cc (range, 0.1–42.3
cc), while the median SRS-VMAT PTV ranged from 3.7 to 133.4 cc
(median, 19.4 cc). The dose/volume constraints (15) were respected in all lesions.
Safety
All patients received prescribed SRS-VMAT treatment
and were included in the safety analysis: 7 patients (29.2%)
experienced grade 1–2 acute toxicity, which however was grade 2 in
only 1 case. Moreover, only 3 patients (12.5%) developed grade 1–2
late toxicity of which only 1 was grade 2. Details concerning the
time of onset, type and severity of complications are provided in
Table III. Acute adverse events
included asymptomatic pneumonitis not requiring intervention (n=1),
grade 1 skin toxicity (n=2) and grade 2 mucositis (n=1) causing
discomfort, oedema and redness successfully treated with topic
medications. Mild pain worsening in the irradiated site (flare-up)
was reported in 3 (12.5%) patients. Regarding late toxicity, we
observed 1 case of grade 2 symptomatic pneumonitis out of 5
patients treated on the thorax, and this patient was treated by
oral steroids. Neither grade 3–4 toxicities nor treatment-related
deaths were reported.
 | Table IIIType of acute and late complications
according to organ system and grade (CTC-AE v.4.0 scale). |
Table III
Type of acute and late complications
according to organ system and grade (CTC-AE v.4.0 scale).
| Organ system
toxicity | n | Type |
|---|
| Acute toxicity
(N=7) |
| Lung | 1 | |
| G1 | 1 | Diagnostic
observation of asymptomatic pneumonitis: intervention not
indicated |
| Skin | 2 | |
| G1 | 2 | Faint erythema or
dry desquamation |
| Mucosal
tissue | 1 | |
| G2 | 1 | Vaginal
inflammation with mild discomfort, edema and redness |
| Other | 3 | |
| G1 | 3 | Flare-up: mild
worsening of the pain in the irradiated site |
| Late toxicity
(N=3) |
| Lung | 2 | |
| G1 | 1 | Diagnostic
observation of asymptomatic pneumonitis: intervention not
indicated |
| G2 | 1 | Symptomatic
pneumonitis: medical intervention indicated; limited instrumental
activities of daily living |
| Liver | 1 | |
| G1 | 1 | Diagnostic
observation of hepatobiliary disorders: intervention not
indicated |
Efficacy
All 36 irradiated lesions were evaluated for best
response (Table IV). The ORR of
target lesions to SRS-VMAT was 77.7% including 16 lesions achieving
complete response (44.4%) and 12 lesions achieving partial response
(33.3%). Stabilization of disease (SD) was observed in 7 lesions
(19.4%), while progression inside SRS-VMAT field was documented in
only 1 lesion (2.8%). The rate of clinical benefit (ORR + SD) was
97.2%.
 | Table IVResponse to SRS-VMAT on a per lesion
basis. |
Table IV
Response to SRS-VMAT on a per lesion
basis.
