Introduction
Pigmented villonodular synovitis (PVNS); also known
as tenosynovial giant cell tumor (TGCT) is a rare synovial
proliferative disease with an incidence of 1.8 cases/million
(1–5). Although the etiology of PVNS is
unclear at present, recent evidence suggests a neoplastic nature
(6–8), and malignant cases have been reported
(9,10). The disease, which can involve all
synovial-lined structures, particularly the knee joint, is
characterized by proliferation of synovial tissues and invasion of
cartilage and bone around the joints. PVNS causes clinical swelling
and pain, and the diffuse form or recurrence can trigger rapid
articular cartilage damage associated with severe functional
impairment, eventually leading to joint replacement or even
amputation (11–14). Although surgical synovectomy, either
via open surgery or arthroscopy, is considered the standard
treatment for PVNS (7,15), post-operative complications and
limitations, including longer hospitalization and rehabilitation
periods, post-operative stiffness, and in particular, high rates of
recurrence are common. The post-operative recurrence rate is as
high as 60% (12,16,17).
Imatinib mesylate (IM), a tyrosine kinase inhibitor,
is the first-line drug of choice for chronic myeloid leukemia
(18) and a gastrointestinal
stromal cell tumor. A number of recent studies have reported a
significant clinical effect of IM on PVNS (1,19–21).
The effect of IM on PVNS was initially documented by Blay et
al (1). In a subsequent
multi-institutional retrospective study on 27 patients (74% with
stable disease) by Cassier et al (19), treatment with IM led to symptomatic
improvement in 73% patients. However, the antitumor mechanism of IM
on PVNS to date is not clear. Therefore, in the present study, we
examined whether IM exerts cellular antiproliferative effects and
induces apoptosis in PVNS synovial cells. We further determined
whether cellular apoptosis induced by IM is dependent on the
mitochondrial apoptosis signaling pathways.
Materials and methods
Antibodies and reagents
IM was purchased from Selleck Chemicals (Houston,
TX, USA). Z-LEHD-FMK, a caspase-9 specific inhibitor, was purchased
from R&D Systems (Minneapolis, MN, USA). Dulbecco's modified
Eagle's medium (DMEM) high glucose medium and fetal bovine serum
(FBS) were obtained from gibco (Gaithersburg, MD, USA). Trypsin and
MTT were purchased from Sigma (St. Louis, MO, USA). Rabbit
monoclonal antibody to caspase-3 and -9 were purchased from Abcam
(Cambridge, UK). Rabbit polyclonal antibody to Bcl-2 and Bax were
purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Cell isolation and culture
PVNS fibroblast-like synoviocytes (PVNS-FLS) were
isolated from synovial tissues obtained from PVNS patients at the
time of knee joint synovectomy. All the patients were biopsy-proven
and diagnosis was made based on conventional histological criteria.
The present study was approved by the Medical Ethics Committee of
the First Affiliated Hospital of China Medical University and
written informed consent was obtained from all patients. FLS were
purified from synovial tissue as previously published (22). Briefly, the tissue samples were
minced and digested with 2 mg/ml collagenase II for 4 h and then
with trypsin for 30 min at 37°C, filtered through a 70 µm
cell strainer, and cultured in DMEM containing 10% FBS, 100 U/ml
penicillin and 100 mg/ml streptomycin in a humidified 5%
CO2 incubator. After overnight culture, non-adherent
cells were removed by medium exchange, and adherent cells were
trypsinized and passaged, and used between passages 3 and 7.
Analysis of cell proliferation and
viability
The viability of PVNS-FLS cells was evaluated by the
MTT [3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium
bromide] method. Cells/well (8×103) of FLS were
incubated in a 96-well plate for 24 h, then cells were treated with
different concentrations of IM with or without the caspase-9
specific inhibitor Z-LEHD-FMK, each concentration for parallel
6-wells. After incubating for another 24, 48 and 72 h, 20 µl
MTT solution was added to each well. After 4 h, the medium was
discarded and 150 µl DMSO was added to dissolve the formazan
crystal. The absorbance was measured with a wavelength of 490 nm.
Survival rate of cells (%) = experimental group A value/control
group A value × 100%.
Morphology of apoptotic cells
Cells were seeded into a 6-well plate and pretreated
with or without the caspase-9 inhibitor Z-LEHD-FMK at 50 µM
for 2 h, then 40 µM IM was added in the test group and
cultured for another 48 h. Changes in cell morphology,
proliferation and ability of adherence were observed and compared
under an inverted phase contrast microscope.
Apoptosis of FLS detected by Hoechst
staining
FLS were seeded on aseptic cover slides into the
24-well plate and were treated with 40 µM IM in the presence
or absence of Z-LEHD-FMK for 48 h. The cells were washed three
times with phosphate-buffered saline (PBS) and fixed for 10 min,
and then stained with Hoechst 33258 staining solution for 5 min.
The staining of cell nucleus were observed under a fluorescence
microscope. The apoptotic rate (%) = the number of apoptotic
cells/the number of total cells × 100%.
Acridine orange/ethidium bromide (AO/EB)
apoptosis assay
An AO/EB staining method was used to detect
apoptosis-related morphologic changes of FLS. Cells were seeded on
cover slides in a 24-well plate as described, 500 µl of
freshly-prepared dual stain containing 10 mg/ml acridine orange
(AO) and 10 mg/ml ethidium bromide (EB) was added to each well
staining for 10 min in the dark at room temperature. The
morphological changes of cell nucleus were observed under a
fluorescence microscope and apoptotic rate was calculated.
Annexin V/PI apoptosis detection
After induction of apoptosis, FLS were detached by
trypsinization and washed twice with cold PBS. The cells were
centrifuged at 500 × g for 5 min and resuspended in 400 µl
binding buffer, then 5 µl Annexin V-FITC and 10 µl
propidium iodide (PI) were added and incubated in the dark at room
temperature for 15 min. The samples were analyzed by a FACSCalibur
(BD Biosciences) flow cytometer immediately and the data were
analyzed using CellQuest software.
Cell cycle analysis
After treated with different doses of imatinib
(10–40 µM) for 24 h, cells were washed twice with cold PBS
and fixed in 70% ethanol for 24 h at 4°C before analysis. Then
cells were centrifuged and stained with 50 µg/ml PI and 100
µg/ml RNase A for 30 min in the dark at room temperature.
The samples were analyzed by BD FACSCalibur flow cytometer and the
data were analyzed using CellQuest software.
