Introduction
Based on epidemiological data in 2020, primary liver
cancer is the third leading cause of cancer-associated death
globally, accounting for ~830,2000 deaths. Hepatocellular carcinoma
(HCC) accounts for 90% of the primary liver cancer incidence
(1,2). Over the past two decades, molecular
targeted agents have emerged as promising therapeutic options for
HCC (3). However, the prognosis of
patients with HCC remains unfavorable due to the aggressive growth
and metastasis of this cancer. Therefore, a deeper understanding of
HCC pathogenesis remains essential.
Chaperonins are a cluster of molecular chaperones
with sizes ranging from 55 to 64 kDa (4) that are divided into groups I and II
according to their distinct encapsulation mechanism. The tailless
complex polypeptide 1 (TCP1) ring complex belongs to group II and
comprises eight distinct subunits (5). The classical functions of the TCP1
ring complex include a chaperone that is key for the correct
folding of polypeptide substrates and an autophagy receptor
involved in regulating the degradation of solid protein aggregates
(6,7). Numerous chaperonin-containing TCP1
(CCT) subunits have been shown to regulate HCC progression;
specifically, knockdown of CCT3 sensitizes HCC cells to sorafenib
and enhances sorafenib-mediated ferroptosis (8). Inhibiting CCT4 induces securin and Bim
accumulation, which causes HCC cell apoptosis (9). Depletion of CCT8 arrests the cell
cycle in the G0/G1 phase by inhibiting CDK2 and cyclin E expression
in Huh-7 cells (10). In
silico analysis has indicated that CCT2 expression is markedly
upregulated in breast and pancreatic cancer and thyroid carcinoma
(11). The clinical value and
biological functions of CCT2 in HCC progression remain poorly
understood, however, CCT2 has been observed to promote tumor
viability, stemness and metastasis in breast and lung cancer
(12,13).
STAT3 is the most well-known member of the STAT
family (14). Phosphorylation of
STAT3 at the Tyr705 and Ser727 residues are prerequisites of STAT3
signaling activation (15).
Although over-activation of STAT3 is noted in numerous types of
liver disease, including viral hepatitis (16), hepatic ischemia-reperfusion injury
(17) and liver fibrosis (18), its role in HCC is notable as the
STAT3 signaling pathway regulates the proliferation, stemness and
metastasis of HCC cells (19).
However, the regulatory effects of CCT2 in STAT3 activation remain
ambiguous. Chen et al (20)
reported that over-activation of STAT3 signaling is crucial for the
CCT2-mediated proliferation and invasion of triple-negative breast
cancer cells. By contrast, Vallin et al (21) found that the levels of STAT3
phosphorylation at Tyr705 increase following CCT2 knockout in the
estrogen receptor positive and progesterone receptor positive MCF-7
breast cancer cell line. Genetic heterogeneity between cell lines
may be how CCT2 achieves different roles in the STAT3 signaling
pathway. The association between CCT2 expression and STAT3
signaling activation in HCC remains unclear.
The present study systematically investigated the
effects of CCT2 knockdown on the malignant properties and STAT3
signaling activation in HCC cells. The findings of the present
study may facilitate the translational application of CCT2 in HCC
molecular targeted therapy in the future.
Materials and methods
Public dataset acquisition
The Cancer Genome Atlas-Liver HCC (TCGA-LIHC)
(22), International Cancer Genome
Consortium-Liver Cancer-Riken, Japan (ICGC-LIRI-JP) (23) and OEP00000321 (24) preprocessed datasets were downloaded
from the HCC database (lifeome.net:809/#/download) (25). Based on the median expression of
CCT2 calculated in HCC tissue (11.17 in TCGA-LIHC, 5.40 in
ICGC-LIRI-JP and 11.41 in NODE-OEP00000321), patients were
stratified into high and low CCT2 expression groups. The
association between CCT2 expression and overall survival was
assessed by Kaplan-Meier analysis using GraphPad Prism 10 software
(Dotmatics).
Cell culture
The HCC cell lines Huh-7 (cat. no. STCC10102G),
Hep3B (cat. no. STCC10103G), Li-7 (cat. no. STCC10107G) and HCCLM3
(cat. no. STCC10111G) were provided by Wuhan Servicebio Technology
Co., Ltd. STR profiling was performed to ensure the authenticity of
cells. Complete culture medium was prepared with DMEM (cat. no.
C11995500BT) supplemented with 10% FBS (cat. no. A5256701; both
Gibco; Thermo Fisher Scientific, Inc.) and 1%
penicillin-streptomycin solution (cat. no. P4333; Sigma-Aldrich;
Merck KGaA). The cells were incubated in complete culture medium at
37°C under a humidified environment with 5% CO2.
Recombinant human IL-6 (cat. no. HY-P7044; MedChemExpress) as used
for STAT3 activation. In brief, Huh-7 and HCCLM3 cells were treated
with IL-6 (50 ng/ml) or PBS at 37°C for 24 h and cells were
collected for further experiments.
Lentivirus transduction
Huh-7 and HCCLM3 cells were seeded into 6-well
plates at a density of 1×105 cells/well and incubated at
37°C overnight. As previously described (26), shRNA plasmids and packaging plasmids
were co-transfected into 293T cells at 37°C for 8 h. Mature
lentiviral particles (1×108 TU/ml) carrying
CCT2-specific short hairpin (sh)RNA (sh-CCT2) or negative control
shRNA (sh-NC) were provided by Shanghai GeneChem Co., Ltd. The
sequence for human CCT2 shRNA was 5′-TTCATCCACAGACCATCATAG-3′. The
sequence for human sh-NC was 5′-TTCTCCGAACGTGTCACGT-3′. The
lentiviral transduction of HCC cells was performed at a
multiplicity of infection of 10. Following 12 h transduction at
37°C, the viral medium was replaced with fresh complete medium. To
select stably transduced cells, puromycin was applied at a
concentration of 5 µg/ml for 48 h. The viable cells were maintained
in culture medium containing 1.25 µg/ml puromycin for continuous
passaging. Expression of CCT2 was detected by reverse
transcription-quantitative PCR (RT-q)PCR and western blot analysis.
Subsequent experiments were performed ≥72 h after lentiviral
transduction.
EdU incorporation assay
Huh-7 and HCCLM3 cells were cultured in 6-well
plates at an initial seeding density of 1×105
cells/well. Following a 3 h light-protected incubation at 37°C with
0.1% EdU reagent (cat. no. C10310-1; Guangzhou RiboBio Co., Ltd.),
the cells were fixed using 4% paraformaldehyde (PFA) for 30 min at
room temperature. The incorporated EdU was detected by treating the
cells with Apollo staining reagent (cat. no. C10371-1; Guangzhou
RiboBio Co., Ltd.) for 30 min at room temperature. The cell nuclei
were stained with Hoechst 33342 reagent for 30 min at room
temperature. Fluorescence images were captured using the Operetta
CLS high-content analysis system (PerkinElmer, Inc.). In total,
five visual fields were chosen at random and the number of EdU
positive cells was evaluated using ImageJ software (version 1.53 K;
National Institutes of Health). The proportion of EdU-positive
cells was calculated as follows: (EdU-positive cells/total cells)
×100%.
Colony formation assay
Huh-7 and HCCLM3 cells were cultured in 6-well
plates at an initial seeding density of 1×103
cells/well. The complete DMEM in the wells was refreshed at 3 day
intervals. Following 14 day culture, 4% PFA and 0.1% crystal violet
were added to the plates for fixation (30 min at room temperature)
and staining (20 min at room temperature), respectively. The cell
colonies consisting of ≥40 cells were observed using the ECLIPSE
Ts2 inverted light microscope (Nikon Corporation) and counted using
ImageJ software.
Flow cytometry
Early and late apoptotic cells were labeled using
Annexin V-PE/7-AAD (cat. no. KGA1104; Nanjing KeyGen Biotech Co.,
Ltd.). Huh-7 and HCCLM3 cells were digested with EDTA-free trypsin.
Then, 1×105 cells were resuspended in 500 µl binding
buffer working solution to prepare a single-cell suspension. For
each 500 µl cell suspension, 1 µl Annexin V-PE and 5 µl 7-AAD were
sequentially added. After mixing by vortexing, the tubes were kept
in the dark for 10 min to ensure complete reactions. The FACSAria
II flow cytometer (BD Biosciences) was used to measure the
percentage of apoptotic cells. Cell apoptosis was analyzed by
FACSDiva Software (v6.1.3, BD Biosciences).
Apo-ONE homogeneous caspase-3/7
assay
Huh-7 or HCCLM3 cells were resuspended in DMEM at a
density of 1×105 cells/ml. Then, 100 µl Apo-ONE
caspase-3/7 substrate working solution (cat. no. G7792; Promega
Corporation) and 100 µl cell suspension were sequentially added to
a black 96-well cell culture plate. The contents were mixed using a
plate shaker. The cells were incubated at room temperature,
protected from the light, for 8 h. The fluorescence of each well
was measured using the FL 6500 spectrofluorometer (PerkinElmer,
Inc.), as previously described (27).