| Clinical
response | Target lesions n
(%) | 95% CI |
|---|
| Complete
response | 16 (44.4) | 28.2–60.6 |
| Partial
response | 12 (33.3) | 17.9–48.7 |
| Objective
response | 28 (77.7) | 63.7–91.7 |
| Stable disease | 7 (19.4) | 6.5–32.3 |
| Clinical
benefit | 35 (97.2) | 91.8–102.6 |
| Progressive
disease | 1 (2.8) | −2.6–8.2 |
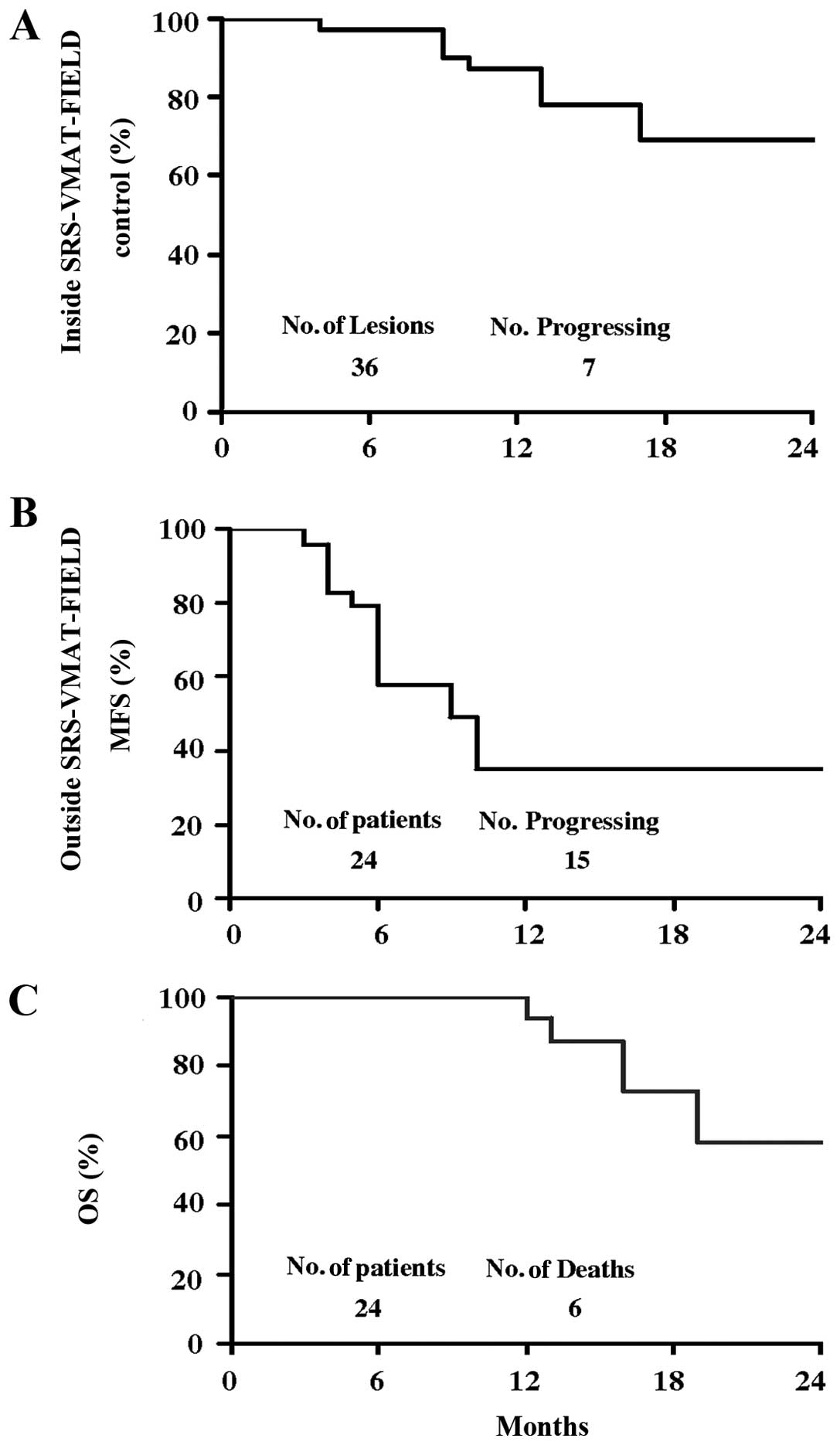
As of October 2013, the median duration of follow-up
was 15.5 months (range, 6–50). Recurrence/progression within the
SRS-VMAT-treated field was observed in 6 patients (total lesions,
7). As shown in Fig. 1, the 2-year
inside SRS-VMAT field disease control expressed on a per lesion
basis, was 69%.
Recurrence/progression of disease outside the
SRS-VMAT field was documented in 15 patients; the 2-year outside
SRS-VMAT field progression-free survival (PFS), expressed on a per
patient basis, was 35%. Death due to disease was documented in 6
patients and the 2-year OS was 58%.
