Introduction
The development and progression of a tumor depends
not only on its proliferation, but also on its interactions with
the local microenvironment (1).
Tumors have hypoxic stress microenvironments (2). An acidic microenvironment [pH
(6.5–6.8) vs. (7.4)] caused by a large amount of anaerobic
glycolysis is an important characteristic of malignant tumors, and
an important factor in inducing their occurrence, metastasis, and
drug resistance (3).
Tumor-associated macrophages (TAMs) are the most
important cell subsets in the aforementioned environment, and their
numbers and functions are affected by many factors related to the
host and tumor (4). TAMs are
primarily divided into the following two subtypes; M1-TAM is a part
of the classical activation pathway, and M2-TAM is a part of the
alternative activation pathway. M1-TAM shows a high expression of
interleukin (IL)-12, low expression of IL-10, and plays an
important role in antitumor activity (4). M2-TAM has low expression of IL-12,
high expression of IL-10, and exerts a tumor-promoting effect.
Experiments have shown that TAMs can play an important positive
regulatory role in the development and progression of tumors
(5). In recent years, with a more
thorough understanding of acidic microenvironments, these are now
considered a new target for tumor diagnosis and treatment,
especially in regulating TAM immune escape (6). Previous studies have shown that lactic
acid can maintain the immunosuppressive tumor microenvironment by
regulating the transformation of macrophages to the M2
type and reducing the cytotoxic function of natural killer cells
(7,8). Previous findings showed that TAMs can
also combine with programmed death-1 (PD-1) expressed on the
surface of T cells through programmed death-ligand 1 (PD-L1) and
mediate the immune escape of tumor cells through the PD-L1/PD-1
pathway to promote tumor progression (9,10).
However, the authenticity of this mechanism is unclear at
present.
In this study, by investigating the effects of
lactic acid on macrophage redistribution and immune function, the
possible mechanism by which tumor cells, including pancreatic
cancer cells, release lactic acid through glycolysis to promote
immune escape was explored, providing a foundation for new
treatment directions.
Materials and methods
Cell cultures and treatments
The human monocyte/macrophage cell line THP-1, human
T lymphocyte cell line H9, and BxPC-3 human pancreatic cancer cell
lines (American Tissue Type Collection) were maintained in
Dulbecco's modified Eagle's medium (DMEM) supplemented with
penicillin (100 U/ml), streptomycin (100 µg/ml), 0.1 mM
nonessential amino acids, 0.2 mM glutamine, 1 mM pyruvate, and 10%
heat-inactivated fetal bovine serum (FBS) and were incubated in 5%
CO2 humidified atmosphere at 37°C. Cells were grown to
80% confluency prior to treatment.
Primary human pancreatic cells were isolated from
the same pancreatic site in pancreatic cancer and non-cancerous
partial pancreatomy specimens, respectively, [tissue samples
obtained from areas within 2.0 cm around the tumor were obtained
from 45 patients undergoing partial pancreaticoduodenectomy
(Whipple resection) for pancreatic cancer at the Department of
Hepatobiliary and Pancreatic Surgery, Affiliated Hospital of the
Xi'an Jiaotong University] from January 2016 to December 2018. Of
the 45 patients, there were 30 men and 15 women. The median age at
the time of surgery was 60.5 years (range 42–77 years). All the
patients in the study signed informed consent. Briefly, each
specimen was collected and transferred to the laboratory. After
several washings with sterile phosphate-buffered saline (PBS), 1
cm2 sections of tissues were placed into the wells of
culture flasks. Once the tissue appeared to adhere to the flasks
(5–6 h), DMEM containing 10% FBS was added gently to the tissue
sections. Specimens were inspected daily and the medium was
exchanged after 24 h for the first time and every third day
thereafter. Tissue samples were then removed from the cultures and
were transferred to larger tissue culture vessels once they had
reached 70% confluency, after approximately 2 weeks. All the cells
used for this study were between passages 3 and 5. Ethics approval
was obtained from the Human Subjects Committee of the Xi'an
Jiaotong University, China. The antibodies against cluster of
differentiation CD68 (sc-20060), CD163 (sc-20066), HIF-1α
(sc-13515), PD-L1 (sc-293425) and β-actin (sc-58673) were purchased
from Santa Cruz Biotechnology.
Proliferation assay
Cell proliferation was determined via the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT;
Sigma) uptake method. The cells were seeded (5×103/well)
in 200 µl of DMEM in 96-well plates and cultured overnight. After
treatment with lactic acid [0-20 mmol/l, the concentration was
chosen based on a previous article (7)] at 37°C with 5% CO2, MTT
reagent (5 mg/ml) was added for the evaluation of cell growth, and
incubation was continued for an additional 4 h. The reaction was
terminated with 150 µl dimethyl sulfoxide (DMSO) per well.
Absorbance values were determined using an MRX Revelation 96-well
multiscanner (Dynex Technologies). The cells cultured in DMEM
served as the control. The cell viability index was calculated
according to the formula: experimental OD value/control OD value.
The experiments were repeated three times.
In situ detection of apoptotic
cells
TUNEL assays were performed using the in situ
cell apoptosis detection kit following the manufacturer's
instructions. Briefly, the cells were placed on cover slips and
fixed with 4% paraformaldehyde for 30 min at 25°C. The non-specific
chromogen reaction, induced by endogenous peroxidase, was inhibited
with 3% hydrogen peroxide (H2O2) for 10 min.
Terminal deoxynucleotidyl transferases (TdTs) were used for the
incorporation of DNA strand breaks in situ for 1 h at 37°C
in a humidified box. Positive control slides were treated with
DNase and negative control slides were treated with
phosphate-buffered saline (PBS) instead of TdT. DNA fragments were
stained using 3,3′-diaminobenzidine (DAB) as a substrate for
peroxidase, and hematoxylin was used as a counterstain. The
apoptotic index was calculated as a ratio of the number of
apoptotic cells to the total number of tumor cells on each slide
(10 of fields analyzed per sample).
Immunofluorescence assay
Exponentially growing cells were seeded on 25 mm
square glass coverslips placed in 35 mm diameter culture dishes.
After treatment, the cells were fixed with 4% formaldehyde for 5
min at 25°C, permeabilized with 0.2% solution of Triton X-100 in
PBS, and blocked with 2% bovine serum albumin-PBS for 30 min at
25°C. Slides were incubated with anti-CD68 and anti-CD163
overnight. Fluorescent imaging was conducted with a confocal laser
scanning microscope (Carl Zeiss MicroImaging, Inc.).
