Introduction
Breast cancer (BC) is a major contributor to global
cancer-related mortality in women. According to the World Cancer
Statistics, it was the second most prevalent cancer among women in
2022, with ~2.3 million new cases reported (~11.6% incidence)
(1,2). The efficacy of BC therapy is often
compromised by the development of drug resistance, a key factor
associated with treatment failure and elevated mortality (3,4). Tumor
cell resistance involves complex molecular mechanisms, including
the inactivation of tumor suppressor genes and the overexpression
of ATP-binding cassette (ABC) transporters. To date, >40 ABC
transporters have been identified and their overexpression is
associated with multidrug resistance (MDR) in cancer therapy
(5). Key ABC transporters
implicated in MDR include ABCB1 (P-glycoprotein), ABCCs
(MDR-associated proteins) and ABCG2 (BC resistance protein)
(6–8). Therefore, developing novel agents that
inhibit ABC transporters to reduce drug efflux may enhance
chemosensitivity. While synthetic inhibitors such as Verapamil
exist (9), it remains unclear
whether natural compounds can simultaneously target multiple ABC
transporters in BC cells.
Quercetin (Que), a natural flavonoid, can inhibit
proliferation and enhance the cytotoxicity of conventional
chemotherapeutic agents such as doxorubicin in BC cell lines,
including MCF-7 and MDA-MB-231 (10–13).
It can also induce cell cycle arrest and apoptosis in
drug-resistant sublines. These properties position Que as a
promising candidate for overcoming chemoresistance. However, the
intricate signaling networks through which Que modulates ABC
transporter expression are not fully understood. The present study
aims to elucidate the role of Que in regulating ABC transporters in
MCF-7 cells and explore the underlying molecular mechanism.
Materials and methods
Cell culture and materials
MCF-7, a human BC cell line, was purchased from
Procell Life Science & Technology Co., Ltd. and cultured in
Dulbecco's Modified Eagle Medium (DMEM; Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin-streptomycin (100
U/ml; Cytiva) at 37°C in a humidified incubator with 5%
CO2 atmosphere. Cells were routinely screened for
mycoplasma contamination. Que (Beijing Solarbio Science &
Technology Co., Ltd.) was dissolved in 100% DMSO to prepare a 100
mM stock solution, which was then diluted in DMEM such that the
final concentration of DMSO was maintained at 0.1% (v/v) across all
working concentrations of Que (30, 60, 90 and 120 µmol/l). The
pLVX-Puro-control (Plvx-con) and pLVX-Puro-human-PTEN (Plvx-PTEN,
NM_000314.6) plasmids were sourced from Xiamen Life Internet
Technology Co., Ltd. and LY294002, Verapamil and Rhodamine 123 was
purchased from MedChemExpress.
Cell transfection
A total of 1×106 MCF-7 cells were
transfected with 5 µg Plvx-con or Plvx-PTEN with the
Lipo8000™ Transfection Reagent (Beyotime Biotechnology)
in 6-cm dishes, strictly according the manufacturer's protocol.
Following transfection, the cells were treated with 60 µmol/l Que
for 24 h at 37°C before subsequent experiments.
Cell Counting Kit-8 (CCK-8) assay
For each treatment group, 5×103 MCF-7
cells were plated in triplicate into 96-well plates and allowed to
adhere for 24 h at 37°C in a CO2 incubator. Cells were
treated with increasing concentrations of Que (0, 30, 60, 90 and
120 µmol/l) for 24 or 48 h at 37°C. For combination treatments,
cells were transfected with 0.2 µg Plvx-con or 0.2 µg Plvx-PTEN for
24 h at 37 °C, followed by treatment with 60 µmol/l Que for another
24 h at 37°C. After that, 10 µl of CCK-8 solution (Beijing Lablead
Trading Co., Ltd.) was added to each well and the cells were
incubated for 2 h at 37°C. The absorbance was measured at 450 nm
using an Infinite M1000 Pro microplate reader (Tecan Trading
Ag).
Apoptosis analysis by flow
cytometry
A total of 1×105 MCF-7 cells were
cultured in six-well plates and treated with Que (0, 30, 60, 90 or
120 µmol/l) for 24 h at 37°C. In a separate experiment, cells were
treated with 60 µmol/l Que, 25 µmol/l LY294002 or a combination of
both for 24 h at 37°C. Cells were then trypsinized, and harvested
via centrifugation at 800 × g (4°C), and stained with 10 µl of
Annexin V-FITC and 5 µl of propidium iodide using an apoptosis
detection kit (Beyotime Biotechnology). Cells were analyzed on a
Beckman Coulter (BC) FC 500 flow cytometer using the CXP software
(version 2.2) for data acquisition and analysis.
Western blot analysis
MCF-7 cells were lysed in cold RIPA lysis buffer
(Beyotime Biotechnology) supplemented with protease and phosphatase
inhibitors (cat. no. C0104, Beijing Lanbolide Trading Co., Ltd.).
The protein concentration was determined using the BCA assay
according to the manufacturer's instructions (Dalian Meilun Biology
Technology Co., Ltd.), and 30 µg protein samples were separated by
8–12% SDS-PAGE, transferred to PVDF membranes (Millipore Sigma) and
blocked at room temperature with blocking buffer (cat. no.
BM10-100; Energenesis Biomedical Co., Ltd.). The membranes were
incubated with primary antibodies overnight at 4°C. Primary
antibodies included PTEN (1:5,000; cat. no. ab267787), Bcl-2
(1:6,000; cat. no. ab196495), Bax (1:6,000; cat. no. ab32503) and
ABCG2 (1:5,000; cat. no. ab108312) from Abcam; p-PI3K (p85)
(1:2,000; cat. no. 4228), p-AKT (Ser473) (1:2,000; cat. no. 4060),
cleaved caspase-3 (1:5,000; cat. no. 9664), ABCC2 (1:1,000; cat.
no. 12559) and ABCB1 (1:2,000; cat. no. 13342) from Cell Signaling
Technology, Inc.; and GAPDH (1:12,000; cat. no. 60004-1-Ig), PI3K
(1:3,000; cat. no. 20584-1-AP), AKT (1:3,000; 10176-2-AP),
Caspase-3 (1:3,000; 19677-1-AP) and β-actin (1:10,000; cat. no.
66009-1-Ig) from Proteintech Group, Inc. After incubation with
HRP-conjugated secondary antibodies (1:10,000; cat. no. SA00001-1
and SA00001-2; Proteintech Group, Inc.), protein bands were
visualized using an ECL reagent (Beijing Lanbolide Trading Co.,
Ltd.) and imaged with a ChemiDoc XRS+ system (Bio-Rad Laboratories,
Inc.).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from MCF-7 cells using
TRIzol® (Thermo Fisher Scientific, Inc.) according to
the manufacturer's instructions. Subsequently, cDNA was synthesized
from the isolated RNA using a commercial reverse transcription kit
(cat. no. F0202; Beijing Lanbolide Trading Co., Ltd.). qPCR was
carried out using SYBR Green master mix (Beijing Lanbolide Trading
Co., Ltd.) on a Roche LightCycler® 96 System (Roche
Diagnostics). The thermal cycling protocol consisted of an initial
denaturation at 95°C for 30 sec, followed by 40 cycles of
denaturation at 95°C for 5 sec and annealing/extension at 60°C for
30 sec. GAPDH was used as the internal control and relative gene
expression was calculated using the 2−ΔΔCq method
(14). Primer sequences are listed
in Table I.
 | Table I.Reverse transcription-quantitative
PCR primer sequences. |
Table I.
