Introduction
An acidic microenvironment is a characteristic
feature of tumors resulting from cancer metabolism, inflammation,
regional hypoxia, and poor vascular perfusion. Previous studies
have reported that local acidosis promotes cancer progression via
accelerated cell proliferation and induces the expression of
proteinases such as cathepsin K, matrix metalloproteinase (MMP)-2
and MMP-9, resulting in local invasion (1–5).
Rofstad et al showed that acidic conditions promoted the
metastasis of human melanoma cells in athymic nude mice in
vivo, which was accompanied by increased secretion of protease
and pro-angiogenic factors (6). In
addition, Nishisho et al demonstrated that expression of a3
isoform vacuolar type H+-ATPase, which regulates proton
transport and creates extracellular acidosis, promotes distant
metastasis of B16 mouse melanoma cells (7). However, the regulatory factors that
function between the acidic microenvironment and cancer cells are
not fully understood.
Interleukin (IL)-8, also known as CXCL8, is a
pro-inflammatory CXC chemokine that activates multiple
intracellular signaling pathways through binding to receptors
(CXCR1 and CXCR2). During inflammation, IL-8 is produced in
macrophages, epithelial cells, and fibroblasts following
stimulation of IL-1 and/or tumor necrosis factor (TNF)-α. Secreted
IL-8 induces the recruitment and activation of neutrophils and
lymphocytes. Increased expression of IL-8 and/or its receptors in
cancer cells has been observed (8,9), which
suggests that IL-8 is a critical molecule in cancer development.
IL-8 signaling promotes the activation of the primary effectors,
phosphatidylinositol-3 kinase or phospholipase C, which in turn
activate AKT, PKC, and the MAPK signaling cascade (8). These pathways stimulate the
proliferation, survival, chemoresistance, adhesion, and migration
of cancer cells. Srivastava et al demonstrated that IL-8
inhibition drastically decreased the proliferation, migration, and
invasiveness of melanoma cells. Furthermore, IL-8 depletion
inhibited endothelial cell proliferation and the formation of
capillary-like structures (10). We
previously demonstrated that IL-8 accelerated the proliferation of
lymphatic endothelial cells, and inhibition of IL-8 diminished tube
formation and cell migration (11).
While various roles for IL-8 in cancer progression have been
reported, the mechanism of IL-8 upregulation and its association
with the acidic microenvironment remain to be elucidated.
Extracellular acidosis stimulates cells via ion
channels such as transient receptor potential vanilloid subtype 1
(TRPV1), acid-sensing ion channel 1–4 (ASIC1-4), and proton-sensing
G-protein coupled receptors (GPCRs) including GPR4, GPR65 (also
known as TDAG8), GPR68 (OGR1), and GPR132 (G2A). TRPV1 and ASICs
are non-selective cation channels that are activated by
extracellular protons and mainly expressed in peripheral sensory
and central nervous system neurons. TRPV1 is activated by
capsaicin, noxious heat, and protons (12), and an acidic microenvironment is
thought to be responsible for TRPV1-mediated pain signals in
patients with inflammation and cancer (13,14).
In the peripheral nervous system, ASICs are involved in nociception
such as pain signaling and mechano-sensation. In the central
nervous system, ASICs have various roles; ASIC1a is involved in
synaptic plasticity, learning, and memory, and ASIC2a is required
for the maintenance of retinal integrity and the survival of
neurons in ischemia (15).
Previous studies have reported that acid-sensing
receptors are expressed in tumor cells and are involved in cell
proliferation, apoptosis, angiogenesis, and metastasis (1,3,16,17).
Studies have demonstrated higher expression of ASIC1 in high-grade
glioma cells compared with normal astrocytes (18,19).
Berdiev et al demonstrated that ASIC1 and ASIC2 are involved
in the proliferation and migration of glioblastoma cells (20). Furthermore, pharmacological block or
knockdown of ASIC1 inhibited acid-induced currents and cell
migration in glioblastoma cells (18,21).
These data suggest the importance of the interactions between the
acidic microenvironment and tumor cell-expressing ASICs in cancer
development. However, the expression of acid-sensing receptors in
breast cancer cells and the role of these receptors in cancer
progression remain unclear.
In the present study, we investigated the
morphological and functional changes in breast cancer cells
stimulated by acidic conditions. We found that acidosis induced the
migration and invasion of MDA-MB-231 cells, and acid-induced IL-8
expression was responsible for these phenomena. We also found that
the expression of ASIC1 was upregulated by acidic stimulation and
ASIC1 was responsible for the induction of IL-8. Our findings will
increase the understanding of the mechanism of acid-mediated
progression in breast cancer.
Materials and methods
Cell culture
The MDA-MB-231 human cell line, which is derived
from triple-negative breast cancer, was purchased from the American
Type Culture Collection (ATCC). The cells were cultured in
Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich; Merck
KGaA) with 10% fetal bovine serum (FBS; Biowest) and 1%
penicillin-streptomycin solution (Thermo Fisher Scientific K.K.) at
37°C in a humidified 5% CO2 incubator.
Acidic stimulation
For the acidic stimulation, pH 6.4 medium was
prepared using lactic acid (FujiFilm Wako Pure Chemical Corp.).
Many studies have shown that the extracellular pH ranges from 6.2
to 6.9 in malignant tumors (22–24).
Furthermore, the pH sensitivity (pH50) of ASIC1a is pH
5.8–6.8 (25). We first
investigated the IL-8 mRNA expression in MDA-MB-231 cells under pH
5.5, 6.0, 6.4, and 6.8. We observed the most prominent expression
at pH 6.0 and 6.4 (data not shown). In addition, we previously
performed studies on lymphatic endothelial cells cultured under pH
6.4 conditions (11). Therefore, we
selected a condition of pH 6.4 for the present study.
Cell staining
For visualization of cellular morphology, cells were
fixed with 4% paraformaldehyde and incubated with
rhodamine-phalloidin (Cytoskeleton, Inc.) for 30 min at room
temperature. After washing with PBS, the cells were counterstained
with DAPI (Vector Laboratories, Inc.).
Cell proliferation assay
Cell growth was measured using a Cell Counting Kit-8
(CCK-8, Dojindo Molecular Technologies, Inc., Japan) according to
the manufacturer's instructions. MDA-MB-231 cells were seeded in
96-well plates and treated with control, pH 6.4, or control medium
containing IL-8 (Peprotech, Inc.). After incubation, the CCK-8
reagent was added to each well and cells were incubated for another
2 h. Absorbance was measured using a microplate reader.
In vitro wound healing assay
MDA-MB-231 cells (5×105/well) were plated
in 6-well plates and incubated for 24 h. After a complete monolayer
had formed, cells were then cultured with serum-free DMEM for 8 h.
The monolayers were scratched using a 200-µl plastic tip, and then
the media were replaced with fresh control (pH 7.4) or acidic (pH
6.4) DMEM without serum. Migration and cell movement throughout the
wound area was observed (at 12, 24, 30, and 48 h after scratching)
with a phase-contrast microscope and images were obtained (×100
magnification). The percentage of the remaining wounded area was
measured on the images.
Transwell migration and invasion
assays
For the migration and invasion assays, 24-well
Transwell plates (Corning Incorporated) were used. First,
sub-confluent MDA-MB-231 cells were incubated with serum-free media
(control or pH 6.4) in 6-cm dishes for 8 h. Cells
(2×104/well) were then resuspended in the appropriate
media and placed in the polycarbonate membrane inserts (8-µm pore
size) coated without or with Matrigel (Corning Incorporated) for
the migration or invasion assays, respectively. The bottom wells
contained DMEM with 10% FBS. After incubation for 18 h (for
migration assays) or 24 h (for invasion assays), cells on the upper
surface of the membrane were removed. The migrated or invaded cells
on the lower side were fixed with 4% paraformaldehyde and stained
with Giemsa. The number of cells that passed to the lower side was
counted in five fields per membrane using a light microscope (×100
magnification) (Nikon Solutions Co., Ltd.).
Gelatin zymography
MDA-MB-231 cells were cultured in 6-well plates and
grown to 70% confluence. Cells were washed twice with serum-free
DMEM and then cultured for 48 h with control or pH 6.4 serum-free
media. Conditioned media were concentrated 30-fold on a Vivaspin 6
(10 kDa cut-off; Cytiva). MMP activity was assessed by gelatin
zymography (Gelatin-zymography kit, Cosmobio Co., Ltd.) according
to the manufacturer's protocol.