Reverse-transcription polymerase chain
reaction
Total RNA was isolated using TRIzol®
reagent (GIBCO BRL), and the quantities were determined
spectrophotometrically. First-strand cDNA was synthesized from 2 µg
of total RNA using a RevertAid Kit (Fermentas MBI). The polymerase
chain reaction (PCR) primer sets were designed 1) for PD-L1,
forward: 5′-CGACATGTGCTAGCATGCTGCTCCTGC-3′ and reverse:
5′-CCCTCGAGGCGGCCGCCTAGATCCTCTTTC-3′; 2) for CD68, forward:
5′-CACGCAGCACAGTGGACATTCT-3′ and reverse:
5′-TGGGGCAGGAGAAACTTTGCC-3′; 3) for CD163, forward:
5′-AGCATGGAAGCGGTCTCTGTGATT-3′ and reverse:
5′-AGCTGACTCATTCCCACGACAAGA-3′; and 4) for β-actin, forward:
5′-ATCGTGCGTGACATTAAGGAGAAG-3′ and reverse:
5′-AGGAAGGAAGGCTGGAAGAGTG-3′. The PCR conditions included an
initial cDNA synthesis reaction at 42°C for 1 h using a RevertAid
Kit (Fermentas MBI) followed by a denaturation step for 5 min at
94°C and 22 cycles: 30 sec at 94°C, 30 sec at 55°C, and 30 sec at
72°C. After the last cycle, a final extension was performed at 72°C
for 10 min. The housekeeping gene β-actin was used as an
internal control.
Western blotting
Briefly, 5×105 cells were incubated on
ice for 30 min in 0.5 ml of ice-cold whole-cell lysate buffer.
Debris were removed by centrifugation at 23 × g, for 10 min at 4°C.
The protein content of the cells was determined, and the cellular
lysates were separated by 10% SDS-PAGE and electro-transferred onto
nitrocellulose membranes. After being blocked with 5% non-fat milk
in TBST, the membranes were incubated with primary antibodies CD68
(1:500, sc-20060), CD163 (1:500, sc-20066), HIF-1α (1:500,
sc-13515), PD-L1 (1:500, sc-293425) at 4°C overnight, followed by
1:2,000 horseradish peroxidase-conjugated secondary antibody (Santa
Cruz, sc-516102) for 2 h. Immunoreactive bands were visualized
using an Enhanced Chemiluminescence Kit (Amersham Pharmacia
Biotech). The western blot signals were quantified by densitometric
analysis using Total Lab Nonlinear Dynamic Image Analysis software
(Nonlinear).
siRNA assay
To inhibit the expression of HIF-1α, siRNA oligos
were used (Qiagen). HIF-1α siRNA target sequence was:
5′-AGGAAGAACTATGAACATAAA-3′, and the sequence of the control siRNA
was: 5′-UUCUCCGAACGUGUCACGUTT-3′; this selection was based on the
results of our previous study (11). The cells (n=2×106) were
transfected with siRNA targeted against HIF-1α (100 nm/l) or a
control siRNA (Qiagen) using Lipofectamine 2000 (Invitrogen). Cells
were covered overnight prior to starvation. This was then followed
by treatment with lactic acid (15 mmol/l) for 24 h. Finally, the
cells were harvested for reverse-transcription polymerase chain
reaction (RT-PCR) and western blotting.
Indirect co-culture model
The T cells were added to Petri dishes at a density
of 1.5×105/ml; after 24 h, TAM-CM was added, and the
cells were cultured for 48 h. Cells in PBS or serum-free medium
were used as controls. ELISA and TUNEL assays were used to observe
the factor and apoptosis of T cells in each Petri dish. Proteins
were extracted from the cells. The co-culture of pancreatic cells,
THP-1 or pancreatic cancer cells was performed in the same
manner.
Statistical analysis
Each experiment was performed at least three times.
Data are provided as means ± standard deviation and differences
were evaluated using Student's t-test and a one-way ANOVA (when the
variance was homogeneous, Tukey's test was used as the post hoc
test; when the variance was uneven, tamhane test was used as the
post hoc test). Homogeneity of variance was determined with Fisher
test. P<0.05 was considered statistically significant. All
statistical analyses were performed using SPSS Version 13.0
statistical software (SPSS).
Results
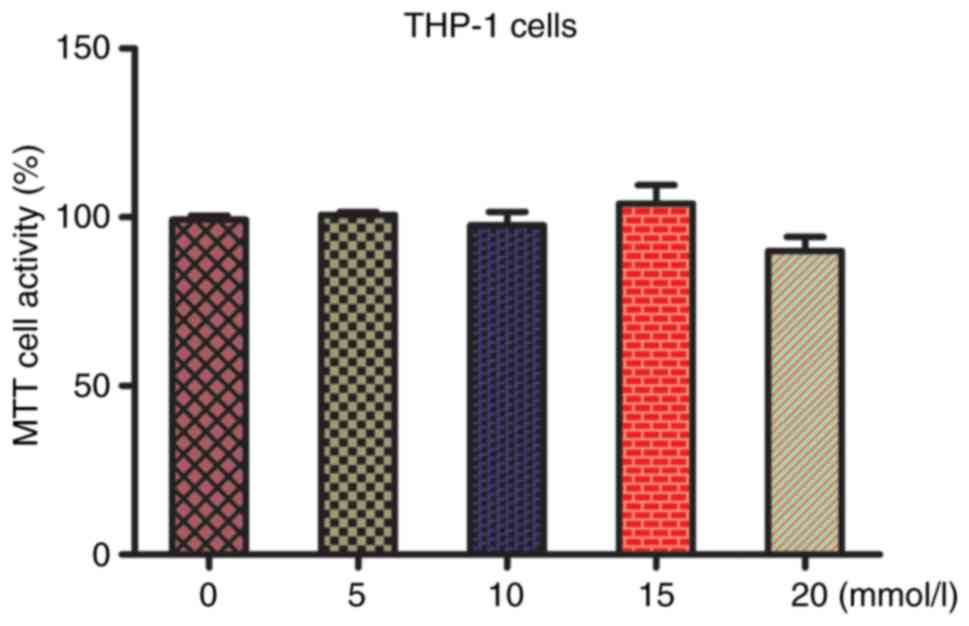
Changes in the proliferation of THP-1
macrophages with different concentrations of lactic acid
We first examined the effects of different
concentrations of lactic acid (0, 5, 10, 15, and 20 mmol/l) on the
proliferation of THP-1 cells by administering different
concentrations for 48 h (7). Cell
proliferation did not change significantly after treatment with
lactic acid concentrations <15 mmol/l (P>0.05), whereas it
was inhibited at 20 mmol/l, indicating drug toxicity. Therefore,
lactic acid concentration of 15 mmol/l was selected for subsequent
experiments (Fig. 1).