Discussion
To the best of our knowledge, this series represents
the first report on the toxicity and activity of SRS-VMAT in breast
and gynecologic cancer recurrences not indicated for resection or
other locally ablative treatments. Despite sample heterogeneity in
terms of doses and histotypes, we found that the single fraction
irradiation approach was feasible and tolerable with encouraging,
although preliminary, clinical results. In particular, despite the
ablative doses delivered (median dose = 18 Gy; BED2 Gy, α/β:
10=50.4 Gy), only 1 case of acute grade 2 toxicity and 1 case
of late grade 2 toxicity were observed; these results have to be
taken into account considering that almost one-third of our cases
had already been irradiated at the SRS-VMAT site. Retrospective
studies employing SBRT for treatment of recurrent gynecological
tumors have been reported in two literature reviews (1,27);
doses ranging between 14 Gy (2 fractions) and 45 Gy (3 fractions)
were delivered with an acceptable safety profile with the exception
of cases already submitted to previous treatments. In particular,
Guckenberger et al (2), who
delivered SBRT (15 Gy with 3 fractions by means of the IMRT
technique) to oligometastatic gynecological patients already
submitted to surgery and/or radiation, reported >G2 late
toxicity in almost 25% of cases with 2 patients developing severe
adverse events (1 grade 4 entero-vaginal fistula and 1 grade 4
small bowel ileus). Although the authors recognized that this rate
of toxicity was similar to those reported with non-3D image guided
brachytherapy ± external beam radiotherapy (28–30),
they also advocated the use of risk-adapted protocols, in which
different SBRT fraction numbers with different single fraction
doses are used depending on the proximity of targets and OARs, as
for pulmonary SBRT in case of centrally located lung cancer
(31).
Concerning breast cancer oligometastatic patients,
systemic therapy or surgery, if feasible, are usually the main
treatment for these patients. However, recently, Habermehl et
al reported the safety of single-dose radiosurgical treatment
for hepatic metastases, with 6 and 12 months local control of 87
and 70% (4). Considering the rapid
increase in the scientific evidence in the relatively new field of
stereotactic radiotherapy, we strongly believe that this technique
in the next few years will be an integral part of the weapons
available for the treatment of oligometastasized breast cancer.
In regards to the preliminary evaluation of
treatment efficacy, the rate of ORR of the target lesions was 77.7%
(complete response: 44.4%); moreover, stabilization of tumor
lesions was ~97%, respectively; these findings are in line with
previously reported data obtained in similar settings with SBRT,
which has provided response rates between 67 and 79% despite the
use of various ablative doses and schedules (1,3,4,9,10,13).
Moreover, we reported a 2-year inside SRS-VMAT field local control
rate of 69%, when expressed on a per lesion basis.
Local control of SBRT-treated oligometastatic
patients has been shown to range between 71 and 96% with 1–2 year
follow up (32–35); explanations for this wide range of
efficacy could be represented by the delivery of different doses
and inclusion of various tumor types and histologies (1,2,12,13).
Moreover, the dose-finding design of our series as well as the
choice to express the local control data on a per lesion basis must
be taken into account when considering the comparison of our
results with previous experiences.
As expected, the 2-year outside SRS-VMAT field-PFS
and the 2-year OS were relatively dismal (35 and 58%,
respectively); as argued by Kunos et al (10) progression of disease elsewhere in
the body shortly after SBRT may signal either progression of
already present occult disease, or inability of SBRT to control
targeted disease prior to disease dissemination, circumstances
which both claim for concurrent chemotherapy administration. The
low toxicity profile of SRS-VMAT technique and its fast
administration makes this approach particularly suitable to be
administered between one chemotherapy cycle and the other, probably
resulting in a more effective, comprehensive approach to the
oligometastatic setting. In this context, a phase I clinical trial
of SBRT plus gemcitabine and carboplatin chemotherapy is underway
(ClinicalTrials. gov identifier: NCT01652794). In addition to the
higher acceptance and compliance to treatment of these vulnerable
patients already faced with distressing experiences with several
previous therapies, other potential advantages of SRS-VMAT must
also be emphasized, such as lack of inter-fraction uncertainties
that translate into a higher treatment reproducibility and reduced
costs in spite of its complexity.
