Introduction
Cholangiocarcinoma (CCA) is a devastating biliary
cancer that poses continuing diagnostic and therapeutic challenges
(1). There are several risk factors
for CCA: mainly liver fluke infection, primary sclerosing
cholangitis, biliary-duct cysts and hepatolithiasis (2). The highest prevalence of liver fluke
Opisthorchis viverrini infection has been reported in
Northeast Thailand, where CCA incidence is also high (3). CCA patients usually present after the
disease is advanced and have a short survival outcome. Chemotherapy
has not been shown to obviously improve survival in patients with
CCA. Therefore, new strategies are needed to improve the treatment
of this cancer.
Apoptosis, a form of programmed cell death, plays an
important role in the homeostasis, development and prevention of
cancer (4). Induction of apoptosis
is considered a major goal of anticancer therapies (5,6).
Several chemopreventive agents exert their oncostatic effects via
the production of reactive oxygen species (ROS), which ultimately
disrupt the redox tone leading to cytostasis and/or cell death
partly via the mitochondrial pathway (7). Mitochondria are believed to be the
major source of ROS production (8).
Furthermore, the mitochondrial pathway of apoptosis is triggered by
various death signals, such as ROS and DNA damage. These signals
promote binding of the pro-apoptotic protein Bax with the outer
mitochondrial membrane, which disrupts the mitochondrial membrane
potential, resulting in the release of apoptogenic factors such as
cytochrome c from the mitochondria to the cytosol. This in
turns leads to activation of the caspase cascades and cell death
(9,10). Thus, enhancement of mitochondrial
apoptosis may be a strategy for the treatment of cancer (11).
Melatonin, the major secretory product of the pineal
gland, may exert differential effects for protection against cancer
under certain conditions (12) with
antioxidant property or oncostatic action (13). The oncostatic effects of melatonin
involve the inhibition of neoplastic cell proliferation,
intensified apoptosis, and decreased capacity to form metastases
(14). Our previous study
demonstrated the chemopreventive effect of melatonin on CCA
hamsters (15). However, the
cytotoxic effect of melatonin on CCA treatment remains unclear.
The present study aimed to investigate the cytotoxic
effect of melatonin on human CCA cell lines. Human CCA cell lines
with poor differentiation (KKU-M055) and well differentiation
(KKU-M214) were treated with melatonin at concentrations of 0.5, 1,
and 2 mM for 48 h. The viability of cells, generation of ROS, DNA
damage formation, and expression of apoptotic-related proteins were
assayed. The study revealed that melatonin exhibited a cytotoxic
effect on CCA cell lines which points to a potential therapeutic
role for melatonin in patients with advanced stage CCA.
Materials and methods
Cell culture and treatment
Human CCA cell lines KKU-M055 (poorly differentiated
CCA) and KKU-M214 (well-differentiated CCA) were used in the
present study. CCA cell lines were established and characterized
from CCA patients hospitalized at the Faculty of Medicine, Khon
Kaen University (16). Cells were
cultured in Ham’s F-12 medium supplemented with 10% fetal bovine
serum, 100 U/ml penicillin and 100 μg/ml streptomycin at 37°C in a
5% CO2 humidified atmosphere. Melatonin (Huanggang
Innovation Biochemicals, Hubei, China) was dissolved in dimethyl
sulfoxide (DMSO) before being diluted with medium with a final DMSO
concentration of 0.1%. Cells were treated with melatonin at
concentrations of 0.5, 1 and 2 mM, or vehicle (0.1% DMSO) for 48
h.
Cell viability assay
Cell viability was assayed by
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT;
Invitrogen Life Technologies, Carlsbad, CA, USA). Briefly, cells
were plated at a density of 1×104 cells/well into
96-well plates. After overnight growth, the cells were treated with
1 nM, 1 μM, 0.5, 1 and 2 mM of melatonin for 24, 48, and 72 h.
After treatment, the cells were incubated for 4 h with 5 mg/ml of
MTT reagent and lysed with DMSO. The absorbance was monitored with
a microplate reader at a wavelength of 570 nm (reference wavelength
at 655 nm).
Measurement of intracellular ROS
Evaluation of intracellular oxidants was carried out
using the fluorescent probe 5-(and
-6)-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate, acetyl
ester (CM-H2DCFDA; Invitrogen Life Technologies).
Briefly, after treatment with melatonin for 48 h, the cells were
incubated with PBS containing CM-H2DCFDA at a final
concentration of 5 μM at 37°C for 30 min. The cells were then
harvested by trypsinization and washed twice with PBS. The cells
were suspended in PBS and analyzed on a flow cytometer (FACScan;
Becton-Dickinson, San Jose, CA, USA).
Immunostaining of 8-oxo-7,
8-dihydro-2′-deoxyguanosine (8-oxodG)
To evaluate the simultaneous localization of 8-oxodG
in nuclear and mitochondrial DNA, the cells were labeled with 100
nM MitoTracker® Red CMXRos probe (mitochondria-specific)
(Invitrogen Life Technologies) for 15 min at 37°C. After staining,
the cells were washed in PBS and fixed with 4% formaldehyde in PBS
for 10 min. The cells were then permeabilized with 0.5% Triton
X-100 in PBS for 3 min and washed with PBS before being blocked
with 5% skim milk for 60 min. Subsequently, the cells were
incubated with mouse monoclonal anti-8-oxodG antibody (Japan
Institute for the Control of Aging, Fukuroi, Japan) at 1:200
dilution overnight at room temperature and then incubated with
Alexa 488-labeled goat antibody against mouse IgG at 1:400 dilution
for 3 h at room temperature. The cells were mounted and visualized
under a fluorescence microscope (Axiovert 200; Carl Zeiss,
Göttingen, Germany). The excitation/emission wavelengths for
MitoTracker® Red CMXRos were 579/599 nm,
respectively.
