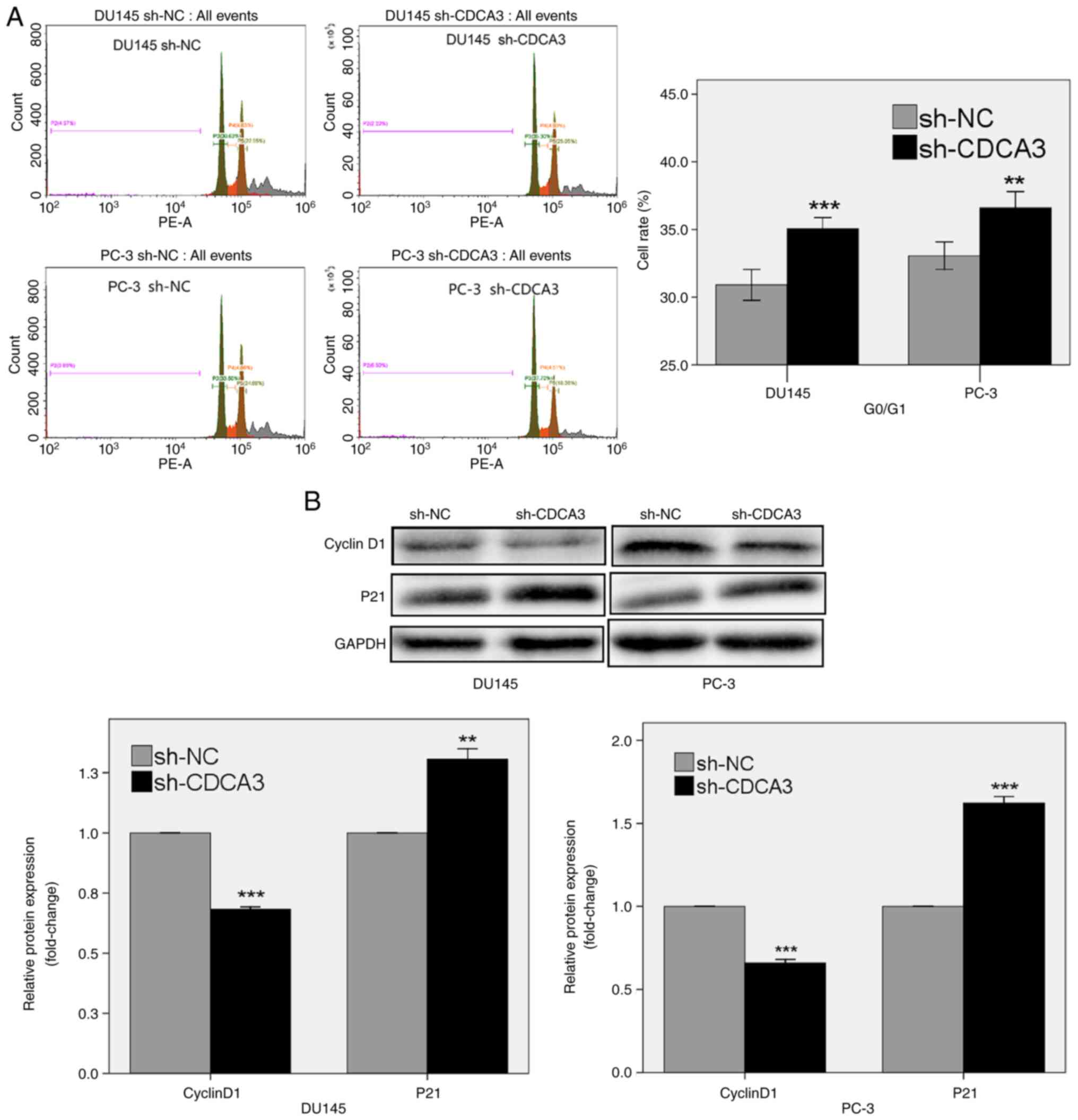
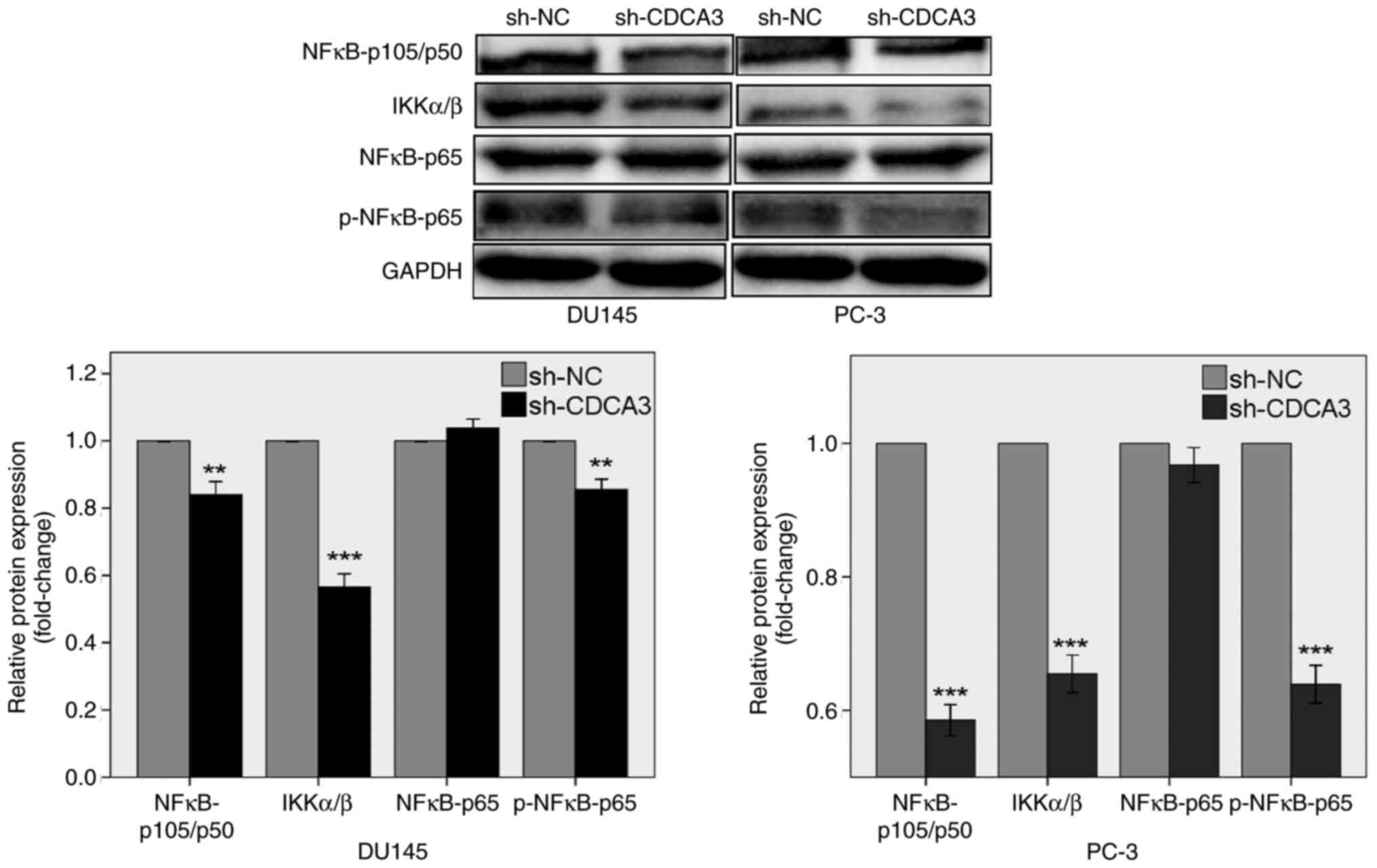
|
1
|
Jin L, Zhou Y, Chen G, Dai G, Fu K, Yang D
and Zhu J: EZH2-TROAP pathway promotes prostate cancer progression
Via TWIST signals. Front Oncol. 10:5922392020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Attard G, Parker C, Eeles RA, Schröder F,
Tomlins SA, Tannock I, Drake CG and de Bono JS: Prostate cancer.
Lancet. 387:70–82. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wei B, Ruan J, Mi Y, Hu J, Zhang J, Wang
Z, Hu Q, Jiang H and Ding Q: Knockdown of TNF receptor-associated
factor 2 (TRAF2) modulates in vitro growth of TRAIL-treated
prostate cancer cells. Biomed Pharmacother. 93:462–469. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
De Angelis ML, Francescangeli F, La Torre
F and Zeuner A: Stem cell plasticity and dormancy in the
development of cancer therapy resistance. Front Oncol. 9:6262019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ahn JY, Hwang HS, Park YS, Kim HR, Jung
HY, Kim JH, Lee SE and Kim MA: Endoscopic and pathologic findings
associated with clinical outcomes of melanoma in the upper
gastrointestinal tract. Ann Surg Oncol. 21:2532–2539. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wu ZH, Fang M and Zhou Y: Comprehensive
analysis of the expression and prognosis for CDCAs in head and neck
squamous cell carcinoma. PLoS One. 15:e02366782020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Phan NN, Wang CY, Li KL, Chen CF, Chiao
CC, Yu HG, Huang PL and Lin YC: Distinct expression of CDCA3,
CDCA5, and CDCA8 leads to shorter relapse free survival in breast
cancer patient. Oncotarget. 9:6977–6992. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Obara W, Sato F, Takeda K, Kato R, Kato Y,
Kanehira M, Takata R, Mimata H, Sugai T, Nakamura Y and Fujioka T:
Phase I clinical trial of cell division associated 1 (CDCA1)
peptide vaccination for castration resistant prostate cancer.