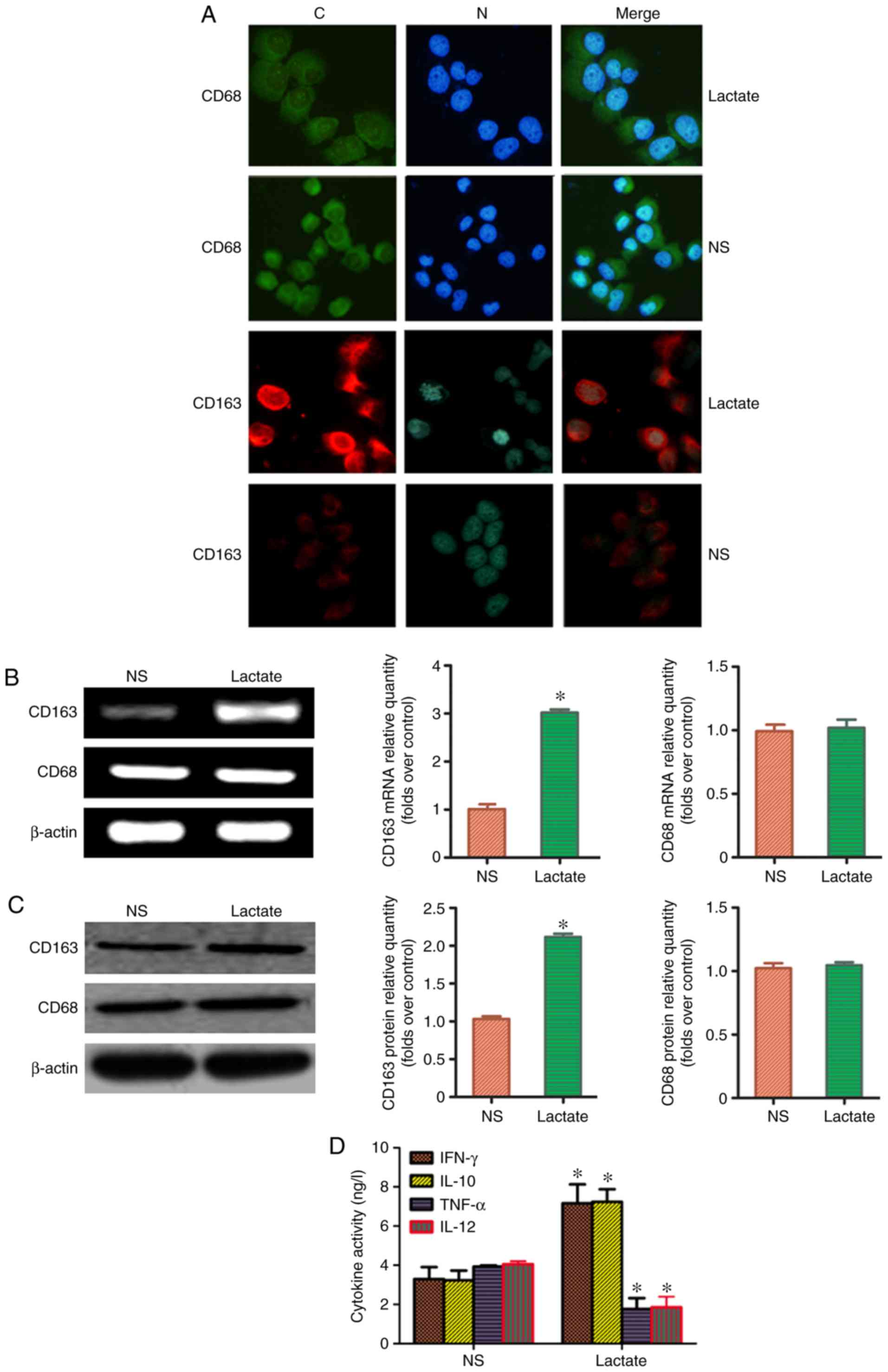
Lactic acid upregulated the expression
of macrophage marker molecules in M2 macrophages
It has previously been reported that CD68 is a
specific molecular marker for macrophages, and CD163 expression is
a marker for M2-TAMs (4). The ratio of CD163:CD68, thus,
represents the proportion of M2-TAMs. To clarify whether
lactic acid can redistribute M2-TAMs, THP-1 macrophages
were treated with 15 mmol/l for 48 h. Immunofluorescence analysis
revealed that the fluorescence intensity of CD163 in the lactic
acid-treated group was significantly higher than that in the blank
control group; by contrast, the fluorescence intensity of CD68 did
not change considerably (Fig. 2A).
RT-PCR and western blot analysis revealed that the ratio of
CD163:CD68 in the lactic acid group was significantly higher than
that in the blank control group (Fig.
2B and C). Similarly, the levels of cytokines, IFN-γ and IL-10,
secreted in the supernatant by M2-TAMs of the lactic
acid-treated group were higher than those secreted by
M2-TAMs of the blank control group (P<0.05).
Additionally, the levels of cytokines, TNF-α and IL-12, secreted by
M1-TAMs decreased (P<0.05; Fig. 2D). These results suggested that
lactic acid can redistribute M2-TAM subsets.
