Introduction
Head and neck squamous cell carcinoma (HNSCC) is the
seventh most common cancer worldwide (1,2).
Treatment depends largely on the stage of the tumor and is based on
a multimodality strategy primarily involving surgery and
radiotherapy. Patients with advanced disease are offered a
cisplatin-based chemotherapy or combination of radiotherapy with
cetuximab, a monoclonal antibody targeting epidermal growth factor
receptor (EGFR) (3). Despite the
progress in therapy, both surgery and radiotherapy, similar to
chemotherapy, patient survival has not substantially improved in
the last few decades. The 5-year overall survival in patients with
advanced HNSCC remains <50% (4).
Hypoxia in HNSCC has been linked with poor response
to treatment, increased metastasis and tumor aggressivity (5,6).
Adaptive responses of cells to hypoxia include stimulation of
angiogenesis and alteration of cellular metabolism (7). The reduced oxygen tension in cells
leads to activation of the hypoxia-inducible factor (HIF)-1
transcription factor, which is constitutively produced and degraded
by the ubiquitin-proteasome system under normoxic conditions but
becomes stabilized and transcriptionally active under hypoxic
conditions (8). HIF-1 regulates
the transcription of hundreds of genes that code for proteins
involved in various aspects of cancer biology (9), such as angiogenesis (10), metabolism (11), genetic instability (12), invasion and metastasis (13,14),
resistance to chemotherapy and radiation (9,10,15).
Ionizing radiation is used extensively and is an integral part of
cancer treatment. The mechanism underlying radiation resistance in
human cancer is not fully understood. However, research into tumor
radioresistance in HNSCC has suggested distinct mechanisms that
have a negative impact on tumor irradiation, including hypoxia
(16). Tumor recurrence after
radiotherapy is a major obstacle to recovery in HNSCC (17).
During metastasis, tumor cells lose the cell-cell
adhesion capacity and acquire the capability of cell motility for
invasion via the epithelial-mesenchymal transition (EMT). In a
majority of tumors, including HNSCC, EMT is tightly linked to
hypoxia, leading to an increased radioresistance (18,19).
Due to the importance of hypoxia in tumors, there is
a requirement for reliable biomarkers for this condition to serve
as a diagnostic marker of hypoxia and a potential therapeutic
target. To date, a number of tumor tissue markers have been
described as potential biomarkers in HNSCC (20). Among them, the expression of
hypoxia markers, including HIF-1a, carbonic anhydrase 9 (CA9) and
glucose transporter (GLUT)1, in HNSCC is associated with poor
prognosis for the patients (21,22).
Several studies using gene expression microarrays
have demonstrated that the expression levels of various genes can
be used to characterize different tumor types and to identify
diagnostic and therapeutic biomarkers (23,24).
In the present study, cDNA microarrays were used to assess the
impact of hypoxia (1% O2) on the global gene expression
in HNSCC and to identify hypoxia-induced differentially expressed
genes possessing potential therapeutic impact.
Materials and methods
Cell lines and culture conditions
In this study, the UT-SCC-14 cell line from the
University of Turku (provided by Professor Reidar Grénman at the
Department of Otorhinolaryngology, Head and Neck Surgery, Turku
University Central Hospital), and two cell lines from University of
Linköping, LK0858 and LK0863 (25), were used. All cell lines were
derived from tissue specimens from patients diagnosed with HNSCC.
The UT-SCC-14 and LK0858 cell lines originate from the tongue,
whereas LK0863 originates from the larynx. The present study was
approved by the Research Ethics Committee of the Linköping
University (approval no. 03–537). Written consent was obtained from
the patients involved in the study.
All cell lines were cultured in keratinocyte
serum-free medium supplemented with antibiotics (50 U/ml
penicillin, 50 µg/ml streptomycin) and 1% FBS (all from Gibco;
Thermo Fisher Scientific, Inc.).
The cells were given fresh culture media twice per
week and were subcultured to confluence after detaching the cells
with 0.25% trypsin + 0.02% EDTA at a weekly split ratio of ~1:2.
Cultures from passages 10 to 25 were used in all experiments. The
cells were screened periodically for mycoplasma contamination using
a Mycoplasma Detection kit (MycoAlert™; Lonza Group, Ltd.).
Introduction of hypoxia
Hypoxic conditions were achieved in a New Brunswick
Galaxy® CO2 incubator 14S at 37°C with
atmospheric conditions of 5% CO2 and 1% O2.
The cells were cultured in hypoxic conditions for 24 h prior to
planned experiments.
Cell proliferation assay
Tumor cells were seeded in 12-well plates at
densities of 300–800 cells/cm2, depending on the plating
efficiency of each cell line. Selected cells were irradiated (2, 4
or 6 Gy) with 4 MeV photons generated by a linear accelerator
(Clinac 4/100; Varian Medical Systems), delivering a dose-rate of
2.0 Gy/min. The cytostatic/cytotoxic effect was determined after
another 9 days. After fixation in 4% paraformaldehyde (20 min; room
temperature), the cells were stained with crystal violet (0.04% in
1% ethanol; room temperature) for 20 min at room temperature, and
were then washed and air-dried. After solubilization in 1% SDS, the
optical density at 550 nm was measured using a Victor plate reader
(EG&G Wallac).
Reverse transcription-quantitative PCR
(RT-qPCR)
RT-qPCR analysis was performed on a 7500 Fast
Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific,
Inc.). Total RNA was extracted from the cells using a RNeasy Mini
kit (Qiagen GmbH), the cDNA was synthesized using a High-Capacity
RNA-to-cDNA kit according to the manufacturer's instructions
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The
expression of hypoxia-responsive genes was analyzed with a panel of
TaqMan® Gene Expression assays [cadherin 1 (CDH1;
Hs01023895_m1); cadherin 2 (CDH2; Hs00983056_m1); vimentin
(VIM; Hs00958111_m1); fibronectin 1 (FN1; Hs01549976_m1); FOXC2
(Hs00270951_s1); TWIST1 (Hs04989912_s1); CD44 (Hs01075864_m1); SOX2
(Hs04234836_s1); NANOG (Hs02387400_g1); GLUT3
(Hs00359840_m1); CA9 (Hs00154208_m1); caspase 14
(CASP14; Hs00201637_m1); serpin family E 1 (SERPINE1;
Hs00167155_m1); lysyl oxidase (LOX; Hs00942480_m1);
amphiregulin (AREG; Hs00950669_m1); epiregulin (EREG;
Hs00914313_m1); all labelled with FAM and purchased from Thermo
Fisher Scientific, Inc.] and amplified using a TaqMan real-time PCR
protocol according to manufacturer's instructions (Thermo Fisher
Scientific, Inc.). The following thermocycling conditions were
used: Initial denaturation at 95°C for 10 min followed by 40 cycles
at 95°C for 15 sec and 60°C for 1 min.
Amplification of both GAPDH [TaqMan Gene
Expression assay (Hs02758991_g1); Thermo Fisher Scientific, Inc.]
and ACTB [TaqMan Gene Expression assay (Hs99999903_m1);
Thermo Fisher Scientific, Inc.] was used as an internal standard.
The comparative Cq method was applied to determine the
fold-difference in expression levels relative to a control sample
(26).
Western blot analysis
Whole cell extracts were prepared from hypoxic and
normoxic HNSCC cells. The cells were lysed with RIPA buffer (cat.
no. 89900) supplemented with protease and phosphatase inhibitor
(cat. no. A32959) (both from Thermo Fisher Scientific, Inc.).
Protein concentration was determined using the DC protein assay
(cat. no. 5000111; BioRad Laboratories, Inc.). For each sample, 20
µg protein was separated using 10% SDS-PAGE (CBS Scientific),
blotted onto a nitrocellulose membrane using an iBlot 2 transfer
device (Thermo Fisher Scientific, Inc.), followed by blocking for
60 min in TBS-Tween-20 (0.1%; TBS-Tween) containing 5% skimmed milk
and 1% BSA (Sigma-Aldrich; Merck KGaA). The membranes were
incubated with anti-HIF-1α (cat. no. 610959; 1:500; BD
Biosciences), anti-CDH2 (cat. no. ab18203; 1:1,000; Abcam),
anti-CA9 (cat. no. NB100-417; 1:1,000; Novus Biologicals, Ltd.) or
anti-SERPINE1 (cat. no. ab125687; 1:1,000; Abcam) antibodies in
TBS-0.1% Tween20 containing 0.1% skimmed milk at overnight at 4°C.
