Lung cancer is the most commonly diagnosed cancer
worldwide, responsible for 19.4% of all cancer-related mortalities
(1). Non-small cell lung cancer
(NSCLC) and SCLC are the main subtypes of lung cancer, with NSCLC
representing ~85% of cases (2).
While the incidence of lung cancer in the US has been declining
since 2005 (1) due to decreased
smoking rates (3), the incidence in
China for the same period has been increasing, particularly in the
female population and the younger generation (4,5).
Smoking and air pollution have been reported to be mainly
responsible for the increase in lung cancer incidence and
mortalities in China (5). The
majority of patients with lung cancer are usually diagnosed at an
advanced stage, leaving limited treatment options (3,6). As a
result, the prognosis of lung cancer patients remains poor, with a
5-year overall survival rate as low as 15%, despite progress being
made in the field of NSCLC (7). A
poor treatment outcome has been associated with the multidrug
resistance-associated protein 1 (MRP1) gene, which is commonly
overexpressed in NSCLC tissues and may limit the efficacy of
chemotherapy (8).
MRP1 was first identified and cloned in the
anthracycline-selected human small-cell lung carcinoma cell line,
H69AR (8). MRP1 is a member of the
subfamily C of ATP-binding cassette (ABC) transporters, and is
hence also known as ABCC1 (9).
Distributed throughout a variety of normal human tissues, MRP1 is
present in organs such as the lungs, spleen, testes, kidneys,
thyroid, bladder and adrenal glands (9,10). In
normal cells, MRP1 mediates the efflux of endogenous metabolites,
including glutathione, cysteinyl leukotriene C4, nitric oxides,
lipid-derived signaling molecules and antioxidants (11,12).
Overexpression of MRP1 is a common phenomenon in various cancer
tissues, reducing cytotoxicity and the efficacy of antineoplastic
agents by boosting the efflux of the drugs, including cisplatin,
vinorelbine and gemcitabine, resulting in shorter tumor-free
survival and overall survival (OS) times in patients with NSCLC
(9,13,14).
Although MRP1 has been known about and studied for
more than two decades, and found to be consistently overexpressed
in the majority of patients with NSCLC, the clinical relevance of
MRP1 expression in these patients remains inconclusive (15). This is not unexpected, as it has
been a challenge to define precisely the relevance of MRP1 in
clinical drug resistance in patients with cancer (12). As aforementioned, MRP1 is an ABC
membrane transport protein implicated in clinical drug resistance,
and is capable of actively decreasing the intracellular drug
concentration in the cells. Thus, MRP1 expression may affect the
clinical outcome of chemotherapy for NSCLC. Indeed, certain studies
showed that MRP1 expression was a significant indicator of a poor
response to chemotherapy and poor OS in NSCLC (16). However, another study showed that
the expression of MRP1 had no correlation with OS or response to
chemotherapy in NSCLC, and no significant correlations between MRP1
expression and the clinicopathological parameters of NSCLC were
found (15). Therefore there is a
requirement to clarify the clinical relevance of MRP1 in NSCLC
patients.
The present meta-analysis aimed to systemically
investigate the epidemiological and clinicopathological
implications associated with increased MRP1 gene in patients
suffering from NSCLC by pooling all available published data.
Findings from this study will advance our current understanding of
the function of MRP1, and in particular, provide novel insight into
the clinical significance of the MRP1 gene in NSCLC.
Two independent researchers screened all the
identified articles and selected those that met the following
inclusion criteria: Clinical studies involving patients with a
confirmed diagnosis of NSCLC and with lung tissue specimens tested
for MRP1 by either immunohistochemistry or reverse
transcription-polymerase chain reaction (RT-PCR). Moreover, the
numbers of MRP1(+) patients against the total number of patients
were recorded. The following categories of articles were excluded
from the meta-analysis: i) Studies of non-NSCLC carcinomas; ii)
lung tissue specimens not tested or analyzed by
immunohistochemistry or RT-PCR; iii) animal studies, cell line
experiments, case reports, meta-analyses and systematic reviews;
and iv) duplicate studies. When overlapping studies were published
by the same author(s), the most informative or the most updated
studies were selected. Disputes of selection were resolved
following discussion with the senior investigators.
The Newcastle-Ottawa Scale (NOS) was adopted to
assess the methodology quality of the 36 studies included in this
meta-analysis (18). Since standard
criteria for the NOS have not been established, the star system of
the NOS (range, 0 to 9 stars) was used for all assessments, with a
score of 7 or more stars considered as high quality.
All statistical analyses were performed using the
Stata software, version 12.0 (StataCorp LP, College Station, TX,
USA). The pooled odds ratios (ORs) with 95% confidence intervals
(CIs) were calculated to assess the strength of the association
between increased MRP1 gene expression and the clinicopathological
features of the patients with NSCLC. The significance of the pooled
ORs was determined by the Z-test, with P<0.05 considered as
statistically significant. The degree of heterogeneity between
studies was assessed using Cochran's Q-statistic and the
I2 test (variation in OR attribuTable to heterogeneity).
An I2 value of 0% indicated no observed heterogeneity,
with larger I2 values indicating increased
heterogeneity; P<0.10 was regarded as indicating a statistically
significant difference. When statistical heterogeneity existed, the
random-effects model was conducted, otherwise the fixed-effects
model was applied (19).
To test the robustness of the results in this
meta-analysis, a sensitivity analysis was performed using the
one-at-a-time method, which omits one study at a time with repeat
meta-analysis to reveal the influence of the individual data sets
to the pooled ORs.
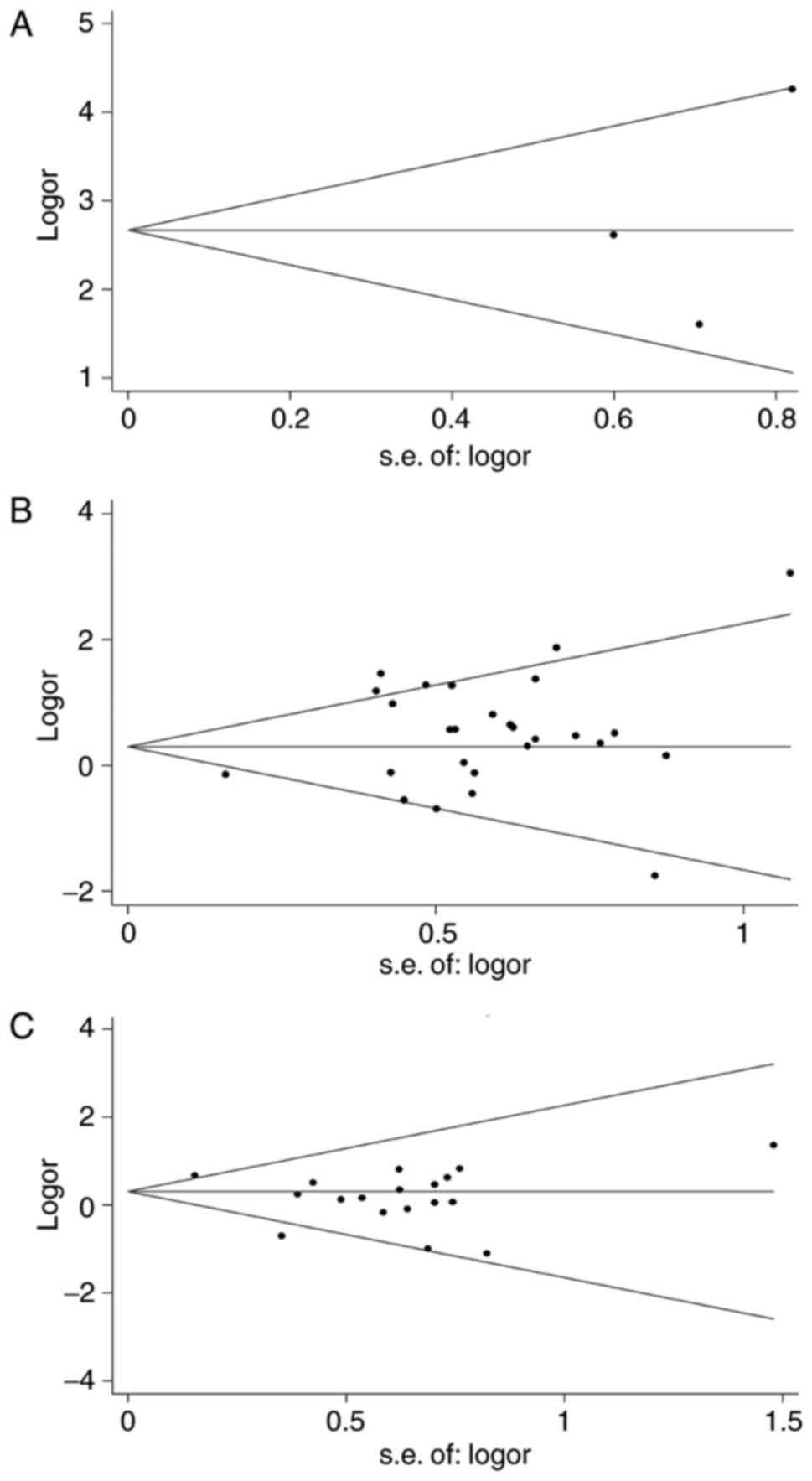
Publication bias was estimated with visual and
statistically significant asymmetry using the funnel plot. Begg's
funnel plot and Egger's test were employed to assess the potential
publication bias in all studies. Publication bias was considered to
be present when Begg's funnel plot was asymmetric or when P<0.05
using Egger's test.
