Introduction
Breast cancer is a disease that affects the health
of women worldwide, and is the leading cause of cancer-associated
deaths in women (1). The incidence
of breast cancer has been increasing in China in recent years. It
was estimated that there were 208,000 female patients with breast
cancer in China in 2010. The incidence rate is highest in
50-year-old women, and the number of deaths due to breast cancer
accounts for 9.6% of global deaths (2). Although surgery, radiotherapy,
chemotherapy as well as other treatments have made significant
progress, the clinical results remain frustrating. Numerous
patients with breast cancer succumb to tumor recurrence or
metastasis due to the systemic side effects of the current
treatments (3). Tumor
immunotherapies are different from traditional treatments, in
which, the antitumor immune response of patients is activated to
kill tumors. It is characterized by high specificity and minor side
effects, which can help patients establish immune memory to exert a
long-term ‘monitoring’ effect. Immunotherapy mainly utilizes the
antigenicity of tumors; thus, finding a suitable antigen as a
target is of importance to immunotherapy (4).
Cancer testis antigens (CTAs) are only expressed in
tumor cells and germ cells (testis, ovarian and placental cells),
while rarely expressed in somatic cells. This restricted expression
makes them a promising tumor antigen of immunotherapy (5). Melanoma-associated antigen (MAGE) was
identified by Professor Vander Bruggen, and belongs to the CTA
subfamily (6). Among the numerous
MAGE family members, the melanoma-associated antigen A family
(MAGE-As) has energetical tumor antigen specificity, and can be
recognized by autoimmune cells to induce specific antitumor humoral
immunity and cellular immunity (7).
The effect of antitumor immunotherapy depends on the specificity
and expression level of tumor antigens (8). Breast cancer tissue can concurrently
express multiple subtypes of the MAGE-A antigen, indicating that
the MAGE-A antigen is a suitable target for breast cancer
immunotherapy (9). However, the
expression level of different subtypes of MAGE-A antigen in breast
cancer tissues is relatively low, which renders treatment based on
tumor vaccines against specific MAGE-A antigen subtypes alone
unable to induce strong cytotoxic T-cell antitumor responses
(10). Increased expression of the
MAGE-A antigen in tumor cells and identification of common
antigenic peptides of MAGE-A antigen should be beneficial for the
recognition and killing of tumor cells by the immune system. Our
previous study revealed that demethylating drugs can increase the
expression of MAGE-A11 in a breast cancer cell line, which enhanced
the killing effects of CTLs on breast cancer (11). Therefore, the identification of
additional drugs that can increase the expression of MAGE-As is
necessary for immunotherapy against breast cancer.
At present, several studies have designed common
antigen peptides (mostly polypeptides) of different subtypes of
MAGE-A and verified their antitumor effectiveness in various types
of tumor cells (12). However, a
major disadvantage of polypeptides is their weak immunogenicity,
which induces a weak immune response to CD8+ T cells
(13). Therefore, it is necessary
to find novel approaches that can improve the immunogenicity of
antigenic peptides to compensate the limitations of peptide
immunity. The enhancement of immunogenicity of breast cancer by
inducing an increase in the effect of T cell response and promotion
of tumor suppressors is important in breast cancer immunotherapy.
Notably, immunological checkpoint molecules are currently the most
investigated, and promising treatment methods for a variety of
tumors and their therapeutic effects have been demonstrated in
animal experiments and in phase II and phase III clinical studies
(14,15). The immunological checkpoint
cytotoxic T-lymphocyte antigen 4 (CTLA-4) was found to inhibit
dendritic cell (DC) function in breast cancer cells, which in turn
rendered T cells unable to be effectively activated; thus, it
greatly reduced tumor killing, and was one of the causes of cancer
cell proliferation and metastasis (16). It has been suggested that breaking
this immunosuppressive state may be useful for restoring DC and
T-cell function (16).
To date, there are no studies using anti-CTLA-4
monoclonal antibody and MAGE-As common antigen peptide as a
combination therapy for treating breast cancer. Therefore, in the
present study, killing experiments on breast cancer were designed
consisting of a co-antigen peptide of MAGE-A antigen combined with
CTLA-4 blockade. Furthermore, the demethylating drug 5DC was used
to pretreat cancer cells, which was found to increase the
expression of MAGE-As and enhance the immunogenicity of the cells.
Hence, MAGE-As co-antigen peptide-specific CTLs in combination with
anti-CTLA-4 monoclonal antibody and 5DC have potent tumor cell
killing effects, which provides a novel theory for the development
of breast cancer therapies.
Materials and methods
Clinical samples
The breast cancer tissues and corresponding adjacent
normal tissues of 115 patients (14 aged 30–40, 27 aged 40–50, 35
aged 50–60, 28 aged 60–70, 9 aged 70–80, 2 >80 years; 112 women
and 3 men) with breast cancer who were hospitalized at the Fourth
Hospital of Hebei Medical University (Shijiazhuang, Hebei, China)
from 2012 to 2019 were collected. The patients were all onset for
the first time, and no radiotherapy, chemotherapy or endocrine
therapy were performed on the patients before this study. All
patients were diagnosed with breast cancer by pathology.
Pathological diagnosis and tumor staging criteria were performed in
accordance with the standards of the World Health Organization and
the Union for International Cancer Control. The present study was
approved by the Ethics Committee of the Affiliated Hospital of
Hebei Medical University. All patients involved in this study were
informed of the experimental content, purpose and significance of
the study, and signed the informed consent form.
Cell culture and treatment
MCF-7, MDA-MB-453, MDA-MB-231 and BT549 cells were
provided by the Research Center of the Fourth Hospital of Hebei
Medical University. Cells were cultured in RPMI-1640 medium (cat.
no. A33823) containing 10% fetal bovine serum (FBS; cat. no.
16140071) and penicillin and streptomycin [P/S; penicillin,
5×105 U/l; streptomycin 100 mg/l; (cat. no. 15070063;
all from Thermo Fisher Scientific, Inc.] at 37°C in a 5%
CO2 incubator.
For the culture of primary breast cancer cells, the
adipose tissue and connective tissue around the breast cancer
tissues were removed, and the breast cancer tissues were washed
with PBS. The tissue was placed in P/S solution (penicillin,
5×105 U/l; streptomycin 100 mg/l) for 20 min. Then, the
tissues were cut into pieces (~1 mm3) and placed into
collagenase I solution (2×105 U/l) at 37°C and 100 × g
for 30 min. The dissociation solution was filtered through a
100-mesh cell sieve and centrifuged at 200 × g for 10 min, and then
the supernatant was discarded. The precipitate was resuspended in
RPMI-1640 medium supplemented with 5% FBS. Repeated differential
adherence method was used to remove fibroblasts as thoroughly as
possible and fibroblasts were collected as control cells according
to a previous study (10).
For 5DC treatment, 2.5, 5 or 10 µM 5DC were added to
the cell culture medium, and the cells were cultured at 37°C for 72
h.
Immunohistochemical (IHC)
staining
The tissue was embedded in paraffin and cut into
3-µm sections. The prepared tissue sections were placed in a 67°C
oven for 10 min. The tissue sections were immersed in xylene I and
II for 15 min, and then sequentially placed in absolute ethanol I
and absolute ethanol II for 20 min, and in 95 and 80% ethanol for
10 min. The sections were treated with high-pressure heat repair
for 4 min, and then 6% methanol hydrogen peroxide solution was
added dropwise on the sections, which were subsequently incubated
at room temperature for 20 min in the dark. Next, 10% goat serum
was added dropwise, and the sections were then incubated at 37°C
for 45 min. Incubation then took place with the primary antibody
including anti-MAGE-A10 (1:500 dilution; product no. 81740) and
anti-CTLA-4 (1:500 dilution; product no. 96399; both from Cell
Signaling Technology, Inc.) overnight at 4°C followed by incubation
with the secondary antibody conjugated to HRP for DAB staining for
1 h at 37°C. The nuclei were stained with hematoxylin and eosin
(H&E) staining for 1 min. The slides were observed under an
Olympus IX53 light microscope (magnification, ×400; Olympus
Corporation).
Ten fields were randomly selected from each section,
and the percentage of positive cells and staining intensity were
evaluated, and the mean values were calculated. Positive cells were
judged by cytoplasm or nucleus containing brownish yellow
particles, and staining intensity scores were performed on positive
cells. A score of 0 indicated no positive staining (similar to the
background color); a score of 1 represented mild positive staining
(light yellow); a score of 2 represented moderate positive staining
(brownish yellow); and a score of 3 represented severe positive
staining (tan). The positive cell percentage score was as follows:
0% corresponded to 0; 1–10% corresponded to 1; 11–50% corresponded
to 2; and >51% corresponded to 3. The sum of the two scores was
considered as the total score of the patient, and a total score
<4 was defined as negative, while ≥4 was defined as
positive.