The membranes were then washed with TBS-Tween (0.1%) and incubated
for 1 h at room temperature with a HRP-conjugated anti-mouse (cat.
no. AP127P) or anti-rabbit (AP156P) antibodies (both 1:3,000 and
from MilliporeSigma). The bands were visualized with Western
Blotting Luminol Reagent (Bio-Rad Laboratories, Inc.) and captured
digitally using the Chemidoc XRS system (Bio-Rad Laboratories,
Inc.). Equal loading was verified by reprobing the membranes for 60
min at room temperature with an HRP-conjugated anti-β-actin
antibody (cat. no. A5441; 1:10,000 Sigma-Aldrich; Merck KGaA).
RNA interference
Cells were seeded at a density of 12,000
cells/cm2 and transfected 24 h later with FlexiTube
small interfering (si)RNA (cat. no. 1027416; Qiagen GmbH) against
AREG (cat. nos. SI00299845 and SI03049683), EREG (cat. no.
SI04199244), HIF-1α (cat. nos. SI02664053 and SI04262041), SERPINE1
(cat. nos. SI00012628 and SI03039715), CDH2 (cat. nos. SI02757335
and SI04434619) and CA9 (cat. nos. SI00023541 and SI00023534), or a
non-targeting siRNA with no homology to any known human gene
(AllStars Negative Control siRNA; cat. no. 1027280) with HiPerFect
transfection reagent (Qiagen GmbH). The list of siRNA target
sequences is provided in Table
SI. A total of two siRNA clones to gene of interest in 1:1
ratio were used for each transfection, except for EREG siRNA. The
UT-SCC-14 cell line was transfected with the HIF-1α, CA9, SERPINE1,
AREG and EREG siRNA. The final siRNA concentration in the culture
medium was 10 nmol/l. At 24 h after transfection, the cells were
sparsely seeded into 12-well plates (Falcon; Corning Life
Sciences). After another 24 h, half of the cultures were moved to
1% O2 and the rest of the cultures were cultured under
standard conditions (20% O2); after an additional 24 h,
the cells were irradiated (2, 4 or 6 Gy). The treated cells were
grown for another 9 days, after which they were fixed and stained
with crystal violet as aforementioned. Knockdown was verified via
RT-qPCR. A reduction in the mRNA level by ≥70% was achieved in all
experiments.
Microarray analysis
Three HNSCC cell lines (UT-SCC-14, LK0859 and
LK0863) were selected for the microarray analysis. Cells were
cultured to 70–80% confluence and exposed to either normoxic (21%
O2) or hypoxic (1% O2) conditions for 24 h
prior to total RNA extraction. The independently derived cell lines
were considered as biological replicates for each experimental
condition. Each experimental condition included a single
hybridization of the same sample in our microarray design. Total
RNA was extracted from the HNSCC cell lines cultured in normoxic
and hypoxic conditions using the RNeasy Mini kit and quantified
using a Nanodrop ND-100 Spectrophotometer (Thermo Fisher
Scientific, Inc.). Total RNA (150 ng) was used to process the
Affymetrix GeneChip® Human Transcriptome 2.0 arrays
(Affymetrix; Thermo Fisher Scientific, Inc.) using a GeneChip WT
Plus Reagent kit (Thermo Fisher Scientific, Inc.) according to
manufacturer's instructions. Hybridized arrays were scanned with an
Affymetrix GeneChip 3000 fluorescent scanner (Affymetrix; Thermo
Fisher Scientific, Inc.).
Robust multiarray analysis was used for
normalization of the microarray data. The raw expression data was
log2 transformed, and the probes with a variance <0.1 were
filtered out before statistical analyses. Differential expression
analyses were performed using the Limma package (version 3.38.3) in
R (version 3.5) using a pairwise comparison between normoxia (n=3)
vs. hypoxia (n=3) conditions. A P-value adjustment was performed
using the Benjamini-Hochberg procedure (27,28).
The P<0.05 and minimum log2 fold change of 2 from the analyzed
data set (normoxia vs. hypoxia) was used to identify
hypoxia-associated genes. Pathway analysis was performed using
Ingenuity Pathways Analysis (IPA) software (build version, 486617M;
content version, 46901286; Qiagen Inc.; http://www.qiagenbioinformatics.com/products/ingenuity-pathway-analysis).
Only significantly (adjusted P<0.05) upregulated and
downregulated genes were considered for IPA-supported analysis.
Statistical analysis
All values obtained were represented as the mean ±
SD of at least three independent experiments. Statistical analysis
was performed with GraphPad Prism software v. 7.0 (GraphPad
Software, Inc.). An unpaired Student's t-test or one-way ANOVA,
followed by a Bonferroni post hoc test was used to analyze the
data. P<0.05 was considered to indicate a statistically
significant difference.
Results
Hypoxia-induced changes in treatment
response
Our previous study demonstrated that hypoxia was
associated with the enhanced survival of HNSCC cells in response to
different treatments (29). To
further evaluate the impact of hypoxia on radiation treatment,
three HNSCC cell lines (UT-SCC-14, LK0858 and LK0863) were cultured
in the presence of 21% O2 (normoxia) or 1% O2
(hypoxia), followed by exposure of the cells to various doses of
ionizing γ-irradiation (2, 4 and 6 Gy). To ensure the presence of
hypoxic conditions during the experimental settings, the expression
of two hypoxia-associated proteins, namely, HIF-1α and CA9, were
assessed using western blotting (Fig.
1A). The cells cultured in the hypoxic conditions exhibited an
increased survival capacity in response to irradiation (Fig. 1B). Furthermore, siRNA-mediated
downregulation of HIF-1α was not associated with sensitization of
hypoxic HNSCC cells towards radiation treatment (Fig. 1C).
