Introduction
Prostate cancer is the most prevalent malignancy and
the second leading cause of cancer-related mortality among men in
the United States with estimated 180,890 new cases and 26,120
estimated disease related death in 2016 (1). In China, the incidence rate of
prostate cancer shows a steady rise over the past years. In 2015,
prostate cancer has become the seventh most common cancer and the
tenth leading cause of cancer death in China men with 60,300
estimated new cancer cases and 26,600 estimated cancer deaths
(2). Currently, hormone ablation
therapy is the most common clinical approach to treat prostate
cancer. However, although patients exhibit an initial high response
rate to androgen withdrawal, most of them ultimately become
resistant to the therapy and develop castration-resistant prostate
cancer, for which there is no curative therapeutic strategies
available at present (3). Hence,
there is an imperative need to elucidate the molecular basis of
prostate cancer initiation and progression to develop novel
therapies against this disease.
Cap-dependent translation plays important roles in
the initiation and progression of many cancers through initiating
the translation of many oncogenic mRNAs such as cyclin D1 and c-MYC
(4). Cap-dependent translation is
controlled by the eIF4F complex, which initiates protein
translation by recruiting the 40S ribosomal subunit to the 5′ end
of mRNA (5). The eIF4F complex
contains eIF4A, eIF4G and eIF4E and its activity is mainly
regulated by the availability of eIF4E, which is controlled by the
phosphorylation of 4E-BP1 and the eIF4E expression level (6). Hypophosphorylated 4E-BP1 prevents the
assembly of the eIF4F complex by binding the same residues on eIF4E
necessary for the eIF4E-eIF4G interaction. Phosphorylation of
4E-BP1 weakens its binding to eIF4E and facilitates the formation
of the eIF4F complex (7).
Phosphorylation of the 4E-BP1 is mainly regulated by mammalian
target of rapamycin complex 1 (mTORC1), the role and mechanism of
which in the initiation and progression of prostate cancer has been
extensively studied (8). eIF4E has
been shown to be overexpressed in prostate cancer tissues and its
overexpression is significantly associated with poor patient
outcomes (9). Silencing eIF4E
expression in prostate cancer cells using an eIF4E-specific
antisense oligonucleotide (4EASO) markedly suppressed xenograft
tumor growth (10). Although the
role of eIF4E in prostate cancer development has been
characterized, little is known about the underlying mechanism
regulating eIF4E expression in the prostate cancer cells.
MicroRNAs (miRNAs), a class of small noncoding RNAs
(19–25 nucleotides), play a crucial role in the
post-transcriptional regulation of gene expression by binding to
the 3′-untranslated region (3′-UTR) of target mRNA, resulting in
either translational repression or mRNA degradation (11). miRNAs regulate different biological
processes, including cell proliferation, apoptosis, differentiation
and metastasis, deregulation of which is implicated in the
initiation and progression of cancer (12). Accumulating evidence demonstrates
that miRNAs are abnormally regulated in various human cancer types
and they can function as oncogenes or tumor suppressors in
tumorigenesis (13). In human
prostate cancer, oncogenic miRNAs, including miR-21, miR-32 and
miR-96, have been shown to be aberrantly overexpressed in tumors
and contribute to prostate carcinogenesis (14–16).
In contrast, miRNAs, such as miR-185, miR-302a and miR-340, are
downregulated in prostate cancer and exert tumor-suppressive roles
in the development of prostate cancer (17–19).
miR-455-3p has been reported to be downregulated in
many tumor tissues, including hepatocellular carcinoma (20), melanoma (21) and thyroid carcinoma (22). However, the exact role of miR-455-3p
in tumorigenesis is still unclear. In this study, we found that
miR-455-3p was downregulated in prostate cancer tissues compared
with normal prostate tissues. We demonstrated that miR-455-3p
suppressed prostate cancer cell proliferation and tumor formation.
In addition, we identified eIF4E as the target gene through which
miR-455-3p exerts its tumor-suppressive function.
Materials and methods
Tissue samples
The study protocol was approved by the ethics
committee of The Fourth Hospital of Baotou. Prostate cancer tissue
samples and normal prostate tissues were collected with informed
consent from the participants at the Department of Urinary Surgery
of The Fourth Hospital of Baotou. Both normal and prostate cancer
samples were confirmed by pathological examination. The tissue
samples were snap-frozen in liquid nitrogen and preserved at −80°C
until use.
Cells and cell culture
RWPE-1 cells were maintained in keratinocyte
serum-free medium (K-SFM, Invitrogen, Carlsbad, CA, USA)
supplemented with 50 mg/ml bovine pituitary extract, 100 U/ml
penicillin and 100 mg/l streptomycin. 22Rv1, LNCaP, C4-2 and PC-3
and DU145 cells were cultured in RPMI 1640 medium (Life
Technologies, Grand Island, NY, USA) supplemented with 10% fetal
bovine serum (FBS, Hyclone, Logan, UT, USA), 100 U/ml penicillin
and 100 mg/l streptomycin. Cells were maintained in a 37°C
incubator at 5% CO2.
To generate cell lines stably overexpressing
miR-455-3p, LNCaP and PC-3 cells were infected with appropriate
amounts of Lentiviral particles harboring hsa-miR-455-3p
(MIMAT0004784) (GeneChem Co., Shanghai, China), followed by
selection with 1 µg/ml puromycin (Sigma-Aldrich, St. Louis, MO,
USA) for two weeks.
Quantitative real-time PCR
miRNA from cell lines and frozen tissue samples was
extracted using a mirVana miRNA Isolation kit (Ambion, Carlsbad,
CA, USA) following the manufacturer's instructions. Expression of
miR-455-3p was determined using the PrimeScript miRNA RT-PCR kit
(Takara, Shiga, Japan) and was normalized to the U6 small nuclear
RNA. The forward primer for miR-455-3p is:
5′-ACACTCCAGCTGGGGCAGTCCACGGGCATATACAC-3′; the reverse primer for
miRNAs is the UnimiRqPCR Primer (Takara). The forward primer for U6
is: 5′-GCGCGTCGTGAAGCGTTC-3′; the reverse primer for U6 is
5′-GTGCAGGGTCCGAGGT-3′.
Total RNA from cell lines was extracted using TRIzol
Trizol (Qiagen, Valencia, CA, USA) and reverse-transcribed into
cDNA using a cDNA Library Construction kit (Takara, Dalian, China)
according to the manufacturer's instructions. RT-PCR was performed
using the QuantiTect SYBR Green PCR kit (Qiagen) according to the
manufacturer's protocols. The primer sequences were as follows:
EIF4E (forward: 5′-TACTAAGAGCGGCTCCACCAC-3′, reverse:
5′-TCGATTGCTTGACGCAGTCTCC-3′); CCND1 (forward:
5′-TGAACTACCTGGACCGCT-3′, reverse: 5′-GCCTCTGGCATTTTGGAG-3′); MYC:
(forward: 5′-GCTGCTTAGACGCTGGATTT-3′, reverse:
5′-CACCGAGTCGTAGTCGAGGT-3′); β-actin: (5′-GCACAGAGCCTCGCCTT-3′;
5′-GTTGTCGACGACGAGCG-3′).
Transient transfection of miR-455-3p
mimics or inhibitors
Chemically synthesized miR-455-3p mimics and
antagomiR-455-3p were purchased from RiboBio (Guangzhou, China).
miR-455-3p mimics or antagomiR-455-3p was transiently transfected
into cells using Lipofectamine RNAiMAX Transfection Reagent
(Invitrogen) according to the manufacturer's instructions and
subsequent measurements were performed 24 or 48 h later.
