Introduction
Worldwide, breast cancer, which is induced by
diverse factors, such as heredity and lifestyle, is the most
frequently diagnosed cancer and the leading cause of cancer-related
deaths among women (1,2). Breast cancer stem-like cells (BCSCs)
are heterogeneous tumor cells with self-renewal ability and
multidifferentiation potential that participate in tumorigenesis,
metastasis and therapy resistance of breast cancer (3). Many studies have demonstrated that
BCSCs play an essential role in the poor clinical outcome of breast
cancer by investigating markers, ALDH(+), CD44(+) and CD24(−)
(4–7). In addition, the BCSC-related signaling
pathways, Wnt, Notch and hedgehog, are involved in malignant tumor
behavior, such as proliferation, colony formation and migration
(8–10). Therefore, drugs targeting these
signaling pathways are promising treatment strategies (11–13).
Hypoxia is a common tumor microenvironment
phenomenon that is characterized by decreased oxygen levels in
tissues. Hypoxia affects the survival of cells, including
disseminated tumor cells (DTCs), through transcriptional, metabolic
and mobility changes (14,15). Mitochondria are the major consumer
organelles, as well as the potential source of reactive oxygen
species (ROS). The lack of oxygen results in mitochondrial fusion
and fission, mitophagy, oxidative phosphorylation and eventually
mitochondrial dysfunction (16).
Hypoxia has been confirmed to exert a crucial effect on the
viability of CSCs (17). It has
become increasingly clear that hypoxia impacts cellular processes
through a group of genetic transcription activators termed
hypoxia-inducible factors (HIFs) (18). HIFs are composed of an alpha (HIF-α)
and a beta (HIF-β) subunit, and the oxygen-dependent HIF-α subunit
is the key protein determining the transcriptional activity of
HIFs, while the HIF-β subunit is structurally expressed in the cell
nucleus (19). A large number of
reports have revealed that oxygen deficit induces the generation of
HIFs in human embryonic stem cells (hESCs), which have the ability
to activate the Wnt and Notch self-renewal signaling pathways
(20,21) and vascular endothelial growth
factors (VEGFs) so that HIFs can maintain cells in an
undifferentiated state and promote tumor vessel formation (22).
MicroRNAs (miRNAs or miRs) are small noncoding
single-stranded RNAs with a length of 22 nucleotides that affect
the regulation of posttranscriptional gene expression (23). During a hypoxic period, specific
miRNAs, called hypoxia-regulated miRNAs (HRMs), are differentially
expressed, some of which are downregulated while others are
upregulated (24). MicroRNA-137
(miRNA-137 or miR-137) is considered a representative HRM (25). Li et al (26) concluded that miR-137 effectively
protected retinal ganglion cells against hypoxia-induced apoptosis
by attenuating Notch1 expression. One study demonstrated that
miR-137 could promote PI3K/AKT/mTOR and ERK pathway activation to
induce mitophagy and autophagy of brain cells exposed to hypoxia
(27). Moreover, microRNA-210
(miRNA-210 or miR-210) is regarded as the main hypoxia-controlled
RNA (24), and Wang et al
discovered that miR-210 exerted a negative effect on HIF1-α protein
expression, accompanied by the activation of T cells (28). Interestingly, results from various
experiments have shown that miRNAs share a strong connection with
mitochondria. Primarily, miRNAs affect mitochondrial metabolism and
dynamics by positively regulating translation (29–32).
Moreover, the levels of some miRNAs are abnormal in different types
of cancer (33). For example,
miR-34 accelerates gastric cancer progression by maintaining cancer
stemness properties (34).
The studies mentioned above provide insight into the
role of hypoxia in malignant tumor behaviors, including the
expression of miRNAs and the function of mitochondria (35–37).
However, the mechanism by which hypoxia regulates mitochondrial
function and mitophagy by affecting miRNA expression remains
largely unclear. In the present study, we examined the
mitochondrial function in BCSCs after deoxygenation and analyzed
the related microRNAs to characterize the underlying regulatory
mechanism.
Materials and methods
Cell culture
The human breast cancer cell lines SKBR-3, MCF-7,
MDA-MB-231 were acquired from the American Type Culture Collection
(ATCC). Cells were maintained in high-glucose Dulbecco's modified
Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific, Inc.)
containing 10% fetal bovine serum (FBS, Gibco; Thermo Fisher
Scientific, Inc.) and incubated at 37°C with 95% humidity and 5%
CO2.
Macrospheroid formation
SKBR-3, MCF-7 and MDA-MB-231 cells were cultured in
serum-free medium (SFM) consisting of DMEM/F-12 (Gibco; Thermo
Fisher Scientific, Inc.) (1:1), 2% B27 (Invitrogen; Thermo Fisher
Scientific, Inc.), 10 ng/ml EGF and 10 ng/ml bFGF (PeproTech,
Inc.). The medium was half-refreshed every three days. Three weeks
later, spheroids were obtained. The collected spheroids were
stained with an ALDEFLUOR™ assay kit (Stem Cell Technologies). The
proportions of newly formed tumor stem cell spheres were isolated
by ALDH-positive cell sorting.
Treatments
CoCl2 (Sigma-Aldrich; Merck KGaA) was
dissolved in DMEM at a concentration of 100 mM. To mimic hypoxic
conditions, 100 mM CoCl2 was added to the three breast
cancer cell lines for 24, 48, and 72 h, and BCSCs were transfected
with miR-137 mimics for 12 and 24 h for further examination. LW-6
(cat. no. S8441; Selleck Chemicals) was dissolved in dimethyl
sulfoxide (DMSO) at a concentration of 25 mM. BCSCs were treated
with DMEM containing 10 ng/ml LW-6 or DMSO (control group) for 12
or 24 h to explore the role of miR-137 mimics.
Cell transfection
BCSCs were cultured in 6-well plates with DMEM
containing 10% FBS. For each well, 5 µl Lipofectamine®
2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.) was
diluted in 250 µl Opti-MEM I reduced serum medium (Gibco; Thermo
Fisher Scientific, Inc.). A total of 62.5 pmol of miR-137 mimics or
miR-137 negative control mimics, miR-210 inhibitor or miR-210
negative control inhibitor (RiboBio) was diluted in 250 µl Opti-MEM
and incubated at room temperature for 5 min. A total of 250 µl
diluted miRNAs and 250 µl diluted reagent Lipofectamine®
2000 were mixed and used according to the manufacturer's
instructions. The transfected cells were incubated at 37°C with 95%
humidity and 5% CO2. After 12 h, when transfection
efficiency >80% was observed by fluorescence microscopy,
relevant experiments were subsequently performed.
Aldefluor assay
The cells were washed with phosphate-buffered saline
(PBS) twice and centrifuged at 150 × g for 5 min. The cell
concentration was adjusted to approximately 2×105
cells/ml suspended in assay buffer. A 1 ml sample suspension was
placed into each test tube. Next, 5 µl of ALDEFLUOR™ DEAB reagent
was placed into the control tube, and 5 µl of activated ALDEFLUOR™
reagent (StemCell Technologies) was added into the test tube. Then,
0.5 ml of the mixed solution was transferred from the test tube to
the control tube. After incubation for 30 min at 37°C, all tubes
were centrifuged at 250 × g for 5 min, and the supernatant was
removed. Finally, the Alde Fluor assay was performed by a 3-laser
Navios flow cytometer (Beckman Coulter). The data files were
analyzed using FlowJo 9.7.6 (FlowJo LLC).
Western blot analysis
The cell lysates were extracted using radio
immunoprecipitation assay (RIPA) lysis buffer and the protein
concentrations were measured using BCA Protein assay kit
(Sigma-Aldrich; Merck KGaA). An amount of 20 µg of total proteins
was separated on 10–15% sodium dodecyl sulfate-polyacrylamide gel
electrophoresis (SDS-PAGE) and transferred onto polyvinylidene
fluoride membranes (PVDF; Thermo Fisher Scientific, Inc.). After
blocking with 5% milk in 0.1% Tween-20 PBS (PBS-T) at room
temperature for 1 h, the membranes were incubated with primary
antibodies for Oct-4 (dilution 1:1,000, cat. no. ab222233), Nanog
(dilution 1:2,000, cat. no. ab218524), Fundc1 (dilution 1:1,000,
cat. no. ab224722), NIX (dilution 1:1,000, cat. no. ab8399), LC3B
(dilution 1:500, cat. no. ab48394), Tomm20 (dilution 1:2,000, cat.
no. ab56783), VADC (dilution 1:2,000, cat. no. ab81673), Tim23
(dilution 1:2,000, cat. no. ab230253) and β-actin (dilution
1:5,000, cat. no. ab8226) at 4°C overnight. Then, the membranes
were washed three times with PBS-T and incubated with secondary
antibodies (goat anti-rabbit IgG antibody, dilution 1:5,000, cat.
no. 6721) at room temperature for 1 h. All antibodies were
purchased from Abcam. After washing with PBS-T three times, blots
were imaged and analyzed on an X-ray film and quantified using
ImageJ software (version 1.46; National Institutes of Health,
Bethesda, MD, USA).