Measurement of cell death
Cell death was quantified using TACS™ Annexin V kits
(Trevigen, Gaithersburg, MD, USA) as per the manufacturer’s
instructions. Briefly, the cells were collected by centrifugation
at 300 × g for 10 min and washed with PBS. The cells were then
resuspended in 100 μl of Annexin V/PI incubation reagent for 15 min
at room temperature, followed by addition of 400 μl of 1X binding
buffer and processing by flow cytometry. The following staining
criterion was used for characterization: cells that were not
stained by either Annexin V or PI were considered healthy, cells
stained by only Annexin V were considered early apoptotic, and
cells stained by both PI and Annexin V were considered late
apoptotic or necrotic.
Western blot analysis
Protein from the whole cell lysates was extracted
using RIPA buffer (50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100,
0.5% sodium deoxycholate and 0.1% SDS). Protein was separated on
12–15% SDS-PAGE, transferred onto a polyvinylidenedifluoride
membrane (GE Healthcare Biosciences Corp., Piscataway, NJ, USA) and
immunoblotted with specific antibodies. The primary antibodies used
were rabbit polyclonal anti-Bcl2 (ab7973), rabbit polyclonal
anti-caspase-3 (ab44976), rabbit polyclonal anti-inducible nitric
oxide synthase (iNOS) (ab15323; Abcam, Cambridge, MA, USA), goat
polyclonal anti-caspase-7 (sc-6138) and rabbit polyclonal
anti-cytochrome c (sc-7159; Santa Cruz Biotechnology, Santa
Cruz, CA, USA). Rabbit polyclonal anti-GAPDH antibody (ab77109;
Abcam) was used as a sample loading control. Immunostained protein
bands were detected with an enhanced chemiluminescence kit (GE
Healthcare Biosciences Corp.).
Statistical analysis
The data are expressed as mean ± SD. Statistical
analysis was conducted using one-way analysis of variance (ANOVA)