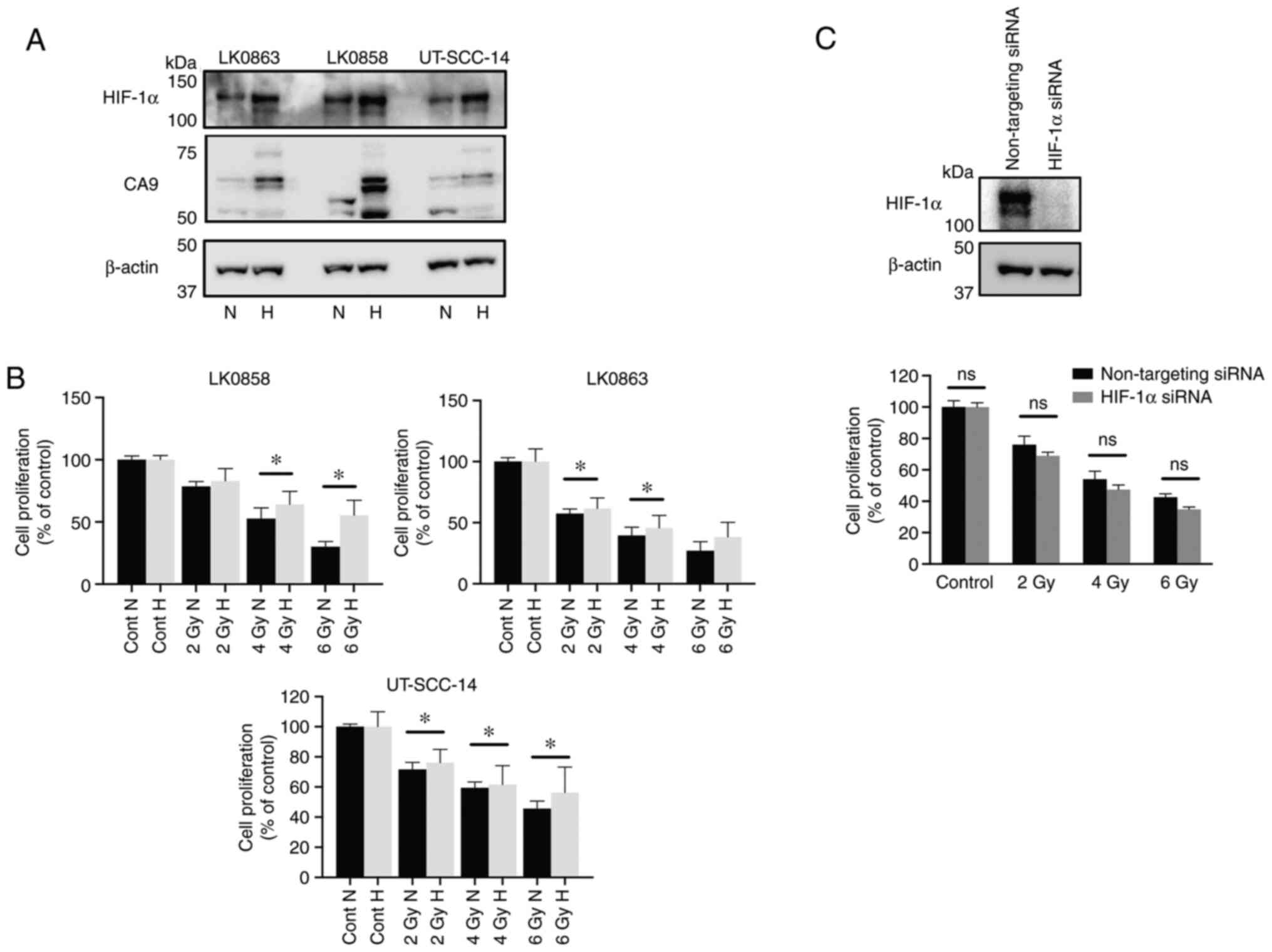 | Figure 1.Hypoxia-induced response to
radiotherapy in three head and neck squamous cell carcinoma cell
lines. The LK0858, LK0863 and UT-SCC-14 cell lines were cultured in
normoxic (21% O2) and hypoxic (1% O2)
conditions. (A) Western blot analysis of HIF-1α and CA9 expression
in cell lines cultured in normoxic and hypoxic conditions. β-actin
was used as the loading control. (B) LK0858, LK0863 and UT-SCC-14
cells were irradiated (2, 4 and 6 Gy) at 48 h after seeding and
subsequently returned to normoxic or hypoxic conditions. The
hypoxic cells were exposed to hypoxia for 24 h prior to
irradiation. After 9 days, the cytotoxic/cytostatic effect on cell
proliferation was determined by a crystal violet assay. (C)
UT-SCC-14 cells transiently transfected with either non-targeting
siRNA or HIF-1α-targeting siRNA were exposed to hypoxia for 24 h
prior to transfection and placed back under hypoxic conditions
after transfection. The efficiency of HIF-1α downregulation was
assessed via western blotting at 48 h post-transfection. In
parallel experimental settings, the UT-SCC-14 cells were irradiated
at 2, 4 and 6 Gy 24 h post-transfection with the respective siRNAs,
followed by re-exposure to hypoxic conditions (1% O2).
After 9 days, the cytotoxic/cytostatic effect on cell proliferation
was determined by a crystal violet assay. Cell proliferation is
presented as the percentage of the untreated controls, and data are
presented as the mean ± SD from three experiments performed in
triplicate. *P<0.05. The data was analyzed using an
unpaired Student's t-test. HIF, hypoxia-inducible factor; CA9,
carbonic anhydrase 9; siRNA, small interfering RNA; N, normoxia; H,
hypoxia; ns, not significant. |
Hypoxia-induced changes in EMT- and
cancer stem cell (CSC)-associated genes
The role of hypoxia in the acquisition of EMT and
cancer stemness as a leading cause of metastasis has been suggested
(19). To investigate this, the
mRNA expression levels of various EMT-associated genes and
CSC-associated genes in hypoxic HNSCC cell lines compared with
cells cultured in normoxic conditions were analyzed via
RT-qPCR.
As presented in Table
I, hypoxia induced significantly increased levels of
EMT-associated CDH2 (UT-SCC-14 and LK0863), vimentin
(UT-SCC-14, LK0858 and LK0863) and fibronectin 1 (FN1;
LK0858 and LK0863). In addition, the CDH1 mRNA expression
level was significantly decreased in UT-SCC-14 in response to
hypoxia, whereas no significant changes were observed in LK0858 or
LK0863 cells. Moreover, hypoxia induced increases in CSC-associated
gene expression in all analyzed cell lines, with significantly
elevated mRNA levels of SOX2 and NANOG transcription
factors.
 | Table I.Relative mRNA expression of
epithelial-mesenchymal transition and cancer stem cell markers in
hypoxic head and neck squamous cell carcinoma cells. |
Table I.
Relative mRNA expression of
epithelial-mesenchymal transition and cancer stem cell markers in
hypoxic head and neck squamous cell carcinoma cells.
|
| Cell line, mean ±
SD expression level |
|---|
|
|
|
|---|
| Gene | UT-SCC-14 | LK0858 | LK0863 |
|---|
| CDH1 |
0.3±0.08a | 1.5±0.19 | 0.9±0.29 |
| CDH2 |
8.5±2.08a | 0.9±0.12 |
3.8±1.06a |
| VIM | 6±2.11a |
2.6±0.44a |
4.6±0.77a |
| FN1 | 1.5±0.25 |
2.4±0.47a |
2.6±0.28a |
| FOXC2 | 1.8±0.53 | 0.7±0.10 | 1±0.39 |
| TWIST1 | 2.5±1.11 | 2.3±0.19 | 1.4±0.23 |
| CD44 | 1.4±0.28 | 0.7±0.08 | 0.7±0.12 |
| SOX2 |
6.8±0.98a | 0.8±0.05 |
2.7±0.35a |
| NANOG |
7.2±0.77a |
4.7±0.75a |
3.2±0.14a |
Of note, silencing of CDH2 with siRNA did not affect
the proliferation of hypoxic HNSCC cells in response to ionizing
γ-irradiation (Fig. S1).
Microarray analysis of
hypoxia-regulated gene expression in HNSCC cell lines
To identify hypoxia-regulated genes, HNSCC cells
cultured in normoxic and hypoxic conditions were analyzed with an
mRNA microarray. The expression profiles generated from the HNSCC
cells (UT-SCC-14, LK0859 and LK0863) cultured in hypoxic conditions
were compared with the HNSCC cells cultured in normoxic conditions.