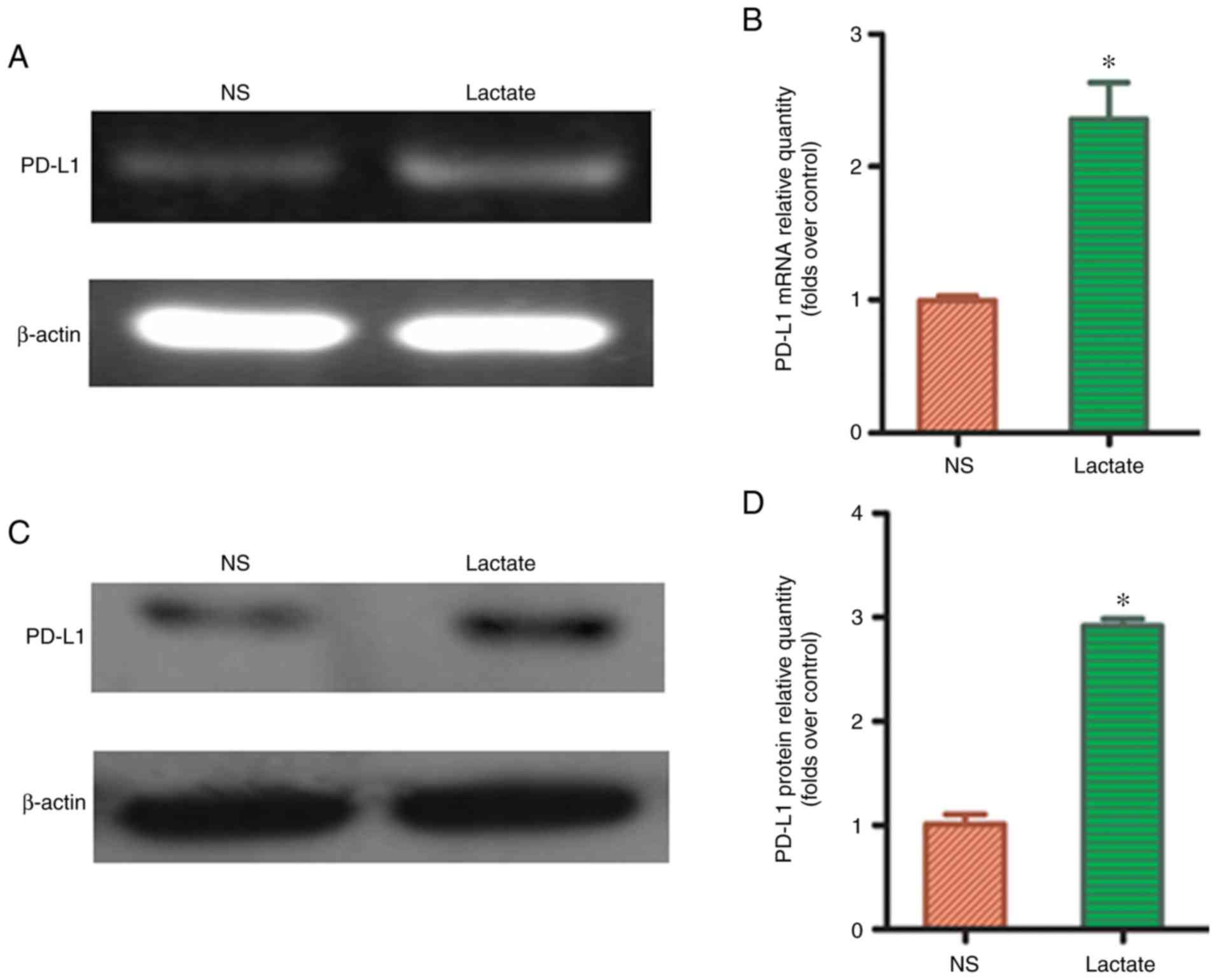
Lactic acid upregulated the expression
of PD-L1 protein in M2-TAMs
To determine whether lactic acid can affect PD-L1
expression in macrophages, 15 mmol/l lactic acid was used to treat
THP-1 macrophages for 48 h. RT-PCR showed that PD-L1 mRNA
expression significantly increased in the lactic acid-treated group
compared to that in the blank control group (P<0.05; Fig. 3A and B). Similarly, western blot
analysis revealed that the expression of PD-L1 protein was
significantly upregulated in the lactic acid-treated group compared
to that in the blank control group (P<0.05; Fig. 3C and D), suggesting that lactic acid
may affect tumor immune escape through the PD-L1/PD-1 pathway,
after redistributing M2-TAM subsets.
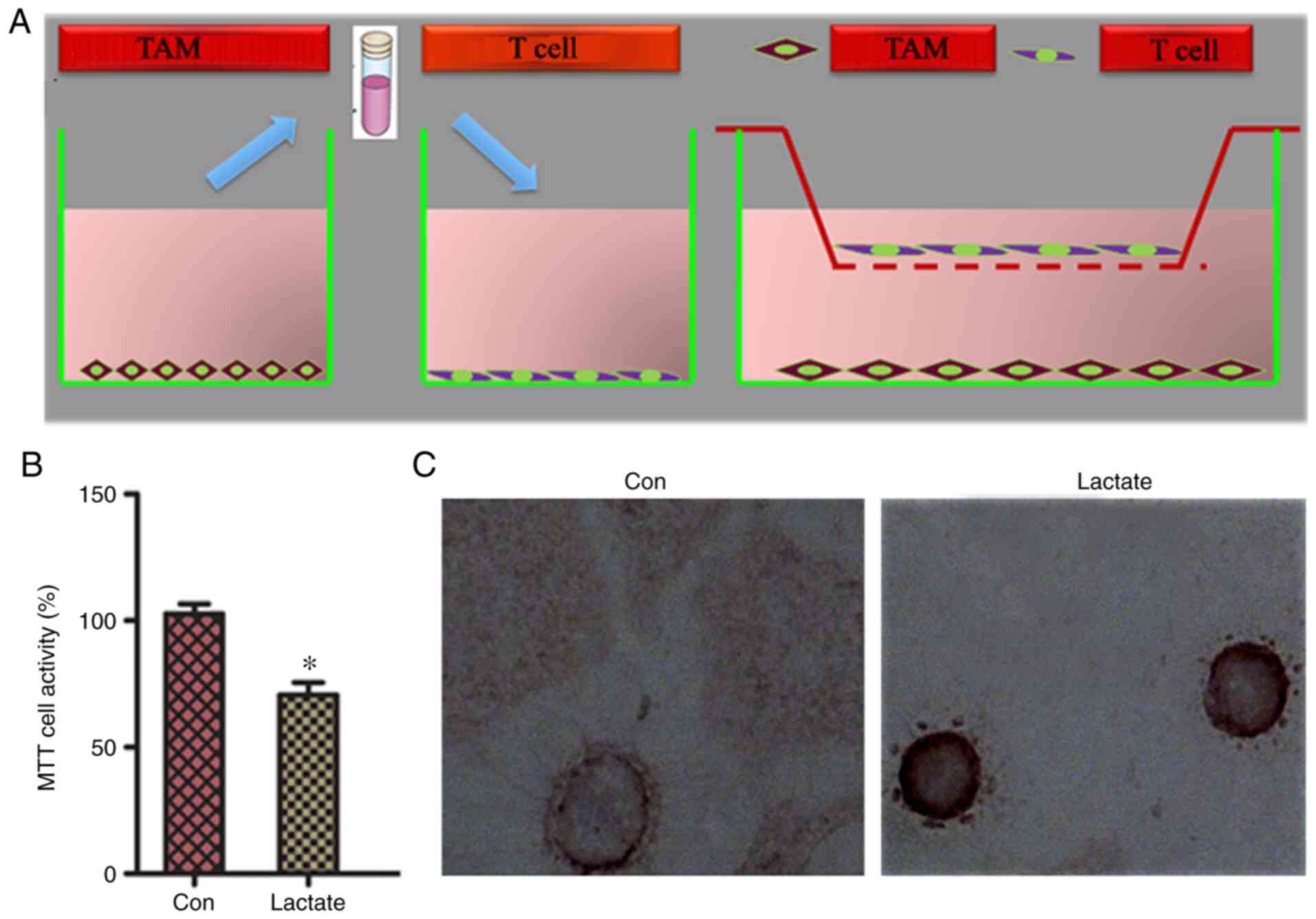
Effect of the lactic acid-induced
redistribution of M2-TAM subsets on the biological
behaviors of T cells
PD-L1 and PD-1 are negative costimulatory molecules
that can induce apoptosis of specific cytotoxic T lymphocytes
(CTLs) and reduce the sensitivity of tumors to the cytotoxic effect
of CTLs, thereby assisting tumor immune escape. To determine
whether M2-TAMs overexpressing PD-L1 following treatment
with lactic acid affected the proliferation and apoptosis of T
cells, T cells and macrophages were first co-cultured indirectly,
then lactic acid/PBS was added to the co-culture (Fig. 4A). M2-TAMs in the lactic acid
treatment group had a significantly lower ability to induce
activation and proliferation of T cells than those co-cultured in
the PBS control group (Fig. 4B).
T-cell apoptosis in the co-cultured group was higher in the lactic
acid treatment group than that in the PBS control group (Fig. 4C; Table
