Introduction
Gliomas are the most common tumor type of the adult
central nervous system (1).
Although some progress has been made in recent years, regarding
surgical resection and a number of adjuvant therapies, such as
combined radiotherapy, chemotherapy and targeted therapy, the
treatment of glioblastoma (GBM) is still unsatisfactory due to the
high recurrence rate of glioma in patients and a median survival
time of only 14 months (2–3).
Increasing evidence has revealed that some
tumor-initiating cells with stem cell characteristics in glioma,
also called glioma stem cells (GSCs), contribute to the high rates
of therapeutic resistance and rapid recurrence (4–5). GSCs
have self-renewal properties, high proliferation rates and high
invasive growth characteristics, leading to increased resistance to
radiotherapy and chemotherapy compared with non-stem glioma cells
(NGSCs) (6–8). Consequently, GSCs are one of the main
reasons of GBM treatment failure, and the treatment of GSCs has
become the focus of current malignant glioma therapeutic
strategies.
A previous study has shown that altered circadian
regulation in cancer stem cells may contribute to disease
progression (9). Endogenous
circadian rhythms are maintained by two feedback loops. Clock
circadian regulator (CLOCK) and brain and muscle Arnt-like
protein-1 (BMAL1) form heterodimers to rhythmically activate the
expression of transcriptional repressor proteins, such as period
1/2/3 and cryptochrome 1/2, which form a complex to inhibit CLOCK
and BMAL1 transcriptional activity (9–11). In
some types of cancer and model systems, period circadian clock 2
(PER2) serves as a tumor suppressor gene. Recent systems analysis
revealed that changes in PER2 expression were correlated with lung
cancer, gastric carcinoma, liver cancer, colon cancer, and head and
neck squamous cell carcinomas (12–19).
Our previous study has also revealed that PER1 and PER2 are
significantly downregulated in glioma tissue compared with normal
brain tissue and their expression is correlated with World Health
Organization grading of glioma (20).
However, whether PER2 and PER1 play a critical role
in GSCs and the circadian properties of human GSCs remains unclear.
Based on the findings of previous studies, the present study
investigated the potential role of PER2 on stemness, tumorigenesis,
cell growth, cell cycle distribution, migration and invasion of
GSCs in glioma. The present results revealed a novel mechanism of
action underlying the role of PER2 in GSCs, as well as a new target
to prevent glioma progression.
Materials and methods
Cell culture and patient samples
Normal human astrocytes (cat. no. 1800; http://www.sciencellonline.com/human-astrocytes.html)
were obtained from ScienCell Research Laboratories, Inc., and last
authenticated by the ScienCell in 2018. The human U87 (GBM of
unknown origin) and U251 glioma cells were obtained from The Cell
Bank Type Culture Collection of the Chinese Academy of Sciences and
last authenticated with short tandem repeat (STR) profiling by an
independent third-party company in 2019. All cells were grown in
DMEM (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10%
fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) at 37°C
in a humidified atmosphere of 5% CO2. The glioma
stem-like cell lines U251s and U87s were isolated from U251 and U87
cells using a serum-free clone formation method. Glioma cells were
dispersed and resuspended in DMEM/F12 (Gibco; Thermo Fisher
Scientific, Inc.) serum-free medium (SFM), B27 (20 mg/ml;
Invitrogen; Thermo Fisher Scientific, Inc.), 20 ng/ml basic
fibroblast growth factor (Sigma-Aldrich; Merck KGaA), 20 ng/ml
epidermal growth factor (PeproTech, Inc.), 100 U/ml penicillin and
100 µg/ml streptomycin (Gibco; Thermo Fisher Scientific, Inc.). All
the cells used for analysis were within passage 10, generally in
passages 3–7. To generate a PER2 overexpression model, the U87s and
U251s cells were infected with lentiviruses packaged in
pGMLV-PE1-PER2.
The present study consisted of 48 glioma tissue
specimens, which were resected at the Department of Neurosurgery of
the General Hospital of Ningxia Medical University between January
2017 and July 2019. None of the patients (17 female and 31 male
patients; aged 12 to 73 years old) received preoperative treatment,
such as irradiation or chemotherapy. All patients provided signed
informed consent and the study was approved by the Ethics Committee
of Ningxia Medical University. All research protocols for the
present study were approved by the Ethics Committee of Ningxia
Medical University.
Western blot analysis of protein
expression
Cells were lysed in RIPA lysis buffer (Thermo Fisher
Scientific, Inc.) and then centrifuged at 14,000 × g for 15 min at
4°C. Protein concentrations were determined using the BCA Protein
assay (Gen5 software; Bio-Tek Instruments, Inc.). The supernatant
was collected and denatured. Proteins (30 µg/lane) were separated
via SDS-PAGE on a 10% gel and transferred onto a polyvinylidene
difluoride (PVDF) membrane. The PVDF membrane was treated with TBS
with Tween-20 containing 50 g/l skim milk at room temperature for 4
h, followed by incubation overnight at 4°C with the following
primary antibodies: PER2 (product code ab94915; dilution 1:1,000;
Abcam), CD133 (cat. no. NB120-16518; dilution 1:500; Bio-Techne),
SOX2 (product code ab79351; dilution 1:1,000), NESTIN (product code
ab6320; dilution 1:500), β-catenin (product code ab16051; dilution
1:500), Wnt7b (product code ab94915; dilution 1:200), matrix
metallopeptidase (MMP)2 (product code ab97779; dilution 1:1,000),
MMP9 (product code ab38898; dilution 1:2,000), c-Myc (product code
ab32072; dilution 1:1,000), cyclin D1 (product code ab16663;
dilution 1:200; all from Abcam) and GAPDH (cat. no. 10494-1-AP;
dilution 1:2,000, ProteinTech Group, Inc.). Goat anti-rabbit IgG
H&L IRDye® 800CW (product code ab216773; dilution
1:5,000) and goat anti-mouse IgG H&L IRDye® 680RD
(product code ab216776; dilution 1:5,000; both from Abcam) were
used as secondary antibodies incubated with the membrane for 2 h at
room temperature. The western blot images were visualized on a
LI-COR Odyssey Fc imaging system (LI-COR Biosciences).
RNA isolation and reverse
transcription-quantitative (RT-q)PCR
Total RNA was extracted using TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) in accordance with the
standard protocols. cDNA was reverse transcribed from total RNA
using a PrimeScript RT Master Mix Perfect Real-Time kit (Takara
Bio, Inc.). mRNA expression was analyzed using SYBR Green PCR
Master mix (Takara Biotechnology Co., Ltd.) and a 7300 Real-Time
PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.).