Western blot assay
Total protein of breast cancer cells was extracted
using RIPA lysis and extraction buffer (Thermo Fischer Scientific,
Inc.). Quantification of proteins was performed by the Bradford
protein concentration quantification method (Beyotime Institute of
Biotechnology). In total, 50 µg proteins were electrophoresed on
10% SDS-PAGE, transferred to a PVDF membrane and blocked in 5%
non-fat milk for 1 h. The membranes were then incubated with
primary antibodies, anti-MAGE-A10 (1:1,000 dilution, product no.
81740) and anti-β-actin (1:5,000 dilution; product no. 4970; both
from Cell Signaling Technology, Inc.) at 4°C overnight. Then, the
membranes were washed with PBS 3 times and incubated with secondary
antibodies anti-rabbit IgG light chain (HRP) (1:10,000 dilution;
product code ab99697; Abcam). The protein expression levels were
measured with an enhanced chemiluminescence detection kit (Thermo
Fisher Scientific, Inc.).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA of breast cancer cells was extracted using
TRIzol (Thermo Fisher Scientific, Inc.). PrimeScript RT Reagent Kit
with gDNA Eraser (Takara Biotechnology Co., Ltd.) was used for RT.
RT-qPCR was conducted using SYBR Green qPCR Master Mix Kit (Takara
Biotechnology Co., Ltd.) according to the manufacturer's
instructions. GADPH was used as an internal standard. The RT-qPCR
conditions for MAGE-As were as follows: 95°C for 5 min, followed by
35 cycles at 95°C for 30 sec, 65°C for 30 sec and 72°C for 30 sec,
and 72°C for 5 min. The RT-qPCR conditions for GAPDH were as
follows: 95°C for 5 min, followed by 22 cycles of 95°C for 20 sec,
58°C for 20 sec and 72°C for 20 sec, and 72°C for 5 min. The
2−∆∆Cq method was used to calculate the relative
expression level according to a previous study (17). The primers were as follows: MAGE-As
forward, 5′-GTGGTCCTAAGATCTACCAAGCA-3′; MAGE-As reverse,
5′-AGGGCAGCAGGTAGGAGTG-3′; GAPDH forward, 5′-AGCCACATCGCTCAGACAC-3′
and GAPDH reverse, 5′-GCCCAATACGACCAAATCC-3′.
MAGE-As-specific induction of
cytotoxic T cells (CTLs)
Peripheral blood mononuclear cells (PBMCs) were
separated by density gradient centrifugation at 400 × g for 30 min,
and the middle white cell layer was aspirated after centrifugation.
The cell concentration was adjusted to 1×106 cells/ml
with DC culture medium (RPMI-1640 medium containing 10% FBS,
5×105 U/l penicillin and 100 mg/l streptomycin), and the
cells were stimulated with IL-2 (50 ng/ml) and IL-17 (50 ng/ml) to
become T cells. For DC induction, PBMCs were incubated with GM-CSF
(100 ng/ml) and IL-4 (50 ng/ml), and their medium was changed every
12 h, followed by the addition of GM-CSF and IL-4. At 5 days
post-culture, LPS (10 ng/ml) and TNF-α (20 ng/ml) were added, and
the cells were cultured for another 2 days to stimulate DC
maturation. MAGE-As polypeptide (10 µM) was added to the mature
DCs, and antigen-loaded DCs were obtained by incubation at 37°C for
24 h. Mature DCs and T cells were mixed at a ratio of 1:10. After
adding IL-2 (1,000 U/ml), the cells were cultured for 2 days; then,
half the volume was changed were every other day and supplemented
with IL-2 (1,000 U/ml). This process was repeated 7 times to
collect CTLs.
Flow cytometric assay
Mature DCs and CTLA-4 expression were detected by
flow cytometry. For detection of mature DCs, PE-labeled anti-CD86
(1:1,000 dilution; product no. 60712; Cell Signaling Technology,
Inc) + anti-CD80 (1:1,000 dilution; cat. no. 15416; Cell Signaling
Technology, Inc.) and PE-labeled anti-CD1a (1:1,000 dilution; cat.
no. NBP2-34731PE; Novus Biologicals) + anti-CD83 (1:1,000 dilution;
product no. 99075S; Cell Signaling Technology, Inc.) were added to
100 µl cell suspension. Then, the mixture was incubated at 4°C for
30 min in the dark and subjected to flow cytometry (BD
Biosciences). For detection of CTLA-4 expression on the breast
cancer cell surface, 5 µl PE-labeled anti-CTLA-4 monoclonal
antibody (1:1,000 dilution; cat. no. 15132; Cell Signaling
Technology, Inc.) was mixed with 100 µl cell suspension, and 5 µl
PE-labeled IgG was added to the control group, followed by
incubation at room temperature for 15 min and then assessment on a
FACS machine (BD FACSCalibur).
CCK-8 assay
CCK-8 assay has been used for evaluating T cell
lysis ability (17). The target
cells included MCF-7 (ER+, HLA-A*0201), MDA-MB-453
(HER-2 overexpression, HLA-A*0201), MDA-MB-231 and BT549 (triple
negative, HLA-A*0201), while the effector cells included
MAGE-As-specific CTLs. Four groups were established as follows: i)
CTL group; ii) CTL + anti-CTLA-4 group; iii) CTL + 5DC group; and
iv) CTL + anti-CTLA-4 + 5DC group. The target cells were seeded in
96-well plates at a density of 1×103 cells/well and
cultured overnight. Various concentrations of effector cells were
added to the experimental wells to achieve different
effector:target ratios (5:1, 10:1 and 20:1). After adding 10 µl
CCK-8 solution per well and incubating at 37°C for 3 h, the
absorbance at 450 nm was detected using Multiskan FC (Thermo Fisher
Scientific, Inc.). The specific lysis rate was calculated according
to the following formula: Specific lysis rate (%)=optical density
[(OD)test group-ODblank
group)]/(ODcontrol group-ODblank group)
×100.
ELISA
The supernatants of the co-cultured cells of each
group were collected, and the IFN-γ level in the supernatant was
analyzed by ELISA. The human IFN-γ ELISA kit was purchased from
DAKEWE Biotech Co., Ltd. ELISA plates were coated with monoclonal
antibodies (cat no. EA-0507; DAKEWE Biotech Co., Ltd.) and
incubated overnight at 4°C. After discarding the coating solution,
the plates were washed three times with PBS-Tween-20 (0.05%). The
samples were diluted (1:5 dilution) and incubated in the ELISA
plates at 37°C for 30 min. Next, biotinylated antibody (1:80
dilution; cat. no. EA-0507; DAKEWE Biotech Co., Ltd.) and
avidin-labeled horseradish peroxidase (DAKEWE Biotech Co., Ltd.)
were added to the ELISA plate, followed by incubation at 37°C for
30 min. The chromogenic reagent (100 µl per well; DAKEWE Biotech
Co., Ltd.) was added to the ELISA plate and incubated 37°C for 15
min in the dark. Then, 50 µl termination solution (100 µl per well;
DAKEWE Biotech Co., Ltd.) was added per well to stop the reaction.
Multiskan FC (Thermo Fisher Scientific, Inc.) was used to measure
the absorbance at 450 nm.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism (version 5; GraphPad Software, Inc.). The association between
the expression of MAGE-As and CTLA-4 and clinicopathological
parameters was analyzed by χ2 test. Survival analysis
was performed using the Kaplan-Meier method with the log-rank test.
Data are expressed as the mean ± standard deviation. Statistical
analysis regarding comparison of different groups was performed by
one-way analysis of variance (ANOVA) test followed by Tukey's
multiple post hoc test. Student's t-tests were performed for
comparison between two groups. A P-value <0.05 was considered to
indicate a statistically significant difference.
