Introduction
Hepatocellular carcinoma (HCC) represents the fifth
most common malignancy and the third most common cause of
cancer-related death worldwide (1).
Hepatic resection is the main treatment option for HCC patients
(2–4). However, even after curative resection,
80% of these patients develop intrahepatic recurrence, and 50% die
within 5 years (5). Intrahepatic
metastasis (IM) refers to HCC foci developing from tumor cells that
spread into the remnant liver via the portal vein before or during
hepatic resection. Multicentric occurrence (MO) refers to new HCC
foci developing due to the presence of chronic active hepatitis,
cirrhosis, or other HCC-relevant risk factors after resection
(6–9). Previous studies have indicated that
the clinical progression and outcomes of these two recurrence types
differ significantly (8–10). Therefore, identification of such
differences is important for both the determination of therapeutic
strategies and prognosis prediction.
Several studies have utilized genetic background
analysis of both recurrent and primary tumors to differentiate
between IM and MO (11,12). The progression and outcome of truly
relapsed HCC are distinct from those of a second primary tumor,
thus clonal analysis of initial and recurrent HCC is of clinical
significance. We previously reported clonal analysis data from the
investigation of recurrent HCC genotypes by detecting mutations of
the mitochondrial genome (13) or
examining patterns of promoter hypermethylation in several
tumor-suppressor genes in tumor cells (14). Our findings from the above-mentioned
studies suggested the distinction between MO and IM.
Although early HCC may be cured by surgical
resection, the major issue of this fatal disease is its tendency to
develop MO (11). Many researchers
have attempted to predict the prognosis of HCC patients by
evaluating only risks associated with the resected tumor tissue.
However, in consideration of the unique MO pattern in HCC, a mere
focus on the tumor tissue would be insufficient. We hypothesized
that molecular changes in the background normal liver tissue
surrounding HCC may also influence patient prognosis. Thus, the
present study was designed to identify a unique biological marker,
especially from those present in the background normal liver tissue
of HCC, with outcome predictive values.
Materials and methods
Sample collection and DNA preparation for
microarray analyses
Control samples, termed supernormal (SN) liver, were
obtained from the normal tissues of 11 patients with metastatic
liver cancer who underwent liver resection at our institution.
Their primary diseases were colorectal cancer (n=5),
gastrointestinal stromal tumor (n=2), and one each of gastric
cancer, esophageal cancer, cervical cancer and tongue cancer. For
comparison, non-neoplastic liver tissue was obtained from a typical
HCC case that resulted from chronic hepatitis C [the corresponding
normal (CN) sample] at the time of primary liver resection. This
patient was a 58-year-old man with liver cirrhosis who had
undergone liver resection but experienced recurrences 3 years after
the primary lesion resection. DNA and RNA were extracted from the
SN and CN tissues after appropriate pathological confirmation that
the CN sample did not contain any cancerous regions. Expression
profiling and methylation array were performed to compare the SN
and CN samples and identify genes with differential expression and
methylation rate.
Microarray procedure
Total RNA from the 11 SN samples were mixed to
eliminate individual background, then labeled with cyanine-3 dye
using a Quick Amp Labeling kit (Agilent Technologies, Mississauga,
ON, Canada), and hybridized to Agilent Whole Human Genome (4×44K)
microarrays for 17 h in a rotating SciGene model 700 oven (SciGene,
Sunnyvale, CA, USA). The arrays were scanned using an Agilent
Technologies DNA Microarray Scanner (Agilent Technologies). Data
were feature-extracted using Feature Extraction Software 10.5.1.1
and statistically analyzed using the default settings on GeneSpring
GX 11.0.1 software (both from Agilent Technologies) (15). Methylation array was performed
according to the Illumina Infinium HumanMethylation27 BeadChip
(Illumina, San Diego, CA, USA) protocol, requiring 500 ng to 1 μg
of bisulfite-converted DNA (16).
Analysis of methylation array data was performed as previously
described (17,18).
HCC cases for real-time quantitative
reverse transcription-polymerase chain reaction analysis
Primary HCC and surrounding non-cancerous tissues
were collected from 100 patients who underwent liver resection at
Nagoya University Hospital between 1994 and 2002. The patient age
ranged from 21 to 78 years (mean, 62.2±9.8 years), and the
male-to-female ratio was 86:14. The median follow-up duration was
65.43 months (range, 0.3–208.9 months). Patient characteristics are
summarized in Table I. All tissue
samples were histologically confirmed as HCC. This study was
approved by our institutional review board, and all patients
provided written informed consent. Patient prognosis was predicted
for all cases according to gene expression results by reverse
transcription-polymerase chain reaction (RT-qPCR).
 | Table IHCC patient characteristics
(n=100). |
Table I
HCC patient characteristics
(n=100).
