Introduction
Currently there is no effective treatment for
patients with breast cancer in an aggressive phase. Breast cancer
is caused by complex biological processes involving multi-gene
mutations. Therefore any breakthrough in breast cancer treatment is
likely to depend on improvement of gene therapy and development of
new targeting drugs. Cytosine deaminase/5-fluorocytosine (CD/5-FC)
and thymidine kinase/ganciclovir (TK/GCV) are the most common
suicide gene therapy treatment systems (1,2).
Single suicide gene therapy often results in tumor relapse and
combination gene therapy may potentially overcome the limitations
of single gene therapy and improve the therapeutic efficacy
(3,4). Our previous study confirmed that the
double suicide gene system (CD/TK) driven by VEGF promoter can
inhibit proliferation of human breast cancer cells and induce
cancer cells apoptosis in vitro (5) yet even this strategy does not
completely eradicate advanced tumors often seen in clinic.
It is well-known now that bystander effect (BSE) is
very helpful in promoting the effectiveness of suicide gene system
for anticancer therapy. Bystander effect elicits its effect mainly
through gap junction-mediated intercellular communication (GJIC)
(6). Gap junctions are an important
gateway of information exchange by promoting transport of small (~1
kDa) molecules like ions or secondary messengers like cyclic AMP. A
gap junction is composed of two connexons, also termed
hemichannels, and each connexon is composed of six connexin
sub-units (7,8). Three connexins (Cx43, Cx26 and Cx32)
have been shown to be expressed in the human breast tissue in
different temporal and spatial patterns. In normal human breast
tissues, Cx26 and Cx32 are present in the mammary epithelium,
whereas Cx43 is localized mainly to myoepithelial cells (9,10). It
was reported that Cxs are often reduced or lost resulting in
significantly decreased GJIC in tumor tissues, thus decreased gap
junctional communication may be involved in oncogenesis (11). Studies have shown that human mammary
tumor MCF-7 cell line has low expression of these connexins,
particularly Cx43 (12). This
suggests that MCF-7 cells may regulate GJIC by downregulation of
gap junction proteins. Several chemicals [like ATRA, cAMP, and
carotenoids (13)] are known for
their induction of Cx43 expression and, subsequently, improvement
of GJIC that consequently leads to an increased bystander effect
seen in suicide gene therapy.
In this study we selected ATRA because it is strong
inducer of GJIC in suicide gene therapy (14). ATRA is a derivative of vitamin A
thus is non-toxic and well tolerated in vivo. We observed
that ATRA significantly improved bystander effect and cell
apoptosis in suicide gene therapy for breast cancer MCF-7 cells due
to upregulation of Cx43 expression and consequently GJIC. The
purpose of our study was to find a more efficient strategy for
improving the efficacy of suicide gene therapy for breast
cancer.
Materials and methods
Human breast cancer cell lines
MCF-7 cells were obtained from the American Type
Culture Collection (ATCC, VA, USA). They were maintained in a
monolayer culture in DMEM (Dulbecco's modified Eagle's medium) with
10% fetal bovine serum and 0.5% penicillin/streptomycin. The cells
were kept in humidified atmosphere of 5% CO2 at
37°C.
Adenovirus vectors and viral
infection
The adenovirus vectors (Ad-VEGFP-CD/TK) had
previously been constructed by our group and successfully amplified
by transient transfection of HEK-293 cells according to published
protocols (15). The titer of
Ad-VEGFP-CD/TK stock was 2.2×1011 pfu/ml analyzed by
end-point dilution assay (16).
MCF-7 cells at logarithmic growth phase were incubated in DMEM
medium with 10% FBS containing the adenovirus vectors at
multiplicities of infection (MOI) of 20, 40, 60, 80, 100 and 200 at
37°C for 24 h. The adenovirus vectors contained green fluorescent
protein (GFP) gene as a marker of transduction. Infected cells were
analyzed for percentage of GFP-positive cells by flow cytometry
(FACS, Becton-Dickinson, San Jose, CA, USA). MCF-7 cells infected
by the adenovirus vectors were termed as MCF-7/CD-TK.
Cell viability MTT assay
The MCF-7 cells and MCF-7/CD-TK cells were collected
at logarithmic growth phase (1×105/ml). MCF-7/CD-TK
cells were trypsinized and mixed with uninfected MCF-7 cells at
ratios of 0, 5, 10, 20, 40 and 80%. Then, the cells were seeded at
a density of 1×104 cells/well in 96-well plates and
incubated in DMEM at 37°C for 24 h, with or without 10−8
and 10−7 mol/l of ATRA. The following day, GCV (final
concentration, 1 mg/l) and 5-FC (final concentration at 80 mg/l)
were added in triplicate into the 96-well plates and further
cultured for 2 days. Finally, the cell viability was assessed using
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
(Sigma, St. Louis, MO, USA) assay. MTT was added at a final
concentration of 0.5 mg/ml for 4 h at 37°C. After incubation, the
supernatant was removed and replaced by DMSO. After the cell plates
were shaken for 10 min, cell proliferation was measured in terms of
optical absorbance per well by a microplate reader at a wavelength
of 490 nm.
Cell apoptosis assay
To investigate the effect of ATRA on apoptosis in
Ad-VEGFP-CD/TK suicide gene, we used JC-1 Assay kit (17) (Molecular Probes, Eugene, OR, USA)
which measures the integrity of mitochondria. MCF-7/CD-TK cells
were mixed with non-transduced parental cells at ratios of 2:3.