I), suggesting that M2-TAMs reduce the cytotoxic
effect of T cells.
 | Table I.T-cell apoptosis of co-cultured group
was higher in the lactic acid treatment group than that in the PBS
control group (mean ± SD). |
Table I.
T-cell apoptosis of co-cultured group
was higher in the lactic acid treatment group than that in the PBS
control group (mean ± SD).
| Group | TUNEL-positive
cells (apoptotlc index) (%) |
|---|
| Control | 1.05±0.27 |
| Lactic acid |
9.46±0.14a |
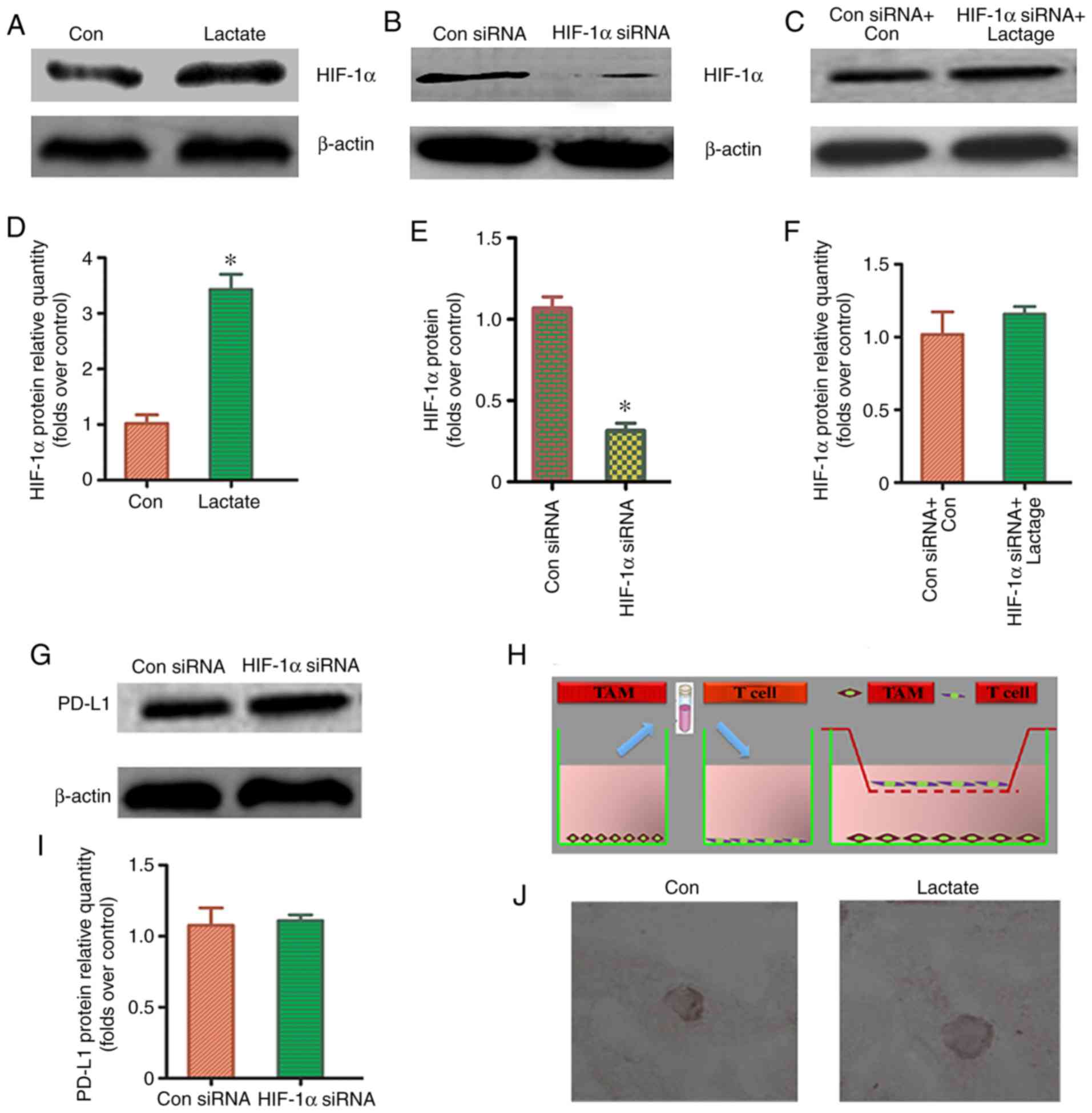
HIF-1α signaling pathway may be the
key regulatory mechanism
To determine whether the HIF-1α signaling pathway
was involved in the lactic acid-induced redistribution of
M2-TAM subsets and PD-L1 overexpression, a western blot
was used to detect HIF-1α protein expression in THP-1 macrophages
treated with lactic acid for 48 h. HIF-1α expression was
significantly increased in the lactic acid-treated group as
compared to that in the blank control group (Fig. 5A and D). This suggests that HIF-1α
plays a regulatory role in the redistribution of M2-TAM
subsets and PD-L1 overexpression. Successful transfection with
HIF-1a siRNA was verified using western blotting (Fig. 5B and E). To further verify the
effect of HIF-1α on M2-TAM subsets and PD-L1
overexpression, we pre-treated macrophages with HIF-1α siRNA and
treated them with lactic acid (Fig. 5C
and F). There was no significant difference in the distribution
of M2-TAM subsets and PD-L1 expression between the
HIF-1α siRNA-treated group and the control group (Fig. 5G-J; Table II). These data indicated that the
HIF-1α signaling pathway participates to some extent in the lactic
acid-induced redistribution of M2-TAM subsets and PD-L1
overexpression.