Following normalization and variance filtering, 20,778 probes were
investigated for further statistical analyses. A total of 71 genes
were found to be upregulated and 147 were found to be downregulated
by hypoxia compared with normoxia (adjusted P<0.05; ±log2 fold
change >2). The profile of hypoxia-regulated gene expression in
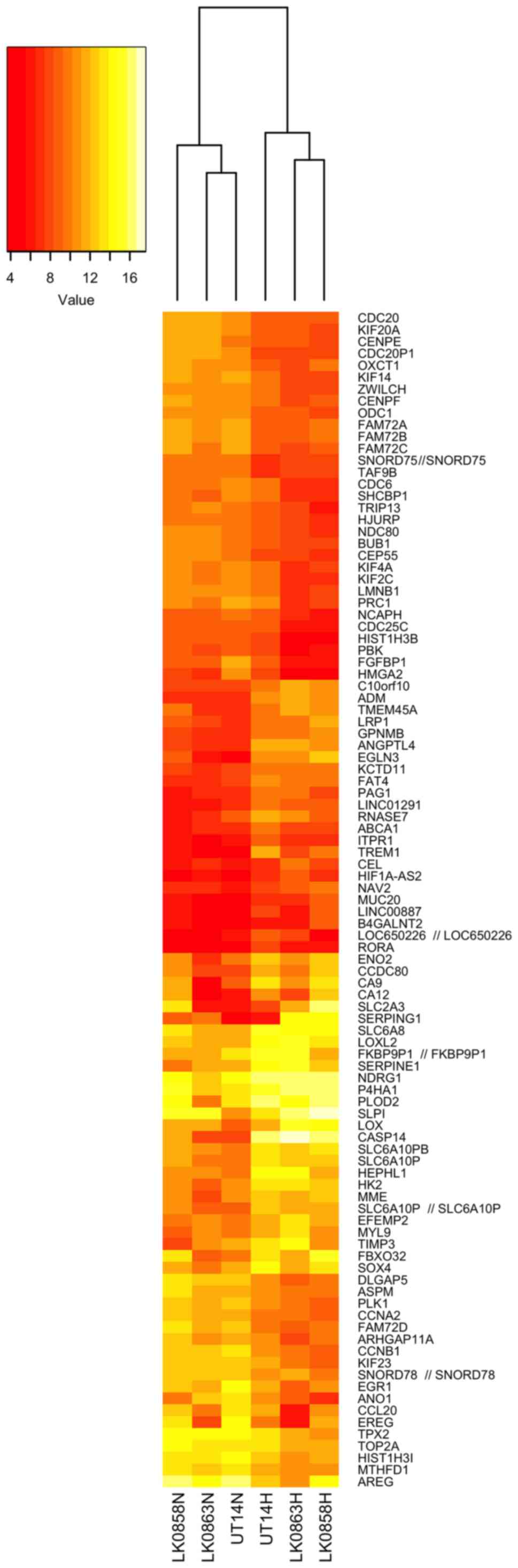
the three HNSCC cell lines is shown in Fig. 2.
The genes highly upregulated by hypoxia included
CASP14, EGLN3, TREM1, CA9, ANGPTL4, SERPING1, ADM, LOX, SLC2A3
(GLUT3) and SERPINE1. The genes that were the most
hypoxia-repressed included AREG, FAM72C/FAM72D, EREG, CCNB1,
ANO1, FGFBP1, HIST1H3B, PLK1, CDC20 and KIF14 (Table II).
 | Table II.Top significantly dysregulated
associated genesa. |
Table II.
Top significantly dysregulated
associated genesa.
| A, Upregulated
genes |
|---|
| Gene symbol | Full gene name | Log2 fold
change |
|---|
| CASP14 | Caspase 14 | 7.372 |
| EGLN3 | Egl-9 family
hypoxia-inducible factor 3 | 5.029 |
| TREM1 | Triggering receptor
expressed on myeloid cells 1 | 4.323 |
| CA9 | Carbonic anhydrase
9 | 4.267 |
| ANGPTL4 | Angiopoietin like
4 | 4.252 |
|
SERPING1 | Serpin family G
member 1 | 3.975 |
| ADM | Adrenomedullin | 3.856 |
| LOX | Lysyl oxidase | 3.362 |
| SLC2A3
(GLUT3) | Solute carrier
family 2 member 3 | 3.321 |
|
SERPINE1 | Serpin family E
member 1 | 3.054 |
|
| B, Downregulated
genes |
|
| Gene
symbol | Full gene
name | Log2 fold
change |
|
| AREG | Amphiregulin | −3.255 |
|
FAM72C/FAM72D | Family with
sequence similarity 72 member C/D | −3.252 |
| EREG | Epiregulin | −3.044 |
| CCNB1 | Cyclin B1 | −2.975 |
| ANO1 | Anoctamin 1 | −2.974 |
| FGFBP1 | Fibroblast growth
factor binding protein 1 | −2.906 |
|
HIST1H3B | Histone cluster 1,
H3b | −2.740 |
| PLK1 | Polo like kinase
1 | −2.671 |
| CDC20 | Cell division cycle
20 | −2.626 |
| KIF14 | Kinesin family
member 14 | −2.602 |
A number of hypoxia-responsive genes from microarray
analysis were selected for validation with RT-qPCR. The cumulative
mRNA data analysis (LK0858, LK0863 and UT-SCC-14 cells) confirmed
upregulation of SERPINE1, CA9, CASP14, LOX and SLC2A3
(GLUT3), as well as downregulation of AREG and
EREG (Fig. 3). The
statistically significant deregulation of hypoxia-induced genes was
found with CA9, CASP14 and EREG.
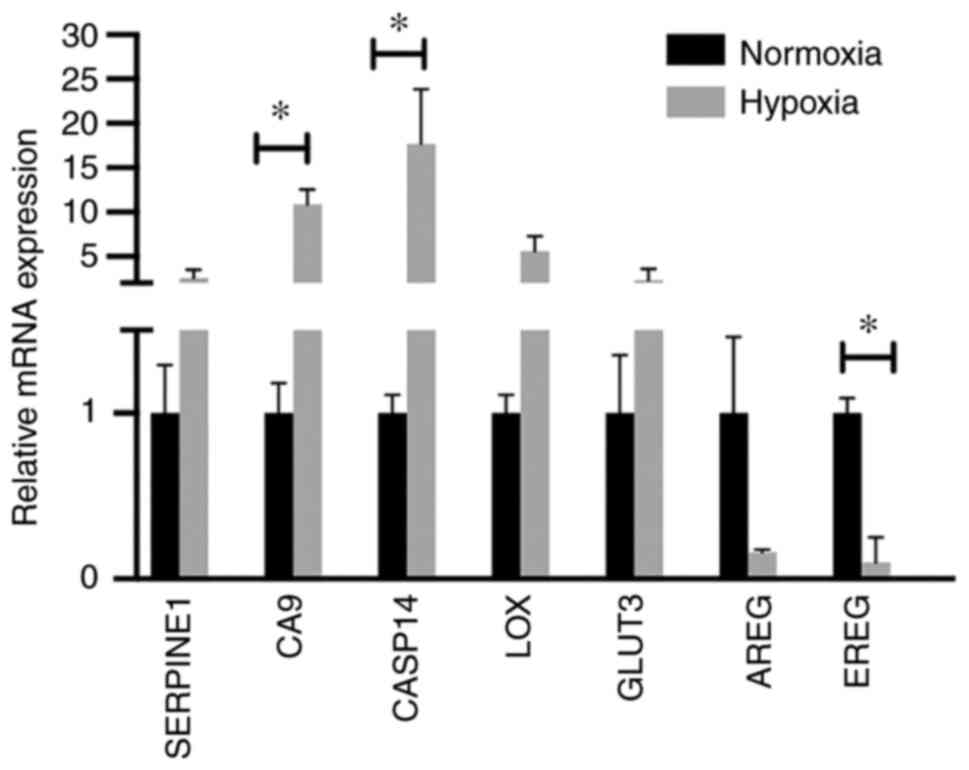 | Figure 3.Validation of microarray analysis.
Reverse transcription-quantitative PCR analysis was used to analyze
the mRNA expression levels of hypoxia-responsive genes in the
LK0858, LK0863 and UT-SCC-14 cells using microarray. The relative
amounts of SERPINE1, CA9, CASP14, LOX, GLUT3, AREG and
EREG mRNA was calculated using the 2−ΔΔCq method
and amplification of both GAPDH and β-actin were used as an
internal standard. The cumulative, relative mRNA levels from the
LK0858, LK0863 and UT-SCC-14 cells are presented as the mean ± SD
relative to cells cultured in normoxia. *P<0.05. The data was
analyzed using an unpaired Student's t-test. SERPINE1, serpin
family E 1; CA9, carbonic anhydrase 9; CASP14, caspase 14; LOX,
lysyl oxidase; GLUT3, glucose transporter 3; AREG, amphiregulin;
EREG, epiregulin. |
With the use of IPA, the top networks assembled from
the hypoxia-regulated genes were cellular growth and proliferation,
cell cycle, cellular assembly and organization, cancer, cell death
and survival, and cellular function and maintenance (Table III). The top regulator effect
network with a consistency score of 22.15 consisted of 14 upstream
regulators (CSF2, E2F2, E3F3, EP400, INSR, JAK2, LIN9, MED1,
NTRK2, TBX2, RARA, RBL2 and RB) that targeted a number
of differentially expressed genes under hypoxic conditions in the
HNSCC cell lines. The connected downstream functions included cell
proliferation of carcinoma cell lines, metabolism of DNA and
segregation of chromosomes, among others (Fig. S2).
 | Table III.Top networks generated by Ingenuity
IPA software. |
Table III.
