Introduction
Osteoarthritis (OA) is one of the commonly observed
diseases of joints caused by the disintegration of cartilage matrix
and death of chondrocytes (1).
There is always an equilibrium maintained by extracellular
cartilage matrix between the formation and loss of chondrocytes
(2,3). Loss of chondrocytes is a limiting
factor for the formation of extracellular cartilage matrix which
subsequently leads to the development of osteoarthritis (4). Many factors such as proteoglycan and
collagen (type II) are involved in the regulation of normal
functioning of chondrocytes (5).
Studies have revealed that chondrocyte loss and extracellular
matrix degradation involve several factors including, generation of
peroxide radicals, TNF-α and interleukins (6,7). All
these factors initiate inflammatory reactions leading to
chondrocyte apoptosis (8).
Expression of interleukin-1β is responsible for the production of
inhibitors of metalloproteinase (TIMPs) leading to the inhibition
of matrix metalloproteinase (MMP) generation and subsequent
chondrocyte death and OA (9–11).
Traditional Chinese medicine (TCM) involves the use
of various herbs as well as dietary ingredients for several
disorders depending upon the type of the syndrome (12). Numerous studies have been performed
to demonstrate the chemotherapeutic potential of TCM against
various types of cancers (13).
Ginseng has a long traditional medicinal importance for the
treatment of cancers, stress and diabetes (14). Ginsenoside-Rg5 obtained by the
processing of ginseng has shown promising anticancer,
anti-inflammatory and anti-aging properties (15–17).
There are several reports which demonstrate the activity of
ginsenoside-Rg5 against cancer (18,19),
dermatis (20), inflammation
(21) and neurotrophic disorders
(22). This study was performed to
investigate the effects of ginsenoside-Rg5 on destruction of
cartilage through onset of cartilage matrix damage and death of
chondrocytes in OA rat model. This study revealed that
ginsenoside-Rg5 significantly prevents destruction of articular
cartilage through inhibition of chondrocyte apoptosis and matrix
damage in OA rats.
Materials and methods
Animals
Male adult Wistar rats aged 8–10 weeks (270–300 g)
were purchased from the Laboratory Animal Center of Sun Yat-Sen
University and were housed in the animal care facility center of
our institute under pathogen-free conditions. The experimental
procedures on animals were performed according to the guidelines of
the National Institutes of Health criteria for the care and use of
laboratory animals. This study was approved by the Laboratory
Animal Care Committee of Sun Yat-sen University (Guangzhou,
China).
Preparation of osteoarthritis rat
model
The animals were subjected to anesthetization using
halothane, knee joint was exposed to dislocate the patella and
subsequently micro-scissors were used for transection of ligament
and resection of the medial meniscus. The rats were randomly
assigned to seven groups of 5 each. The five treatment groups were
given 1, 2, 5, 10 or 15 mg/g doses of ginsenoside-Rg5
intragastrically daily for one month. The rats in the normal and
untreated groups received equal volume of normal saline at the same
time.
Histological analysis
On day 31 animals were sacrificed to extract the
tibia and femur bones from each of the animals. The dissected bones
were subjected to fixing in paraformaldehyde followed by
decalcification and paraffin embedment. The bones were then cut
into thin sections, deparaffined and subjected to hematoxylin and
eosin staining for microscopic examination. The cartilage damage
was monitored on the Mankin scale ranging from 0 to 12. The
quantification of synovial lining damage was performed using
Image-Pro Plus 6.0 system (IPP) image analysis system (Media
Cybernetics, Rockville, MD, USA).
Terminal deoxynucleotidyl
transferase-mediated deoxyuridine triphosphate nick-end labeling
(TUNEL) staining
Antigen retrieval was conducted according to the
manufacturer's instructions (Roche) and after dewaxing. The
cartilage sections were washed twice with PBST and then
permeabilized using 0.1% Triton X-100. Incubation of the sections
was performed using fluorescein-labeled dUTP and terminal
deoxynucleotidyl transferase (TdT) mixture. Antifluorescein
antibody in combination with alkaline phosphatase was used for
probing purposes. The Olympus OX31 microscope (Olympus, Tokyo,
Japan) was used for the analysis of the apoptosis of the
chondrocytes. Quantification of the apoptotic chondrocytes was
performed using Image-Pro Plus 6.0 system (IPP).
Immunoblotting analysis
The paraffin-embedded sections were cut into thin
sections followed by de-paraffinization in boiling xylene. The
sections were treated by hydrogen peroxide and then incubated with
blocking serum (Vectastain® ABC kit, Vector
Laboratories, Burlingame, CA, USA) for 45 min. The sections were
incubated with mouse monoclonal antibodies against type II
collagen, MMP-13 and TIMP-1 (dilution 1/30; Beijing Biosynthesis
Biotechnology Co., Ltd., Beijing, China) overnight at 4°C. The
sections were washed with PBS an subsequently incubated with horse
secondary antibody (Zhonshan Golden Bridge Biotechnology, Beijing,
China) for 1 h. The 3,3′-diaminobenzidine (DAB; Sigma, St. Louis,
MO, USA) stained sections were analyzed using Image-Pro Plus 6.0
system (IPP).
Flow cytometry for analysis of
apoptosis induction
Chondrocytes treated with ginsenoside-Rg5 were
collected, rinsed in ice-cold PBS. Chondrocytes were then suspended
at a density of 2×106 per ml into the binding buffer and
subjected to incubation. Then 100 ml samples were put into the
fulcrum tubes and treated with Annexin V-FITC (5 µl) and propidium
iodide (10 µl) for 30 min under dark atmosphere. Following
incubation, chondrocytes were treated with binding buffer and then
analyzed using flow cytometry.