Gap closure assay
Ibidi Culture-Insert 25 Well plates (cat. no. 80209;
ibidi GmbH) were used for the gap closure assay. 3×105
Huh-7 or HCCLM3 cells were cultured in the wells until they formed
100% confluent monolayers, at which point the inserts were removed.
The cells were cultured in serum-free DMEM (Thermo Fisher
Scientific, Inc.) at 37°C for an additional 48 h. In total, five
independent repeats were performed for each group. Images were
captured by the ECLIPSE Ts2 inverted light microscope at 0 and 48
h. The gap area was measured by ImageJ software and calculated as
follows: Gap closure ratio (%)=[1-(area at 48 h/area at 0 h)]
×100%.
Transwell assay
DMEM-Matrigel (10%; cat. no. 354234; Corning, Inc.)
was used to precoat the upper surface of permeable Transwell insert
membranes (cat. no. 3464; Corning, Inc.) for 2 h at 37°C. The
inserts were placed in a 24-well plate. Next, 2×104
Huh-7 or HCCLM3 cells were resuspended in 200 µl serum-free DMEM
and added to the upper chamber of the Transwell insert, while the
lower chamber was filled with 600 µl complete DMEM. Following a 24
h culture at 37°C, the inserts were incubated with 4% PFA and 0.1%
crystal violet for fixation (30 min at room temperature) and
staining (20 min at room temperature), respectively. The
successfully invaded cells were counted under the ECLIPSE Ts2
inverted light microscope.
Tumor sphere formation assay
Suspended single Huh-7 or HCCLM3 cells
(1×103) were cultured with Tumor Stem Cell Pellet
Culture Medium (serum-free; cat. no. P2401; Shanghai QiDa
Biotechnology Co., Ltd) in ultra-low attachment plates (24-well;
cat. no. 3473; Corning, Inc.). Following 14 days incubation at
37°C, cell spheres were counted under the ECLIPSE Ts2 inverted
light microscope. The spheroid formation efficiency (SFE) was
calculated as follows: SFE (%)=number of spheres/1,000×100%.
Furthermore, tumor spheres were harvested and digested into a
single-cell suspension for EdU incorporation assay and Transwell
assay.
RT-qPCR
Total RNA from Huh-7 and HCCLM3 cells was extracted
using TRIzol™ reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's instructions. RT-qPCR was performed
using the SuperScript™ III Platinum™ One-Step qRT-PCR kit (cat. no.
11732020; Invitrogen; Thermo Fisher Scientific, Inc.). The one-step
thermocycling conditions were as follows: 50°C for 15 min to
synthesize cDNA; 95°C for 2 min to denaturalize the cDNA and
activate the Taq DNA Polymerase; 40 cycles of 95°C for 15 sec
(denaturation) and 60°C for 30 sec (annealing and extension). The
2−∆∆Cq method was used to calculate the relative
expression of the target genes (28,29).
The primers were as follows: CCT2 forward,
5′-GCACTACCTCTGTTACCGTTTT-3′ and reverse,
5′-CTTCTCTCCAACCCGCTATGA-3′ and β-actin (reference gene) forward,
5′-CATGTACGTTGCTATCCAGGC-3′ and reverse,
5′-CTCCTTAATGTCACGCACGAT-3′.
Western blotting
RIPA buffer (cat. no. P0013B) and the BCA Protein
Assay kit (cat. no. P0012; both Beyotime Institute of
Biotechnology) were used to extract the total protein from Huh-7
and HCCLM3 cells and measure the protein concentration,
respectively. The same mass of protein (40 µg/lane) was separated
by 4–20% SDS-PAGE (cat. no. P0468S; Beyotime Institute of
Biotechnology) and then transferred onto PVDF membranes (cat. no.
IPVH00010; MilliporeSigma). The membranes were washed with TBS for
10 min at room temperature. The membranes were blocked with 5%
fat-free milk for 1 h at room temperature followed by incubation
with primary antibodies (1:1,000 using 5% fat-free milk) at 4°C
overnight. The primary antibodies were as follows: CCT2 (cat. no.
24896-1-AP), β-actin (cat. no. 66009-1-Ig), MMP2 (cat. no.
10373-2-AP), myeloid cell leukemia sequence 1 (MCL1; cat. no.
16225-1-AP) and SRY-box transcription factor 2 (SOX2; cat. no.
11064-1-AP; all Proteintech Group, Inc.) and STAT3 (cat. no. 4904)
and phosphorylated (p-)STAT3 (Tyr705; cat. no. 4113; both Cell
Signaling Technology, Inc.) The membranes were washed three times
in TBST (0.1% Tween-20) for 5 min each at room temperature. The
proteins were labeled with HRP-conjugated secondary antibodies
(1:5,000 in 5% fat-free milk; cat. nos. SA00001-1 and SA00001-2;
Proteintech Group, Inc.) for 1 h at room temperature. The membranes
were washed three times in TBST for 5 min each at room temperature.
Chemiluminescent signals were detected by an enhanced
chemiluminescence kit (cat. no. WBKLS0500; MilliporeSigma). The
band densitometry was quantified by ImageJ software.
Subcutaneous xenograft and
hematogenous lung metastasis model
A total of 20 BALB/c (age, 4–6 weeks; weight, 16–18
g) male nude mice were acquired from SPF Biotechnology Co. Ltd. and
raised in a specialized pathogen-free environment (25°C, 45%
humidity, 12/12-h light/dark period and ad libitum access to
diet and water). The institutional animal care and use committee of
SPF Biotechnology Co., Ltd (Beijing, China). approved the animal
experiment protocol (approval no. AWE2023102504). Nude mice were
randomly assigned into four groups (sh-NC and sh-CCT2 for the
xenograft and hematogenous lung metastasis model; n=5/group) using
random number table method. For the xenograft model, a 150 µl
single-cell suspension containing 2×106 transduced
HCCLM3 cells (with sh-NC or sh-CCT2) was administered via
subcutaneous injection into the left forelimb. Once the
subcutaneous tumors were palpable, they were monitored three times
per week. Tumor diameters were measured using a vernier caliper.
Tumor volumes were determined as follows: Volume (mm3)=a
× b2x0.5, where a indicates the length and b indicates
the width. The tumor volume did not exceed 2,000 mm3, in
compliance with animal ethics requirements. For the lung metastasis
model, a single cell suspension of 1×105 transduced
HCCLM3 cells (with sh-NC or sh-CCT2) in 100 µl sterile saline was
administered into mice via tail vein injection. After 28 days, all
animals were placed in a chamber ventilated with CO2 at
30–70% of its volume/min. The mice were left in the chamber for ≥5
min after respiratory arrest and observed to ensure the cessation
of breathing and heartbeat. Cervical dislocation (with the
confirmation of a gap between the skull and spinal column) was used
to ensure the death. For the xenograft model, subcutaneous tumors
were dissected and weighed. For the lung metastasis model, both
lungs were dissected. The tumor and lung were fixed with 4% PFA for
30 min at room temperature for further experiments.
Immunohistochemistry and hematoxylin
and eosin (H&E) staining
PFA-fixed subcutaneous tumor and lung tissue were
first embedded in paraffin and then sliced into 4-µm-thick
sections. The sections were dewaxed in xylene (three times for 10
min each) and rehydrated through graded ethanol (100, 95, 90, 80
and 70%, 5 min each). For immunohistochemistry, antigen retrieval
was performed by microwaving at 95°C for 10 min with Tris-EDTA
buffer (pH 9.0; cat. no. PR30002; Proteintech Group, Inc.).
Endogenous peroxidase activity quenching and non-specific antigen
blocking were performed using IHC Detect kit for Rabbit/Mouse
Primary Antibody (cat. no. PK10006; Proteintech Group, Inc.). The
primary antibodies, including p-STAT3 (cat. no. 4113, Cell
Signaling Technology), MCL1 (cat. no. 16225-1-AP), MMP2 (cat. no.
10373-2-AP) and SOX2 (cat. no. 11064-1-AP; all Proteintech Group,
Inc.), were diluted 1:100 in antibody diluent (cat. no. PR30016;
Proteintech Group, Inc.) and applied at 4°C overnight. Afterward,
the sections were washed with PBS to eliminate any unbound primary
antibodies and subsequently incubated with 100 µl ready-to-use
HRP-conjugated polymer secondary antibodies (cat. no. RGAU011,
Proteintech Group, Inc.) for 30 min at room temperature. DAB was
used to visualize proteins. Hematoxylin reagent was utilized to
stain the nuclei for 1 min at room temperature.
For H&E staining, the sections were dipped in
hematoxylin reagent for 5 min at room temperature followed by
washing in flowing tap water. Then, the sections were immersed in
acid alcohol (1% hydrochloric acid in 70% alcohol) for 5 sec and
washed in flowing tap water until they turned blue. Eosin solution
(1%) was used to stain the sections for 20 sec at room
temperature.
All sections were imaged using the ECLIPSE Ts2
inverted light microscope. The mean optical density was calculated
by ImageJ software to assess the expression of the target protein
in the subcutaneous tumor tissues. The number of lung nodules was
counted to evaluate the tumor metastatic capacity.
