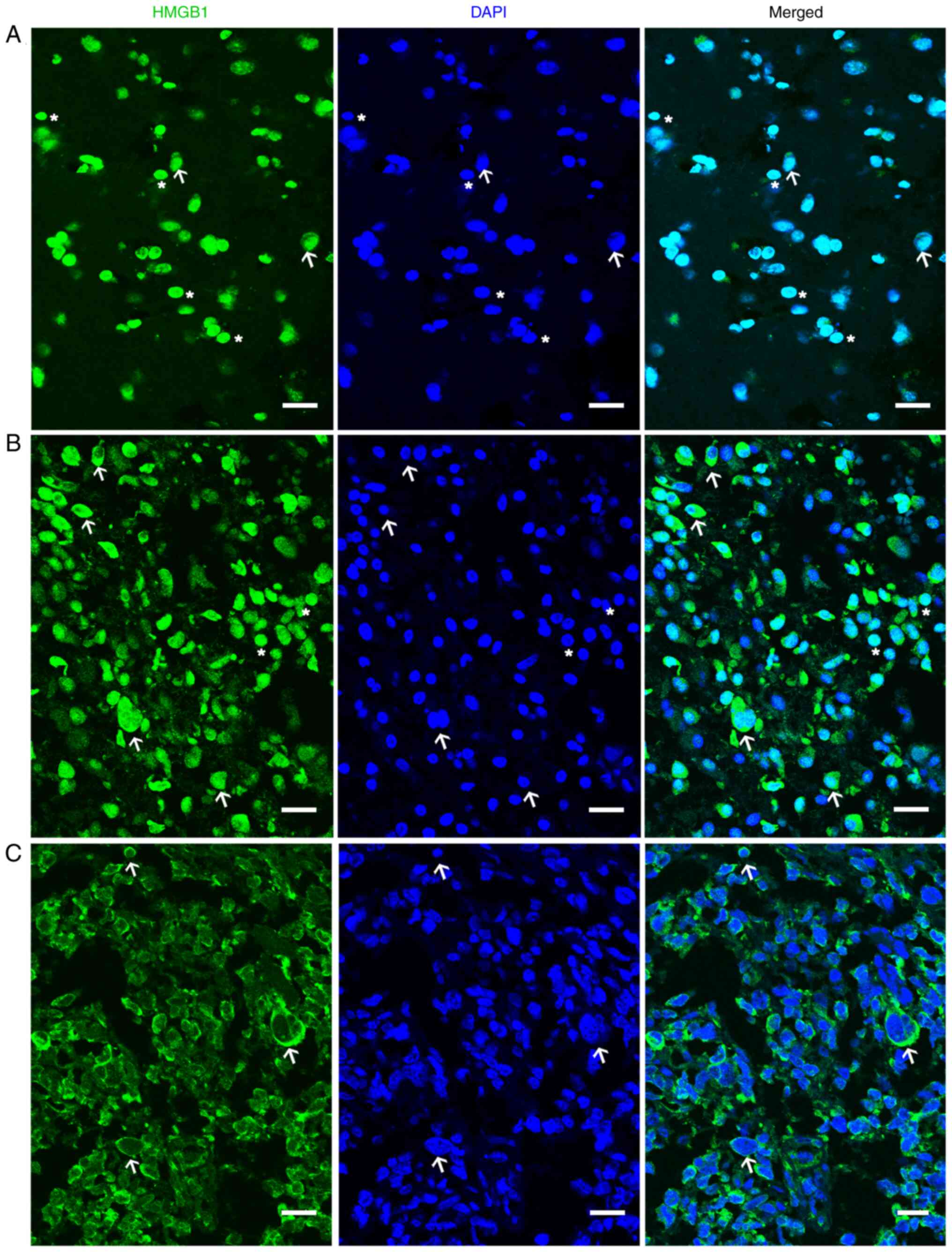
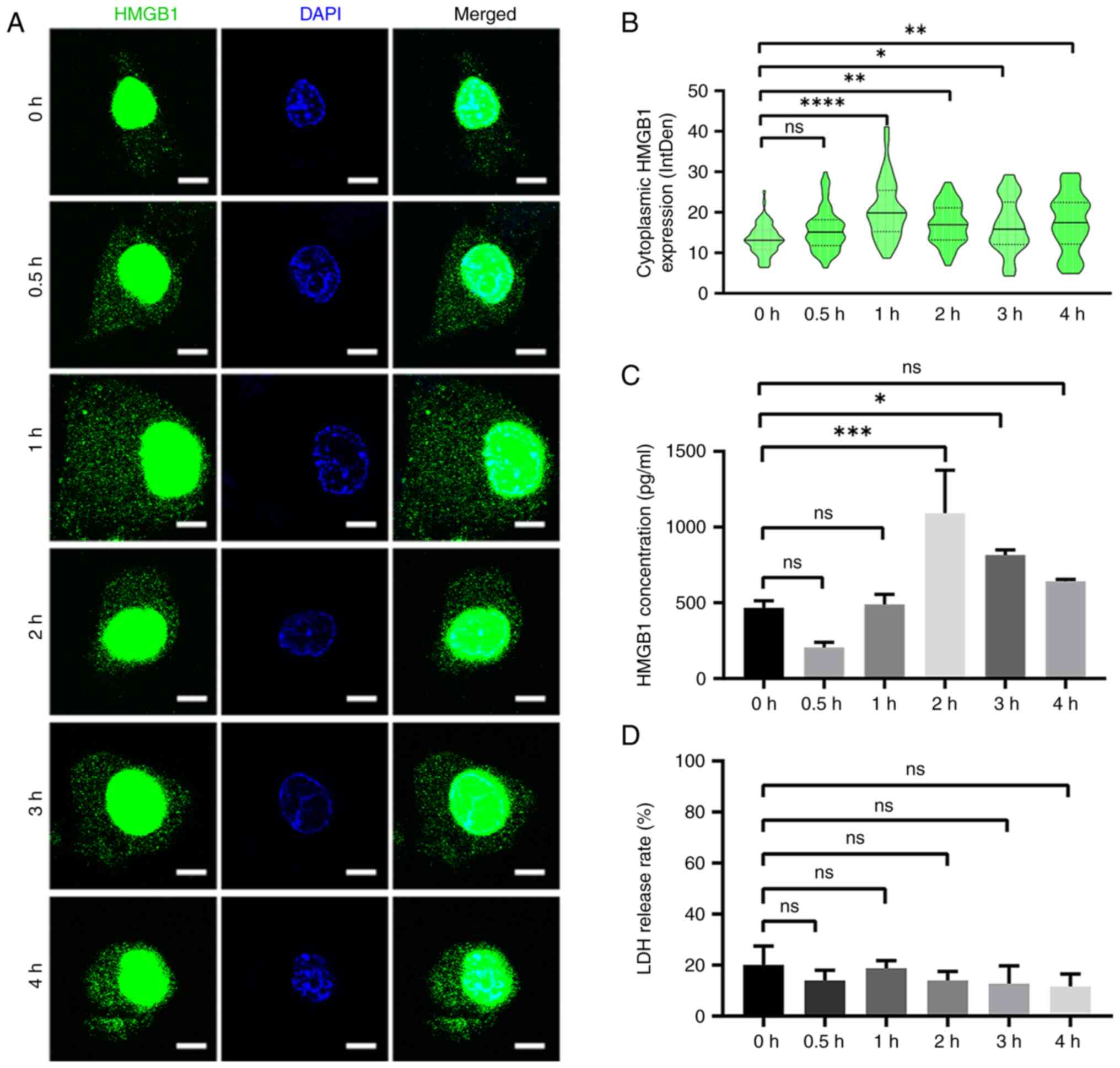
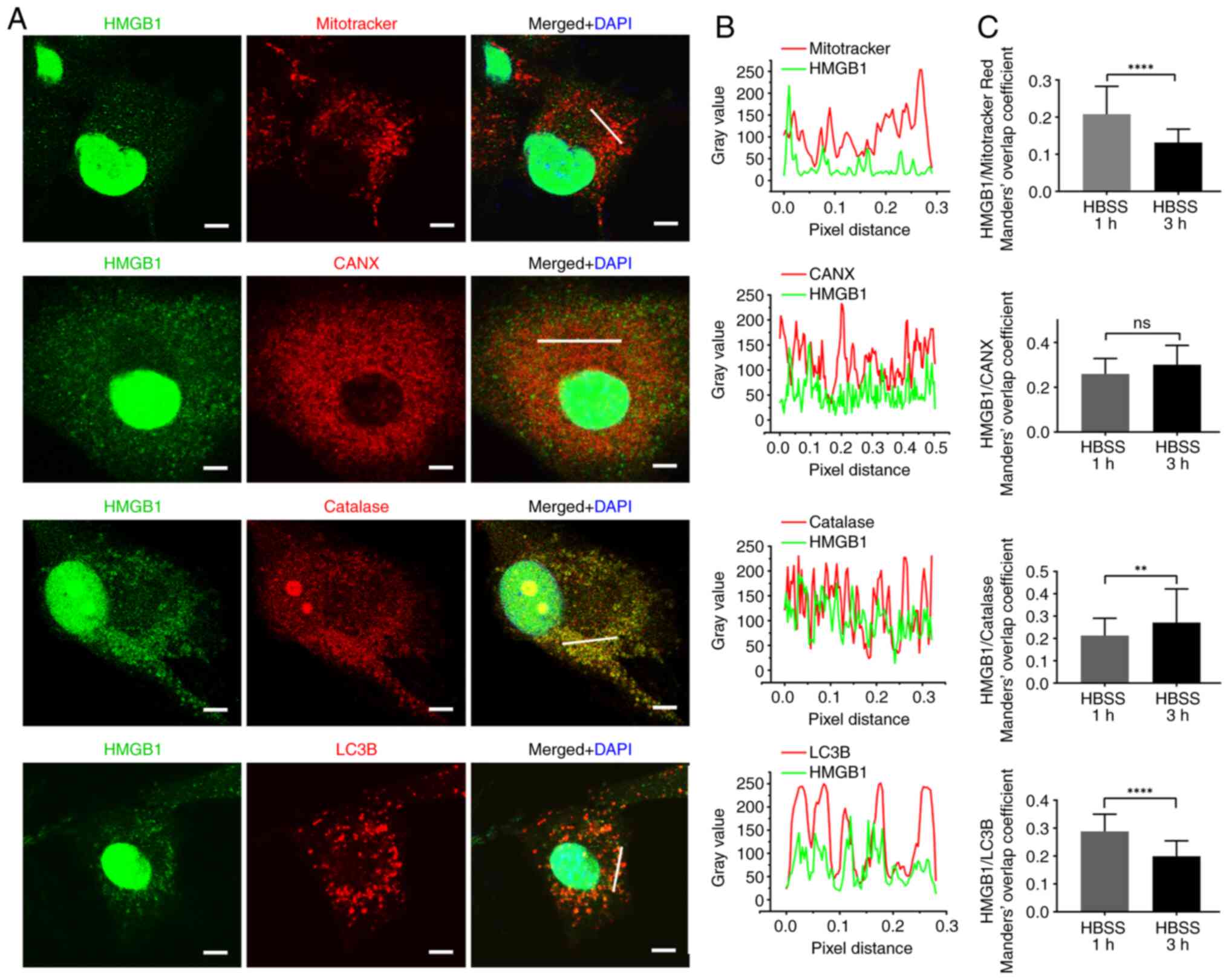
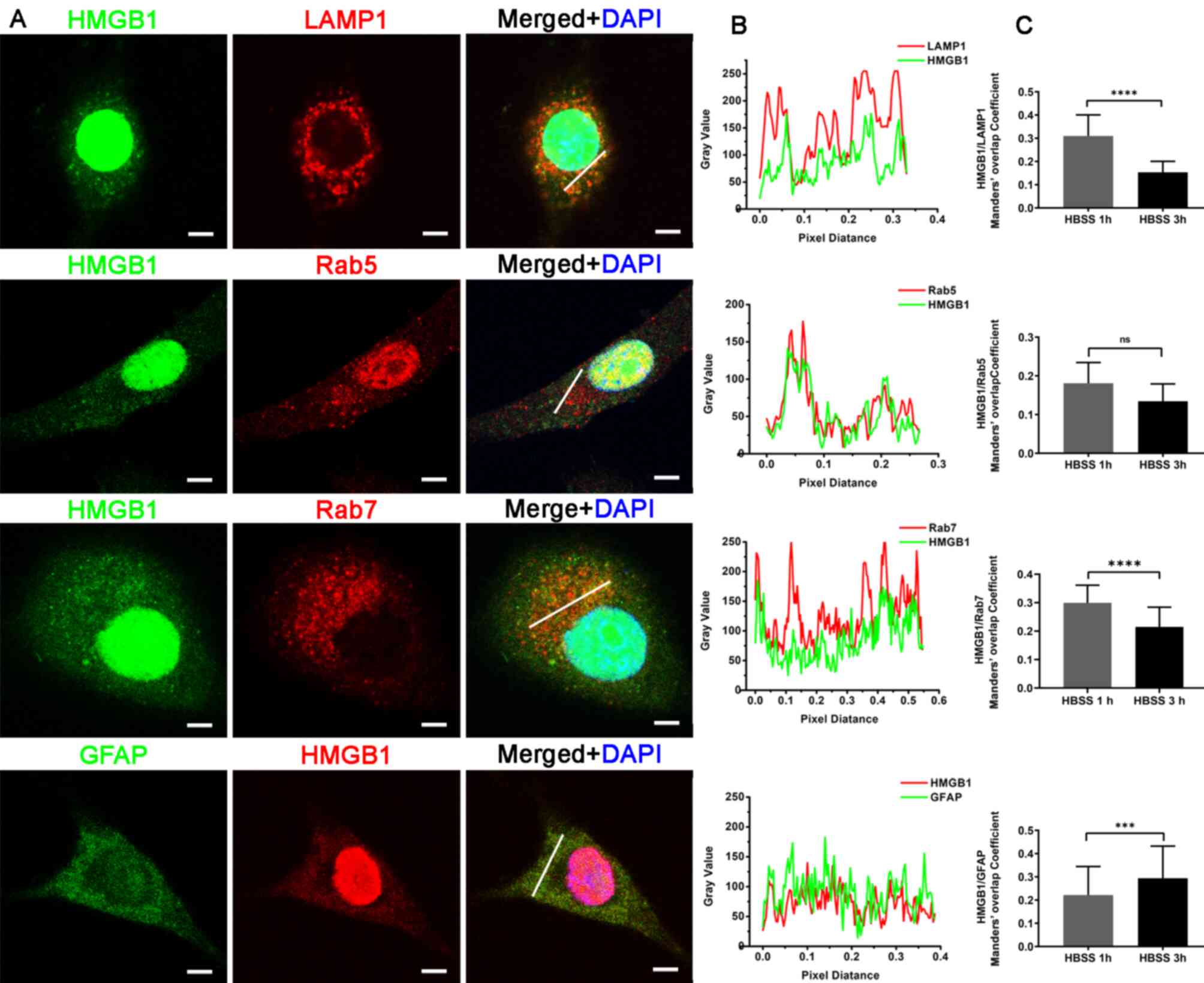
|
1
|
Perry A and Wesseling P: Histologic
classification of gliomas. Handb Clin Neurol. 134:71–95. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xu S, Tang L, Li X, Fan F and Liu Z:
Immunotherapy for glioma: Current management and future
application. Cancer Lett. 476:1–12. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Louis DN, Perry A, Reifenberger G, von
Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD,
Kleihues P and Ellison DW: The 2016 World Health Organization
Classification of Tumors of the central nervous system: A summary.
