Introduction
Fanconi anemia (FA) is a heterogeneous autosomal
recessive disease characterized by congenital malformations,
progressive bone marrow failure and an increased incidence of
cancer. In contrast to normal fibroblasts, FA fibroblasts display
elevated spontaneous chromosomal breaks and deletions and nuclear
extracts that have substantially decreased plasmid-rejoining
activity (1,2). Epanchintsev et al demonstrated
the overproduction of secretory factors such as interleukin (IL)-6,
IL-8, matrix metalloproteinase (MMP)-2, and MMP-9 in FA and showed
that these overexpressed secretory factors were effective in
promoting the proliferation, migration and invasion of surrounding
tumor cells (3). Ibáňez et
al described an anomalous high level of the pro-inflammatory
cytokine IL-1β present in the serum of FA patients which activated
the proliferation of tumor cells (4). Increased levels of MMP-9 have been
shown to be associated with cancer progression and poorer patient
prognosis due to the significant role MMP-9 plays in tumor cell
invasion and metastasis by digesting the basement membrane and
components of the extracellular matrix (5–7). MMP
activity is regulated by and dependent upon environmental
influences from surrounding stroma cells, ECM proteins, systemic
hormones and other factors (5,8,9).
Furthermore, MMPs are regulated at multiple levels, including
transcription, modulation of messenger RNA half-life (translation),
secretion, localization, activation and inhibition (10).
In the present study we investigated the effects of
selected cytokines, inducers and inhibitors affecting cancer cell
metabolism on the regulation of MMP-2 and MMP-9 activities in FA
fibroblast cell lines.
Materials and methods
Materials
Human FA fibroblast cell lines A:PD20 and A:PD220
were obtained from the Fanconi Anemia Research Fund, Oregon Health
& Science University (Portland, OR, USA). Antibiotics,
penicillin and fetal bovine serum (FBS), were obtained from
Gibco-BRL (Long Island, NY, USA). Twenty-four well tissue culture
plates were obtained from Costar (Cambridge, MA, USA). Gelatinase
zymography was performed on 10% Novex pre-cast SDS polyacrylamide
gel (Invitrogen Inc., Carlsbad, CA, USA) with 0.1% gelatin in
non-reducing conditions. IL-1β, tumor necrosis factor-α (TNF-α),
phorbol 12-myristate 13-acetate (PMA), lipopolysaccharide (LPS),
doxycycline, epigallocatechin gallate (EGCG), actinomycin-D,
cyclohexamide, retinoic acid and dexamethasone were purchased from
Sigma-Aldrich (St. Louis, MO, USA). The nutrient mixture (NM),
prepared by VitaTech (Hayward, CA, USA) was composed of the
following ingredients in the relative amounts indicated: vitamin C
(as ascorbic acid and as Mg, Ca and palmitate ascorbate) 700 mg;
L-lysine 1,000 mg; L-proline 750 mg; L-arginine 500 mg;
N-acetyl cysteine 200 mg; standardized green tea extract
(80% polyphenol) 1,000 mg; selenium 30 µg; copper 2 mg; manganese 1
mg. All other reagents used were of high quality and were obtained
from Sigma-Aldrich, unless otherwise indicated.
Cell cultures
FA fibroblasts were grown in Dulbeccos modified
Eagles medium (DMEM), supplemented with 15% FBS, 100 U/ml
penicillin and 100 µg/ml streptomycin in 24-well tissue culture
plates. The cells were plated at a density of 1×105
cells/ml and grown to confluency in a humidified atmosphere of 5%
CO2 at 37°C. Serum-supplemented media were removed and
the cell monolayer was washed once with phosphate-buffered saline
(PBS) and with the recommended serum-free media. The cells were
then incubated in 0.5 ml of serum-free medium with various
cytokines, mitogens, inducers and inhibitors in triplicates, as
indicated: PMA (10, 25, 50 and 100 ng/ml); TNF-α (0.1, 1, 10 and 25
ng/ml); IL-1β (0.1, 1, 10 and 25 ng/ml); LPS (10, 25, 50 and 100
µg/ml); EGCG (10, 25, 50 and 100 µM) without and with PMA 100
ng/ml; doxycycline (10, 25, 50 and 100 µM) without and with PMA 100
ng/ml; NM (10, 50, 100, 500 and 1,000 µg/ml) with PMA 100 ng/ml,
retinoic acid (50 µM); dexamethasone (50 µM); actinomycin-D and
cyclohexamide (2 and 4 µg/ml). The plates were then returned to the
incubator. The conditioned medium from each treatment was
separately collected, pooled and centrifuged at 4°C for 10 min at
3,000 rpm to remove cells and cell debris. The clear supernatant
was collected and used for gelatinase zymography as described
below.
Gelatinase zymography
Gelatinase zymography was utilized due to its high
sensitivity to gelatinolytic enzymatic activity and ability to
detect both pro- and active forms of MMP-2 and MMP-9. Upon
renaturation of the enzyme, the gelatinases digest the gelatin in
the gel and reveal clear bands against an intensely stained
background. Gelatinase zymography was performed on 10% Novex
pre-cast SDS polyacrylamide gel in the presence of 0.1% gelatin
under non-reducing conditions. Culture media (20 µl) were mixed
with sample buffer and loaded for SDS-PAGE with Tris-glycine-SDS
buffer, as suggested by the manufacturer (Novex). Samples were not
boiled before electrophoresis. Following electrophoresis the gels
were washed twice in 2.5% Triton X-100 for 30 min at room
temperature to remove SDS. The gels were then incubated at 37°C
overnight in a substrate buffer containing 50 mM Tris-HCl and 10 mM
CaCl2 at pH 8.0 and stained with 0.5% Coomassie Blue
R250 in 50% methanol and 10% glacial acetic acid for 30 min and
destained. Protein standards were run concurrently and approximate
molecular weights were determined by plotting the relative
mobilities of known proteins. Gelatinase zymograms were scanned
using CanoScan 9950F Canon scanner at 300 dpi. The intensity of the
bands was evaluated using the pixel-based densitometer program
Un-Scan-It, version 5.1, 32-bit, by Silk Scientific Corporation
(Orem, UT, USA), at a resolution of 1 scanner unit (1/100 of an
inch for an image that was scanned at 100 dpi).