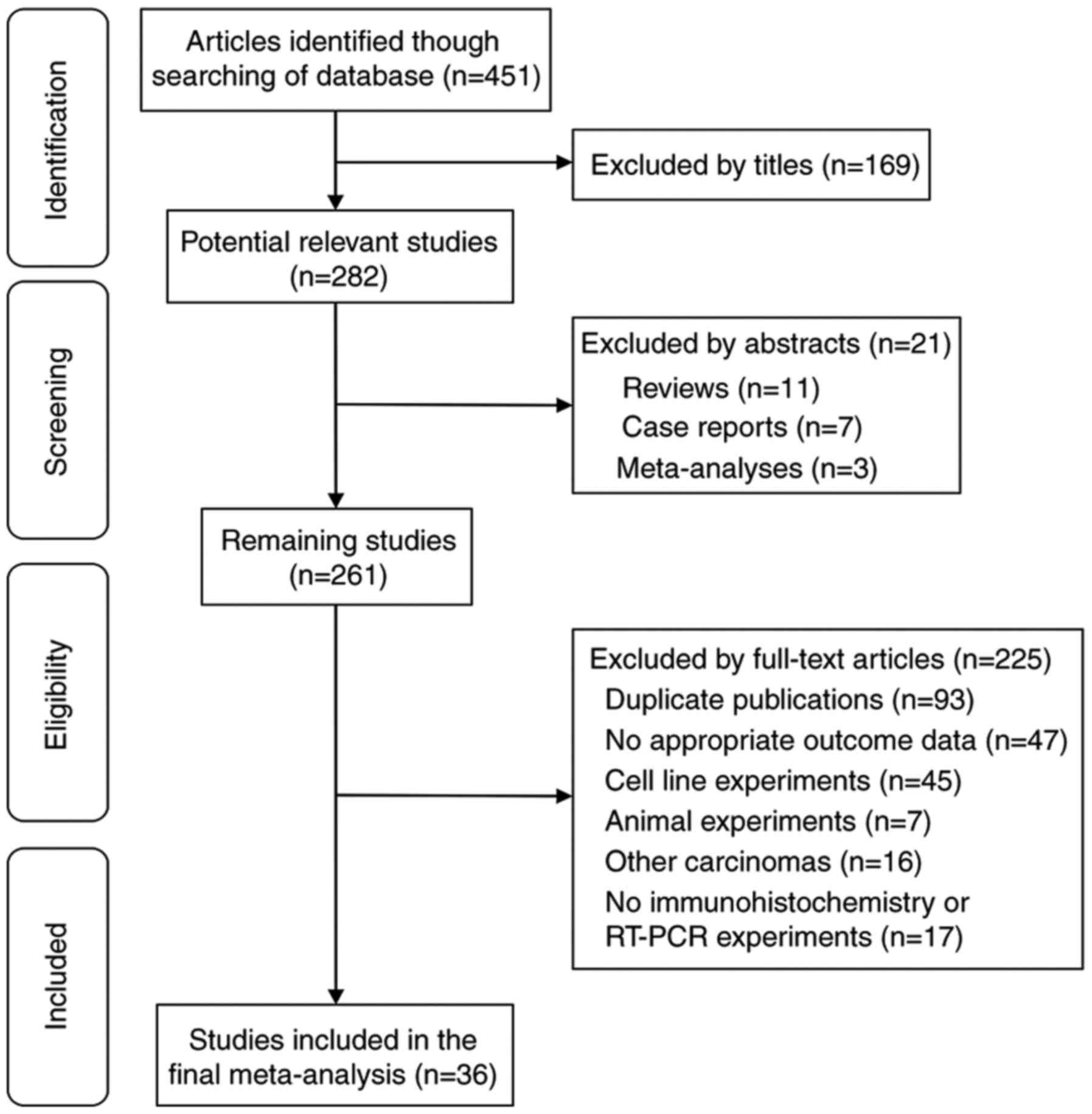
A total of 36 clinical studies, involving 3,278
patients, were included in the present meta-analysis, with 2,259
(68.9%) of the total cohort being Asian (20–55).
Fig. 1 outlines the results of the
selection criteria and the search strategies. Table I summarizes the baseline
characteristics of the included studies. Table II highlights the quality of
publications included in the study.
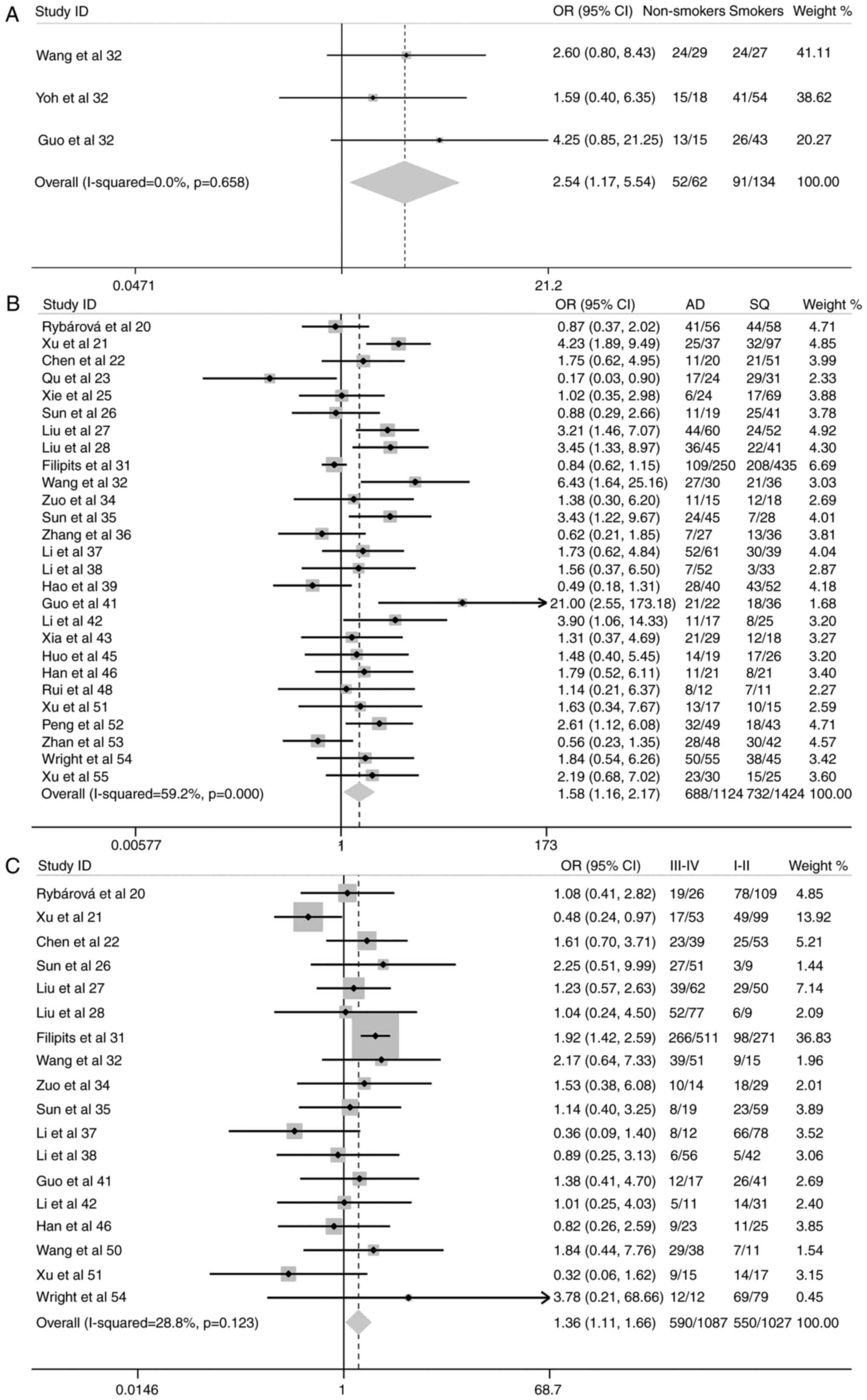
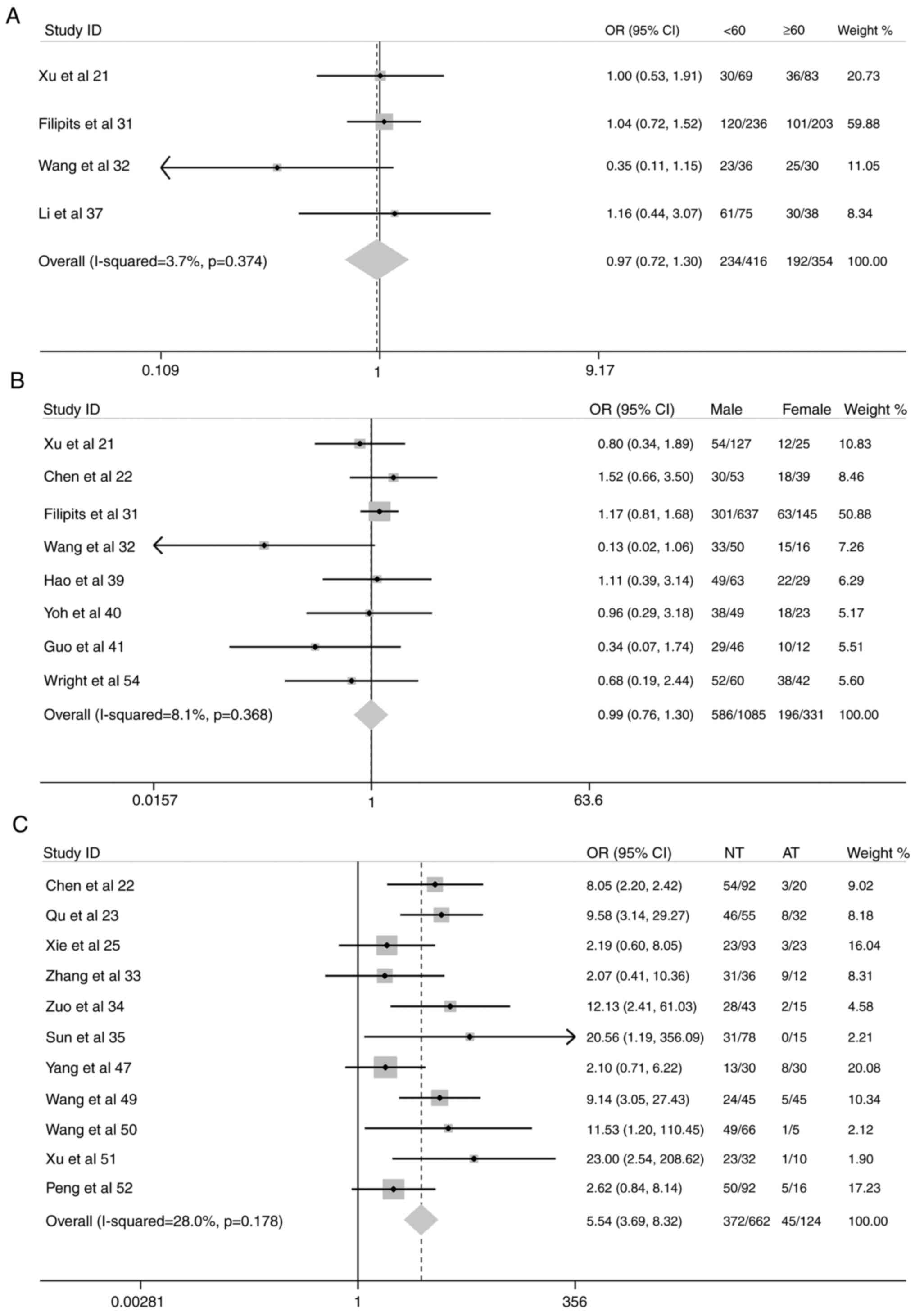
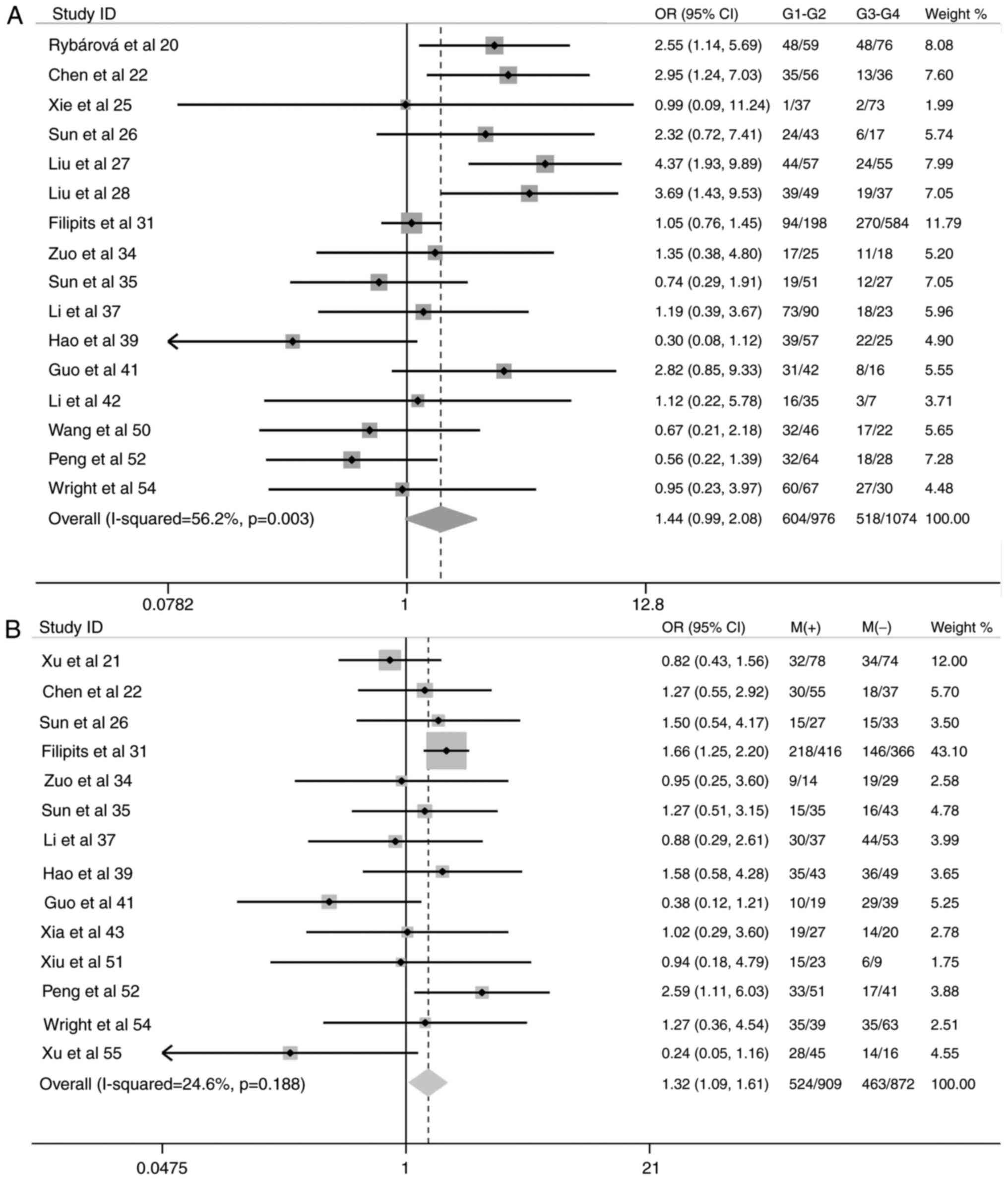
Increased expression of the MRP1 gene was also