Western blot analysis
The methods for western blotting have been described
(23). Briefly, the protein samples
were resolved by SDS-PAGE and transferred to PVDF membranes. After
transfer, the PVDF membranes were blocked with 5% (w/v) non-fat
milk in TBST at room temperature for 2 h, and then incubated with
the primary antibodies in the TBST buffer containing 1% (w/v) BSA
at 4°C for 16 h. Following three washes in TBST, the membranes were
incubated with the appropriate secondary antibody in TBST for 2 h
at room temperature. Proteins were detected by the ECL system.
Determination of the invasion
ability
The invasive ability of PVNS-FLS was evaluated by
the Transwell invasion system. The chamber was washed with
serum-free medium, then 20 µl Matrigel was added to cover
the bottom of the chamber. A Matrigel membrane was created by
incubating at 37°C for 30 min. Cells (2×105) with 200
µl serum-free DMEM medium was seeded in the upper chamber of
the Transwell invasion system and 500 µl DMEM with 10% FBS
was added into the lower chamber. The Transwell invasion system was
incubated in a cell culture incubator for 24 h, and then the upper
chamber was taken out and the cells removed from the upper surface
of the membrane. Then cells invaded to the lower surface of the
membrane were stained with 0.1% crystal violet and observed under
an inverted phase contrast microscope. The results are presented as
the mean ± SD, with three repeated experiments for each group.
Statistical analysis
Statistical analyses were performed using one-way
analysis of variance (one-way ANOVA) and Bonferroni's multiple
comparisons post tests were used as needed. Data are presented as
the mean ± SD from a minimum of three independent experiments
performed in triplicate. P<0.05 was considered to indicate a
statistically significant result.
Results
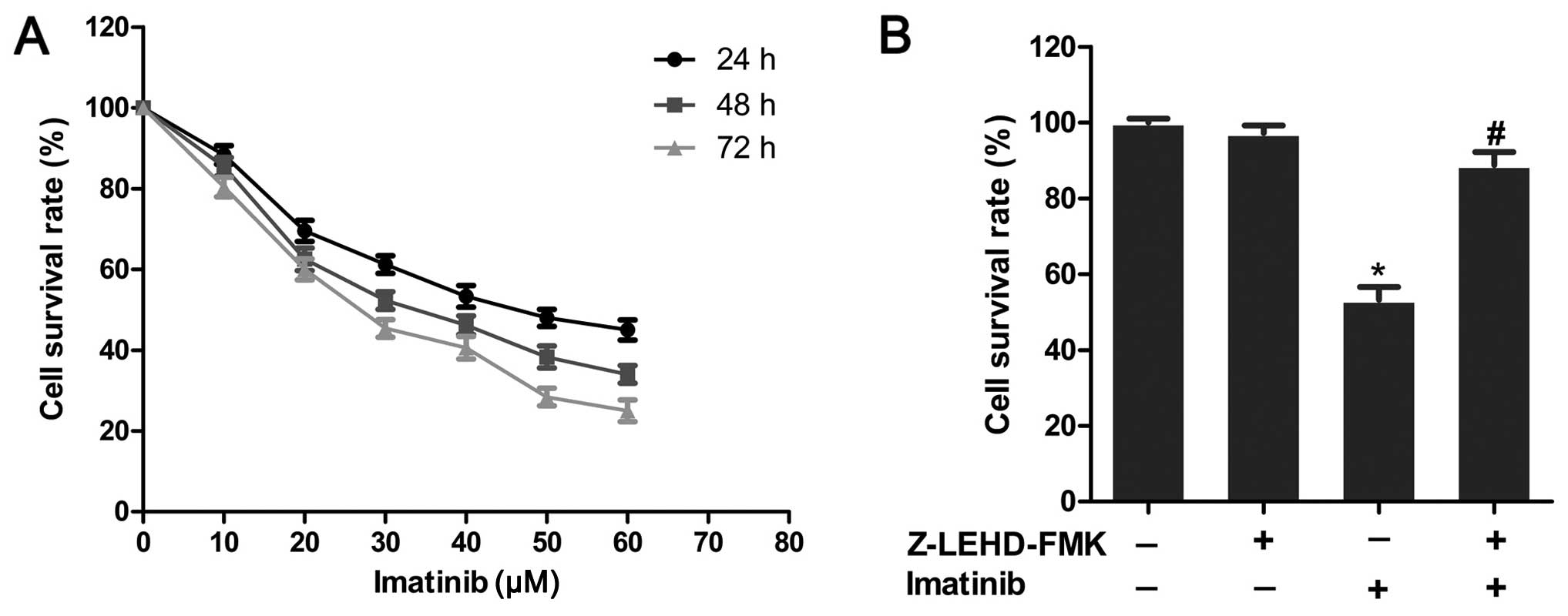
IM reduces the viability of PVNS-FLS
To determine the effects of IM on proliferation of
PVNS-FLS, the MTT assay was carried out with different
concentrations of IM (10–60 µM) for 24, 48 and 72 h. As
shown in Fig. 1A, IM significantly
inhibited the survival of PVNS-FLS cells in a dose- and
time-dependent manner. The IC50 value for 48 h was ~40
µM, which was taken as the effective drug concentration in
subsequent experiments. Upon pre-treatment of cells with the
caspase-9-specific inhibitor Z-LEHD-FMK (50 µM), the
antiproliferative effect was blocked (P<0.05), and survival
rates were markedly enhanced (Fig.
1B). We observed no differences in survival rates (P>0.05),
compared with the control group, upon the addition of Z-LEHD-FMK
alone, suggesting a critical role of caspase-9 in IM-induced
apoptosis of PVNS-FLS.