| Patient
characteristics | Values |
|---|
| Age (years), mean ±
SD (range) | 62.2±9.8
(21–78) |
| Gender (n) |
| Male | 86 |
| Female | 14 |
| Viral infection
(n) |
| HBV | 18 |
| HCV | 75 |
| Non-HBV/HCV | 7 |
| Child-Pugh
classification (n) |
| A | 87 |
| B | 8 |
| Liver damage
classification (n) |
| A | 62 |
| B | 28 |
| Albumin (mg/dl),
mean ± SD | 3.91±0.38 |
| Total bilirubin
(mg/dl), mean ± SD | 0.82±0.33 |
| PT (%), mean ±
SD | 75.9±12.8 |
| AFP (ng/ml), mean ±
SD | 3,069±13,841.8 |
| Tumor size (cm),
mean ± SD (range) | 4.47±3.08
(0.8–15) |
| Tumor multiplicity
(n) |
| Solitary | 66 |
| Multiple | 30 |
| ICG-R15, mean ±
SD | 12.49±9.82 |
| Japanese stage
(n) |
| I | 9 |
| I | 47 |
| III | 21 |
| IV | 15 |
DNA preparation and RNA isolation from
tissues
Surgically obtained tissue samples were immediately
frozen in liquid nitrogen and stored at −80°C until analysis.
Genomic DNA was purified from the tissue samples by proteinase K
digestion, followed by phenol/chloroform extraction. Total RNA was
extracted from the SN and CN samples using a Qiagen miRNeasy Mini
kit (Qiagen, Toronto, ON, Canada). RNA quality was confirmed with
an RNA integrity number of ≥8 measured by an Agilent 2100
Bioanalyzer (Agilent Technologies).
RT-qPCR
The absolute quantification method was used to
determine the input copy number by relating the PCR signal to a
standard curve owing to its advantages in studies with a large
number of samples. PCR was performed using a SYBR-Green PCR Core
Reagent kit (Perkin-Elmer Applied Biosystems, Foster City, CA, USA)
under the following conditions: 50°C for 2 min, 95°C for 10 min,
and 45 cycles of 95°C for 15 sec and 60°C for 30 sec. SYBR-Green
signal was detected in real-time using an ABI Prism 7000 Sequence
Detector (Perkin-Elmer Applied Biosystems). The PCR primers used
were for a 93-bp fragment of Janus kinase 2 (JAK2) (sense,
5′-ACCTCCAGCCGTGCTTGA-3′ in exon 11; antisense,
5′-TGATTACCTGCTTTCTTCAGTTTACTAATG-3′ in exon 12). GAPDH
expression was quantified in each sample for standardization
purposes. All RT-qPCR experiments were performed at least three
times, including negative controls without a template. JAK2
expression was determined as the value of JAK2
×103/GAPDH in each sample.
Statistical analysis
Continuous variables are expressed as median (range)
and were compared using the Mann-Whitney U-test. Categorical
variables were compared using the χ2 or Fisher’s exact
test, as appropriate. Overall survival rates were estimated by the
Kaplan-Meier method and compared using the log-rank test. All
statistical analyses were performed using JMP Pro software version
11.0.0 (SAS Institute Inc., Cary, NC, USA). The level of
statistical significance was set at P<0.05.
Results
Expression profiling and methylation
array analysis
To identify novel tumor-related genes in the
background normal liver tissue, we focused on genes with an
increased expression in CN tissues compared with the corresponding
SN tissue expression. Array analysis revealed that JAK2,
which is involved in the JAK/STAT pathway, was strongly upregulated
by 2.378-fold (log2 ratio) in the CN tissues (Table IIA).
 | Table IIExpression profile and methylation
array. |
Table II
Expression profile and methylation
array.
| A, Expression
profile |
|---|
|
|---|
| Gene symbol | Fold-change CN vs.
SN | Regulation CN vs.
SN | Raw CN | Raw SN | Flags CN | Flags SN |
|---|
| JAK2 | 2.378 | Upregulated | 220.579 | 76.974 | Detected | Detected |
|
| B, Methylation
array |
|
| Chromosome | Gene symbol | SN value | CN value | | | |
|
| 9 | JAK2 | 0.748964 | 0.125348 | | | |
Methylation array analysis showed continuous β
values of 0.125 for CN vs. 0.748 for SN tissues, indicating a high
degree of demethylation in the CN sample of the hepatitis C virus
(HCV)-positive case (Table IIB).
The result was of particular interest as it may imply that a
possible mechanism of JAK2 upregulation was demethylation of
its promoter region in the HCV normal tissue.