Cell proliferation assays
Transfected LNCaP or PC-3 cells were seeded in
96-well plates at 2000 cells per well. Cell proliferation was
measured using Cell Counting Kit-8 (CCK-8) (Dojindo Laboratories,
Tokyo, Japan) following the manufacturer's instructions. Absorbance
was measured at 450 nm using a multilabel plate reader. Each
experiment was performed with three replicates per sample.
Colony formation assay
Transfected LNCaP or PC-3 cells were seeded in a
6-well plate at 1000 cells per well containing RPMI-1640/10% FBS
medium. After two weeks, the cells were fixed with methanol for 20
min and stained with 1% crystal violet (Sigma-Aldrich) for 20 min.
The colony numbers were analyzed using ImageJ (Bethesda, MD, USA).
Each experiment was performed with three replicates per sample.
Soft agar assay
The cells were plated in 6-well dish at 5000 cells
per well, with a base layer of 0.5% agarose containing
RPMI-1640/10% FBS medium and a top layer of 0.35% agarose in the
same medium. The cells were allowed to grow for three weeks in a
37°C incubator at 5% CO2 before imaging and
counting.
Cloning and Luciferase reporter
assay
The full-length wild-type 3′-UTR of eIF4E was
amplified by PCR with the cDNA from PC3 cells and cloned into
pMIR-Reporter plasmid (Life Technologies). Mutations in miR-455-3p
binding site of 3′-UTR of eIF4E was introduced using a QuickChange
site-directed Mutagenesis kit (Stratagene, La Jolla, CA, USA)
following the manufacturer's instructions. All vectors were
confirmed by sequencing.
The cells were seeded into 24-well plates at
4×104 cells per well. The following day, the cells were
co-transfected with luciferase reporter plasmids with wild-type
3′-UTR or mutant 3′-UTR of eIF4E, and miR-455-3p mimics or
antagomiR-455-3p using Lipofectamine 2000 (Invitrogen, New York,
NY, USA). At 48 h after transfection, luciferase activities were
determined using the Dual-Luciferase Reporter assay system
(Promega, Madison, WI, USA). The pRL-TK vector was used as an
internal control. Assays were performed in triplicate and results
are presented as means ± standard deviation (SD).
Western blotting
Cells were lysed in ice-cold RIPA buffer (50 mM
Tris-HCl, pH 7.5, 150 mM NaCl, 1% sodium deoxycholate, 1% NP-40,
0.1% SDS, 1 mM EDTA, 1 mM EGTA) containing protease/phosphatase
inhibitor cocktail (Roche, San Francisco, CA, USA). After the
measurement of protein concentration using the Pierce BCA protein
assay kit (Thermo Fisher Scientific, Waltham, MA, USA), equal
amounts of total protein were separated on 10% SDS-PAGE and
transferred to nitrocellulose membranes (Bio-Rad, Richmond, CA,
USA). After being blocked with 5% non-fat milk in Tris-buffered
saline containing 0.05% Tween-20 for 1 h at room temperature, the
members were probed with primary antibodies against eIF4E, cyclin
D1 and c-Myc (Cell Signaling Technology, Beverly, MA, USA) or
Tubulin (Santa Cruz Biotechnologies, Dallas, TX, USA) overnight at
4°C, followed by incubation with HRP-conjugated secondary
antibodies for 2 h at room temperature. Detection was performed
using a Super Signal Enhanced Chemiluminescence kit (Pierce
Biotechnology, Inc., Rockford, IL, USA).
Cap-dependent translational activity
assay
Cap-dependent translational activity in the cells is
determined using pcDNA3-rLuc-PolioIRES-fLuc plasmid, which directs
cap-dependent translation of the Renilla luciferase gene and
cap-independent Polio IRES-mediated translation of the firefly
gene. LNCaP or PC-3 cells were transiently co-transfected with
pcDNA3-rLuc-PolioIRES-fLuc plasmid and miR-455-3p mimics or
antagomiR-455-3p. At 48 h after transfection, cells were harvested
for luciferase activity assay using the Dual-Luciferase Reporter
assay system (Promega) according to the manufacturer's
instructions. Cap-dependent translational activity was measured by
calculating the ratio of Renilla/firefly luciferase
activity. Assays were performed in triplicate.
In vivo tumorigenesis assay
Male BALB/c nude mice (5–6-week-old) were purchased
from the Animal Center for Vitalriver (Beijing, China). To
determine the effect of miR-455-3p overexpression on the tumor
growth, PC3 cells stably expressed miR-455-3p or control cells in
0.1 ml serum-free medium were injected subcutaneously in the right
flank of nude mice, respectively. To determine the effect of
miR-455-3p inhibition on the tumor growth, PC3 cells were injected
subcutaneously in the right flank of nude mice. When the average
tumor volume had reached approximately 100 mm3, the mice
were randomly divided to 2 groups (n=6/group) according to body
weights and tumor volumes and intratumorally injected
antagomir-455-3p (10 nM of antagomir-455-3p diluted in 50 µl PBS)
or antagomir-NC with two times per week for 3 weeks. Tumor sizes
were recorded at indicated times.
Statistical analysis
All data were analyzed with the unpaired Student
t-test using SPSS 17.0 software (SPSS, Chicago, IL, USA). The data
are presented as mean ± SD. For all analyses, P-values <0.05
were considered to indicate statistically significant
differences.