The thermocycling program was as follows: 95°C for 1 min; (95°C for
15 sec; 60°C for 30 sec; 72°C for 30 sec) ×40 cycles. The primer
sequences used were as follows: BMAL1 forward,
5′-CTCCTCCAATGTGGGCATCAA-3′, BMAL1 reverse,
5′-GGTGGCACCTCTTAATGTTTTCA-3′; CLOCK forward,
5′-TGCGAGGAACAATAGACCCAA-3′ and reverse,
5′-ATGGCCTATGTGTGCGTTGTA-3′; TIMELESS forward,
5′-TCTGATCCGCTATTTGAGGCA-3′ and reverse,
5′-GGCAGAAGGTCGCTCTGTAG-3′; NPAS2 forward,
5′-CGTGTTGGAAAAGGTCATCGG-3′ and reverse,
5′-TCCAGTCTTGCTGAATGTCAC-3′; Cry1 forward,
5′-CTCCTCCAATGTGGGCATCAA-3′ and reverse,
5′-CCACGAATCACAAACAGACGG-3′; Cry2 forward,
5′-TCCCAAGGCTGTTCAAGGAAT-3′ and reverse,
5′-TGCATCCCGTTCTTTCCCAAA-3′; PER1 forward,
5′-AGTCCGTCTTCTGCCGTATCA-3′ and reverse,
5′-AGCTTCGTAACCCGAATGGAT-3′; PER2 forward,
5′-GTGAAAGTGAGGAGAAAGGCAACC-3′ and reverse,
5′-CACCTCTTCCGAGCACCGTC-3′; PER3 forward,
5′-GCAGAGGAAATTGGCGGACA-3′ and reverse,
5′-GGTTTATTGCGTCTCTCCGAG-3′; and GAPDH forward,
5′-GGCTAGCACCAACCAAAAGTATAGTCATCCGG-3′ and reverse,
5′-GGAATTCGTCTTAAAATTCAATTTATTATAAA-3′. Fold changes in mRNA
expression were quantified with the 2−∆∆Cq relative
quantification method using GAPDH as a housekeeping control gene
(21).
Neurosphere formation assay
GSCs were plated at 1,000 cells/well in 96-well
plates. After culture for 7 days, the number of neurospheres that
contained >20 cells was determined in each well. Wells
containing no cells or >1 cell were excluded, and those with 1
cell were marked and monitored daily under a microscope. The number
of tumor spheres per well was recorded, and the tumor sphere
formation rate was calculated.
Cell proliferation assay and GSC flow
cytometric assay
Cell proliferation was measured using a CCK-8 assay
(Dojindo Molecular Technologies., Inc.) according to the
manufacturer's protocol. The absorbance was then measured at 450 nm
using a microplate reader (Tecan Group, Ltd.). Three independent
experiments were carried out, and the proliferation fold was
calculated as follows: (OD value at 24, 48 or 96 h-OD value at 0
h)/(OD value at 0 h).
To study the cell cycle, when GSCs reached ~70%
confluency, they were detached with trypsin after washing twice
with phosphate-buffered saline (PBS), centrifuged at 1,000 × g for
5 min, and then fixed with cold 70% alcohol and frozen at −20°C. In
addition, cell cycle analysis was performed by standard PI staining
(BD Biosciences). Briefly, cells were fixed, stained with PI
solution for 30 min at 37°C, spun down and resuspended in PBS.
After staining, the samples (5×104 cells/200 µl) were
analyzed using a BD FACSCalibur™ Flow Cytometer (BD Biosciences).
The data were analyzed with a BD FACS Diva (version 7.0; BD
Biosciences).
Immunofluorescence and
immunohistochemical staining
GSCs were fixed with 4% paraformaldehyde at room
temperature, and PBS was used to wash the cells. Triton X-100
(0.1%) was used to permeabilize the cells, following which they
were blocked with 5% BSA. Cells were incubated with the primary
antibody for 20 h at 4°C (stem marker antibody concentrations were
according to the manufacturer's instructions). Next, the samples
were washed three times with PBS and incubated with fluorescent
secondary antibodies at 37°C for 1 h in the dark: Goat anti-mouse
IgG H&L (product code ab150117; dilution 1:500), goat
anti-mouse IgG H&L (product code ab150115; dilution 1:1,000)
and goat anti-rabbit IgG H&L (product code ab150079; dilution
1:1,000; all from Abcam). Then, the samples were washed three times
with PBS, and DAPI was used to counterstain the cells at room
temperature for 10 min. Finally, a fluorescence microscope (Leica
Microsystems GmbH) at ×40 magnification was used to examine the
cells.
For immunohistochemical (IHC) staining, brain tumor
sections were incubated with the following primary antibodies: PER2
(product code ab94915; 1:500), Ki-67 (product code ab15580; 1:100;
both from Abcam) and CD133 (cat. no. NB120-16518; 1:500,
Bio-Techne), for 1 h at room temperature after deparaffinization,
rehydration, antigen retrieval, endogenous peroxidase quenching and
blocking. The sections were incubated with HRP-conjugated polymer
(Dako; Agilent Technologies, Inc.) for 40 min and then with
diaminobenzidine using an Ultravision DAB Plus Substrate Detection
System (Thermo Fisher Scientific, Inc.) for 1–10 min at room
temperature, followed by hematoxylin staining at room temperature
for 2 min. Finally, a Leica DM6 light microscope (Leica
Microsystems GmbH) at magnification ×10 and ×20 was used to examine
the sections.
Migration and invasion assays
GSCs (1×105) in 200 µl of SFM were seeded
into the top chamber of cell culture inserts coated with or without
Matrigel (BD Biosciences), while the lower chamber was filled with
400 µl medium containing 8% FBS as a chemoattractant. After
incubation at 37°C for 24 h, the cells that passed through the
membrane were fixed in 100% methanol for 20 min at room temperature
and stained with 0.1% crystal violet solution for 10 min at room
temperature. Three independent fields of cells adhered to the lower
side of the Transwell filters were photographed by an inverted
light microscope (Nikon Corporation) at ×20 magnification for each
well.
RNA-Sequencing (RNA-Seq)
Total RNA from U251s-control and U251s-PER2
overexpression cells was extracted using TRIzol (Invitrogen; Thermo
Fisher Scientific, Inc.) and treated with DEPC water. RNA-Seq was
performed by Allwegene Technology Co., Ltd. (Beijing, China). The
reference genome was from http://www.ensembl.org/Homo_sapiens/Info/Index. The
data were analyzed on the online Majorbio I-Sanger Cloud Platform
(www.i-sanger.com). The differential expression
analysis was conducted with DEGseq 1.30.0 software. Genes with
adjusted P-values (P-adjust) ≤0.05 were considered differentially
expressed genes (DEGs). Kyoto Encyclopedia of Genes and Genomes
(KEGG) pathway and Gene Ontology (GO) analyses of DEGs were
analyzed with R package software (P<0.05).
