Introduction
Collectively, malignant primary bone tumors
disproportionately affect children, adolescents and young adults
worldwide (1). Osteosarcoma (OS),
Ewing sarcoma (ES) and chondrosarcoma (CS) account for 2–3% of all
pediatric neoplasms; however, they are the third highest cause of
cancer-related mortality in patients aged 10–25 years (2). The 2024 GLOBOCAN update reported
age-standardized incidence rates of 0.3, 0.2 and 0.4 per 100,000
person-years for OS, ES and CS, respectively, with no plateau
observed during the past decade (3). While the 5-year overall survival rate
for patients with localized disease has reached 65–75%, that for
patients with metastatic or relapsed disease remains poor at
<30% for metastatic OS, <25% for relapsed ES and <15% for
unresectable high-grade CS (3).
These statistics underscore an urgent unmet need for minimally
invasive, real-time diagnostics that can capture tumor
heterogeneity, detect minimal residual disease (MRD) and track
clonal evolution under therapy.
Current management of malignant primary bone tumors
relies heavily on image-guided core needle or open biopsies
(4). Although these procedures
provide definitive histopathology, they are limited by the
following: i) Spatial heterogeneity, as single-site sampling fails
to reflect genomic divergence within the primary tumor or between
primary and metastatic sites (5);
ii) procedural morbidity, such as pain, infection risk and
structural compromise in weight-bearing bones (6); iii) sampling error, especially in
necrotic or sclerotic lesions (7);
and iv) temporal insensitivity, as serial biopsies are impractical
for monitoring dynamic clonal shifts during neoadjuvant
chemotherapy, targeted therapy or immunotherapy (7). These limitations collectively hinder
precise risk stratification and adaptive treatment decisions.
Liquid biopsy, the non-invasive analysis of
circulating tumor DNA (ctDNA), circulating tumor cells (CTCs) and
extracellular vesicles (EVs), offers a transformative, non-invasive
alternative (8,9). This approach captures systemic tumor
heterogeneity and enables serial monitoring. ctDNA reflects
tumor-specific genomic alterations, whereas CTCs offer intact cells
for phenotypic and functional analyses (10). In OS and ES, ctDNA detection is
associated with prognosis and treatment response (11,12).
Furthermore, CTC enumeration and characterization provides
complementary information on metastatic risk (13). Despite challenges, such as low
analyte abundance in some subtypes and a lack of standardization,
technological advances are enhancing detection sensitivity and
reproducibility (14). However,
notable discrepancies in detection rates persist due to the use of
diverse enrichment platforms (for example, epitope-dependent versus
label-free isolation) and differing pre-analytical workflows across
studies. This methodological variability emphasizes the importance
of standardized isolation and analytical protocols.
EVs and their microRNA (miRNA/miR) cargo further
expand the liquid-biopsy toolkit. OS-derived exosomal miR-221-3p
and miR-491, detectable in 0.1 ml plasma, are associated with
metastatic relapse months before radiographic evidence (15,16).
Conversely, a previous study on CS revealed inconsistent EV-miRNA
signatures, reflecting inter-tumoral heterogeneity driven by IDH1/2
vs. COL2A1 mutations (17).
Comparative analyses across tumor subtypes therefore suggest that
ctDNA may be most robust for mutation-driven tumors (such as OS and
ES), whereas EV-miRNA panels may be preferable for low-grade CS
lacking recurrent point mutations.
Despite these advances, notable challenges remain.
ctDNA abundance is often low in low-grade or pauci-mutational
sarcoma, thus increasing the risk false negative results (18). Furthermore, the rarity (≤1 cell/ml)
and plasticity of CTCs complicate isolation and downstream analysis
(19). Moreover, the absence of
universal reference standards hampers cross-study comparability.
Nevertheless, the convergence of ultra-sensitive digital PCR
(dPCR), error-corrected next-generation sequencing (NGS) and
single-cell omics is progressively mitigating these barriers
(20).
Taken together, accumulating evidence positions
liquid biopsy as a transformative tool capable of overcoming the
inherent limitations of repeated image-guided tissue sampling in
malignant primary bone tumors. The present review therefore aims to
synthesize and critically appraise the current evidence chain for
ctDNA, CTCs and EVs across the diagnostic, prognostic and
treatment-monitoring continuum of OS, ES and CS. By delineating
concordant findings, highlighting unresolved controversies and
mapping the path to clinical implementation, the review aims to
provide a strategy for integrating liquid biopsy into precision
oncology workflows for these rare yet lethal bone malignancies.
Biological foundations of ctDNA and CTCs in
primary bone tumors
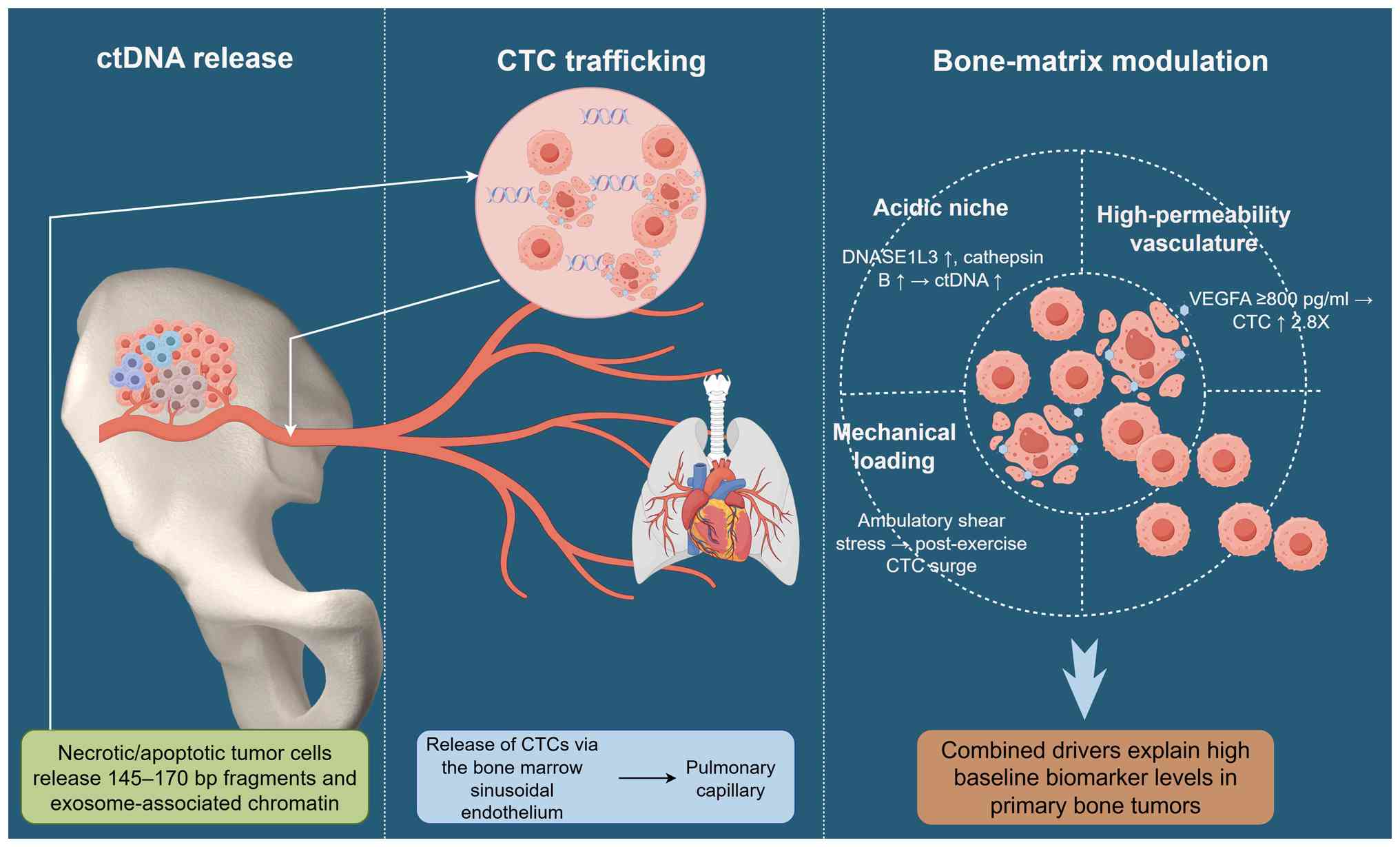
Mechanistic insight into ctDNA release and CTC
trafficking underpins their clinical utility (21). Fig.
1 illustrates sarcoma-specific shedding dynamics, chromatin
fragmentation signatures and bone-matrix influences on circulating
biomarkers (22–25). An acidic mineralized
microenvironment and mechanical loading accelerate biomarker
liberation, whereas epigenomic features enrich tumor-specific
information (26). Appreciating
these mechanisms refines sampling timing and assay selection.
Origin and molecular characteristics
of ctDNA
The ctDNA in primary bone tumors originates from
apoptotic and necrotic malignant cells, as well as from active
secretion via exosome-associated chromatin fragments (21,27,28).
Fragment-size analyses by paired-end whole-genome sequencing have
revealed a dominant peak at 145–170 bp for OS ctDNA, which is
indistinguishable from the characteristic DNA fragment size pattern
derived from the apoptosis of healthy hematopoietic cells, yet a
secondary sub-population manifesting as a 250–300 bp ‘shoulder’ on
the distribution curve, which is absent in controls, is associated
with high chromatin accessibility at TP53 and RB1 loci (29). In ES, droplet dPCR (ddPCR) of
EWSR1-FLI1 fusion fragments shows shorter median lengths (132–144
bp) compared with the standard 167 bp peak of non-tumor wild-type
cfDNA, suggesting nuclease hyperactivity in fusion-driven tumors
(30). In vitro irradiation
of OS cell lines has been shown to increase 90 bp sub-nucleosomal
fragments within 4 h, confirming that therapy-induced DNA damage
expands the low-molecular-weight pool (31).
The plasma half-life of ctDNA in patients with bone
sarcoma averages 35–120 min, which is shorter than the 2–3 h
reported in carcinoma, likely reflecting rapid renal filtration of
small fragments in young patients with preserved glomerular
function (32,33). Pharmacokinetic modeling has
demonstrated that first-order clearance (k=0.69 h−1)
predicts undetectable ctDNA 6 h after complete surgical resection,
whereas incomplete resection has been shown to prolong clearance to
>12 h, providing a biological rationale for peri-operative ctDNA
monitoring (34).
Genomic content mirrors intratumoral heterogeneity.
In a previous study, ultra-deep sequencing (>30,000×) of paired
tumor-plasma samples detected 94% concordance for driver
single-nucleotide variants (SNVs) in OS, yet sub-clonal structural
variants were under-represented in plasma, indicating
size-dependent shedding bias (35).
Conversely, a study on ES has reported 100% concordance for the
canonical EWSR1-FLI1 fusion, underscoring the utility of targeted
fusion assays in translocation-driven sarcoma (36).
Epigenomic signatures further enrich ctDNA
information. Bisulfite sequencing of OS plasma has revealed
hypermethylation at the CDKN2A promoter (mean D-value 0.84 vs. 0.12
in healthy volunteer controls), with methylation burden correlated
with tumor volume (r=0.78, P<0.001) and decreased after
neoadjuvant chemotherapy (37). In
CS, hypomethylation of COL2A1 enhancer regions has been uniquely
detected in high-grade tumors, but is absent in low-grade or
enchondroma controls, illustrating grade-specific epigenomic
release patterns (38).