RNA preparation and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted using a NucleoSpin RNA kit
(Macherey-Nagel) and single-stranded cDNA was synthesized using
PrimeScript RT Master Mix (Takara Bio Inc.) according to the
manufacturer's instructions. Real-time PCR was performed using the
SYBR Green detection protocol and Thermal Cycler Dice Real Time
System (Takara Bio Inc.). The following conditions were used for
quantitative PCR: initial denaturation for 30 sec at 95°C, followed
by 40 cycles of denaturation for 5 sec at 95°C and annealing for 30
sec at 60°C. The primers used for amplification were as follows:
IL-1β forward, 5′-AGGCACAAGGCACAACAGGCT-3′ and reverse,
5′-AACAACTGACGCGGCCTGCC-3′; IL-6 forward, 5′-TGGCTGCAGGACATGACAA-3′
and reverse, 5′-TGAGGTGCCCATGCTACATTT-3′, IL-8 forward,
5′-CGTGGCTCTCTTGGCAGCCTTC-3′ and reverse,
5′-TTCCTTGGGGTCCAGACAGAGCTC-3′; ASIC1 forward,
5′-GGATGGAGGTCTACCCTCGA-3′ and reverse,
5′-GACCTCAGCTTCTGCCTGTCA-3′; glyceraldehyde-3-phosphate
dehydrogenase (GAPDH) forward, 5′-CCCTTCATTGACCTCAACTACATGGT-3′ and
reverse, 5′-TGATGACAAGCTTCCCGTTCTCAG-3′. Relative gene expression
was determined and normalized to GAPDH mRNA expression.
RT-PCR
Total RNA from MDA-MB-231 cells was extracted using
a NucleoSpin RNA kit (Macherey-Nagel) and single-stranded cDNA was
synthesized using PrimeScript RT Master Mix (Takara Bio Inc.)
according to the manufacturer's instructions. PCR amplifications
were performed using DNA polymerase kit (Takara Ex Taq; Takara Bio
Inc.) and the following primer pairs: TRPV1 forward,
5′-CTCCTACAACAGCCTGTAC-3′ and reverse, 5′-AAGGCCTTCCTCATGCACT-3′;
ASIC1 forward, 5′-GGATGGAGGTCTACCCTCGA-3′ and reverse,
5′-GACCTCAGCTTCTGCCTGTCA-3′; ASIC2 forward,
5′-GCAACCTAACCCGCTACAAC-3′ and reverse, 5′-AGCAGGCAATCTCCTCCAAG-3′;
ASIC3 forward, 5′-TATGAGACCGTGGAGCAG-3′ and reverse,
5′-TGTGTGACAAGGTAGCAGG-3′; OGR1 forward,
5′-ACTTCGGCTACCTGCAGATCAA-3′ and reverse,
5′-AGCCCACGCTGATGTAGATGTT-3′; TDAG8 forward,
5′-TGCCGTTGATCGGTATTTGGCT-3′ and reverse,
5′-TTGCATAGCCTGTACACGTCCT-3′; GPR4 forward,
5′-ATACCACAGCTCACTGGCTTTC-3′ and reverse,
5′-TCATGGCTTTGGCTGTGCTGTT-3′; G2A forward,
5′-TGCAACATCTACGTCAGCATCC-3′ and reverse,
5′-ATCTGCAGCATGTCAAAGCAGG-3′; GAPDH forward,
5′-CCCTTCATTGACCTCAACTACATGGT-3′ and reverse,
5′-TGATGACAAGCTTCCCGTTCTCAG-3′. The following thermocycling
conditions were used: initial denaturation for 5 min at 95°C,
followed by 30 cycles of denaturation at 95°C for 30 sec, annealing
at 60°C for 30 sec, and elongation at 72°C for 30 sec. The PCR
products were separated on 2% agarose gels containing ethidium
bromide and visualized under ultraviolet light.
Enzyme-linked immunosorbent assay
(ELISA)
MDA-MB-231 cells (1×105/well) were seeded
in 24-well plates and incubated with serum-free medium for 12 h.
The medium was changed to fresh control or pH 6.4 serum-free media
and then cells were incubated for 24, 48, or 72 h. Culture
supernatants were collected and analyzed for IL-8 levels using a
human IL-8 ELISA kit (Quantikine ELISA; R&D Systems) according
to the manufacturer's instructions.
Knockdown of IL-8 and ASIC1
Small interfering RNA (siRNA) targeting IL-8
(siIL-8), ASIC1 (siASIC1) and negative control siRNA (siNC) were
purchased from Thermo Fisher Scientific K.K. MDA-MB-231 cells were
cultured in DMEM containing 10% FBS without antibiotics, and siIL-8
was transfected using Lipofectamine RNAiMAX reagent (Thermo Fisher
Scientific K.K.) according to the manufacturer's protocol. After
incubation for 12 h, the transfected cells were pre-incubated with
serum-free control or pH 6.4 media for 8 h and then used for
migration or invasion assays. siASIC1 was transfected by
electroporation using the Neon Transfection System (Thermo Fisher
Scientific K.K.) according to the manufacturer's protocol.
Knockdown of IL-8 and ASIC1 were examined by real-time PCR after 24
h.
Anti-IL-8 treatment
Anti-human IL-8/CXCL8 purified polyclonal goat IgG
and normal goat IgG control were purchased from R&D Systems,
Inc. For migration and invasion assays, MDA-MB-231 cells were
suspended in serum-free control or low pH media with anti-IL-8
antibody (100 ng/ml) or control IgG, and seeded in the inserts.
After incubation for 18 h (for migration assays) or 24 h (for
invasion assays) at 37°C, migrated or invaded cells on the lower
side were investigated.
Treatment with NF-κB inhibitors
The NF-κB inhibitor BAY11-7082 and dexamethasone
were purchased from FujiFilm Wako Pure Chemical Corporation.
MDA-MB-231 cells were preincubated for 10 min with BAY11-7082 (10
µM) or dexamethasone (1 µM), and treated with control or low pH
media containing inhibitors or vehicle (ethanol) for 24 h at
37°C.
Western blotting
MDA-MB-231 cells were rinsed with PBS and lysed in
lysis buffer (FujiFilm Wako Pure Chemical Industries Ltd.). The
lysates were centrifuged at 15,000 × g for 20 min at 4°C and then
boiled in SDS sample buffer for 5 min. The proteins were separated
by 4–20% SDS-polyacrylamide gel electrophoresis (Bio-Rad
Laboratories) and transferred to polyvinylidene difluoride
membranes. After blocking in 5% BSA for 2 h at room temperature,
the membranes were incubated with primary antibodies, followed by
incubation with horseradish peroxidase-coupled anti-rabbit IgG
antibodies, and then bands were visualized using the ECL detection
kit (Amersham ECL Prime, Cytiva). Primary antibodies included
rabbit anti-nuclear factor (NF)-κB p65 (cat. no. 8242; 1:1,000
dilution, Cell Signaling Technology Inc.),
rabbit-anti-phospho-NF-κB p65 (cat. no. 3033; 1:1,000 dilution,
Cell Signaling Technology), and mouse anti-β-actin (product no.
A1978; 1:4,000 dilution, Sigma-Aldrich; Merck KGaA). β-actin was
used for reference protein. Secondary antibodies were as follows:
HRP-conjugated goat anti-rabbit IgG (H+L) (product code
111-036-003, 1:5,000 dilution, Jackson Immuno Research Laboratories
Inc.) and HRP-conjugated goat anti-mouse IgG (H+L) (product code
115-036-003, 1:10,000, Jackson Immuno Research Laboratories Inc.).
The bands were analyzed using Densitograph software CS Analyzer ver
3.0 (Atto Corporation).
Statistical analysis
Data are presented as the mean ± SD. Statistical
analyses were performed using data obtained from three independent
experiments. Student's t-test was used to compare data between two
groups. For more than three groups, we used a one-way ANOVA
followed by the Tukey-Kramer test or Dunnett test (JMP Pro software
version 14.1, SAS Institute Inc.). P-values of <0.05 were
considered to indicate statistical significance.