Top networks generated by Ingenuity
IPA software.
| Score | Focus genes | Associated network
functions |
|---|
| 49 |
ABCE1a, BLMHa, BZW2a, CDCA7a, CENPEa, CENPFa, CST6b, CTPS1a, DCTPP1a, DCXRa, DIMT1a, IDH3Ba, MRPL24a, MTFR2a, MYCa, NUF2a, PAICSa, PLBD2b, PSMA3a, PSMB2a, PSMB3a, RBMS3b, SLC25A19a, SRMa, STX3b, ZWINTa | Cellular growth and
proliferation, cell cycle |
| 41 |
AIFM1a, C1QBPa, CASP14b, COQ2a, COQ3a, DNASE2b, GARTa, GPNMBb, GSSa, MBOAT1a, MCCC2a, NDUFA9a, NDUFAF1a NDUFV1a, NUAK1b, OXCT1a, PPATa, SIGMAR1a, STOML2a, TUFMa, UCA1b | Cellular assembly
and organization, nucleic acid metabolism |
| 37 |
AAASa, BAG2a, BTG1b, BUB1a, CCT2a, CCT3a, CCT5a, CCT7a, CDC20a, CLCA2b, DKC1a, ELAC2a, INCENPa, NDC1a, NUP88a, NUP93a, PCGF5b, PFKFB3b, PRMT3a, RAB8Bb, SEH1La, TSC22D2b, VLDLRb, WDR77a | Cellular assembly
and organization, molecular transport, RNA trafficking |
| 19 |
ALDH1A3a, CA9b, CHRNA5a, DHX33a, ETS2b, GSRa, HAT1a, HIST1H2BJa, HIST1H3Ba, HMGA2a, PNPT1a, PPP1R13Lb, SAAL1a, SLPIb, TIMP3b, WSB1b, ZNF160b | Cancer, cell death
and survival, cellular function and maintenance |
| 19 |
ABCC4a, ASPMa, FBXO32b, FHa, FNIP1b, GCLCa, KRT18a, NQO1a, PDHBa, SERPINE1b, SLC25A10a, SLC6A8b, TALDO1a, TP53INP1b | Cancer, cell death
and survival, organismal injury and abnormalities |
Effect of microarray-revealed target
genes' downregulation on the radiotherapy response
As radiation resistance is associated with hypoxia,
whether the highly upregulated genes in hypoxia, namely, CA9
and SERPINE1, affected radioresistance in HNSCC cells was
explored. The results showed that silencing CA9 or
SERPINE1 with siRNA did not sensitize hypoxic HNSCC cells to
ionizing γ-irradiation (Fig. 4A and
B).
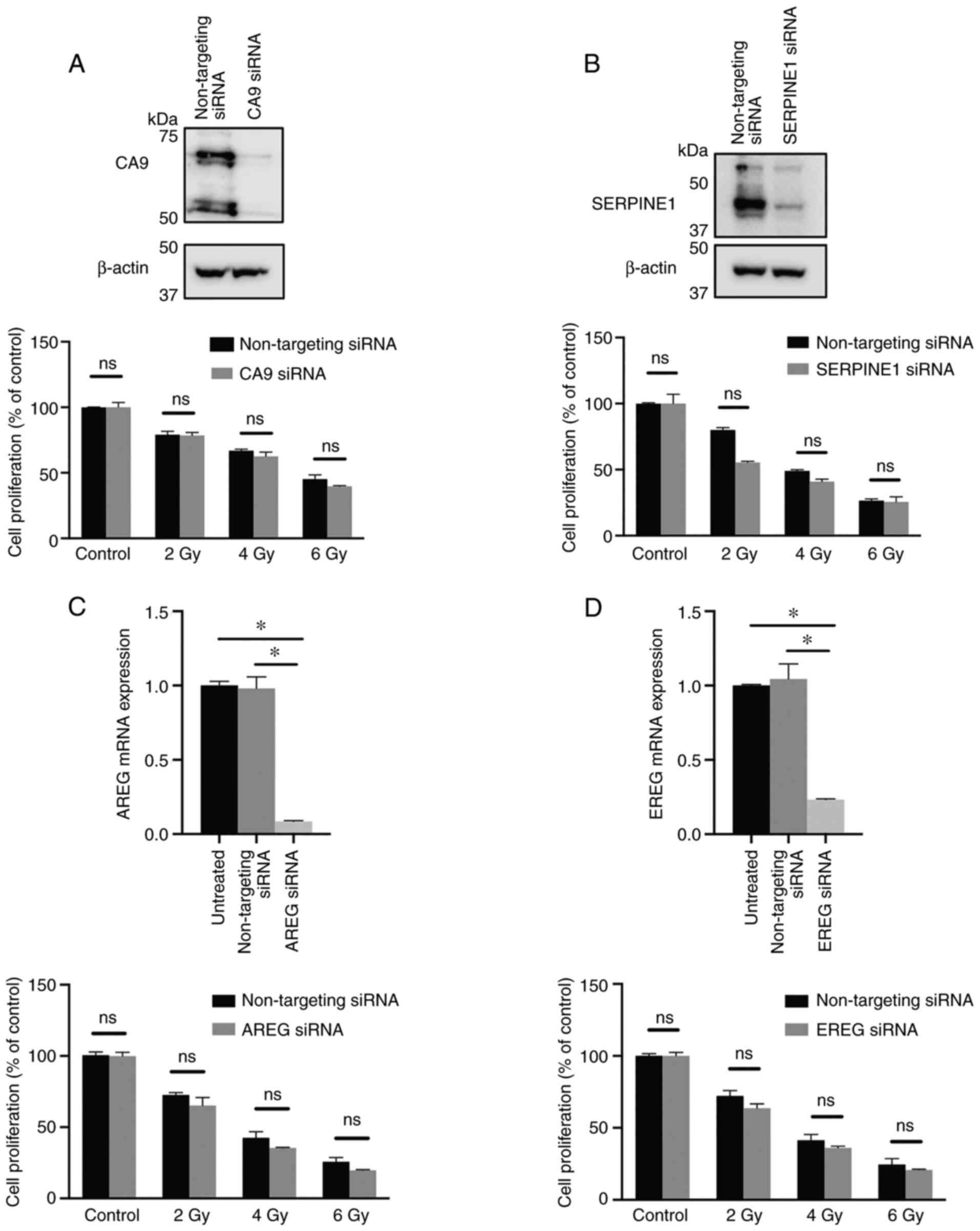 | Figure 4.Effect of CA9, SERPINE1, AREG and
EREG downregulation on the radiation response in head and neck
squamous cell carcinoma cells. UT-SCC-14 cells were transiently
transfected with either non-targeting siRNA or siRNA interfering
with expression of (A) CA9, (B) SERPINE1, (C) AREG and (D) EREG.