Results
Expression of MAGE-As in breast cancer
tissues and cells, and its influence on patient prognosis
To explore the role of MAGE-As in the progression of
breast cancer, the expression of MAGE-As in breast cancer tissues
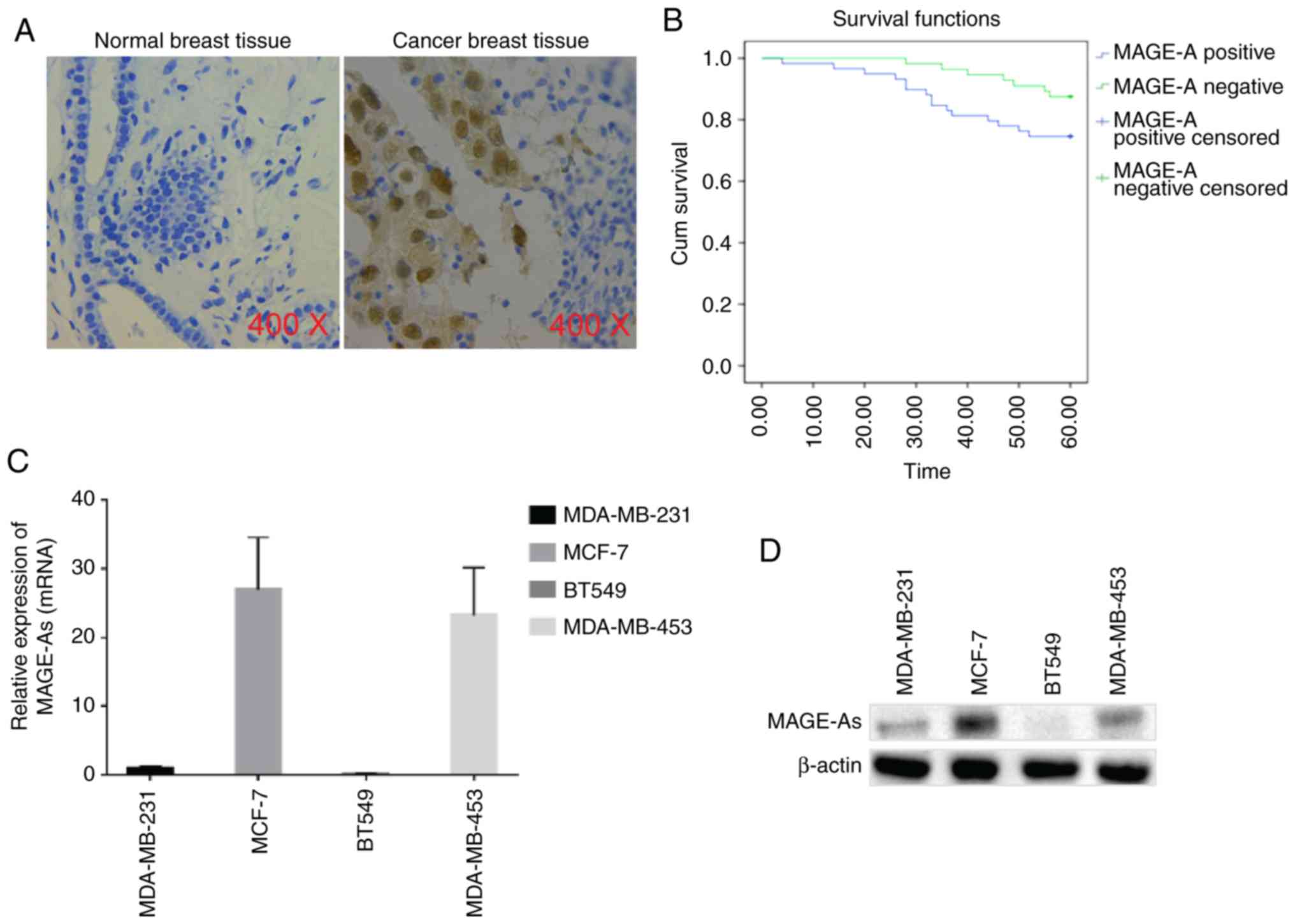
and cells was evaluated in the present study. As assessed by
immunohistochemical (IHC) staining, MAGE-As expression could not be
detected in normal breast tissue, while it was positive in breast
cancer tissues (both in the cytoplasm and nucleus), and the overall
positive expression rate was 51.3% (Fig. 1A). The association between MAGE-As
positive expression and the clinicopathological parameters of
patients with breast cancer is presented in Table I. The results revealed that
lymphatic metastasis as well as recurrence and metastasis were both
associated with MAGE-As expression. The results of Kaplan-Meier
analysis revealed that there was no significant difference in
overall survival between patients with breast cancer who exhibited
positive MAGE-As expression and those with negative MAGE-As
expression (Fig. 1B). Furthermore,
the expression levels of MAGE-As in breast cancer cell lines,
including MDA-MB-231, MCF-7, BT549 and MDA-MB-453, was detected by
RT-qPCR and western blotting. As revealed in Fig. 1C and D, the highest expression of
MAGE-As was observed in MCF-7 cells, followed by MDA-MB-453 and
MDA-MB-231 cells, while the lowest expression of MAGE-As was
detected in BT549 cells (n=6). Collectively, these data indicated
that MAGE-As was highly expressed in breast cancer tissues and had
no significant effect on the prognosis of patients with breast
cancer. The expression levels of MAGE-As varied across different
breast cancer cell lines.
 | Table I.The associations between MAGE-As
expression and clinicopathological parameters in breast cancer
patients. |
Table I.
The associations between MAGE-As
expression and clinicopathological parameters in breast cancer
patients.
| Parameters | n | MAGE-A
positive | MAGE-A
negative | χ2 | P-value |
|---|
| Age/years |
|
|
| 0.257 | 0.621 |
|
>60 | 50 | 27 | 23 |
|
|
|
≤60 | 65 | 32 | 33 |
|
|
| Sex |
|
|
| 0.000 | 0.059 |
|
Female | 112 | 57 | 55 |
|
|
|
Male | 3 | 2 | 1 |
|
|
| Tumor size
(cm) |
|
|
| 0.432 | 0.511 |
| ≥3
cm | 45 | 25 | 20 |
|
|
| <3
cm | 70 | 34 | 36 |
|
|
| Lymphatic
metastasis |
|
|
| 16.818 | <0.001 |
|
Yes | 85 | 52 | 33 |
|
|
| No | 30 | 7 | 23 |
|
|
| Recurrence and
metastasis |
|
|
| 6.369 | 0.012 |
|
Yes | 21 | 16 | 5 |
|
|
| No | 94 | 43 | 51 |
|
|
| Vascular
invasion |
|
|
| 0.355 | 0.551 |
|
Yes | 40 | 19 | 21 |
|
|
| No | 75 | 40 | 35 |