Results
Acidic conditions induce morphological
changes, migration activity, and invasion activity of MDA-MB-231
cells
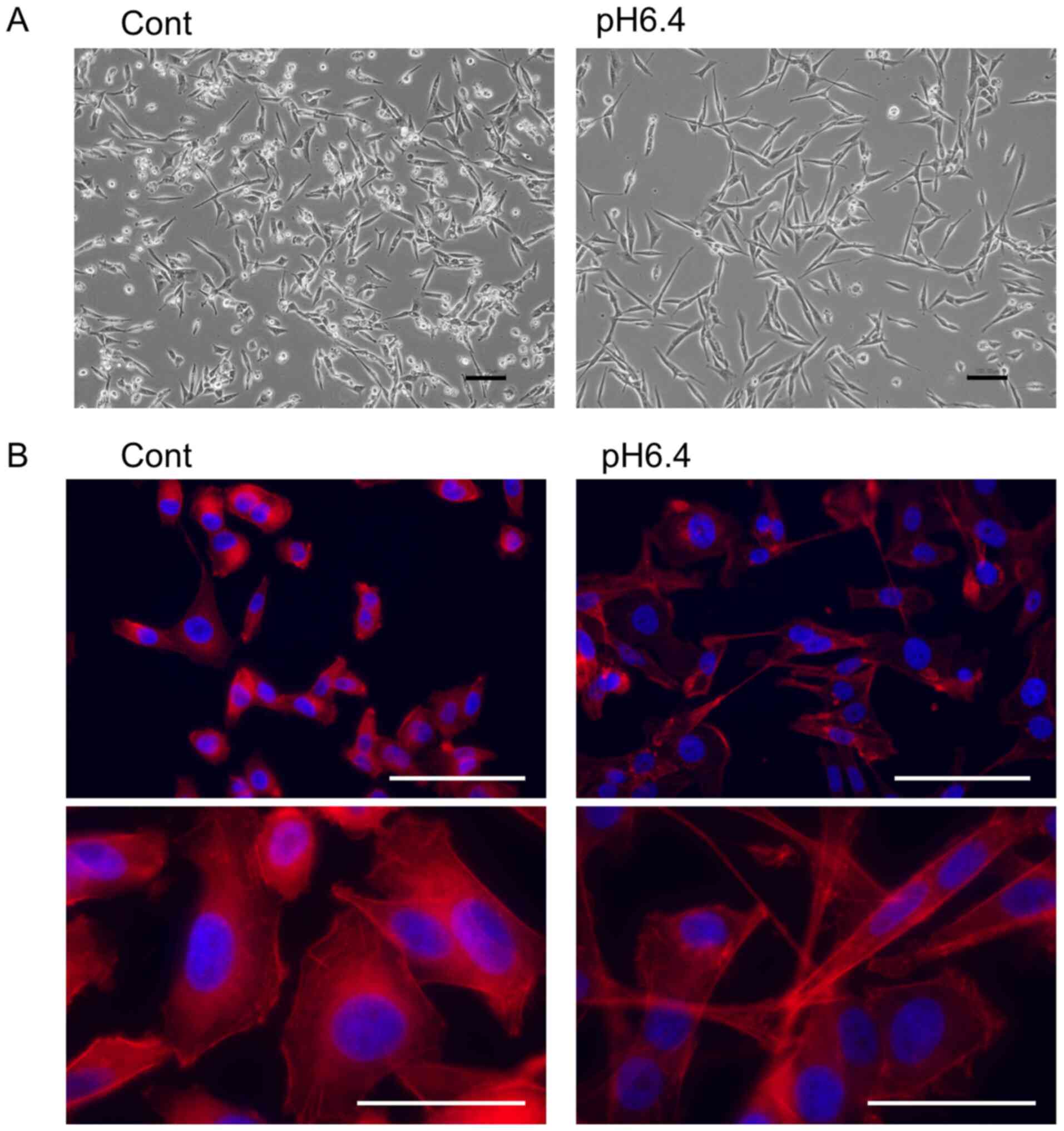
As shown in Fig. 1A,
the culture of MDA-MB-231 cells in low pH medium (pH 6.4) induced
morphological changes to spindle-shaped cells with processes. These
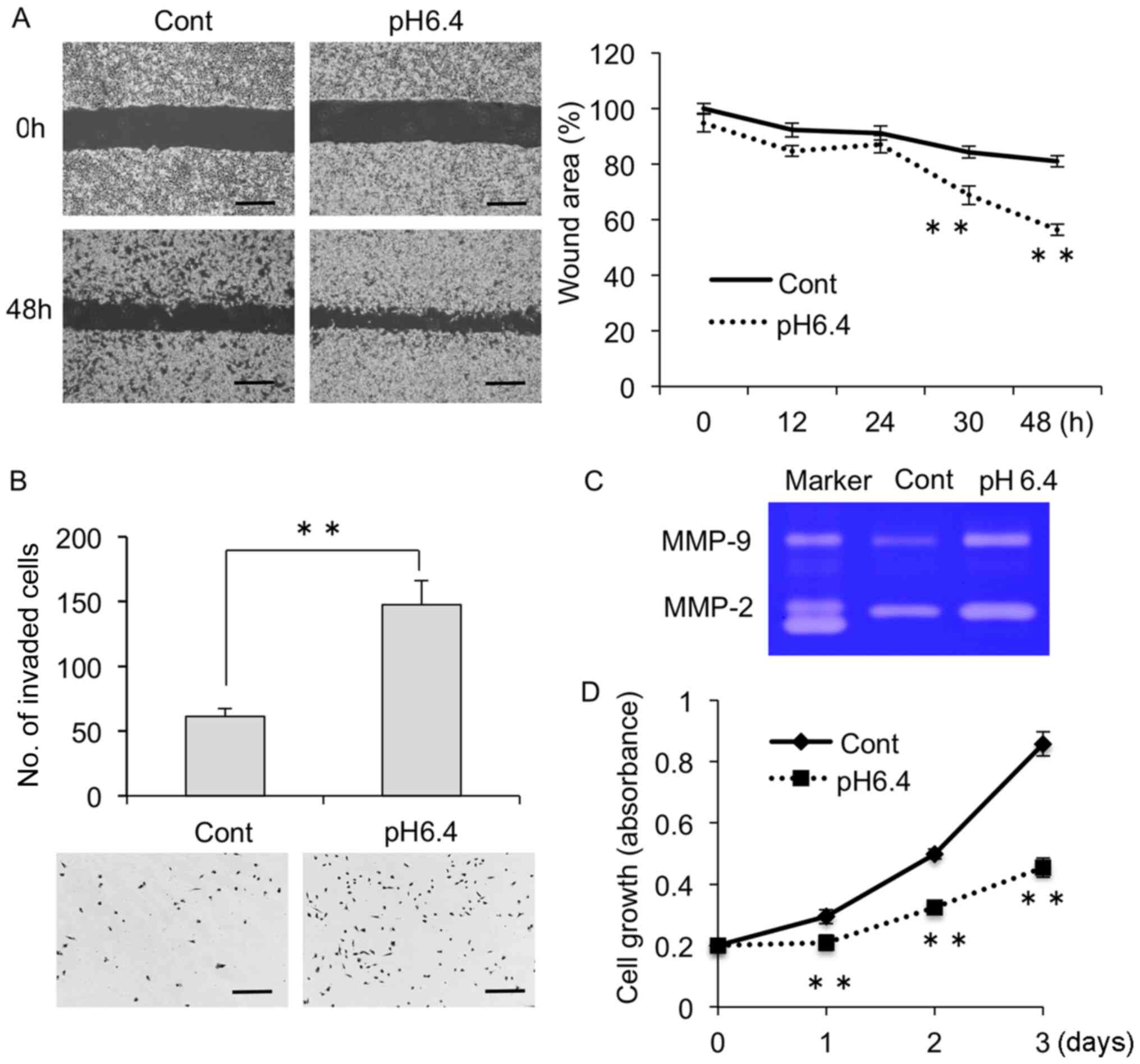
structures were also detected by Phalloidin staining (Fig. 1B). Wound healing assay revealed that
the migration activity was markedly increased in the MDA-MB-231
cells cultured in pH 6.4 medium (Fig.
2A). We also found that the invasion activity was increased in
cells cultured in acidic conditions, as determined by invasion
assays using a Transwell plate coated with Matrigel (Fig. 2B). In addition, conditioned medium
from cells cultured in acidic medium showed increased activity of
gelatinases such as MMP-2 and MMP-9 compared with medium from cells
cultured in normal medium (Fig.
2C). However, proliferation was suppressed in cells cultured
under acidic conditions compared with the control cells (Fig. 2D).