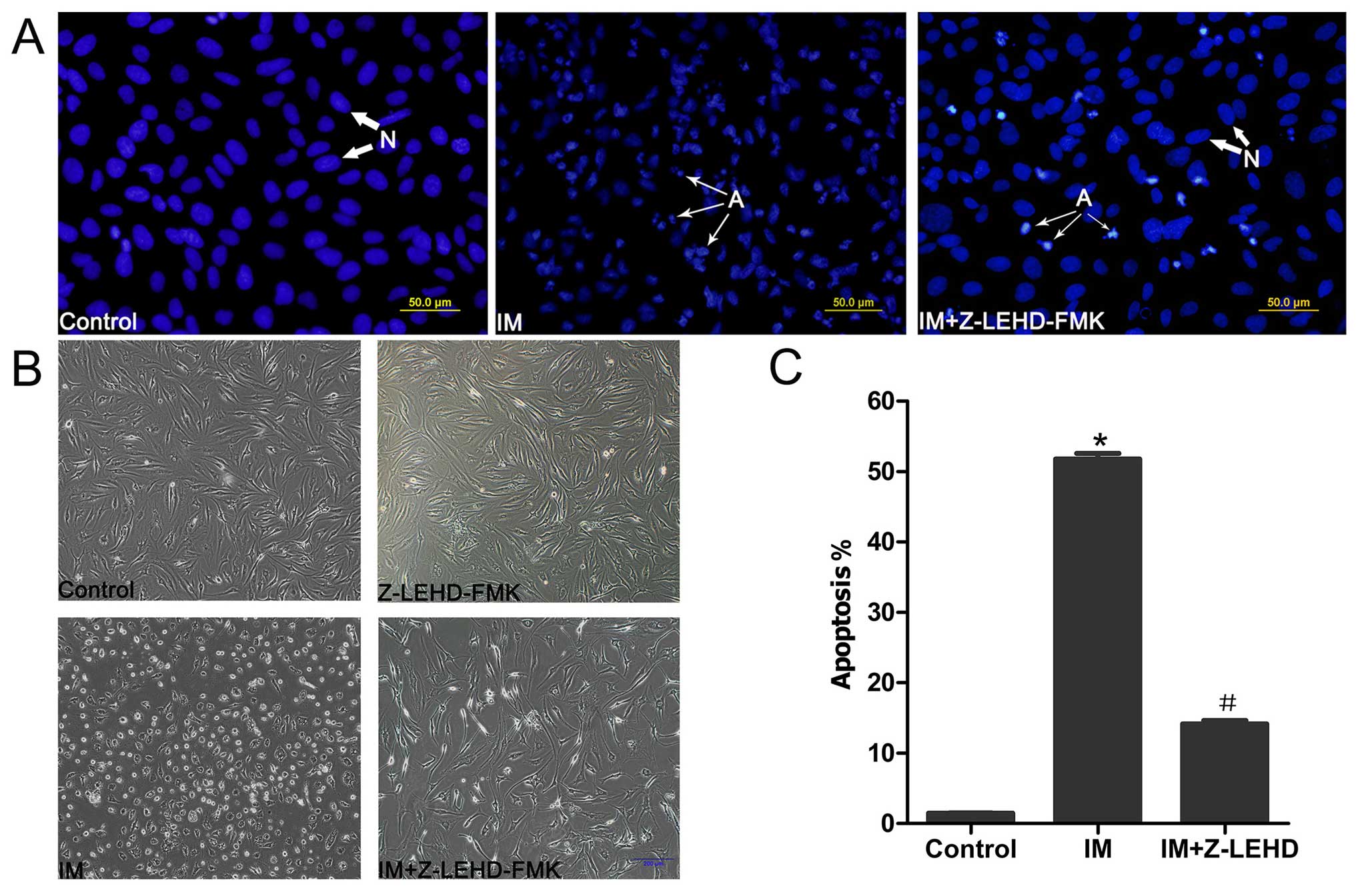
Morphological changes of apoptotic
cells
Morphological changes in FLS after apoptosis
induction with IM were examined under an inverted phase-contrast
microscope. Cells in the control and Z-LEHD-FMK groups presented a
slender fiber-like appearance and attached uniformly to the dish.
In contrast, in the drug-treated groups, the cell shape became
small and round, with some cells being non-adherent and suspended
in medium (Fig. 2B). To further
resolve the morphology of apoptotic nuclei, Hoechst 33258 staining
was performed. FLS in the control group showed regular round and
normal nuclei with almost no sign of apoptosis. In contrast, cells
treated with IM exhibited condensed, fragmented and crescent-shaped
nuclei characteristic of apoptosis (Fig. 2A). Apoptosis was blocked upon
pretreatment with the caspase-9 inhibitor Z-LEHD-FMK (50
µM). The percentage of apoptosis index was significantly
different between groups. The control group contained a limited
percentage of apoptotic cells (1.45±0.097%), which was markedly
increased in the drug-treated group (51.74±1.75%). After the
addition of Z-LEHD-FMK, the apoptotic rate was markedly decreased
in the IM-treated group (14.14±1.07%; Fig. 2C).
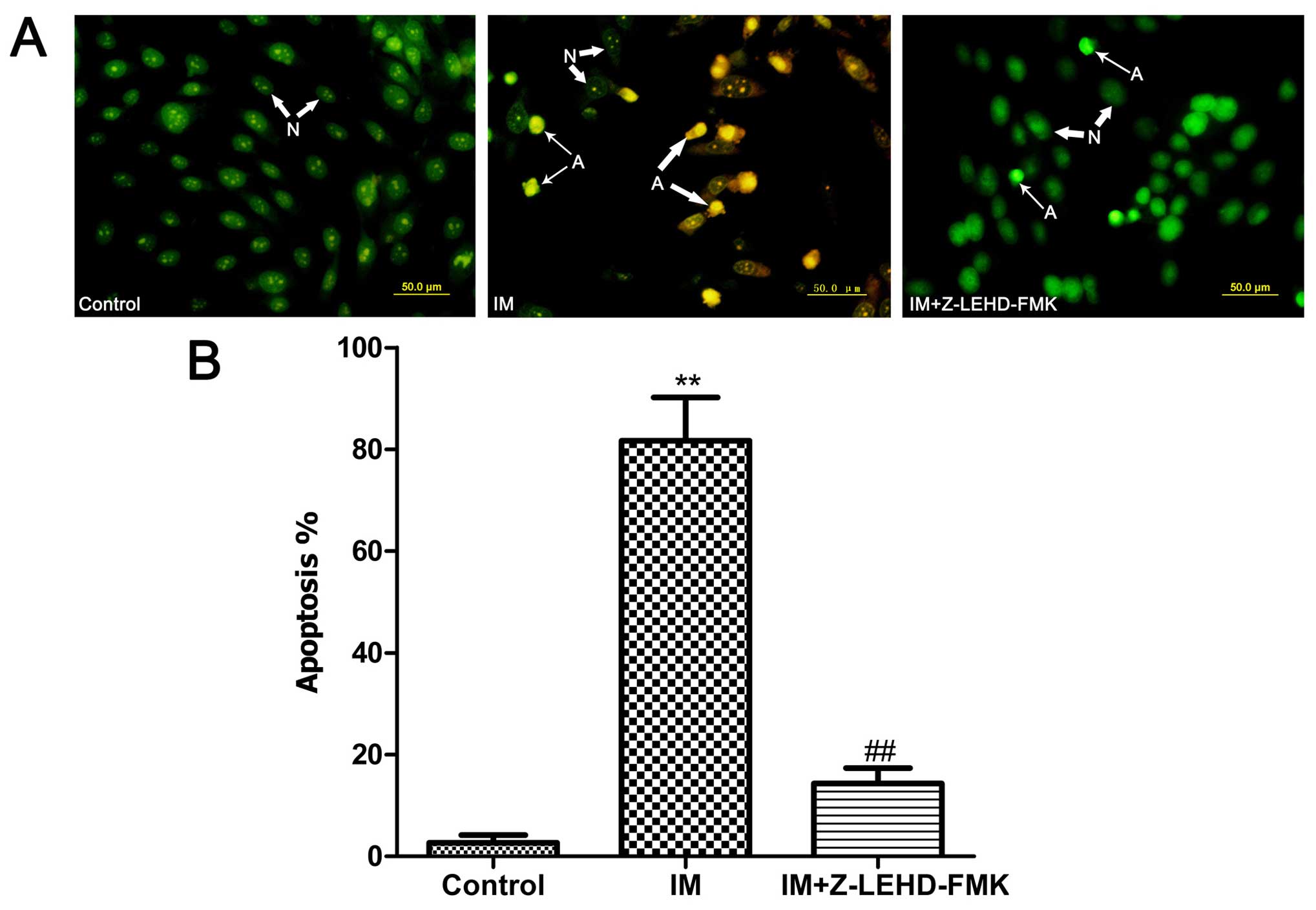
Apoptotic changes of cell nuclei stained
with AO/EB staining
In order to observe the changes in nuclear
morphology of apoptosis more clearly, an AO/EB double staining
method was used. As shown in Fig.
