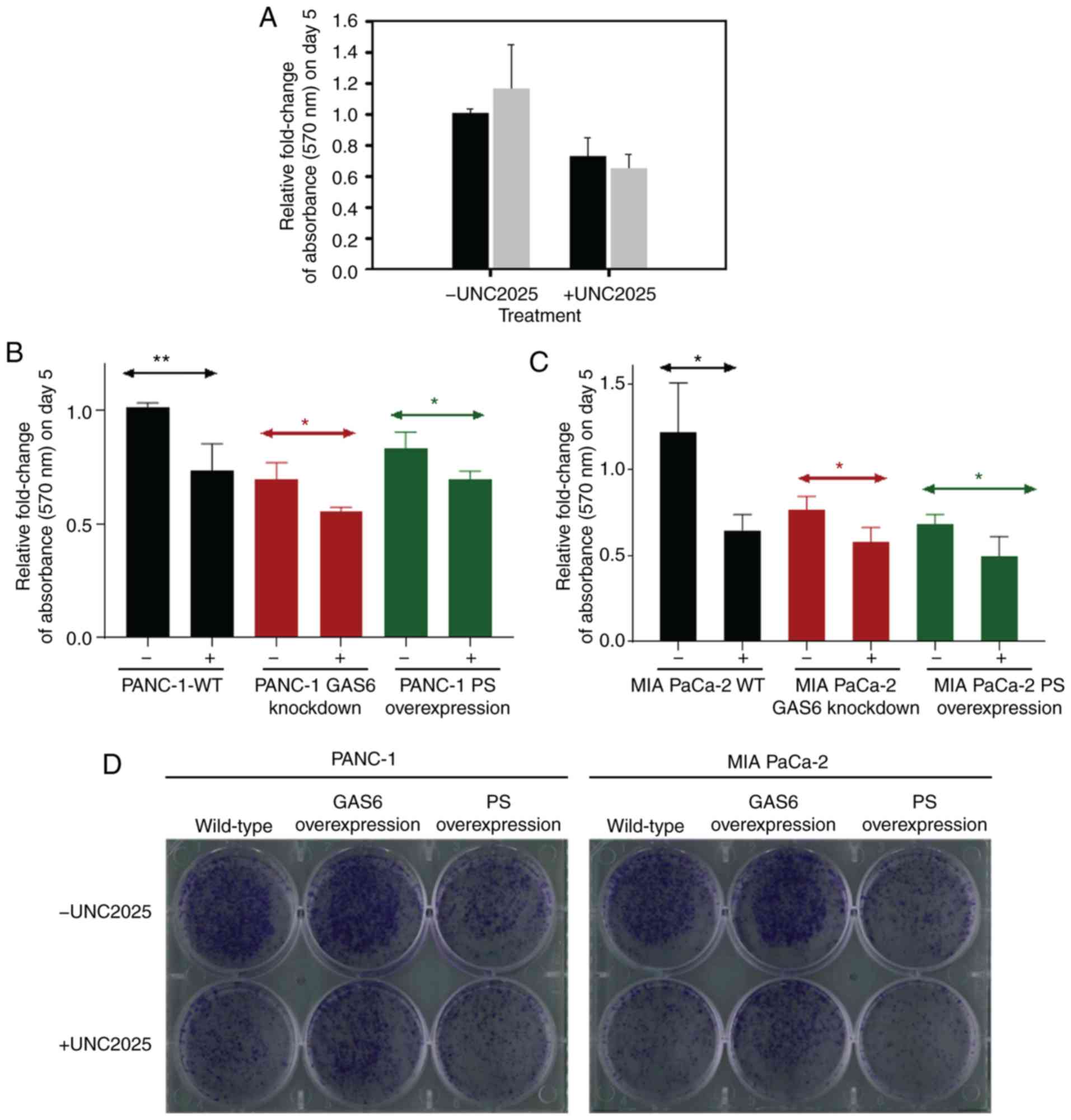
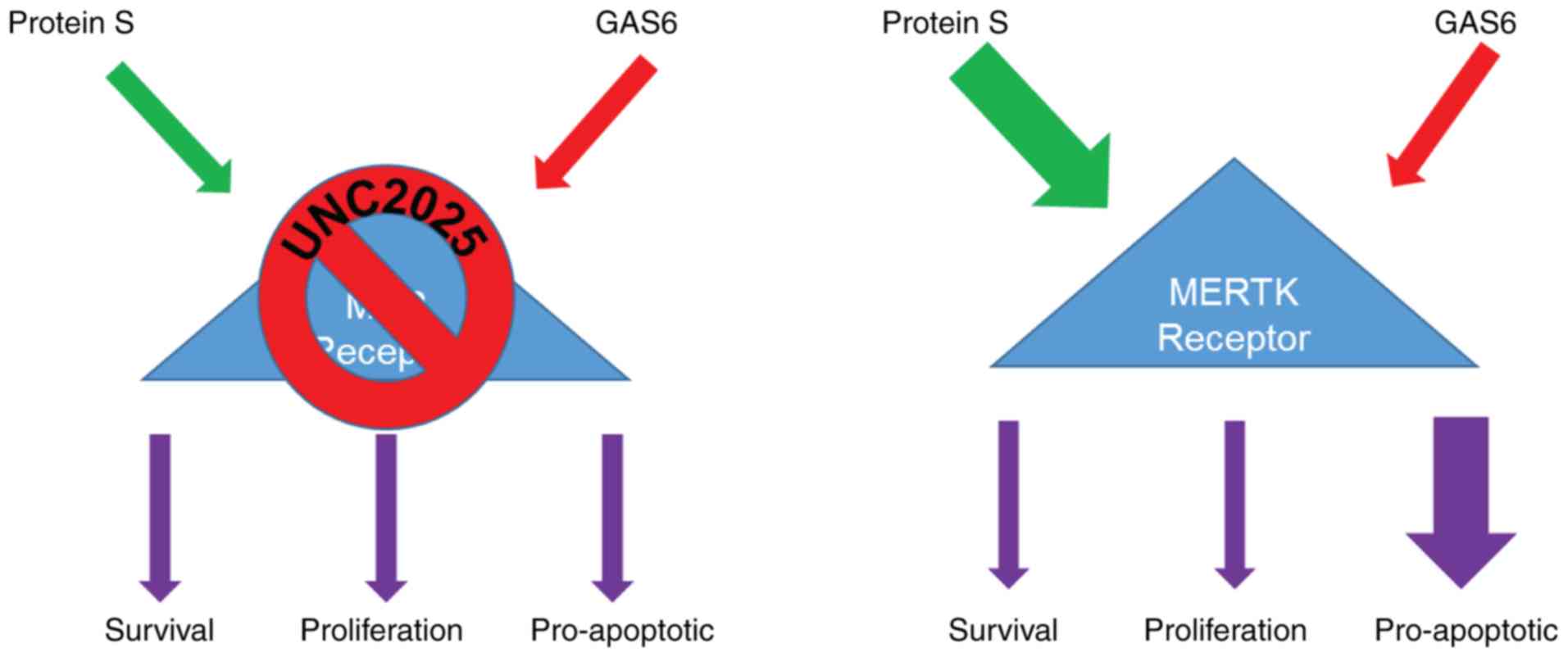
|
1
|
Arnljots B and Dahlbäck B: Protein S as an
in vivo cofactor to activated protein C in prevention of
microarterial thrombosis in rabbits. J Clin Invest. 95:1987–1993.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hackeng TM, Seré KM, Tans G and Rosing J:
Protein S stimulates inhibition of the tissue factor pathway by
tissue factor pathway inhibitor. Proc Natl Acad Sci USA.
103:3106–3111. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rosing J, Maurissen LF, Tchaikovski SN,
Tans G and Hackeng TM: Protein S is a cofactor for tissue factor
pathway inhibitor. Thromb Res. 122 (Suppl 1):S60–S63. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Suleiman L, Négrier C and Boukerche H:
Protein S: A multifunctional anticoagulant vitamin K-dependent
protein at the crossroads of coagulation, inflammation,
angiogenesis, and cancer. Crit Rev Oncol Hematol. 88:637–654. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lemke G: Biology of the TAM receptors.
Cold Spring Harb Perspect Biol. 5:a0090762013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lemke G and Rothlin CV: Immunobiology of
the TAM receptors. Nat Rev Immunol. 8:327–336. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Anderson HA, Maylock CA, Williams JA,
Paweletz CP, Shu H and Shacter E: Serum-derived protein S binds to
phosphatidylserine and stimulates the phagocytosis of apoptotic
cells. Nat Immunol. 4:87–91. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Burstyn-Cohen T: TAM receptor signaling in
development. Int J Dev Biol. 61:215–224. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Burstyn-Cohen T, Heeb MJ and Lemke G: Lack
of protein S in mice causes embryonic lethal coagulopathy and
vascular dysgenesis. J Clin Invest. 119:2942–2953. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fraineau S, Monvoisin A, Clarhaut J,
Talbot J, Simonneau C, Kanthou C, Kanse SM, Philippe M and
Benzakour O: The vitamin K-dependent anticoagulant factor, protein
S, inhibits multiple VEGF-A-induced angiogenesis events in a Mer-
and SHP2-dependent manner. Blood. 120:5073–5083. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schneider C, King RM and Philipson L:
Genes specifically expressed at growth arrest of mammalian cells.