associated with the following subgroups: NSCLC tissue vs. normal
pulmonary tissue adjacent to NSCLC (OR, 5.54; 95% CI, 3.69–8.32;
P<0.001) (Fig. 3); increased
tendency of lymph node metastases (OR, 1.32; 95% CI, 1.09–1.61;
P=0.005) (Fig. 4); compromised
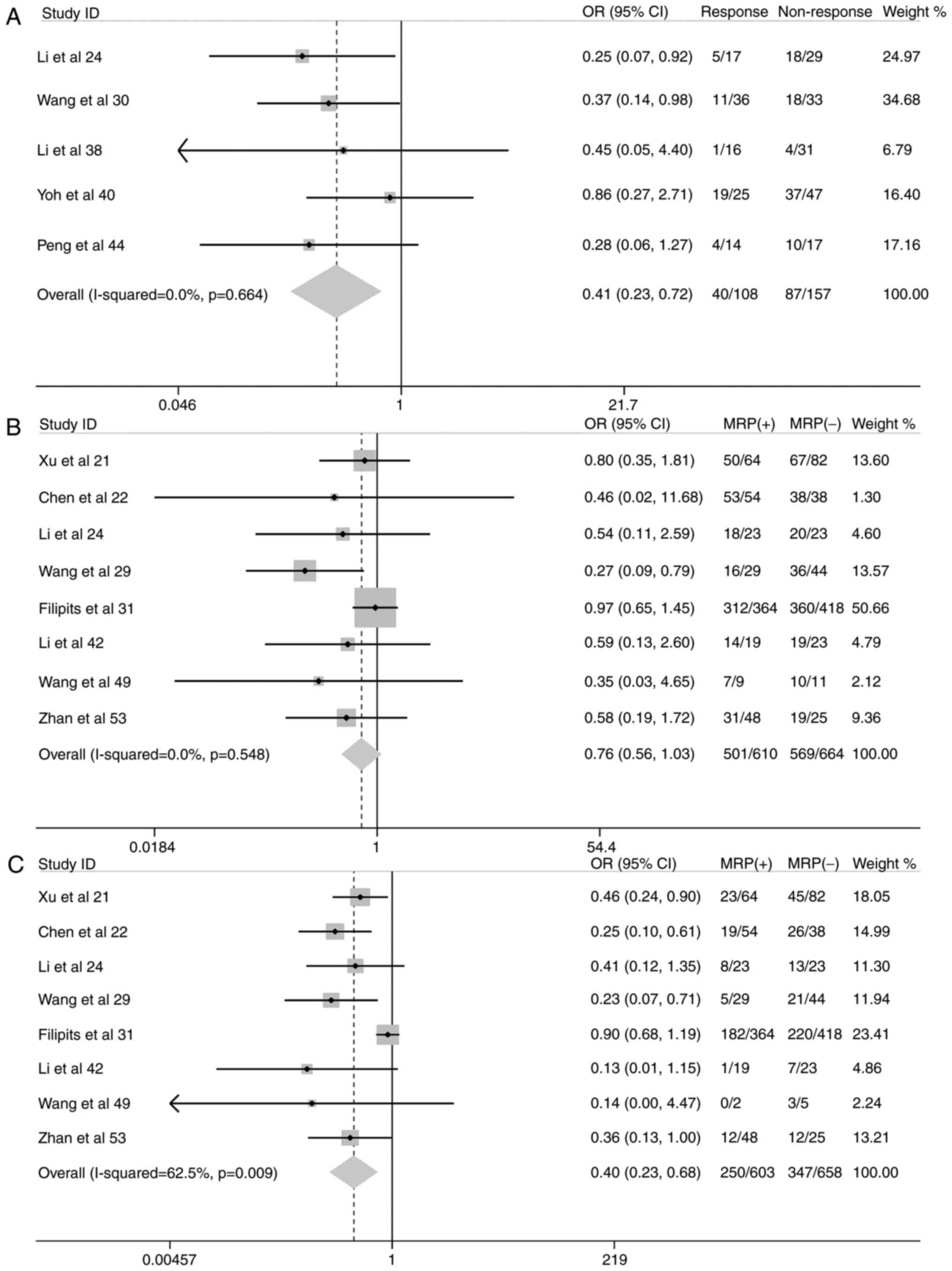
response to chemotherapy (OR, 0.41; 95% CI, 0.23–0.72; P=0.002);
and decreased 3-year survival rate (OR, 0.40; 95% CI, 0.23–0.68;
P=0.001) (Fig. 5).
There were no significant differences in MRP1 gene
expression in the following subgroups: Male vs. female, patients
≥60 years old vs. <60 years old, NSCLC cohorts with cytological