Increased IL-8 expression in
MDA-MB-231 cells under acidic conditions
To identify the potential factors involved in the
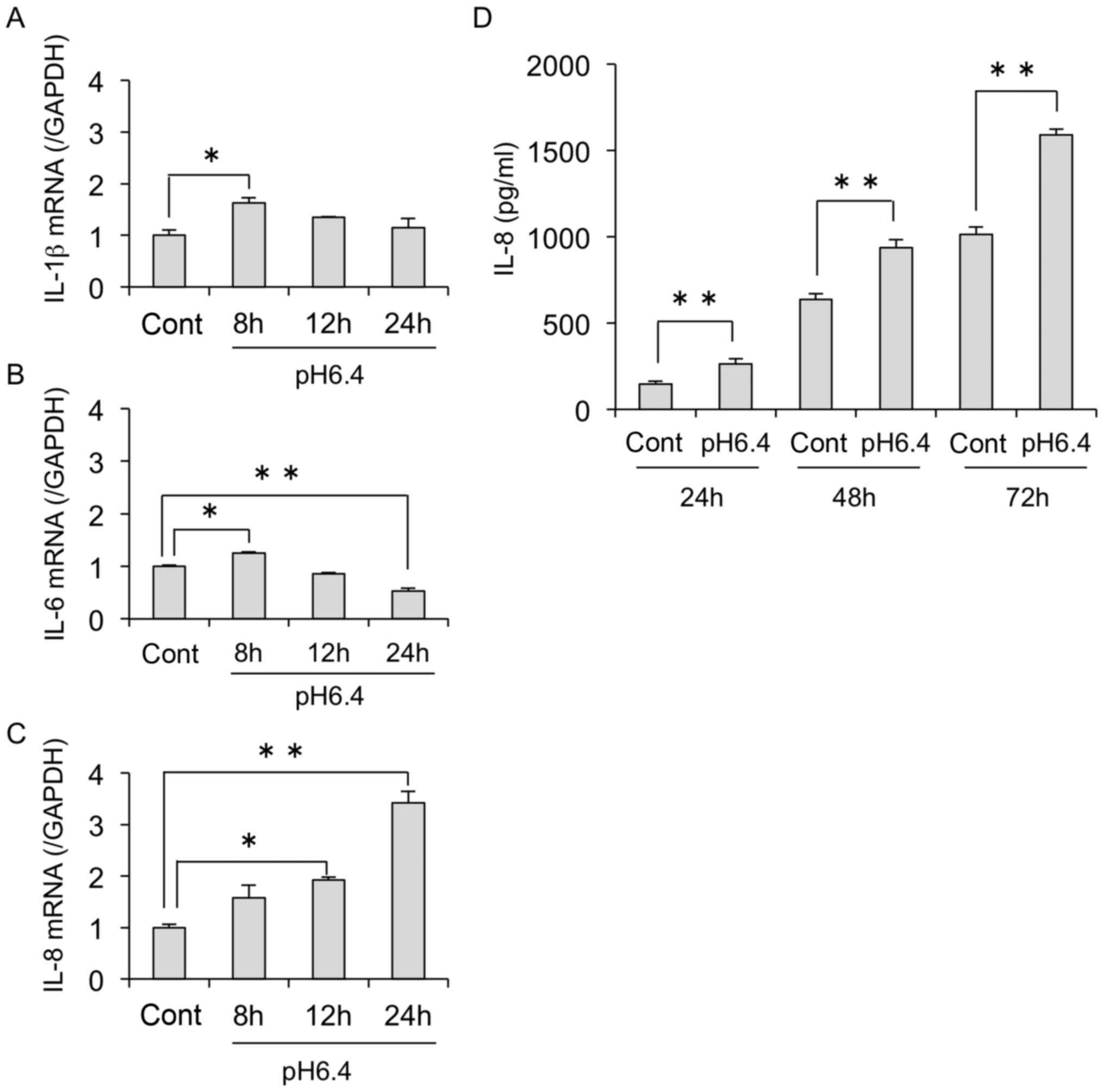
altered activity of cells under acidic conditions, we analyzed
changes in the expression of several cytokines induced by acidic
stimulation. The results showed that acidic conditions upregulated
the mRNA expressions of IL-8 but not IL-1β and
IL-6 in the MDA-MB-231 cells (Fig. 3A-C). In addition, we found that IL-8
secretion was also increased in a time-dependent manner in acidic
conditions, as determined by ELISA (Fig. 3D).
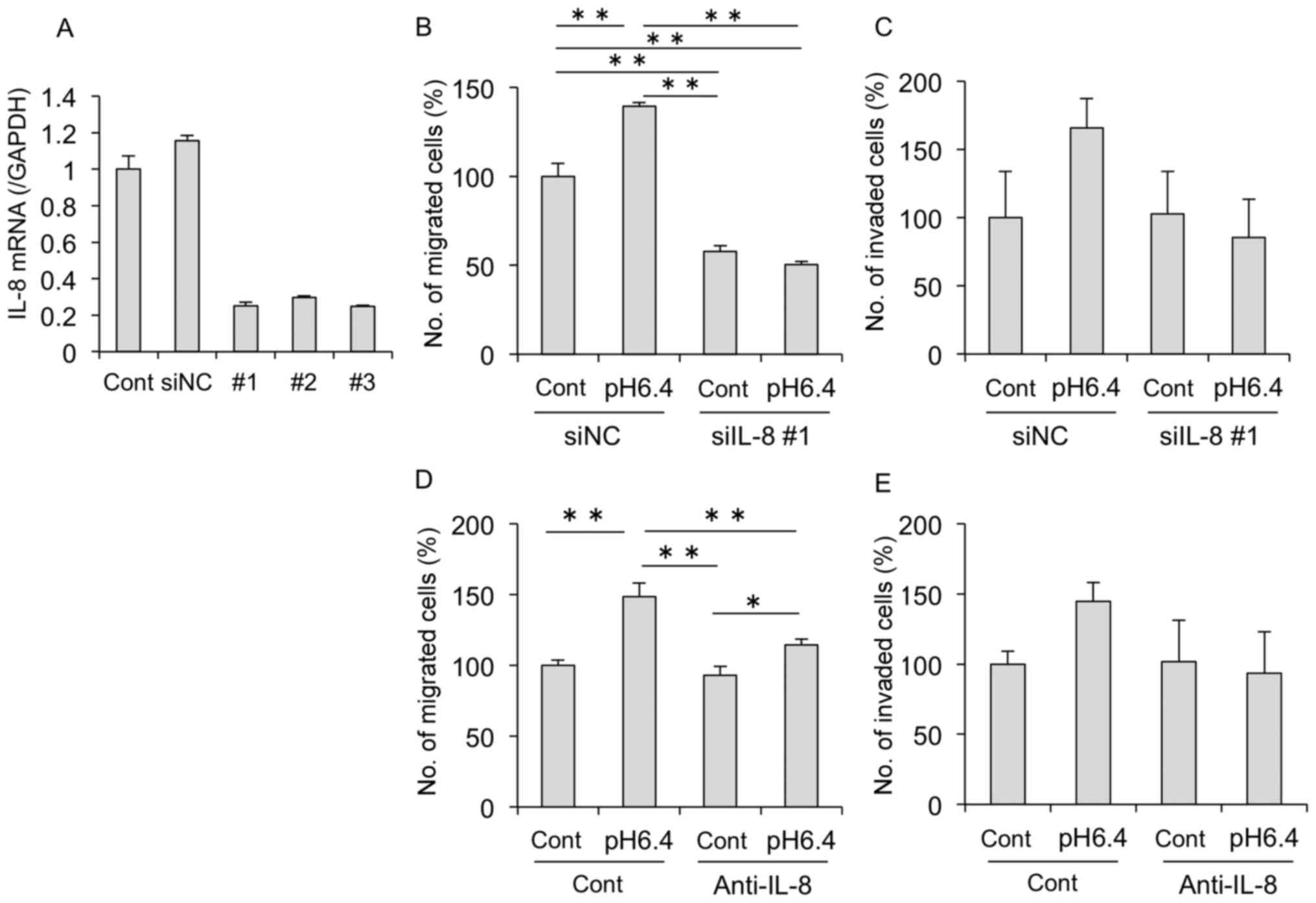
IL-8 is critical for migration and
invasion of MDA-MB-231 cells
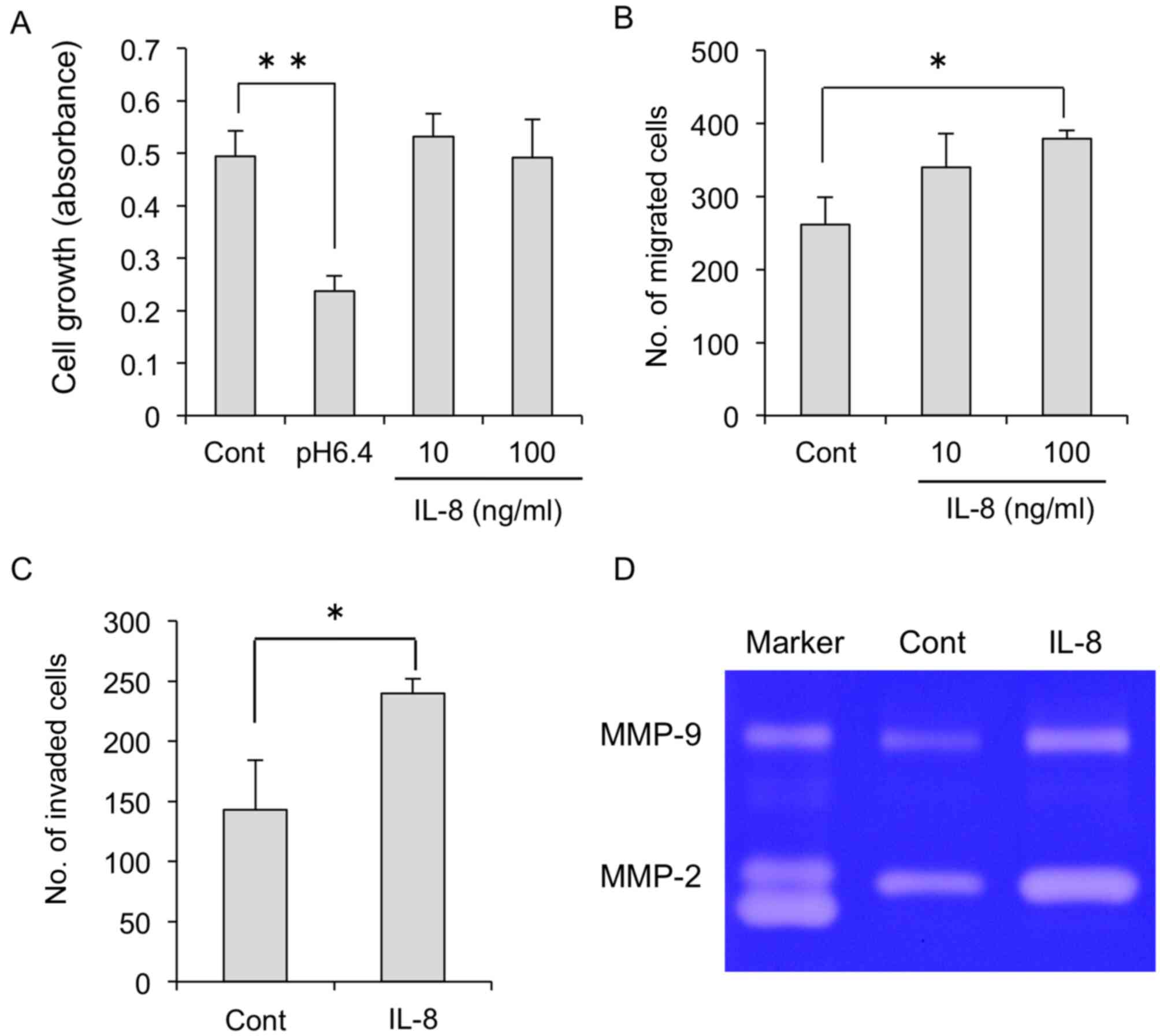
We next verified the direct effects of IL-8 on the
migration and invasion of MDA-MB-231 cells. As shown in Fig. 4A, IL-8 had no effects on cell
proliferation. In contrast, migration and invasion were
significantly increased by IL-8 addition (Fig. 4B and C). Of note, gelatinase
activity was also increased in MDA-MB-231 cells treated with IL-8
(Fig. 4D).
Therefore, we next confirmed the role of IL-8 by
knockdown experiments using siRNA targeting human IL-8 in
MDA-MB-231 cells. We confirmed no significant differences in
IL-8 mRNA expression between the untransfected cells and
those transfected with the negative control siRNA (siNC). The
expression of IL-8 mRNA was clearly decreased in cells