Cell. 54:787–793. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nagata K, Ohashi K, Nakano T, Arita H,
Zong C, Hanafusa H and Mizuno K: Identification of the product of
growth Arrest-specific Gene 6 as a common ligand for Axl, Sky, and
mer receptor tyrosine kinases. J Biol Chem. 271:30022–30027. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mark MR, Chen J, Hammonds RG, Sadick M and
Godowsk PJ: Characterization of Gas6, a member of the superfamily
of G domain-containing proteins, as a ligand for rse and Axl. J
Biol Chem. 271:9785–9789. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
van der Meer JH, van der Poll T and van't
Veer C: TAM receptors, Gas6, and protein S: Roles in inflammation
and hemostasis. Blood. 123:2460–2469. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zagórska A, Través PG, Lew ED, Dransfield
I and Lemke G: Diversification of TAM receptor tyrosine kinase
function. Nat Immunol. 15:920–928. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dransfield I and Farnworth S: Axl and mer
receptor tyrosine kinases: Distinct and nonoverlapping roles in
inflammation and cancer? Adv Exp Med Biol. 930:113–132. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bosurgi L, Bernink JH, Delgado Cuevas V,
Gagliani N, Joannas L, Schmid ET, Booth CJ, Ghosh S and Rothlin CV:
Paradoxical role of the proto-oncogene Axl and Mer receptor
tyrosine kinases in colon cancer. Proc Natl Acad Sci USA.
110:13091–13096. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bilimoria KY, Bentrem DJ, Ko CY, Stewart
AK, Winchester DP and Talamonti MS: National failure to operate on
early stage pancreatic cancer. Ann Surg. 246:173–180. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cronin-Fenton DP, Søndergaard F, Pedersen
LA, Fryzek JP, Cetin K, Acquavella J, Baron JA and Sørensen HT:
Hospitalisation for venous thromboembolism in cancer patients and
the general population: a population-based cohort study in Denmark,
1997–2006. Br J Cancer. 103:947–953. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Walker AJ, Card TR, West J, Crooks C and
Grainge MJ: Incidence of venous thromboembolism in patients with
cancer-a cohort study using linked United Kingdom databases. Eur J
Cancer. 49:1404–1413. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Horsted F, West J and Grainge MJ: Risk of
venous thromboembolism in patients with cancer: A systematic review
and meta-analysis. PLoS Med. 9:e10012752012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lindahl AK, Odegaard OR, Sandset PM and
Harbitz TB: Coagulation inhibition and activation in pancreatic
cancer. Changes during progress of disease. Cancer. 70:2067–2072.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gradiz R, Silva HC, Carvalho L, Botelho MF
and Mota-Pinto A: MIA PaCa-2 and PANC-1-pancreas ductal
adenocarcinoma cell lines with neuroendocrine differentiation and
somatostatin receptors. Sci Rep. 6:216482016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tan MH, Nowak NJ, Loor R, Ochi H, Sandberg
AA, Lopez C, Pickren JW, Berjian R, Douglass HO Jr and Chu TM:
Characterization of a new primary human pancreatic tumor line.
Cancer Invest. 4:15–23. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yunis AA, Arimura GK and Russin DJ: Human
pancreatic carcinoma (MIA PaCa-2) in continuous culture:
Sensitivity to asparaginase. Int J Cancer. 19:128–135. 1977.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tai MH, Olson LK, Madhukar BV, Linning KD,
Van Camp L, Tsao MS and Trosko JE: Characterization of gap
junctional intercellular communication in immortalized human
pancreatic ductal epithelial cells with stem cell characteristics.
Pancreas. 26:e18–e26. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Qian J, Niu J, Li M, Chiao PJ and Tsao MS:
In vitro modeling of human pancreatic duct epithelial cell
transformation defines gene expression changes induced by K-ras
oncogenic activation in pancreatic carcinogenesis. Cancer Res.