Reverse transcription-quantitative
PCR primer sequences.
| Gene name | Forward sequence
(5′-3′) | Reverse sequence
(5′-3′) |
|---|
| GAPDH |
TGACCACAGTCCATGCCATCAC |
CGCCTGCTTCACCACCTTCTT |
| ABCB1 |
CGTAGGAGTGTCCGTGGATCA |
GCGAGCCTGGTAGTCAATGC |
| Bax |
CCAAGAAGCTGAGCGAGTGTCT |
AGATGGTGAGTGAGGCGGTGAG |
| Bcl-2 |
TTCGCCGAGATGTCCAGCCA |
GCATCCCAGCCTCCGTTATCCT |
| PTEN |
GCTGGAAAGGGACGAACTGGTG |
ACAGGTAACGGCTGAGGGAACT |
| ABCG2 |
TCTTCTTCCTGACGACCAACCA |
CACACTCTGACCTGCTGCTATG |
| ABCC2 |
TCACTTCAGCGAGACCGTATCA |
ATGTCATCCTCACCAGCCAGTT |
Hoechst 33342 staining
Transfected and Que-treated MCF-7 cells were fixed
with 4% paraformaldehyde for 30 min at room temperature and stained
with Hoechst 33342 solution (Beijing Solarbio Science &
Technology co., Ltd.) for 30 min at room temperature. Cell nuclei
were visualized and imaged under an inverted fluorescence
microscope (Leica Microsystems GmbH).
Immunofluorescence analysis
A total of 1×105 cells were seeded into
3.5-cm confocal dishes, transfected and subsequently treated with
60 µmol/l Que and/or 25 µmol/l LY294002 for 24 h at 37°C. After
washing with PBS, cells were fixed with 4% paraformaldehyde for 30
min at room temperature, blocked with 2% BSA (Beijing Lanbolide
Trading Co., Ltd.) for 1 h at room temperature and incubated
overnight at 4°C with primary antibody against ABCG2 (1:400). Cells
were then incubated with Alexa Fluor® 488-conjugated
secondary antibody (1:1,000; cat. no. ab150077; Abcam) at room
temperature for 1 h, and the nuclei was stained with
DAPI-containing mounting medium (cat. no. ab104139; Abcam) for 10
min at room temperature. Fluorescence images were captured using a
Leica SP8 confocal microscope (Leica Microsystems GmbH).
Rhodamine 123 accumulation assay
MCF-7 cells were treated as following groups: i)
Control group, ii) 100 µmol/l Verapamil group, iii) 60 µmol/l Que
group, iv) 25 µmol/l LY294002 group and v) 25 µmol/l LY294002 + 60
µmol/l Que group. MCF-7 cells were treated with the indicated
agents and cultured for 24 h at 37°C. Notably, Verapamil (100
µmol/l) was added to the corresponding group 1 h prior to Rhodamine
123 staining. Following the 24 h treatment, the cells were washed
three times with Hanks' balanced salt solution (HBSS; Beijing
Solarbio Science & Technology co., Ltd.) and incubated with 100
µmol/l Rhodamine 123 for 15 min at 37°C (No fixation or other
staining steps were performed). After three additional washed with
HBSS, fluorescence images were acquired using an inverted
fluorescence microscope (Leica Microsystems GmbH) and fluorescence
intensity was quantified to assess ABC transporter activity.
Statistical analysis
Statistical analyses were performed using the