transfected with IL-8 siRNA (siIL-8 #1, #2, #3) compared
with the siNC-transfected cells (Fig.
5A). As shown in Fig. 5B and C,
acidic conditions increased the migration and invasion of the
MDA-MB-231 cells, while knockdown of IL-8 inhibited these
effects. Furthermore, similar results were confirmed when cells
were treated with an anti-IL-8 antibody (Fig. 5D and E). Treatment with the
anti-IL-8 antibody markedly suppressed acid-induced migration;
however, no significant difference was observed with invasion due
to the large SD. Therefore, these results suggest that IL-8 induced
by acidic stimulation is important for cell motility and
invasion.
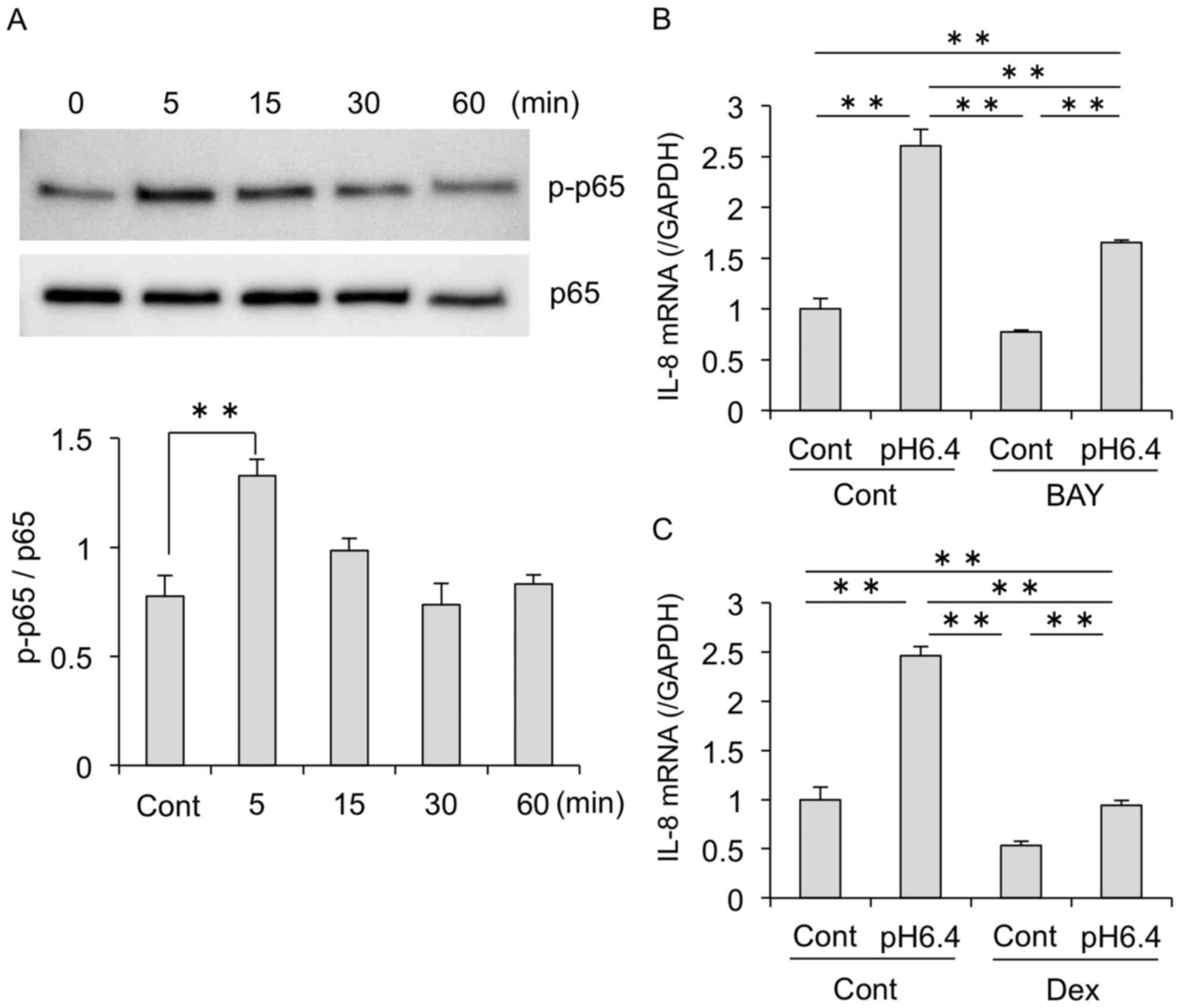
The NF-kB pathway is involved in
acid-induced IL-8 expression
Our results showed that acidic stimulation
upregulated IL-8 expression in MDA-MB-231 cells. Previous studies
have shown that NF-κB signaling regulates IL-8 induction in various
cell types (8,10,11,31).
To investigate the potential factors involved in acidic stimulation
of IL-8, we next examined whether acidic stimulation affects NF-κB
activation. Western blot analysis revealed that acidic conditions
increased the phosphorylation of NF-κB p65 (Fig. 6A). Moreover, treatment with the
NF-κB inhibitor BAY11-7082 decreased the expression of IL-8
mRNA induced by acidic conditions (Fig.
6B). Acid-induced IL-8 mRNA was also markedly decreased
by treatment with dexamethasone, another inhibitor of NF-κB
signaling (Fig. 6C).