In conclusion, clinical radiation practice with
SRS-VMAT for metastatic or previously irradiated gynecologic
cancers appears promising and is likely to translate into a greater
benefit for female cancer patient survival, although results from
phase II trials will better assess tumor response.
References
|
1
|
Higginson DS, Morris DE, Jones EL, et al:
Stereotactic body radiotherapy (SBRT): technological innovation and
application in gynecologic oncology. Gynecol Oncol. 120:404–412.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Guckenberger M, Bachmann J, Wulf J, et al:
Stereotactic body radiotherapy for local boost irradiation in
unfavourable locally recurrent gynaecological cancer. Radiother
Oncol. 94:53–59. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Deodato F, Macchia G, Grimaldi L, et al:
Stereotactic radiotherapy in recurrent gynecological cancer: A case
series. Oncol Rep. 22:415–419. 2009.PubMed/NCBI
|
|
4
|
Habermehl D, Herfarth KK, Bermejo JL, et
al: Single-dose radiosurgical treatment for hepatic metastases -
therapeutic outcome of 138 treated lesions from a single
institution. Radiat Oncol. 8:1752013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Seung SK, Larson DA, Galvin JM, et al:
American College of Radiology (ACR) and American Society for
Radiation Oncology (ASTRO) practice guideline for the performance
of stereotactic radiosurgery (SRS). Am J Clin Oncol. 36:310–315.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Solberg TD, Balter JM, Benedict SH, et al:
Quality and safety considerations in stereotactic radiosurgery and
stereotactic body radiation therapy: executive summary. Pract
Radiat Oncol. 2:2–9. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Potters L, Kavanagh B, Galvin JM, et al:
American Society for Therapeutic Radiology and Oncology (Astro) and
American College of Radiology (ACR) practice guideline for the
performance of stereotactic body radiation therapy. Int J Radiat
Oncol Biol Phys. 76:326–332. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Molla M, Escude L, Nouet P, et al:
Fractionated stereotactic radiotherapy boost for gynecologic
tumors: an alternative to brachytherapy? Int J Radiat Oncol Biol
Phys. 62:118–124. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kunos C, Chen W, DeBernardo R, et al:
Stereotactic body radiosurgery for pelvic relapse of gynecologic
malignancies. Technol Cancer Res Treat. 8:393–400. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kunos CA, Brindle J, Waggoner S, et al:
Phase II clinical trial of robotic stereotactic body radiosurgery
for metastatic gynecologic malignancies. Front Oncol. 2:1812012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kunos CA and Spelic M: Role of
stereotactic radiosurgery in gynecologic cancer. Curr Opin Oncol.
25:532–538. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jorcano S, Molla M, Escude L, et al:
Hypofractionated extracranial stereotactic radiotherapy boost for
gynecologic tumors: a promising alternative to high-dose rate
brachytherapy. Technol Cancer Res Treat. 9:509–514. 2010.
View Article : Google Scholar
|
|
13
|
Choi CW, Cho CK, Yoo SY, et al:
Image-guided stereotactic body radiation therapy in patients with
isolated para-aortic lymph node metastases from uterine cervical
and corpus cancer. Int J Radiat Oncol Biol Phys. 74:147–153. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Otto K: Volumetric modulated arc therapy:
IMRT in a single gantry arc. Med Phys. 35:310–317. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Deodato F, Cilla S, Macchia G, et al:
Extracranial radiosurgery with volumetric modulated arc therapy:
Feasibility evaluation of a phase I trial. Oncol Lett. 5:1889–1896.
2013.PubMed/NCBI
|
|
16
|
Macchia G, Morganti AG, Cilla S, et al:
Quality of life and toxicity of stereotactic radiotherapy in
pancreatic tumors: a case series. Cancer Invest. 30:149–155. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hurkmans CW, Cuijpers JP, Lagerwaard FJ,
et al: Recommendations for implementing stereotactic radiotherapy
in peripheral stage IA non-small cell lung cancer: report from the
quality assurance working party of the randomised phase III ROSEL
study. Radiat Oncol. 4:12009. View Article : Google Scholar
|
|
18
|
Cilla S, Viola P, Azario L, et al:
Comparison of measured and computer portal dose for IMRT treatment.