|
|
| Histological
grade |
|
|
| 0.000 | 0.063 |
| I | 19 | 6 | 13 |
|
|
| II | 69 | 35 | 34 |
|
|
|
III | 27 | 18 | 9 |
|
|
| TNM stage |
|
|
| 0.000 | 0.364 |
| I | 45 | 22 | 23 |
|
|
| II | 52 | 25 | 27 |
|
|
|
III | 18 | 12 | 6 |
|
|
| Tumor pathological
type |
|
|
| 1.932 | 0.165 |
|
Invasive ductal carcinoma | 113 | 57 | 56 |
|
|
|
Other | 2 | 2 | 0 |
|
|
| Nipple invaded |
|
|
| 0.149 | 0.700 |
|
Yes | 15 | 7 | 8 |
|
|
| No | 100 | 52 | 48 |
|
|
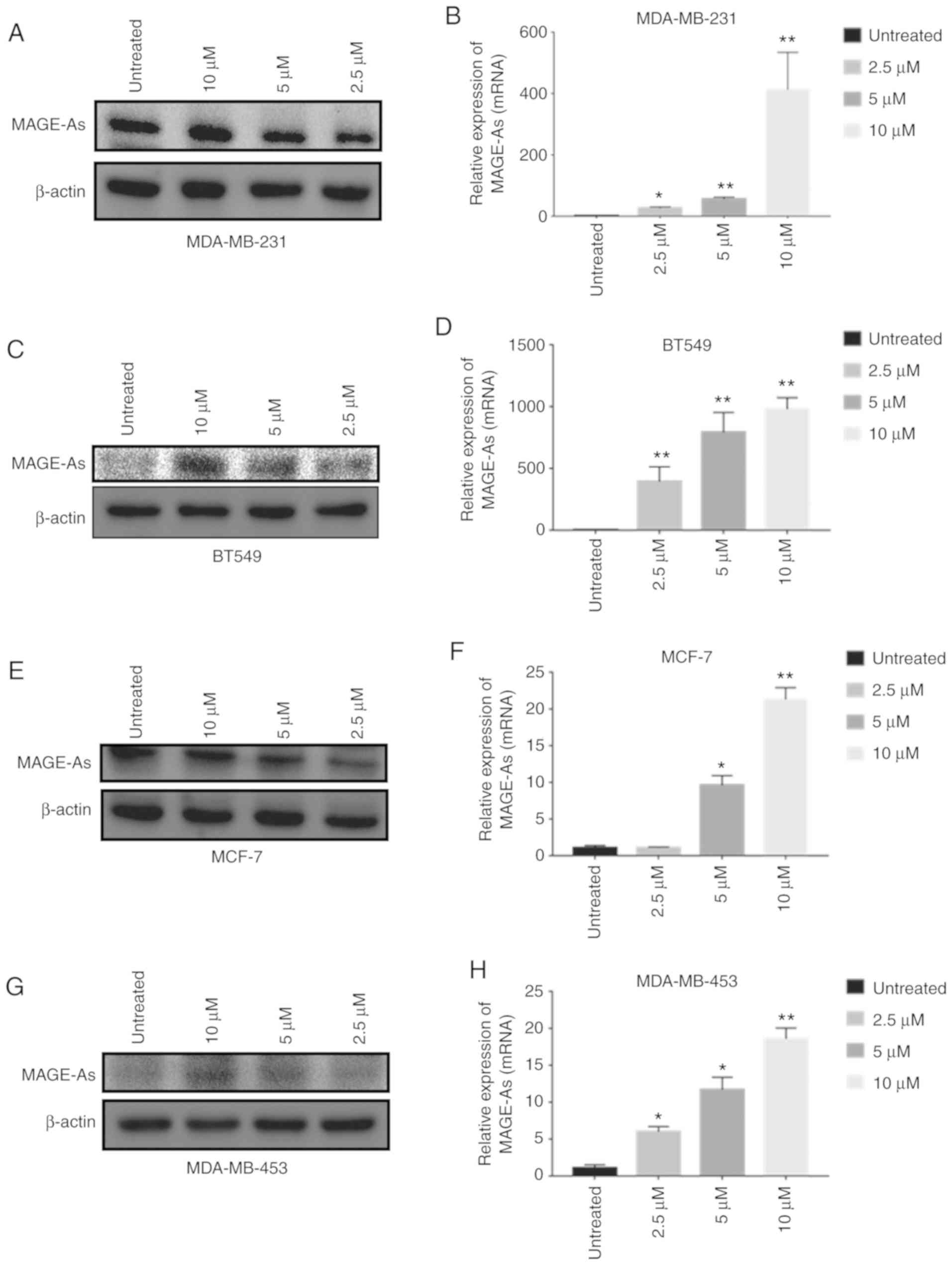
5DC treatment promotes MAGE-As
expression in breast cancer cells
An increase in the expression of MAGE-As proteins in
breast cancer cells can enhance antigen-specific T cell killing
(18); thus, the identification of
drugs that can increase the expression of MAGE-As is important. For
that aim, in the present study, breast cancer cells were treated
with 2.5, 5 and 10 µM 5DC for 72 h. In the four breast cancer cell
lines evaluated (MDA-MB-231, MDA-MB-453, BT549 and MCF-7 cell
lines), the expression level of MAGE-As was significantly
up-regulated in a dose-dependent manner (n=6; P<0.05 and
P<0.01; Fig. 2A-H). The
aforementioned data indicated that 5DC treatment upregulated
MAGE-As expression in breast cancer cells.
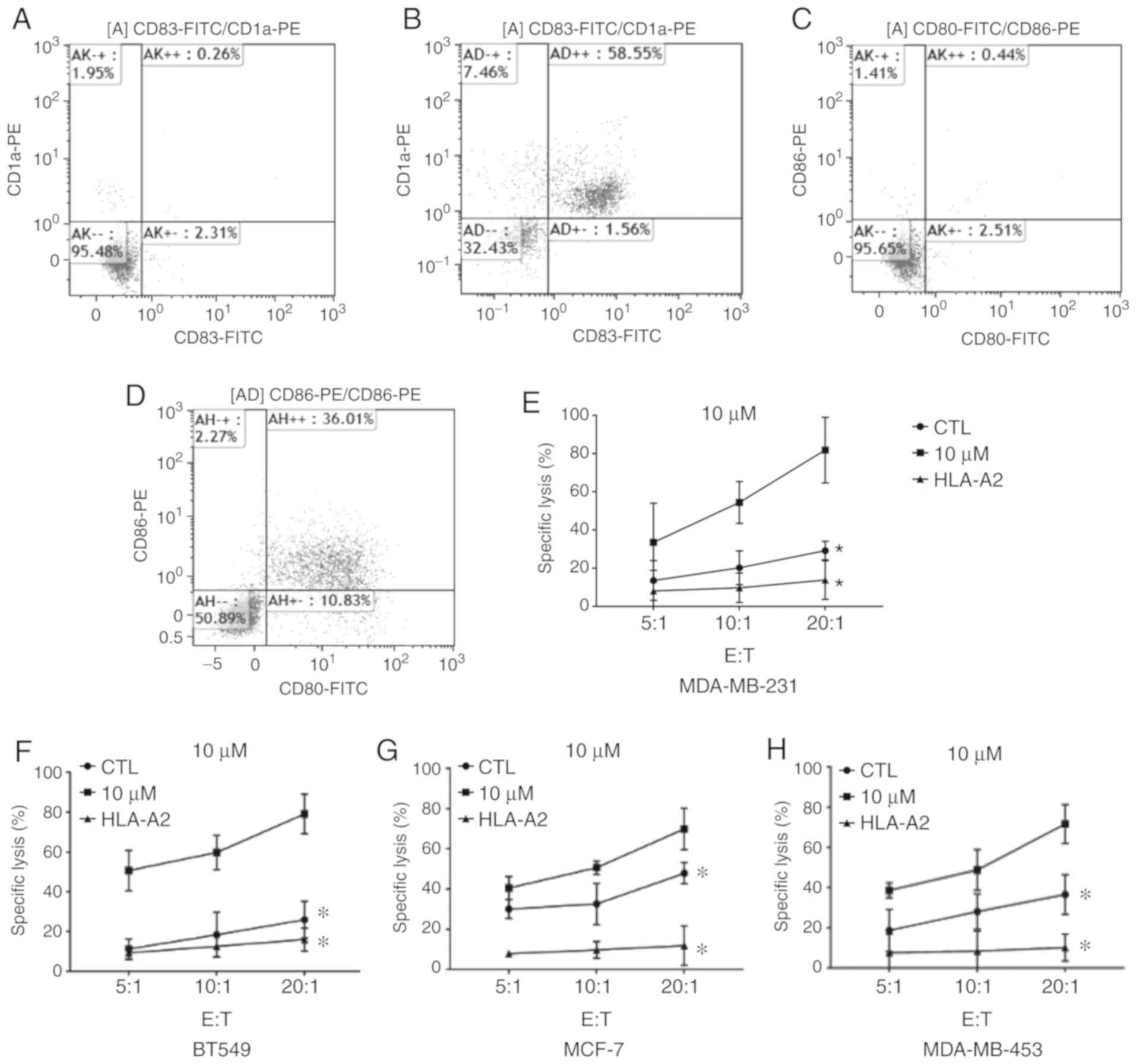
Effect of MAGE-As on the immune
response against tumors
To further explore the role of MAGE-As on immune
responses involved in killing tumors, mature DCs were successfully
obtained and verified by flow cytometry. As revealed in Fig. 3A-D,
CD80+/CD86+ cells and
CD83+/CD1a+ cells accounted for ~36 and 59%
of mature DCs, respectively (n=6). The mature DCs were loaded with
MAGE-As antigen peptide, and further co-incubated with T cells to
induce CTLs. Subsequently, the effect of MAGE-As on the lysis
efficiency of breast cancer cells was examined by CCK-8 assay. The
lysis efficiency of CTLs on breast cancer cells increased with the
increase in CTL concentration (n=6; P<0.05; Fig. 3E-H). The lysis efficiency of the CTL
+ 10 µM 5DC group was higher than that of the CTL group under the
same effector:target ratio (n=6; P<0.05; Fig. 3E-H). In addition, the lysis rate of
CTLs on breast cancer cells in the HLA-A2 blockade group was
significantly lower than that in the CTL and CTL + 10 µM 5DC
groups, indicating that CTLs had HLA restriction on the lysis of
breast cancer cells (n=6; P<0.05; Fig. 3E-H). Collectively, these data
demonstrated that upregulation of MAGE-As expression specifically
enhanced the ability of CTLs to kill breast cancer cells, and that
MAGE-As-specific CTLs had HLA restriction on the lysis of breast
cancer cells.