3, after treated with IM, the number of apoptotic cells was
increased greatly, and this trend is blocked with the addition of
Z-LEHD-FMK, which is consistent with the previous results.
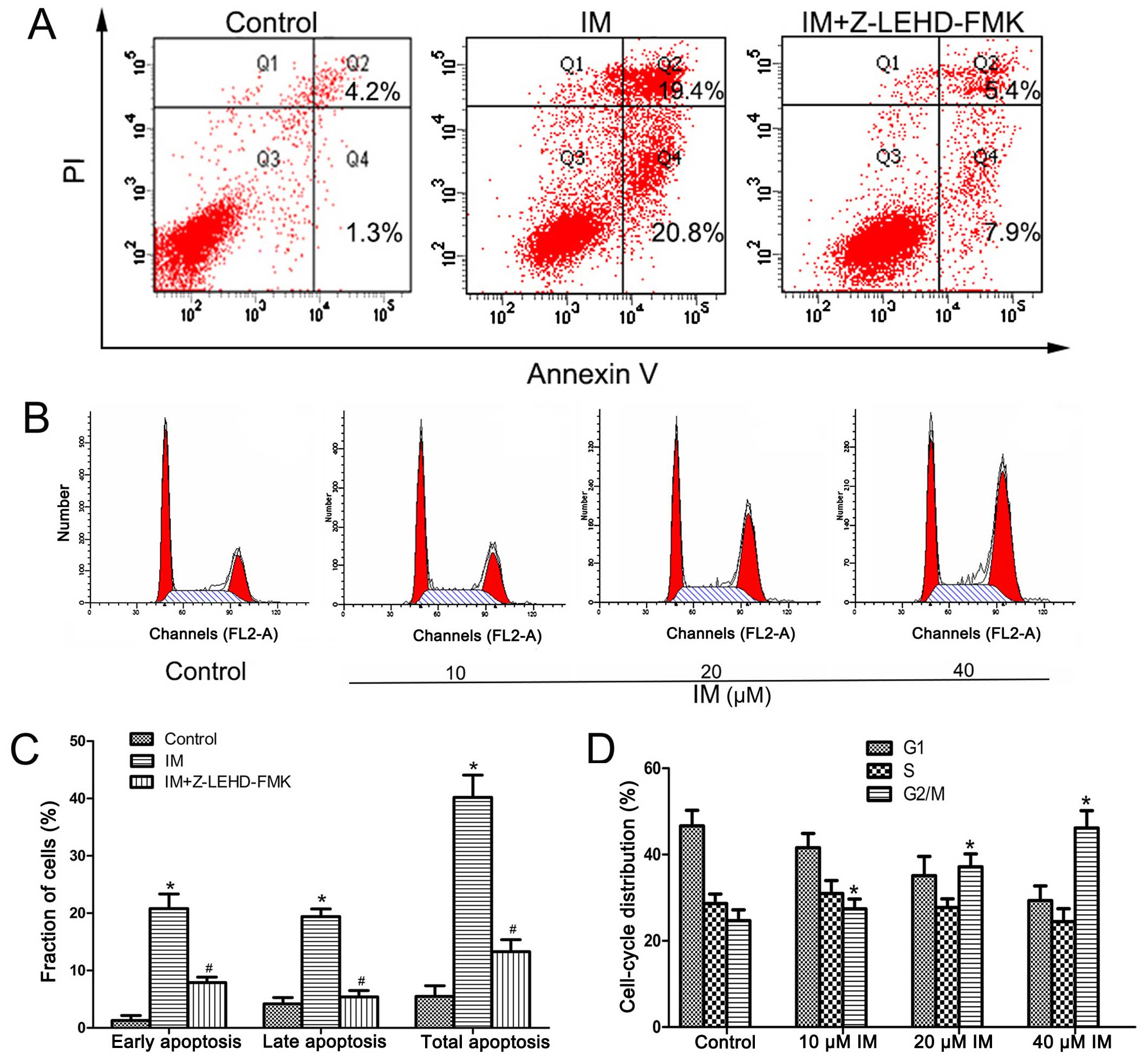
IM induces caspase-9-dependent apoptosis
in PVNS-FLS
The Annexin V/PI staining assay was employed to
further confirm the apoptotic effect of imatinib. As shown in
Fig. 4A, the apoptosis ratio of the
IM-treated group was significantly higher than that of the control
group (P<0.01). The apoptotic cell population was significantly
reduced in the presence of Z-LEHD-FMK (from 41.52±3.87 to
14.25±2.08%) (Fig. 4C), clearly
indicating that apoptosis is induced by IM via a
caspase-9-dependent pathway.
IM induces G2/M arrest in PVNS-FLS
PI staining data revealed that IM causes cell cycle
arrest at the G2/M phase in PVNS-FLS. Upon exposure of cells to
different concentrations of drugs as above, the cell number at the
G2/M phase was substantially increased from 24.68±2.53 to
46.17±4.62% in the 40 µM IM-treated group while the number
of cells at the G1 phase decreased from 46.66±3.66 to 29.35±3.40%
(Fig. 4D).