J Appl Clin Med Phys. 7:65–79. 2006.PubMed/NCBI
|
|
19
|
Morganti AG, Deodato F, Zizzari S, et al:
Complexity index (COMIX) and not type of treatment predicts
undetected errors in radiotherapy planning and delivery. Radiother
Oncol. 89:320–329. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors. European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Young H, Baum R, Cremerius U, et al:
Measurement of clinical and subclinical response using
[18F]-fluorodeoxiglucose and positron emission tomography: review
and 1999 EORTC recommendations. European Organisation for Research
and Treatment of Cancer (EORTC) PET study group. Eur J Cancer.
35:1773–1782. 1999.
|
|
22
|
National Institutes of Health National
Cancer Institute. Common Terminology Criteria for Adverse Events
(CTCAE), version 4.03. Published on February 22, 2012.
|
|
23
|
Sutherland HJ, Walker P and Till JE: The
development of a method for determining oncology patients’
emotional distress using linear analogue scales. Cancer Nurs.
11:303–308. 1988.
|
|
24
|
Melzack R: The McGill pain questionnaire:
major properties and scoring methods. Pain. 1:277–299. 1975.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Salazar OM, Sandhu T, da Motta NW, et al:
Fractionated half-body irradiation (HBI) for the rapid palliation
of widespread, symptomatic, metastatic bone disease: a randomized
phase III trial of the International Atomic Energy Agency (IAEA).
Int J Radiat Oncol Biol Phys. 50:765–775. 2001. View Article : Google Scholar
|
|
26
|
Kaplan EL and Meier P: Non-parametric
estimation from incomplete observations. J Am Statist Assoc.
53:457–481. 1985. View Article : Google Scholar
|
|
27
|
Mayr NA, Huang Z, Sohn JW, et al: Emerging
application of stereotactic body radiation therapy for gynecologic
malignancies. Expert Rev Anticancer Ther. 11:1069–1075.
2011.PubMed/NCBI
|
|
28
|
Grigsby PW: Radiotherapy for pelvic
recurrence after radical hysterectomy for cervical cancer. Radiat
Med. 23:327–330. 2005.PubMed/NCBI
|
|
29
|
Haasbeek CJ, Uitterhoeve AL, van der
Velden J, et al: Long-term results of salvage radiotherapy for the
treatment of recurrent cervical carcinoma after prior surgery.
Radiother Oncol. 89:197–204. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ito H, Shigematsu N, Kawada T, et al:
Radiotherapy for centrally recurrent cervical cancer of the vaginal
stump following hysterectomy. Gynecol Oncol. 67:154–161. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Guckenberger M, Wulf J, Mueller G, et al:
Dose-response relationship for image-guided stereotactic body
radiotherapy of pulmonary tumors: relevance of 4D dose calculation.
Int J Radiat Oncol Biol Phys. 74:47–54. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rusthoven KE, Kavanagh BD, Cardenes H, et
al: Multi-institutional phase I/II trial of stereotactic body
radiation therapy for liver metastases. J Clin Oncol. 27:1572–1578.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rusthoven KE, Kavanagh BD, Burri SH, et
al: Multi-institutional phase I/II trial of stereotactic body
radiation therapy for lung metastases. J Clin Oncol. 27:1579–1584.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mendez Romero A, Wunderink W, Hussain SM,
et al: Stereotactic body radiation therapy for primary and
metastatic liver tumors: a single institution phase i–ii study.
Acta Oncol. 45:831–837. 2006.
|
|
35
|
Herfarth KK, Debus J, Lohr F, et al:
Stereotactic single-dose radiation therapy of liver tumors: results
of a phase I/II trial. J Clin Oncol. 19:164–170. 2001.PubMed/NCBI
|