Cell viability assay
Cells (1×104) were seeded into 96-well
plates and allowed to attach overnight. To detect cell viability,
the medium was replaced with fresh medium, and 10 µl of Cell
Counting Kit-8 (CCK-8) (Sigma-Aldrich; Merck KGaA) solution was
added to each well for 1 h at 37°C. The optical density (OD) was
analyzed using a multifunctional microplate reader (FlexStation 3;
Molecular Devices) within 2 h.
Reverse transcription and quantitative
PCR (RT-qPCR)
Cells were lysed using SoniConvert®
sonicator (DocSense) following the manufacturer's instructions and
then total RNA was isolated using the miRcute miRNA Isolation kit
[Tiangen Biotech (Beijing) Co., Ltd.]. Reverse transcription was
performed using SYBR® Green PCR Master Mix (Thermo
Fisher Scientific, Inc.) following the manufacturer's instructions.
The primer sequences were as follows: miR-137 (forward
5′-GCGCGCTTATTGCTTAAGAATAC-3′ and reverse
5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCTACGC-3′); miR-210
(forward 5′-CCAGTGCAGGGTCCGAGGTATTC-3′ and reverse
5′-TGTCGGTGTGACAGGCGC-3′); miR-9 (forward
5′-CGTTCGTCTCTTTGGTTATCTAGC-3′ and reverse
5′-AATGGTTGATCTGCTCTCTCTCTC-3′); mtF805
(5′-CCACGGGAAACAGCAGTGATT-3′); mtR927
(5′-CTATTGACTTGGGTTAATCGTGTGA-3′); COXI (upstream
5′-AGCAGGAATAGTAGGGA-3′ and downstream 5′-GGAATCAGTGGACGAA-3′); ND1
(upstream 5′-ATACGCCCTCTAACCACC-3′ and downstream
5′-GAATTTGAGGCTCATCCC-3′); β-actin (forward
5′-CATGTACGTTGCTATCCAGGC-3′ and reverse
5′-CTCCTTAATGTCACGCACGAT-3′). All primers were purchased from
Sangon Biotech. Quantitative real-time polymerase chain reaction
(qPCR) of miR-137 in cells was conducted using the Applied
Biosystems 7500 Fast Real-Time PCR system (Applied Biosystems). The
amplification conditions were as follow: 40 cycles of denaturation
at 95°C for 15 sec, annealing and extension at 60°C for 1 min. The
calculations of expression of target genes were relative
quantification, and 2−ΔΔCq represented relative gene
expression (38). Each sample was
tested three times. β-actin was used as an internal control for
normalization.
Cell cycle assay
A total of 2×106 cells were fixed with
75% ice-cold ethanol for 12 h at 4°C. Fixed cells were pelleted by
centrifugation and washed with ice-cold PBS. Then, 400 µl PBS
containing 50 µg/ml propidium iodide (PI) (Sigma-Aldrich; Merck
KGaA), 100 µg/ml RNase A and 0.2% Triton X-100 was added to the
cells and coincubated for 30 min in the dark. Cells were analyzed
for cell cycle phase distribution by a 3-laser Navios flow
cytometer (Beckman Coulter). The data were analyzed using FlowJo
9.7.6 (FlowJo LLC).
Apoptosis assay
A total of 1×106 cells were suspended in
100 µl 1X binding buffer, 5 µl Annexin V fluorescein isothiocyanate
and 5 µl propidium iodide (PI; BD Biosciences) in the dark for 20
min at room temperature. Then, 400 µl of 1X binding buffer was
added, and stained cells were analyzed by using a 3-laser Navios
flow cytometer (Beckman Coulter). The data were analyzed using
FlowJo 9.7.6.
Measurement of mitochondrial ROS in
BCSCs
Cells were washed with PBS three times and incubated
with working solution containing 5 µM MitoSOX™ reagent (Thermo
Fisher Scientific, Inc.) for 10 min at 37°C in the dark. Then, the
cells were washed with PBS and imaged using an X71 (U-RFL-T)
fluorescence microscope at ×400 magnification (Olympus).
Measurement of intracellular ROS in
BCSCs
After being washed with ice-cold PBS three times,
the cells were resuspended in pre-warmed PBS containing 5 µM
intracellular ROS-specific fluorescent probe staining reagent
H2DCFDA (Thermo Fisher Scientific, Inc.) and then
incubated in the dark for 30 min at 37°C. After being washed three
times with pre-warmed PBS, the cells were imaged using an X71
(U-RFL-T) fluorescence microscope at ×400 magnification.
Citrate synthase activity assay
A total of 5×104 cells were collected and
rinsed with ice-cold PBS twice. The cell pellet was solubilized at
a concentration of 2×107/ml in extraction buffer and
then incubated on ice for 20 min. After 1,600 × g centrifugation
for 20 min, the supernatants were transferred into a new tube.
Thereafter, 100 µl of sample was added to each well and incubated
hermetically for 3 h to capture the enzyme. The suspension was
extracted and washed twice using 300 µl 1X washing buffer. Then,
100 µl 1X Citrate Synthase Activity Solution (cat. no. ab119692,
Abcam) was added. Finally, the absorbance was analyzed for 5–30 min
at 20-sec intervals by a microplate reader (Synergy 2 Multi-Mode
Microplate Reader, BioTek) at a wavelength of 412 nm.
ATP synthesis assay
A total of 1×104 cells were collected and
incubated with 200 µl of cell lysis buffer according to the
instructions of the ATP Detection kit (Beyotime Institute of
Biotechnology). The cells were pipetted up and down several times
and centrifuged at 1,200 × g for 5 min. Then, 100 µl supernatant
was moved into the prepared well for 5 min at room temperature.
Then, the luminescence of ATP was measured by a microplate reader
(Synergy 2 Multi-Mode Microplate Reader, BioTek).
Evaluation of GFP-LC3
fluorescence
A total of 1×105 cells were transfected
with the GFP-LC3 plasmid (Addgene) for 24 h and then re-transfected
with miR-137 mimics or NC mimics for another 24 h. Then, the cells
were washed with PBS three times and incubated with 5 µM
MitoTracker Red (Thermo Fisher Scientific, Inc.) for 30 min at 37°C
in the dark. Cells were fixed with 4% paraformaldehyde for 10 min
at room temperature and re-stained with
4′6-diamidino-2-phenylindole (DAPI, diluted 1:5,000; Thermo Fisher
Scientific, Inc.). The colocalization of GFP-LC3 (green dots) and
mitochondria (red dots) was imaged using an X71 (U-RFL-T)
fluorescence microscope at ×400 magnification.
Statistical analysis
All data were analyzed using SPSS 20.0 software (IBM
Corp.). Data are presented as the means ± SD. The experimental
group and control group were compared using Student's t-test. ANOVA
followed by Tukey's analysis was used to determine significance for
multiple group comparisons. P<0.05 was assigned to indicate a
significant difference.
