Introduction
Adrenocortical carcinoma (ACC) is a rare but
aggressive endocrine malignancy with an incidence of ~1–2/million
annually and a 5-year survival rate of 16–44% (1–3).
Currently, there is limited information concerning the molecular
pathology and pathogenesis of ACC and there is no effective
therapy. In addition, distinguishing ACC from adrenocortical
adenoma (ACA) using conventional histology and imaging can be
difficult. Presently, no molecular pathological markers are able to
reliably distinguish between the two cancers. Thus, local invasion
or distant metastasis is the only absolute demonstration of
malignancy (4). Given the rarity
and poor survival rate of ACC and the high prevalence of ACA in the
general population (5), as well as
the different treatments used (6),
the use of biomarkers to confirm the type of adrenocortical tumors
(ACT) is required. These markers are also therapeutic targets and
prognostic indicators for ACC.
MicroRNAs (miRNAs) are 19–25 nucleotide small,
non-coding RNAs involved in 30% of human gene expression via
binding to target mRNAs (7,8). Previous data showed that small RNAs
are misregulated in human tumors and can serve as either oncogenes
or tumor suppressor genes (9–11).
Functional studies suggest that aberrant miRNA expression is a key
to cancer development, including cell proliferation, apoptosis and
differentiation (12). Due to
specific expression patterns and tremendous regulatory capacity,
miRNAs are being assessed as potential biomarkers to help diagnose
and treat different types of cancers, including ACC. However,
limited miRNA expression analyses have been performed on ACC.
We noted downregulation of miR-205 expression in ACC
samples compared to ACAs. Overexpression of miR-205 not only
induced apoptosis and impaired proliferation of ACC SW-13 cell
in vitro, but also inhibited tumor growth in vivo.
Furthermore, miR-205 inhibited Bcl-2 mRNA and protein expression
via 3′-untranslated region (3′UTR) interaction; this activated the
intrinsic apoptosis pathway in SW-13 cells. Therefore, Bcl-2 is a
critical anti-apoptotic gene involved in the intrinsic apoptotic
pathway (13), and it is a target
via which miR-205 inhibits SW-13 cell proliferation.
Materials and methods
Cell line, tissues and mice
A SW-13 cell line was purchased from the Cell
Repository of the Chinese Academy of Science and originated from
the American Type Culture Collection (ATCC; Manassas, VA, USA).
Cells were cultured in Leibovitz-15 (L-15) media at 37°C without
CO2. Then, 32 cases of ACC and ACA tissue samples were
obtained from the Guangzhou General Hospital of People's Liberation
Army. Clinical characteristics of the study cohort are summarized
in Table I. Tumors were classified
as ACC when the Weiss criteria were ≥3. Tumors were classified as
benign when the Weiss criteria were <3 (14). Female nude mice 4 weeks of age were
provided by the Animal Center of Southern Medical University.
 | Table IClinical characteristics of the study
cohort. |
Table I
Clinical characteristics of the study
cohort.
|
Characteristics | ACC | ACA |
|---|
| No. of
patients | 11 | 21 |
| Age (average ±
SD) | 53±12 | 48±16 |
| Gender
(female/male) | 8/3 | 14/7 |
| Syndrome |
| Cushing's | 7 | 7 |
| Subclinical
Cushing's | 0 | 1 |
| Conn's | 0 | 3 |
|
Non-functioning | 4 | 10 |
Quantitative real-time PCR (qRT-PCR)
According to the manufacturer's protocol, total RNA
was extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA),
and first strand cDNA was synthesized using miScript Reverse
Transcription kit (Qiagen). miR-205 expression was quantified by
qRT-PCR using TaqMan microRNA assays (Applied Biosystems, Carlsbad,
CA, USA) and U6 was used for normalization. Bcl-2 expression was
quantified by qRT-PCR using SYBR-Green assays (Applied Biosystems)
and β-actin was used for normalization. Real-time PCR was performed
on the ABI 7500 Sequence Detection System (Applied Biosystems), and
relative expression was calculated using the 2−ΔΔCt
method. All experiments were performed in triplicate independently.
The sequences of primers are depicted in Table II.
 | Table IIPrimers used in the present
study. |
Table II
Primers used in the present
study.
| Primers | Sequence |
|---|
| miR-205 | F
5′-ACACTCCAGCTGGGTAGGTAGTTTCATGTTGTT-3′ |
| miR-205 | R
5′-CTCAACTGGTGTCGTGGA-3′ |
| U6 | F
5′-CTCGCTTCGGCAGCACA-3′ |
| U6 | R
5′-AACGCTTCACGAATTTGCGT-3′ |
| Bcl-2 | F
5′-ATTTTGTAGTCACCCACCTCTAAGG-3′ |
| Bcl-2 | R
5′-CATCTCCCTTCACAGCAGAACTTAAC-3′ |
| β-actin | F
5′-GGGAAATCGTGCGTGACATTAAGG-3′ |
| β-actin | R
5′-CAGGAAGGAAGGCTGGAAGAGTC-3′ |
miRNA mimics and transfection
miR-205 mimics, the negative control pre-miR
(miR-NC) and the miR-205 inhibitors provided by RiboBio (Guangzhou,
China) were transfected into SW-13 cells using Lipofectamine 2000
(Invitrogen) in accordance with the manufacturer's manual.
Cell proliferation assay
SW-13 cells transiently transfected with miR-205,
miR-NC and miR-205 inhibitors were cultured in 96-well plates
(4×103 cells/well) for 24 h. Cell proliferation was
measured via MTT assay (KeyGen, China) and further verified using a
5-ethynyl-2′-deoxyuridine (EdU) assay (RiboBio) following the
manufacturer's instructions. SW-13 cells incorporating EdU were
observed under fluorescent microscopy. Cell proliferation was
measured using the following formula: SW-13 cell proliferation =
number of EdU-positive cells/all cells × 100.
