Introduction
Lung cancer, predominantly non-small-cell lung
cancer (NSCLC), is one of the most common types of malignant cancer
and the primary cause of tumor-associated mortality and morbidity
worldwide with a high incidence rate and a 5-year survival rate of
18% (1). Platinum-based
chemotherapy regimens are currently the standard adjuvant treatment
strategy for advanced NSCLC following surgical resection (2). As a widely used platinum-coordinated
complex in cancer therapy, cisplatin (DDP) is the first-line
chemotherapeutic drug against NSCLC due to its therapeutic
advantages such as high efficiency and mild side effects (3). However, the development of drug
resistance, which limits the clinical therapeutic efficacy of DDP,
is currently a major impediment to successful chemotherapy
(2,4). The mechanisms underlying DDP
resistance are extremely complex and multi-factorial (5). Therefore, elucidating these molecular
mechanisms is clinically important to develop therapeutic
approaches for the treatment of lung cancer and to improve the
therapeutic efficacy of lung cancer chemotherapy.
Human genome sequence data indicates that only 2% of
the genome encodes proteins, and the majority of genes are
transcribed into non-coding RNAs (ncRNAs) (6,7). Among
the various types of ncRNAs, long ncRNAs (lncRNAs) are defined as
non-protein coding RNA transcripts that are >200 nucleotides
long (8). LncRNAs serve regulatory
roles in gene expression at the transcriptional,
post-transcriptional and epigenetic levels, and thus are implicated
in a range of physiological and pathological processes, including
the pathogenesis and development of tumors (9). A previous study has demonstrated that
upregulation of lncRNA HOX transcript antisense RNA (HOTAIR) may
repress the chemosensitivity of lung cancer cells resistant to
cisplatin (8). Antisense ncRNA in
the inhibitor of cyclin-dependent kinase 4 (INK4) locus (ANRIL),
also termed CDKN2B antisense RNA1, is transcribed as a 3.8-kb
lncRNA in the antisense orientation of the INK4B-ARF-INK4A
gene cluster at chromosome 9p21 (10). A number of studies have demonstrated
that ANRIL is aberrantly upregulated in several types of
malignancy, such as lung, breast and gastric cancer (11–13).
Accumulating evidence has suggested that ANRIL exhibits oncogenic
activity and acts as a key player in a variety of carcinomas,
including NSCLC (11,12,14).
However, the detailed roles of ANRIL in DDP resistance of lung
cancer and the underlying mechanisms remain to be elucidated.
microRNAs (miRNAs) are a superfamily of small ncRNAs
that are 19–25 nucleotides long and serve important roles in the
regulation of gene expression at the post-transcriptional level by
binding to the 3′ untranslated region (UTR) of the target mRNA,
resulting in mRNA degradation or translation repression (15). An increasing number of studies have
reported that miRNAs participate in the regulation of various
biological processes, such as cell proliferation, invasion and
apoptosis (16,17). In addition, dysregulation of miRNA
expression has been revealed to be involved in the chemoresistance
of various types of cancer, including NSCLC (18,19).
Specifically, miR-98 has been demonstrated to be downregulated in
DDP-resistant NSCLC cells, and high expression of miR-98 leads to
high sensitivity to DDP (20). A
novel regulatory mechanism regarding the competing endogenous (ce)
RNA hypothesis has suggested that lncRNAs can act as miRNA sponges
via competition for shared miRNA-response elements to suppress the
expression and activity of miRNAs (21). Therefore, the present study focused
on the interaction between NARIL and miR-98 in lung cancer.
The present study aimed to investigate the
functional roles of ANRIL in DDP resistance of lung cancer and the
underlying mechanism involved in the ceRNA regulatory network.
Materials and methods
Tissue sample collection
A total of 26 NSCLC tissues and paired adjacent
normal lung tissues from patients (mean age, 58 years; age range,
40–72 years) undergoing resection were collected from the First
Affiliated Hospital of Zhengzhou University (Zhengzhou, China),
between January 2017 and December 2018. The patients received no
preoperative chemotherapy or radiotherapy and were diagnosed with
NSCLC by histopathological evaluation. The distance between
cancerous lesions and adjacent healthy tissues was ≥5 cm. All
tissue specimens were immediately frozen in liquid nitrogen and
stored at −80°C until RNA extraction. The present study was
reviewed and approved by the Research Ethics Committee of the First
Affiliated Hospital of Zhengzhou University. Written consent was
obtained from all patients. The clinicopathological characteristics
of the patients with NSCLC are list in Table I.
 | Table I.Clinicopathological characteristics
of patients with non-small cell lung cancer. |
Table I.
Clinicopathological characteristics
of patients with non-small cell lung cancer.
| ID | Age | Sex | TNM stage | Smoking status | Prior therapy |
|---|
| 1 | 64 | Male | IV | Former | Surgery |
| 2 | 40 | Male | II | Current | Surgery |
| 3 | 53 | Male | IV | Former | Surgery |
| 4 | 56 | Male | IV | Former | None |
| 5 | 46 | Male | IV | Current | None |
| 6 | 49 | Male | II | Current | Surgery |
| 7 | 58 | Male | IV | Current | Surgery |
| 8 | 64 | Female | IV | Current | Surgery |
| 9 | 63 | Female | IV | Current | Surgery |
| 10 | 67 | Male | III | Current | Surgery |
| 11 | 45 | Male | III | Current | None |
| 12 | 43 | Male | I | Current | None |
| 13 | 44 | Male | I | Current | None |
| 14 | 69 | Female | IV | Former | Surgery |
| 15 | 64 | Female | IV | Current | Surgery |
| 16 | 63 | Male | IV | Former | Surgery |
| 17 | 59 | Female | IV | Current | Surgery |
| 18 | 66 | Male | II | Current | None |
| 19 | 67 | Male | IV | Current | Surgery |
| 20 | 54 | Female | II | Former | None |
| 21 | 58 | Male | IV | Current | Surgery |
| 22 | 48 | Male | II | Current | Surgery |
| 23 | 72 | Male | IV | Former | Surgery |
| 24 | 67 | Male | IV | Former | Surgery |
| 25 | 64 | Male | IV | Former | Surgery |
| 26 | 65 | Male | IV | Former | Surgery |
Cell culture and transfection
Normal lung epithelial cells (BEAS-2B) were obtained
from the American Type Culture Collection. The human lung
adenocarcinoma cell line A549 and DDP-resistant A549/DDP cells were
purchased from Nanjing Keygen Biotechnology Co. Ltd. All cells were
cultured in RPMI-1640 medium (Invitrogen; Thermo Fisher Scientific,
Inc.) supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo
Fisher Scientific, Inc.), 1% L-glutamine and 1%
penicillin/streptomycin (Invitrogen; Thermo Fisher Scientific,
Inc.) at 37°C in a humidified atmosphere with 5% CO2.
A549/DDP cells were cultured in RPMI-1640 medium containing 1 µg/ml
DDP (Sigma Aldrich; Merck KGaA) to maintain the resistance of this
cell line to DDP prior to the experiments. Cells in the logarithmic
growth phase were used for the experiments.
pcDNA-ANRIL, pcDNA vector (vector), small
interfering (si) RNAs targeting ANRIL (si-ANRIL-1, si-ANRIL-2 and
si-ANRIL-3), scrambled non-targeting siRNA negative control
(si-NC), miR-98 mimics (miR-98) and scrambled non-targeting miRNA
negative control (miR-NC) were purchased from Shanghai Genepharma
Co., Ltd. The sequences of si-ANRIL and miR-98 mimics were as
follows: si-ANRIL-1, 5′-ACAGAAUAGUGUUCUUCUGCU-3′; si-ANRIL-2,
5′-UUUCUAAUAAUUCCAUUUGUC-3′; si-ANRIL-3,
5′-UCUGUUUAAAUUAUGAAUGUG-3′; si-NC, 5′-TTCTCCGAACGTGTCACGT-3′;
miR-98 mimics, 5′-UGAGGUAGUAAGUUGUAUUGUU-3′; and miR-NC,
5′-UAAAGUGCUUAUAGUGCAGGUAG-3′. A549 and A549/DDP cells were seeded
in 6-well plates at a density of 1×105 cells/well and
grown to 70% confluence prior to transfection. According to
manufacturer's instructions, cells were transfected with
oligonucleotides (40 nM) or plasmids (2 µg/ml) using
Lipofectamine® 2000 transfection reagent (Invitrogen;
Thermo Fisher Scientific, Inc.) at room temperature, and the
transfected cells were cultured for the indicted times at 37°C in a
humidified atmosphere with 5% CO2.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from tissues and cultured
cells using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). The first strand of complementary DNA (cDNA) was
synthesized from ~1 µg of the extracted total RNA sample using the
Prime-Script RT reagent kit (Takara Bio, Inc.) at 37°C for 15 min,
85°C for 5 sec and 4°C until the removal of the RT products. The
expression levels of ANRIL and miR-98 were quantified PCR using the
TaqMan Power SYBR® Green PCR Mix (Invitrogen; Thermo
Fisher Scientific, Inc.) and the SYBR® Green Master Mix
(cat#: 4444556; Thermo Fisher Scientific), respectively. The qPCR
was performed using the Applied Biosystems 7500 Real-time PCR
system (Applied Biosystems; Thermo Fisher Scientific, Inc.) under
the following thermocycling conditions: 94°C for 2 min; followed by
35 cycles of 94°C for 45 sec, 56°C for 30 sec and 72°C for 45 sec;
and an extension at 72°C for 10 min. The relative gene expression
levels were calculated and normalized to the expression of
GAPDH (for lncRNAs) or U6 small nuclear RNA (for
miRNAs) using the 2−ΔΔCq method (22). The primers used in the present study
are listed in Table II.