Results
Inducers and cytokines
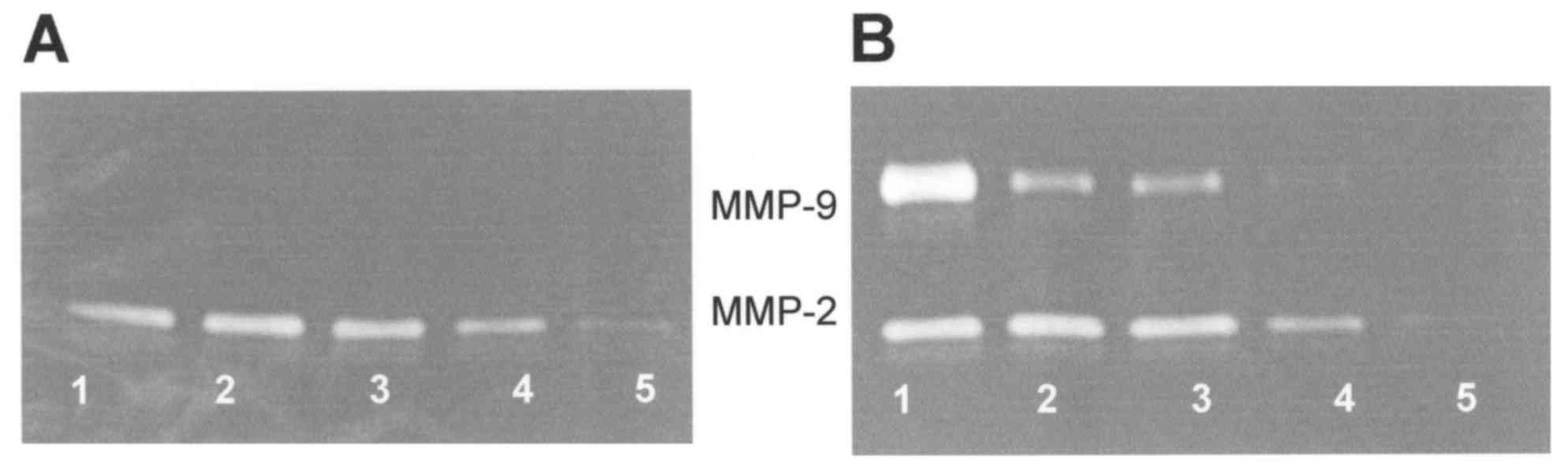
Both FA cell lines A:PD20 and A:PD220 expressed only
one band corresponding to MMP-2. Cytokines, mitogens, inducers and
inhibitors had a similar effect on MMP-2 and PMA-induced MMP-9
expression in both FA fibroblasts. Therefore, only data for FA
A:PD20 is presented. Table I shows
the quantitative densitometry results from the effects of PMA,
TNF-α, IL-1β and LPS on MMP-2 and MMP-9 expression in the FA
fibroblasts.
 | Table I.Effect of inducers on Fanconi anemia
fibroblast MMP-2 and MMP-9 secretion. |
Table I.
Effect of inducers on Fanconi anemia
fibroblast MMP-2 and MMP-9 secretion.
| Inducers | MMP-2 (%) | MMP-9 (%) |
|---|
| PMA (ng/ml) |
|
|
| Control | 100 | 0 |
| 10 | 126 | 16.5 |
| 25 | 217 | 33.5 |
| 50 | 236 | 32 |
| 100 | 167 | 19 |
| TNF-α (ng/ml) |
|
|
| Control | 100 | 100 |
| 0.1 | 156 | 365 |
| 1 | 140 | 622 |
| 10 | 90 | 5,660 |
| 25 | 92 | 6,487 |
| IL-1β (ng/ml) |
|
|
| Control | 100 | 100 |
| 0.1 | 82 | 145 |
| 1 | 121 | 880 |
| 10 | 173 | 1,216 |
| 25 | 111 | 700 |
| LPS (µg/ml) |
|
|
| Control | 100 |
|
| 10 | 118 |
|
| 25 | 170 |
|
| 50 | 104 |
|
| 100 | 65 |
|
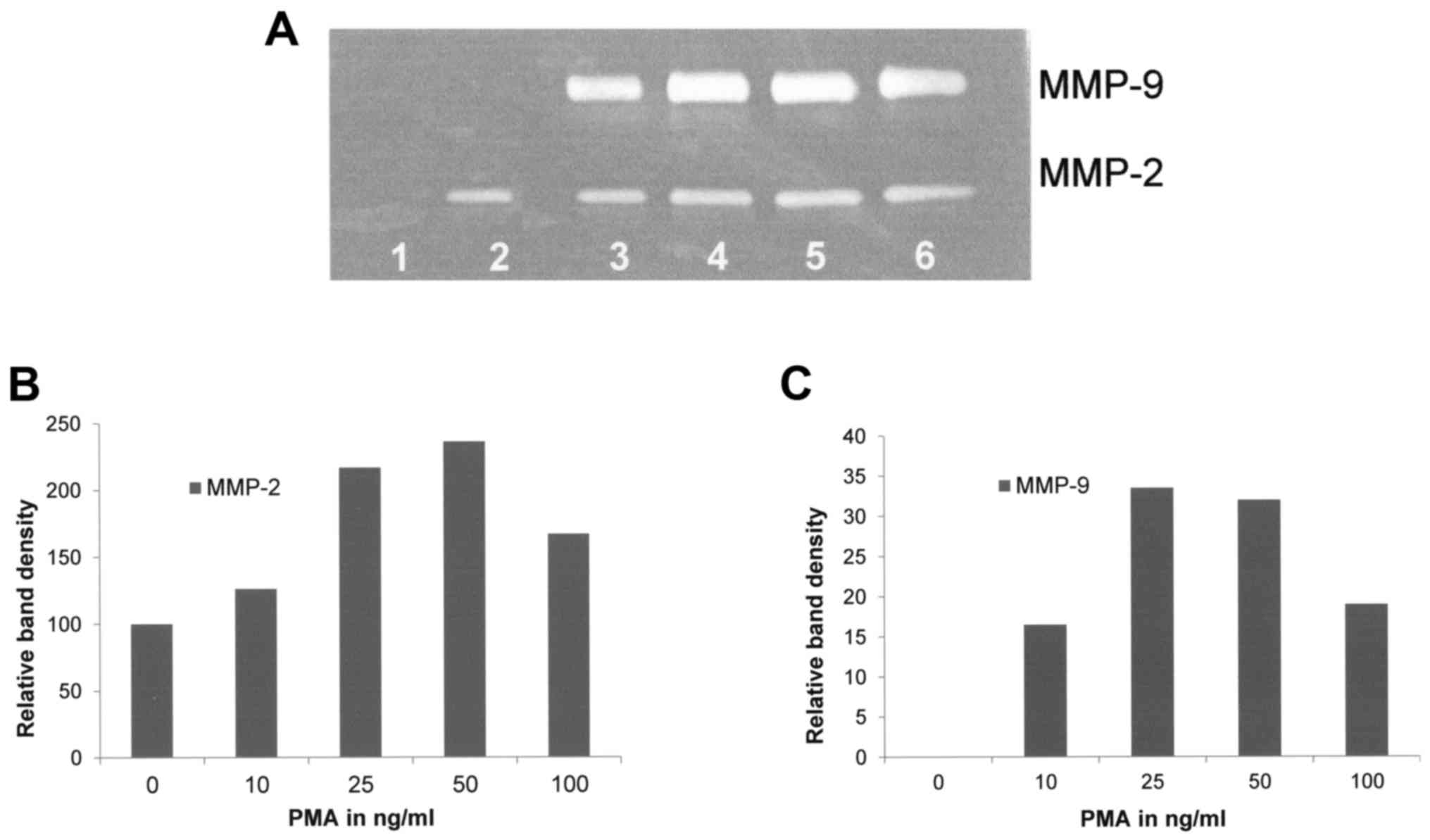
Effect of PMA on FA fibroblast
secretion of MMPs
Upon gelatinase zymography, FA fibroblasts
demonstrated moderate expression of MMP-2 and no expression of
MMP-9. PMA treatment had a moderate stimulatory effect on the
expression of MMP-2 (linear trend R2=0.4446) and
strongly stimulated MMP-9 expression in a dose-dependent manner
(linear trend R2=0.4084) as shown in Fig. 1.