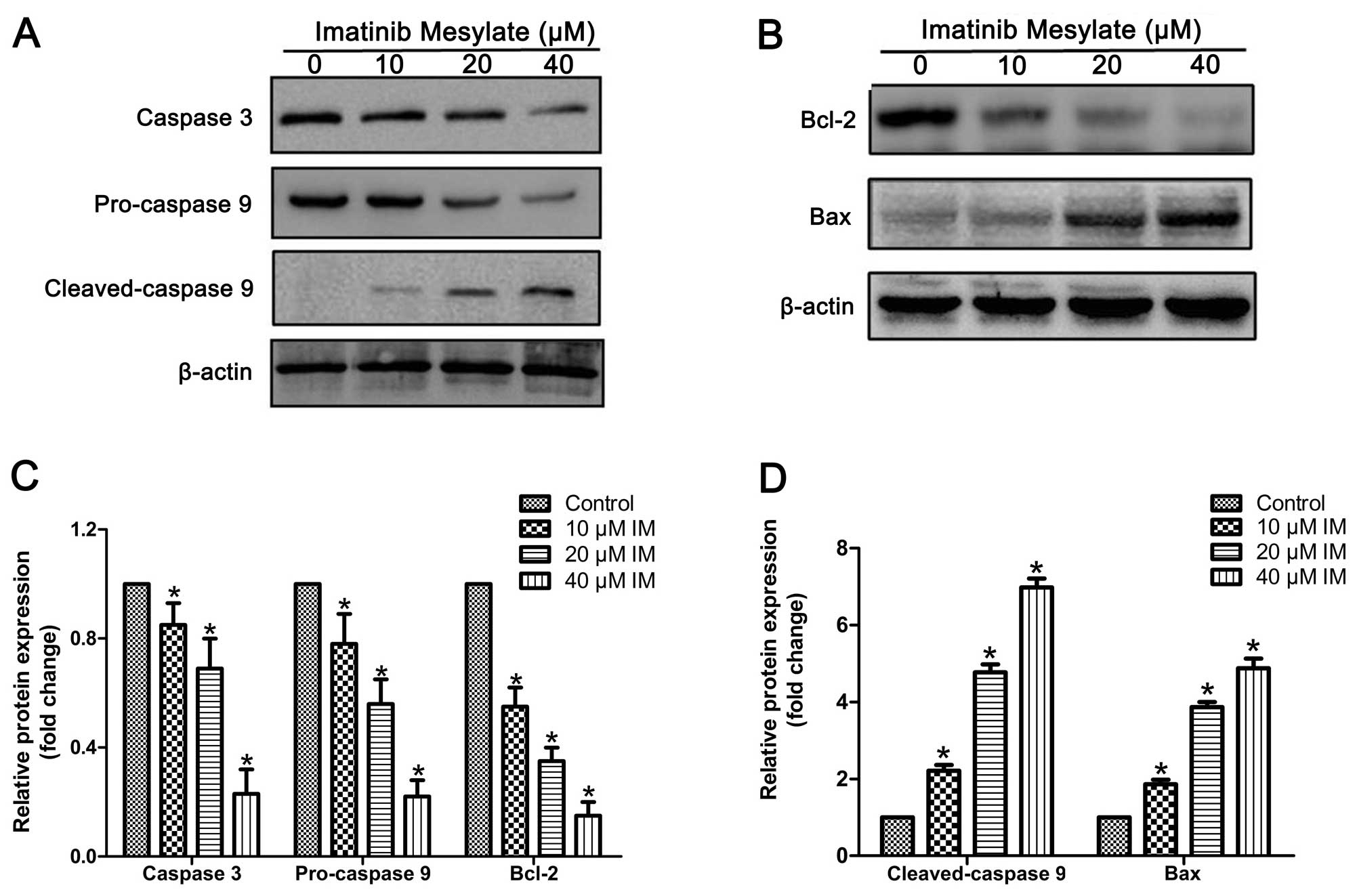
IM activates caspase-9/-3 and the Bcl-2
family in PVNS-FLS
To further clarify the specific signaling pathways
involved in the apoptotic effects of IM, we evaluated the
appropriate protein expression patterns using western blotting. As
shown in Fig. 5, expression of
cleaved caspase-9 (35 kDa) was markedly increased upon IM treatment
in a dose-dependent manner. Conversely, we observed a significant
decrease in pro-caspase-9 and pro-caspase-3 levels, indicating
activation of caspase-9 and -3. The key members of the Bcl-2
family, Bcl-2 and Bax, were analyzed to determine the effects of IM
on expression patterns of upstream apoptotic proteins. Expression
of Bax was considerably increased while that of Bcl-2 was decreased
by IM in a dose-dependent manner.
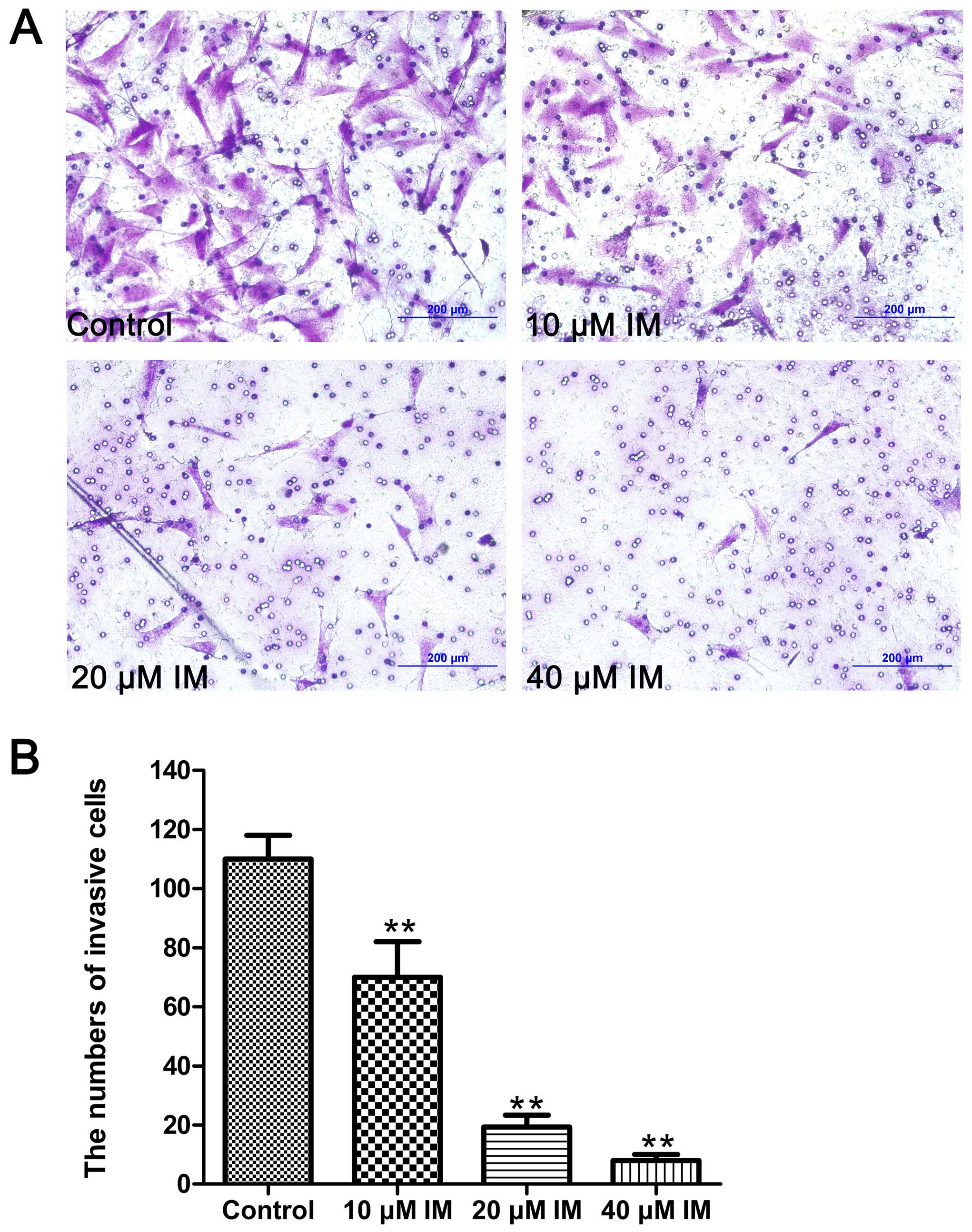
IM inhibits the invasion ability of
PVNS-FLS
A Transwell invasion assay was used to evaluate the
invasion ability of PVNS-FLS after treated with different drugs. As
shown in Fig. 6, cells in the drug
groups that crossed through the polycarbonate membrane of the
Transwell invasion chamber were significantly less than that in the
control group (P<0.01), and showed a dose-dependent manner.
These data indicated that IM had a strong inhibitory effect on the
invasion ability of PVNS-FLS.
Discussion
Pigmented villonodular synovitis (PVNS) is a rare
synovial proliferative disorder, which is almost always
monoarticular, and in 80% of cases, affects the knee joint. The
disorder can present either as a localized (LPVNS) or diffuse form
(DPVNS), the latter being more common (5,24–27).
Although its etiology is unclear, recent studies have suggested a
type of sarcoma. The disease, particularly the diffuse form,
displays several tumor characteristics. PVNS presents as an
aggressive tumor that invades adjacent articular cartilage and
extra-articular tissues and causes rapid, irreversible joint
impairment. Unfortunately, it is almost impossible to excise the
extensive mass in its entirety via synovectomy without causing
joint damage, since almost no boundaries exist between the lesions
and normal tissues. Hence, there remains an urgent unmet medical
need for safe and effective treatments for this disease.
Imatinib mesylate (IM) is a known antitumor agent
for chronic myloid leukemia (CML) (28) and gastrointestinal stromal tumors
(GIST) (29). Recent research has
revealed a critical role in controlling tumor growth and relieving
clinical symptoms of patients with PVNS, supporting its utility as
a novel therapeutic agent for the disease (1,19–21).
Blay et al (1) reported the
effect of IM on a PVNS patient with a relapse of PVNS of the right
elbow for the first time. Complete remission was observed after
treatment with imatinib at a dose of 400 mg/day for 5 months.
Subsequent studies by Ravi et al (20) and Cassier et al (19) further confirmed the therapeutic