Results
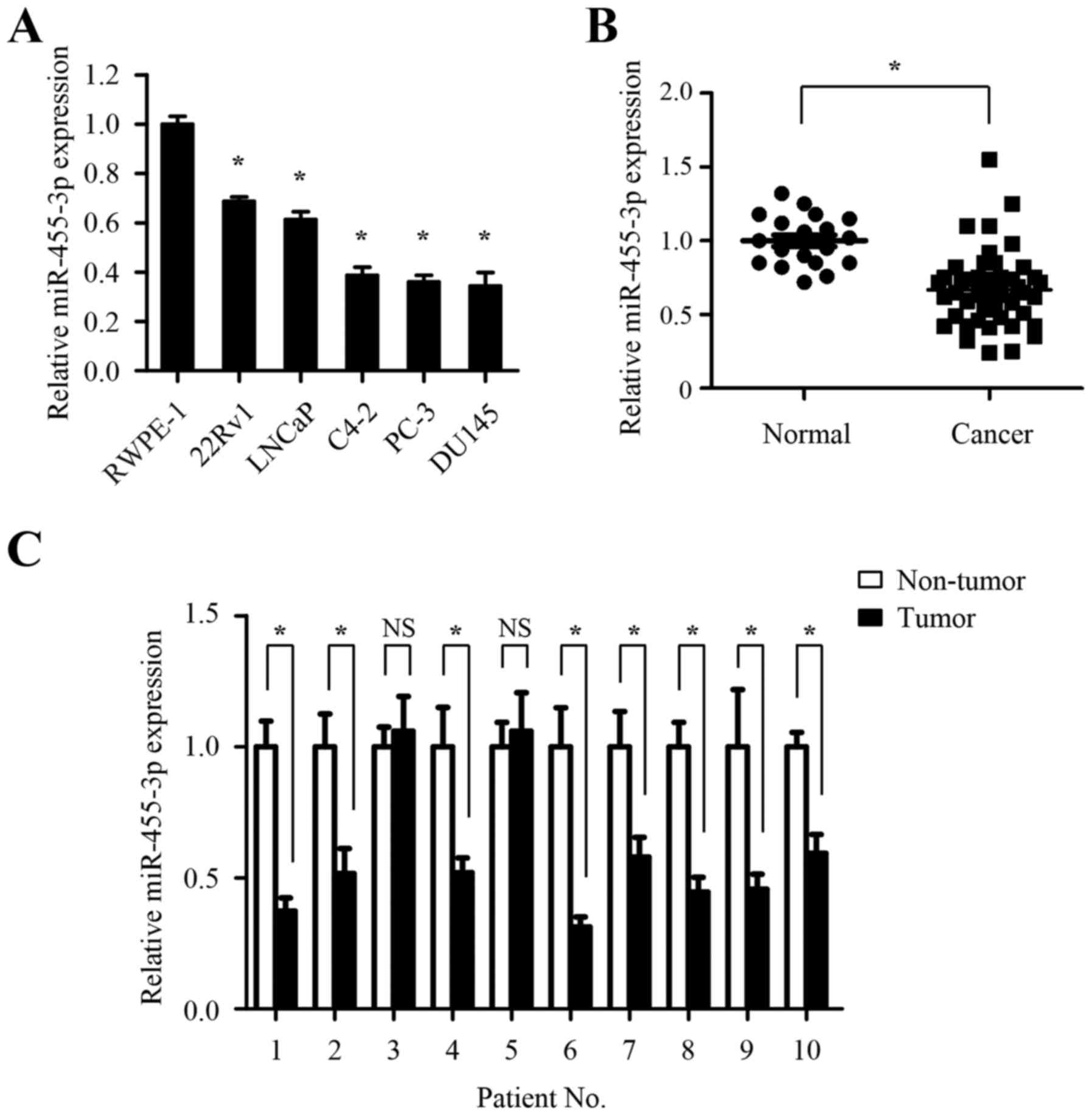
miR-455-3p is significantly
downregulated in prostate cancer cell lines and tissues
To investigate the significance of miR-455-3p in
prostate carcinogenesis, the expression of miR-455-3p was measured
in one benign prostate cell line (RWPE-1) and five prostate cancer
cell lines (22Rv1, LNCaP, C4-2, PC-3 and DU145) using quantitative
PCR (qPCR). As shown in Fig. 1A,
the expression of miR-455-3p in prostate cancer cell lines was
significantly reduced compared with benign prostate cell lines.
Next, we investigated whether miR-455-3p is downregulated in
clinical prostate cancer tissues. The abundance of miR-455-3p was
detected in 65 clinical prostatic tissues, including 18 normal
prostate tissues and 47 prostate cancer tissues. As shown in
Fig. 1B, the expression of
miR-455-3p in prostate cancer tissues was profoundly decreased
compared with that in normal prostate tissues, suggesting a
potential involvement of miR-455-3p in prostate cancer initiation.
To verify the suppression of miR-455-3p in prostate cancer
patients, we determined the levels of miR-455-3p in 10 randomly
selected prostate cancer samples and matched benign adjacent
tissues. Compared with matched benign tissues, 8 of 10 prostate
cancer samples (80%) exhibited marked downregulation of miR-455-3p
(Fig. 2C). Taken together, these
results from cell lines and clinical specimens suggested that
miR-455-3p is aberrantly downregulated in prostate cancer cells and
its low expression may be associated with biological process of
tumorigenesis.
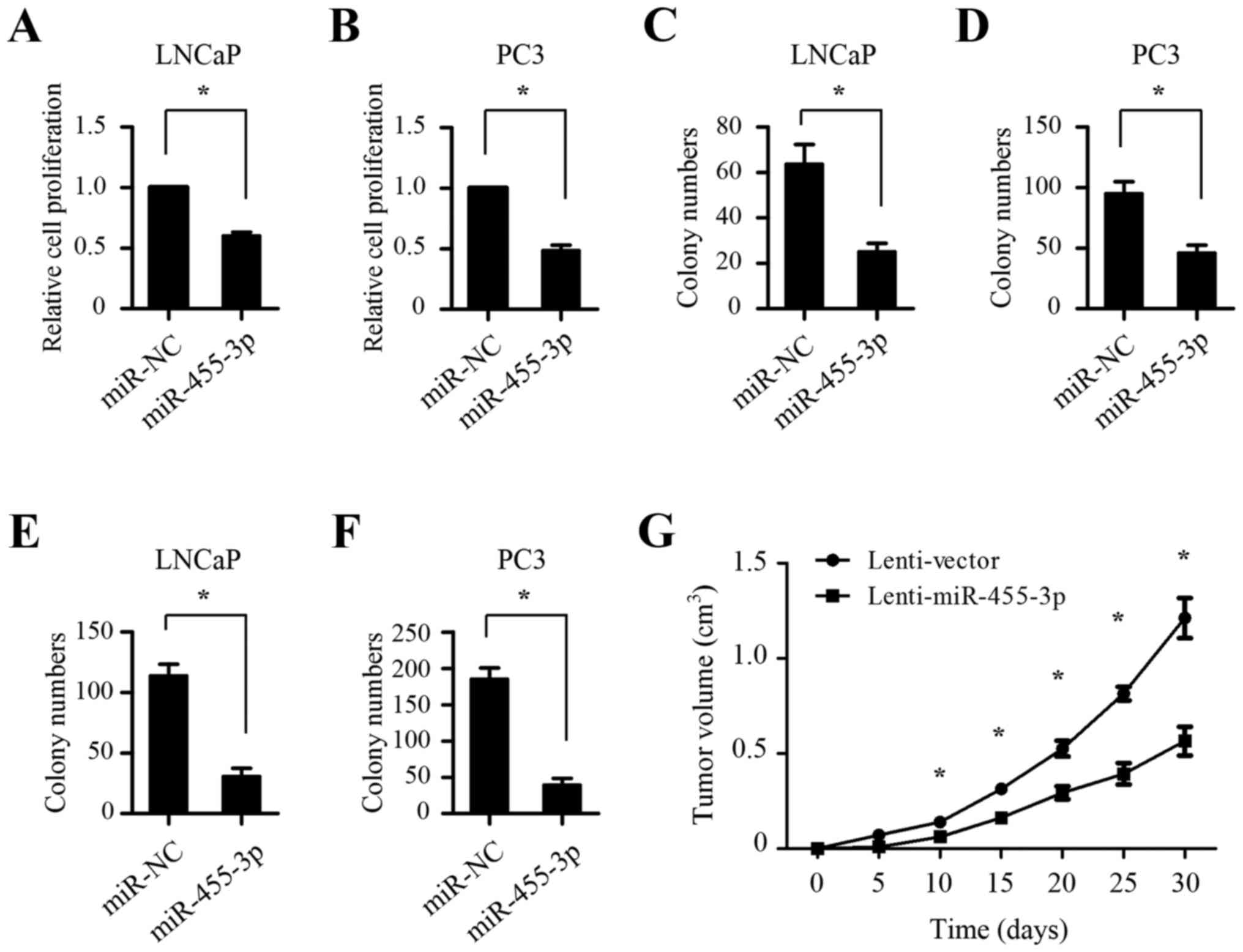
Upregulation of miR-455-3p suppresses
prostate cancer cell growth
The low expression of miR-455-3p in prostate cancer
indicates that miR-455-3p may function as tumor suppressor in
prostate tumorigenesis. To investigate the role of miR-455-3p, the
miR-455-3p mimics were transfected into LNCaP and PC-3 cells. As
shown in Fig. 2A-D, CCK8 and colony
formation assays demonstrated that miR-455-3p overexpression
significantly inhibited the growth of both LNCaP and PC-3 cells.