Statistical analysis
Quantitative data are reported as the mean ±
standard deviation of ≥3 independent experimental repeats. The
unpaired Student's t-test was performed to evaluate differences
between two groups when data passed Shapiro-Wilk normality and
equal variance test; otherwise, the Mann-Whitney test was used. The
differences between >2 groups were compared by one-way ANOVA,
with Tukey's post hoc test. GraphPad Prism 10 software (Dotmatics)
was used for data analysis. P<0.05 was considered to indicate a
statistically significant difference.
Results
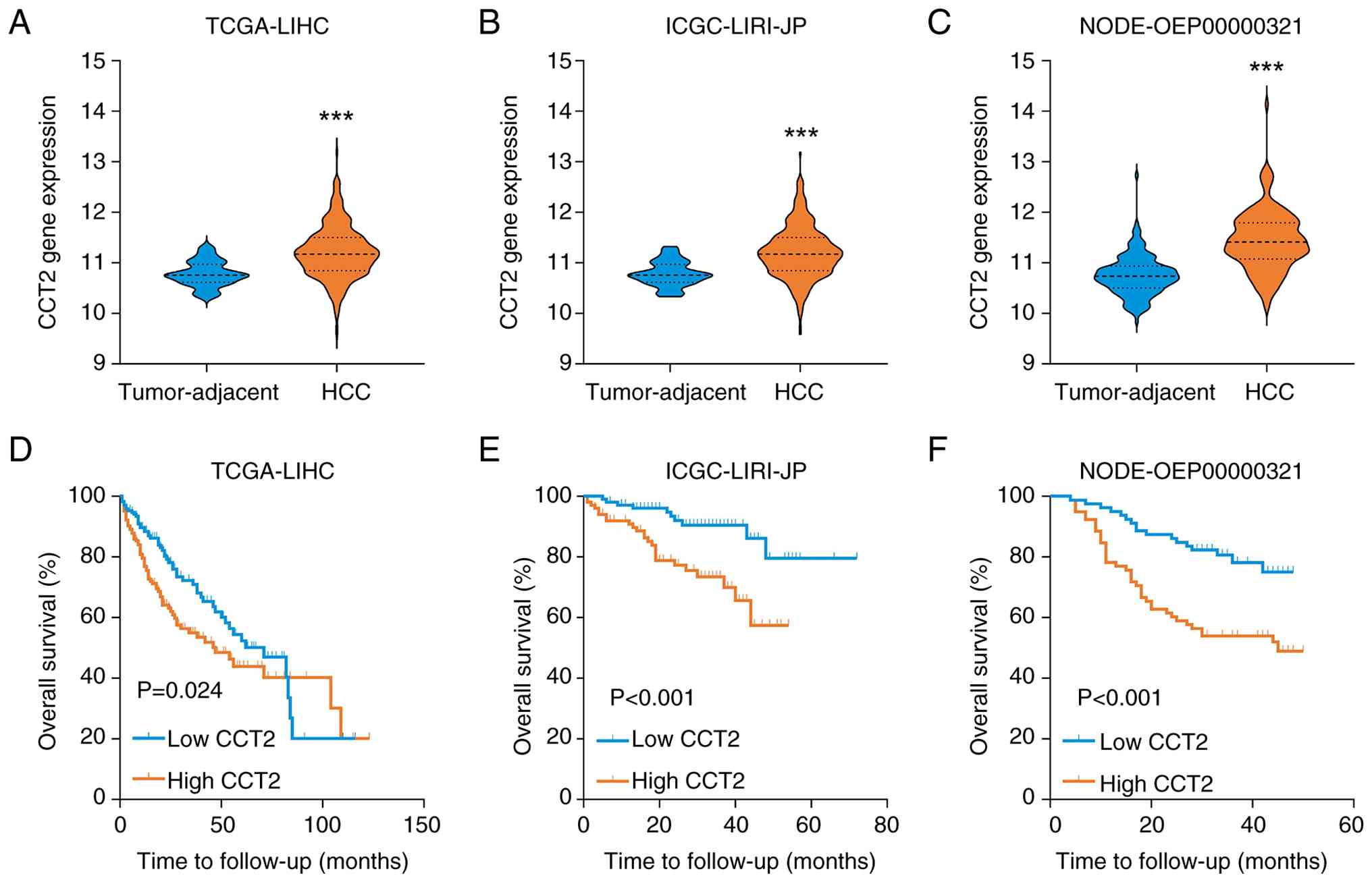
Upregulated CCT2 expression is
associated with poor prognosis in HCC
To evaluate the clinical significance of CCT2 in
HCC, the association between CCT2 expression and OS was analyzed
using multiple public HCC datasets. The results across all datasets
demonstrated CCT2 expression was upregulated in HCC tissues
compared with tumor-adjacent tissue (Fig. 1A-C). Kaplan-Meier curve analyses
demonstrated that patients with HCC with high levels of CCT2 had a
poorer OS prognosis. The hazard ratios (HRs) for OS were as
follows: HR=1.51 [95% confidence interval (CI), 1.06–2.16; P=0.024;
Fig. 1D] in TCGA-LIHC cohort,
HR=3.12 (95% CI, 1.59–6.11; P<0.001; Fig. 1E) in the ICGC-LIRI-JP cohort and
HR=2.59 (95% CI, 1.51–4.43; P<0.001; Fig. 1F) in the NODE-OEP00000321 cohort.
Taken together, high expression of CCT2 may be considered a risk
factor for patients with HCC.
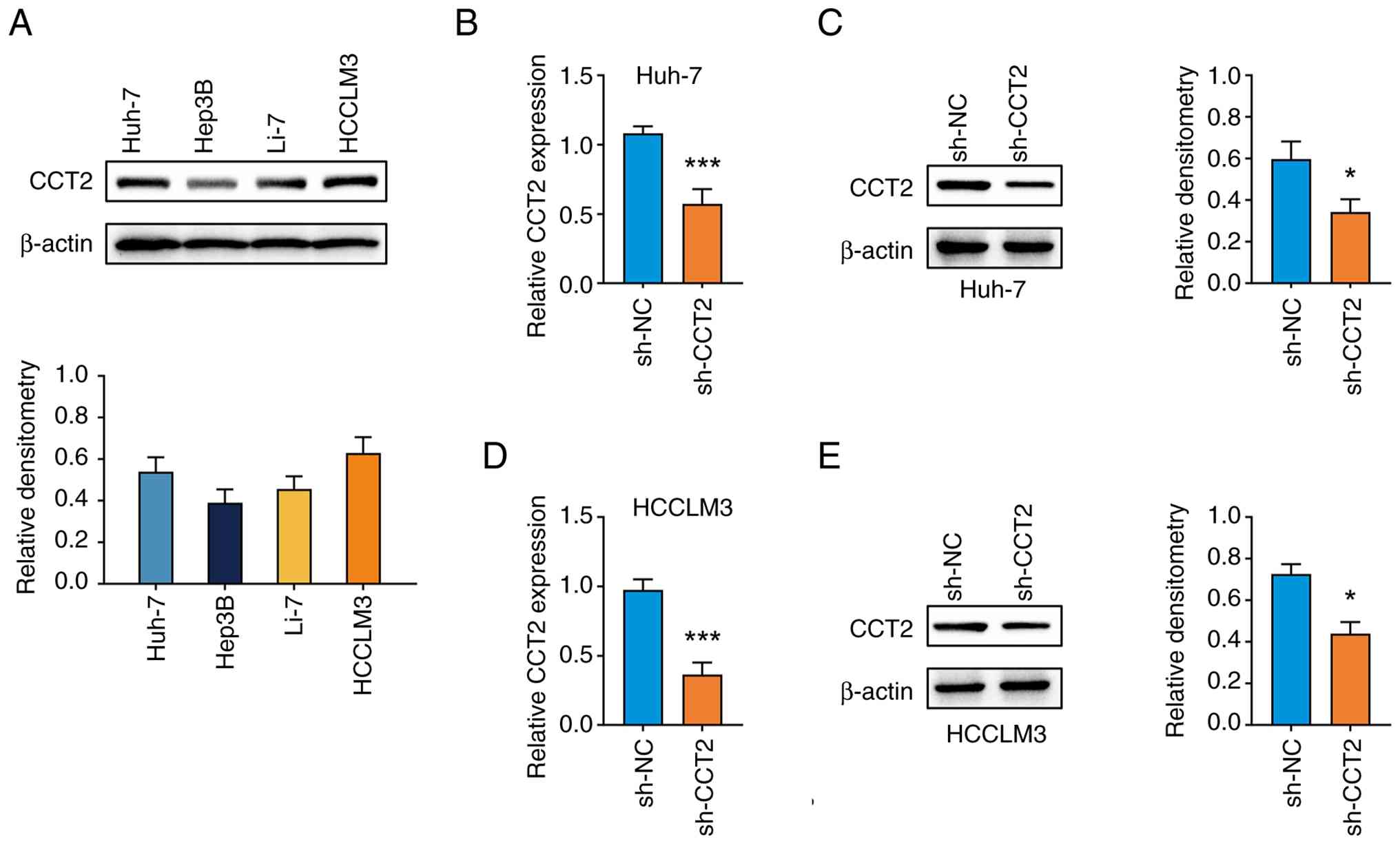
CCT2 expression is inhibited in Huh-7
and HCCLM3 cells by lentivirus transduction
To elucidate the biological functions of CCT2 in HCC
malignancy, the expression of CCT2 in HCC cell lines was detected
by western blotting (Fig. 2A).