Cells were seeded at a density of 1×106 cells in a
25-cm2 culture bottle and incubated in DMEM at 37°C for
24 h, with or without 10−8 and 10−7 mol/l
ATRA. The following day, GCV (1 mg/l) and 5-FC (80 mg/l) were added
to cells for 2 days. Pure MCF-7 cells in the control group were
cultured in DMEM for 2 days. Then, the cells were collected and
incubated with JC-1 working solution (JC-1 stock solution and assay
buffer at a 1,100) at 37°C in the dark for 20 min. Cells were then
collected by centrifugation at room temperature and washed twice
with PBS. Apoptosis was measured a flow cytometry (Beckman Coulter,
USA) to detect 5, 5′, 6, 6′-tetrachloro1, 1′, 3,
3′-tetraethyl-benzimidazolylcarbocyanine iodide (JC-1) staining,
which emits green fluorescence (detected in FL1 channel) when it is
in monomer (indication of cell apoptosis) and red (detected in FL2
channel) when it aggregates (which detects healthy cells).
Apoptosis was indicated by an increase in the green/red
fluorescence intensity ratio (18).
Flow cytometry analysis of GJIC in MCF-7
cells under ATRA treatment
GJIC was functionally evaluated by means of flow
cytometry, as described by Peterson-Roth et al (19). MCF-7 (3×105) cells per
well were seeded into 6-well plates. The next day, ATRA was added
to MCF-7 cells at 10−8 or 10−7 mol/l ATRA for
the next 72 h. Cells without added ATRA served as a control. The
donor cells were preloaded with fluorescent dyes calcein/AM
(Molecular Probes) and
1,10-dioctadecyl-3,3,30,30-tetramethylindocarbocyanine perchlorate
(DiI) (Molecular Probes) for 20 min in a 5% CO2
incubator at 37°C. After ATRA treatment for 72 h, the preloaded
donor cells were added to the unlabeled recipient cells at a ratio
of 1:50 and were co-cultured for 5 h in the presence of ATRA. After
incubation, cells were collected by trypsinization, washed and
resuspended in PBS and analyzed by flow cytometer. Data were
analyzed by flow analysis software (FACSAria, FACSDiva Software, BD
Biosciences, NJ, USA).
Real-time RT-PCR
MCF-7 cells were incubated in DMEM with
10−8 or 10−7 mol/l ATRA at 37°C for 72 h. The
control group was decoid of ATRA. Total RNA from experimental and
control group was prepared using the TRIzol reagent (Invitrogen,
Carlsbad, CA, USA) according to the manufacturer's instructions.
RT-PCR was performed on 0.5 µg total RNA using a QuantiTect
Rev Transcription kit (Qiagen). Quantitative PCR was performed
using an ABI 7500 Real-Time PCR system and a QuantiTect SYBR Green
PCR kit (Qiagen); β-actin was used as an endogenous reference, and
each sample was normalized to its β-actin content. All experiments
were performed in duplicate and repeated twice. Primers for
quantitative PCR are shown in Table
I.
 | Table IPrimer set used for real-time PCR. |
Table I
Primer set used for real-time PCR.
| Primer set | Sequence | Target gene |
|---|
| qCx43 | Sense:
5′-AAATAGACAGGTCTGAGTGCCTGAA-3′
Antisense: 5′-CCTCCAGCAGTTGAGTAGGCTTG-3′ | Homo sapiens
gap junction protein (NM_000165.4)
Amplicon: 181 bp |
| qβ-actin | Sense:
5′-TGGCACCCAGCACAATGAA-3′
Antisense: 5′-CTAAGTCATAGTCCGCCTAGAAGCA-3′ | Actin, β
(NM_001101)
Amplicon: 186 bp |
Flow cytometry analysis of Cx43
expression
MCF-7 cells were seeded at a density of
5×105 cells/well in a 50-ml cell culture bottle and
incubated in DMEM at 37°C for 24 h. Cells were infected with
Ad-VEGFP-CD/TK vector for 24 h following exposure to ATRA (0,
10−8 and 10−7 mol/l) for 72 h. Cells were
then trypsinized, collected, and re-suspended in cold PBS
(106 cells/ml). Polyclonal anti-Cx43 antibody (1:1,000,
ProteinTech Group, Chicago, IL, USA) was added to cells for 30 min
and cells were stained on ice. Cells were washed in FACS buffer
(PBS+2% FBS) and FITC-labeled goat anti-rabbit IgG (1:500,
Molecular Probes) was added or 30 min on ice. Cells were
centrifuged and washed with PBS. The cells were harvested and
analysis was performed using a Beckman Coulter Flow Cytometer
(Beckman Coulter). The data were analyzed by Multicycle software
(Phonenix Flow Systems, USA).
Statistics
All data were expressed as means ± SD and analyzed
with SPSS version 16.0. A Dunnett's t-test and a one-way ANOVA were
performed to assess the statistical significance between different
groups. Significance for all tests was established at
p<0.05.
Results
Infection efficiency of Ad-VEGFP-CD/TK in
MCF-7 breast cancer cells
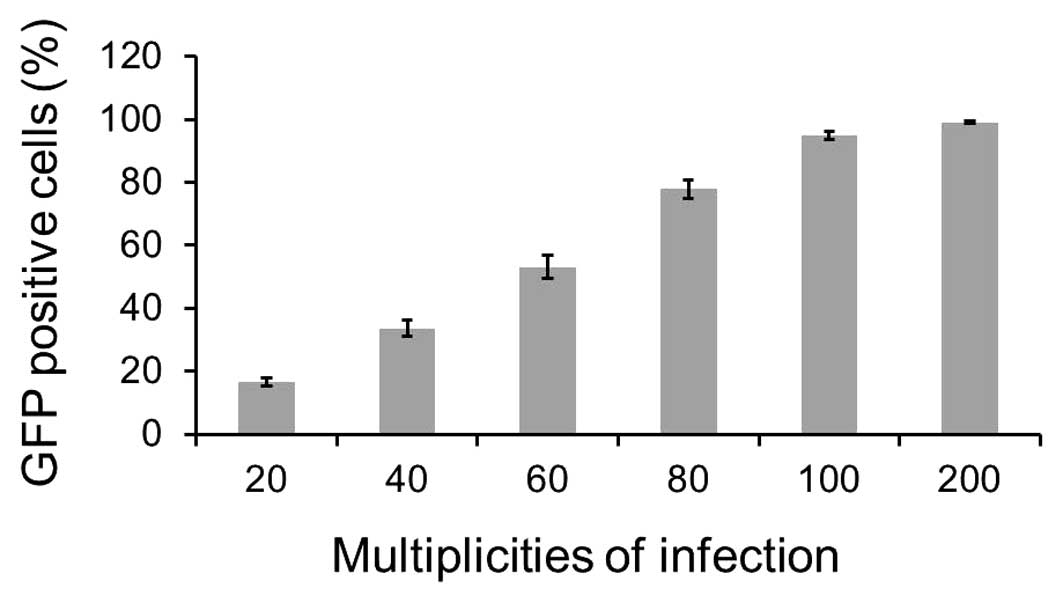
As shown in Fig. 1,
one day after infection, >16% of GFP-MCF-7 cells at a MOI of 20
were identified by flow cytometry. The infected GFP-positive cells
were as high as 95.05% at MOI of 100. More than 99% of the infected
cells expressed green fluorescence at an MOI of 200. The result
indicated that the Ad-VEGFP-CD/TK vector was able to effectively
infect MCF-7 cells in vitro.