Cell apoptosis assay
Apoptosis of transfected SW-13 cells was measured
using the Annexin V/Propidium Iodide (PI) Detection kit (KeyGen)
following the manufacturer's instructions. Data were analyzed by
flow cytometry (Becton-Dickinson, Franklin Lakes, NJ, USA). In
addition, SW-13 cell apoptosis was measured via TUNEL assay. Cells
were stained with TUNEL (Sigma, St. Louis, MO, USA) to quantify
apoptotic nuclei. At least five visual fields were observed under a
fluorescent microscope for each sample.
Plasmid construction and stable
overexpression of miR-205
The pre-hsa-miR-205 sequence was synthesized with
the following primers by PCR: 5′-CCCAAGCTTCTGGGTGGCTGTTTTGAAAAC-3′
(F), and 5′-CCGCTCGAGGAAGCACGCACACTCCAGATG-3′ (R), and subcloned
into the pcDNA3.1(+) plasmid (Invitrogen) following the digestion
of EcoRI and BamHI to generate the recombinant
plasmid pcDNA3.1(+)-miR-205. SW-13 cells were cultured to reach
60–80% confluence, and were then transfected with
pcDNA3.1(+)-miR-205 according to the Lipofectamine 2000
instructions into 24-well plates. Then, transfected cells were
sorted by G418 (200 mg/l) and cultured. The expression of miR-205
was measured by qRT-PCR.
Tumor formation in nude mice
Stably transfected SW-13 cells (1×108)
were subcutaneously injected into the flank of each nude mouse (3
groups of 6 mice). All animal experiments were performed following
the NIH Guide for the Care and Use of Laboratory Animals. Tumor
volume (V) was calculated using the formula: (L × W2) ×
0.5 (L, length; W, width) with a Vernier caliper.
miRNA target prediction
The predicted target genes and their conserved sites
of the seed region binding with each miRNA were investigated using
TargetScan (http://www.targetscan.org) and miRSVR
(http://www.microrna.org).
Plasmid construction and luciferase
reporter assay
mRNA of wild-type Bcl-2–3′UTR (3,906–3,928 nt;
GenBank accession no. NM_000633) containing binding sequence
complementary to miR-205 was digested by XbaI and
FseI and cloned downstream of the pGL3-REPORT luciferase
vector (Promega, Madison, WI, USA). Based on the wild-type plasmid,
site-specific mutagenesis generated mutated Bcl-2–3′UTR
complementary to miR-205. For the luciferase reporter assay, SW-13
cells were co-transfected with wild-type or mutated Bcl-2–3′UTR and
miR-205 transiently, and the luciferase was measured with a
Dual-Luciferase Assay System (Promega) 48 h later. Each reporter
plasmid was transfected at least three times.
Western blotting
After being stably transfected with plasmid or
miR-205, proteins extracted from SW-13 cells were subjected to
SDS-PAGE and transferred to PVDF membranes. The membrane was
incubated with specific primary antibodies against Bcl-2, Bax
(Santa Cruz Biotechnology, Santa Cruz, CA, USA), caspase-9 and -3
(Cell Signaling Technology, Danvers, MA, USA) at 4°C overnight.
After washing with phosphate-buffered saline (PBS), the blotted
membrane was incubated with HRP-conjugated anti-mouse or
anti-rabbit IgG (1:2,000) (RiboBio) at room temperature for 2 h.
β-actin was used as an internal control. The signal was detected as
previously described (15).
Immunohistochemical (IHC) assay
IHC staining of xeno-graft tumor slices was
performed according to published methods. Then, 3-µm-thick slices
were incubated with diluted primary antibody against Bcl-2 (Santa
Cruz Biotechnology) at 4°C overnight. After removing the residual
primary antibody, HRP-polymer-conjugated secondary antibody was
applied at 37°C for 1 h. Next, slices were counterstained with
hematoxylin. Three fields were selected for quantification of
percentages of positive tumors and the staining intensity.
Statistical analysis
Data are presented as means ± SEM. Differences among
groups were detected with the Student's t-test and ANOVA. P-values
<0.05 were considered to indicate a statistically significant
result.