grades 1 and 2 vs. cytological grades 3 and 4, and 1-year survival
rate (Figs. 3–5).
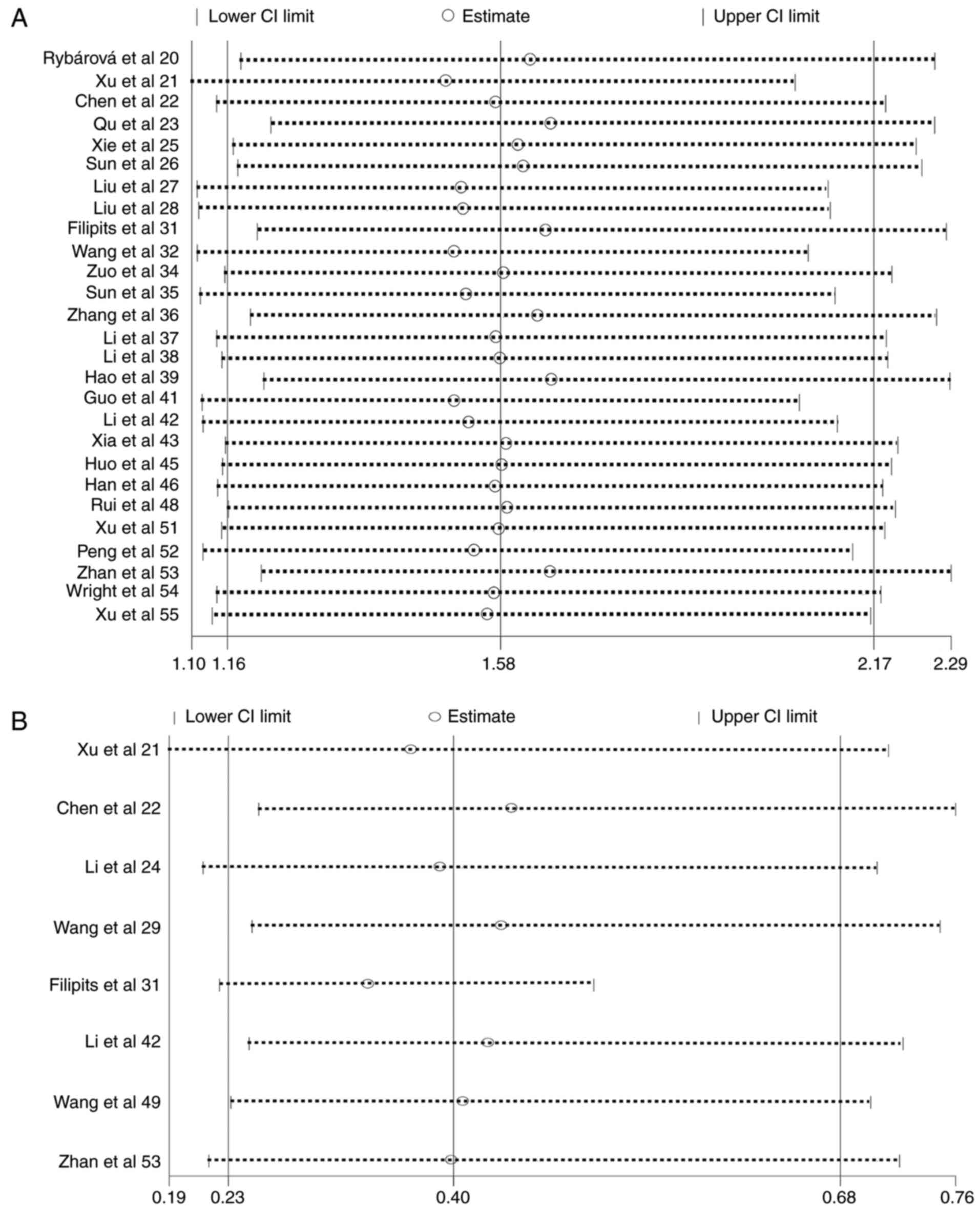
Since the heterogeneity among studies included in
the analysis of the associations of MRP1 expression with
pathological type and 3-year survival rate were significantly high
(I2 value of 59.2 and 62.5%, respectively; Table III), a sensitivity analysis was
conducted to assess the stability of the results. The analysis
showed that the significance of the results was not affected by any
single study, which is visually depicted in Fig. 6, indicating the statistical
robustness of the results.