Effect of ATRA on prodrug
cytotoxicity
ATRA at a final concentration of 10−7 and
10−8 mol/l had no toxicity to MCF-7 cells. This
concentration was established based on previous studies (20). We detected the ability of ATRA
(10−7 and 10−8 mol/l) to enhance the
bystander effect of our suicide gene system in mixed cultures of
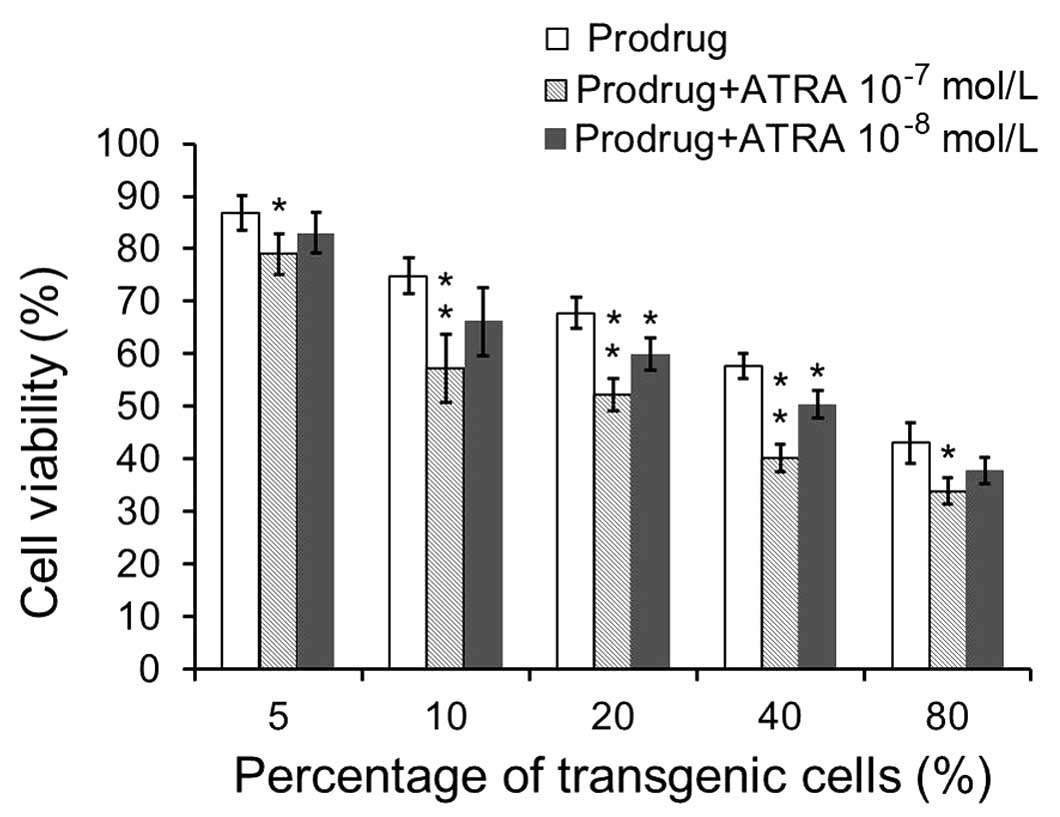
transgenic and non-transgenic cells at various ratios. As shown in
Fig. 2, the MTT assays showed 79,
57, 52, 40 and 34% cell viability after prodrug and ATRA
(10−7 mol/l) treatment for a ratio of 5, 10, 20, 40 and
80% infected cells, respectively, and 83, 66, 60, 50 and 38% cell
viability after prodrug and ATRA (10−8 mol/l) treatment,
respectively, and 87, 75, 68, 58 and 43% cell viability after
prodrug alone treatment, respectively. The data showed that MCF-7
cell survival rate decreased along with the increasing ratios of
CD/TK-transgenic cells. This result indicates that bystander
effects exist in MCF-7 cells exposed to suicide gene system. In
addition, the cell survival rate under treatment with the prodrug
(5-FC and GCV) and ATRA (10−7 mol/l) was significantly
lower than those of the cells treated with the prodrug alone
(p<0.05). In addition, the cell survival rate in the treatment
with the prodrug and ATRA (10−8 mol/l) was significantly
lower than the prodrug alone at ratios of 1:5 and 2:5, of the
infected cells to the non-infected cells (p<0.05). The result
demonstrates ATRA can promote the bystander effects of the suicide
gene system for breast cancer cells.