GraphPad Prism 6.0 (Dotmatics). Data are presented as the mean ±
SEM from at least three independent experiments. Comparisons
between groups were conducted using an unpaired Student's t-test or
one-/two-way ANOVA, as appropriate. For two-way ANOVA, Bonferroni's
post-hoc test was used to evaluate multiple comparisons. P<0.05
was considered to indicate a statistically significant
difference.
Results
Que inhibits viability, induces
apoptosis and downregulates ABC transporters in MCF-7 cells
As assessed by the CCK-8 assay, Que treatment caused
a marked suppression of MCF-7 cell viability, and the inhibitory
effect was dependent on both the concentration and duration of
exposure (Fig. 1A). Furthermore,
Que downregulated the mRNA and protein expression of ABCB1, ABCC2
and ABCG2 (Fig. 1B-E). It also
promoted apoptosis, as evidenced by decreased Bcl-2 and increased
Bax and cleaved caspase-3 expression (Fig. 1C-D), as well as an increased
percentage of apoptotic cells in flow cytometry analysis (Fig. 1F and G). Based on these results, 60
µmol/l Que was selected for subsequent experiments.
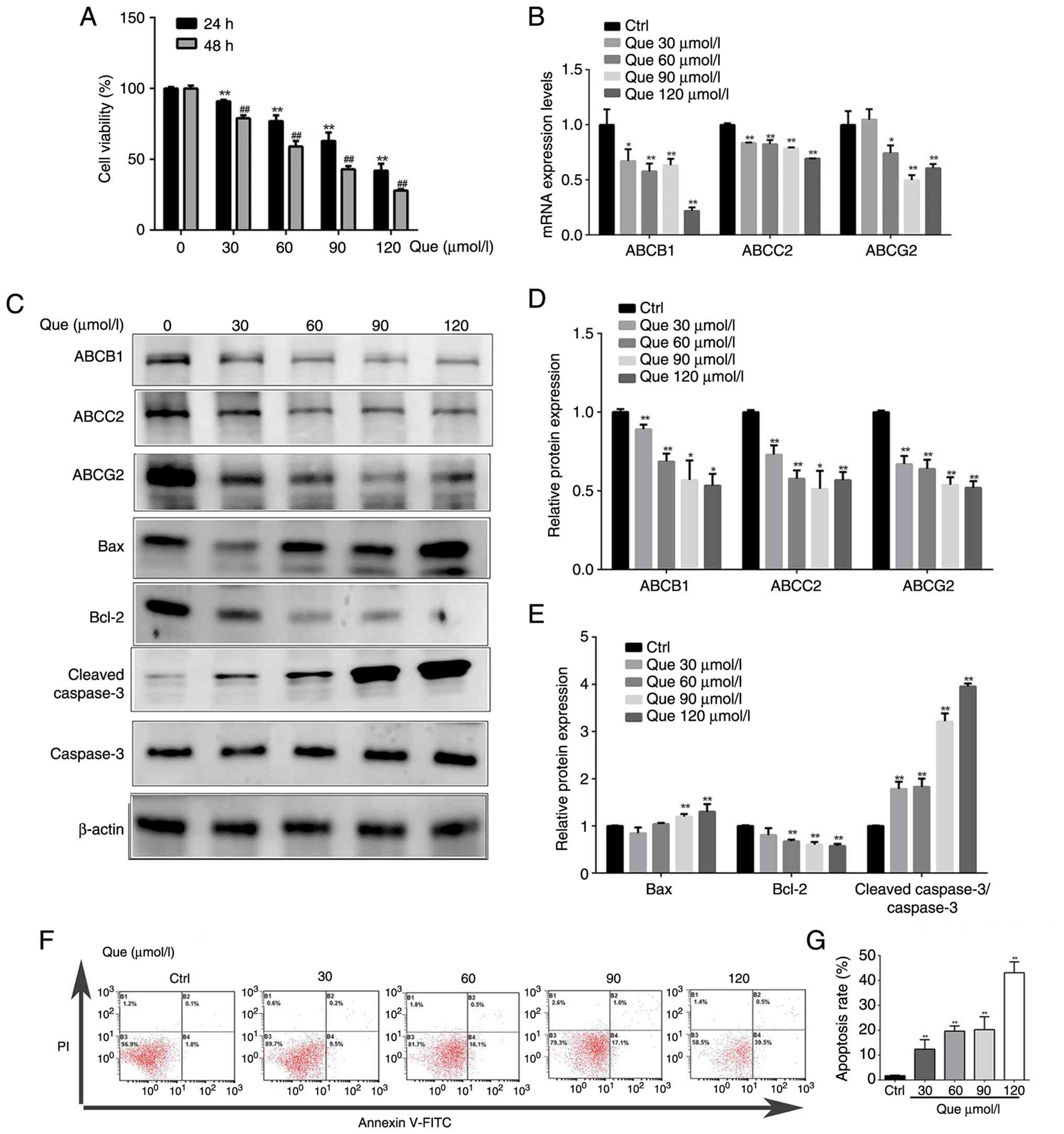 | Figure 1.Effects of Que on cell viability,
mRNA levels, apoptosis and protein expression in MCF-7 cells. (A)
Cell viability of MCF-7 cells treated with the indicated
concentrations of Que for 24 and 48 h, as determined by the Cell
Cycle Kit-8 assay. (B) mRNA expression levels of ABCB1, ABCC2 and
ABCG2 following Que treatment. (C) Representative western blotting
images, (D) protein expression levels of ABCB1, ABCC2 and ABCG2,
and (E) Bax, Bcl-2, and cleaved caspase-3 were assessed by western
blotting. (F) Representative images of flow cytometry and (G)
apoptosis rate was analyzed by flow cytometry after Annexin
V-FITC/PI staining. Data are presented as the mean ± SEM of at
least three independent experiments. Statistical significance was
determined by one/two-way ANOVA followed by Bonferroni's post hoc
test for multiple comparisons. *P<0.05, **P<0.01 compared
with the control group, ##P<0.01 compared with the
Que (24 h) group. Que, Quercetin; Ctrl, control. |
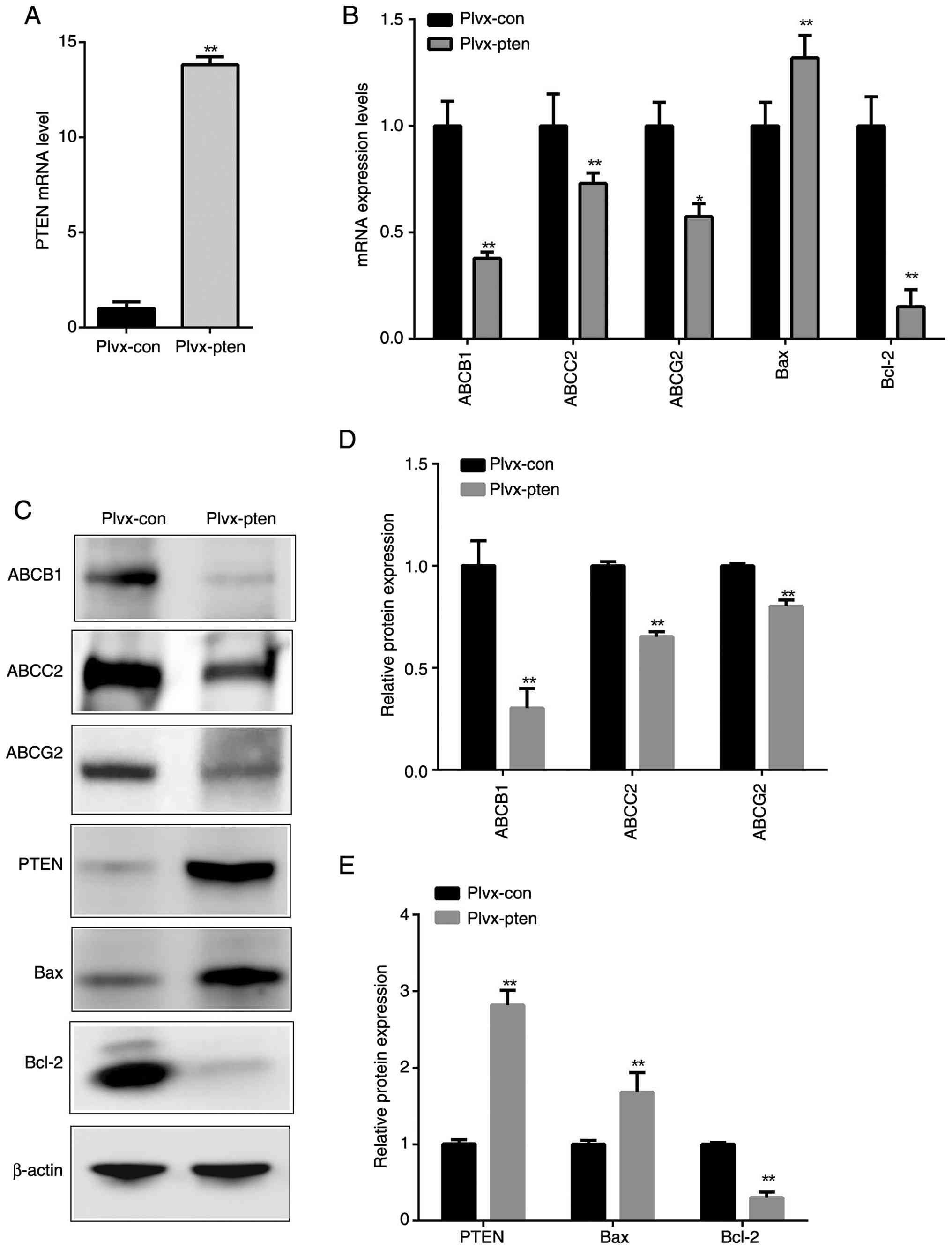
PTEN overexpression suppresses ABC
transporter expression and modulates apoptosis-related
proteins
PTEN inactivation or mutation is implicated in
various types of cancer, including BC (15–18).
Patients with PTEN mutations have a higher risk of primary and
secondary types of BC (19). The
present study verified successful PTEN overexpression, which
significantly reduced the mRNA and protein levels of ABCB1, ABCC2
and ABCG2 (Fig. 2A-E). It also
reduced the anti-apoptotic protein Bcl-2 and increased the level of
the pro-apoptotic protein Bax (Fig. 2C
and E), mirroring the effects of Que.