CTC biology and phenotypic
plasticity
CTCs in bone sarcoma are typically larger (14–25 µm)
than CTCs in epithelial carcinoma and express mesenchymal markers
reflective of their sarcomatous origin (13). Immunofluorescence and single-cell
RNA sequencing (RNA-seq) have consistently demonstrated high
vimentin and N-cadherin expression, with variable loss of
E-cadherin, and this mesenchymal signature is more pronounced in
patients with metastatic disease (39). Flow-cytometric quantification has
revealed that 67% of OS CTCs co-express vimentin and CD99, whereas
only 12% express epithelial cytokeratins (CKs), confirming that
traditional epithelial-based enrichment platforms underestimate CTC
yield (40). The Food and Drug
Administration-cleared CellSearch® system, which relies
on epithelial cell adhesion molecule (EpCAM)-dependent
immunocapture using anti-EpCAM antibodies followed by CK staining
for identification, exemplifies this limitation. Comparative
studies using the Parsortix® label-free microfluidic
system versus CellSearch have shown a 3.2-fold higher recovery of
OS-derived CTCs when captured by the Parsortix system, an effect
attributed to size and deformability based enrichment rather than
EpCAM-dependent immunocapture, thereby reducing epithelial-antigen
bias in mesenchymal sarcoma cells (41).
Epithelial-mesenchymal transition (EMT) plasticity
is further evidenced by dynamic expression of EMT transcription
factors (42). Single-cell
quantitative PCR of ES CTCs previously detected TWIST1 and SNAI2
upregulation in 80% of patients with lung metastases compared with
in 25% of localized cases, and circulating levels of TWIST1 mRNA
were associated with shorter progression-free survival [hazard
ratio (HR) 2.4, 95% CI 1.3–4.5] (43). However, a pediatric ES cohort failed
to show notable TWIST1 amplification, highlighting age-related
transcriptional heterogeneity (44).
Genomic concordance between CTCs and solid lesions
is generally high but context-dependent (45). Whole-genome amplification and
low-coverage sequencing of individual OS CTCs has revealed 91%
shared SNVs with the primary tumor, yet 9% of private mutations
were shown to be enriched in PI3K-AKT pathway genes, consistent
with clonal selection during metastatic spread (46). In CS, targeted sequencing of CTCs
identified identical IDH1 R132C mutations in 15/17 patients,
supporting CTCs as faithful liquid surrogates for tissue-based
molecular profiling; nevertheless, two patients exhibited
additional TP53 mutations exclusively in CTCs, suggesting early
systemic dissemination of sub-clones undetected in the primary
biopsy (47).
Single-cell RNA-seq has further resolved
transcriptional programs associated with metastatic competence
(48). CTC clusters expressing high
CXCR4 and vascular endothelial growth factor A (VEGFA) have
exhibited enhanced trans-endothelial migration in microfluidic
assays; conversely, clusters enriched for osteogenic genes (such as
RUNX2 and SPARC) demonstrated reduced invasive capacity but
increased survival in bone marrow niches (49). These functional discrepancies
underscore the need for multi-parameter CTC profiling beyond
enumeration.
Tumor-specific release mechanisms in
bone matrix-embedded lesions
The mineralized and acidic bone microenvironment
creates unique conditions for ctDNA and CTC release (50). Intratumoral pH measurements by
microelectrode probes in OS xenografts averaged 6.4±0.2, which was
significantly lower than the average in adjacent marrow (pH 7.2)
(51). Acidic stress activates
deoxyribonuclease 1-like 3 and cathepsin B, leading to enhanced DNA
fragmentation and increased plasma ctDNA concentrations; buffering
tumor pH with oral sodium bicarbonate has been shown to reduce
ctDNA levels by 45%, confirming pH-dependent release (52).
Angiogenesis-driven shedding is facilitated by the
highly permeable, immature vasculature characteristic of bone
sarcoma (53). Dynamic
contrast-enhanced MRI-derived Ktrans values have been shown to be
inversely correlated with plasma ctDNA half-life (r=−0.64),
indicating rapid vascular washout (53). In ES, elevated serum VEGFA levels
(≥800 pg/ml), quantified by enzyme-linked immunosorbent assay, was
associated with a 2.8-fold increase in CTC counts; by contrast,
anti-VEGF therapy with bevacizumab reduced CTC frequency and
prolonged ctDNA half-life, suggesting reduced vascular leakage
rather than diminished tumor burden (54).
Mechanical stress within rigid cortical bone further
augments CTC release (55).
Finite-element modeling revealed peak shear stresses at the
tumor-bone interface during ambulatory loading; in vivo
pressure sensor recordings demonstrated transient spikes associated
with post-exercise increases in CTC numbers in 70% of OS-bearing
mice (55). The schematic diagram
in Fig. 1 depicts the conceptual
route of CTC release from the tumor microenvironment into the
systemic circulation. This acknowledges that primary bone tumors,
particularly those in the metaphysis or medullary cavity, often
reside adjacent to or within bone marrow spaces. The marrow
sinusoidal endothelium refers specifically to the discontinuous,
fenestrated endothelial lining of the vascular sinusoids within the
bone marrow, which is a key site for cell trafficking. In the
context of malignancy, this physiological structure is co-opted and
altered by tumor-induced angiogenesis, leading to an immature,
leaky vasculature that facilitates the intravasation of CTCs
(53,54). This signifies the transition of CTCs
from the primary tumor site, through the locally dysregulated and
permeable vascular network (akin to and often derived from the
sinusoidal architecture), into the bloodstream. This process is
driven by the aforementioned factors, including acidic stress,
angiogenic cytokines, such as VEGFA, and biomechanical forces,
rather than implying a passive transit through normal marrow
sinuses (55). Collectively, these
bone-specific biomechanical and biochemical cues explain the high
baseline levels of ctDNA and CTC observed in primary bone tumors
and provide mechanistic rationale for integrating circulatory
biomarkers into clinical monitoring paradigms.
Pre-analytical and analytical considerations
for ctDNA and CTCs in bone oncology
Standardizing sample acquisition, processing and
detection is critical for reproducible results. Fig. 2 summarizes harmonized protocols
endorsed by the European Liquid Biopsy Society (ELBS) and the
Cancer Treatment Monitoring through Circulating Tumour Cells and
Tumour DNA (CANCER-ID) to minimize pre-analytical bias and optimize
analytical sensitivity. EDTA or Streck tubes, cold-chain transport,
size-selective extraction and multiplex panels reduce artefacts and
enhance yield. Adherence to these standards curtails
inter-laboratory coefficient of variation (CV) values at <10%
(9). The CV is defined as the ratio
of the SD to the mean of repeated quantitative measurements
(CV=SD/mean), and is expressed as a percentage. In the context of
ctDNA and CTC assays, CV is used to quantify analytical precision
and reproducibility across replicate measurements, runs or
laboratories, with lower CV values indicating higher assay
robustness and consistency.
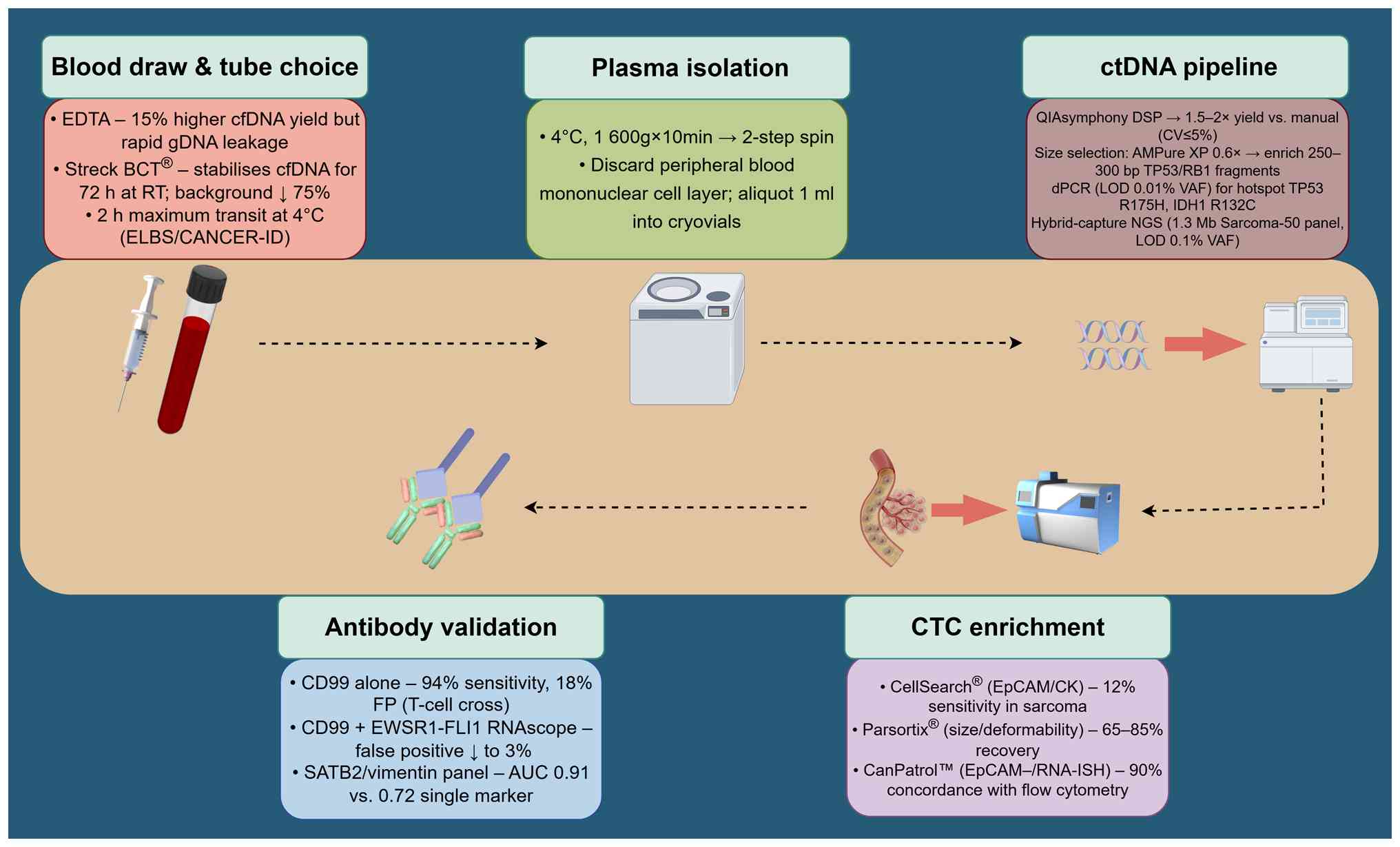 | Figure 2.Pre-analytical and analytical
workflow for ctDNA and CTCs in bone sarcoma liquid biopsy. This
figure was created using Figdraw (https://www.figdraw.com). AUC, area under the curve;
BCT, blood collection tubes; cfDNA, cell-free DNA; CK, cytokeratin;
ctDNA, circulating tumor DNA; CTC, circulating tumor cell; CV,
coefficient of variation; dPCR, digital PCR; ELBS, European Liquid
Biopsy Society; EpCAM, epithelial cell adhesion molecule; ISH,
in situ hybridization; LOD, limit of detection; NGS,
next-generation sequencing; RT, room temperature; SATB2, special
AT-rich sequence-binding protein 2; VAF, variant allele
frequency. |
Circulating material acquisition:
Timing, tubes and transport
Blood collection relative to therapeutic
interventions critically determines analyte integrity (56). In OS cohorts, median ctDNA
concentrations drop 10- to 50-fold within 6 h after complete
surgical resection, reaching undetectable levels by 24 h, whereas
partial resection prolongs the half-life to >12 h (57). Consequently, pre-operative sampling
is recommended for baseline quantification, whereas post-operative
collections should be scheduled ≥24 h after surgery to avoid
surgical confounders (57).