using SPSS version 11.5 (SPSS Inc., Chicago, IL, USA). A P<0.05
was considered to indicate a statistically significant result.
Results
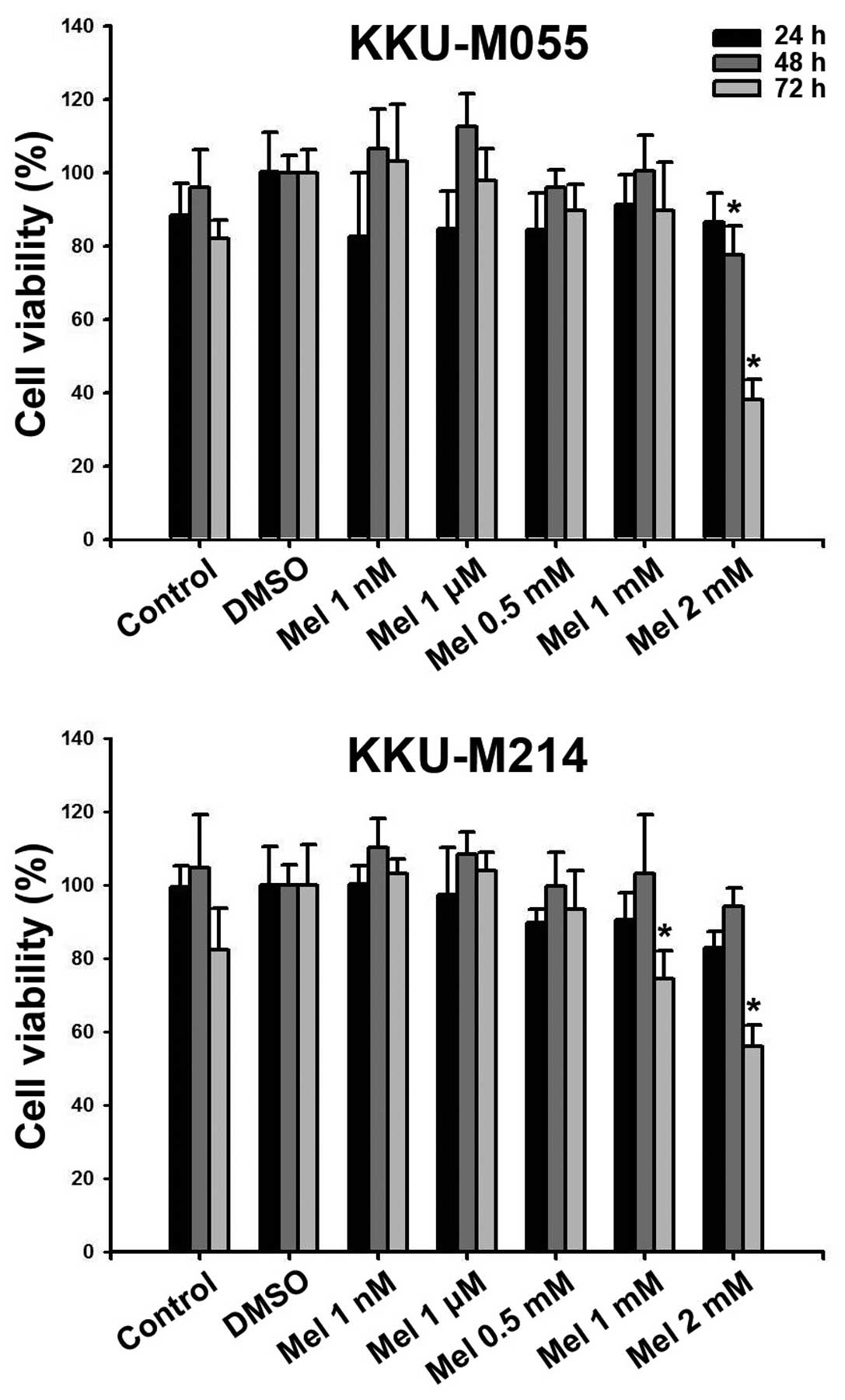
Melatonin decreases cell viability
To investigate the effect of the concentration and
the duration of melatonin treatment on the viability of cells,
KKU-M055 and KKU-M214 cells were treated with different
concentrations of melatonin (1 nM, 1 μM and 0.5, 1 and 2 mM) for
24, 48, and 72 h. The result of the MTT assay showed that melatonin
decreased the percentage of viable cells in a concentration and
time-dependent manner. Treatment with physiological (1 nM) or low
pharmacological (1 μM) concentrations of melatonin did not
demonstrate a significant effect. Treatment with high
pharmacological concentrations of melatonin (1 and 2 mM)
significantly decreased cell viability compared to the DMSO-treated
group (Fig. 1). Therefore,
concentrations of 0.5, 1 and 2 mM and an incubation time of 48 h
were used in the subsequent experiments.
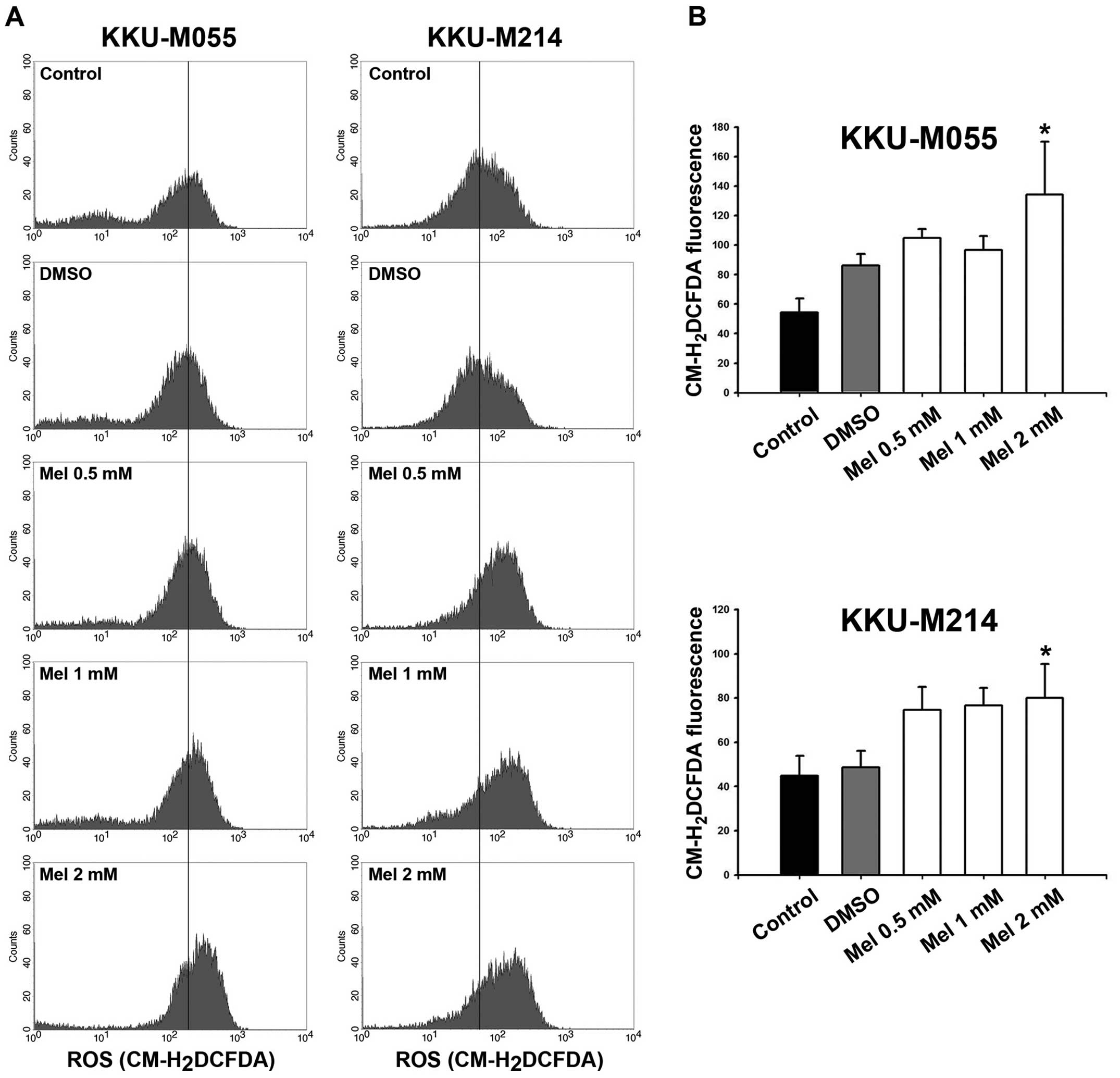
Melatonin enhances ROS production
The ROS generation in KKU-M055 and KKU-M214 cells
after treatment with different concentrations (0.5, 1, and 2 mM) of
melatonin for 48 h was measured by CM-H2DCFDA
fluorescence. The result showed that melatonin treatment increased
ROS production in a concentration-dependent manner (Fig. 2). At a high concentration (2 mM) of
melatonin treatment, it significantly increased
CM-H2DCFDA fluorescence when compared with the
DMSO-treated group (Fig. 2B). We
examined the protein expression of iNOS, an enzyme that generates
free radical nitric oxide (NO) by western blot analysis. The result
revealed that melatonin treatment increased iNOS expression in a
concentration-dependent manner (Fig.