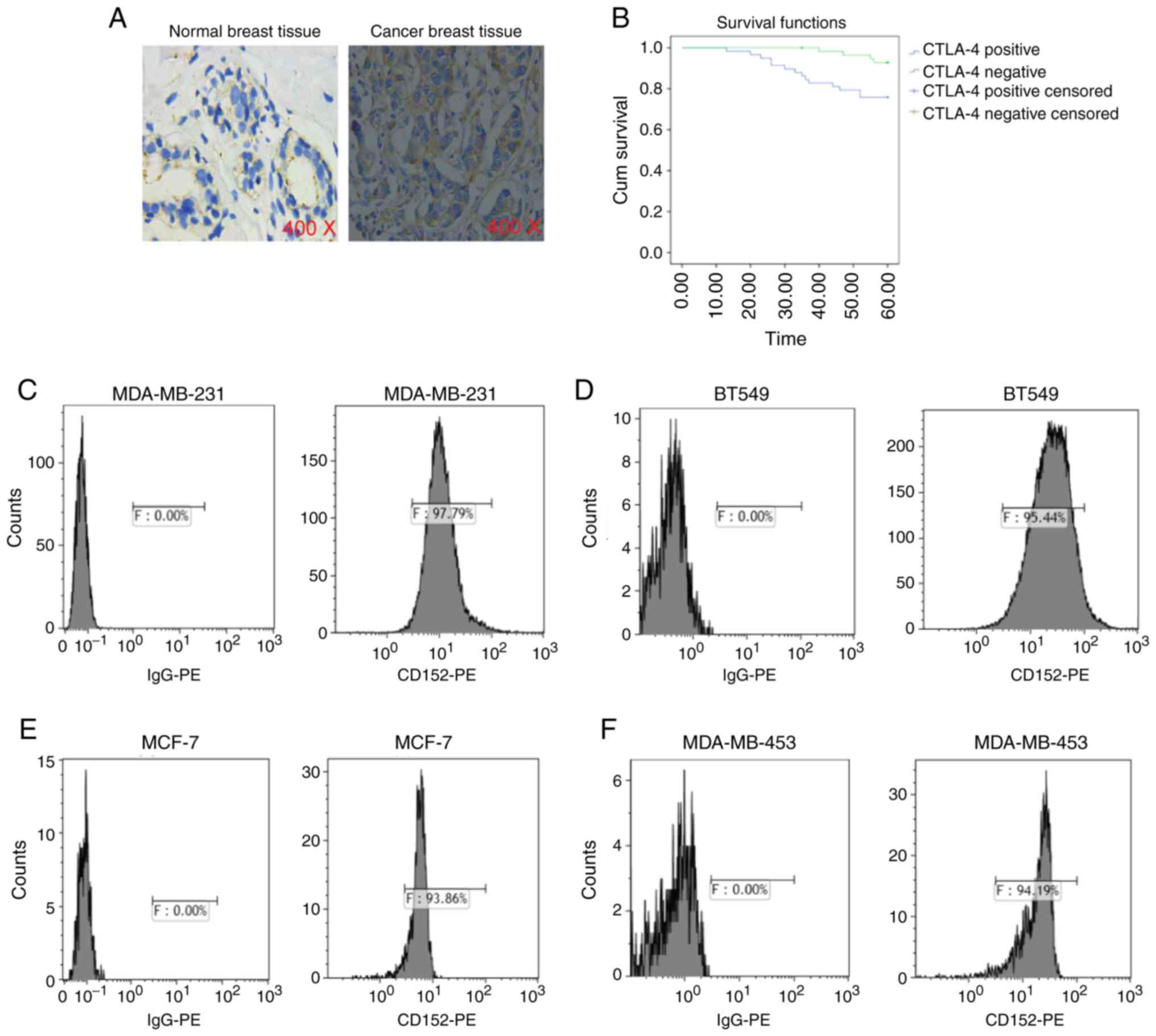
Expression of CTLA-4 in breast cancer
tissues and cells, and its influence on patient prognosis
IHC staining revealed that the expression level of
CTLA-4 in breast cancer tissues was higher than that in normal
breast tissues, and it was mainly expressed in the cytoplasm and
membrane of breast cancer cells (Fig.
4A). The association between CTLA-4 positive expression and
clinicopathological parameters in patients with breast cancer is
presented in Table II. The results
revealed that tumor size, lymphatic metastasis, recurrence and
metastasis as well as histological grade were associated with CTL-4
expression. Kaplan-Meier analysis indicated that the overall
survival of patients with breast cancer who exhibited positive
CTLA-4 expression was notably lower than that of patients with
CTLA-4 negative expression (Fig.
4B). Flow cytometry was used to detect the expression of CTLA-4
in MDA-MB-231, BT549, MCF-7 and MDA-MB-453 cells. The positive
expression rate of CTLA-4 in MDA-MB-231, BT549, MCF-7 and
MDA-MB-453 cells was 97.79, 95.44, 93.86 and 94.19%, respectively
(n=6; Fig. 4C-F). Overall, these
results demonstrated that CTLA-4 was highly expressed in breast
cancer tissues and cells, and that positive expression of CTLA-4
decreased the overall survival of patients with breast cancer.
 | Table II.The associations between CTLA-4
expression and clinicopathological parameters in breast cancer
patients. |
Table II.
The associations between CTLA-4
expression and clinicopathological parameters in breast cancer
patients.
| Parameters | n | CTLA-4
positive | CTLA-4
negative | χ2 | P-value |
|---|
| Age/years |
|
|
| 1.096 | 0.295 |
|
>60 | 50 | 27 | 23 |
|
|
|
≤60 | 65 | 41 | 24 |
|
|
| Sex |
|
|
| 0.747 | 0.388 |
|
Female | 112 | 65 | 47 |
|
|
|
Male | 3 | 3 | 0 |
|
|
| Tumor size
(cm) |
|
|
| 4.391 | 0.036 |
| ≥3
cm | 45 | 32 | 13 |
|
|
| <3
cm | 70 | 36 | 34 |
|
|
| Lymphatic
metastasis |
|
|
| 9.606 | 0.002 |
|
Yes | 85 | 56 | 29 |
|
|
| No | 30 | 12 | 18 |
|
|
| Recurrence and
metastasis |
|
|
| 10.446 | 0.001 |
|
Yes | 21 | 19 | 2 |
|
|
| No | 94 | 49 | 45 |
|
|
| Vascular
invasion |
|
|
| 0.874 | 0.350 |
|
Yes | 40 | 26 | 14 |
|
|
| No | 75 | 42 | 33 |
|
|
| Histological
grade |
|
|
| 0.000 | 0.022 |
| I | 19 | 6 | 13 |
|
|
| II | 69 | 43 | 26 |
|
|
|
III | 27 | 19 | 8 |
|
|
| TNM stage |
|
|
| 1.398 | 0.497 |
| I | 45 | 23 | 22 |
|
|
| II | 52 | 34 | 18 |
|
|
|
III | 18 | 11 | 7 |
|
|
| Tumor pathological
type |
|
|
| 0.212 | 0.645 |
|
Invasive ductal carcinoma | 113 | 66 | 47 |
|
|
|
Other | 2 | 2 | 0 |
|
|
| Nipple invaded |
|
|
| 2.463 | 0.117 |
|
Yes | 15 | 11 | 4 |
|
|
| No | 100 | 57 | 43 |
|
|
Effects of anti-CTLA-4 monoclonal
antibody alone and anti-CTLA-4 monoclonal antibody combined with
5DC on the cytotoxic function of MAGE-As-specific CTLs
To demonstrate the effects of anti-CTLA-4 monoclonal
antibody alone and in combination with 5DC on the cytotoxic
function of CTLs, the specific lysis rate of MDA-MB-231,
MDA-MB-453, BT549 and MCF-7 cells that underwent different
treatments (including control, 5DC alone treatment, anti-CTLA
antibody alone treatment, and combined treatment of 5DC and
anti-CTLA antibody) was determined by CCK-8 assay. The specific
lysis rate of the CTL + anti-CTLA-4 group was significantly higher
than that of the CTL group (n=6; P<0.05; Fig. 5A-D). By comparing the specific lysis
rates of different treatments at the same effector:target ratio, it
was revealed that the CTL + anti-CTLA-4 + 10 µM 5DC group had the
highest cell lysis rate (n=6; P<0.05; Fig. 5A-D). However, at an effector:target
ratio of 20:1, CTLs exhibited no significant difference in the
lysis rate of MDA-MB-231, MDA-MB-453 and MCF-7 cells compared with
that of the CTL + anti-CTLA-4 + 10 µM 5DC and CTL + 10 µM 5DC
groups, while no significant difference was found in the lysis rate
of BT549 cells between the CTL + anti-CTLA-4 + 10 µM 5DC, CTL +
anti-CTLA-4 and CTL + 10 µM 5DC groups (n=6; Fig. 5A-D). Subsequently, the level of
IFN-γ in the supernatant of each group was assessed by ELISA. As
revealed in Fig. 5E-H (n=6;
P<0.05, P<0.01 and P<0.001), it was revealed that the
level of IFN-γ in the cell culture supernatant was, from high to
low, as follows: CTL + anti-CTLA-4 + 10 µM 5DC group, CTL + 10 µM
5DC group, CTL + anti-CTLA-4 group and CTL group. Collectively,
CTLA-4 promoted the lysis efficiency of CTLs on breast cancer
cells, and the combination of anti-CTLA-4 and 10 µM 5DC enhanced
the lytic ability of CTLs.