 | Table II.T-cell apoptosis of HIF-1α
siRNA-macrophages co-culturea. |
Table II.
T-cell apoptosis of HIF-1α
siRNA-macrophages co-culturea.
| Group | TUNEL-positive
cells (apoptotlc index) (%) |
|---|
| Control | 1.13±0.16 |
| Lactic acid |
1.22±0.20b |
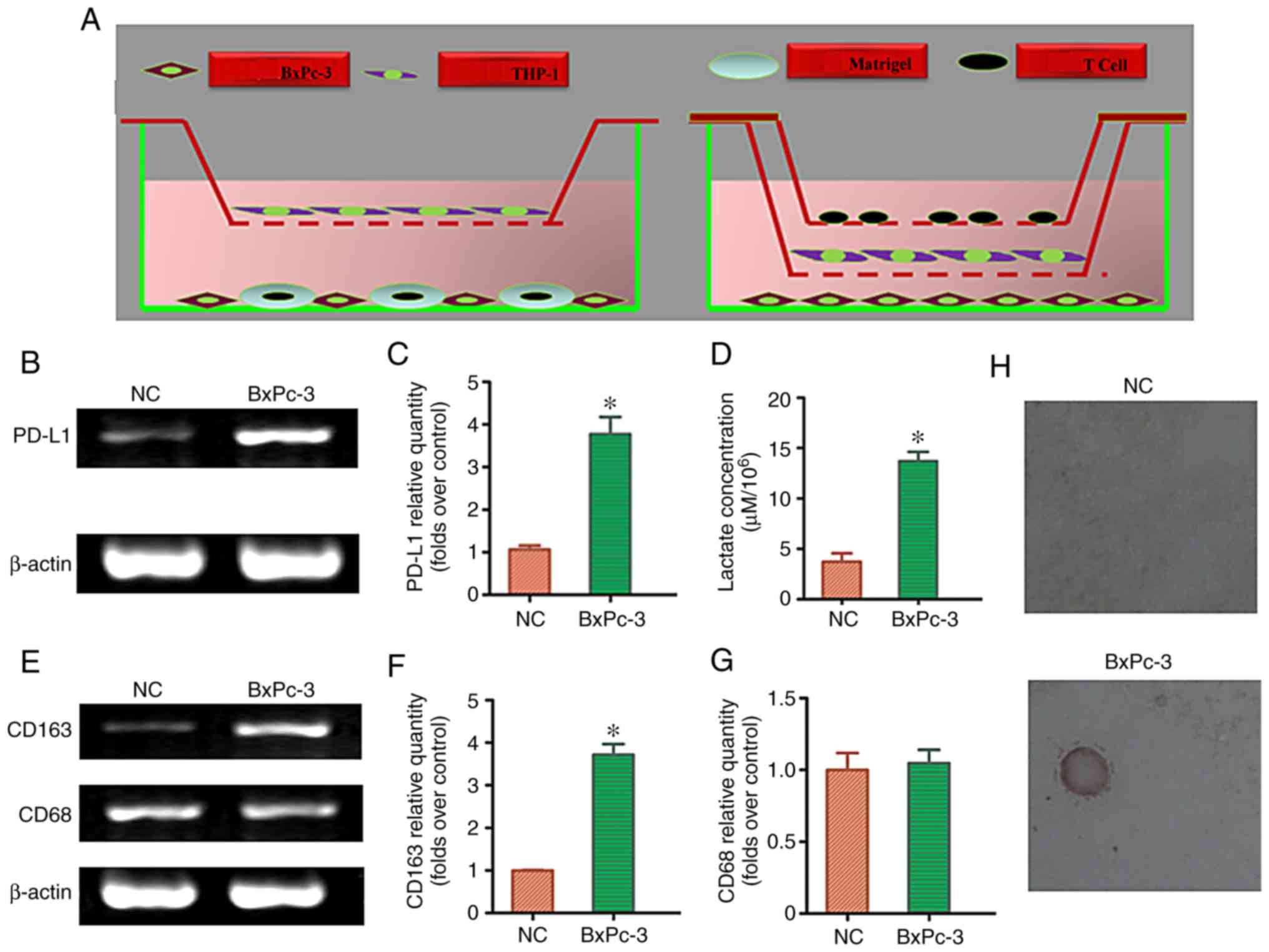
Redistribution of M2-TAM
subsets and effects on T cell apoptosis after the co-culture of
pancreatic cancer cells, macrophages, and T cells
To clarify whether lactic acid released by
pancreatic cancer cells redistributes M2-TAM subsets and
regulates immune function, we first detected the lactic acid
concentration in the supernatant after the co-culturing of
pancreatic cancer cells, macrophages, and T cells using a direct or
indirect triple-cell culture model (Fig. 6A). Compared with the normal
pancreatic cell culture group, the lactic acid concentration in the
pancreatic cancer cell co-culture group significantly increased
(P<0.05; Fig. 6D). To determine
whether lactic acid released by pancreatic cancer cells
redistributed M2-TAMs, the co-cultured cells were
collected to detect CD163/CD68 mRNA expression. RT-PCR results
showed that the ratio of CD163:CD68 mRNA was significantly
upregulated in the pancreatic cancer cell co-culture group compared
with that in the normal pancreatic cell culture group (P<0.05)
(Fig. 6E and G). Similarly, RT-PCR
results showed that PD-L1 mRNA expression was significantly
upregulated in the pancreatic cancer cell co-culture group as
compared to that in the normal pancreatic cell culture group
(P<0.05) (Fig. 6B and C).