 | Table II.Primers used for reverse
transcription-quantitative PCR. |
Table II.
Primers used for reverse
transcription-quantitative PCR.
| Primer | Sequences
(5′→3′) |
|---|
| U6 | F:
CTCGCTTCGGCAGCACA |
|
| R:
AACGCTTCACGAATTTGCGT |
| miR-98 | F:
TGAGGTAGTAAGTTGTAT |
|
| R:
AACATGTACAGTCCATGGATG |
| ANRIL | F:
CTTATTTTATTCCTGGCTCCCCT |
|
| R:
ATCATTCTCCTCAAATTACAGAG |
| GAPDH | F:
GTCAACGGATTTGGTCTGTATT |
|
| R:
AGTCTTCTGGGTGGCAGTGAT |
Western blot assay
Cultured A549 and A549/DDP cells were lysed using a
Radio-Immunoprecipitation Assay (RIPA) protein extraction reagent
(Cell Signaling Technology, Inc.) containing phenylmethanesulfonyl
fluoride. The bicinchoninic acid (BCA) protein assay was performed
to determine the protein concentration using a BCA assay kit
(Thermo Fisher Scientific, Inc.). Protein extracts (~30 µg/lane)
were separated by SDS-PAGE (10% gel) and transferred to
polyvinylidene difluoride membranes (EMD Millipore). The membranes
were blocked with 5% non-fat milk for 1 h at room temperature. The
membranes were incubated with primary antibodies against Ki67
(1:1,500; cat. no. sc-23900; Santa Cruz Biotechnology, Inc.),
cleaved caspase-3 (1:2,000; cat. no. 9661; Cell Signaling
Technology, Inc.), mitogen-activated protein kinase kinase (MEK;
1:1,000; cat. no. 4694; Cell Signaling Technology, Inc.),
phosphorylated (p-)MEK (1:1,000; cat. no. 2338; Cell Signaling
Technology, Inc.), mitogen-activated protein kinase 1/2 (ERK1/2;
1:1,000; cat. no. ab17942; Abcam), p-ERK1/2 (1:2,000; #4370; Cell
Signaling Technology) or β-actin (1:1,500; cat. no. ZRB1312;
Sigma-Aldrich; Merck KGaA) at 4°C overnight. The membranes were
washed twice with PBS and incubated with a horseradish
peroxidase-conjugated anti-mouse IgG secondary antibody (1:1,000;
cat. no. sc-2357; Santa Cruz Biotechnology, Inc.) and visualized
using an ECL system (Amersham; Cytiva). The data were quantified
using ImageJ software (version 1.8.0.112; National Institutes of
Health).
MTT assay
A549 and A549/DDP cells (2×103) at the
logarithmic growth phase were plated into 96-well plates at 48 h
post-transfection and treated with or without 1 or 2 µg/ml DDP at
37°C for 0, 24, 48 and 72 h. Following treatment, 20 µl MTT
solution (5 mg/ml; Sigma-Aldrich; Merck KGaA) was added into each
well for 4 h at 37°C. Subsequently, the supernatant of each well
was removed, and 150 µl DMSO (Sigma-Aldrich; Merck KGaA) was added
to dissolve the formazan crystal. The absorbance value at 490 nm
was detected using a plate reader (Bio-Rad Laboratories, Inc.).
Apoptosis analysis by flow
cytometry
The transfected A549 and A549/DDP cells were
incubated with culture medium with or without 1 or 2 µg/ml DDP at
37°C for 48 h. For apoptosis analysis, the cells were digested
using trypsin without EDTA for 1–2 min, and 1 ml RPMI-1640 medium
with 10% FBS were added to terminate the digestion. Then, the cells
were centrifuged at 111 × g for 5 min at 4°C and collected. The
collected cells were double-stained with a Annexin V-FITC Apoptosis
Detection kit (BD Biosciences) according to the manufacturer's
protocol. The apoptotic cells were analyzed using a FACScan flow
cytometer (Becton, Dickinson and Company) with Cell Quest software
version 7.5.3 (BD Biosciences). All experiments were repeated in
triplicate.
Dual luciferase reporter assay
Prediction using bioinformatics online software
miRcode (http://www.mircode.org/) and Starbase
2.0 (http://starbase.sysu.edu.cn/mirLncRNA.php) identified
that miR-98 contained putative binding sites with ANRIL. The
fragments of ANRIL containing the potential miR-98 binding sites
were amplified and cloned into pGL3 plasmids (Promega Corporation)
to produce an ANRIL-WT plasmid. The luciferase reporter plasmids
containing the mutated miR-98 binding sites in ANRIL were
constructed using the Quick-Change Site Directed Mutagenesis kit
(Invitrogen; Thermo Fisher Scientific, Inc.) and termed ANRIL-MUT.
For the luciferase reporter assay, cells transfected with 20 nM
miR-98 mimics (miR-98) and miR-NC were seeded into 96-well plates
at a density of 1,000 cells/well and transfected with the vector,
ANRIL-WT or ANRIL-MUT (50 ng) and the internal control
Renilla luciferase plasmid using Lipofectamine®
2000 transfection reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) at 37°C for 48 h. At 48 h post-transient transfection, the
luciferase activity was measured with a Dual-Luciferase Reporter
Assay System (Promega Corporation) and normalized to Renilla
luciferase activity.
RNA immunoprecipitation (RIP)
In order to confirm the association between ANRIL
and miR-98, RIP assay was performed using a Magna RIP™ RNA-Binding
Protein Immunoprecipitation kit (EMD Millipore). A549 and A549/DDP
cells (1×107) were lysed using complete RNA lysis buffer
containing 1 mM PMSF (Sigma-Aldrich; Merck KGaA), 1 mM DTT
(Invitrogen; Thermo Fisher Scientific, Inc.), 1% protease inhibitor
(Sigma-Aldrich; Merck KGaA) and 200 U/ml RNase inhibitor. Then, 100
µl whole cell lysis solution was co-incubated with the RIP buffer
containing A + G magnetic beads conjugated with 8 µg anti-human
argonaute 2 (Ago2) antibody (1:40; cat. no. ab186733; Abcam) and
normal mouse IgG (1:30; cat. no. AP503P; EMD Millipore) as a
negative control. To avoid non-specific binding, the samples were
incubated with 10 mg/ml Proteinase K, and the immunoprecipitated
RNA was isolated using TRIzol® reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). Co-precipitated RNAs were
subjected to RT-qPCR analysis as aforementioned.
Statistical analysis
Data are presented as the mean ± standard deviation
(SD). All experiments were repeated separately at least three
times. Statistical analysis was performed using SPSS 13.0 (SPSS,
Inc.). Differences among three or more groups were analyzed by
one-way analysis of variance with Tukey's post hoc test.
Correlation analysis was performed by Pearson's correlation test.
Significant differences between two groups were estimated by
unpaired and paired two-tailed Student's t-test. P<0.05 was
considered to indicate a statistically significant difference.