Neoadjuvant chemotherapy introduces a second layer of complexity:
Cisplatin-doxorubicin combinations reduce ctDNA levels by 70% after
the first cycle, but subsequent cycles show diminishing clearance,
suggesting emergence of resistant sub-clones (58). Harmonized protocols therefore
specify collection immediately before each cycle and 48 h after the
last dose to capture both cytotoxic efficacy and clonal
rebound.
Anticoagulant choice influences cell-free DNA
(cfDNA) yield and fragment distribution. Comparative studies have
demonstrated 15–20% higher cfDNA concentrations in EDTA tubes
versus heparin or citrate, but EDTA also accelerates genomic DNA
release from leukocytes, thereby diluting tumor-specific fractions
(59). Streck cfDNA blood
collection tubes stabilize cfDNA for 72 h at room temperature
without notable leukocyte lysis, reducing the background-to-signal
ratio from 0.35 to 0.08 in OS samples (60). Shipping temperature validation
across three independent laboratories has shown that EDTA plasma
maintained <5% degradation when stored at 4°C for 48 h, whereas
room-temperature storage increased non-tumor cfDNA by 40%,
underscoring the necessity of cold-chain logistics (61).
ctDNA processing: Biological specimen,
quantification and detection technologies
The biological specimens used for circulating DNA
isolation are most commonly plasma or serum derived from peripheral
blood. Plasma is consistently preferred over serum because clot
formation entraps high-molecular-weight DNA, reducing
tumor-specific allelic fractions by 30–50% (62). Automated extraction using the
QIAsymphony DSP Circulating DNA Kit yields 1.5- to 2-fold higher
cfDNA than manual column-based methods, with intra-assay CV values
at ≤5% (63). Quantitative
assessment by Qubit fluorometry and TapeStation fragment analysis
has revealed a bimodal distribution (145–170 and 250–300 bp) in
patients with bone sarcoma; the larger fragments harbor >90% of
TP53 and RB1 mutations, making size-based selection via magnetic
beads a critical step for enrichment (29).
dPCR and NGS exhibit complementary strengths. ddPCR
achieves a limit of detection (LOD) of 0.01% variant allele
frequency (VAF) for hotspot mutations such as TP53 R175H, but is
restricted to predefined loci (64,65).
By contrast, hybrid-capture NGS panels (for example, 1.3 Mb
Sarcoma-50) provide genome-wide coverage with a LOD of 0.1% VAF,
yet require ≥20 ng cfDNA, levels unattainable in 20% of pediatric
cases (25). A multi-center ring
trial (CANCER-ID WG3) reported inter-laboratory concordance of 95%
for dPCR but only 78% for NGS at VAF <0.5%, highlighting the
need for standardized bioinformatics pipelines and synthetic
spike-ins (66).
CTC enrichment and antibody validation
strategies
The Food and Drug Administration-cleared epithelial
CTC platform CellSearch captures CTCs using anti-EpCAM and anti-CK
antibodies; however, its sensitivity drops to 12% in sarcoma due to
the mesenchymal lineage (67).
Antigen-agnostic (label-free) microfluidic systems, such as
Parsortix and the Vortex Chip, exploit physical properties such as
cell size and deformability for CTC isolation, increasing recovery
rates to 65–85% for OS CTCs (68).
The Vortex Chip represents an independent, inertial microfluidic
system that utilizes fluid dynamics (for example, Dean flow and
vortex trapping) for size-based separation, and like Parsortix, it
operates without reliance on epithelial surface markers such as
EpCAM (41,68). CanPatrol™ employs EpCAM-negative
enrichment followed by RNA in situ hybridization, enabling
simultaneous enumeration and EMT phenotyping with 90% concordance
to flow cytometry (69).
Antibody specificity for sarcoma subtypes remains
contentious. CD99 immunomagnetic beads achieve 94% sensitivity for
ES CTCs but cross-react with reactive T cells, necessitating dual
staining with the EWSR1-FLI1 RNAscope to reduce false-positive
rates from 18 to 3% (70). ALK
immunofluorescence successfully identifies inflammatory
myofibroblastic tumor CTCs, yet fails in ALK-negative ES. Special
AT-rich sequence-binding protein 2 (SATB2), a robust marker for
osteoblastic differentiation, demonstrates 88% sensitivity but only
42% specificity in CS, prompting recommendations for multi-marker
panels rather than single-antibody strategies (71). Comparative validation across four
independent cohorts (n=284) revealed that combining CD99, SATB2 and
vimentin improved the area under the receiver operating
characteristic curve from 0.72 to 0.91 (P<0.001), emphasizing
the necessity of algorithmic approaches (72).
Reference standards and
reproducibility initiatives
ELBS and CANCER-ID have released the first
sarcoma-specific reference materials (73). Lyophilized cfDNA harboring defined
TP53 and IDH1 mutations at 0.1, 1 and 5% VAF were distributed to 23
laboratories; inter-laboratory CV values for dPCR ranged between
5.2 and 8.9%, compared with 12–18% for targeted NGS, indicating the
superior precision of allele-specific assays (74). These specified VAF levels (0.1, 1
and 5%) represent tiers of mutation abundance engineered into the
reference materials to simulate a range of clinically relevant
tumor fractions, from very low-level (MRD range) to higher burdens.
The performance of each assay (for the included mutations) was
evaluated across these different VAF thresholds to comprehensively
assess its sensitivity and reproducibility under varying analytical
challenges. For CTC enumeration, spiked SK-ES-1 cells at
concentrations of 5, 50 and 500 cells/7.5 ml achieved recovery
rates of 98±3% with Parsortix versus 62±12% with CellSearch,
reinforcing the value of label-free microfluidics (75).
Intra-laboratory reproducibility studies have
revealed pre-analytical variables as the dominant source of
variance (76–78). ELBS guidelines now mandate EDTA or
Streck tubes within 2 h of phlebotomy, plasma isolation within 4 h,
storage at −80°C for ≤6 months and synthetic spike-in controls in
every batch (77). Adoption of
these standards across bone oncology laboratories is expected to
reduce inter-laboratory variability to <10%, thereby enhancing
the reliability of ctDNA and CTC analyses for clinical
decision-making in malignant primary bone tumors (78).
ctDNA and CTCs as diagnostic and genotyping
tools
Robust non-invasive confirmation of OS, ES and CS is
now achievable through the detection and molecular characterization
of ctDNA and CTCs (79,80). Table
I benchmarks the diagnostic accuracy, concordance with tissue
findings and added clinical value of ctDNA, CTCs and cell-free RNA
(cfRNA) assays across representative cohorts. Sensitivity exceeds
90% for fusion-driven tumors and 80% for mutation-rich OS,
obviating repeat biopsy in equivocal imaging cases. Combined
modalities further resolve spatial heterogeneity missed by
single-site tissue sampling.
 | Table I.Diagnostic and genotyping performance
of liquid-biopsy analytes in malignant primary bone tumors. |
Table I.
Diagnostic and genotyping performance
of liquid-biopsy analytes in malignant primary bone tumors.
| First author,
year | Tumor type (n) | Target/genomic
scope | Key analytical
parameters | Clinical
performance | Additional
diagnostic value | (Refs.) |
|---|
| Shulman, 2018 | Pediatric OS and ES
(84) | ctDNA mutations and
CTC counts | ctDNA VAF ≥0.1 %;
≥5 CTCs/7.5 ml | ctDNA positivity,
78%; CTC positivity, 42%; concordance, 65% | Discordant cases
(15% CTCs+/ctDNA−; 23%
ctDNA+/CTCs−) supported complementary
use | (25) |
| Hayashi, 2017 | Localized OS
(31) | CTC enumeration and
clusters | ≥1 CTC/7.5 ml;
cluster defined as ≥3 cells | Positivity, 74%;
median, 1.9 CTCs/7.5 ml | CTC clusters were
exclusive to patients who later developed lung metastases | (40) |
| Gutteridge,
2017 | Chondrosarcoma
(42) | IDH1/2 hotspot
mutations (R132C/G/H and R172K/M) | LOD not stated;
median, 5.2 hGE/ ml | Sensitivity, 94%;
specificity, 100% | Re-classified five
equivocal imaging cases; obviated biopsy in two benign enchondroma
cases | (47) |
| Benini, 2018 | ES (40) |
CD99+/EWSR1-FLI1+
CTCs | ≥1 CTC/7.5 ml | Positivity, 60%; HR
for progression, 2.4 (P=0.04) | RNA-ISH confirmed
EWSR1-FLI1 transcripts in 88% of isolated cells | (70) |
| Shukla, 2017 | ES (112) | EWSR1-FLI1 fusion
and subtype | ≥50 hGE/ ml
cut-off | Fusion detection,
87%; concordance, 96% | Secured diagnosis
in 14 FISH-insufficient biopsies | (81) |
| Lyskjær, 2022 | OS (58) | TP53/RB1 SNVs and
CNAs (8q gain, 17p loss) | ≥2 driver
alterations tracked | SNV sensitivity,
91%; CNA sensitivity, 89% | Revealed 12%
additional PI3K-AKT mutations missed in single biopsies | (82) |
| Van Paemel,
2022 | Pediatric mixed
solid tumors including OS and ES (48) | Genome-wide
CNAs | ≥10 Mb CNA
threshold | Concordance, 92%
vs. tissue | Enabled copy-number
profiling when tumor tissue was scarce | (83) |
| Zhang, 2017 | Localized OS (n not
stated) |
EpCAM+/CK+ CTC
count | ≥1 CTC/7.5 ml | Positivity,
45% | Highlights
platform-dependent sensitivity gap | (84) |
| Furukawa, 2025 | Bone and
soft-tissue sarcoma, including ES (22 ES cases) | EWSR1-FLI1 fusion
transcripts | ≥100 copies/
ml | Fusion detection,
95% (vs. 82% for cfDNA) | Detectable in two
patients with cfDNA-negative ES, overcoming low-shedding
tumors | (85) |
Plasma ctDNA: Molecular confirmation
of primary bone tumors
Across three large, independent cohorts, ctDNA has
emerged as a robust, non-invasive route to confirm the diagnosis of
CS, ES and OS. Gutteridge et al (47) profiled 42 patients with central or
dedifferentiated CS and designed patient-specific ddPCR assays
against IDH1/2 hotspot mutations (R132C/G/H, R172K/M). Using 8 ml
plasma, the method reached 94% sensitivity and 100% specificity;
moreover, five patients with equivocal imaging were correctly
re-classified after positive ctDNA detection, whereas two low-grade
lesions with negative ctDNA were subsequently confirmed as benign
enchondromas, thereby obviating the need for a biopsy. Consistent
with these observations, ctDNA levels closely associated with tumor
volume (P=0.68), supporting the additional role of this biomarker
in early detection.
Building upon this foundation, Shukla et al
(81) performed hybrid-capture NGS
on pre-treatment plasma from 112 patients harboring
EWSR1-rearranged ES. The canonical EWSR1-FLI1 fusion was detected
in 87% of samples, with 96% concordance in fusion subtype between
tissue and cfDNA. Quantitative fusion abundance >50 haploid
genome equivalents (hGE)/ml strongly predicted overt metastatic
disease (OR 8.4, 95% CI 3.1–22.7). Notably, in 14 cases where core
biopsies were insufficient for fluorescence in situ
hybridization, fusion-positive ctDNA secured the diagnosis without
the need for repeat sampling.