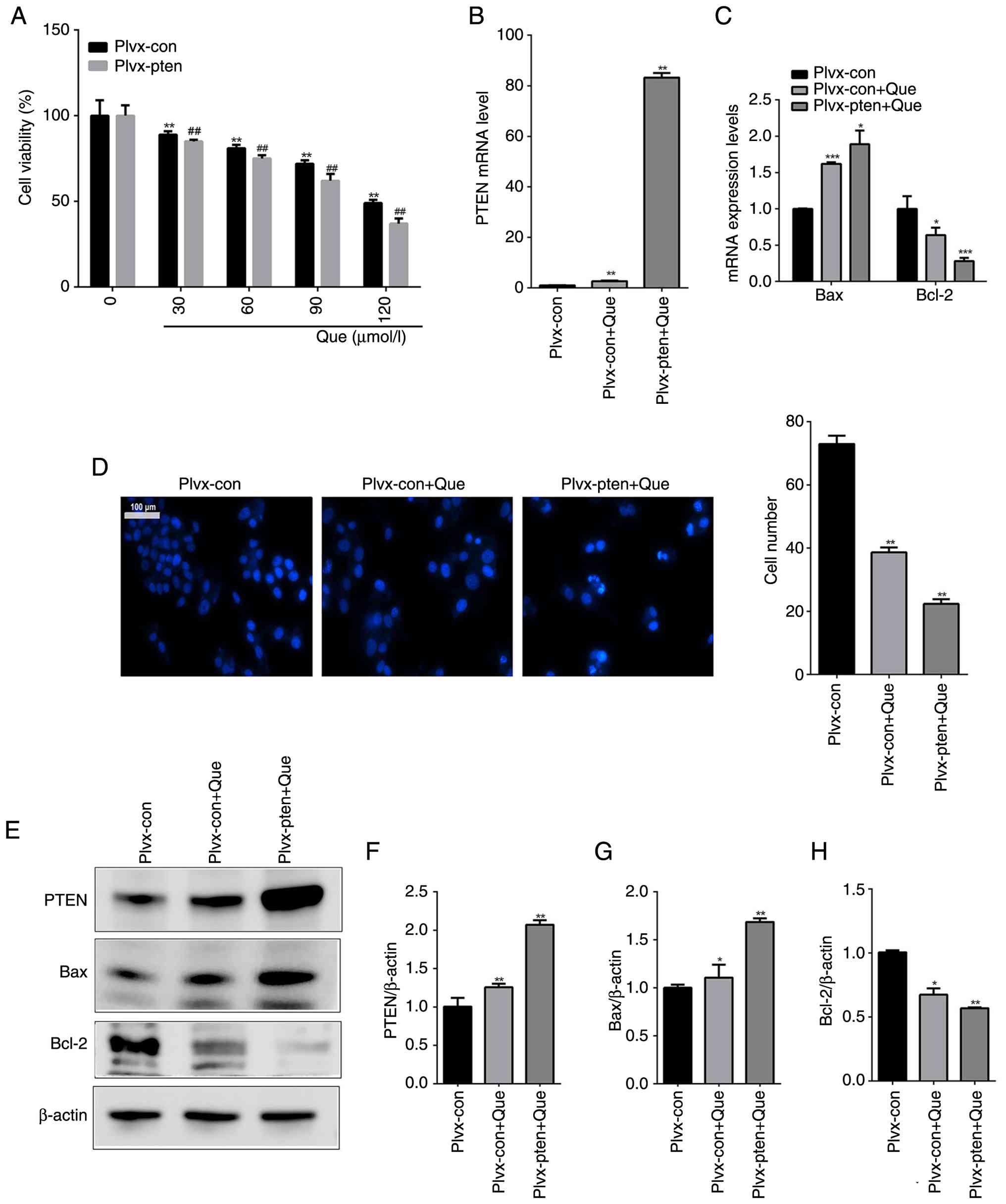
PTEN overexpression synergizes with
Que to enhance anti-proliferative and pro-apoptotic effects
The combination of PTEN overexpression and Que
treatment resulted in a greater suppression of cell viability
compared with Que treatment alone (Fig.
3A). This combination also led to a more pronounced
upregulation of PTEN and Bax and downregulation of Bcl-2 at the
gene and protein level (Fig. 3B, C,
E-H). Hoechst 33342 staining analysis revealed a marked
reduction in viable cells in the combination group (Fig. 3D).
The combination of PTEN overexpression
and Que suppresses ABC transporters via the PI3K/AKT pathway
Clinical studies have shown that overexpression of
ABC efflux transporters, such as ABCB1, ABCC2 and ABCG2,
contributes to MDR in cancer by reducing intracellular drug
concentrations (20,21). Therefore, the present study examined
whether PTEN expression influences Que-mediated regulation of ABC
transporters. Analysis revealed that PTEN overexpression
potentiates Que-induced downregulation of ABCB1, ABCC2 and ABCG2
mRNA and protein (Fig. 4A-E).
Concurrently, the combination treatment led to a significant
reduction in the levels of phosphorylated PI3K and AKT (Fig. 4D, F and G), indicating inhibition of
the PI3K/AKT signaling pathway.