The present meta-analysis of 36 clinical studies of
3,278 patients with NSCLC suggested that increased expression of
the MRP1 gene in NSCLC patients is significantly associated with
the following specific subgroups: Non-smokers, adenocarcinoma and
advanced clinical stage. Increased MRP1 gene expression is also
significantly associated with decreased 3-year survival rate
(P=0.001) and a compromised response to chemotherapy (P=0.002).
These findings highlight the updated clinical implications of MRP1
gene expression in patients with NSCLC. To the best our knowledge,
this is by far the most extensive study summarizing current
evidence on the clinicopathological relevance of MRP1 in NSCLC
patients.
The present study showed that non-smoking patients
with NSCLC tend to have increased expression of MRP1 compared with
their smoking counterparts. Apart from the data extracted from the
included studies (32,40,41),
there is currently no other study directly comparing the MRP1
expression in smoking and non-smoking patients with NSCLC. An in
vitro study has demonstrated that cigarette smoke extracts
diminish MRP1 activity in the human bronchial epithelial cells
(56). A previous study by Leslie
et al (57) also suggested
that MRP1-mediated activity of uptake of organic anions can be
inhibited by nicotine glucuronide conjugates. By contrast, another
study claimed that the physiological functions of MRP1 are unlikely
to be substantially decreased by nicotine glucuronide metabolites
at concentrations achievable in human serum (58). More convincing data derived from
studies based on patients with chronic obstructive pulmonary
disease (COPD), a condition strongly associated with smoking
history, found that diminished MRP1 expression was observed in the
bronchial epithelium and lung tissues in the COPD group (59,60).
It was concluded that MRP1 appeared to be a protective protein for
COPD development. The exact role of increased MRP1 expression in
non-smoking NSCLC patients is unclear and requires further
study.
In agreement with other previous studies,
significantly enhanced MRP1 expression in lung cancer tissue of
NSCLC patients was observed (OR, 5.54; 95% CI, 3.69–8.32;
P<0.0001), in comparison to the non-tumor pulmonary tissue
adjacent to the tumor, in the present study. The published studies
have overwhelmingly shown that MRP1 is highly expressed and
functionally active in NSCLC cells, and is potentially associated
with negative treatment outcomes in NSCLC patients (12–14,22,24,36,49,52).
It is unclear how MRP1 expression is regulated in NSCLC (61) and in other tissues (12). However, increased expression of MRP1
in NSCLC tissues could be explained in two ways. Firstly, it maybe
a result of p53 mutant expression in NSCLC, as mutant p53-positive
NSCLC in patients has shown a significant correlation with MRP1
overexpression (61,62). Secondly, it is possible that MRP1
could be induced by cancer chemotherapy or radiotherapy, as
evidence has clearly shown that the mRNA levels of MRP1 in
recurrent tumors and residual tumors following chemotherapeutic
treatment are higher than those in untreated primary tumors
(63,64). These explanations can also be
applied to other findings from the current study, which showed an
increase in MRP1 gene expression was associated with patients in
advanced clinical stages (stage III–IV) and with lymph node
metastases. It should be noted that these findings contradict the
results from one study by Berger et al (64), which stated that MRP1 expression
levels were highest in stage I and declined with advanced stage.
Although this could be associated with ethnicity, the discrepancy
is mainly due to the differences in sample size. The present
results were derived from 19 studies involving 2,114 patients,
while the sample size in the study by Berger et al was
limited to 126 patients. Therefore the results presented in this
report are much more extensive and show better representation.
MRP1(+) patients with NSCLC were found to have a
compromised response to platinum-based chemotherapy and decreased
3-year survival rates when compared with their MRP1(−)
counterparts. A clear trend was observed linking MRP1 expression to
decreased 1-year survival, although this was not statistically
significant (P=0.07). It has been demonstrated that MRP1 is highly
expressed in patients with NSCLC and associated with a defect in
platinum accumulation in cisplatin-resistant cell lines (65,66).
Triller et al (67) observed
a significant negative correlation between MRP1 expression and a
more favorable response rate to chemotherapy in patients with
NSCLC. Similarly, it is worth noting that increased expression of
ABCG4 and transforming growth factor β receptor type 2 are also
identified as novel poor prognostic factors of chemotherapy in
NSCLC patients (68,69). It is possible that an increase in
MRP1-mediated efflux of anti-neoplastic agents reduces the
intracellular concentration, and thereby decreases the therapeutic
efficacy of the agents. RNA interference-based knockdown of MRP1
reversed MDR efficiently by decreasing the efflux ability and
increasing the DDP-induced apoptosis in A549/DDP cells (70). These studies provided a solid ground
for the clinical use of the MRP1 inhibitor for the treatment of
NSCLC patients with enhanced expression of MRP1. It is predicted
that the combined use of MRP1 inhibitor(s) with anti-neoplastic
agents would increase the intracellular drug concentration, and
thus possibly increase the curative effect of the agents. Indeed, a
recent study demonstrated that meloxicam, a COX-2 inhibitor,
increased the intracellular accumulation of doxorubicin and
enhanced doxorubicin-induced cytotoxicity in the human lung cancer
A549 cell line via downregulation of MRP1 (71).
The present analysis clearly shows that the MRP1
expression in patients with adenocarcinoma is significantly higher
as opposed to that in patients with squamous cell carcinoma, and
this is consistent with numerous other studies (72–77).
However, it is noted that in a few studies, no significant
differences in MRP1 expression between different histological
subtypes of NSCLC could be detected (64). In the present study, 61.2%
(688/1,124) of patients with adenocarcinoma were MRP1(+), whereas
the presence of epidermal growth factor receptor (EGFR) mutations
ranged from 40.3 to 64.5% in patients with adenocarcinoma (6,76,78–82).
Currently, mutant EGFR is considered as a good predictor of
clinical response to tyrosine kinase inhibitors (TKIs).
Administration of TKIs has been shown to have a superior
therapeutic value to chemotherapy regimens in non-smoking Asian
patients with pulmonary adenocarcinoma harboring a higher rate of
EGFR mutations (76,83). The use of TKIs is indicated in NSCLC
patients harboring EGFR mutations, rather than in those with
increased expression of the MRP1 gene. However, there is certain
evidence suggesting that EGF induces MRP1 gene expression and
increases MRP1 promoter activity (84). TKIs have become promising MRP1
inhibitors. Ibrutinib, a Bruton's TKI, was shown to significantly
increase the efficacy of chemotherapeutic agents in
MRP1-overexpressing cells of leukemia by antagonizing the efflux
function of the MRP1 transporter (9). In addition, interactions of human MRP1
with TKIs, including imatinib and AG1393, have also been reported
to inhibit its transportation activity (9,85).
Although the exact association between EGFR and MRP1 in NSCLC is
uncertain at present, it is tempting to postulate that the combined
use of TKIs and MRP1 inhibitors may have a synchronous effect in
treating NSCLC patients with increased gene expression for
MRP1.