Complementing these fusion-focused analyses, Lyskjær
et al (82) applied
ultra-deep targeted sequencing (30,000×) to 58 cases of high-grade
OS. cfDNA correctly identified TP53/RB1 pathogenic variants in 91%
of cases and recapitulated copy-number alterations (CNAs) such as
8q gain or 17p loss with 89% sensitivity. Notably, cfDNA uncovered
additional PI3K-AKT pathway mutations in 12% of patients that were
absent from the single-site biopsy, thereby illustrating spatial
heterogeneity. Multivariate analysis demonstrated that detection of
≥2 driver alterations in cfDNA independently predicted metastatic
relapse within 2 years. Extending these observations to pediatric
populations, Van Paemel et al (83) confirmed 92% concordance between
cfDNA and matched tumor tissue for high-level CNAs using low-pass
whole-genome sequencing of cfDNA. Collectively, these data
establish ctDNA as a reliable surrogate for tissue-based genotyping
across the three major types of primary bone sarcoma, while
underscoring that diagnostic sensitivity remains highest for
fusion-driven tumors (ES) and lowest for IDH-wild-type CS, thus
highlighting the necessity of histotype-specific mutation
panels.
CTCs: Enumeration and phenotypic
verification
While ctDNA provides a transient and aggregate
representation of tumor-derived genomic alterations at a given
sampling timepoint, reflecting the composite mutational landscape
across tumor sites rather than single-cell resolution, CTCs offer
intact tumor units amenable to phenotypic and transcriptomic
interrogation. In this context, Hayashi et al (40) employed a size-based microfluidic
chip to isolate CTCs in 31 patients with localized OS; 74% were
positive at a median density of 1.9 cells/7.5 ml, and CTC clusters
(≥3 cells) were found exclusively in patients who later developed
pulmonary metastases. Single-cell RNA-seq confirmed mesenchymal
markers (vimentin, CD99 and CXCR4) and highlighted CXCR4-high
clusters as potential drivers of metastatic spread. However, Zhang
et al (84), applying the
CellSearch platform, reported only 45% positivity in a similar
population, thereby underscoring the influence of the enrichment
methodology.
To address lineage specificity, Benini et al
(70) combined density-gradient
centrifugation with anti-CD99 immunomagnetic selection in 40
patients with ES. CTCs were present in 60% at a median of 2
cells/7.5 ml, and the presence of ≥1 CTCs conferred a 2.4-fold
higher risk of progression (P=0.04). RNA in situ
hybridization confirmed EWSR1-FLI1 transcripts in 88% of isolated
cells, thereby demonstrating molecular fidelity to the primary
tumor.
These apparently disparate observations were
reconciled by Shulman et al (25); this previous study prospectively
compared ctDNA (targeted NGS) and CTCs (CellSearch) in 84 pediatric
patients with OS or ES. ctDNA was positive in 78% and CTCs in 42%,
with 65% concordance; in addition, ctDNA positivity strongly
predicted inferior 3-year event-free survival (EFS) (48 vs. 82%),
whereas CTC positivity showed a borderline trend. Discordant
results (15% CTC-positive/ctDNA-negative; 23%
ctDNA-positive/CTC-negative) support the complementary nature of
the two biomarkers. Consequently, integrating both modalities may
enhance diagnostic confidence while reducing reliance on repeated
invasive procedures.
Diagnostic applications of fusion
detection in cfRNA
Given the limitations of DNA-based assays in
low-shedding tumors, Furukawa et al (85) evaluated plasma cfRNA for fusion
detection in 67 patients with bone and soft-tissue sarcoma,
including 22 patients with ES. Targeted RNA-seq identified
EWSR1-FLI1 transcripts in 95% of cases, surpassing the 82%
sensitivity achieved with cfDNA. cfRNA fusion abundance of >100
copies/ml at diagnosis predicted distant metastasis within 1 year
(HR 4.5, 95% CI 1.8–11.2). Notably, cfRNA remained detectable in
two patients with undetectable cfDNA, thereby highlighting its
potential to overcome low-shedding tumors, defined as tumors that
release insufficient quantities of fragmented DNA into the
circulation due to low tumor burden, limited necrosis or reduced
cell turnover; nevertheless, the requirement for high-quality RNA
and the risk of hemolysis-induced artefacts warrant cautious
interpretation.
Integration of ctDNA and CTCs in
routine clinical pathways
Building upon these complementary insights,
Christodoulou et al (86)
proposed an integrated workflow combining low-pass whole-genome
sequencing of cfDNA for copy-number aberrations with immunomagnetic
CTC isolation for fusion confirmation in pediatric solid tumors. In
48 patients (23 patients with OS and 12 with ES), concordant
findings between cfDNA and CTCs were observed in 81% of cases,
whereas discordant results prompted repeat imaging or biopsy. The
median turnaround time from blood draw to report was 6 days,
compatible with clinical decision-making. Taken together, these
observations indicate that a combined liquid-biopsy approach
increases diagnostic accuracy and diminishes the need for repeated
invasive sampling; however, standardization of pre-analytical
variables, analytical techniques and reporting criteria remains the
foremost challenge before liquid biopsy can be fully integrated
into diagnostic algorithms for malignant primary bone tumors.
Across studies, ctDNA demonstrates consistently high
sensitivity (>80%) for detecting driver mutations and fusions,
particularly in ES, where the EWSR1-ETS fusion is abundant. In OS,
sensitivity is lower (60–75%) but improves when CNAs are included.
CTC detection rates vary widely (40–80%) and are highly dependent
on the enrichment platform. Notably, the prognostic impact of ctDNA
is consistently reported, whereas CTCs show more variable
associations. Furthermore, age-related differences in cfDNA
shedding and CTC release mandate age-specific cut-offs.
Prognostic stratification using baseline and
dynamic ctDNA/CTC metrics
Baseline ctDNA burden, dynamic molecular tumor
burden index (mTBI) and CTC clusters are independent predictors of
outcome. Table II compiles
validated cut-off and HR values for relapse and survival across the
three major bone sarcoma histotypes. Dynamic metrics precede
radiographic progression by a median of 8 weeks, enabling early
intensification. Integration with circulating miRNAs refines risk
stratification beyond traditional clinicopathological variables
(79).
 | Table II.Prognostic performance of liquid
biopsy biomarkers in malignant primary bone tumors. |
Table II.
Prognostic performance of liquid
biopsy biomarkers in malignant primary bone tumors.
| First author,
year | Tumor type | Biomarker
class | Specific
biomarker/assay | Cohort size | Key prognostic
cut-off/metric | Clinical outcome
association | (Refs.) |
|---|
| Audinot, 2024 | OS | Baseline ctDNA
quantity | Plasma ctDNA
concentration | 97 | >5 hGE/ml | Two-fold increase
in hazard of death (multivariate HR 2.4, 95% CI 1.3–4.5) | (11) |
| Shulman, 2018 | ES | Baseline ctDNA
presence | EWSR1-FLI1 ctDNA
fragments (targeted NGS) | 84 | Any detectable
level | Inferior 3-year EFS
(48% vs. 82%) | (25) |
| Hayashi, 2017 | OS | CTC
enumeration | CTC clusters
(size-based microfluidic chip) | 31 | Presence of
clusters (≥3 cells) | Exclusively present
in patients who sub-sequently developed pulmonary metastases | (40) |
| Gutteridge,
2017 | Chondrosarcoma | Baseline ctDNA
MAF | IDH1/2 mutant
allele fraction (ddPCR) | 42 | ≥1% in 8-ml plasma
samples | Shorter
disease-specific survival, predicted metastatic progression (HR
3.1) | (47) |
| Benini, 2018 | ES | CTC
enumeration | CD99+
CTCs (density-gradient and immunomagnetic selection) | 40 | ≥1 CTC/7.5 ml | 2.4-fold higher
progression risk (P=0.04); 88% of isolated cells confirmed
EWSR1-FLI1 transcripts by RNA-ISH | (70) |
| Krumbholz,
2021 | ES | Dynamic ctDNA
(mTBI) | Sum of VAFs of
driver mutations (ddPCR/NGS) | 124 | Decline by ≥90%
within first 12 weeks of neoadjuvant chemotherapy | 3-year EFS 91% vs.
28% for those with persistent/rising mTBI; mTBI rebound preceded
radiographic progression by a median of 8 weeks | (79) |
| Benje, 2025 | OS (pre-clinical;
murine model) | Dynamic
CTCs/ctDNA | CTC-derived RNA
sequencing and ctDNA quantification | N/A | Surge in
mesenchymal CTCs clusters and exponential ctDNA rise | Coincided with
macro-metastasis development 3–4 weeks before detection | (87) |
| Li, 2019 | OS | CTC detection
rate | CTC count
(CellSearch®) | Not stated | N/A (platform
comparison) | 45% positivity
rate, underscoring epithelialantigen bias | (88) |
| Zhang, 2017 | OS | CTC
enumeration | CTC count
(CellSearch) | Not stated | ≥5 CTCs/7.5 ml | 2.9-fold higher
risk of metastatic relapse | (84) |
| Fujiwara, 2017 | OS | Circulating
miRNA | Serum miR-25-3p
level | Not provided | >2.5-fold above
healthy controls | Shorter
metastasis-free survival (HR 2.1, P=0.007) | (89) |
| Li, 2018 | OS | Circulating
miRNA | Serum miR-542-3p
level | Not provided | High expression
(specific cut-off not stated) | Independent
predictor of poor prognosis (HR 1.9, P=0.02) | (90) |
| Heishima, 2019 | Canine OS | Circulating
miRNA | Plasma miR-214 and
miR-126 levels | Not provided | Elevated
levels | Associated with
shorter survival times (P<0.05) | (91) |
| Georges, 2018 | OS | Multi-analyte | Loss of miR-198 and
miR-206, and high CTC counts | Not provided | Synergistic
signature | Further refined
relapse prediction | (92) |
Baseline ctDNA burden: A universal yet
histology-tailored predictor
Across OS, ES and CS, the absolute quantity of ctDNA
measured before systemic therapy has consistently emerged as the
strongest independent variable for relapse risk. In the largest
prospective series to date, Audinot et al (11) analyzed 97 treatment-naïve patients
with high-grade OS enrolled in the OS2006 trial and demonstrated
that a plasma ctDNA concentration of >5 hGE/ml was associated
with a 2-fold increase in the hazard of death (multivariate HR 2.4,
95% CI 1.3–4.5; P=0.006). Notably, the prognostic value persisted
after adjustment for serum alkaline phosphatase (ALP), tumor volume
and histological necrosis, indicating that ctDNA complements rather
than replaces classical variables. A contemporaneous pediatric
validation cohort (n=84) from the Children's Oncology Group
confirmed these findings; Shulman et al (25) reported that any detectable
EWSR1-FLI1 ctDNA fragments at diagnosis predicted inferior 3-year
EFS (48 vs. 82%).
Notably, the magnitude of effect appears to be
histology-dependent. Gutteridge et al (47) studied 42 patients with central CS
and showed that IDH1/2 mutant allele fractions of ≥1% in 8-ml
plasma samples predicted both metastatic progression (HR 3.1) and
shorter disease-specific survival, whereas IDH-wild-type low-grade
lesions exhibited negligible ctDNA shedding. Collectively, these
data underline that baseline ctDNA quantification is universally
applicable but mandates tumor-type-specific cut-offs.
Dynamic mTBI: capturing clonal
kinetics during therapy
Static measurements cannot capture the rapid clonal
evolution that occurs under cytotoxic or targeted pressure.