Intracranial tumor formation in
vivo
For intracranial implantation, U251 GSCs
(1×105) transfected with lentiviral vectors expressing
GFP-PER2 or GFP-control were implanted into the right frontal lobes
of 6–8-week-old male BALB/c athymic nude mice (a total of 4
mice/group, Experimental Animals Center of Ningxia Medical
University, Yinchuan, China) after anesthesia (1.5–2.5%
isoflurane/oxygen inhalation for anesthetic maintenance). After 4,
10 and 15 days, a Xenogen in vivo imaging system (IVIS) was
used to visualize the tumors, and photon measurement was defined
around the tumor area and quantified using Living Image software
(Caliper Life Science SA). The nude mice were raised under specific
pathogen-free conditions in a temperature-controlled room (22±2°C;
humidity, 50±10%) with a 12-h light/dark cycle, and with ad
libitum access to sterilized water and mice food. The health
and behavior of mice were monitored daily. After 2 weeks,
tumor-bearing nude mice exhibited clinical signs of a brain tumor,
such as spine curvature or dyskinesia, and thus the mice were
sacrificed with transcardial perfusion under anesthesia (1.5–2.5%
isoflurane/oxygen inhalation) on the 15th day after the
implantation. The death of mice was verified by assessing cardiac
arrest. Brain tumors in the mice were removed after transcardial
perfusion with normal saline and then 4% paraformaldehyde (PFA) and
were fixed in formalin or post-fixed in 4% PFA overnight at 4°C for
OCT frozen tissue blocks. All mice included in the present study
exhibited a single tumor, the maximum level of cachexia observed
was a body weight loss <5%. The maximum tumor diameter was 6 mm,
the volume was 78.45 mm3 (the tumor volume was
calculated according to the formula: Tv=π/6 × tumor length × tumor
width2) and the wet weight was 0.15 g, as <1% of
total mice body weight (Fig. S1A).
All mice experiments were performed with the approval of the
Ningxia Medical University Experimental Animals Center IACUC.
Statistical analysis
The SPSS 20.0 software (IBM Corp.) was used for
statistical analysis. All experiments were performed independently
in triplicate, and all values were expressed as the mean ± standard
deviation. The data were analyzed using two-tailed Student's t-test
(for two-group comparisons) or one-way analysis of variance (ANOVA)
with Tukey's post hoc test (for multiple comparisons). P<0.05
and P<0.01 were considered to indicate statistically significant
differences.
Results
PER2 expression is downregulated in
GSCs
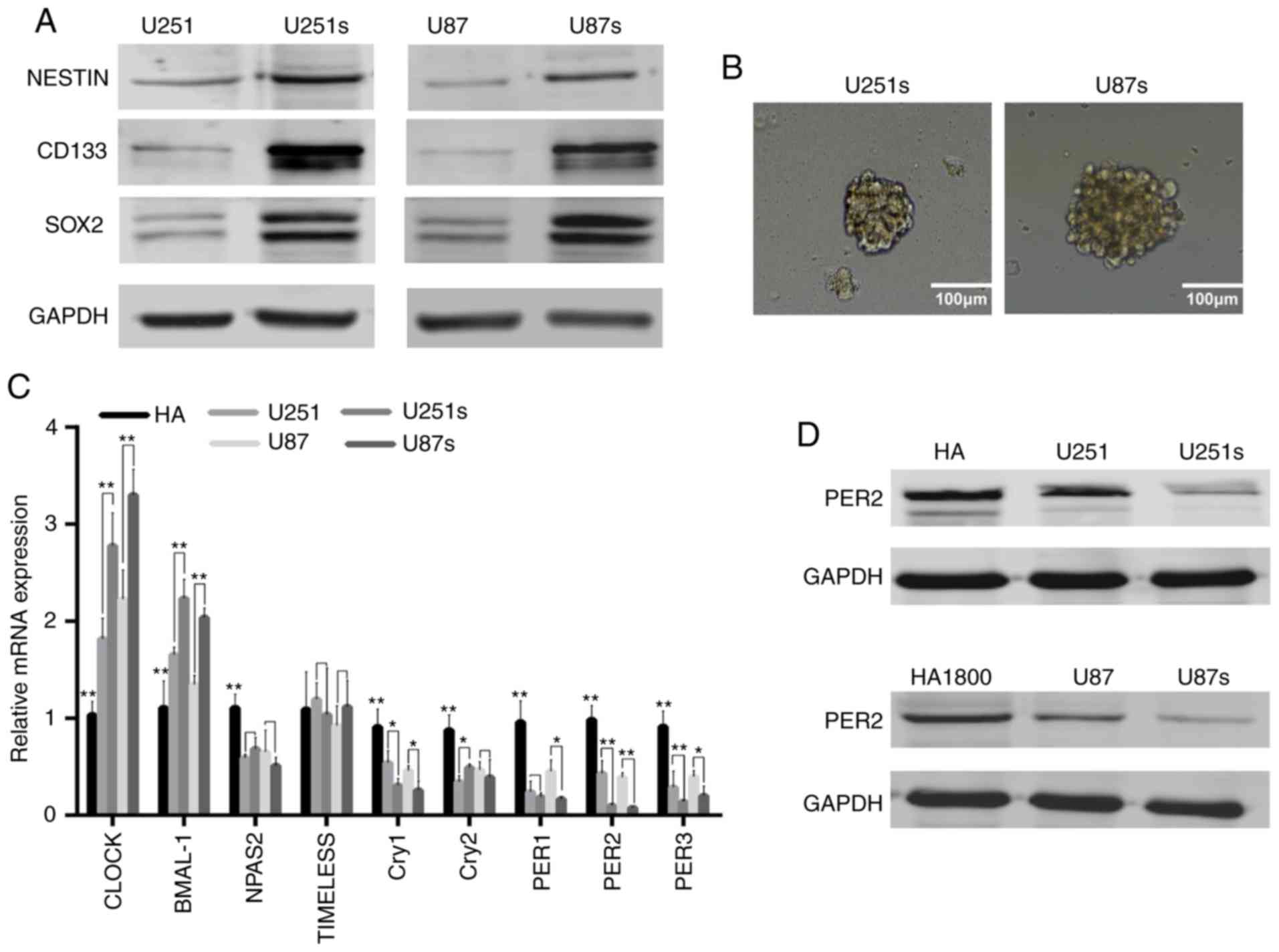
Based on previous study, GSCs could be enriched by
sphere formation (22). Preliminary
analysis of western blot data indicated that, as anticipated, the
expression of critical GSC markers, such as CD133, NESTIN and SOX2,
were markedly upregulated in U251 and U87 sphere-forming cells
(Fig. 1A). The representative
images of these GSCs are presented in Fig. 1B. These data confirmed that GSCs
were enriched in U251 and U87 sphere-forming cells. Next, it was
investigated whether circadian genes were ectopically expressed in
GSCs. RT-qPCR and western blotting were performed to identify the
mediator of GSCs stemness, it was demonstrated that PER2 was one of
the most significantly downregulated core circadian genes in GSCs
compared with NGSCs or human astrocyte cell line (Fig. 1C and D). These results indicated
that PER2 expression was associated with GSCs, indicating that PER2
may be involved in the malignant process of glioma and the function
of GSCs.