Results
ANRIL is upregulated and miR-98 is
downregulated in lung cancer compared with healthy tissues
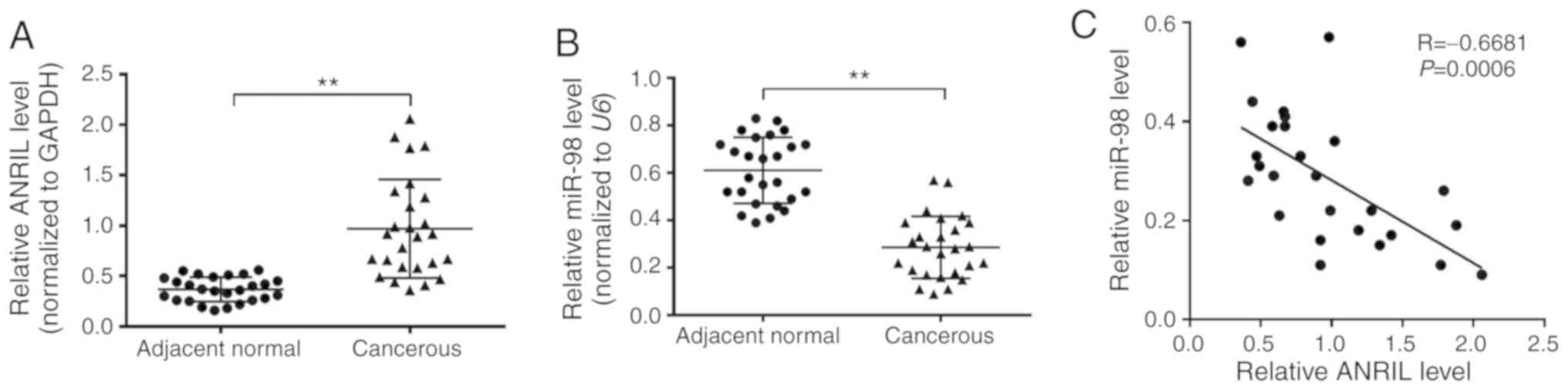
In order to investigate whether the expression
levels of ANRIL and miR-98 were altered in NSCLC tissues, the
present study first detected the expression levels in 26 pairs of
NSCLC tissues and paired adjacent normal lung tissues. RT-qPCR
analysis revealed that compared with the normal tissues, NSCLC
tissues exhibited a significant increase in ANRIL expression and a
significant decrease in miR-98 expression (Fig. 1A and B). In addition, Pearson's
correlation analysis revealed that the expression of ANRIL was
significantly negatively correlated with the expression of miR-98
in lung cancer tissues (Fig. 1C).
These results suggested that abnormal expression of ANRIL and
miR-98 may serve crucial roles in the development of NSCLC.
ANRIL is upregulated and miR-98 is
downregulated in DDP-resistant lung cancer cells
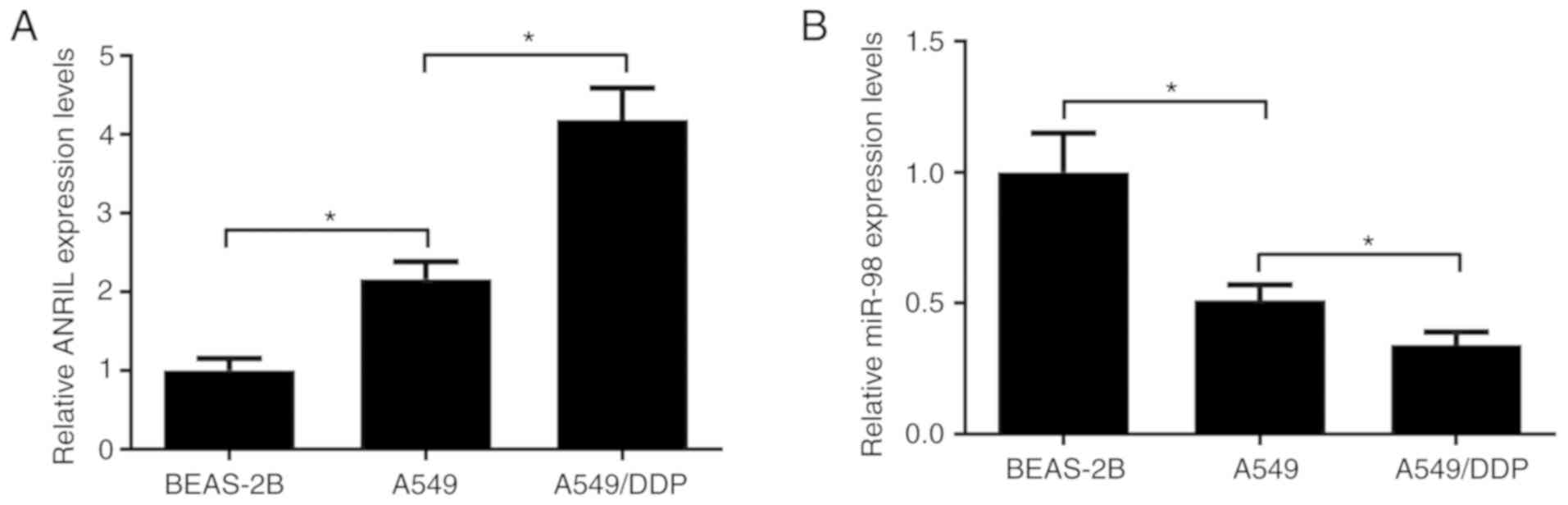
The expression levels of ANRIL and miR-98 in
parental lung cancer cells A549 and DDP-resistant A549/DDP cells
were assessed by RT-qPCR. Compared with normal lung epithelial
cells BEAS-2B, ANRIL expression was significantly increased in A549
cells (Fig. 2A). miR-98 expression
was significantly decreased in A549 cells compared with that in
BEAS-2B cells (Fig. 2B).
Furthermore, ANRIL expression in A549/DDP cells was higher compared
with that in their parental A549 cell line (Fig. 2A), and miR-98 expression was lower
in A549/DDP cells compared with that in A549 cells (Fig. 2B). These results suggested that
ANRIL and miR-98 may be involved in the development of DDP
resistance in lung cancer cells.
ANRIL overexpression promotes DDP
resistance in lung cancer cells
To determine the functional roles of ANRIL in DDP
resistance of lung cancer, ANRIL was overexpressed in A549 cells by
transfecting them with pcDNA-ANRIL or an empty control vector. The
results of the RT-qPCR analysis revealed that A549 cells
transfected with pcDNA-ANRIL exhibited a >20-fold increase in
ANRIL expression compared with vector-treated cells (Fig. 3A). As presented in Fig. 3B, the MTT assay revealed that DDP
administration led to a marked decrease in cell proliferation at 48
and 72 h in A549 cells, whereas ectopic expression of ANRIL
significantly alleviated the inhibitory effects of DDP on cell
proliferation at 48 and 72 h. In addition, DDP administration led
to a marked decrease in Ki67 expression, and overexpression of
ANRIL alleviated the inhibitory effects of DDP on Ki67 expression
in A549 cells (Fig. 3C). The
results of the present study demonstrated that ANRIL overexpression
significantly alleviated the inhibitory effects of DDP on the
activation of the MEK/ERK pathway (Fig.
3D). Flow cytometry analysis results demonstrated that DDP
treatment induced the apoptosis of A549 cells, whereas
overexpression of ANRIL partially impeded the DDP-induced apoptosis
in A549 cells compared with that in the vector group (Fig. 3E). In addition, DDP treatment
resulted in increased expression of the apoptosis-associated
protein cleaved caspase-3, and ANRIL overexpression partially
reversed the promotive effects of DDP on cleaved caspase-3
expression in A549 cells (Fig. 3F).
Collectively, these results demonstrated that ANRIL overexpression
increased DDP resistance in lung cancer cells.