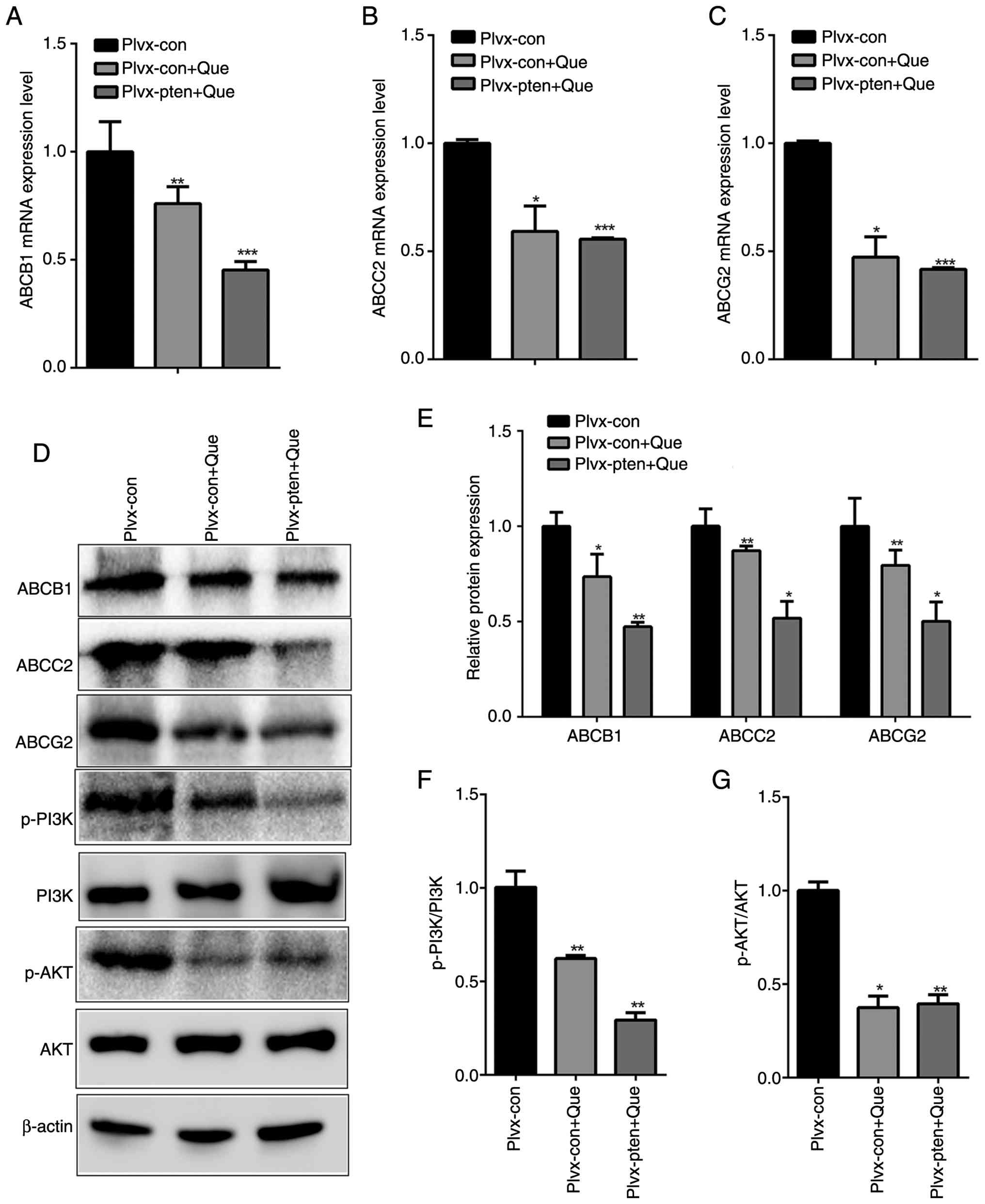 | Figure 4.Effects of combined PTEN
overexpression and Que treatment on ABC transporters and the
PI3K/AKT signaling pathway in MCF-7 cells. mRNA expression levels
of (A) ABCB1, (B) ABCC2 (C) and ABCG2 in different treatment
groups. (D) Representative images and (E) protein expression levels
of ABCB1, ABCC2, ABCG2, (F) p-PI3K, and (G) p-AKT were evaluated by
western blotting. The results are presented as the mean ± SEM of at
least three independent experiments and analyzed using one-way
ANOVA followed by Bonferroni's post hoc test. *P<0.05,
**P<0.01, ***P<0.001 compared with the Plvx-con group. Que,
Quercetin; con, control; p, phosphorylated. |
Inhibition of PI3K/AKT signaling
enhances the effects of Que on apoptosis and ABC transporters
The PI3K/AKT signaling pathway is involved in cell
proliferation and the expression of drug-resistant proteins,
including ABC transporters (22–24).
To explore the relationship between Que and PI3K/AKT signaling in
regulating apoptosis and ABC transporters, MCF-7 cells were treated
with LY294002 (a PI3K inhibitor), Que or a combination of both.
Treatment with LY294002 alone enhanced the effects of cell
apoptosis and inhibited ABC transporter expression. The combination
of Que and LY294002 resulted in the most substantial reduction of
ABCB1, ABCC2 and ABCG2 mRNA and protein, as well as the strongest
inhibition of PI3K/AKT signaling (Fig.
5A-F). Immunofluorescence analysis corroborated the
downregulation of ABCG2 protein (Fig.
5G) and flow cytometry revealed that the highest rate of
apoptosis occurred in the combination group (Fig. 5H and I).
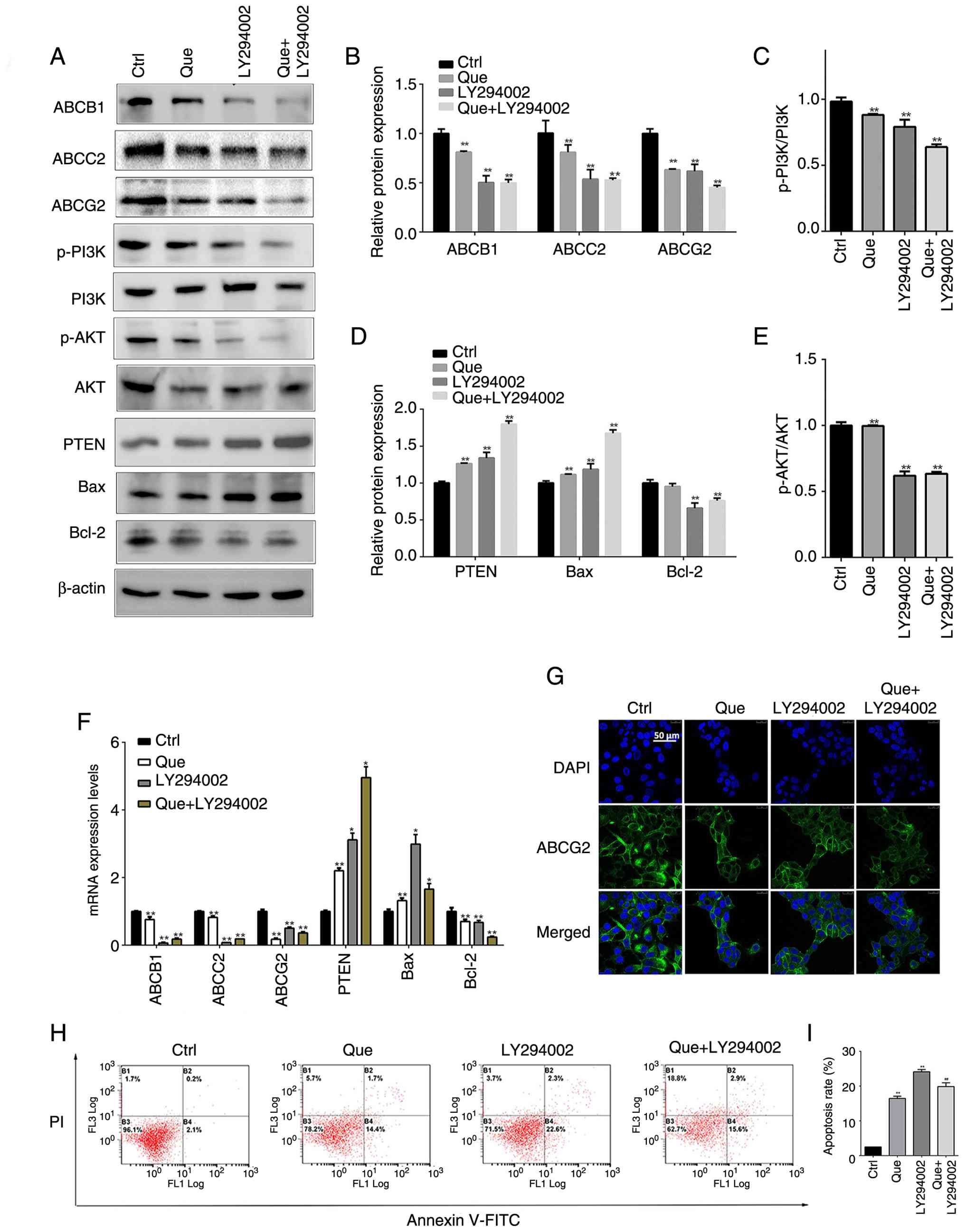 | Figure 5.Effects of combined LY294002 and Que
treatment on apoptosis and protein expression in MCF-7 cells. (A)
Representative western blotting images and Protein expression
levels of (B) ABCB1, ABCC2, ABCG2, (C) p-PI3K (D) PTEN, Bax, Bcl-2
and (E) p-AKT in different treatment groups. (F) mRNA expression
levels of ABCB1, ABCC2, ABCG2, PTEN, Bax and Bcl-2 in different
treatment groups. (G) Representative immunofluorescence images of
ABCG2 expression (green). Nuclei were counterstained with DAPI
(blue). Scale bar, 50 µm. (H) Representative flow cytometry images
and (I) Apoptosis rate was analyzed by flow cytometry after Annexin