effect of imatinib on PVNS in clinic. However, the antitumor
mechanism is unknown and related basic research is lacking.
In the present study, we treated PVNS synovial cells
with different concentrations of IM for specific periods of time.
Survival rates showed a clear dose- and time-dependent trend. After
48 h of treatment, the survival rate of cells was reduced from
85.42±2.37 to 34.0±2.58% with increasing concentrations of IM from
10 to 60 µM. The survival rate declined from 53.37±3.86 to
40.68±2.59% with extended incubation times at a drug concentration
of 40 µM (Fig. 1A). Previous
studies have speculated that the caspase-9-based mitochondrial
apoptotic pathway may play a key role in the cell killing effect of
imatinib in CML (30). To ascertain
whether the mechanism underlying the antiproliferative effects of
IM in PVNS-FLS involves activation of caspase-9, cells were treated
with Z-LEHD-FMK, a specific caspase-9 inhibitor. Upon treatment
with Z-LEHD-FMK alone, no differences in cell viability were
evident, compared with the control group, suggesting no effect of
the inhibitor on proliferation. However, treatment with Z-LEHD-FMK
in combination with IM led to a significant improvement in cell
survival rates (Fig. 1B),
indicating a central role of caspase-9 in IM-induced apoptosis.
Significant changes were observed in the general morphology of
cells treated with IM, including loss of normal shape and adhesion
ability (Fig. 2B). The results
collectively suggest that IM has an obvious killing effect on
PVNS-FLS and effectively reduces cell viability in vitro.
Hoechst and AO/EB staining data further supported the apoptotic
effect of IM. Characteristic morphological changes of apoptosis
appeared gradually with the addition of IM. Nuclei became condensed
and crescent shaped with subsequent chromatin dissolution,
breakdown, and fragmentation, indicative of the late stages of
apoptosis (Figs. 2A and 3).
The Annexin V/PI staining assay additionally
validated the apoptosis-inducing effect of IM. Upon IM treatment,
we observed a significant increase in apoptosis rates, both early
and late-stage, compared with the control group (Fig. 4A and C). Consistent with previous
results, apoptosis was blocked upon the addition of Z-LEHD-FMK. PI
staining data revealed that IM causes cell cycle arrest in FLS.
Treated cells displayed apparent G2/M phase arrest with increasing
drug concentrations (Fig. 4B and
D). Our findings suggest that when cells are treated with IM,
mitosis is blocked, which affects the proliferation and viability
of PVNS-FLS. With increasing drug concentrations, the killing
effect is fulfilled through induction of apoptosis. Pretreatment of
cells with the caspase-9 inhibitor Z-LEHD-FMK, blocked apoptosis,
implying that IM-induced cell death occurs via a
caspase-9-dependent pathway.