Huh-7 and HCCLM3 cell lines were selected for subsequent
experiments due to their relatively high levels of CCT2 expression.
CCT2 expression was knocked down in Huh-7 and HCCLM3 cells by
lentivirus-mediated sh-CCT2 transduction. RT-qPCR and western
blotting confirmed that, compared with the sh-NC group, the mRNA
(Fig. 2B and D) and protein
(Fig. 2C and E) levels of CCT2 were
significantly downregulated in the sh-CCT2 group.
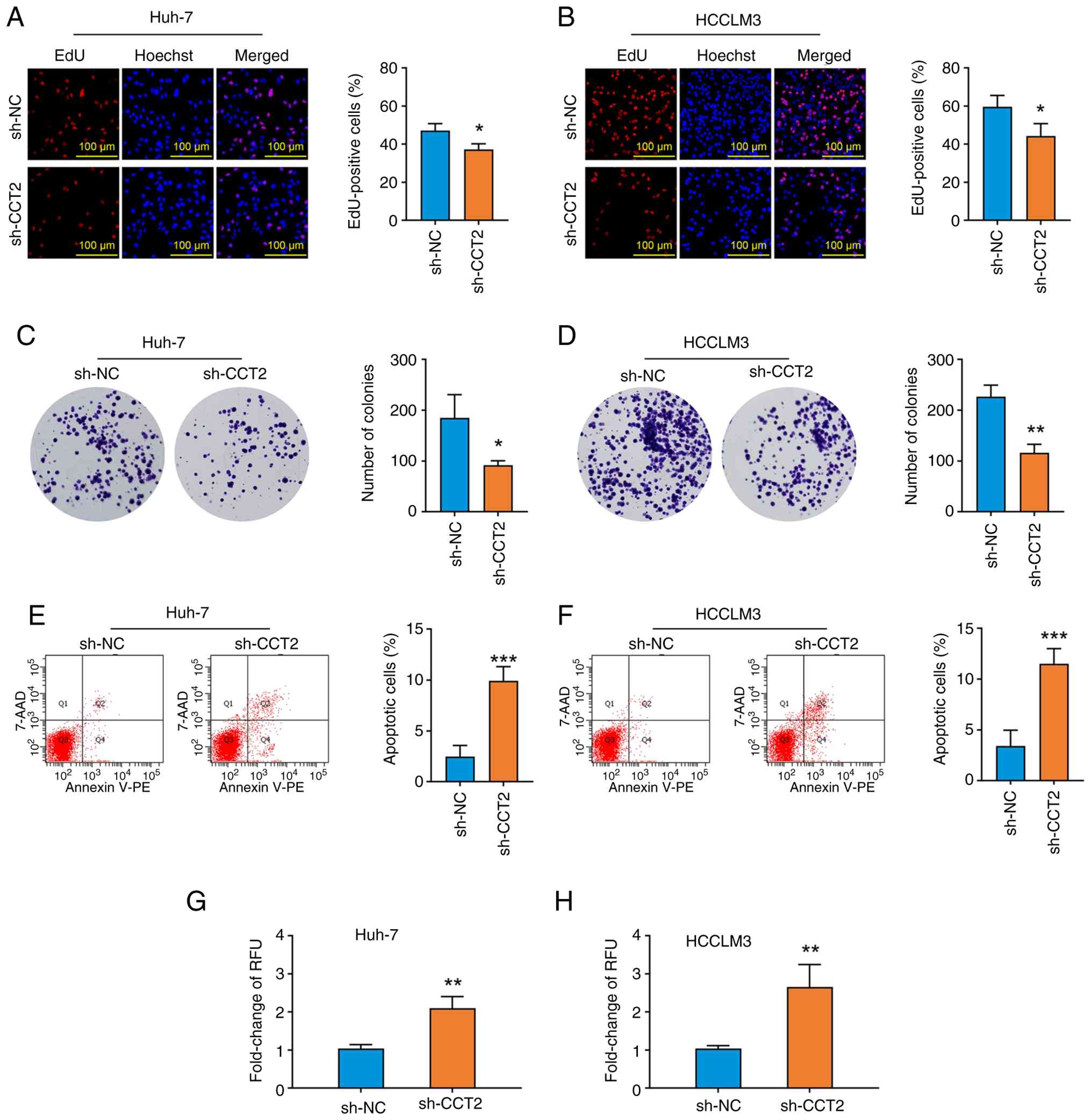
Knockdown of CCT2 inhibits
proliferation and promotes the apoptosis of HCC cells
Functional experiments were performed to determine
whether CCT2 knockdown affected HCC cell viability. CCT2 knockdown
significantly decreased the percentage of EdU-positive cells
(Fig. 3A and B) and the number of
colonies formed (Fig. 3C and D).
Additionally, compared with the sh-NC group, the proportion of
apoptotic cells (Fig. 3E and F) and
the activity of caspase-3/7 (Fig. 3G
and H) increased in the CCT2 knockdown group. Collectively, the
findings indicated that CCT2 may be involved in the regulation of
cell proliferation and apoptosis in HCC.
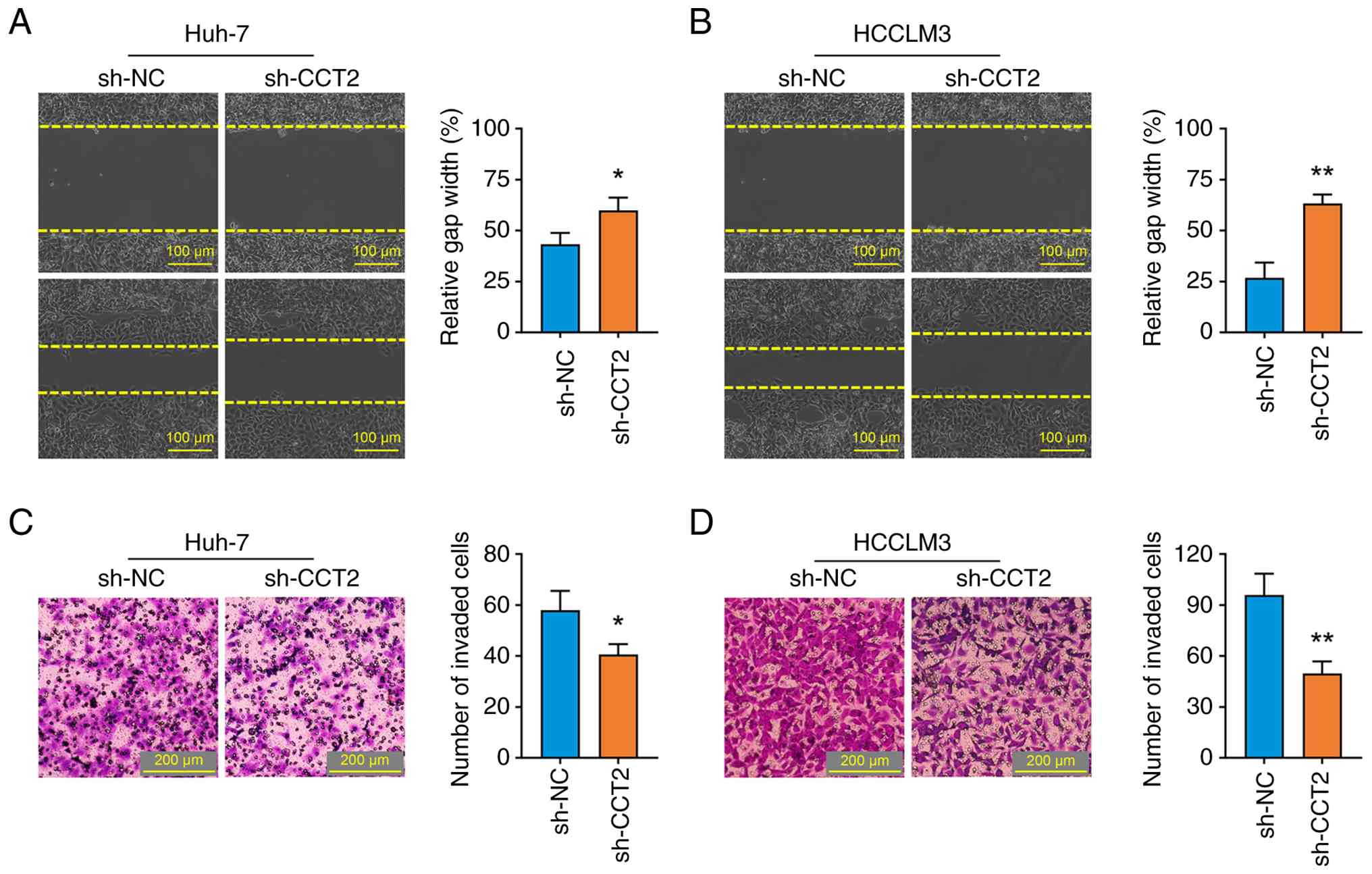
Knockdown of CCT2 inhibits the
migration and invasion of HCC cells
The effects of CCT2 knockdown on cancer cell
motility were investigated. CCT2 knockdown significantly suppressed
the migration and invasion of Huh-7 and HCCLM3 cells, as shown by
the gap closure (Fig. 4A and B) and
Transwell invasion (Fig. 4C and D)
assays. These data demonstrated that CCT2 may be involved in the
migration and invasive characteristics of HCC.