Western blot assay
After incubation of the chondrocytes with
ginsenoside-Rg5 for 48 h, 300 µl lysis buffer (Wuhan Boster
Biological Technology, Ltd., Wuhan, China) was added. Then cell
lysate was treated with 3 µl protease inhibitor and the mortar for
45 min on ice followed by centrifugation for 30 min at 12,000 × g.
The supernatant was collected for the determination of
concentration of proteins. To this mixture was added 4X sample
buffer solution, followed by boiling for 45 min. The centrifuged
mixture was subjected to isolation on SDS-PAGE and the protein
transfer to polyvinylidene difluoride membranes. The membranes were
blocked using 5% skimmed milk and subsequently incubated with mouse
anti-MMP-13 monoclonal antibody (1:100; sc-189; Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA) and rabbit anti-TIMP-1
polyclonal antibody (1:100; sc-189; Santa Cruz Biotechnology,
Inc.). After overnight incubation, the membranes were washed twice
with PBS and then incubated for 1 h with horseradish
peroxide-coupled sheep anti-mouse secondary antibody (Beijing
Kangwei Technology Group Co., Ltd., Beijing, China). The blots were
treated with chemiluminescent reagent (GS009; Beyotime Institute of
Biotechnology, Shanghai, China) and then images were captured.
Quantitative polymerase chain reaction
(qPCR)
The chondrocytes were treated with ginsenoside-Rg5
for 48 h. Following incubation, total RNA from the chondrocytes was
isolated using RNA extraction kit (Invitrogen Life Technologies,
Carlsbad, CA, USA). After extraction and subsequent purification,
the RNA concentration was measured using a spectrophotometer. The
2-µg RNA samples were then subjected to reverse transcription using
reverse transcription reagent (Takara Biotechnology Co., Ltd.,
Dalian, China). Quantification and analysis of the transcripts was
performed using QuantiTect SYBR Green PCR kit (Qiagen, Tokyo,
Japan) and Applied Biosystems® 7500 Fast Real-Time PCR
system (Applied Biosystems Inc.), respectively. PCR was performed
using the following reaction conditions: pre-denaturation was
performed at 95°C for 30 sec; at 95°C denaturation was carried out
for 3 sec; and at 60°C annealing was done for 30 sec. The data were
obtained directly from the real-time fluorescent quantitative PCR
instrument (Bio-Rad Laboratories, Inc., Hercules, CA, USA). PCR
primers used were: TGAGGA TACAGGCAAGACTCT (forward),
CAATACGGTTACTCC AGATGC (reverse) for MMP-13 and CTTCTGGCATCCTGT
TGTTG (forward), AGAAGGCCGTCTGTGGGT (reverse) for TIMP-1.
Statistical analysis
The data presented were analyzed using SPSS
software, version 12.0 (SPSS, Inc., Chicago, IL, USA). Analysis of
the data was performed using one-way analysis of variance and
subsequent Tukey post hoc comparisons. Statistically significant
differences were considered at a two-tailed P<0.05.
Results
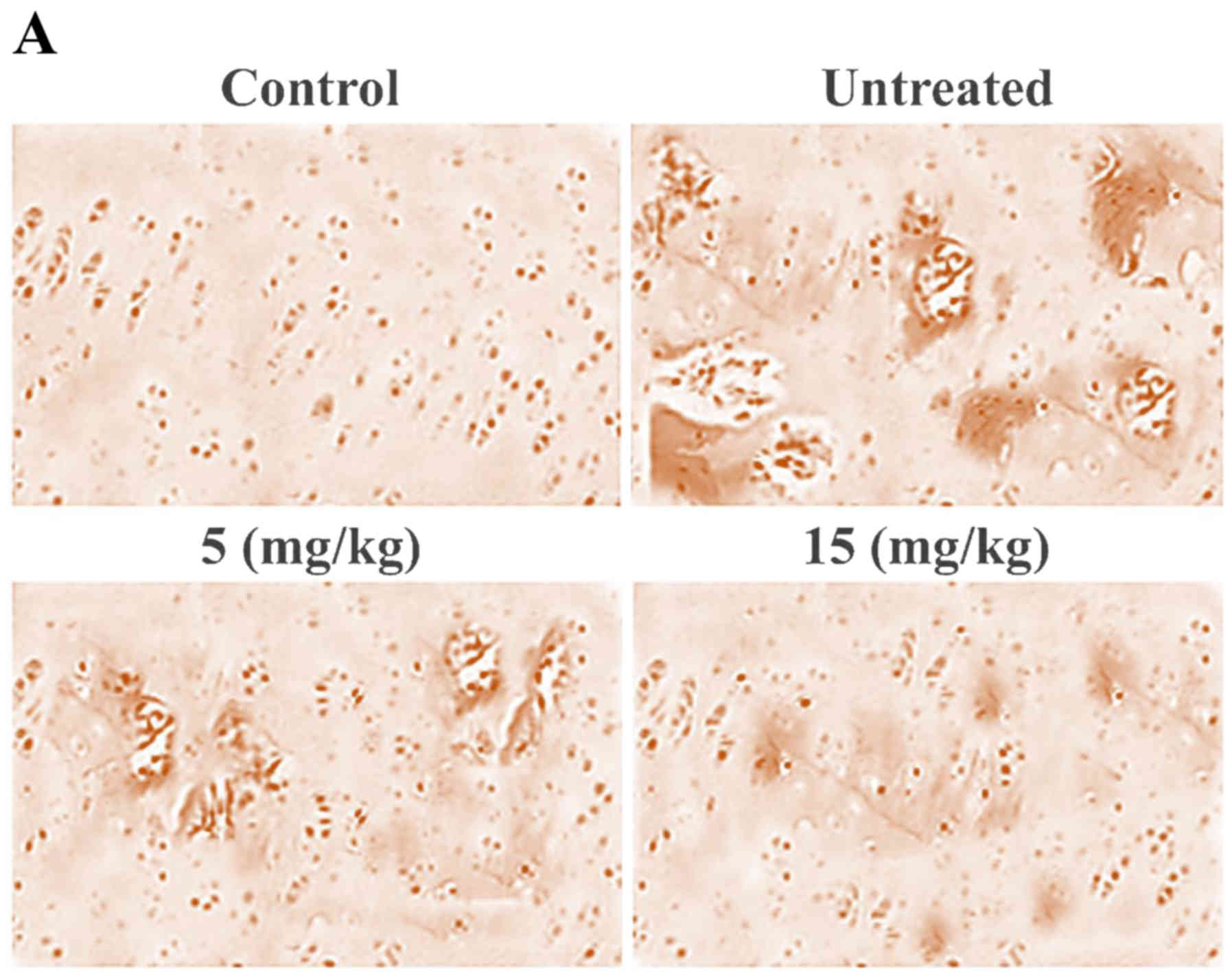
Ginsenoside-Rg5 treatment prevents
damage to articular knee cartilage in OA-rat model
Histopathological analysis of the OA-rat cartilage
revealed marked changes such as presence of wide spaces,
dissociated cells and rough surface. However, ginsenoside-Rg5
treatment prevented damage to cartilage in the OA-rats following
one month of treatment (Fig. 1A).