Compared with the normal pancreatic cell culture group, the
pancreatic cancer cell co-culture group significantly induced T
cell apoptosis (P<0.05; Fig. 6H;
Table III). The results of the
triple-cell culture suggested that lactic acid released by
pancreatic cancer cells can redistribute M2-TAM subsets
and regulate immune escape.
 | Table III.T-cell apoptosis was higher in the
pancreatic cancer cell co-culture group compared with the normal
pancreatic cell culture group (mean ± SD). |
Table III.
T-cell apoptosis was higher in the
pancreatic cancer cell co-culture group compared with the normal
pancreatic cell culture group (mean ± SD).
| Group | TUNEL-positive
cells (apoptotlc index) (%) |
|---|
| Normal cell | 1.07±0.62 |
| Cancer cell |
7.00±0.52a |
Discussion
Tumor cells consist of an acidic microenvironment
(2). Lactic acid, a glycolytic
metabolite, is believed to be a key factor in redistributing
macrophage polarization in the tumor microenvironment (3). Previously, it was shown that a
high-lactic acid environment in pancreatic cancer tissue samples is
consistent with the distribution of M2-TAM subsets
(7). The results of this in
vitro experiment confirmed that lactic acid could redistribute
M2-TAM subsets (7),
which is consistent with the results of the current study. This
suggests that lactic acid may be an initiating factor for
M2-TAMs to promote tumor progression by their
transformation from a ‘good’ macrophage into a ‘bad’
macrophage.
The development and progression of solid tumors are
immune regulation processes (12).
Various factors in the tumor microenvironment interact with each
other, interfering with the balance of the immune system, resulting
in the immune escape of tumors (13). According to previous findings,
macrophages act as checkpoints by determining whether to initiate
an immune response or induce immune tolerance, thus, serving as a
bridge between the immune state and tumor evolution (4). The mechanism underlying the
M2-TAM modulation of immune escape has not yet been
fully elucidated, and previous findings have shown that PD-L1 may
be involved in this process (9).
PD-L1 belongs to the B7 family and contains IgV-like, IgC-like, and
transmembrane regions, and a cytoplasmic tail (14). The latter is involved in
intracellular signal transduction, while the IgV and IgC regions
are involved in intercellular signal transduction. The binding of
PD-L1 to PD-1 on T cells promotes the phosphorylation of tyrosine
in the ITSM domain of PD-1, causes the dephosphorylation of
downstream Syk kinase and phosphoinositide 3-kinase, inhibits the
activation of downstream AKT, extracellular signal-activated
kinase, and other pathways, and ultimately inhibits the
transcription and translation of the genes and cytokines required
for T-cell activation (14). These
activities play a role in negatively regulating T-cell activity.
For example, Dong et al (15), based on the results of an in
vitro experiment utilizing mouse models, reported that
activation of the PD-L1/PD-1 signaling pathway may induce the
specific apoptosis of CTLs, which reduces the sensitivity of the
CTL cytotoxic killing effect and promotes the immune escape of
tumor cells. Zhou et al (16), found that in patients with liver
cancer, TAM also inhibits functions of tumor-infiltrating
lymphocytes through the highly expressed PD-L1 on its surface,
leading to the escape of tumor cells from immune cell attack.
Consistent with this finding, we observed that the high expression
of PD-L1 in the TAM of pancreatic cancer was consistent with CD163.
Moreover, correlation analysis of clinical factors revealed that
the overexpression of PD-L1 in TAM was positively associated with
lymph node metastasis and tumor staging. In the analysis of
prognostic factors of pancreatic cancer, PD-L1 in TAM may serve as
an independent risk factor. Patients with PD-L1 overexpression had
a significantly worse prognosis than patients with low expression,
suggesting that the PD-L1/PD-1 signaling pathway is involved in the
mechanism underlying the M2-TAM regulation of tumor
immune escape (17). However,
results of this study still need to be verified through relevant
in vitro experiments.
The abovementioned results show that modulation of
lactic acid level may initiate the polarization of macrophages.
Previous findings regarding this mechanism have reported that
lactic acid is absorbed and oxidized by macrophage mitochondria
through MCT1 into pyruvate (18).
The latter inhibits prolyl hydroxylase domain (PHD) activity
through direct competition with the PHD substrate, leading to the
stable accumulation of HIF-1α, and subsequently, inducing the
transformation of the M2-TAM subsets (19). In the present study, we found that
the expression of HIF-1α protein significantly increased after the
treatment of THP-1 macrophages with lactic acid. There was no
significant difference in the distribution of M2-TAM
cell subsets and PD-L1 expression after macrophages were pretreated
with HIF-1α siRNA then treated with lactic acid. This indicates
that the HIF-1α signaling pathway participates to some extent in
the redistributing of M2-TAM subsets by lactic acid and
PD-L1 overexpression, which is consistent with the finding that
HIF-1α is involved in the redistribution of M2-TAM
subsets and regulation of the immune function. Moreover, we used a
triple-cell culture model consisting of pancreatic cancer cells,
macrophages, and T cells to simulate the real microenvironment of
pancreatic cancer, and again verified that lactic acid released by
glycolysis in pancreatic cancer cells could redistribute
M2-TAMs and further regulate PD-L1/PD-1 signals to
mediate T-cell apoptosis, thereby assisting immune escape. Results
of the present study demonstrated the significance of the
involvement of lactic acid, a key metabolite, in tumor progression.
SiRNA is an important tool for genetic analysis of mammalian cells.
SiRNA-induced gene silencing in mammalian cells can effectively
inhibit the expression of specific genes. In this experiment,
single siRNA of HIF-1α corresponding to target genes were prepared
and detected. Further research with more siRNAs of HIF-1α may be
useful to obtain more reliable results.