V-FITC/PI staining. The results are presented as the mean ± SEM of
at least three independent experiments and analyzed using two-way
ANOVA followed by Bonferroni's post hoc test. *P<0.05,
**P<0.01 compared with the control group; ##P<0.05
compared with the Que group. Que, Quercetin; Ctrl, control; p,
phosphorylated. |
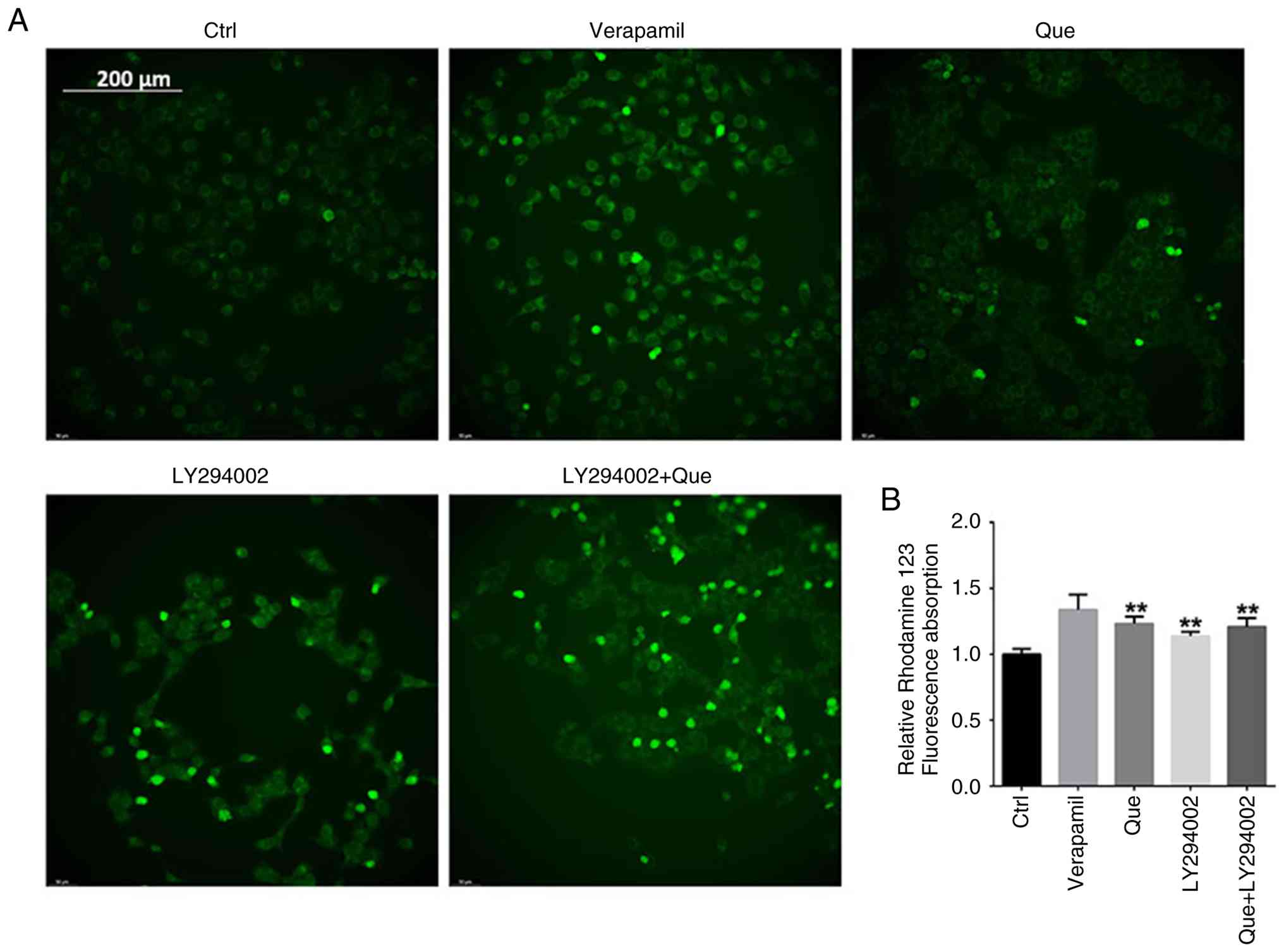
Que and LY294002 increase
intracellular Rhodamine 123 accumulation
Rhodamine 123, a substrate for ABC transporters, is
used to assess transporter activity (25). The present study investigated the
functional consequence of ABC transporter downregulation using the
Rhodamine 123 accumulation assay. Analysis revealed that both Que
and LY294002 increased intracellular Rhodamine 123 retention, with
the combination treatment showing the most significant effect
(Fig. 6A and B), comparable to the
positive control Verapamil. These results indicate that both Que
and LY294002 inhibit the efflux activity of ABC transporters. The
observed increase in fluorescent dye accumulation is consistent
with a decrease in ABC transporter protein expression, as confirmed
in Figs. 1B and C and 5A and B. This suggests that the
downregulation of ABC transporters by Que and LY294002 may enhance
the intracellular accumulation and efficacy of co-administered
therapeutic agents.
Discussion
Despite considerable advances in BC treatment,
discovering effective strategies to overcome drug resistance
remains important. Modern tyrosine kinase inhibitors such as
lapatinib and sorafenib are effective against early-stage BC but
are of limited value in advanced stages, often leading to drug
resistance and side effects such as neuropathy and bone marrow
suppression (26,27). Therefore, identifying novel
therapeutic agents that mitigate side effects and prevent drug
resistance is essential.
Que a naturally occurring flavonoid with
anti-inflammatory and anti-aging properties, has been shown to
induce autophagy and cell death in various types of cancer
including myeloma, prostate cancer and lung cancer (28–30).
However, its effects on drug-resistant proteins such as ABC
transporters in BC cells remain poorly understood. The present
study demonstrates that Que enhances drug uptake in MCF-7 cells by
targeting ABC transporters through the PTEN/PI3K/AKT axis.