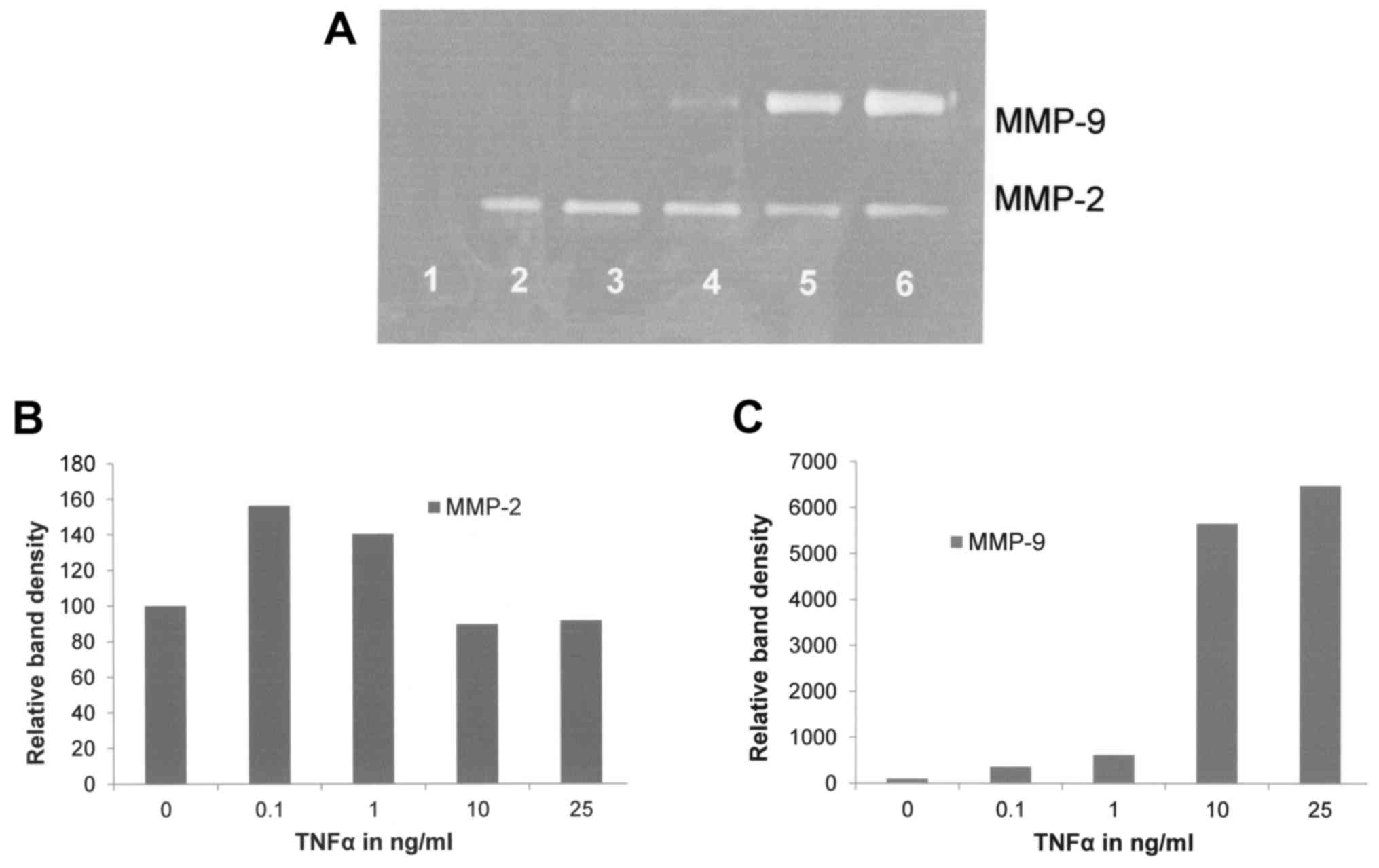
Effect of TNFα on FA fibroblast
secretion of MMPs
TNF-α had a negligible effect on MMP-2
(R2=0.1844) and a significant stimulatory dose-dependent
effect on MMP-9 (R2=0.824) as shown in Fig. 2.
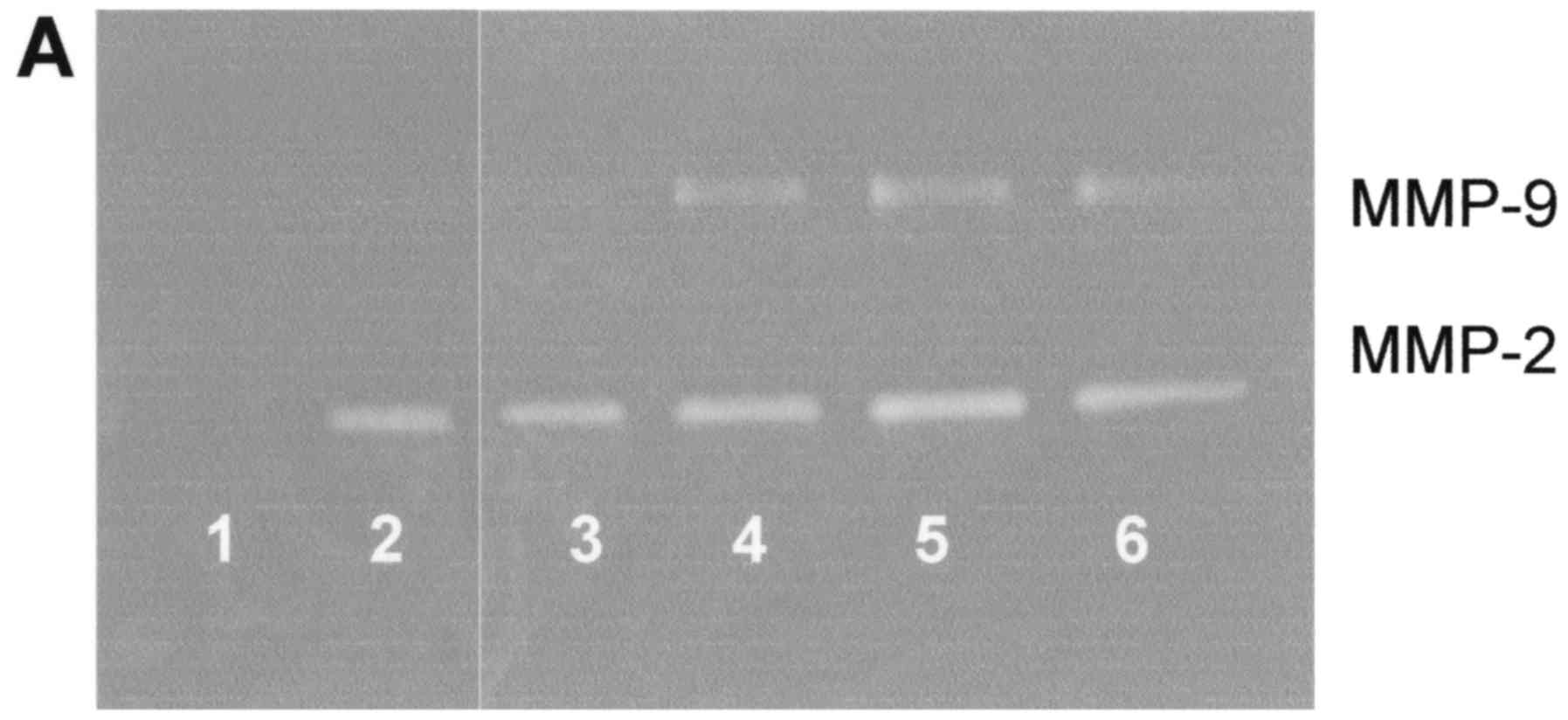
Effect of IL-1β on FA fibroblast
secretion of MMPs
IL-1β caused slight stimulation of MMP-2 at 1 and 10
ng/ml (R2=0.273), and had significant stimulatory
dose-dependent effects on MMP-9 (R2=0.558) as shown in
Fig. 3.
