|
1
|
Enriquez C, Cancila V, Ferri R, Sulsenti
R, Fischetti I, Milani M, Ostano P, Gregnanin I, Mello-Grand M,
Berrino E, et al: Castration-induced downregulation of SPARC in
stromal cells drives neuroendocrine differentiation of prostate
cancer. Cancer Res. 81:4257–4274. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhu S, Tian H, Niu X, Wang J, Li X, Jiang
N, Wen S, Chen X, Ren S, Xu C, et al: Neurotensin and its receptors
mediate neuroendocrine transdifferentiation in prostate cancer.
Oncogene. 38:4875–4884. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sreekumar A and Saini S: Role of MicroRNAs
in neuroendocrine prostate cancer. Non-coding RNA. 8:252022.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bhagirath D, Liston M, Patel N, Akoto T,
Lui B, Yang TL, To DM, Majid S, Dahiya R, Tabatabai ZL and Saini S:
MicroRNA determinants of neuroendocrine differentiation in
metastatic castration-resistant prostate cancer. Oncogene.
39:7209–7223. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang Y, Wei W, Li C, Yan S, Wang S, Xiao
S, He C, Li J, Qi Z, Li B, et al: Idarubicin combats abiraterone
and enzalutamide resistance in prostate cells via targeting XPA
protein. Cell Death Dis. 13:10342022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Patel GK, Chugh N and Tripathi M:
Neuroendocrine differentiation of prostate cancer-An intriguing
example of tumor evolution at play. Cancers (Basel). 11:14052019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wishahi M: Treatment-induced
neuroendocrine prostate cancer and de novo neuroendocrine
prostate cancer: Identification, prognosis and survival, genetic
and epigenetic factors. World J Clin Cases. 12:2143–2146. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Conteduca V, Oromendia C, Eng KW, Bareja
R, Sigouros M, Molina A, Faltas BM, Sboner A, Mosquera JM, Elemento
O, et al: Clinical features of neuroendocrine prostate cancer. Eur
J Cancer. 121:7–18. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang Y and Wang Y, Ci X, Choi SYC, Crea F,
Lin D and Wang Y: Molecular events in neuroendocrine prostate
cancer development. Nat Rev Urol. 18:581–596. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fine SW: Neuroendocrine tumors of the
prostate. Mod Pathol. 31 (Suppl):S122–S132. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Merkens L, Sailer V, Lessel D, Janzen E,
Greimeier S, Kirfel J, Perner S, Pantel K, Werner S and von Amsberg
G: Aggressive variants of prostate cancer: Underlying mechanisms of
neuroendocrine transdifferentiation. J Exp Clin Cancer Res.
41:462022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Beltran H, Prandi D, Mosquera JM, Benelli
M, Puca L, Cyrta J, Marotz C, Giannopoulou E, Chakravarthi BV,
Varambally S, et al: Divergent clonal evolution of
castration-resistant neuroendocrine prostate cancer. Nat Med.
22:298–305. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yamasaki M, Nomura T, Sato F and Mimata H:
Chronic hypoxia induces androgen-independent and invasive behavior
in LNCaP human prostate cancer cells. Urologic Oncology: Seminars
and Original Investigations Elsevier. 1124–1131. 2013. View Article : Google Scholar
|
|
14
|
Danza G, Di Serio C, Rosati F, Lonetto G,
Sturli N, Kacer D, Pennella A, Ventimiglia G, Barucci R, Piscazzi
A, et al: Notch signaling modulates hypoxia-induced neuroendocrine
differentiation of human prostate cancer cells. Mol Cancer Res.
10:230–238. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ammirante M, Shalapour S, Kang Y, Jamieson
CA and Karin M: Tissue injury and hypoxia promote malignant
progression of prostate cancer by inducing CXCL13 expression in
tumor myofibroblasts. Proc Natl Acad Sci. 111:14776–14781. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
White RWd, Vinall RL, Tepper CG and Shi
X-B: MicroRNAs and their potential for translation in prostate
cancer. Urologic Oncology: Seminars and Original Investigations
Elsevier. 307–311. 2009. View Article : Google Scholar
|
|
17
|
Shao L, Chen Z, Soutto M, Zhu S, Lu H,
Romero-Gallo J, Peek R, Zhang S and El-Rifai W: Helicobacter
pylori-induced miR-135b-5p promotes cisplatin resistance in gastric
cancer. FASEB J. 33:264–274. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu D, Jin Y, Wu J, Zhu H and Ye D:
MiR-135b-5p is an oncogene in pancreatic cancer to regulate GPRC5A
expression by targeting transcription factor KLF4. Cell Death
Discov. 8:232022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aakula A, Leivonen SK, Hintsanen P,
Aittokallio T, Ceder Y, Børresen-Dale AL, Perälä M, Östling P and
Kallioniemi O: MicroRNA-135b regulates ERα, AR and HIF1AN and
affects breast and prostate cancer cell growth. Mol Oncol.