RT-qPCR analysis of SN and background
normal tissues from 100 HCC cases
However, when 100 HCC cases were analyzed, the
overall JAK2 expression (relative to GAPDH) was
significantly lower in the adjacent normal than in the SN tissues
(median ± SD of JAK2 ×103/GAPDH ratio,
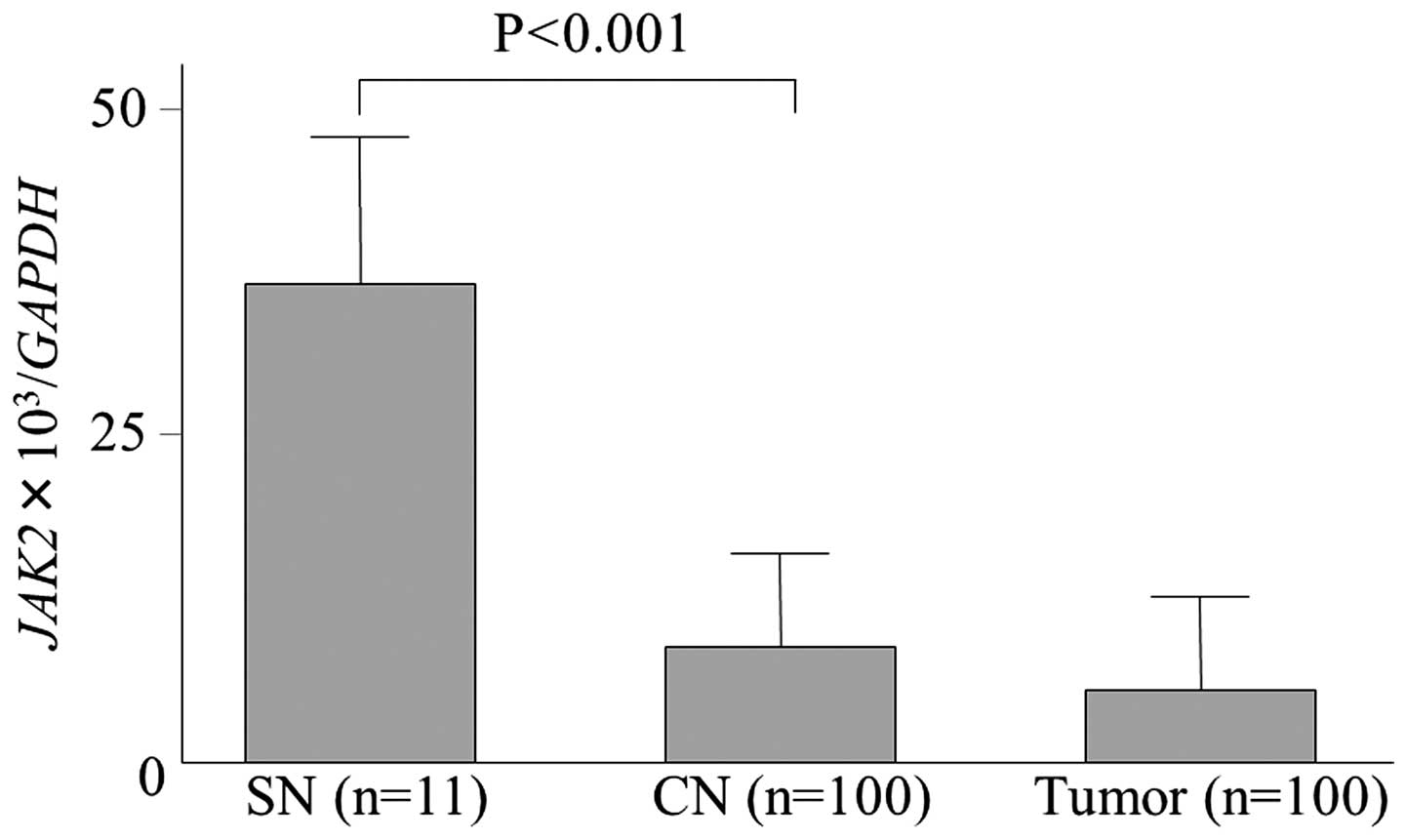
9.24±6.43 vs. 35.21±21.38; P<0.001) (Fig. 1). In addition, JAK2
expression in the tumor tissue was 5.21±5.71. Thus, the HCC case
examined by array analysis was thought to be a rare incidence of
upregulated and demethylated JAK2. As a result of the
unexpected findings, we decided not to investigate the methylation
status of JAK2 in the CN tissues and instead focus on the
clinical relevance of JAK2 expression by evaluating its
relationship with HCC prognosis. First, as we investigated
JAK2 expression in the tumor tissue, the HCC cases were
divided into two groups: one with higher JAK2 tumor tissue
expression than the average value (n=28) and the other with
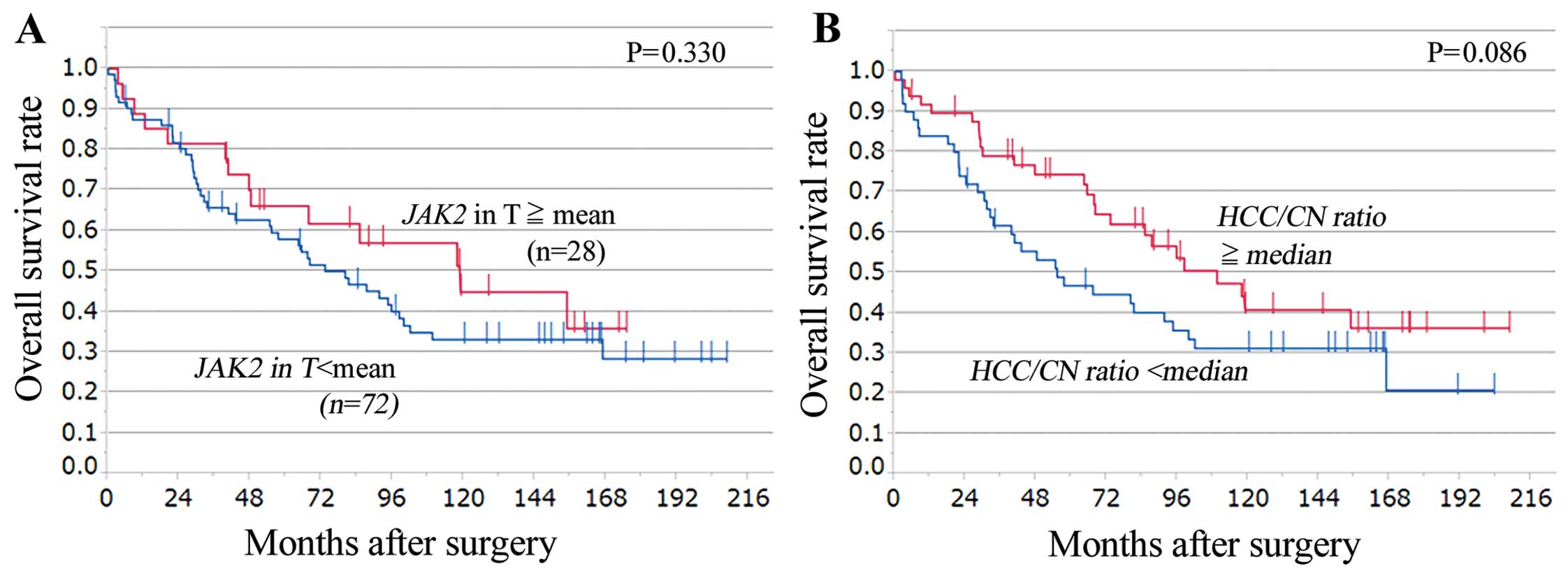
lower-than-average JAK2 tumor expression (n=72). No
significant differences in overall survival were observed between
the two groups (Fig. 2A).
Subsequently, the ratio of JAK2 expression in the tumor
tissue and that in the normal sample (HCC/CN) was calculated for
each case to evaluate the risk of non-cancerous tissue. The group
with a higher than the median HCC/CN ratio (n=50) tended to have
