Introduction
Epithelial-mesenchymal transition (EMT) is
characterized by the loss of epithelial markers with a reciprocal
gain in mesenchymal phenotype and migratory potential (1). While EMT is essential for embryonic
development and adult tissue maintenance (2,3), it is
also necessary for desmoplasia and cancer cell migration (4). Conversely, mesenchymal-epithelial
transition (MET) is a physiological and embryological phenomenon
induced by cytokines (5,6). During EMT, transcription factors, such
as Snail (7) and Slug (8) are upregulated and induce mesenchymal
gene expression and suppress that of epithelial genes (9).
Transforming growth factor-β (TGF-β) is an important
inducer of EMT (10,11). Bone morphogenetic proteins (BMPs)
are members of the TGF-β superfamily, of which 20 have been
discovered in humans to date (12).
BMPs were first identified for their pro-osteogenic effects, but
recent studies have revealed their additional significance as
tissue morphogenetic factors (13),
particularly for BMP-2, −4 and −7 (14). In particular, BMP-2 is a cytokine
used to treat bone defects and is being investigated in
regenerative studies (15,16).
BMP signaling is induced when a heterodimeric
membrane kinase binds BMP and subsequently triggers Smad protein
phosphorylation, similar to the mechanism of TGF-β pathway
activation. However, downstream BMP-induced signals are mediated by
Smad1/5/9, whereas TGF-β signaling is mediated by Smad2/3. These
receptor-regulated Smad complexes (Smad1/5/9 and Smad2/3) bind the
common mediator Smad4, and collectively translocate into the
nucleus. The Smad complex then binds the DNA promoter region
upstream of target genes to induce their expression (17). The effects of BMP on cancer cells
are relatively uncharacterized when compared to those of TGF-β, but
generally thought to promote EMT (18,19).
However, BMP-2 was recently reported to suppress EMT in the
presence of TGF-β (20). Thus, the
overall effects of BMP on EMT likely depend on the cellular
context.
Squamous cell carcinoma is the most common cancer of
the oral cavity in humans (21,22).
Although BMPs are thought to be involved in cancer metastasis, the
mechanisms underlying BMP-induced EMT/MET have not yet been
clarified at the molecular level (12). Nevertheless, a few studies have
demonstrated the positive effects of BMP on cancer progression
using human oral squamous cell carcinoma (hOSCC) cell lines and
carcinoma tissue (23–26). Moreover, while bone is a major
target for hOSCC metastasis (26,27),
the cellular and molecular mechanisms facilitating this process
remain to be elucidated.
Previous studies by our group revealed that TGF-β1
responsiveness was correlated with EMT-related gene expression in
six hOSCC cell lines. Notably, TGF-β1 enhanced the migration of
HSC-4 hOSCC cells via the Slug/Wnt-5b/MMP-10 and integrin α3β1/FAK
signaling axes (28,29). Since cells are subjected to multiple
simultaneous signals from extracellular ligands and must then
integrate and interpret them, this study investigated the
independent and collective effects of TGF-β1 and BMP-2 on EMT and
MET in HSC-4 cells. In addition, we evaluated how TGF-β1 affects
the BMP-2-induced MET in HSC-4 cells at the molecular level.
Materials and methods
Materials
Cell lines were obtained from the Japanese
Collection of Research Bioresources (Osaka, Japan). Recombinant
human TGF-β1 and BMP-2 were purchased from Peprotech, Inc. (Rocky
Hill, NJ, USA). The TGF-βR1 kinase inhibitor SB431542 was provided
by Merck-Millipore (Frankfurt, Germany). The BMP type I receptor
kinase inhibitor LDN-193189 was purchased from Selleck Chemical
(Houston, TX, USA). The proteasome inhibitor MG132 was obtained
from Merck Millipore. The protease inhibitor cocktail and
phosphatase inhibitor cocktail 1 and 2 were purchased from
Sigma-Aldrich (St. Louis, MO, USA). All of the other reagents were
of analytical grade.
Cell culture
All of the cell lines were grown at 37°C in 5%
CO2. Human HSC-2 and HSC-4 squamous cell carcinoma cells
were cultured in MEM (Sigma-Aldrich) supplemented with 10% fetal
bovine serum (FBS) (Gibco, Rockville, MD, USA). SAS cells were
cultured in RPMI-1640 medium (Gibco) supplemented with 10% FBS.
HSC-3 cells were cultured in DMEM (Sigma-Aldrich) containing 10%
FBS. The culture medium was replaced with serum-free medium 24 h
prior to cytokine-stimulated experiments. In the majority of
experiments, 2.0×105 hOSCC cells were cultured in 500 µl
serum-free media containing 10 ng/ml TGF-β1 or 20 ng/ml BMP-2 for
the indicated time-periods in 24-well tissue culture plates.
Western blot analysis
For Smad phosphorylation analysis,
3.0×106 cells were lysed in RIPA buffer (Sigma-Aldrich)
containing a protease and phosphatase inhibitor cocktail
(Sigma-Aldrich) and the protein content was assessed with a BCA
reagent (Thermo Fisher Scientific, Waltham, MA, USA). To examine
marker protein expression, 1.0×106 cells were cultured
in a 6-well plate in serum-free MEM with or without 10 ng/ml TGF-β1
for the indicated time-points. Harvested cells were homogenized in
SDS sample buffer containing a protease and phosphatase inhibitor
cocktail (Sigma-Aldrich). Proteins were separated by 12.5% SDS-PAGE
(ATTO Co., Tokyo, Japan) and subsequently transferred onto PVDF
membranes (Merck Millipore). The membranes were probed with the
primary antibodies, including rabbit anti-Smad1 (#6944) and rabbit
anti-Smad5 (#9517; both from Cell Signaling Technology, Beverly,
MA, USA), rabbit anti-Smad9 [Smad8 (R-64); Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA] and rabbit
anti-phospho-Smad1 (Ser463/465)/Smad5 (Ser463/465)/Smad9
(Ser465/467) (#13820; Cell Signaling Technology). Mouse
anti-β-actin (clone C4; Santa Cruz Biotechnology, Inc.) was used as
a loading control in the Smad phosphorylation experiments. The
blots were then incubated with alkaline phosphatase-conjugated
secondary antibody and signals were visualized using an alkaline
phosphatase substrate kit (BCIP/NBT substrate kit; Vector
Laboratories, Inc., Burlingame, CA, USA).