One of the potential limitations of this
meta-analysis is its potential risk of sampling bias: 34 out of the
36 studies were conducted in Asia, with the majority of the
patients being Chinese. Therefore it is unknown whether a study
with larger samples covering multiple ethnic groups would lead to
the same conclusions. Another potential limitation is the
relatively small sampling size in certain subgroups, including the
MRP1 gene expression and smoking status subgroup, which contained 3
studies and only 196 patients (Fig.
2). Although the results of this meta-analysis are overall
extremely promising, larger-scale clinical studies would provide
more robust evidence-based results.
In conclusion, increased expression of the MRP1 gene
is associated with a non-smoking status, adenocarcinoma, advanced
clinical stages and a poor prognosis to chemotherapy in patients
with NSCLC, indicating that the MRP1 gene serves a significant role
in the development of NSCLC. The present results suggest further
research on the implications of MRP1 expression in NSCLC is
required to validate the important clinical significance of MRP1
expression that may influence the treatment of NSCLC. In
particular, the fact that enhanced MRP1 expression strongly
associates with a poor prognosis and advanced clinical stages of
NSCLC provides a compelling foundation to continue investigating
the potential use of MRP1 as a biomarker/clinical indicator for
NSCLC.
The authors would like to thank Dr Eileen McGowan of
University of Technology Sydney (Sydney, New South Wales,
Australia) for providing critical reading of the manuscript.
The study was supported by a research grant from the
National Natural Science Foundation of China (no. 81273814) and by
a Guangdong Provincial Key Scientific Research Grant (no.
2013A02210031).
All data generated or analyzed during this study are
included in this published article.
YL, MX, PH and PTYW conceived and designed the
study. PH and PTYW executed the study. YL, MX, PH and PTYW analyzed
and interpreted the data, and drafted and edited the manuscript.
QZ, LH, WN and SC analyzed and interpreted data, and revised and
edited the manuscript. All authors read and approved the final
manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Molina JR, Yang P, Cassivi SD, Schild SE
and Adjei AA: Non-small cell lung cancer: Epidemiology, risk
factors, treatment, and survivorship. Mayo Clin Proc. 83:584–594.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Meza R, Meernik C, Jeon J and Cote ML:
Lung cancer incidence trends by gender, race and histology in the
United States, 1973–2010. PLoS One. 10:e01213232015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guan WJ, Zheng XY, Chung KF and Zhong NS:
Impact of air pollution on the burden of chronic respiratory
diseases in China: Time for urgent action. Lancet. 388:1939–1951.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou C: Lung cancer molecular epidemiology
in China: Recent trends. Transl Lung Cancer Res. 3:270–279.
2014.PubMed/NCBI
|
|
7
|
Stewart B and Wild CP: World cancer report
2014. Health (NY); 2017
|
|
8
|
Cole SP, Bhardwaj G, Gerlach JH, Mackie
JE, Grant CE, Almquist KC, Stewart AJ, Kurz EU, Duncan AM and
Deeley RG: Overexpression of a transporter gene in a
multidrug-resistant human lung cancer cell line. Science.
258:1650–1654. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang H, Patel A, Ma SL, Li XJ, Zhang YK,
Yang PQ, Kathawala RJ, Wang YJ, Anreddy N, Fu LW and Chen ZS: In
vitro, in vivo and ex vivo characterization of ibrutinib: A potent
inhibitor of the efflux function of the transporter MRP1. Br J
Pharmacol. 171:5845–5857. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Flens MJ, Zaman GJ, van der Valk P,
Izquierdo MA, Schroeijers AB, Scheffer GL, van der Groep P, de Haas
M, Meijer CJ and Scheper RJ: Tissue distribution of the multidrug
resistance protein. Am J Pathol. 148:1237–1247. 1996.PubMed/NCBI
|
|
11
|
Lok HC, Rahmanto Suryo Y, Hawkins CL,
Kalinowski DS, Morrow CS, Townsend AJ, Ponka P and Richardson DR:
Nitric oxide storage and transport in cells are mediated by
glutathione S-transferase P1-1 and multidrug resistance protein 1
via dinitrosyl iron complexes. J Biol Chem. 287:607–618. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cole SP: Targeting multidrug resistance
protein 1 (MRP1, ABCC1): Past, present, and future. Annu Rev
Pharmacol Toxicol. 54:95–117. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Keppler D: Multidrug resistance proteins
(MRPs, ABCCs): Importance for pathophysiology and drug therapy.
Handb Exp Pharmacol. 299–323. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li XQ, Li J, Shi SB, Chen P, Yu LC and Bao
QL: Expression of MRP1, BCRP, LRP and ERCC1 as prognostic factors
in non-small cell lung cancer patients receiving postoperative
cisplatin-based chemotherapy. Int J Biol Markers. 24:230–237. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lu JF, Pokharel D and Bebawy M: MRP1 and
its role in anticancer drug resistance. Drug Metab Rev. 47:406–419.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Munoz M, Henderson M, Haber M and Norris
M: Role of the MRP1/ABCC1 multidrug transporter protein in cancer.
IUBMB Life. 59:752–757. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Moher D, Liberati A, Tetzlaff J and Altman
DG: PRISMA Group: Preferred reporting items for systematic reviews
and meta-analyses: The PRISMA statement. Int J Surg. 8:336–341.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Myung SK, Ju W, McDonnell DD, Lee YJ,
Kazinets G, Cheng CT and Moskowitz JM: Mobile phone use and risk of
tumors: A meta-analysis. J Clin Oncol. 27:5565–5572. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sutton AJ, Song F, Gilbody SM and Abrams
KR: Modelling publication bias in meta-analysis: A review. Stat
Methods Med Res. 9:421–445. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rybárová S, Hodorová I, Mihalik J and
Mirossay L: MRP1 and GSTp1 expression in non-small cell lung cancer
does not correlate with clinicopathological parameters: A Slovakian
population study. Acta Histochem. 116:1390–1398. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu Y, Wang L, Zheng X, Liu G, Wang Y, Lai
X and Li J: Positive expression of p53, c-erbB2 and MRP proteins is
correlated with survival rates of NSCLC patients. Mol Clin Oncol.
1:487–492. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen ZJ, Le HB, Zhang YK, Qian LY, Sekhar
KR and Li WD: Lung resistance protein and multidrug resistance
protein in non-small cell lung cancer and their clinical
significance. J Int Med Res. 39:1693–1700. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qu Z, Jiang J, Qu J, Tian R and Zhao N: A
study on the expression and biological significance of PTEN and MRP
in NSCLC tissues. Chin J Geriatrics. 31:22011.
|
|
24
|
Li J, Li ZN, Yu LC, Bao QL, Wu JR, Shi SB
and Li XQ: Association of expression of MRP1, BCRP, LRP and ERCC1
with outcome of patients with locally advanced non-small cell lung
cancer who received neoadjuvant chemotherapy. Lung Cancer.
69:116–122. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xie H, Yu X, Ke M and Jiang Y: A study on
the expression of multidrug resistance-associated protein genes in
lung cancer tissues and other malignant tumor. Pract J Cancer.
25:150–153. 2010.
|
|
26
|
Sun C, Shi H and Yuan D: Study on the
correlation between expression of p53/MDR and drug resistance in
non-small cell lung cancer. Hainan Med J. 21:45–48. 2010.
|
|
27
|
Liu X, Zhang S, Xu S, Wei H and Liu Y:
Expression and clinical significance of LRP and MRP in non-small
cell lung cancer tissues by bronchoscopy biopsy. Chin J Lung
Cancer. 12:571–573. 2009.
|
|
28
|
Liu Y, Zhang S, Xing Q, Yang Y, Wei H and
Xu S: Expression and clinical significance of P-gp, LRP, MRP and
GST-π in non-small cell lung cancer tissues. J Modern Oncol.