longer overall survival than the one with lower than the median
ratio (n=50), but the differences were not statistically
significant (P=0.086) (Fig. 2B).
Based on these findings, JAK2 expression in the background
normal tissue of HCC, but not in the tumor tissue itself, appeared
to relate to the overall survival of HCC patients. We then sorted
these HCC cases according to JAK2 expression and divided
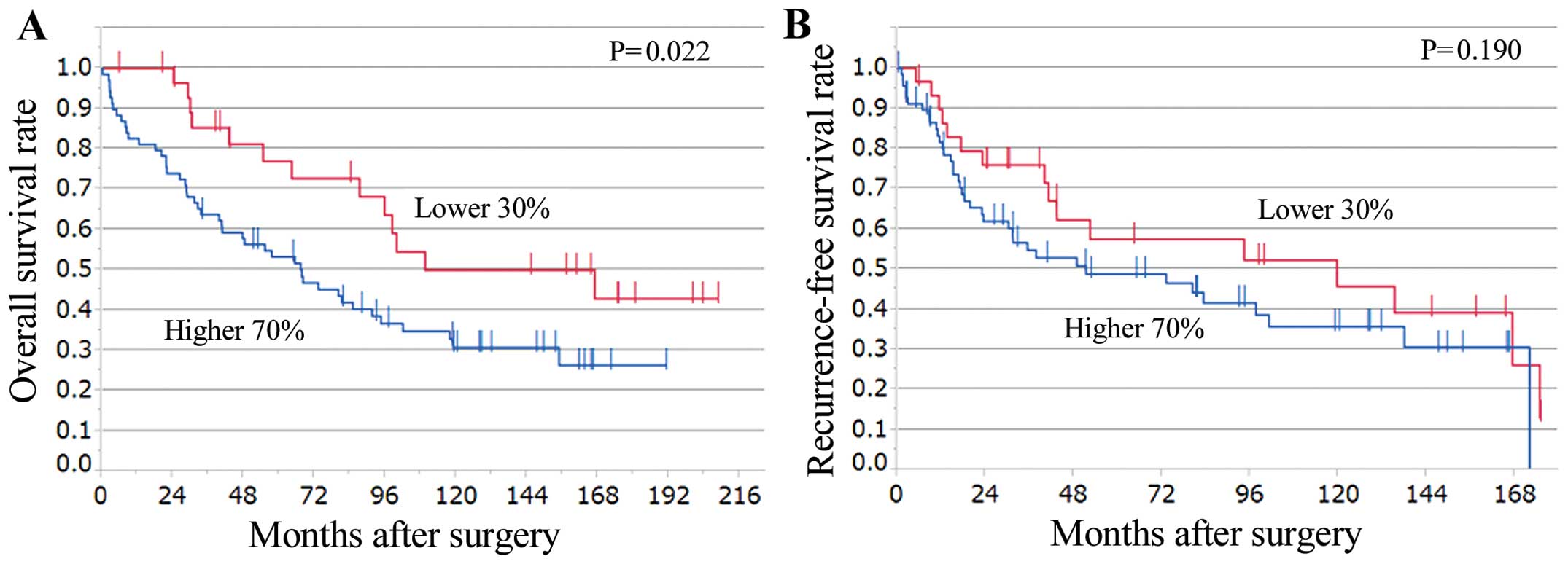
them into two groups: the higher 70% group, which consisted of the
upper 70% of cases (n=70), and the lower 30% group containing the
remaining 30% of cases (n=30). The higher 70% group showed
significantly worse overall survival than the lower 30% group
(P=0.022) (Fig. 3A). Additionally,
the higher 70% group tended to have worse recurrence-free survival
than the lower 30% group, but the differences were not
statistically significant (Fig.
3B).
Correlation between upregulated JAK2
expression in background normal tissue and clinicopathological
characteristics of HCC
JAK2 expression was significantly different
between well/moderately and poorly differentiated tissues (8.86 vs.
13.25; P=0.014) (Table III).
 | Table IIIClinicopathological findings of 100
HCC patients according to JAK2 expression. |
Table III
Clinicopathological findings of 100
HCC patients according to JAK2 expression.
| Adjacent normal
tissue |
|---|
|
|
|---|
| Clinicopathological