Acta Neuropathol. 131:803–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mendonça Gorgulho C, Murthy P, Liotta L,
Espina V and Lotze MT: Different measures of HMGB1 location in
cancer immunology. Methods Enzymol. 629:195–217. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Le Y, Wang Y, Zhou L, Xiong J, Tian J,
Yang X, Gai X and Sun Y: Cigarette smoke-induced HMGB1
translocation and release contribute to migration and NF-κB
activation through inducing autophagy in lung macrophages. J Cell
Mol Med. 24:1319–1331. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang S and Zhang Y: HMGB1 in inflammation
and cancer. J Hematol Oncol. 13:1162020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Völp K, Brezniceanu ML, Bösser S, Brabletz
T, Kirchner T, Göttel D, Joos S and Zörnig M: Increased expression
of high mobility group box 1 (HMGB1) is associated with an elevated
level of the antiapoptotic c-IAP2 protein in human colon
carcinomas. Gut. 55:234–242. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Akaike H, Kono K, Sugai H, Takahashi A,
Mimura K, Kawaguchi Y and Fujii H: Expression of high mobility
group box chromosomal protein-1 (HMGB-1) in gastric cancer.
Anticancer Res. 27:449–457. 2007.PubMed/NCBI
|
|
9
|
Ranganathan A, Gunnarsson O and Casarett
D: Palliative care and advance care planning for patients with
advanced malignancies. Ann Palliat Med. 3:144–149. 2014.PubMed/NCBI
|
|
10
|
Seidu RA, Wu M, Su Z and Xu H: Paradoxical
role of high mobility group box 1 in glioma: A suppressor or a
promoter? Oncol Rev. 11:3252017.PubMed/NCBI
|
|
11
|
Xue J, Suarez JS, Minaai M, Li S, Gaudino
G, Pass HI, Carbone M and Yang H: HMGB1 as a therapeutic target in
disease. J Cell Physiol. 236:3406–3419. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao M, Zhang Y, Jiang Y, Wang K, Wang X,
Zhou D, Wang Y, Yu R and Zhou X: YAP promotes autophagy and
progression of gliomas via upregulating HMGB1. J Exp Clin Cancer
Res. 40:992021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang XJ, Zhou SL, Fu XD, Zhang YY, Liang
B, Shou JX, Wang JY and Ma J: Clinical and prognostic significance
of high-mobility group box-1 in human gliomas. Exp Ther Med.
9:513–518. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gao XY, Zang J, Zheng MH, Zhang YF, Yue
KY, Cao XL, Cao Y, Li XX, Han H, Jiang XF and Liang L: Temozolomide
treatment induces HMGB1 to promote the formation of glioma stem
cells via the TLR2/NEAT1/Wnt pathway in glioblastoma. Front Cell
Dev Biol. 9:6208832021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Z, Fu WJ, Chen XQ, Wang S, Deng RS,
Tang XP, Yang KD, Niu Q, Zhou H, Li QR, et al: Autophagy-based
unconventional secretion of HMGB1 in glioblastoma promotes
chemosensitivity to temozolomide through macrophage M1-like
polarization. J Exp Clin Cancer Res. 41:742022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Grasmann G, Smolle E, Olschewski H and
Leithner K: Gluconeogenesis in cancer cells-repurposing of a
starvation-induced metabolic pathway? Biochim Biophys Acta Rev
Cancer. 1872:24–36. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Finicle BT, Jayashankar V and Edinger AL:
Nutrient scavenging in cancer. Nat Rev Cancer. 18:619–633. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Komori T: The 2016 WHO classification of
tumours of the central nervous system: The major points of
revision. Neurol Med Chir (Tokyo). 57:301–311. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fan W, Song Y, Ren Z, Cheng X, Li P, Song
H and Jia L: Glioma cells are resistant to inflammation-induced
alterations of mitochondrial dynamics. Int J Oncol. 57:1293–1306.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Prajapati P, Wang WX, Nelson PT and
Springer JE: Methodology for subcellular fractionation and MicroRNA
examination of mitochondria, mitochondria associated ER membrane
(MAM), ER, and cytosol from human brain. Methods Mol Biol.