Among the various doses used to treat the OA-rats, the effect was
significant (P=0.005) at 15 mg/kg concentration of ginsenoside-Rg5.
In comparison to the untreated group of OA-rats, the Mankin score
in the ginsenoside-Rg5 treated group was significantly lower.
Synovial examination showed irregular surface and higher population
of the inflammatory cells in the OA-rats. However, ginsenoside-Rg5
treatment prevented the disintegration of synovial membrane to a
significant (P=0.005) extent compared to the untreated group
(Fig. 1B).
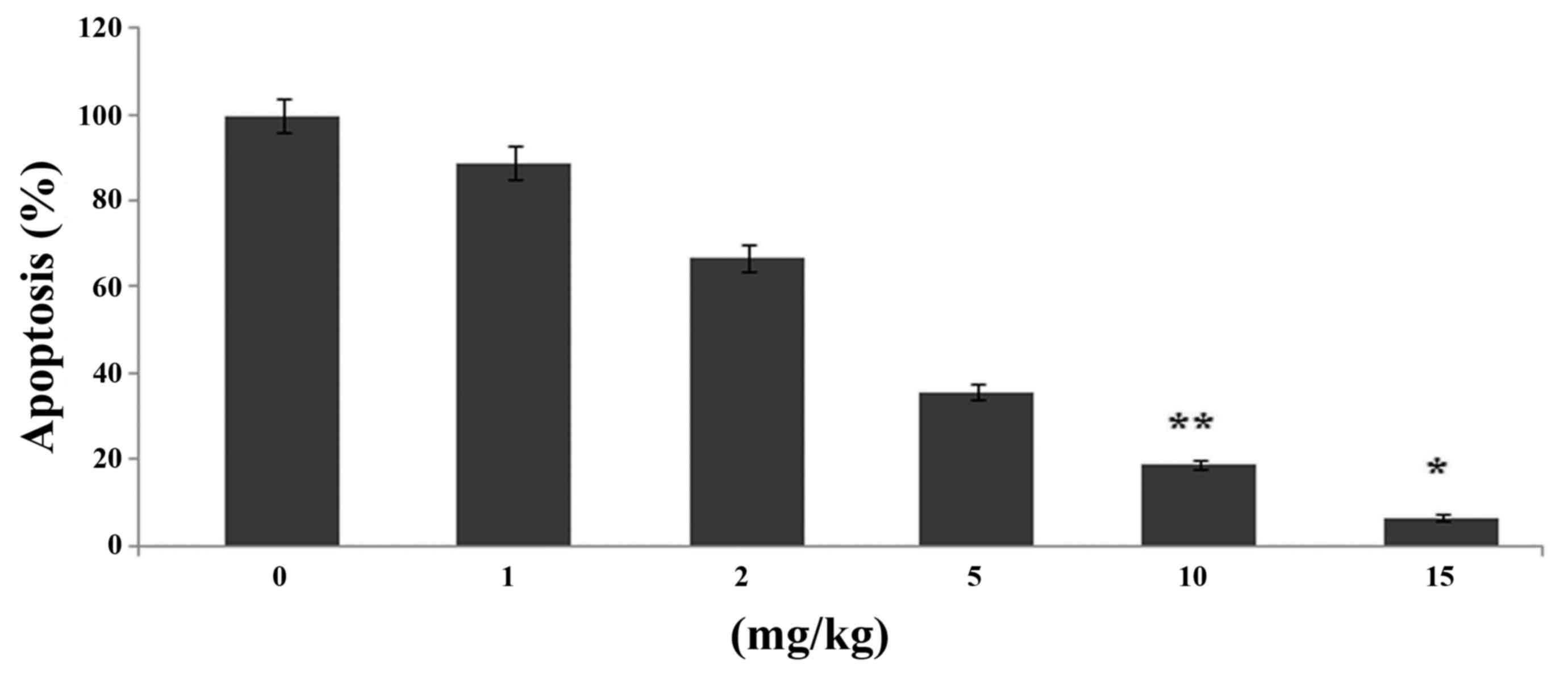
Effects of ginsenoside-Rg5 on knee
chondrocyte apoptosis in the OA-rat model
Ginsenoside-Rg5 treatment caused a significant
(P<0.05) reduction in the proportion of apoptotic cells in the
cartilage compared to the control group (Fig. 2). Ginsenoside-Rg5 treatment for one
month caused reduction in the proportion of apoptotic cells to 7%
in the knee joints compared to the control.