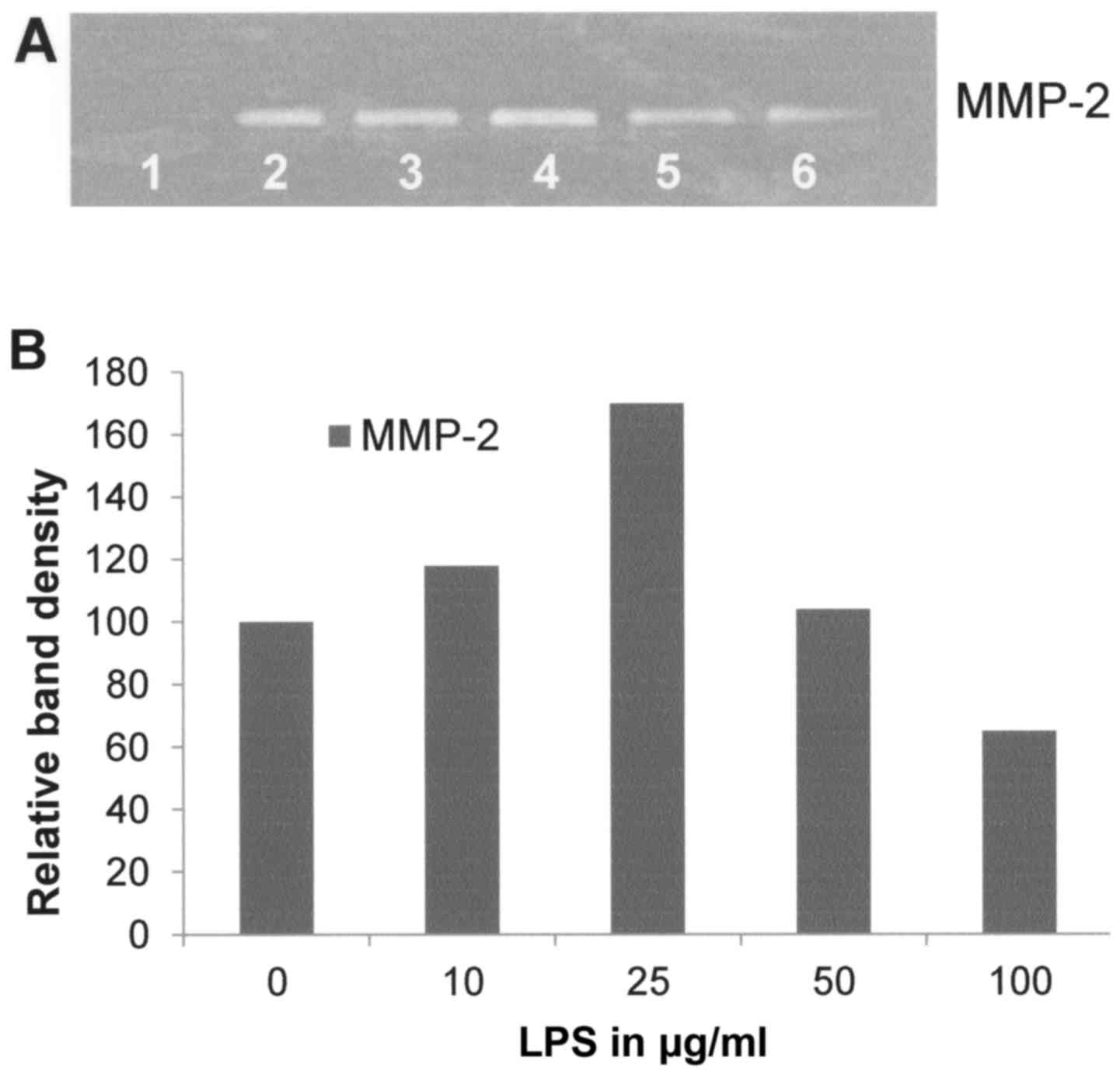
Effect of LPS on FA fibroblast
secretion of MMPs
LPS had a moderate stimulatory effect on MMP-2
secretion below 50 µg/ml and an inhibitory effect at 50 and 100
µg/ml (Fig. 4) and no effect on
MMP-9 (data not shown).
Chemical inhibitors
Table II shows the
quantitative densitometry results from the effects of the chemical
inhibitors doxycycline, dexamethasone and actinomycin-D on MMP-2
and MMP-9 expression in FA fibroblast cell lines.
 | Table II.Effect of inhibitors on Fanconi
anemia fibroblast MMP-2 and MMP-9 secretion. |
Table II.