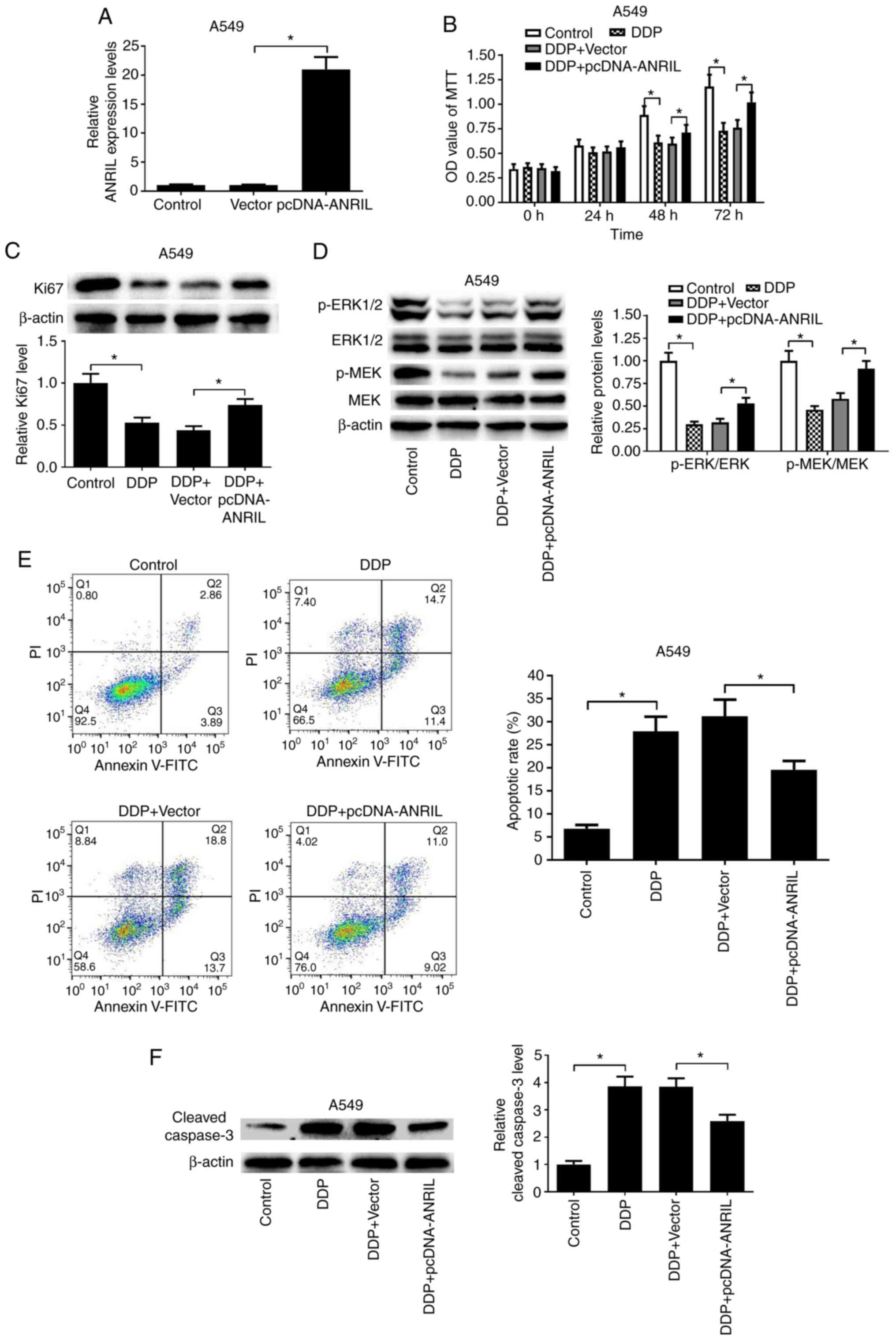 | Figure 3.ANRIL overexpression enhances DDP
resistance in lung cancer cells. A549 cells were transfected with
pcDNA-ANRIL or an empty vector. (A) The expression levels of ANRIL
in transfected A549 cells were estimated by reverse
transcription-quantitative PCR. (B) MTT assay was performed to
detect cell proliferation after the transfected A549 cells were
treated with 1 µg/ml DDP for 0, 24, 48 and 72 h. (C and D) Western
blotting was performed to assess the expression levels of Ki67,
MEK, p-MEK, ERK1/2 and p-ERK1/2 after the transfected A549 cells
were exposed to 1 µg/ml DDP for 48 h. (E) Flow cytometry analysis
was performed to determine the apoptotic rates after the
transfected A549 cells were treated with 1 µg/ml DDP for 48 h. (F)
Western blot analysis of the expression of cleaved caspase-3 was
performed after the transfected A549 cells were exposed to 1 µg/ml
DDP for 48 h. *P<0.05. ANRIL, antisense non-coding RNA in the
inhibitor of cyclin-dependent kinase 4 locus; DDP, cisplatin; ERK,
mitogen-activated protein kinase; MEK-mitogen-activated protein
kinase kinase; p, phosphorylated; PI, propidium iodide. |
ANRIL knockdown inhibits DDP
resistance in A549/DDP cells
To further confirm the association between ANRIL and
DDP resistance in lung cancer cells, ANRIL-knockdown A549/DDP cells
were established by siRNA. As presented in Fig. 4A, si-ANRIL-1, si-ANRIL-2 and
si-ANRIL-3 effectively reduced ANRIL expression in A549/DDP cells
compared with that in the si-NC group. si-ANRIL-3 exhibited the
highest knockdown efficiency and thus was selected for further
experiments. MTT assay results revealed that cell proliferation
following DDP treatment was repressed at 48 and 72 h in A549/DDP
cells compared with that in the untreated control, and ANRIL
knockdown exacerbated the DDP-induced cell proliferation inhibition
at 24, 48 and 72 h (Fig. 4B). In
addition, compared with that in the control group, DDP treatment
significantly inhibited the expression of Ki67, and ANRIL knockdown
significantly enhanced this inhibitory effect in A549/DDP cells
(Fig. 4C). Knockdown of ANRIL also
promoted the inhibitory effects of DDP on the activation of the
MEK/ERK pathway (Fig. 4D). In
addition, DDP treatment significantly promoted the apoptosis of
A549 cells compared with that in the control group, whereas ANRIL
knockdown reinforced the DDP-induced apoptosis, as demonstrated by
flow cytometry analysis (Fig. 4E).
Additionally, DDP treatment significantly induced the expression of
cleaved caspase-3 compared with that in the control group, and
ANRIL knockdown enhanced these effects in A549/DDP cells (Fig. 4F). Taken together, these results
revealed that ANRIL knockdown significantly inhibited DDP
resistance in A549/DDP cells.