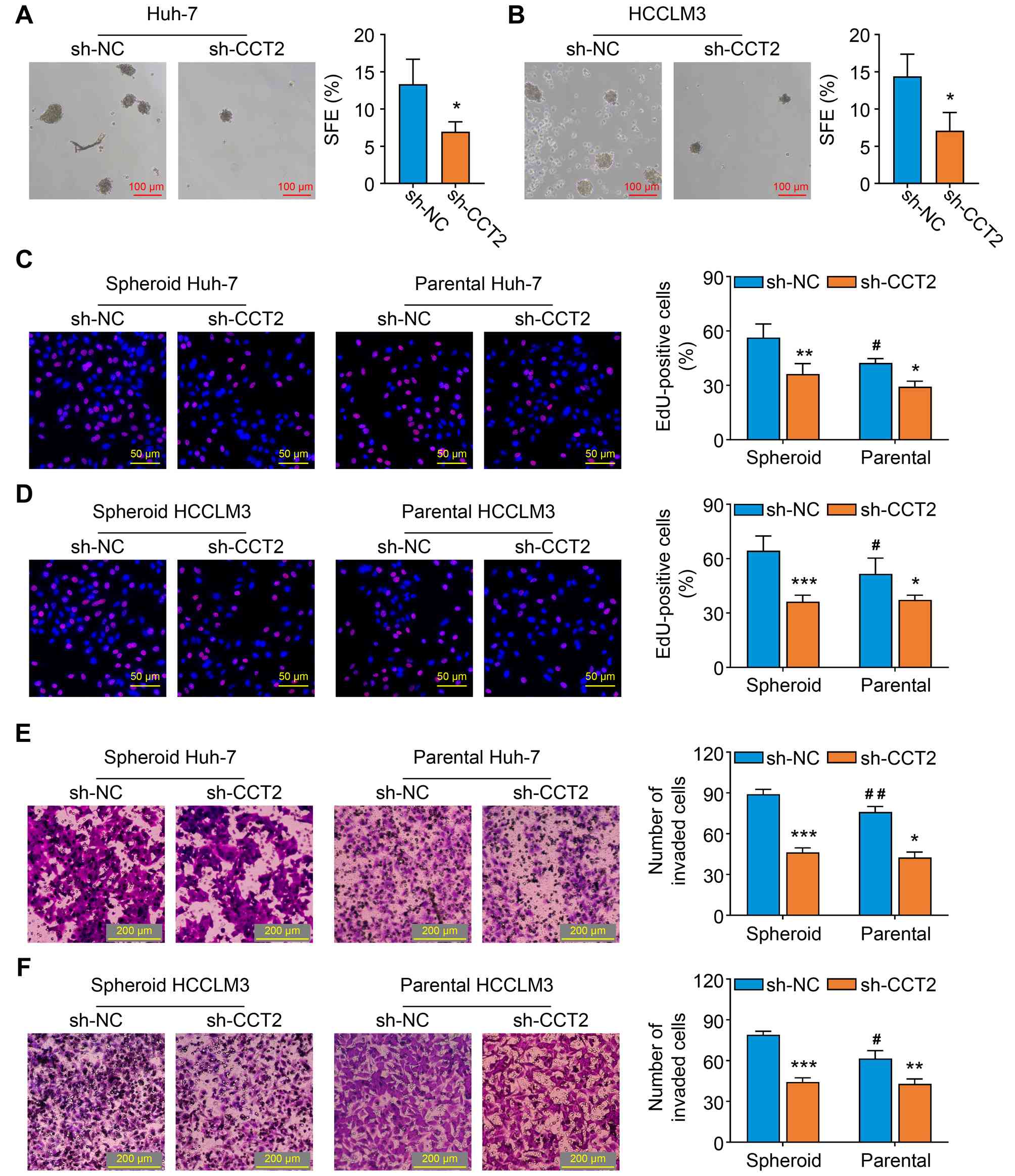
Knockdown of CCT2 inhibits the
stemness traits of HCC cells
The effect of CCT2 on cancer cell self-renewal was
detected by the tumor-sphere formation assay. The number of spheres
was lower in the sh-CCT2 compared with the sh-NC group (Fig. 5A and B). Compared with the parental
cells in sh-NC group, the spheroid cells in sh-NC group exhibited
enhanced proliferative and invasive capacities (Fig. 5C-F). Furthermore, knockdown of CCT2
inhibited these malignant behaviors in both parental and spheroid
cells (Fig. 5C-F). Taken together,
these findings demonstrated that CCT2 may regulate the stemness
traits of HCC cells.
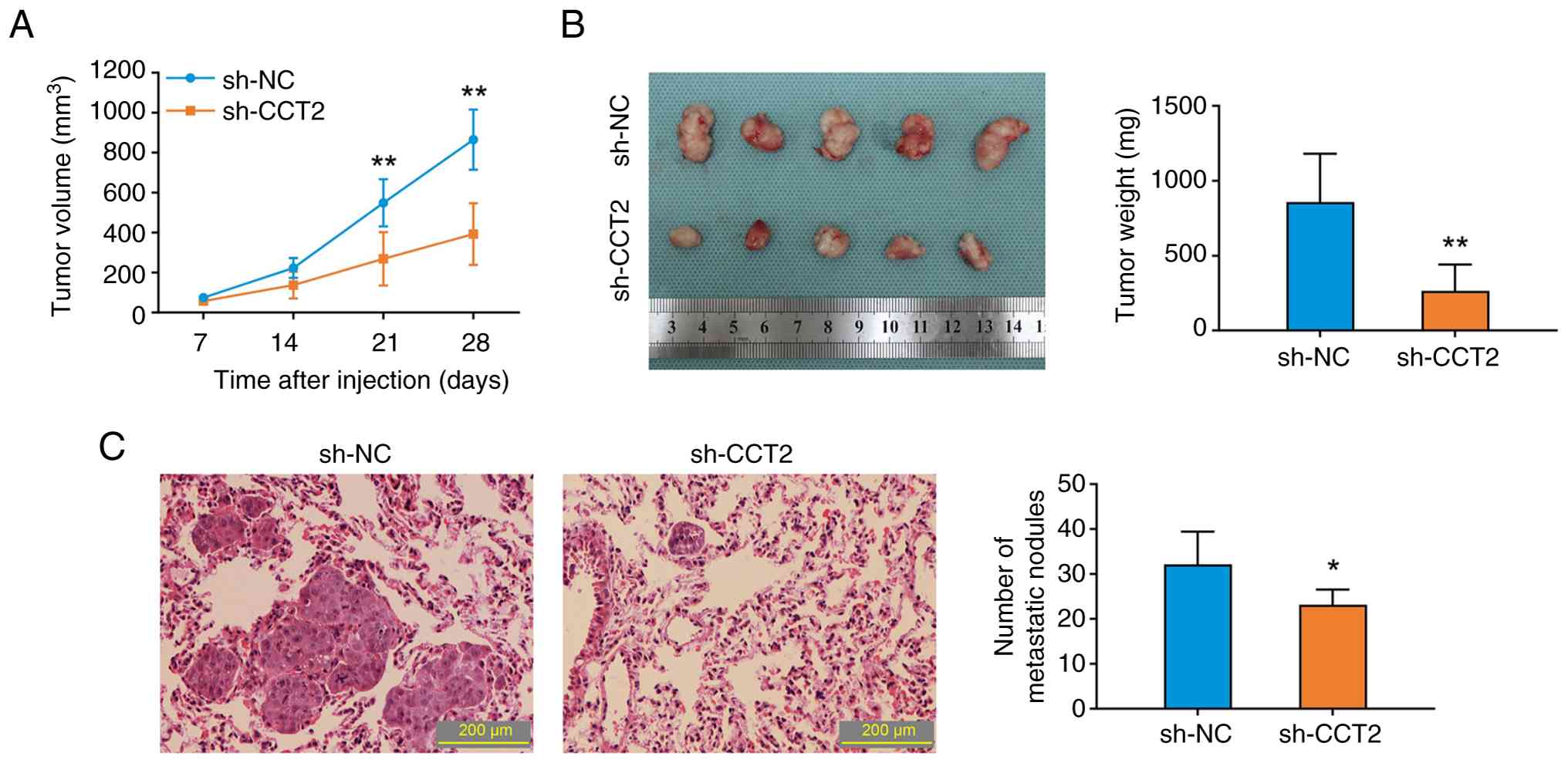
Knockdown of CCT2 inhibits the
proliferation and metastasis of HCCLM3 cells in vivo
Subcutaneous xenograft and hematogenous lung
metastasis models were used to verify the anticancer effects of
CCT2 knockdown in vivo. Knockdown of CCT2 in HCCLM3 cells
significantly decreased the tumor proliferation rate and final
tumor weight in xenograft mice (Fig. 6A
and B). Moreover, compared with the sh-NC group, the number of
lung metastatic nodules decreased in the sh-CCT2 group (Fig. 6C). In brief, the in vivo
results confirmed the roles of CCT2 in HCC progression.