2063:139–154. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang M, Kenny SJ, Ge L, Xu K and Schekman
R: Translocation of interleukin-1β into a vesicle intermediate in
autophagy-mediated secretion. Elife. 4:e112052015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Penjweini R, Deville S, Haji Maghsoudi O,
Notelaers K, Ethirajan A and Ameloot M: Investigating the effect of
poly-l-lactic acid nanoparticles carrying hypericin on the
flow-biased diffusive motion of HeLa cell organelles. J Pharm
Pharmacol. 71:104–116. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Suwannakul N, Midorikawa K, Du C, Qi YP,
Zhang J, Xiang BD, Murata M and Ma N: Subcellular localization of
HMGB1 in human cholangiocarcinoma: Correlation with tumor stage.
Discov Oncol. 12:492021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Satoh TK: The role of HMGB1 in
inflammatory skin diseases. J Dermatol Sci. 107:58–64. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang X, Zhou W, Li X, Ren J, Ji G, Du J,
Tian W, Liu Q and Hao A: Graphene oxide suppresses the growth and
malignancy of glioblastoma stem cell-like spheroids via epigenetic
mechanisms. J Transl Med. 18:2002020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
van Asperen JV, Robe PAJT and Hol EM: GFAP
alternative splicing and the relevance for disease-a focus on
diffuse gliomas. ASN Neuro. 14:175909142211020652022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Uceda-Castro R, van Asperen JV, Vennin C,
Sluijs JA, van Bodegraven EJ, Margarido AS, Robe PAJ, van Rheenen J
and Hol EM: GFAP splice variants fine-tune glioma cell invasion and
tumour dynamics by modulating migration persistence. Sci Rep.
12:4242022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pastorek L, Sobol M and Hozák P:
Colocalization coefficients evaluating the distribution of
molecular targets in microscopy methods based on pointed patterns.
Histochem Cell Biol. 146:391–406. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Khushi M, Napier CE, Smyth CM, Reddel RR
and Arthur JW: MatCol: A tool to measure fluorescence signal
colocalisation in biological systems. Sci Rep. 7:88792017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Le Vasseur M, Chen VC, Huang K, Vogl WA
and Naus CC: Pannexin 2 localizes at ER-mitochondria contact sites.
Cancers (Basel). 11:3432019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Klionsky DJ, Abdel-Aziz AK, Abdelfatah S,
Abdellatif M, Abdoli A, Abel S, Abeliovich H, Abildgaard MH, Abudu
YP, Acevedo-Arozena A, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy (4th edition).
Autophagy. 17:1–382. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Morciano G, Marchi S, Morganti C, Sbano L,
Bittremieux M, Kerkhofs M, Corricelli M, Danese A,
Karkucinska-Wieckowska A, Wieckowski MR, et al: Role of
mitochondria-associated ER membranes in calcium regulation in
cancer-specific settings. Neoplasia. 20:510–523. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Murley A, Sarsam RD, Toulmay A, Yamada J,
Prinz WA and Nunnari J: Ltc1 is an ER-localized sterol transporter
and a component of ER-mitochondria and ER-vacuole contacts. J Cell
Biol. 209:539–548. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Eisenberg-Bord M, Shai N, Schuldiner M and
Bohnert M: A tether is a tether is a tether: Tethering at membrane
contact sites. Dev Cell. 39:395–409. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang Z, Zhou H, Zheng H, Zhou X, Shen G,
Teng X, Liu X, Zhang J, Wei X, Hu Z, et al: Autophagy-based
unconventional secretion of HMGB1 by keratinocytes plays a pivotal
role in psoriatic skin inflammation. Autophagy. 17:529–552. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kim YH, Kwak MS, Lee B, Shin JM, Aum S,
Park IH, Lee MG and Shin JS: Secretory autophagy machinery and
vesicular trafficking are involved in HMGB1 secretion. Autophagy.
17:2345–2362. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hu Y, Chen H, Zhang L, Lin X, Li X, Zhuang
H, Fan H, Meng T, He Z, Huang H, et al: The AMPK-MFN2 axis
regulates MAM dynamics and autophagy induced by energy stresses.