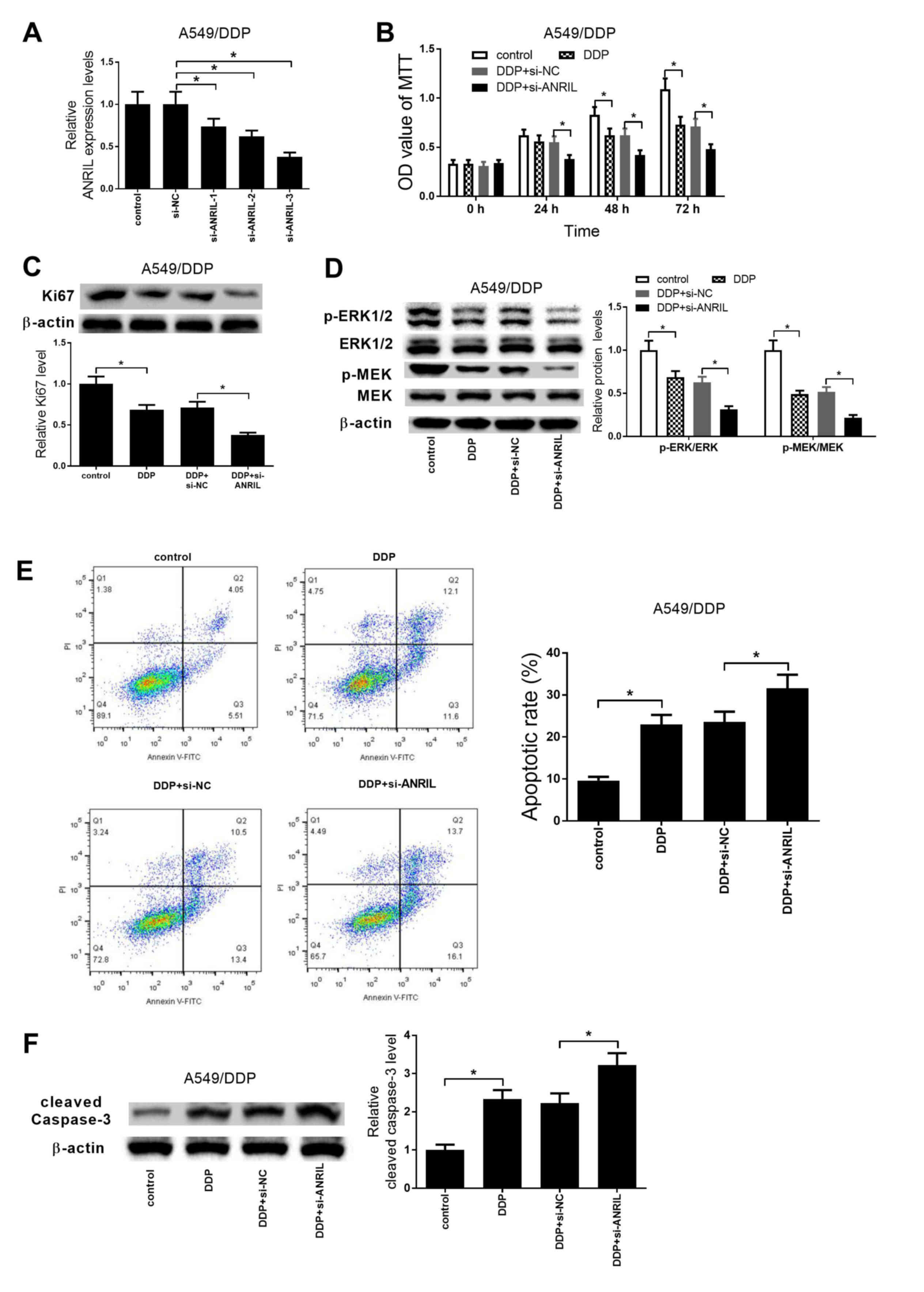 | Figure 4.ANRIL knockdown significantly
inhibits DDP resistance in A549/DDP cells. (A) The expression of
ANRIL in A549/DDP cells transfected with si-NC, si-ANRIL-1,
si-ANRIL-2 or si-ANRIL-3 was examined by reverse
transcription-quantitative PCR. (B) MTT assay was applied to
determine the cell proliferation after A549 cells were transfected
with si-ANRIL or si-NC, followed by treatment with 2 µg/ml DDP for
0, 24, 48 and 72 h. (C and D) Western blot analysis of the
expression of Ki67, MEK, p-MEK, ERK1/2 and p-ERK1/2 was performed
after the transfected A549/DDP cells were exposed to 2 µg/ml DDP
for 48 h. (E) Flow cytometry analysis was conducted to detect
apoptosis after si-ANRIL- or si-NC-transfected A549/DDP cells were
treated with 2 µg/ml DDP for 48 h. (F) Western blot analysis of the
expression of cleaved caspase-3 was performed after the transfected
A549/DDP cells were exposed to 2 µg/ml DDP for 48 h. *P<0.05.
ANRIL, antisense non-coding RNA in the inhibitor of
cyclin-dependent kinase 4 locus; siRNA, small interfering RNA; NC,
negative control; DDP, cisplatin; ERK, mitogen-activated protein
kinase; MEK-mitogen-activated protein kinase kinase; p,
phosphorylated; PI, propidium iodide. |
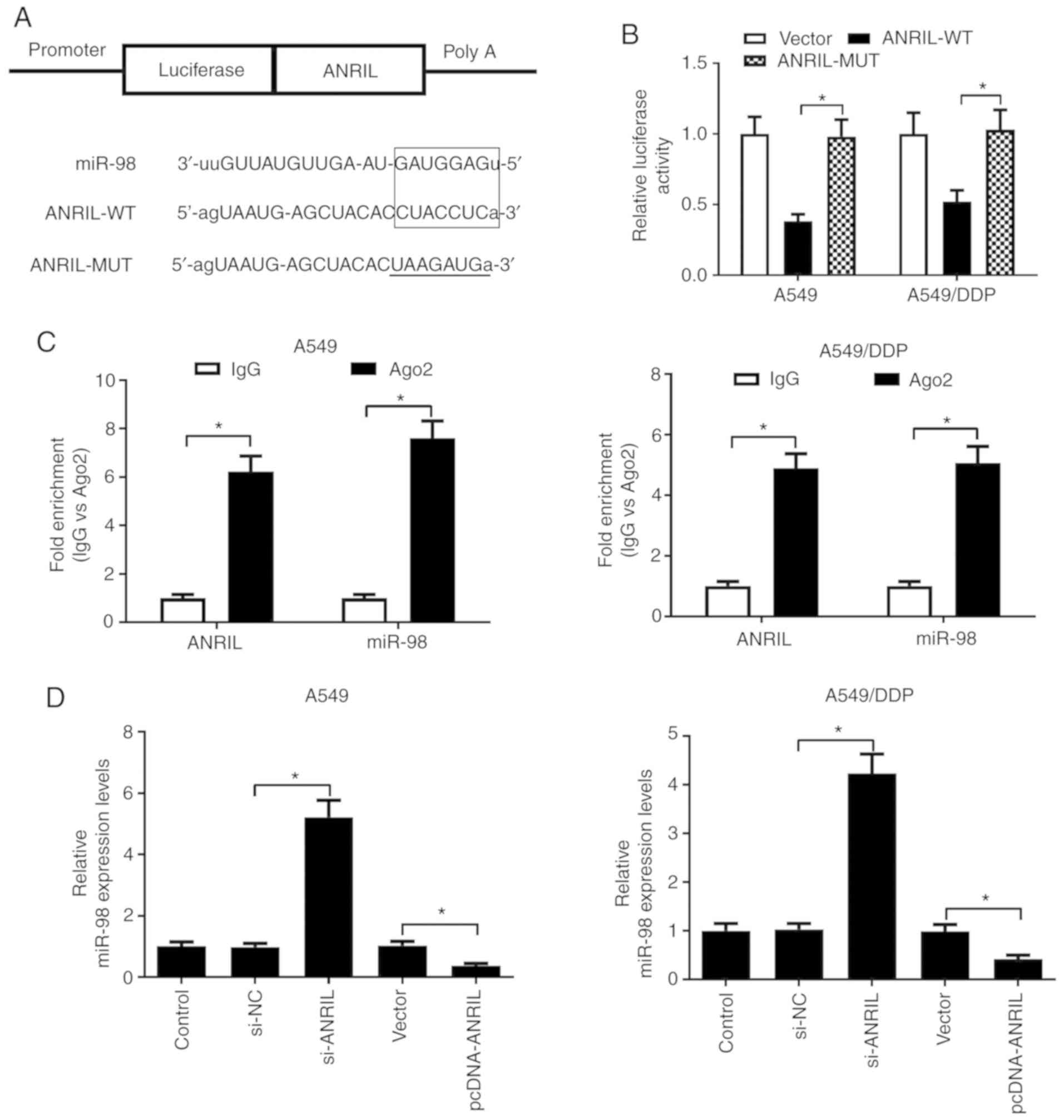
ANRIL directly interacts with miR-98
and suppresses miR-98 expression in lung cancer cells
In view of the inverse expression trend of ANRIL and
miR-98 in the parental and DDP-resistant lung cancer cells, the
present study further investigated the potential interaction
between ANRIL and miR-98. Through bioinformatics online software
analyses by miRcode and Starbase 2.0, the present study discovered
that miR-98 contained putative binding sites with ANRIL, as
presented in Fig. 5A. The
luciferase reporter assay results revealed that miR-98
significantly repressed the luciferase activity in A549 and
A549/DDP cells transfected with ANRIL-WT, but did not suppress
luciferase activity in A549 and A549/DDP cells transfected with
ANRIL-MUT (Fig. 5B), suggesting a
direct interaction between miR-98 and ANRIL. To confirm whether
ANRIL and miR-98 were in the same RNA-induced silencing complex
(RISC), a RIP assay was performed in A549 and A549/DDP cell
extracts using an antibody against Ago2, and the immunoprecipitated
RNA levels were assessed by RT-qPCR. ANRIL and miR-98 were enriched
in the Ago2 pellet in both A549 and A549/DDP cell extracts relative
to the control IgG immunoprecipitates (Fig. 5C). In order to further investigate
the regulatory effect of ANRIL on miR-98 expression, RT-qPCR was
used to evaluate the expression of miR-98 in A549 and A549/DDP
cells transfected with pcDNA-ANRIL, si-ANRIL or the respective
controls. The results demonstrated that ANRIL knockdown increased,
whereas overexpression of ANRIL decreased miR-98 expression in A549
and A549/DDP cells (Fig. 5D).
Collectively, these results demonstrated that ANRIL may function as
a molecular sponge of miR-98 and inhibit its expression in lung
cancer cells.