Soft agar assays showed that ectopic expression of miR-455-3p
markedly decreased anchorage-independent growth ability of both
LNCaP and PC-3 cells (Fig. 2E and
F). To further determine whether miR-455-3p could suppress
tumor growth in vivo, PC-3 cells stably overexpressing
miR-455-3p with a lentiviral vector and control cells were injected
into the flanks of nude mice. The results showed that miR-455-3p
overexpression also markedly decreased the growth of subcutaneous
xenograft tumors (Fig. 2G). Taken
together, these data indicate that miR-455-3p has a suppressor role
in prostate cancer cell growth.
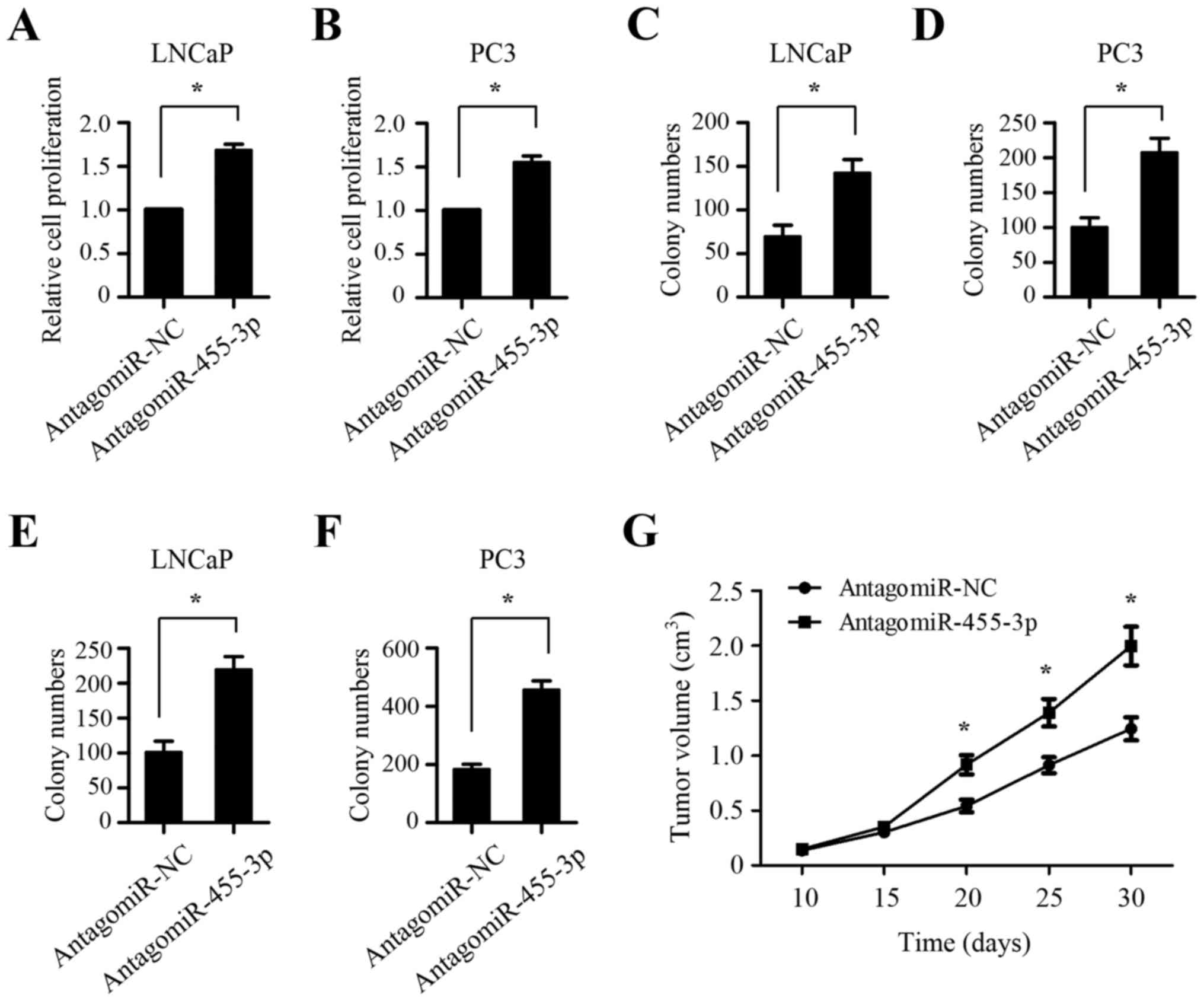
Silencing endogenous miR-455-3p
promotes prostate cancer cell growth in vitro and in vivo
To confirm the inhibitory role of miR-455-3p in
prostate cancer cell growth, we silenced endogenous miR-455-3p
using the miRNA antagonist antagomir-455-3p and determined the
effect of miR-455-3p downregulation on prostate cancer cell growth.
CCK8 and colony formation assays revealed that miR-455-3p
inhibition drastically promoted the growth of both LNCaP and PC-3
cells (Fig. 3A-D). Soft agar assays
showed that miR-455-3p depletion markedly increased
anchorage-independent growth ability of both LNCaP and PC-3 cells
(Fig. 3E and F). We next detected
the role of silencing endogenous miR-455-3p on prostate cancer cell
tumorigenesis. Nude mice bearing PC-3- cell-derived tumors were
injected with antagomir-455-3p or negative control through the
lateral tail vein every three days for 3 weeks. As shown in
Fig. 3E, antagomir-455-3p treatment
significantly enhanced prostate cancer cell tumorigenic ability
in vivo. Taken together, these results indicate that
silencing endogenous miR-455-3p promotes prostate cancer cell
growth in vitro and in vivo.