Results
Expression of miR-205 decreases in ACC
tissues
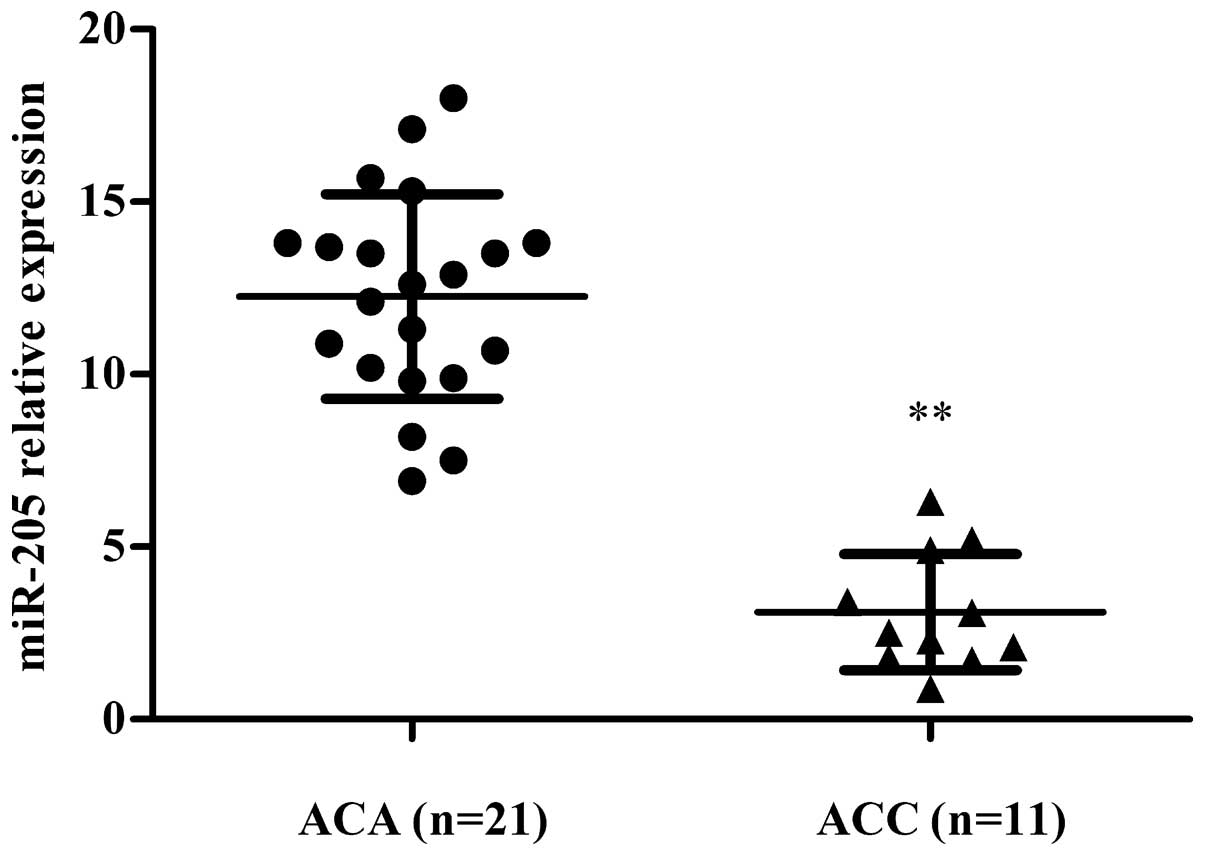
To understand the role of miR-205 in ACC, expression
of miR-205 was measured in both ACC (n=11) and ACA tissues (n=21)
using qRT-PCR. We noted less expression of miR-205 in ACC tissues
compared to ACAs (P=0.008) (Fig.
1). Thus, miR-205 has a tumor-suppressor role in ACC.
miR-205 reduces proliferation and induces
apoptosis of SW-13 cells
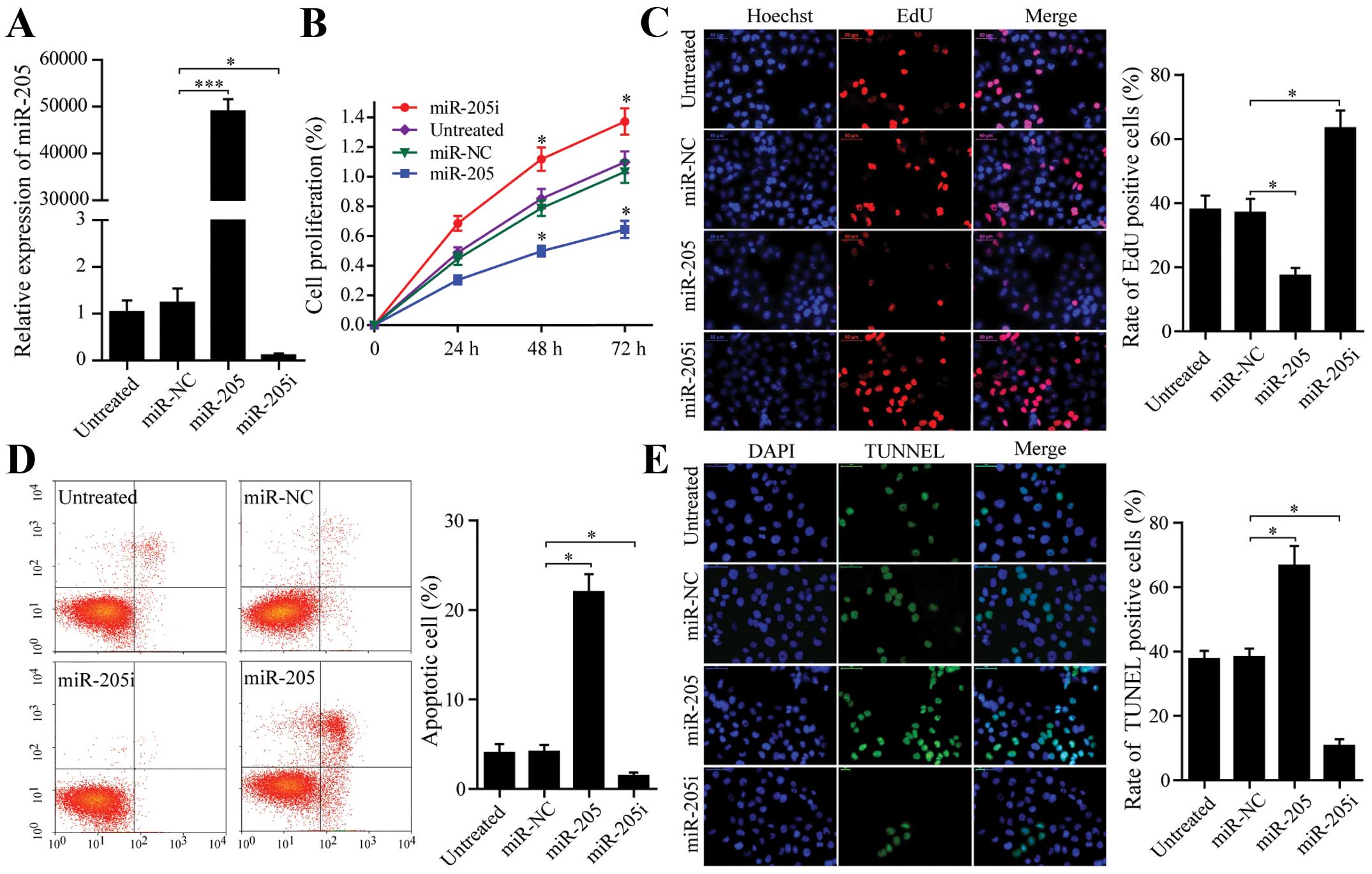
SW-13 cells were transfected with miR-205 mimics,
miR-NC and miR-205 inhibitors and miR-205 expression was measured
by qRT-PCR (Fig. 2A). In an MTT
assay, significantly decreased proliferation was observed over time
in cells expressing miR-205 compared with cells expressing miR-NC.