ANRIL-miR-98 axis participates in the
regulation of DDP resistance in DDP-resistant lung cancer
cells
Since upregulation of miR-98 was demonstrated to
sensitize lung cancer cells to DDP, the present study investigated
the functional effects of the ANRIL-miR-98 axis on DDP resistance
of lung cancer cells. A549/DDP cells were transfected with miR-98
mimics, and miR-98 expression levels were significantly elevated in
the miR-98 transfected A549/DDP cells compared with those in the
miR-NC group (Fig. 6A). As
presented in Fig. 6B and C, miR-98
mimics promoted the inhibitory effects of DDP on cell proliferation
and Ki67 expression in A549/DDP cells, which was partially reversed
by overexpression of ANRIL, which was determined by MTT and western
blot assays. In addition, miR-98 mimics promoted the inhibitory
effects of DDP on the MEK/ERK signaling pathway, which was
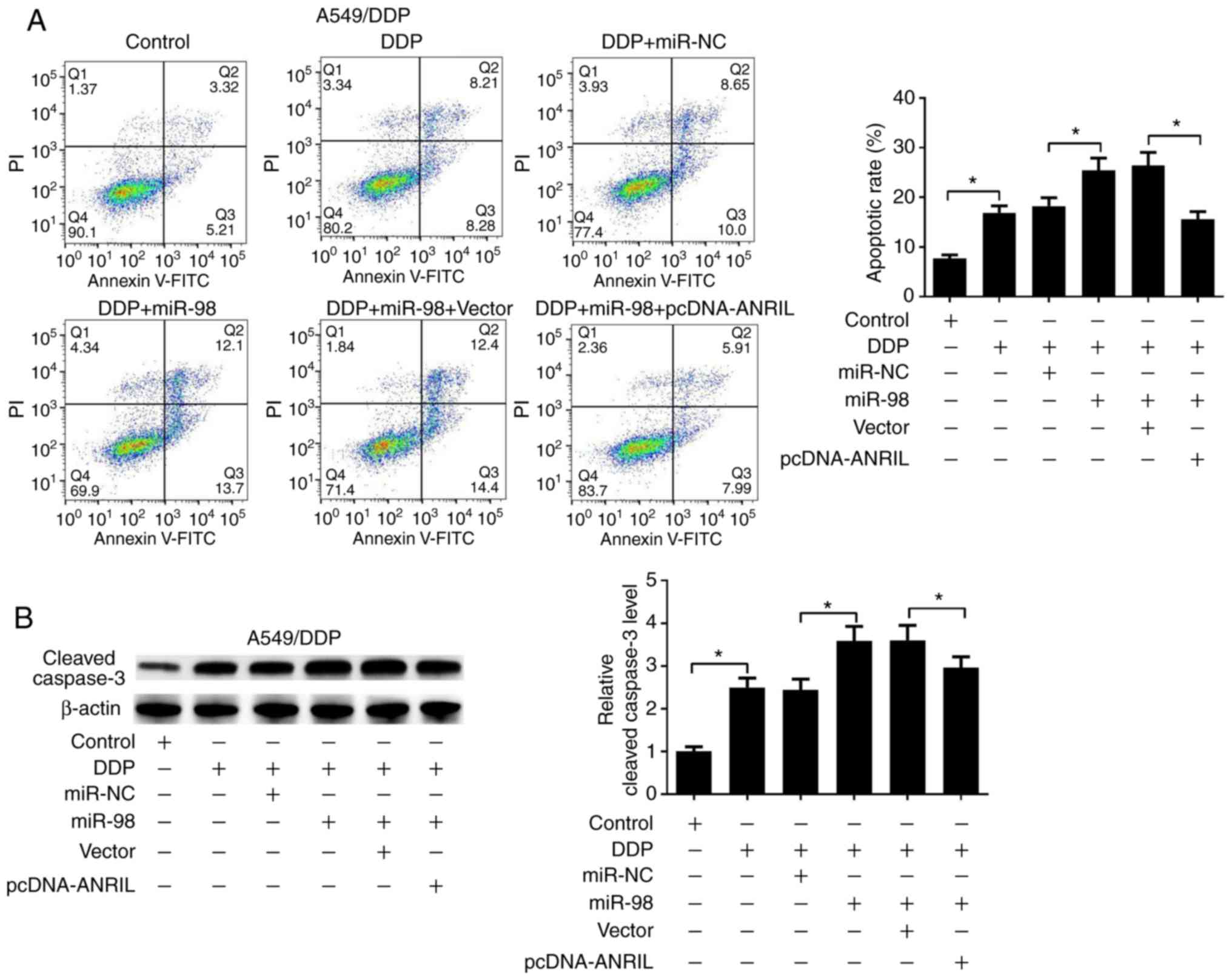
partially reversed by ANRIL overexpression (Fig. 6D). Additionally, flow cytometry
analysis results demonstrated that miR-98 mimics significantly
enhanced the DDP-induced apoptosis of A549/DDP cells, whereas ANRIL
overexpression abolished the promotive effect of miR-98 on the
DDP-induced apoptosis (Fig. 7A).
Furthermore, cleaved caspase-3 expression was upregulated in
A549/DDP cells transfected with miR-98 mimics, and ANRIL
overexpression partially inversed this phenomenon (Fig. 7B). Therefore, the present study
concluded that ANRIL overexpression reversed miR-98
overexpression-mediated DDP resistance repression in DDP-resistant
lung cancer cells.
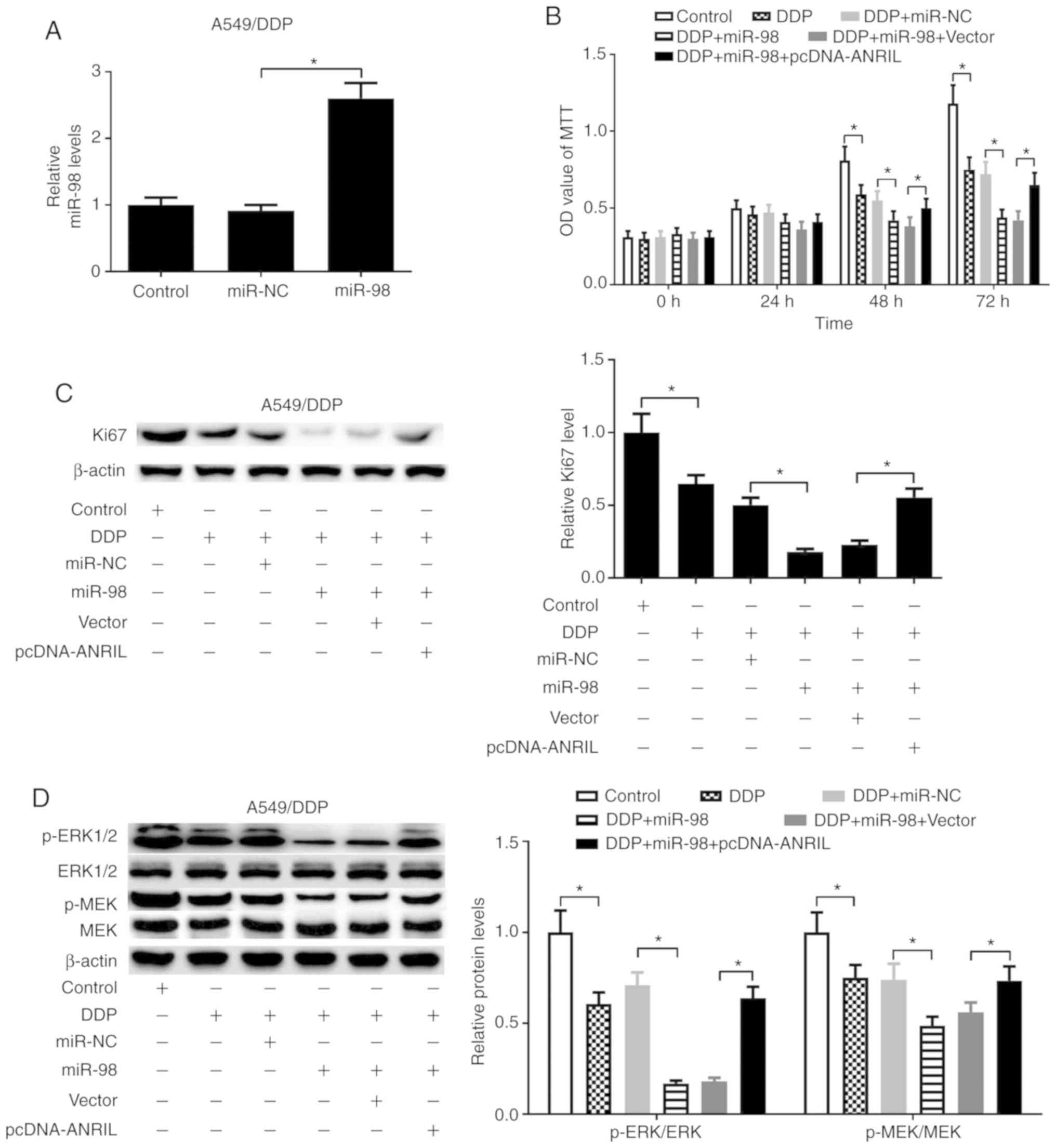 | Figure 6.ANRIL-miR-98 axis regulates DDP
resistance in DDP-resistant lung cancer cells by modulating cell
proliferation. A549/DDP cells were transfected with miR-NC, miR-98
or combined with the vector or pcDNA-ANRIL and cultured for 48 h.