Consistent with previous reports (10–13),
the present study revealed that Que inhibits cell proliferation and
promotes apoptosis. More importantly, the present study identified
a previously unreported mechanism: Que downregulates the expression
of the key ABC transporters ABCB1, ABCC2 and ABCG2. This effect was
functionally corroborated by enhanced intracellular accumulation of
Rhodamine 123, a known substrate of these transporters (31), indicating that Que may augment drug
retention in cancer cells.
While previous research has primarily focused on its
pro-apoptotic role, the present study revealed a dual-acting
mechanism. The present study demonstrated, for the first time: that
Que not only promotes apoptosis but also concurrently overcomes a
key driver of chemoresistance by transcriptionally repressing key
ABC transporters. This effect may be mechanistically driven by the
restoration of PTEN and subsequent inhibition of the PI3K/AKT
pathway, which implicates a previously unreported signaling axis,
PTEN/PI3K/AKT/ABC transporters. This repositions Que as a potential
double agent that may target both cell survival and drug efflux
pathways which may contribute to counteracting MDR.
To further elucidate this axis, the present study
investigated the role of PTEN in more detail. As a tumor suppressor
that is frequently downregulated in several types of cancer
(32,33), PTEN contributes to drug resistance
by regulating drug influx, efflux and anti-apoptotic processes
(34,35). Results of the present study indicate
that Que upregulates PTEN expression while suppressing
phosphorylation of PI3K and AKT, suggesting a modulatory role in
this pathway. Given that PI3K/AKT inhibition can alleviate
chemoresistance (36), these
findings position Que as a potential candidate for resistance
prevention. This conclusion is further supported by the observation
that PTEN overexpression potentiated the effects of Que and that
the PI3K inhibitor LY294002 phenocopied these outcomes (37). The synergy between Que and both
genetic and pharmacological inhibition of the pathway suggests the
PTEN/PI3K/AKT axis as a central target of Que.
The functional outcome of targeting the
PTEN/PI3K/AKT axis is the inhibition of ABC transporter activity.
ABC transporters, which facilitate drug efflux, are
well-established mediators of MDR (20,38).
In line with this, the Rhodamine 123 accumulation assay confirmed
that Que, either alone or in combination with LY294002, inhibits
ABC transporter activity, leading to increased drug retention.
Collectively, the results of the present study provide functional
data that Que may enhance chemosensitivity in MCF-7 cells through
transcriptional and functional repression of ABC transporters,
mediated by the PTEN/PI3K/AKT pathway.
The present study has limitations. The findings of
the present study are completely based on in vitro
experiments using the estrogen receptor-positive MCF-7 cell line.
Consequently, the general applicability of this mechanism to other
BC subtypes, particularly triple-negative BC, remains to be
investigated. Furthermore, the mechanisms identified here need to
be validated in more complex systems. A key future direction will
be to address the lack of in vivo data and to assess
essential translation parameters, such as a central role for the
PTEN/PI3K/AKT axis in the potential of Que to prevent
chemoresistance in BC treatment. Having established this core
mechanism, future work will explore the broader signaling landscape
and systematically investigate the interplay with other key
pathways such as MAPK and Wnt/β-catenin to uncover potential
synergistic nodes for overcoming multi-pathway driven
resistance.
In conclusion, the present study revealed a novel
PTEN/PI3K/AKT/ABC transporter axis as a mechanism through which Que
may counteract MDR. By functionally associating the suppression of
this pathway to the enhanced intracellular accumulation of
chemotherapeutic drugs, the present study provides an explanation
for the efficacy of Que. These findings highlight the dual-acting
potential of Que, targeting both cell survival and drug efflux
pathways, and support its further investigation as a potential
adjunctive agent to re-sensitize BC cells to conventional
chemotherapy.
Acknowledgements
Not applicable.
Funding
This work was supported by the Natural Science Foundation of
Fujian Province, Fujian, China (grant no. 2024J011398), the Fujian
Province Science and Technology Innovation Joint Fund Project,
Fujian, China (grant no. 2024Y9712), the Xiamen Science and
Technology Bureau Foundation of Science and Technology Project for
Medicine and Health, Xiamen, China (grant no. 3502Z20244ZD1139) and
college students' innovation and entrepreneurship program of Xiamen
Medical College (grant nos. 202512631044, 202512631046 and
202512631064).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
WF contributed to methodology and conceptualization.
LL contributed to methodology and investigation. JL contributed to
data curation and investigation. FQ contributed to investigation
and formal analysis. CC contributed to visualization and
investigation. YinCai contributed to investigation and data
curation. YilCai contributed to methodology and investigation. YH
contributed to methodology. WY contributed to Investigation. SZ
contributed to reviewing and editing, writing the original draft,
validation, methodology, data curation, conceptualization and
project administration.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Filho AM, Laversanne M, Ferlay J, Colombet
M, Piñeros M, Znaor A, Parkin DM, Soerjomataram I and Bray F: The
GLOBOCAN 2022 cancer estimates: Data sources, methods, and a
snapshot of the cancer burden worldwide. Int J Cancer.
156:1336–1346. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal J: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
3
|
Wang LB, Zhao JF, Cao J, Tao X, Wang J, Ma
JR, Huang Q, Yu JJ, Tian JH and Zhang QH: Integrative analysis of
differentially expressed mRNAs and proteins induced by PGC-1β in
breast cancer cells. Biochem Biophys Res Commun. 637:73–82. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jiang YZ, Ma D, Suo C, Shi JX, Xue MZ, Hu
X, Xiao Y, Yu KD, Liu YR, YU Y, et al: Genomic and transcriptomic
landscape of triple-negative breast cancers: Subtypes and treatment
strategies. Cancer Cell. 35:428–440.e5. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Muriithi W, Macharia LW, Heming CP,
Echevarria JL, Nyachieo A, Filho PN and Neto VM: ABC transporters
and the hallmarks of cancer: Roles in cancer aggressiveness beyond
multidrug resistance. Cancer Biol Med. 17:253–269. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dean M, Rzhetsky A and Allikmets R: The
human ATP-binding cassette (ABC) transporter superfamily. Genome
Res. 42:1156–1166. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu X: ABC family transporters. Adv Exp
Med Biol. 1141:13–100. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Goda K, Bacsó Z and Szabó G: Multidrug
resistance through the spectacle of P-glycoprotein. Curr Cancer
Drug Targets. 9:281–297. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wegler C, Gazit M, Issa K, Subramaniam S,
Artursson P and Karlgren M: Expanding the efflux in vitro assay
toolbox: A CRISPR-Cas9 edited MDCK cell line with human BCRP and
completely lacking canine MDR1. J Pharm Sci. 110:388–396. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Almohammad Aljabr B, Zihlif M, Abu-Dahab R
and Zalloum H: Effect of quercetin on doxorubicin cytotoxicity in
sensitive and resistant human MCF7 breast cancer cell lines. Biomed
Rep. 20:582024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tang H, Kuang Y, Wu W, Peng B and Fu Q:
Quercetin inhibits the metabolism of arachidonic acid by inhibiting
the activity of CYP3A4, thereby inhibiting the progression of
breast cancer. Mol Med. 29:1272023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Safi A, Heidarian E and Ahmadi R:
Quercetin synergistically enhances the anticancer efficacy of
docetaxel through induction of apoptosis and modulation of
PI3K/AKT, MAPK/ERK, and JAK/STAT3 signaling pathways in MDA-MB-231
breast cancer cell line. Int J Mol Cell Med. 10:11–22.