miR-205 inhibitors promoted cell proliferation as well (P<0.05,
Fig. 2B). In addition, an EdU assay
was used to measure effectiveness of miR-205 on SW-13 cell
proliferation. EdU, acting as a thymidine analogue replaces thymine
(T) in replicating DNA (16). We
observed that SW-13 cells incorporating EdU in the miR-205 group
were significantly decreased when compared to the miR-NC group. In
contrast, miR-205 inhibitors increased SW-13 cell DNA replication
(P<0.05, Fig. 2C). Thus, SW-13
DNA replication was inversely related to miR-205 expression.
To identify the cause of reduced cell proliferation,
we measured apoptosis using Annexin V/PI and TUNEL assays. The
proportion of apoptotic SW-13 cells transfected with miR-205 was
greater than the miR-NC group. miR-205 inhibitors decreased
apoptotic proportions according to the Annexin V/PI assays
(P<0.05, Fig. 2D). Similarly,
SW-13 apoptosis was significantly increased due to miR-205 and was
reduced due to miR-205 inhibitor according to TUNEL assay data
(Fig. 2E, P<0.05). Thus,
inhibition of SW-13 cell proliferation by miR-205 was associated
with increased apoptosis.
miR-205 inhibits tumor growth in
vivo
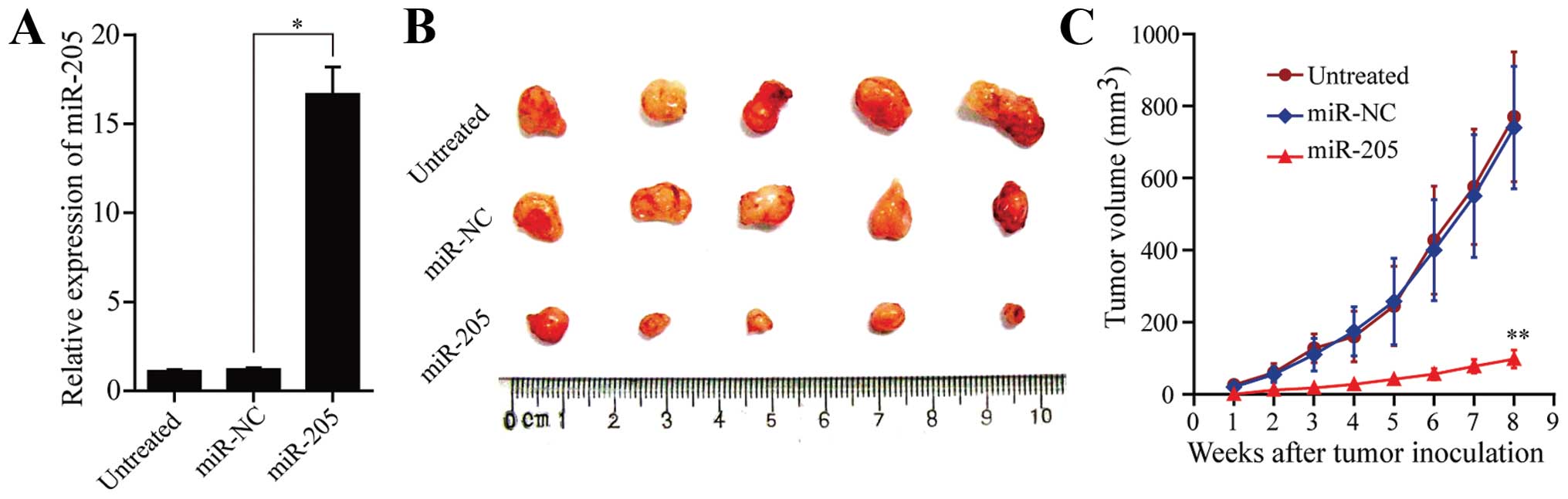
To investigate the effect of miR-205 on tumor
formation, we established SW-13 cells stably overexpressing miR-205
by the eukaryotic expression vector pcDAN3.1(+). Then, miR-205
expression was measured by qRT-PCR (Fig. 3A). Tumors in the subcutaneous nude
mouse model were palpable in ~7 days in the miR-NC and untreated
groups, whereas tumors in the miR-205 group were palpable ~2 weeks
after inoculation. All mice developed tumors at the end of the
experiment (Fig. 3B). The mean
tumor volume of the miR-205 group was reduced by >80% when
compared to the miR-NC group (P<0.05, Fig. 3C). miR-NC and untreated animals were
not different with respect to tumor volume. Thus, elevated miR-205
in SW-13 cells reduced their ability to form tumors and miR-205
suppresses proliferation of SW-13 cells in the animal model.
miR-205 induces SW-13 cell apoptosis by
targeting the 3′UTR of Bcl-2
To learn how miR-205 induces SW-13 cell apoptosis,
bioinformatics databases were used to predict several target genes
of miR-205, including Lin28, Bak1, Cbx7 and Bcl-2. Bcl-2, an
anti-apoptotic gene that is important to numerous tumors. We
hypothesized that miR-205 promotes apoptosis and inhibits
proliferation of SW-13 cells by targeting the Bcl-2 gene.