Results
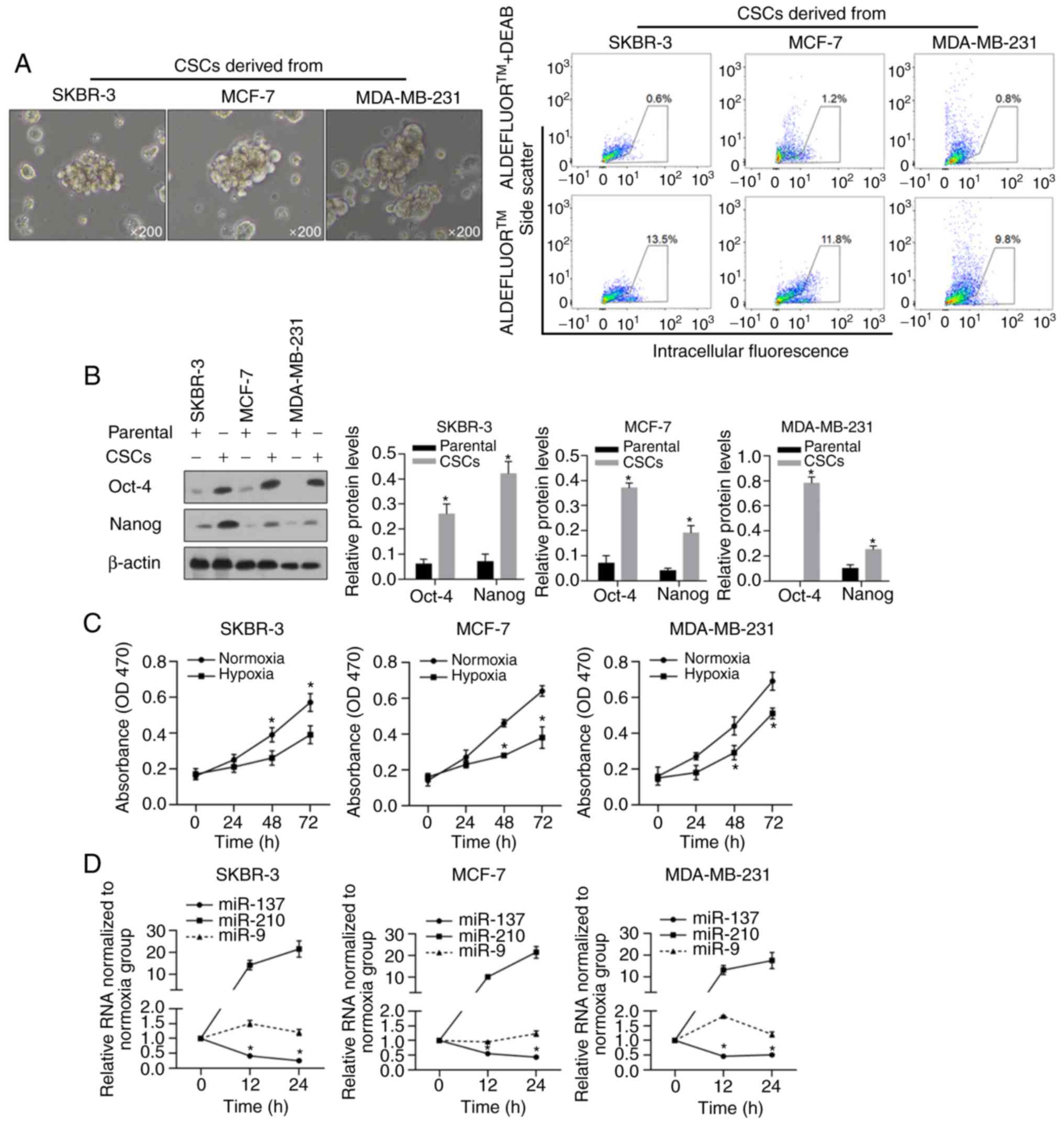
Enrichment and characterization of
BCSCs
To enrich the BCSCs, we cultured human breast cancer
cells, SKBR-3, MCF-7, and MDA-MB-231, in serum-free medium with the
necessary supplementary cytokines as described previously, and
macrospheroids were observed in all of these cell cultures
(Fig. 1A, left panel), all of which
presented enriched ALDH-positive subpopulations (Fig. 1A, right panel). By performing
western blotting, the widely reported stem cell markers Nanog and
Oct-4, which have been positively linked with the
epithelial-mesenchymal transition (EMT) of cancer stem cells and
poor outcomes (39–42), presented significantly higher
expression levels in macrospheroids compared to their levels in
parental cells (Fig. 1B,
P<0.05). Then, hypoxic conditions were mimicked by culturing
cells in medium supplemented with 100 mM CoCl2, and cell
viability was evaluated at the 0, 24, 48 and 72 h time points. As
shown in Fig. 1C, hypoxic
conditions significantly decreased the viability of all BCSCs, as
expected. To detect the expression of miR-137 and miR-210, which
have been reported as two hypoxia-responsive microRNAs, the
expression in these cells was detected at 12 and 24 h after hypoxia
exposure. As expected, without interfering with miR-9, miR-210 was
upregulated, while miR-137 was downregulated, in all assessed cell
lines (Fig. 1D, P<0.05).
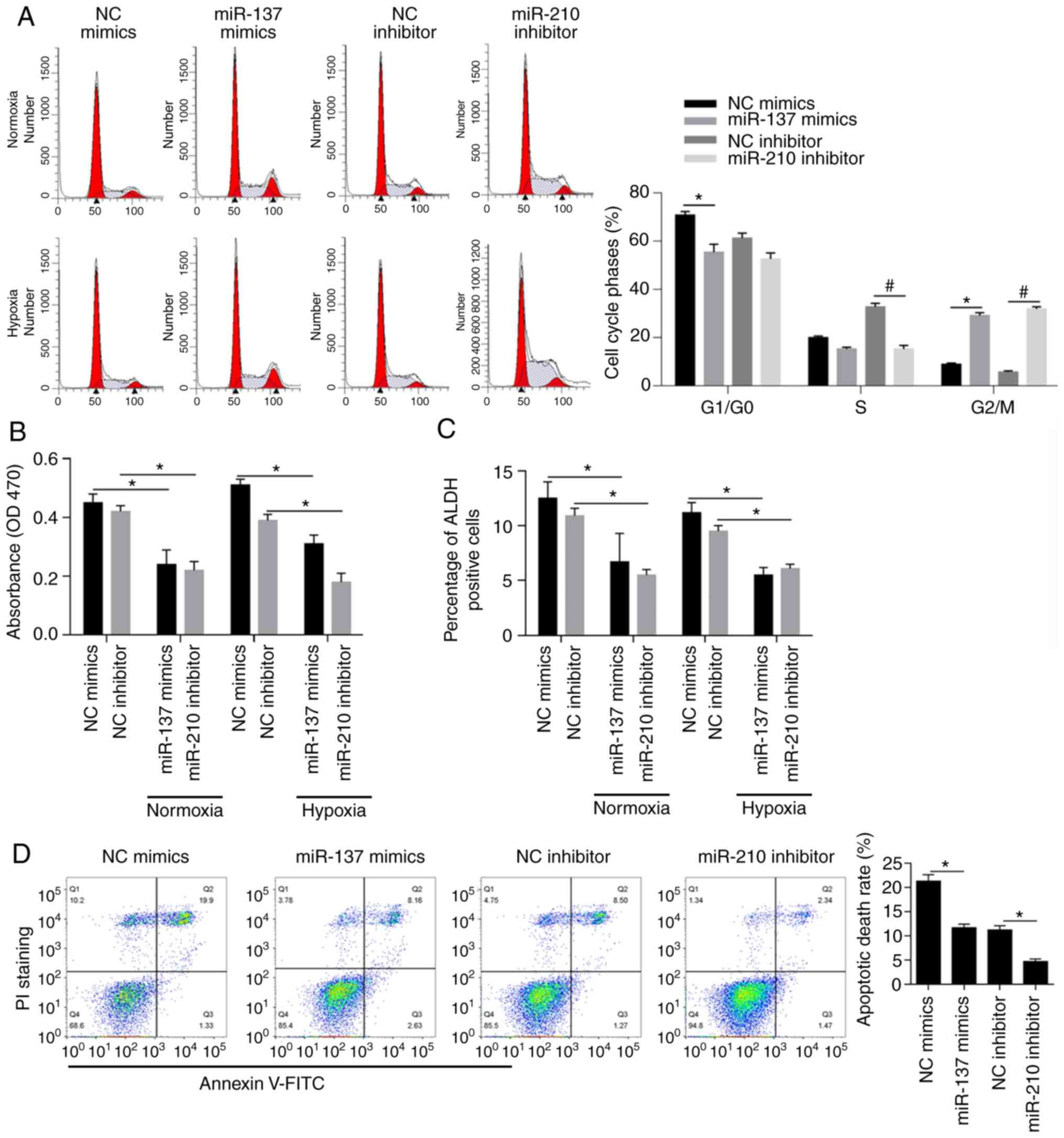
Effects of miR-137 and miR-210 on cell
cycle distribution, cell viability, cell stemness and apoptosis in
normoxic and hypoxic conditions
The abovementioned macrospheroids presented similar
gene expression profiles, which prompted us to conduct an analysis
of hypoxia-responsive microRNAs focusing on MCF-7 BCSCs. To
determine the effects of miR-137 and miR-210 on the distribution of
cells in cell cycle phases, mimics targeted to miR-137 (miR-137
mimics) or negative control mimics (NC mimics) were transfected
into cells, and miR-210 inhibitor or negative control inhibitor (NC
inhibitor) was also used for transfection. To confirm the
introduction efficacy, after 24 h, qPCR was performed and the
results indicated the significant overexpression of miR-137 and
miR-210 inhibitor (Fig. S1). The
results of PI staining followed by flow cytometry showed that
miR-137 overexpression and miR-210 inhibition promoted cell cycle
entry by decreasing BCSCs in the G1/G0 phase accompanied with
significant increase in G2/M phase simultaneously (Fig. 2A, P<0.05). Moreover, the decrease
in cell viability shown in Fig. 2B
(P<0.05) demonstrated that high levels of miR-137 or low levels
of miR-210 exerted inhibitory effects on BCSCs in normoxic and
hypoxic conditions respectively. ALDH-positive cells, known to be
closely associated with the stemness of BCSCs (43–45),
were found to be decreased upon overexpression of miR-137 or
inhibition of miR-210 (Fig. 2C,
P<0.05) both in normoxia and hypoxia. As expected, the apoptotic
rate was negatively associated with overexpression of miR-137 and
inhibition of miR-210 (Fig. 2D,
P<0.05). Overall, miR-137 and miR-210 exerted protective effects
on MCF-7 BCSCs.