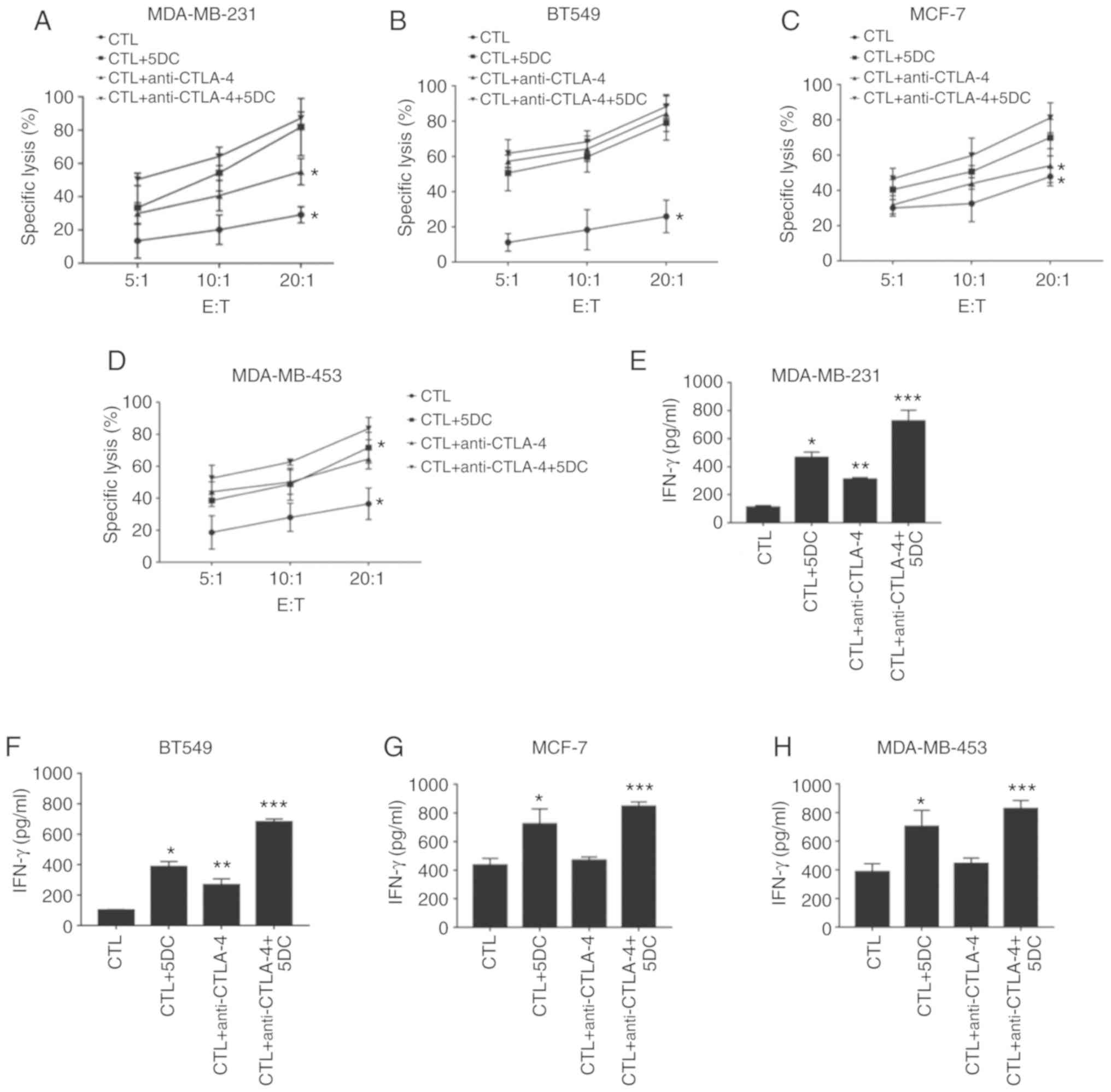 | Figure 5.Effects of anti-CTLA-4 monoclonal
antibody and anti-CTLA-4 monoclonal antibody combined with 5DC on
the cytotoxic function of MAGE-As-specific CTLs. MDA-MB-231, BT549,
MCF-7 and MDA-MB-453 cells were divided into 5 groups: i) the CTL
group; ii) the CTL + 10 µM 5DC group; iii) the CTL + anti-CTLA-4
group; and iv) the CTL + anti-CTLA-4 + 10 µM 5DC group. CCK-8
assays were used to detect the specific lytic efficiency of CTLs in
various cells including (A) MDA-MB-231, (B) BT549, (C) MCF-7, and
(D) MDA-MB-451 when the effector:target ratio was 5:1, 10:1 and
20:1, respectively. ELISA was performed to assess the level of
IFN-γ in the supernatant of (E) MDA-MB-231, (F) BT549, (G) MCF-7
and (H) MDA-MB-453 cells (n=6). Data are presented as the mean ±
standard deviation. *P<0.05, **P<0.01 and ***P<0.001 vs.
the CTL group. MAGE-As, melanoma-associated antigen A family; CTL,
cytotoxic T cell; 5DC, 5-aza-2′-deoxycytidine. |
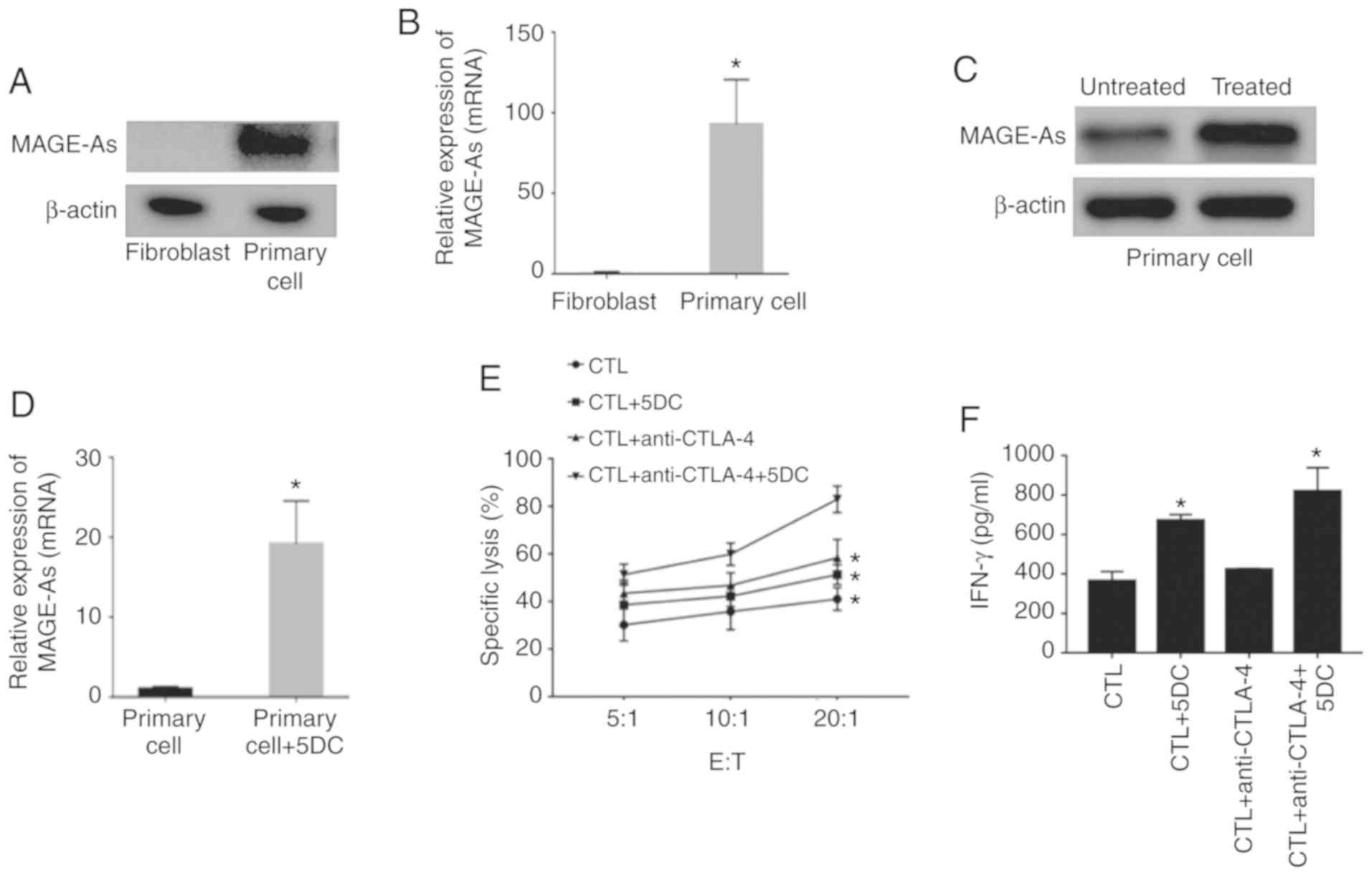
Combination of anti-CTLA-4 antibody
and 5DC enhances the killing effect of CTLs on primary breast
cancer cells
To further verify the effects of the combination of
anti-CTLA-4 antibody and 5DC on the killing effect of CTLs in
primary breast cancer cells, fibroblasts and primary breast cancer
cells were cultured, and the expression levels of MAGE-As in
fibroblasts and primary cells were assessed by western blotting and
RT-qPCR. As revealed in Fig. 6A and
B (n=6; P<0.05), MAGE-As levels in primary cells were
notably higher than those in fibroblasts. In addition, 10 µM 5DC
treatment successfully induced the expression of MAGE-As in primary
breast cancer cells (n=6; P<0.05; Fig. 6C and D). A CCK-8 assay revealed that
the specific lysis efficiency of CTLs in the CTL + 10 µM 5DC and
CTL + anti-CTLA-4 groups was higher than that in the CTL group, and
the lysis rate was highest in the CTL + anti-CTLA-4 + 10 µM 5DC
group (n=6; P<0.05; Fig. 6E).