Effect of ATRA on apoptosis of breast
cancer cells
When transgenic MCF-7 cells were mixed with
non-transgenic MCF-7 cells at ratios of 40% and treated by prodrug
and/or the ATRA, there was a significantly different apoptosis rate
between the prodrug only group and ATRA plus the prodrug group
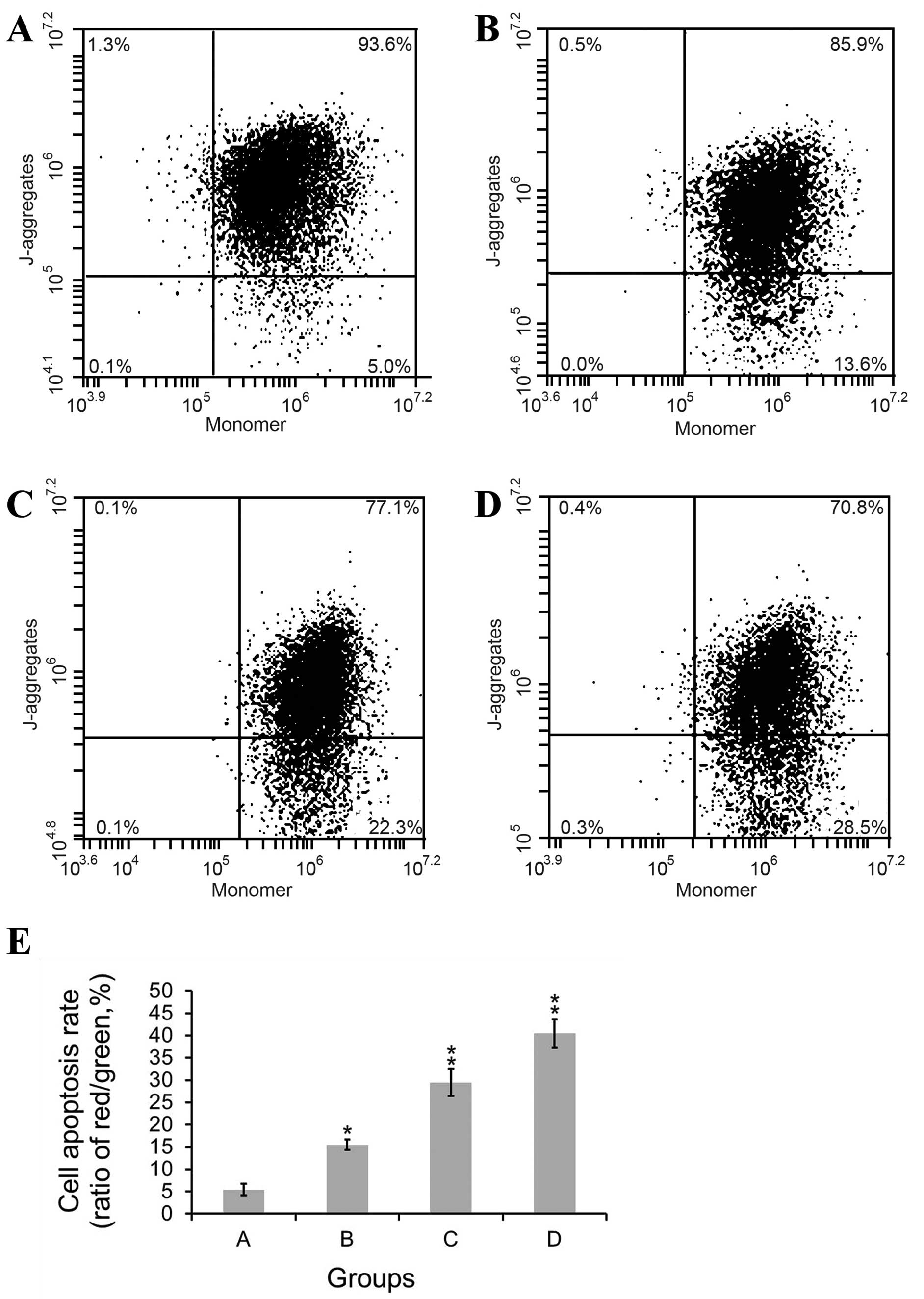
(Fig. 3). In control group, MCF-7
cells showed a low rate of apoptosis of 5.45%. Under the treatment
of 5-FC and GCV, cell apoptosis rate was 15.50%. Under the combined
treatment of the prodrug and ATRA (the concentration of ATRA was
10−8 mol/l or 10−7 mol/l), cell apoptosis
rate was 29.48 and 40.46%, respectively. The results indicate that
there is statistically significant difference among four groups
(F=125.277). The data suggested that the prodrug only exerted some
cell killing effects in mixed cells. The apoptotic rate was
significantly increased with the addition of ATRA, especially when
the concentration of ATRA increased from 10−8 mol/l to
10−7 mol/l. The effect of ATRA showed a
concentration-dependent manner. These results demonstrated that the
drug plus ATRA therapy was superior to prodrug only.