factor | N | Expression
value | P-value |
|---|
| Age (years) |
| ≥65 | 52 | 9.15 | 0.518 |
| <65 | 48 | 9.32 | |
| Gender |
| Male | 84 | 9.11 | 0.316 |
| Female | 16 | 9.85 | |
| HCV |
| + | 73 | 9.93 | 0.077 |
| − | 25 | 7.48 | |
| HBV |
| + | 18 | 7.46 | 0.184 |
| − | 80 | 9.72 | |
| Liver
cirrhosis |
| + | 45 | 9.30 | 0.658 |
| − | 48 | 9.41 | |
| Tumor size |
| ≥5 cm | 35 | 10.36 | 0.434 |
| <5 cm | 59 | 8.64 | |
| Tumor
multiplicity |
| Solitary | 66 | 9.35 | 0.399 |
| Multiple | 30 | 9.19 | |
| Septal
formation |
| + | 74 | 8.92 | 0.165 |
| − | 26 | 10.12 | |
| Growth form |
| Expansive | 79 | 9.19 | 0.123 |
| Infiltrative | 14 | 10.33 | |
| Vascular
invasion |
| + | 25 | 9.67 | 0.383 |
| − | 68 | 9.13 | |
| Differentiation
1 |
| Well | 16 | 8.26 | 0.873 |
| Others | 76 | 9.33 | |
| Differentiation
2 |
| Well,
moderately | 86 | 8.86 | 0.014 |
| Poorly | 6 | 13.25 | |
| AFP |
| ≥20 ng/ml | 57 | 9.25 | 0.952 |
| <20 ng/ml | 39 | 9.17 | |
| Japanese stage |
| I, II | 58 | 9.65 | 0.717 |
| II, IV | 37 | 8.73 | |
Univariate analysis revealed significant correlation
of overall survival with liver cirrhosis (P=0.018), vascular
invasion (P=0.001), α-fetoprotein levels of >20 ng/ml (P=0.038),
and higher JAK2 expression (>70 vs. <30%, P<0.017)
(Table IV).
 | Table IVUnivariate and multivariate analyses
of overall survival. |
Table IV
Univariate and multivariate analyses
of overall survival.