(A) The expression of miR-98 was determined by reverse
transcription-quantitative PCR in A549/DDP cells transfected with
miR-NC or miR-98 at 48 h. (B) Cell proliferation was determined by
MTT assay in transfected A549/DDP cells following 2 µg/ml DDP
treatment for 0, 24, 48 or 72 h. (C and D) Western blotting was
used to detect the expression levels of Ki67, MEK, p-MEK, ERK1/2
and p-ERK1/2 in the transfected A549/DDP cells treated with or
without 2 µg/ml DDP for 48 h. *P<0.05. ANRIL, antisense
non-coding RNA in the inhibitor of cyclin-dependent kinase 4 locus;
miR, microRNA; NC, negative control; DDP, cisplatin; ERK,
mitogen-activated protein kinase; MEK-mitogen-activated protein
kinase kinase; p, phosphorylated. |
Discussion
Accumulating evidence has demonstrated that
dysregulated lncRNAs are involved in various aspects of cancer
biology and serve functional roles in the development of drug
resistance in numerous types of cancer, including lung cancer
(23,24). For example, lncRNA regulator of
reprogramming (ROR) has been demonstrated to be expressed at high
levels in A549/DDP cells, and ROR silencing improves the
sensitivity of NSCLC to DDP by inhibiting the PI3K/Akt/mTOR
signaling pathway (25). In
addition, lncRNA HOTAIR has been demonstrated to be aberrantly
upregulated in A549/DDP cells, and inhibition of HOTAIR expression
increases the sensitivity of A549/DDP cells to DDP (26). LncRNA maternally expressed gene 3
(Meg3) is revealed to be downregulated in A549/DDP cells and
enhance DDP resistance in lung cancer cells through the activation
of the WNT/β-catenin signaling pathway (27). However, the potential function of
ANRIL in regulating DDP resistance in lung cancer remains
unclear.
An increasing amount of evidence has reported that
ANRIL is deregulated and exerts oncogenic activities in the
tumorigenesis and drug resistance of several types of cancer. For
instance, ANRIL has been demonstrated to be upregulated in
nasopharyngeal carcinoma (NPC) cells; knockdown of ANRIL represses
the proliferation, promotes the apoptosis, improves the
radiosensitivity and enhances the DDP-induced cytotoxicity in NPC
cells (28,29). ANRIL is highly expressed in gastric
cancer tissues from DDP- and 5-fluorouracil (5-FU)-resistant
patients, and knockdown of ANRIL in gastric cancer cells inhibits
the development of multidrug resistance (30). The results of the present study
demonstrated that ANRIL was upregulated in lung cancer tissues and
A549/DDP cells compared with normal lung tissues and cells.
Functional analyses demonstrated that ANRIL overexpression reversed
the DDP-induced suppression of cell proliferation and apoptosis,
whereas ANRIL knockdown exhibited the opposite effects, suggesting
that ANRIL participated in the development of DDP resistance in
lung cancer cells. Similarly, a previous study has demonstrated
that ANRIL functions as a potential oncogene as it is upregulated
and promotes the acquisition of paclitaxel resistance in lung
adenocarcinoma A549 cells (31).
Although in the present study ANRIL was demonstrated
to be involved in DDP resistance of lung cancer cells, the
underlying mechanism remains to be further elucidated. It has been
reported that miRNAs exert their function through the RISC that
contains Ago2 (32). A ceRNA
regulatory hypothesis has been proposed, which suggests that
lncRNAs serve an important role in multiple processes of cancer
cells by functioning as endogenous miRNA sponges and regulating
miRNA expression and biological functions (33). For example, a lncRNA termed
upregulated in CRC may act as an endogenous sponge by competing for
miR-143 to regulate the targets of this miRNA, therefore promoting
colorectal cancer progression (34). LncRNA urothelial
carcinoma-associated 1 enhances the cell proliferation and 5-FU
resistance in colorectal cancer by sponging endogenous miR-204-5p
(32). ANRIL acts as a molecular
sponge of miR-186 to contribute to cervical cancer tumorigenesis
(35). In pediatric
medulloblastoma, ANRIL inhibition represses cell proliferation and
migration, but promotes apoptosis by acting as a molecular sponge
of miR-323 (36). In addition,
ANRIL knockdown represses tumorigenicity and enhances DDP
sensitivity in NPC cells by negatively regulating miRNA let-7a
(37). The results of the present
study demonstrated that in NSCLC tissues, there was a significant
upregulation in ANRIL expression and downregulation in miR-98
expression compared with those in adjacent normal tissues. Of note,
ANRIL expression exhibited a significant negative correlation with
that of miR-98 in lung cancer tissues. In order to improve the
current understanding of the underlying mechanism of the
lncRNA/miRNA regulatory function, the present study performed RIP
assay, luciferase reporter assay and RT-qPCR analysis, and
demonstrated that ANRIL functioned as a molecular sponge of miR-98
in lung cancer cells. In accordance with a previous study (20), the present study confirmed that
miR-98 was downregulated in A549/DDP cells, and miR-98 mimics
promoted the DDP-induced cell proliferation inhibition and
apoptosis of A549/DDP cells. Certain studies have reported that the
MEK/ERK signaling pathway is involved in the progression and drug
resistance of multiple types of cancer (29,38).
In the present study, the phosphorylation levels of MEK and ERK
were reduced by DDP treatment in lung cancer cells, and ANRIL
negatively regulated the miR-98 mimic-induced repression on the
MEK/ERK signaling pathway. Mechanistic analyses further revealed
that overexpression of ANRIL reversed the miR-98 mimic-induced
suppression of DDP resistance in lung cancer cells, indicating that
ANRIL regulated the development of DDP resistance by functioning as
a ceRNA of miR-98.
In the present study, due to the limitation of the
conditions, the animal experiments to confirm the results in
vivo have not been performed. These experiments will be
performed in our future study.
In summary, the present study demonstrated the
upregulation of ANRIL and downregulation of miR-98 in lung cancer
tissues and DDP-resistant lung cancer cells compared with normal
lung tissues and cells. Functional and mechanistic analyses
demonstrated that ANRIL knockdown inhibited the development of DDP
resistance by functioning as a ceRNA of miR-98, providing novel
insights into the molecular mechanism of ANRIL involved in the DDP
resistance of lung cancer cells. The present study reported that
ANRIL may be a novel potential therapeutic target to overcome DDP
resistance in lung cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets generated and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
XW, GZ, ZC, LD and ML designed the study, performed
the experiments and drafted the manuscript. XJ, HW and RZ analyzed
the data and revised the manuscript. TJ, YY, LJ and MY provided
technical support and revised the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was reviewed and approved by the
Research Ethics Committee of the First Affiliated Hospital of
Zhengzhou University. Written consent was obtained from all
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Riaz MK, Zhang X, Wong KH, Chen H, Liu Q,
Chen X, Zhang G, Lu A and Yang Z: Pulmonary delivery of transferrin
receptors targeting peptide surface-functionalized liposomes
augments the chemotherapeutic effect of quercetin in lung cancer
therapy. Int J Nanomedicine. 14:2879–2902. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Romero-Vielva L, Viteri S, Moya-Horno I,
Toscas JI, Maestre-Alcácer JA, Ramón Y, Cajal S and Rosell R:
Salvage surgery after definitive chemo-radiotherapy for patients
with non-Small cell lung cancer. Lung Cancer. 133:117–122. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen QY, Jiao DM, Wang J, Hu H, Tang X,
Chen J, Mou H and Lu W: miR-206 regulates cisplatin resistance and
EMT in human lung adenocarcinoma cells partly by targeting MET.