Cells transfected with either non-targeting siRNA or siRNA
targeting CA9 or SERPNE1 were exposed to hypoxia for 24 h prior
transfection and placed back under hypoxic (1% O2)
conditions after transfection. The protein expression was evaluated
48 h post-transfection using western blotting. β-actin was used as
the loading control. In parallel experimental settings, cells
transfected with CA9 or SERPINE1 siRNA were irradiated at 2, 4 or 6
Gy 24 h post-transfection, followed by re-exposure to hypoxic
conditions, whereas cells transfected with AREG or EREG siRNA were
exposed to normoxic conditions (21% O2) after
irradiation. The efficiency of AREG and EREG downregulation with
specific siRNA was assessed via reverse transcription-quantitative
PCR. After 9 days, the cytotoxic/cytostatic effect on cell
proliferation was determined by a crystal violet assay. Cell
proliferation is presented as the percentage of the untreated
controls, and the data are presented as the mean ± SD from three
experiments performed in triplicate. All results are shown as the
mean ± SD. *P<0.05 vs. non-targeting siRNA. The data was
analyzed either using one-way ANOVA followed by Bonferroni's
multiple comparison test or an unpaired Student's t-test. SERPINE1,
serpin family E 1; CA9, carbonic anhydrase 9; AREG, amphiregulin;
EREG, epiregulin; siRNA, small interfering RNA; ns, not
significant. |
To further explore an association between
hypoxia-mediated downregulation of genes and radiotherapy response,
the study subsequently focused on the two genes notably repressed
by hypoxia: AREG and EREG. UT-SCC-14 cells were
transfected with AREG or EREG siRNA prior to culture under normoxic
conditions, as the expression of these genes was downregulated
under hypoxic conditions. Significant changes in the proliferation
rate of HNSCC cells in response to ionizing γ-irradiation upon AREG
or EREG targeting was not observed when compared with cells
transfected with non-targeting siRNA (Fig. 4C and D). This suggested that the
hypoxia-mediated downregulation of AREG and EREG expression did not
have a direct effect on the response of HNSCC cells to radiation
therapy.
Discussion
Several studies have indicated that hypoxia is
associated with cancer progression and the development of
resistance to chemotherapy and radiotherapy (30–32).
Oxygenation status of head and neck tumors appears to be important,
as hypoxic tumors exhibit a poorer response to surgery and
radiotherapy (33,34). Identification of biomarkers for
hypoxia possessing predictive parameters is essential and targeting
tumor hypoxia could therefore improve the response to radiotherapy
in HNSCC. HIF-1α is a well-recognized member of the HIF family that
mediates a cellular response to hypoxia, and numerous reports have
identified HIF-1α as a leading factor promoting radioresistance in
tumor cells (35,36). The present study observed a link
between hypoxia and the response of HNSCC cells to radiation
treatment; however, in the present experimental settings, the
increased survival capacity of hypoxic HNSCC cells compared with
cells cultured in normoxic conditions after radiation did not
appear to be HIF-1α-dependent. Thus, targeting hypoxia via
preirradiation inhibition of HIF-1α in order to improve the
response of HNSCC to radiotherapy may not be sufficient.
Furthermore, findings by Harada et al (37) suggested that the timing of HIF-1α
inhibition determined whether a HIF-1 inhibitor can suppress or
enhance the effect of radiation treatment, with the authors
highlighting the therapeutic significance of inhibiting HIF-1α
action after radiation treatment. Hypoxia is known to modulate
radiation response and may have an effect on the expression of
hypoxia-responsive genes, particularly HIF-1α. There are reports
showing a slight increase in HIF-1α after irradiation (38,39).
In the current experimental design, the impact of downregulation of
selected hypoxia-associated genes on radiotherapy response was
explored via siRNA-mediately silencing prior to exposure to hypoxia
and irradiation. The siRNA-mediated downregulation was validated
via RT-qPCR and western blotting prior to irradiation. However,
irradiation-induced increases in HIF-1α expression as a response to
oxidative stress cannot be excluded.
It is widely hypothesized in cancer research that
hypoxia triggers the EMT, which has been associated with metastasis
in cancer, including HNSCC (19,40).
There are a number of established EMT-associated markers, and their
role in tumorigenesis has been well described. Our previous study
reported that the EMT marker FN1 can serve as a biomarker
for intrinsic radiosensitivity in HNSCC (41). As hypoxia and HIF-1α signaling
drives the EMT, the role of CDH2 in the hypoxia-mediated response
to radiation treatment was investigated. CDH2 is one of the
EMT-associated markers that promotes tumor cell survival, migration
and invasion, and high levels are often associated with a poor
prognosis (40,42). However, sensitization of hypoxic
HNSCC cells towards radiation was not observed upon the silencing
of CDH2 with siRNA. This suggested that CDH2, one of numerous
important players in such a dynamic process as EMT, cannot be
considered as a determinant of the radiation response upon hypoxia
treatment.
Next, the effect of hypoxia on global gene
expression in the HNSCC cell lines was investigated via microarray
analysis. Implementation of a hypoxia-associated gene expression
signature in predicting the radiotherapy response is desirable in
HNSCC treatment (43–45). Similar to other studies regarding
tumor hypoxia, the commonly accepted marker for hypoxic cells,
CA9, was also found to be highly upregulated in the present
experimental setting (46–48). Among the 71 upregulated genes
observed in hypoxic HNSCC cell lines, CASP14, EGLN3, TREM1, CA9,
ANGPTL4, SERPING1, ADM, LOX, SLC2A3 (GLUT3) and SERPINE1
were most prominent. CASP14 has been most extensively
described in breast cancer, where its high expression is a marker
of breast cancer aggressiveness in association with proliferation
and cancer stemness (49).
Induction of ADM by hypoxia in turn is associated with
increased invasiveness of pancreatic cancer cells and may influence
angiogenesis (50), but its role
in hypoxic HNSCC cells has not been described. Moreover, hypoxia
signaling also controls the establishment of the premetastatic
niche, for which LOX activity is indispensable (51,52).
Increased expression of hypoxia-regulated
GLUT3 has been observed in different types of tumors,
including head and neck cancer. GLUT1 and GLUT3 protein expression
is associated with a poor prognosis in oral squamous cell
carcinoma, most likely due to the increased glycolytic metabolism
of more aggressive cancer cells (53). CA9 is highly expressed in response
to hypoxia and is very often associated with increased invasiveness
of tumors (54). However, the
prognostic and predictive role of CA9 in radiotherapy of HNSCC is
quite inconclusive (55–57). Moreover, dysregulation of SERPINE1
has been linked to the activation of hypoxia-related factors and
radiation resistance in head and neck cancer (58–60).
As CA9 and SERPINE1 were upregulated in all analyzed
HNSCC cell lines when cultured under hypoxic conditions, it was
hypothesized that they had a direct impact on the radiosensitivity
of HNSCC cells. However, subsequent experiments did not support
this hypothesis, as the targeting of CA9 and SERPINE1
with siRNA did not lead to resensitization of hypoxic HNSCC cells
to irradiation when compared with cells transfected with
non-targeting siRNA. Taking into consideration the number of
upregulated genes under hypoxic conditions and the complexity of
the hypoxia-mediated radioresistance, it is proposed that a panel
of hypoxia-responsive genes should be considered as a predictive
factor of radiotherapy in HNSCC.
Among the genes repressed by hypoxia, AREG
and EREG have been reported to serve a role in cancer
progression; AREG and EREG act as ligands of EGFR, and their
tumorigenic action and implication in treatment resistance have
been already reported (61,62).
Our previous study reported that the expression of AREG and EREG
influences the treatment (cetuximab and cisplatin) sensitivity of
HNSCC cells and may be useful as predictive markers (25). For functional analyses, AREG and
EREG downregulation with siRNA under normoxic conditions was
performed in order to assess their role in mediating
radioresistance in HNSCC cells. The results showed that
hypoxia-mediated downregulation of AREG and EREG
expression was not a determinant of radiotherapy resistance in
HNSCC.
As AREG and EREG were downregulated in
hypoxia-exposed cells, their interactions with other signaling
molecules may be different than those in normoxic conditions. It
would be informative to test in the future if the forced expression
of these genes in hypoxia-exposed and irradiated cells affects
their survival. However, these genes cannot be excluded from the
panel of genes that affect the radiotherapy response under
hypoxia.
In conclusion, a number of hypoxia-regulated genes
were identified in HNSCC that may promote tumorigenesis and
radiotherapy resistance. In addition, the identified genes were
involved in multiple biological functions, such as the cell cycle,
DNA replication, cellular development, cellular growth and
proliferation. The hypoxia-associated gene pattern observed in the
present study, in combination with previous reports, may be useful
for outcome prediction in HNSCC.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Professor Reidar
Grénman (Department of Otorhinolaryngology, University of Turku,
Finland) for providing the UT-SCC-14 cell line and Dr Fredrik
Levander (National Bioinformatics Infrastructure, SciLifeLab, Lund
University, Sweden) for their assistance with the pre-processing of
the microarray data.