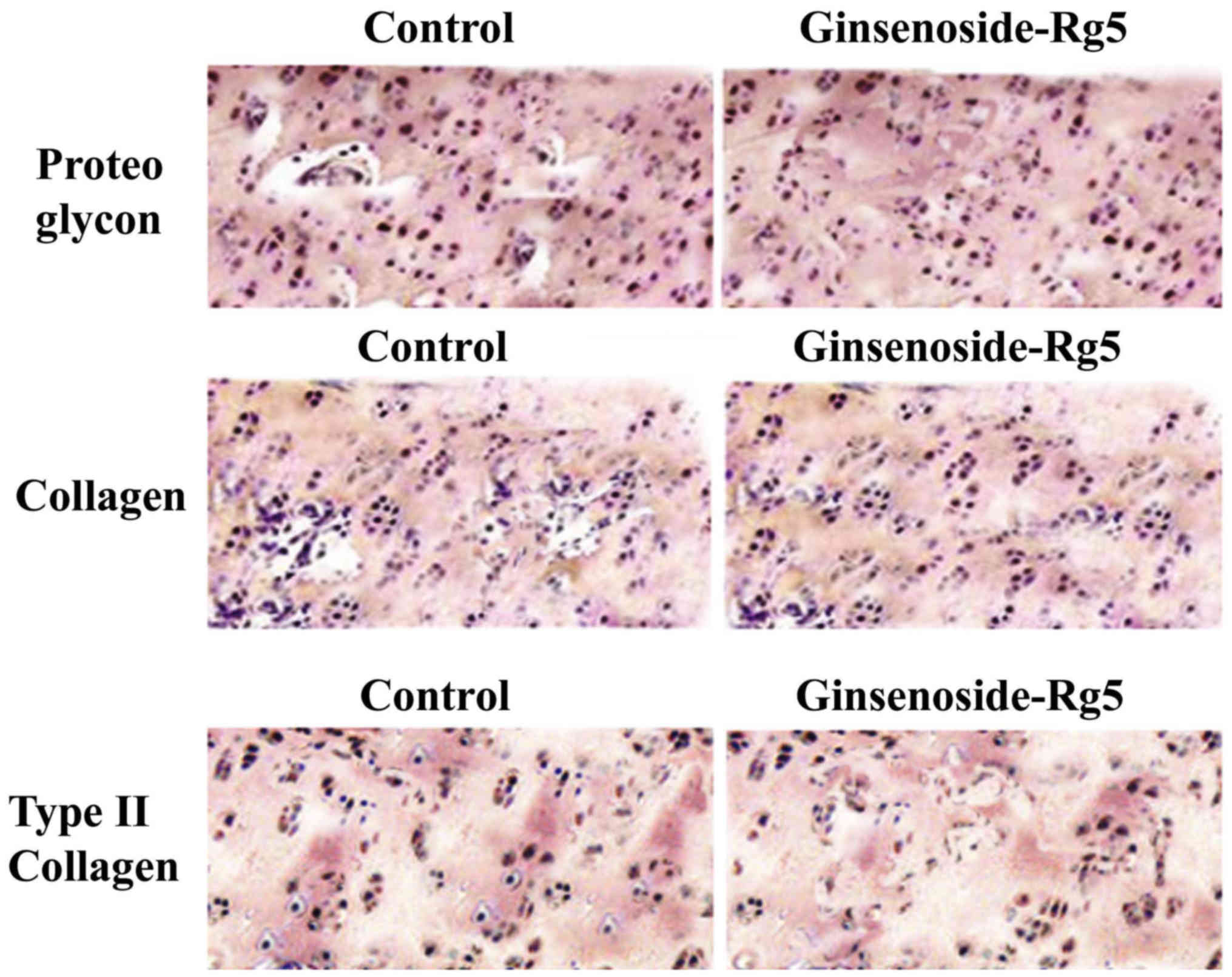
Effects of ginsenoside-Rg5 on knee
cartilage matrix in the OA-rat model
Treatment of the rats with ginsenoside-Rg5 for one
month caused a significant prevention of the degradation of
cartilage matrix compared to those in the control group. The levels
of proteins including, proteoglycan, collagen and type II collagen
were increased by 5-, 3- and 4-fold compared to the control group
(Fig. 3).
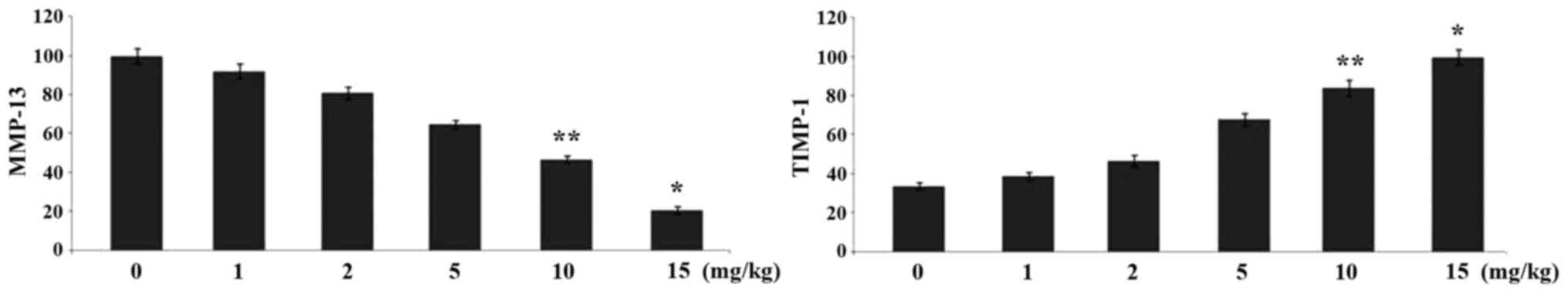
Ginsenoside-Rg5 treatment alters the
expression levels of MMP-13 and TIMP-1 in the OA-rat model
Immunohistochemistry revealed a significant
alteration in the expression levels of MMP-13 and TIMP-1 in the
rats on treatment with ginsenoside-Rg5 compared to the control
group after one month. The level of MMP-13 was reduced to 45%
compared to the control and that of TIMP-1 was increased by 67% in
the rats treated with ginsenoside-Rg5 for one month (Fig. 4).