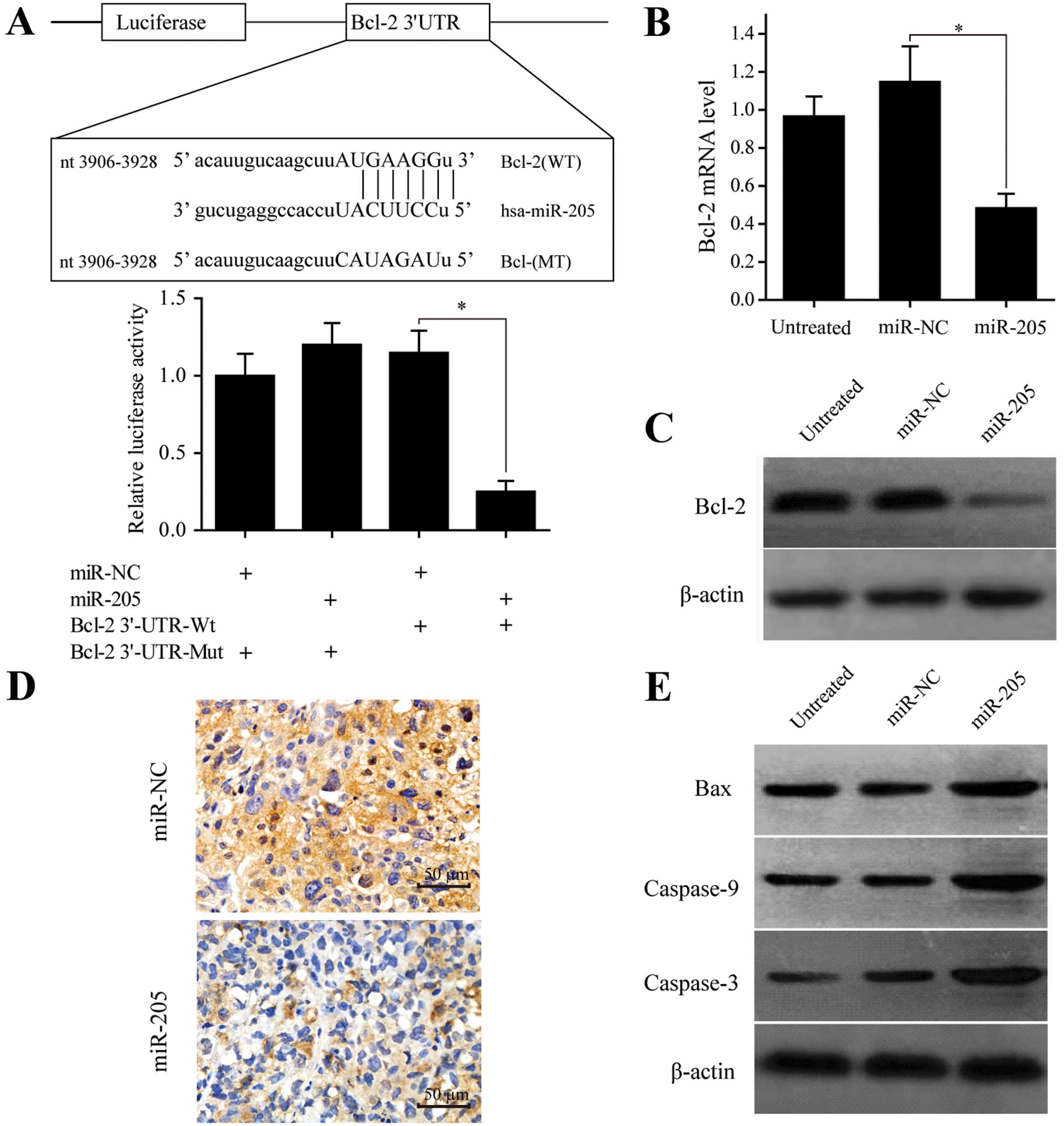
First, the 3′UTR of Bcl-2 containing either a
wild-type or mutant binding sequence complementary to miR-205 was
cloned into a luciferase reporter plasmid (Fig. 4A). miR-205 reduced luciferase
activity of wild-type Bcl-2 (~80%) in SW-13 cells (P<0.05,
Fig. 4B), yet it failed to repress
the mutated one. To estimate whether miR-205 downregulated Bcl-2
expression functionally, qRT-PCR analysis was used to confirm that
stably overexpressing miR-205 reduced mRNA expression of Bcl-2
compared to the miR-NC group (P<0.05, Fig. 4B). Western blotting was used to
quantify expression of Bcl-2 protein due to miR-205 overexpression
(Fig. 4C). IHC staining confirmed
that Bcl-2 expression was negatively correlated with that of the
miR-205 in xenograft tumor tissues (Fig. 4D). Therefore, miR-205 targets Bcl-2
through 3′UTR and regulates mRNA and protein expression.