17:1444–1447. 2009.
|
|
29
|
Wang X, Jiang H, Zhong H and Yu X:
Relationship between prognosis and expression of MRP and pS3 in
non-small cell lung cancer alter neoadjuvant chemotherapy. J Clin
Pulm Med. 13:150–153. 2008.
|
|
30
|
Wang X, Jiang H, Zhong H and Yu X:
Pathological characteristics and expression of MRP and p53 in
non-small cell lung cancer after neoadjuvant chemotherapy. Clin Med
(Northfield Il). 28:109–111. 2008.
|
|
31
|
Filipits M, Haddad V, Schmid K, Huynh A,
Dunant A, André F, Brambilla E, Stahel R, Pignon JP, Soria JC, et
al: Multidrug resistance proteins do not predict benefit of
adjuvant chemotherapy in patients with completely resected
non-small cell lung cancer: International Adjuvant Lung Cancer
Trial Biologic Program. Clin Cancer Res. 13:3892–3898. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang H, Wang Q, Fan Q, et al: Expression
of multidrug resistance proteins P-glycoprotein, multidrug
resistance associated protein 1 and breast cancer resistance
protein in non-small cell lung cancer:correlation with pro gnosis.
Central Plains Med J. 34:1–5. 2007.
|
|
33
|
Zhang Y, Wang C, Sun J and Song G:
Expressions and significance of MRP1 mRNA and GST-π mRNA in NSCLC
tissues. Chin J Cancer Prev Treat. 14:913–917. 2007.
|
|
34
|
Zuo Y, Huang J, Mu C and Shen D: The
expression and significance of the multidrug resistance-associated
genes MDR1, MRP1, LRP mRNA and their proteins P-gp, MRP, LRP in
human non-small cell lung cancer tissues. Chin Clin Oncol.
11:921–929. 2006.
|
|
35
|
Sun Z, Yu H, Liu H, Li Y, He J and Sun J:
Expression of P-glycoprotein, multidrug resistance-associated
protein, lung resistance-related protein, P53 and cerbB-2 in
non-small cell lung cancer. Acad J Sec Mil Med Univ. 27:474–478.
2006.
|
|
36
|
Zhang L, Yang H and Dong D: Study on the
relationship among multidrug resistance factor expression of lung
cancer tissues and clinicopathological characteristics in patients
with lung cancer. Chin J Lung Cancer. 9:60–64. 2006.
|
|
37
|
Li L, Xiong YY, Liu L, Chen TX, Yao XF and
Wang YW: Relarionships among expressions of hTERT, MDR1, MRPmRNA,
and C-myc protein in non-small cell lung cancer. Ai Zheng.
24:53–57. 2005.(In Chinese). PubMed/NCBI
|
|
38
|
Li C, Hou M, Zhao Y and Zhou Q: Expression
of multidrug resistance-associated proteins in non-small cell lung
cancer tissues and its clinical significance. Chin J Lung Cancer.
8:523–527. 2005.
|
|
39
|
Hao J, Wang H, Wang E, Qiu X, Li Q and Liu
Y: Expression of multi-drug resistance-associated protein (MRP) and
its relationship with clinicopathological factors in non-small cell
lung cancer. Chin J Cancer Res. 16:34–39. 2004. View Article : Google Scholar
|
|
40
|
Yoh K, Ishii G, Yokose T, Minegishi Y,
Tsuta K, Goto K, Nishiwaki Y, Kodama T, Suga M and Ochiai A: Breast
cancer resistance protein impacts clinical outcome in
platinum-based chemotherapy for advanced non-small cell lung
cancer. Clin Cancer Res. 10:1691–1697. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Guo Y and Wang Y, Wang X, Yang H and Wang
Y: Expression and clinical significance of resistance-related genes
in non-small cell lung cancer tissue. Chin J Clin Oncol.
31:391–396. 2004.
|
|
42
|
Li Q, Lu X, Hu J, Jin XL and Zhao X:
Expression and clinical significance of multi-drug resistance
associated protein and P-glycoprotein in non-small cell lung
cancer. Clinical Focus. 19:906–909. 2004.
|
|
43
|
Xia S, Yu S, Yuan X and Fu X: The relative
study on the expression of HIF-1a, P-gp, and MRP in non-small cell
lung cancer. Chin J Exp Surg. 21:1515–1518. 2004.
|
|
44
|
Peng ZM, Luo J, Wang WB, Wang XH, Chen JH
and Lan SM: Predictive value of drug resistance-related genes
expression in neoadjuvant chemotherapy in patients with non-small
cell lung cancer of stage III. Ai Zheng. 23:963–967. 2004.(In
Chinese). PubMed/NCBI
|
|
45
|
Huo J, Che C and Huang Q: Coexpression and
clinical significance of multi-drug resistance factors in lung
cancer. Zhongguo Fei Ai Za Zhi. 7:218–221. 2004.(In Chinese).
PubMed/NCBI
|
|
46
|
Han B, Liao M, Su J, Feng J, Wang E and
Dong Q: The relationship between drug sensitivity and expression of
drug resistance gene mutations in non-small cell lung cancer.
Zhonghua Jie He He Hu Xi Za Zhi. 25:727–731. 2002.(In Chinese).
PubMed/NCBI
|
|
47
|
Yang J, Dai W, Shi T and Wei X: Expression
of MDR1-mRNA, MRP-mRNA and LRP-mRNA in patients with non-small cell
lung cancer. Zhongguo Fei Ai Za Zhi. 4:175–177. 2001.(In Chinese).
PubMed/NCBI
|
|
48
|
Rui M, Li L, Liu L, Wang J and Zhang Z:
Expression of P-gp, MRP and p53 and their clinical significance in
human lung cancer tissues. Zhongguo Fei Ai Za Zhi. 4:58–62.
2001.(In Chinese). PubMed/NCBI
|
|
49
|
Wang Y, Wang A, Li X, Zhang Y and Wang W:
Expression of MDR-1 and MRP gene in non-small cell lung caner and
their relations to prognosis of the patients. Chin Clin Oncol.
6:303–308. 2001.
|
|
50
|
Wang J, Liu S and Jiang W: Expression of
LRP, MRP and MDR1 in non-small-cell lung cancer and its clinical
significance. Zhonghua Zhong Liu Za Zhi. 22:304–307. 2000.(In
Chinese). PubMed/NCBI
|
|
51
|
Xu M, Li J and Xia Q: Expression of
multidrug resistance-associated protein gene in non-small cell lung
cancer. Zhonghua Jie He He Hu Xi Za Zhi. 22:268–270. 1999.(In
Chinese). PubMed/NCBI
|
|
52
|
Peng X, Feng F, Zhang W, Wu B, Chang S and
Jin S: Expression of multidrug resistance-associated protein in
human non-small cell lung cancer. Chin J Tuberc Respir Dis.
22:655–659. 1999.
|
|
53
|
Zhan M, X L, Li J and Jiang W: Clinical
significance of detecting MRP in 110 patients with lung cancer.
Chin J Clin Oncol. 26:732–735. 1999.
|
|
54
|
Wright SR, Boag AH, Valdimarsson G,
Hipfner DR, Campling BG, Cole SP and Deeley RG: Immunohistochemical
detection of multidrug resistance protein in human lung cancer and
normal lung. Clin Cancer Res. 4:2279–2289. 1998.PubMed/NCBI
|
|
55
|
Xu M, Li J and Xie J: Expression and
clinical importance of multidrug resistance-associated protein gene
in non-small cell lung cancer. China J Mod Med. 8:4–8. 1998.
|
|
56
|
van der Deen M, de Vries EG, Visserman H,
Zandbergen W, Postma DS, Timens W and Timmer-Bosscha H: Cigarette
smoke extract affects functional activity of MRP1 in bronchial
epithelial cells. J Biochem Mol Toxicol. 21:243–251. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Leslie EM, Ito K, Upadhyaya P, Hecht SS,
Deeley RG and Cole SP: Transport of the beta -O-glucuronide
conjugate of the tobacco-specific carcinogen
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) by the
multidrug resistance protein 1 (MRP1). Requirement for glutathione
or a non-sulfur-containing analog. J Biol Chem. 276:27846–27854.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Létourneau IJ, Bowers RJ, Deeley RG and
Cole SP: Limited modulation of the transport activity of the human
multidrug resistance proteins MRP1, MRP2 and MRP3 by nicotine
glucuronide metabolites. Toxicol Lett. 157:9–19. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
van der Deen M, Marks H, Willemse BW,
Postma DS, Müller M, Smit EF, Scheffer GL, Scheper RJ, de Vries EG
and Timens W: Diminished expression of multidrug
resistance-associated protein 1 (MRP1) in bronchial epithelium of
COPD patients. Virchows Arch. 449:682–688. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Budulac SE, Postma DS, Hiemstra PS, Kunz
LI, Siedlinski M, Smit HA, Vonk JM, Rutgers B, Timens W and Boezen
HM: Groningen Leiden Universities Corticosteroids in Obstructive
Lung Disease (GLUCOLD) study group: Multidrug resistance-associated
protein-1 (MRP1) genetic variants, MRP1 protein levels and severity
of COPD. Respir Res. 11:602010. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Oshika Y, Nakamura M, Tokunaga T,
Fukushima Y, Abe Y, Ozeki Y, Yamazaki H, Tamaoki N and Ueyama Y:
Multidrug resistance-associated protein and mutant p53 protein
expression in non-small cell lung cancer. Mod Pathol. 11:1059–1063.