Effect of inhibitors on Fanconi
anemia fibroblast MMP-2 and MMP-9 secretion.
|
| Untreated | PMA-treated (100
ng/ml) |
|---|
|
|
|
|
|---|
| Inhibitors | MMP-2 (%) | MMP-2 (%) | MMP-9 (%) |
|---|
| Doxycycline
(µM) |
|
|
|
|
Control | 100 | 100 | 100 |
| 10 | 265 | 59 | 4 |
| 25 | 255 | 55 | 7 |
| 50 | 49 | 8 | 0.5 |
| 100 | 1 | 0.5 | 0.5 |
| EGCG (µM) |
|
|
|
|
| Control | 100 | 100 | 100 |
| 10 | 201 | 118 | 16 |
| 25 | 148 | 107 | 13 |
| 50 | 75 | 39 | 1 |
| 100 | 17 | 0.5 | 0.5 |
| NM (µg/ml) |
|
|
|
|
| Controls | 100 | 100 | 100 |
| 10 | 112 | 98 | 53 |
| 50 | 80 | 102 | 30 |
| 100 | 19 | 49 | 10 |
| 500 | 1 | 1 | 1 |
| 1,000 | 1 | 1 | 1 |
| Dexamethasone
(µM) |
|
|
|
|
| Control | 100 |
|
|
| 50 | 12 |
|
|
| Retinoic acid
(µM) |
|
|
|
| Control | 100 |
|
|
| 50 | 7 |
|
|
| Actinomycin-D
(µM) |
|
|
|
| Control | 100 |
|
|
| 2 | 56 |
|
|
| 4 | 67 |
|
|
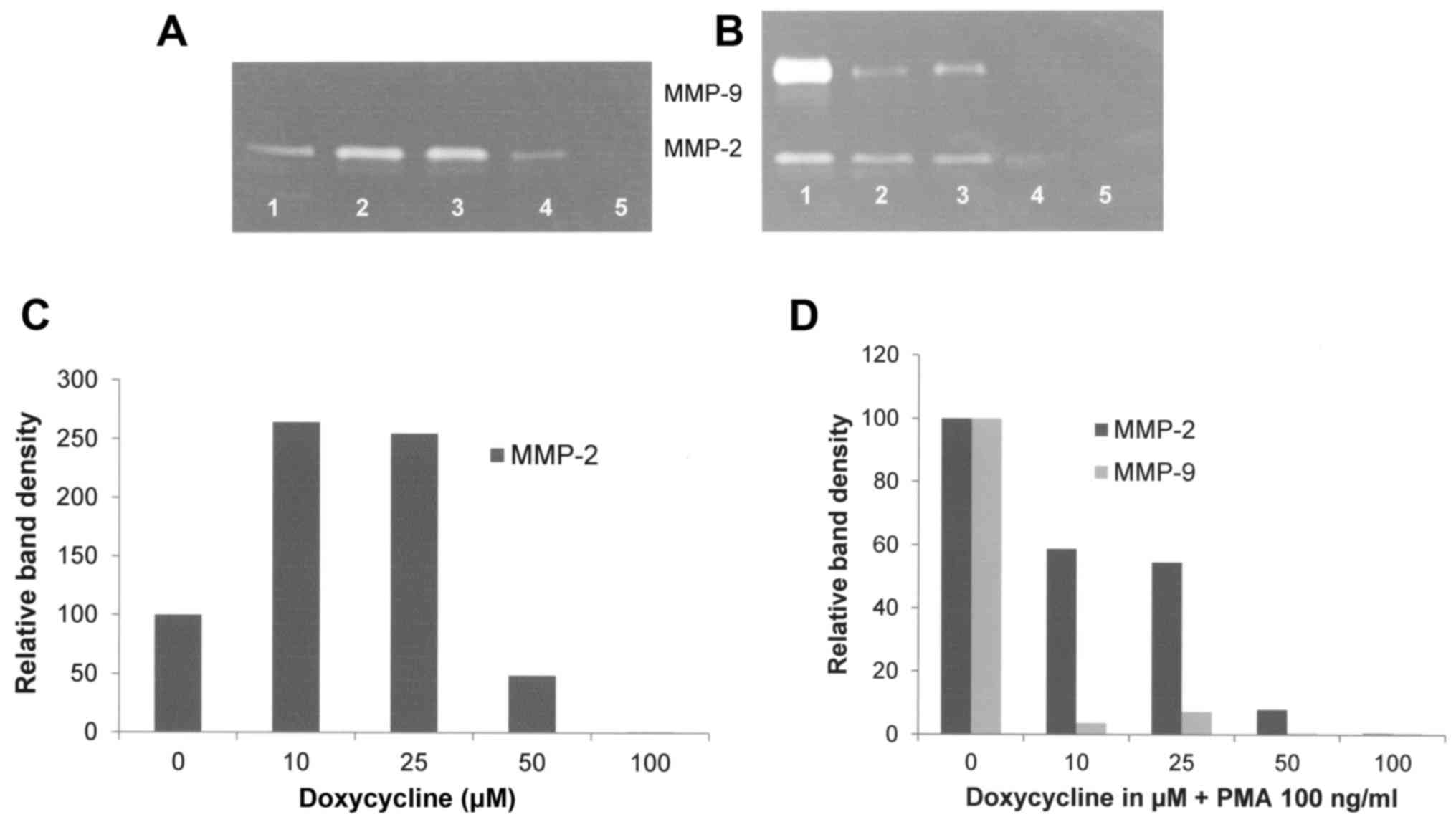
Doxycycline showed increased FA fibroblast MMP-2
secretion at 10 and 25 µM, and decreased secretion at 50 and 100 µM
with virtual total blockage at 100 µM (R2=0.296). When
treated with PMA 100 ng/ml, doxycycline downregulated the
expression of FA fibroblast MMP-2 and MMP-9 in a dose-dependent
manner, with virtual total blockage of MMP-2 at 100 µM
(R2=0.9378) and of MMP-9 at 50 µM (R2=0.5403)
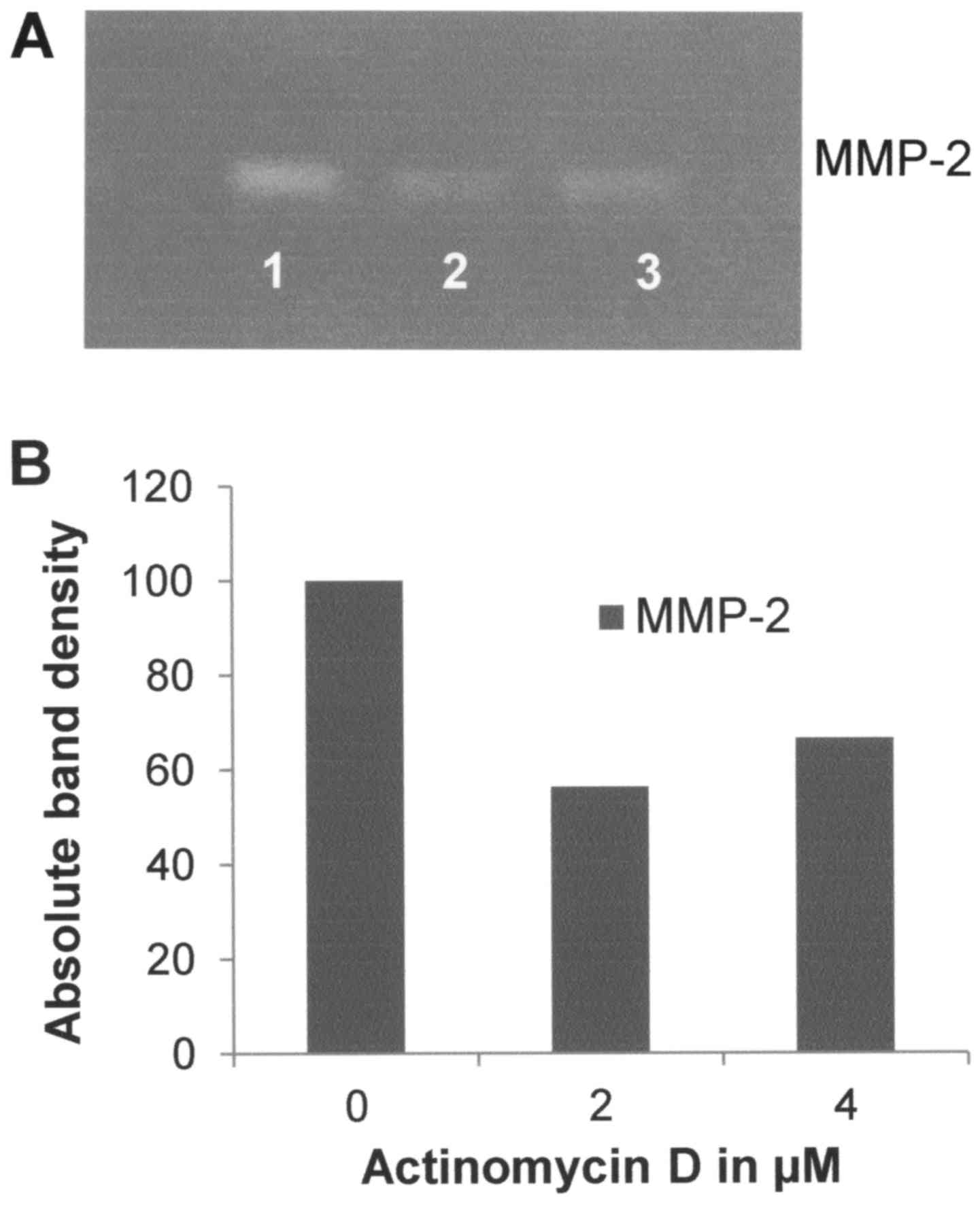