5).
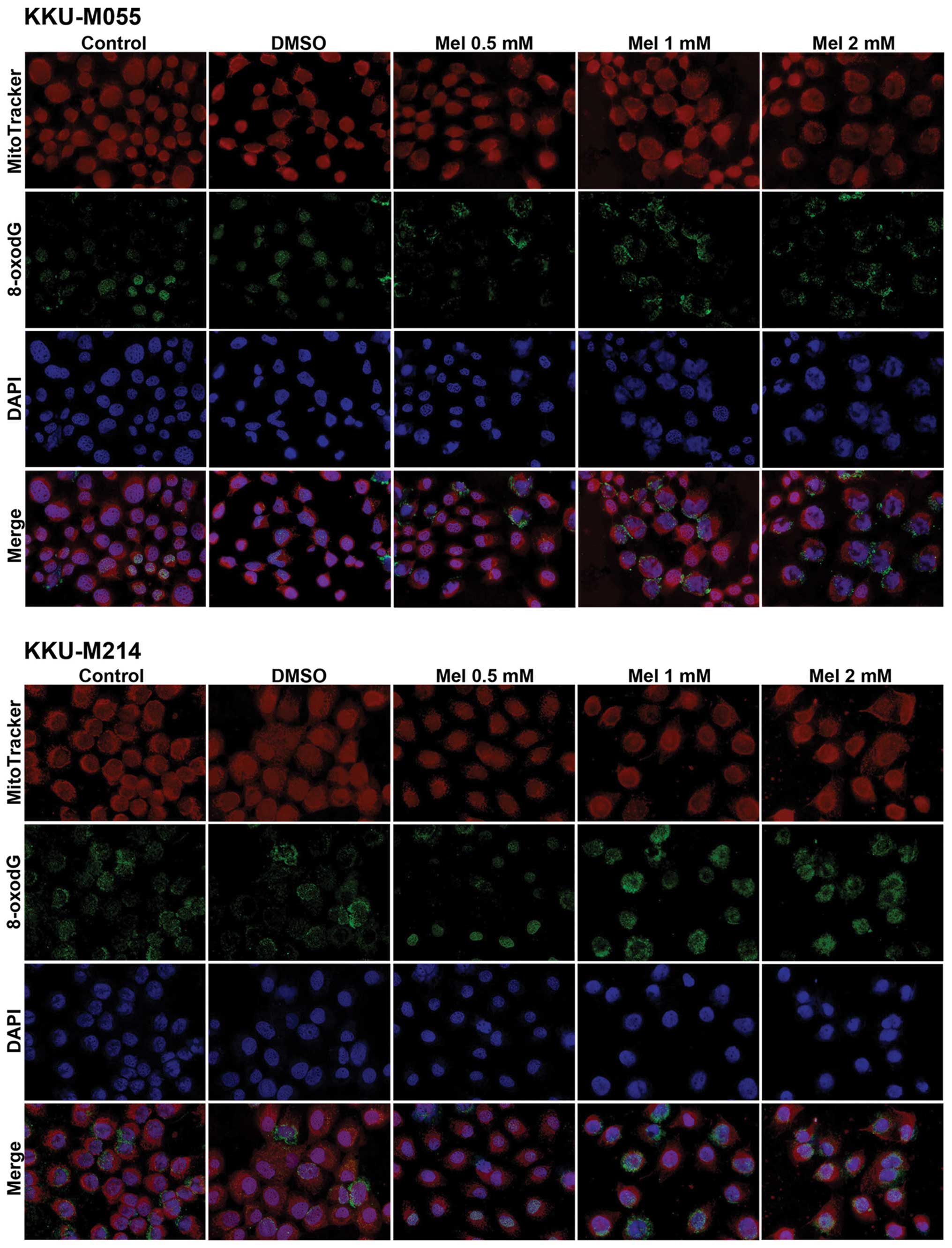
Melatonin induces DNA damage
Since ROS can cause oxidative damage to DNA, we
measured the damage of DNA caused by melatonin treatment by
immunostaining of 8-oxodG for 48 h after melatonin treatment. The
results showed that melatonin treatment coincided with an increase
in 8-oxodG-positive cells in a concentration-dependent manner.
Notably, the 8-oxodG-positive area in the control and DMSO-treated
groups was mostly found in the nucleus while this area in the
melatonin-treated group was distributed to the cytoplasm and
mitochondria, as shown by the result of merging the 8-oxodG and
MitoTracker signals (Fig. 3).