Reverse transcriptase quantitative-PCR
(RT-qPCR)
For total RNA preparation, 2.0×105 cells
were cultured in 24-well tissue culture plates. Total RNA was
isolated using the Isogen reagent (Nippon Gene, Co., Ltd., Toyama,
Japan) according to the manufacturer's instructions. RNA was
reverse transcribed into first-strand cDNA with an RT-PCR system
kit (Takara Bio, Inc., Shiga, Japan). qPCR was performed on a
Thermal Cycler Dice Real-Time System (Takara Bio, Inc.) using
SYBR® Premix Ex Taq™ II (Takara Bio, Inc.) with human
gene-specific primers (Table I).
Target gene expression was normalized to an internal β-actin
reference and expressed in terms of fold-change relative to the
control sample (30).
 | Table I.Primer sequences for the qPCR
analysis. |
Table I.
Primer sequences for the qPCR
analysis.
| Genes | Primer
sequences |
|---|
| BMP-2 | F |
5′-AAGATTCCTAAGGCATGCTGTGTC-3′ |
|
| R |
5′-TCGTCAAGGTACAGCATCGAGA-3′ |
| Cytokeratin 9 | F |
5′-TCAGCTGACTGGGCAGAACA-3′ |
|
| R |
5′-ACCTCATGCTCGATCTGGGTTA-3′ |
| Cytokeratin 18 | F |
5′-AGGAGTATGAGGCCCTGCTGAA-3′ |
|
| R |
5′-TTGCATGGAGTTGCTGCTGTC-3′ |
| E-cadherin | F |
5′-TACACTGCCCAGGAGCCAGA-3′ |
|
| R |
5′-TGGCACCAGTGTCCGGATTA-3′ |
| ID1 | F |
5′-CGGAATCTGAGGGAGAACAAG-3′ |
|
| R |
5′-CTGAGAAGCACCAAACGTGA-3′ |
| N-cadherin | F |
5′-CGAATGGATGAAAGACCCATCC-3′ |
|
| R |
5′-TCGTCAAGGTACAGCATCGAGA-3′ |
| NEDD4 | F |
5′-GATTTGTAAACCGAATCCAGAAGCA-3′ |
|
| R |
5′-CCAGTCATTCACATCAACATCTCC-3′ |
| NEDD4L | F |
5′-CCAATGGGTCAGAAATAATGGTCA-3′ |
|
| R |
5′-AAGGCGTTCATCTGCTTCTGG-3′ |
| Smad1 | F |
5′-ACAGTCTGTGAACCATGGATTTGA-3′ |
|
| R |
5′-TGAGGTGAACCCATTTGAGTAAGAA-3′ |
| Samd5 | F |
5′-GCTTTCATCCCACCACTGTCTGTA-3′ |
|
| R |
5′-CCTGCCGGTGATATTCTGCTC-3′ |
| Smad6 | F |
5′-GAGCTGAGCCGAGAGAAAGA-3′ |
|
| R |
5′-AGATGCACTTGGAGCGAGTT-3′ |
| Smad7 | F |
5′-TGCAACCCCCATCACCTTAG-3′ |
|
| R |
5′-TCGTCAAGGTACAGCATCGAGA-3′ |
| Smad9 | F |
5′-TGGCCCAGTCAGTTCACCAC-3′ |
|
| R |
5′-CATGAAGATGAATCTCAATCCAGCA-3′ |
| Smurf1 | F |
5′-CCGCATCGAAGTGTCCAGAG-3′ |
|
| R |
5′-CCCACGGAATTTCACCATCAG-3′ |
| Smurf2 | F |
5′-TGCACTAACAACCTGCCGAAAG-3′ |
|
| R |
5′-CTTGTCATTCCACAGCAAATCCAC-3′ |
| Snail | F |
5′-GACCACTATGCCGCGCTCTT-3′ |
|
| R |
5′-TCGCTGTAGTTAGGCTTCCGATT-3′ |
| β-actin | F |
5′-GGAGATTACTGCCCTGGCTCCTA-3′ |
|
| R |
5′-GACTCATCGTACTCCTGCTTGCTG-3′ |
Proliferation assay
HSC-4 cell proliferation was evaluated by
alamarBlue® assay (AbD Serotec, Oxon, UK) according to
the manufacturer's instructions. Briefly, 1.0×104
cells/well were subcultured in 96-well plates in MEM supplemented
with 10% FBS for 24 h. The culture medium was then replaced with
MEM supplemented without FBS with or without TGF-β1 (10 ng/ml) or
BMP-2 (20 ng/ml) for 2 days. The cells were washed once with PBS
and then incubated with 100 µl alamarBlue® (10%
alamarBlue® in Ham's F-12) at 37°C in 5% CO2
for 1.5 h. The absorbances at 570 and 600 nm were used to measure
the reduced and oxidized forms of reagent, respectively, using a
microplate reader. Assays were performed independently at least 3
times (n=6).
Cell migration assays
Boyden chamber migration assays were performed using
cells transfected with Slug siRNA as aforementioned. Then, cells
were treated with 10 ng/ml TGF-β1 or 20 ng/ml BMP-2 under
serum-free conditions for 72 h and subsequently plated at a density
of 1.0×105 cells in the upper chamber of a Boyden
apparatus in serum-free media, and were allowed to migrate into the
lower chamber containing medium supplemented with 10% FBS for 24 h
at 37°C. The chamber filter was fixed in 4% paraformaldehyde and
stained with DAPI (1:500; Sigma-Aldrich) for 10 min. Migrating
cells were counted in 9 fields on the membrane. Values indicate the
mean number of migrating cells compared to the controls.
Statistical analysis
All experiments were performed at least 3 times
independently. Results are expressed as the means ± standard
deviation (SD). Data were analyzed using two-tailed, independent
Student's t-tests. P<0.05 was considered statistically
significant.