as shown in Fig. 5. Actinomycin-D
had a slight inhibitory effect on MMP-2 (R2=0.5355) with
33% inhibition at 4 µM as shown in Fig.
6. Dexamethasone had a potent inhibitory effect on MMP-2, with
inhibition of 88% at 50 µM compared to the control (data not
shown). Cyclohexamide had no effect on MMP-2 secretion by FA
fibroblasts (data not shown).
Natural inhibitors
Table II shows the
quantitative densitometry results from the effects of natural
inhibitors EGCG, NM and retinoic acid on MMP-2 and MMP-9 expression
in FA fibroblast cell lines.
EGCG downregulated the expression of MMP-2 at and
over 50 µM, with 83% block at 100 µM (R2=0.4337) as
shown in Fig. 7A and C. EGCG showed
inhibition of PMA-induced (100 ng/ml) MMP-9 (R2=0.6554)
and of MMP-2 (R2=0.7476) in a dose-dependent manner with
virtual total block of both at 100 µM as shown in Fig. 7B and D.
NM inhibited the secretion of MMP-2 by uninduced FA
fibroblast cells in a dose-dependent manner, with a linear trend of
R2=0.8706 (Fig. 8A and
C). NM showed dose-dependent inhibition of MMP-2 and MMP-9
expression in PMA-treated cells with virtual total blockage of both
at 500 µg/ml as shown in Fig. 8B and
C, with linear trends R2=0.8479 and 0.8597,
respectively.
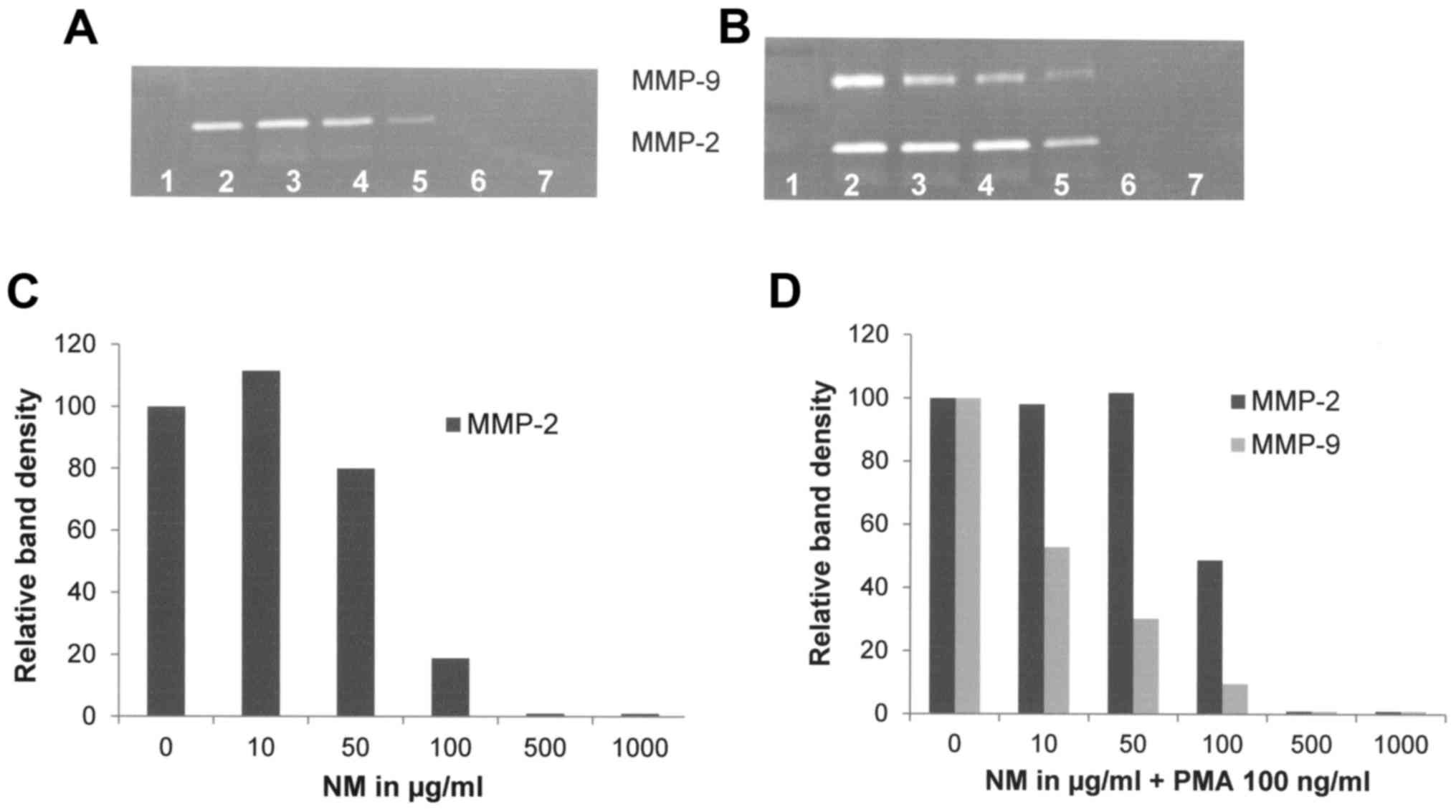 | Figure 8.Effect of NM on MMP-2 and MMP-9
secretion of normal and PMA-treated (100 ng/ml) FA-A:PD20 cells.