Knockdown of CCT2 impairs the
activation of STAT3 in HCC cells
Phosphorylation of STAT3, particularly at residue
Tyr705, serves as a hallmark of its canonical activation (15). Western blotting showed that
knockdown of CCT2 attenuated the phosphorylation levels of STAT3
rather than changing its total protein levels (Fig. 7A and B). In addition, the protein
levels of MCL1, MMP2 and SOX2 were downregulated following CCT2
knockdown (Fig. 7A and B).
Immunohistochemical staining of subcutaneous tumor tissue also
demonstrated that the p-STAT3, MCL1, MMP2 and SOX2 levels were
decreased in the sh-CCT2 group (Fig.
7C-F). These results suggested that the STAT3 signaling pathway
may be a key downstream pathway regulated by CCT2 in HCC cells.
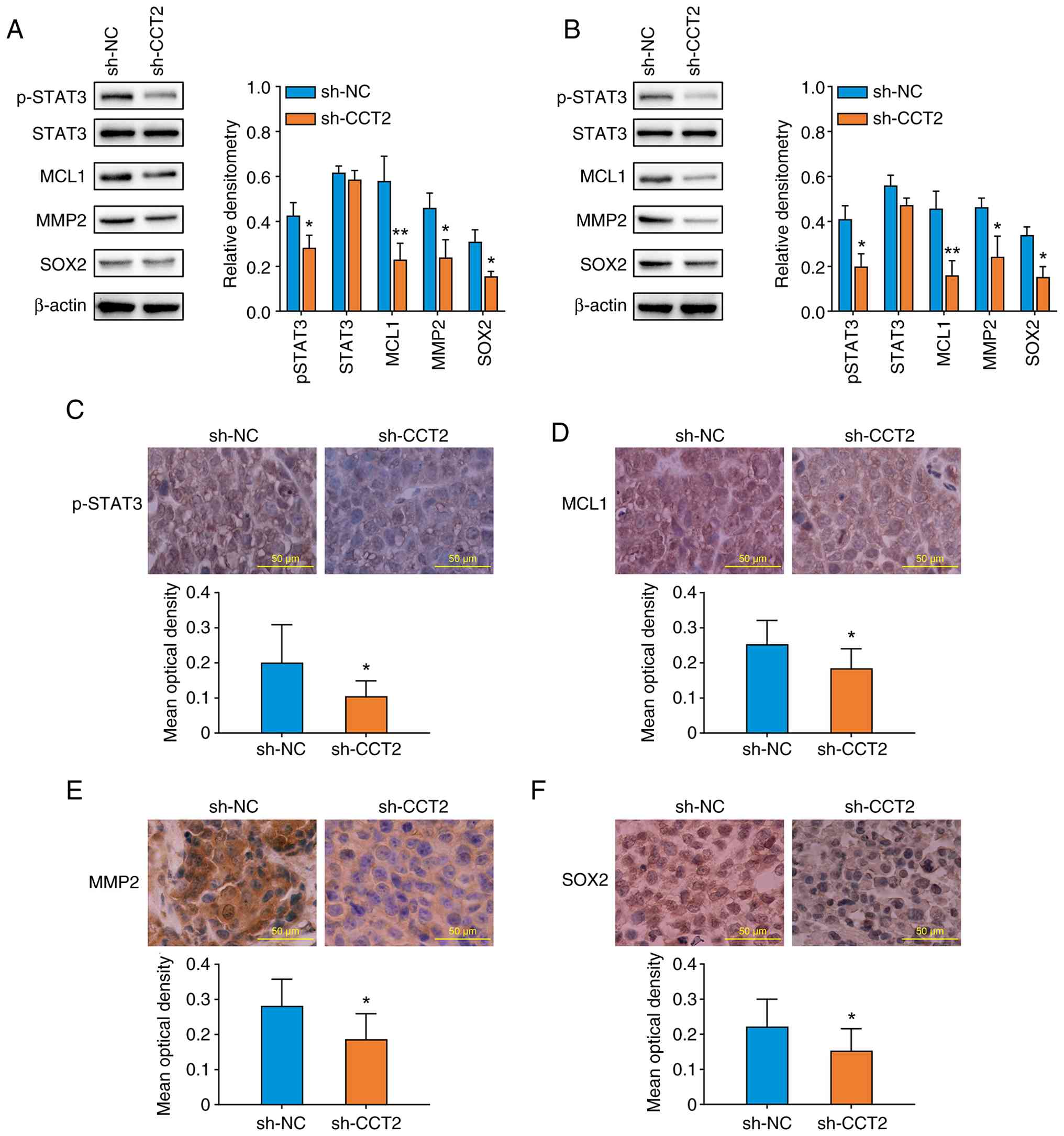 | Figure 7.Knockdown of CCT2 inhibits STAT3
signaling activation in hepatocellular carcinoma cells. The protein
levels of STAT3, p-STAT3, MCL1, MMP2 and SOX2 in (A) Huh-7 and (B)
HCCLM3 cells were measured by western blotting. The protein levels
of (C) p-STAT3, (D) MCL1, (E) MMP2 and (F) SOX2 in subcutaneous
tumor tissue were detected by immunohistochemical staining.
*P<0.05, **P<0.01 vs. sh-NC. CCT2, chaperonin containing TCP1
subunit 2; sh, short hairpin; NC, negative control; p,
phosphorylated; MCL1, myeloid cell leukemia sequence 1; SOX2,
SRY-box transcription factor 2. |
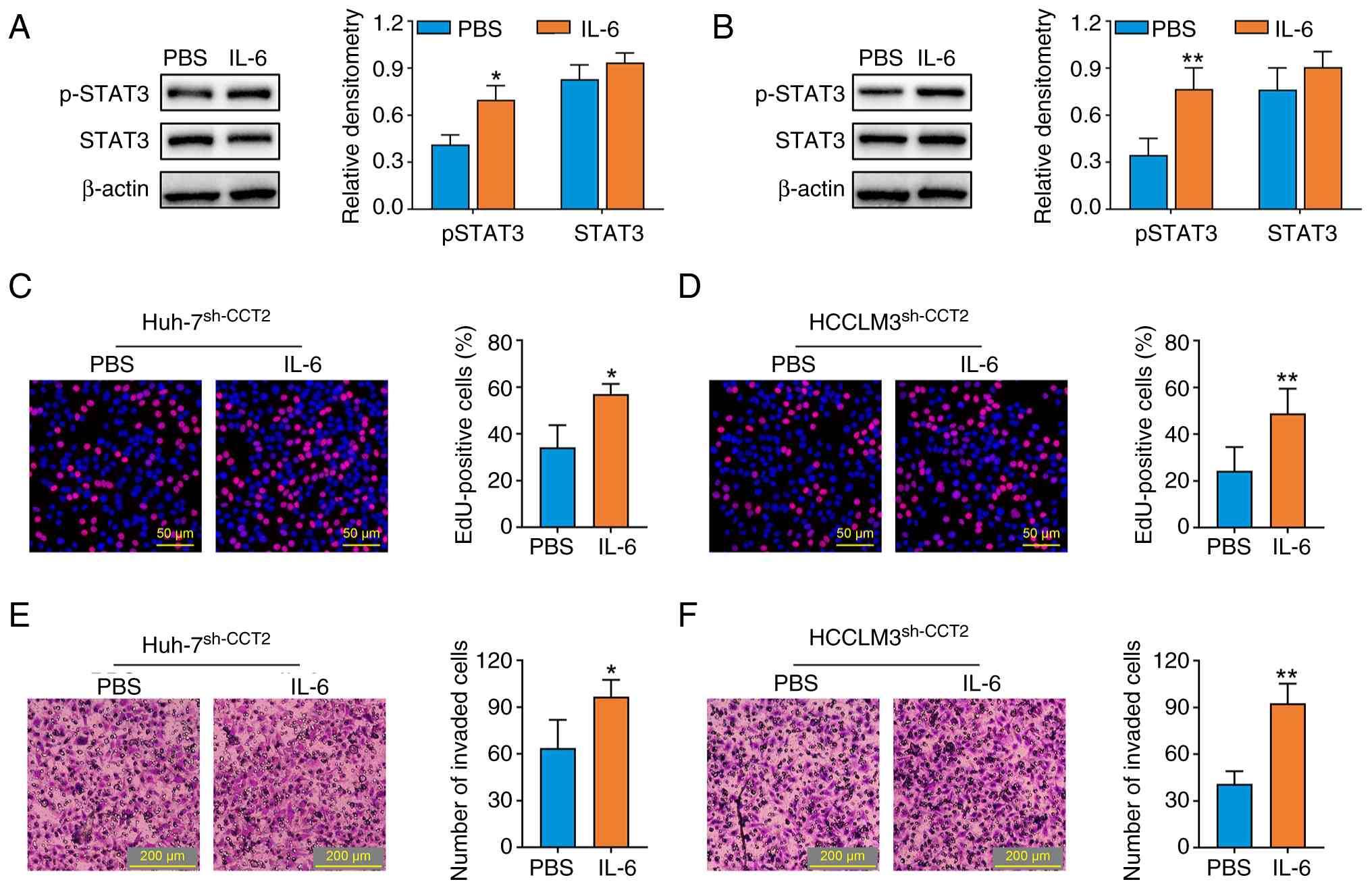
IL-6 abolishes the effects of CCT2
knockdown in HCC cells
To assess whether the oncogenic effects of CCT2 were
dependent on the activation of STAT3, recombinant human IL-6 (50
ng/ml) was used to activate STAT3 signaling in HCC cells. The
western blotting results confirmed that IL-6 treatment increased
the levels of p-STAT3 in Huh-7 and HCCLM3 cells (Fig. 8A and B). Furthermore, compared with
the PBS treatment group, the inhibitory effects of CCT2 knockdown
on the proliferation (Fig. 8C and
D) and invasion (Fig. 8E and F)
of Huh-7 and HCCLM3 cells were significantly abrogated by IL-6
treatment. These results indicated that CCT2 exerts its
tumor-promoting effects via STAT3 activation.