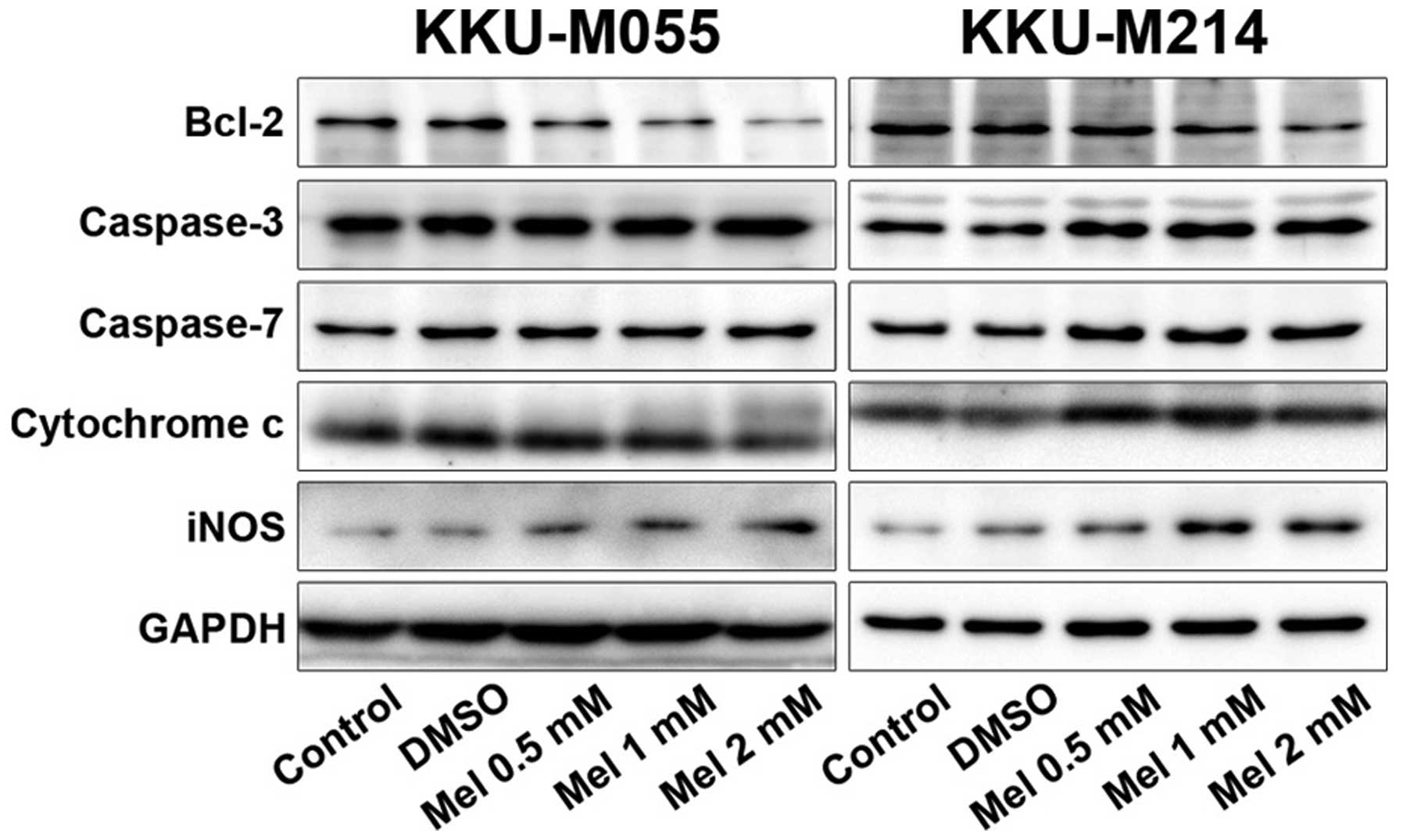
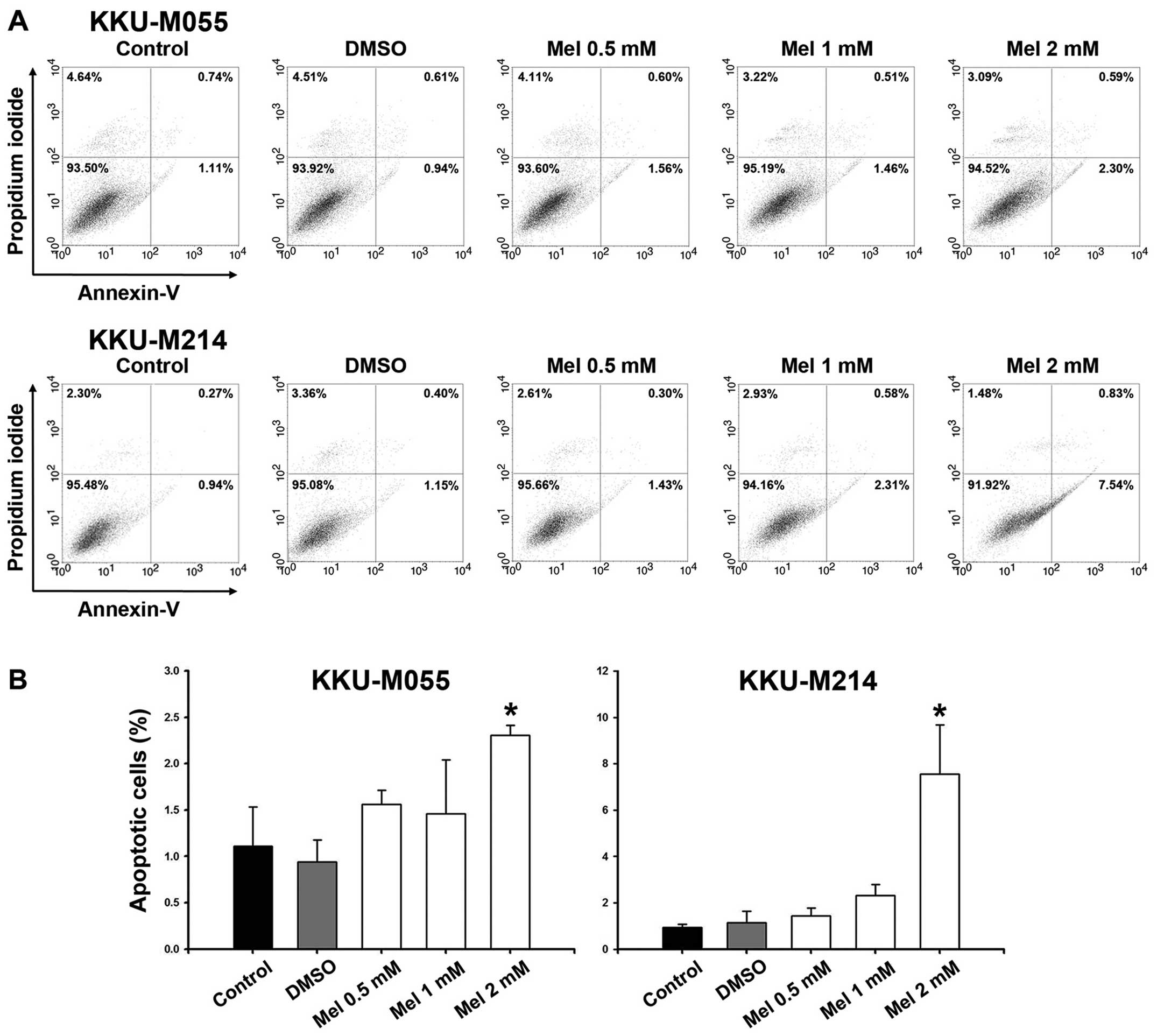
Melatonin induces apoptosis
The apoptosis caused by different concentrations of
melatonin was evaluated by flow cytometry of Annexin V/PI. The
results showed that melatonin increased the percentage of apoptotic
cells in a concentration-dependent manner. Compared to the
DMSO-treated cells, there was a significant increase in the
proportion of cells in early apoptosis (Annexin
V+/PI−) after 2 mM melatonin treatment
(Fig. 4). Furthermore, we examined
the expression of apoptosis-related proteins by western blot
analysis. Melatonin treatment promoted the activation of caspase-3
and caspase-7, and cytochrome c while inhibiting the
expression of the anti-apoptotic protein Bcl-2 (Fig. 5).
Discussion
Melatonin has been shown to have a wide range of
physiological functions in humans and has been implicated in the
disruption of neoplastic processes (17,18).
The physiological actions of melatonin are partly attributed to its
antioxidant activity (19). Under
certain conditions, melatonin may also exert oxidant effects,
particularly in cancer cells (20).
In the present study, we demonstrated that high pharmacological
concentrations of melatonin exhibited a cytotoxic effect on human
CCA cell lines. Our results showed that melatonin enhanced
ROS-dependent DNA damage, leading to apoptosis and reducing the
viability of cells. This suggests that melatonin induces tumor cell
death by its pro-oxidant activity (20,21).
Melatonin may act as either a pro-oxidant or an
antioxidant in different contexts in order to exert an antitumor
effect (12). It is well known as
an antioxidant, free radical scavenger and oncostatic agent in