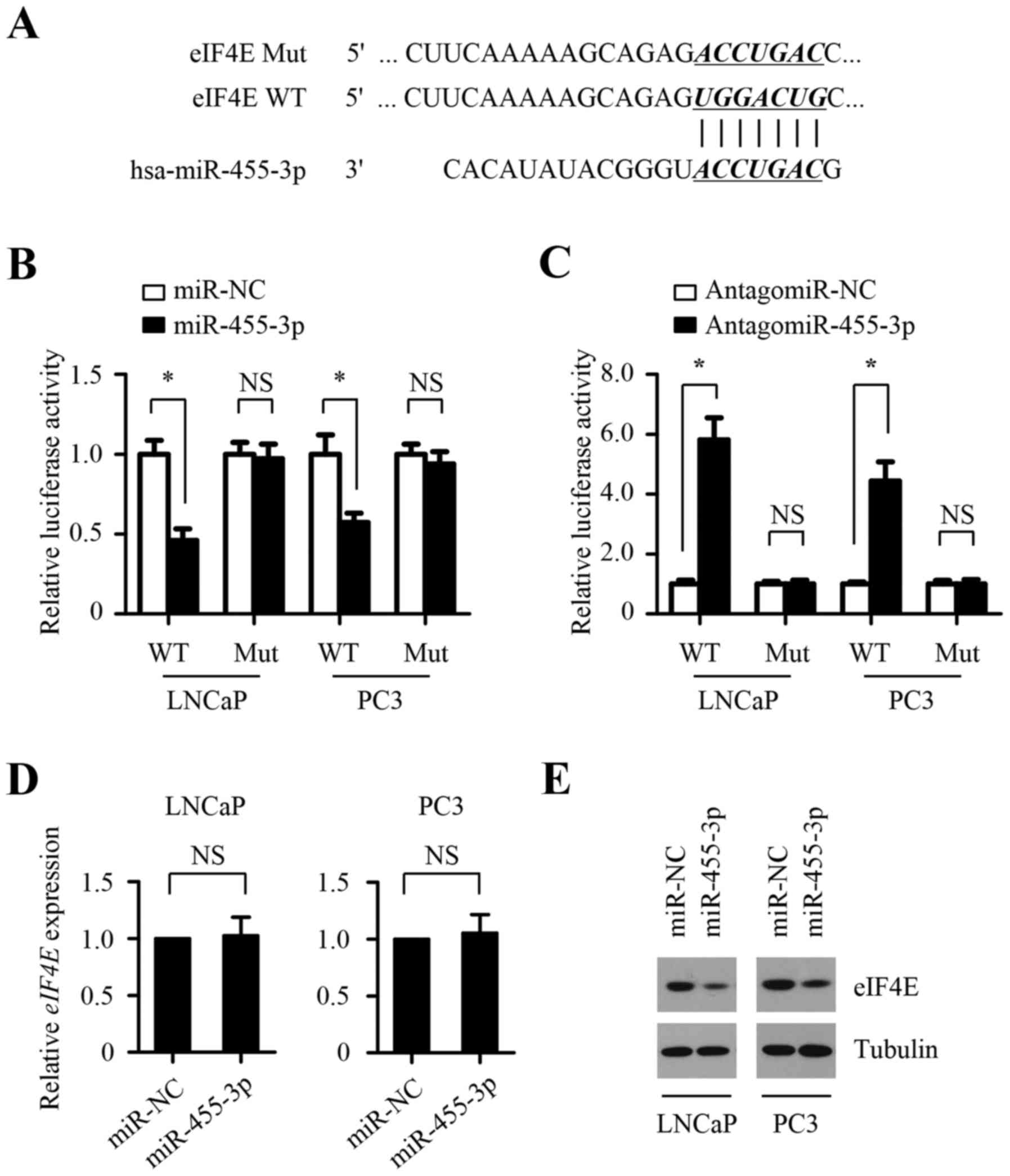
miR-455-3p negatively regulates the
expression of eIF4E at the post-transcriptional level by directly
binding to the 3′-UTR of eIF4E mRNA
To explore the underlying molecular mechanism by
which miR-455-3p inhibits prostate cancer cell proliferation, we
explored its target genes by employing two bioinformatics tools,
TargetScan and miRanda. EIF4E was chosen as a preferred candidate
target gene of miR-455-3p due to its role as oncogene in prostate
cancer progression and one possible binding site of miR-455-3p in
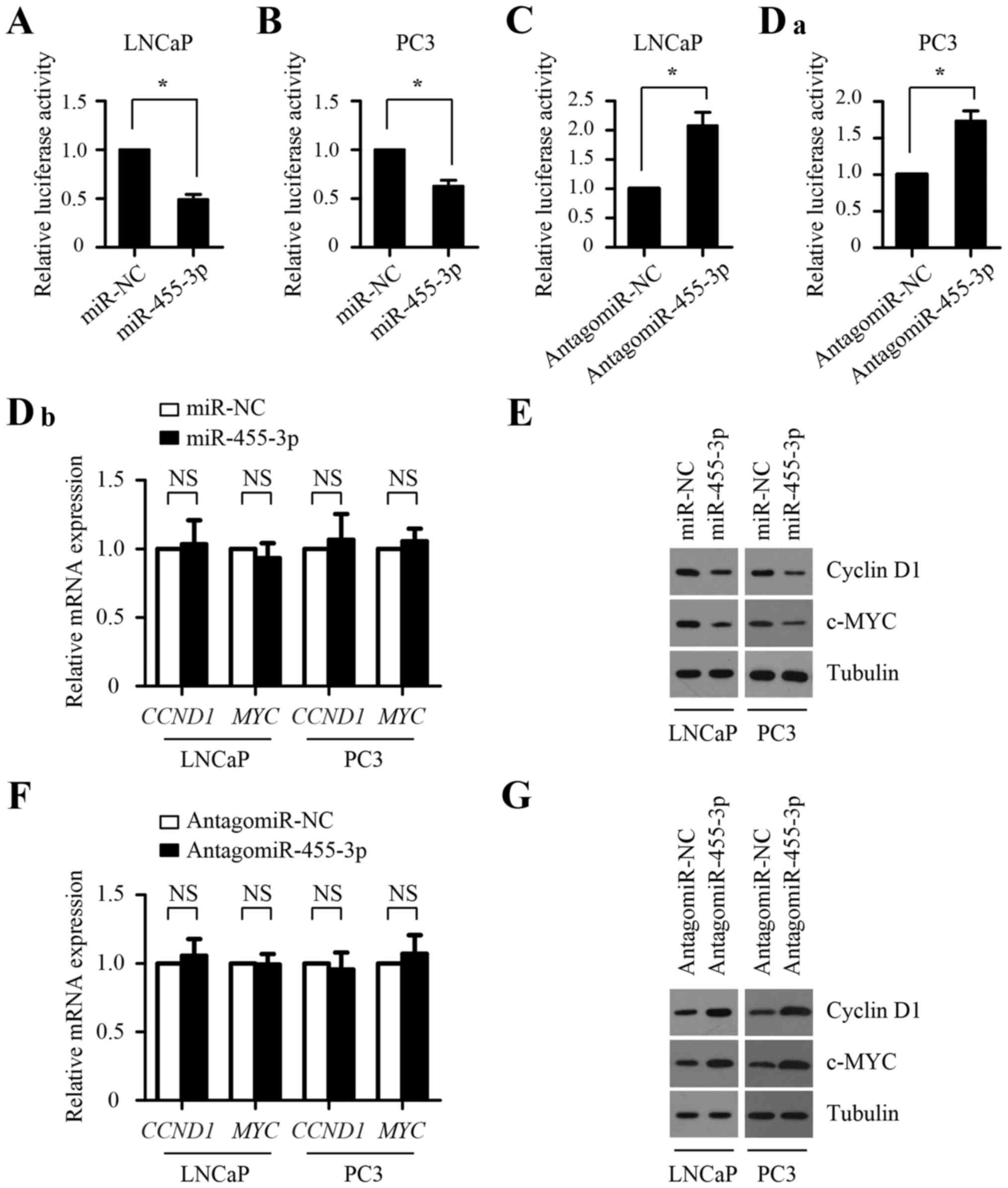
its 3′-untranslated region (3′-UTR) (Fig. 4A). To determine whether EIF4E is a
direct target of miR-455-3p, luciferase activity assays were
performed with luciferase reporters carrying putative EIF4E 3′-UTR
binding sites or mutant binding sites downstream of a luciferase
reporter gene (Fig. 4A). As shown
in Fig. 4B, miR-455-3p
overexpression decreased luciferase activity in both LNCaP and PC-3
cells, whereas this suppressive effects was abolished by the
potential miR-455-3p binding site mutation in the 3′-UTRs of EIF4E,
indicating that miR-455-3p directly targets EIF4E. The opposite
result was observed in cells transfected with antagomir-455-3p
(Fig. 4C), suggesting that the
3′-UTRs of EIF4E was suppressed by endogenous miR-455-3p.