Discussion
Despite targeted therapy improving the prognosis of
patients with HCC, a notable number of patients experience
suboptimal outcomes owing to drug resistance or
treatment-associated toxicities (30). Identification of novel molecular
targets to improve the outcome of these patients is clinically
meaningful. Several studies have investigated the participation of
the CCT subunits in oncogenesis mechanisms and found an association
between these subunits and hallmarks of malignant cells (31,32).
The present study showed that CCT2 promoted cell functions typical
of malignancy in HCC.
In the present study, in silico analysis
revealed that CCT2 expression was typically elevated in HCC tissues
and associated with a poorer OS across multiple independent
clinical cohorts. In agreement with these findings, CCT2
upregulation is a hallmark of poor prognosis in gallbladder
(33), breast (34) and gastric (35) cancer. These results suggest CCT2 may
exert key oncogenic roles in HCC progression. In colorectal cancer,
CCT2 knockdown inhibits cell proliferation, migration and invasion
by interrupting the folding of GLI family zinc finger 1 (36). In the present study,
lentiviral-mediated shRNA transduction was used to knockdown the
expression of CCT2 in HCC cells. CCT2 knockdown not only attenuated
the malignant behaviors, including proliferation, migration and
invasion, but also induced the apoptosis of HCC cells. Cancer stem
cells promote tumor proliferation and invasion (37,38).
The present study found CCT2 knockdown impaired self-renewal as
well as the proliferative and invasive properties of spheroid HCC
cells. Moreover, in both the subcutaneous xenograft and
hematogenous lung metastasis models, CCT2 knockdown inhibited the
proliferation and lung metastasis of HCC cells in vivo. To
the best of our knowledge, the present study is the first to report
that CCT2 serves as a driver of HCC proliferation, stemness and
invasion.
STAT3 is an oncogene in cancer development.
Kasembeli et al (39) found
that CCT2 knockdown decreases the expression of total STAT3 protein
in HS-578T breast cancer cells. However, knockdown of CCT3
sensitizes A549 lung adenocarcinoma cells to cisplatin treatment by
inhibiting the phosphorylation of STAT3 (40). Additionally, overexpression of CCT4
partly rescues the inactivation of p-STAT3 induced by anticarin-β
in MG-63 osteosarcoma cells (41).
The aforementioned reports suggested that the regulation of STAT3
by CCT subunits is mediated by its phosphorylated activation. The
present study demonstrated that CCT2 knockdown inactivated the
STAT3 signaling pathway in HCC. CCT2 knockdown decreased the levels
of p-STAT3, MCL1, MMP2 and SOX2 in HCC cells. As downstream targets
of STAT3, MCL1, MMP2 and SOX2 promote HCC progression by driving
cell viability, extracellular matrix degradation and self-renewal
(14). Exogenous IL-6 treatment
rescued the phosphorylation of STAT3 and abrogated the
tumor-suppressive effects induced by CCT2 knockdown. Collectively,
the present study indicated that CCT2 promotes the malignant
behaviors of HCC cells by activating the STAT3 signaling pathway.
Notably, CCT2 promotes cancer progression through multiple
molecular mechanisms. For example, CCT2 directly binds to KRAS,
leading to the increased stability, viability and proliferation of
glioblastoma multiforme cells (42). Additionally, epithelial-mesenchymal
transition (EMT) causes cancer cells to acquire aggressive
phenotypes. CCT2 promotes the EMT process by upregulating the
expression of EMT-associated transcription factors, such as zinc
finger E-box binding homeobox 1 (43) and Snail/Slug (44). These findings provide a basis to
investigate the mechanisms by which CCT2 promotes HCC
progression.
The present study had several limitations that
should be acknowledged. First, the comprehensive mechanisms by
which CCT2 enhances STAT3 phosphorylation remain unclear.
Subsequent studies should assess protein-protein interactions to
explore how CCT2 activates STAT3 signaling, such as by stabilizing
its structure or facilitating its folding. Second, the clinical
significance of CCT2 was not evaluated using a prospective patient
cohort. A high-quality case-control study should be performed to
assess the clinical value of CCT2 in distinguishing healthy
controls and HCC.
In conclusion, the present study suggested that CCT2
promoted the malignant behaviors of HCC cells by activating the
STAT3 signaling pathway. CCT2 may serve as a promising therapeutic
target for patients with HCC.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the Beijing
Hospitals Authority Youth Programme (grant no. QML20210906) and the
Natural Science Basic Research Program of Shaanxi (grant no.
2021JQ-397).
Availability of data and materials
The data generated in this study can be requested
from the corresponding author.
Authors' contributions
CL and HQ conceptualized the present study. CL wrote
the manuscript. CL, LY and YZ performed experiments. CL, BZ and HQ
analyzed the data. All authors have read and approved the final
manuscript. CL and HQ confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
The animal experiments were approved by the
Institutional Animal Care and Use Committee of SPF Biotechnology
Co. Ltd. (approval no. AWE2023102504; Beijing, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hwang SY, Danpanichkul P, Agopian V, Mehta
N, Parikh ND, Abou-Alfa GK, Singal AG and Yang JD: Hepatocellular
carcinoma: Updates on epidemiology, surveillance, diagnosis and
treatment. Clin Mol Hepatol. 31 (Suppl):S228–S254. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mak LY, Liu K, Chirapongsathorn S, Yew KC,
Tamaki N, Rajaram RB, Panlilio MT, Lui R, Lee HW, Lai JC, et al:
Liver diseases and hepatocellular carcinoma in the Asia-Pacific
region: Burden, trends, challenges and future directions. Nat Rev
Gastroenterol Hepatol. 21:834–851. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Huang A, Yang XR, Chung WY, Dennison AR
and Zhou J: Targeted therapy for hepatocellular carcinoma. Signal
Transduct Target Ther. 5:1462020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim H, Park J and Roh SH: The structural
basis of eukaryotic chaperonin TRiC/CCT: Action and folding. Mol
Cells. 47:1000122024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gruber R and Horovitz A: Allosteric
mechanisms in chaperonin machines. Chem Rev. 116:6588–6606. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Date Y, Matsuura A and Itakura E:
Disruption of actin dynamics induces autophagy of the eukaryotic
chaperonin TRiC/CCT. Cell Death Discov. 8:372022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gestaut D, Zhao Y, Park J, Ma B, Leitner
A, Collier M, Pintilie G, Roh SH, Chiu W and Frydman J: Structural
visualization of the tubulin folding pathway directed by human
chaperonin TRiC/CCT. Cell. 185:4770–4787.e20. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu HH, Liu QH, Meng QN, Zhang LJ, Ju SW,
Lang JH, Zhu DH, Chen YX, Aishan N, Ouyang XX, et al:
CCT3/ACTN4/TFRC axis protects hepatocellular carcinoma cells from
ferroptosis by inhibiting iron endocytosis. J Exp Clin Canc Res.
43:2452024. View Article : Google Scholar
|
|
9
|
Li F, Liu CS, Wu P, Ling AS, Pan Q and Li
XN: CCT4 suppression inhibits tumor growth in hepatocellular
carcinoma by interacting with Cdc20. Chin Med J (Engl).