ASIC1 is involved in acid-induced
changes in MDA-MB-231 cells
To elucidate the mechanism of the acid-induced
effects on in MDA-MB-231 cells, we next investigated the expression
of acid-sensing receptors. Previous studies have shown that
extracellular acidity activates acid receptors including ion
channels such as TRPV1 and ASICs (ASIC1–4), and GPCRs (OGR1, TDAG8,
GPR4, G2A). RT-PCR analysis revealed that MDA-MB-231 cells
expressed various pH-sensing receptors including TRPV1, ASIC1,
ASIC3, OGR1, TDAG8 and G2A (Fig. 7A). Furthermore, qPCR analysis
revealed that ASIC1 and ASIC3 mRNA expression was
significantly increased by acidic stimulation, whereas other
receptors were unchanged (Fig. 7B).
Therefore, we focused on ASIC1. Unexpectedly, acid stimulation
partially restored ASIC1 mRNA expression even when
ASIC1 was knocked down (Fig.
7C). Furthermore, we also found that knockdown of ASIC1
decreased IL-8 mRNA expression induced by acidic stimulation
(Fig. 7D). We also performed
experiments using siRNA against OGR1, which is strongly
expressed in MDA-MB-231; however, no inhibitory effect was observed
on the acid-induced expression of IL-8 mRNA (data not
shown). Together these results suggest that ASIC1 plays an
important role in acid-induced IL-8 expression.
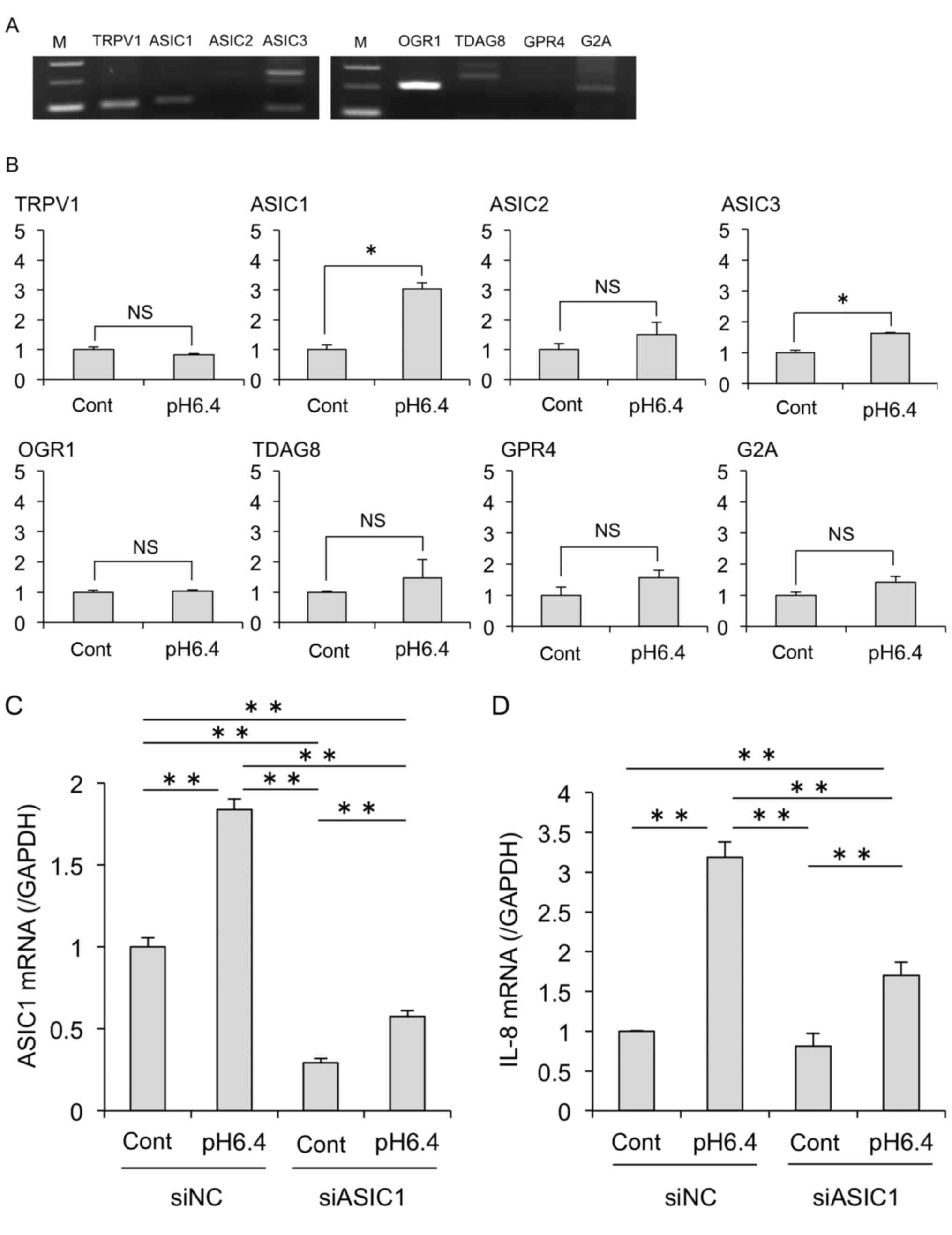 | Figure 7.Acid-sensing receptor ASIC1 is
critical to acid-induced IL-8 expression. (A) RT-PCR analysis
revealed that MDA-MB-231 cells express genes encoding various acid
receptors including ion channels and GPCRs. M, marker. (B) The
effect of acidic stimulation on the gene expression of acid
receptors was evaluated by real-time PCR. *P<0.05; NS, not
significant. (C and D) At 24 h after transfection of siRNA (siNC or
siASIC1) by electroporation, MDA-MB-231 cells were treated with
control or acidic medium. After 24 h, mRNA expression levels of
ASIC1 (C) and IL-8 (D) were analyzed by real-time
PCR. Data are shown as the fold expression normalized to the
control transfected with siNC (mean ± SD). *P<0.05, **P<0.01.
IL, interleukin; ASIC1, acid-sensing ion channel 1; GPCRs,
G-protein coupled receptors; TRPV1, transient receptor potential
vanilloid subtype 1; OGR1, also known as GPR68; G2A, also known as
GPR132; TDAG8, also known asGPR4. |
Discussion
Local acidosis is a characteristic feature of
cancer. The Warburg effect is a phenomenon in which tumors consume
significant amounts of glucose and produce lactate via the
anaerobic glycolytic pathway (4,26,27).
Because of the increased lactate, the tumor microenvironment
exhibits acidification, with the extracellular pH ranging between
6.0 and 6.5. This acidosis promotes processes such as metastasis,
angiogenesis, and immunosuppression, which have been associated
with a poor clinical prognosis (1–6).
We found that acidic stimulation induced
morphological changes in MDA-MB-231 cells to more spindle-shaped
cells with protrusions. Recently, Chen et al showed that the
acidic microenvironment induced invasion of prostate cancer cells
through induction of Snail (SNAI1) and Twist (TWIST1)
mRNA (28). We also examined the
expression of EMT-related genes, such as CDH1, CDH2, VIM,
SNAI1, and TWIST1, but there were no significant changes
in MDA-MB-231 cells following acidic treatment (data not shown).
Because MDA-MB-231 cells originally exhibit a mesenchymal phenotype
with higher levels of VIM and lower levels of CDH1,
EMT-related genes may have no relationship with the morphological
changes in this cell line. Therefore, we focused on protease
activity as a cause of migration and invasion. Under acidic
conditions, there were no significant changes in MMP2 and
MMP9 mRNA expression (data not shown). However, we observed
that cells cultured in acidic medium exhibited increased secretion
of gelatinase compared with cells in normal medium.
In this study, the proliferation was suppressed in
cells under acidic conditions. We initially expected that acidic
conditions promote cell growth. In fact, we found that the growth
of other cell lines was promoted by acidic conditions (data not
shown). We speculated that acidic stimulation of MDA-MB-231 cells
would preferentially induce cell survival pathways rather than
proliferation, although we did not examine apoptosis and cell
death. A recent study showed that extracellular acidosis induced
upregulation of p21, G1/G0 cell cycle arrest, and a reduction in
proliferation in human melanoma cells (29). In addition, the authors detected no
induction of cellular apoptosis but an increase in senescence
characteristics. Wojtkowiak et al reported that acute
exposure to acidic medium (pH 6.7 for 3 days) reduced the
proliferation of cells and promoted autophagy as a survival
adaptation in MDA-MB-231 cells (30). Furthermore, the authors showed that
long-term exposure (3 months) to low pH restored cellular
proliferative activity. We believe that changes in cellular
function associated with the acidic microenvironment need to be
investigated in more detail.