1998.PubMed/NCBI
|
|
62
|
Zhou CZ, Li Y and Xu J: Correlation
between p53 gene mutation and the expression of tumor drug
resistance genes in lung cancer and its clinical significance.
Zhonghua Jie He He Hu Xi Za Zhi. 27:678–682. 2004.(In Chinese).
PubMed/NCBI
|
|
63
|
Tada Y, Wada M, Migita T, Nagayama J,
Hinoshita E, Mochida Y, Maehara Y, Tsuneyoshi M, Kuwano M and Naito
S: Increased expression of multidrug resistance-associated proteins
in bladder cancer during clinical course and drug resistance to
doxorubicin. Int J Cancer. 98:630–635. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Berger W, Setinek U, Hollaus P, Zidek T,
Steiner E, Elbling L, Cantonati H, Attems J, Gsur A and Micksche M:
Multidrug resistance markers P-glycoprotein, multidrug resistance
protein 1, and lung resistance protein in non-small cell lung
cancer: Prognostic implications. J Cancer Res Clin Oncol.
131:355–363. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Liang XJ, Shen DW, Garfield S and
Gottesman MM: Mislocalization of membrane proteins associated with
multidrug resistance in cisplatin-resistant cancer cell lines.
Cancer Res. 63:5909–5916. 2003.PubMed/NCBI
|
|
66
|
Young LC, Campling BG, Cole SP, Deeley RG
and Gerlach JH: Multidrug resistance proteins MRP3, MRP1, and MRP2
in lung cancer: Correlation of protein levels with drug response
and messenger RNA levels. Clin Cancer Res. 7:1798–1804.
2001.PubMed/NCBI
|
|
67
|
Triller N, Korosec P, Kern I, Kosnik M and
Debeljak A: Multidrug resistance in small cell lung cancer:
Expression of P-glycoprotein, multidrug resistance protein 1 and
lung resistance protein in chemo-naive patients and in relapsed
disease. Lung Cancer. 54:235–240. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yang G, Wang XJ, Huang LJ, Zhou YA, Tian
F, Zhao JB, Chen P, Liu BY, Wen MM, Li XF, et al: High ABCG4
expression is associated with poor prognosis in non-small-cell lung
cancer patients treated with cisplatin-based chemotherapy. PLoS
One. 10:e01355762015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Han Y, Jia C, Cong X, Yu F, Cai H, Fang S,
Cai L, Yang H, Sun Y, Li D, et al: Increased expression of
TGFβR2 is associated with the clinical outcome of non-small
cell lung cancer patients treated with chemotherapy. PLoS One.
10:e01346822015. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Shao SL, Cui TT, Zhao W, Zhang WW, Xie ZL,
Wang CH, Jia HS and Liu Q: RNAi-based Knockdown of multidrug
resistance-associated protein 1 is sufficient to reverse multidrug
resistance of human lung cells. Asian Pac J Cancer Prev.
15:10597–10601. 2015. View Article : Google Scholar
|
|
71
|
Chen SF, Zhang ZY and Zhang JL: Meloxicam
increases intracellular accumulation of doxorubicin via
downregulation of multidrug resistance-associated protein 1 (MRP1)
in A549 cells. Genet Mol Res. 14:14548–14560. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Boch C, Kollmeier J, Roth A,
Stephan-Falkenau S, Misch D, Grüning W, Bauer TT and Mairinger T:
The frequency of EGFR and KRAS mutations in non-small cell lung
cancer (NSCLC): Routine screening data for central Europe from a
cohort study. BMJ Open. 3:e0025602013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Inoue A, Suzuki T, Fukuhara T, Maemondo M,
Kimura Y, Morikawa N, Watanabe H, Saijo Y and Nukiwa T: Prospective
phase II study of gefitinib for chemotherapy-naive patients with
advanced non-small-cell lung cancer with epidermal growth factor
receptor gene mutations. J Clin Oncol. 24:3340–3346. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Sequist LV, Martins RG, Spigel D, Grunberg
SM, Spira A, Jänne PA, Joshi VA, McCollum D, Evans TL, Muzikansky
A, et al: First-line gefitinib in patients with advanced
non-small-cell lung cancer harboring somatic EGFR mutations.
J Clin Oncol. 26:2442–2449. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Tamura K, Okamoto I, Kashii T, Negoro S,
Hirashima T, Kudoh S, Ichinose Y, Ebi N, Shibata K, Nishimura T, et
al: Multicentre prospective phase II trial of gefitinib for
advanced non-small cell lung cancer with epidermal growth factor
receptor mutations: Results of the West Japan Thoracic Oncology
Group trial (WJTOG0403). Br J Cancer. 98:907–914. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Mok TS, Wu YL, Thongprasert S, Yang CH,
Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, et
al: Gefitinib or carboplatin-paclitaxel in pulmonary
adenocarcinoma. N Engl J Med. 361:947–957. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Kim DW, Lee SH, Lee JS, Lee MA, Kang JH,
Kim SY, Shin SW, Kim HK and Heo DS: A multicenter phase II study to
evaluate the efficacy and safety of gefitinib as first-line
treatment for Korean patients with advanced pulmonary
adenocarcinoma harboring EGFR mutations. Lung Cancer. 71:65–69.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
An SJ, Chen ZH, Su J, Zhang XC, Zhong WZ,
Yang JJ, Zhou Q, Yang XN, Huang L, Guan JL, et al: Identification
of enriched driver gene alterations in subgroups of non-small cell
lung cancer patients based on histology and smoking status. PLoS
One. 7:e401092012. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Pan Y, Zhang Y, Li Y, Hu H, Wang L, Li H,
Wang R, Ye T, Luo X, Zhang Y, et al: ALK, ROS1 and
RET fusions in 1139 lung adenocarcinomas: A comprehensive
study of common and fusion pattern-specific clinicopathologic,
histologic and cytologic features. Lung Cancer. 84:121–126. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Xia N, An J, Jiang QQ, Li M, Tan J and Hu
CP: Analysis of EGFR, EML4-ALK, KRAS, and c-MET
mutations in Chinese lung adenocarcinoma patients. Exp Lung Res.
39:328–335. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Shi Y, Au JS, Thongprasert S, Srinivasan
S, Tsai CM, Khoa MT, Heeroma K, Itoh Y, Cornelio G and Yang PC: A
prospective, molecular epidemiology study of EGFR mutations in
Asian patients with advanced non-small-cell lung cancer of
adenocarcinoma histology (PIONEER). J Thorac Oncol. 9:154–162.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Wang J, Dong Y, Cai Y, Zhou L, Wu S, Liu
G, Su D, Li X, Qin N, Nong J, et al: Clinicopathologic
characteristics of ALK rearrangements in primary lung
adenocarcinoma with identified EGFR and KRAS status. J Cancer Res
Clin Oncol. 140:453–460. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Yatabe Y: EGFR mutations and the terminal
respiratory unit. Cancer Metastasis Rev. 29:23–36. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Garcia R, Franklin RA and McCubrey JA: EGF
induces cell motility and multi-drug resistance gene expression in
breast cancer cells. Cell Cycle. 5:2820–2826. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Hegedus T, Orfi L, Seprodi A, Váradi A,
Sarkadi B and Kéri G: Interaction of tyrosine kinase inhibitors
with the human multidrug transporter proteins, MDR1 and MRP1.
Biochim Biophys Acta. 1587:318–325. 2002. View Article : Google Scholar : PubMed/NCBI
|