There are some limitations to our study. First,
lactic acid alone (without macrophage co-culture) may affect T-cell
proliferation and apoptosis, and lactic acid alone was not applied
as a control group to exclude this bias. Second, we performed TUNEL
assays to access T-cell apoptosis. However, the results could be
further verified if other methods such as annexin-V staining could
also be used. Third, we did not evaluate the mechanism for
reshaping and immune function of M2-TAM subsets. This remains to be
studied in further experiments.
In conclusion, this in vitro experiment
further explored the hypothesis that tumor cells use lactic acid, a
glycolysis product, as a weapon to activate M2-TAMs via
the bystander effect and, thus, further regulate PD-L1/PD-1
signal-mediated immune escape. These results suggest that lactic
acid, macrophages, and PD-L1 are intrinsically linked, and provide
an important theoretical basis for the treatment of pancreatic
cancer.
Acknowledgements
Not applicable.
Funding
This study was supported by the Innovation Talent
Promotion Project Foundation of Shaanxi Province (grant no.
2018KJXX-058); the Hospital Fund of the Second Affiliated Hospital
of the Health Science Center, Xi'an Jiaotong University [grant no.
RC(GG)201708]; and the National Natural Science Foundation of
China, NSFC (grant no. 81402583); Natural Science Foundation of
Shaanxi Province (grant no. 2019JQ-969); Xi'an Jiaotong University
Education Foundation, XJTUEF (grant no. xjj2018141).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
TS, SC and XC conceived and designed the
experiments. TS, SC, TW and WL performed the experiments. XC, SL,
JZ and YY analyzed the data. JM, WL, XC, WL and YK contributed to
the conducting the experiments. TS wrote the paper and XC, WL and
YK revised the article. All authors approved the final version of
the manuscript.
Ethics approval and consent to
participate
Ethics approval was obtained from the Human Subjects
Committee of the Xi'an Jiaotong University, China. All the patients
in the study provided signed informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Moore A: Cancer ecology: The intracellular
interactome makes little sense without the intercellular one.
Bioessays. 40:e18002022018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yan L, Raj P, Yao W and Ying H: Glucose
metabolism in pancreatic cancer. Cancers (Basel). 11:14602019.
View Article : Google Scholar
|
|
3
|
Rawat D, Chhonker SK, Naik RA, Mehrotra A,
Trigun SK and Koiri RK: Lactate as a signaling molecule: Journey
from dead end product of glycolysis to tumor survival. Front Biosci
(Landmark Ed). 24:366–381. 2019. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Najafi M, Goradel NH, Farhood B, Farhood
B, Salehi E, Nashtaei MS, Khanlarkhani N, Khezri Z, Majidpoor J,
Abouzaripour M, et al: Macrophage polarity in cancer: A review. J
Cell Biochem. 120:2756–2765. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xu JY, Wang WS, Zhou J, Liu CY, Shi JL, Lu
PH and Ding JL: The importance of a conjoint analysis of
tumor-associated macrophages and immune checkpoints in pancreatic
cancer. Pancreas. 48:904–912. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bohn T, Rapp S, Luther N, Klein M, Bruehl
TJ, Kojima N, Aranda Lopez P, Hahlbrock J, Muth S, Endo S, et al:
Tumor immunoevasion via acidosis-dependent induction of regulatory
tumor-associated macrophages. Nat Immunol. 19:1319–1329. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Colegio OR, Chu NQ, Szabo AL, Chu T,
Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC,
Phillips GM, et al: Functional polarization of tumour-associated
macrophages by tumour-derived lactic acid. Nature. 513:559–563.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Terren I, Orrantia A, Vitalle J,
Zenarruzabeitia O and Borrego F: NK cell metabolism and tumor
microenvironment. Front Immunol. 10:22782019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cai J, Qi Q, Qian X, Han J, Zhu X, Zhang Q
and Xia R: The role of PD-1/PD-L1 axis and macrophage in the
progression and treatment of cancer. J Cancer Res Clin Oncol.
145:1377–1385. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sau S, Alsaab HO, Bhise K, Alzhrani R,
Nabil G and Iyer AK: Multifunctional nanoparticles for cancer
immunotherapy: A groundbreaking approach for reprogramming
malfunctioned tumor environment. J Control Release. 274:24–34.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shan T, Cui X, Li W, Lin W, Li Y, Chen X
and Wu T: Novel regulatory program for norepinephrine-induced
epithelial-mesenchymal transition in gastric adenocarcinoma cell
lines. Cancer Sci. 105:847–856. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bolli E, D'Huyvetter M, Murgaski A, Berus
D, Stangé G, Clappaert EJ, Arnouk S, Pombo Antunes AR, Krasniqi A,
Lahoutte T, et al: Stromal-targeting radioimmunotherapy mitigates
the progression of therapy-resistant tumors. J Control Release.
314:1–11. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kalinski P and Talmadge JE: Tumor
immuno-environment in cancer progression and therapy. Adv Exp Med
Biol. 1036:1–18. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun C, Mezzadra R and Schumacher TN:
Regulation and function of the PD-L1 checkpoint. Immunity.
48:434–452. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dong H, Strome SE, Salomao DR, Tamura H,
Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, et al:
Tumor-associated B7-H1 promotes T-cell apoptosis: A potential
mechanism of immune evasion. Nat Med. 8:793–800. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhou G, Sprengers D, Boor PPC, Doukas M,
Schutz H, Mancham S, Pedroza-Gonzalez A, Polak WG, de Jonge J,
Gaspersz M, et al: Antibodies against immune checkpoint molecules
restore functions of tumor-infiltrating T cells in hepatocellular
carcinomas. Gastroenterology. 153:1107–1119.e10. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang L, Ma Q, Chen X, Guo K, Li J and
Zhang M: Clinical significance of B7-H1 and B7-1 expressions in
pancreatic carcinoma. World J Surg. 34:1059–1065. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Silva LS, Poschet G, Nonnenmacher Y,
Becker HM, Sapcariu S, Gaupel AC, Schlotter M, Wu Y, Kneisel N,
Seiffert M, et al: Branched-chain ketoacids secreted by
glioblastoma cells via MCT1 modulate macrophage phenotype. EMBO
Rep. 18:2172–2185. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wei L, Zhou Y, Yao J, Qiao C, Ni T, Guo R,
Guo Q and Lu N: Lactate promotes PGE2 synthesis and gluconeogenesis
in monocytes to benefit the growth of inflammation-associated
colorectal tumor. Oncotarget. 6:16198–16214. 2015. View Article : Google Scholar : PubMed/NCBI
|