Results
BMP-2 responsiveness in the hOSCC cell
lines
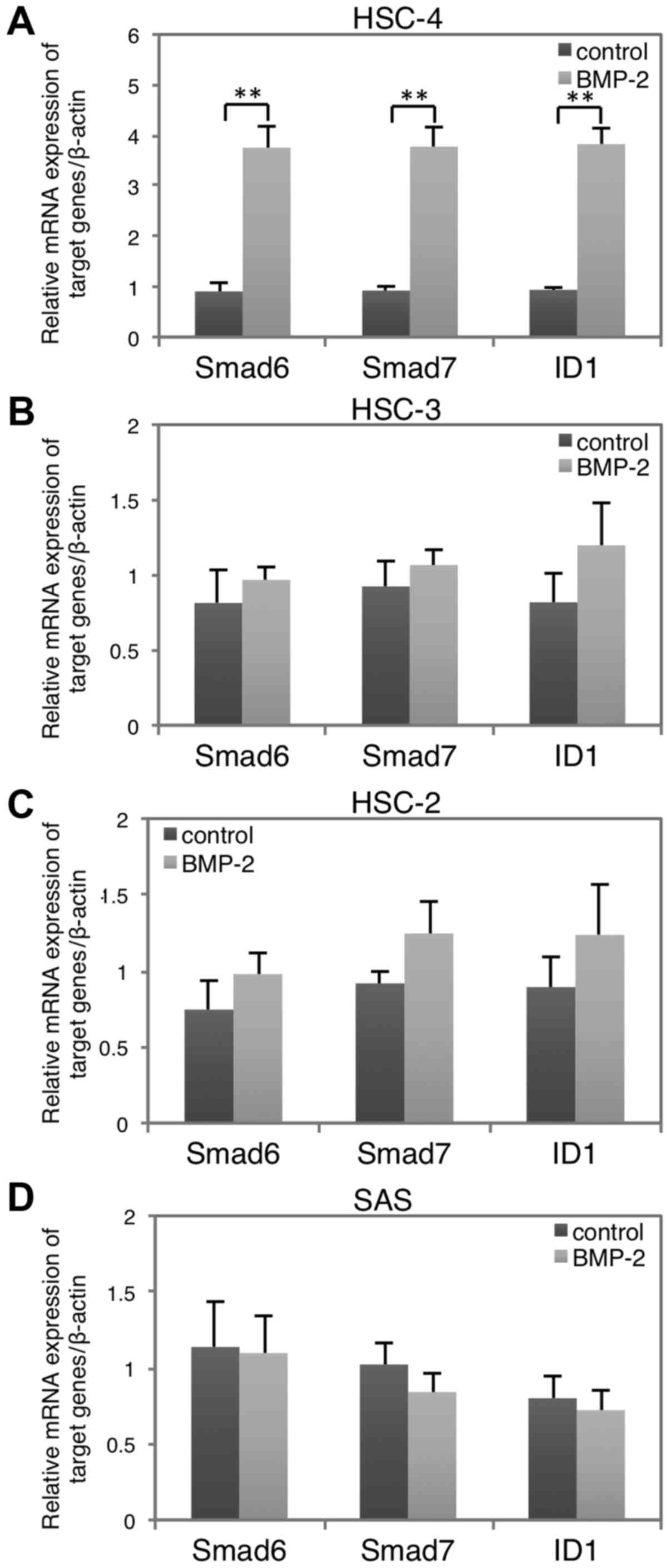
We first studied the cellular response to BMP-2 in
four hOSCC cell lines: HSC-2, HSC-3, HSC-4, and SAS. Smad6, Smad7
and ID1 are targets of BMP signaling (31). Expression analysis revealed that
BMP-2 induced a significant upregulation of all three target genes
in the HSC-4 cells (Fig. 1A), but
no marked changes were observed in the other cell lines (Fig. 1B-D). Thus, we used HSC-4 cells to
investigate the effects of BMP-2 on MET in the following
experiments. In addition, HSC-4 cells are also responsive to TGF-β1
stimulation based on our previous examinations (28).
BMP-2-induced response in HSC-4
cells
To confirm BMP-2 signaling in HSC-4 cells, the time
course of Smad6 and ID1 gene expression was investigated in the
presence or absence of LDN-193189, a selective inhibitor of activin
receptor-like kinase (ALK)-2/3 (Fig. 2A
and B). The results showed that both genes were upregulated 1 h
after BMP-2 treatment, peaking at 3 h post-stimulation, which was
significantly inhibited by LDN-193189. Moreover, Smad1/5/9
phosphorylation was increased in the BMP-2-treated HSC-4 cells, but
inhibited in the presence of LDN-193189 (Fig. 2C). Thus, these results indicated
that BMP-2 elicits Smad1/5/9 activation and target gene expression
in HSC-4 cells.