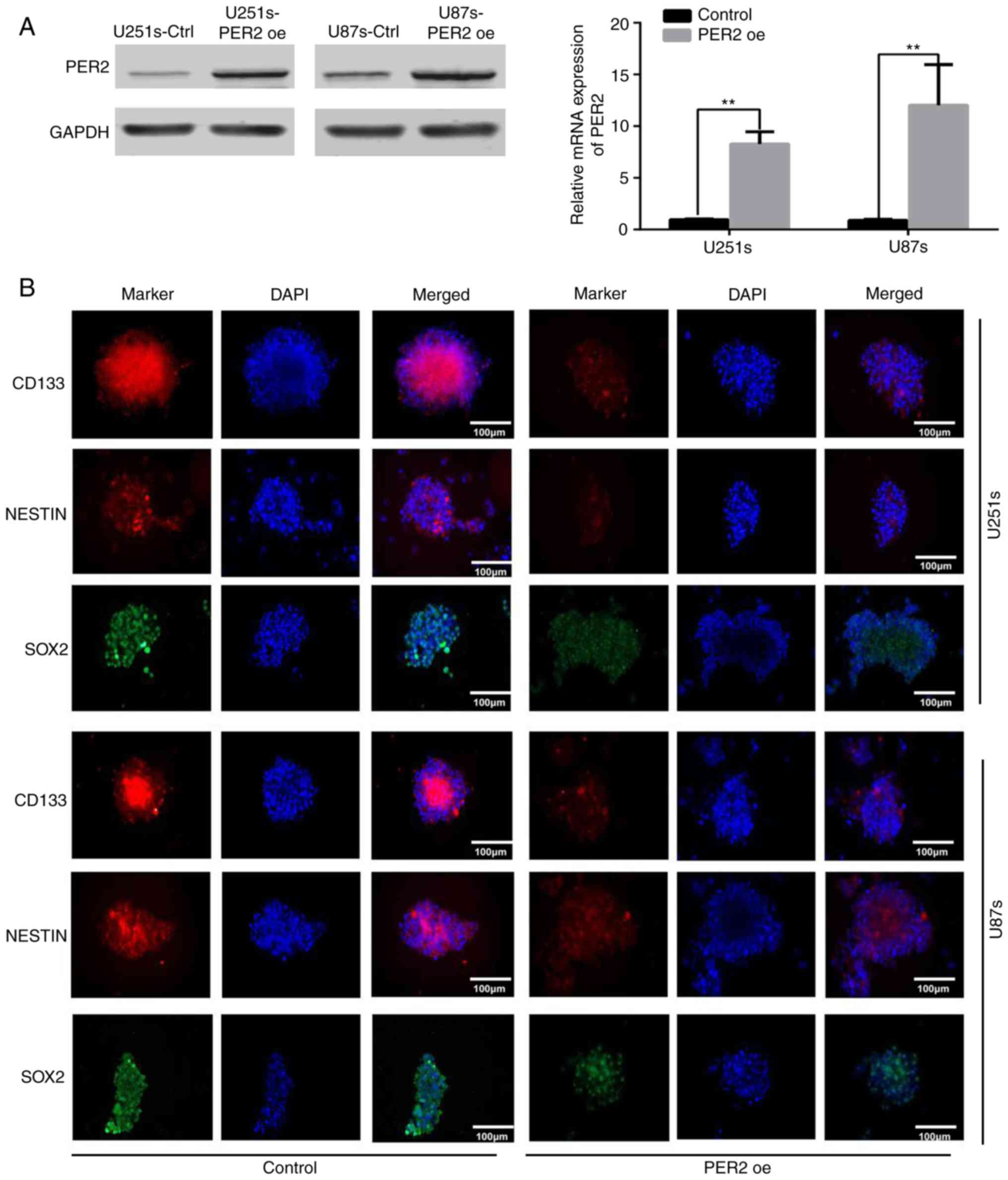
PER2 overexpression reduces the
stemness and self-renewal of GSCs
Maintenance of pluripotency is crucial for the
proliferation and survival of GSCs during cancer development
(4,7). To explore the functional significance
of PER2 in inhibiting GSC stemness, GSCs were infected with
lentivirus-PER2. Western blotting and qRT-PCR data indicated that,
as anticipated, PER2 in these cell lines was overexpressed upon
lentivirus-PER2 transfection (Fig.
2A). Following overexpression of PER2, the expression of
stemness markers, such as CD133, NESTIN, and SOX2, was decreased in
GSCs according to immunofluorescence (Fig. 2B) and western blot analysis
(Fig. S1B). Furthermore,
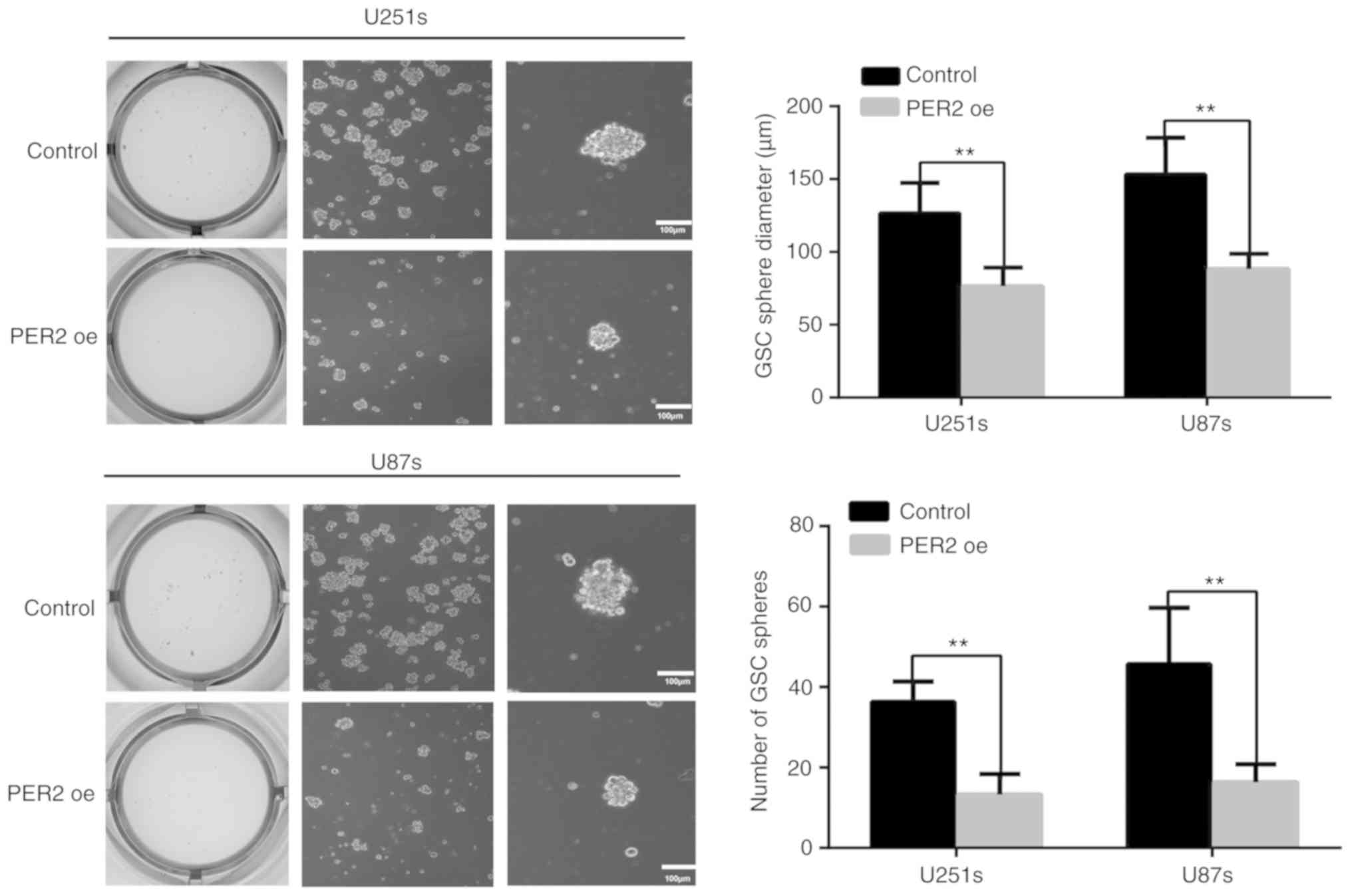
neurosphere formation revealed that PER2 could inhibit the
self-renewal ability of GSCs. Both the neurosphere diameter and the
number of GSC spheres were reduced after treatment with
lentivirus-PER2 (Fig. 3). These
results indicated that PER2 could inhibit the stemness and
self-renewal capability of GSCs.