Funding
This study was supported by the Swedish Cancer Society (grant
no. 2017/301), the County Council of Östergötland, the Research
Funds of Linköping University Hospital and the Cancer Foundation of
Östergötland.
Availability of data and materials
The datasets generated and/or analyzed during the
current study (https://www.ncbi.nlm.gov/geo/query/acc.cgi?acc=GSE182734)
are available in the Gene Expression Omnibus repository.
Authors' contributions
EW and KR designed the study. EW, NM and KR
performed the experiments. EW, NM, AA and KR analyzed the data. EW,
NM and KR wrote the manuscript. All authors have read and approved
the final manuscript. EW, NM and KR confirm the authenticity of all
the raw data.
Ethics approval and consent to
participate
The present study was approved by the Research
Ethics Committee of the Linköping University (approval no. 03-537).
Written consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Saman DM: A review of the epidemiology of
oral and pharyngeal carcinoma: Update. Head Neck Oncol. 4:12012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Koukourakis G, Kouloulias V, Koukourakis
M, Kouvaris J, Zacharias G and Gouliamos A: The efficacy of
combined treatment with cetuximab (erbitux) and radiation therapy
in patients with head and neck cancer. J BUON. 14:19–25.
2009.PubMed/NCBI
|
|
4
|
Pulte D and Brenner H: Changes in survival
in head and neck cancers in the late 20th and early 21st century: A
period analysis. Oncologist. 15:994–1001. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Alsahafi E, Begg K, Amelio I, Raulf N,
Lucarelli P, Sauter T and Tavassoli M: Clinical update on head and
neck cancer: Molecular biology and ongoing challenges. Cell Death
Dis. 10:5402019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bredell MG, Ernst J, El-Kochairi I, Dahlem
Y, Ikenberg K and Schumann DM: Current relevance of hypoxia in head
and neck cancer. Oncotarget. 7:50781–50804. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lv X, Li J, Zhang C, Hu T, Li S, He S, Yan
H, Tan Y, Lei M, Wen M and Zuo J: The role of hypoxia-inducible
factors in tumor angiogenesis and cell metabolism. Genes Dis.
4:19–24. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Salceda S and Caro J: Hypoxia-inducible
factor 1alpha (HIF-1alpha) protein is rapidly degraded by the
ubiquitin-proteasome system under normoxic conditions. Its
stabilization by hypoxia depends on redox-induced changes. J Biol
Chem. 272:22642–22647. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Semenza GL: Defining the role of
hypoxia-inducible factor 1 in cancer biology and therapeutics.
Oncogene. 29:625–634. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Koukourakis MI, Giatromanolaki A, Sivridis
E, Simopoulos C, Turley H, Talks K, Gatter KC and Harris AL:
Hypoxia-inducible factor (HIF1A and HIF2A), angiogenesis, and
chemoradiotherapy outcome of squamous cell head-and-neck cancer.
Int J Radiat Oncol Biol Phys. 53:1192–1202. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Al Tameemi W, Dale TP, Al-Jumaily RMK and
Forsyth NR: Hypoxia-modified cancer cell metabolism. Front Cell Dev
Biol. 7:42019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Luoto KR, Kumareswaran R and Bristow RG:
Tumor hypoxia as a driving force in genetic instability. Genome
Integr. 4:52013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rankin EB, Nam JM and Giaccia AJ: Hypoxia:
Signaling the metastatic cascade. Trends Cancer. 2:295–304. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Erler JT, Bennewith KL, Nicolau M,
Dornhöfer N, Kong C, Le QT, Chi JT, Jeffrey SS and Giaccia AJ:
Lysyl oxidase is essential for hypoxia-induced metastasis. Nature.
440:1222–1226. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Burroughs SK, Kaluz S, Wang D, Wang K, Van
Meir EG and Wang B: Hypoxia inducible factor pathway inhibitors as
anticancer therapeutics. Future Med Chem. 5:553–572. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Perri F, Pacelli R, Della Vittoria
Scarpati G, Cella L, Giuliano M, Caponigro F and Pepe S:
Radioresistance in head and neck squamous cell carcinoma:
Biological bases and therapeutic implications. Head Neck.
37:763–770. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Higgins GS, O'Cathail SM, Muschel RJ and
McKenna WG: Drug radiotherapy combinations: Review of previous
failures and reasons for future optimism. Cancer Treat Rev.
41:105–113. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dudas J, Ladanyi A, Ingruber J,
Steinbichler TB and Riechelmann H: Epithelial to mesenchymal
transition: A mechanism that fuels cancer radio/chemoresistance.
Cells. 9:4282020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yeo CD, Kang N, Choi SY, Kim BN, Park CK,
Kim JW, Kim YK and Kim SJ: The role of hypoxia on the acquisition
of epithelial-mesenchymal transition and cancer stemness: A
possible link to epigenetic regulation. Korean J Intern Med.
32:589–599. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hsieh JC, Wang HM, Wu MH, Chang KP, Chang
PH, Liao CT and Liau CT: Review of emerging biomarkers in head and
neck squamous cell carcinoma in the era of immunotherapy and
targeted therapy. Head Neck. 41 (Suppl 1):S19–S45. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Swartz JE, Pothen AJ, Stegeman I, Willems
SM and Grolman W: Clinical implications of hypoxia biomarker
expression in head and neck squamous cell carcinoma: A systematic
review. Cancer Med. 4:1101–1116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Koukourakis MI, Giatromanolaki A, Sivridis
E, Simopoulos K, Pastorek J, Wykoff CC, Gatter KC and Harris AL:
Hypoxia-regulated carbonic anhydrase-9 (CA9) relates to poor
vascularization and resistance of squamous cell head and neck
cancer to chemoradiotherapy. Clin Cancer Res. 7:3399–3403.
2001.PubMed/NCBI
|
|
23
|
Russo G, Zegar C and Giordano A:
Advantages and limitations of microarray technology in human
cancer. Oncogene. 22:6497–6507. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kurahashi I, Fujita Y, Arao T, Kurata T,
Koh Y, Sakai K, Matsumoto K, Tanioka M, Takeda K, Takiguchi Y, et
al: A microarray-based gene expression analysis to identify
diagnostic biomarkers for unknown primary cancer. PLoS One.
8:e632492013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jedlinski A, Ansell A, Johansson AC and
Roberg K: EGFR status and EGFR ligand expression influence the
treatment response of head and neck cancer cell lines. J Oral
Pathol Med. 42:26–36. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Irizarry RA, Hobbs B, Collin F,
Beazer-Barclay YD, Antonellis KJ, Scherf U and Speed TP:
Exploration, normalization, and summaries of high density
oligonucleotide array probe level data. Biostatistics. 4:249–264.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW,
Shi W and Smyth GK: Limma powers differential expression analyses
for RNA-sequencing and microarray studies. Nucleic Acids Res.
43:e472015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wiechec E, Hansson KT, Alexandersson L,
Jonsson JI and Roberg K: Hypoxia mediates differential response to
Anti-EGFR therapy in HNSCC cells. Int J Mol Sci. 18:9432017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Muz B, de la Puente P, Azab F and Azab AK:
The role of hypoxia in cancer progression, angiogenesis,
metastasis, and resistance to therapy. Hypoxia (Auckl). 3:83–92.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sorensen BS and Horsman MR: Tumor hypoxia:
Impact on radiation therapy and molecular pathways. Front Oncol.
10:5622020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sebestyen A, Kopper L, Danko T and Timar
J: Hypoxia signaling in cancer: From basics to clinical practice.