Caspase-9, an essential initiator caspase, is a
primary caspase of the mitochondrial apoptosis pathway. The
mitochondrial pathway is initiated within cells, and the
mitochondrion is a key location (31). Following exposure to various
apoptotic stimuli in mitochondria, Bcl-2 family members are
activated, including the anti-apoptotic protein, Bcl-2 and the
pro-apoptotic protein, Bax. Consequently, the Bax/Bcl-2 ratio is
increased, which enhances the permeability of the mitochondrial
outer membrane. As a result, a type of soluble mitochondrial
intra-membrane protein (SIMP), cytochrome c (cyt c),
is released into the cytoplasm that binds the apoptosis
protease-activating factor-1 (Apaf-1). Subsequently, pro-caspase-9
interacts with the caspase recruitment domain (CARD) of Apaf-1,
forming the apoptosome whereby caspase-9 is activated and
stimulates the caspase-3 effector, in turn, driving the terminal
events of apoptosis (31–34).
To elucidate the mechanism underlying apoptosis
induction by IM in PVNS-FLS, we employed western blot analysis in
the current investigation. IM-treated PVNS-FLS showed significant
activation of caspase-9 and -3 in a dose-dependent manner. The
level of cleaved caspase-9 (35 kDa) was greatly increased, and
conversely, pro-caspase-9 and pro-caspase-3 levels were
significantly decreased. Expression of pro-apoptotic Bax was
significantly increased while that of anti-apoptotic Bcl-2 was
reduced, indicating activation of the Bcl-2 family members as
described above. Our findings collectively suggest that
caspase-related apoptosis plays a critical role in the IM-induced
cell killing effect, particularly the mitochondrial pathway where
caspase-9 is a central player. The inhibitory effects of Z-LEHD-FMK
on apoptosis further confirmed this theory.
In addition, invasion to tissues around joints is
the main feature of PVNS. Synovial lesions invade the ligaments,
bone and cartilage around the joints, leading to rapid degeneration
and functional impairment of the joints. In addition, this is also
the main reason why surgical synovectomy can not excise the
extensive lesions and eventually cause a high rate of recurrence.
Thus, a treatment to inhibit the synovial lesions invasion is
urgently needed. In the present study, we found that IM exhibited
significant inhibition on the invasion ability of PVNS-FLS in
vitro detected by a Transwell invasion assay (Fig. 6), which makes it a potential
treatment of the disease.
Strong erosion of adjacent tissue and higher
recurrence rate has been a critical dilemma for surgeons dealing
with PVNS in the clinic. Traditional surgical and radiation therapy
procedures are not effective and increase the suffering of
patients. Several chemotherapy drugs have been shown to prevent
cell proliferation or kill tumor cells by inducing apoptosis in the
clinic. It remains to be established whether IM has a similar
effect on PVNS. In our experiments, IM exerted significant
inhibitory activity on the viability and proliferation of PVNS-FLS
in vitro, and apoptotic effects were evident from the
morphological changes of cells under the fluorescence microscope
and results of Annexin V/PI staining. Accordingly, we concluded
that the cell killing effect of IM is attributable to induction of
apoptosis. To further establish the specific signaling pathways
underlying IM-induced apoptosis, cells were treated with the
caspase-9 specific inhibitor Z-LEHD-FMK. IM-induced apoptosis was
significantly blocked upon co-treatment with Z-LEHD-FMK, implying a
key role of caspase-9. Expression of cleaved caspase-9 was
significantly enhanced and the Bcl-2 family and caspase-3 activated
following treatment of PVNS-FLS with IM. Furthermore, IM also
exerted significant inhibition on the invasion ability of
PVNS-FLS.
In summary, IM exerts a significant
antiproliferative effect on PVNS-FLS in vitro through
induction of apoptosis dependent on mitochondrial apoptosis pathway
by the activation of caspase-9/-3 and the Bcl-2 family. The present
study is the first to report the induction of apoptosis of IM on
PVNS-FLS, which provides theoretical bases for clinical treatment
of patients with PVNS. We also observed that IM greatly inhibits
the invasion ability of PVNS-FLS, which may eventually inhibit the
erosion to tissue around joints in clinical. However, further
studies, including in vivo trials, are required to confirm
the current findings and elucidate the precise mechanisms
underlying IM-induced apoptosis. We believe that extended
biomedicine and biotechnology analyses will facilitate the
development of a novel, clinically applicable IM-based therapy for
PVNS.
Acknowledgments
This study was supported by the National Natural
Science Foundation of China (81071449).
References
|
1
|
Blay JY, El Sayadi H, Thiesse P, Garret J
and Ray-Coquard I: Complete response to imatinib in relapsing
pigmented villonodular synovitis/tenosynovial giant cell tumor
(PVNS/TGCT). Ann Oncol. 19:821–822. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gu HF, Zhang SJ, Zhao C, Chen Y and Bi Q:
A comparison of open and arthroscopic surgery for treatment of
diffuse pigmented villonodular synovitis of the knee. Knee Surg
Sports Traumatol Arthrosc. 22:2830–2836. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ottaviani S, Ayral X, Dougados M and
Gossec L: Pigmented villonodular synovitis: A retrospective
single-center study of 122 cases and review of the literature.
Semin Arthritis Rheum. 40:539–546. 2011. View Article : Google Scholar
|
|
4
|
Lavrador JP, Oliveira E, Gil N, Francisco
AF and Livraghi S: C1-C2 pigmented villonodular synovitis and clear
cell carcinoma: Unexpected presentation of a rare disease and a
review of the literature. Eur Spine J. 24(Suppl 4): S465–S471.
2015. View Article : Google Scholar
|
|
5
|
Myers BW, Masi AT and Feigenbaum SL:
Pigmented villo-nodular synovitis and tenosynovitis: A clinical
epidemiologic study of 166 cases and literature review. Medicine.
59:223–238. 1980.
|
|
6
|
West RB, Rubin BP, Miller MA, Subramanian
S, Kaygusuz G, Montgomery K, Zhu S, Marinelli RJ, De Luca A,
Downs-Kelly E, et al: A landscape effect in tenosynovial giant-cell
tumor from activation of CSF1 expression by a translocation in a
minority of tumor cells. Proc Natl Acad Sci USA. 103:690–695. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nassar WA, Bassiony AA and Elghazaly HA:
Treatment of diffuse pigmented villonodular synovitis of the knee
with combined surgical and radiosynovectomy. HSS J. 5:19–23. 2009.
View Article : Google Scholar :
|
|
8
|
Fiocco U, Sfriso P, Lunardi F, Pagnin E,
Oliviero F, Scagliori E, Cozzi L, Vezzù M, Molena B, Scanu A, et
al: Molecular pathways involved in synovial cell inflammation and
tumoral proliferation in diffuse pigmented villonodular synovitis.