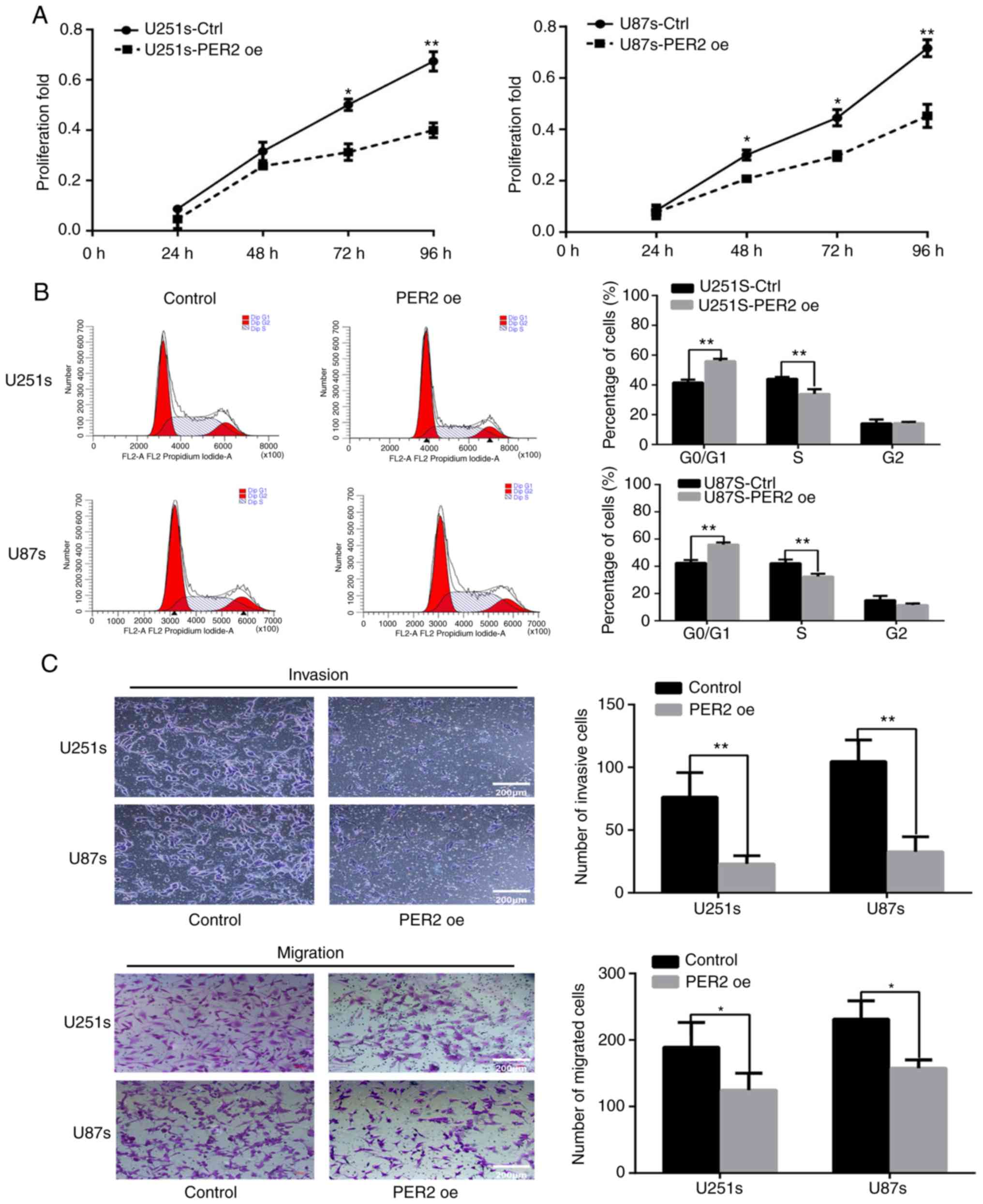
PER2 inhibits viability, cell cycle
progression, migration, and invasion in GSCs
To investigate the role of PER2 in proliferation,
CCK-8 assays were performed at different time-points (24, 48, 72
and 96 h) after transfection with an empty lentiviral vector
(control) or lentiviral-PER2. The proliferation of GSCs was
significantly inhibited by PER2 overexpression (Fig. 4A). Then, flow cytometry was
performed to explore the function of PER2 in the cell cycle. The
results indicated that the percentage of S-phase cells was
significantly decreased after PER2 overexpression, and G0/G1 cycle
arrest was observed in this group of cells (Fig. 4B). Previous research has suggested
that cancer stem cells may possess higher invasive activity than
differentiated cancer cells (8).
Therefore, the effects of PER2 on the invasion and migration
abilities of GCSs were detected. Transwell chamber assays revealed
that overexpression of PER2 inhibited the invasion and migration
abilities of GSCs (Fig. 4C).