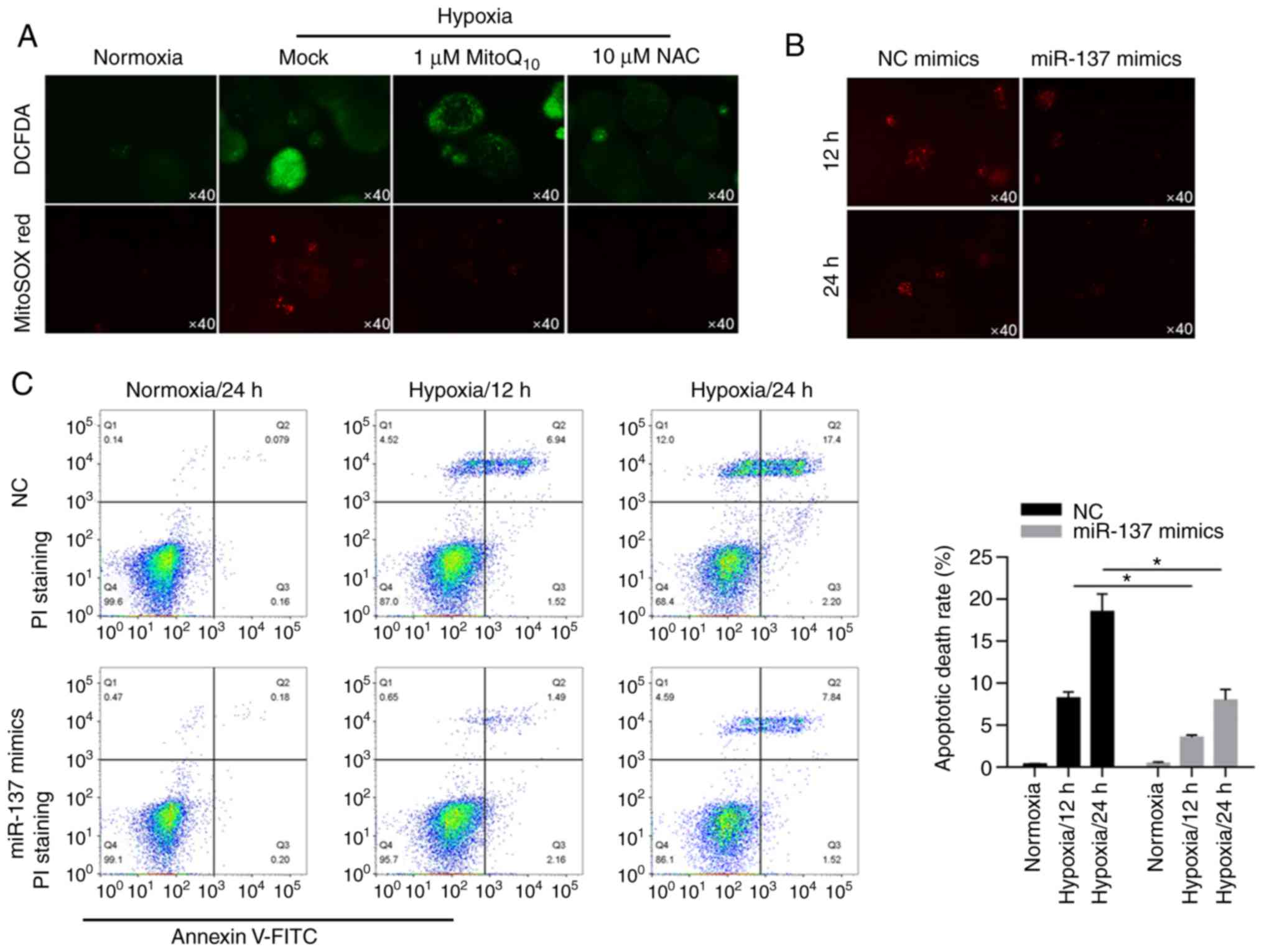
miR-137 significantly inhibits the
increase in ROS caused by hypoxia to suppress apoptotic cell
death
By performing fluorescent staining experiments, we
found that hypoxia induced ROS accumulation in cells and
mitochondria compared with the two antioxidant enzyme groups
(Fig. 3A). Interestingly,
overexpression of miR-137 resulted in less production of ROS under
hypoxic conditions (Fig. 3B). To
further explore the influence of miR-137 on hypoxia-induced
apoptotic cell death, apoptotic cell death was evaluated by Annexin
V/PI double staining followed by flow cytometric analysis. These
data indicated that in BCSCs, miR-137 had a protective effect on
apoptotic cell death (Fig. 3C,
P<0.05).
miR-137 restores cell mitochondrial
dysfunction by inhibiting mitophagy
As numerous articles have reported previously,
hypoxia clearly stimulates mitophagy and leads to mitochondrial
inactivation (46,47), which prompted us to investigate the
role of miR-137 induced by hypoxia in the regulation of MCF-7 BCSC
mitochondrial dysfunction. Fundc1, NIX and LC3, representative
mitophagy receptor proteins and autophagy-related proteins
(25,48,49),
were markedly decreased (Fig. 4A).
In contrast, Tom20 and VADC, mitochondrial-specific proteins
(50,51), were obviously increased.
Accordingly, an increase in mitochondrial DNA levels was detected
in hypoxic BCSCs after miR-137 transfection by qPCR, which
illustrated that miR-137 had a positive correlation with
mitochondrial functions (Fig. 4B,
P<0.05). As shown in Fig. 4C and
D (P<0.05), citrate synthase activity and the levels of
synthesized ATP were notably upregulated by the overexpression of
miR-137 relative to those in the NC group. Finally, we performed
mtDNA transcript analysis in normoxic, mock-, negative control- and
miR-137 mimic-transfected cell extracts using COXI and ND1
(Fig. 4E). It was concluded that
hypoxia-mediated inhibition of mitophagy and mitochondrial
dysfunction were significantly reversed by overexpression of
miR-137 (P<0.05).