Furthermore, western blot assays showed that miR-455-3p
overexpression suppressed the protein expression level of EIF4E in
both LNCaP and PC-3 cells, whereas miR-455-3p inhibition promoted
its protein expression level (Fig. 4E
and G). However, miR-455-3p expression has little effect on the
mRNA level of EIF4E (Fig. 4D and
F). Taken together, these results indicate that miR-455-3p
directly targets the EIF4E 3′-UTR to suppress it protein expression
at the post-transcriptional level.
miR-455-3p inhibits cap-dependent
translation in prostate cancer cells
Given that the expression level of eIF4E is the
rate-limiting factor for the assembly of the eIF4F complex and
cap-dependent translation and we have established the inhibitory
role of miR-455-3p on the expression of eIF4E, we presumed that
miR-455-3p may suppress cap-dependent translation in prostate
cancer cells. To test this hypothesis, the cap-dependent
translation rate in prostate cancer cells transfected with
miR-455-3p mimics or antagomir-455-3p was determined using a
dual-luciferase reporter system which can monitor the ratio between
cap-dependent (Renilla luciferase) and cap-independent
IRES-mediated (firefly luciferase) translation initiation. As shown
in Fig. 5A-Da, miR-455-3p
overexpression inhibited cap-dependent translation in both LNCaP
and PC-3 cells, whereas miR-455-3p inhibition promoted
cap-dependent translation. We next analyzed the effect of
miR-455-3p on the expression of cyclin D1 and c-MYC, two well-known
oncogenic genes regulated by cap-dependent translation. As shown in
Fig. 5E and G, miR-455-3p
overexpression led to decreased protein levels of cyclin D1 and
c-MYC in LNCaP and PC-3 cells, whereas miR-455-3p inhibition
resulted in increased protein levels of c-MYC. Contrary to the
inhibition of cyclin D1 and c-MYC at the protein level by
miR-455-3p mimics and the increase by antagomir-455-3p, the
miR-455-3p mimics or antagomir-455-3p have little effect on the
mRNA levels of CCND1 and MYC (Fig. 5Db and F), suggesting that the
inhibitory effect of cyclin D1 and c-MYC expression by miR-455-3p
results from the repression of their translational inhibition
rather than that of their gene transcription. Taken together, our
results suggest that miR-455-3p inhibits cap-dependent translation
in prostate cancer cells.
miR-455-3p inhibits cap-dependent
translation and the proliferation of prostate cancer cells through
targeting EIF4E
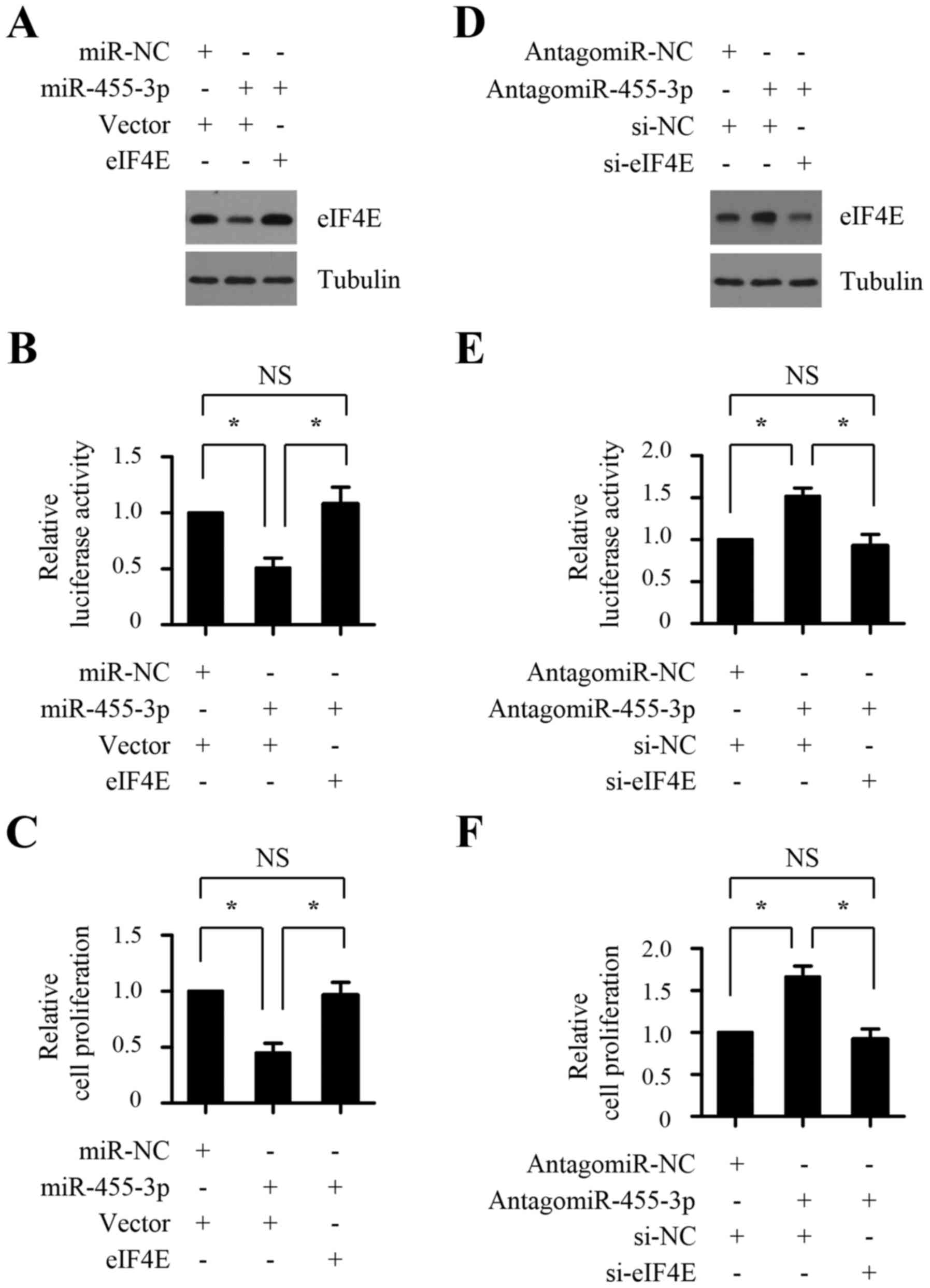
To explore the functional significance of EIF4E in
cap-dependent translation and the proliferation of prostate cancer
cells suppressed by miR-455-3p, 3′ UTR-deleted eIF4E plasmid was
introduced into PC-3 cells transfected with miR-455-3p mimics, and
then cap-dependent translation and cell proliferation was
determined by dual-luciferase reporter system and CCK8 assays,
respectively. As shown in Fig.
6A-C, ectopic overexpression of miR-455-3p resulted in reduced
cap-dependent translation rates and suppressed proliferation of
PC-3 cells, whereas co-transfection with eIF4E plasmid reversed
inhibition of cap-dependent translation and cell proliferation
caused by miR-455-3p mimics. We next analyzed the effect of eIF4E
silencing on antagomir-455-3p-mediated promotion of cap-dependent
translation and cell proliferation of PC-3 cells. As shown in
Fig. 6D-F, PC-3 cells transfected
with antagomir-455-3p exhibited markedly increased cap-dependent
translation rates and cell proliferation, whereas silencing eIF4E
expression markedly abrogated these increase. Taken together, these
results suggest that miR-455-3p inhibits cap-dependent translation
and the proliferation of prostate cancer cells through targeting
EIF4E.