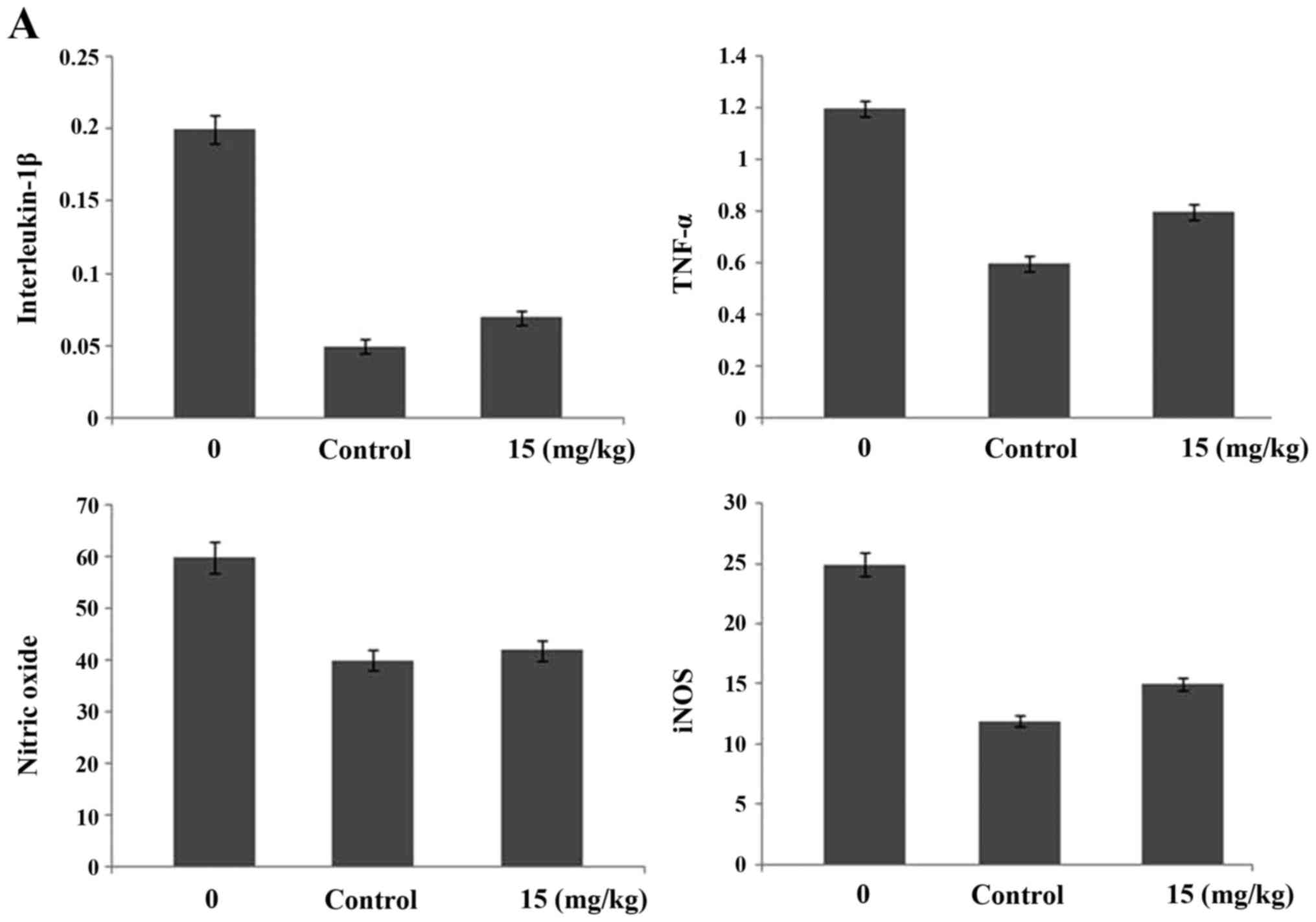
Ginsenoside-Rg5 treatment alters the
expression of interleukin-1β, tumor necrosis factor-α, nitric
oxide, inducible nitric oxide synthetase, BMP and TGF-β in the
serum of OA-rat model
Treatment of the OA-rats with ginsenoside-Rg5 for
one month caused a significant reduction in the expression levels
of interleukin-1β, tumor necrosis factor-α, nitric oxide and
inducible nitric oxide synthetase. The levels of interleukin-1β,
tumor necrosis factor-α, nitric oxide and inducible nitric oxide
synthetase were reduced by 67, 54, 32 ad 49%, respectively, after
one month of treatment with 5 mg/kg dose of ginsenoside-Rg5.
Ginsenoside-Rg5 treatment for one month led to a significant
enhancement in the expression levels of BMP-2 and TGF-β1 in the
OA-rats. The levels were increased to 67 and 52% for BMP-2 and
TGF-β1, respectively after one month (Fig. 5).