Furthermore, the level of IFN-γ in the culture medium of the CTL +
anti-CTLA-4 + 5DC group was the highest, followed by the CTL + 10
µM 5DC, CTL + anti-CTLA-4, and CTL groups, respectively (n=6;
P<0.05; Fig. 6F). These results
were consistent with the trend observed in MDA-MB-231, MDA-MB-453,
BT549 and MCF-7 cells. In summary, the combination of anti-CTLA-4
antibody and 5DC enhanced the killing effect of CTLs on primary
breast cancer cells.
Discussion
In recent years, although various treatments against
breast cancer have progressed, the prognosis is still not
optimistic, and a considerable proportion of patients experience
recurrence and even death (2).
Unlike traditional chemoradiotherapy, tumor immunotherapy is aimed
to stimulate the immune system in the body, and has less systemic
side effects than chemoradiotherapy (18). The most critical point of
immunotherapy is finding the right target antigen. MAGE-A is the
first human tumor-associated antigen found at the molecular level,
and belongs to the CTA antigen family (10). MAGE-A was considered an ideal target
antigen in immunotherapy due to its characteristics of high
expression in tumor tissues, no expression in normal somatic cells
and restricted expression in germ cells (6). In the present study, it was revealed
that MAGE-As was highly expressed in breast cancer tissues, and
that 5DC treatment promoted MAGE-As expression in breast cancer
cells. There was no significant difference in overall survival
between patients with breast cancer who exhibited positive MAGE-As
expression and those with negative MAGE-As expression, which may be
due to the limited number of patients in our study. Notably, CTLA-4
was highly expressed in breast cancer tissues and cells. In
addition, the present study revealed that the combination of
anti-CTLA-4 antibody and 5DC enhanced the killing effect of CTLs on
primary breast cancer cells.
The MAGE-As antigen belongs to the MAGE family, and
includes -A1, -A2, -A3, -A4, -A6, -A10 and -A12. The MAGE-A gene
consists of 12 highly homologous genes located on the Xq28
chromosome (19,20). MAGE-As is widely expressed in a
range of cancer types of different tissue origin, including breast,
ovarian, bladder, lung, prostate and thyroid cancer, and is
associated with poor prognosis of patients (21–23).
Aberrant high expression of MAGE-A3 and -A6 in breast cancer was
revealed to be associated with estrogen receptor (ER), progesterone
receptor (PR), tumor size and adverse outcome (24,25).
Consistently, the results of the present study revealed that
MAGE-As were not expressed in normal breast tissue, but were
abnormally highly expressed in breast cancer tissues, and were
associated with lymph node metastasis, recurrence and metastasis.
Abd-Elsalam and Ismaeli revealed that the positive expression rate
of MAGE-A1-A6 and MAGE-A12 mRNA in venous blood of patients with
breast cancer was associated with the TNM stage of tumors (26). Although it appears that the present
study has a certain discrepancy with the aforementioned studies, it
is certain that the expression of MAGE-As detected by the two
experimental methods was closely associated with the clinical
parameters of the patients.
The expression of the MAGE-A family genes is
regulated by promoter methylation. Demethylating agents can
increase the expression of MAGE-A antigen, thereby enhancing the
killing function of tumor-specific T cells (26). Two currently used demethylating
drugs, decitabine and 5-azacytidine, have been demonstrated to
promote the expression of MAGE-A antigen (27,28).
The positive expression of antigen on the surface of cancer cells
is a prerequisite for peptide-based immunotherapy. Our previous
study found that the novel methyl inhibitor 5DC can induce the
expression of MAGE-A11 in breast cancer cell lines (11). Consistently, in the present study,
it was revealed that treatment of breast cancer cell lines with the
demethylating drug 5DC could induce the expression of MAGE-As
antigen in breast cancer cells. Theoretically, the combination of
other methods could improve the immunogenicity of antigenic
peptides, thereby overcoming some limitations of peptide immunity.
The present results indicated that the increased expression level
of MAGE-As by 5DC treatment could improve the immunotherapeutic
effect of the MAGE-As common antigen peptide in breast cancer
cells.
CTLA-4 is an immunological checkpoint molecule and
is an important T lymphocyte surface molecule. It is mainly
expressed on the surface of activated immune effector T cells and
regulatory T cells (28). Previous
studies have found that CTLA-4 is associated with immune disorders
in patients with breast cancer, and have reported that breast tumor
cells highly express CTLA-4, and that the expression of CTLA-4 in
the peripheral blood mononuclear cells (PBMCs) of patients with
breast cancer is higher than that that of the normal control group
(29,30). In vitro experiments revealed
that CTLA-4 can inhibit the response of T cells to antigen, and
blocking soluble CTLA-4 can significantly enhance the response of
PBMCs to antigen (31). The present
study revealed that CTLA-4 exhibited low expression in normal
breast tissue, but was abnormally highly expressed in breast cancer
tissues, and that CTLA-4 positive expression was associated with
tumor size, lymph node metastasis, recurrence and metastasis.
Patients with breast cancer exhibiting positive CTLA-4 expression
had a lower overall survival rate at 60 months.
It is well known that cellular immunity is one of
the main antitumor processes (32);
thus, another important goal of immunotherapy is to stimulate
tumor-reactive T cells. The combined application of multiple
immunotherapeutic methods against specific tumors can improve
antitumor efficacy, delay the development of the disease and
prolong patient survival (32).
Immunological checkpoint inhibitors have demonstrated efficacy in
immunotherapy against melanoma and lung cancer, thus laying the
foundation for the clinical development of agents that target these
immune escape mechanisms in various solid tumors (32). The present study revealed that the
combination of anti-CTLA-4 antibody and 10 µM 5DC exhibited marked
enhancement on the lytic ability of CTLs in breast cancer cell
lines. Similar observations were further verified in primary breast
cancer cells.
In summary, the present study demonstrated that
MAGE-As common antigen peptide-specific CTLs in combination with
anti-CTLA-4 monoclonal antibody and a demethylating drug (5DC) can
produce potent tumor cell killing function on breast cancer. It
provides important insights into the future research of breast
cancer-targeted therapy. However, there are some limitations in the
present study. First, the limited number of IHC analyses may affect
the association between the positive expression of MAGE-As and the
prognosis of patients with breast cancer. Second, the immune
environment in vivo is complex, which may affect the
specific CTL killing function of MAGE-As thus affecting the results
of this study. The present results revealed that CTLA-4 blockade,
demethylating drug 5DC or a combination of both can improve the
killing effect of MAGE-As-specific CTLs. However, despite CTLA-4
exhibiting a highly positive expression rate in the four breast
cancer cell lines evaluated in the present study, there was no
significant difference between the CTL + anti-CTLA-4 + 5DC group
and the CTL + 5DC group. Further investigation should be conducted
to clarify the reason for these results. One possible explanation
may be the difference between in vitro and in vivo
environments. For example, in the body, an anti-CTLA-4 monoclonal
antibody may directly act on the tumor microenvironment, which can
act on activated T cells and on CTLA-4-positive cancer cells, and
relieve the immunosuppression of DCs by CTLA-4 molecules in the
tumor microenvironment. Second, the interaction time between an
anti-CTLA-4 monoclonal antibody and T cells in vitro is
limited by the culture time of cells, which is different from the
mechanism of repeated administration after a period of time in
vivo. Therefore, further studies on the effectiveness of CTLA-4
combined with MAGE-As common antigenic peptide in antitumor
therapy, and the new combined therapy mode and appropriate drug
concentration are required. Finally, combined treatment based on
MAGE-As and anti-CTLA-4 antibodies may be a simple, effective and
safer therapeutic method against cancer.