Discussion
Growing evidence indicates that miRNAs exhibit
altered expression levels in many cancers and are emerging as key
regulators in the occurrence and progression of cancer. Therefore,
miRNAs have been extensively investigated in cancer research for
identifying novel diagnostic and prognostic biomarkers and
therapeutic targets. In prostate cancer, many miRNAs have been
demonstrated to be aberrantly expressed in prostate cancer tissues
and exhibit oncogenic function or tumor suppressor role in prostate
tumorigenesis. For example, miR-21 has been shown to be
overexpressed in prostate cancer tissues and promote both
hormone-dependent and hormone-independent tumor growth of prostate
cancer cells (14). Further studies
demonstrated that miR-21 promotes the survival and invasion of
prostate cancer cells by directly targeting MARCKS and RECK
(23,24). It was reported that miRNA-302a
expression is significantly downregulated in prostate cancer tissue
compared with normal prostate tissue and its overexpression
markedly inhibits prostate cancer growth in vitro and in
vivo. Moreover, AKT was identified as the direct target gene
through which miRNA-302a exerts tumor suppressor role in prostate
carcinogenesis (19).
Recent studies demonstrate that miR-455-3p is
involved in many physiological and pathological processes.
miR-455-3p has been identified as an important regulator of brown
adipogenesis and promotes the brown adipogenic program and
mitochondrial biogenesis by targeting HIF1an-AMPK-PGC1a signaling
network (25). It was demonstrated
that miR-455-3p activates early chondrogenic differentiation by
directly targeting Runx2 (26). In
addition, miR-455-3p was showed to be significantly downregulated
in placentas from preeclampsia patients and be linked to aberrant
hypoxia signaling in preeclampsia (27). Compared with normal tissues,
miR-455-3p was demonstrated to be aberrantly expressed in various
cancer tissues, including hepatocellular carcinoma (20), melanoma (21) and thyroid carcinoma (22). However, little is known about the
miR-455-3p status in prostate cancer tissues and the possible roles
in prostate carcinogenesis. In the present study, we determined the
expression of miR-455-3p in prostate cancer tissues and found that
its expression in prostate cancer tissues was profoundly decreased
compared with that in normal prostate tissues, indicating that
miR-455-3p is associated with prostate carcinogenesis and it may
function as a tumor suppressor. Further gain-of-function and
loss-of-function results reveal that miR-455-3p suppresses prostate
cancer cell growth in vitro and in vivo. All these
studies demonstrate that miR-455-3p exerts tumor suppressor role in
prostate cancer progression.
Although the oncogenic role of eIF4E has been well
characterized, the mechanisms by which the eIF4E expression is
regulated remain incompletely defined. c-MYC, a well-known
oncogene, has been reported to increase the eIF4E expression at
transcriptional level (28). HSP27
enhances eIF4E protein stability by decreasing eIF4E ubiquitination
and proteasomal degradation, thus promoting the development of
castration resistant prostate cancer (29). Recent studies demonstrate that
miRNAs play an important role in the regulation of eIF4E
expression. miR-34c-3p was reported to be downregulated in
non-small cell lung cancer (NSCLC) tissues and to inhibit NSCLC
cell proliferation and migration by targeting eIF4E (30). It was reported that repression of
microRNA-768-3p by MEK/ERK signaling enhances the eIF4E expression
and protein synthesis in melanoma cells (31). miR-141 was shown to repress the
eIF4E expression and confer docetaxel chemoresistance of breast
cancer cells (32).
Using a database search, we identified that the
3′-UTR of EIF4E mRNA contains a putative miR-455-3p binding site.
Reporter assay and western blotting demonstrated that miR-455-3p
negatively regulates the expression of EIF4E at the
post-transcriptional level by directly binding to the 3′-UTR of
EIF4E mRNA. EIF4E, as a component of the eIF4F complex, determines
the cap-dependent translation activity and plays an important role
in tumorigenesis through controlling the translation initiation of
a wide range of proteins involving hallmarks of cancers, such as
cell cycle, survival, migration and angiogenesis. In agreement with
its critical role in tumorigenesis, elevated expression of eIF4E
has been shown in prostate cancer patient tissues and eIF4E
depletion inhibits prostate cancer cell growth in vitro and
in vivo (9,10). Given that we have found that
miR-455-3p suppresses the expression of eIF4E in prostate cancer
cells, we next determined the effect of miR-455-3p on the
cap-dependent translation activity. Using bicistronic luciferase
reporter assays, we found that miR-455-3p suppresses the
cap-dependent translation activity in prostate cancer cells.
Further results demonstrate that the inhibitory
effect of miR-455-3p mimics on cap-dependent translation and cell
proliferation in prostate cancer cells can be effectively reversed
by eIF4E overexpression, indicating the functional significance of
EIF4E in mediating the tumor suppressor role of miR-455-3p. Taken
together, our results suggest miR-455-3p inhibits cap-dependent
translation and the proliferation of prostate cancer cells through
targeting eIF4E. Whether the expression level of miR-455-3p is
negatively associated with that of eIF4E in clinical tissues
remains to be addressed and is a research direction we are
currently pursuing. It has been reported that eIF4E shows a higher
expression in advanced prostate cancer tissues when compared with
low-grade prostate cancer tissues, and promote the development of
castration resistant prostate cancer (CRPC) (29,33).
Therefore, it will be interesting to determine whether miR-455-3p
is involved in CRPC progression by targeting eIF4E in a future
study.
In summary, in this study we present evidence that
miR-455-3p acts as a tumor suppressor by limiting eIF4E expression
in prostate cancer progression. miR-455-3p was markedly
downregulated in prostate cancer tissues and suppressed prostate
cancer cell growth in vitro and in vivo. Although
further studies are required to determine the mechanism by which
the expression of miR-455-3p is repressed in prostate cancer, our
results indicate that miR-455-3p plays a critical role in prostate
malignant transformation and may serve as a novel therapy target
for prostate cancer treatment.
Acknowledgements
This work was supported by Baotou Medical and Health
Fund (WSIJ2014038 to H.J.).
Glossary
Abbreviations
Abbreviations:
|
miRNAs
|
microRNAs
|
|
miR-455-3p
|
microRNA-455-3p
|
|
eIF4E
|
eukaryotic translation initiation
factor 4E
|
|
3′-UTR
|
the 3′-untranslated region
|
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Watson PA, Arora VK and Sawyers CL:
Emerging mechanisms of resistance to androgen receptor inhibitors
in prostate cancer. Nat Rev Cancer. 15:701–711. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pettersson F, Del Rincon SV and Miller WH
Jr: Eukaryotic translation initiation factor 4E as a novel
therapeutic target in hematological malignancies and beyond. Expert
Opin Ther Targets. 18:1035–1048. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Spilka R, Ernst C, Mehta AK and Haybaeck
J: Eukaryotic translation initiation factors in cancer development
and progression. Cancer Lett. 340:9–21. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pelletier J, Graff J, Ruggero D and
Sonenberg N: Targeting the eIF4F translation initiation complex: A
critical nexus for cancer development. Cancer Res. 75:250–263.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Siddiqui N and Sonenberg N: Signalling to
eIF4E in cancer. Biochem Soc Trans. 43:763–772. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bitting RL and Armstrong AJ: Targeting the
PI3K/Akt/mTOR pathway in castration-resistant prostate cancer.