Oncotarget. 7:24510–24526. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guo L, Song P, Xue X, Guo C, Han L, Fang
Q, Ying J, Gao S and Li W: Variation of programmed death ligand 1
expression after platinum-based neoadjuvant chemotherapy in lung
cancer. J Immunother. 42:215–220. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang Q, Zhang Z, Xu H and Ma C: Lidocaine
alleviates cytotoxicity-resistance in lung cancer A549/DDP cells
via down-regulation of miR-21. Mol Cell Biochem. 456:63–72. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Djebali S, Davis CA, Merkel A, Dobin A,
Lassmann T, Mortazavi A, Tanzer A, Lagarde J, Lin W, Schlesinger F,
et al: Landscape of transcription in human cells. Nature.
489:101–108. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Martens-Uzunova ES, Böttcher R, Croce CM,
Jenster G, Visakorpi T and Calin GA: Long noncoding RNA in
prostate, bladder, and kidney cancer. Eur Urol. 65:1140–1151. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pan JJ, Xie XJ, Li X and Chen W: Long
non-coding RNAs and drug resistance. Asian Pac J Cancer Prev.
16:8067–8073. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang Z, Zhu Z, Watabe K, Zhang X, Bai C,
Xu M, Wu F and Mo YY: Negative regulation of lncRNA GAS5 by miR-21.
Cell Death Differ. 20:1558–1568. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tano K and Akimitsu N: Long non-coding
RNAs in cancer progression. Front Genet. 3:2192012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang EB, Kong R, Yin DD, You LH, Sun M,
Han L, Xu TP, Xia R, Yang JS, De W and Chen Jf: Long noncoding RNA
ANRIL indicates a poor prognosis of gastric cancer and promotes
tumor growth by epigenetically silencing of miR-99a/miR-449a.
Oncotarget. 5:2276–2292. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nie FQ, Sun M, Yang JS, Xie M, Xu TP, Xia
R, Liu YW, Liu XH, Zhang EB, Lu K and Shu YQ: Long noncoding RNA
ANRIL promotes non-small cell lung cancer cell proliferation and
inhibits apoptosis by silencing KLF2 and P21 expression. Mol Cancer
Ther. 14:268–277. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Iranpour M, Soudyab M, Geranpayeh L,
Mirfakhraie R, Azargashb E, Movafagh A and Ghafouri-Fard S:
Expression analysis of four long noncoding RNAs in breast cancer.
Tumour Biol. 37:2933–2940. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wan G, Mathur R, Hu X, Liu Y, Zhang X,
Peng G and Lu X: Long non-coding RNA ANRIL (CDKN2B-AS) is induced
by the ATM-E2F1 signaling pathway. Cell Signal. 25:1086–1095. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu P, Guo M and Hay BA: MicroRNAs and the
regulation of cell death. Trends Genet. 20:617–624. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheng AM, Byrom MW, Shelton J and Ford LP:
Antisense inhibition of human miRNAs and indications for an
involvement of miRNA in cell growth and apoptosis. Nucleic Acids
Res. 33:1290–1297. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ma J, Dong C and Ji C: MicroRNA and drug
resistance. Cancer Gene Ther. 17:523–531. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
MacDonagh L, Gray SG, Finn SP, Cuffe S,
O'Byrne KJ and Barr MP: The emerging role of microRNAs in
resistance to lung cancer treatments. Cancer Treat Rev. 41:160–169.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xiang Q, Tang H, Yu J, Yin J, Yang X and
Lei X: MicroRNA-98 sensitizes cisplatin-resistant human lung
adenocarcinoma cells by up-regulation of HMGA2. Pharmazie.
68:274–281. 2013.PubMed/NCBI
|
|
21
|
Ren K, Li Y, Lu H, Li Z, Li Z, Wu K, Li Z
and Han X: Long noncoding RNA HOTAIR controls cell cycle by
functioning as a competing endogenous RNA in esophageal squamous
cell carcinoma. Transl Oncol. 9:489–497. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia H and Hui KM: Mechanism of cancer drug
resistance and the involvement of noncoding RNAs. Curr Med Chem.
21:3029–3041. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen QN, Wei CC, Wang ZX and Sun M: Long
non-coding RNAs in anti-cancer drug resistance. Oncotarget.
8:1925–1936. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shi H, Pu J, Zhou XL, Ning YY and Bai C:
Silencing long non-coding RNA ROR improves sensitivity of
non-small-cell lung cancer to cisplatin resistance by inhibiting
PI3K/Akt/mTOR signaling pathway. Tumour Biol.
39:10104283176975682017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu MY, Li XQ, Gao TH, Cui Y, Ma N, Zhou Y
and Zhang GJ: Elevated HOTAIR expression associated with cisplatin
resistance in non-small cell lung cancer patients. J Thorac Dis.
8:3314–3322. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xia Y, He Z, Liu B, Wang P and Chen Y:
Downregulation of Meg3 enhances cisplatin resistance of lung cancer
cells through activation of the WNT/β-catenin signaling pathway.
Mol Med Rep. 12:4530–4537. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hu X, Jiang H and Jiang X: Downregulation
of lncRNA ANRIL inhibits proliferation, induces apoptosis, and
enhances radiosensitivity in nasopharyngeal carcinoma cells through
regulating miR-125a. Cancer Biol Ther. 18:331–338. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang Y, Cheng N and Luo J: Downregulation
of lncRNA ANRIL represses tumorigenicity and enhances
cisplatin-induced cytotoxicity via regulating microRNA let-7a in
nasopharyngeal carcinoma. J Biochem Mol Toxicol. 31:e219042017.
View Article : Google Scholar
|
|
30
|
Lan WG, Xu DH, Xu C, Ding CL, Ning FL,
Zhou YL, Ma LB, Liu CM and Han X: Silencing of long non-coding RNA
ANRIL inhibits the development of multidrug resistance in gastric
cancer cells. Oncol Rep. 36:263–270. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu R, Mao Y, Chen K, He W, Shi W and Han
Y: The long noncoding RNA ANRIL acts as an oncogene and contributes
to paclitaxel resistance of lung adenocarcinoma A549 cells.
Oncotarget. 8:39177–39184. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bian Z, Jin L, Zhang J, Yin Y, Quan C, Hu
Y, Feng Y, Liu H, Fei B, Mao Y, et al: LncRNA-UCA1 enhances cell
proliferation and 5-fluorouracil resistance in colorectal cancer by
inhibiting miR-204-5p. Sci Rep. 6:238922016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tay Y, Rinn J and Pandolfi PP: The
multilayered complexity of ceRNA crosstalk and competition. Nature.
505:344–352. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Huang FT, Chen WY, Gu ZQ, Zhuang YY, Li
CQ, Wang LY, Peng JF, Zhu Z, Luo X, Li YH, et al: The novel long
intergenic noncoding RNA UCC promotes colorectal cancer progression
by sponging miR-143. Cell Death Dis. 8:e27782017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang JJ, Wang DD, Du CX and Wang Y: Long
noncoding RNA ANRIL promotes cervical cancer development by acting
as a sponge of miR-186. Oncol Res. 26:345–352. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang H, Wang X and Chen X: Potential role
of long non-coding RNA ANRIL in pediatric medulloblastoma through
promotion on proliferation and migration by targeting miR-323. J
Cell Biochem. 118:4735–4744. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nickols NG, Nazarian R, Zhao SG, Tan V,
Uzunangelov V, Xia Z, Baertsch R, Neeman E, Gao AC, Thomas GV, et
al: MEK-ERK signaling is a therapeutic target in metastatic
castration resistant prostate cancer. Prostate Cancer Prostatic
Dis. 22:531–538. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li Q, Wang C, Wang Y, Sun L, Liu Z, Wang
L, Song T, Yao Y, Liu Q and Tu K: HSCs-derived COMP drives
hepatocellular carcinoma progression by activating MEK/ERK and
PI3K/AKT signaling pathways. J Exp Clin Cancer Res. 37:231. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shroff GS, Viswanathan C, Carter BW,
Benveniste MF, Truong MT and Sabloff BS: Staging lung cancer:
Metastasis. Radiol Clin North Am. 56:411–418. 2018. View Article : Google Scholar : PubMed/NCBI
|