Based on the aforementioned data, it was
hypothesized that PER2 could inhibit the malignant characteristics
of GSCs in vivo. To test this hypothesis, the constructed
U251-GSC line was implanted into nude mice by intracranial
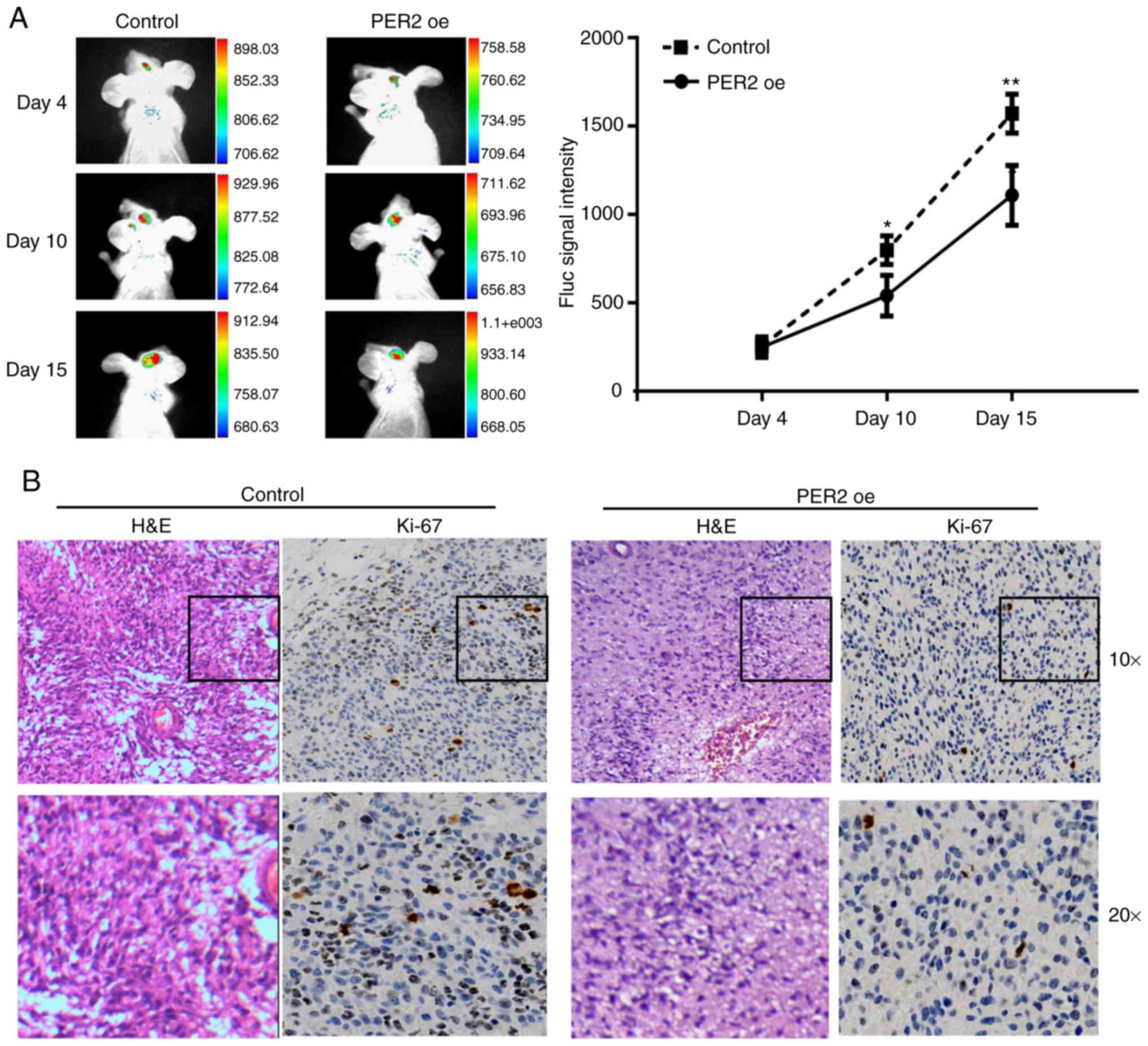
injection. As anticipated, IVIS (an imaging system) analysis and
tumor volume assessment revealed that PER2 overexpression decreased
tumor sizes at 4, 10, and 15 days after GSC transplantation
(Fig. 5A) and the final tumor
volume (Fig. S1A). Furthermore,
fewer Ki-67-positive cells were observed in the PER2 overexpression
group than in the control group (Fig.
5B).
Next, it was determined whether PER2 also affected
the phenotype of GSCs formed in humans. PER2 expression was
analyzed in 48 paraffin-embedded glioma cancer tissue specimens.
CD133 is the most well-accepted stemness marker of GSCs (22,23),
and CD133 IHC revealed a significant negative association between
PER2 expression and the stemness of glioma cells. High
CD133-positive samples exhibited low or non-detectable PER2
staining. In contrast, samples with high PER2 staining did not have
CD133 membrane staining (Fig.
S1C).
PER2 suppresses the stem cell-like
phenotype of GBM by dysregulating Wnt/β-catenin signaling
To characterize the molecular mechanisms of PER2 in
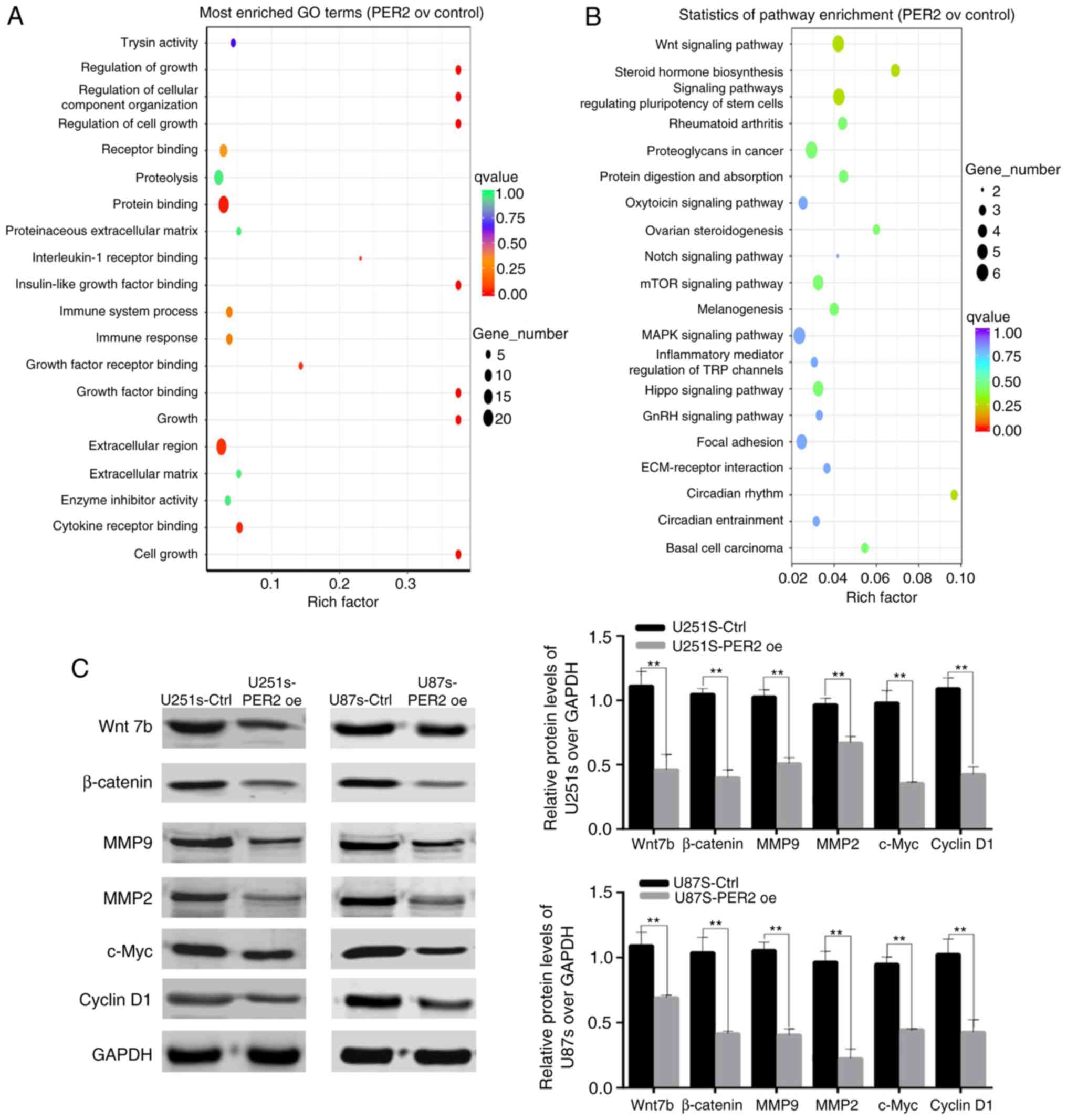
GSCs, RNA-Seq analysis of control and PER2-overexpressed U251 GSCs
was performed. Analysis of the RNA-Seq data indicated that PER2
overexpression decreased the expression of Wnt family genes,
including Wnt7b and other Wnt family ligands and receptors
(Fig. S2). To explore the
molecular mechanisms underlying PER2 function, KEGG and GO analyses
of the RNA-Seq data were also performed. The GO analysis revealed
that PER2 altered the expression of genes involved in cell growth,
metabolism and organismal systems, that aberrant signal
transduction was closely related to the initiation and development
of glioma (Fig. 6A). The results of
the KEGG analysis also revealed that PER2 was significantly
involved in the regulation of the Wnt signaling pathway (Fig. 6B; P=0.00694). Based on these data,
it was hypothesized that PER2 may target the Wnt signaling pathway
in order to regulate the development and progression of GSCs.