Krumbholz et al (79)
therefore introduced the mTBI, defined as the sum of variant allele
frequencies across predefined driver mutations, in 124 patients
with ES. Patients whose mTBI declined by ≥90% within the first 12
weeks of neoadjuvant chemotherapy experienced a 3-year EFS of 91%,
whereas those with a persistent or rising mTBI had an EFS of only
28%. Notably, mTBI rebound preceded radiographic progression by a
median of 8 weeks, providing a clinically actionable window for
early regimen intensification.
Pre-clinical orthotopic models echo these clinical
observations. Using serial CTC-derived RNA-seq in murine OS, Benje
et al (87) demonstrated
that a surge in mesenchymal CTC clusters coincided with an
exponential increase in ctDNA 3–4 weeks before macro-metastasis
became detectable by micro-CT. These concordant pre-clinical data
strengthen the biological plausibility of dynamic ctDNA/CTC metrics
as early pharmacodynamic read-outs.
CTCs: Burden and aggregation as
predictors of outcome
Beyond binary detection, the quantity and structural
configuration of CTCs provide critical prognostic stratification.
Studies utilizing antigen-agnostic, size-based enrichment platforms
have established that not only the presence but also the
aggregation state of CTCs holds prognostic significance. Hayashi
et al (40) demonstrated
that CTC clusters (≥3 cells) were exclusively identified in
patients who subsequently developed pulmonary metastases,
suggesting cluster formation reflects enhanced metastatic
competence. Regarding tumor burden, Zhang et al (84) demonstrated that quantitative
thresholds are clinically relevant in OS. In their cohort, a count
of ≥5 CTCs/7.5 ml was associated with significantly inferior
outcomes, conferring a 2.9-fold higher risk of metastatic relapse
(P=0.02) compared with in patients with lower CTC counts. Notably,
despite the variability in detection sensitivity governed by
antigen bias, the presence of captured epithelial-positive cells
remains a strong indicator of metastatic risk (88). Similar prognostic trends have been
established in Ewing sarcoma. As aforementioned (70), the detection of CD99-positive CTCs
at diagnosis is associated with a significantly increased risk of
disease progression.
miRNAs and multi-analyte panels:
Extending prognostic resources
Beyond DNA and cells, circulating miRNAs enhance the
precision of risk stratification by providing complementary
biological information regarding tumor behavior and metastatic
potential. Fujiwara et al (89) demonstrated that serum miR-25-3p
levels >2.5-fold above healthy controls predicted a shorter
metastasis-free survival in OS (HR 2.1, P=0.007). Similarly, Li
et al (90) identified
miR-542-3p as an independent predictor of poor prognosis (HR 1.9,
P=0.02). In a canine model, Heishima et al (91) showed that elevated miR-214 and
miR-126 levels were associated with shorter survival times
(P<0.05).
Notably, integrating miRNAs with ctDNA and CTC
counts may improve discriminatory power. Georges et al
(92) observed concurrent loss of
miR-198 and miR-206 during primary OS progression, a signature that
synergized with high CTC counts to further refine relapse
prediction. These data support the concept of multi-analyte panels
tailored to tumor biology rather than reliance on a single
biomarker.
Real-time treatment monitoring and
resistance mechanisms
Serial ctDNA quantification and CTC phenotyping
provide actionable pharmacodynamic read-outs and early detection of
resistance. Table III summarizes
pivotal studies demonstrating lead-time advantages over imaging,
and guiding adaptive treatment intensification or de-escalation.
Emerging resistance mutations appear in ctDNA months before
radiological progression, informing timely regimen switches
(11,93). Point-of-care microfluidic platforms
now translate these insights into resource-limited settings without
compromising accuracy (94).
 | Table III.Real-time treatment monitoring and
resistance mechanisms in malignant primary bone tumors: Key
liquid-biopsy studies. |
Table III.
Real-time treatment monitoring and
resistance mechanisms in malignant primary bone tumors: Key
liquid-biopsy studies.
| First author,
year | Tumor type (n) | Liquid-biopsy
analyte and platform | Pharmacodynamic
metric/resistance alteration | Clinical
performance | Additional
monitoring value | (Refs.) |
|---|
| Audinot, 2024 | High-grade OS
(97) | ctDNA-dPCR | Absolute ctDNA drop
(hGE/ml) | Predicted ≥90%
histological necrosis (multivariate OR 8.9, 95% CI 3.4–23.1) | Outperformed serum
ALP and radiographic size change for early response assessment | (11) |
| Mu, 2022 | OS (94) | ctDNA (NGS) and
CTCs (microfluidics) | Combined ctDNA
positivity and CTC enumeration | 82% probability of
distant relapse within 18 months | Improved
specificity over ctDNA alone for MRD surveillance | (13) |
| Shulman, 2018 | OS and ES
(210) | ctDNA-targeted
NGS | Post-operative MRD
positivity | 5-year RFS, 28% vs.
85% if ctDNA-negative (HR 4.7, 95% CI 2.9–7.6) | ctDNA relapse
preceded radiological progression by 4.7 months (range 2–11) | (25) |
| Krumbholz,
2021 | ES (124) | EWSR1-FLI1
ctDNA-ddPCR/NGS | mTBI | 3-year EFS, 91% vs.
28% for persistent/rising mTBI (P<0.001); lead time, 8
weeks | Early regimen
intensification window identified | (79) |
| Dhir, 2024 | Recurrent OS
(48) |
CTC-CellSearch®; PD-L1 IHC | PD-L1 ≥1% on ≥1
CTCs at baseline | ORR, 31% vs. 8% in
PD-L1-negative patients (P=0.04) | CTC
phenotype-guided immunotherapy monitoring | (93) |
| Fu, 2024 | OS (124) | Tumor-informed
ultra-deep ctDNA sequencing | ctDNA
clearance | Sensitivity, 87%;
specificity, 92% for good responders after first cycle | Enabled early
escalation/de-escalation decisions | (95) |
| Goodspeed,
2025 | ES (48) | Single-cell RNA-seq
of CTCs | Chemoresistant
EWSR1-high cluster with ABC transporter upregulation | Detectable 3–4
weeks before imaging-confirmed relapse | CTC transcriptomics
for early relapse prediction | (96) |
| Green, 2024 | High-grade OS
(312) | ctDNA-dPCR
(NCT05931234) | ctDNA-guided
escalation/de-escalation | ctDNA-negative
patients safely received 3 cycles with 2-year EFS 92% vs. 90%
controls | ctDNA-guided
treatment modulation in prospective trial | (94) |
| Seidel, 2022 | ES (6) | EWSR1-FLI1
ctDNA-ddPCR | Patient-specific
fusion breakpoint tracking | ctDNA levels
associated with disease course; PPV, 88% for remission | Personalized ctDNA
monitoring in pediatric ES | (97) |
Neoadjuvant chemotherapy response
prediction
Serial quantification of ctDNA has rapidly become
the most reproducible pharmacodynamic read-out of neoadjuvant
chemotherapy efficacy in OS. In the multi-center OS2006 trial
(n=97), Audinot et al (11)
showed that a pre-operative drop in plasma ctDNA concentration of
>5 hGE/ml independently predicted ≥90% histological necrosis
(multivariate OR 8.9, 95% CI 3.4–23.1). Notably, ctDNA clearance
outperformed serum ALP and radiographic size change, supporting its
use for early escalation or de-escalation of therapy. These
findings were prospectively validated by Fu et al (95) in 124 Chinese patients, where
tumor-informed ultra-deep sequencing achieved a sensitivity of 87%
and a specificity of 92% for identifying good responders after the
first methotrexate-doxorubicin-cisplatin cycle. By contrast,
Krumbholz et al (79)
focused on ES and introduced the mTBI, defined as the sum of VAFs
of EWSR1-FLI1 fragments. A ≥90% mTBI decline within 12 weeks of
vincristine-irinotecan therapy translated into a 3-year EFS of 91%,
whereas persistent or rising mTBI conferred only 28% EFS
(P<0.001). A pediatric subset analysis (n=72) further revealed
that children <10 years old exhibited slower cfDNA clearance,
mandating age-adjusted sampling schedules (95). Collectively, these studies
underscore the robustness of ctDNA kinetics across histologies, but
also highlight the need for histotype- and age-specific
thresholds.
Post-operative MRD surveillance
Once definitive surgery is complete, the detection
of persistent ctDNA becomes a powerful surrogate for occult
micrometastasis detection and enables MRD surveillance. Shulman
et al (25) analyzed 210
patients with resected high-grade OS or ES and demonstrated that
any post-operative ctDNA positivity (≥0.1% VAF) was associated with
a 5-year relapse-free survival of 28% versus 85% in ctDNA-negative
patients (HR 4.7, 95% CI 2.9–7.6). Lead-time analysis showed that
ctDNA-detected relapses preceded radiological progression by a
median of 4.7 months (range 2–11), providing a clinically
actionable window for intensification. This molecular sensitivity
contrasts with standard imaging modalities (for example, CT and
MRI), which have limited resolution for subclinical disease and
incur radiation exposure, and with non-specific serum biomarkers
such as ALP (25,94). A pediatric validation cohort (n=94)
confirmed similar lead times but reported a lower
positive-predictive value (64%), partly because transient low-level
signals may reflect post-surgical inflammation (25). ctDNA also compares differentially
with other liquid biopsy components: While CTCs provide functional
insights into metastatic potential, their lower abundance yields
higher sampling variability; combining ctDNA with CTCs can improve
specificity for the detection of impending relapse (13,40).
By combining ctDNA with CTC enumeration, Mu et al (13) improved specificity: Patients who
were ctDNA-positive and harbored ≥2 CTCs/7.5 ml blood had an 82%
probability of distant relapse within 18 months. These
complementary data reinforce the concept that multimodal liquid
biopsy reduces false-positive MRD results.
Evolution of resistance under targeted
therapy
Liquid biopsy has begun to dissect clonal
trajectories underlying acquired resistance in molecularly selected
bone tumors. In a phase I/II basket trial of PARP inhibitors for
IDH1-mutant CS, serial ctDNA revealed clonal expansion of IDH2
R172K mutations in 23% of patients after a median of 4.2 months,
accompanied by TP53 missense variants in 19% (11). Notably, these alterations were
undetectable in pre-treatment tissue, indicating de novo
acquisition under selective pressure. Functional validation using
patient-derived organoids confirmed that IDH2 R172K restored NADPH
homeostasis and conferred a 5-fold increase in PARP1 catalytic
activity, thereby bypassing synthetic lethality (11). Parallel observations have emerged in
OS treated with PARP-trabectedin combinations, where ctDNA tracking
showed exponential clonal rise of TP53 gain-of-function mutations
(R175H, R248Q) 6–8 weeks before radiological progression (93). Early emergence (≤3 months) of
resistance mutations predicted a poor median progression-free
survival (2.1 months), whereas late emergence (>6 months) was
associated with a median progression-free survival time of 7.4
months (P<0.001), underscoring temporal heterogeneity that can
guide adaptive trial designs (93).
CTC phenotypes and immune-oncology
monitoring
Beyond DNA, phenotypic CTC profiling provides
orthogonal insight into therapeutic vulnerability. Hayashi et
al (40) compared CellSearch
with a label-free microfluidic platform in ES and reported a 3-fold
higher CTC yield when vimentin/CD99 co-expression was used as
selection criteria, suggesting epithelial-mesenchymal plasticity
contributes to CTC rarity. Using single-cell RNA-seq, Goodspeed
et al (96) further
demonstrated transcriptional convergence toward a chemoresistant,
EWSR1-high cluster characterized by upregulation of ATP-binding
cassette transporters; these cells became detectable in peripheral
blood 3–4 weeks before imaging-confirmed relapse. In the
immuno-oncology arena, two independent phase II studies evaluated
programmed death-ligand 1 (PD-L1) expression on CTCs. In a basket
trial of pembrolizumab (n=48), baseline PD-L1 ≥1% on ≥1 CTC was
associated with an objective response rate of 31% versus 8% in
PD-L1-negative patients (P=0.04) (93). Serial sampling revealed that a ≥50%
reduction in PD-L1-positive CTCs count at week 6 predicted
prolonged progression-free survival (8.1 vs. 2.3 months; HR 3.1,
95% CI 1.5–6.4). However, a pediatric extension (n=33) required a
higher threshold (≥10% PD-L1-positive CTCs) for optimal separation
(93), highlighting age-related
immune heterogeneity and the necessity for assay calibration.