| | Univariate
analysis | Multivariate
analysis |
|---|
| |
|
|
|---|
| Clinicopathological
factor | N | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age (years) |
| ≥65 | 52 | 1 | | | | | |
| <65 | 48 | 1.17 | 0.706–1.98 | 0.529 | | | |
| Gender |
| Male | 84 | 1 | | | | | |
| Female | 16 | 1.1 | 0.543–2.04 | 0.77 | | | |
| HCV |
| + | 73 | 1.88 | 0.997–3.96 | 0.051 | | | |
| − | 25 | 1 | | | | | |
| HBV |
| + | 18 | 1 | | | | | |
| − | 80 | 1.7 | 0.827–4.12 | 0.157 | | | |
| Liver
cirrhosis |
| + | 45 | 1.88 | 1.11–3.20 | 0.018 | 2.09 | 1.18–3.71 | 0.01 |
| − | 48 | 1 | | | | | |
| Tumor size |
| ≥5 cm | 35 | 1.28 | 0.745–2.17 | 0.359 | | | |
| <5 cm | 59 | 1 | | | | | |
| Tumor
multiplicity |
| Solitary | 66 | 1 | | | | | |
| Multiple | 30 | 1.44 | 0.828–2.44 | 0.19 | | | |
| Septal
formation |
| + | 74 | 1 | | | | | |
| − | 26 | 1.08 | 0.574–1.93 | 0.786 | | | |
| Growth form |
| Expansive | 79 | 1 | | | | | |
| Infiltrative | 14 | 1.38 | 0.633–2.69 | 0.39 | | | |
| Vascular
invasion |
| + | 25 | 2.63 | 1.49–4.51 | 0.001 | 3.1 | 1.70–5.51 | 0.0003 |
| − | 68 | 1 | | | | | |
| Differentiation
1 |
| Well | 16 | 1 | | | | | |
| Others | 76 | 1.98 | 0.921–5.17 | 0.082 | | | |
| Differentiation
2 |
| Well,
moderately | 86 | 1 | | | | | |
| Poorly | 6 | 1.71 | 0.594–3.89 | 0.286 | | | |
| AFP |
| ≥20 ng/ml | 57 | 1.76 | 1.02–3.12 | 0.038 | 1.5 | 0.860–2.72 | 0.152 |
| <20 ng/ml | 39 | 1 | | | | | |
| Japanese stage |
| I, II | 58 | 1 | | | | | |
| II, IV | 37 | 1.62 | 0.961–2.72 | 0.069 | | | |
| JAK2
expression in CN |
| Higher 70% | 70 | 2.04 | 1.12–3.96 | 0.017 | 1.91 | 1.04–3.74 | 0.032 |
| Lower 30% | 30 | 1 | | | | | |
Furthermore, multivariate analysis confirmed
significant correlation of overall survival with liver cirrhosis
(P=0.01), vascular invasion (P=0.0003), and higher JAK2
expression in the background normal liver tissue of the HCC cases
(P=0.032) (Table IV).
Discussion
A major obstacle for HCC treatment is the high
frequency of tumor recurrence even after curative resection and
liver transplantation (19). In
cases of small and well-differentiated tumors, the recurrence rate
remains high (20). We previously
reported that MO was more common than IM in HCC (13,14).
Accordingly, the detection of metachronous multicentric recurrent
carcinoma at an earlier stage and the instigation of appropriate
additional therapy may prolong survival in patients with MO
(21). Furthermore, molecular
elucidation of the risks for MO using CN liver tissue could provide
useful information alongside evaluation of the cancer tissue
itself.
In the present study, a multi-combination array
analysis, using SN liver tissue obtained from surgically resected
specimens of metastatic liver cancer as a control, revealed that
JAK2 overexpression and promoter region demethylation were
striking characteristics of one particular CN specimen.
Constitutive JAK2 activation is known to be largely
responsible for the development of polycythemia vera and
myeloproliferative neoplasms (22,23).
JAK family members, including JAK1, JAK2, JAK3 and TYK2, have also
been detected in the human liver by both immunohistochemistry and
western blotting (24). JAK2
protein expression is relatively high in normal human liver tissue
compared with other intestinal organs according to GeneCards
(25). Although JAK2
phosphorylation is not detected in the normal liver tissue, its
expression is significantly increased from the surrounding
non-neoplastic liver to HCC (26).
The activation of the JAK/STAT pathway in HCC was previously
demonstrated by measuring the phosphorylation of JAK/STAT proteins
(26). On the basis of the array
analysis results and subsequent literature review, we hypothesized
that JAK2 overexpression in the background normal liver
tissue adjacent to HCC could be a novel molecular candidate risk
factor for HCC. Since there have been few studies demonstrating
differential JAK2 expression among HCC, CN and SN liver
tissues, we proceeded to evaluate JAK2 expression using
surgically resected specimens from patients with HCC.