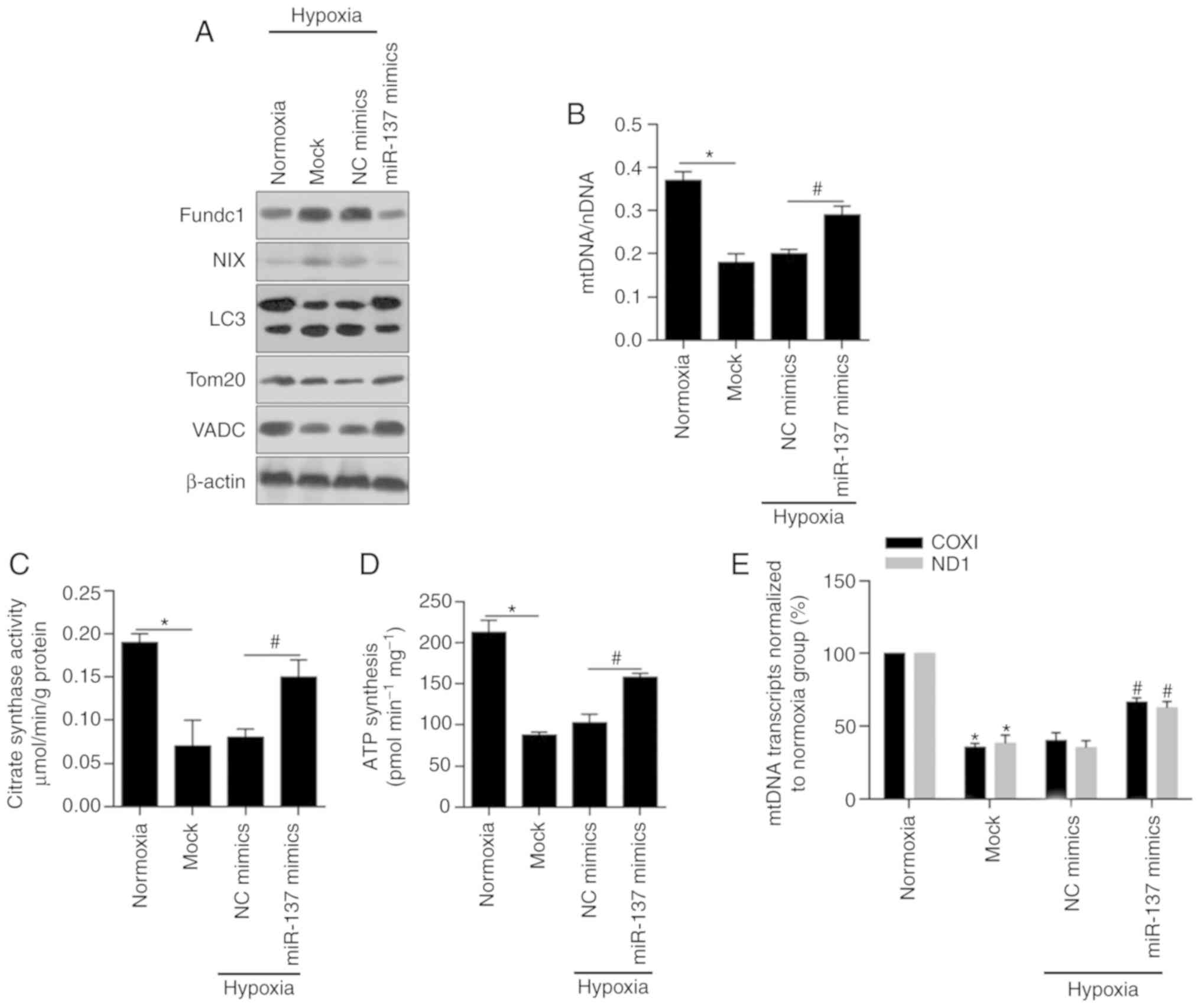 | Figure 4.miR-137 restores the cell
mitochondrial dysfunction by inhibiting mitophagy. (A) Western
blotting was performed to detect the protein levels of Fundc1, NIX,
LC3, Tom20 and VADC in BCSCs after introduction of miR-137 mimics.
(B) The mtDNA levels in BCSCs after introduction of miR-137 mimics
were assessed by RT-qPCR. *P<0.05, vs. Normoxia group;
#P<0.05, vs. NC mimics group. (C) Citrate synthase
activity was measured by Citrate Synthase Activity Colorimetric
Assay Kit. *P<0.05, vs. Normoxia group; #P<0.05,
vs. NC mimics group. (D) Synthesized ATP was analyzed by ATP
synthase Assay Kit. *P<0.05, vs. Normoxia group;
#P<0.05, vs. NC mimics group. (E) The mtDNA
transcription activity was measured by assessing the expression of
COXI and ND1. *P<0.05, vs. Normoxia group;
#P<0.05, vs. NC mimics group. BCSCs, breast cancer
stem-like cells; mtDNA, mitochondrial DNA; NIX, also known as
beclin-2 (BCL2); Fundc1, Fun14 domain-containing protein 1; VADC,
voltage-dependent anion channel; LC3, light chain 3; Tom20,
mitochondrial import receptor subunit TOM20 homolog; COXI,
cytochrome c oxidase I; ND1, NADH-ubiquinone oxidoreductase
chain 1; NC, negative control. |
miR-137 inhibits mitophagy by
posttranscriptionally regulating Fundc1
Considering previously reported findings (25,52),
Fundc1, a substrate of mitochondrially localized E3 ubiquitin
ligase, participates in hypoxia-induced mitophagy and its 3′UTR has
been proven to contain sites complimentary to miR-137. To further
confirm the posttranscriptional regulation of Fundc1 protein levels
by miR-137, we analyzed Fundc1 protein expression after
transfection of miR-137 mimics, and it was observed that the Fundc1
protein level was significantly decreased after miR-137 mimic
transfection (Fig. 5A, P<0.05).
A decrease in Fundc1 protein was observed to be inversely
associated with Tom20 and Tim23, indicating that miR-137
potentially affected mitochondrial mass by regulating Fundc1
(Fig. 5A). Notably, treatment with
LW-6, an inhibitor of the transcriptional activity of HIF-1,
exerted similar effects as the introduced miR-137 mimics,
indicating that the decrease in miR-137 was potentially caused by
transcriptional activity of HIF-1 under hypoxia (Fig. 5A). In experiments on LC3, decreased
LC3-II was detected after both miR-137 mimic transfection and LW-6
treatment, confirming that miR-137 mimics inhibited mitophagy
(Fig. 5B, P<0.05). Furthermore,
mitochondrial staining with MitoTracker Red, GFP-LC3 and DAPI
revealed that both miR-137 transfection and LW-6 treatment
decreased the colocalization of GFP-LC3 (green dots) with
mitochondria (red dots, Fig. 5C,
P<0.05). Overall, the HIF-1-mediated decrease in miR-137 induced
by hypoxia may inhibit mitophagy by targeting Fundc1.