Endocr Relat Cancer. 20:R83–R99. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kremer CL, Klein RR, Mendelson J, Browne
W, Samadzedeh LK, Vanpatten K, Highstrom L, Pestano GA and Nagle
RB: Expression of mTOR signaling pathway markers in prostate cancer
progression. Prostate. 66:1203–1212. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Graff JR, Konicek BW, Vincent TM, Lynch
RL, Monteith D, Weir SN, Schwier P, Capen A, Goode RL, Dowless MS,
et al: Therapeutic suppression of translation initiation factor
eIF4E expression reduces tumor growth without toxicity. J Clin
Invest. 117:2638–2648. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mao B and Wang G: MicroRNAs involved with
hepatocellular carcinoma (Review). Oncol Rep. 34:2811–2820.
2015.PubMed/NCBI
|
|
12
|
Gambari R, Brognara E, Spandidos DA and
Fabbri E: Targeting oncomiRNAs and mimicking tumor suppressor
miRNAs: Νew trends in the development of miRNA therapeutic
strategies in oncology (Review). Int J Oncol. 49:5–32.
2016.PubMed/NCBI
|
|
13
|
Garzon R and Marcucci G: Potential of
microRNAs for cancer diagnostics, prognostication and therapy. Curr
Opin Oncol. 24:655–659. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ribas J, Ni X, Haffner M, Wentzel EA,
Salmasi AH, Chowdhury WH, Kudrolli TA, Yegnasubramanian S, Luo J,
Rodriguez R, et al: miR-21: An androgen receptor-regulated microRNA
that promotes hormone-dependent and hormone-independent prostate
cancer growth. Cancer Res. 69:7165–7169. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Eberlein C, Kendrew J, McDaid K, Alfred A,
Kang JS, Jacobs VN, Ross SJ, Rooney C, Smith NR, Rinkenberger J, et
al: A human monoclonal antibody 264RAD targeting αvβ6 integrin
reduces tumour growth and metastasis, and modulates key biomarkers
in vivo. Oncogene. 32:4406–4416. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fendler A, Jung M, Stephan C, Erbersdobler
A, Jung K and Yousef GM: The antiapoptotic function of miR-96 in
prostate cancer by inhibition of FOXO1. PLoS One. 8:e808072013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Qu F, Cui X, Hong Y, Wang J, Li Y, Chen L,
Liu Y, Gao Y, Xu D and Wang Q: MicroRNA-185 suppresses
proliferation, invasion, migration, and tumorigenicity of human
prostate cancer cells through targeting androgen receptor. Mol Cell
Biochem. 377:121–130. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang GM, Bao CY, Wan FN, Cao DL, Qin XJ,
Zhang HL, Zhu Y, Dai B, Shi GH and Ye DW: MicroRNA-302a suppresses
tumor cell proliferation by inhibiting AKT in prostate cancer. PLoS
One. 10:e01244102015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang K, Tang Y, He L and Dai Y:
MicroRNA-340 inhibits prostate cancer cell proliferation and
metastasis by targeting the MDM2-p53 pathway. Oncol Rep.
35:887–895. 2016.PubMed/NCBI
|
|
20
|
Shubbar E, Kovács A, Hajizadeh S, Parris
TZ, Nemes S, Gunnarsdóttir K, Einbeigi Z, Karlsson P and Helou K:
Elevated cyclin B2 expression in invasive breast carcinoma is
associated with unfavorable clinical outcome. BMC Cancer. 13:12013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Segura MF, Belitskaya-Lévy I, Rose AE,
Zakrzewski J, Gaziel A, Hanniford D, Darvishian F, Berman RS,
Shapiro RL, Pavlick AC, et al: Melanoma microRNA signature predicts
post-recurrence survival. Clin Cancer Res. 16:1577–1586. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Swierniak M, Wojcicka A, Czetwertynska M,
Stachlewska E, Maciag M, Wiechno W, Gornicka B, Bogdanska M,
Koperski L, de la Chapelle A, et al: In-depth characterization of
the microRNA transcriptome in normal thyroid and papillary thyroid
carcinoma. J Clin Endocrinol Metab. 98:E1401–E1409. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li T, Li D, Sha J, Sun P and Huang Y:
MicroRNA-21 directly targets MARCKS and promotes apoptosis
resistance and invasion in prostate cancer cells. Biochem Biophys
Res Commun. 383:280–285. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Reis ST, Pontes-Junior J, Antunes AA,
Dall'Oglio MF, Dip N, Passerotti CC, Rossini GA, Morais DR,
Nesrallah AJ, Piantino C, et al: miR-21 may acts as an oncomir by
targeting RECK, a matrix metalloproteinase regulator, in prostate
cancer. BMC Urol. 12:142012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang H, Guan M, Townsend KL, Huang TL, An
D, Yan X, Xue R, Schulz TJ, Winnay J, Mori M, et al: MicroRNA-455
regulates brown adipogenesis via a novel HIF1an-AMPK-PGC1α
signaling network. EMBO Rep. 16:1378–1393. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang Z, Hou C, Meng F, Zhao X, Zhang Z,
Huang G, Chen W, Fu M and Liao W: MiR-455-3p regulates early
chondrogenic differentiation via inhibiting Runx2. FEBS Lett.
589:3671–3678. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lalevée S, Lapaire O and Bühler M: miR455
is linked to hypoxia signaling and is deregulated in preeclampsia.
Cell Death Dis. 5:e14082014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rosenwald IB, Rhoads DB, Callanan LD,
Isselbacher KJ and Schmidt EV: Increased expression of eukaryotic
translation initiation factors eIF-4E and eIF-2 alpha in response
to growth induction by c-myc. Proc Natl Acad Sci USA. 90:6175–6178.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Andrieu C, Taieb D, Baylot V, Ettinger S,
Soubeyran P, De-Thonel A, Nelson C, Garrido C, So A, Fazli L, et
al: Heat shock protein 27 confers resistance to androgen ablation
and chemotherapy in prostate cancer cells through eIF4E. Oncogene.
29:1883–1896. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu F, Wang X, Li J, Gu K, Lv L, Zhang S,
Che D, Cao J, Jin S and Yu Y: miR-34c-3p functions as a tumour
suppressor by inhibiting eIF4E expression in non-small cell lung
cancer. Cell Prolif. 48:582–592. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jiang CC, Croft A, Tseng HY, Guo ST, Jin
L, Hersey P and Zhang XD: Repression of microRNA-768-3p by MEK/ERK
signalling contributes to enhanced mRNA translation in human
melanoma. Oncogene. 33:2577–2588. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yao YS, Qiu WS, Yao RY, Zhang Q, Zhuang
LK, Zhou F, Sun LB and Yue L: miR-141 confers docetaxel
chemoresistance of breast cancer cells via regulation of EIF4E
expression. Oncol Rep. 33:2504–2512. 2015.PubMed/NCBI
|
|
33
|
Graff JR, Konicek BW, Lynch RL, Dumstorf
CA, Dowless MS, McNulty AM, Parsons SH, Brail LH, Colligan BM, Koop
JW, et al: eIF4E activation is commonly elevated in advanced human
prostate cancers and significantly related to reduced patient
survival. Cancer Res. 69:3866–3873. 2009. View Article : Google Scholar : PubMed/NCBI
|