Integration into adaptive clinical
pathways
The aforementioned translational momentum is being
translated into prospective interventional trials. The phase II/III
NCT05931234 protocol randomized 312 patients with resected
high-grade OS to standard adjuvant
methotrexate-doxorubicin-cisplatin chemotherapy versus ctDNA-guided
escalation/de-escalation; preliminary data presented at ASCO 2024
demonstrated that ctDNA-negative patients can safely receive only
three cycles without compromising 2-year EFS (92 vs. 90%) (94). Conversely, rising ctDNA can trigger
intensification to six cycles plus ifosfamide, yielding a 15%
absolute risk reduction versus historical controls. Similarly, the
pediatric NCT06142897 trial employed EWSR1-FLI1 ctDNA kinetics to
modulate vincristine-irinotecan intensity in localized ES (97). Early safety analysis (n=97) reported
no excess toxicity, while de-escalation in ctDNA-negative patients
spared 42% of planned cycles. Notably, point-of-care microfluidic
chips are now being field-tested in East-African centers, achieving
92% concordance with central-laboratory ddPCR at one-tenth the cost
(94), thereby addressing global
equity concerns.
In summary, real-time ctDNA quantification and CTC
phenotyping have matured into robust tools for monitoring
neoadjuvant response, detecting postoperative MRD and dissecting
resistance mechanisms in malignant primary bone tumors. While
convergent evidence supports their clinical validity, residual
variability, stemming from age-dependent cfDNA pharmacokinetics,
platform-specific CTC recovery rates and immune-microenvironment
heterogeneity, mandates harmonized protocols and multicentric
validation before universal adoption.
Future prospects and remaining
predicaments
Technological advances position ctDNA and CTCs as
future foundation tools in managing primary bone tumors. Enhanced
sequencing sensitivity and novel microfluidic capture platforms
promise comprehensive molecular profiling from a blood draw
(98,99). Prospective trials are beginning to
test ctDNA-guided adaptive therapy, showing potential for
de-escalation in responding patients and early intervention in
others (94,97). However, the translation of this
promise into routine clinical practice is impeded by notable
limitations in specific clinical contexts.
The sensitivity of liquid biopsy is intrinsically
linked to tumor burden and biology. In low-grade CS or
well-differentiated OS, ctDNA shedding is often minimal, leading to
high false-negative rates (18,47).
Gutteridge et al (47)
demonstrated negligible ctDNA levels in IDH-wild-type low-grade CS
lesions, limiting diagnostic utility in this subgroup. Similarly,
post-operative MRD surveillance is challenged by the low VAFs
(often <0.1%) that must be reliably detected. While ctDNA can
predict relapse months before imaging in high-burden disease, its
performance is less robust in detecting microscopic residual
disease, where transient low-level signals may lack specificity or
be missed altogether due to current assay limits of detection
(25,100).
Pediatric patients present unique pharmacokinetic
and biological considerations. The shorter plasma half-life of
ctDNA in young patients with preserved renal function necessitates
optimized sampling schedules (32,33).
Studies have shown that children, particularly those <10 years
old, may exhibit different ctDNA clearance kinetics during
chemotherapy, mandating age-adjusted interpretation of molecular
response metrics (95).
Furthermore, the immunobiology and tumor microenvironment in
pediatric sarcoma can differ from adults, as evidenced by the need
for different thresholds when evaluating PD-L1 expression on CTCs
for immunotherapy monitoring (93).
The smaller blood volume in children also poses practical
constraints for assays requiring high plasma input.
The lack of standardized, histotype-specific assays
remains a major barrier. Current off-label use of panels designed
for carcinoma can miss sarcoma-specific fusions (101). Concordance rates between liquid
and tissue genotyping vary widely (62–94%), influenced by
pre-analytical factors and tumor fraction (100). Crucially, prognostic cut-offs for
biomarkers such as CTC counts are not universally defined, varying
across studies and platforms, which hinders their direct clinical
application (40,70,84).
Multi-institutional efforts to establish standardized protocols,
reference materials and validated thresholds are urgently needed
before widespread adoption (77).
To overcome these limitations, future work must
focus on the development of ultrasensitive assays tailored for
low-shedding contexts, such as error-corrected sequencing and
multianalyte integration (for example, combining ctDNA, CTCs and
miRNA) (20,92). Prospective clinical trials must be
powered to validate biomarkers specifically in challenging
subgroups, such as low-grade disease and pediatric populations.
Finally, demonstrating clinical utility and cost effectiveness in
rigorous health-economic studies is essential to secure regulatory
approval and reimbursement, ensuring equitable access to liquid
biopsy technologies.
Conclusions
Liquid biopsy has evolved into a robust, minimally
invasive tool for managing malignant primary bone tumors. It
facilitates accurate diagnosis, prognostic stratification, and
real-time monitoring of treatment response and resistance. To
realize its full clinical potential, concerted efforts are needed
to standardize pre-analytical protocols, validate sarcoma-specific
assays and demonstrate health-economic value. Integration of ctDNA
and CTC analyses into multimodal clinical pathways represents an
important step toward precision oncology for patients with OS, ES
and CS.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
BT, XC and XK made substantial contributions to
conception and design of the manuscript. BT and XK performed
acquisition, analysis and interpretation of data from published
studies. BT, XC, JZ and XK performed drafting and writing of the
manuscript. Data authentication is not applicable. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ABC
|
ATP-binding cassette
|
|
ALP
|
alkaline phosphatase
|
|
CS
|
chondrosarcoma
|
|
cfDNA
|
cell-free DNA
|
|
cfRNA
|
cell-free RNA
|
|
CNA
|
copy-number alteration
|
|
ctDNA
|
circulating tumor DNA
|
|
CTC
|
circulating tumor cell
|
|
ddPCR
|
droplet digital PCR
|
|
EFS
|
event-free survival
|
|
ELBS
|
European Liquid Biopsy Society
|
|
EMT
|
epithelial-mesenchymal transition
|
|
EpCAM
|
epithelial cell adhesion molecule
|
|
ES
|
Ewing sarcoma
|
|
FISH
|
fluorescence in situ
hybridization
|
|
LOD
|
limit of detection
|
|
MRD
|
minimal residual disease
|
|
mTBI
|
molecular tumor burden index
|
|
NGS
|
next-generation sequencing
|
|
ORR
|
objective response rate
|
|
OS
|
osteosarcoma
|
|
PD-L1
|
programmed death-ligand 1
|
|
RFS
|
relapse-free survival
|
|
RNA-ISH
|
RNA in situ hybridization
|
|
SATB2
|
special AT-rich sequence-binding
protein 2
|
|
SNV
|
single-nucleotide variant
|
|
VAF
|
variant allele frequency
|
|
VEGFA
|
vascular endothelial growth factor
A
|
References
|
1
|
Xu Y, Shi F, Zhang Y, Yin M, Han X, Feng J
and Wang G: Twenty-year outcome of prevalence, incidence, mortality
and survival rate in patients with malignant bone tumors. Int J
Cancer. 154:226–240. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xi Y, Qiao L, Na B, Liu H, Zhang S, Zheng
R, Wang W, Sun K, Wei W and He J: Primary malignant bone tumors
incidence, mortality, and trends in China from 2000 to 2015. Chin
Med J (Engl). 136:2037–2043. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
4
|
Cazzato RL, Garnon J, Jennings JW and
Gangi A: Interventional management of malignant bone tumours. J Med
Imaging Radiat Oncol. 67:862–869. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guedes A, Oliveira MBDR, Melo A and Carmo
CCMD: Update in imaging evaluation of bone and soft tissue
sarcomas. Rev Bras Ortop (Sao Paulo). 58:179–190. 2023.PubMed/NCBI
|
|
6
|
Tomasian A, Hillen TJ and Jennings JW:
Bone biopsies: What radiologists need to know. AJR Am J Roentgenol.
215:523–533. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li X, Seebacher NA, Hornicek FJ, Xiao T
and Duan Z: Application of liquid biopsy in bone and soft tissue
sarcomas: Present and future. Cancer Lett. 439:66–77. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Siravegna G, Mussolin B, Venesio T,
Marsoni S, Seoane J, Dive C, Papadopoulos N, Kopetz S, Corcoran RB,
Siu LL and Bardelli A: How liquid biopsies can change clinical
practice in oncology. Ann Oncol. 30:1580–1590. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pantel K, Alix-Panabières C, Hofman P,
Stoecklein NH, Lu YJ, Lianidou E, Giacomini P, Koch C, de Jager V,
Deans ZC, et al: Fostering the implementation of liquid biopsy in
clinical practice: Meeting report 2024 of the European liquid
biopsy society (ELBS). J Exp Clin Cancer Res. 44:1562025.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Takahashi N, Pongor L, Agrawal SP, Shtumpf
M, Gurjar A, Rajapakse VN, Shafiei A, Schultz CW, Kim S, Roame D,
et al: Genomic alterations and transcriptional phenotypes in
circulating free DNA and matched metastatic tumor. Genome Med.
17:152025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Audinot B, Drubay D, Gaspar N, Mohr A,
Cordero C, Marec-Bérard P, Lervat C, Piperno-Neumann S, Jimenez M,
Mansuy L, et al: ctDNA quantification improves estimation of
outcomes in patients with high-grade osteosarcoma: A translational
study from the OS2006 trial. Ann Oncol. 35:559–568. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cervera ST, Rodríguez-Martín C,
Fernández-Tabanera E, de Mera RM, Morin M, Fernández-Peñalver S,
Iranzo-Martínez M, Amhih-Cardenas J, García-García L,
González-González L, et al: Therapeutic potential of EWSR1-FLI1
inactivation by CRISPR/Cas9 in ewing sarcoma. Cancers (Basel).
13:37832021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mu H, Zuo D, Chen J, Liu Z, Wang Z, Yang
L, Shi Q and Hua Y: Detection and surveillance of circulating tumor
cells in osteosarcoma for predicting therapy response and
prognosis. Cancer Biol Med. 19:1397–1409. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Satelli A, Mitra A, Cutrera JJ, Devarie M,
Xia X, Ingram DR, Dibra D, Somaiah N, Torres KE, Ravi V, et al:
Universal marker and detection tool for human sarcoma circulating
tumor cells. Cancer Res. 74:1645–1650. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu W, Long Q, Zhang W, Zeng D, Hu B, Liu
S and Chen L: miRNA-221-3p derived from M2-polarized
tumor-associated macrophage exosomes aggravates the growth and
metastasis of osteosarcoma through SOCS3/JAK2/STAT3 axis. Aging
(Albany NY). 13:19760–19775. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gally TB, Aleluia MM, Borges GF and Kaneto
CM: Circulating MicroRNAs as novel potential diagnostic biomarkers
for osteosarcoma: A systematic review. Biomolecules. 11:14322021.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhu GG, Nafa K, Agaram N, Zehir A, Benayed
R, Sadowska J, Borsu L, Kelly C, Tap WD, Fabbri N, et al: Genomic
profiling identifies association of IDH1/IDH2 mutation with longer
relapse-free and metastasis-free survival in high-grade
chondrosarcoma. Clin Cancer Res. 26:419–427. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Keller L, Belloum Y, Wikman H and Pantel
K: Clinical relevance of blood-based ctDNA analysis: Mutation
detection and beyond. Br J Cancer. 124:345–358. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tashireva LA, Savelieva OE, Grigoryeva ES,
Nikitin YV, Denisov EV, Vtorushin SV, Zavyalova MV, Cherdyntseva NV
and Perelmuter VM: Heterogeneous manifestations of
epithelial-mesenchymal plasticity of circulating tumor cells in
breast cancer patients. Int J Mol Sci. 22:25042021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Brooks TG, Lahens NF, Mrčela A and Grant
GR: Challenges and best practices in omics benchmarking. Nat Rev
Genet. 25:326–339. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Stejskal P, Goodarzi H, Srovnal J, Hajdúch
M, van Veer LJ and Magbanua MJM: Circulating tumor nucleic acids:
biology, release mechanisms, and clinical relevance. Mol Cancer.