Cancer Sci. 108:1452–1457. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang Y, Cheng Y, Zhang Z, Bai Z, Jin H,
Guo X, Huang X, Li M, Wang M, Shu XS, et al: CDCA2 inhibits
apoptosis and promotes cell proliferation in prostate cancer and is
directly regulated by HIF-1α pathway. Front Oncol. 10:7252020.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Clermont PL, Crea F, Chiang YT, Lin D,
Zhang A, Wang JZ, Parolia A, Wu R, Xue H, Wang Y, et al:
Identification of the epigenetic reader CBX2 as a potential drug
target in advanced prostate cancer. Clin Epigenetics. 8:162016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Song Z, Huang Y, Zhao Y, Ruan H, Yang H,
Cao Q, Liu D, Zhang X and Chen K: The identification of potential
biomarkers and biological pathways in prostate cancer. J Cancer.
10:1398–1408. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Qian W, Zhang Z, Peng W, Li J, Gu Q, Ji D,
Wang Q, Zhang Y, Ji B, Wang S, et al: CDCA3 mediates p21-dependent
proliferation by regulating E2F1 expression in colorectal cancer.
Int J Oncol. 53:2021–2033. 2018.PubMed/NCBI
|
|
14
|
Zhang W, Lu Y and Li X, Zhang J, Zheng L,
Zhang W, Lin C, Lin W and Li X: CDCA3 promotes cell proliferation
by activating the NF-κB/cyclin D1 signaling pathway in colorectal
cancer. Biochem Biophys Res Commun. 500:196–203. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang Y, Yin W, Cao W, Chen P, Bian L and
Ni Q: CDCA3 is a potential prognostic marker that promotes cell
proliferation in gastric cancer. Oncol Rep. 41:2471–2481.
2019.PubMed/NCBI
|
|
16
|
Adams MN, Burgess JT, He Y, Gately K,
Snell C, Zhang SD, Hooper JD, Richard DJ and O'Byrne KJ: Expression
of CDCA3 Is a prognostic biomarker and potential therapeutic target
in non-small cell lung cancer. J Thorac Oncol. 12:1071–1084. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Uchida F, Uzawa K, Kasamatsu A, Takatori
H, Sakamoto Y, Ogawara K, Shiiba M, Tanzawa H and Bukawa H:
Overexpression of cell cycle regulator CDCA3 promotes oral cancer
progression by enhancing cell proliferation with prevention of G1
phase arrest. BMC Cancer. 12:3212012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zou RC, Guo ZT, Wei D, Shi ZT, Ye ZC, Zhai
G, Zhong C, Tang B, Wang L and Ge JY: Downregulation of CDCA3
expression inhibits tumor formation in pancreatic cancer.
Neoplasma. 67:1223–1232. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen J, Zhu S, Jiang N, Shang Z, Quan C
and Niu Y: HoxB3 promotes prostate cancer cell progression by
transactivating CDCA3. Cancer Lett. 330:217–224. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu L, Guo K, Liang Z, Li F and Wang H:
Identification of candidate genes that may contribute to the
metastasis of prostate cancer by bioinformatics analysis. Oncol
Lett. 15:1220–1228. 2018.PubMed/NCBI
|
|
21
|
Guo L, Liu Y, Ding Z, Sun W and Yuan M:
Signal transduction by M3 muscarinic acetylcholine receptor in
prostate cancer. Oncol Lett. 11:385–392. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gu P, Yang D, Zhu J, Zhang M and He X:
Bioinformatics analysis identified hub genes in prostate cancer
tumorigenesis and metastasis. Math Biosci Eng. 18:3180–3196. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chandrashekar DS, Bashel B, Balasubramanya
SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi BVSK and
Varambally S: UALCAN: A portal for facilitating tumor subgroup gene
expression and survival analyses. Neoplasia. 19:649–658. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chin CH, Chen SH, Wu HH, Ho CW, Ko MT and
Lin CY: cytoHubba: Identifying hub objects and sub-networks from
complex interactome. BMC Syst Biol. 8 (Suppl 4):S112014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou Y, Gu P, Li J, Li F, Zhu J, Gao P,
Zang Y, Wang Y, Shan Y and Yang D: Suppression of STIM1 inhibits
the migration and invasion of human prostate cancer cells and is
associated with PI3K/Akt signaling inactivation. Oncol Rep.