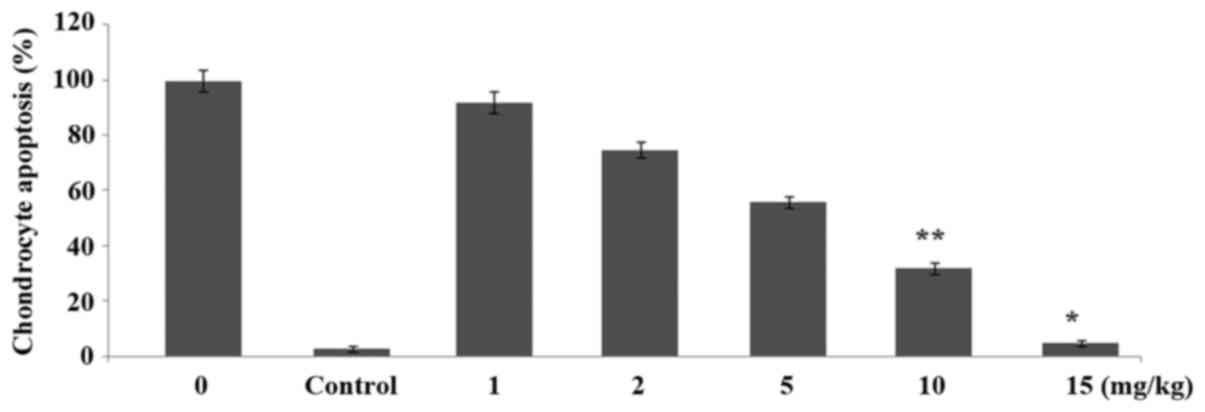
Ginsenoside-Rg5 treatment inhibits
chondrocyte apoptosis
Incubation of the chondrocytes with interleukin-1β
caused a marked increase in the proportion of apoptotic cells.
However, treatment of the chondrocytes with 1, 2, 5, 10 and 15 µM
doses of ginsenoside-Rg5 for 48 h prevented the interleukin-1β
induced apoptosis of chondrocytes in a dose-dependent manner
(Fig. 6). Apoptosis of chondrocytes
was reduced to 56, 42, 29, 11 and 2% on treatment with 1, 2, 5, 10
and 15 µM, respectively doses of ginsenoside-Rg5 after 48 h.
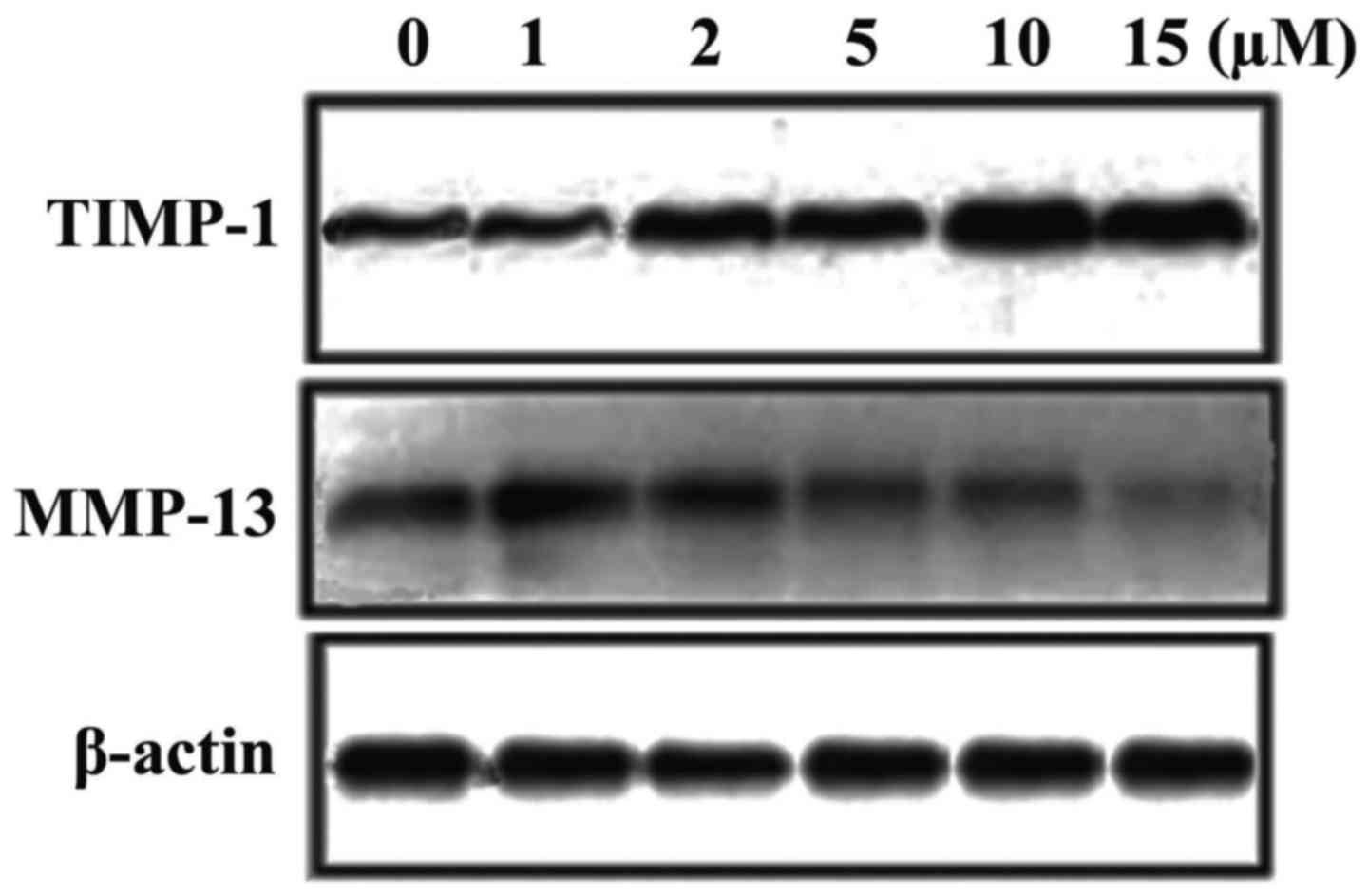
Ginsenoside-Rg5 treatment of
chondrocytes alters the expression of MMP-13 and TIMP-1
Treatment of chondrocytes with ginsenoside-Rg5
caused a concentration-dependent decrease in the interleukin-1β
induced expression of MMP-13. The expression of MMP-13 was reduced
to 82, 76, 58, 34 and 12% on treatment of chondrocytes with 1, 2,
5, 10 and 15 µM, respectively, doses of ginsenoside-Rg5 for 48 h.