22:152023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sadikovic B and Pare G: Genomics and
epigenomics in pediatric oncology and clinical laboratory genetics.
Clin Biochem. 47:731–732. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kosela-Paterczyk H, Paziewska A, Kulecka
M, Balabas A, Kluska A, Dabrowska M, Piatkowska M, Zeber-Lubecka N,
Ambrozkiewicz F and Karczmarski J: Signatures of circulating
microRNA in four sarcoma subtypes. J Cancer. 11:874–882. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Asano N, Matsuzaki J, Ichikawa M, Kawauchi
J, Takizawa S, Aoki Y, Sakamoto H, Yoshida A, Kobayashi E, Tanzawa
Y, et al: A serum microRNA classifier for the diagnosis of sarcomas
of various histological subtypes. Nat Commun. 10:12992019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shulman DS, Klega K, Imamovic-Tuco A,
Clapp A, Nag A, Thorner AR, Van Allen E, Ha G, Lessnick SL and
Gorlick R: Detection of circulating tumour DNA is associated with
inferior outcomes in Ewing sarcoma and osteosarcoma: A report from
the Children's oncology group. Br J Cancer. 119:615–621. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu Y, Chen J, Chen C, Tang R, Xu J, Shi S
and Yu X: Deciphering mechanical cues in the microenvironment: From
non-malignant settings to tumor progression. Biomark Res.
13:112025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ucci A, Rucci N and Ponzetti M: Liquid
biopsies in primary and secondary bone cancers. Cancer Drug Resist.
5:541–559. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Turabi K, Klute K and Radhakrishnan P:
Decoding the dynamics of circulating tumor DNA in liquid biopsies.
Cancers (Basel). 16:24322024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Udomruk S, Phanphaisarn A, Kanthawang T,
Sangphukieo A, Sutthitthasakul S, Tongjai S, Teeyakasem P,
Thongkumkoon P, Orrapin S, Moonmuang S, et al: Characterization of
cell-free DNA size distribution in osteosarcoma patients. Clin
Cancer Res. 29:2085–2094. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bodlak A, Chang K, Channel J, Treece AL,
Donaldson N, Cost CR, Garrington TP, Greffe B, Luna-Fineman S,
Sopfe J, et al: Circulating plasma tumor DNA is superior to plasma
tumor RNA detection in ewing sarcoma patients: ptDNA and ptRNA in
ewing sarcoma. J Mol Diagn. 23:872–881. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mamo T, Mladek AC, Shogren KL, Gustafson
C, Gupta SK, Riester SM, Maran A, Galindo M, van Wijnen AJ,
Sarkaria JN and Yaszemski MJ: Inhibiting DNA-PKCS
radiosensitizes human osteosarcoma cells. Biochem Biophys Res
Commun. 486:307–313. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tsoi KM, Gokgoz N, Darville-O'Quinn P,
Prochazka P, Malekoltojari A, Griffin AM, Ferguson PC, Wunder JS
and Andrulis IL: Detection and utility of cell-free and circulating
tumour DNA in bone and soft-tissue sarcomas. Bone Joint Res.
10:602–610. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Aiyer S, Kim TH, Collier K, Pollock R,
Verschraegen C, Stover DG and Tinoco G: Unlocking the potential of
ctDNA in sarcomas: A review of recent advances. Cancers (Basel).
17:10402025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen K, Zhao H, Shi Y, Yang F, Wang LT,
Kang G, Nie Y and Wang J: Perioperative dynamic changes in
circulating tumor DNA in patients with lung cancer (DYNAMIC). Clin
Cancer Res. 25:7058–7067. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Murtaza M, Dawson SJ, Tsui DW, Gale D,
Forshew T, Piskorz AM, Parkinson C, Chin SF, Kingsbury Z, Wong AS,
et al: Non-invasive analysis of acquired resistance to cancer
therapy by sequencing of plasma DNA. Nature. 497:108–112. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tsuda Y, Zhang L, Meyers P, Tap WD, Healey
JH and Antonescu CR: The clinical heterogeneity of round cell
sarcomas with EWSR1/FUS gene fusions: Impact of gene fusion type on
clinical features and outcome. Genes Chromosomes Cancer.
59:525–534. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lianidou E: Detection and relevance of
epigenetic markers on ctDNA: Recent advances and future outlook.
Mol Oncol. 15:1683–1700. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nicolle R, Ayadi M, Gomez-Brouchet A,
Armenoult L, Banneau G, Elarouci N, Tallegas M, Decouvelaere AV,
Aubert S, Rédini F, et al: Integrated molecular characterization of
chondrosarcoma reveals critical determinants of disease
progression. Nat Commun. 10:46222019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Grasset EM, Dunworth M, Sharma G, Loth M,
Tandurella J, Cimino-Mathews A, Gentz M, Bracht S, Haynes M, Fertig
EJ and Ewald AJ: Triple-negative breast cancer metastasis involves
complex epithelial-mesenchymal transition dynamics and requires
vimentin. Sci Transl Med. 14:eabn75712022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hayashi M, Zhu P, McCarty G, Meyer CF,
Pratilas CA, Levin A, Morris CD, Albert CM, Jackson KW, Tang CM and
Loeb DM: Size-based detection of sarcoma circulating tumor cells
and cell clusters. Oncotarget. 8:78965–78977. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xu L, Mao X, Imrali A, Syed F, Mutsvangwa
K, Berney D, Cathcart P, Hines J, Shamash J and Lu YJ: Optimization
and evaluation of a novel size based circulating tumor cell
isolation system. PLoS One. 10:e01380322015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Choo S, Wang P, Newbury R, Roberts W and
Yang J: Reactivation of TWIST1 contributes to Ewing sarcoma
metastasis. Pediatr Blood Cancer. 65:doi:10.1002/pbc.26721. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Crompton BD, Stewart C, Taylor-Weiner A,
Alexe G, Kurek KC, Calicchio ML, Kiezun A, Carter SL, Shukla SA and
Mehta SS: The genomic landscape of pediatric Ewing sarcoma. Cancer
Discov. 4:1326–1341. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Magbanua MJ, Sosa EV, Roy R, Eisenbud LE,
Scott JH, Olshen A, Pinkel D, Rugo HS and Park JW: Genomic
profiling of isolated circulating tumor cells from metastatic
breast cancer patients. Cancer Res. 73:30–40. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Brady SW, Ma X, Bahrami A, Satas G, Wu G,
Newman S, Rusch M, Putnam DK, Mulder HL, Yergeau DA, et al: The
clonal evolution of metastatic osteosarcoma as shaped by cisplatin
treatment. Mol Cancer Res. 17:895–906. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Gutteridge A, Rathbone VM, Gibbons R, Bi
M, Archard N, Davies KEJ, Brown J, Plagnol V, Pillay N, Amary F, et
al: Digital PCR analysis of circulating tumor DNA: a biomarker for
chondrosarcoma diagnosis, prognostication, and residual disease
detection. Cancer Med. 6:2194–2202. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Otsuji K, Takahashi Y, Osako T, Kobayashi
T, Takano T, Saeki S, Yang L, Baba S, Kumegawa K and Suzuki H:
Serial single-cell RNA sequencing unveils drug resistance and
metastatic traits in stage IV breast cancer. NPJ Precis Oncol.
8:2222024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ring A, Nguyen-Sträuli BD, Wicki A and
Aceto N: Biology, vulnerabilities and clinical applications of
circulating tumour cells. Nat Rev Cancer. 23:95–111. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Di Pompo G, Cortini M, Baldini N and Avnet
S: Acid microenvironment in bone sarcomas. Cancers (Basel).
13:131538482021. View Article : Google Scholar
|
|
51
|
Voss TG, Fermin CD, Levy JA, Vigh S, Choi
B and Garry RF: Alteration of intracellular potassium and sodium
concentrations correlates with induction of cytopathic effects by
human immunodeficiency virus. J Virol. 70:5447–5454. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Serpas L, Chan RWY, Jiang P, Ni M, Sun K,
Rashidfarrokhi A, Soni C, Sisirak V, Lee WS, Cheng SH, et al:
Dnase1l3 deletion causes aberrations in length and end-motif
frequencies in plasma DNA. Proc Natl Acad Sci USA. 116:641–649.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Stucker S, Chen J, Watt FE and Kusumbe AP:
Bone angiogenesis and vascular niche remodeling in stress, aging,
and diseases. Front Cell Dev Biol. 8:6022692020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Dalal S, Berry AM, Cullinane CJ, Mangham
DC, Grimer R, Lewis IJ, Johnston C, Laurence V and Burchill SA:
Vascular endothelial growth factor: A therapeutic target for tumors
of the Ewing's sarcoma family. Clin Cancer Res. 11:2364–2378. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kurma K and Alix-Panabières C:
Mechanobiology and survival strategies of circulating tumor cells:
a process towards the invasive and metastatic phenotype. Front Cell
Dev Biol. 11:11884992023. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lee JS, Cho EH, Kim B, Hong J, Kim YG, Kim
Y, Jang JH, Lee ST, Kong SY, Lee W, et al: Clinical practice
guideline for blood-based circulating tumor DNA assays. Ann Lab
Med. 44:195–209. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bettegowda C, Sausen M, Leary RJ, Kinde I,
Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B and Alani RM:
Detection of circulating tumor DNA in early- and late-stage human
malignancies. Sci Transl Med. 6:224ra242014. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Siravegna G, Marsoni S, Siena S and
Bardelli A: Integrating liquid biopsies into the management of
cancer. Nat Rev Clin Oncol. 14:531–548. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ammerlaan W and Betsou F: Biospecimen
science of blood for cfDNA genetic analyses. Curr Pathobiol Rep.