Autophagy. 17:1142–1156. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bjørkøy G, Lamark T, Brech A, Outzen H,
Perander M, Overvatn A, Stenmark H and Johansen T: p62/SQSTM1 forms
protein aggregates degraded by autophagy and has a protective
effect on huntingtin-induced cell death. J Cell Biol. 171:603–614.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Komatsu M, Waguri S, Koike M, Sou YS, Ueno
T, Hara T, Mizushima N, Iwata J, Ezaki J, Murata S, et al:
Homeostatic levels of p62 control cytoplasmic inclusion body
formation in autophagy-deficient mice. Cell. 131:1149–1163. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pankiv S, Clausen TH, Lamark T, Brech A,
Bruun JA, Outzen H, Øvervatn A, Bjørkøy G and Johansen T:
p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of
ubiquitinated protein aggregates by autophagy. J Biol Chem.
282:24131–24145. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Turkoz Uluer E, Kilicaslan Sonmez P,
Akogullari D, Onal M, Tanriover G and Inan S: Do wortmannin and
thalidomide induce apoptosis by autophagy inhibition in 4T1 breast
cancer cells in vitro and in vivo? Am J Transl Res. 13:6236–6247.
2021.PubMed/NCBI
|
|
42
|
Mauthe M, Orhon I, Rocchi C, Zhou X, Luhr
M, Hijlkema KJ, Coppes RP, Engedal N, Mari M and Reggiori F:
Chloroquine inhibits autophagic flux by decreasing
autophagosome-lysosome fusion. Autophagy. 14:1435–1455. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang D, Liu K, Fukuyasu Y, Teshigawara K,
Fu L, Wake H, Ohtsuka A and Nishibori M: HMGB1 translocation in
neurons after ischemic insult: Subcellular localization in
mitochondria and peroxisomes. Cells. 9:6432020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jia L, Song Y, Song H, Wang G, Fan W, Li
X, Zheng H and Yao A: Overexpression of high mobility group box 1
(HMGB1) has no correlation with the prognosis in glioma. Biomark
Med. 13:851–863. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fu L, Liu K, Wake H, Teshigawara K,
Yoshino T, Takahashi H, Mori S and Nishibori M: Therapeutic effects
of anti-HMGB1 monoclonal antibody on pilocarpine-induced status
epilepticus in mice. Sci Rep. 7:11792017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kostova N, Zlateva S, Ugrinova I and
Pasheva E: The expression of HMGB1 protein and its receptor RAGE in
human malignant tumors. Mol Cell Biochem. 337:251–258. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Taguchi A, Blood DC, del Toro G, Canet A,
Lee DC, Qu W, Tanji N, Lu Y, Lalla E, Fu C, et al: Blockade of
RAGE-amphoterin signalling suppresses tumour growth and metastases.
Nature. 405:354–360. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bald T, Quast T, Landsberg J, Rogava M,
Glodde N, Lopez-Ramos D, Kohlmeyer J, Riesenberg S, van den
Boorn-Konijnenberg D, Hömig-Hölzel C, et al:
Ultraviolet-radiation-induced inflammation promotes angiotropism
and metastasis in melanoma. Nature. 507:109–113. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
New J and Thomas SM: Autophagy-dependent
secretion: Mechanism, factors secreted, and disease implications.
Autophagy. 15:1682–1693. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Andersson U, Yang H and Harris H:
High-mobility group box 1 protein (HMGB1) operates as an alarmin
outside as well as inside cells. Semin Immunol. 38:40–48. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Elmaci I, Alturfan EE, Cengiz S, Ozpinar A
and Altinoz MA: Neuroprotective and tumoricidal activities of
cardiac glycosides. Could oleandrin be a new weapon against stroke
and glioblastoma? Int J Neurosci. 128:865–877. 2018.PubMed/NCBI
|
|
52
|
Fraser J, Simpson J, Fontana R,
Kishi-Itakura C, Ktistakis NT and Gammoh N: Targeting of early
endosomes by autophagy facilitates EGFR recycling and signalling.