(A) Gelatinase zymograms of normal FA fibroblast cells and (B)
PMA-treated FA fibroblast cells. (C) Densitometry analysis of
normal FA fibroblast cells and (D) PMA-treated FA fibroblast cells.
Lane 1, markers; lane 2, control; lanes 3–7: 10, 50, 100, 500 and
1,000 µg/ml of NM, respectively. NM, nutrient mixture; MMP, matrix
metalloproteinase; PMA, phorbol 12-myristate 13-acetate; FA,
Fanconi anemia. |
Retinoic acid inhibited FA fibroblast MMP-2
secretion by 93% at 50 µM (data not shown).
Discussion
Elevated MMP levels correlate with tumor progression
and metastasis, as documented in experimental and clinical studies
(5,6). Epanchintsev et al reported the
overproduction of secretory factors in Fanconi anemia (FA), such as
IL-6, IL-8, MMP-2 and MMP-9 and that overexpression of these
secretory factors promoted the proliferation, migration and
invasion of surrounding tumor cells (3). Thus, knowledge of MMP regulation is of
importance for developing therapeutic strategies for FA.
Extracellular factors, including cytokines, growth factors,
inducers and inhibitors, have been implicated in the regulation of
MMP expression in different types of tumor cells (11,12).
In the present study, we compared MMP secretion
patterns by cytokines, PMA and LPS in FA immortalized cell lines.
In addition, we investigated the effect of inhibitors doxycycline,
EGCG, nutrient mixture (NM) and others, such as dexamethasone,
retinoic acid and agents that affect transcription and translation
levels, such as actinomycin-D.
Among the inducers and cytokines, PMA treatment had
a moderate stimulatory effect on MMP-2 and strong stimulation of
MMP-9 secretion, TNFα had a negligible effect on MMP-2 and a
significant stimulatory dose-dependent effect on MMP-9, IL-1β had
slight stimulation on MMP-2 at 1 and 10 ng/ml and significant
stimulatory dose-dependent effects on MMP-9, and LPS showed a
moderate stimulatory effect on MMP-2 secretion below 50 µM and an
inhibitory effect at 50 and 100 µM and no effect on MMP-9.
Among the chemical inhibitors, doxycycline
downregulated the secretion of FA fibroblast MMP-2 and MMP-9 in a
dose-dependent manner, with virtual total blockage of MMP-2 at 100
µM and of MMP-9 at 50 µM. In contrast, actinomycin-D had a slight
inhibitory effect on MMP-2 and a strong stimulatory effect on MMP-9
secretion. Dexamethasone had a potent inhibitory effect on
MMP-2.
Among the natural inhibitors, EGCG downregulated the
expression of MMP-2 and PMA induced MMP-9 expression in a
dose-dependent manner with virtual total blockage of both at 100
µM. Similarly, NM showed dose-dependent inhibition of MMP-2 and
MMP-9 expression in PMA-treated cells with virtual total blockage
of both at 500 µg/ml. Retinoic acid strongly inhibited FA
fibroblast MMP-2 secretion.
NM, which contains micronutrients such as lysine,
proline, ascorbic acid, and green tea extract, has demonstrated
antitumor and anti-invasive potential in vivo and in
vitro (13). The usage of
combinations of micronutrients expands metabolic targets mediated
by different pathways, and thus maximizes the biological impact
with lower doses of components. For example, a previous comparative
study on the effects of NM and its components such as green tea
extract and EGCG on the inhibition of MMP-2 and MMP-9 secretion of
different cancer cell lines with varying MMP secretion patterns,
revealed the superior potency of NM over green tea extract and EGCG
at equivalent doses (14).
We designed NM by selecting nutrients that act on
critical physiological targets in cancer progression and
metastasis, as documented in both clinical and experimental
studies. Adequate levels of ascorbic acid, lysine and proline are
essential for supporting proper synthesis and hydroxylation of
collagen fibers to optimize ECM structure. In addition, lysine
contributes to ECM stability as a natural inhibitor of
plasmin-induced proteolysis (15,16).
Manganese and copper also contribute to collagen formation. Green
tea extract has been shown to be potent in modulating cancer cell
growth, metastasis, angiogenesis and other aspects of cancer
progression (17–21). N-acetyl cysteine and selenium
have been documented to suppress tumor cell MMP-9 and invasive
activities, in addition to migration of endothelial cells through
the ECM (22–24). Ascorbic acid has been documented to
modulate cancer cell and tumor growth as well as to prevent
metastasis (25–30) and low levels of ascorbic acid are
found in cancer patients (31,32).
Low levels of arginine limit NO production, an inducer of apoptosis
(33).
In conclusion, our results demonstrated that
cytokines, mitogens and inhibitors modulated FA fibroblast MMP-2
and MMP-9 secretion, suggesting the clinical use of MMP inhibitors,
particularly potent and non-toxic ones such as NM and its component
EGCG in the management of FA cancers.
Acknowledgements
The present study was funded by Dr. Rath Health
Foundation (Santa Clara, CA, USA), a non-profit organization.
References
|
1
|
Donahue SL and Campbell C: A DNA double
strand break repair defect in Fanconi anemia fibroblasts. J Biol
Chem. 277:46243–46247. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Donahue SL, Lundberg R, Saplis R and
Campbell C: Deficient regulation of DNA double-strand break repair
in Fanconi anemia fibroblasts. J Biol Chem. 278:29487–29495. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Epanchintsev A, Shyamsunder P, Verma RS
and Lyakhovich A: IL-6, IL-8, MMP-2, MMP-9 are overexpressed in
Fanconi anemia cells through a NF-κB/TNF-α dependent mechanism. Mol
Carcinog. 54:1686–1699. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ibáñez A, Río P, Casado JA, Bueren JA,
Fernández-Luna JL and Pipaón C: Elevated levels of IL-1β in Fanconi
anaemia group A patients due to a constitutively active
phosphoinositide 3-kinase-Akt pathway are capable of promoting
tumour cell proliferation. Biochem J. 422:161–170. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liotta LA, Tryggvason K, Garbisa S, Hart
I, Foltz CM and Shafie S: Metastatic potential correlates with
enzymatic degradation of basement membrane collagen. Nature.
284:67–68. 1980. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stetler-Stevenson WG: The role of matrix
metalloproteinases in tumor invasion, metastasis, and angiogenesis.
Surg Oncol Clin N Am. 10:383–392. 2001.PubMed/NCBI
|
|
7
|
Stetler-Stevenson WG: Type IV collagenases
in tumor invasion and metastasis. Cancer Metastasis Rev. 9:289–303.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sato T, Sakai T, Noguchi Y, Takita M,
Hirakawa S and Ito A: Tumor-stromal cell contact promotes invasion
of human uterine cervical carcinoma cells by augmenting the
expression and activation of stromal matrix metalloproteinases.