Effects of BMP-2 or TGF-β1 on
epithelial and mesenchymal status in HSC-4 cells
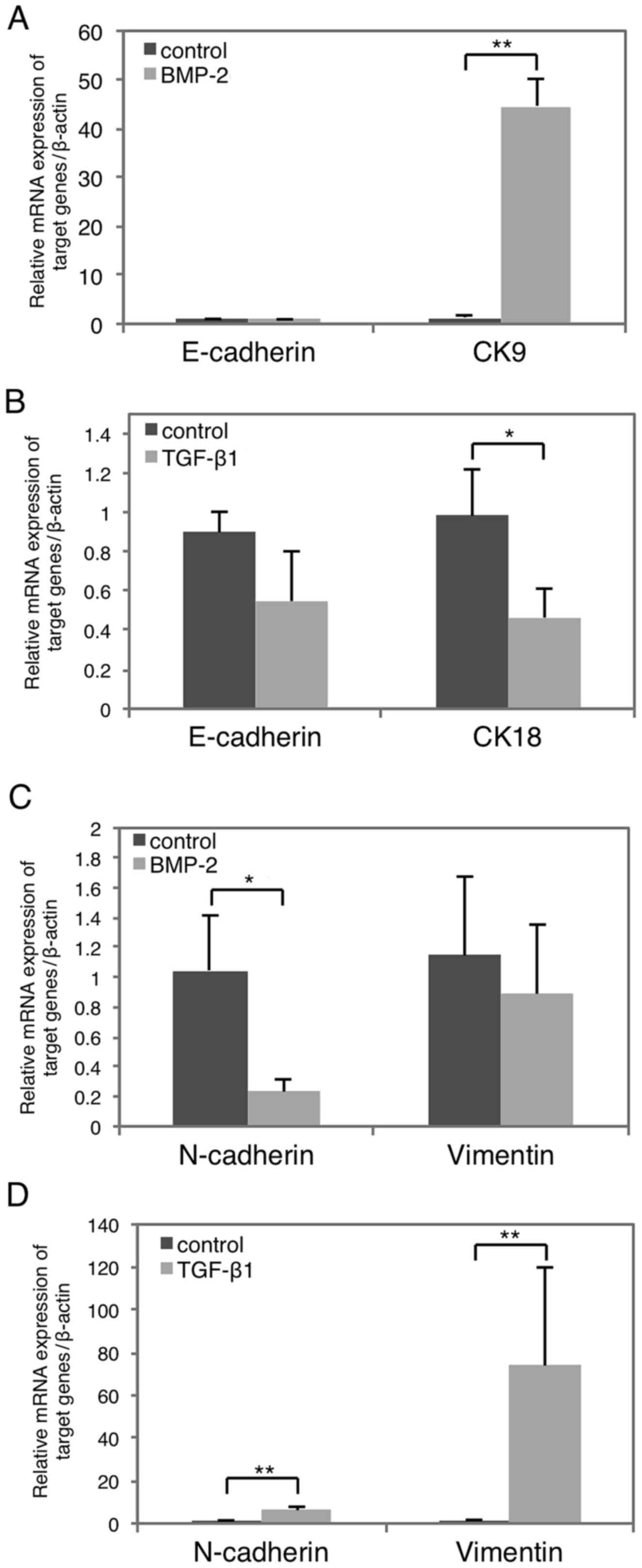
Next, we examined how BMP-2 alters the epithelial
and mesenchymal characteristics of HSC-4 cells by examining marker
expression with RT-qPCR (Fig. 3).
Notably, the epithelial marker cytokeratin 9 (CK9) was clearly
upregulated following BMP-2-stimulation (Fig. 3A, right), whereas that of N-cadherin
was significantly suppressed (Fig.
3C, left). Conversely, TGF-β1 stimulation resulted in the
significant suppression of epithelial cytokeratin 18 (CK18) marker
(Fig. 3B, right), as well as the
induction of the mesenchymal markers N-cadherin and vimentin
(Fig. 3D). However, the expression
of epithelial marker E-cadherin was not affected by either BMP-2 or
TGF-β1 stimulation (Fig. 3A and B,
left), and mesenchymal vimentin was not significantly affected by
BMP-2 (Fig. 3C, right). Moreover,
the morphology of BMP-2-treated HSC-4 cells was not different from
that of control cells (data not shown). Collectively, these data
indicated that TGF-β1 suppresses epithelial marker expression and
promotes that of mesenchymal markers, resulting in EMT. In
contrast, BMP-2 has the opposite effect promoting MET.
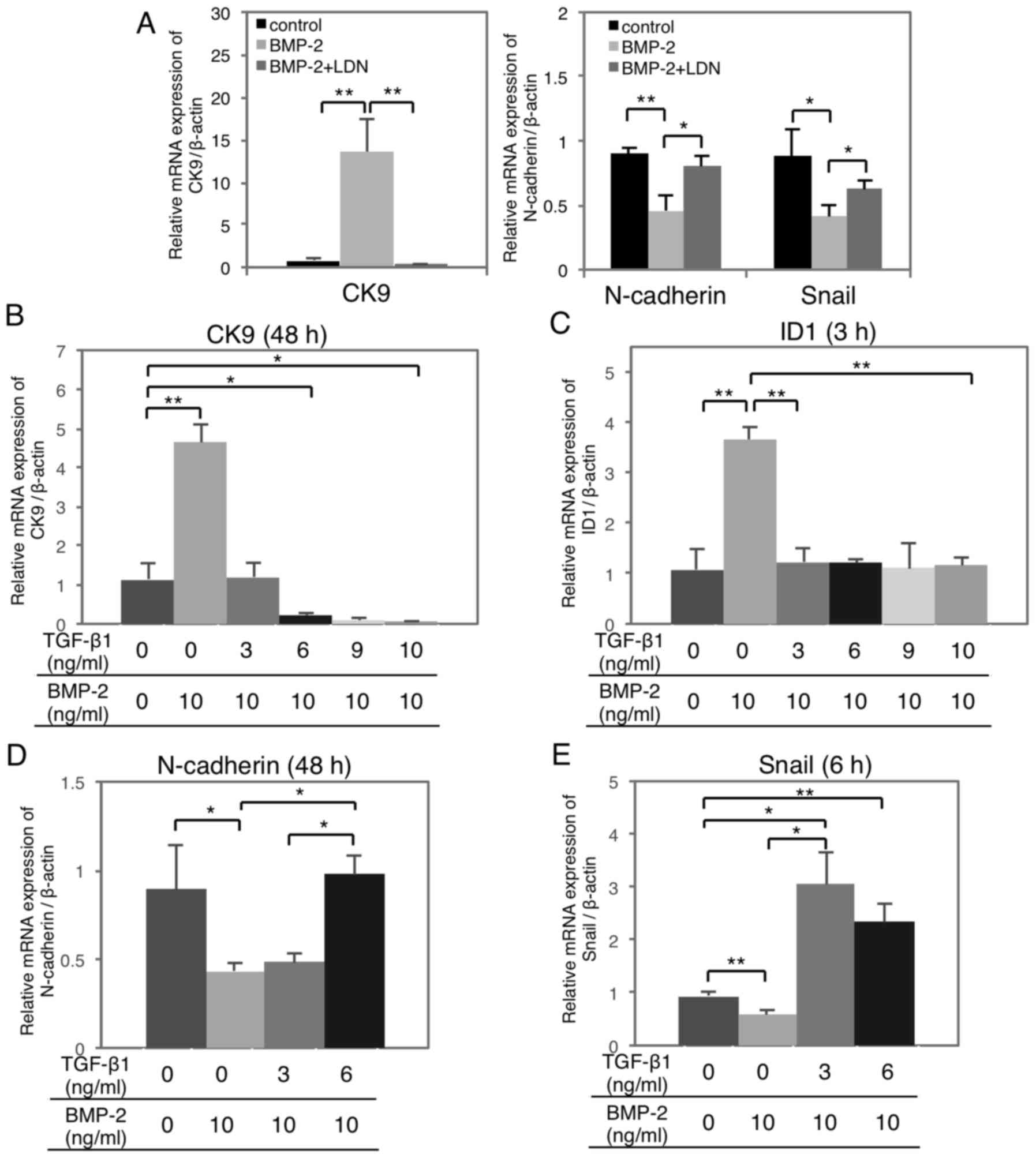
TGF-β1 abrogates BMP-2-mediated
effects on MET/EMT gene expression in a dose-dependent manner
To examine the function of TGF-β1 on BMP-2-induced
MET, HSC-4 cells were treated with increasing doses of TGF-β1 with
or without BMP-2 stimulation. BMP-2-induced epithelial CK9
(Fig. 4A, left) and ID1
upregulation (Fig. 2B), whereas the
N-cadherin and Snail downregulation (Fig. 4A, right) was significantly
suppressed by LDN-193189. Moreover, BMP-2-induced CK9 and ID1
upregulation was significantly suppressed by TGF-β1 in a
dose-dependent manner (Fig. 4B and
C). Similarly, BMP-2-induced N-cadherin and Snail
downregulation were significantly suppressed by TGF-β1 stimulation
in a dose-dependent manner (Fig. 4D and
E).