Impact of ATRA on GJIC
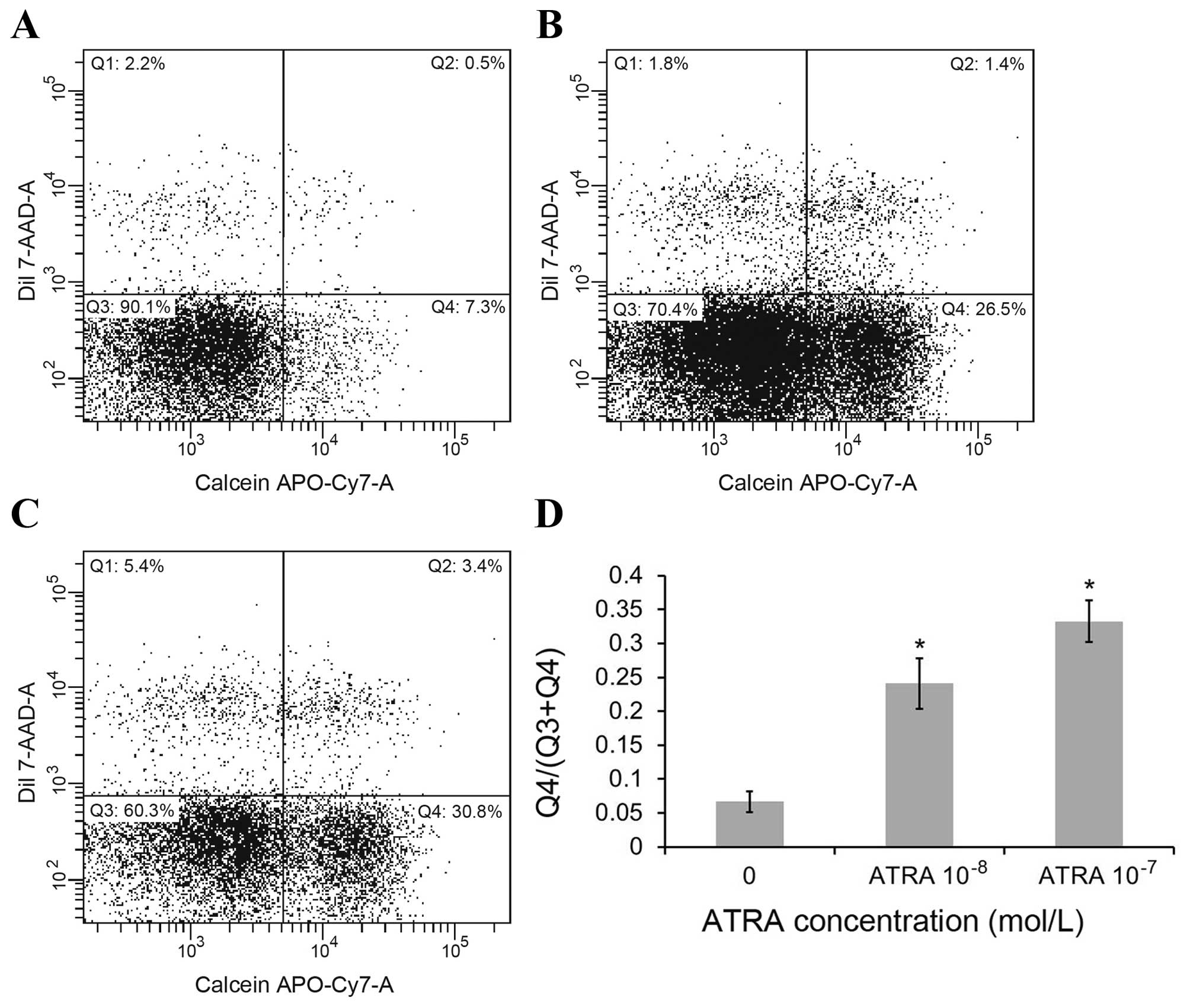
To detect the change in GJIC caused by ATRA in MCF-7
cells, we used flow cytometry quantification of calcein/CM-DiI
assays. DiI cannot pass through gap junctions, while calcein passes
through gap junctions and transfer cells preloaded with calcein to
a population of unloaded cells. In the FACS plot (Fig. 4), quadrants Q1 represents cell
populations positive for DiI, and the cells population in quadrants
Q2 represents preloaded cells which are positive for both dyes, and
the population in quadrants Q4 represents positive cells for only
calcein which are the unlabeled cells that received the calcein dye
through GJIC. The population in quadrants Q3 represents the
calcein-negative unlabeled cells. The ratio of MCF-7 cell numbers
in quadrants Q4 to Q3 plus Q4 was used to evaluate the transfer of
calcein as an indication of GJIC function. As seen in Fig. 4D, the ratio of Q4 to (Q3+Q4) was
0.07, 0.24 and 0.33 in the control group, 10−8 and
10−7 mol/l ATRA treatment group, respectively
(p<0.01). The results show that ATRA significantly increased GJ
transmission in MCF-7 cells in a concentration-dependent manner,
which indicates GJIC function in the MCF-7 cells is significantly
improved by ATRA treatment.
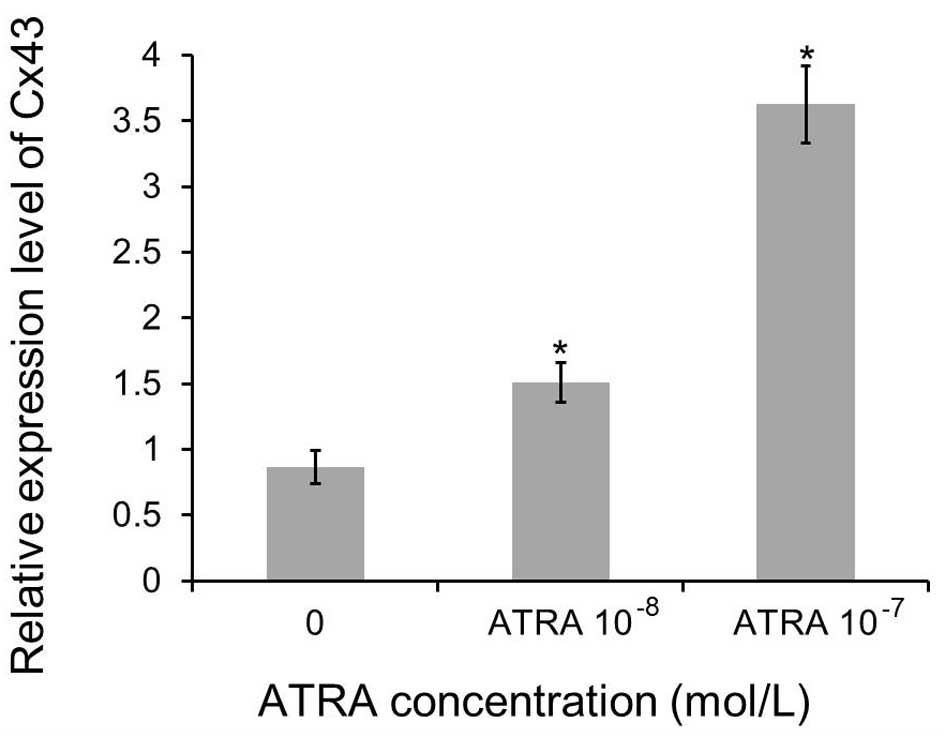
ATRA treatment upregulates connexin 43
gene expression
Our previous experiment indicated increased GJIC
being an effect of treatment with ATRA. It was crucial to delineate
whether it is a sole enhancement in the intercellular communication
or whether this is an effect on increased gap junctions resulting
with increased expression of Cx43. Transgenic MCF-7 cells were
treated with increasing concentrations of ATRA (0, 10−7
and 10−8 mol/l) for 72 h to determine the ATRA effect on
the expression of Cx43 gene by real-time PCR. The upregulation of
the Cx43 gene was pronounced, yielding a 1.5-fold increase
(10−8 mol/l ATRA) and 3.6-fold (10−7 mol/l
ATRA), respectively. Furthermore, the results indicate that the
influence is dose-dependent. Compared with levels in control group,
the mRNA expression levels of Cx43 in the transgenic MCF-7 cells
treated with ATRA were increased significantly (p<0.01, Fig. 5).