EMBO Rep. 20:e477342019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Okuma Y, Becker LB, Hayashida K, Aoki T,
Saeki K, Nishikimi M, Shoaib M, Miyara SJ, Yin T and Shinozaki K:
Effects of post-resuscitation normoxic therapy on oxygen-sensitive
oxidative stress in a rat model of cardiac arrest. J Am Heart
Assoc. 10:e0187732021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Liu Y, Yan W, Tohme S, Chen M, Fu Y, Tian
D, Lotze M, Tang D and Tsung A: Hypoxia induced HMGB1 and
mitochondrial DNA interactions mediate tumor growth in
hepatocellular carcinoma through Toll-like receptor 9. J Hepatol.
63:114–121. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhang GZ, Zhang K, Yang SQ, Zhang Z, Chen
S, Hou BJ and Yuan JY: VASPIN reduces inflammation and endoplasmic
reticulum stress of renal tubular epithelial cells by inhibiting
HMGB1 and relieves renal ischemia-reperfusion injury. Eur Rev Med
Pharmacol Sci. 24:8968–8977. 2020.PubMed/NCBI
|
|
56
|
Tu L, Long X, Song W, Lv Z, Zeng H, Wang
T, Liu X, Dong J and Xu P: MiR-34c acts as a tumor suppressor in
non-small cell lung cancer by inducing endoplasmic reticulum stress
through targeting HMGB1. Onco Targets Ther. 12:5729–5739. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Di Cara F, Andreoletti P, Trompier D,
Vejux A, Bülow MH, Sellin J, Lizard G, Cherkaoui-Malki M and Savary
S: Peroxisomes in immune response and inflammation. Int J Mol Sci.
20:38772019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lismont C, Revenco I and Fransen M:
Peroxisomal hydrogen peroxide metabolism and signaling in health
and disease. Int J Mol Sci. 20:36732019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Fan H, Tang HB, Chen Z, Wang HQ, Zhang L,
Jiang Y, Li T, Yang CF, Wang XY, Li X, et al: Inhibiting HMGB1-RAGE
axis prevents pro-inflammatory macrophages/microglia polarization
and affords neuroprotection after spinal cord injury. J
Neuroinflammation. 17:2952020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Muro S: Alterations in cellular processes
involving vesicular trafficking and implications in drug delivery.
Biomimetics (Basel). 3:192018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Nguyen TMT, Kim J, Doan TT, Lee MW and Lee
M: APEX proximity labeling as a versatile tool for biological
research. Biochemistry. 59:260–269. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lam SS, Martell JD, Kamer KJ, Deerinck TJ,
Ellisman MH, Mootha VK and Ting AY: Directed evolution of APEX2 for
electron microscopy and proximity labeling. Nat Methods. 12:51–54.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Jiang J, Zhang L, Chen H, Lei Y, Zhang T,
Wang Y, Jin P, Lan J, Zhou L, Huang Z, et al: Regorafenib induces
lethal autophagy arrest by stabilizing PSAT1 in glioblastoma.
Autophagy. 16:106–122. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Song L, Tang S, Han X, Jiang Z, Dong L,
Liu C, Liang X, Dong J, Qiu C, Wang Y and Du Y: KIBRA controls
exosome secretion via inhibiting the proteasomal degradation of
Rab27a. Nat Commun. 10:16392019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wall AA, Condon ND, Luo L and Stow JL:
Rab8a localisation and activation by Toll-like receptors on
macrophage macropinosomes. Philos Trans R Soc Lond B Biol Sci.
374:201801512019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Wu PH, Onodera Y, Giaccia AJ, Le QT,
Shimizu S, Shirato H and Nam JM: Lysosomal trafficking mediated by
Arl8b and BORC promotes invasion of cancer cells that survive
radiation. Commun Biol. 3:6202020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Tian X, Zheng P, Zhou C, Wang X, Ma H, Ma
W, Zhou X, Teng J and Chen J: DIPK2A promotes STX17- and
VAMP7-mediated autophagosome-lysosome fusion by binding to VAMP7B.
Autophagy. 16:797–810. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Sleiman M, Stevens DR, Chitirala P and
Rettig J: Cytotoxic granule trafficking and fusion in
synaptotagmin7-deficient cytotoxic T lymphocytes. Front Immunol.
11:10802020. View Article : Google Scholar : PubMed/NCBI
|