To verify this hypothesis, western blotting was
performed to assess the expression of some genes within the Wnt
signaling pathway in the GSC cell lines. PER2 decreased the
expression of Wnt7b and its downstream target genes, such as
β-catenin, c-Myc, MMP2, MMP9, and cyclin D1. Consequently, PER2
suppressed the stemness of GSCs, at least partly, via the
Wnt/β-catenin signaling pathway (Fig.
6C).
Discussion
Increasing evidence is showing that circadian genes
play critical roles in the biological processes of cancer cells
(12-14-24). Some of these genes also regulate cancer stem cell
differentiation and cell stemness maintenance (25,26).
Therefore, circadian genes may be potential targets for cancer stem
cell treatment (27). Despite these
insights into the role of circadian dysregulation in cancer, few
novel circadian gene-targeting GSC therapies have emerged. Thus,
nine core circadian gene expression profiles were detected in GSCs,
normal glioma cells and human astrocyte cell lines, and PER2 was
screened as the most significantly altered circadian gene involved
in the malignant process of glioma. Consistent with data from our
previous study, PER2 expression was notably downregulated in glioma
samples compared with adjacent noncancerous tissues (20). These findings led to the development
of the present study, which aimed to further explore the role of
PER2 in GSCs and its potential mechanism of action.
In the present study, two GSC cell lines (U251s and
U87s) were successfully established. Consistent with a previous
study (6), our results confirmed
that the stemness markers CD133, SOX2, and NESTIN were
significantly increased in sphere-forming glioma cells compared
with normal glioma cells. Next, it was demonstrated that PER2
overexpression led to the significant inhibition of stemness marker
expression, decreased self-renewal capability and cell cycle arrest
at the G0/G1 phase in GSCs. Furthermore, it was observed that PER2
suppressed the malignant characteristics of GSCs in vitro
and in vivo. These results are consistent with a previous
study revealing that the PER family gene PER3 could regulate
self-renewal and chemoresistance in colorectal CSCs (28). Notably, we studied the malignant
characteristics of GSCs in vivo with non-whole brain tumor
pathology which could moderately demonstrate the result. However,
pathological sections of the whole brain of tumor-bearing mice will
provide relative position information of tumors in the brain which
can further confirm that PER2 could inhibit the malignant
characteristics of GSCs.
Previous research has demonstrated that PER2 acts as
a potential anti-oncogene by targeting multiple genes, such as p53,
AKT, OCT1 as well as several others (13,29–31).
By analyzing the RNA-Seq results, it was determined that PER2 was
involved in regulating the Wnt signaling pathway in GSCs. Griveau
et al demonstrated that Wnt7b can affect glioma
proliferation and invasion (32).
As an important downstream component of the Wnt/β-catenin pathway,
β-catenin can promote the uncontrolled proliferation and stemness
of GBM cells (33). Tumor invasion
is often due to the activities of metalloproteinases, which
efficiently digest the extracellular matrix, such as MMP2 and MMP9
(34,35). It has been reported that c-Myc is
required for the diverse features and pluripotency of GSCs
(36). Therefore, in the present
study, the downregulation of Wnt7b, β-catenin, MMP2, MMP9, and
c-Myc induced by PER2 may reduce the invasive potential and
stemness of GSCs. Cyclin D1 is a dominant cell cycle regulator and
is involved in cell cycle control (37). which could explain the G0/G1 phase
arrest observed in the PER2-overexpressing GSC cell lines.
In conclusion, the present study identified an
important function of PER2 in regulating the stemness, cell growth,
cell cycle progression and migration of GSCs in vitro and
in vivo via the downregulation of Wnt/β-catenin
signaling-associated gene expression. These findings indicated that
PER2 may serve as a novel target in therapeutic strategies to
overcome the effects of GSCs.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by a grant from the
National Natural Science Foundation of China (grant no.
81660419).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HX and HF conceived and designed the experiments. DM
and LH performed the experiments. XJ and HL provided technical
support, critical comments and suggestions. ZN analyzed the data.
DM wrote the manuscript. All authors read and approved the final
manuscript and agree to be accountable for all aspects of the work
in ensuring that questions related to the accuracy or integrity of
any part of the work are appropriately investigated and
resolved.
Ethics approval and consent to
participate
All patients provided written informed consent, and
the study was approved by the Ethics Committee of Ningxia Medical
University. All mice experiments were performed with the approval
of the Ningxia Medical University Experimental Animals Center
IACUC.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cenciarini M, Valentino M, Belia S, Sforna
L, Rosa P, Ronchetti S, D'Adamo MC and Pessia M: Dexamethasone in
glioblastoma multiforme therapy: Mechanisms and controversies.
Front Mol Neurosci. 12:652019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Eisemann T, Costa B, Strelau J,
Mittelbronn M, Angel P and Peterziel H: An advanced glioma cell
invasion assay based on organotypic brain slice cultures. BMC
Cancer. 18:1032018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Babu R, Komisarow JM, Agarwal VJ,
Rahimpour S, Iyer A, Britt D, Karikari IO, Grossi PM, Thomas S,
Friedman AH and Adamson C: Glioblastoma in the elderly: The effect
of aggressive and modern therapies on survival. J Neurosurg.
124:998–1007. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Singh SK, Hawkins C, Clarke ID, Squire JA,
Bayani J, Hide T, Henkelman RM, Cusimano MD and Dirks PB:
Identification of human brain tumour initiating cells. Nature.