2021.PubMed/NCBI
|
|
13
|
Kıyga E, Şengelen A, Adıgüzel Z and Önay
Uçar E: Investigation of the role of quercetin as a heat shock
protein inhibitor on apoptosis in human breast cancer cells. Mol
Biol Rep. 47:4957–4967. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Thies KA, Lefler JE, Leone G and Ostrowski
MC: PTEN in the Stroma. Cold Spring Harb Perspect Med.
9:a0361112019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lefler JE, Seward C and Ostrowski MC: PTEN
in cancer associated fibroblasts. Adv Cancer Res. 154:203–226.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bhave MA, Quintanilha JCF, Tukachinsky H,
Li G, Scott T, Ross JS, Pasquina L, Huang RSP, McArthur H, Levy MA,
et al: Comprehensive genomic profiling of ESR1, PIK3CA, AKT1, and
PTEN in HR(+)HER2(-) metastatic breast cancer: Prevalence along
treatment course and predictive value for endocrine therapy
resistance in real-world practice. Breast Cancer Res Treat.
207:599–609. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ngeow J, Stanuch K, Mester JL,
Barnholtz-Sloan JS and Eng C: Second malignant neoplasms in
patients with Cowden syndrome with underlying germline PTEN
mutations. J Clin Oncol. 32:1818–1824. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Molina-Montes E, Requena M,
Sánchez-Cantalejo E, Fernández MF, Arroyo-Morales M, Espín J,
Arrebola JP and Sánchez MJ: Risk of second cancers cancer after a
first primary breast cancer: A systematic review and meta-analysis.
Gynecol Oncol. 136:158–171. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pote M and Gacche RN: ATP-binding cassette
efflux transporters and MDR in cancer. Drug Discov Today.
28:1035372023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Garrido-Castro AC, Lin NU and Polyak K:
Insights into molecular classifications of triple-negative breast
cancer: Improving patient selection for treatment. Cancer Discov.
9:176–198. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gu J, Huang W, Wang X, Zhang J, Tao T,
Zheng Y, Liu S, Yang J, Chen ZS, Cai CY, et al:
Hsa-miR-3178/RhoB/PI3K/Akt, a novel signaling pathway regulates ABC
transporters to reverse gemcitabine resistance in pancreatic
cancer. Mol Cancer. 21:1122022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qiu F, Chen J, Cao J, Diao F and Huang P:
Low-intensity low-frequency ultrasound enhances the
chemosensitivity of gemcitabine-resistant ASPC-1 cells via
PI3K/AKT/NF-κB pathway mediated ABC transporters. Oncol Rep.
44:1158–1168. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sui H, Pan SF, Feng Y, Jin BH, Liu X, Zhou
H, Hou FG, Wang WH, Fu XL, Han ZF, et al: Zuo Jin Wan reverses
P-gp-mediated drug-resistance by inhibiting activation of the
PI3K/Akt/NF-κB pathway. BMC Complement Altern Med. 14:2792014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dong W, Liao ZG, Zhao GW, Guan XJ, Zhang
J, Liang XL and Yang M: Reversal effect of oxypeucedanin on
P-glycoprotein-mediated drug transport. Molecules. 23:18412018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ryan Q, Ibrahim A, Cohen MH, Johnson J, Ko
CW, Sridhara R, Justice R and Pazdur R: FDA drug approval summary:
Lapatinib in combination with capecitabine for previously treated
metastatic breast cancer that overexpresses HER-2. Oncologist.
13:1114–1119. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bao X, Lu S, Liow JS, Morse CL, Anderson
KB, Zoghbi SS, Innis RB and Pike VW: [¹¹C]Rhodamine-123:
Synthesis and biodistribution in rodents. Nucl Med Biol.
39:1128–1136. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bronte G, Andrei Ds, Bravaccini S, Maltoni
R, Cecconetto L, Schirone A, Farolfi A, Fedeli A, Serra P, Donati
C, et al: Sorafenib for the treatment of breast cancer. Expert Opin
Pharmacother. 18:621–630. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ren MX, Deng XH, Ai F, Yuan GY and Song
HY: Effect of quercetin on the proliferation of the human ovarian
cancer cell line SKOV-3 in vitro. Exp Ther Med. 10:579–583. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang P, Henning SM, Magyar CE, Elshimali
Y, Heber D and Vadgama JV: Green tea and quercetin sensitize PC-3
xenograft prostate tumors to docetaxel chemotherapy. J Exp Clin
Cancer Res. 35:732016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kundur S, Prayag A, Selvakumar P, Nguyen
H, McKee L, Cruz C, Srinivasan A, Shoyele S and Lakshmikuttyamma A:
Synergistic anticancer action of quercetin and curcumin against
triple-negative breast cancer cell lines. J Cell Physiol.
237:11103–11118. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gao X, Qin T, Mao J, Zhang J, Fan S, Lu Y,
Sun Z, Zhang Q, Song B and Li L: Correction: PTENP1/miR-20a/PTEN
axis contributes to breast cancer progression by regulating PTEN
via PI3K/AKT pathway. J Exp Clin Cancer Res. 42:2022023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Qian YY, Li K, Liu QY and Liu ZS: Long
non-coding RNA PTENP1 interacts with miR-193a-3p to suppress cell
migration and invasion through the PTEN pathway in hepatocellular
carcinoma. Oncotarget. 8:107859–107869. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cretella D, Digiacomo G, Giovannetti E and
Cavazzoni A: PTEN alterations as a potential mechanism for tumor
cell escape from PD-1/PD-L1 inhibition. Cancers (Basel).
11:13182019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huang FF, Zhang L, Wu DS, Yuan XY, Yu YH,
Zhao XL, Chen FP and Zeng H: PTEN regulates BCRP/ABCG2 and the side
population through the PI3K/Akt pathway in chronic myeloid
leukemia. PLoS One. 9:e882982014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Miricescu D, Balan DG, Tulin A, Stiru O,
Ivacaroiu IA, Mihai DA, Popa CC, Papacoce RL, Enyedi M, Sorin NA,
et al: PI3K/AKT/mTOR signalling pathway involvement in renal cell
carcinoma pathogenesis (review). Exp Ther Med. 21:5402021.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Choi HJ, Heo JH, Park JY, Jeong JY, Cho
HJ, Park KS, Kim SH, Moon YW, Kim JS and An HJ: A novel PI3K/mTOR
dual inhibitor, CMG002, overcomes the chemoresistance in ovarian
cancer. Gynecol Oncol. 153:135–148. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X: Transporter-mediated drug-drug
interactions and their significance In: Drug Transporters in Drug
Disposition. Effects and Toxicity. Advances in Experimental
Medicine and Biology. Liu X and Pan G: Vol 1141. Springer;
Singapore: pp. 241–291. 2019, View Article : Google Scholar : PubMed/NCBI
|