65:5045–5053. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rezende SM, Razzari C and Simmonds RE: In
vitro high level protein S expression after modification of protein
S cDNA. Thromb Haemost. 90:1214–1215. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Levkovitz Y and Baraban JM: A dominant
negative inhibitor of the Egr family of transcription regulatory
factors suppresses cerebellar granule cell apoptosis by blocking
c-Jun activation. J Neurosci. 21:5893–5901. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Weber K, Bartsch U, Stocking C and Fehse
B: A multicolor panel of novel lentiviral ‘gene ontology’ (LeGO)
vectors for functional gene analysis. Mol Ther. 16:698–706. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Weber K, Mock U, Petrowitz B, Bartsch U
and Fehse B: Lentiviral gene ontology (LeGO) vectors equipped with
novel drug-selectable fluorescent proteins: New building blocks for
cell marking and multi-gene analysis. Gene Ther. 17:511–520. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Feoktistova M, Geserick P and Leverkus M:
Crystal violet assay for determining viability of cultured cells.
Cold Spring Harb Protoc 2016. pdb.prot087379. 2016. View Article : Google Scholar
|
|
35
|
Niyazi M, Niyazi I and Belka C: Counting
colonies of clonogenic assays by using densitometric software.
Radiat Oncol. 2:42007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lee E, Decker AM, Cackowski FC, Kana LA,
Yumoto K, Jung Y, Wang J, Buttitta L, Morgan TM and Taichman RS:
Growth Arrest-Specific 6 (GAS6) promotes prostate cancer survival
by g1 arrest/s phase delay and inhibition of apoptosis during
chemotherapy in bone marrow. J Cell Biochem. 117:2815–2824. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gustafsson A, Bostrom AK, Ljungberg B,
Axelson H and Dahlbäck B: Gas6 and the receptor tyrosine kinase Axl
in clear cell renal cell carcinoma. PLoS One. 4:e75752009.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cummings CT, Deryckere D, Earp HS and
Graham DK: Molecular pathways: MERTK signaling in cancer. Clin
Cancer Res. 19:5275–5280. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Keating AK, Salzberg DB, Sather S, Liang
X, Nickoloff S, Anwar A, Deryckere D, Hill K, Joung D, Sawczyn KK,
et al: Lymphoblastic leukemia/lymphoma in mice overexpressing the
Mer (MerTK) receptor tyrosine kinase. Oncogene. 25:6092–6100. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sufit A, Lee-Sherick AB, DeRyckere D,
Rupji M, Dwivedi B, Varella-Garcia M, Pierce AM, Kowalski J, Wang
X, Frye SV, et al: MERTK inhibition induces polyploidy and promotes
cell death and cellular senescence in glioblastoma multiforme. PLoS
One. 11:e01651072016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu J, Frady LN, Bash RE, Cohen SM,
Schorzman AN, Su YT, Irvin DM, Zamboni WC, Wang X, Frye SV, et al:
MerTK as a therapeutic target in glioblastoma. Neuro Oncol.
20:92–102. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Waizenegger JS, Ben-Batalla I, Weinhold N,
Meissner T, Wroblewski M, Janning M, Riecken K, Binder M,
Atanackovic D, Taipaleenmaeki H, et al: Role of Growth
arrest-specific gene 6-Mer axis in multiple myeloma. Leukemia.
29:696–704. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mandalà M, Falanga A and Roila F:
Management of venous thromboembolism (VTE) in cancer patients: ESMO
Clinical Practice Guidelines. Ann Oncol. 22 (Suppl 6):vi85–vi92.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hisada Y, Geddings JE, Ay C and Mackman N:
Venous thrombosis and cancer: From mouse models to clinical trials.
J Thromb Haemost. 13:1372–1382. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sheth RA, Niekamp A, Quencer KB, Shamoun
F, Knuttinen MG, Naidu S and Oklu R: Thrombosis in cancer patients:
Etiology, incidence, and management. Cardiovasc Diagn Ther. 7
(Suppl 3):S178–S185. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Iodice S, Gandini S, Löhr M, Lowenfels AB
and Maisonneuve P: Venous thromboembolic events and organ-specific
occult cancers: A review and meta-analysis. J Thromb Haemost.
6:781–788. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sohail MA and Saif MW: Role of
anticoagulation in the management of pancreatic cancer. JOP.
10:82–87. 2009.PubMed/NCBI
|
|
48
|
Walker FJ: Regulation of activated protein
C by a new protein. A possible function for bovine protein S. J
Biol Chem. 255:5521–5524. 1980.PubMed/NCBI
|
|
49
|
Walker FJ, Chavin SI and Fay PJ:
Inactivation of factor VIII by activated protein C and protein S.