Autoimmun Rev. 9:780–784. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bertoni F, Unni KK, Beabout JW and Sim FH:
Malignant giant cell tumor of the tendon sheaths and joints
(malignant pigmented villonodular synovitis). Am J Surg Pathol.
21:153–163. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Imakiire N, Fujino T, Morii T, Honya K,
Mochizuki K, Satomi K and Fujioka Y: Malignant pigmented
villonodular synovitis in the knee - report of a case with rapid
clinical progression. Open Orthop J. 5:13–16. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kaneko K, Nakahara D, Tobe M, Iwase H,
Inoue Y, Ohbayashi O and Kurosawa H: Pigmented villonodular
synovitis of the ankle in an adolescent. Int Orthop. 24:234–237.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Park G, Kim YS, Kim JH, Lee SW, Song SY,
Choi EK, Yi SY and Ahn SD: Low-dose external beam radiotherapy as a
postoperative treatment for patients with diffuse pigmented
villonodular synovitis of the knee: 4 recurrences in 23 patients
followed for mean 9 years. Acta Orthop. 83:256–260. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Snoots WM, Watkins D, Dockery D, Mennel R
and Cheek BS: Pigmented villonodular synovitis responsive to
imatinib therapy. Proc. 24:134–138. 2011.
|
|
14
|
Ray RA, Morton CC, Lipinski KK, Corson JM
and Fletcher JA: Cytogenetic evidence of clonality in a case of
pigmented villonodular synovitis. Cancer. 67:121–125. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen WM, Wu PK and Liu CL: Simultaneous
anterior and posterior synovectomies for treating diffuse pigmented
villonodular synovitis. Clin Orthop Relat Res. 470:1755–1762. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nakahara H, Matsuda S, Harimaya K,
Sakamoto A, Matsumoto Y, Okazaki K, Tashiro Y and Iwamoto Y:
Clinical results of open synovectomy for treatment of diffuse
pigmented villonodular synovitis of the knee: Case series and
review of literature. Knee. 19:684–687. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nishida Y, Tsukushi S, Nakashima H,
Sugiura H, Yamada Y, Urakawa H, Arai E and Ishiguro N:
Osteochondral destruction in pigmented villonodular synovitis
during the clinical course. J Rheumatol. 39:345–351. 2012.
View Article : Google Scholar
|
|
18
|
Liu XY, Yang YF, Wu CT, Xiao FJ, Zhang QW,
Ma XN, Li QF, Yan J, Wang H and Wang LS: Spred2 is involved in
imatinib-induced cytotoxicity in chronic myeloid leukemia cells.
Biochem Biophys Res Commun. 393:637–642. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cassier PA, Gelderblom H, Stacchiotti S,
Thomas D, Maki RG, Kroep JR, van der graaf WT, Italiano A, Seddon
B, Dômont J, et al: Efficacy of imatinib mesylate for the treatment
of locally advanced and/or metastatic tenosynovial giant cell
tumor/pigmented villonodular synovitis. Cancer. 118:1649–1655.
2012. View Article : Google Scholar
|
|
20
|
Ravi V, Wang WL and Lewis VO: Treatment of
tenosynovial giant cell tumor and pigmented villonodular synovitis.
Curr Opin Oncol. 23:361–366. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Stacchiotti S, Crippa F, Messina A,
Pilotti S, Gronchi A, Blay JY and Casali PG: Response to imatinib
in villonodular pigmented synovitis (PVNS) resistant to nilotinib.
Clin Sarcoma Res. 3:82013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zimmermann T, Kunisch E, Pfeiffer R, Hirth
A, Stahl HD, Sack U, Laube A, Liesaus E, Roth A, Palombo-Kinne E,
et al: Isolation and characterization of rheumatoid arthritis
synovial fibroblasts from primary culture - primary culture cells
markedly differ from fourth-passage cells. Arthritis Res. 3:72–76.
2001. View Article : Google Scholar :
|
|
23
|
Wessel D and Flügge UI: A method for the
quantitative recovery of protein in dilute solution in the presence
of detergents and lipids. Anal Biochem. 138:141–143. 1984.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Granowitz SP, D'Antonio J and Mankin HL:
The pathogenesis and long-term end results of pigmented
villonodular synovitis. Clin Orthop Relat Res. 114:335–351.
1976.PubMed/NCBI
|
|
25
|
Schwartz HS, Unni KK and Pritchard DJ:
Pigmented villonodular synovitis. A retrospective review of
affected large joints. Clin Orthop Relat Res. 247:243–255.
1989.PubMed/NCBI
|
|
26
|
Tyler WK, Vidal AF, Williams RJ and Healey
JH: Pigmented villonodular synovitis. J Am Acad Orthop Surg.
14:376–385. 2006.PubMed/NCBI
|
|
27
|
Dines JS, DeBerardino TM, Wells JL, Dodson
CC, Shindle M, DiCarlo EF and Warren RF: Long-term follow-up of
surgically treated localized pigmented villonodular synovitis of
the knee. Arthroscopy. 23:930–937. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guilhot F: Indications for imatinib
mesylate therapy and clinical management. Oncologist. 9:271–281.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sawyers CL: Imatinib GIST keeps finding
new indications: Successful treatment of dermatofibrosarcoma
protuberans by targeted inhibition of the platelet-derived growth
factor receptor. J Clin Oncol. 20:3568–3569. 2002.PubMed/NCBI
|
|
30
|
Du Y, Wang K, Fang H, Li J, Xiao D, Zheng
P, Chen Y, Fan H, Pan X, Zhao C, et al: Coordination of intrinsic,
extrinsic, and endoplasmic reticulum-mediated apoptosis by imatinib
mesylate combined with arsenic trioxide in chronic myeloid
leukemia. Blood. 107:1582–1590. 2006. View Article : Google Scholar
|
|
31
|
Hu Q, Wu D, Chen W, Yan Z, Yan C, He T,
Liang Q and Shi Y: Molecular determinants of caspase-9 activation
by the Apaf-1 apoptosome. Proc Natl Acad Sci USA. 111:16254–16261.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jin Z and El-Deiry WS: Overview of cell
death signaling pathways. Cancer Biol Ther. 4:139–163. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Luo X, Budihardjo I, Zou H, Slaughter C
and Wang X: Bid, a Bcl2 interacting protein, mediates cytochrome c
release from mitochondria in response to activation of cell surface
death receptors. Cell. 94:481–490. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mutlu A, Gyulkhandanyan AV, Freedman J and
Leytin V: Activation of caspases-9, -3 and -8 in human platelets
triggered by BH3-only mimetic ABT-737 and calcium ionophore A23187:
Caspase-8 is activated via bypass of the death receptors. Br J
Haematol. 159:565–571. 2012.PubMed/NCBI
|