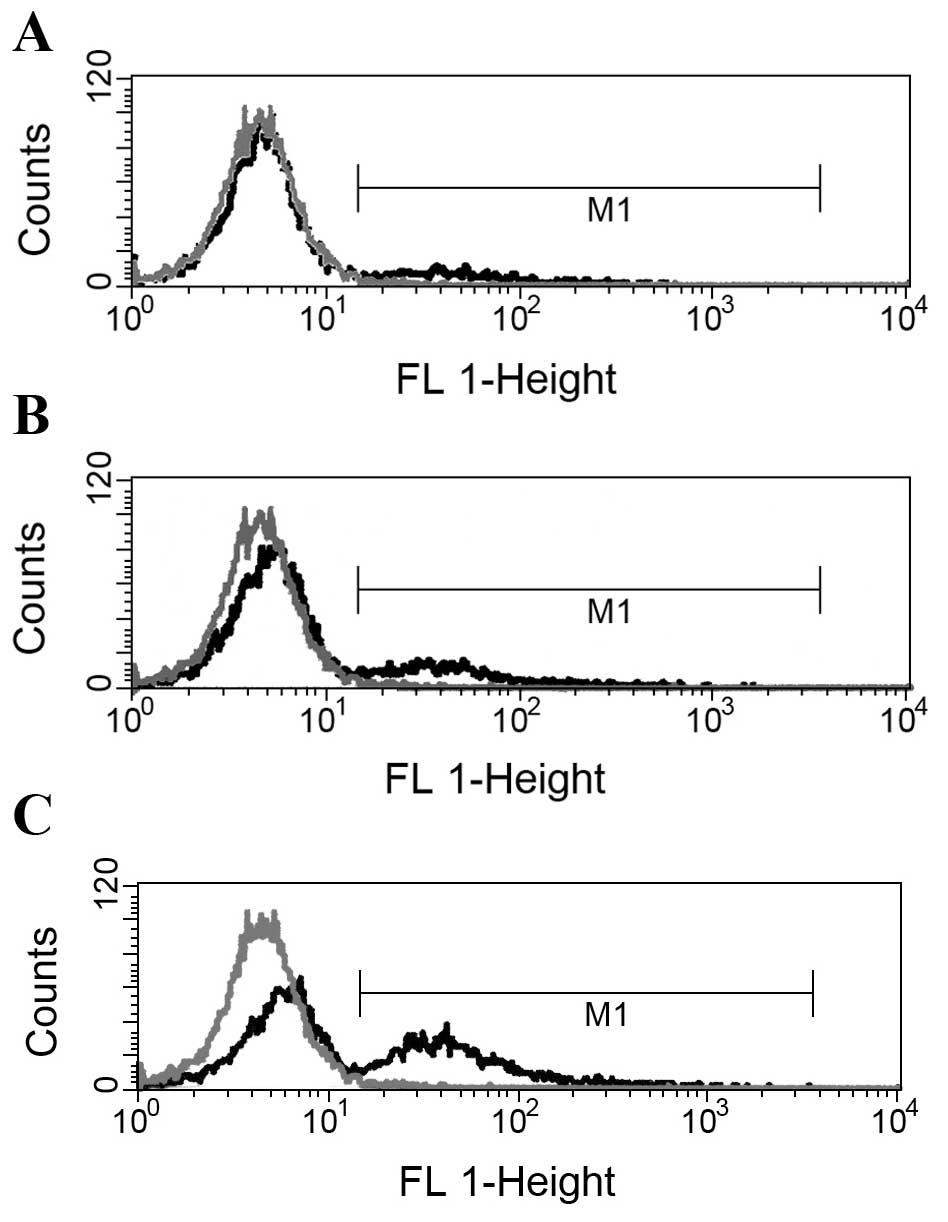
Flow cytometry analysis of Cx43 protein
expression in MCF-7 cells
To examine whether or not Cx43 protein expression in
MCF-7-CD/TK cells changes under the influence of ATRA, different
concentrations of ATRA (0, 10−7 and 10−8
mol/l) were added to cultures of MCF-7 cells. Flow cytometry
analysis showed that positive rates of Cx43 protein expression in
transgenic MCF-7 treated with the different concentrations of 0,
10−8 or 10−7 mol/l ATRA were (6.27±1.75),
(15.58±1.41) and (27.32±2.85)%, respectively. A significant
difference was noted between the experimental group
(10−8 or 10−7 mol/l ATRA) and the untreated
control group. As shown in Fig. 6,
at 72 h after ATRA exposure, the percentages of Cx43 protein
detected using flow cytometry were also significantly higher in
cells of the experimental group than in the control cells both
transfected with Ad-VEGFP-CD/TK vector.