Gynecol Oncol. 92:47–56. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pyke C, Kristensen P, Ralfkiaer E,
Grøndahl-Hansen J, Eriksen J, Blasi F and Danø K: Urokinase-type
plasminogen activator is expressed in stromal cells and its
receptor in cancer cells at invasive foci in human colon
adenocarcinomas. Am J Pathol. 138:1059–1067. 1991.PubMed/NCBI
|
|
10
|
Vincenti MP, White LA, Schroen DJ, Benbow
U and Brinckerhoff CE: Regulating expression of the gene for matrix
metalloproteinase-1 (collagenase): Mechanisms that control enzyme
activity, transcription and mRNA stability. Crit Rev Eukaryot Gene
Expr. 6:391–411. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ray JM and Stetler-Stevenson WG: The role
of matrix metalloproteases and their inhibitors in tumour invasion,
metastasis and angiogenesis. Eur Respir J. 7:2062–2072.
1994.PubMed/NCBI
|
|
12
|
Apodaca G, Rutka JT, Bouhana K, Berens ME,
Giblin JR, Rosenblum ML, McKerrow JH and Banda MJ: Expression of
metalloproteinases and metalloproteinase inhibitors by fetal
astrocytes and glioma cells. Cancer Res. 50:2322–2329.
1990.PubMed/NCBI
|
|
13
|
Niedzwiecki A, Roomi MW, Kalinovsky T and
Rath M: Micronutrient synergy - a new tool in effective control of
metastasis and other key mechanisms of cancer. Cancer Metastasis
Rev. 29:529–542. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Roomi MW, Monterrey JC, Kalinovsky T, Rath
M and Niedzwiecki A: Comparative effects of EGCG, green tea and a
nutrient mixture on the patterns of MMP-2 and MMP-9 expression in
cancer cell lines. Oncol Rep. 24:747–757. 2010.PubMed/NCBI
|
|
15
|
Rath M and Pauling L: Plasmin-induced
proteolysis and the role of apoprotein(a), lysine and synthetic
analogs. Orthomolecular Med. 7:17–23. 1992.
|
|
16
|
Sun Z, Chen YH, Wang P, Zhang J, Gurewich
V, Zhang P and Liu JN: The blockage of the high-affinity lysine
binding sites of plasminogen by EACA significantly inhibits
prourokinase-induced plasminogen activation. Biochim Biophys Acta.
1596:182–192. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Valcic S, Timmermann BN, Alberts DS,
Wächter GA, Krutzsch M, Wymer J and Guillén JM: Inhibitory effect
of six green tea catechins and caffeine on the growth of four
selected human tumor cell lines. Anticancer Drugs. 7:461–468. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mukhtar H and Ahmad N: Tea polyphenols:
Prevention of cancer and optimizing health. Am J Clin Nutr. 71
Suppl 6:1698S–1704S. 2000.PubMed/NCBI
|
|
19
|
Yang GY, Liao J, Kim K, Yurkow EJ and Yang
CS: Inhibition of growth and induction of apoptosis in human cancer
cell lines by tea polyphenols. Carcinogenesis. 19:611–616. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Taniguchi S, Fujiki H, Kobayashi H, Go H,
Miyado K, Sadano H and Shimokawa R: Effect of (−)-epigallocatechin
gallate, the main constituent of green tea, on lung metastasis with
mouse B16 melanoma cell lines. Cancer Lett. 65:51–54. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hara Y: Green tea: Health Benefits and
Applications. Marcel Dekker; New York, Basel: 2001, http://dx.doi.org/10.1201/9780203907993
View Article : Google Scholar
|
|
22
|
Kawakami S, Kageyama Y, Fujii Y, Kihara K
and Oshima H: Inhibitory effect of N-acetylcysteine on invasion and
MMP-9 production of T24 human bladder cancer cells. Anticancer Res.
21:213–219. 2001.PubMed/NCBI
|
|
23
|
Morini M, Cai T, Aluigi Mg, Noonan DM,
Masiello L, De Flora S, D'Agostini F, Albini A and Fassina G: The
role of the thiol N-acetylcysteine in the prevention of tumor
invasion and angiogenesis. Int J Biol markers. 14:268–271.
1999.PubMed/NCBI
|
|
24
|
Yoon SO, Kim MM and Chung AS: Inhibitory
effect of selenite on invasion of HT1080 tumor cells. J Biol Chem.
276:20085–20092. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cha J, Roomi MW, Ivanov V, Kalinovsky T,
Niedzwiecki A and Rath M: Ascorbate supplementation inhibits growth
and metastasis of B16FO melanoma and 4T1 breast cancer cells in
vitamin C-deficient mice. Int J Oncol. 42:55–64. 2013.PubMed/NCBI
|
|
26
|
Naidu KA, Karl RC, Naidu KA and Coppola D:
Antiproliferative and proapoptotic effect of ascorbyl stearate in
human pancreatic cancer cells: Association with decreased
expression of insulin-like growth factor 1 receptor. Dig Dis Sci.
48:230–237. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Anthony HM and Schorah CJ: Severe
hypovitaminosis C in lung-cancer patients: The utilization of
vitamin C in surgical repair and lymphocyte-related host
resistance. Br J Cancer. 46:354–367. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Maramag C, Menon M, Balaji KC, Reddy PG
and Laxmanan S: Effect of vitamin C on prostate cancer cells in
vitro: Effect on cell number, viability, and DNA synthesis.
Prostate. 32:188–195. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Koh WS, Lee SJ, Lee H, Park C, Park MH,
Kim WS, Yoon SS, Park K, Hong SI, Chung MH, et al: Differential
effects and transport kinetics of ascorbate derivatives in leukemic
cell lines. Anticancer Res. 18:2487–2493. 1998.PubMed/NCBI
|
|
30
|
Chen Q, Espey Mg, Krishna MC, Mitchell JB,
Corpe CP, Buettner GR, Shacter E and Levine M: Pharmacologic
ascorbic acid concentrations selectively kill cancer cells: Action
as a pro-drug to deliver hydrogen peroxide to tissues. Proc Natl
Acad Sci USA. 102:13604–13609. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Núñez M, Artín C and de Apodaca y Ruiz A
Ortiz: Ascorbic acid in the plasma and blood cells of women with
breast cancer. The effect of the consumption of food with an
elevated content of this vitamin. Nutr Hosp. 10:368–372. 1995.(In
Spanish). PubMed/NCBI
|
|
32
|
Kurbacher CM, Wagner U, Kolster B,
Andreotti PE, Krebs D and Bruckner HW: Ascorbic acid (vitamin C)
improves the antineoplastic activity of doxorubicin, cisplatin, and
paclitaxel in human breast carcinoma cells in vitro. Cancer Lett.
103:183–189. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cooke JP and Dzau VJ: Nitric oxide
synthase: Role in the genesis of vascular disease. Annu Rev Med.
48:489–509. 1997. View Article : Google Scholar : PubMed/NCBI
|