38:2629–2636. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Qie S and Diehl JA: Cyclin D1, cancer
progression, and opportunities in cancer treatment. J Mol Med
(Berl). 94:1313–1326. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma T, Chen H, Wang P, Yang N and Bao J:
Downregulation of lncRNA ZEB1-AS1 represses cell proliferation,
migration, and invasion through mediating PI3K/AKT/mTOR signaling
by miR-342-3p/CUL4B Axis in prostate cancer. Cancer Biother
Radiopharm. 35:661–672. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nguyen DP, Li J, Yadav SS and Tewari AK:
Recent insights into NF-κB signalling pathways and the link between
inflammation and prostate cancer. BJU Int. 114:168–176. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dominska K, Kowalska K, Matysiak ZE,
Pluciennik E, Ochedalski T and Piastowska-Ciesielska AW: Regulation
of mRNA gene expression of members of the NF-κB transcription
factor gene family by angiotensin II and relaxin 2 in normal and
cancer prostate cell lines. Mol Med Rep. 15:4352–4359. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ji J, Shen T, Li Y, Liu Y, Shang Z and Niu
Y: CDCA5 promotes the progression of prostate cancer by affecting
the ERK signalling pathway. Oncol Rep. 45:921–932. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zheng P, Wu K, Gao Z, Li H, Li W, Wang X,
Shi Z, Xiao F, Wang K, Li Z and Han Q: KIF4A promotes the
development of bladder cancer by transcriptionally activating the
expression of CDCA3. Int J Mol Med. 47:992021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bi L, Zhou B, Li H, He L, Wang C, Wang Z,
Zhu L, Chen M and Gao S: A novel miR-375-HOXB3-CDCA3/DNMT3B
regulatory circuitry contributes to leukemogenesis in acute myeloid
leukemia. BMC Cancer. 18:1822018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu Y, Cheng G, Huang Z, Bao L, Liu J,
Wang C, Xiong Z, Zhou L, Xu T, Liu D, et al: Long noncoding RNA
SNHG12 promotes tumour progression and sunitinib resistance by
upregulating CDCA3 in renal cell carcinoma. Cell Death Dis.
11:5152020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jin M, Duan J, Liu W, Ji J, Liu B and
Zhang M: Feedback activation of NF-KB signaling leads to adaptive
resistance to EZH2 inhibitors in prostate cancer cells. Cancer Cell
Int. 21:1912021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jung AR, Kim GE, Kim MY, Ha US, Hong SH,
Lee JY, Kim SW and Park YH: HMGB1 promotes tumor progression and
invasion through HMGB1/TNFR1/NF-κB axis in castration-resistant
prostate cancer. Am J Cancer Res. 11:2215–2227. 2021.PubMed/NCBI
|
|
36
|
Staal J and Beyaert R: Inflammation and
NF-κB signaling in prostate cancer: Mechanisms and clinical
implications. Cells. 7:1222018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
He S, Chen M, Lin X, Lv Z, Liang R and
Huang L: Triptolide inhibits PDGF-induced proliferation of ASMCs
through G0/G1 cell cycle arrest and suppression of the
AKT/NF-κB/cyclinD1 signaling pathway. Eur J Pharmacol.
867:1728112020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang R, Li J, Yan X, Jin K, Li W, Liu X,
Zhao J, Shang W and Zhao X: Long noncoding RNA MLK7AS1 promotes
proliferation in human colorectal cancer via downregulation of p21
expression. Mol Med Rep. 19:1210–1221. 2019.PubMed/NCBI
|
|
39
|
Cui X, Cui M, Asada R, Kanemoto S, Saito
A, Matsuhisa K, Kaneko M and Imaizumi K: The androgen-induced
protein AIbZIP facilitates proliferation of prostate cancer cells
through downregulation of p21 expression. Sci Rep. 6:373102016.
View Article : Google Scholar : PubMed/NCBIPubMed/NCBI
|