Acknowledgements
Not applicable.
Funding
The present study was supported by Hebei Natural
Science Foundation (grant no. H2016206410).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
WL and BS conceived and designed the experiments.
WL, MS, XH and YW performed the majority of the experiments. WL
performed the data analysis. BS wrote the manuscript. All authors
read the manuscript and approved the final version.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Affiliated Hospital of Hebei Medical University.
All patients involved in this study were informed of the
experimental content, purpose and significance of the study, and
signed the informed consent form.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zeng H, Zheng R, Zhang S, Zou X and Chen
W: Female breast cancer statistics of 2010 in China: Estimates
based on data from 145 population-based cancer registries. J Thorac
Dis. 6:466–470. 2014.PubMed/NCBI
|
|
3
|
Kakimi K, Karasaki T, Matsushita H and
Sugie T: Advances in personalized cancer immunotherapy. Breast
Cancer. 24:16–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang Y: Cancer immunotherapy: Harnessing
the immune system to battle cancer. J Clin Invest. 125:3335–3337.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Milani A, Sangiolo D, Aglietta M and
Valabrega G: Recent advances in the development of breast cancer
vaccines. Breast Cancer (Dove Med Press). 6:159–168.
2014.PubMed/NCBI
|
|
6
|
van der Bruggen P, Traversari C, Chomez P,
Lurquin C, De Plaen E, Van den Eynde B, Knuth A and Boon T: A gene
encoding an antigen recognized by cytolytic T lymphocytes on a
human melanoma. Science. 254:1643–1647. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Schooten E, Di Maggio A, van Bergen En
Henegouwen PMP and Kijanka MM: MAGE-A antigens as targets for
cancer immunotherapy. Cancer Treat Rev. 67:54–62. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Finn OJ: Human tumor antigens yesterday,
today, and tomorrow. Cancer Immunol Res. 5:347–354. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Adams S, Greeder L, Reich E, Shao Y,
Fosina D, Hanson N, Tassello J, Singh B, Spagnoli GC, Demaria S and
Jungbluth AA: Expression of cancer testis antigens in human
BRCA-associated breast cancers: potential targets for
immunoprevention? Cancer Immunol Immunother. 60:999–1007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Otte M, Zafrakas M, Riethdorf L,
Pichlmeier U, Löning T, Jänicke F and Pantel K: MAGE-A gene
expression pattern in primary breast cancer. Cancer Res.
61:6682–6687. 2001.PubMed/NCBI
|
|
11
|
Hou SY, Sang MX, Geng CZ, Liu WH, Lü WH,
Xu YY and Shan BE: Expressions of MAGE-A9 and MAGE-A11 in breast
cancer and their expression mechanism. Arch Med Res. 45:44–51.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Graff-Dubois S, Faure O, Gross DA, Alves
P, Scardino A, Chouaib S, Lemonnier FA and Kosmatopoulos K:
Generation of CTL recognizing an HLA-A*0201-restricted epitope
shared by MAGE-A1, -A2, -A3, -A4, -A6, -A10, and -A12 tumor
antigens: Implication in a broad-spectrum tumor immunotherapy. J
Immunol. 169:575–580. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Garrido F, Aptsiauri N, Doorduijn EM,
Garcia Lora AM and van Hall T: The urgent need to recover MHC class
I in cancers for effective immunotherapy. Curr Opin Immunol.
39:44–51. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Villarreal DO, Chin D, Smith MA, Luistro
LL and Snyder LA: Combination GITR targeting/PD-1 blockade with
vaccination drives robust antigen-specific antitumor immunity.
Oncotarget. 8:39117–39130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Davila E, Kennedy R and Celis E:
Generation of antitumor immunity by cytotoxic T lymphocyte epitope
peptide vaccination, CpG-oligodeoxynucleotide adjuvant, and CTLA-4
blockade. Cancer Res. 63:3281–3288. 2003.PubMed/NCBI
|
|
16
|
Chen X, Shao Q, Hao S, Zhao Z, Wang Y, Guo
X, He Y, Gao W and Mao H: CTLA-4 positive breast cancer cells
suppress dendritic cells maturation and function. Oncotarget.
8:13703–13715. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Emens LA: Breast cancer immunotherapy:
Facts and hopes. Clin Cancer Res. 24:511–520. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chomez P, De Backer O, Bertrand M, De
Plaen E, Boon T and Lucas S: An overview of the MAGE gene family
with the identification of all human members of the family. Cancer
Res. 61:5544–5551. 2001.PubMed/NCBI
|
|
19
|
De Plaen E, Arden K, Traversari C, Gaforio
JJ, Szikora JP, De Smet C, Brasseur F, van der Bruggen P, Lethé B,
Lurquin C, et al: Structure, chromosomal localization, and
expression of 12 genes of the MAGE family. Immunogenetics.
40:360–369. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Simpson AJ, Caballero OL, Jungbluth A,
Chen YT and Old LJ: Cancer/testis antigens, gametogenesis and
cancer. Nat Rev Cancer. 5:615–625. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sang M, Wang L, Ding C, Zhou X, Wang B,
Wang L, Lian Y and Shan B: Melanoma-associated antigen genes-an
update. Cancer Lett. 302:85–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Scanlan MJ, Gure AO, Jungbluth AA, Old LJ
and Chen YT: Cancer/testis antigens: An expanding family of targets
for cancer immunotherapy. Immunol Rev. 188:22–32. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ayyoub M, Scarlata CM, Hamai A, Pignon P
and Valmori D: Expression of MAGE-A3/6 in primary breast cancer is
associated with hormone receptor negative status, high histologic
grade, and poor survival. J Immunother. 37:73–76. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yang F, Zhou X, Miao X, Zhang T, Hang X,
Tie R, Liu N, Tian F, Wang F and Yuan J: MAGEC2, an
epithelial-mesenchymal transition inducer, is associated with
breast cancer metastasis. Breast Cancer Res Treat. 145:23–32. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Abd-Elsalam EA and Ismaeil NA:
Melanoma-associated antigen genes: A new trend to predict the
prognosis of breast cancer patients. Med Oncol. 31:2852014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Moreno-Bost A, Szmania S, Stone K, Garg T,
Hoerring A, Szymonifka J, Shaughnessy J Jr, Barlogie B, Prentice HG
and van Rhee F: Epigenetic modulation of MAGE-A3 antigen expression
in multiple myeloma following treatment with the demethylation
agent 5-azacitidine and the histone deacetlyase inhibitor MGCD0103.
Cytotherapy. 13:618–628. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goodyear O, Agathanggelou A,
Novitzky-Basso I, Siddique S, McSkeane T, Ryan G, Vyas P, Cavenagh
J, Stankovic T, Moss P and Craddock C: Induction of a CD8+ T-cell
response to the MAGE cancer testis antigen by combined treatment
with azacitidine and sodium valproate in patients with acute
myeloid leukemia and myelodysplasia. Blood. 116:1908–1918. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Baecher-Allan C, Brown JA, Freeman GJ and
Hafler DA: CD4+CD25 high regulatory cells in human peripheral
blood. J Immunol. 167:1245–1253. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mao H, Zhang L, Yang Y, Zuo W, Bi Y, Gao
W, Deng B, Sun J, Shao Q and Qu X: New insights of CTLA-4 into its
biological function in breast cancer. Curr Cancer Drug Targets.
10:728–736. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jaberipour M, Habibagahi M, Hosseini A,
Habibabad SR, Talei A and Ghaderi A: Increased CTLA-4 and FOXP3
transcripts in peripheral blood mononuclear cells of patients with
breast cancer. Pathol Oncol Res. 16:547–551. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ward FJ, Dahal LN, Wijesekera SK,
Abdul-Jawad SK, Kaewarpai T, Xu H, Vickers MA and Barker RN: The
soluble isoform of CTLA-4 as a regulator of T-cell responses. Eur J
Immunol. 43:1274–1285. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Marmé FL: Immunotherapy in breast cancer.
Oncol Res Treat. 39:335–345. 2016. View Article : Google Scholar : PubMed/NCBI
|