Discussion
Suicide gene therapy is a potential way of targeting
cancer, which is much less harmful to healthy cells. Many studies
also have achieved good results in the application of suicide gene
system in multiple malignancy tumors, such as glioblastoma,
prostate cancer, and breast cancer (21–23).
Moreover, some clinical trial of suicide gene therapy for patients
with certain malignant tumors showed encouraging effects (24,25).
However, the efficacy of the suicide gene therapy for the treatment
of cancer is limited because of the low killing activity. The
bystander effect in suicide gene therapy offers a way of promoting
the effectiveness of suicide gene system for cancer. ATRA can
enhance Cx43 expression and upregulate GJIC in various tumor cells.
To enhance the antitumor activity, we determined whether suicide
gene system combined with ATRA could potentiate the destruction of
breast cancer.
Three connexin proteins have been identified in
breast tissues, including Cx43, Cx26 and Cx32 (9). Expression of Cx26 and Cx32 are
enhanced during pregnancy and further increased during lactation in
normal breast tissues (10). Cx43
is the most widely studied of these connexin proteins because of
its high expression in many cells (26) including breast cancer cells although
human MCF-7 cells express low level of GJIC (12). In this study, QPCR and FACS methods
detected upregulation of Cx43 gene and protein in MCF-7 cells under
ATRA treatment. At the same time, ATRA was found to significantly
increase GJ transmission in MCF-7 cells as a results of ATRA
elevated Cx43 expression.
In the present study, we observed that the cell
survival rate of transgenic MCF-7 cells treated with the prodrug
and ATRA was significantly lower than those of the cells treated
with the prodrug alone (p<0.05). Our findings suggest that the
therapeutic bystander effect of VEGFP-TK/CD system in MCF-7 cells
was enhanced by combination with ATRA administration via
overexpression of Cx43 and enhanced GJIC. We also found that
compared with control group, the suicide gene system induced a
marked increase in cell apoptosis rate in MCF-7 cells under the
prodrug plus ATRA treatment group when MCF-7/CD-TK cells were mixed
with non-transgenic MCF-7 cells at ratios of 40%. Intercellular
communication through gap junctions plays an important role in
maintaining tissue homeostasis by allowing transfer of ions and
small molecules (molecular weight <1 kDa) by GJIC (27). So, the present study suggested that
the transfer of apoptotic vesicles from dying cells to the
surrounding non-transgenic tumor cells through gap junctions is one
of the mechanistic actions of the suicide gene system on the BSE in
cancer cells.
In conclusion, we demonstrated that ATRA can augment
bystander effect and increase apoptosis in MCF-7 cells during
suicide gene therapy against breast cancer. The effect was
associated with enhanced GJIC via the upregulation of Cx43
expression driven by ATRA induction. Our study suggested the
upregulation of gap junctional communication by ATRA may provide a
novel pathway of chemical inducer for improving suicide gene system
therapy for breast cancer.
Acknowledgments
This study was supported by Shenzhen Council for
Scientific and Technological Innovation under grant
JCYJ20130402151227177 and Shenzhen Nanshan District Science and
Technology Project under grant 2015009.
References
|
1
|
Nguyen Thai QA, Sharma N, Luong H, Sodhi
SS, Kim JH, Kim N, Oh SJ and Jeong DK: Targeted inhibition of
osteosarcoma tumor growth by bone marrow-derived mesenchymal stem
cells expressing cytosine deaminase/5-fluorocytosine in
tumor-bearing mice. J Gene Med. 17:87–99. 2015. View Article : Google Scholar
|
|
2
|
Zu B, Shi Y, Xu M, You G, Huang Z, Gao M
and Feng W: ARE/SUZ12 dual specifically-regulated adenoviral TK/GCV
system for CML blast crisis cells. J Exp Clin Cancer Res.
34:562015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Niu J, Xing C, Yan C, Liu H, Cui Y, Peng
H, Chen Y, Li D, Jiang C, Li N, et al: Lentivirus-mediated CD/TK
fusion gene transfection neural stem cell therapy for C6
glioblastoma. Tumour Biol. 34:3731–3741. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ibrišimović M, Lion T and Klein R:
Combinatorial targeting of 2 different steps in adenoviral DNA
replication by herpes simplex virus thymidine kinase and artificial
microRNA expression for the inhibition of virus multiplication in
the presence of ganciclovir. BMC Biotechnol. 13:542013. View Article : Google Scholar
|
|
5
|
Kong H, Tao L, Qi K, Wang Y, Li Q, Du J
and Huang Z: Thymidine kinase/ganciclovir and cytosine
deaminase/5-fluorocytosine suicide gene therapy-induced cell
apoptosis in breast cancer cells. Oncol Rep. 30:1209–1214.
2013.PubMed/NCBI
|
|
6
|
Sato T, Neschadim A, Lavie A, Yanagisawa T
and Medin JA: The engineered thymidylate kinase (TMPK)/AZT
enzyme-prodrug axis offers efficient bystander cell killing for
suicide gene therapy of cancer. PLoS One. 8:e787112013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gabashvili AN, Baklaushev VP, Grinenko NF,
Levinskii AB, Mel'nikov PA, Cherepanov SA and Chekhonin VP:
Functionally active gap junctions between connexin 43-positive
mesenchymal stem cells and glioma cells. Bull Exp Biol Med.
159:173–179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vaiyapuri S, Flora GD and Gibbins JM: Gap
junctions and connexin hemichannels in the regulation of
haemostasis and thrombosis. Biochem Soc Trans. 43:489–494. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rakib MA, Lee WS, Kim GS, Han JH, Kim JO
and Ha YL: Antiproliferative action of conjugated linoleic acid on
human MCF-7 breast cancer cells mediated by enhancement of gap
junctional intercellular communication through inactivation of
NF-κB. Evid Based Complement Alternat Med. 2013:4293932013.