Arch Biochem Biophys. 252:322–328. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ndonwi M and Broze G Jr: Protein S
enhances the tissue factor pathway inhibitor inhibition of factor
Xa but not its inhibition of factor VIIa-tissue factor. J Thromb
Haemost. 6:1044–1046. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Chattopadhyay R, Sengupta T and Majumder
R: Inhibition of intrinsic Xase by protein S: A novel regulatory
role of protein S independent of activated protein C. Arterioscler
Thromb Vasc Biol. 32:2387–2393. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Plautz WE, Sekhar Pilli VS, Cooley BC,
Chattopadhyay R, Westmark PR, Getz T, Paul D, Bergmeier W, Sheehan
JP and Majumder R: Anticoagulant Protein S targets the factor IXa
Heparin-binding exosite to prevent thrombosis. Arterioscler Thromb
Vasc Biol. 38:816–828. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Saller F, Brisset AC, Tchaikovski SN,
Azevedo M, Chrast R, Fernández JA, Schapira M, Hackeng TM, Griffin
JH and Angelillo-Scherrer A: Generation and phenotypic analysis of
protein S-deficient mice. Blood. 114:2307–2314. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Burstyn-Cohen T, Lew ED, Traves PG,
Burrola PG, Hash JC and Lemke G: Genetic dissection of TAM
receptor-ligand interaction in retinal pigment epithelial cell
phagocytosis. Neuron. 76:1123–1132. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Nakamura YS, Hakeda Y, Takakura N, Kameda
T, Hamaguchi I, Miyamoto T, Kakudo S, Nakano T, Kumegawa M and Suda
T: Tyro 3 receptor tyrosine kinase and its ligand, gas6, stimulate
the function of osteoclasts. Stem Cells. 16:229–238. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hafizi S and Dahlbäck B: Gas6 and Protein
S. Vitamin K-dependent ligands for the Axl receptor tyrosine kinase
subfamily. FEBS J. 273:5231–5244. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Linger RM, Keating AK, Earp HS and Graham
DK: TAM receptor tyrosine kinases: Biologic functions, signaling,
and potential therapeutic targeting in human cancer. Adv Cancer
Res. 100:35–83. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Graham DK, Salzberg DB, Kurtzberg J,
Sather S, Matsushima GK, Keating AK, Liang X, Lovell MA, Williams
SA, Dawson TL, et al: Ectopic expression of the proto-oncogene mer
in pediatric t-cell acute lymphoblastic leukemia. Clin Cancer Res.
12:2662–2669. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Shankar SL, O'Guin K, Kim M, Varnum B,
Lemke G, Brosnan CF and Shafit-Zagardo B: Gas6/Axl Signaling
activates the phosphatidylinositol 3-Kinase/Akt1 survival pathway
to protect oligodendrocytes from tumor necrosis factor
alpha-induced apoptosis. J Neurosci. 26:5638–5648. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Shankar SL, O'Guin K, Cammer M, McMorris
FA, Stitt TN, Basch RS, Varnum B and Shafit-Zagardo B: The growth
arrest-specific gene product Gas6 promotes the survival of human
oligodendrocytes via a phosphatidylinositol 3-kinase-dependent
pathway. J Neurosci. 23:4208–4218. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Moody G, Belmontes B, Masterman S, Wang W,
King C, Murawsky C, Tsuruda T, Liu S, Radinsky R and Beltran PJ:
Antibody-mediated neutralization of autocrine Gas6 inhibits the
growth of pancreatic ductal adenocarcinoma tumors in vivo. Int J
Cancer. 139:1340–1349. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Song X and Wang H, Logsdon CD, Rashid A,
Fleming JB, Abbruzzese JL, Gomez HF, Evans DB and Wang H:
Overexpression of receptor tyrosine kinase Axl promotes tumor cell
invasion and survival in pancreatic ductal adenocarcinoma. Cancer.
117:734–743. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Koorstra JB, Karikari C, Feldmann G, Bisht
S, Rojas PL, Offerhaus GJ, Alvarez H and Maitra A: The Axl receptor
tyrosine kinase confers an adverse prognostic influence in
pancreatic cancer and represents a new therapeutic target. Cancer
Biol Ther. 8:618–626. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Shen Y, Chen X, He J, Liao D and Zu X: Axl
inhibitors as novel cancer therapeutic agents. Life Sci.
198:99–111. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Kim HA, Nam JY, Jung JY, Bae CB, An JM,
Jeon JY, Kim BS and Suh CH: Serum growth arrest-specific protein 6
levels are elevated in adult-onset Still's disease. Clin Rheumatol.
33:865–868. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Suh CH, Hilliard B, Li S, Merrill JT and
Cohen PL: TAM receptor ligands in lupus: Protein S but not Gas6
levels reflect disease activity in systemic lupus erythematosus.
Arthritis Res Ther. 12:R146. 2010. View
Article : Google Scholar : PubMed/NCBI
|