Pathol Oncol Res. 27:16098022021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Isa AY, Ward TH, West CM, Slevin NJ and
Homer JJ: Hypoxia in head and neck cancer. Br J Radiol. 79:791–798.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Swartz JE, Pothen AJ, van Kempen PM,
Stegeman I, Formsma FK, Cann EM, Willems SM and Grolman W: Poor
prognosis in human papillomavirus-positive oropharyngeal squamous
cell carcinomas that overexpress hypoxia inducible factor-1α. Head
Neck. 38:1338–1346. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Harada H: Hypoxia-inducible factor
1-mediated characteristic features of cancer cells for tumor
radioresistance. J Radiat Res. 57 (Suppl 1):i99–i105. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Moeller BJ and Dewhirst MW: HIF-1 and
tumour radiosensitivity. Br J Cancer. 95:1–5. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Harada H, Itasaka S, Zhu Y, Zeng L, Xie X,
Morinibu A, Shinomiya K and Hiraoka M: Treatment regimen determines
whether an HIF-1 inhibitor enhances or inhibits the effect of
radiation therapy. Br J Cancer. 100:747–757. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gu Q, He Y, Ji J, Yao Y, Shen W, Luo J,
Zhu W, Cao H, Geng Y, Xu J, et al: Hypoxia-inducible factor 1α
(HIF-1α) and reactive oxygen species (ROS) mediates
radiation-induced invasiveness through the SDF-1α/CXCR4 pathway in
non-small cell lung carcinoma cells. Oncotarget. 6:10893–10907.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moeller BJ, Cao Y, Li CY and Dewhirst MW:
Radiation activates HIF-1 to regulate vascular radiosensitivity in
tumors: Role of reoxygenation, free radicals, and stress granules.
Cancer Cell. 5:429–441. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Thierauf J, Veit JA and Hess J:
Epithelial-to-Mesenchymal transition in the pathogenesis and
therapy of head and neck cancer. Cancers (Basel). 9:762017.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jerhammar F, Ceder R, Garvin S, Grenman R,
Grafstrom RC and Roberg K: Fibronectin 1 is a potential biomarker
for radioresistance in head and neck squamous cell carcinoma.
Cancer Biol Ther. 10:1244–1251. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Derycke LD and Bracke ME: N-cadherin in
the spotlight of cell-cell adhesion, differentiation,
embryogenesis, invasion and signalling. Int J Dev Biol. 48:463–476.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Toustrup K, Sorensen BS, Alsner J and
Overgaard J: Hypoxia gene expression signatures as prognostic and
predictive markers in head and neck radiotherapy. Semin Radiat
Oncol. 22:119–127. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang L and West CM: Hypoxia gene
expression signatures as predictive biomarkers for personalising
radiotherapy. Br J Radiol. 92:201800362019.PubMed/NCBI
|
|
45
|
Toustrup K, Sørensen BS, Nordsmark M, Busk
M, Wiuf C, Alsner J and Overgaard J: Development of a hypoxia gene
expression classifier with predictive impact for hypoxic
modification of radiotherapy in head and neck cancer. Cancer Res.
71:5923–5931. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sung FL, Hui EP, Tao Q, Li H, Tsui NB, Lo
YM, Ma BB, To KF, Harris AL and Chan AT: Genome-wide expression
analysis using microarray identified complex signaling pathways
modulated by hypoxia in nasopharyngeal carcinoma. Cancer Lett.
253:74–88. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Beasley NJ, Wykoff CC, Watson PH, Leek R,
Turley H, Gatter K, Pastorek J, Cox GJ, Ratcliffe P and Harris AL:
Carbonic anhydrase IX, an endogenous hypoxia marker, expression in
head and neck squamous cell carcinoma and its relationship to
hypoxia, necrosis, and microvessel density. Cancer Res.
61:5262–5267. 2001.PubMed/NCBI
|
|
48
|
Wykoff CC, Beasley NJ, Watson PH, Turner
KJ, Pastorek J, Sibtain A, Wilson GD, Turley H, Talks KL, Maxwell
PH, et al: Hypoxia-inducible expression of tumor-associated
carbonic anhydrases. Cancer Res. 60:7075–7083. 2000.PubMed/NCBI
|
|
49
|
Handa T, Katayama A, Yokobori T, Yamane A,
Horiguchi J, Kawabata-Iwakawa R, Rokudai S, Bao P, Gombodorj N,
Altan B, et al: Caspase14 expression is associated with triple
negative phenotypes and cancer stem cell marker expression in
breast cancer patients. J Surg Oncol. 116:706–715. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Keleg S, Kayed H, Jiang X, Penzel R, Giese
T, Büchler MW, Friess H and Kleeff J: Adrenomedullin is induced by
hypoxia and enhances pancreatic cancer cell invasion. Int J Cancer.
121:21–32. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Erler JT, Bennewith KL, Cox TR, Lang G,
Bird D, Koong A, Le QT and Giaccia AJ: Hypoxia-induced lysyl
oxidase is a critical mediator of bone marrow cell recruitment to
form the premetastatic niche. Cancer Cell. 15:35–44. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu W, Shen SM, Zhao XY and Chen GQ:
Targeted genes and interacting proteins of hypoxia inducible
factor-1. Int J Biochem Mol Biol. 3:165–178. 2012.PubMed/NCBI
|
|
53
|
Ayala FR, Rocha RM, Carvalho KC, Carvalho
AL, da Cunha IW, Lourenço SV and Soares FA: GLUT1 and GLUT3 as
potential prognostic markers for oral squamous cell carcinoma.
Molecules. 15:2374–2387. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Benej M, Pastorekova S and Pastorek J:
Carbonic anhydrase IX: Regulation and role in cancer. Subcell
Biochem. 75:199–219. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kwon OJ, Park JJ, Ko GH, Seo JH, Jeong BK,
Kang KM, Woo SH, Kim JP, Hwa JS and Carey TE: HIF-1α and CA-IX as
predictors of locoregional control for determining the optimal
treatment modality for early-stage laryngeal carcinoma. Head Neck.
37:505–510. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hwa JS, Kwon OJ, Park JJ, Woo SH, Kim JP,
Ko GH, Seo JH and Kim RB: The prognostic value of
immunohistochemical markers for oral tongue squamous cell
carcinoma. Eur Arch Otorhinolaryngol. 272:2953–2959. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Eriksen JG and Overgaard J; Danish Head
and Neck Cancer Study Group (DAHANCA), . Lack of prognostic and
predictive value of CA IX in radiotherapy of squamous cell
carcinoma of the head and neck with known modifiable hypoxia: An
evaluation of the DAHANCA 5 study. Radiother Oncol. 83:383–388.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lee YC, Yu CC, Lan C, Lee CH, Lee HT, Kuo
YL, Wang PH and Chang WW: Plasminogen activator inhibitor-1 as
regulator of tumor-initiating cell properties in head and neck
cancers. Head Neck. 38 (Suppl 1):E895–E904. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Pavon MA, Arroyo-Solera I, Cespedes MV,
Casanova I, Leon X and Mangues R: uPA/uPAR and SERPINE1 in head and
neck cancer: Role in tumor resistance, metastasis, prognosis and
therapy. Oncotarget. 7:57351–57366. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Bayer C, Schilling D, Hoetzel J, Egermann
HP, Zips D, Yaromina A, Geurts-Moespot A, Sprague LD, Sweep F,
Baumann M, et al: PAI-1 levels predict response to fractionated
irradiation in 10 human squamous cell carcinoma lines of the head
and neck. Radiother Oncol. 86:361–368. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Busser B, Sancey L, Brambilla E, Coll JL
and Hurbin A: The multiple roles of amphiregulin in human cancer.
Biochim Biophys Acta. 1816:119–131. 2011.PubMed/NCBI
|
|
62
|
Cluckey A, Perino AC, Yunus FN, Leef GC,
Askari M, Heidenreich PA, Narayan SM, Wang PJ and Turakhia MP:
Efficacy of ablation lesion sets in addition to pulmonary vein
isolation for paroxysmal atrial fibrillation: Findings from the
SMASH-AF meta-analysis study cohort. J Am Heart Assoc.
8:e0099762019. View Article : Google Scholar : PubMed/NCBI
|