Treatment of the chondrocytes with ginsenoside-Rg5 caused a
significant increase in the interleukin-1β inhibited expression of
TIMP-1 (Fig. 7). Chondrocytes were
treated with 1, 2, 5, 10 and 15 µM doses of ginsenoside-Rg5 and the
expression of TIMP-1 was found to be 2, 3, 3.7, 4.5 and 6-fold,
respectively compared to the interleukin-1β treated chondrocytes
(Fig. 7).
Discussion
This study was performed to investigate the effects
of ginsenoside-Rg5 on destruction of cartilage through onset of
cartilage matrix damage and death of chondrocytes in OA rat model.
The results from this study demonstrated that ginsenoside-Rg5
prevents degradation of cartilage and inhibits inflammation of
synovium in the OA rats. OA is characterized by the dissociation
and death of chondrocytes, damage to extracellular cartilage matrix
and production of free radical leading to inflammatory
reactions.
During OA a marked loss of chondrocytes is observed
in the articular cartilage which is evident by the formation of
wide spaces and rough cartilage surface (23). The current study revealed a
significant increase in the chondrocyte apoptosis and degradation
of extracellular matrix in the OA rats. However, ginsenoside-Rg5
treatment led to inhibition of chondrocyte apoptosis and prevention
of extracellular matrix damage in OA rats after one month.
Apoptosis was also induced in the chondrocytes by incubation with
interleukin-1β and the cells were then treated with
ginsenoside-Rg5. The results revealed that ginsenoside-Rg5
treatment significantly inhibited the interleukin-1β induced
apoptosis in the chondrocytes. Degradation of cartilage is caused
by the inhibition of proteoglycan which is the major component of
extracellular matrix and provides it strength (24,25).
Another factor of extracellular matrix providing strength to the
articular is the type II collagen (26). The degradation of type II collagen
is caused by MMP-13, however, its activity is suppressed by TIMP-1
(27,28). Our current study revealed that
ginsenoside-Rg5 treatment for one month significantly increased the
expression of TIMP-1. The increased expression of TIMP-1 was
evident by a marked reduction in the MMP-13 expression in OA-rats.
Treatment of the chondrocytes with interleukin-1β caused a
significant reduction in the expression of TIMP-1. However,
treatment of the chondrocytes with ginsenoside-Rg5 led to a marked
increase in the expression of TIMP-1 in following incubation with
interleukin-1β. Initial stage of OA is characterized by the
inflammation and higher level of proinflammatory cytokines such as
interleukin-1β and the tumor necrosis factor (29). In addition, the level of nitric
oxide radical and inducible nitric oxide synthetase is also higher
which induced chondrocyte death (30). Our results revealed that
ginsenoside-Rg5 treatment caused a significant reduction in the
expression of interleukin-1β, tumor necrosis factor, nitric oxide
radical and inducible nitric oxide synthetase in the OA-rats after
one month.
Thus, this study revealed that ginsenoside-Rg5
significantly prevents destruction of articular cartilage through
inhibition of chondrocyte apoptosis and matrix damage in OA rats.
Therefore, ginsenoside-Rg5 can be used for the treatment of
osteoarthritis.
References
|
1
|
Loeser RF: Aging and osteoarthritis: The
role of chondrocyte senescence and aging changes in the cartilage
matrix. Osteoarthritis Cartilage. 17:971–979. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Goldring MB: The role of the chondrocyte
in osteoarthritis. Arthritis Rheum. 43:1916–1926. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Roughley PJ: Articular cartilage and
changes in arthritis: Noncollagenous proteins and proteoglycans in
the extracellular matrix of cartilage. Arthritis Res. 3:342–347.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aigner T and Kim HA: Apoptosis and
cellular vitality: Issues in osteoarthritic cartilage degeneration.
Arthritis Rheum. 46:1986–1996. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Eyre D: Collagen of articular cartilage.
Arthritis Res. 4:30–35. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Aizawa T, Kon T, Einhorn TA and
Gerstenfeld LC: Induction of apoptosis in chondrocytes by tumor
necrosis factor-alpha. J Orthop Res. 19:785–796. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fernandes JC, Martel-Pelletier J and
Pelletier JP: The role of cytokines in osteoarthritis
pathophysiology. Biorheology. 39:237–246. 2002.PubMed/NCBI
|
|
8
|
Kapoor M, Martel-Pelletier J, Lajeunesse
D, Pelletier JP and Fahmi H: Role of proinflammatory cytokines in
the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 7:33–42.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Attur M, Al-Mussawir HE, Patel J, Kitay A,
Dave M, Palmer G, Pillinger MH and Abramson SB: Prostaglandin E2
exerts catabolic effects in osteoarthritis cartilage: Evidence for
signaling via the EP4 receptor. J Immunol. 181:5082–5088. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abramson SB: Osteoarthritis and nitric
oxide. Osteoarthritis Cartilage. 16 Suppl 2:S15–S20. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Burger D, Rezzonico R, Li JM, Modoux C,
Pierce RA, Welgus HG and Dayer JM: Imbalance between interstitial
collagenase and tissue inhibitor of metalloproteinases 1 in
synoviocytes and fibroblasts upon direct contact with stimulated T
lymphocytes: Involvement of membrane-associated cytokines.