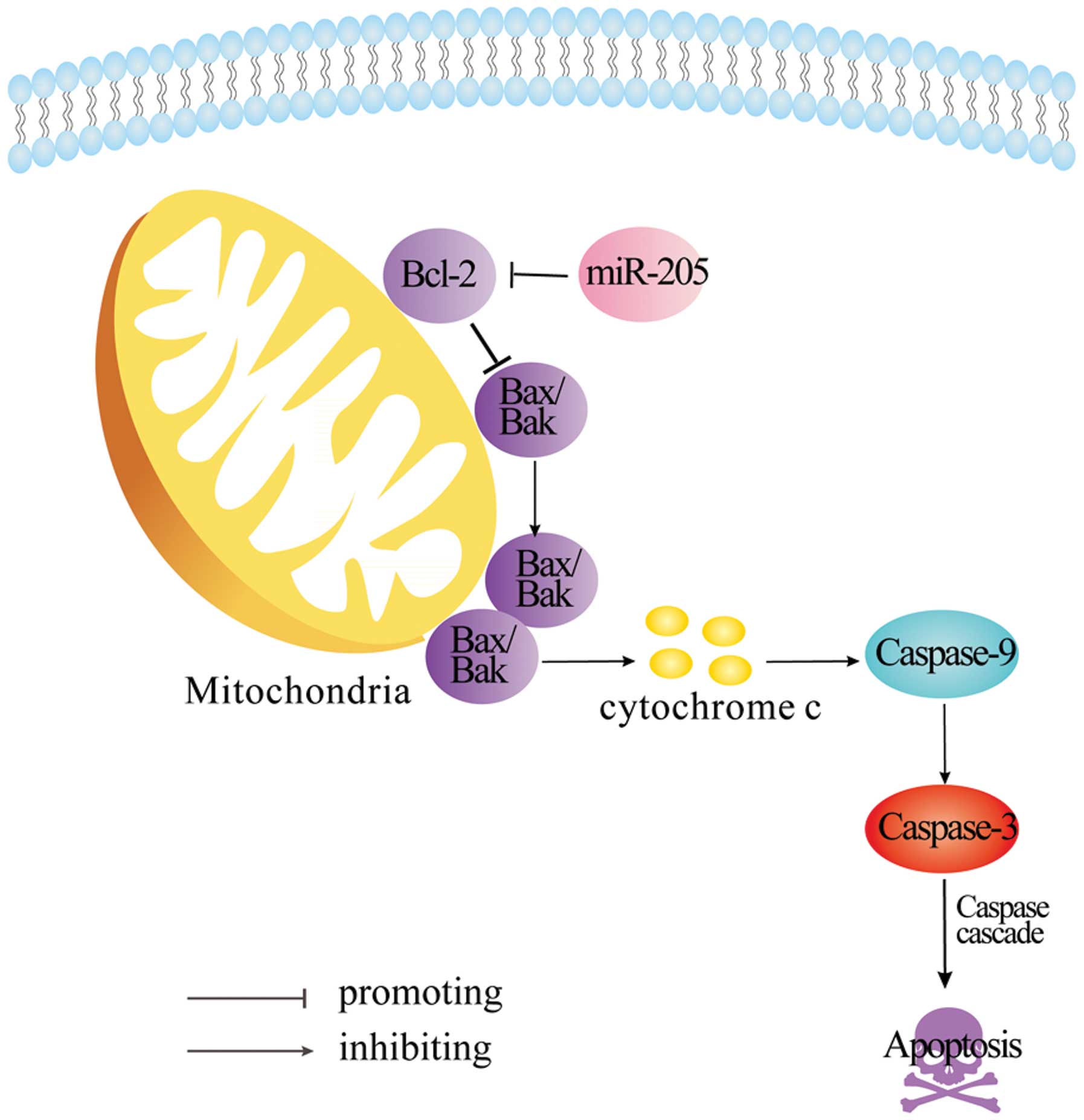
miR-205 inhibits Bcl-2 expression via the
intrinsic apoptotic pathway
Bcl-2 is reported to be a vital anti-apoptotic gene.
Activation of Bcl-2 inhibits apoptosis by cleaving Bax, releasing
cytochrome c, caspase-9 and -3 which are involved in the
intrinsic apoptotic pathway (17).
To determine whether the intrinsic apoptotic pathway is also
activated through miR-205-mediated suppression of Bcl-2 in SW-13
cells, we measured Bax, caspase-9 and -3 expression via western
blotting and noted that these genes were upregulated at the protein
level in miR-205-overexpressing SW-13 cells (Fig. 4E). Therefore, the Bcl-2-mediated
intrinsic apoptotic pathway plays an important role in inducing
SW-13 cell apoptosis due to miR-205.
Discussion
Research shows that miRNAs are critical regulators
in various biological pathways and pathologic processes, including
initiation and progression of cancer (9). miRNAs in tumors are thought also to
modulate oncogenic or tumor suppressive pathways (18). Recently, the expression of miRNAs
was confirmed in adrenocortical tumors and the goal was to identify
miRNAs to differentiate adenomas from carcinomas. For instance,
miR-483 was upregulated and miR-195 was downregulated in ACC
compared with ACA (4,19), indicating a potential value of
miRNAs in diagnosing ACC. Despite these advances, little is known
about the composition and characteristic of miRNAs in ACC, and few
targets of miRNAs in ACC have been identified.
We reported that miR-205 is downregulated in ACC
compared to ACA, and overexpression of miR-205 not only induced
apoptosis and impaired proliferation of SW-13 cells, but also
repressed tumor growth in vivo. Thus, miR-205 functions as
an anti-oncogene in ACC. The role of miRNAs in tumors depends on
the tumor origin and tissue specificity. The known functions of
miR-205 include inhibition of epithelial to mesenchymal transition
(EMT) via significant suppression of E-cadherin, which is related
to metastasis (20,21). In contrast, miR-205 regulated target
genes by direct cleavage of mRNA or inhibition of protein synthesis
through complementarity with 3′UTR. At this time, few genes have
been identified as targets of miR-205, including anti-oncogenes
PTEN (22) and SHIP2 (23), the oncogenes HER3 (24) and PKCε (25), and the angiogenic factor VEGFA
(26), and finally the
pro-metastatic genes Zeb1 and Zeb2 (27). In the present study, we propose that
the Bcl-2 gene is a target of miR-205 through bioinformatics
prediction software, and these data were verified by luciferase
assay. mRNA and protein expression of Bcl-2 were negatively related
to miR-205 in SW-13 cells and xenograft tumor tissues, suggesting
that miR-205 inhibits tumor progression through targeting the 3′UTR
of Bcl-2 in ACC. In addition, miR-205 was a tumor-suppressing gene
by targeting Bcl-2 which has been documented in prostate cancer
(28).