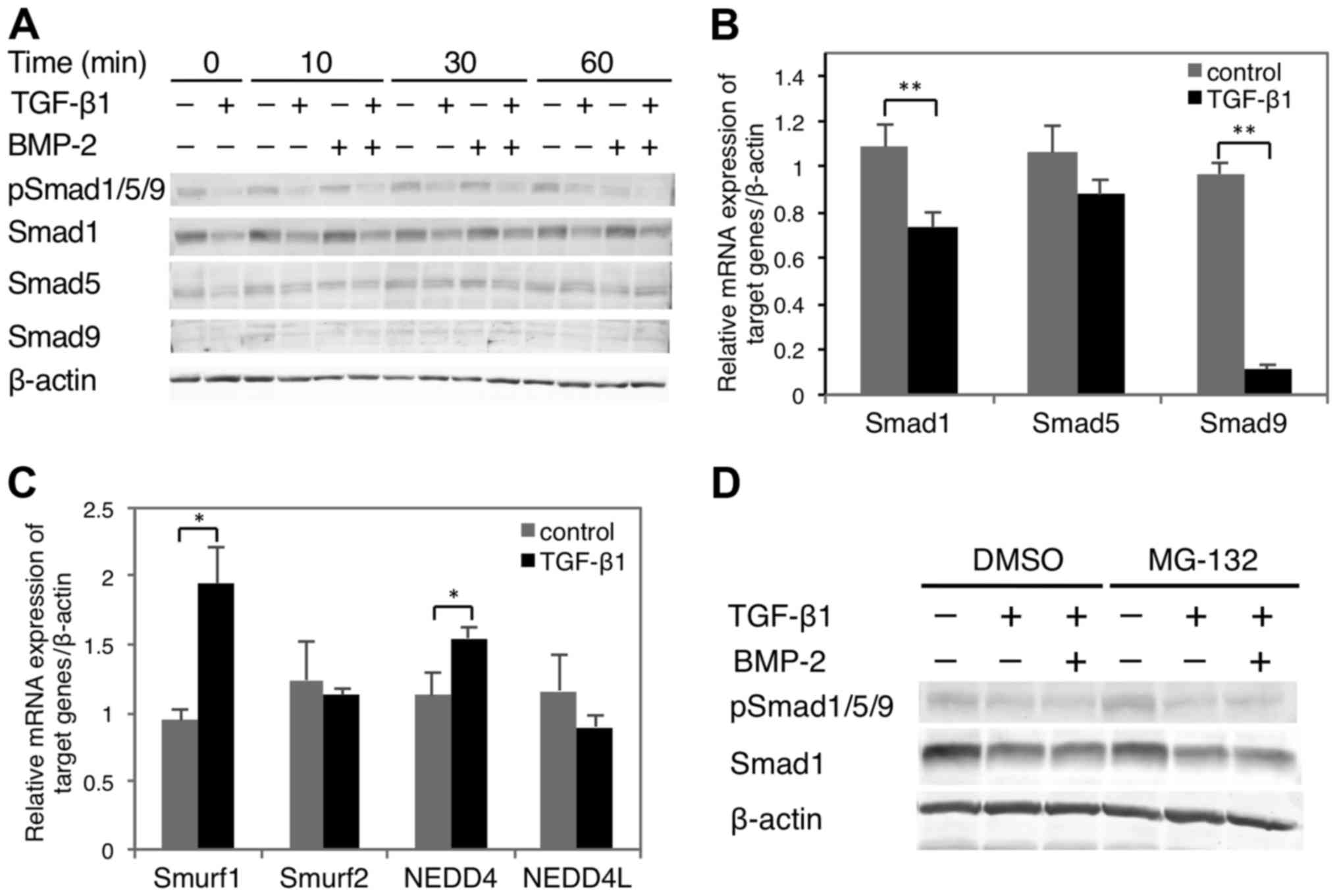
Effect of TGF-β1 on Smad1/5/9
expression and phosphorylation
Western blot analysis showed that TGF-β1 hindered
Smad1/5/9 phosphorylation in HSC-4 cells in the presence or absence
of BMP-2 (Fig. 5A). In addition,
while TGF-β1 suppressed Smad1 expression independently of BMP-2, it
had no marked effect on Smad5 expression. Smad9 expression was
undetectable regardless of exogenous stimulation. With respect to
RNA expression, both Smad1 and 9 were significantly decreased by
TGF-β1 stimulation, whereas that of Smad5 was unaffected (Fig. 5B). TGF-β1 stimulation alters the
levels of phosphorylated (p)Smad1/5 and pSmad2 by regulating the E3
ligase activities of Smurf1 (32,33),
NEDD4 (34) Smurf2 (35) and NEDD4L (36), respectively. Notably, Smurf1 and
NEDD4 were significantly upregulated by TGF-β1 stimulation, whereas
Smurf2 and NEDD4L were mostly unchanged (Fig. 5C). However, the TGF-β1-mediated
degradation of BMP-2-induced pSmad1/5/9 remained intact after
treatment with MG132 proteasome inhibitor. This suggested that BMP
signal attenuation by TGF-β1 occurs in a proteasome-independent
manner (Fig. 5D), possibly through
the Smad1/9 downregulation (Fig.