9:1287–1300. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hu CD, Choo R and Huang J: Neuroendocrine
differentiation in prostate cancer: A mechanism of radioresistance
and treatment failure. Front Oncol. 5:902015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mohamed OA, Tesen HS, Hany M, Sherif A,
Abdelwahab MM and Elnaggar MH: The role of hypoxia on prostate
cancer progression and metastasis. Mol Biol Rep. 50:3873–3884.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Javan FN, Aryana K and Askari E: Prostate
cancer with neuroendocrine differentiation recurring after
treatment with 177Lu-PSMA: A chance for: 177: Lu-DOTATATE Therapy?
Clin Nucl Med. 46:e480–e482. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xing N, Qian J, Bostwick D, Bergstralh E
and Young CY: Neuroendocrine cells in human prostate over-express
the anti-apoptosis protein survivin. Prostate. 48:7–15. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gong J, Lee J, Akio H, Schlegel PN and
Shen R: Attenuation of apoptosis by chromogranin A-induced Akt and
survivin pathways in prostate cancer cells. Endocrinology.
148:4489–4499. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Segal NH, Cohen RJ, Haffejee Z and Savage
N: BCL-2 proto-oncogene expression in prostate cancer and its
relationship to the prostatic neuroendocrine cell. Arch Pathol Lab
Med. 118:616–618. 1994.PubMed/NCBI
|
|
27
|
Gururajan M, Cavassani KA, Sievert M, Duan
P, Lichterman J, Huang JM, Smith B, You S, Nandana S, Chu GC, et
al: SRC family kinase FYN promotes the neuroendocrine phenotype and
visceral metastasis in advanced prostate cancer. Oncotarget.
6:44072–44083. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Vlachostergios PJ, Puca L and Beltran H:
Emerging variants of castration-resistant prostate cancer. Curr
Oncol Rep. 19:322017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Vaupel P and Mayer A: Hypoxia in cancer:
Significance and impact on clinical outcome. Cancer Metastasis Rev.
26:225–239. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Semenza GL: Oxygen sensing,
hypoxia-inducible factors, and disease pathophysiology. Annu Rev
Pathol. 9:47–71. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin TP, Chang YT, Lee SY, Campbell M, Wang
TC, Shen SH, Chung HJ, Chang YH, Chiu AW, Pan CC, et al: REST
reduction is essential for hypoxia-induced neuroendocrine
differentiation of prostate cancer cells by activating autophagy
signaling. Oncotarget. 7:26137–26151. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Nallamshetty S, Chan SY and Loscalzo J:
Hypoxia: A master regulator of microRNA biogenesis and activity.
Free Radic Biol Med. 64:20–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Davies A, Zoubeidi A and Selth LA: The
epigenetic and transcriptional landscape of neuroendocrine prostate
cancer. Endocr Relat Cancer. 27:R35–R50. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lu M, Huang Y, Sun W, Li P and Li L and Li
L: miR-135b-5p promotes gastric cancer progression by targeting
CMTM3. Int J Oncol. 52:589–598. 2017.PubMed/NCBI
|
|
35
|
Yang Y, Ishak Gabra MB, Hanse EA, Lowman
XH, Tran TQ, Li H, Milman N, Liu J, Reid MA, Locasale JW, et al:
MiR-135 suppresses glycolysis and promotes pancreatic cancer cell
adaptation to metabolic stress by targeting phosphofructokinase-1.
Nat Commun. 10:8092019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shao Y, Zhang S, Pan Y, Peng Z and Dong Y:
miR-135b: A key role in cancer biology and therapeutic targets.