To determine the factors involved in acid-induced
migration and invasion, we investigated several cytokines that have
been implicated in the expression of the malignant phenotype in
cancer cells. We found that acidic stimulation upregulated the mRNA
expression and secretion of IL-8 in MDA-MB-231 cells. Végran et
al revealed that lactic acid from tumor cells stimulates the
IL-8 pathway in endothelial cells, resulting in angiogenesis and
tumor growth (31). In addition, we
previously showed that the acidic microenvironment induced IL-8
production in lymphatic endothelial cells and promoted cellular
proliferation and tube formation (11). These results suggest that increased
IL-8 expression in the tumor microenvironment has an important role
in cancer progression.
Therefore, we next investigated the direct effects
of IL-8 on several activities of MDA-MB-231 cells. Although a
previous study showed that the secretion of IL-8 from cancer cells
enhances the proliferation and survival of cancer cells via the
autocrine pathway (8), the
proliferative activity of MDA-MB-231 cells was not altered by IL-8
treatment in this study. In contrast, we found that the migration
and invasion of MDA-MB-231 cells were markedly increased by IL-8
treatment. Moreover, MMP-2/-9 activity was upregulated following
IL-8 treatment, which suggests a relationship between IL-8 and
acid-induced functional changes in MDA-MB-231 cells. Noteworthy,
IL-8 knockdown markedly inhibited these acid-induced
effects. These results showed that the acid-induced IL-8 production
accelerated migration and invasion via the autocrine pathway in
MDA-MB-231 cells.
The expression of IL-8 has been shown to be
regulated by many stimuli, such as inflammatory signals, chemical
and environmental stresses, and steroid hormones, and is primarily
induced via activator proteins and/or NF-κB-mediated
transcriptional activity (8,32). We
observed that acidic stimulation induced the phosphorylation of p65
within 15 min in MDA-MB-231 cells. In addition, acid-induced IL-8
expression was markedly decreased by treatment with NF-κB
inhibitors. Recently, Chen et al showed that acidosis
induced NF-κB activation through ERK signaling in prostate cancer
cells (28). However, even in the
presence of inhibitors, IL-8 expression levels in acidic conditions
were higher compared with the control. These results suggest that
signaling pathways other than NF-κB may also be involved in the
acid-induced IL-8 expression.
We demonstrated that MDA-MB-231 cells express
various acid-sensing receptors and that ASIC1 has an important
function in the regulation of IL-8 expression. Several reports have
described a relationship between breast cancer and the acid-sensing
ion channel TRPV1 (33–36). Weber et al showed that breast
cancer cells express functional TRPV1 and that TRPV1 activation
inhibited cell proliferation and induced apoptosis and necrosis
(34). In addition, Lozano et
al showed that intracellular aggregated TRPV1 is associated
with a shorter survival in breast cancer patients (35). These reports indicate that TRPV1 has
antitumor activity in breast cancer cells. In this study, we also
observed TRPV1 expression in MDA-MB-231 cells. However, whether
TRPV1 was involved in acid-induced IL-8 expression is unknown. In
addition to TRPV1, MDA-MB-231 cells also highly express OGR1.
Several studies suggest that OGR1 expressed in breast cancer cells
also functions as a tumor suppressor, promoting apoptosis and
inhibiting cell proliferation and migration (37–39).
In the present study, we also observed that acid-induced IL-8
expression was not altered by OGR1 knockdown (data not
shown). Further studies are needed to clarify the role of
acid-sensing GPCRs in breast cancer development.
Of note, we observed that acidic conditions induced
the expression of ASIC1 mRNA, and acid-induced IL-8
expression was markedly inhibited by ASIC1 knockdown.
Unexpectedly, ASIC1 mRNA was partially restored by acidic
stimulation, despite the siRNA knockdown. The mechanism by which
acid stimulation induces ASIC mRNA expression is not yet
known, but we speculate that this phenomenon is the cause of the
increased IL-8 expression in the siASIC1-transfected group.
Several studies suggest that ASIC1 expression is involved in breast
cancer progression (16,40). Chen et al reported that ASIC1
expression is upregulated in prostate cancer cases, and knockdown
of ASIC1 significantly suppressed cell proliferation and
invasion in vitro and in vivo (28). In addition, some reports have
indicated a relationship between ASIC1 expression and the
development of glioma (18,20). Collectively, these findings suggest
that ASIC1 has unique functions that differ from other acid
receptors and that there are important implications of ASIC1
expression in cancer progression in the acidic
microenvironment.
Acidosis in the cancer microenvironment is formed by
protons and lactic acid produced by cancer cells and the
surrounding stromal cells (2,27,41).
In this study, we focused on proton-sensing receptors, but it is
also necessary to study the effects of lactate via the lactate
receptors or transporters. Kolesnik et al revealed that
lactic acidosis promoted the survival and proliferation of Lewis
lung cancer cells through inhibition of autophagy and apoptosis
(42). In addition, Romero-Garcia
et al showed that lung cancer cells consume lactate and
induce mitochondrial biogenesis to support survival and
proliferation in lactic acidosis conditions (43). Monocarboxylate transporter (MCT)1-4
are lactate transporters, and overexpression of MCTs is a common
feature of some cancers with high metabolic rates (27,44).
Furthermore, some studies have reported that several GPCRs function
as sensors of lactate, and the expression levels of these GPCRs
correlate with tumor growth and metastasis (45,46).
Further investigation of the role of lactate receptors in IL-8
expression and cellular functions in breast cancer cells is
required.
In summary, we showed that an acidic
microenvironment induced IL-8 expression and invasion activity
through the activation of ASIC1 in breast cancer cells. These
findings suggest a critical role for ASIC1 in the cancer
microenvironment and indicate that ASIC1 and IL-8 may be novel
therapeutic targets for cancer progression.
Acknowledgements
We thank H. Nikki March and Gabrielle White Wolf for
editing a draft of this manuscript.
Funding
This work was supported in part by a Grant-in-Aid
for Scientific Research (MN) from the Japan Society for the
Promotion of Science (JSPS).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
MN designed the study. MN, AK, HY, NK and KN
performed the experiments. MN and YM analyzed and interpreted the
data, and wrote the manuscript. All authors read and approved the
manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interest.
References
|
1
|
Justus CR, Dong L and Yang LV: Acidic
tumor microenvironment and pH-sensing G protein-coupled receptors.
Front Physiol. 4:3542013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Estrella V, Chen T, Lloyd M, Wojtkowiak J,
Cornnell HH, Ibrahim-Hashim A, Bailey K, Balagurunathan Y, Rothberg
JM, Sloane BF, et al: Acidity generated by the tumor
microenvironment drives local invasion. Cancer Res. 73:1524–1535.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Damaghi M, Wojtkowiak JW and Gillies RJ:
pH sensing and regulation in cancer. Front Physiol. 4:3702013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kato Y, Ozawa S, Miyamoto C, Maehata Y,
Suzuki A, Maeda T and Baba Y: Acidic extracellular microenvironment
and cancer. Cancer Cell Int. 13:89–96. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boedtkjer E and Pedersen SF: The acidic
tumor microenvironment as a driver of cancer. Annu Rev Physiol.
82:103–126. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rofstad EK, Mathiesen B, Kindem K and
Galappathi K: Acidic extracellular pH promotes experimental
metastasis of human melanoma cells in athymic nude mice. Cancer
Res. 66:6699–6707. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nishisho T, Hata K, Nakanishi M, Morita Y,
Sun-Wada GH, Wada Y, Yasui N and Yoneda T: The a3 isoform vacuolar
type H(+)-ATPase promotes distant metastasis in the mouse B16
melanoma cells. Mol Cancer Res. 9:845–855. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Waugh DJ and Wilson C: The interleukin-8
pathway in cancer. Clin Cancer Res. 14:6735–6741. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zarogoulidis P, Katsikogianni F, Tsiouda
T, Sakkas A, Katsikogiannis N and Zarogoulidis K: Interleukin-8 and
interleukin-17 for cancer. Cancer Invest. 32:197–205. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Srivastava SK, Bhardwaj A, Arora S, Tyagi
N, Singh AP, Carter JE, Scammell JG, Fodstad Ø and Singh S:
Interleukin-8 is a key mediator of FKBP51-induced melanoma growth,
angiogenesis and metastasis. Br J Cancer. 112:1772–1781. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nakanishi M, Morita Y, Hata K and Muragaki
Y: Acidic microenvironments induce lymphangiogenesis and IL-8
production via TRPV1 activation in human lymphatic endothelial
cells. Exp Cell Res. 345:180–189. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Caterina MJ, Schumacher MA, Tominaga M,
Rosen TA, Levine JD and Julius D: The capsaicin receptor: A
heat-activated ion channel in the pain pathway. Nature.