5B). Thus, the effect of TGF-β1 on BMP-2 signaling likely
results from the suppression of Smad1/9 expression rather than
phosphorylation.
Effect of BMP-2 and TGF-β1 on HSC-4
cell migration and proliferation
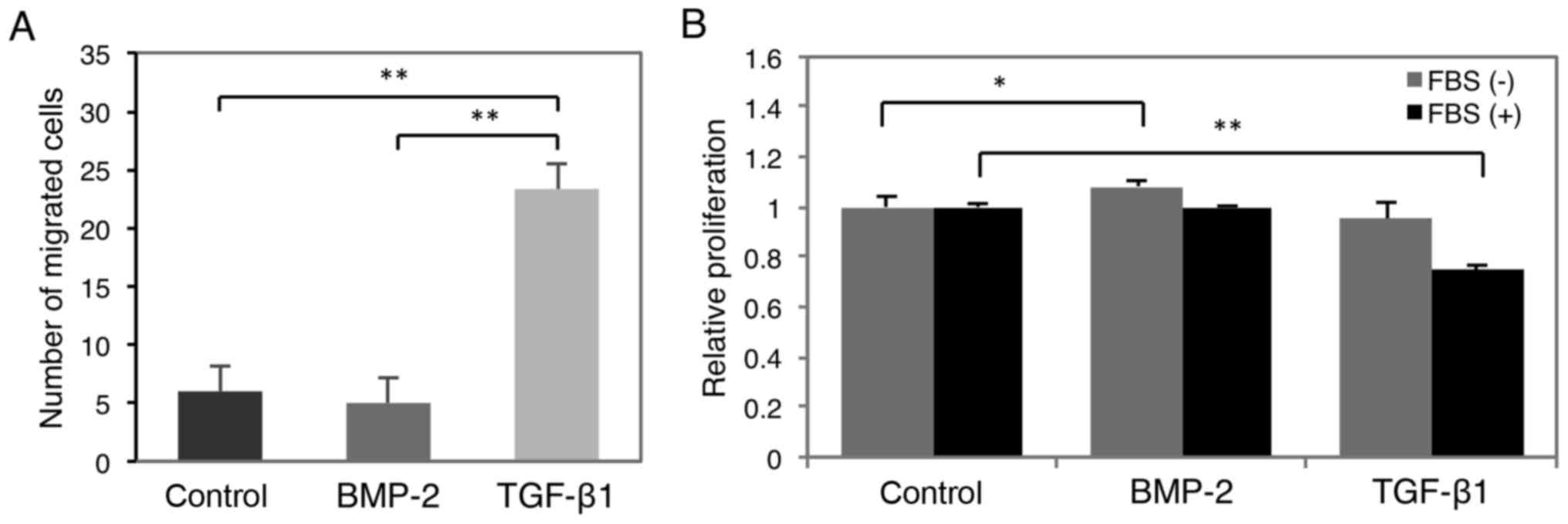
The effect of BMP-2 on cell migration and
proliferation was investigated to assess its potential effect on
hOSCC progression (Fig. 6). This
analysis revealed that TGF-β1 significantly enhanced the migratory
capacity of HSC-4 cells 72 h after TGF-β1 stimulation, whereas
BMP-2 did not (Fig. 6A).
Alternatively, BMP-2 facilitated cell proliferation 48 h after
stimulation, but was significantly suppressed in the presence of
TGF-β1 (Fig. 6B).
Discussion
In order to detect BMP-2-responsive hOSCC, Smad6 and
ID1 expression (Fig. 1) and
Smad1/5/9 phosphorylation (Fig. 2)
were examined in four hOSCC cell lines after BMP-2 stimulation.
HSC-4 cells were the most responsive to both BMP-2 and TGF-β1 and
thus selected for further analysis. Results from the present study
demonstrate that BMP-2 promoted the expression of epithelial CK9,
but suppressed that of the mesenchymal markers N-cadherin and
vimentin (Fig. 3C), suggesting that
it promotes MET. Moreover, these BMP-2-inducible effects were
significantly inhibited by TGF-β1 stimulation in HSC-4 cells in a
dose-dependent manner (Fig. 4B-E).
Collectively, these results strongly suggest that TGF-β1 inhibits
BMP-2-induced MET in hOSCC.
Further analysis revealed that TGF-β1 suppressed
BMP-2-induced Smad1/5/9 phosphorylation (Fig. 5A) and Smad1/9 expression (Fig. 5B) and promoted the expression of E3
protein ligases that target BMP-2 pathway effectors in HSC-4 cells
(Fig. 5C). As such, TGF-β1 is
likely a key regulator of Smad1/5/9 pathway suppression downstream
of BMP-2 in hOSCC cells. ID (inhibitor of DNA binding) proteins are
a family of four transcriptional regulators, including ID1
(31). ID1 expression is reportedly
enhanced by BMP-2-mediated Smad1/5 pathway activity (Figs. 1A, 2B and 2C)
(37,38). ID proteins have significant
implications in cancer progression as Stankic et al reported
that ID1 induced MET during metastatic breast cancer cell
colonization (39). Moreover, Del
Pozo Martin et al (40)
reported that metastatic colonization is induced by the interaction
between mesenchymal cancer cells and stromal fibroblasts, which
secrete factors to induce MET via BMP/Smad1/5 signaling. Our study
showed that ID1 protein expression was increased when cultured in
activated fibroblast-conditioned media, but was blocked by
LDN-193189 treatment. Based on these data, TGF-β1 may suppress MET
by disrupting BMP-2-mediated Smad1/5/9 signaling, resulting in ID1
downregulation in HSC-4 cells. On the other hand, Snail is
upregulated during EMT and generates a positive feedback loop
(10). Notably, Snail expression
was significantly suppressed by BMP-2 in HSC-4 cells (Fig. 4A, right and 4E); however, whether
BMP-2-induced Smad1/5/9 signaling plays an important role in Snail
suppression in HSC-4 cells remains unclear.