Non-coding RNA Res. 12:67–80. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hudson C: Regulation of hypoxia-inducible
factor 1: Expression and function by the mammalian target of
rapamycin. Mol Cell. 11:14912003.
|
|
39
|
Wu F, Chen Z, Liu J and Hou Y: The
Akt-mTOR network at the interface of hematopoietic stem cell
homeostasis. Exp Hematol. 103:15–23. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shorning BY, Dass MS, Smalley MJ and
Pearson HB: The PI3K-AKT-mTOR pathway and prostate cancer: at the
crossroads of AR, MAPK, and WNT signaling. Int J Mol Sci.
21:45072020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhu Y, Zang Y, Zhao F, Li Z, Zhang J, Fang
L, Li M, Xing L, Xu Z and Yu J: Inhibition of HIF-1α by PX-478
suppresses tumor growth of esophageal squamous cell cancer in vitro
and in vivo. Am J Cancer Res. 7:1198–1212. 2017.PubMed/NCBI
|















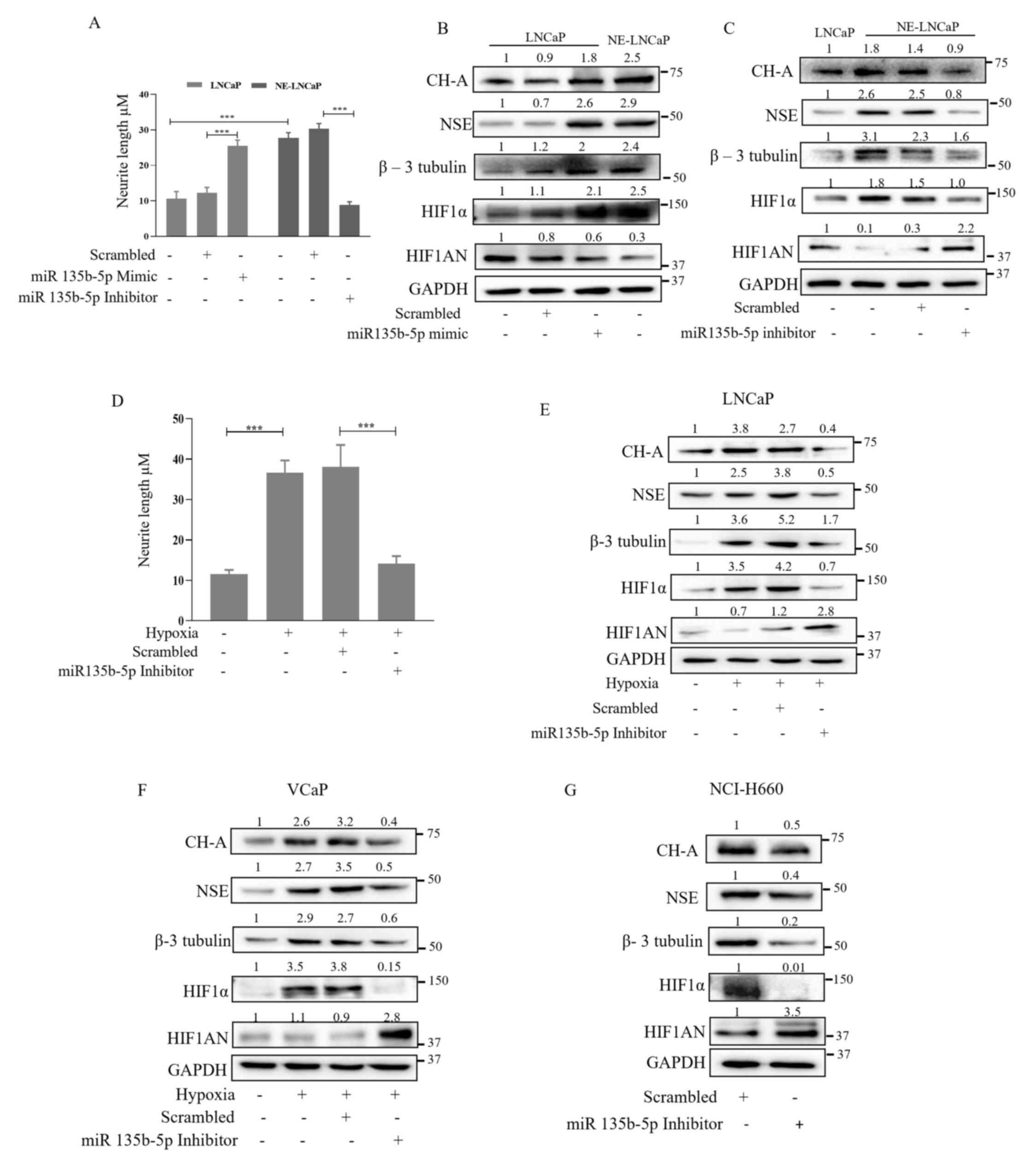