View Article : Google Scholar
|
|
10
|
Stewart MK, Plante I, Bechberger JF, Naus
CC and Laird DW: Mammary gland specific knockdown of the
physiological surge in Cx26 during lactation retains normal mammary
gland development and function. PLoS One. 9:e1015462014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang J, Liu B, Wang Q, Yuan D, Hong X,
Yang Y and Tao L: Connexin 32 and its derived homotypic gap
junctional inter-cellular communication inhibit the migration and
invasion of transfected HeLa cells via enhancement of intercellular
adhesion. Mol Med Rep. 4:971–979. 2011.PubMed/NCBI
|
|
12
|
Talhouk RS, Fares MB, Rahme GJ, Hariri HH,
Rayess T, Dbouk HA, Bazzoun D, Al-Labban D and El-Sabban ME:
Context dependent reversion of tumor phenotype by connexin-43
expression in MDA-MB231 cells and MCF-7 cells: Role of
β-catenin/connexin43 association. Exp Cell Res. 319:3065–3080.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Piccoli C, D'Aprile A, Scrima R, Ambrosi
L, Zefferino R and Capitanio N: Subcytotoxic mercury chloride
inhibits gap junction intercellular communication by a redox- and
phosphorylation-mediated mechanism. Free Radic Biol Med.
52:916–927. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hu C, Chen Z, Zhao W, Wei L, Zheng Y, He
C, Zeng Y and Yin B: Vesicular stomatitis virus G glycoprotein and
ATRA enhanced bystander killing of chemoresistant leukemic cells by
herpes simplex virus thymidine kinase/ganciclovir. Biomol Ther
(Seoul). 22:114–121. 2014. View Article : Google Scholar
|
|
15
|
Chen JF, Huang ZH, Huang YY, Song HJ and
Che XY: Construction of recombinant adenoviruses encoding TK
suicide gene driven by VEGF promoter using efficient AdEasier-1
system. Ai Zheng. 23:1093–1097. 2004.In Chinese. PubMed/NCBI
|
|
16
|
Sugiyama N, Murayama A, Suzuki R, Watanabe
N, Shiina M, Liang TJ, Wakita T and Kato T: Single strain isolation
method for cell culture-adapted hepatitis C virus by end-point
dilution and infection. PLoS One. 9:e981682014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang J, Zhao YM, Zhang B and Guo CY:
Protective effect of total phenolic compounds from Inula helenium
on hydrogen peroxide-induced oxidative stress in SH-SY5Y Cells.
Indian J Pharm Sci. 77:163–169. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abu N, Akhtar MN, Yeap SK, Lim KL, Ho WY,
Zulfadli AJ, Omar AR, Sulaiman MR, Abdullah MP and Alitheen NB:
Flavokawain A induces apoptosis in MCF-7 and MDA-MB231 and inhibits
the metastatic process in vitro. PLoS One. 9:e1052442014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Peterson-Roth E, Brdlik CM and Glazer PM:
Src-Induced cisplatin resistance mediated by cell-to-cell
communication. Cancer Res. 69:3619–3624. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Stefanska B, Salamé P, Bednarek A and
Fabianowska-Majewska K: Comparative effects of retinoic acid,
vitamin D and resveratrol alone and in combination with adenosine
analogues on methylation and expression of phosphatase and tensin
homologue tumour suppressor gene in breast cancer cells. Br J Nutr.
107:781–790. 2012. View Article : Google Scholar
|
|
21
|
de Melo SM, Bittencourt S, Ferrazoli EG,
da Silva CS, da Cunha FF, da Silva FH, Stilhano RS, Denapoli PM,
Zanetti BF, Martin PK, et al: The anti-tumor effects of adipose
tissue mesenchymal stem cell transduced with HSV-Tk gene on
U-87-driven brain tumor. PLoS One. 10:e01289222015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kubo M, Satoh T, Tabata KI, Tsumura H,
Iwamura M, Baba S, Thompson TC and Obata F: Enhanced central memory
cluster of differentiation 8(+) and tumor antigen-specific T cells
in prostate cancer patients receiving repeated in situ
adenovirus-mediated suicide gene therapy. Mol Clin Oncol.
3:515–521. 2015.PubMed/NCBI
|
|
23
|
Yi BR, Choi KJ, Kim SU and Choi KC:
Therapeutic potential of stem cells expressing suicide genes that
selectively target human breast cancer cells: Evidence that they
exert tumoricidal effects via tumor tropism (Review). Int J Oncol.
41:798–804. 2012.PubMed/NCBI
|
|
24
|
Kim KH, Dmitriev I, O'Malley JP, Wang M,
Saddekni S, You Z, Preuss MA, Harris RD, Aurigemma R, Siegal GP, et
al: A phase I clinical trial of Ad5.SSTR/TKRGD, a novel
infectivity-enhanced bicistronic adenovirus, in patients with
recurrent gynecologic cancer. Clin Cancer Res. 18:3440–3451. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maury S, Rosenzwajg M, Redjoul R, Marcais
A, Xhaard A, Cherai M, Cabanne L, Churlaud G, Suarez F, Socié G, et
al: Lymphodepletion followed by infusion of suicide gene-transduced
donor lymphocytes to safely enhance their antitumor effect: A phase
I/II study. Leukemia. 28:2406–2410. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ghosh S, Kumar A and Chandna S:
Connexin-43 downregulation in G2/M phase enriched tumour cells
causes extensive low-dose hyper-radiosensitivity (HRS) associated
with mitochondrial apoptotic events. Cancer Lett. 363:46–59. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kleopa KA and Sargiannidou I: Connexins,
gap junctions and peripheral neuropathy. Neurosci Lett. 596:27–32.
2015. View Article : Google Scholar
|