Cancer metastasis is the result of cancer cell MET,
as well as their proliferative burst after homing to these
metastatic sites (12,40). As shown in Fig. 6B, BMP-2 significantly induced HSC-4
cell proliferation, which was not observed following TGF-β1
treatment. If hOSCC cells are susceptible to BMP-2 stimulation at
metastatic sites, they likely retain a high proliferative capacity
to promote secondary tumor formation. Collectively, this evidence
supports that BMP-2 positively regulates metastatic colonization in
hOSCC. On the other hand, TGF-β1 induces EMT (28) and increases cell migration (Fig. 6A) and invasion (29) in primary hOSCC tumors. In addition,
TGF-β1 may inhibit tumor progression by attenuating BMP-2-induced
MET at metastatic sites.
Yang et al (20) reported that BMP-2 suppresses EMT in
TGF-β1-induced renal interstitial fibrosis. Interestingly, BMP-2
attenuated TGF-β1-induced EMT of NRK-49F kidney fibroblasts
downregulating Snail expression. Alternatively, we found that the
BMP-2-induced Snail downregulation was significantly inhibited by
TGF-β1 stimulation in a dose-dependent manner (Fig 4E), suggesting that TGF-β1 suppresses
the BMP-2-induced MET by disrupting the induction of Snail in hOSCC
cells. Recently, it was reported that BMP-4 may inhibit
TGF-β1-induced EMT in primary retinal pigment epithelium cells
through the Smad2/3 pathway (41).
Therefore, it will be necessary to determine whether TGF-β1-induced
EMT is inhibited by BMP stimulation in hOSCC cells in the
future.
Our findings partly clarify the molecular mechanisms
underlying EMT and MET in hOSCC and may facilitate the discovery of
molecular drug targets to attenuate hOSCC progression.
Acknowledgements
We would like to thank Editage (www.editage.jp) for English language editing. This
study was supported in part by a Grant-in-aid for Scientific
Research (no. 90118274 to S.K., no. 26293426 to T.S. and no.
2667052 and 16H05534 to A.I.) from the Ministry of Education,
Culture, Sports, Science, and Technology of Japan; Grant-in-aid for
the Strategic Medical Science Research Center from the Ministry of
Education, Culture, Sports, Science and Technology of Japan,
2010–2014.
Glossary
Abbreviations
Abbreviations:
|
BMP
|
bone morphogenetic protein
|
|
CK9
|
cytokeratin 9
|
|
CK18
|
cytokeratin 18
|
|
EMT
|
epithelial-mesenchymal transition
|
|
hOSCC
|
human oral squamous cell carcinoma
|
|
MET
|
mesenchymal-epithelial transition
|
|
TGF-β
|
transforming growth factor-β
|
References
|
1
|
Smith A, Teknos TN and Pan Q: Epithelial
to mesenchymal transition in head and neck squamous cell carcinoma.
Oral Oncol. 49:287–292. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial- mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Weber CE, Li NY, Wai PY and Kuo PC:
Epithelial-mesenchymal transition, TGF-β, and osteopontin in wound
healing and tissue remodeling after injury. J Burn Care Res.
33:311–318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
He J, Xu Y, Koya D and Kanasaki K: Role of
the endothelial-to-mesenchymal transition in renal fibrosis of
chronic kidney disease. Clin Exp Nephrol. 17:488–497. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fabregat I, Malfettone A and Soukupova J:
New insights into the crossroads between EMT and stemness in the
context of cancer. J Clin Med. 5:372016. View Article : Google Scholar
|
|
6
|
Moustakas A and Heldin P: TGFβ and
matrix-regulated epithelial to mesenchymal transition. Biochim
Biophys Acta. 1840:2621–2634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Batlle E, Sancho E, Francí C, Domínguez D,
Monfar M, Baulida J and De Herreros A García: The transcription
factor snail is a repressor of E-cadherin gene expression in
epithelial tumour cells. Nat Cell Biol. 2:84–89. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Medici D, Hay ED and Olsen BR: Snail and
Slug promote epithelial-mesenchymal transition through
β-catenin-T-cell factor-4-dependent expression of transforming
growth factor-β3. Mol Biol Cell. 19:4875–4887. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zeisberg M and Neilson EG: Biomarkers for
epithelial-mesenchymal transitions. J Clin Invest. 119:1429–1437.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Peinado H, Olmeda D and Cano A: Snail, Zeb
and bHLH factors in tumour progression: An alliance against the
epithelial phenotype? Nat Rev Cancer. 7:415–428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wakefield LM and Hill CS: Beyond TGFβ:
Roles of other TGFβ superfamily members in cancer. Nat Rev Cancer.
13:328–341. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Simic P and Vukicevic S: Bone
morphogenetic proteins in development and homeostasis of kidney.