Arthritis Rheum. 41:1748–1759. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang G, Li X, Li X, Wang L, Li J, Song X,
Chen J, Guo Y, Sun X, Wang S, et al: Traditional Chinese medicine
in cancer care: A review of case series published in the chinese
literature. Evid Based Complement Alternat Med. 2012:7510462012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu J, Li X, Liu J, Ma L, Li X and Fønnebø
V: Traditional Chinese medicine in cancer care: A review of case
reports published in Chinese literature. Forsch Komplement Med.
18:257–263. 2011. View Article : Google Scholar
|
|
14
|
Kwon SW, Han SB, Park IH, Kim JM, Park MK
and Park JH: Liquid chromatographic determination of less polar
ginsenosides in processed ginseng. J Chromatogr A. 921:335–339.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim SN, Ha YW, Shin H, Son SH, Wu SJ and
Kim YS: Simultaneous quantification of 14 ginsenosides in Panax
ginseng C.A. Meyer (Korean red ginseng) by HPLC-ELSD and its
application to quality control. J Pharm Biomed Anal. 45:164–170.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yun TK, Lee YS, Lee YH, Kim SI and Yun HY:
Anticarcinogenic effect of Panax ginseng C.A. Meyer and
identification of active compounds. J Korean Med Sci. 16
Suppl:S6–S18. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kang KS, Kim HY, Baek SH, Yoo HH, Park JH
and Yokozawa T: Study on the hydroxyl radical scavenging activity
changes of ginseng and ginsenoside-Rb2 by heat processing. Biol
Pharm Bull. 30:724–728. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nag SA, Qin JJ, Wang W, Wang MH, Wang H
and Zhang R: Ginsenosides as anticancer agents: In vitro and in
vivo activities, structure-activity relationships, and molecular
mechanisms of action. Front Pharmacol. 3:252012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee KY, Lee YH, Kim SI, Park JH and Lee
SK: Ginsenoside-Rg5 suppresses cyclin E-dependent protein kinase
activity via up-regulating p21Cip/WAF1 and down-regulating cyclin E
in SK-HEP-1 cells. Anticancer Res. 17A:1067–1072. 1997.
|
|
20
|
Shin YW, Bae EA and Kim DH: Inhibitory
effect of ginsenoside Rg5 and its metabolite ginsenoside Rh3 in an
oxazolone-induced mouse chronic dermatitis model. Arch Pharm Res.
29:685–690. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim TW, Joh EH, Kim B and Kim DH:
Ginsenoside Rg5 ameliorates lung inflammation in mice by inhibiting
the binding of LPS to toll-like receptor-4 on macrophages. Int
Immunopharmacol. 12:110–116. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim EJ, Jung IH, Van Le TK, Jeong JJ, Kim
NJ and Kim DH: Ginsenosides Rg5 and Rh3 protect scopolamine-induced
memory deficits in mice. J Ethnopharmacol. 146:294–299. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kühn K, D'Lima DD, Hashimoto S and Lotz M:
Cell death in cartilage. Osteoarthritis Cartilage. 12:1–16. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jubb RW and Fell HB: The breakdown of
collagen by chondrocytes. J Pathol. 130:159–167. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rosenberg AE: Bones, joints, and soft
tissue tumorsRobbins Pathologic Basis of Disease. 6th edition.
Cotran RS, Kumar C and Collins T: WB Saunders; Philadelphia, PA:
pp. 12531999
|
|
26
|
Naito K, Watari T, Muta T, Furuhata A,
Iwase H, Igarashi M, Kurosawa H, Nagaoka I and Kaneko K:
Low-intensity pulsed ultrasound (LIPUS) increases the articular
cartilage type II collagen in a rat osteoarthritis model. J Orthop
Res. 28:361–369. 2010.PubMed/NCBI
|
|
27
|
Goldring MB, Otero M, Plumb DA, Dragomir
C, Favero M, El Hachem K, Hashimoto K, Roach HI, Olivotto E, Borzì
RM, et al: Roles of inflammatory and anabolic cytokines in
cartilage metabolism: Signals and multiple effectors converge upon
MMP-13 regulation in osteoarthritis. Eur Cell Mater. 21:202–220.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wetzel M, Li L, Harms KM, Roitbak T,
Ventura PB, Rosenberg GA, Khokha R and Cunningham LA: Tissue
inhibitor of metalloproteinases-3 facilitates Fas-mediated neuronal
cell death following mild ischemia. Cell Death Differ. 15:143–151.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hashimoto S, Nishiyama T, Hayashi S,
Fujishiro T, Takebe K, Kanzaki N, Kuroda R and Kurosaka M: Role of
p53 in human chondrocyte apoptosis in response to shear strain.
Arthritis Rheum. 60:2340–2349. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Del Carlo M Jr and Loeser RF: Nitric
oxide-mediated chondrocyte cell death requires the generation of
additional reactive oxygen species. Arthritis Rheum. 46:394–403.
2002. View Article : Google Scholar : PubMed/NCBI
|