134:2721–2729. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang XD, Wang XX, Cheng C, Cai J, He S,
Wang H, Liu F, Zhu CL, Ding ZM, Huang XT, et al: Chaperonin
containing TCP1, subunit 8 (CCT8) is upregulated in hepatocellular
carcinoma and promotes HCC proliferation. Apmis. 122:1070–1079.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lv W, Shi L, Pan J and Wang S:
Comprehensive prognostic and immunological analysis of CCT2 in
pan-cancer. Front Oncol. 12:9869902022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Showalter AE, Martini AC, Nierenberg D,
Hosang K, Fahmi NA, Gopalan P, Khaled AS, Zhang W and Khaled AR:
Investigating Chaperonin-containing TCP-1 subunit 2 as an essential
component of the chaperonin complex for tumorigenesis. Sci Rep.
10:7982020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Carr AC, Khaled AS, Bassiouni R, Flores O,
Nierenberg D, Bhatti H, Vishnubhotla P, Manuel JP, Santra S and
Khaled AR: Targeting chaperonin containing TCP1 (CCT) as a
molecular therapeutic for small cell lung cancer. Oncotarget.
8:110273–110288. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hu Y, Dong Z and Liu K: Unraveling the
complexity of STAT3 in cancer: Molecular understanding and drug
discovery. J Exp Clin Cancer Res. 43:232024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Berkley K, Zalejski J and Sharma A:
Targeting STAT3 for cancer therapy: Focusing on Y705, S727, or dual
inhibition? Cancers (Basel). 17:7552025. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu J, Zeng X, Huang J, Ma S, Li K, Yang S,
Naz W, Yousaf T, Yuan S, Liu Y, et al: Dual-specificity
tyrosine-regulated kinase 4 modulates the STAT3-FOS signaling axis
to inhibit hepatitis B virus replication via autophagy. Int J Biol
Sci. 21:2415–2429. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tulahong A, Xu X, Zhou T, Ruze R, Yuan Z,
Qiao P, Jiang T, Aji T and Shao Y: Empagliflozin alleviates hepatic
ischemia-reperfusion injury by inhibiting c-Myc through the
JAK1-STAT3 signaling pathway. Int Immunopharmacol. 165:1155082025.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nian F, Chen Y, Chen J, Jiang Q, Meng F,
Chen Z, Lu X, Shen X and Li Y: Carpaine alleviates NASH-related
fibrosis by targeting Nid1 to inhibit IL-6/JAK/STAT3 signaling and
macrophage M1 polarization. Int J Biol Macromol. 337:1494512025.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hashemi M, Sabouni E, Rahmanian P,
Entezari M, Mojtabavi M, Raei B, Zandieh MA, Behroozaghdam M,
Mirzaei S, Hushmandi K, et al: Deciphering STAT3 signaling
potential in hepatocellular carcinoma: Tumorigenesis, treatment
resistance, and pharmacological significance. Cell Mol Biol Lett.
28:332023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen X, Ma CN, Li YM, Liang YR, Chen T,
Han DW, Luo D, Zhang N, Zhao WJ, Wang LJ, et al: Trim21-mediated
CCT2 ubiquitination suppresses malignant progression and promotes
CD4+T cell activation in breast cancer. Cell Death Dis.
15:5422024. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vallin J, Córdoba-Beldad CM and Grantham
J: Sequestration of the transcription factor STAT3 by the molecular
chaperone CCT: A potential mechanism for modulation of STAT3
phosphorylation. J Mol Biol. 433:1669582021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cancer Genome Atlas Research Network.
Electronic address, . simplewheeler@bcm.edu and Cancer
Genome Atlas Research Network: Comprehensive and integrative
genomic characterization of hepatocellular carcinoma. Cell.
169:1327–1341.e23. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fujimoto A, Furuta M, Totoki Y, Tsunoda T,
Kato M, Shiraishi Y, Tanaka H, Taniguchi H, Kawakami Y, Ueno M, et
al: Whole-genome mutational landscape and characterization of
noncoding and structural mutations in liver cancer. Nat Genet.
48:500–509. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gao Q, Zhu H, Dong L, Shi W, Chen R, Song
Z, Huang C, Li J, Dong X, Zhou Y, et al: Integrated Proteogenomic
characterization of HBV-related hepatocellular carcinoma. Cell.
179:561–577. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang Z, Wu Y, Miao Y, Deng K, Yang F, Xu
S, Wang Y, You R, Zhang L, Fan Y, et al: HCCDB v2.0: Decompose
expression variations by Single-cell RNA-seq and spatial
Transcriptomics in HCC. Genomics Proteomics Bioinformatics.
22:qzae0112024. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li C, Cheng X and Jiang Y:
Deuterium-depleted water inhibits the malignant progression of
colorectal cancer cells by modulating oxidative stress. Oncol Rep.
53:702025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li C, Miao R, Zhang J, Qu K and Liu C:
Long non-coding RNA KCNQ1OT1 mediates the growth of hepatocellular
carcinoma by functioning as a competing endogenous RNA of miR-504.
Int J Oncol. 52:1603–1612. 2018.PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ranjbar R, Behzadi P and Mammina C:
Respiratory tularemia: Francisella tularensis and microarray probe
designing. Open Microbiol J. 10:176–182. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qin Y, Han S, Yu Y, Qi D, Ran M, Yang M,
Liu Y and Li Y, Lu L, Liu Y and Li Y: Lenvatinib in hepatocellular
carcinoma: Resistance mechanisms and strategies for improved
efficacy. Liver Int. 44:1808–1831. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Macario AJL and Conway de Macario E:
Chaperonins in cancer: Expression, function, and migration in
extracellular vesicles. Semin Cancer Biol. 86:26–35. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Grantham J: The molecular chaperone
CCT/TRiC: An essential component of proteostasis and a potential
modulator of protein aggregation. Front Genet. 11:1722020.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zou Q, Yang ZL, Yuan Y, Li JH, Liang LF,
Zeng GX and Chen SL: Clinicopathological features and CCT2 and
PDIA2 expression in gallbladder squamous/adenosquamous carcinoma
and gallbladder adenocarcinoma. World J Surg Oncol. 11:1432013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu Q, Qi Y, Kong X, Wang X, Zhang W, Zhai
J, Yang Y, Fang Y and Wang J: Molecular and clinical
characterization of CCT2 expression and prognosis via Large-scale
transcriptome profile of breast cancer. Front Oncol. 11:6144972021.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ma X, Qiu SP, Tang X, Song QY, Wang PC,
Wang JW, Xia QC, Wang ZJ, Zhao QH and Lu M: TSPAN31 regulates the
proliferation, migration, and apoptosis of gastric cancer cells
through the METTL1/CCT2 pathway. Transl Oncol. 20:1014232022.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Park SH, Jeong S, Kim BR, Jeong YA, Kim
JL, Na YJ, Jo MJ, Yun HK, Kim DY, Kim BG, et al: Activating CCT2
triggers Gli-1 activation during hypoxic condition in colorectal
cancer. Oncogene. 39:136–150. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li YF, Su S, Luo Y, Wei C, He J, Song LD,
Han K, Wang J, Gan X and Wang DL: Widespread activation and
critical role of EMT and stemness in the neuroendocrine
differentiation of prostate cancer (Review). Oncol Rep. 54:1092025.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ham A, Cho MH, Won HS, Jo J and Lee KE:
β-catenin blockers enhance the effect of CDK4/6 inhibitors on
stemness and proliferation suppression in endocrine-resistant
breast cancer cells. Oncol Rep. 48:1302022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kasembeli M, Lau WCY, Roh SH, Eckols TK,
Frydman J, Chiu W and Tweardy DJ: Modulation of STAT3 folding and
function by TRiC/CCT Chaperonin. PLoS Biol. 12:e10018442014.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xu DN, Zeng JZ, Sun HM, Pan YL, Yang CC
and Lu YD: Chaperonin containing TCP1 subunit 3 (CCT3) promotes
cisplatin resistance of lung adenocarcinoma cells through targeting
the Janus kinase 2/signal transducers and activators of
transcription 3 (JAK2/STAT3) Pathway. Bioengineered. 12:7335–7347.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang G, Zhang M, Meng P, Long CB, Luo XD,
Yang XW, Wang YF, Zhang ZY, Mwangi J, Kamau PM, et al: Anticarin-β
shows a promising anti-osteosarcoma effect by specifically
inhibiting CCT4 to impair proteostasis. Acta Pharm Sin B.
12:2268–2279. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhao FH, Yao Z, Li YQ, Zhao WB, Sun YF,
Yang XB, Zhao ZM, Huang B, Wang J, Li XA, et al: Targeting the
molecular chaperone CCT2 inhibits GBM progression by influencing
KRAS stability. Cancer Lett. 590:2168442024. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chen JY, Hu Q, Zhou CH and Jin DW: CCT2
prevented β-catenin proteasomal degradation to sustain cancer stem
cell traits and promote tumor progression in epithelial ovarian
cancer. Mol Biol Rep. 51:542024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu WP, Lin RZ, Zhu CM, Chen YXZ, Gao QA
and Zhong JJ: CCT2 regulates ZEB1-induced EMT gene transcription to
promote the metastasis and tumorigenesis of papillary thyroid
carcinoma. Discov Med. 36:1819–1830. 2024. View Article : Google Scholar : PubMed/NCBI
|