Cytokine Growth Factor Rev. 16:299–308. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bragdon B, Moseychuk O, Saldanha S, King
D, Julian J and Nohe A: Bone morphogenetic proteins: A critical
review. Cell Signal. 23:609–620. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu M, Chen G and Li YP: TGF-β and BMP
signaling in osteoblast, skeletal development and bone formation,
homeostasis and disease. Bone Res. 4:160092016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kopf J, Paarmann P, Hiepen C, Horbelt D
and Knaus P: BMP growth factor signaling in a biomechanical
context. Biofactors. 40:171–187. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen G, Deng C and Li YP: TGF-β and BMP
signaling in osteoblast differentiation and bone formation. Int J
Biol Sci. 8:272–288. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
McCormack N and O'Dea S: Regulation of
epithelial to mesenchymal transition by bone morphogenetic
proteins. Cell Signal. 25:2856–2862. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kang MH, Kim JS, Seo JE, Oh SC and Yoo YA:
BMP2 accelerates the motility and invasiveness of gastric cancer
cells via activation of the phosphatidylinositol 3-kinase
(PI3K)/Akt pathway. Exp Cell Res. 316:24–37. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang YL, Ju HZ, Liu SF, Lee TC, Shih YW,
Chuang LY, Guh JY, Yang YY, Liao TN, Hung TJ, et al: BMP-2
suppresses renal interstitial fibrosis by regulating
epithelial-mesenchymal transition. J Cell Biochem. 112:2558–2565.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Graves CA, Abboodi FF, Tomar S, Wells J
and Pirisi L: The translational significance of
epithelial-mesenchymal transition in head and neck cancer. Clin
Transl Med. 3:602014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lambert R, Sauvaget C, de Camargo Cancela
M and Sankaranarayanan R: Epidemiology of cancer from the oral
cavity and oropharynx. Eur J Gastroenterol Hepatol. 23:633–641.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kokorina NA, Lewis JS Jr, Zakharkin SO,
Krebsbach PH and Nussenbaum B: rhBMP-2 has adverse effects on human
oral carcinoma cell lines in vivo. Laryngoscope. 122:95–102. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kokorina NA, Zakharkin SO, Krebsbach PH
and Nussenbaum B: Treatment effects of rhBMP-2 on invasiveness of
oral carcinoma cell lines. Laryngoscope. 121:1876–1880.
2011.PubMed/NCBI
|
|
25
|
Sand JP, Kokorina NA, Zakharkin SO, Lewis
JS Jr and Nussenbaum B: BMP-2 expression correlates with local
failure in head and neck squamous cell carcinoma. Otolaryngol Head
Neck Surg. 150:245–250. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kejner AE, Burch MB, Sweeny L and
Rosenthal EL: Bone morphogenetic protein 6 expression in oral
cavity squamous cell cancer is associated with bone invasion.
Laryngoscope. 123:3061–3065. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Quan J, Johnson NW, Zhou G, Parsons PG,
Boyle GM and Gao J: Potential molecular targets for inhibiting bone
invasion by oral squamous cell carcinoma: A review of mechanisms.
Cancer Metastasis Rev. 31:209–219. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Saito D, Kyakumoto S, Chosa N, Ibi M,
Takahashi N, Okubo N, Sawada S, Ishisaki A and Kamo M: Transforming
growth factor-β1 induces epithelial-mesenchymal transition and
integrin α3β1-mediated cell migration of HSC-4 human squamous cell
carcinoma cells through Slug. J Biochem. 153:303–315. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hino M, Kamo M, Saito D, Kyakumoto S,
Shibata T, Mizuki H and Ishisaki A: Transforming growth factor-β1
induces invasion ability of HSC-4 human oral squamous cell
carcinoma cells through the Slug/Wnt-5b/MMP-10 signalling axis. J
Biochem. 159:631–640. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT Method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lasorella A, Benezra R and Iavarone A: The
ID proteins: Master regulators of cancer stem cells and tumour
aggressiveness. Nat Rev Cancer. 14:77–91. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shi W, Chen H, Sun J, Chen C, Zhao J, Wang
YL, Anderson KD and Warburton D: Overexpression of Smurf1
negatively regulates mouse embryonic lung branching morphogenesis
by specifically reducing Smad1 and Smad5 proteins. Am J Physiol
Lung Cell Mol Physiol. 286:L293–L300. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Murakami G, Watabe T, Takaoka K, Miyazono
K and Imamura T: Cooperative inhibition of bone morphogenetic
protein signaling by Smurf1 and inhibitory Smads. Mol Biol Cell.
14:2809–2817. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim BG, Lee JH, Yasuda J, Ryoo HM and Cho
JY: Phospho-Smad1 modulation by nedd4 e3 ligase in BMP/TGF-β
signaling. J Bone Miner Res. 26:1411–1424. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin X, Liang M and Feng XH: Smurf2 is a
ubiquitin E3 ligase mediating proteasome-dependent degradation of
Smad2 in transforming growth factor-β signaling. J Biol Chem.
275:36818–36822. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gao S, Alarcón C, Sapkota G, Rahman S,
Chen PY, Goerner N, Macias MJ, Erdjument-Bromage H, Tempst P and
Massagué J: Ubiquitin ligase Nedd4L targets activated Smad2/3 to
limit TGF-β signaling. Mol Cell. 36:457–468. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ogata T, Wozney JM, Benezra R and Noda M:
Bone morphogenetic protein 2 transiently enhances expression of a
gene, Id (inhibitor of differentiation), encoding a
helix-loop-helix molecule in osteoblast-like cells. Proc Natl Acad
Sci USA. 90:9219–9222. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Langenfeld EM, Kong Y and Langenfeld J:
Bone morphogenetic protein 2 stimulation of tumor growth involves
the activation of Smad-1/5. Oncogene. 25:685–692. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Stankic M, Pavlovic S, Chin Y, Brogi E,
Padua D, Norton L, Massagué J and Benezra R: TGF-β-Id1 signaling
opposes Twist1 and promotes metastatic colonization via a
mesenchymal-to-epithelial transition. Cell Reports. 5:1228–1242.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Del Pozo Martin Y, Park D, Ramachandran A,
Ombrato L, Calvo F, Chakravarty P, Spencer-Dene B, Derzsi S, Hill
CS, Sahai E, et al: Mesenchymal cancer cell-stroma crosstalk
promotes niche activation, epithelial reversion, and metastatic
colonization. Cell Reports. 13:2456–2469. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yao H, Li H, Yang S, Li M, Zhao C, Zhang
J, Xu G and Wang F: Inhibitory effect of bone morphogenetic protein
4 in retinal pigment epithelial-mesenchymal transition. Sci Rep.
6:321822016. View Article : Google Scholar : PubMed/NCBI
|