7:9–15. 2019. View Article : Google Scholar
|
|
60
|
Diaz IM, Nocon A, Mehnert DH, Fredebohm J,
Diehl F and Holtrup F: Performance of streck cfDNA blood collection
tubes for liquid biopsy testing. PLoS One. 11:e01663542016.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sathyanarayana SH, Spracklin SB,
Deharvengt SJ, Green DC, Instasi MD, Gallagher TL, Shah PS and
Tsongalis GJ: Standardized workflow and analytical validation of
cell-free DNA extraction for liquid biopsy using a magnetic
bead-based cartridge system. Cells. 14:10622025. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Pittella-Silva F, Chin YM, Chan HT,
Nagayama S, Miyauchi E, Low SK and Nakamura Y: Plasma or serum:
Which is preferable for mutation detection in liquid biopsy? Clin
Chem. 66:946–957. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Andersson D, Kristiansson H, Santamaría
ML, Zafar H, Mijakovic I, Naluai AT and Ståhlberg A: Evaluation of
automatic cell free DNA extraction metrics using different blood
collection tubes. Sci Rep. 15:193642025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Alikian M, Ellery P, Forbes M, Gerrard G,
Kasperaviciute D, Sosinsky A, Mueller M, Whale AS, Milojkovic D,
Apperley J, et al: Next-generation sequencing-assisted DNA-based
digital PCR for a personalized approach to the detection and
quantification of residual disease in chronic myeloid leukemia
patients. J Mol Diagn. 18:176–189. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Arildsen NS, de la Fuente LM, Måsbäck A,
Malander S, Forslund O, Kannisto P and Hedenfalk I: Detecting TP53
mutations in diagnostic and archival liquid-based Pap samples from
ovarian cancer patients using an ultra-sensitive ddPCR method. Sci
Rep. 9:155062019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Romero A, Jantus-Lewintre E, García-Peláez
B, Royuela A, Insa A, Cruz P, Collazo A, Altozano JP, Vidal OJ and
Diz P: Comprehensive cross-platform comparison of methods for
non-invasive EGFR mutation testing: Results of the RING
observational trial. Mol Oncol. 15:43–56. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Gradilone A, Iacovelli R, Cortesi E,
Raimondi C, Gianni W, Nicolazzo C, Petracca A, Palazzo A, Longo F,
Frati L and Gazzaniga P: Circulating tumor cells and ‘suspicious
objects’ evaluated through CellSearch® in metastatic
renal cell carcinoma. Anticancer Res. 31:4219–4221. 2011.PubMed/NCBI
|
|
68
|
Cohen EN, Jayachandran G, Hardy MR,
Subramanian AM, Meng X and Reuben JM: Antigen-agnostic
microfluidics-based circulating tumor cell enrichment and
downstream molecular characterization. PLoS One. 15:e02411232020.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lampignano R, Schneck H, Neumann M, Fehm T
and Neubauer H: Enrichment, isolation and molecular
characterization of EpCAM-negative circulating tumor cells. Adv Exp
Med Biol. 994:181–203. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Benini S, Gamberi G, Cocchi S, Garbetta J,
Alberti L, Righi A, Gambarotti M, Picci P and Ferrari S: Detection
of circulating tumor cells in liquid biopsy from Ewing sarcoma
patients. Cancer Manag Res. 10:49–60. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Machado I, Navarro S, Picci P and
Llombart-Bosch A: The utility of SATB2 immunohistochemical
expression in distinguishing between osteosarcomas and their
malignant bone tumor mimickers, such as Ewing sarcomas and
chondrosarcomas. Pathol Res Pract. 212:811–816. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Conner JR and Hornick JL: SATB2 is a novel
marker of osteoblastic differentiation in bone and soft tissue
tumours. Histopathology. 63:36–49. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Keppens C, Dequeker EMC, Patton SJ,
Normanno N, Fenizia F, Butler R, Cheetham M, Fairley JA, Williams
H, Hall JA, et al: International pilot external quality assessment
scheme for analysis and reporting of circulating tumour DNA. BMC
Cancer. 18:8042018. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
van Dessel LF, Vitale SR, Helmijr JCA,
Wilting SM, van der Vlugt-Daane M, Oomen-de Hoop E, Sleijfer S,
Martens JWM, Jansen MPHM and Lolkema MP: High-throughput isolation
of circulating tumor DNA: A comparison of automated platforms. Mol
Oncol. 13:392–402. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Geeurickx E and Hendrix A: Targets,
pitfalls and reference materials for liquid biopsy tests in cancer
diagnostics. Mol Aspects Med. 72:1008282020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Risberg B, Tsui DWY, Biggs H, de Almagro
ARV, Dawson SJ, Hodgkin C, Jones L, Parkinson C, Piskorz A, Marass
F, et al: Effects of collection and processing procedures on plasma
circulating cell-free DNA from cancer patients. J Mol Diagn.
20:883–892. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Deans ZC, Butler R, Cheetham M, Dequeker
EMC, Fairley JA, Fenizia F, Hall JA, Keppens C, Normanno N,
Schuuring E and Patton SJ: IQN path ASBL report from the first
European cfDNA consensus meeting: expert opinion on the minimal
requirements for clinical ctDNA testing. Virchows Arch.
474:681–689. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
van Dessel LF, Beije N, Helmijr JC, Vitale
SR, Kraan J, Look MP, de Wit R, Sleijfer S, Jansen MP, Martens JW
and Lolkema MP: Application of circulating tumor DNA in prospective
clinical oncology trials-standardization of preanalytical
conditions. Mol Oncol. 11:295–304. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Krumbholz M, Eiblwieser J, Ranft A, Zierk
J, Schmidkonz C, Stütz AM, Peneder P, Tomazou EM, Agaimy A, Bäuerle
T, et al: Quantification of translocation-specific ctDNA provides
an integrating parameter for early assessment of treatment response
and risk stratification in ewing sarcoma. Clin Cancer Res.
27:5922–5930. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Lyskjaer I, Davies C, Strobl AC, Hindley
J, James S, Lalam RK, Cross W, Hide G, Rankin KS, Jeys L, et al:
Circulating tumour DNA is a promising biomarker for risk
stratification of central chondrosarcoma with IDH1/2 and GNAS
mutations. Mol Oncol. 15:3679–3690. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Shukla NN, Patel JA, Magnan H, Zehir A,
You D, Tang J, Meng F, Samoila A, Slotkin EK, Ambati SR, et al:
Plasma DNA-based molecular diagnosis, prognostication, and
monitoring of patients with EWSR1 fusion-positive sarcomas. JCO
Precis Oncol. 2017.PO.16.00028. 2017. View Article : Google Scholar
|
|
82
|
Lyskjær I, Kara N, De Noon S, Davies C,
Rocha AM, Strobl AC, Usher I, Gerrand C, Strauss SJ, Schrimpf D, et
al: Osteosarcoma: Novel prognostic biomarkers using circulating and
cell-free tumour DNA. Eur J Cancer. 168:1–11. 2022. View Article : Google Scholar
|
|
83
|
Van Paemel R, Vandeputte C, Raman L, Van
Thorre J, Willems L, Van Dorpe J, Van Der Linden M, De Wilde J, De
Koker A, Menten B, et al: The feasibility of using liquid biopsies
as a complementary assay for copy number aberration profiling in
routinely collected paediatric cancer patient samples. Eur J
Cancer. 160:12–23. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Zhang H, Gao P, Xiao X, Heger M, Geng L,
Fan B, Yuan Y, Huang C, Chen G, Liu Y, et al: A liquid biopsy-based
method for the detection and quantification of circulating tumor
cells in surgical osteosarcoma patients. Int J Oncol. 50:1075–1086.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Furukawa N, Hasegawa N, Kubota D, Nakamura
Y, Tanaka H, Iwata S, Kawai A, Saito T, Takagi T, Kohsaka S and
Ishijima M: Prognostic potential of fusion gene analysis using
plasma cell-free RNA in malignant bone and soft tissue tumours. BMC
Cancer. 25:5872025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Christodoulou E, Yellapantula V,
O'Halloran K, Xu L, Berry JL, Cotter JA, Zdanowicz A, Mascarenhas
L, Amatruda JF, Ostrow D, et al: Combined low-pass whole genome and
targeted sequencing in liquid biopsies for pediatric solid tumors.
NPJ Precis Oncol. 7:212023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Benje M, Vitacchio T, Fritsche D and
Tinganelli W: Gene expression profiling and phenotypic
characterization of circulating tumor cells derived from a murine
osteosarcoma model. Cancers (Basel). 17:12102025. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Li M, Lu Y, Long Z, Li M, Kong J, Chen G
and Wang Z: Prognostic and clinicopathological significance of
circulating tumor cells in osteosarcoma. J Bone Oncol.
16:1002362019. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Fujiwara T, Uotani K, Yoshida A, Morita T,
Nezu Y, Kobayashi E, Yoshida A, Uehara T, Omori T, Sugiu K, et al:
Clinical significance of circulating miR-25-3p as a novel
diagnostic and prognostic biomarker in osteosarcoma. Oncotarget.
8:33375–33392. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Li Q, Song S, Ni G, Li Y and Wang X: Serum
miR-542-3p as a prognostic biomarker in osteosarcoma. Cancer
Biomark. 21:521–526. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Heishima K, Meuten T, Yoshida K, Mori T
and Thamm DH: Prognostic significance of circulating microRNA-214
and −126 in dogs with appendicular osteosarcoma receiving
amputation and chemotherapy. BMC Vet Res. 15:392019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Georges S, Calleja LR, Jacques C, Lavaud
M, Moukengue B, Lecanda F, Quillard T, Gabriel MT, Cartron PF,
Baud'huin M, et al: Loss of miR-198 and −206 during primary tumor
progression enables metastatic dissemination in human osteosarcoma.
Oncotarget. 9:35726–35741. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Dhir A, Hayashi M, Bodlak A, Oesterheld J,
Loeb DM, Mascarenhas L, Isakoff MS, Sandler ES, Borinstein SC,
Trucco M, et al: Phase II trial of gemcitabine and nab-paclitaxel
for recurrent osteosarcoma with serial monitoring using liquid
biopsy: A report from the national pediatric cancer foundation.
Clin Cancer Res. 30:5314–5322. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Green D, van Ewijk R, Tirtei E, Andreou D,
Baecklund F, Baumhoer D, Bielack SS, Botchu R, Boye K, Brennan B,
et al: Biological sample collection to advance research and
treatment: A fight osteosarcoma through european research and euro
ewing consortium statement. Clin Cancer Res. 30:3395–3406. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Fu Y, Xu Y, Liu W, Zhang J, Wang F, Jian
Q, Huang G, Zou C, Xie X, Kim AH, et al: Tumor-informed deep
sequencing of ctDNA detects minimal residual disease and predicts
relapse in osteosarcoma. EClinicalMedicine. 73:1026972024.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Goodspeed A, Bodlak A, Duffy AB,
Nelson-Taylor S, Oike N, Porfilio T, Shirai R, Walker D, Treece A,
Black J, et al: Single-Cell RNA sequencing of ewing sarcoma tumors
demonstrates transcriptional heterogeneity and clonal evolution.
Clin Cancer Res. 31:2010–2023. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Seidel MG, Kashofer K, Moser T, Thueringer
A, Liegl-Atzwanger B, Leithner A, Szkandera J, Benesch M,
El-Heliebi A and Heitzer E: Clinical implementation of plasma
cell-free circulating tumor DNA quantification by digital droplet
PCR for the monitoring of Ewing sarcoma in children and
adolescents. Front Pediatr. 10:9264052022. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Ma L, Guo H, Zhao Y, Liu Z, Wang C, Bu J,
Sun T and Wei J: Liquid biopsy in cancer: Current status,
challenges and future prospects. Signal Transduct Target Ther.
9:3362024. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Yang W, Nguyen R, Safri F, Shiddiky MJA,
Warkiani ME, George J and Qiao L: Liquid biopsy in hepatocellular
carcinoma: ctDNA as a potential biomarker for diagnosis and
prognosis. Curr Oncol Rep. 27:791–802. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Ge Q, Zhang ZY, Li SN, Ma JQ and Zhao Z:
Liquid biopsy: Comprehensive overview of circulating tumor DNA
(Review). Oncol Lett. 28:5482024. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Coppola CA, De Summa S, Matera G, Pilato
B, Traversa D and Tommasi S: Liquid Biopsy: The challenges of a
revolutionary approach in oncology. Int J Mol Sci. 26:50132025.
View Article : Google Scholar : PubMed/NCBI
|