389:816–824. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nakanishi M, Hata K, Nagayama T, Sakurai
T, Nishisho T, Wakabayashi H, Hiraga T, Ebisu S and Yoneda T: Acid
activation of Trpv1 leads to an up-regulation of calcitonin
gene-related peptide expression in dorsal root ganglion neurons via
the CaMK-CREB cascade: A potential mechanism of inflammatory pain.
Mol Biol Cell. 21:2568–2577. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yoneda T, Hata K, Nakanishi M, Nagae M,
Nagayama T, Wakabayashi H, Nishisho T, Sakurai T and Hiraga T:
Involvement of acidic microenvironment in the pathophysiology of
cancer-associated bone pain. Bone. 48:100–105. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sherwood TW, Frey EN and Askwith CC:
Structure and activity of the acid-sensing ion channels. Am J
Physiol Cell Physiol. 303:C699–C710. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wu Y, Gao B, Xiong Q, Wang Y, Huang D and
Wu W-N: Acid sensing ion channels contribute to the effect of
extracellular acidosis in proliferation and migration of A549
cells. Tumour Biol. 39:10104283177057502017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhu S, Zhou HY, Deng SC, Deng SJ, He C, Li
X, Chen J-Y, Jin Y, Hu Z-L, Wang F, et al: ASIC1 and ASIC3
contribute to acidity-induced EMT of pancreatic cancer through
activating Ca(2+)/RhoA pathway. Cell Death Dis. 8:e28062017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rooj AK, McNicholas CM, Bartoszewski R,
Bebok Z, Benos DJ and Fuller CM: Glioma-specific cation conductance
regulates migration and cell cycle progression. J Biol Chem.
287:4053–4065. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tian Y, Bresenitz P, Reska A, El Moussaoui
L, Beier CP and Gründer S: Glioblastoma cancer stem cell lines
express functional acid sensing ion channels ASIC1a and ASIC3. Sci
Rep. 7:136742017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Berdiev BK, Xia J, McLean LA, Markert JM,
Gillespie GY, Mapstone TB, Naren AP, Jovov B, Bubien JK, Ji H-L, et
al: Acid-sensing ion channels in malignant gliomas. J Biol Chem.
278:15023–15034. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kapoor N, Bartoszewski R, Qadri YJ, Bebok
Z, Bubien JK, Fuller CM and Benos DJ: Knockdown of ASIC1 and
epithelial sodium channel subunits inhibits glioblastoma whole cell
current and cell migration. J Biol Chem. 284:24526–24541. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gatenby RA and Gawlinski ET: A
reaction-diffusion model of cancer invasion. Cancer Res.
56:5745–5753. 1996.PubMed/NCBI
|
|
23
|
Stubbs M, McSheehy PM, Griffiths JR and
Bashford CL: Causes and consequence of tumour acidity and
implications for treatment. Mol Med Today. 6:15–19. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hashim AI, Zhang X, Wojtkowiak JW,
Martinez GV and Gillies RJ: Imaging pH and metastasis. NMR Biomed.
24:582–591. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wemmie JA, Taugher RJ and Kreple CJ:
Acid-sensing ion channels in pain and disease. Nat Rev Neurosci.
14:461–471. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Matsuo T and Sadzuka Y: Extracellular
acidification by lactic acid suppresses glucose deprivation-induced
cell death and autophagy in B16 melanoma cells. Biochem Biophys Res
Commun. 496:1357–1361. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
de la Cruz-López KG, Castro-Muñoz LJ,
Reyes-Hernández DO, García-Carrancá A and Manzo-Merino J: Lactate
in the regulation of tumor microenvironment and therapeutic
approaches. Front Oncol. 9:11432019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen B, Liu J, Ho TT, Ding X and Mo YY:
ERK-mediated NF-kappaB activation through ASIC1 in response to
acidosis. Oncogenesis. 5:e2792016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Böhme I and Bosserhoff A: Extracellular
acidosis triggers a senescence-like phenotype in human melanoma
cells. Pigment Cell Melanoma Res. 33:41–51. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wojtkowiak JW, Rothberg JM, Kumar V,
Schramm KJ, Haller E, Proemsey JB, Lloyd MC, Sloane BF and Gillies
RJ: Chronic autophagy is a cellular adaptation to tumor acidic pH
microenvironments. Cancer Res. 72:3938–3947. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Végran F, Boidot R, Michiels C, Sonveaux P
and Feron O: Lactate influx through the endothelial cell
monocarboxylate transporter MCT1 supports an NF-κB/IL-8 pathway
that drives tumor angiogenesis. Cancer Res. 71:2550–2560. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Brat DJ, Bellail AC and Van Meir EG: The
role of interleukin-8 and its receptors in gliomagenesis and
tumoral angiogenesis. Neuro-oncol. 7:122–133. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pecze L, Josvay K, Blum W, Petrovics G,
Vizler C, Oláh Z and Schwaller B: Activation of endogenous TRPV1
fails to induce overstimulation-based cytotoxicity in breast and
prostate cancer cells but not in pain-sensing neurons. Biochim
Biophys Acta. 1863:2054–2064. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Weber LV, Al-Refae K, Wölk G, Bonatz G,
Altmüller J, Becker C, Gisselmann G and Hatt H: Expression and
functionality of TRPV1 in breast cancer cells. Breast Cancer (Dove
Med Press). 8:243–252. 2016.PubMed/NCBI
|
|
35
|
Lozano C, Córdova C, Marchant I, Zúñiga R,
Ochova P, Ramírez-Barrantes R, González-Arriagada WA, Rodriguez B
and Olivero P: Intracellular aggregated TRPV1 is associated with
lower survival in breast cancer patients. Breast Cancer (Dove Med
Press). 10:161–168. 2018.PubMed/NCBI
|
|
36
|
So CL, Milevskiy MJG and Monteith GR:
Transient receptor potential cation channel subfamily V and breast
cancer. Lab Invest. 100:199–206. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Singh LS, Berk M, Oates R, Zhao Z, Tan H,
Jiang Y, Zhou A, Kirmani K, Steinmetz R, Lindner D, et al: Ovarian
cancer G protein-coupled receptor 1, a new metastasis suppressor
gene in prostate cancer. J Natl Cancer Inst. 99:1313–1327. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li J, Guo B, Wang J, Cheng X, Xu Y and
Sang J: Ovarian cancer G protein coupled receptor 1 suppresses cell
migration of MCF7 breast cancer cells via a
Gα12/13-Rho-Rac1 pathway. J Mol Signal. 8:62013.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang J, Che L, Sun W, Shang J, Hao M and
Tian M: Correlation of OGR1 with proliferation and apoptosis of
breast cancer cells. Oncol Lett. 17:4335–4340. 2019.PubMed/NCBI
|
|
40
|
Gupta SC, Singh R, Asters M, Liu J, Zhang
X, Pabbidi MR, Watabe K and Mo Y-Y: Regulation of breast
tumorigenesis through acid sensors. Oncogene. 35:4102–4111. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
García-Cañaveras JC, Chen L and Rabinowitz
JD: The tumor metabolic microenvironment: Lessons from lactate.
Cancer Res. 79:3155–3162. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kolesnik DL, Pyaskovskaya ON and Solyanik
GI: Impact of lactic acidosis on the survival of Lewis lung
carcinoma cells. Exp Oncol. 39:112–116. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Romero-Garcia S, Prado-Garcia H,
Valencia-Camargo AD and Alvarez-Pulido A: Lactic acidosis promotes
mitochondrial biogenesis in lung adenocarcinoma cells, supporting
proliferation under normoxia or survival under hypoxia. Front
Oncol. 9:10532019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Pinheiro C, Garcia EA, Morais-Santon F,
Scapulatempo-Neto C, Mafra A, Steenbergen RDM, Boccardo E, Villa
LL, Baltazar F and Longatto-Filho A: Lactate transporters and
vascular factors in HPV-induced squamous cell carcinoma of the
uterine cervix. BMC Cancer. 14:7512014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Roland CL, Arumugam T, Deng D, Liu SH,
Philip B, Gomez S, Burns WR, Ramachandran V, Wang H,
Cruz-Monserrate Z, et al: Cell surface lactate receptor GPR81 is
crucial for cancer cell survival. Cancer Res. 74:5301–5310. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen P, Zuo H, Xiong H, Kolar MJ, Chu Q,
Saghatelian A, Siegwart DJ and Wan Y: Gpr132 sensing of lactate
mediates tumor-macrophage interplay to promote breast cancer
metastasis. Proc Natl Acad Sci USA. 114:580–585. 2017. View Article : Google Scholar : PubMed/NCBI
|