In the embryo stage, the fetal cortex accounts for
85% of the adrenal cortex but disappears quickly due to apoptosis
after birth (29,31). Thus, it was speculated that
similarities may exist between normal cortex and ACC which
originated from defective apoptosis of the fetal cortex (31–33).
Consequently, the research on apoptosis-related genes could be of
great significance not only to provide new views to the biology of
ACC, yet also to search potential prognostic and therapeutic
markers. Bcl-2 predominantly localizes to the mitochondria which is
the central coordinator of the apoptotic pathway (34). Bcl-2 is reported to vary from ACC to
ACA at the mRNA expression level (35), yet the mechanism of Bcl-2 action is
unclear. In the present study, we found that miR-205 is an upstream
regulator inhibiting Bcl-2 expression. Also, the intrinsic
apoptotic pathway was activated and this promoted caspase-dependent
apoptosis in SW-13 cells (Fig. 5).
Notably, other miRNAs target Bcl-2 to induce cancer-cell apoptosis,
among them, miR-15/miR-16 and miR-148a negatively regulated Bcl-2
expression to induce leukemic and colorectal cancer cell apoptosis,
respectively (36,37). In addition, miR-181b modulates
multidrug resistance by targeting Bcl-2 in lung cancer cell lines
(38). Thus, miR-205 with other
miRNAs, targets Bcl-2 to create a network to regulate tumor cell
death.
In conclusion, miRNA-205 serves as a
tumor-suppressor in ACC by targeting anti-apoptotic Bcl-2, which
induces SW-13 cell apoptosis via the intrinsic apoptotic pathway.
Our results may provide a foundation for considering miR-205 as a
biomarker to aid in the diagnosis and treatment of ACC. Extensive
studies are required to confirm our findings and to validate the
diagnostic accuracy of these miRNAs.
Acknowledgments
The present study was supported by the China
National Natural Science Foundation (nos. 81172421 and 81372744),
and the Guangdong Province Natural Science Foundation of China (no.
S2012010010009).
References
|
1
|
Lehmann T and Wrzesinski T: The molecular
basis of adrenocortical cancer. Cancer Genet. 205:131–137. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bilimoria KY, Shen WT, Elaraj D, Bentrem
DJ, Winchester DJ, Kebebew E and Sturgeon C: Adrenocortical
carcinoma in the United States: Treatment utilization and
prognostic factors. Cancer. 113:3130–3136. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kutikov A, Mallin K, Canter D, Wong YN and
Uzzo RG: Effects of increased cross-sectional imaging on the
diagnosis and prognosis of adrenocortical carcinoma: Analysis of
the National Cancer Database. J Urol. 186:805–810. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Patterson EE, Holloway AK, Weng J, Fojo T
and Kebebew E: MicroRNA profiling of adrenocortical tumors reveals
miR-483 as a marker of malignancy. Cancer. 117:1630–1639. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chiodini I: Clinical review: Diagnosis and
treatment of subclinical hypercortisolism. J Clin Endocrinol Metab.
96:1223–1236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kuruba R and Gallagher SF: Current
management of adrenal tumors. Curr Opin Oncol. 20:34–46. 2008.
View Article : Google Scholar
|
|
7
|
Maas S: Gene regulation through RNA
editing. Discov Med. 10:379–386. 2010.PubMed/NCBI
|
|
8
|
Garzon R, Marcucci G and Croce CM:
Targeting microRNAs in cancer: Rationale, strategies and
challenges. Nat Rev Drug Discov. 9:775–789. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Etheridge A, Lee I, Hood L, Galas D and
Wang K: Extracellular microRNA: A new source of biomarkers. Mutat
Res. 717:85–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Iorio MV and Croce CM: MicroRNAs in
cancer: Small molecules with a huge impact. J Clin Oncol.
27:5848–5856. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ferracin M, Veronese A and Negrini M:
Micromarkers: miRNAs in cancer diagnosis and prognosis. Expert Rev
Mol Diagn. 10:297–308. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gilad S, Meiri E, Yogev Y, Benjamin S,
Lebanony D, Yerushalmi N, Benjamin H, Kushnir M, Cholakh H, Melamed
N, et al: Serum microRNAs are promising novel biomarkers. PLoS One.
3:e31482008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kang MH and Reynolds CP: Bcl-2 inhibitors:
Targeting mitochondrial apoptotic pathways in cancer therapy. Clin
Cancer Res. 15:1126–1132. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aubert S, Wacrenier A, Leroy X, Devos P,
Carnaille B, Proye C, Wemeau JL, Lecomte-Houcke M and Leteurtre E:
Weiss system revisited: A clinicopathologic and immunohistochemical
study of 49 adrenocortical tumors. Am J Surg Pathol. 26:1612–1619.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang B, Li Z, Zhang W, Wang H, Zhi X,
Feng J, Chen Z, Zhu Y, Yang L, Xu H, et al: miR-874 inhibits cell
proliferation, migration and invasion through targeting aquaporin-3
in gastric cancer. J Gastroenterol. 49:1011–1025. 2014. View Article : Google Scholar
|
|
16
|
Chehrehasa F, Meedeniya AC, Dwyer P,
Abrahamsen G and Mackay-Sim A: EdU, a new thymidine analogue for
labelling proliferating cells in the nervous system. J Neurosci
Methods. 177:122–130. 2009. View Article : Google Scholar
|
|
17
|
Skommer J, Brittain T and Raychaudhuri S:
Bcl-2 inhibits apoptosis by increasing the time-to-death and
intrinsic cell-to-cell variations in the mitochondrial pathway of
cell death. Apoptosis. 15:1223–1233. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Suzuki H, Maruyama R, Yamamoto E and Kai
M: DNA methylation and microRNA dysregulation in cancer. Mol Oncol.