Contrary to our expectations, RT-qPCR revealed lower
JAK2 expression in CN than in SN. Indeed, only one of 100 CN
specimens exhibited higher JAK2 expression than the mean
value of all SN samples. Thus, the HCC case chosen for
multi-combination array analysis turned out to be a rare incidence
of higher JAK2 expression through promoter demethylation.
However, subsequent evaluation of JAK2 expression in the
clinical specimens of HCC did produce some significant findings.
First, we assessed the correlation between JAK2 expression
in tumor tissue and survival as JAK2 reportedly contributed to
oncogenesis through activation of STAT3 in a previous study
involving various human solid tumor cell lines (27). Additionally, high expression of
phosphorylated JAK2 in HCC tissue was found to associate
with poor prognosis (28). However,
in the present study, we found that JAK2 in tumor tissue was
not significantly correlated with survival in HCC patients. We also
observed that a greater HCC/CN ratio tended to associate with
better overall survival. Thus, we speculated that in HCC cases, the
JAK2 expression level in CN but not in cancerous tissues
affected overall survival, which we subsequently demonstrated in
this study. Lower JAK2 expression levels in CN tissue may
reflect liver conditions such as chronic inflammation, liver
cirrhosis or viral infection. However, some studies showed that DNA
binding activity of STAT3 was markedly increased in the remnant rat
liver immediately after partial hepatectomy (27,28).
Higher JAK2 expression in SN tissue may also reflect the
better regeneration potential of an intact liver (29). Since an intact liver generally
exhibits high JAK2 expression after liver resection, varying
levels of JAK2 expression can only be observed in patients
with a damaged liver and thus suppressed JAK2 expression.
Nonetheless, the reason why higher JAK2 expression persisted
despite viral or inflammatory damage inherent to CN tissue and the
mechanism underlying its association with poor prognosis observed
in this study remain unclear. Recent findings suggest that high
JAK2 expression could reflect carcinogenesis potential in the
background liver tissue and may indicate the existence of hormones
or cytokines upstream of JAK2 that potentially may affect HCC
outcomes (30,31).
In high-risk HCC cases, such as those with viral
hepatitis or steatohepatitis, measurement of JAK2 expression
in liver biopsies or surgically resected specimens may enable the
prediction of early disease recurrence via higher JAK2
expression. Patients with high JAK2 expression could then be
offered a more intense follow-up program, such as frequent
examination with ultrasonography or computed tomography. Further
study is needed to elucidate how genes such as JAK2 in
adjacent normal liver tissue response to HCC development and
recurrence. Such knowledge could provide excellent opportunity for
novel approaches to prediction, prevention, and exploitation of
molecular-targeted therapy for HCC.
In conclusion, our findings suggest that higher
JAK2 expression in the background normal liver tissue of HCC
may be a good prognostic biomarker for resected HCC. Thus, its
combination with other tumor prognostic factors may lead to a more
accurate prediction of HCC prognosis.
Acknowledgements
This study was supported by the Japan Society for
the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Scientific
Research (C) no. 22591427.
References
|
1
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rahbari NN, Mehrabi A, Mollberg NM, et al:
Hepatocellular carcinoma: current management and perspectives for
the future. Ann Surg. 253:453–469. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kobayashi A, Kawasaki S, Miyagawa S, et
al: Results of 404 hepatic resections including 80 repeat
hepatectomies for hepatocellular carcinoma. Hepatogastroenterology.
53:736–741. 2006.PubMed/NCBI
|
|
4
|
Chen MS, Li JQ, Zheng Y, et al: A
prospective randomized trial comparing percutaneous local ablative
therapy and partial hepatectomy for small hepatocellular carcinoma.
Ann Surg. 243:321–328. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Taura K, Ikai I, Hatano E, Fujii H, Uyama
N and Shimahara Y: Implication of frequent local ablation therapy
for intrahepatic recurrence in prolonged survival of patients with
hepatocellular carcinoma undergoing hepatic resection: an analysis
of 610 patients over 16 years old. Ann Surg. 244:265–273. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen PJ, Chen DS, Lai MY, et al: Clonal
origin of recurrent hepatocellular carcinomas. Gastroenterology.
96:527–529. 1989.PubMed/NCBI
|
|
7
|
Imamura H, Matsuyama Y, Tanaka E, et al:
Risk factors contributing to early and late phase intrahepatic
recurrence of hepatocellular carcinoma after hepatectomy. J
Hepatol. 38:200–207. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Portolani N, Coniglio A, Ghidoni S, et al:
Early and late recurrence after liver resection for hepatocellular
carcinoma: prognostic and therapeutic implications. Ann Surg.
243:229–235. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cucchetti A, Piscaglia F, Caturelli E, et
al: Comparison of recurrence of hepatocellular carcinoma after
resection in patients with cirrhosis to its occurrence in a
surveilled cirrhotic population. Ann Surg Oncol. 16:413–422. 2009.