preventing tumors at initiation, promotion and progression steps in
a variety of cancer types such as breast, colorectal,
hepatocellular, and bile duct cancer (22–25).
In our previous study, we showed that melatonin exerted
chemopreventive effects on O. viverrini infection (26) and on O. viverrini plus
N-nitrosodimethylamine-induced CCA in hamsters (15) by reducing oxidative and nitrative
DNA damage, liver injury, and tumor mitochondrial pathways of
apoptosis. On the other hand, here, we found that melatonin
exhibited pro-oxidant activity instead of an antioxidant effect in
CCA cells, suggesting that melatonin may be a double-edged sword
acting in cancer under certain conditions (12).
Although there is much evidence that melatonin
exerts antioxidant activity, a few studies using cultured cells
have revealed that melatonin may generate ROS at pharmacologically
active concentrations (μM to mM range) and therefore may have
pro-oxidant activity under some conditions (12). Our results showed that melatonin was
able to stimulate production of intracellular ROS as evidenced by
the increase in CM-H2DCFDA fluorescence. Treatment of
human CCA cell lines with melatonin at a concentration of 2 mM
increased ROS generation, but this effect was not observed at 0.5
or 1 mM of melatonin. Stimulation of ROS production by 2 mM
melatonin in CCA cell lines was similar to the previous finding
that high-dose (1 mM) melatonin caused stimulation of ROS
production in human leukaemia cells, but did not after treatment
with a melatonin dose lower than 10 μM (27). Taken together, these results
demonstrate that melatonin induces the production of ROS that may
account for the cytotoxicity of melatonin at high concentrations
(20,21,27).