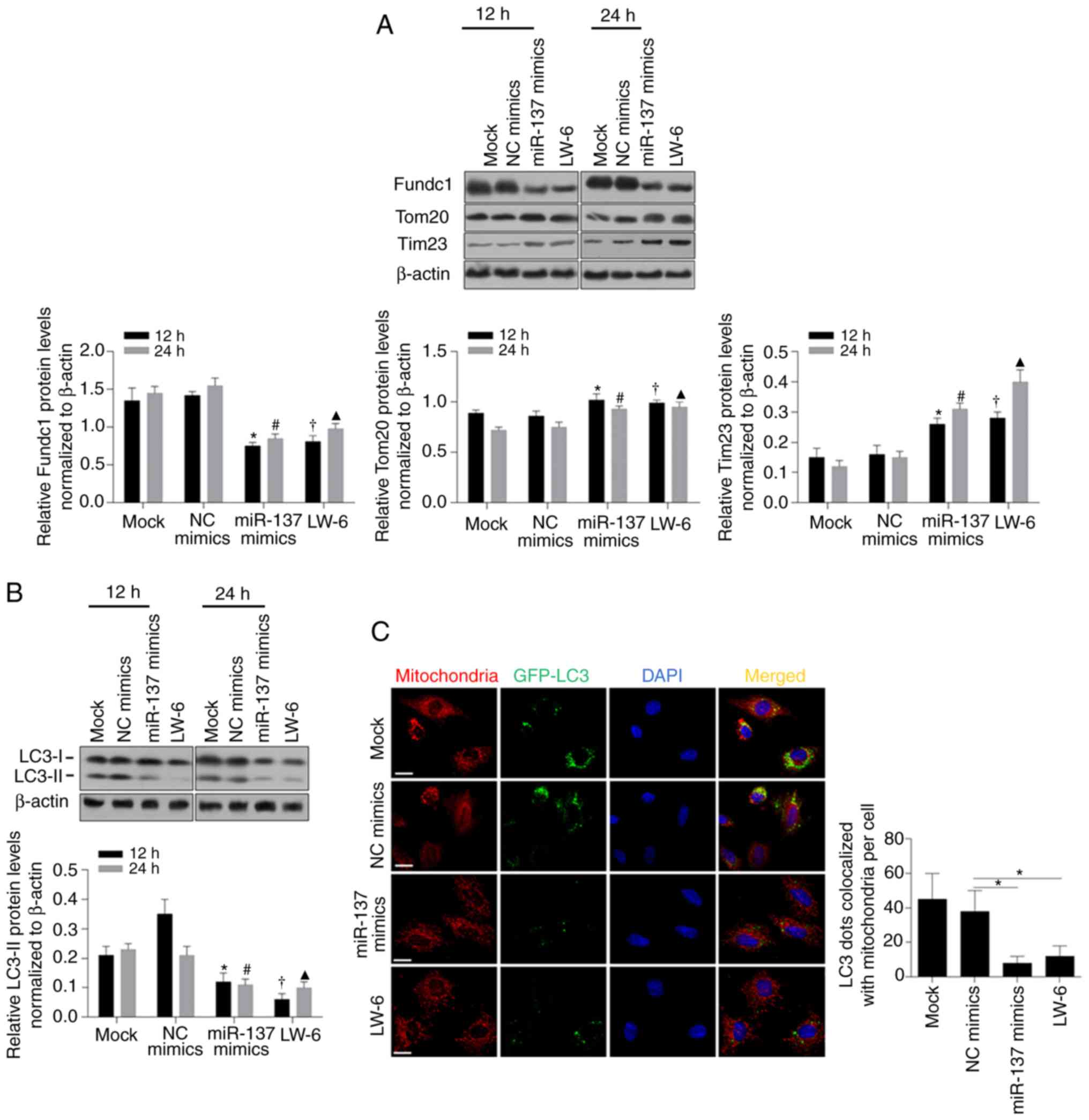 | Figure 5.miR-137 regulates mitophagy via
targeting Fundc1. (A) Western blotting was performed to detect
Fundc1, Tom20, and Tim23 after introduction of miR-137 mimics or
treatment with LW-6 after 12 or 24 h (upper panel). Fundc1, Tom20,
and Tim23 protein expression by statistical analysis (lower
panels). *P<0.05, vs. 12 h NC mimics group;
#P<0.05, vs. 24 h NC mimics group;
†P<0.05, vs. 12 h Mock group; ▲P<0.05,
vs. 24 h Mock group. (B) The protein expression level of LC3 (upper
panel). LC3-II protein expression by statistical analysis (lower
panel). *P<0.05, vs. 12 h NC mimics group;
#P<0.05, vs. 24 h NC mimics group;
†P<0.05, vs. 12 h Mock group; ▲P<0.05,
vs. 24 h Mock group. (C) The colocalization of LC3 with
mitochondria (left panel). LC3 dots colocalized with mitochondria
per cell by statistical analysis (right panel). *P<0.05, vs. NC
mimics group. Fundc1, Fun14 domain-containing protein 1; LC3, light
chain 3; Tom20, mitochondrial import receptor subunit TOM20
homolog; Tim23, mitochondrial import inner membrane translocase
subunit Tim23; NC, negative control. |
Discussion
In the present study, by performing analysis of stem
cell-like side populations, western blotting, CCK-8 and RT-PCR, it
was demonstrated that the macrospheroids of breast cancer stem-like
cells (BCSCs) were formed successfully when the cells were cultured
with three cell cytokines, and hypoxia reduced cell viability, as
anticipated. Additionally, low oxygen induced the downregulation of
miR-137 in breast cancer stem-like cell lines. To verify the
phenomenon described above, we further detected the expression of
miR-137 in BCSCs under hypoxic conditions. Overexpression of
miR-137 promoted BCSC survival but inhibited cell proliferation,
viability and stemness. To investigate the relationship between
miR-137 and BCSC mitochondria in hypoxia, we implemented a series
of mitochondria-relevant experiments. These data indicated that
overexpression of miR-137 led to a significant reduction in ROS in
abnormal mitochondria induced by hypoxia and suppressed mitophagy
while decreasing the protein levels of FUN14 domain containing 1
(Fundc1) and NIX. The function of intracellular mitochondria was
demonstrated to improve under treatment with miR-137.
ROS accumulation induced by hypoxia is frequently a
major cause of cell injury and thus promotes apoptotic cell death
in cancer cells (53). One of the
main reasons for its fatal effects is that ROS disrupt
mitochondrial homeostasis by decreasing mitochondrial membrane
potential, thus leading to mitochondrial dysfunction, including a
decrease in ATP synthesis and transcriptional activity (54). Notably, according to our results,
the introduction of miR-137 mimics decreased mitochondrial ROS
under hypoxia exposure and simultaneously significantly inhibited
the proliferation of BCSCs, which is considered to be
controversial. These conflicting results may be due to miR-137
scavenging ROS and inhibition of proliferation in two independent
mechanisms. Another possibility is that miR-137 may sensitize cells
to accumulated ROS, resulting in low cell viability and low ROS
levels.
miR-137, found mainly in the brain (55,56),
is involved in many regulatory mechanisms of different types of
cancer. In ulcerative colitis-associated colorectal cancer
patients, methylation of miR-137 showed a rising trend related to
age (57). Cui et al also
reported lower expression of miR-137 in hepatocellular carcinoma in
pathological tissue compared with peripheral tissue (58). Another study showed that miR-137
inhibited the growth of oligodendroglial tumor cells by targeting
CSE1L (59). miR-137 was found to
play a negative role in the therapeutic resistance to drugs in
breast cancer cell lines, including fulvestrant (60) and FSTL1 targeting chemoresistance
(61). In addition, miR-137 was
also found to play an important role in the EMT of breast cancer
cells concurrent with the reduction in bone morphogenetic protein-7
(BMP7) (62), the upregulation of
E-cadherin and the downregulation of N-cadherin and vimentin
(63). Notably, many studies have
identified miR-137 regulation of cancer stem-like cell stemness in
recent years. For example, He et al (64) found that targeting of KLF12 by
miR-137 inhibited the CSC phenotype of pancreatic cancer cells by
blocking Wnt/β-catenin signaling activity. In addition, miR-137
suppressed the Wnt/β-catenin signaling pathway, breast cancer cell
stemness, and multi-drug chemoresistance by negatively regulating
FSTL1 expression (61).
However, their physiological roles have, until now,
not been elucidated in BCSCs. Thus, we used CoCl2 to
create a low-oxygen environment, which showed that hypoxia could
enhance BCSC mitophagy. Notably, after overexpression of miR-137,
BCSCs exhibited the opposite tendencies in cell growth and cell
apoptosis. These findings showed that upregulation of miR-137 not
only improved BCSC survival but also inhibited BCSC stemness.
Subsequently, we explored the intracellular changes in mitochondria
in BCSCs cocultured with miR-137 mimics. Our data revealed that the
overexpression of miR-137 inhibited cell apoptosis by reducing
hypoxia-induced abnormal mitochondrial homeostasis. Moreover, after
carrying out further protein experiments, we found an inhibitory
role of miR-137 in mitophagy in BCSCs. In the present study, we
established for the first time a new regulatory pathway for
miR-137, malignant behavior, and mitochondrial activity in BCSCs.
Interestingly, our findings that the miR-137-mediated
downregulation of mitophagy inhibited hypoxia-induced BCSC
apoptosis and maintained mitochondrial function are related to the
study by Chen et al showing that overexpression of miR-137
inhibited stemness of triple negative breast cancer (TNBC) cells
and tumor formation by downregulating B-cell lymphoma/leukaemia 11A
(65). However, Chen et al
indicated that the miR-137-mediated disruption of the B-cell
lymphoma/leukaemia 11A-DNA methyltransferase 1 interaction impaired
BCSC stemness and tumorigenesis by suppressing ISL-1. Similarly, we
confirmed the negative regulation of mitochondrial metabolism by
miR-137, which resulted in failure to provide enough energy to
maintain BCSC stemness under nonphysiological hypoxia conditions,
which demonstrated another regulatory effect of miR-137 on BCSC
stemness and tumor development. The results of the present study
further confirmed the protective effect of miR-137 in
hypoxia-induced BCSC mitochondrial dysfunction, which establishes a
connection between microRNA and mitophagy receptors bound to LC3
involved in mitophagy.
Although important findings have been revealed by
the present study, there are also limitations. It should be noted
that this study examined only protein alterations of Fundc1. The
results are incomplete and cannot be taken as evidence that Fundc1
is a direct target of miR-137. Bioinformatics analysis and dual
luciferase reporter assays should be performed to evaluate the
direct binding of miR-137 with its targets. Moreover, no study was
conducted using nude mice, and whether miR-137 is effective in the
prevention of breast cancer stemness and tumor formation still
requires further research and verification. In addition, miR-210
has been reported to negatively regulate E-cadherin in hypoxic
BCSCs to promote cell metastasis, proliferation, and self-renewal
(66). In the present study, we
also discovered the activation of miR-210, which indicated the
possibility of targeting miR-210 in hypoxia-induced cell injury.