6:567–578. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Soon PS, Tacon LJ, Gill AJ, Bambach CP,
Sywak MS, Campbell PR, Yeh MW, Wong SG, Clifton-Bligh RJ, Robinson
BG, et al: miR-195 and miR-483–5p identified as predictors of poor
prognosis in adrenocortical cancer. Clin Cancer Res. 15:7684–7692.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gregory PA, Bert AG, Paterson EL, Barry
SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y and Goodall GJ:
The miR-200 family and miR-205 regulate epithelial to mesenchymal
transition by targeting ZEB1 and SIP1. Nat Cell Biol. 10:593–601.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fassina A, Cappellesso R, Guzzardo V,
Dalla Via L, Piccolo S, Ventura L and Fassan M:
Epithelial-mesenchymal transition in malignant mesothelioma. Mod
Pathol. 25:86–99. 2012. View Article : Google Scholar
|
|
22
|
Greene SB, Herschkowitz JI and Rosen JM:
The ups and downs of miR-205: Identifying the roles of miR-205 in
mammary gland development and breast cancer. RNA Biol. 7:300–304.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yu J, Ryan DG, Getsios S,
Oliveira-Fernandes M, Fatima A and Lavker RM: MicroRNA-184
antagonizes microRNA-205 to maintain SHIP2 levels in epithelia.
Proc Natl Acad Sci USA. 105:19300–19305. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Iorio MV, Casalini P, Piovan C, Di Leva G,
Merlo A, Triulzi T, Ménard S, Croce CM and Tagliabue E:
microRNA-205 regulates HER3 in human breast cancer. Cancer Res.
69:2195–2200. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gandellini P, Folini M, Longoni N, Pennati
M, Binda M, Colecchia M, Salvioni R, Supino R, Moretti R, Limonta
P, et al: miR-205 exerts tumor-suppressive functions in human
prostate through downregulation of protein kinase Cepsilon. Cancer
Res. 69:2287–2295. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wu H, Zhu S and Mo YY: Suppression of cell
growth and invasion by miR-205 in breast cancer. Cell Res.
19:439–448. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gregory PA, Bert AG, Paterson EL, Barry
SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y and Goodall GJ:
The miR-200 family and miR-205 regulate epithelial to mesenchymal
transition by targeting ZEB1 and SIP1. Nat Cell Biol. 10:593–601.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Verdoodt B, Neid M, Vogt M, Kuhn V,
Liffers ST, Palisaar RJ, Noldus J, Tannapfel A and
Mirmohammadsadegh A: MicroRNA-205, a novel regulator of the
anti-apoptotic protein Bcl2, is downregulated in prostate cancer.
Int J Oncol. 43:307–314. 2013.PubMed/NCBI
|
|
29
|
Mesiano S and Jaffe RB: Role of growth
factors in the developmental regulation of the human fetal adrenal
cortex. Steroids. 62:62–72. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
West AN, Neale GA, Pounds S, Figueredo BC,
Rodriguez Galindo C, Pianovski MA, Oliveira Filho AG, Malkin D,
Lalli E, Ribeiro R, et al: Gene expression profiling of childhood
adrenocortical tumors. Cancer Res. 67:600–608. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Spencer SJ, Mesiano S, Lee JY and Jaffe
RB: Proliferation and apoptosis in the human adrenal cortex during
the fetal and perinatal periods: Implications for growth and
remodeling. J Clin Endocrinol Metab. 84:1110–1115. 1999.PubMed/NCBI
|
|
32
|
Coulter CLL: Fetal adrenal development:
Insight gained from adrenal tumors. Trends Endocrinol Metab.
16:235–242. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
El Wakil A, Doghman M, Latre De Late P,
Zambetti GP, Figueiredo BC and Lalli E: Genetics and genomics of
childhood adrenocortical tumors. Mol Cell Endocrinol. 336:169–173.
2011. View Article : Google Scholar
|
|
34
|
Plati J, Bucur O and Khosravi-Far R:
Apoptotic cell signaling in cancer progression and therapy. Integr
Biol Camb. 3:279–296. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Fonseca AL, Kugelberg J, Starker LF,
Scholl U, Choi M, Hellman P, Åkerström G, Westin G, Lifton RP,
Björklund P, et al: Comprehensive DNA methylation analysis of
benign and malignant adrenocortical tumors. Genes Chromosomes
Cancer. 51:949–960. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cimmino A, Calin GA, Fabbri M, Iorio MV,
Ferracin M, Shimizu M, Wojcik SE, Aqeilan RI, Zupo S, Dono M, et
al: miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl
Acad Sci USA. 102:13944–13949. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang H, Li Y, Huang Q, Ren X, Hu H, Sheng
H and Lai M: MiR-148a promotes apoptosis by targeting Bcl-2 in
colorectal cancer. Cell Death Differ. 18:1702–1710. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhu W, Shan X, Wang T, Shu Y and Liu P:
miR-181b modulates multidrug resistance by targeting BCL2 in human
cancer cell lines. Int J Cancer. 127:2520–2529. 2010. View Article : Google Scholar : PubMed/NCBI
|