View Article : Google Scholar
|
|
10
|
Matsuda M, Fujii H, Kono H and Matsumoto
Y: Surgical treatment of recurrent hepatocellular carcinoma based
on the mode of recurrence: repeat hepatic resection or ablation are
good choices for patients with recurrent multicentric cancer. J
Hepatobiliary Pancreat Surg. 8:353–359. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Morimoto O, Nagano H, Sakon M, et al:
Diagnosis of intrahepatic metastasis and multicentric
carcinogenesis by microsatellite loss of heterozygosity in patients
with multiple and recurrent hepatocellular carcinomas. J Hepatol.
39:215–221. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nakata T, Seki N, Miwa S, et al:
Identification of genes associated with multiple nodules in
hepatocellular carcinoma using cDNA microarray: multicentric
occurrence or intrahepatic metastasis? Hepatogastroenterology.
55:865–872. 2008.PubMed/NCBI
|
|
13
|
Nomoto S, Yamashita K, Koshikawa K, Nakao
A and Sidransky D: Mitochondrial D-loop mutations as clonal markers
in multicentric hepatocellular carcinoma and plasma. Clin Cancer
Res. 8:481–487. 2002.PubMed/NCBI
|
|
14
|
Nomoto S, Kinoshita T, Kato K, et al:
Hypermethylation of multiple genes as clonal markers in
multicentric hepatocellular carcinoma. Br J Cancer. 97:1260–1265.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stangegaard M: Gene expression analysis
using agilent DNA microarrays. Methods Mol Biol. 529:133–145. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bibikova M and Fan JB: GoldenGate assay
for DNA methylation profiling. Methods Mol Biol. 507:149–163. 2009.
View Article : Google Scholar
|
|
17
|
Okamura Y, Nomoto S, Hayashi M, et al:
Identification of the bleomycin hydrolase gene as a methylated
tumor suppressor gene in hepatocellular carcinoma using a novel
triple-combination array method. Cancer Lett. 312:150–157. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Takai D and Jones PA: The CpG island
searcher: a new WWW resource. In Silico Biol. 3:235–240.
2003.PubMed/NCBI
|
|
19
|
Wang Z, Zhang G, Wu J and Jia M: Adjuvant
therapy for hepatocellular carcinoma: current situation and
prospect. Drug Discov Ther. 7:137–143. 2013.PubMed/NCBI
|
|
20
|
Takenaka K, Adachi E, Nishizaki T, et al:
Possible multicentric occurrence of hepatocellular carcinoma: a
clinicopathological study. Hepatology. 19:889–894. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kumada T, Nakano S, Takeda I, et al:
Patterns of recurrence after initial treatment in patients with
small hepatocellular carcinoma. Hepatology. 25:87–92. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Amato D and Wang C: Resolution of
thrombocytopenia with JAK2 mutation in a patient with Gaucher
disease. Blood. 122:4287–4288. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kralovics R, Passamonti F, Buser AS, et
al: A gain-of-function mutation of JAK2 in myeloproliferative
disorders. N Engl J Med. 352:1779–1790. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hellgren G, Jansson JO, Carlsson LM and
Carlsson B: The growth hormone receptor associates with Jak1, Jak2
and Tyk2 in human liver. Growth Horm IGF Res. 9:212–218. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
GeneCards. Weizmann Institute of Science.
2014, Available from http://www.genecards.org.
|
|
26
|
Calvisi DF, Ladu S, Gorden A, et al:
Ubiquitous activation of Ras and Jak/Stat pathways in human HCC.
Gastroenterology. 130:1117–1128. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cressman DE, Diamond RH and Taub R: Rapid
activation of the Stat3 transcription complex in liver
regeneration. Hepatology. 21:1443–1449. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Trautwein C, Rakemann T, Niehof M,
Rose-John S and Manns MP: Acute-phase response factor, increased
binding, and target gene transcription during liver regeneration.
Gastroenterology. 110:1854–1862. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ono M, Chia DJ, Merino-Martinez R,
Flores-Morales A, Unterman TG and Rotwein P: Signal transducer and
activator of transcription (Stat) 5b-mediated inhibition of
insulin-like growth factor binding protein-1 gene transcription: a
mechanism for repression of gene expression by growth hormone. Mol
Endocrinol. 21:1443–1457. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yeh YT, Lee KT, Tsai CJ, Chen YJ and Wang
SN: Prolactin promotes hepatocellular carcinoma through Janus
kinase 2. World J Surg. 36:1128–1135. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yoshikawa H, Matsubara K, Qian GS, et al:
SOCS-1, a negative regulator of the JAK/STAT pathway, is silenced
by methylation in human hepatocellular carcinoma and shows
growth-suppression activity. Nat Genet. 28:29–35. 2001. View Article : Google Scholar : PubMed/NCBI
|