This finding prompted us to determine whether an inhibitor of
miR-210 exerts its therapeutic effects by regulating the expression
of mitophagy-related proteins consistent with the introduction of
miR-137. Overall, we can clearly outline our next steps. In
summary, considering all the molecular analyses of BCSCs mentioned
above, we tentatively propose that miR-137 may be a potential
therapeutic target for breast cancer. Moreover, further
investigations are needed to clarify whether miR-137 is an
effective and safe therapy for breast cancer in humans and
elucidate its comprehensive molecular regulatory mechanism on BCSC
mitophagy.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural
Science Foundation of China (nos. 81601835 and 81973684), the
Department of Science and Technology of Sichuan Province (no.
18YYJC0689) and the Health and Family Planning Commission of
Sichuan Province (no. 18ZD039).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
ZZ and CX contributed to the study design. QH and YY
contributed to data collection and figure construction. YW and YH
contributed to the literature search. All authors read and approved
the manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors state that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Momenimovahed Z and Salehiniya H:
Epidemiological characteristics of and risk factors for breast
cancer in the world. Breast Cancer (Dove Med Press). 11:151–164.
2019.PubMed/NCBI
|
|
3
|
Sousa B, Ribeiro AS and Paredes J:
Heterogeneity and plasticity of breast cancer stem cells. Adv Exp
Med Biol. 1139:83–103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kong Y, Lyu N, Wu J, Tang H and Xie X,
Yang L, Li X, Wei W and Xie X: Breast cancer stem cell markers CD44
and ALDH1A1 in serum: Distribution and prognostic value in patients
with primary breast cancer. J Cancer. 9:3728–3735. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sjöström M, Hartman L, Honeth G, Grabau D,
Malmström P, Hegardt C, Fernö M and Niméus E: Stem cell biomarker
ALDH1A1 in breast cancer shows an association with prognosis and
clinicopathological variables that is highly cut-off dependent. J
Clin Pathol. 68:1012–1019. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang F, Cao L, Sun Z, Jin J, Fang H, Zhang
W and Guan X: Evaluation of breast cancer stem cells and intratumor
stemness heterogeneity in triple-negative breast cancer as
prognostic factors. Int J Biol Sci. 12:1568–1577. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Seo AN, Lee HJ, Kim EJ, Jang MH, Kim YJ,
Kim JH, Kim SW, Ryu HS, Park IA, Im SA, et al: Expression of breast
cancer stem cell markers as predictors of prognosis and response to
trastuzumab in HER2-positive breast cancer. Br J Cancer.
114:1109–1116. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sulaiman A, McGarry S, Li L, Jia D, Ooi S,
Addison C, Dimitroulakos J, Arnaout A, Nessim C, Yao Z, et al: Dual
inhibition of Wnt and Yes-associated protein signaling retards the
growth of triple-negative breast cancer in both mesenchymal and
epithelial states. Mol Oncol. 12:423–440. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yuan X, Wu H, Xu H, Xiong H, Chu Q, Yu S,
Wu GS and Wu K: Notch signaling: An emerging therapeutic target for
cancer treatment. Cancer Lett. 369:20–27. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
O'Toole SA, Machalek DA, Shearer RF,
Millar EK, Nair R, Schofield P, McLeod D, Cooper CL, McNeil CM,
McFarland A, et al: Hedgehog overexpression is associated with
stromal interactions and predicts for poor outcome in breast
cancer. Cancer Res. 71:4002–4014. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Palomeras S, Ruiz-Martínez S and Puig T:
Targeting breast cancer stem cells to overcome treatment
resistance. Molecules. 23:21932018. View Article : Google Scholar
|
|
12
|
Nwabo Kamdje AH, Seke Etet PF, Vecchio L,
Muller JM, Krampera M and Lukong KE: Signaling pathways in breast
cancer: Therapeutic targeting of the microenvironment. Cell Signal.
26:2843–2856. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Maruthanila VL, Elancheran R, Kunnumakkara
AB, Kabilan S and Kotoky J: Recent development of targeted
approaches for the treatment of breast cancer. Breast Cancer.
24:191–219. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nobre AR, Entenberg D, Wang Y, Condeelis J
and Aguirre-Ghiso JA: The different routes to metastasis via
hypoxia-regulated programs. Trends Cell Biol. 28:941–956. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Abe H, Semba H and Takeda N: The roles of
hypoxia signaling in the pathogenesis of cardiovascular diseases. J
Atheroscler Thromb. 24:884–894. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fuhrmann DC and Brüne B: Mitochondrial
composition and function under the control of hypoxia. Redox Biol.
12:208–215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tong WW, Tong GH and Liu Y: Cancer stem
cells and hypoxia-inducible factors (Review). Int J Oncol.
53:469–476. 2018.PubMed/NCBI
|
|
18
|
Keith B and Simon MC: Hypoxia-inducible
factors, stem cells, and cancer. Cell. 129:465–472. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gonzalez FJ, Xie C and Jiang C: The role
of hypoxia-inducible factors in metabolic diseases. Nat Rev
Endocrinol. 15:21–32. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mazumdar J, O'Brien WT, Johnson RS,
LaManna JC, Chavez JC, Klein PS and Simon MC: O2
regulates stem cells through Wnt/β-catenin signalling. Nat Cell
Biol. 12:1007–1013. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang Z, Han H, Rong Y, Zhu K, Zhu Z, Tang
Z, Xiong C and Tao J: Hypoxia potentiates gemcitabine-induced
stemness in pancreatic cancer cells through AKT/Notch1 signaling. J
Exp Clin Cancer Res. 37:2912018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Catalano V, Turdo A, Di Franco S, Dieli F,
Todaro M and Stassi G: Tumor and its microenvironment: A
synergistic interplay. Semin Cancer Biol. 23:522–532. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mohr AM and Mott JL: Overview of microRNA
biology. Semin Liver Dis. 35:3–11. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bandara KV, Michael MZ and Gleadle JM:
MicroRNA biogenesis in hypoxia. Microrna. 6:80–96. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li W, Zhang X, Zhuang H, Chen HG, Chen Y,
Tian W, Wu W, Li Y, Wang S, Zhang L, et al: MicroRNA-137 is a novel
hypoxia-responsive microRNA that inhibits mitophagy via regulation
of two mitophagy receptors FUNDC1 and NIX. J Biol Chem.
289:10691–10701. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li H, Zhu Z, Liu J, Wang J and Qu C:
MicroRNA-137 regulates hypoxia-induced retinal ganglion cell
apoptosis through Notch1. Int J Mol Med. 41:1774–1782.
2018.PubMed/NCBI
|
|
27
|
Chang J, Yan X and Zeng Y: Propofol
weakens hypoxia-aroused apoptosis and autophagy via elevating
microRNA-137 in neurocytes. Exp Mol Pathol. 112:1043272020.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang H, Flach H, Onizawa M, Wei L, McManus
MT and Weiss A: Negative regulation of Hif1a expression and TH17
differentiation by the hypoxia-regulated microRNA miR-210. Nat
Immunol. 15:393–401. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
el Azzouzi H, Leptidis S, Dirkx E, Hoeks
J, van Bree B, Brand K, McClellan EA, Poels E, Sluimer JC, van den
Hoogenhof MM, et al: The hypoxia-inducible microRNA cluster
miR-199a-214 targets myocardial PPARδ and impairs mitochondrial
fatty acid oxidation. Cell Metab. 18:341–354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chan SY, Zhang YY, Hemann C, Mahoney CE,
Zweier JL and Loscalzo J: MicroRNA-210 controls mitochondrial
metabolism during hypoxia by repressing the iron-sulfur cluster
assembly proteins ISCU1/2. Cell Metab. 10:273–284. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang X, Ji R, Liao X, Castillero E,
Kennel PJ, Brunjes DL, Franz M, Möbius-Winkler S, Drosatos K,
George I, et al: MicroRNA-195 regulates metabolism in failing
myocardium via alterations in sirtuin 3 expression and
mitochondrial protein acetylation. Circulation. 137:2052–2067.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li H, Zhang X, Wang F, Zhou L, Yin Z, Fan
J, Nie X, Wang P, Fu XD, Chen C and Wang DW: MicroRNA-21 lowers
blood pressure in spontaneous hypertensive rats by upregulating
mitochondrial translation. Circulation. 134:734–751. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ullmann P, Nurmik M, Begaj R, Haan S and
Letellier E: Hypoxia- and microRNA-induced metabolic reprogramming
of tumor-initiating cells. Cells. 8:5282019. View Article : Google Scholar
|
|
34
|
Jafari N and Abediankenari S: MicroRNA-34
dysregulation in gastric cancer and gastric cancer stem cell.