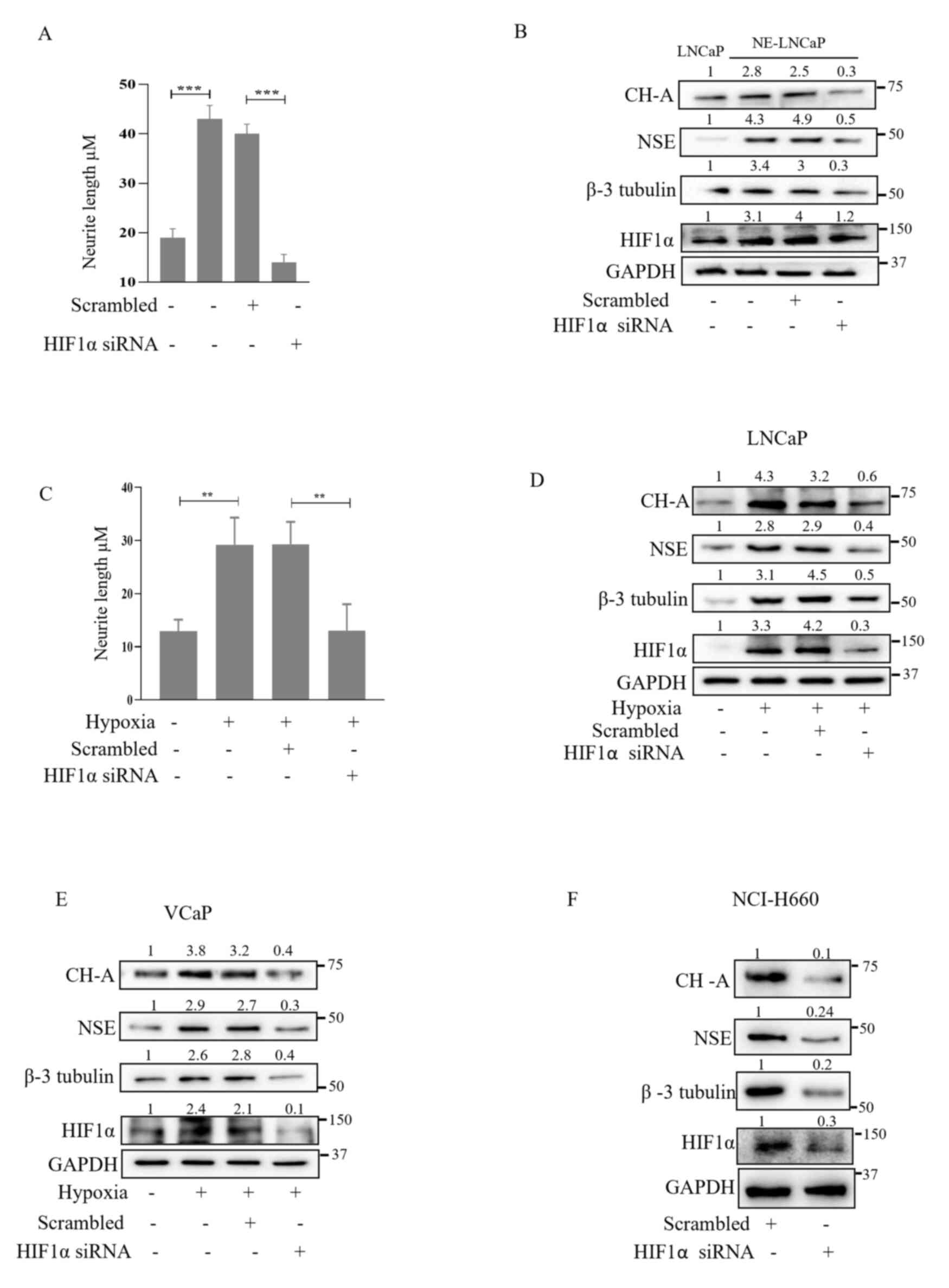
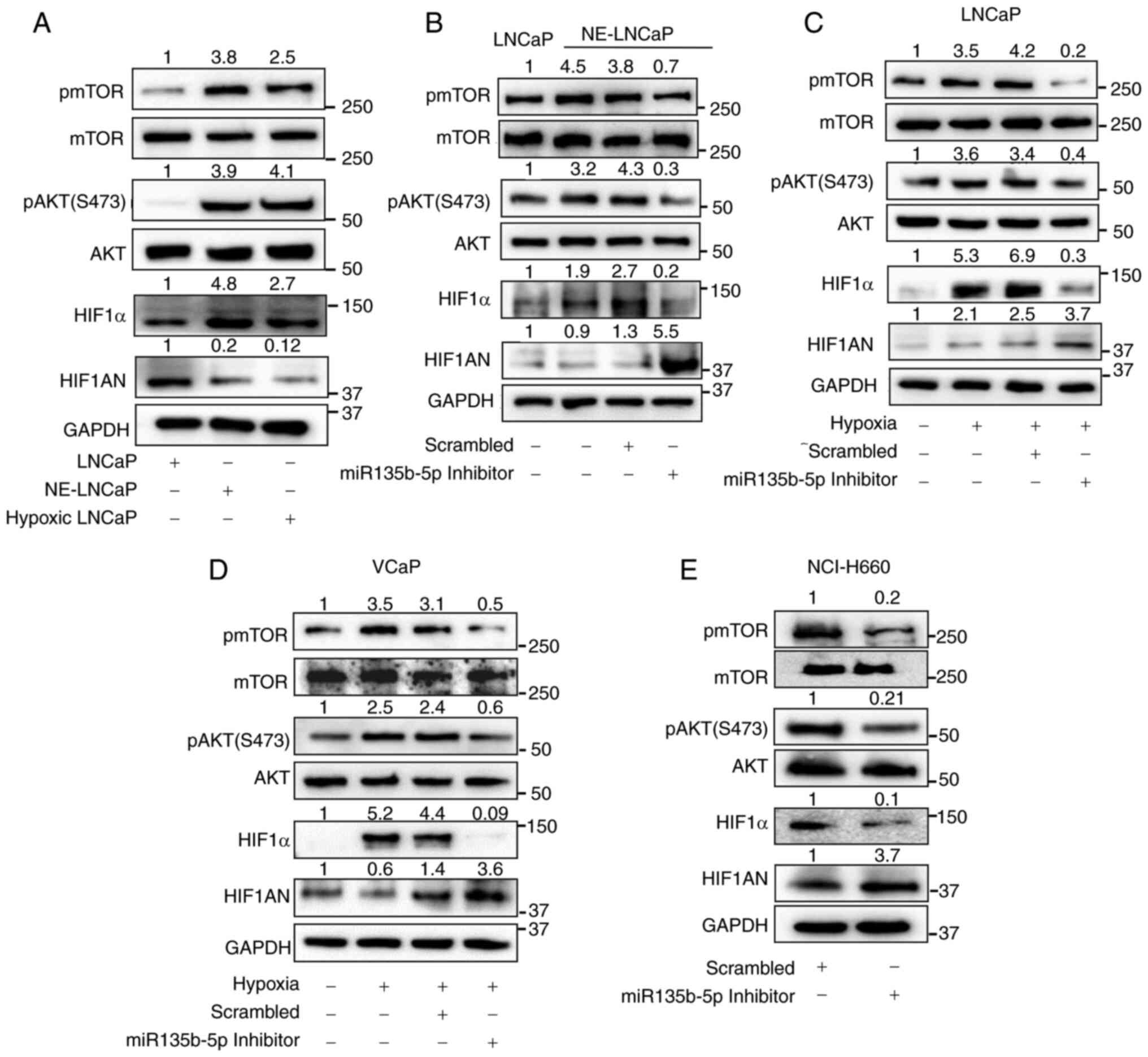
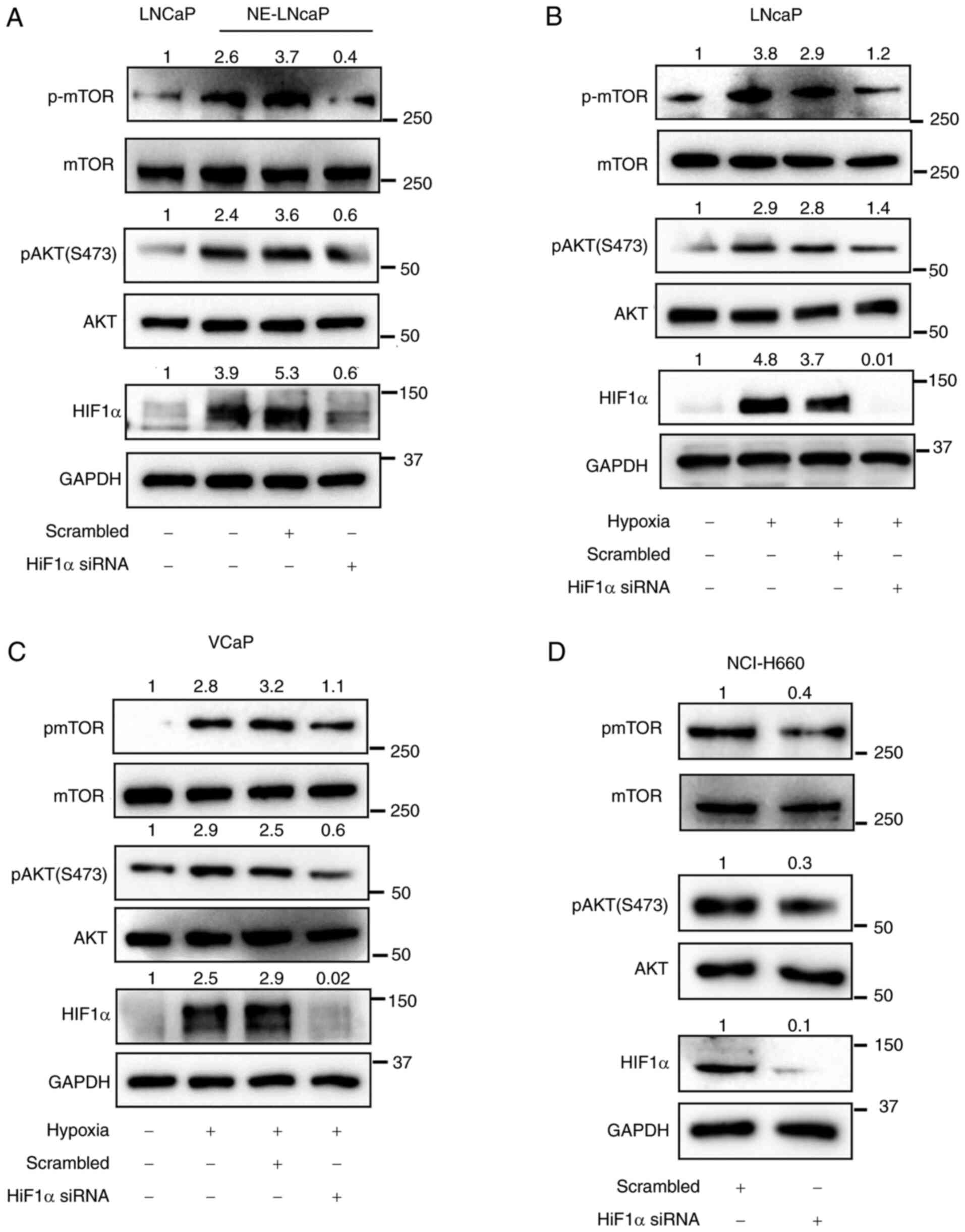
![PX-478, a HIF1α inhibitor, suppresses
NE differentiation of PCa cells. (A and B) NE-LNCaP cells were
treated with PX-478, and the expression of NE markers, AKT/mTOR
pathway components, and HIF1α was assessed by western blotting.
(C-F) LNCaP and VCaP cells were treated with PX-478 under hypoxic
conditions, and changes in NE markers, AKT/mTOR pathway proteins,
and HIF1α were analyzed by western blotting. (G and H) NCI-H660
cells were treated with PX-478, and the expression of NE markers,
AKT/mTOR pathway components, and HIF1α was determined by western
blotting. PX-478, S-2-amino-3-[4′-N, N,-bis (chloroethyl) amino]
phenyl propionic acid N-oxide dihydrochloride; NE, neuroendocrine;
HIF1α, hypoxia inducible factor alpha-1; CH-A chromogranin A.](/article_images/or/55/4/or-55-04-09079-g06.jpg)