Mechanistically, the production of ROS by melatonin
in high doses may be explained by its interaction with calmodulin,
the mitochondrial complex III or the mitochondrial transition pore
(12). The pro-oxidant, cytotoxic
and pro-apoptotic effects of melatonin on human CCA cell lines were
similar to the effects of melatonin treatment on human
promyelocytic leukaemia HL-60 cells observed previously (20,28).
Thus, the pro-oxidant action of melatonin may assist in restraining
CCA cell growth, which may be useful for treating patients with
otherwise untreatable metastatic solid tumors (29).
Treatment with melatonin induced ROS production,
which was concomitant with apoptosis and a loss of CCA cell
viability. These results are consistent with previous observations
that other cancer cells are sensitive to melatonin, particularly
liver cancer cells (30,31). Melatonin treatment also induced a
decrease in the expression of the anti-apoptotic protein Bcl-2 and
an increase in the expression of cytochrome c and
apoptotic-associated caspases (caspase-3 and caspase-7), all of
which are indicators of apoptotic cell death. Moreover, 8-oxodG in
melatonin-treated cells was localized to the cytoplasm and
mitochondria as shown by the result of merging 8-oxodG and
MitoTracker (Fig. 3), while in the
control and DMSO-treated groups 8-oxodG was mostly found in the
nucleus, indicating that melatonin induced mitochondrial DNA
damage. Taken together, these data suggest that the increase in ROS
production is responsible for the pro-apoptotic effect of high
concentrations of melatonin in CCA cells.
We previously demonstrated that melatonin can
attenuate apoptosis in hamster CCA. In contrast, we here found that
treatment with melatonin at 2 mM induced apoptosis in human CCA
cells, but at lower concentrations of 0.5 and 1 mM melatonin
treatment had no effect on apoptosis. Previous studies have shown
that melatonin treatment of non-small cell lung cancers (NSCLCs)
after genotoxic stress induced by irradiation with UV increased
apoptosis, but no apoptotic effect was observed in NSCLC cell lines
without genotoxic damage (32).
Furthermore, induction of apoptosis by melatonin was dose-dependent
in the high stress conditions, the predominance of DNA damage, in
NSCLC cell lines. Taken together, we suggest that melatonin exerts
effects on the apoptosis pathway according to stress conditions and
the dose of melatonin. This phenomenon may be caused by increased
oxidative stress of cancer cells as a result of their elevated
metabolism. Thus, melatonin treatment may be useful in impairing
the ROS buffering capacity of cancer cells (28). We did not test the cytotoxic effect
of melatonin on normal cholangiocytes in the present study;
however, previous studies have shown that melatonin does not confer
any observable cytotoxic effects on hepatocytes in normal hamsters
(26).
In conclusion, melatonin treatment in human CCA cell
lines increased intracellular ROS, which was associated with
significant cytotoxicity and activation of caspase activities.
Melatonin may exert a pro-oxidant cytotoxic effect on human CCA
cell lines and thereby reduce the viability of cancer cells by
activating ROS-dependent DNA damage and inducing apoptosis. The
activation of ROS production by melatonin is associated with
cytotoxicity in CCA cells, but only at high concentrations of
melatonin. We suggest that melatonin could be used as a supplement
to classical anticancer drugs in therapies used to treat CCA
patients. Confirming the efficacy and safety of melatonin in cancer
treatment will require further study in CCA patients.
Acknowledgements
The present study was supported by The Thailand
Research Fund through Royal Golden Jubilee Ph.D. Program (to U.L.
and S.P.), the Thailand Research Fund (RMU5380016), and The Higher
Education Research Promotion and National Research University
Project of Thailand, Office of the Higher Education Commission,
through the Health Cluster (SHeP-GMS), Khon Kaen University. This
study was partly supported by Grants-in-Aids for Scientific
Research from the Ministry of Education, Culture, Sports, Science
and Technology of Japan (no. 25293149). We thank Dr Justin Reese,
Research Affairs, Faculty of Medicine, Khon Kaen University,
Thailand for critically reading and improving this manuscript.
References
|
1
|
Anderson CD, Pinson CW, Berlin J and Chari
RS: Diagnosis and treatment of cholangiocarcinoma. Oncologist.
9:43–57. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tyson GL and El-Serag HB: Risk factors for
cholangiocarcinoma. Hepatology. 54:173–184. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
IARC. A Review of Human Carcinogens:
Opisthorchis viverrini and Clonorchis sinensis. IARC Monogr Eval
Carcinog Risks Hum. 100B:341–370. 2012.
|
|
4
|
Gerl R and Vaux DL: Apoptosis in the
development and treatment of cancer. Carcinogenesis. 26:263–270.
2005. View Article : Google Scholar
|
|
5
|
Kaufmann SH and Earnshaw WC: Induction of
apoptosis by cancer chemotherapy. Exp Cell Res. 256:42–49. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hannun YA: Apoptosis and the dilemma of
cancer chemotherapy. Blood. 89:1845–1853. 1997.PubMed/NCBI
|
|
7
|
Hail N Jr and Lotan R: Cancer
chemoprevention and mitochondria: targeting apoptosis in
transformed cells via the disruption of mitochondrial
bioenergetics/redox state. Mol Nutr Food Res. 53:49–67. 2009.