Tumour Biol. 39:10104283177016522017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Grosso S, Doyen J, Parks SK, Bertero T,
Paye A, Cardinaud B, Gounon P, Lacas-Gervais S, Noël A, Pouysségur
J, et al: miR-210 promotes a hypoxic phenotype and increases
radioresistance in human lung cancer cell lines. Cell Death Dis.
4:e5442013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gee HE, Ivan C, Calin GA and Ivan M:
HypoxamiRs and cancer: From biology to targeted therapy. Antioxid
Redox Signal. 21:1220–1238. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Qin Q, Furong W and Baosheng L: Multiple
functions of hypoxia-regulated miR-210 in cancer. J Exp Clin Cancer
Res. 33:502014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Almozyan S, Colak D, Mansour F, Alaiya A,
Al-Harazi O, Qattan A, Al-Mohanna F, Al-Alwan M and Ghebeh H: PD-L1
promotes OCT4 and Nanog expression in breast cancer stem cells by
sustaining PI3K/AKT pathway activation. Int J Cancer.
141:1402–1412. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yin X, Zhang BH, Zheng SS, Gao DM, Qiu SJ,
Wu WZ and Ren ZG: Coexpression of gene Oct4 and Nanog initiates
stem cell characteristics in hepatocellular carcinoma and promotes
epithelial-mesenchymal transition through activation of Stat3/Snail
signaling. J Hematol Oncol. 8:232015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang D, Lu P, Zhang H, Luo M, Zhang X, Wei
X, Gao J, Zhao Z and Liu C: Oct-4 and Nanog promote the
epithelial-mesenchymal transition of breast cancer stem cells and
are associated with poor prognosis in breast cancer patients.
Oncotarget. 5:10803–10815. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jeter CR, Yang T, Wang J, Chao HP and Tang
DG: Concise review: NANOG in cancer stem cells and tumor
development: An update and outstanding questions. Stem Cells.
33:2381–2390. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Charafe-Jauffret E, Ginestier C, Iovino F,
Tarpin C, Diebel M, Esterni B, Houvenaeghel G, Extra JM, Bertucci
F, Jacquemier J, et al: Aldehyde dehydrogenase 1-positive cancer
stem cells mediate metastasis and poor clinical outcome in
inflammatory breast cancer. Clin Cancer Res. 16:45–55. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ginestier C, Hur MH, Charafe-Jauffret E,
Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG,
Liu S, et al: ALDH1 is a marker of normal and malignant human
mammary stem cells and a predictor of poor clinical outcome. Cell
Stem Cell. 1:555–567. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Balicki D: Moving forward in human mammary
stem cell biology and breast cancer prognostication using ALDH1.
Cell Stem Cell. 1:485–487. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wu H and Chen Q: Hypoxia activation of
mitophagy and its role in disease pathogenesis. Antioxid Redox
Signal. 22:1032–1046. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhang H, Gao P, Fukuda R, Kumar G,
Krishnamachary B, Zeller KI, Dang CV and Semenza GL: HIF-1 inhibits
mitochondrial biogenesis and cellular respiration in VHL-deficient
renal cell carcinoma by repression of C-MYC activity. Cancer Cell.
11:407–420. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Liu L, Sakakibara K, Chen Q and Okamoto K:
Receptor-mediated mitophagy in yeast and mammalian systems. Cell
Res. 24:787–795. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wu X, Wu FH, Wu Q, Zhang S, Chen S and
Sima M: Phylogenetic and molecular evolutionary analysis of
mitophagy receptors under hypoxic conditions. Front Physiol.
8:5392017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
van der Laan M, Rissler M and Rehling P:
Mitochondrial preprotein translocases as dynamic molecular
machines. FEMS Yeast Res. 6:849–861. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
DeHart DN, Fang D, Heslop K, Li L,
Lemasters JJ and Maldonado EN: Opening of voltage dependent anion
channels promotes reactive oxygen species generation, mitochondrial
dysfunction and cell death in cancer cells. Biochem Pharmacol.
148:155–162. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen Z, Siraj S, Liu L and Chen Q:
MARCH5-FUNDC1 axis fine-tunes hypoxia-induced mitophagy. Autophagy.
13:1244–1245. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hielscher A and Gerecht S: Hypoxia and
free radicals: Role in tumor progression and the use of
engineering-based platforms to address these relationships. Free
Radic Biol Med. 79:281–291. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Shadel GS and Horvath TL: Mitochondrial
ROS signaling in organismal homeostasis. Cell. 163:560–569. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Arakawa Y, Yokoyama K, Tasaki S, Kato J,
Nakashima K, Takeyama M, Nakatani A and Suzuki M: Transgenic mice
overexpressing miR-137 in the brain show schizophrenia-associated
behavioral deficits and transcriptome profiles. PLoS One.
14:e02203892019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Guella I, Sequeira A, Rollins B, Morgan L,
Torri F, van Erp TG, Myers RM, Barchas JD, Schatzberg AF, Watson
SJ, et al: Analysis of miR-137 expression and rs1625579 in
dorsolateral prefrontal cortex. J Psychiatr Res. 47:1215–1221.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Toiyama Y, Okugawa Y, Tanaka K, Araki T,
Uchida K, Hishida A, Uchino M, Ikeuchi H, Hirota S, Kusunoki M, et
al: A panel of methylated MicroRNA biomarkers for identifying
high-risk patients with ulcerative colitis-associated colorectal
cancer. Gastroenterology. 153:1634–1646.e8. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Cui S, Sun Y, Liu Y, Liu C, Wang J, Hao G
and Sun Q: MicroRNA-137 has a suppressive role in liver cancer via
targeting EZH2. Mol Med Rep. 16:9494–9502. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Li KK, Yang L, Pang JC, Chan AK, Zhou L,
Mao Y, Wang Y, Lau KM, Poon WS, Shi Z and Ng HK: MIR-137 suppresses
growth and invasion, is downregulated in oligodendroglial tumors
and targets CSE1L. Brain Pathol. 23:426–439. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Guo J, He K, Zeng H, Shi Y, Ye P, Zhou Q,
Pan Z and Long X: Differential microRNA expression profiles
determined by next-generation sequencing in three
fulvestrant-resistant human breast cancer cell lines. Oncol Lett.
17:3765–3776. 2019.PubMed/NCBI
|
|
61
|
Cheng S, Huang Y, Lou C, He Y, Zhang Y and
Zhang Q: FSTL1 enhances chemoresistance and maintains stemness in
breast cancer cells via integrin β3/Wnt signaling under miR-137
regulation. Cancer Biol Ther. 20:328–337. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Ying X, Sun Y and He P: MicroRNA-137
inhibits BMP7 to enhance the epithelial-mesenchymal transition of
breast cancer cells. Oncotarget. 8:18348–18358. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Han Y, Bi Y, Bi H, Diao C, Zhang G, Cheng
K and Yang Z: miR-137 suppresses the invasion and procedure of EMT
of human breast cancer cell line MCF-7 through targeting CtBP1. Hum
Cell. 29:30–36. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
He Z, Guo X, Tian S, Zhu C, Chen S, Yu C,
Jiang J and Sun C: MicroRNA-137 reduces stemness features of
pancreatic cancer cells by targeting KLF12. J Exp Clin Cancer Res.
38:1262019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chen F, Luo N, Hu Y, Li X and Zhang K:
miR-137 suppresses triple-negative breast cancer stemness and
tumorigenesis by perturbing BCL11A-DNMT1 interaction. Cell Physiol
Biochem. 47:2147–2158. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tang T, Yang Z, Zhu Q, Wu Y, Sun K,
Alahdal M, Zhang Y, Xing Y, Shen Y, Xia T, et al: Up-regulation of
miR-210 induced by a hypoxic microenvironment promotes breast
cancer stem cells metastasis, proliferation, and self-renewal by
targeting E-cadherin. FASEB J. Sep 6–2018.(Epub ahead of print).
View Article : Google Scholar
|