432:396–401. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Almenawer SA, Badhiwala JH, Alhazzani W,
Greenspoon J, Farrokhyar F, Yarascavitch B, Algird A, Kachur E,
Cenic A, Sharieff W, et al: Biopsy versus partial versus gross
total resection in older patients with high-grade glioma: A
systematic review and meta-analysis. Neuro Oncol. 17:868–881. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q,
Hjelmeland AB, Dewhirst MW, Bigner DD and Rich JN: Glioma stem
cells promote radioresistance by preferential activation of the DNA
damage response. Nature. 444:756–760. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen J, Li Y, Yu TS, McKay RM, Burns DK,
Kernie SG and Parada LF: A restricted cell population propagates
glioblastoma growth after chemotherapy. Nature. 488:522–526. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bao S, Wu Q, Sathornsumetee S, Hao Y, Li
Z, Hjelmeland AB, Shi Q, McLendon RE, Bigner DD and Rich JN: Stem
cell-like glioma cells promote tumor angiogenesis through vascular
endothelial growth factor. Cancer Res. 66:7843–7848. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kohler F and Rodriguez-Paredes M: DNA
methylation in epidermal differentiation, aging, and cancer. J
Invest Dermatol. 140:38–47. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lowrey PL and Takahashi JS: Mammalian
circadian biology: Elucidating genome-wide levels of temporal
organization. Annu Rev Genomics Hum Genet. 5:407–441. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikegami K, Refetoff S, Van Cauter E and
Yoshimura T: Interconnection between circadian clocks and thyroid
function. Nat Rev Endocrinol. 15:590–600. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shostak A: Circadian clock, cell division,
and cancer: From molecules to organism. Int J Mol Sci. 18:E8732017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hwang-Verslues WW, Chang PH, Jeng YM, Kuo
WH, Chiang PH, Chang YC, Hsieh TH, Su FY, Lin LC, Abbondante S, et
al: Loss of corepressor PER2 under hypoxia up-regulates
OCT1-mediated EMT gene expression and enhances tumor malignancy.
Proc Natl Acad Sci USA. 110:12331–12336. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Xiang R, Cui Y, Wang Y, Xie T, Yang X,
Wang Z, Li J and Li Q: Circadian clock gene Per2 downregulation in
nonsmall cell lung cancer is associated with tumour progression and
metastasis. Oncol Rep. 40:3040–3048. 2018.PubMed/NCBI
|
|
15
|
Xiong H, Yang Y, Yang K, Zhao D, Tang H
and Ran X: Loss of the clock gene PER2 is associated with cancer
development and altered expression of important tumor-related genes
in oral cancer. Int J Oncol. 52:279–287. 2018.PubMed/NCBI
|
|
16
|
Deng F and Yang K: Current status of
research on the period family of clock genes in the occurrence and
development of cancer. J Cancer. 10:1117–1123. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu ML, Yeh KT, Lin PM, Hsu CM, Hsiao HH,
Liu YC, Lin HY, Lin SF and Yang MY: Deregulated expression of
circadian clock genes in gastric cancer. BMC Gastroenterol.
14:672014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mazzoccoli G, Panza A, Valvano MR, Palumbo
O, Carella M, Pazienza V, Biscaglia G, Tavano F, Di Sebastiano P,
Andriulli A and Piepoli A: Clock gene expression levels and
relationship with clinical and pathological features in colorectal
cancer patients. Chronobiol Int. 28:841–851. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rahman S, Kraljević Pavelić S and
Markova-Car E: Circadian (De)regulation in head and neck squamous
cell carcinoma. Int J Mol Sci. 20:E26622019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xia HC, Niu ZF, Ma H, Cao SZ, Hao SC, Liu
ZT and Wang F: Deregulated expression of the Per1 and Per2 in human
gliomas. Can J Neurol Sci. 37:365–370. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Singh SK, Clarke ID, Terasaki M, Bonn VE,
Hawkins C, Squire J and Dirks PB: Identification of a cancer stem
cell in human brain tumors. Cancer Res. 63:5821–5828.
2003.PubMed/NCBI
|
|
23
|
Lv D, Ma QH, Duan JJ, Wu HB, Zhao XL, Yu
SC and Bian XW: Optimized dissociation protocol for isolating human
glioma stem cells from tumorspheres via fluorescence-activated cell
sorting. Cancer Lett. 377:105–115. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Maiese K: Moving to the rhythm with clock
(circadian) genes, autophagy, mTOR, and SIRT1 in degenerative
disease and cancer. Curr Neurovasc Res. 14:299–304. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang J, Morita Y, Han B, Niemann S,
Löffler B and Rudolph KL: Per2 induction limits lymphoid-biased
haematopoietic stem cells and lymphopoiesis in the context of DNA
damage and ageing. Nat Cell Biol. 18:480–490. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Puram RV, Kowalczyk MS, de Boer CG,
Schneider RK, Miller PG, McConkey M, Tothova Z, Tejero H, Heckl D,
Järås M, et al: Core circadian clock genes regulate leukemia stem
cells in AML. Cell. 165:303–316. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ríos-Arrabal S, Muñoz-Gámez JA,
Jiménez-Ruíz SM, Casado-Ruíz J, Artacho-Cordón F and León-López J:
Circadian Regulation of Colon Cancer Stem Cells. Implications for
Therapy. 2016.
|
|
28
|
Zhang F, Sun H, Zhang S, Yang X, Zhang G
and Su T: Overexpression of PER3 inhibits self-renewal capability
and chemoresistance of colorectal cancer stem-like cells via
inhibition of notch and β-catenin signaling. Oncol Res. 25:709–719.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Miki T, Matsumoto T, Zhao Z and Lee CC:
p53 regulates period 2 expression and the circadian clock. Nat
Commun. 4:24442013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang X, He X, Yang Z and Jabbari E:
Mammalian PER2 regulates AKT activation and DNA damage response.
Biochem Cell Biol. 90:675–682. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gotoh T, Kim JK, Liu J, Vila-Caballer M,
Stauffer PE, Tyson JJ and Finkielstein CV: Model-driven
experimental approach reveals the complex regulatory distribution
of p53 by the circadian factor period 2. Proc Natl Acad Sci USA.
113:13516–13521. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Griveau A, Seano G, Shelton SJ, Kupp R,
Jahangiri A, Obernier K, Krishnan S, Lindberg OR, Yuen TJ, Tien AC,
et al: A glial signature and Wnt7 signaling regulate
glioma-vascular interactions and tumor microenvironment. Cancer
Cell. 33:874–889.e7. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang N, Wei P, Gong A, Chiu WT, Lee HT,
Colman H, Huang H, Xue J, Liu M, Wang Y, et al: FoxM1 promotes
β-catenin nuclear localization and controls Wnt target-gene
expression and glioma tumorigenesis. Cancer Cell. 20:427–442. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guichet PO, Guelfi S, Teigell M, Hoppe L,
Bakalara N, Bauchet L, Duffau H, Lamszus K, Rothhut B and Hugnot
JP: Notch1 stimulation induces a vascularization switch with
pericyte-like cell differentiation of glioblastoma stem cells. Stem
Cells. 33:21–34. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mikheev AM, Mikheeva SA, Severs LJ, Funk
CC, Huang L, McFaline-Figueroa JL, Schwensen J, Trapnell C, Price
ND, Wong S and Rostomily RC: Targeting TWIST1 through loss of
function inhibits tumorigenicity of human glioblastoma. Mol Oncol.
12:1188–1202. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Di J, Duiveman-de Boer T, Zusterzeel PL,
Figdor CG, Massuger LF and Torensma R: The stem cell markers Oct4A,
Nanog and c-Myc are expressed in ascites cells and tumor tissue of
ovarian cancer patients. Cell Oncol (Dordr). 36:363–374. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim JK and Diehl JA: Nuclear cyclin D1: An
oncogenic driver in human cancer. J Cell Physiol. 220:292–296.
2009. View Article : Google Scholar : PubMed/NCBI
|