View Article : Google Scholar
|
|
8
|
Turrens JF: Mitochondrial formation of
reactive oxygen species. J Physiol. 552:335–344. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Green DR and Reed JC: Mitochondria and
apoptosis. Science. 281:1309–1312. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Desagher S and Martinou JC: Mitochondria
as the central control point of apoptosis. Trends Cell Biol.
10:369–377. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kasibhatla S and Tseng B: Why target
apoptosis in cancer treatment? Mol Cancer Ther. 2:573–580.
2003.PubMed/NCBI
|
|
12
|
Zhang HM and Zhang Y: Melatonin: a
well-documented anti-oxidant with conditional pro-oxidant actions.
J Pineal Res. 57:131–146. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bartsch C, Bartsch H and Karasek M:
Melatonin in clinical oncology. Neuro Endocrinol Lett. 23:30–38.
2002.PubMed/NCBI
|
|
14
|
Jung B and Ahmad N: Melatonin in cancer
management: progress and promise. Cancer Res. 66:9789–9793. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Laothong U, Pinlaor P, Boonsiri P, et al:
Melatonin inhibits cholangiocarcinoma and reduces liver injury in
Opisthorchis viverrini-infected and N-nitrosodimethylamine-treated
hamsters. J Pineal Res. 55:257–266. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sripa B, Leungwattanawanit S, Nitta T, et
al: Establishment and characterization of an
opisthorchiasis-associated cholangiocarcinoma cell line (KKU-100).
World J Gastroenterol. 11:3392–3397. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sanchez-Barcelo EJ, Cos S, Fernandez R and
Mediavilla MD: Melatonin and mammary cancer: a short review. Endocr
Relat Cancer. 10:153–159. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hill SM, Frasch T, Xiang S, Yuan L,
Duplessis T and Mao L: Molecular mechanisms of melatonin anticancer
effects. Integr Cancer Ther. 8:337–346. 2009. View Article : Google Scholar
|
|
19
|
Barrenetxe J, Delagrange P and Martinez
JA: Physiological and metabolic functions of melatonin. J Physiol
Biochem. 60:61–72. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bejarano I, Espino J, Barriga C, Reiter
RJ, Pariente JA and Rodriguez AB: Pro-oxidant effect of melatonin
in tumour leucocytes: relation with its cytotoxic and pro-apoptotic
effects. Basic Clin Pharmacol Toxicol. 108:14–20. 2011. View Article : Google Scholar
|
|
21
|
Rodriguez C, Martin V, Herrera F, et al:
Mechanisms involved in the pro-apoptotic effect of melatonin in
cancer cells. Int J Mol Sci. 14:6597–6613. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carbajo-Pescador S, Garcia-Palomo A,
Martin-Renedo J, Piva M, Gonzalez-Gallego J and Mauriz JL:
Melatonin modulation of intracellular signaling pathways in
hepatocarcinoma HepG2 cell line: role of the MT1 receptor. J Pineal
Res. 51:463–471. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Han Y, Demorrow S, Invernizzi P, et al:
Melatonin exerts by an autocrine loop antiproliferative effects in
cholangiocarcinoma: its synthesis is reduced favoring
cholangiocarcinoma growth. Am J Physiol Gastrointest Liver Physiol.
301:G623–G633. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Motilva V, Garcia-Maurino S, Talero E and
Illanes M: New paradigms in chronic intestinal inflammation and
colon cancer: role of melatonin. J Pineal Res. 51:44–60. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Proietti S, Cucina A, Reiter RJ and
Bizzarri M: Molecular mechanisms of melatonin’s inhibitory actions
on breast cancers. Cell Mol Life Sci. 70:2139–2157. 2013.
View Article : Google Scholar
|
|
26
|
Laothong U, Pinlaor P, Hiraku Y, et al:
Protective effect of melatonin against Opisthorchis
viverrini-induced oxidative and nitrosative DNA damage and liver
injury in hamsters. J Pineal Res. 49:271–282. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Buyukavci M, Ozdemir O, Buck S, Stout M,
Ravindranath Y and Savasan S: Melatonin cytotoxicity in human
leukemia cells: relation with its pro-oxidant effect. Fundam Clin
Pharmacol. 20:73–79. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bejarano I, Espino J, Marchena AM, et al:
Melatonin enhances hydrogen peroxide-induced apoptosis in human
promyelocytic leukaemia HL-60 cells. Mol Cell Biochem. 353:167–176.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lissoni P, Paolorossi F, Tancini G, et al:
A phase II study of tamoxifen plus melatonin in metastatic solid
tumour patients. Br J Cancer. 74:1466–1468. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Martin-Renedo J, Mauriz JL, Jorquera F,
Ruiz-Andres O, Gonzalez P and Gonzalez-Gallego J: Melatonin induces
cell cycle arrest and apoptosis in hepatocarcinoma HepG2 cell line.
J Pineal Res. 45:532–540. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Osseni RA, Rat P, Bogdan A, Warnet JM and
Touitou Y: Evidence of prooxidant and antioxidant action of
melatonin on human liver cell line HepG2. Life Sci. 68:387–399.
2000. View Article : Google Scholar
|
|
32
|
Kim W, Jeong JW and Kim JE: CCAR2
deficiency augments genotoxic stress-induced apoptosis in the
presence of melatonin in non-small cell lung cancer cells. Tumour
Biol. 135:10919–10929. 2014. View Article : Google Scholar
|